Radicle emergence (%)
|
|
|
- Melinda Scott
- 5 years ago
- Views:
Transcription
1 A MDCA (µm) Radicle emergence (%) B 5 µm 75 µm 1 µm 125 µm 15 µm 175 µm 2 µm Figure S1. Effect of MDCA on germination of Arabidopsis. (A) Radicle emergence (%) of seeds 2 DAG grown on.5xms-medium supplemented with different concentrations of MDCA (n>25) (B) Phenotype of seedlings (12 DAG) grown on.5xms-medium supplemented with different concentrations of MDCA (n>25) (scale bar:.5 cm).
2 Compound Top-1 Retention time (min) 1 µm MDCA Average St. Dev. Average St. Dev. Fold change MDCA/ 1. Cinnamoyl aspartate 1 2. MDCA glutamate 1 3. MDCA aspartate 1 4. Cinnamoyl aspartate 2 5. MDCA aspartate 2 6. MDCA glutamate 2 7. Unknown 8. Cinnamoyl glutamate 9. Cinnamoyl aspartate 1 (fragment) 1. MDCA aspartate 2 (heterodimer) Compound Top-1 DWN 1. 4-methylthio-N-butyl-glucosinolate 2. 1-methoxyindol-3-ylmethyl-glucosinolate 3. G(8--4)ferulic acid + hexose + hexose 4. G(8--4)ferulic acid + hexose + hexose 5. 8-methylsulfinyloctyl-glucosinolate 6. Kaempferol + hexose + deoxyhexose 7. 5-methylthio-N-pentyl-glucosinolate 8. Unknown 9. Quercetin + hexose + deoxyhexose 1. 8-methylthio-N-octyl-glucosinolate (dimer) Figure S2. Metabolites with an altered abundance in 1 μm MDCA-treated Arabidopsis seedlings. An overview of the top-1 significantly differential compounds with at least a 1-fold increase () or decrease (DWN) in 1 μm MDCA-treated seedlings as compared to -treated seedlings (n=6). The top-1 defined as those compounds with the highest normalized peak area in MDCA-treated seedlings, whereas the top-1 DWN were defined as those compounds with the highest normalized peak area in -treated seedlings. The retention time is expressed in minutes. For each compound, normalized average peak areas (unitless) of - and 1 μm MDCA-treated seedlings are given. Peak areas are normalized relative to the dry weight of the pellet remaining after methanol extraction. The term implies that a peak could only be detected in 1 μm MDCA-treated seedlings and not in -treated seedlings.
3 A PA + hexose (formic acid adduct) (5.92 min, ) 1 intensity (%) ; in source fragment, [M-H + -dehyrated hexose] - = [PA-H + ] theoretical [M+ formic acid - H + ] - = retention time (min) B MS/MS @ 5.92 min (in-source fragment of PA + hexose) [PA-H + -C 2 -CH 2 ] hexose H H intensity (%) [PA-H + -C 2 ] [PA-H + ] C PA + malate (9.71 min, ) 1 intensity (%) ; in source fragment, [M-H + -dehyrated malate] - = [PA-H + ] theoretical [M -H + ] - = retention time (min) D MS/MS @ 9.71 min (in-source fragment of PA + malate) [PA-H + -C 2 -CH 2 ] [PA-H + -C 2 ] H H intensity (%) [PA-H + ] Figure S3. UHPLC-MS based detection of PA + hexose and PA + malate in MDCA treated Arabidopsis seedlings. Structural identification of the compounds was done via accurate match and MS/MS fragmentation spectra. (A) PA + hexose was detected at 5.92 min as formate adduct and as an in-source fragment. (B) The MS/MS of the in-source fragment of PA + hexose with eluting at 5.92 min showed characteristic fragmentation pattern of PA. (C) PA + malate was detected at 9.71 min as molecular ion and as an in-source fragment. (D) The MS/MS of the in-source fragment of PA + malate with eluting at 9.71 min showed characteristic fragmentation pattern of PA.
4 Compound Retention time (min) 1 µm MDCA Average St. Dev. Average St. Dev. Fold change MDCA/ Cinnamic acid derivatives Cinnamoyl hexose (formic acid adduct) Cinnamoyl malate 1 Cinnamoyl malate *** p-coumaric acid derivatives p-coumaric acid 4--hexoside p-coumaroyl hexose 1 p-coumaroyl hexose 2 Dihydro-p-coumaric acid + hexose *.1516***.4744*** Caffeic acid derivatives Caffeic acid 1 Caffeic acid 2 Caffeic acid 3/4--hexoside 3 Caffeoyl hexose 1 Caffeoyl hexose 3/4--hexoside 1 Caffeoyl malate 1 Caffeoyl malate *** ***
5 Compound Retention time (min) 1 µm MDCA Average St. Dev. Average St. Dev. Fold change MDCA/ Ferulic acid derivatives Ferulic acid Feruloyl hexose 1 Feruloyl hexose 2 Ferulic acid -4-hexoside Feruloyl malate 1 Feruloyl malate ***.68***.1733***.2158 Sinapic acid derivatives Sinapic acid Sinapoyl hexose 1 Sinapoyl hexose 2 Disinapoyl hexose 1 Disinapoyl hexose 2 Sinapoyl malate 1 Sinapoyl malate ** Varia Coniferin (formic acid adduct) Scopolin (formic acid adduct) ***.2442***
6 Compound Retention time (min) 1 µm MDCA Average St. Dev. Average St. Dev. Fold change MDCA/ Coniferyl alcohol - ferulic acid conjugates G(8--4) ferulic acid G(8--4) ferulic acid + hexose 1 G(8--4) ferulic acid + hexose 2 G(8--4) ferulic acid + hexose 3 G(8--4) ferulic acid + hexose 4 G(8--4) ferulic acid + hexose 5 G(8--4) ferulic acid + hexose 6 G(8--4) ferulic acid + malate 1 G(8--4) ferulic acid + malate 2 G(8--4) ferulic acid + malate 3 G(8--4) ferulic acid + malate 4 G(8--4) ferulic acid Da 1 G(8--4) ferulic acid Da *** 1.798*** ***.6178**.6748*.2275***.3841***.448***.542*** G(8-5) feruloyl hexose 1 G(8-5) feruloyl hexose 2 G(8-5) feruloyl hexose 3 G(8-5) feruloyl malate G(8-5) feruloyl malate -4-hexoside 1 G(8-5) feruloyl malate -4-hexoside ***.2451***.4154*.4891**.319***.5487
7 Compound Retention time (min) 1 µm MDCA Average St. Dev. Average St. Dev. Fold change MDCA/ Hexosylated oligolignols G(8-5)G + hexose G 4--hexoside(8-5)G G(red8-5)G + hexose G(red8-8)G 8/4--hexoside G(8-8)G hexoside 1 G(8-8)G hexoside 2 G(8-8)G + hexose + hexose G(8--4)G(red8-5)G + hexose 1 G(8--4)G(red8-5)G + hexose ***.1814***.2128***.542***.475***.17*** *** Flavonol glycosides Isorhamnetin + hexose + deoxyhexose Isorhamnetin-3--rhamnoside-7--glucoside Isorhamnetin + deoxyhexose + deoxyhexose Isorhamnetin + hexose + deoxyhexose ***.431***.87.37***
8 Compound Retention time (min) 1 µm MDCA Average St. Dev. Average St. Dev. Fold change MDCA/ Flavonol glycosides Kaempferol-3--rhamnosyl(1-->2)- glucoside -7--rhamnoside Kaempferol + deoxyhexose + deoxyhexose Kaempferol + hexose + hexose+ deoxyhexose Kaempferol-3--rhamnoside-7--glucoside Kaempferol + hexose + deoxyhexose + deoxyhexose Kaempferol-3--rhamnoside-7--rhamnoside Kaempferol + hexose + deoxyhexose Kaempferol + hexose Kaempferol + deoxyhexose Kaempferol + deoxyhexose *.786**.3747** ***.386***.338***.4919 Quercetin + hexose + deoxyhexose Quercetin-3--rhamnosyl(1-->2)- glucoside -7--rhamnoside Quercetin + hexose + hexose+ deoxyhexose Quercetin + hexose + deoxyhexose + deoxyhexose Quercetin-3--rhamnoside-7--glucoside Quercetin + hexose + deoxyhexose Quercetin + deoxyhexose **.6289**.683***.58**.6831*.37***.754*** Figure S4. Metabolites with an altered abundance in 1 μm MDCA-treated Arabidopsis seedlings. A targeted approach was used to investigate which of the identified compounds are altered in 1 μm MDCA-treated seedlings in comparison with -treated seedlings (n=6). For each compound, normalized average peak areas (unitless) of - and 1 μm MDCA-treated seedlings are given. Peak areas are normalized relative to the dry weight of the pellet remaining after methanol extraction. The retention time is expressed in minutes. Asterisks represent significant differences between 1 μm MDCA-treated and -treated plants as determined by Dunnett s test. Dunnett s test P-values: *.1 P <.5, **.1 P <.1, *** P <.1. The term implies that the compound could only be detected in 1 μm MDCA-treated seedlings and not in -treated seedlings.
