Plant Physiology Preview. Published on March 9, 2018, as DOI: /pp
|
|
|
- Derrick Parker
- 6 years ago
- Views:
Transcription
1 Plant Physiology Preview. Published on March 9, 2018, as DOI: /pp Research area: Cell biology Short title: AP-3 mediates pollen tube growth For correspondence: Yan Zhang, State Key Laboratory of Crop Biology, College of Life Sciences, Shandong Agricultural University, Tai an, , China Tel: (86) yzhang@sdau.edu.cn Author contributions Y.Z. conceived and supervised the project; Q.F. performed the experiments with the assistance of X.L.; Y.Z., S.L., and Q.F. designed the experiments and analyzed the data; Y.Z. and Q.F. wrote the article Footnotes: This work was supported by the Natural Science Foundation of China ( and to Y.Z., to S.L.), by Natural Science Foundation of Shandong Province (ZR2014CM027 to S.L.), and by China Postdoctoral Science Foundation (2015M and 2016T90643 to S.L.). Y.Z. s laboratory is partially supported by the Tai-Shan Scholar Program of the Shandong Provincial Government Copyright 2018 by the American Society of Plant Biologists
2 The ADAPTOR PROTEIN-3 complex mediates pollen tube growth by coordinating vacuolar targeting and organization Qiang-Nan Feng, Xin Liang, Sha Li, Yan Zhang* State Key Laboratory of Crop Biology, College of Life Sciences, Shandong Agricultural University, Tai an, , China One Sentence Summary: The dynamic organization of vacuoles and the association of tonoplast cargo protein PAT10 are impaired and lead to reduced pollen tube growth in adaptor protein-3 mutants Abstract Pollen tube growth is an essential step for successful plant reproduction. Vacuolar trafficking and dynamic organization are important for pollen tube growth; however, the key proteins involved in these processes are not well understood. Here, we report that the ADAPTOR PROTEIN-3 (AP-3) complex and its tonoplast cargo PROTEIN S-ACYL TRANSFERASE10 (PAT10) are critical for pollen tube growth in Arabidopsis (Arabidopsis thaliana). AP-3 is a heterotetrameric protein complex consisting of four subunits, δ, β, µ, and σ. AP-3 regulates tonoplast targeting of several cargoes, such as PAT10. We show that functional loss of any of the four AP-3 subunits reduces plant fertility. In ap-3 mutants, pollen development was normal but pollen tube growth was compromised, leading to reduced male transmission. Functional loss of PAT10 caused a similar reduction in pollen tube growth, suggesting that the tonoplast association of PAT10 mediated by AP-3 is crucial for this process. Indeed, Ca 2+ gradient during pollen tube growth was significantly reduced due to AP-3 loss-of-function, consistent with the abnormal targeting of Calcinuerin B-like2 (CBL2) and CBL3, whose tonoplast association depends on PAT10. Further, we show that the pollen tubes of ap-3 mutants have vacuoles with simplified tubules and bulbous structures, indicating that AP-3 affects vacuolar organization. Our results demonstrate a role for AP-3 in plant reproduction and provide insights into the role of vacuoles in polarized cell growth Keywords: pollen tube growth, vacuolar trafficking, male fertility, CBL2, calcium - 2 -
3 Introduction Sexual reproduction of flowering plants is a complex, multi-step process involving gametophytic development, interactions between male and female gametophytes, and interactions between male and female gametes (McCormick, 2004). Due to the immobility of sperm cells in angiosperms, the delivery of sperm into embryo sacs depends on pollen tubes, long cylindrical extensions initiated from the growing tip of pollen grains. Pollen tubes penetrate female sporophytic tissues, target the micropyle by sensing and responding to female guidance cues, and finally burst to release sperm cells into embryo sacs (Johnson and Preuss, 2002). Tip growth of pollen tubes is a specialized polar growth that involves complex intracellular activities, including a tip-focused Ca 2+ gradient, spatial organization of endomembrane systems, and dynamic organization of the cytoskeleton (Hepler et al., 2001; Cheung and Wu, 2007; Cheung et al., 2008; Cheung and Wu, 2008; Hepler et al., 2012). Pollen tube growth accompanies dynamic vacuolar organization and trafficking (Hicks et al., 2004). Vacuoles, as extensive tubular extensions, fill with growing pollen tubes, except at the very apex (Hicks et al., 2004; Wudick et al., 2014). Functional loss of VACUOLESS1 (VCL1), a gene essential for vacuolar biogenesis (Rojo et al., 2001), resulted in reduced male gametophytic transmission although the vacuoles in vcl1 pollen tubes seemed normal (Hicks et al., 2004). Overexpression of PTEN, a phosphatase that downregulates PtdIns3P, the phosphoinositide critical for vacuolar fusion, resulted in defective pollen tube growth by disrupting vacuolar consumption of autophagic bodies in Arabidopsis (Arabidopsis thaliana) (Zhang et al., 2011). In addition, mutations at VPS41, encoding a component of the homotypic fusion and vacuolar protein sorting (HOPS) complex that mediates vesicle-vacuole fusion, caused defective pollen tube growth in response to female cues (Hao et al., 2016). These results suggested that vacuolar trafficking and organization are critical for the polar and directional growth of pollen tubes. However, the key proteins targeted to vacuoles and critical for pollen tube growth are yet unknown. Proper targeting of proteins within the endomembrane system is crucial for cell growth and responses to the environment. Adaptor proteins (AP) are key players mediating protein sorting; they recognize both cargo proteins and coat proteins - 3 -
4 during vesicle formation (Boehm and Bonifacino, 2001; Robinson, 2004; Bassham et al., 2008). APs are heterotetrameric protein complexes. There are five AP complexes in eukaryotes, differing in their subcellular targeting and functionalities: AP-1, AP-2, AP-3, AP-4, and AP-5 (Boehm and Bonifacino, 2001; Robinson, 2004; Bassham et al., 2008; Hirst et al., 2011). In plants, except for the newly discovered AP-5, the other types of APs have been characterized. AP-1 associates with the trans-golgi network/early endosome (TGN/EE) to sort proteins to the plasma membrane (PM), to the forming cell plate, or to vacuoles (Park et al., 2013; Teh et al., 2013). Functional loss of AP-1 reduced male and female fertility (Park et al., 2013; Teh et al., 2013; Wang et al., 2013). AP-2 participates in clathrin-mediated endocytosis and is required for reproductive organ development (Fan et al., 2013; Kim et al., 2013; Yamaoka et al., 2013). Another class of ancient adaptor complex termed TPLATE/TSET (Gadeyne et al., 2014; Hirst et al., 2014) was reported to mediate reproduction, as mutations in those components caused pollen developmental defects (Van Damme et al., 2006; Gadeyne et al., 2014). AP-4 was recently reported to mediate vacuolar trafficking (Fuji et al., 2016). Several studies indicated that AP-3 mediates vacuolar targeting of several protein cargoes, including SUCROSE TRANSPORTER4 (SUC4) (Wolfenstetter et al., 2012), the R-SNARE subfamily members VAMP713 (Ebine et al., 2014) and VAMP711 (Feng et al., 2017), as well as PROTEIN S-ACYL TRANSFERASE10 (PAT10) (Feng et al., 2017b). Functional loss of AP-3 resulted in reduced germination potential of seeds (Feraru et al., 2010; Zwiewka et al., 2011) and slight defects in gravitropic responses (Feraru et al., 2010). It is unclear whether AP-3 affects reproductive processes. In this study, we analyze mutants of all four AP-3 subunits and show that AP-3 and its tonoplast cargo PAT10 are important for pollen tube growth. Functional loss of single AP-3 subunits reduced male transmission and caused reduced seed set. Pollen development was not affected by AP-3 loss-of-function. However, the rapid growth of pollen tubes both in vitro and in vivo was compromised in the ap-3 mutants. Functional loss of PAT10 caused a similar reduction in pollen tube growth, suggesting that AP-3-mediated vacuolar targeting of PAT10 is crucial for this process. We further show that AP-3 loss-of-function affected the vacuolar organization of pollen tubes, as vacuoles of mutant pollen tubes were less complex tubules, with - 4 -
5 attached bulbous structures. Our results demonstrate a role for AP-3 in plant reproduction and provide insights into the role of vacuoles in polarized cell growth Results AP-3 loss-of-function results in reduced seed set To determine the potential roles of AP-3 during plant reproduction, we characterized mutants of each AP-3 component, i.e., AP-3δ, AP-3β, AP-3µ, and AP-3σ (Fig. 1A). Each component is encoded by a single gene in Arabidopsis (Bassham et al., 2008). Mutants of AP-3δ, AP-3β, and AP-3µ used in this study were previously designated as protein affected trafficking4 (pat4-2), pat2-2, and ap-3µ-2, respectively, and were proven to be null mutants (Niihama et al., 2009; Feraru et al., 2010; Zwiewka et al., 2011). Only one T-DNA insertion mutant was available within the genomic region of AP-3σ, in which the full-length transcript of AP-3σ was below RT-qPCR detection (Feng et al., 2017b). To be consistent with their functional roles as AP-3 components, and for simplicity, we refer to these mutants as ap-3δ, ap-3β, ap-3µ, and ap-3σ. Each of the mutants was similar to wild type during the vegetative stage, as reported (Feraru et al., 2010; Zwiewka et al., 2011). However, during the reproductive stage, each mutant showed significantly reduced seed set (Fig. 1B), with undeveloped ovules distributed predominantly at the bottom of the siliques (Fig. 1C, 1D). These results showed that AP-3 is important for reproduction Functional loss of AP-3 reduces male transmission but does not affect pollen development To determine the reason for the reduced seed set, we performed reciprocal crosses between the wild type and each heterozygous mutant: ap-3δ, ap-3β, ap-3µ, or ap-3σ. Segregation ratios of the F1 progenies showed that each mutant showed reduced male but not female transmission (Table 1). These results demonstrated that AP-3 is involved in male gametophytic function. Several steps lead to proper male gametophytic function, specifically, pollen development, pollen germination, polar and directional growth of pollen tubes, and pollen tube reception (McCormick, 2004). To determine the causes of compromised male gametophytic function in the ap-3 mutants, we first analyzed pollen - 5 -
