A missense mutation in CHS1, a TIR-NB protein, induces chilling sensitivity in Arabidopsis
|
|
|
- Elvin Caldwell
- 5 years ago
- Views:
Transcription
1 The Plant Journal (2013) 75, doi: /tpj A missense mutation in CHS1, a TIR-NB protein, induces chilling sensitivity in Arabidopsis Yuancong Wang 1, Yao Zhang 1, Zheng Wang 1, Xiaoyan Zhang 1 and Shuhua Yang 1,2,3, * 1 State Key Laboratory of Plant Physiology and Biochemistry, College of Biological Sciences, China Agricultural University, Beijing , China, 2 Coordinated Research Center for Crop Biology, China Agricultural University, Beijing , China, and 3 National Plant Gene Research Center, Beijing , China Received 6 March 2013; revised 28 April 2013; accepted 2 May 2013; published online 8 May *For correspondence ( yangshuhua@cau.edu.cn). SUMMARY Low temperature is an environmental factor that affects plant growth and development and plant pathogen interactions. How temperature regulates plant defense responses is not well understood. In this study, we characterized chilling-sensitive mutant 1 (chs1), and functionally analyzed the role of the CHS1 gene in plant responses to chilling stress. The chs1 mutant displayed a chilling-sensitive phenotype, and also displayed defense-associated phenotypes, including extensive cell death, the accumulation of hydrogen peroxide and salicylic acid, and an increased expression of PR genes: these phenotypes indicated that the mutation in chs1 activates the defense responses under chilling stress. A map-based cloning analysis revealed that CHS1 encodes a TIR-NB-type protein. The chilling sensitivity of chs1 was fully rescued by pad4 and eds1, but not by ndr1. The overexpression of the TIR and NB domains can suppress the chs1 conferred phenotypes. Interestingly, the stability of the CHS1 protein was positively regulated by low temperatures independently of the 26S proteasome pathway. This study revealed the role of a TIR-NB-type gene in plant growth and cell death under chilling stress, and suggests that temperature modulates the stability of the TIR-NB protein in Arabidopsis. Keywords: CHS1, TIR-NB-type protein, chilling stress, defense responses, Arabidopsis. INTRODUCTION To survive in complex environments, plants have evolved a sophisticated mechanism to deal with the different stresses, including biotic and abiotic stresses. Plants must keep pathogens out during their whole lives. Previous studies have identified that there are at least two layers of inducible defense responses beside physical barriers (Chisholm et al., 2006; Jones and Dangl, 2006). The first is called microbial- or pathogen-associated molecular patterns (MAMPs or PAMPs)-triggered immunity (PTI). Plants trigger general but relatively weak resistance by using transmembrane pattern recognition receptors (PRRs) to sense the slowly evolving molecular patterns, called MAMPs or PAMPs. The second is termed effector-triggered immunity (ETI), which is triggered when host disease resistance (R) proteins recognize pathogen effectors. These effectors function to help the pathogens overcome the PTI and invade the hosts. Unlike PTI, ETI is specific for the pathogens whose effectors are recognized by the hosts, and triggers a much stronger resistance than is triggered The Plant Journal 2013 John Wiley & Sons Ltd by PTI (Jones and Dangl, 2006). ETI is often accompanied by: the accumulation of the defense hormone salicylic acid (SA); the induction of PATHOGENESIS-RELATED (PR) genes; the production of reactive oxygen species (ROS); and programmed cell death, termed the hypersensitive response (Hammond-Kosack and Jones, 1996). The largest class of R proteins consists of proteins containing a nucleotide-binding leucine-rich repeat (NB-LRR) domain at the C terminus and either a coiled-coil (CC) domain or Toll/Interleukin 1 receptor (TIR) domain at the N terminus (Dangl and Jones, 2001). Besides these R proteins, there are an additional 57 proteins that lack LRR domains but are considered to be related to R proteins; 49 of them contain a TIR domain at the N-terminus. These proteins are divided into two subclasses. Proteins harboring an NB domain are called TIR-NB types, and the others, which lack both an LRR and an NB domain, are called TIR X types (Meyers et al., 2002). Several key factors have been identified to be crucial to the functions of these R genes in hosts. 553
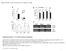
2 554 Yuancong Wang et al. R proteins belonging to the TIR class require the function of ENHANCED DISEASE SUSCEPTIBILITY 1 (EDS1) and PHY- TOALEXIN-DEFICIENT 4 (PAD4) (Glazebrook et al., 1996; Aarts et al., 1998), and those belonging to the CC class depend on the action of NON-RACE SPECIFIC DISEASE RESISTANCE 1 (NDR1) (Aarts et al., 1998; Glazebrook, 2001). It has been reported that the subcellular localization of some R proteins is critical for disease resistance. For instance, in order to trigger plant resistance responses, some R proteins need to accumulate in the nucleus, including N, MILDEWA 10 (MLA 10), RESISTANCE TO PSEUDO- MONAS SYRINGAE 4 (RPS4) and SUPPRESSOR OF npr1 1 CONSTITUTIVE 1 (SNC1) (Burch-Smith et al., 2007; Shen et al., 2007; Wirthmueller et al., 2007; Cheng et al., 2009). Emerging evidence has demonstrated a close link between biotic and cold stress signaling (Alcazar and Parker, 2011). Recent studies have shown that temperatures that are higher than the normal growth range can cause reduced PTI against virulent pathogens and NB-LRRconditioned ETI against avirulent pathogens (Yang and Hua, 2004; Wang et al., 2009). Enhanced disease resistance and inhibited plant growth, caused by the transcriptional accumulation of EDS1 and PAD4 in an auto-active immune background, can be suppressed by high temperatures (Yang and Hua, 2004; Rietz et al., 2011). Plant pathogen interactions have been demonstrated to be regulated differently at different temperatures (Garrett et al., 2006). The R gene Mi can confer nematode resistance to tomatoes (Solanum lycopersicum), and this resistance is lost when the temperature is above 32 C (Hwang et al., 2000). Other evidence supporting this notion includes N in tobacco (Nicotiana tabacum; Someya et al., 2004), and SSI4, RPW8 and the RPP1-like TIR-NB-LRR cluster in Arabidopsis (Xiao et al., 2003; Bomblies et al., 2007; Zhou et al., 2008; Alcazar et al., 2009). Some auto-active resistance mutants are also typically temperature-dependent. Different mutations in SNC1, chilling sensitive 2 (CHS2) and CHS3 show variant auto-active immune phenotypes in a temperature-dependent manner (Zhang et al., 2003; Yang and Hua, 2004; Huang et al., 2010; Yang et al., 2010; Zhu et al., 2010). A recent study showed that ABA-deficient mutations enhance resistance mediated by SNC1 and RPS4 through promoting their nuclear accumulation, thus suggesting an intersection between ABA and disease resistance through the localization of R proteins (Mang et al., 2012). Previous studies reported that one chs1 mutant grew normally at 22 C; however, when it was transferred to the chilling temperature the chs1 mutant showed leaf chlorosis, and did not survive (Hugly et al., 1990). Analyses of microarray data and comparative protein and lipid profiling indicate that the chs1 mutation also disrupts the chloroplast metabolism and lipid trafficking (Hugly et al., 1990; Patterson et al., 1993; Provart et al., 2003). In this report, we show that a mutated TIR-NB-type gene (CHS1) confers sensitivity to chilling temperatures. The function of the CHS1 gene in plant growth and cell death under chilling stress was investigated, and our results revealed that the accumulation of the TIR-NB protein is regulated by low temperatures in Arabidopsis. RESULTS Characterization of the chilling-sensitive mutant chs1 To better understand the molecular regulation of the plant response to chilling stress, we investigated the chs1 1 and chs1 2 mutants, which were isolated previously (Hugly et al., 1990). Because these two alleles showed similar phenotypes, we chose to examine chs1 2 in this study. The chs1 2 mutant was indistinguishable from wild-type Col plants when grown at a normal temperature (22 C; Hugly et al., 1990). However, chs1 2 showed chillingsensitive phenotypes when germinated and grown at chilling temperatures (16 C or below), or when moved from 22 to 16 C (Hugly et al., 1990) (Figure 1a,b). To better analyze the speed of death of chs1 2, compared with the wild type, the levels of chlorophyll a and b were measured in Col and chs1 2. Two-week-old chs1 2 seedlings grown at 22 C were transferred to 16 C and maintained for the indicated time. After 3 days at 16 C, the levels of chlorophyll a and b were dramatically decreased in chs1 2 (which did not show any visible defect phenotype), compared with the wild type; the levels decreased continually with the chilling treatment (Figure 1c). Because chs1 2 grown at 16 C showed a chlorotic phenotype, an ion leakage assay was performed on 2 week-old chs1 2 seedlings before and after the chilling treatment. No obvious changes in ion leakage were observed in the wild type before the chilling treatment. In contrast, ion leakage in the chs1 2 seedlings was significantly increased after 3 days at 16 C, and the leakage continued to increase after that (Figure 1d). We then examined whether chs1 2 conferred cell death at 16 C by performing trypan blue staining. At 22 C, chs1 2 resembled the wild-type Col in morphology, and neither leaves of chs1 2 nor leaves of Col displayed visible trypan blue staining. However, after transferring the plants to 16 C for 7 days, cell death was evident in chs1 2 but not in Col (Figure 1e). Electron transport chains can be perturbed by low temperatures, thus inducing ROS production (Fryer et al., 2002; Hideg et al., 2002; Pfannschmidt et al., 2003). Therefore, we tested the accumulation of hydrogen peroxide (H 2 O 2 ) by using 3,3 diaminobenzidine (DAB) staining. Strong staining was detected in chs1 2, but not in the wild type, following the chilling treatment at 16 C (Figure 1e), indicating that the chs1 2 mutant accumulates a higher level of H 2 O 2 than the wild type under chilling stress. We next determined whether the expression of defenseassociated genes was altered in chs1 2 under chilling
3 chs1 mediated chilling sensitivity 555 (a) (b) (e) (c) (f) (g) Figure 1. Phenotype of the chs1 2 mutant. (a, b) Chilling sensitivity of the chs1 2 mutant. (a) Wild-type Col and chs1 2 mutant plants were grown on MS medium at 16 C for 3 weeks. (b) Col and chs1 2 were grown in soil at 22 C for 2 weeks before transfer to 16 C for 1 week. (c) Measurement of chlorophyll a and b in Col and chs1 2 after they were grown at 22 C for 2 weeks and treated at 16 C for the indicated time. Ion leakage of seedlings described in (c). (e) Two-week-old wild-type Col and chs1 2 plants were grown at 22 C before transfer to 16 C for 7 days; detached leaves were stained with trypan blue (upper panel) and 3,3 diaminobenzidine (DAB) (lower panel). Scale bar: 1 mm. (f) The expression of PR1 and PR2 in Col and chs1 2 grown at 22 C for 2 weeks and transferred to 16 C for the indicated time, as measured by qrt-pcr. The data represent the means of four replicates SDs. (g) Free and total salicylic acid (SA) accumulation in Col and chs1 2. The data represent the means of four replicates SDs. Similar results were observed in three independent experiments. conditions. Total RNA was extracted from 2 week-old chs1 2 and Col plants grown at 22 C at 0 and 3 days after transfer to 16 C, and PR1 and PR2 expression was analyzed by quantitative real-time PCR (qrt-pcr). The expression levels of both PR1 and PR2 were dramatically increased in chs1 2 after the transfer to 16 C, whereas no obvious change in the PR1 or PR2 transcript levels was detected in Col plants under the same conditions (Figure 1f). Because induced PR gene expression is often associated with SA signaling, the endogenous levels of SA in chs1 2 and Col were measured. The levels of both total and free SA were similar to those of the wild type at 22 C. In contrast, the chilling-treated chs1 2 plants accumulated 10- and eight-fold higher levels of total and free SA than the wild type (Figure 1g). These results demonstrated that the mutation in CHS1 results in the activation of the defense response under chilling stress. ROS-associated gene expression in chs1 2 under chilling conditions The above data showed that chs1 2 experienced cell death and H 2 O 2 accumulation at 16 C (Figure 1e). Excess H 2 O 2 induces the expression of genes involved in oxidative stress (Iba, 2002; Mittler et al., 2004; Rizhsky et al., 2004). We next used qrt-pcr to examine the expression of ROS-associated genes, including genes encoding the ROS-producing enzyme NADPH oxidase (RbohD) and ROS-scavenging enzymes, such as ascorbate peroxidase (APX1), catalase (CAT1) and glutathione reductase (GR1), after a 16 C treatment for 3 days. No obvious differences