9 Number of endodermal cells *** ** 1 MDCA (µm) no compl. compl. compl. MDCA 1 µm compl.+ MDCA 1 µm Figure S5. The impact of the MDCA-induced lignin reduction on the plant phenotype. Visualization of the effect of MDCA treatment on the Casparian strip formation (white; no compl.) in Arabidopsis seedlings 5 DAG and complementation by exogenous application of two monolignols (grey; compl.): 5 μm of each coniferyl alcohol and sinapyl alcohol, which allows for the formation of a functional Casparian strip (n=1). See manuscript for additional explanation on this experiment. Error bars represent standard deviations and asterisks were used to indicate statistically significant differences compared to the corresponding -treated control sample as determined by Dunnett's test P-values: *P <.5, **P <.1, *** P <.1. (B)
10 Peak area/dry weight (mg -1 ) * MDCA 1 µm Figure S6. Detection of salicylic acid in 1 μm MDCA-treated Arabidopsis seedlings. The average peak area of salicylic acid (SA) in - and 1 μm MDCA-treated seedlings 12 DAG (n=1). The unitless peak areas were normalized to the dry weight of the pellet remaining after methanol extraction (in mg). Error bars represent standard deviations. The asterisk represents significant difference in SA levels between 1 μm MDCA-treated and -treated plants (.1 < P-value <.5) as determined by Dunnett's test.
11 A H N H B IAA MDCA x z H y z x y C Physiochemical Properties MM pka clogp([octanol]/[water]) partition #H-acceptors #H-donors Total surface area Polar surface area Rotable bonds Aromatic rings D IAA MDCA E Figure S7. Chemical and physical characteristics and docking of MDCA for TIR1. (A) The molecular structure of IAA and MDCA. (B) Top view of energy minimized structures represented as sticks with bicyclic ring structures planar. The IAA carboxylic acid group does not position in the plane of the aromatic ring, whilst for MDCA, the carboxylic acid is fixed planar to the ring due to the trans-alkene bond. (C) Physiochemical properties of IAA and MDCA. (D) The binding pocket of TIR1 shown as a surface representation in relation to the whole structure whereby the binding region is defined as an 18Å x 18Å x 18Å box. Docking of IAA from the crystal structure (purple) with that of the docked result (grey) showed almost identical and superimposable results. (E) The best possible pose for MDCA in the lower region of the TIR1 pocket.
12 MDCA 5 µm MDCA 1 µm Indole-3-actetic acid (IAA) 15.5 (2.5) 2.6 (2.3) * 27.8 (2.6) *** Precursors Anthranilate 26.5 (6.4) 36.5 (7.8) 38.1 (9.3) Tryptophan Tryptamine Indole-3-acetamide Indole-3-acetonitrile Indole-3-acetaldoxime Indole-3-acetaldehyde (58.9).18 (.5) 1.13 (.26) (79.5) 12.5 (2.4) 95.3 (24.6) (389.1).28 (.7) * 1.86 (.37) ** (126.7) * 16.9 (3.) * 126. (2.8) (648.9).3 (.8) * 2.71 (.61) *** 19.8 (189.1) *** 2.2 (3.4) ** 16. (43.1) * Indole-3-pyruvic acid (15.3) (22.6) 149. (39.) Conjugates, catabolites 2-oxindole-3-acetic acid IAA-glutamate IAA-aspartate (26.1).94 (.16).85 (.2) (2.22) 1.66 (.36) ** 1.7 (.37) ** (26.82) * 2.24 (.28) *** 2.66 (.62) *** Figure S8. Concentration of auxin and auxin metabolites after MDCA treatment of Arabidopsis seedlings. The concentration of free IAA, IAA-precursors, IAA-amino acid conjugates and catabolites in seedlings (12 DAG) grown on.5xms-medium supplemented with MDCA (5 μm and 1 μm). Each biological replicate represents ten seedlings that were pooled and analyzed (n=6). Standard deviations are mentioned inbetween brackets and asterisks represent statistically significant differences between MDCA-treated and -treated plants as determined by Dunnett's test. P-values: *P <.5, **P <.1, *** P <.1.
13 Indole-3-actetic acid (IAA) WT 9.1 (.7) ref3-2 mutant 13.3 (1.9) *** Precursors Anthranilate Tryptophan Tryptamine Indole-3-acetamide Indole-3-acetonitrile Indole-3-acetaldoxime Indole-3-acetaldehyde Indole-3-pyruvic acid 26.8 (7.4) (183.3) 1. (.3) 1.2 (.3) (117.8) 7.5 (1.4) 69.8 (1.7) 218 (42) 15.6 (2.6) ** 43.4 (137.8) 1.1 (.2) 2.2 (.5) ** (71.1) ** 4. (.9) *** (42.7) * 344 (87) ** Conjugates, catabolites 2-oxindole-3-acetic acid IAA-glutamate IAA-aspartate 65.2 (15.6) 2.5 (.6) 6.5 (1.8) 93.4 (2.) * 9.5 (2.1) *** 72.8 (13.9) *** Figure S9. Concentration of auxin and auxin metabolites in the c4h mutant ref3-2. The concentration of free IAA, IAA-precursors, IAA-amino acid conjugates and catabolites in the leaves of 2 months old ref3-2 plants. Each biological replicate represents one plant (n=6). Standard deviations are mentioned inbetween brackets and asterisks represent statistically significant differences between wild type (WT) and c4h mutant ref3-2 plants as determined by Dunnett's test. P-values: *P <.5, **P <.1, *** P <.1.
14 A.25 Accumulation of [ 3 H]-NAA (pmol per 1 mg FW) B Accumulation of [ 3 H]-2,4-D (pmol per 1 mg FW) MDCA 5 µm Time (min) MDCA 5 µm Time (min) Figure S1. The impact of MDCA on auxin transport in suspension-grown Arabidopsis T87 cells. (A-B) Effect of MDCA on the net accumulation of (A) [ 3 H]-NAA or (B) [ 3 H]-2,4-D in four-day old suspension-grown Arabidopsis cells (2 minute uptake period). Arrows point at time of application of MDCA. Error bars in (A-B) represent standard deviations (n=4).