6 development using Alexander staining for cytoplasmic viability, DAPI staining for nuclear structure, and scanning electron microscopy (SEM) for pollen coat structure (Johnson-Brousseau and McCormick, 2004). The ap-3 mutants were no different from the wild type (Supplemental Fig. 1), indicating that AP-3 is not involved in pollen development AP-3 loss-of-function causes a reduction in pollen tube growth but not in guidance To determine the cause of reduced male transmission in the ap-3 mutants, we next performed in vitro pollen germination (Fig. 2A-E). All ap-3 mutants were comparable to wild type in germination potential (Fig. 2F), indicating that AP-3 is not essential for pollen germination. In the first two hours of pollen germination, there was no difference in pollen tube length between the ap-3 mutants and the wild type (Supplemental Fig. 2). However, at later stages (4 hr and 6 hrs), pollen tube growth in the ap-3 mutants was significantly reduced (Fig. 2B-E) compared to the wild type (Fig. 2A) at the same time points (Fig. 2G, Supplemental Fig. 2). In addition, pollen tube width of the ap-3 mutants was slightly but significantly increased (Fig. 2H), suggesting compromised tube polarity. To test whether pollen tube growth in the ap-3 mutants was also affected in vivo, we emasculated and hand-pollinated wild-type pistils with pollen from the ap-3 mutants or wild type. Using aniline blue staining of pistils at 9 hours after pollination (HAP), we determined that wild-type pollen tubes could grow to the bottom of the pistils (Fig. 3A), whereas pollen tubes of the ap-3 mutants could not (Fig. 3B-E). Among the ap-3 mutants, ap-3σ showed the least severe phenotypic defect in pollen tube growth in vivo (Fig. 3E), consistent with its seed set reduction (Fig. 1D, 1E) and pollen tube growth in vitro (Fig. 2E, 2G). Even at 24 HAP, pollen tubes of the ap-3 mutants could hardly reach the bottom of the pistils (Supplemental Fig. 3), which explains the incidence of unfertilized ovules at the bottom of the pistils (Fig. 1). Despite the reduced growth, pollen tubes of ap-3 mutants were able to reach and properly target ovules that were accessible (Fig. 3G-J), similarly to the wild type (Fig. 3F). SEM of wild-type pistils emasculated and hand-pollinated with ap-3 pollen also confirmed normal pollen tube guidance (Fig. 3K-O). Because ap-3δ contained Pro LAT52 :GUS in the T-DNA, we could perform histochemical GUS staining of wild-type - 6 -
7 pistils pollinated either with Pro LAT52 :GUS pollen or ap-3δ pollen. All ovules accessible to mutant pollen tubes were targeted (Supplemental Fig. 4), confirming intact pollen tube guidance in the ap-3 mutants AP-3 loss-of-function compromises the vacuolar organization of pollen tubes Pollen tube growth requires dynamic vacuolar organization (Hicks et al., 2004; Wudick et al., 2014), which is likely crucial for turgor regulation and ion homeostasis. Functional loss of AP-3 affected vacuolar organization in seeds and root cells (Feraru et al., 2010; Zwiewka et al., 2011). Therefore, we were interested in determining the vacuolar organization of ap-3 mutant pollen tubes. To this purpose, we used a fluorescent-fusion of the V-ATPase a3 subunit, VHA-a3-YFP, to label the tonoplast. VHA-a3 is targeted to the tonoplast via an AP-3-independent route (Viotti et al., 2013; Feng et al., 2017). In wild-type pollen tubes, VHA-a3-labeled tubular vacuoles were extensive and penetrated to the subapical area right beneath the clear zone (Fig. 4A, Supplemental Movie 1). In contrast, VHA-a3-labeled vacuoles in ap-3σ pollen tubes were less complex and far behind the subapical region (Fig. 4B, Supplemental Movie 2). In addition to tubular structures resembling those in wild type (Fig. 4A, Supplemental Movie 3), there were often VHA-a3-labeled ring-shaped or bulbous structures associated with tubular vacuoles in ap-3σ pollen tubes (Fig. 4B, Supplemental Movie 4). These results showed that functional loss of AP-3 compromises vacuolar organization in pollen tubes PAT10, whose tonoplast association depends on AP-3, mediates pollen tube growth Because AP-3 functions through sorting vacuolar cargoes at the Golgi (Bassham et al., 2008; Uemura and Ueda, 2014), we questioned whether mis-targeting of some AP-3 cargoes resulted in reduced pollen tube growth. In Arabidopsis, AP-3 was reported to mediate the tonoplast localization of SUC4 (Wolfenstetter et al., 2012), VAMP713 (Ebine et al., 2014), VAMP711 (Feng et al., 2017), and PAT10 (Feng et al., 2017b). To determine whether these AP-3 cargoes were involved in pollen tube growth, we first examined their expression in mature pollen grains by RT-qPCR. We found that SUC4 was barely expressed in pollen, while PAT10 and the genes encoding R-SNAREs were expressed (Supplemental Fig. 5)
8 Functional loss of PAT10 showed reproductive defects, specifically reduced male transmission (Zhou et al., 2013). Although the role of PAT10 in sporophytically-controlled pollen development has been characterized (Zhou et al., 2013), its roles during pollen tube growth are not known. First, we introduced PAT10g-GFP (Zhou et al., 2013) into ap-3δ and examined PAT10 targeting in ap-3δ pollen tubes. VHA-a3-RFP was co-expressed to label the tonoplast both in wild-type and ap-3δ pollen tubes. In wild type, PAT10-GFP labeled dynamic tubular structures overlapping those of VHA-a3-RFP (Fig. 5A), indicative of the tonoplast (Hicks et al., 2004). However, PAT10-GFP was distributed into punctate vesicles in ap-3δ pollen tubes, whereas VHA-a3-RFP still showed tubular tonoplast localization (Fig. 5B). The tonoplast association of PAT10-GFP in ap-3δ pollen tubes, as well as reduced pollen tube length, were restored by introducing AP-3δg-RFP (Fig. 5C-D), confirming AP-3δ-dependent tonoplast targeting of PAT10 and tube growth. Next, we used PAT10g-GFP/-;pat10-2 plants for pollen tube growth studies in vitro, in which fluorescent pollen tubes were equivalent to wild type and non-fluorescent ones were pat10-2. Indeed, non-fluorescent pollen tubes from the PAT10g-GFP/-;pat10-2 plants were significantly shorter than GFP-expressing pollen tubes (Supplemental Fig. 6), demonstrating the role of PAT10 in pollen tube growth. PAT10 mediates the S-acylation-dependent tonoplast association of CALCINEURIN B-LIKE2 (CBL2) and CBL3 (Zhou et al., 2013; Zhang et al., 2015). To determine whether CBL2 and CBL3 were mis-targeted in the ap-3 pollen tubes due to mis-targeting of PAT10, we analyzed the subcellular localization of CBL2-RFP in each ap-3 mutant. In contrast to the tubular distribution of CBL2-RFP in wild-type pollen tubes (Fig. 5E), RFP signals were distributed into the PM and cytoplasmic vesicles in the ap-3 pollen tubes (Fig. 5F-I). Because mutations at CBL2 and CBL3 cause defects in pollen tube growth (Steinhorst et al., 2015), these results further supported the idea that the mis-targeting of PAT10 and its downstream components contributes to the reduced pollen tube growth of the ap-3 mutants AP-3 loss-of-function compromises the tip-focused Ca 2+ gradient in pollen tubes Pollen tube growth relies on a tip-focused Ca 2+ gradient (Konrad et al., 2011; Steinhorst and Kudla, 2013), while CBL2 and CBL3 are calcium sensors functioning in - 8 -
9 pollen tube growth (Steinhorst et al., 2015). Thus, we hypothesized that ap-3 pollen tubes might be compromised in their Ca 2+ gradient or signaling, leading to reduced pollen tube growth. To test this hypothesis, we introduced a Pro UBQ10 :YC3.6 transgene (Monshausen et al., 2008; Behera et al., 2015), which expresses the Förster (fluorescence) resonance energy transfer (FRET)-based genetically modified Ca 2+ indicator Yellow Cameleon YC3.6, into ap-3δ. FRET analysis showed that compared to wild-type pollen tubes (Fig. 6A, 6C), growing ap-3δ pollen tubes exhibited a significantly reduced Ca 2+ gradient at the tip (Fig. 6B, 6C), consistent with the reduced growth of mutant pollen tubes. To provide further evidence that a reduced Ca 2+ gradient resulted in compromised pollen tube growth in the ap-3 mutants, we tested the effect of enhanced Ca 2+ concentration by supplementing the growth medium with a higher concentration of extracellular Ca 2+, using 5 mm instead of 2 mm Ca 2+ in the regular germination medium. Although both wild-type and ap-3δ pollen tubes responded to the increased Ca 2+ by enhanced tube growth (Fig. 6D), ap-3δ pollen tubes were more sensitive than those of wild type. The ap-3δ pollen tubes doubled their length after 4 hr germination, whereas those of wild type only increased around 40% (Fig. 6D). This result supports the idea that there is reduced Ca 2+ signaling in ap-3δ pollen tubes Discussion The roles of AP-1 and AP-2 in plants have been well characterized, especially during reproductive processes (Fan et al., 2013; Park et al., 2013; Wang et al., 2016). Here we demonstrated that AP-3, the AP complex that sorts proteins at the Golgi to vacuoles, mediates pollen tube growth and male fertility (Fig. 1, Fig. 2). Mutants in each AP-3 component showed a reduction of pollen tube growth both in vitro and in vivo (Fig. 3), suggesting AP-3 functions as a complex. The phenotypic defects of ap-3σ were the weakest (Fig. 2, Fig. 3). Because the σ subunit stabilizes AP complexes (Boehm and Bonifacino, 2001; Robinson, 2004), we speculate that this subunit is not essential for AP-3. Alternatively, the σ subunits of other APs may perform redundant roles as a compensation mechanism. Among the established tonoplast cargoes that are sorted by AP-3 during their vacuolar trafficking, PAT10 is an important player in pollen tube growth (Fig. 5, - 9 -
10 Supplemental Fig. 6); functional loss of PAT10 affected male fertility (Zhou et al., 2013). By using sporophytically-complemented transgenic materials, we demonstrated that PAT10 functions in male gametophytes to promote pollen tube growth (Supplemental Fig. 6). CBL2 and CBL3, two calcium sensors, rely on PAT10 for their tonoplast association and functionality (Zhou et al., 2013; Zhang et al., 2015). Indeed, functional loss of both CBL2 and CBL3 impaired male fertility (Steinhorst et al., 2015). The CBL2/CBL3 study was performed using a homozygous cbl2 cbl3 mutant, in which sporophytic defects would have enhanced the defects of pollen tube growth (Steinhorst et al., 2015). Nevertheless, these results suggest a key role for the tonoplast calcium sensors in pollen tubes. Indeed, by FRET analysis of the YC3.6 probe, we demonstrated that growing pollen tubes with a functional loss of AP-3 contained a reduced Ca 2+ gradient (Fig. 6), which is consistent with the reduced pollen tube growth (Hepler et al., 2012). Functional loss of AP-3 caused not only mis-targeting of PAT10, which is important for pollen tube growth, but also defects in dynamic vacuolar organization (Fig. 4). By using an AP-3-independent tonoplast protein VHA-a3, we demonstrated that pollen tubes of the ap-3 mutants contained a simplified structure of vacuoles (Fig. 4). Unlike the extensive tubular vacuoles in wild-type pollen tubes, which extended to the subapical region right behind the apex, those in ap-3 pollen tubes were excluded from the subapical zone (Fig. 4). Indeed, mutations in other AP-3 subunits also resulted in defects in morphology and function of lytic vacuoles in root cells (Feraru et al., 2010; Zwiewka et al., 2011). Because of the key role of vacuoles in providing turgor pressure and facilitating ion homeostasis, such defective vacuolar organization might have contributed to the reduction in pollen tube growth. Despite the role of AP-3 in pollen tube growth, defects of ap-3 mutants have low penetrance (Table 1), whereas mutations at the HOPS complex, involved in vesicle-vacuole fusion, resulted in male gametophytic lethality (Hao et al., 2016). A likely explanation is that compensating pathways of vacuolar trafficking, such as Rab5-mediated (Cui et al., 2014; Ebine et al., 2014; Singh et al., 2014) and AP-4-mediated (Fuji et al., 2016), might help to maintain the dynamic organization of vacuoles and provide ion homeostasis