4 556 Yuancong Wang et al. were detected in the expression of these genes between Col and chs1 2 plants grown at 22 C; however, all four of these ROS-associated genes were upregulated between two- and threefold in chs1 2 compared with the wild type following the chilling treatment (Figure 2). ZAT12 is a C 2 H 2 zinc finger-type transcription factor gene that plays an important role in regulating several genes involved in plant responses to oxidative stress (Davletova et al., 2005). ZAT12 was upregulated (approximately 50 fold) in chs1 2 following the 16 C treatment (Figure 2). The FER1 gene encoding ferritin, which is pivotal for protecting cells against oxidative damage (Ravet et al., 2009), was also highly expressed in the chs1 2 plants subjected to chilling stress (Figure 2). Taken together, these results suggest that the chs1 conferred chilling-sensitive phenotype links the over-activation of ROS production and detoxification with the oxidative signaling pathways caused by the mutation of CHS1. CHS1 encodes a TIR-NB-type protein The chs1 2 mutant was previously reported to be a recessive mutant in a single nuclear locus (Hugly et al., 1990). To identify the CHS1 gene, we generated a segregating population by crossing chs1 2 with Landsberg erecta (Ler). A total of 2500 chilling-sensitive mutant plants were selected for mapping from the segregating F 2 population. The CHS1 locus was mapped to a 275 kb region between markers F28G4 and F11A6 on chromosome I (Figure 3a). A DNA sequencing analysis in this region showed that that a G? A substitution at nucleotide 28 was present in the At1 g17610 gene; this substitution results in an alanine (Ala)? threonine (Thr) substitution (Figures 3b and S1). The same nucleotide mutation was found in the chs1 1 mutant. CHS1 encodes a TIR-NB-type protein. A construct harboring the CHS1 coding region, driven by its native promoter into chs1 2 plants (approximately 2 kb upstream of CHS1), can fully rescue the chilling-sensitive phenotype of chs1 2, cell death and PR expression (Figure 3c f). These results indicate that the chs1 2 mutant phenotype is caused by the mutation observed in At1g To dissect the nature of the chs1 2 mutation, we transformed Col plants with the CHS1 coding region fused with the green fluorescent protein (GFP) gene under the control of the Super promoter (CHS1-GFP). Of the eight independent transgenic lines generated, six showed overexpression of CHS1 (Figure 4a), and they exhibited similar phenotypes, so we chose two lines for further study (lines 3 and 4). Intriguingly, the other two lines (1 and 2) displayed underexpression of CHS1 compared with the wild type Col (Figure 4a), which may be caused by a genesilencing effect. After growing at 16 C for 3 weeks, underexpressing lines showed smaller and weaker stature, whereas overexpressing lines tended to grow better and bigger than the wild type (Figure 4b). These results implied that chs1 2 is a recessive, negative-interfering mutation. Next, transgenic plants overexpressing the mutated chs1 GFP in Col were generated (Figure 4c). These plants showed higher expression of PR1, small stature, obvious lesions and cell-death phenotypes (Figure 4c e). Furthermore, the overexpression of CHS1-GFP in the chs1 2 background at least partially restored the chilling-sensitive phenotype of chs1 2 (Figure 4f, g). F 1 progeny of chs1 2 and Ler showed semi-chilling sensitivity, intermediate between that of Col/Ler and chs1 2 plants (Figure 4h). Taken together, these results suggest that the chilling sensitivity of chs1 2 is dosage dependent. A genetic screen for suppressors of chs1 2 was performed to identify mutants that restore the chs1 2 phenotype to wild-type morphology. Two suppressors of chs1 2, named chs1 r1 and chs1 r2, were mapped to the original CHS1 locus. A sequencing analysis revealed that the mutations of G152R and A138V in chs1 r1 and chs1 r2 were both located in the TIR domain of CHS1 (Figure S2). Both chs1 r1 and chs1 r2 fully restored the chs1 2 chillingsensitive phenotype (Figure S3a). Moreover, the cell death, H 2 O 2 accumulation and expression of PR genes in chs1 r1 and chs1 r2 all recovered to wild-type levels (Figure S3b,c). Figure 2. Expression of reactive oxygen species (ROS)-associated genes in Col and chs1 2 plants under chilling stress, as measured by qrt-pcr. Plants were grown at 22 C for 2 weeks and transferred to 16 C for the indicated time. The data represent the means of three replicates SDs. All of the experiments were repeated three times with similar results.
5 chs1 mediated chilling sensitivity 557 (a) (c) (b) (e) (f) Figure 3. Map-based cloning of CHS1. (a) Genetic map of the CHS1 locus. The markers used for mapping are shown with the number of recombinants. Predicted genes are indicated by arrows, indicating the direction of transcription. (b) The genomic structure of the CHS1 gene. The filled box and unfilled box present the coding region and untranslated region of CHS1 exon, respectively. The positions of the chs1 mutations are shown. (c, d) Complementation test of the chs1 2 mutant. A genomic fragment of CHS1 driven by its native promoter was transformed into chs1 2 plants. (c) Twoweek-old plants grown at 22 C were transferred to 16 C for 1 week. Seedlings grown on MS medium for 3 weeks at 16 C. (e) Trypan blue staining of true leaves from plants that were described in. Scale bar: 100 lm. (f). The PR1 and PR2 transcript levels in the plants grown at 22 C for 2 weeks and transferred to 16 C for 3 days, as measured by qrt-pcr. The data represent the means of three replicates SDs. Similar results were observed in three independent experiments. These results imply that the second mutations in chs1 r1 and chs1 r2 abolish or weaken the negative interference of the chs1 2 mutation. The expression pattern of CHS1 To examine the expression pattern of CHS1, we transformed Col plants with a CHS1:GUS (b glucuronidase) construct carrying the GUS reporter gene driven by a 2.0 kb fragment of the CHS1 promoter. In these transgenic lines, strong GUS staining was primarily observed in cauline leaves, flowers (mainly in sepals) and old rosette leaves (Figure 5b e). GUS signals were also observed in the stems, but not in the siliques or roots (Figure 5a,d f). In addition, semiquantitative RT-PCR demonstrated that CHS1 transcripts were primarily detected in the leaves and flowers (Figure 5g), which is consistent with the GUS staining results. The subcellular localization of CHS1 To investigate the function of CHS1, we examined the subcellular localization of the CHS1-GFP protein in Arabidopsis protoplasts. As a control, a nuclear-localized marker, AT-hook motif nuclear-localized protein 22 (AHL22), fused with red fluorescent protein (RFP), was co-expressed in Arabidopsis protoplasts (Figure 5h). CHS1-GFP was detected in both the cytoplasm and the nucleus (Figure 5h). In some cases, the activation of the TIR-NB-LRR protein that is caused by point substitutions is known to alter its subcellular localization. For example, the accumulation of mutated SNC1 in the nucleus results in the constitutive defense response and dwarf phenotype (Zhu et al., 2010). To determine whether the mutation of CHS1 also affects its subcellular localization, we transiently co-expressed chs1 GFP and AHL22-RFP in Arabidopsis protoplasts. chs1 GFP
6 558 Yuancong Wang et al. (a) (b) (c) (e) (h) (f) (g) Figure 4. Phenotypes of Super:CHS1-GFP and Super:chs1 GFP plants in Col or chs1 2 backgrounds at 16 C. (a) Expression of CHS1 in Col, chs1 2 and four independent transgenic plants of Super:CHS1-GFP grown at 16 C for 2 weeks, as measured by qrt-pcr. (b) Phenotypes of 4 week-old plants described in (a). (c) Expression of CHS1 and PR1 in Col, chs1 2 and three independent transgenic plants of Super:chs1 GFP grown at 16 C for 2 weeks, as measured by qrt-pcr. Phenotypes of the 4 week-old plants described in (c). (e) Trypan blue staining of true leaves from the plants described in. Scale bar: 100 lm. (f) RT-PCR analysis of CHS1 in Col and two independent transgenic lines of chs1/super:gfp and chs1/super:chs1-gfp. EF1a was used as a control. (g) Phenotypes of the 4 week-old plants described in (f). (h) Phenotypes of Col, Ler, chs1 2 and F 1 progeny of chs1 2 crossed with Ler grown at 22 C for 4 weeks before transfer to 16 C for an additional 8 days. accumulated in the cytoplasm and nucleus (Figure 5h). The mutated chs1 did not obviously change the subcellular localization of this protein. Overexpression of CHS1-TIR and CHS1-NB domains can suppress the phenotype of chs1 2 A BLAST analysis showed that CHS1 contains TIR and NB domains. To gain insight into the function of each domain, we generated two constructs carrying the coding region that encodes the TIR or NB domain fused with GFP under the control of the Super promoter, and we transformed them into chs1 2. The transgenic lines indeed overexpressed the TIR and NB domains by RT-PCR (Figure 6a). The transgenic lines carrying TIR-GFP could partially rescue the chilling-sensitive phenotype of chs1 2, whereas the transgenic lines carrying NB-GFP could completely restore the sensitive phenotype of chs1 2 following a 16 C treatment (Figure 6b). Trypan blue and DAB staining showed that the cell death and H 2 O 2 accumulation phenotypes in chs1 2 were partially compromised by the overexpression
7 chs1 mediated chilling sensitivity 559 (a) (b) (c) (g) (h) (e) (f) crossed with plants expressing the nahg gene, which encodes an enzyme that degrades SA to catechol. Col, chs1 2 and chs1 2 nahg plants were grown at 16 C for 3 weeks, and nahg almost completely rescued the chillingsensitive phenotype of chs1 2 (Figure 7a). Consistent with the phenotype, cell death and H 2 O 2 accumulation were clearly suppressed (Figure 7b,c), and PR1 expression also reverted to the wild-type level in the chs1 2 nahg plants (Figure 7d). To further determine the role of SA in the chs1 2 phenotype, chs1 2 was crossed with sid2 1, a mutant with defects in SA biosynthesis (Wildermuth et al., 2001), and npr1 1, a mutant defective in SA signaling (Durrant and Dong, 2004). The chilling-sensitive phenotype and PR1 expression in chs1 2 were partially suppressed by sid2 1 and npr1 1, and the rate of cell death in chs1 2 sid1 2 and chs1 2 npr1 1 was slightly slower than in chs1 2; however, the eventual cell death phenotype was not substantially affected (Figure 7a). These results indicate that the chilling sensitivity and defense activation in chs1 2 partially requires SA. The chs1 2 phenotype is dependent on EDS1 and PAD4 Figure 5. Expression pattern and subcellular localization of CHS1. (a f) GUS expression in the CHS1:GUS transgenic lines. b glucuronidase (GUS) activity was analyzed in the different seedling stages as follows: the two-leaf (a) and eight-leaf (b) stages; (c) the leaves in (b); inflorescences and flowers; (e) a zoom in of the area designated by the arrow in ; and (f) siliques. (g) RT-PCR analysis of CHS1 in the roots (RT), stems (ST), mature rosette leaves (RL), cauline leaves (CL), flowers (FL) and siliques (SL). (h) Subcellular localization of CHS1 and mutated chs1 in Arabidopsis protoplasts. CHS1-GFP or chs1 GFP was co-transformed with a nuclear marker gene (AHL22-RFP) into protoplast prepared from 15 day-old seedlings grown on MS medium. Scale bars: 20 lm. of TIR-GFP, and were completely suppressed by the overexpression of NB-GFP (Figure 6c). We also tested the mrna levels of PR1 and PR2 by qrt-pcr. The transcript level of PR1 in the chs1/nb-gfp lines showed no visible differences from the wild type, and the mrna level of PR1 in the chs1/tir-gfp line was between those of the wild type and chs1 2. The PR2 transcript level in the chs1/tir-gfp lines was partially or completely restored to the wild-type level (Figure 6d). These data indicate that overexpression of the TIR or NB domain, respectively, can partially or completely rescue the chillingsensitive phenotype of chs1 2. The chs1 2 phenotype is partially dependent upon SA To determine whether an increased SA level in chs1 2 is required for the chilling-sensitive phenotype, chs1 2 was Previous studies have demonstrated that the function of traditional TIR-NB-LRR R proteins requires EDS1 and PAD4 (Glazebrook et al., 1996; Aarts et al., 1998). Because CHS1 contains TIR-NB domains, we tested whether the phenotype of chs1-2 also requires EDS1 and PAD4 by crossing chs1 2 with pad4 1 (Jirage et al., 1999) and eds1 2 (in the Col background) (Bartsch et al., 2006). We found that the eds1 and pad4 mutations completely suppressed the phenotype of chs1 2 at 16 C (Figure 7a). The double mutants displayed no obvious cell-death phenotype, H 2 O 2 accumulation or constitutive PR1 gene expression (Figure 7b d). However, the chs1 2 ndr1 1 double mutant behaved similarly to chs1 2 with respect to both morphological and cell-death phenotypes (Figure 7a d). Therefore, the chs1 conferred phenotypes under chilling stress require EDS1 and PAD4, but not NDR1. The CHS1 protein accumulates under chilling stress Because the phenotype of chs1 2 was dependent upon a low temperature, we wondered whether the CHS1 transcript level was temperature-regulated. A semiquantitative RT-PCR analysis showed that CHS1 expression in wild-type plants grown at 22 C and at 16 C remained at the same levels (Figure 8a), indicating that the CHS1 transcript level is not regulated by temperature. We then investigated whether the CHS1 protein level is influenced by temperature. The transgenic lines overexpressing GFP, CHS1-GFP, or chs1 GFP were grown at 16 and 22 C for 7 days, and the GFP signal was detected under a confocal microscope. The GFP signals from the roots of CHS1-GFP and chs1-gfp seedlings grown at 16 C were stronger than in those grown at 22 C (Figure 8b). As
8 560 Yuancong Wang et al. (a) (b) (c) Figure 6. Overexpression of the TIR and NB domains of CHS1 suppresses the chs1 2 phenotype. Wild-type Col, chs1 2 and transgenic plants overexpressing the TIR and NB domains of CHS1 driven by the Super promoter in chs1 2 (chs1/tir-gfp and chs1/nb-gfp) were grown at 16 C. (a) RT-PCR analysis of TIR and NB domains in 2 week-old Col and chs1 2 plants, and in two independent transgenic lines of chs1/tir-gfp and chs1/nb-gfp, respectively. EF1a was used as a control. (b) Phenotypes of the plants described in (a) grown at 16 C for 3 weeks. (c) Trypan blue (upper panel) and DAB (lower panel) staining of true leaves from the plants described in (a) grown at 16 C for 3 weeks. Scale bar: 100 lm. Expression of PR1 and PR2 genes in the plants described in (a) grown at 16 C for 2 weeks, as measured by qrt-pcr. The data represent the means of three replicates SDs. Similar results were observed in three independent experiments. a control, the GFP signals in the GFP seedlings grown at 22 C were similar to those grown at 16 C (Figure 8c). To further determine which domain of CHS1 was sensitive to temperature, we observed the GFP signals in the TIR-GFP and NB-GFP seedlings grown at 22 and 16 C, respectively. TIR-GFP showed reduced expression at 22 C compared with 16 C, whereas no obvious difference in the NB-GFP signals was observed at 16 or 22 C (Figure 8b). Consistently, an immunoblot analysis revealed that the protein levels of CHS1, chs1 and TIR were lower at 22 C than at 16 C, whereas the NB-GFP and GFP protein levels remained unchanged at 16 and 22 C (Figure 8c). These results indicate that the CHS1 protein is low-temperature regulated at the post-transcriptional level. To further study the kinetics of temperature-dependent CHS1-GFP degradation, 14 day-old seedlings of GFP, CHS1-GFP, TIR-GFP and NB-GFP grown at 16 C were transferred to higher temperature (28 C) conditions, which can accelerate CHS1 degradation. Total protein was extracted from these seedlings at 0, 1, 3 and 6 h after treatment at 28 C. The CHS1-GFP and TIR-GFP proteins were indeed degraded following the treatment at 28 C, whereas the GFP and NB-GFP proteins displayed no notable changes under the same treatment (Figure 8d). Notably, the TIR-GFP protein degraded much faster than the CHS1- GFP protein (Figure 8d), suggesting that the NB domain of CHS1 may stabilize the CHS1 protein. To determine whether the TIR domain of CHS1 protein degradation is dependent on the 26S ubiquitin-proteasome pathway, we treated TIR1-GFP seedlings with the proteasome inhibitor MG132 (Smalle and Vierstra, 2004). MG132 treatment did not suppress high temperature-induced degradation of the TIR domain (Figure 8e). These results suggest that the temperature-dependent degradation of the CHS1 protein and its TIR domain is not mediated by the 26S proteasome pathway.