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated
 Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Additional file 1. Martin Schäfer, Christoph Brütting, Ian T. Baldwin and Mario Kallenbach
 Additional file 1 High-throughput quantification of more than 100 primary- and secondarymetabolites, and phytohormones by a single solid-phase extraction based sample preparation with analysis by UHPLC-HESI-MS/MS
Additional file 1 High-throughput quantification of more than 100 primary- and secondarymetabolites, and phytohormones by a single solid-phase extraction based sample preparation with analysis by UHPLC-HESI-MS/MS
Ethylene is critical to the maintenance of primary root growth and Fe. homeostasis under Fe stress in Arabidopsis
 Ethylene is critical to the maintenance of primary root growth and Fe homeostasis under Fe stress in Arabidopsis Guangjie Li, Weifeng Xu, Herbert J. Kronzucker, Weiming Shi * Supplementary Data Supplementary
Ethylene is critical to the maintenance of primary root growth and Fe homeostasis under Fe stress in Arabidopsis Guangjie Li, Weifeng Xu, Herbert J. Kronzucker, Weiming Shi * Supplementary Data Supplementary
Assembly of a lignin modification toolbox
 Assembly of a lignin modification toolbox Alan Friedman Kevin Dolan Nick Fico Nick Anderson Nick Bonawitz Jo Cusumano Shiva Hemmati Shinyoung Lee Sirius Li Yi Li Junko Maeda Jody Banks Mike Ladisch Shawn
Assembly of a lignin modification toolbox Alan Friedman Kevin Dolan Nick Fico Nick Anderson Nick Bonawitz Jo Cusumano Shiva Hemmati Shinyoung Lee Sirius Li Yi Li Junko Maeda Jody Banks Mike Ladisch Shawn
Evaluation and application of a targeted SPE-LC-MS method for quantifying plant hormones and phenolics in Arabidopsis
 1.171/FP163_AC CSIRO 17 Supplementary Material: Functional Plant Biology, 17, 44(6), 624-634. Evaluation and application of a targeted SPE-LC-MS method for quantifying plant hormones and phenolics in Arabidopsis
1.171/FP163_AC CSIRO 17 Supplementary Material: Functional Plant Biology, 17, 44(6), 624-634. Evaluation and application of a targeted SPE-LC-MS method for quantifying plant hormones and phenolics in Arabidopsis
Supplementary Figure 1 Characterization of wild type (WT) and abci8 mutant in the paddy field.
 Supplementary Figure 1 Characterization of wild type (WT) and abci8 mutant in the paddy field. A, Phenotypes of 30-day old wild-type (WT) and abci8 mutant plants grown in a paddy field under normal sunny
Supplementary Figure 1 Characterization of wild type (WT) and abci8 mutant in the paddy field. A, Phenotypes of 30-day old wild-type (WT) and abci8 mutant plants grown in a paddy field under normal sunny
1) Which of the following represents the breaking of a noncovalent interaction? Topic: The Nature of Noncovalent Interactions
 Multiple Choice Questions 1) Which of the following represents the breaking of a noncovalent interaction? A) hydrolysis of an ester B) dissolving of salt crystals C) ionization of water D) decomposition
Multiple Choice Questions 1) Which of the following represents the breaking of a noncovalent interaction? A) hydrolysis of an ester B) dissolving of salt crystals C) ionization of water D) decomposition
Analysis - HPLC A.136. Primesep 5 µm columns B.136
 Primesep 5 µm columns Primesep columns feature double functionality of the bonding i.e : alkyl chain with anionic or cationic group, chelating group. This feature creates unique selectivities when using
Primesep 5 µm columns Primesep columns feature double functionality of the bonding i.e : alkyl chain with anionic or cationic group, chelating group. This feature creates unique selectivities when using
Aromatic Hydrocarbons
 Aromatic Hydrocarbons Aromatic hydrocarbons contain six-membered rings of carbon atoms with alternating single and double carbon-carbon bonds. The ring is sometimes shown with a circle in the center instead
Aromatic Hydrocarbons Aromatic hydrocarbons contain six-membered rings of carbon atoms with alternating single and double carbon-carbon bonds. The ring is sometimes shown with a circle in the center instead
Supplemental Information. Molecular Basis of Spectral Diversity. in Near-Infrared Phytochrome-Based. Fluorescent Proteins
 Chemistry & Biology, Volume 22 Supplemental Information Molecular Basis of Spectral Diversity in Near-Infrared Phytochrome-Based Fluorescent Proteins Daria M. Shcherbakova, Mikhail Baloban, Sergei Pletnev,
Chemistry & Biology, Volume 22 Supplemental Information Molecular Basis of Spectral Diversity in Near-Infrared Phytochrome-Based Fluorescent Proteins Daria M. Shcherbakova, Mikhail Baloban, Sergei Pletnev,
Application Note. Edgar Naegele. Abstract
 Fast identification of main drug metabolites by quadrupole time-of-flight LC/MS Measuring accurate MS and MS/MS data with the Agilent 651 Q-TOF LC/MS and identification of main meta-bolites by comparison
Fast identification of main drug metabolites by quadrupole time-of-flight LC/MS Measuring accurate MS and MS/MS data with the Agilent 651 Q-TOF LC/MS and identification of main meta-bolites by comparison
Supplemental Figure 1: Increased Fe deficiency gene expression in roots of nas4x-2
 Supplemental Figure 1: Increased Fe deficiency gene expression in roots of nas4x-2 IRT1, FRO2 and FIT expression levels in roots of the wild-type, nas4x- 1 and nas4x-2, showing that in both nas mutants
Supplemental Figure 1: Increased Fe deficiency gene expression in roots of nas4x-2 IRT1, FRO2 and FIT expression levels in roots of the wild-type, nas4x- 1 and nas4x-2, showing that in both nas mutants
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL
 GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature10534 Supplementary Fig. 1. Diagrammatic representation of the N-end rule pathway of targeted proteolysis (after Graciet and Wellmer 2010 9 ). Tertiary, secondary