11 Materials and Methods Plant materials, growth, and transformation The T-DNA insertion lines ap-3δ/pat4-2 (SALK_069881) (Niihama et al., 2009; Zwiewka et al., 2011), ap-3β/pat2-2 (SAIL_1258_G03) (Niihama et al., 2009; Feraru et al., 2010), ap-3µ (SALK_064486) (Niihama et al., 2009), and ap-3σ (SAIL_269_F04) were obtained from the Arabidopsis Biological Resource Center (ABRC). Other materials, including Pro LAT52 :GUS (Li et al., 2013), Pro UBQ10 :YC3.6 (Behera et al., 2015), PAT10g-GFP (Zhou et al., 2013), Pro UBQ10 :VHA-a3-YFP (Feng et al., 2017), and Pro UBQ10 :CBL2-RFP (Zhang et al., 2015) were described previously. The Arabidopsis (Arabidopsis thaliana) Columbia-0 ecotype was used as the wild type. Transgenic plants were selected on half-strength MS medium supplemented with 30 µg/ml Basta salts (Sigma-Aldrich) or 50 µg/ml Hygromycin (Roche) RT-qPCR The extraction of total RNA, reverse transcription, and RT-qPCR were performed as described (Zhou et al., 2013). Primers for all AP-3 mutants were described (Feng et al., 2017b). Primers used for RT-qPCR were ZP5314/ZP5315 for VAMP711, ZP5316/ZP5317 for VAMP712, ZP5318/ZP5319 for VAMP713, ZP5363/ZP5364 for SUC4, and ZP691/ZP692 for PAT10. Primers for GAPDH and TUBULIN2 were as described (Zhou et al., 2013). All primers are listed in Supplemental Table DNA constructs Primers used for cloning of AP-3δ are ZP4427/ZP4428. The entry vector for AP-3δ was generated in the pentr/d/topo backbone (Invitrogen). The entry vectors for VHA-a3 (Feng et al., 2017) and CBL2 (Zhang et al., 2015) were described previously. Expression vectors were generated by combining entry vectors and the destination vector Pro UBQ10 :GW-RFP (Zhang et al., 2015) in LR reactions using LR Clonase II (Invitrogen). PCR amplifications used Phusion hot-start high-fidelity DNA polymerase with the annealing temperature and extension times recommended by the manufacturer (Thermofisher). Entry vectors were sequenced, and sequences were analyzed using Vector NTI. All primers are listed in Supplemental Table
12 Phenotypic analysis of pollen development and tube growth Methods to analyze pollen development, including Alexander staining, DAPI staining, and SEM, were performed as described (Johnson-Brousseau and McCormick, 2004; Li et al., 2013). Methods to analyze pollen tube growth in vitro were performed as described (Boavida and McCormick, 2007). Methods to analyze pollen tube growth in vivo by aniline blue staining and by histochemical GUS staining were performed as described (Li et al., 2013) Fluorescence microscopy Fluorescent images were captured using a Zeiss LSM 880 confocal laser scanning microscope (CLSM) with a 40/1.3 oil objective. GFP and YFP fusions were excited at 488 nm with a VIS-argon laser; RFP fusions were excited at 561 nm with a VIS-DPSS561 laser diode. Pollen tubes double-labeled with GFP and RFP fusions were captured using alternate line switching mode with a multi-track function. Fluorescence was detected using a 505- to 550-nm band-pass filter for GFP/YFP/OG or a 575- to 650-nm band-pass filter for RFP. Z-stack images were recorded with a step size of 0.3 μm, image dimension of , pinhole at 1 airy unit. Time-lapse imaging for generating movie clips was performed as an interval of 0.5 sec. Each movie clip was generated from 100 slides of still images. Image processing was performed with the Zeiss LSM image processing software (Zeiss). 3-D surface renderings were performed with Imaris7.0 software Accession Numbers. Arabidopsis Genome Initiative locus identifiers for the genes mentioned in this article are: AP-3δ, At1g48760; AP-3β, At3g55480; AP-3µ, At1g56590; AP-3σ, At3g50860; CBL2, At5g55990; PAT10, At3g51390; SUC4, At1g09960; VAMP711, At4g32150; VAMP712, At2g25340; VAMP713, At5g11150; VHA-a3, At4g Supplemental Data The following materials are available in the online version of this article. Supplemental Fig. 1. Functional loss of AP-3 does not compromise pollen development
13 Supplemental Fig. 2. Functional loss of AP-3 reduces pollen tube growth in vitro. Supplemental Fig. 3. Functional loss of AP-3 reduces pollen tube growth in vivo. Supplemental Fig. 4. Functional loss of AP-3δ does not impair pollen tube guidance. Supplemental Fig. 5. Expression of genes encoding cellular cargos of AP-3 in pollen. Supplemental Fig. 6. AP-3-mediated tonoplast protein PAT10 is important for pollen tube growth. Supplemental Movie 1. Vacuolar dynamics in a wild-type pollen tube. Supplemental Movie 2. Vacuolar dynamics in an ap-3δ pollen tube. Supplemental Movie 3. 3D surface rendering of VHA-a3-YFP-labeled vacuoles in a wild-type pollen tube. Supplemental Movie 4. 3D surface rendering of VHA-a3-YFP-labeled vacuoles in an ap-3δ pollen tube. Supplemental Table 1. Oligos used in this study Acknowledgements We thank ABRC for plant materials. We are grateful for language editing by Prof. Sheila McCormick. The authors declare that there is no conflict of interest. Table 1. AP-3 loss-of-function resulted in defective male transmission. Parents F1 progenies Female X Male Genotype Expected Ratio Observed Ratio ap-3δ +/- X wild type ap-3δ +/+:+/- 1:1 74:68 wild type X ap-3δ +/- ap-3δ +/+:+/- 1:1 87:7 a ap-3δ +/- X ap-3δ +/- ap-3δ +/+:+/-:-/- 1:2:1 13:20:2 b ap-3β +/- X wild type R:S* 1:1 94:87 wild type X ap-3β +/- R:S* 1:1 11:159 a ap-3β +/- X ap-3β +/- R:S* 3:1 369:211 c ap-3μ +/- X wild type ap-3μ +/+:+/- 1:1 99:
14 wild type X ap-3μ +/- ap-3μ +/+:+/- 1:1 170:8 a ap-3μ +/- X ap-3μ +/- ap-3μ +/+:+/-:-/- 1:2:1 42:38:16 b ap-3σ +/- X wild type R:S* 1:1 125:110 wild type X ap-3σ +/- R:S* 1:1 39:56 a ap-3σ +/- X ap-3σ +/- R:S* 3:1 409:150 c a Significantly different from the segregation ratio 1:1 ( 2, P<0.01 ). b Significantly different from the segregation ratio 1:2:1 ( 2, P<0.01). c Significantly different from the segregation ratio 3:1 ( 2 2, P<0.01). *: R for Basta-resistant and S for Basta-sensitive. Basta sensitivity was used to distinguish the mutant copy of ap-3β or ap-3σ, resistant to Basta salt, from its wild-type copy Figure Legends Figure 1. Mutations at AP 3 subunits result in reduced seed set. (A) Schematic illustration of T-DNA insertions within the genomic regions of AP-3 subunit-encoding genes, AP-3δ, AP-3β, AP-3µ, and AP-3σ. Arrowheads point at the T-DNA insertion sites. Arrows at the genomic region of AP-3σ indicate the binding sites of RT-qPCR primers. (B) Quantitative analysis of seed set in wild type (WT) and the ap-3 mutants. Results are means ± standard deviation (SD, N=20). Each mutant is significantly different from wild type in seed set as indicated by asterisks (One-way ANOVA, Dunnett s multiple comparison test, P<0.05). (C-D) From left to right: a representative silique from WT, ap-3δ, ap-3β, ap-3µ, or ap-3σ. Close-up of the bottom part of (C) is shown in (D). Arrowheads point at undeveloped ovules. Bars = 2 mm for (C); 500 µm for (D). Figure 2. Functional loss of AP 3 reduces pollen tube growth in vitro. (A-E) In vitro pollen tubes from WT (A), ap-3δ (B), ap-3β (C), ap-3µ (D), or ap-3σ (E) at 4 hr after germination. Two representative pollen tubes from each genotype are highlighted in pink. (F-H) The germination ratio (F), length (G), or apical width (H) of pollen tubes after 4 hours incubation in germination medium. Results shown are means ± SD (n=300 for F, n= 100 for G, n=60 for H). All mutants are not significantly different from wild type in the germination ratio (One-way ANOVA, Dunnett s multiple comparison test, P>0.05). Each mutant is significantly different from wild type in the length or width of pollen tubes as indicated by asterisks (One-way ANOVA, Dunnett s multiple comparison test, P<0.05). Bars = 50 µm. Figure 3. Functional loss of AP 3 affects the growth but not the guidance of pollen tubes in vivo. (A-J) Representative aniline blue staining of wild-type pistils emasculated and hand-pollinated with pollen from wild type (A, F), ap-3δ (B, G), ap-3β (C, H), ap-3µ (D, I), or ap-3σ (E, J) at 9 hours after pollination (HAP). Arrows point at the front of in
15 vivo growing pollen tubes; arrowheads point at the micropyle where pollen tubes arrive. The relative distance pollen tubes travel within the transmitting tract is quantified with 15 pistils. Results are shown at the bottom (means ± SD). Each mutant is significantly different from wild type in the length of in vivo growing pollen tubes (One-way ANOVA, Dunnett s multiple comparison test, P<0.05). (K-O) Scanning electron micrographs (SEMs) of wild-type ovules hand-pollinated with pollen from wild type (K), ap-3δ (L), ap-3β (M), ap-3µ (N), or ap-3σ (O) at 9 HAP. Pollen tubes growing toward the micropyle are false-colored pink. Bars=200 μm for (A-E); 100 μm for (F-J); 20 μm for (K-O). Figure 4. Vacuolar organization is compromised in ap 3 pollen tubes. (A-B) a growing wild-type (A) or ap-3δ pollen tube (B) expressing VHA-a3-YFP. From top to bottom: the YFP channel image at the mid-optical plane; merge of the YFP and transmission channels; 3D-surface rendering of CLSM images. Dotted lines illustrate the silhouettes of the pollen tubes. Over 30 pollen tubes for each genotype were examined with similar results. Bars = 10 μm. Figure 5. Tonoplast association of PAT10 and CBL2 relies on AP 3 in pollen tubes. (A-B) Confocal laser scanning fluorescence micrographs (CLSMs) of a pollen tube expressing PAT10g-GFP;VHA-a3-RFP in wild type (A) or in ap-3δ (B). (C) CLSM of a pollen tube expressing PAT10g-GFP in the complemented AP-3δ-RFP;ap-3δ. (D) Pollen tube length. Results shown are means ± SD (n=100). Means with different letters indicate significant difference of pollen tube length (One-way ANOVA, Dunnett s multiple comparison test, P<0.05). (E-F) A pollen tube stably transformed with Pro UBQ10 :CBL2-RFP in wild type (E) or in ap-3δ (F). (G-I) A pollen tube stably transformed with Pro UBQ10 :CBL2-RFP;PAT10g-GFP in ap-3β (G), ap-3µ (H), or ap-3σ (I). G/T or R/T indicates merge of the GFP or RFP channel and the transmission channel; G/R/T indicates merge of the GFP channel, the RFP channel, and the transmission channel. Over 30 pollen tubes for each genotype were examined with similar results for data shown in (A-C) and (E-I). Bars = 10 μm. Figure 6. AP 3 loss of function compromises the tip focused Ca2+ gradient. (A-B) CLSM of a pollen tube expressing the Ca 2+ sensor YC3.6 in wild type (A) or in ap-3δ (B). Images are representative of 20 pollen tubes for each genotype. Cytosolic Ca 2+ levels were calibrated as described in Materials and Methods and pseudocolored according to the scale at the left. (C) Quantification of fluorescence intensity as the ratio between YFP signals and CFP signals. Results are means SD (n=20). Asterisk indicates significant difference (t-test, P<0.01). (D) Pollen tube length after 4 hr germination in a medium containing 2 mm Ca 2+ or 5 mm Ca 2+. Results are means ± SD (n=100). Asterisk indicates significantly different responses of pollen tube growth upon increased exogenous Ca 2+ levels (t-test, P<0.01). Bars = 10 μm. Reference Bassham DC, Brandizzi F, Otegui MS, Sanderfoot AA (2008) The secretory system of Arabidopsis. Arabidopsis Book 6: e0116 Behera S, Wang N, Zhang C, Schmitz-Thom I, Strohkamp S, Schultke S, Hashimoto K, Xiong L, Kudla J (2015) Analyses of Ca 2+ dynamics using a ubiquitin-10 promoter-driven