9 chs1 mediated chilling sensitivity 561 (a) (b) (c) Figure 7. Phenotypes of Col, chs1 2 and double mutants grown at 16 C. (a) The phenotypes of Col, chs1 2 and double mutants were grown at 16 C for 3 weeks. (b, c) Trypan blue (b) and DAB (c) staining of the plants described in (a). Scale bars: 100 lm. Expression of PR1 in the plants described in (a) grown at 16 C for 2 weeks, as measured by qrt-pcr. The data represent the means of three replicates SDs. Similar results were observed in three independent experiments. DISCUSSION In this study we characterized the chilling-sensitive mutant chs1, which was previously reported upon by Hugly et al. (1990). This mutant exhibits growth arrest and a seedlinglethal phenotype when grown at chilling temperatures of 16 C or below. CHS1 encodes a TIR-NB-type protein. The overexpression of the TIR and NB domains can suppress the chs1-confered phenotypes. Furthermore, the accumulation of CHS1 protein was positively regulated by low temperatures in a 26S proteasome pathway-independent manner. Recent studies reported that the nuclear accumulation of SNC1 and N may contribute to the resistance mediated by them. The mutated snc1 promotes its nuclear accumulation and triggers the auto-defense response in Arabidopsis (Zhu et al., 2010). In this study, however, CHS1 and mutated chs1 are both localized in the nucleus and cytoplasm, suggesting that other factors affect CHS1 function rather than change its localization. One previous study showed that the LRR domain of CC-NB-LRR-type potato R protein Rx1 can promote localization in the cytoplasm (Slootweg et al., 2010). One mutation of SNC1 that changes the ratio of nuclear to cytoplasm distribution also occurs in the LRR domain of SNC1 (Zhu et al., 2010). Thus, the lack of an LRR domain of CHS1 may contribute to the stable subcellular localization of CHS1. Previous studies have demonstrated that the functions of TIR-NB-LRR and CC-TIR-LRR R proteins are dependent upon EDS1 and NDR1, respectively (Glazebrook et al., 1996; Aarts et al., 1998; Glazebrook, 2001). Our genetic analysis demonstrated that the CHS1 conferred chilling sensitive phenotype at 16 C is dependent upon EDS1 and PAD4, but not upon NDR1, which is consistent with the characteristics of most of the TIR-type R proteins. Therefore, although CHS1 does not contain an LRR domain like traditional R proteins, it can activate similar pathways like a TIR-NB-LRR-type R protein. It is possible that unknown TIR-NB-LRR-type protein(s) may participate in the pathway that causes the phenotype of chs1. Several lines of evidence support the hypothesis that chs1 2 appears to be a recessive, negative-interfering mutation in a dosage-dependent manner. First, chs1 2 is a recessive mutation (Hugly et al., 1990). Second, the growth of seedlings is promoted in transgenic plants overexpressing CHS1, and inhibited in plants underexpressing CHS1. Third, the wild-type plants become chilling-sensitive when the mutated chs1 GFP is overexpressed, whereas the chilling sensitivity of chs1 mutants is recovered when CHS1- GFP is overexpressed. Furthermore, F 1 progeny of chs1 2 and Ler showed semi-chilling sensitivity that was intermediate between that of Col/Ler and chs1 2. In traditional NB-LRR R proteins, the NB domain has been shown to represent a switch that can turn the R
10 562 Yuancong Wang et al. (a) (c) (b) (e) Figure 8. CHS1 is degraded at high temperatures. (a) Expression of CHS1 in response to low temperature by semiquantitative RT-PCR. Two-week-old seedlings grown at 22 C were transferred to 16 C for the indicated times. (b) GFP signals in roots from 7 day-old transgenic seedlings grown at 22 and 16 C were visualized with a confocal microscope. Scale bar: 50 lm. (c) Immunoblot analysis of CHS1 protein and different domains of CHS1 in seedlings described in (a). Total protein was extracted from 7 day-old transgenic lines grown at 22 and 16 C, and analyzed by immunoblotting with antibodies against GFP. Immunoblot analysis of CHS1 and different domains of CHS1 under temperature-shifted conditions. Seven-day-old seedlings grown at 16 C were transferred to 28 C for the indicated times. Total protein was extracted and analyzed by immunoblotting with antibodies against GFP. (e) Immunoblot analysis of the TIR domain of CHS1 in the presence of MG132 under temperature-shifted conditions. Seven-day-old seedlings grown at 16 C were transferred to 28 C in the presence of MG132 or DMSO (as a control) for the indicated time. Total protein was extracted and analyzed by immunoblotting with antibodies against GFP. proteins on or off by binding ATP or ADP, respectively (Lukasik and Takken, 2009). The mutation of CHS2 (an active form of RPP4) is in the NB domain and may constitutively activate RPP4 at low temperatures (Huang et al., 2010). However, in CHS1 we found that the mutation causing the functional change of CHS1 is close to the N terminus of the TIR domain. The TIR domain has been identified as an intracellular part of Toll-like receptors (TLRs) in animals (Tapping, 2009). The TIR domain also occurs at an N terminal location in a major subclass of cytoplasmic NB-LRR-plant R proteins. These R proteins can perceive the pathogen effectors and trigger defense
11 chs1 mediated chilling sensitivity 563 responses to protect the plants from pathogens (Chisholm et al., 2006; Jones and Dangl, 2006; Rafiqi et al., 2009; Dodds and Rathjen, 2010). Evidence suggests that the TIR domain is dispensable for the effector recognition essential for defense signaling (Bernoux et al., 2011). Effector-independent cell death can be triggered by the overexpression of the TIR domain plus amino acids from some TIR-NB-LRR proteins of Arabidopsis, tobacco and flax (Linum usitatissimum; Frost et al., 2004; Michael Weaver et al., 2006; Swiderski et al., 2009; Krasileva et al., 2010). Based on our results, we speculate that both wildtype CHS1 and mutant chs1 can form a heterodimer with a TIR-NB-LRR-type protein through its TIR domain, but whereas the heterodimer containing mutated chs1 may activate this TIR-NB-LRR protein, the wild-type CHS1 does not. When overexpressed TIR domain of wild-type CHS1 in chs1-2, it competes with the mutated chs1 in forming heterodimer with one or more unknown TIR-NB-LRR proteins, and thereby reducing the ratio of the dimer containing mutate chs1. Since the dimer containing wild-type TIR domain cannot activate the TIR-NB-LRR proteins, it can partially restore the phenotype of chs1-2. It has been reported that the overexpression of other domains can suppress the activation of R or R-like proteins. For instance, the phenotype of chs3 1 is suppressed by overexpressing the LIM domain of CHS3, suggesting that the LIM domain might repress N terminal R like activity (Yang et al., 2010). Overexpressing the NB-ARC domain can also inhibit the auto-activity and self-association of the L6 TIR domain (Bernoux et al., 2011). Consistently, the overexpression of the NB ARC domain of CHS1 may also repress the dimerization, thus totally suppressing the chilling-sensitive phenotype of chs1 2. Furthermore, we tested the transcription level of CHS1 in chs1/super:tir- GFP and chs1/super:nb-gfp plants, and found that CHS1 expression in all transgenic lines remained the same compared with the wild type (Figure S3). These results exclude the possibility that the gene silencing of CHS1 contributes to the suppression of the chs1 phenotype. Although the chs1 mutant displays extensive cell death at low temperatures (16 C or below), it grows normally at normal temperatures (22 C). This phenomenon prompted us to test the effect of temperature on CHS1 protein stability. Previous study reported both the transcriptional and protein levels of SNC1 were higher at 22 C than 28 C (Yang and Hua, 2004; Zhu et al., 2010). But for CHS1, unlike SNC1, the mrna level remained unchanged at 16 C; however, CHS1-GFP and TIR-GFP proteins accumulated to a greater extent at 16 C than at 22 C, whereas NB-GFP was maintained at a consistent level at 22 and 16 C. Furthermore, the CHS1 protein was degraded several hours after the plants were transferred from 22 to 28 C. Intriguingly, we noticed that the TIR domain of CHS1 degraded much faster than that of the full-length protein. In contrast, the NB domain was not responsive to the temperature change. These results imply that one function of the NB domain is to stabilize CHS1 at high temperatures. EXPERIMENTAL PROCEDURES Plant materials and growth conditions Arabidopsis thaliana plants of the Columbia (Col) and Landsberg erecta (Ler) ecotypes were used in this study. The chs1 1 and chs1 2 mutants (Hugly et al., 1990) were obtained from the Arabidopsis Biological Resource Center (ABRC, The mutants sid2 1 (Wildermuth et al., 2001), npr1 1 (Durrant and Dong, 2004), pad4 1 (Jirage et al., 1999), eds1 2 (in Col background) (Bartsch et al., 2006) and ndr1 1 (Century et al., 1995) are in the Col background. The Arabidopsis plants used in this study were grown at 22 or 16 C under a 16 h light/8 h dark photoperiod (100 lmol m 2 sec 1 ), with 50 70% relative humidity. The Arabidopsis seeds were planted on MS medium (Sigma-Aldrich, supplemented with 2% sucrose and 0.8% agar before they were transferred to soil. Map-based cloning of the CHS1 gene To map the chs1 2 mutation, chs1 2 (Col background) was crossed with Ler. The F 1 plants from the cross were self-fertilized, and the resulting F 2 seeds were collected. A total of 2500 chs1 2 mutant plants were chosen from the segregating F 2 population based on their hypersensitivity at 16 C. Genomic DNA was used to perform PCR-based mapping with simple sequence length polymorphism (SSLP) and cleaved amplified polymorphic sequence (CAPS), and derived CAPS (dcaps) markers. Plasmid construction and plant transformation A 3.55 kb genomic fragment containing the CHS1 promoter, coding region and 3 untranslated region (3 UTR) region was amplified from Col genomic DNA using the primers CHS1-1-1F and CHS1-1-3R; the fragment was cloned into the pcambia1300 vector (CAMBIA) to generate the CHS1:CHS1 construct. To generate the CHS1:GUS construct, a 2.07 kb genomic fragment upstream of the CHS1 ATG was amplified using primers CHS1-1-1F and CHS1-1-2R, and was cloned into the pzpgus2 vector (Diener et al., 2000). To construct Super:TIR-GFP, Super:NB-GFP and Super:CHS1-GFP, the DNA fragments were amplified from Col by PCR using the primers CHS1-2-TIR-F and CHS1-2-TIR-R, CHS1-2-NB-F and CHS1-2-NB-R, and CHS1-2-TIR-F and CHS1-2-NB-R, respectively; the fragments were cloned into the pcambia1300 vector containing the Super promoter (Ni et al., 1995). The mutated chs1 DNA fragment used to construct Super:chs1 GFP was amplified from the chs1 2 mutant. The primers are listed in Table S1. All of the constructs were transformed into the designated genotypes via floral-dip transformation (Clough and Bent, 1998). Electrolyte leakage assays and chlorophyll measurement Two-week-old seedlings grown at 22 C were transferred to 16 C for 0, 3, 6 and 9 d. The electrolyte leakage assay was performed as described previously (Lee et al., 2002). The contents of chlorophyll a and b were extracted and measured as previously described (Huang et al., 2009).