SUPPLEMENTARY INFORMATION doi:10.1038/nature10534 Supplementary Fig. 1. Diagrammatic representation of the N-end rule pathway of targeted proteolysis (after Graciet and Wellmer 2010 9 ). Tertiary, secondary
Supplemental Data. Perrella et al. (2013). Plant Cell /tpc
 Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
The new Water Screening PCDL
 The new Water Screening PCDL Content and integration in suspect and non-target screening Dr. Thomas Glauner Senior LC/MS Applications Scientist EMEA Market Development Team 1 Accurate mass screening and
The new Water Screening PCDL Content and integration in suspect and non-target screening Dr. Thomas Glauner Senior LC/MS Applications Scientist EMEA Market Development Team 1 Accurate mass screening and
Analysis of Synthetic Cannabinoids and Metabolites: Adding New Compounds to an Existing LC-MS/MS Method
 Analysis of Synthetic Cannabinoids and Metabolites: Adding New Compounds to an Existing LC-MS/MS Method By Sharon Lupo and Frances Carroll Abstract The analysis of synthetic cannabinoids and their metabolites
Analysis of Synthetic Cannabinoids and Metabolites: Adding New Compounds to an Existing LC-MS/MS Method By Sharon Lupo and Frances Carroll Abstract The analysis of synthetic cannabinoids and their metabolites
Crystal structure of DL-Tryptophan at 173K
 Cryst. Res. Technol. 39, No. 3, 274 278 (2004) / DOI 10.1002/crat.200310182 Crystal structure of DL-Tryptophan at 173K Ch. B. Hübschle, M. Messerschmidt, and P. Luger* Institut für Chemie / Kristallographie,
Cryst. Res. Technol. 39, No. 3, 274 278 (2004) / DOI 10.1002/crat.200310182 Crystal structure of DL-Tryptophan at 173K Ch. B. Hübschle, M. Messerschmidt, and P. Luger* Institut für Chemie / Kristallographie,
Supplemental Data. Gao et al. (2012). Plant Cell /tpc
 Supplemental Figure 1. Plant EMP Proteins. (A) The Accession numbers of the 12 EMP members from Arabidopsis. (B) Phylogenetic analysis of EMP proteins from Arabidopsis, human and yeast using the Mac Vector
Supplemental Figure 1. Plant EMP Proteins. (A) The Accession numbers of the 12 EMP members from Arabidopsis. (B) Phylogenetic analysis of EMP proteins from Arabidopsis, human and yeast using the Mac Vector
Viewing and Analyzing Proteins, Ligands and their Complexes 2
 2 Viewing and Analyzing Proteins, Ligands and their Complexes 2 Overview Viewing the accessible surface Analyzing the properties of proteins containing thousands of atoms is best accomplished by representing
2 Viewing and Analyzing Proteins, Ligands and their Complexes 2 Overview Viewing the accessible surface Analyzing the properties of proteins containing thousands of atoms is best accomplished by representing
GFP GAL bp 3964 bp
 Supplemental Data. Møller et al. (2009) Shoot Na + exclusion and increased salinity tolerance engineered by cell type-specific alteration of Na + transport in Arabidopsis Supplemental Figure 1. Salt-sensitive
Supplemental Data. Møller et al. (2009) Shoot Na + exclusion and increased salinity tolerance engineered by cell type-specific alteration of Na + transport in Arabidopsis Supplemental Figure 1. Salt-sensitive
CHAPTER 29 HW: AMINO ACIDS + PROTEINS
 CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
Carboxylic Acids and Nitriles
 Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Table S1 List of primers used for genotyping and qrt-pcr.
 Table S1 List of primers used for genotyping and qrt-pcr. genotyping! allele! ligomer*! 5'-sequence-3'! rice! d10-2! F! TTGGCTTTGCCTCGTTTC!!! R! AGCCTCCACTTGTACTGTG! Arabidopsis! max2-3, max2-4! F! ACTCTCTCCGACCTCCCTGACG!!!
Table S1 List of primers used for genotyping and qrt-pcr. genotyping! allele! ligomer*! 5'-sequence-3'! rice! d10-2! F! TTGGCTTTGCCTCGTTTC!!! R! AGCCTCCACTTGTACTGTG! Arabidopsis! max2-3, max2-4! F! ACTCTCTCCGACCTCCCTGACG!!!
Numerical Data. Effect of copper on nutrients content (mg g 1 dry wt.) of radish (45th day) N P K Na Ca Mg
 Numerical Data Effect of copper on nutrients (mg g 1 dry wt.) of radish (45th day) Copper added in the soil N P K Na Ca Mg (mg kg 1 ) Control 32.66 6.31 43.52 1.70 13.56 3.99 50 37.35(+14.36) 6.97(+10.45)
Numerical Data Effect of copper on nutrients (mg g 1 dry wt.) of radish (45th day) Copper added in the soil N P K Na Ca Mg (mg kg 1 ) Control 32.66 6.31 43.52 1.70 13.56 3.99 50 37.35(+14.36) 6.97(+10.45)
Acid-Base Chemistry. Introduction to Reaction Mechanism
 Acid-Base Chemistry Introduction to Reaction Mechanism What is an acid and what is a base? Bronsted-Lowry definition Acids are proton donors (A-H) e.g. HCl, H 2 SO 4, HBr, HNO 3, HI etc. Bronsted-Lowry
Acid-Base Chemistry Introduction to Reaction Mechanism What is an acid and what is a base? Bronsted-Lowry definition Acids are proton donors (A-H) e.g. HCl, H 2 SO 4, HBr, HNO 3, HI etc. Bronsted-Lowry
Supplemental Data. Perea-Resa et al. Plant Cell. (2012) /tpc
 Supplemental Data. Perea-Resa et al. Plant Cell. (22)..5/tpc.2.3697 Sm Sm2 Supplemental Figure. Sequence alignment of Arabidopsis LSM proteins. Alignment of the eleven Arabidopsis LSM proteins. Sm and
Supplemental Data. Perea-Resa et al. Plant Cell. (22)..5/tpc.2.3697 Sm Sm2 Supplemental Figure. Sequence alignment of Arabidopsis LSM proteins. Alignment of the eleven Arabidopsis LSM proteins. Sm and
NECTAR COLLECTION AND ANALYSES. One significant consideration when performing nectar collection is timing, both
 NECTAR COLLECTION AND ANALYSES COLLECTION PROCEDURES Important Considerations One significant consideration when performing nectar collection is timing, both developmental and circadian. This is because
NECTAR COLLECTION AND ANALYSES COLLECTION PROCEDURES Important Considerations One significant consideration when performing nectar collection is timing, both developmental and circadian. This is because