16 Yellow Cameleon 3.6 indicator reveal reliable transgene expression and differences in cytoplasmic Ca 2+ responses in Arabidopsis and rice (Oryza sativa) roots. New Phytol 206: Boavida LC, McCormick S (2007) Temperature as a determinant factor for increased and reproducible in vitro pollen germination in Arabidopsis thaliana. Plant J 52: Boehm M, Bonifacino JS (2001) Adaptins: the final recount. Mol Biol Cell 12: Cheung AY, Duan QH, Costa SS, de Graaf BH, Di Stilio VS, Feijo J, Wu HM (2008) The dynamic pollen tube cytoskeleton: live cell studies using actin-binding and microtubule-binding reporter proteins. Mol Plant 1: Cheung AY, Wu HM (2007) Structural and functional compartmentalization in pollen tubes. J Exp Bot 58: Cheung AY, Wu HM (2008) Structural and signaling networks for the polar cell growth machinery in pollen tubes. Annu Rev Plant Biol 59: Cui Y, Zhao Q, Gao C, Ding Y, Zeng Y, Ueda T, Nakano A, Jiang L (2014) Activation of the Rab7 GTPase by the MON1-CCZ1 complex is essential for PVC-to-vacuole trafficking and plant growth in Arabidopsis. Plant Cell 26: Ebine K, Inoue T, Ito J, Ito E, Uemura T, Goh T, Abe H, Sato K, Nakano A, Ueda T (2014) Plant vacuolar trafficking occurs through distinctly regulated pathways. Curr Biol 24: Fan L, Hao H, Xue Y, Zhang L, Song K, Ding Z, Botella MA, Wang H, Lin J (2013) Dynamic analysis of Arabidopsis AP2σ subunit reveals a key role in clathrin-mediated endocytosis and plant development. Development 140: Feng QN, Song SJ, Yu SX, Wang JG, Li S, Zhang Y (2017) Adaptor Protein-3-dependent vacuolar trafficking involves a subpopulation of COPII and HOPS tethering proteins. Plant Physiol 174: Feng QN, Zhang Y, Li S (2017) Tonoplast targeting of VHA-a3 relies on a Rab5-mediated but Rab7-independent vacuolar trafficking route. J Integr Plant Biol 59: Feraru E, Paciorek T, Feraru MI, Zwiewka M, De Groodt R, De Rycke R, Kleine-Vehn J, Friml J (2010) The AP-3 β adaptin mediates the biogenesis and function of lytic vacuoles in Arabidopsis. Plant Cell 22: Fuji K, Shirakawa M, Shimono Y, Kunieda T, Fukao Y, Koumoto Y, Takahashi H, Hara-Nishimura I, Shimada T (2016) The adaptor complex AP-4 regulates vacuolar protein sorting at the trans-golgi network by interacting with VACUOLAR SORTING RECEPTOR1. Plant Physiol 170: Gadeyne A, Sanchez-Rodriguez C, Vanneste S, Di Rubbo S, Zauber H, Vanneste K, Van Leene J, De Winne N, Eeckhout D, Persiau G, Van De Slijke E, Cannoot B, Vercruysse L, Mayers JR, Adamowski M, Kania U, Ehrlich M, Schweighofer A, Ketelaar T, Maere S, Bednarek SY, Friml J, Gevaert K, Witters E, Russinova E, Persson S, De Jaeger G, Van Damme D (2014) The TPLATE adaptor complex drives clathrin-mediated endocytosis in plants. Cell 156: Hao L, Liu J, Zhong S, Gu H, Qu LJ (2016) AtVPS41-mediated endocytic pathway is essential for pollen tube-stigma interaction in Arabidopsis. Proc Natl Acad Sci U S A 113: Hepler PK, Kunkel JG, Rounds CM, Winship LJ (2012) Calcium entry into pollen tubes. Trends
17 Plant Sci 17: Hepler PK, Vidali L, Cheung AY (2001) Polarized cell growth in higher plants. Annu Rev Cell Dev Biol 17: Hicks GR, Rojo E, Hong S, Carter DG, Raikhel NV (2004) Geminating pollen has tubular vacuoles, displays highly dynamic vacuole biogenesis, and requires VACUOLESS1 for proper function. Plant Physiol 134: Hirst J, D. Barlow L, Francisco GC, Sahlender DA, Seaman MNJ, Dacks JB, Robinson MS (2011) The fifth adaptor protein complex. PLoS Biol 9: e Hirst J, Schlacht A, Norcott JP, Traynor D, Bloomfield G, Antrobus R, Kay RR, Dacks JB, Robinson MS (2014) Characterization of TSET, an ancient and widespread membrane trafficking complex. Elife 3: e02866 Johnson-Brousseau SA, McCormick S (2004) A compendium of methods useful for characterizing Arabidopsis pollen mutants and gametophytically-expressed genes. Plant J 39: Johnson MA, Preuss D (2002) Plotting a course: multiple signals guide pollen tubes to their targets. Dev Cell 2: Kim SY, Xu ZY, Song K, Kim DH, Kang H, Reichardt I, Sohn EJ, Friml J, Juergens G, Hwang I (2013) Adaptor protein complex 2-mediated endocytosis is crucial for male reproductive organ development in Arabidopsis. Plant Cell 25: Konrad KR, Wudick MM, Feijo JA (2011) Calcium regulation of tip growth: new genes for old mechanisms. Curr Opin Plant Biol 14: Li S, Ge FR, Xu M, Zhao XY, Huang GQ, Zhou LZ, Wang JG, Kombrink A, McCormick S, Zhang XS, Zhang Y (2013) Arabidopsis COBRA-LIKE 10, a GPI-anchored protein, mediates directional growth of pollen tubes. Plant J 74: McCormick S (2004) Control of male gametophyte development. Plant Cell 16 Suppl: S Monshausen GB, Messerli MA, Gilroy S (2008) Imaging of the Yellow Cameleon 3.6 indicator reveals that elevations in cytosolic Ca 2+ follow oscillating increases in growth in root hairs of Arabidopsis. Plant Physiol 147: Niihama M, Takemoto N, Hashiguchi Y, Tasaka M, Morita MT (2009) ZIP genes encode proteins involved in membrane trafficking of the TGN-PVC/vacuoles. Plant Cell Physiol 50: Park M, Song K, Reichardt I, Kim H, Mayer U, Stierhof Y-D, Hwang I, Jürgens G (2013) Arabidopsis μ-adaptin subunit AP1M of adaptor protein complex 1 mediates late secretory and vacuolar traffic and is required for growth. Proc Nat Acad Sci USA Robinson MS (2004) Adaptable adaptors for coated vesicles. Trends Cell Biol 14: Rojo E, Gillmor CS, Kovaleva V, Somerville CR, Raikhel NV (2001) VACUOLELESS1 is an essential gene required for vacuole formation and morphogenesis in Arabidopsis. Dev Cell 1: Singh MK, Kruger F, Beckmann H, Brumm S, Vermeer JE, Munnik T, Mayer U, Stierhof YD, Grefen C, Schumacher K, Jurgens G (2014) Protein delivery to vacuole requires SAND protein-dependent Rab GTPase conversion for MVB-vacuole fusion. Curr Biol 24: Steinhorst L, Kudla J (2013) Calcium - a central regulator of pollen germination and tube
18 growth. Biochim Biophys Acta 1833: Steinhorst L, Mahs A, Ischebeck T, Zhang C, Zhang X, Arendt S, Schultke S, Heilmann I, Kudla J (2015) Vacuolar CBL-CIPK12 Ca 2+ -sensor-kinase complexes are required for polarized pollen tube growth. Curr Biol 25: Teh OK, Shimono Y, Shirakawa M, Fukao Y, Tamura K, Shimada T, Hara-Nishimura I (2013) The AP-1 µ adaptin is required for KNOLLE localization at the cell plate to mediate cytokinesis in Arabidopsis. Plant Cell Physiol 54: Uemura T, Ueda T (2014) Plant vacuolar trafficking driven by RAB and SNARE proteins. Curr Opin Plant Biol 22: Van Damme D, Coutuer S, De Rycke R, Bouget FY, Inze D, Geelen D (2006) Somatic cytokinesis and pollen maturation in Arabidopsis depend on TPLATE, which has domains similar to coat proteins. Plant Cell 18: Viotti C, Kruger F, Krebs M, Neubert C, Fink F, Lupanga U, Scheuring D, Boutte Y, Frescatada-Rosa M, Wolfenstetter S, Sauer N, Hillmer S, Grebe M, Schumacher K (2013) The Endoplasmic reticulum is the main membrane source for biogenesis of the lytic vacuole in Arabidopsis. Plant Cell 25: Wang JG, Li S, Zhao XY, Zhou LZ, Huang GQ, Feng C, Zhang Y (2013) HAPLESS13, the Arabidopsis µ1 adaptin, is essential for protein sorting at the trans-golgi network/early endosome. Plant Physiol 162: Wang T, Liang L, Xue Y, Jia PF, Chen W, Zhang MX, Wang YC, Li HJ, Yang WC (2016) A receptor heteromer mediates the male perception of female attractants in plants. Nature Wolfenstetter S, Wirsching P, Dotzauer D, Schneider S, Sauer N (2012) Routes to the tonoplast: the sorting of tonoplast transporters in Arabidopsis mesophyll protoplasts. Plant Cell 24: Wudick MM, Luu DT, Tournaire-Roux C, Sakamoto W, Maurel C (2014) Vegetative and sperm cell-specific aquaporins of Arabidopsis highlight the vacuolar equipment of pollen and contribute to plant reproduction. Plant Physiol 164: Yamaoka S, Shimono Y, Shirakawa M, Fukao Y, Kawase T, Hatsugai N, Tamura K, Shimada T, Hara-Nishimura I (2013) Identification and dynamics of Arabidopsis adaptor protein-2 complex and its involvement in floral organ development. Plant Cell 25: Zhang Y, Li S, Zhou L-Z, Fox E, Pao J, Sun W, Zhou C, McCormick S (2011) Overexpression of Arabidopsis thaliana PTEN caused accumulation of autophagic bodies in pollen tubes by disrupting phosphatidylinositol 3-phosphate dynamics. Plant J 68: Zhang YL, Li E, Feng QN, Zhao XY, Ge FR, Zhang Y, Li S (2015) Protein palmitoylation is critical for the polar growth of root hairs in Arabidopsis. BMC Plant Biol 15: 50 Zhou LZ, Li S, Feng QN, Zhang YL, Zhao X, Zeng YL, Wang H, Jiang L, Zhang Y (2013) PROTEIN S-ACYL TRANSFERASE10 is critical for development and salt tolerance in Arabidopsis. Plant Cell 25: Zwiewka M, Feraru E, Moller B, Hwang I, Feraru MI, Kleine-Vehn J, Weijers D, Friml J (2011) The AP-3 adaptor complex is required for vacuolar function in Arabidopsis. Cell Res 21:
19 Figure 1. Mutations at AP 3 subunits resulted in reduced seed set. (A) Schematic illustration of T DNA insertions within the genomic regions of AP 3 subunit encoding genes, AP 3δ, AP 3β, AP 3µ, and AP 3σ. Arrowheads point at the T DNA insertion sites. Arrows at the genomic region of AP 3σ indicate the binding sites of RT PCR primers. (B) Quantitative analysis of seed set in wild type (WT) and the ap 3 mutants. Results are means ± standard deviation (SD, N=20). Each mutant is significantly different from wild type in seed set as indicated by asterisks (One way ANOVA, Dunnett s multiple comparison test, P<0.05). (C D) From left to right: a representative silique from WT, ap 3δ, ap 3β, ap 3µ, or ap 3σ. Close up of the bottom part of (C) is shown in (D). Arrowheads point at undeveloped ovules. Bars = 2 mm for (C); 500 µm for (D).
20 Figure 2. Functional loss of AP 3 reduced pollen tube growth in vitro. (A E) In vitro pollen tubes from WT (A), ap 3δ (B), ap 3β (C), ap 3µ (D), or ap 3σ (E) at 4 hr after germination. Two representative pollen tubes from each genotype are highlighted in pink. (F H) The germination ratio (F), length (G), or apical width (H) of pollen tubes after 4 hours incubation in germination medium. Results shown are means ± SD (n=300 for F, n= 100 for G, n=60 for H). All mutants are not significantly different from wild type in the germination ratio (One way ANOVA, Dunnett s multiple comparison test, P>0.05). Each mutant is significantly different from wild type in the length or width of pollen tubes as indicated by asterisks (One way ANOVA, Dunnett s multiple comparison test, P<0.05). Bars = 50 µm.