12 564 Yuancong Wang et al. Salicylic acid (SA) measurement Free and total SA were extracted and measured from 2 week-old seedlings grown at 22 C and then transferred to 16 C for 3 days, as previously described (Li et al., 1999). Histochemistry The histochemical detection of GUS activity was performed as previously described (Yang et al., 2006). Trypan blue and DAB staining was also performed as previously described (Bowling et al., 1997; Thordal-Christensen et al., 1997). Quantitative real-time PCR (qrt-pcr) Two-week-old seedlings grown at 22 C were transferred to 16 C for 3 days. Total RNA was prepared using the TRIzol reagent (Invitrogen, and was reverse transcribed using M MLV reverse transcriptase (Promega, qrt-pcr was performed using a SYBR Green PCR Master Mix kit (TaKaRa, The relative expression levels were calculated as previously described (Huang et al., 2010). The primers used are listed in Table S1. Protoplast transformation Protoplast isolation and transformation were performed as previously described (Zhai et al., 2009). Briefly, protoplasts were prepared from 10 day-old wild-type seedlings grown on MS medium. After transformation, the protoplasts were incubated at 16 C, and the GFP signals were observed after h of incubation. Confocal laser microscopy Transgenic plants expressing Super:CHS1-GFP, Super:CHS1-TIR- GFP, Super:CHS1-NB-GFP or Super:chs1 GFP were grown at 22 or 16 C for 7 days. GFP fluorescence in the roots of the transgenic plants or in transgenic protoplasts was visualized using a confocal laser scanning microscope (LSM510; Zeiss, Immunoblot analysis Total protein was prepared from 7 day-old transgenic plants that were grown at 22 or 16 C, or were transferred from 16 to 22 C for different time periods, as previously described (Huang et al., 2009). The CHS1-GFP fusion proteins were visualized on immunoblots using an anti-gfp antibody (Sigma-Aldrich). Rubisco was stained with Ponceau S as a control. ACKNOWLEDGEMENTS We thank Jane E. Parker and ABRC for mutant seeds. This work was supported by the National Key Basic Research Program of China (2011CB915401), China National Funds for Distinguished Young Scientists ( ), the National Natural Science Foundation of China ( ), and the Ministry of Agriculture of China for transgenic research (2011ZX ). SUPPORTING INFORMATION Additional Supporting Information may be found in the online version of this article. Figure S1. Amino-acid sequence of CHS1. Figure S2. Phenotypes of Col, chs1 2 chs1 r1 and chs1 r2. Figure S3. Expression of CHS1 in Super:TIR-GFP and Super: NB-GFP plants grown at 16 C for 2 weeks by semiquantitative RT-PCR. Table S1. Gene-specific primers used in this study. REFERENCES Aarts, N., Metz, M., Holub, E., Staskawicz, B. J., Daniels, M. J. and Parker, J. E. (1998) Different requirements for EDS1 and NDR1 by disease resistance genes define at least two R gene-mediated signaling pathways in Arabidopsis. Proc. Natl Acad. Sci. USA, 95, Alcazar, R. and Parker, J. E. (2011) The impact of temperature on balancing immune responsiveness and growth in Arabidopsis. Trends Plant Sci. 16, Alcazar, R., Garcia, A. V., Parker, J. E. and Reymond, M. (2009) Incremental steps toward incompatibility revealed by Arabidopsis epistatic interactions modulating salicylic acid pathway activation. Proc. Natl Acad. Sci. USA, 106, Bartsch, M., Gobbato, E., Bednarek, P., Debey, S., Schultze, J. L., Bautor, J. and Parker, J. E. (2006) Salicylic acid-independent ENHANCED DISEASE SUSCEPTIBILITY1 signaling in Arabidopsis immunity and cell death is regulated by the monooxygenase FMO1 and the Nudix hydrolase NUDT7. Plant Cell, 18, Bernoux, M., Ve, T., Williams, S., Warren, C., Hatters, D., Valkov, E., Zhang, X., Ellis, J. G., Kobe, B. and Dodds, P. N. (2011) Structural and functional analysis of a plant resistance protein TIR domain reveals interfaces for self-association, signaling, and autoregulation. Cell Host Microbe, 9, Bomblies, K., Lempe, J., Epple, P., Warthmann, N., Lanz, C., Dangl, J. L. and Weigel, D. (2007) Autoimmune response as a mechanism for a Dobzhansky-Muller-type incompatibility syndrome in plants. PLoS Biol. 5, e236. Bowling, S. A., Clarke, J. D., Liu, Y., Klessig, D. F. and Dong, X. (1997) The cpr5 mutant of Arabidopsis expresses both NPR1-dependent and NPR1- independent resistance. Plant Cell, 9, Burch-Smith, T. M., Schiff, M., Caplan, J. L., Tsao, J., Czymmek, K. and Dinesh-Kumar, S. P. (2007) A novel role for the TIR domain in association with pathogen-derived elicitors. PLoS Biol. 5, e68. Century, K. S., Holub, E. B. and Staskawicz, B. J. (1995) NDR1, a locus of Arabidopsis thaliana that is required for disease resistance to both a bacterial and a fungal pathogen. Proc. Natl Acad. Sci. USA, 92, Cheng, Y. T., Germain, H., Wiermer, M. et al. (2009) Nuclear pore complex component MOS7/Nup88 is required for innate immunity and nuclear accumulation of defense regulators in Arabidopsis. Plant Cell, 21, Chisholm, S. T., Coaker, G., Day, B. and Staskawicz, B. J. (2006) Hostmicrobe interactions: shaping the evolution of the plant immune response. Cell, 124, Clough, S. J. and Bent, A. F. (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, Dangl, J. L. and Jones, J. D. (2001) Plant pathogens and integrated defence responses to infection. Nature, 411, Davletova, S., Schlauch, K., Coutu, J. and Mittler, R. (2005) The zinc-finger protein Zat12 plays a central role in reactive oxygen and abiotic stress signaling in Arabidopsis. Plant Physiol. 139, Diener, A. C., Li, H., Zhou, W., Whoriskey, W. J., Nes, W. D. and Fink, G. R. (2000) Sterol methyltransferase 1 controls the level of cholesterol in plants. Plant Cell, 12, Dodds, P. N. and Rathjen, J. P. (2010) Plant immunity: towards an integrated view of plant-pathogen interactions. Nat. Rev. Genet. 11, Durrant, W. E. and Dong, X. (2004) Systemic acquired resistance. Annu. Rev. Phytopathol. 42, Frost, D., Way, H., Howles, P., Luck, J., Manners, J., Hardham, A., Finnegan, J. and Ellis, J. (2004) Tobacco transgenic for the flax rust resistance gene L expresses allele-specific activation of defense responses. Mol. Plant Microbe Interact. 17, Fryer, M. J., Oxborough, K., Mullineaux, P. M. and Baker, N. R. (2002) Imaging of photo-oxidative stress responses in leaves. J. Exp. Bot. 53, Garrett, K. A., Dendy, S. P., Frank, E. E., Rouse, M. N. and Travers, S. E. (2006) Climate change effects on plant disease: genomes to ecosystems. Annu. Rev. Phytopathol. 44, Glazebrook, J. (2001) Genes controlling expression of defense responses in Arabidopsis 2001 status. Curr. Opin. Plant Biol. 4,
13 chs1 mediated chilling sensitivity 565 Glazebrook, J., Rogers, E. E. and Ausubel, F. M. (1996) Isolation of Arabidopsis mutants with enhanced disease susceptibility by direct screening. Genetics, 143, Hammond-Kosack, K. E. and Jones, J. D. (1996) Resistance gene-dependent plant defense responses. Plant Cell, 8, Hideg, E., Barta, C., Kalai, T., Vass, I., Hideg, K. and Asada, K. (2002) Detection of singlet oxygen and superoxide with fluorescent sensors in leaves under stress by photoinhibition or UV radiation. Plant Cell Physiol. 43, Huang, X., Zhang, X. and Yang, S. (2009) A novel chloroplast-localized protein EMB1303 is required for chloroplast development in Arabidopsis. Cell Res. 19, Huang, X., Li, J., Bao, F., Zhang, X. and Yang, S. (2010) A gain-of-function mutation in the Arabidopsis disease resistance gene RPP4 confers sensitivity to low temperature. Plant Physiol. 154, Hugly, S., McCourt, P., Browse, J., Patterson, G. W. and Somerville, C. (1990) A chilling sensitive mutant of Arabidopsis with altered steryl-ester metabolism. Plant Physiol. 93, Hwang, C. F., Bhakta, A. V., Truesdell, G. M., Pudlo, W. M. and Williamson, V. M. (2000) Evidence for a role of the N terminus and leucine-rich repeat region of the Mi gene product in regulation of localized cell death. Plant Cell, 12, Iba, K. (2002) Acclimative response to temperature stress in higher plants: approaches of gene engineering for temperature tolerance. Annu. Rev. Plant Biol. 53, Jirage, D., Tootle, T. L., Reuber, T. L., Frost, L. N., Feys, B. J., Parker, J. E., Ausubel, F. M. and Glazebrook, J. (1999) Arabidopsis thaliana PAD4 encodes a lipase-like gene that is important for salicylic acid signaling. Proc. Natl Acad. Sci. USA, 96, Jones, J. D. and Dangl, J. L. (2006) The plant immune system. Nature, 444, Krasileva, K. V., Dahlbeck, D. and Staskawicz, B. J. (2010) Activation of an Arabidopsis resistance protein is specified by the in planta association of its leucine-rich repeat domain with the cognate oomycete effector. Plant Cell, 22, Lee, H., Guo, Y., Ohta, M., Xiong, L., Stevenson, B. and Zhu, J. K. (2002) LOS2, a genetic locus required for cold-responsive gene transcription encodes a bi-functional enolase. EMBO J. 21, Li, X., Zhang, Y., Clarke, J. D., Li, Y. and Dong, X. (1999) Identification and cloning of a negative regulator of systemic acquired resistance, SNI1, through a screen for suppressors of npr1-1. Cell, 98, Lukasik, E. and Takken, F. L. (2009) STANDing strong, resistance proteins instigators of plant defence. Curr. Opin. Plant Biol. 12, Mang, H. G., Qian, W., Zhu, Y., Qian, J., Kang, H. G., Klessig, D. F. and Hua, J. (2012) Abscisic acid deficiency antagonizes high-temperature inhibition of disease resistance through enhancing nuclear accumulation of resistance proteins SNC1 and RPS4 in Arabidopsis. Plant Cell, 24, Meyers, B. C., Morgante, M. and Michelmore, R. W. (2002) TIR-X and TIR- NBS proteins: two new families related to disease resistance TIR-NBS- LRR proteins encoded in Arabidopsis and other plant genomes. Plant J. 32, Michael Weaver, L., Swiderski, M. R., Li, Y. and Jones, J. D. (2006) The Arabidopsis thaliana TIR-NB-LRR R-protein, RPP1A; protein localization and constitutive activation of defence by truncated alleles in tobacco and Arabidopsis. Plant J. 47, Mittler, R., Vanderauwera, S., Gollery, M. and Van Breusegem, F. (2004) Reactive oxygen gene network of plants. Trends Plant Sci. 9, Ni, M., Cui, D., Einstein, J., Narasimhulu, S., Vergara, C. E. and Gelvin, S. B. (1995) Strength and tissue specificity of chimeric promoters derived from the octopine and mannopine synthase genes. Plant J. 7, Patterson, G., Hugly, S. and Harrison, D. (1993) Sterols and phytyl esters of Arabidopsis thaliana under normal and chilling temperatures. Phytochemistry, 33, Pfannschmidt, T., Schutze, K., Fey, V., Sherameti, I. and Oelmuller, R. (2003) Chloroplast redox control of nuclear gene expression a new class of plastid signals in interorganellar communication. Antioxid. Redox Signal. 5, Provart, N. J., Gil, P., Chen, W., Han, B., Chang, H. S., Wang, X. and Zhu, T. (2003) Gene expression phenotypes of Arabidopsis associated with sensitivity to low temperatures. Plant Physiol. 132, Rafiqi, M., Bernoux, M., Ellis, J. G. and Dodds, P. N. (2009) In the trenches of plant pathogen recognition: role of NB-LRR proteins. Semin. Cell Dev. Biol. 20, Ravet, K., Touraine, B., Boucherez, J., Briat, J. F., Gaymard, F. and Cellier, F. (2009) Ferritins control interaction between iron homeostasis and oxidative stress in Arabidopsis. Plant J. 57, Rietz, S., Stamm, A., Malonek, S., Wagner, S., Becker, D., Medina-Escobar, N., Vlot, A. C., Feys, B. J., Niefind, K. and Parker, J. E. (2011) Different roles of Enhanced Disease Susceptibility1 (EDS1) bound to and dissociated from Phytoalexin Deficient4 (PAD4) in Arabidopsis immunity. New Phytol. 