** * * * Col-0 cau1 CAU1. Actin2 CAS. Actin2. Supplemental Figure 1. CAU1 affects calcium accumulation.
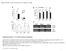 Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
How proteins separate on reverse-phase HPLC
 1 Reverse Phase How proteins separate on reverse-phase HPLC RP chromatography separates proteins through the interaction of the hydrophobic foot of the protein with a nonpolar surface of the particle RP
1 Reverse Phase How proteins separate on reverse-phase HPLC RP chromatography separates proteins through the interaction of the hydrophobic foot of the protein with a nonpolar surface of the particle RP
2017 Reaction of cinnamic acid chloride with ammonia to cinnamic acid amide
 217 Reaction of cinnamic acid chloride with ammonia to cinnamic acid amide O O Cl NH 3 NH 2 C 9 H 7 ClO (166.6) (17.) C 9 H 9 NO (147.2) Classification Reaction types and substance classes reaction of
217 Reaction of cinnamic acid chloride with ammonia to cinnamic acid amide O O Cl NH 3 NH 2 C 9 H 7 ClO (166.6) (17.) C 9 H 9 NO (147.2) Classification Reaction types and substance classes reaction of
Content : Properties of amino acids.. Separation and Analysis of Amino Acids
 قسم الكيمياء الحيوية.دولت على سالمه د. استاذ الكيمياء الحيوية ٢٠١٥-٢٠١٤ المحاضرة الثانية 1 Content : Properties of amino acids.. Separation and Analysis of Amino Acids 2 3 Physical Properties of Amino
قسم الكيمياء الحيوية.دولت على سالمه د. استاذ الكيمياء الحيوية ٢٠١٥-٢٠١٤ المحاضرة الثانية 1 Content : Properties of amino acids.. Separation and Analysis of Amino Acids 2 3 Physical Properties of Amino
2/28/2011. Chapter 6 An Overview of Organic Reactions. Organic Chemical Reactions. 6.1 Kinds of Organic Reactions
 John E. McMurry http://www.cengage.com/chemistry/mcmurry Chapter 6 An Overview of Organic Reactions CHP 6 Problems: 6.1-13, 17-36. Richard Morrison University of Georgia, Athens Organic Chemical Reactions
John E. McMurry http://www.cengage.com/chemistry/mcmurry Chapter 6 An Overview of Organic Reactions CHP 6 Problems: 6.1-13, 17-36. Richard Morrison University of Georgia, Athens Organic Chemical Reactions
CH 3 CH 2 OH +H 2 O CHO. 2e + 2H + + O 2 H 2 O +HCOOH
 2 4 H CH 3 2e + 2H + + 2 H 2 2 H CH 2 H 2e + 2H + + 2 H 2 2 H +H 2 CH 2e + 2H + + 2 H 2 2 H +HCH Supplemental Figure S. The three-step 4DM reaction, each step requires two reducing equivalents from ADPH
2 4 H CH 3 2e + 2H + + 2 H 2 2 H CH 2 H 2e + 2H + + 2 H 2 2 H +H 2 CH 2e + 2H + + 2 H 2 2 H +HCH Supplemental Figure S. The three-step 4DM reaction, each step requires two reducing equivalents from ADPH
CHM 292 Final Exam Answer Key
 CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
Paul A. Ullucci, Bruce Bailey, Ian N. Acworth, Christopher Crafts, Marc Plante Thermo Fisher Scientific, Chelmsford, MA, USA
 The Spectro-Electro Array: A Novel Platform for the Measurement of Secondary Metabolites in Botanicals, Supplements, Foods and Beverages - Part : Theory and Concepts Paul A. Ullucci, Bruce Bailey, Ian
The Spectro-Electro Array: A Novel Platform for the Measurement of Secondary Metabolites in Botanicals, Supplements, Foods and Beverages - Part : Theory and Concepts Paul A. Ullucci, Bruce Bailey, Ian
Profiling of Diferulates (Plant Cell Wall Cross- Linkers) Using Ultrahigh-performance Liquid. Chromatography-Tandem Mass Spectrometry
 Supporting Information for: Profiling of Diferulates (Plant Cell Wall Cross- Linkers) Using Ultrahigh-performance Liquid Chromatography-Tandem Mass Spectrometry Ramin Vismeh a,b, Fachuang Lu c,d, Shishir
Supporting Information for: Profiling of Diferulates (Plant Cell Wall Cross- Linkers) Using Ultrahigh-performance Liquid Chromatography-Tandem Mass Spectrometry Ramin Vismeh a,b, Fachuang Lu c,d, Shishir
SUPPLEMENTARY INFORMATION
 doi:1.138/nature1737 Supplementary Table 1 variant Description FSEC - 2B12 a FSEC - 6A1 a K d (leucine) c Leucine uptake e K (wild-type like) K (Y18F) K (TS) K (TSY) K288A mutant, lipid facing side chain
doi:1.138/nature1737 Supplementary Table 1 variant Description FSEC - 2B12 a FSEC - 6A1 a K d (leucine) c Leucine uptake e K (wild-type like) K (Y18F) K (TS) K (TSY) K288A mutant, lipid facing side chain
Sunhats for plants. How plants detect dangerous ultraviolet rays
 Sunhats for plants How plants detect dangerous ultraviolet rays Anyone who has ever suffered sunburn will know about the effects of too much ultraviolet (UV) radiation, in particular UV-B (from 280-315
Sunhats for plants How plants detect dangerous ultraviolet rays Anyone who has ever suffered sunburn will know about the effects of too much ultraviolet (UV) radiation, in particular UV-B (from 280-315
The Phenylpropanoid Pathway in Arabidopsis
 The Phenylpropanoid Pathway in Arabidopsis Author(s): Christopher M. Fraser and Clint Chapple Source: The Arabidopsis Book, Published By: The American Society of Plant Biologists https://doi.org/10.1199/tab.0152
The Phenylpropanoid Pathway in Arabidopsis Author(s): Christopher M. Fraser and Clint Chapple Source: The Arabidopsis Book, Published By: The American Society of Plant Biologists https://doi.org/10.1199/tab.0152
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor
 LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2
 Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry s Organic Chemistry, 7 th edition The
Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry s Organic Chemistry, 7 th edition The
Applying MRM Spectrum Mode and Library Searching for Enhanced Reporting Confidence in Routine Pesticide Residue Analysis
 PO-CON1768E Applying MRM Spectrum Mode and Library Searching for Enhanced Reporting Confidence in Routine Pesticide ASMS 2017 TP-194 David Baker 1, Christopher Titman 1, Neil Loftus 1, Jonathan Horner
PO-CON1768E Applying MRM Spectrum Mode and Library Searching for Enhanced Reporting Confidence in Routine Pesticide ASMS 2017 TP-194 David Baker 1, Christopher Titman 1, Neil Loftus 1, Jonathan Horner
23-. Shoot and root development depend on ratio of IAA/CK
 Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Actions of auxin. Hormones: communicating with chemicals History: Discovery of a growth substance (hormone- auxin)
 Hormones: communicating with chemicals History- discovery of plant hormone. Auxin Concepts of hormones Auxin levels are regulated by synthesis/degradation, transport, compartmentation, conjugation. Polar
Hormones: communicating with chemicals History- discovery of plant hormone. Auxin Concepts of hormones Auxin levels are regulated by synthesis/degradation, transport, compartmentation, conjugation. Polar
Content : Properties of amino acids.. Separation and Analysis of Amino Acids
 قسم الكيمياء الحيوية.دولت على سالمه د استاذ الكيمياء الحيوية ٢٠١٥-٢٠١٤ المحاضرة الثانية Content : Properties of amino acids.. Separation and Analysis of Amino Acids 2 -3 A. Physical properties 1. Solubility:
قسم الكيمياء الحيوية.دولت على سالمه د استاذ الكيمياء الحيوية ٢٠١٥-٢٠١٤ المحاضرة الثانية Content : Properties of amino acids.. Separation and Analysis of Amino Acids 2 -3 A. Physical properties 1. Solubility:
Scission of Dinitrogen by a Molybdenum(III) Xylidene Complex. CHM 5.33 Fall 2005
 Scission of Dinitrogen by a Molybdenum(III) Xylidene Complex CHM 5.33 Fall 2005 Introduction The experiment is based on research performed in the laboratory of Professor Cummins during the early 90 s.
Scission of Dinitrogen by a Molybdenum(III) Xylidene Complex CHM 5.33 Fall 2005 Introduction The experiment is based on research performed in the laboratory of Professor Cummins during the early 90 s.