21 Figure 3. Functional loss of AP 3 affected the growth but not guidance of pollen tubes in vivo. (A J) Representative aniline blue staining of wild type pistils emasculated and hand pollinated with pollen from wild type (A, F), ap 3δ (B, G), ap 3β (C, H), ap 3µ (D, I), or ap 3σ (E, J) at 9 hours after pollination (HAP). Arrows point at the front of in vivo growing pollen tubes; arrowheads point at the micropyle where pollen tubes arrive. The relative distance pollen tubes travel within the transmitting tract is quantified with 15 pistils. Results are shown at the bottom (means ± SD). Each mutant is significantly different from wild type in the length of in vivo growing pollen tubes (One way ANOVA, Dunnett s multiple comparison test, P<0.05). (K O) Scanning electron micrographs (SEMs) of wild type ovules hand pollinated with pollen from wild type (K), ap 3δ (L), ap 3β (M), ap 3µ (N), or ap 3σ (O) at 9 HAP. Pollen tubes growing toward the micropyle are false colored pink. Bars=200 μm for (A E); 100 μm for (F J); 20 μm for (K O).
22 Figure 4. Vacuolar organization was compromised in ap 3 pollen tubes. (A B) a growing wild type (A) or ap 3δ pollen tube (B) expressing VHA a3 YFP. From top to bottom: the YFP channel image at the mid optical plane; merge of the YFP and transmission channels; 3D surface rendering of CLSM images. Dotted lines illustrate the silhouettes of the pollen tubes. Bars = 10 μm.
23
24 Figure 6. AP 3 loss of function compromised the tip focused Ca 2+ gradient. (A B) CLSM of a pollen tube expressing the Ca 2+ sensor YC3.6 in wild type (A) or in ap 3δ (B). Cytosolic Ca 2+ levels were calibrated as described in Materials and Methods and pseudocolored according to the scale at the left. (C) Quantification of fluorescence intensity as the ratio between YFP signals and CFP signals. Results are means SD (n=20). Asterisk indicates significant difference (t test, P<0.01). (D) Pollen tube length after 4 hr germination in a medium containing 2 mm Ca 2+ or 5 mm Ca 2+. Results are means ± SD (n=100). Asterisk indicates significantly different responses of pollen tube growth upon increased exogenous Ca 2+ levels (t test, P<0.01). Bars = 10 μm.
25 Parsed Citations Bassham DC, Brandizzi F, Otegui MS, Sanderfoot AA (2008) The secretory system of Arabidopsis. Arabidopsis Book 6: e0116 Behera S, Wang N, Zhang C, Schmitz-Thom I, Strohkamp S, Schultke S, Hashimoto K, Xiong L, Kudla J (2015) Analyses of Ca2+ dynamics using a ubiquitin-10 promoter-driven Yellow Cameleon 3.6 indicator reveal reliable transgene expression and differences in cytoplasmic Ca2+ responses in Arabidopsis and rice (Oryza sativa) roots. New Phytol 206: Boavida LC, McCormick S (2007) Temperature as a determinant factor for increased and reproducible in vitro pollen germination in Arabidopsis thaliana. Plant J 52: Boehm M, Bonifacino JS (2001) Adaptins: the final recount. Mol Biol Cell 12: Cheung AY, Duan QH, Costa SS, de Graaf BH, Di Stilio VS, Feijo J, Wu HM (2008) The dynamic pollen tube cytoskeleton: live cell studies using actin-binding and microtubule-binding reporter proteins. Mol Plant 1: Cheung AY, Wu HM (2007) Structural and functional compartmentalization in pollen tubes. J Exp Bot 58: Cheung AY, Wu HM (2008) Structural and signaling networks for the polar cell growth machinery in pollen tubes. Annu Rev Plant Biol 59: Cui Y, Zhao Q, Gao C, Ding Y, Zeng Y, Ueda T, Nakano A, Jiang L (2014) Activation of the Rab7 GTPase by the MON1-CCZ1 complex is essential for PVC-to-vacuole trafficking and plant growth in Arabidopsis. Plant Cell 26: Ebine K, Inoue T, Ito J, Ito E, Uemura T, Goh T, Abe H, Sato K, Nakano A, Ueda T (2014) Plant vacuolar trafficking occurs through distinctly regulated pathways. Curr Biol 24: Fan L, Hao H, Xue Y, Zhang L, Song K, Ding Z, Botella MA, Wang H, Lin J (2013) Dynamic analysis of Arabidopsis AP2σ subunit reveals a key role in clathrin-mediated endocytosis and plant development. Development 140: Feng QN, Song SJ, Yu SX, Wang JG, Li S, Zhang Y (2017) Adaptor Protein-3-dependent vacuolar trafficking involves a subpopulation of COPII and HOPS tethering proteins. Plant Physiol 174: Feng QN, Zhang Y, Li S (2017) Tonoplast targeting of VHA-a3 relies on a Rab5-mediated but Rab7-independent vacuolar trafficking route. J Integr Plant Biol 59: Feraru E, Paciorek T, Feraru MI, Zwiewka M, De Groodt R, De Rycke R, Kleine-Vehn J, Friml J (2010) The AP-3 β adaptin mediates the biogenesis and function of lytic vacuoles in Arabidopsis. Plant Cell 22:
Wave line information sheet
 Wave line information sheet Niko Geldner May 200 Wave constructs and lines, as well as pnigel vector series were published: Rapid, combinatorial analysis of membrane compartments in intact plants with
Wave line information sheet Niko Geldner May 200 Wave constructs and lines, as well as pnigel vector series were published: Rapid, combinatorial analysis of membrane compartments in intact plants with
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins
 13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
CELB40060 Membrane Trafficking in Animal Cells. Prof. Jeremy C. Simpson. Lecture 2 COPII and export from the ER
 CELB40060 Membrane Trafficking in Animal Cells Prof. Jeremy C. Simpson Lecture 2 COPII and export from the ER Today s lecture... The COPII coat - localisation and subunits Formation of the COPII coat at
CELB40060 Membrane Trafficking in Animal Cells Prof. Jeremy C. Simpson Lecture 2 COPII and export from the ER Today s lecture... The COPII coat - localisation and subunits Formation of the COPII coat at
EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA
 EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA Item Type text; Electronic Thesis Authors Bergstrand, Lauren Janel Publisher The University of Arizona. Rights Copyright is held by the author. Digital
EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA Item Type text; Electronic Thesis Authors Bergstrand, Lauren Janel Publisher The University of Arizona. Rights Copyright is held by the author. Digital
The Pollen Tube: A Model System for Cell and Molecular Biology Studies
 Plant Cell Monogr (3) R. Malhó: The Pollen Tube DOI 10.1007/7089_041/Published online: 26 January 2006 Springer-Verlag Berlin Heidelberg 2006 The Pollen Tube: A Model System for Cell and Molecular Biology
Plant Cell Monogr (3) R. Malhó: The Pollen Tube DOI 10.1007/7089_041/Published online: 26 January 2006 Springer-Verlag Berlin Heidelberg 2006 The Pollen Tube: A Model System for Cell and Molecular Biology
The neuron as a secretory cell
 The neuron as a secretory cell EXOCYTOSIS ENDOCYTOSIS The secretory pathway. Transport and sorting of proteins in the secretory pathway occur as they pass through the Golgi complex before reaching the
The neuron as a secretory cell EXOCYTOSIS ENDOCYTOSIS The secretory pathway. Transport and sorting of proteins in the secretory pathway occur as they pass through the Golgi complex before reaching the
Cell Biology Review. The key components of cells that concern us are as follows: 1. Nucleus
 Cell Biology Review Development involves the collective behavior and activities of cells, working together in a coordinated manner to construct an organism. As such, the regulation of development is intimately
Cell Biology Review Development involves the collective behavior and activities of cells, working together in a coordinated manner to construct an organism. As such, the regulation of development is intimately
Protein Sorting, Intracellular Trafficking, and Vesicular Transport
 Protein Sorting, Intracellular Trafficking, and Vesicular Transport Noemi Polgar, Ph.D. Department of Anatomy, Biochemistry and Physiology Email: polgar@hawaii.edu Phone: 692-1422 Outline Part 1- Trafficking
Protein Sorting, Intracellular Trafficking, and Vesicular Transport Noemi Polgar, Ph.D. Department of Anatomy, Biochemistry and Physiology Email: polgar@hawaii.edu Phone: 692-1422 Outline Part 1- Trafficking
Pollen Tubes Lacking a Pair of K + Transporters Fail to Target Ovules in Arabidopsis C W OA
 This article is a Plant Cell Advance Online Publication. The date of its first appearance online is the official date of publication. The article has been edited and the authors have corrected proofs,
This article is a Plant Cell Advance Online Publication. The date of its first appearance online is the official date of publication. The article has been edited and the authors have corrected proofs,
23-. Shoot and root development depend on ratio of IAA/CK
 Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
** * * * Col-0 cau1 CAU1. Actin2 CAS. Actin2. Supplemental Figure 1. CAU1 affects calcium accumulation.
 Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Chapter 26 Profiling the GCS1 -Based Gamete Fusion Mechanism
 Chapter 26 Profiling the GCS1 -Based Gamete Fusion Mechanism Toshiyuki Mori Abstract Angiosperm double fertilization is composed of two sets of gamete fusion, in which two sperm cells released from a pollen
Chapter 26 Profiling the GCS1 -Based Gamete Fusion Mechanism Toshiyuki Mori Abstract Angiosperm double fertilization is composed of two sets of gamete fusion, in which two sperm cells released from a pollen
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL
 GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
AUXIN BINDING PROTEIN 1 (ABP1): A matter of fact. Chun-Ming Liu, Professor
 Commentary AUXIN BINDING PROTEIN 1 (ABP1): A matter of fact Chun-Ming Liu, Professor Editor-in-Chief Journal of Integrative Plant Biology (JIPB) Correspondence: cmliu@ibcas.ac.cn This article has been
Commentary AUXIN BINDING PROTEIN 1 (ABP1): A matter of fact Chun-Ming Liu, Professor Editor-in-Chief Journal of Integrative Plant Biology (JIPB) Correspondence: cmliu@ibcas.ac.cn This article has been
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
Supplemental Data. Chen and Thelen (2010). Plant Cell /tpc
 Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
Molecular Genetics of. Plant Development STEPHEN H. HOWELL CAMBRIDGE UNIVERSITY PRESS
 Molecular Genetics of Plant Development STEPHEN H. HOWELL CAMBRIDGE UNIVERSITY PRESS Contents Preface A Word on Genetic Nomenclature page xiii xvii 1 Approaches to the Study of Plant Development 1 Pattern
Molecular Genetics of Plant Development STEPHEN H. HOWELL CAMBRIDGE UNIVERSITY PRESS Contents Preface A Word on Genetic Nomenclature page xiii xvii 1 Approaches to the Study of Plant Development 1 Pattern
Supplementary Figure 1. Phenotype of the HI strain.
 Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
Reinforcement Unit 3 Resource Book. Meiosis and Mendel KEY CONCEPT Gametes have half the number of chromosomes that body cells have.
 6.1 CHROMOSOMES AND MEIOSIS KEY CONCEPT Gametes have half the number of chromosomes that body cells have. Your body is made of two basic cell types. One basic type are somatic cells, also called body cells,
6.1 CHROMOSOMES AND MEIOSIS KEY CONCEPT Gametes have half the number of chromosomes that body cells have. Your body is made of two basic cell types. One basic type are somatic cells, also called body cells,
Supplemental Data. Wang et al. (2014). Plant Cell /tpc
 Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
Supplementary information
 Supplementary information Statistical organelle dissection of Arabidopsis guard cells using image database LIPS Takumi Higaki *, Natsumaro Kutsuna, Yoichiroh Hosokawa, Kae Akita, Kazuo Ebine, Takashi Ueda,
Supplementary information Statistical organelle dissection of Arabidopsis guard cells using image database LIPS Takumi Higaki *, Natsumaro Kutsuna, Yoichiroh Hosokawa, Kae Akita, Kazuo Ebine, Takashi Ueda,
!"#$%&'%()*%+*,,%-&,./*%01%02%/*/3452*%3&.26%&4752*,,*1%%
 !"#$%&'%()*%+*,,%-&,./*%01%02%/*/3452*%3&.26%&4752*,,*1%% !"#$%&'(")*++*%,*'-&'./%/,*#01#%-2)#3&)/% 4'(")*++*% % %5"0)%-2)#3&) %%% %67'2#72'*%%%%%%%%%%%%%%%%%%%%%%%4'(")0/./% % 8$+&'&,+"/7 % %,$&7&/9)7$*/0/%%%%%%%%%%
!"#$%&'%()*%+*,,%-&,./*%01%02%/*/3452*%3&.26%&4752*,,*1%% !"#$%&'(")*++*%,*'-&'./%/,*#01#%-2)#3&)/% 4'(")*++*% % %5"0)%-2)#3&) %%% %67'2#72'*%%%%%%%%%%%%%%%%%%%%%%%4'(")0/./% % 8$+&'&,+"/7 % %,$&7&/9)7$*/0/%%%%%%%%%%
. Supplementary Information
 . Supplementary Information Supplementary Figure S1. Mature embryo sac observations. Supplementary Figure S2. STT observations. Supplementary Figure S3. Comparison of the PTB1 cdna with that of the mutant.