191, Rizhsky, L., Davletova, S., Liang, H. and Mittler, R. (2004) The zinc finger protein Zat12 is required for cytosolic ascorbate peroxidase 1 expression during oxidative stress in Arabidopsis. J. Biol. Chem. 279, Shen, Q. H., Saijo, Y., Mauch, S., Biskup, C., Bieri, S., Keller, B., Seki, H., Ulker, B., Somssich, I. E. and Schulze-Lefert, P. (2007) Nuclear activity of MLA immune receptors links isolate-specific and basal disease-resistance responses. Science, 315, Slootweg, E., Roosien, J., Spiridon, L. N. et al. (2010) Nucleocytoplasmic distribution is required for activation of resistance by the potato NB-LRR receptor Rx1 and is balanced by its functional domains. Plant Cell, 22, Smalle, J. and Vierstra, R. D. (2004) The ubiquitin 26S proteasome proteolytic pathway. Annu. Rev. Plant Biol. 55, Someya, N., Niinuma, K., Kimura, M., Yamaguchi, I. and Hamamoto, H. (2004) Pattern of N gene-mediated systemic hypersensitive response and turnover of viral replicase protein in tobacco. Arch. Virol. 149, Swiderski, M. R., Birker, D. and Jones, J. D. (2009) The TIR domain of TIR-NB-LRR resistance proteins is a signaling domain involved in cell death induction. Mol. Plant Microbe Interact. 22, Tapping, R. I. (2009) Innate immune sensing and activation of cell surface Toll-like receptors. Semin. Immunol. 21, Thordal-Christensen, H., Zhang, Z., Wei, Y. and Collinge, D. B. (1997) Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley-powdery mildew interaction. Plant J. 11, Wang, Y., Bao, Z., Zhu, Y. and Hua, J. (2009) Analysis of temperature modulation of plant defense against biotrophic microbes. Mol. Plant Microbe Interact. 22, Wildermuth, M. C., Dewdney, J., Wu, G. and Ausubel, F. M. (2001) Isochorismate synthase is required to synthesize salicylic acid for plant defence. Nature, 414, Wirthmueller, L., Zhang, Y., Jones, J. D. and Parker, J. E. (2007) Nuclear accumulation of the Arabidopsis immune receptor RPS4 is necessary for triggering EDS1-dependent defense. Curr. Biol. 17, Xiao, S., Charoenwattana, P., Holcombe, L. and Turner, J. G. (2003) The Arabidopsis genes RPW8.1 and RPW8.2 confer induced resistance to powdery mildew diseases in tobacco. Mol. Plant Microbe Interact. 16, Yang, S. and Hua, J. (2004) A haplotype-specific Resistance gene regulated by BONZAI1 mediates temperature-dependent growth control in Arabidopsis. Plant Cell, 16, Yang, H., Li, Y. and Hua, J. (2006) The C2 domain protein BAP1 negatively regulates defense responses in Arabidopsis. Plant J. 48, Yang, H., Shi, Y., Liu, J., Guo, L., Zhang, X. and Yang, S. (2010) A mutant CHS3 protein with TIR-NB-LRR-LIM domains modulates growth, cell death and freezing tolerance in a temperature-dependent manner in Arabidopsis. Plant J. 63, Zhai, Z., Sooksa-nguan, T. and Vatamaniuk, O. K. (2009) Establishing RNA interference as a reverse-genetic approach for gene functional analysis in protoplasts. Plant Physiol. 149, Zhang, Y., Goritschnig, S., Dong, X. and Li, X. (2003) A gain-of-function mutation in a plant disease resistance gene leads to constitutive activation of downstream signal transduction pathways in suppressor of npr1-1, constitutive 1. Plant Cell, 15, Zhou, F., Mosher, S., Tian, M., Sassi, G., Parker, J. and Klessig, D. F. (2008) The Arabidopsis gain-of-function mutant ssi4 requires RAR1 and SGT1b differentially for defense activation and morphological alterations. Mol. Plant Microbe Interact. 21, Zhu, Y., Qian, W. and Hua, J. (2010) Temperature modulates plant defense responses through NB-LRR proteins. PLoS Pathog. 6, e
Arabidopsis HSP90 protein modulates RPP4-mediated temperature-dependent cell death and defense responses
 Research Arabidopsis HSP90 protein modulates RPP4-mediated temperature-dependent cell death and defense responses Fei Bao 1, Xiaozhen Huang 1, Chipan Zhu 2, Xiaoyan Zhang 1, Xin Li 2 and Shuhua Yang 1
Research Arabidopsis HSP90 protein modulates RPP4-mediated temperature-dependent cell death and defense responses Fei Bao 1, Xiaozhen Huang 1, Chipan Zhu 2, Xiaoyan Zhang 1, Xin Li 2 and Shuhua Yang 1
Looking for LOV: Location of LOV1 function in Nicotiana benthamiana cells
 Looking for LOV: Location of LOV1 function in Nicotiana benthamiana cells By: Patrick Rutledge 1 Dr. Jennifer Lorang 2,3, Dr. Marc Curtis 2,3, Dr. Thomas Wolpert 2,3 BioResource Research 1, Botany and
Looking for LOV: Location of LOV1 function in Nicotiana benthamiana cells By: Patrick Rutledge 1 Dr. Jennifer Lorang 2,3, Dr. Marc Curtis 2,3, Dr. Thomas Wolpert 2,3 BioResource Research 1, Botany and
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated
 Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Nature Genetics: doi: /ng Supplementary Figure 1. The phenotypes of PI , BR121, and Harosoy under short-day conditions.
 Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach
 Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport
 Ph.D. thesis Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport Zsigmond Laura Supervisor: Dr. Szabados László Arabidopsis Molecular Genetic Group Institute of Plant
Ph.D. thesis Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport Zsigmond Laura Supervisor: Dr. Szabados László Arabidopsis Molecular Genetic Group Institute of Plant
A mutant CHS3 protein with TIR-NB-LRR-LIM domains modulates growth, cell death and freezing tolerance in a temperature-dependent manner in Arabidopsis
 The Plant Journal (2010) 63, 283 296 doi: 10.1111/j.1365-313X.2010.04241.x A mutant CHS3 protein with TIR-NB-LRR-LIM domains modulates growth, cell death and freezing tolerance in a temperature-dependent
The Plant Journal (2010) 63, 283 296 doi: 10.1111/j.1365-313X.2010.04241.x A mutant CHS3 protein with TIR-NB-LRR-LIM domains modulates growth, cell death and freezing tolerance in a temperature-dependent
Supplemental material
 Supplemental material Table 1- Segregation analysis of sgt1a sgt1b double mutant plants Parental genotype No. of lines Normal seeds Phenotype Abnormal seeds Ratio of abnormal seeds (%) WT 3 171 2 1.16
Supplemental material Table 1- Segregation analysis of sgt1a sgt1b double mutant plants Parental genotype No. of lines Normal seeds Phenotype Abnormal seeds Ratio of abnormal seeds (%) WT 3 171 2 1.16
Host-Pathogen Interaction. PN Sharma Department of Plant Pathology CSK HPKV, Palampur
 Host-Pathogen Interaction PN Sharma Department of Plant Pathology CSK HPKV, Palampur-176062 PATHOGEN DEFENCE IN PLANTS A BIOLOGICAL AND MOLECULAR VIEW Two types of plant resistance response to potential
Host-Pathogen Interaction PN Sharma Department of Plant Pathology CSK HPKV, Palampur-176062 PATHOGEN DEFENCE IN PLANTS A BIOLOGICAL AND MOLECULAR VIEW Two types of plant resistance response to potential
Supplemental Data. Perrella et al. (2013). Plant Cell /tpc
 Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
PAMP-triggered immunity (PTI)
 PAMP-triggered immunity (PTI) PAMP-triggered immunity (PTI) Recognition of danger signals - Distinguish self or damaged self versus non-self fundamental to any immune system - PAMP or MAMP pathogen/microbe-associated
PAMP-triggered immunity (PTI) PAMP-triggered immunity (PTI) Recognition of danger signals - Distinguish self or damaged self versus non-self fundamental to any immune system - PAMP or MAMP pathogen/microbe-associated
Lipid transfer proteins confer resistance to trichothecenes
 Lipid transfer proteins confer resistance to trichothecenes John McLaughlin and Anwar Bin-Umer Tumer Laboratory National Fusarium Head Blight Forum December 6th, 2012 FY09-11: Identify trichothecene resistance
Lipid transfer proteins confer resistance to trichothecenes John McLaughlin and Anwar Bin-Umer Tumer Laboratory National Fusarium Head Blight Forum December 6th, 2012 FY09-11: Identify trichothecene resistance
Supplemental Data. Chen and Thelen (2010). Plant Cell /tpc
 Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
23-. Shoot and root development depend on ratio of IAA/CK
 Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
The sucrose non-fermenting-1-related protein kinases SAPK1 and SAPK2 function collaboratively as positive regulators of salt stress tolerance in rice
 Lou et al. BMC Plant Biology (2018) 18:203 https://doi.org/10.1186/s12870-018-1408-0 RESEARCH ARTICLE Open Access The sucrose non-fermenting-1-related protein kinases SAPK1 and SAPK2 function collaboratively
Lou et al. BMC Plant Biology (2018) 18:203 https://doi.org/10.1186/s12870-018-1408-0 RESEARCH ARTICLE Open Access The sucrose non-fermenting-1-related protein kinases SAPK1 and SAPK2 function collaboratively
Host-Pathogen interaction-ii. Pl Path 604 PN Sharma Department of Plant Pathology CSK HPKV, Palampur
 Host-Pathogen interaction-ii Pl Path 604 PN Sharma Department of Plant Pathology CSK HPKV, Palampur-176062 It was originally believed that gene-for-gene resistance was conferred by a direct interaction
Host-Pathogen interaction-ii Pl Path 604 PN Sharma Department of Plant Pathology CSK HPKV, Palampur-176062 It was originally believed that gene-for-gene resistance was conferred by a direct interaction
Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering
 Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering by Valverde et. Al Published in Science 2004 Presented by Boyana Grigorova CBMG 688R Feb. 12, 2007 Circadian Rhythms: The Clock Within
Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering by Valverde et. Al Published in Science 2004 Presented by Boyana Grigorova CBMG 688R Feb. 12, 2007 Circadian Rhythms: The Clock Within
Quantitative Nature of Arabidopsis Responses during Compatible and Incompatible Interactions with the Bacterial Pathogen Pseudomonas syringae
 The Plant Cell, Vol. 15, 317 330, February 2003, www.plantcell.org 2003 American Society of Plant Biologists Quantitative Nature of Arabidopsis Responses during Compatible and Incompatible Interactions
The Plant Cell, Vol. 15, 317 330, February 2003, www.plantcell.org 2003 American Society of Plant Biologists Quantitative Nature of Arabidopsis Responses during Compatible and Incompatible Interactions
TIME-LINE OF INFECTION
 Review of Lecture 8: Getting inside the host is a critical step in disease development Fungal pathogens use contact and chemical tropisms to guide their way to a site where infection is possible Pathogens
Review of Lecture 8: Getting inside the host is a critical step in disease development Fungal pathogens use contact and chemical tropisms to guide their way to a site where infection is possible Pathogens
 Supplemental Data. Tameling et al. (2010). Plant Cell 10.1105/tpc.110.077461 Supplemental Table 1. Primers used in plasmid construction Primer Code Sequence eo1 5 -GCCATGGCTCATGCAAGTGTGGCTTCTC-3 eo2 5
Supplemental Data. Tameling et al. (2010). Plant Cell 10.1105/tpc.110.077461 Supplemental Table 1. Primers used in plasmid construction Primer Code Sequence eo1 5 -GCCATGGCTCATGCAAGTGTGGCTTCTC-3 eo2 5
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL
 GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
** * * * Col-0 cau1 CAU1. Actin2 CAS. Actin2. Supplemental Figure 1. CAU1 affects calcium accumulation.
 Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Genetic interaction and phenotypic analysis of the Arabidopsis MAP kinase pathway mutations mekk1 and mpk4 suggests signaling pathway complexity
 Genetic interaction and phenotypic analysis of the Arabidopsis MAP kinase pathway mutations mekk1 and mpk4 suggests signaling pathway complexity Shih-Heng Su, Maria Cristina Suarez-Rodriguez, Patrick Krysan
Genetic interaction and phenotypic analysis of the Arabidopsis MAP kinase pathway mutations mekk1 and mpk4 suggests signaling pathway complexity Shih-Heng Su, Maria Cristina Suarez-Rodriguez, Patrick Krysan
THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING. AnitaHajdu. Thesis of the Ph.D.
 THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING AnitaHajdu Thesis of the Ph.D. dissertation Supervisor: Dr. LászlóKozma-Bognár - senior research associate Doctoral
THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING AnitaHajdu Thesis of the Ph.D. dissertation Supervisor: Dr. LászlóKozma-Bognár - senior research associate Doctoral
Supplementary Figure 1. Phenotype of the HI strain.
 Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family
 Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family GENES & DEVELOPMENT (2000) 14: 108 117 INTRODUCTION Flower Diagram INTRODUCTION Abscission In plant, the process by which a plant
Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family GENES & DEVELOPMENT (2000) 14: 108 117 INTRODUCTION Flower Diagram INTRODUCTION Abscission In plant, the process by which a plant
EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA
 EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA Item Type text; Electronic Thesis Authors Bergstrand, Lauren Janel Publisher The University of Arizona. Rights Copyright is held by the author. Digital
EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA Item Type text; Electronic Thesis Authors Bergstrand, Lauren Janel Publisher The University of Arizona. Rights Copyright is held by the author. Digital
Introduction. Gene expression is the combined process of :
 1 To know and explain: Regulation of Bacterial Gene Expression Constitutive ( house keeping) vs. Controllable genes OPERON structure and its role in gene regulation Regulation of Eukaryotic Gene Expression
1 To know and explain: Regulation of Bacterial Gene Expression Constitutive ( house keeping) vs. Controllable genes OPERON structure and its role in gene regulation Regulation of Eukaryotic Gene Expression
Regulation of Phosphate Homeostasis by microrna in Plants
 Regulation of Phosphate Homeostasis by microrna in Plants Tzyy-Jen Chiou 1 *, Kyaw Aung 1,2, Shu-I Lin 1,3, Chia-Chune Wu 1, Su-Fen Chiang 1, and Chun-Lin Su 1 Abstract Upon phosphate (Pi) starvation,
Regulation of Phosphate Homeostasis by microrna in Plants Tzyy-Jen Chiou 1 *, Kyaw Aung 1,2, Shu-I Lin 1,3, Chia-Chune Wu 1, Su-Fen Chiang 1, and Chun-Lin Su 1 Abstract Upon phosphate (Pi) starvation,
BIOLOGY STANDARDS BASED RUBRIC
 BIOLOGY STANDARDS BASED RUBRIC STUDENTS WILL UNDERSTAND THAT THE FUNDAMENTAL PROCESSES OF ALL LIVING THINGS DEPEND ON A VARIETY OF SPECIALIZED CELL STRUCTURES AND CHEMICAL PROCESSES. First Semester Benchmarks:
BIOLOGY STANDARDS BASED RUBRIC STUDENTS WILL UNDERSTAND THAT THE FUNDAMENTAL PROCESSES OF ALL LIVING THINGS DEPEND ON A VARIETY OF SPECIALIZED CELL STRUCTURES AND CHEMICAL PROCESSES. First Semester Benchmarks:
Arabidopsis thaliana. Lucia Strader. Assistant Professor, Biology
 Arabidopsis thaliana Lucia Strader Assistant Professor, Biology Arabidopsis as a genetic model Easy to grow Small genome Short life cycle Self fertile Produces many progeny Easily transformed HIV E. coli
Arabidopsis thaliana Lucia Strader Assistant Professor, Biology Arabidopsis as a genetic model Easy to grow Small genome Short life cycle Self fertile Produces many progeny Easily transformed HIV E. coli
Ph.D. thesis. Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana
 Ph.D. thesis Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana Written by: Edit Ábrahám Temesváriné Supervisors: Dr. László Szabados
Ph.D. thesis Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana Written by: Edit Ábrahám Temesváriné Supervisors: Dr. László Szabados
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/9/452/ra106/dc1 Supplementary Materials for Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots Hyo-Jun Lee, Jun-Ho
www.sciencesignaling.org/cgi/content/full/9/452/ra106/dc1 Supplementary Materials for Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots Hyo-Jun Lee, Jun-Ho
Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function
 Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function Brian D. Gregory Department of Biology Penn Genome Frontiers Institute University of Pennsylvania
Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function Brian D. Gregory Department of Biology Penn Genome Frontiers Institute University of Pennsylvania
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. HSP21 expression in 35S:HSP21 and hsp21 knockdown plants. (a) Since no T- DNA insertion line for HSP21 is available in the publicly available T-DNA collections,
Supplementary Figure 1 Supplementary Figure 1. HSP21 expression in 35S:HSP21 and hsp21 knockdown plants. (a) Since no T- DNA insertion line for HSP21 is available in the publicly available T-DNA collections,
Supplemental Data. Wang et al. (2014). Plant Cell /tpc
 Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
7. Advances in host-pathogen molecular interactions: rust effectors as targets for recognition
 7. Advances in host-pathogen molecular interactions: rust effectors as targets for recognition Peter Dodds 1, Greg Lawrence 1, Rohit Mago 1, Michael Ayliffe 1, Narayana Upadhyaya 1, Les Szabo 2, Robert
7. Advances in host-pathogen molecular interactions: rust effectors as targets for recognition Peter Dodds 1, Greg Lawrence 1, Rohit Mago 1, Michael Ayliffe 1, Narayana Upadhyaya 1, Les Szabo 2, Robert
Life Science Journal 2014;11(9) Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis
 Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis Sung-Il Kim 1, Sang Ik Song 3, Hak Soo Seo 1, 2, 4 * 1 Department of Plant Science and Research Institute of Agriculture
Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis Sung-Il Kim 1, Sang Ik Song 3, Hak Soo Seo 1, 2, 4 * 1 Department of Plant Science and Research Institute of Agriculture
HRS1 Acts as a Negative Regulator of Abscisic Acid Signaling to Promote Timely Germination of Arabidopsis Seeds
 HRS1 Acts as a Negative Regulator of Abscisic Acid Signaling to Promote Timely Germination of Arabidopsis Seeds Chongming Wu 1,2., Juanjuan Feng 1,2., Ran Wang 1,2, Hong Liu 1,2, Huixia Yang 1,2, Pedro
HRS1 Acts as a Negative Regulator of Abscisic Acid Signaling to Promote Timely Germination of Arabidopsis Seeds Chongming Wu 1,2., Juanjuan Feng 1,2., Ran Wang 1,2, Hong Liu 1,2, Huixia Yang 1,2, Pedro
SUPPLEMENTARY INFORMATION
 Figure S1. Haploid plant produced by centromere-mediated genome elimination Chromosomes containing altered CENH3 in their centromeres (green dots) are eliminated after fertilization in a cross to wild
Figure S1. Haploid plant produced by centromere-mediated genome elimination Chromosomes containing altered CENH3 in their centromeres (green dots) are eliminated after fertilization in a cross to wild
Biological Roles of Cytokinins
 Direct Control of Shoot Meristem Activity by a Cytokinin-Activating Enzyme By Kurakawa et. Al. Published in Nature Presented by Boyana Grigorova Biological Roles of Cytokinins Cytokinins are positive regulators
Direct Control of Shoot Meristem Activity by a Cytokinin-Activating Enzyme By Kurakawa et. Al. Published in Nature Presented by Boyana Grigorova Biological Roles of Cytokinins Cytokinins are positive regulators
Supplemental Data. Hou et al. (2016). Plant Cell /tpc
 Supplemental Data. Hou et al. (216). Plant Cell 1.115/tpc.16.295 A Distance to 1 st nt of start codon Distance to 1 st nt of stop codon B Normalized PARE abundance 8 14 nt 17 nt Frame1 Arabidopsis inflorescence
Supplemental Data. Hou et al. (216). Plant Cell 1.115/tpc.16.295 A Distance to 1 st nt of start codon Distance to 1 st nt of stop codon B Normalized PARE abundance 8 14 nt 17 nt Frame1 Arabidopsis inflorescence
Induced resistance: an enhancement of basal resistance?
 Induced Resistance in Plants against Insects and Diseases 10BCIwprs Bull. Vol. 25(6) 2002 pp. 133-137 Induced resistance: an enhancement of basal resistance? Jurriaan Ton, Sy Ike Davison, Martin De Vos,
Induced Resistance in Plants against Insects and Diseases 10BCIwprs Bull. Vol. 25(6) 2002 pp. 133-137 Induced resistance: an enhancement of basal resistance? Jurriaan Ton, Sy Ike Davison, Martin De Vos,
Genetic Characterization and Functional Analysis of the GID1 Gibberellin Receptors in Arabidopsis W
 The Plant Cell, Vol. 18, 3399 3414, December 2006, www.plantcell.org ª 2006 American Society of Plant Biologists Genetic Characterization and Functional Analysis of the GID1 Gibberellin Receptors in Arabidopsis
The Plant Cell, Vol. 18, 3399 3414, December 2006, www.plantcell.org ª 2006 American Society of Plant Biologists Genetic Characterization and Functional Analysis of the GID1 Gibberellin Receptors in Arabidopsis
Innate Immunity of Plants, Animals and Humans
 Nucleic Acids and Molecular Biology 21 Innate Immunity of Plants, Animals and Humans Bearbeitet von Holger Heine 1. Auflage 2007. Buch. xiv, 241 S. Hardcover ISBN 978 3 540 73929 6 Format (B x L): 15,5
Nucleic Acids and Molecular Biology 21 Innate Immunity of Plants, Animals and Humans Bearbeitet von Holger Heine 1. Auflage 2007. Buch. xiv, 241 S. Hardcover ISBN 978 3 540 73929 6 Format (B x L): 15,5
Valley Central School District 944 State Route 17K Montgomery, NY Telephone Number: (845) ext Fax Number: (845)
 Valley Central School District 944 State Route 17K Montgomery, NY 12549 Telephone Number: (845)457-2400 ext. 18121 Fax Number: (845)457-4254 Advance Placement Biology Presented to the Board of Education
Valley Central School District 944 State Route 17K Montgomery, NY 12549 Telephone Number: (845)457-2400 ext. 18121 Fax Number: (845)457-4254 Advance Placement Biology Presented to the Board of Education
Penghui Li, Beibei Chen, Gaoyang Zhang, Longxiang Chen, Qiang Dong, Jiangqi Wen, Kirankumar S. Mysore and Jian Zhao
 New Phytologist Supporting Information Regulation of anthocyanin and proanthocyanidin biosynthesis by Medicago truncatula bhlh transcription factor MtTT8 Penghui Li, Beibei Chen, Gaoyang Zhang, Longxiang
New Phytologist Supporting Information Regulation of anthocyanin and proanthocyanidin biosynthesis by Medicago truncatula bhlh transcription factor MtTT8 Penghui Li, Beibei Chen, Gaoyang Zhang, Longxiang
Figure 18.1 Blue-light stimulated phototropism Blue light Inhibits seedling hypocotyl elongation
 Blue Light and Photomorphogenesis Q: Figure 18.3 Blue light responses - phototropsim of growing Corn Coleoptile 1. How do we know plants respond to blue light? 2. What are the functions of multiple BL
Blue Light and Photomorphogenesis Q: Figure 18.3 Blue light responses - phototropsim of growing Corn Coleoptile 1. How do we know plants respond to blue light? 2. What are the functions of multiple BL
Cytokinin. Fig Cytokinin needed for growth of shoot apical meristem. F Cytokinin stimulates chloroplast development in the dark
 Cytokinin Abundant in young, dividing cells Shoot apical meristem Root apical meristem Synthesized in root tip, developing embryos, young leaves, fruits Transported passively via xylem into shoots from
Cytokinin Abundant in young, dividing cells Shoot apical meristem Root apical meristem Synthesized in root tip, developing embryos, young leaves, fruits Transported passively via xylem into shoots from
Supplemental Data. Perea-Resa et al. Plant Cell. (2012) /tpc
 Supplemental Data. Perea-Resa et al. Plant Cell. (22)..5/tpc.2.3697 Sm Sm2 Supplemental Figure. Sequence alignment of Arabidopsis LSM proteins. Alignment of the eleven Arabidopsis LSM proteins. Sm and
Supplemental Data. Perea-Resa et al. Plant Cell. (22)..5/tpc.2.3697 Sm Sm2 Supplemental Figure. Sequence alignment of Arabidopsis LSM proteins. Alignment of the eleven Arabidopsis LSM proteins. Sm and
AP Biology Essential Knowledge Cards BIG IDEA 1
 AP Biology Essential Knowledge Cards BIG IDEA 1 Essential knowledge 1.A.1: Natural selection is a major mechanism of evolution. Essential knowledge 1.A.4: Biological evolution is supported by scientific
AP Biology Essential Knowledge Cards BIG IDEA 1 Essential knowledge 1.A.1: Natural selection is a major mechanism of evolution. Essential knowledge 1.A.4: Biological evolution is supported by scientific
Last time: Obtaining information from a cloned gene
 Last time: Obtaining information from a cloned gene Objectives: 1. What is the biochemical role of the gene? 2. Where and when is the gene expressed (transcribed)? 3. Where and when is the protein made?