A rapid and highly selective colorimetric method for direct detection of tryptophan in proteins via DMSO acceleration
 A rapid and highly selective colorimetric method for direct detection of tryptophan in proteins via DMSO acceleration Yanyan Huang, Shaoxiang Xiong, Guoquan Liu, Rui Zhao Beijing National Laboratory for
A rapid and highly selective colorimetric method for direct detection of tryptophan in proteins via DMSO acceleration Yanyan Huang, Shaoxiang Xiong, Guoquan Liu, Rui Zhao Beijing National Laboratory for
Supplementary Figure S1. Amino acid alignment of selected monocot FT-like and TFL-like sequences. Sequences were aligned using ClustalW and analyzed
 Supplementary Figure S1. Amino acid alignment of selected monocot FT-like and TFL-like sequences. Sequences were aligned using ClustalW and analyzed using the Geneious software. Accession numbers of the
Supplementary Figure S1. Amino acid alignment of selected monocot FT-like and TFL-like sequences. Sequences were aligned using ClustalW and analyzed using the Geneious software. Accession numbers of the
NH 2. Biochemistry I, Fall Term Sept 9, Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter
 Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
1014NSC Fundamentals of Biochemistry Semester Summary
 1014NSC Fundamentals of Biochemistry Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Water & ph - Protein Diversity - Nucleic Acids - DNA Replication - Transcription
1014NSC Fundamentals of Biochemistry Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Water & ph - Protein Diversity - Nucleic Acids - DNA Replication - Transcription
Dental Biochemistry EXAM I
 Dental Biochemistry EXAM I August 29, 2005 In the reaction below: CH 3 -CH 2 OH -~ ethanol CH 3 -CHO acetaldehyde A. acetoacetate is being produced B. ethanol is being oxidized to acetaldehyde C. acetaldehyde
Dental Biochemistry EXAM I August 29, 2005 In the reaction below: CH 3 -CH 2 OH -~ ethanol CH 3 -CHO acetaldehyde A. acetoacetate is being produced B. ethanol is being oxidized to acetaldehyde C. acetaldehyde
Evaluating the performance of low-lignin transgenic bioenergy feedstocks in the field
 University of Tennessee, Knoxville Trace: Tennessee Research and Creative Exchange Masters Theses Graduate School 5-2014 Evaluating the performance of low-lignin transgenic bioenergy feedstocks in the
University of Tennessee, Knoxville Trace: Tennessee Research and Creative Exchange Masters Theses Graduate School 5-2014 Evaluating the performance of low-lignin transgenic bioenergy feedstocks in the
Supporting Information. Rec. Nat. Prod. 7:4 (2013)
 Supporting Information Rec. Nat. Prod. 7:4 (213) 312-319 Phenolic Compounds of Pinus brutia Ten.: Chemical Investigation and Quantitative Analysis Using an Ultra-Performance Liquid Chromatography Tandem
Supporting Information Rec. Nat. Prod. 7:4 (213) 312-319 Phenolic Compounds of Pinus brutia Ten.: Chemical Investigation and Quantitative Analysis Using an Ultra-Performance Liquid Chromatography Tandem
WADA Technical Document TD2015IDCR
 MINIMUM CRITERIA FOR CHROMATOGRAPHIC-MASS SPECTROMETRIC CONFIRMATION OF THE IDENTITY OF ANALYTES FOR DOPING CONTROL PURPOSES. The ability of a method to identify an analyte is a function of the entire
MINIMUM CRITERIA FOR CHROMATOGRAPHIC-MASS SPECTROMETRIC CONFIRMATION OF THE IDENTITY OF ANALYTES FOR DOPING CONTROL PURPOSES. The ability of a method to identify an analyte is a function of the entire
NB-DNJ/GCase-pH 7.4 NB-DNJ+/GCase-pH 7.4 NB-DNJ+/GCase-pH 4.5
 SUPPLEMENTARY TABLES Suppl. Table 1. Protonation states at ph 7.4 and 4.5. Protonation states of titratable residues in GCase at ph 7.4 and 4.5. Histidine: HID, H at δ-nitrogen; HIE, H at ε-nitrogen; HIP,
SUPPLEMENTARY TABLES Suppl. Table 1. Protonation states at ph 7.4 and 4.5. Protonation states of titratable residues in GCase at ph 7.4 and 4.5. Histidine: HID, H at δ-nitrogen; HIE, H at ε-nitrogen; HIP,
mau 200 ph min
 mau ph - 6 8 TAKE A LOOK INSIDE TWIN Technology TWIN (Two-In-One ) Technology is what gives Gei its superior performance edge. During the final stage of silica manufacturing, a unique silica-organic layer
mau ph - 6 8 TAKE A LOOK INSIDE TWIN Technology TWIN (Two-In-One ) Technology is what gives Gei its superior performance edge. During the final stage of silica manufacturing, a unique silica-organic layer
C Glucose Qualitative Flux Analysis in HepG2 cells
 Application Note Metabolomics 13 C Glucose Qualitative Flux Analysis in HepG2 cells Using an Agilent 6546 LC/Q-TOF and VistaFlux Authors Siriluck Wattanavanitchakorn and Sarawut Jitrapakdee Department
Application Note Metabolomics 13 C Glucose Qualitative Flux Analysis in HepG2 cells Using an Agilent 6546 LC/Q-TOF and VistaFlux Authors Siriluck Wattanavanitchakorn and Sarawut Jitrapakdee Department
Title Allantoin by Inosine in Nutrient So. Author(s) Toshihiro; Yokoi, Daisuke; Osaki, M
 Title Rice Root Growth with Increasing in Allantoin by Inosine in Nutrient So Author(s) Tokuhisa, Dai; Okazaki, Keiki; Shin Toshihiro; Yokoi, Daisuke; Osaki, M Citation The Proceedings of the Internationa
Title Rice Root Growth with Increasing in Allantoin by Inosine in Nutrient So Author(s) Tokuhisa, Dai; Okazaki, Keiki; Shin Toshihiro; Yokoi, Daisuke; Osaki, M Citation The Proceedings of the Internationa
DIA-Umpire: comprehensive computational framework for data independent acquisition proteomics
 DIA-Umpire: comprehensive computational framework for data independent acquisition proteomics Chih-Chiang Tsou 1,2, Dmitry Avtonomov 2, Brett Larsen 3, Monika Tucholska 3, Hyungwon Choi 4 Anne-Claude Gingras
DIA-Umpire: comprehensive computational framework for data independent acquisition proteomics Chih-Chiang Tsou 1,2, Dmitry Avtonomov 2, Brett Larsen 3, Monika Tucholska 3, Hyungwon Choi 4 Anne-Claude Gingras
Analysis of Polyphenols in Saffron Petal Extracts with UHPLC/UV/MS
 APPLICATION NOTE Liquid Chromatography/ Mass Spectrometry Authors: Roberto Troiano Milan, Italy Wilhad M. Reuter Shelton, CT Analysis of Polyphenols in Saffron Petal Extracts with UHPLC/UV/MS Introduction
APPLICATION NOTE Liquid Chromatography/ Mass Spectrometry Authors: Roberto Troiano Milan, Italy Wilhad M. Reuter Shelton, CT Analysis of Polyphenols in Saffron Petal Extracts with UHPLC/UV/MS Introduction
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. The asymmetric unit in para-iodio-phenylalanine crystal. The 50% probability ellipsoid representation was prepared using the Mercury Software. Colors are as
Supplementary Figures Supplementary Figure 1. The asymmetric unit in para-iodio-phenylalanine crystal. The 50% probability ellipsoid representation was prepared using the Mercury Software. Colors are as
Supplemental Data. Chen and Thelen (2010). Plant Cell /tpc
 Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. HSP21 expression in 35S:HSP21 and hsp21 knockdown plants. (a) Since no T- DNA insertion line for HSP21 is available in the publicly available T-DNA collections,