. Supplementary Information Supplementary Figure S1. Mature embryo sac observations. Supplementary Figure S2. STT observations. Supplementary Figure S3. Comparison of the PTB1 cdna with that of the mutant.
Regulation of Phosphate Homeostasis by microrna in Plants
 Regulation of Phosphate Homeostasis by microrna in Plants Tzyy-Jen Chiou 1 *, Kyaw Aung 1,2, Shu-I Lin 1,3, Chia-Chune Wu 1, Su-Fen Chiang 1, and Chun-Lin Su 1 Abstract Upon phosphate (Pi) starvation,
Regulation of Phosphate Homeostasis by microrna in Plants Tzyy-Jen Chiou 1 *, Kyaw Aung 1,2, Shu-I Lin 1,3, Chia-Chune Wu 1, Su-Fen Chiang 1, and Chun-Lin Su 1 Abstract Upon phosphate (Pi) starvation,
Heather Currinn, Benjamin Guscott, Zita Balklava, Alice Rothnie and Thomas Wassmer*
 Online Resources APP controls the formation of PI(3,5)P 2 vesicles through its binding of the PIKfyve complex. Cellular and Molecular Life Sciences Heather Currinn, Benjamin Guscott, Zita Balklava, Alice
Online Resources APP controls the formation of PI(3,5)P 2 vesicles through its binding of the PIKfyve complex. Cellular and Molecular Life Sciences Heather Currinn, Benjamin Guscott, Zita Balklava, Alice
CURRICULUM VITAE. Fellowships : JSPS Postdoctoral Fellowship for Research Abroad
 CURRICULUM VITAE Kentaro Tamura Birth: 22 Aug. 1976 Nationality: Japanese School of Food and Nutritional Sciences, University of Shizuoka Shizuoka 422-8526, Japan Phone: +81 (0)54 264 5707 E-mail: tamura@u-shizuoka-ken.ac.jp
CURRICULUM VITAE Kentaro Tamura Birth: 22 Aug. 1976 Nationality: Japanese School of Food and Nutritional Sciences, University of Shizuoka Shizuoka 422-8526, Japan Phone: +81 (0)54 264 5707 E-mail: tamura@u-shizuoka-ken.ac.jp
Chapter 6: A Tour of the Cell
 AP Biology Reading Guide Fred and Theresa Holtzclaw Chapter 6: A Tour of the Cell Name Period Chapter 6: A Tour of the Cell Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry
AP Biology Reading Guide Fred and Theresa Holtzclaw Chapter 6: A Tour of the Cell Name Period Chapter 6: A Tour of the Cell Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry
Mapping interactions between subunits of the endocytic TPLATE Adaptor Complex. Daniël Van Damme Advanced live cell imaging group October 9 th 2018
 Mapping interactions between subunits of the endocytic TPLATE Adaptor Complex Daniël Van Damme Advanced live cell imaging group October 9 th 2018 endocytosis - ubiquitous throughout plant life but mechanistically
Mapping interactions between subunits of the endocytic TPLATE Adaptor Complex Daniël Van Damme Advanced live cell imaging group October 9 th 2018 endocytosis - ubiquitous throughout plant life but mechanistically
The Plant Cell, November. 2017, American Society of Plant Biologists. All rights reserved
 The Genetics of Floral Development Teaching Guide Overview The development of flowers in angiosperm plants provided a critical evolutionary advantage, allowing more options for pollen dispersal and seed
The Genetics of Floral Development Teaching Guide Overview The development of flowers in angiosperm plants provided a critical evolutionary advantage, allowing more options for pollen dispersal and seed
Report. Protein Delivery to Vacuole Requires SAND Protein-Dependent Rab GTPase Conversion for MVB-Vacuole Fusion
 Current Biology 24, 1383 1389, June 16, 2014 ª2014 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2014.05.005 Protein Delivery to Vacuole Requires SAND Protein-Dependent Rab GTPase Conversion
Current Biology 24, 1383 1389, June 16, 2014 ª2014 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2014.05.005 Protein Delivery to Vacuole Requires SAND Protein-Dependent Rab GTPase Conversion
A diploid somatic cell from a rat has a total of 42 chromosomes (2n = 42). As in humans, sex chromosomes determine sex: XX in females and XY in males.
 Multiple Choice Use the following information for questions 1-3. A diploid somatic cell from a rat has a total of 42 chromosomes (2n = 42). As in humans, sex chromosomes determine sex: XX in females and
Multiple Choice Use the following information for questions 1-3. A diploid somatic cell from a rat has a total of 42 chromosomes (2n = 42). As in humans, sex chromosomes determine sex: XX in females and
Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family
 Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family GENES & DEVELOPMENT (2000) 14: 108 117 INTRODUCTION Flower Diagram INTRODUCTION Abscission In plant, the process by which a plant
Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family GENES & DEVELOPMENT (2000) 14: 108 117 INTRODUCTION Flower Diagram INTRODUCTION Abscission In plant, the process by which a plant
AP Biology Essential Knowledge Cards BIG IDEA 1
 AP Biology Essential Knowledge Cards BIG IDEA 1 Essential knowledge 1.A.1: Natural selection is a major mechanism of evolution. Essential knowledge 1.A.4: Biological evolution is supported by scientific
AP Biology Essential Knowledge Cards BIG IDEA 1 Essential knowledge 1.A.1: Natural selection is a major mechanism of evolution. Essential knowledge 1.A.4: Biological evolution is supported by scientific
Protein palmitoylation is critical for the polar growth of root hairs in Arabidopsis
 Zhang et al. BMC Plant Biology (2015) 15:50 DOI 10.1186/s12870-015-0441-5 RESEARCH ARTICLE Protein palmitoylation is critical for the polar growth of root hairs in Arabidopsis Yu-Ling Zhang, En Li, Qiang-Nan
Zhang et al. BMC Plant Biology (2015) 15:50 DOI 10.1186/s12870-015-0441-5 RESEARCH ARTICLE Protein palmitoylation is critical for the polar growth of root hairs in Arabidopsis Yu-Ling Zhang, En Li, Qiang-Nan
Sporic life cycles involve 2 types of multicellular bodies:
 Chapter 3- Human Manipulation of Plants Sporic life cycles involve 2 types of multicellular bodies: -a diploid, spore-producing sporophyte -a haploid, gamete-producing gametophyte Sexual Reproduction in
Chapter 3- Human Manipulation of Plants Sporic life cycles involve 2 types of multicellular bodies: -a diploid, spore-producing sporophyte -a haploid, gamete-producing gametophyte Sexual Reproduction in
Apical hyphal growth. (1) the unidirectional flow of vesicles in the cytoplasm fusing with the plasma membrane at the apex,
 Apical hyphal growth The fungal cell wall is a dynamic structure that protects the cell from changes in osmotic pressure and other environmental stresses, while allowing the fungal cell to interact with
Apical hyphal growth The fungal cell wall is a dynamic structure that protects the cell from changes in osmotic pressure and other environmental stresses, while allowing the fungal cell to interact with
Exam 1 PBG430/
 1 Exam 1 PBG430/530 2014 1. You read that the genome size of maize is 2,300 Mb and that in this species 2n = 20. This means that there are 2,300 Mb of DNA in a cell that is a. n (e.g. gamete) b. 2n (e.g.
1 Exam 1 PBG430/530 2014 1. You read that the genome size of maize is 2,300 Mb and that in this species 2n = 20. This means that there are 2,300 Mb of DNA in a cell that is a. n (e.g. gamete) b. 2n (e.g.
for pollen viability in Arabidopsis thaliana
 The Plant Journal (2006) 48, 193 205 doi: 10.1111/j.1365-313X.2006.02863.x The protein kinase genes MAP3Ke1 and MAP3Ke2 are required for pollen viability in Arabidopsis thaliana Suraphon Chaiwongsar 1,
The Plant Journal (2006) 48, 193 205 doi: 10.1111/j.1365-313X.2006.02863.x The protein kinase genes MAP3Ke1 and MAP3Ke2 are required for pollen viability in Arabidopsis thaliana Suraphon Chaiwongsar 1,
Loss-of-Function Mutations of Retromer Large Subunit Genes Suppress the Phenotype of an Arabidopsis zig Mutant That Lacks Qb-SNARE VTI11 C W
 The Plant Cell, Vol. 22: 159 172, January 2010, www.plantcell.org ã 2010 American Society of Plant Biologists Loss-of-Function Mutations of Retromer Large Subunit Genes Suppress the Phenotype of an Arabidopsis
The Plant Cell, Vol. 22: 159 172, January 2010, www.plantcell.org ã 2010 American Society of Plant Biologists Loss-of-Function Mutations of Retromer Large Subunit Genes Suppress the Phenotype of an Arabidopsis
GFP GAL bp 3964 bp
 Supplemental Data. Møller et al. (2009) Shoot Na + exclusion and increased salinity tolerance engineered by cell type-specific alteration of Na + transport in Arabidopsis Supplemental Figure 1. Salt-sensitive
Supplemental Data. Møller et al. (2009) Shoot Na + exclusion and increased salinity tolerance engineered by cell type-specific alteration of Na + transport in Arabidopsis Supplemental Figure 1. Salt-sensitive
Lipid transfer proteins confer resistance to trichothecenes
 Lipid transfer proteins confer resistance to trichothecenes John McLaughlin and Anwar Bin-Umer Tumer Laboratory National Fusarium Head Blight Forum December 6th, 2012 FY09-11: Identify trichothecene resistance
Lipid transfer proteins confer resistance to trichothecenes John McLaughlin and Anwar Bin-Umer Tumer Laboratory National Fusarium Head Blight Forum December 6th, 2012 FY09-11: Identify trichothecene resistance
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
Zimmerman AP Biology CBHS South Name Chapter 7&8 Guided Reading Assignment 1) What is resolving power and why is it important in biology?
 Zimmerman AP Biology CBHS South Name Chapter 7&8 Guided Reading Assignment 1) What is resolving power and why is it important in biology? 2) How does an electron microscope work and what is the difference
Zimmerman AP Biology CBHS South Name Chapter 7&8 Guided Reading Assignment 1) What is resolving power and why is it important in biology? 2) How does an electron microscope work and what is the difference
DISCOVERIES OF MACHINERY REGULATING VESICLE TRAFFIC, A MAJOR TRANSPORT SYSTEM IN OUR CELLS. Scientific Background on the Nobel Prize in Medicine 2013
 DISCOVERIES OF MACHINERY REGULATING VESICLE TRAFFIC, A MAJOR TRANSPORT SYSTEM IN OUR CELLS Scientific Background on the Nobel Prize in Medicine 2013 Daniela Scalet 6/12/2013 The Nobel Prize in Medicine
DISCOVERIES OF MACHINERY REGULATING VESICLE TRAFFIC, A MAJOR TRANSPORT SYSTEM IN OUR CELLS Scientific Background on the Nobel Prize in Medicine 2013 Daniela Scalet 6/12/2013 The Nobel Prize in Medicine
2. Which of the following are NOT prokaryotes? A) eubacteria B) archaea C) viruses D) ancient bacteria
 1. Which of the following statements is FALSE? A) Errors in chromosome separation are rarely a problem for an organism. B) Errors in chromosome separation can result in a miscarriage. C) Errors in chromosome
1. Which of the following statements is FALSE? A) Errors in chromosome separation are rarely a problem for an organism. B) Errors in chromosome separation can result in a miscarriage. C) Errors in chromosome
Name Class Date. KEY CONCEPT Gametes have half the number of chromosomes that body cells have.