Last time: Obtaining information from a cloned gene Objectives: 1. What is the biochemical role of the gene? 2. Where and when is the gene expressed (transcribed)? 3. Where and when is the protein made?
CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON
 PROKARYOTE GENES: E. COLI LAC OPERON CHAPTER 13 CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON Figure 1. Electron micrograph of growing E. coli. Some show the constriction at the location where daughter
PROKARYOTE GENES: E. COLI LAC OPERON CHAPTER 13 CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON Figure 1. Electron micrograph of growing E. coli. Some show the constriction at the location where daughter
An F-box gene, CPR30, functions as a negative regulator of the defense response in Arabidopsis
 The Plant Journal (2009) 60, 757 770 doi: 10.1111/j.1365-313X.2009.03995.x An F-box gene, CPR30, functions as a negative regulator of the defense response in Arabidopsis Mingyue Gou 1,2,, Nan Su 1,2,,
The Plant Journal (2009) 60, 757 770 doi: 10.1111/j.1365-313X.2009.03995.x An F-box gene, CPR30, functions as a negative regulator of the defense response in Arabidopsis Mingyue Gou 1,2,, Nan Su 1,2,,
Enhanced seed production under prolonged heat stress conditions in Arabidopsis thaliana plants deficient in cytosolic ascorbate peroxidase 2
 Journal of Experimental Botany, Vol. 64, No. 1, pp. 253 263, 2012 doi:10.1093/jxb/ers335 Advance Access publication 26 November, 2012 This paper is available online free of all access charges (see http://jxb.oxfordjournals.org/open_access.html
Journal of Experimental Botany, Vol. 64, No. 1, pp. 253 263, 2012 doi:10.1093/jxb/ers335 Advance Access publication 26 November, 2012 This paper is available online free of all access charges (see http://jxb.oxfordjournals.org/open_access.html
16 CONTROL OF GENE EXPRESSION
 16 CONTROL OF GENE EXPRESSION Chapter Outline 16.1 REGULATION OF GENE EXPRESSION IN PROKARYOTES The operon is the unit of transcription in prokaryotes The lac operon for lactose metabolism is transcribed
16 CONTROL OF GENE EXPRESSION Chapter Outline 16.1 REGULATION OF GENE EXPRESSION IN PROKARYOTES The operon is the unit of transcription in prokaryotes The lac operon for lactose metabolism is transcribed
Abstract. Introduction
 Dual Regulation of Gene Expression Mediated by Extended MAPK Activation and Salicylic Acid Contributes to Robust Innate Immunity in Arabidopsis thaliana Kenichi Tsuda 1,2 *, Akira Mine 1, Gerit Bethke
Dual Regulation of Gene Expression Mediated by Extended MAPK Activation and Salicylic Acid Contributes to Robust Innate Immunity in Arabidopsis thaliana Kenichi Tsuda 1,2 *, Akira Mine 1, Gerit Bethke
Principles of QTL Mapping. M.Imtiaz
 Principles of QTL Mapping M.Imtiaz Introduction Definitions of terminology Reasons for QTL mapping Principles of QTL mapping Requirements For QTL Mapping Demonstration with experimental data Merit of QTL
Principles of QTL Mapping M.Imtiaz Introduction Definitions of terminology Reasons for QTL mapping Principles of QTL mapping Requirements For QTL Mapping Demonstration with experimental data Merit of QTL
Abiotic Stress in Crop Plants
 1 Abiotic Stress in Crop Plants Mirza Hasanuzzaman, PhD Professor Department of Agronomy Sher-e-Bangla Agricultural University E-mail: mhzsauag@yahoo.com Stress Stress is usually defined as an external
1 Abiotic Stress in Crop Plants Mirza Hasanuzzaman, PhD Professor Department of Agronomy Sher-e-Bangla Agricultural University E-mail: mhzsauag@yahoo.com Stress Stress is usually defined as an external
Transgenic plants over-expressing HbCuZnSOD cytosolic isoform are more tolerant to a water deficit
 Transgenic plants over-expressing HbCuZnSOD cytosolic isoform are more tolerant to a water deficit J. Leclercq, F. Martin, C. Sanier, A. Clément-Vidal, D. Fabre, G. Oliver, L. Lardet, A. Ayar, M. Peyramard
Transgenic plants over-expressing HbCuZnSOD cytosolic isoform are more tolerant to a water deficit J. Leclercq, F. Martin, C. Sanier, A. Clément-Vidal, D. Fabre, G. Oliver, L. Lardet, A. Ayar, M. Peyramard
Multiple Choice Review- Eukaryotic Gene Expression
 Multiple Choice Review- Eukaryotic Gene Expression 1. Which of the following is the Central Dogma of cell biology? a. DNA Nucleic Acid Protein Amino Acid b. Prokaryote Bacteria - Eukaryote c. Atom Molecule
Multiple Choice Review- Eukaryotic Gene Expression 1. Which of the following is the Central Dogma of cell biology? a. DNA Nucleic Acid Protein Amino Acid b. Prokaryote Bacteria - Eukaryote c. Atom Molecule
SUPPLEMENTARY INFORMATION
 reverse 3175 3175 F L C 318 318 3185 3185 319 319 3195 3195 315 8 1 315 3155 315 317 Supplementary Figure 3. Stability of expression of the GFP sensor constructs return to warm conditions. Semi-quantitative
reverse 3175 3175 F L C 318 318 3185 3185 319 319 3195 3195 315 8 1 315 3155 315 317 Supplementary Figure 3. Stability of expression of the GFP sensor constructs return to warm conditions. Semi-quantitative
Gene expression in prokaryotic and eukaryotic cells, Plasmids: types, maintenance and functions. Mitesh Shrestha
 Gene expression in prokaryotic and eukaryotic cells, Plasmids: types, maintenance and functions. Mitesh Shrestha Plasmids 1. Extrachromosomal DNA, usually circular-parasite 2. Usually encode ancillary
Gene expression in prokaryotic and eukaryotic cells, Plasmids: types, maintenance and functions. Mitesh Shrestha Plasmids 1. Extrachromosomal DNA, usually circular-parasite 2. Usually encode ancillary
Chapter 39: Plant Responses to Internal and External Signals
 AP Biology Reading Guide Name Chapter 39: Plant Responses to Internal and External Signals Concept 39.1 Signal transduction pathways link signal reception to response This concept brings together the general
AP Biology Reading Guide Name Chapter 39: Plant Responses to Internal and External Signals Concept 39.1 Signal transduction pathways link signal reception to response This concept brings together the general
Supplemental Figures. Supplemental Data. Sugliani et al. Plant Cell (2016) /tpc Clades RSH1. Rsh1[HS] RSH2 RSH3. Rsh4[HS] HYD TGS ACT
![Supplemental Figures. Supplemental Data. Sugliani et al. Plant Cell (2016) /tpc Clades RSH1. Rsh1[HS] RSH2 RSH3. Rsh4[HS] HYD TGS ACT Supplemental Figures. Supplemental Data. Sugliani et al. Plant Cell (2016) /tpc Clades RSH1. Rsh1[HS] RSH2 RSH3. Rsh4[HS] HYD TGS ACT](/thumbs/81/84002827.jpg) Supplemental Figures Clades RSH1 TP - HYD TGS ACT Rsh1[HS] RSH2 TP HYD SYN TGS Rsh2[HS] RSH3 TP HYD SYN TGS CRSH TP - SYN EFh Rsh4[HS] Supplemental Figure 1. Arabidopsis RSH domain structure. Schematic
Supplemental Figures Clades RSH1 TP - HYD TGS ACT Rsh1[HS] RSH2 TP HYD SYN TGS Rsh2[HS] RSH3 TP HYD SYN TGS CRSH TP - SYN EFh Rsh4[HS] Supplemental Figure 1. Arabidopsis RSH domain structure. Schematic
Supp- Figure 2 Confocal micrograph of N. benthamiana tissues transiently expressing 35S:YFP-PDCB1. PDCB1 was targeted to plasmodesmata (twin punctate
 Supplemental Data. Simpson et al. (009). n rabidopsis GPI-anchor plasmodesmal neck protein with callosebinding activity and potential to regulate cell-to-cell trafficking. 5 0 stack Supp- Figure Confocal
Supplemental Data. Simpson et al. (009). n rabidopsis GPI-anchor plasmodesmal neck protein with callosebinding activity and potential to regulate cell-to-cell trafficking. 5 0 stack Supp- Figure Confocal
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins
 13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
SUPPLEMENTARY INFORMATION
 Supplemental Methods Isolation and mapping of SPCH An EMS-mutagenized population of tmm-1 (Col);E1728 (an enhancer trap GFP marking guard cells) was created. M2 seeds were collected from M1 s planted in
Supplemental Methods Isolation and mapping of SPCH An EMS-mutagenized population of tmm-1 (Col);E1728 (an enhancer trap GFP marking guard cells) was created. M2 seeds were collected from M1 s planted in
GCD3033:Cell Biology. Transcription
 Transcription Transcription: DNA to RNA A) production of complementary strand of DNA B) RNA types C) transcription start/stop signals D) Initiation of eukaryotic gene expression E) transcription factors
Transcription Transcription: DNA to RNA A) production of complementary strand of DNA B) RNA types C) transcription start/stop signals D) Initiation of eukaryotic gene expression E) transcription factors
Supplementary Figure S1. Amino acid alignment of selected monocot FT-like and TFL-like sequences. Sequences were aligned using ClustalW and analyzed
 Supplementary Figure S1. Amino acid alignment of selected monocot FT-like and TFL-like sequences. Sequences were aligned using ClustalW and analyzed using the Geneious software. Accession numbers of the
Supplementary Figure S1. Amino acid alignment of selected monocot FT-like and TFL-like sequences. Sequences were aligned using ClustalW and analyzed using the Geneious software. Accession numbers of the
Supplementary Figure 1 Characterization of wild type (WT) and abci8 mutant in the paddy field.
 Supplementary Figure 1 Characterization of wild type (WT) and abci8 mutant in the paddy field. A, Phenotypes of 30-day old wild-type (WT) and abci8 mutant plants grown in a paddy field under normal sunny
Supplementary Figure 1 Characterization of wild type (WT) and abci8 mutant in the paddy field. A, Phenotypes of 30-day old wild-type (WT) and abci8 mutant plants grown in a paddy field under normal sunny
Big Idea 1: The process of evolution drives the diversity and unity of life.
 Big Idea 1: The process of evolution drives the diversity and unity of life. understanding 1.A: Change in the genetic makeup of a population over time is evolution. 1.A.1: Natural selection is a major
Big Idea 1: The process of evolution drives the diversity and unity of life. understanding 1.A: Change in the genetic makeup of a population over time is evolution. 1.A.1: Natural selection is a major
Supplementary Methods
 Supplementary Methods Microarray analysis Grains of 7 DAP of the wild-type and gif1 were harvested for RNA preparation. Microarray analysis was performed with the Affymetrix (Santa Clara, CA) GeneChip
Supplementary Methods Microarray analysis Grains of 7 DAP of the wild-type and gif1 were harvested for RNA preparation. Microarray analysis was performed with the Affymetrix (Santa Clara, CA) GeneChip
AP Curriculum Framework with Learning Objectives
 Big Ideas Big Idea 1: The process of evolution drives the diversity and unity of life. AP Curriculum Framework with Learning Objectives Understanding 1.A: Change in the genetic makeup of a population over
Big Ideas Big Idea 1: The process of evolution drives the diversity and unity of life. AP Curriculum Framework with Learning Objectives Understanding 1.A: Change in the genetic makeup of a population over
Enduring understanding 1.A: Change in the genetic makeup of a population over time is evolution.
 The AP Biology course is designed to enable you to develop advanced inquiry and reasoning skills, such as designing a plan for collecting data, analyzing data, applying mathematical routines, and connecting
The AP Biology course is designed to enable you to develop advanced inquiry and reasoning skills, such as designing a plan for collecting data, analyzing data, applying mathematical routines, and connecting
 1 of 13 8/11/2014 10:32 AM Units: Teacher: APBiology, CORE Course: APBiology Year: 2012-13 Chemistry of Life Chapters 1-4 Big Idea 1, 2 & 4 Change in the genetic population over time is feedback mechanisms
1 of 13 8/11/2014 10:32 AM Units: Teacher: APBiology, CORE Course: APBiology Year: 2012-13 Chemistry of Life Chapters 1-4 Big Idea 1, 2 & 4 Change in the genetic population over time is feedback mechanisms
Epigenetics and Flowering Any potentially stable and heritable change in gene expression that occurs without a change in DNA sequence
 Epigenetics and Flowering Any potentially stable and heritable change in gene expression that occurs without a change in DNA sequence www.plantcell.org/cgi/doi/10.1105/tpc.110.tt0110 Epigenetics Usually
Epigenetics and Flowering Any potentially stable and heritable change in gene expression that occurs without a change in DNA sequence www.plantcell.org/cgi/doi/10.1105/tpc.110.tt0110 Epigenetics Usually
A MicroRNA as a Translational Repressor of APETALA2 in Arabidopsis Flower Development
 A MicroRNA as a Translational Repressor of APETALA2 in Arabidopsis Flower Development Xuemei Chen Waksman Institute, Rutgers University, Piscataway, NJ 08854, USA. E-mail: xuemei@waksman.rutgers.edu Plant
A MicroRNA as a Translational Repressor of APETALA2 in Arabidopsis Flower Development Xuemei Chen Waksman Institute, Rutgers University, Piscataway, NJ 08854, USA. E-mail: xuemei@waksman.rutgers.edu Plant
3% three branches, n 661) compared to the corresponding wild-type WS (0% unbranched, 10% two branches, Correspondence: M. Hülskamp
 Brief Communication 1891 CPR5 is involved in cell proliferation and cell death control and encodes a novel transmembrane protein V. Kirik*, D. Bouyer*, U. Schöbinger*, N. Bechtold, M. Herzog, J.-M. Bonneville
Brief Communication 1891 CPR5 is involved in cell proliferation and cell death control and encodes a novel transmembrane protein V. Kirik*, D. Bouyer*, U. Schöbinger*, N. Bechtold, M. Herzog, J.-M. Bonneville
Increasing Processing Tomato Fruit Soluble Solids
 Increasing Processing Tomato Fruit Soluble Solids Diane M Beckles Department of Plant Sciences dmbeckles@ucdavis.edu Processing Tomato Conference @ UC Davis December 13 th 2018 Soil Micronutrients Cultivar
Increasing Processing Tomato Fruit Soluble Solids Diane M Beckles Department of Plant Sciences dmbeckles@ucdavis.edu Processing Tomato Conference @ UC Davis December 13 th 2018 Soil Micronutrients Cultivar
Bio/Life: Cell Biology
 Bio/Life: Cell Biology 1a The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism's cells. As a basis for understanding
Bio/Life: Cell Biology 1a The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism's cells. As a basis for understanding
Agrobacterium tumefasciens, the Ti Plasmid, and Crown Gall Tumorigenesis
 Agrobacterium tumefasciens, the Ti Plasmid, and Crown Gall Tumorigenesis BOM-11: 10.9 Plasmids: General Principles (review) p. 274 10.11 Conjugation: Essential Features (review) p. 278 19.21 Agrobacterium
Agrobacterium tumefasciens, the Ti Plasmid, and Crown Gall Tumorigenesis BOM-11: 10.9 Plasmids: General Principles (review) p. 274 10.11 Conjugation: Essential Features (review) p. 278 19.21 Agrobacterium
GFP GAL bp 3964 bp
 Supplemental Data. Møller et al. (2009) Shoot Na + exclusion and increased salinity tolerance engineered by cell type-specific alteration of Na + transport in Arabidopsis Supplemental Figure 1. Salt-sensitive
Supplemental Data. Møller et al. (2009) Shoot Na + exclusion and increased salinity tolerance engineered by cell type-specific alteration of Na + transport in Arabidopsis Supplemental Figure 1. Salt-sensitive
Gene Control Mechanisms at Transcription and Translation Levels
 Gene Control Mechanisms at Transcription and Translation Levels Dr. M. Vijayalakshmi School of Chemical and Biotechnology SASTRA University Joint Initiative of IITs and IISc Funded by MHRD Page 1 of 9
Gene Control Mechanisms at Transcription and Translation Levels Dr. M. Vijayalakshmi School of Chemical and Biotechnology SASTRA University Joint Initiative of IITs and IISc Funded by MHRD Page 1 of 9
Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids
 Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids Plant growth conditions The soil was a 1:1 v/v mixture of loamy soil and organic compost. Initial soil water content was determined
Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids Plant growth conditions The soil was a 1:1 v/v mixture of loamy soil and organic compost. Initial soil water content was determined
Ethylene is critical to the maintenance of primary root growth and Fe. homeostasis under Fe stress in Arabidopsis
 Ethylene is critical to the maintenance of primary root growth and Fe homeostasis under Fe stress in Arabidopsis Guangjie Li, Weifeng Xu, Herbert J. Kronzucker, Weiming Shi * Supplementary Data Supplementary
Ethylene is critical to the maintenance of primary root growth and Fe homeostasis under Fe stress in Arabidopsis Guangjie Li, Weifeng Xu, Herbert J. Kronzucker, Weiming Shi * Supplementary Data Supplementary
POTASSIUM IN PLANT GROWTH AND YIELD. by Ismail Cakmak Sabanci University Istanbul, Turkey
 POTASSIUM IN PLANT GROWTH AND YIELD by Ismail Cakmak Sabanci University Istanbul, Turkey Low K High K High K Low K Low K High K Low K High K Control K Deficiency Cakmak et al., 1994, J. Experimental Bot.
POTASSIUM IN PLANT GROWTH AND YIELD by Ismail Cakmak Sabanci University Istanbul, Turkey Low K High K High K Low K Low K High K Low K High K Control K Deficiency Cakmak et al., 1994, J. Experimental Bot.
Maria V. Yamburenko, Yan O. Zubo, Radomíra Vanková, Victor V. Kusnetsov, Olga N. Kulaeva, Thomas Börner
 ABA represses the transcription of chloroplast genes Maria V. Yamburenko, Yan O. Zubo, Radomíra Vanková, Victor V. Kusnetsov, Olga N. Kulaeva, Thomas Börner Supplementary data Supplementary tables Table
ABA represses the transcription of chloroplast genes Maria V. Yamburenko, Yan O. Zubo, Radomíra Vanková, Victor V. Kusnetsov, Olga N. Kulaeva, Thomas Börner Supplementary data Supplementary tables Table
USE OF ARABIDOPSIS FOR GENETIC DISSECTION OF PLANT DEFENSE RESPONSES
 Annu. Rev. Genet. 1997. 31:547 69 Copyright c 1997 by Annual Reviews Inc. All rights reserved USE OF ARABIDOPSIS FOR GENETIC DISSECTION OF PLANT DEFENSE RESPONSES J. Glazebrook Center for Agricultural
Annu. Rev. Genet. 1997. 31:547 69 Copyright c 1997 by Annual Reviews Inc. All rights reserved USE OF ARABIDOPSIS FOR GENETIC DISSECTION OF PLANT DEFENSE RESPONSES J. Glazebrook Center for Agricultural
7.06 Cell Biology EXAM #3 April 21, 2005
 7.06 Cell Biology EXAM #3 April 21, 2005 This is an open book exam, and you are allowed access to books, a calculator, and notes but not computers or any other types of electronic devices. Please write
7.06 Cell Biology EXAM #3 April 21, 2005 This is an open book exam, and you are allowed access to books, a calculator, and notes but not computers or any other types of electronic devices. Please write
Prereq: Concurrent 3 CH
 0201107 0201101 General Biology (1) General Biology (1) is an introductory course which covers the basics of cell biology in a traditional order, from the structure and function of molecules to the structure
0201107 0201101 General Biology (1) General Biology (1) is an introductory course which covers the basics of cell biology in a traditional order, from the structure and function of molecules to the structure
Effect of light and the cer10 mutation on the growth rate of Arabidopsis thaliana
 Effect of light and the cer10 mutation on the growth rate of Arabidopsis thaliana Jenna K. Bains, Selam Joseph, Shayan Shokoohi, Ian A. Villamin Abstract The presence of light and the presence of a mutation
Effect of light and the cer10 mutation on the growth rate of Arabidopsis thaliana Jenna K. Bains, Selam Joseph, Shayan Shokoohi, Ian A. Villamin Abstract The presence of light and the presence of a mutation
Resistance gene analogues of Arabidopsis thaliana: recognition by structure
 J. Appl. Genet. 44(3), 2003, pp. 311-321 Resistance gene analogues of Arabidopsis thaliana: recognition by structure Jerzy CHE KOWSKI, Grzegorz KOCZYK Institute of Plant Genetics, Polish Academy of Sciences,
J. Appl. Genet. 44(3), 2003, pp. 311-321 Resistance gene analogues of Arabidopsis thaliana: recognition by structure Jerzy CHE KOWSKI, Grzegorz KOCZYK Institute of Plant Genetics, Polish Academy of Sciences,
Chapter 1 Introduction
 Chapter 1 Introduction 1. INTRODUCTION Plants being sessile are exposed to environmental stresses mainly abiotic, caused by non-living effects of environment (temperature extremes, drought, and salinity)
Chapter 1 Introduction 1. INTRODUCTION Plants being sessile are exposed to environmental stresses mainly abiotic, caused by non-living effects of environment (temperature extremes, drought, and salinity)
Major Plant Hormones 1.Auxins 2.Cytokinins 3.Gibberelins 4.Ethylene 5.Abscisic acid
 Plant Hormones Lecture 9: Control Systems in Plants What is a Plant Hormone? Compound produced by one part of an organism that is translocated to other parts where it triggers a response in target cells
Plant Hormones Lecture 9: Control Systems in Plants What is a Plant Hormone? Compound produced by one part of an organism that is translocated to other parts where it triggers a response in target cells
Map of AP-Aligned Bio-Rad Kits with Learning Objectives
 Map of AP-Aligned Bio-Rad Kits with Learning Objectives Cover more than one AP Biology Big Idea with these AP-aligned Bio-Rad kits. Big Idea 1 Big Idea 2 Big Idea 3 Big Idea 4 ThINQ! pglo Transformation
Map of AP-Aligned Bio-Rad Kits with Learning Objectives Cover more than one AP Biology Big Idea with these AP-aligned Bio-Rad kits. Big Idea 1 Big Idea 2 Big Idea 3 Big Idea 4 ThINQ! pglo Transformation
Flowering Time Control in Plants -How plants know the time to flower?
 Advanced Molecular and Cell Biology II, 2015/12/04 Flowering Time Control in Plants -How plants know the time to flower? Masaki NIWA Grad. Sch. Biostudies, Kyoto Univ. Why can plants bloom every year in
Advanced Molecular and Cell Biology II, 2015/12/04 Flowering Time Control in Plants -How plants know the time to flower? Masaki NIWA Grad. Sch. Biostudies, Kyoto Univ. Why can plants bloom every year in
RECOGNITION AND RESPONSE IN THE PLANT IMMUNE SYSTEM
 Annu. Rev. Genet. 2003. 37:579 609 doi: 10.1146/annurev.genet.37.110801.142628 Copyright c 2003 by Annual Reviews. All rights reserved First published online as a Review in Advance on August 6, 2003 RECOGNITION
Annu. Rev. Genet. 2003. 37:579 609 doi: 10.1146/annurev.genet.37.110801.142628 Copyright c 2003 by Annual Reviews. All rights reserved First published online as a Review in Advance on August 6, 2003 RECOGNITION
Common Effects of Abiotic Stress Factors on Plants
 Common Effects of Abiotic Stress Factors on Plants Plants are living organisms which lack ability of locomotion. Animals can move easily from one location to other. Immovable property of plants makes it
Common Effects of Abiotic Stress Factors on Plants Plants are living organisms which lack ability of locomotion. Animals can move easily from one location to other. Immovable property of plants makes it