 Section 1: Chromosomes and Meiosis KEY CONCEPT Gametes have half the number of chromosomes that body cells have. VOCABULARY somatic cell autosome fertilization gamete sex chromosome diploid homologous
Section 1: Chromosomes and Meiosis KEY CONCEPT Gametes have half the number of chromosomes that body cells have. VOCABULARY somatic cell autosome fertilization gamete sex chromosome diploid homologous
Supplemental Data. Perrella et al. (2013). Plant Cell /tpc
 Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
The AP-3 b Adaptin Mediates the Biogenesis and Function of Lytic Vacuoles in Arabidopsis C W
 The Plant Cell, Vol. 22: 2812 2824, August 2010, www.plantcell.org ã 2010 American Society of Plant Biologists The AP-3 b Adaptin Mediates the Biogenesis and Function of Lytic Vacuoles in Arabidopsis C
The Plant Cell, Vol. 22: 2812 2824, August 2010, www.plantcell.org ã 2010 American Society of Plant Biologists The AP-3 b Adaptin Mediates the Biogenesis and Function of Lytic Vacuoles in Arabidopsis C
Anatomy of Plants Student Notes
 Directions: Fill in the blanks. Anatomy of Plants Student Notes Plant Cell Biology Segment 1. Plants Plants are organisms are incapable of movement produce food through 2. Animals Animals are multicellular
Directions: Fill in the blanks. Anatomy of Plants Student Notes Plant Cell Biology Segment 1. Plants Plants are organisms are incapable of movement produce food through 2. Animals Animals are multicellular
Supporting Information
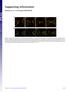 Supporting Information Fleissner et al. 10.1073/pnas.0907039106 Fig. S1. (A) MAK-2-GFP localized to CATs tips is not bound by membrane. his-3::pccg1 mak-2-gfp; mak-2 strain labeled with membrane dye FM4
Supporting Information Fleissner et al. 10.1073/pnas.0907039106 Fig. S1. (A) MAK-2-GFP localized to CATs tips is not bound by membrane. his-3::pccg1 mak-2-gfp; mak-2 strain labeled with membrane dye FM4
Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering
 Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering by Valverde et. Al Published in Science 2004 Presented by Boyana Grigorova CBMG 688R Feb. 12, 2007 Circadian Rhythms: The Clock Within
Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering by Valverde et. Al Published in Science 2004 Presented by Boyana Grigorova CBMG 688R Feb. 12, 2007 Circadian Rhythms: The Clock Within
Analysis of regulatory function of circadian clock. on photoreceptor gene expression
 Thesis of Ph.D. dissertation Analysis of regulatory function of circadian clock on photoreceptor gene expression Tóth Réka Supervisor: Dr. Ferenc Nagy Biological Research Center of the Hungarian Academy
Thesis of Ph.D. dissertation Analysis of regulatory function of circadian clock on photoreceptor gene expression Tóth Réka Supervisor: Dr. Ferenc Nagy Biological Research Center of the Hungarian Academy
HRS1 Acts as a Negative Regulator of Abscisic Acid Signaling to Promote Timely Germination of Arabidopsis Seeds
 HRS1 Acts as a Negative Regulator of Abscisic Acid Signaling to Promote Timely Germination of Arabidopsis Seeds Chongming Wu 1,2., Juanjuan Feng 1,2., Ran Wang 1,2, Hong Liu 1,2, Huixia Yang 1,2, Pedro
HRS1 Acts as a Negative Regulator of Abscisic Acid Signaling to Promote Timely Germination of Arabidopsis Seeds Chongming Wu 1,2., Juanjuan Feng 1,2., Ran Wang 1,2, Hong Liu 1,2, Huixia Yang 1,2, Pedro
7.06 Problem Set #4, Spring 2005
 7.06 Problem Set #4, Spring 2005 1. You re doing a mutant hunt in S. cerevisiae (budding yeast), looking for temperaturesensitive mutants that are defective in the cell cycle. You discover a mutant strain
7.06 Problem Set #4, Spring 2005 1. You re doing a mutant hunt in S. cerevisiae (budding yeast), looking for temperaturesensitive mutants that are defective in the cell cycle. You discover a mutant strain
AP Biology Curriculum Framework
 AP Biology Curriculum Framework This chart correlates the College Board s Advanced Placement Biology Curriculum Framework to the corresponding chapters and Key Concept numbers in Campbell BIOLOGY IN FOCUS,
AP Biology Curriculum Framework This chart correlates the College Board s Advanced Placement Biology Curriculum Framework to the corresponding chapters and Key Concept numbers in Campbell BIOLOGY IN FOCUS,
Essential knowledge 1.A.2: Natural selection
 Appendix C AP Biology Concepts at a Glance Big Idea 1: The process of evolution drives the diversity and unity of life. Enduring understanding 1.A: Change in the genetic makeup of a population over time
Appendix C AP Biology Concepts at a Glance Big Idea 1: The process of evolution drives the diversity and unity of life. Enduring understanding 1.A: Change in the genetic makeup of a population over time
Structures and Functions of Living Organisms
 Structures and Functions of Living Organisms 6.L.1 Understand the structures, processes and behaviors of plants that enable them to survive and reproduce. 6.L.1.1 Summarize the basic structures and functions
Structures and Functions of Living Organisms 6.L.1 Understand the structures, processes and behaviors of plants that enable them to survive and reproduce. 6.L.1.1 Summarize the basic structures and functions
Biology, 7e (Campbell) Chapter 13: Meiosis and Sexual Life Cycles
 Biology, 7e (Campbell) Chapter 13: Meiosis and Sexual Life Cycles Chapter Questions 1) What is a genome? A) the complete complement of an organism's genes B) a specific sequence of polypeptides within
Biology, 7e (Campbell) Chapter 13: Meiosis and Sexual Life Cycles Chapter Questions 1) What is a genome? A) the complete complement of an organism's genes B) a specific sequence of polypeptides within
DNA Structure and Function
 DNA Structure and Function Nucleotide Structure 1. 5-C sugar RNA ribose DNA deoxyribose 2. Nitrogenous Base N attaches to 1 C of sugar Double or single ring Four Bases Adenine, Guanine, Thymine, Cytosine
DNA Structure and Function Nucleotide Structure 1. 5-C sugar RNA ribose DNA deoxyribose 2. Nitrogenous Base N attaches to 1 C of sugar Double or single ring Four Bases Adenine, Guanine, Thymine, Cytosine
AP Biology Unit 6 Practice Test 1. A group of cells is assayed for DNA content immediately following mitosis and is found to have an average of 8
 AP Biology Unit 6 Practice Test Name: 1. A group of cells is assayed for DNA content immediately following mitosis and is found to have an average of 8 picograms of DNA per nucleus. How many picograms
AP Biology Unit 6 Practice Test Name: 1. A group of cells is assayed for DNA content immediately following mitosis and is found to have an average of 8 picograms of DNA per nucleus. How many picograms
SUPPLEMENTARY INFORMATION
 PRC2 represses dedifferentiation of mature somatic cells in Arabidopsis Momoko Ikeuchi 1 *, Akira Iwase 1 *, Bart Rymen 1, Hirofumi Harashima 1, Michitaro Shibata 1, Mariko Ohnuma 1, Christian Breuer 1,
PRC2 represses dedifferentiation of mature somatic cells in Arabidopsis Momoko Ikeuchi 1 *, Akira Iwase 1 *, Bart Rymen 1, Hirofumi Harashima 1, Michitaro Shibata 1, Mariko Ohnuma 1, Christian Breuer 1,
Supplemental Data. Perea-Resa et al. Plant Cell. (2012) /tpc
 Supplemental Data. Perea-Resa et al. Plant Cell. (22)..5/tpc.2.3697 Sm Sm2 Supplemental Figure. Sequence alignment of Arabidopsis LSM proteins. Alignment of the eleven Arabidopsis LSM proteins. Sm and
Supplemental Data. Perea-Resa et al. Plant Cell. (22)..5/tpc.2.3697 Sm Sm2 Supplemental Figure. Sequence alignment of Arabidopsis LSM proteins. Alignment of the eleven Arabidopsis LSM proteins. Sm and
You are required to know all terms defined in lecture. EXPLORE THE COURSE WEB SITE 1/6/2010 MENDEL AND MODELS
 1/6/2010 MENDEL AND MODELS!!! GENETIC TERMINOLOGY!!! Essential to the mastery of genetics is a thorough knowledge and understanding of the vocabulary of this science. New terms will be introduced and defined
1/6/2010 MENDEL AND MODELS!!! GENETIC TERMINOLOGY!!! Essential to the mastery of genetics is a thorough knowledge and understanding of the vocabulary of this science. New terms will be introduced and defined
M P BHOJ (OPEN) UNIVERSITY, BHOPAL ASSIGNMENT QUESTION PAPER
 PAPER :I Cell and molecular Biology of plants Q.1 Describe various models of Plasma membrane with its significance? OR Discuss role of Plasmodesmata in movements of moleculer and macromolecules? Q.2 Give
PAPER :I Cell and molecular Biology of plants Q.1 Describe various models of Plasma membrane with its significance? OR Discuss role of Plasmodesmata in movements of moleculer and macromolecules? Q.2 Give
Overexpression of the Arabidopsis Syntaxin PEP12/SYP21 Inhibits Transport from the Prevacuolar Compartment tothelyticvacuoleinvivo W
 The Plant Cell, Vol. 18, 2275 2293, September 2006, www.plantcell.org ª 2006 American Society of Plant Biologists Overexpression of the Arabidopsis Syntaxin PEP12/SYP21 Inhibits Transport from the Prevacuolar
The Plant Cell, Vol. 18, 2275 2293, September 2006, www.plantcell.org ª 2006 American Society of Plant Biologists Overexpression of the Arabidopsis Syntaxin PEP12/SYP21 Inhibits Transport from the Prevacuolar
Biology of Fungi. Fungal Structure and Function. Lecture: Structure/Function, Part A BIOL 4848/ Fall Overview of the Hypha
 Biology of Fungi Fungal Structure and Function Overview of the Hypha The hypha is a rigid tube containing cytoplasm Growth occurs at the tips of hyphae Behind the tip, the cell is aging Diagram of hyphal
Biology of Fungi Fungal Structure and Function Overview of the Hypha The hypha is a rigid tube containing cytoplasm Growth occurs at the tips of hyphae Behind the tip, the cell is aging Diagram of hyphal
Campbell Biology AP Edition 11 th Edition, 2018
 A Correlation and Narrative Summary of Campbell Biology AP Edition 11 th Edition, 2018 To the AP Biology Curriculum Framework AP is a trademark registered and/or owned by the College Board, which was not
A Correlation and Narrative Summary of Campbell Biology AP Edition 11 th Edition, 2018 To the AP Biology Curriculum Framework AP is a trademark registered and/or owned by the College Board, which was not
CAPE Biology Unit 1 Scheme of Work
 CAPE Biology Unit 1 Scheme of Work 2011-2012 Term 1 DATE SYLLABUS OBJECTIVES TEXT PAGES ASSIGNMENTS COMMENTS Orientation Introduction to CAPE Biology syllabus content and structure of the exam Week 05-09
CAPE Biology Unit 1 Scheme of Work 2011-2012 Term 1 DATE SYLLABUS OBJECTIVES TEXT PAGES ASSIGNMENTS COMMENTS Orientation Introduction to CAPE Biology syllabus content and structure of the exam Week 05-09
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated
 Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry
 Name Period Chapter 6: A Tour of the Cell Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry 1. The study of cells has been limited by their small size, and so they were
Name Period Chapter 6: A Tour of the Cell Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry 1. The study of cells has been limited by their small size, and so they were
Meiosis and Sexual Life Cycles
 Chapter 13 Meiosis and Sexual Life Cycles PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from
Chapter 13 Meiosis and Sexual Life Cycles PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from
Warm-Up Pairs Discuss the diagram What Where Which Why
 Warm-Up In Pairs Discuss the diagram What is it? Where does it come from? Which parts can you label? (in pencil) Why do you think you will learn about it? 5 m Eukaryote: Organelles, Structure and Function
Warm-Up In Pairs Discuss the diagram What is it? Where does it come from? Which parts can you label? (in pencil) Why do you think you will learn about it? 5 m Eukaryote: Organelles, Structure and Function
BIOL 305L Spring 2018 Laboratory Seven
 Please print Full name clearly: BIOL 305L Spring 2018 Laboratory Seven Flowering and reproduction Introduction Flowers are not simple structures, and the diversity of flower shape, color, and fragrance
Please print Full name clearly: BIOL 305L Spring 2018 Laboratory Seven Flowering and reproduction Introduction Flowers are not simple structures, and the diversity of flower shape, color, and fragrance
SUPPLEMENTARY INFORMATION
 Figure S1. Haploid plant produced by centromere-mediated genome elimination Chromosomes containing altered CENH3 in their centromeres (green dots) are eliminated after fertilization in a cross to wild
Figure S1. Haploid plant produced by centromere-mediated genome elimination Chromosomes containing altered CENH3 in their centromeres (green dots) are eliminated after fertilization in a cross to wild
Nature Genetics: doi: /ng Supplementary Figure 1. ssp mutant phenotypes in a functional SP background.
 Supplementary Figure 1 ssp mutant phenotypes in a functional SP background. (a,b) Statistical comparisons of primary and sympodial shoot flowering times as determined by mean values for leaf number on
Supplementary Figure 1 ssp mutant phenotypes in a functional SP background. (a,b) Statistical comparisons of primary and sympodial shoot flowering times as determined by mean values for leaf number on
Meiosis and Sexual Life Cycles
 Chapter 13 Meiosis and Sexual Life Cycles Lecture Outline Overview Living organisms are distinguished by their ability to reproduce their own kind. Offspring resemble their parents more than they do less
Chapter 13 Meiosis and Sexual Life Cycles Lecture Outline Overview Living organisms are distinguished by their ability to reproduce their own kind. Offspring resemble their parents more than they do less
Structures and Functions of Living Organisms
 Structures and Functions of Living Organisms Date: 6.L.1 Understand the structures, processes and behaviors of plants that enable them to survive and reproduce. 6.L.1.1 Summarize the basic structures and
Structures and Functions of Living Organisms Date: 6.L.1 Understand the structures, processes and behaviors of plants that enable them to survive and reproduce. 6.L.1.1 Summarize the basic structures and
Cell Division: the process of copying and dividing entire cells The cell grows, prepares for division, and then divides to form new daughter cells.
 Mitosis & Meiosis SC.912.L.16.17 Compare and contrast mitosis and meiosis and relate to the processes of sexual and asexual reproduction and their consequences for genetic variation. 1. Students will describe
Mitosis & Meiosis SC.912.L.16.17 Compare and contrast mitosis and meiosis and relate to the processes of sexual and asexual reproduction and their consequences for genetic variation. 1. Students will describe
Plant hormones: a. produced in many parts of the plant b. have many functions
 Plant hormones: a. produced in many parts of the plant b. have many functions Illustrated with 4 plant hormones: Gibberellins Auxin Cytokinins Ethylene Gibberellins Gibberellins illustrate how plant hormones
Plant hormones: a. produced in many parts of the plant b. have many functions Illustrated with 4 plant hormones: Gibberellins Auxin Cytokinins Ethylene Gibberellins Gibberellins illustrate how plant hormones
Nature Biotechnology: doi: /nbt Supplementary Figure 1. Overexpression of YFP::GPR-1 in the germline.
 Supplementary Figure 1 Overexpression of YFP::GPR-1 in the germline. The pie-1 promoter and 3 utr were used to express yfp::gpr-1 in the germline. Expression levels from the yfp::gpr-1(cai 1.0)-expressing
Supplementary Figure 1 Overexpression of YFP::GPR-1 in the germline. The pie-1 promoter and 3 utr were used to express yfp::gpr-1 in the germline. Expression levels from the yfp::gpr-1(cai 1.0)-expressing
Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in Section B and ONE question from Section C.
 UNIVERSITY OF EAST ANGLIA School of Biological Sciences Main Series UG Examination 2017-2018 GENETICS BIO-5009A Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in Section
UNIVERSITY OF EAST ANGLIA School of Biological Sciences Main Series UG Examination 2017-2018 GENETICS BIO-5009A Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in Section
Interactive Biology Multimedia Courseware Mendel's Principles of Heredity. Copyright 1998 CyberEd Inc.
 Interactive Biology Multimedia Courseware Mendel's Principles of Heredity Copyright 1998 CyberEd Inc. Mendel's Principles of Heredity TEACHING OBJECTIVES The following subject areas are illustrated throughout
Interactive Biology Multimedia Courseware Mendel's Principles of Heredity Copyright 1998 CyberEd Inc. Mendel's Principles of Heredity TEACHING OBJECTIVES The following subject areas are illustrated throughout
POTASSIUM IN PLANT GROWTH AND YIELD. by Ismail Cakmak Sabanci University Istanbul, Turkey
 POTASSIUM IN PLANT GROWTH AND YIELD by Ismail Cakmak Sabanci University Istanbul, Turkey Low K High K High K Low K Low K High K Low K High K Control K Deficiency Cakmak et al., 1994, J. Experimental Bot.
POTASSIUM IN PLANT GROWTH AND YIELD by Ismail Cakmak Sabanci University Istanbul, Turkey Low K High K High K Low K Low K High K Low K High K Control K Deficiency Cakmak et al., 1994, J. Experimental Bot.
Maximizing productivity of wheat and barley under dry-land systems HYBRIDIZATION. Faddel Ismail NCARE June,
 Maximizing productivity of wheat and barley under dry-land systems HYBRIDIZATION Faddel Ismail NCARE June, 8-10 2015 Hybridization Hybridization is the process of crossing two genetically different individuals
Maximizing productivity of wheat and barley under dry-land systems HYBRIDIZATION Faddel Ismail NCARE June, 8-10 2015 Hybridization Hybridization is the process of crossing two genetically different individuals
Nature Genetics: doi: /ng Supplementary Figure 1. The phenotypes of PI , BR121, and Harosoy under short-day conditions.
 Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Lecture 6 - Intracellular compartments and transport I
 01.26.11 Lecture 6 - Intracellular compartments and transport I Intracellular transport and compartments 1. Protein sorting: How proteins get to their appropriate destinations within the cell 2. Vesicular
01.26.11 Lecture 6 - Intracellular compartments and transport I Intracellular transport and compartments 1. Protein sorting: How proteins get to their appropriate destinations within the cell 2. Vesicular
Chapter 3: Structure and Function of the Cell
 Chapter 3: Structure and Function of the Cell I. Functions of the Cell A. List and describe the main functions of the cell: 1. 2. 3. 4. 5. II. How We See Cells A. Light microscopes allow us to B. Electron
Chapter 3: Structure and Function of the Cell I. Functions of the Cell A. List and describe the main functions of the cell: 1. 2. 3. 4. 5. II. How We See Cells A. Light microscopes allow us to B. Electron
Chapter 6: A Tour of the Cell
 Chapter 6: A Tour of the Cell 1. The study of cells has been limited by their small size, and so they were not seen and described until 1665, when Robert Hooke first looked at dead cells from an oak tree.
Chapter 6: A Tour of the Cell 1. The study of cells has been limited by their small size, and so they were not seen and described until 1665, when Robert Hooke first looked at dead cells from an oak tree.
16 The Cell Cycle. Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization
 The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
Chromosome Chr Duplica Duplic t a ion Pixley
 Chromosome Duplication Pixley Figure 4-6 Molecular Biology of the Cell ( Garland Science 2008) Figure 4-72 Molecular Biology of the Cell ( Garland Science 2008) Interphase During mitosis (cell division),
Chromosome Duplication Pixley Figure 4-6 Molecular Biology of the Cell ( Garland Science 2008) Figure 4-72 Molecular Biology of the Cell ( Garland Science 2008) Interphase During mitosis (cell division),
Chapter 8. The Continuity of Life: How Cells Reproduce. Gregory Ahearn. Lectures by. Ammended by John Crocker. University of North Florida
 Chapter 8 The Continuity of Life: How Cells Reproduce Lectures by Gregory Ahearn University of North Florida Ammended by John Crocker Copyright 2009 Pearson Education, Inc. Review Questions for Chapters
Chapter 8 The Continuity of Life: How Cells Reproduce Lectures by Gregory Ahearn University of North Florida Ammended by John Crocker Copyright 2009 Pearson Education, Inc. Review Questions for Chapters
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2647 Figure S1 Other Rab GTPases do not co-localize with the ER. a, Cos-7 cells cotransfected with an ER luminal marker (either KDEL-venus or mch-kdel) and mch-tagged human Rab5 (mch-rab5,
DOI: 10.1038/ncb2647 Figure S1 Other Rab GTPases do not co-localize with the ER. a, Cos-7 cells cotransfected with an ER luminal marker (either KDEL-venus or mch-kdel) and mch-tagged human Rab5 (mch-rab5,
Enduring understanding 1.A: Change in the genetic makeup of a population over time is evolution.
 The AP Biology course is designed to enable you to develop advanced inquiry and reasoning skills, such as designing a plan for collecting data, analyzing data, applying mathematical routines, and connecting
The AP Biology course is designed to enable you to develop advanced inquiry and reasoning skills, such as designing a plan for collecting data, analyzing data, applying mathematical routines, and connecting
Supplemental Data. Hou et al. (2016). Plant Cell /tpc
 Supplemental Data. Hou et al. (216). Plant Cell 1.115/tpc.16.295 A Distance to 1 st nt of start codon Distance to 1 st nt of stop codon B Normalized PARE abundance 8 14 nt 17 nt Frame1 Arabidopsis inflorescence
Supplemental Data. Hou et al. (216). Plant Cell 1.115/tpc.16.295 A Distance to 1 st nt of start codon Distance to 1 st nt of stop codon B Normalized PARE abundance 8 14 nt 17 nt Frame1 Arabidopsis inflorescence
Trans-Golgi Network-Located AP1 Gamma Adaptins Mediate Dileucine Motif-Directed Vacuolar Targeting in Arabidopsis C W
 The Plant Cell, Vol. 26: 4102 4118, October 2014, www.plantcell.org ã 2014 American Society of Plant Biologists. All rights reserved. Trans-Golgi Network-Located AP1 Gamma Adaptins Mediate Dileucine Motif-Directed
The Plant Cell, Vol. 26: 4102 4118, October 2014, www.plantcell.org ã 2014 American Society of Plant Biologists. All rights reserved. Trans-Golgi Network-Located AP1 Gamma Adaptins Mediate Dileucine Motif-Directed
Plant Cell Advance Publication. Published on March 3, 2016, doi: /tpc
 Plant Cell Advance Publication. Published on March 3, 2016, doi:10.1105/tpc.15.00794 1 2 3 4 5 6 7 8 9 10 HLB1 is a Tetratricopeptide Repeat Domain-Containing Protein that Operates at the Intersection
Plant Cell Advance Publication. Published on March 3, 2016, doi:10.1105/tpc.15.00794 1 2 3 4 5 6 7 8 9 10 HLB1 is a Tetratricopeptide Repeat Domain-Containing Protein that Operates at the Intersection
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach
 Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
is the scientific study of. Gregor Mendel was an Austrian monk. He is considered the of genetics. Mendel carried out his work with ordinary garden.
 11-1 The 11-1 Work of Gregor Mendel The Work of Gregor Mendel is the scientific study of. Gregor Mendel was an Austrian monk. He is considered the of genetics. Mendel carried out his work with ordinary
11-1 The 11-1 Work of Gregor Mendel The Work of Gregor Mendel is the scientific study of. Gregor Mendel was an Austrian monk. He is considered the of genetics. Mendel carried out his work with ordinary