Supplementary Figure 1 Supplementary Figure 1. HSP21 expression in 35S:HSP21 and hsp21 knockdown plants. (a) Since no T- DNA insertion line for HSP21 is available in the publicly available T-DNA collections,
Experiment 8 Optical Isomers. In this experiment you will be given the opportunity to see the 3-dimensional aspects of
 Experiment 8 Optical Isomers In this experiment you will be given the opportunity to see the 3-dimensional aspects of stereochemistry and optical isomers. Previously in class you were exposed to the concept
Experiment 8 Optical Isomers In this experiment you will be given the opportunity to see the 3-dimensional aspects of stereochemistry and optical isomers. Previously in class you were exposed to the concept
Method Development in Solid Phase Extraction using Non-Polar ISOLUTE SPE Columns for the Extraction of Aqueous Samples
 Technical Note 101 Method Development in Solid Phase Extraction using Non-Polar ISOLUTE SPE Columns for the Extraction of Aqueous Samples This technical note includes by specific information on the extraction
Technical Note 101 Method Development in Solid Phase Extraction using Non-Polar ISOLUTE SPE Columns for the Extraction of Aqueous Samples This technical note includes by specific information on the extraction
Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies
 Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies The hydrogen stretch region (3600 2500 cm 1 ). Absorption in this region is associated with the stretching vibration of hydrogen
Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies The hydrogen stretch region (3600 2500 cm 1 ). Absorption in this region is associated with the stretching vibration of hydrogen
Accelerating the Metabolite Identification Process Using High Resolution Q-TOF Data and Mass-MetaSite Software
 Accelerating the Metabolite Identification Process Using High Resolution Q-TOF Data and Mass-MetaSite Software Application ote Drug discovery and development: Metabolite Identifi cation Authors Yuqin Dai,
Accelerating the Metabolite Identification Process Using High Resolution Q-TOF Data and Mass-MetaSite Software Application ote Drug discovery and development: Metabolite Identifi cation Authors Yuqin Dai,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10244 a O07391_MYCAV/127-243 NLPC_HAEIN/80-181 SPR_SHIFL/79-183 P74160_SYNY3/112-245 O24914_HELPY/301-437 Q51835_PORGI/68-178 DPP6_BACSH/163-263 YKFC_BACSU/185-292 YDHO_ECOLI/153-263
doi:10.1038/nature10244 a O07391_MYCAV/127-243 NLPC_HAEIN/80-181 SPR_SHIFL/79-183 P74160_SYNY3/112-245 O24914_HELPY/301-437 Q51835_PORGI/68-178 DPP6_BACSH/163-263 YKFC_BACSU/185-292 YDHO_ECOLI/153-263
A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of
 Questions with Answers- Amino Acids & Peptides A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of 5.0-6.5 (Questions 1-4) 1. Which
Questions with Answers- Amino Acids & Peptides A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of 5.0-6.5 (Questions 1-4) 1. Which
Growth and development of Arabidopsis thaliana under single-wavelength red
 1 Supplementary Information 2 3 4 Growth and development of Arabidopsis thaliana under single-wavelength red and blue laser light 5 6 7 8 Authors Amanda Ooi 1 *, Aloysius Wong 1 *, Tien Khee Ng 2, Claudius
1 Supplementary Information 2 3 4 Growth and development of Arabidopsis thaliana under single-wavelength red and blue laser light 5 6 7 8 Authors Amanda Ooi 1 *, Aloysius Wong 1 *, Tien Khee Ng 2, Claudius
Appendix II- Bioanalytical Method Development and Validation
 A2. Bioanalytical method development 1. Optimization of chromatographic conditions Method development and optimization of chromatographic parameters is of utmost important for validating a method in biological
A2. Bioanalytical method development 1. Optimization of chromatographic conditions Method development and optimization of chromatographic parameters is of utmost important for validating a method in biological
Water. Water participates in H-bonding with biomolecules.
 Water Most biochemical reactions occur in an aqueous environment. Water is highly polar because of its bent geometry. Water is highly cohesive because of intermolecular hydrogen bonding. Water participates
Water Most biochemical reactions occur in an aqueous environment. Water is highly polar because of its bent geometry. Water is highly cohesive because of intermolecular hydrogen bonding. Water participates
Supplemental Data. Wang et al. (2014). Plant Cell /tpc
 Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
CHEMISTRY 112A FALL 2015 EXAM 1 SEPTEMBER 27, 2016 NAME- WRITE BIG STUDENT ID: SECTION AND/OR GSI IF YOU ARE IN THE LABORATORY COURSE:
 CHEMISTRY 112A FALL 2015 EXAM 1 SEPTEMBER 27, 2016 NAME- WRITE BIG STUDENT ID: SECTIN AND/R GSI IF YU ARE IN THE LABRATRY CURSE: You will have 75 minutes in which to work. BE NEAT! Non-legible structure
CHEMISTRY 112A FALL 2015 EXAM 1 SEPTEMBER 27, 2016 NAME- WRITE BIG STUDENT ID: SECTIN AND/R GSI IF YU ARE IN THE LABRATRY CURSE: You will have 75 minutes in which to work. BE NEAT! Non-legible structure
Experiment 1: Thin Layer Chromatography
 Experiment 1: Thin Layer Chromatography Part A: understanding R f values Part B: R f values & solvent polarity Part C: R f values & compound functionality Part D: identification of commercial food dye
Experiment 1: Thin Layer Chromatography Part A: understanding R f values Part B: R f values & solvent polarity Part C: R f values & compound functionality Part D: identification of commercial food dye
Metabolic Phenotyping Using Atmospheric Pressure Gas Chromatography-MS
 Metabolic Phenotyping Using tmospheric Pressure Gas hromatography-ms Vladimir Shulaev, 2 Ghaste Manoj, 2,3 Steven Lai, 1 arolina Salazar, 2 Nobuhiro Suzuki, 2 Janna rossley, 2 Feroza Kaneez oudhury, 2
Metabolic Phenotyping Using tmospheric Pressure Gas hromatography-ms Vladimir Shulaev, 2 Ghaste Manoj, 2,3 Steven Lai, 1 arolina Salazar, 2 Nobuhiro Suzuki, 2 Janna rossley, 2 Feroza Kaneez oudhury, 2
Questions for Biology IIB (SS 2006) Wilhelm Gruissem
 Questions for Biology IIB (SS 2006) Plant biology Wilhelm Gruissem The questions for my part of Biology IIB, Plant Biology, are provided for self-study and as material for the exam. Please note that the
Questions for Biology IIB (SS 2006) Plant biology Wilhelm Gruissem The questions for my part of Biology IIB, Plant Biology, are provided for self-study and as material for the exam. Please note that the
CHERRY HILL TUITION OCR A CHEMISTRY A2 PAPER 19 MARK SCHEME ANNOTATIONS MUST BE USED CH 3 CH 3 CH 3 H + correct products
 ERRY ILL TUITIN R A EMISTRY A2 PAPER 19 MARK SEME ALLW Kekulé structures throughout 1 (a) ANNTATINS MUST BE USED ALLW skeletal ALLW + + N 2 R N 2 ALLW 1st curly arrow from the ring R from within the ring
ERRY ILL TUITIN R A EMISTRY A2 PAPER 19 MARK SEME ALLW Kekulé structures throughout 1 (a) ANNTATINS MUST BE USED ALLW skeletal ALLW + + N 2 R N 2 ALLW 1st curly arrow from the ring R from within the ring
Supplementary Figure 1. Phenotype of the HI strain.
 Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Phenyl-Hexyl. UHPLC Columns. Alternate, complementary selectivity to C18 and C8 bonded phases
 Phenyl-Hexyl UHPLC Columns Alternate, complementary selectivity to C8 and C8 bonded phases Particularly recommended for compounds containing aromatic groups Excellent bonded phase stability for durable,
Phenyl-Hexyl UHPLC Columns Alternate, complementary selectivity to C8 and C8 bonded phases Particularly recommended for compounds containing aromatic groups Excellent bonded phase stability for durable,
Analysis of Serum 17-Hydroxyprogesterone, Androstenedione, and Cortisol by UPLC-MS/MS for Clinical Research
 Analysis of Serum 17-Hydroxyprogesterone, Androstenedione, and Cortisol by UPLC-MS/MS for Clinical Research Heather A Brown, 1 Claudia Rossi, 2 and Lisa J Calton 1 1 Waters Corporation, Wilmslow, UK 2
Analysis of Serum 17-Hydroxyprogesterone, Androstenedione, and Cortisol by UPLC-MS/MS for Clinical Research Heather A Brown, 1 Claudia Rossi, 2 and Lisa J Calton 1 1 Waters Corporation, Wilmslow, UK 2
Chemistry 201. MW 12pm 1:15pm Examination #1 July 22 nd Bronco ID. Question Score Possible Points. 1 (10pts) 2 (24pts) 3 (14pts) 4...
 Chemistry 201 MW 12pm 1:15pm Examination #1 July 22 nd 2015 Name Bronco ID. Question Score Possible Points 1 (10pts) 2 (24pts) 3 (14pts) 4... (22pts) 5 (30pts). Total (100pts) 1. Read each question carefully.
Chemistry 201 MW 12pm 1:15pm Examination #1 July 22 nd 2015 Name Bronco ID. Question Score Possible Points 1 (10pts) 2 (24pts) 3 (14pts) 4... (22pts) 5 (30pts). Total (100pts) 1. Read each question carefully.
Identifying Interaction Hot Spots with SuperStar
 Identifying Interaction Hot Spots with SuperStar Version 1.0 November 2017 Table of Contents Identifying Interaction Hot Spots with SuperStar... 2 Case Study... 3 Introduction... 3 Generate SuperStar Maps
Identifying Interaction Hot Spots with SuperStar Version 1.0 November 2017 Table of Contents Identifying Interaction Hot Spots with SuperStar... 2 Case Study... 3 Introduction... 3 Generate SuperStar Maps
Nature Methods: doi: /nmeth Supplementary Figure 1. Fragment indexing allows efficient spectra similarity comparisons.
 Supplementary Figure 1 Fragment indexing allows efficient spectra similarity comparisons. The cost and efficiency of spectra similarity calculations can be approximated by the number of fragment comparisons
Supplementary Figure 1 Fragment indexing allows efficient spectra similarity comparisons. The cost and efficiency of spectra similarity calculations can be approximated by the number of fragment comparisons
Nature Genetics: doi: /ng Supplementary Figure 1. The phenotypes of PI , BR121, and Harosoy under short-day conditions.
 Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
An alcohol is a compound obtained by substituting a hydoxyl group ( OH) for an H atom on a carbon atom of a hydrocarbon group.
 Derivatives of Hydrocarbons A functional group is a reactive portion of a molecule that undergoes predictable reactions. All other organic compounds can be considered as derivatives of hydrocarbons (i.e.,
Derivatives of Hydrocarbons A functional group is a reactive portion of a molecule that undergoes predictable reactions. All other organic compounds can be considered as derivatives of hydrocarbons (i.e.,
Useful Propagation Terms. Propagation The application of specific biological principles and concepts in the multiplication of plants.
 Useful Propagation Terms Propagation The application of specific biological principles and concepts in the multiplication of plants. Adventitious Typically describes new organs such as roots that develop
Useful Propagation Terms Propagation The application of specific biological principles and concepts in the multiplication of plants. Adventitious Typically describes new organs such as roots that develop
HW #5: 16.20, 16.28, 16.30, 16.32, 16.40, 16.44, 16.46, 16.52, 16.60, 16.62, 16.64, 16.68
 hemistry 131 Lecture 10: Aldehydes and Ketones: Structure, Nomenclature, Physical Properties, and Reactivity hapter 16 in McMurry, Ballantine, et. al. 7 th edition W #5: 16.20, 16.28, 16.30, 16.32, 16.40,
hemistry 131 Lecture 10: Aldehydes and Ketones: Structure, Nomenclature, Physical Properties, and Reactivity hapter 16 in McMurry, Ballantine, et. al. 7 th edition W #5: 16.20, 16.28, 16.30, 16.32, 16.40,
Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers
 Chapter Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers Ch 1-Structure and bonding Ch 2-Polar covalent bonds: Acids and bases McMurry, J. (2004) Organic Chemistry 6 th Edition
Chapter Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers Ch 1-Structure and bonding Ch 2-Polar covalent bonds: Acids and bases McMurry, J. (2004) Organic Chemistry 6 th Edition
THE CRYSTAL STRUCTURE OF THE SGT1-SKP1 COMPLEX: THE LINK BETWEEN
 THE CRYSTAL STRUCTURE OF THE SGT1-SKP1 COMPLEX: THE LINK BETWEEN HSP90 AND BOTH SCF E3 UBIQUITIN LIGASES AND KINETOCHORES Oliver Willhoft, Richard Kerr, Dipali Patel, Wenjuan Zhang, Caezar Al-Jassar, Tina
THE CRYSTAL STRUCTURE OF THE SGT1-SKP1 COMPLEX: THE LINK BETWEEN HSP90 AND BOTH SCF E3 UBIQUITIN LIGASES AND KINETOCHORES Oliver Willhoft, Richard Kerr, Dipali Patel, Wenjuan Zhang, Caezar Al-Jassar, Tina
Page 2. The tripeptide shown is formed from the amino acids alanine, threonine and lysine.
 Q1.(a) The tripeptide shown is formed from the amino acids alanine, threonine and lysine. Draw a separate circle around each of the asymmetric carbon atoms in the tripeptide. Draw the zwitterion of alanine.
Q1.(a) The tripeptide shown is formed from the amino acids alanine, threonine and lysine. Draw a separate circle around each of the asymmetric carbon atoms in the tripeptide. Draw the zwitterion of alanine.
A rapid identification of anthocyanins in various plants from Burkina Faso by HPTLC/MS
 A rapid identification of anthocyanins in various plants from Burkina Faso by HPTLC/MS Presented by Rosella SPINA July 2-4, 2014 Lyon Background- Anthocyanidins chemistry A B Background- Anthocyanins >
A rapid identification of anthocyanins in various plants from Burkina Faso by HPTLC/MS Presented by Rosella SPINA July 2-4, 2014 Lyon Background- Anthocyanidins chemistry A B Background- Anthocyanins >
Packings for HPLC. Packings for HPLC
 Summary of packings for HPLC In analytical HPLC, packings with particle sizes of 3 to 10 µm are preferred. For preparative separation tasks, also particles with diameters larger than 10 µm are applied.
Summary of packings for HPLC In analytical HPLC, packings with particle sizes of 3 to 10 µm are preferred. For preparative separation tasks, also particles with diameters larger than 10 µm are applied.
Supplemental Data. Fernández-Calvo et al. Plant Cell. (2011) /tpc
 Supplemental Data. Fernández-Calvo et al. Plant Cell. (2011). 10.1105/tpc.110.080788 Supplemental Figure S1. Phylogenetic tree of MYC2-related proteins from Arabidopsis and other plants. Phenogram representation
Supplemental Data. Fernández-Calvo et al. Plant Cell. (2011). 10.1105/tpc.110.080788 Supplemental Figure S1. Phylogenetic tree of MYC2-related proteins from Arabidopsis and other plants. Phenogram representation
Medicinal Chemistry/ CHEM 458/658 Chapter 4- Computer-Aided Drug Design
 Medicinal Chemistry/ CHEM 458/658 Chapter 4- Computer-Aided Drug Design Bela Torok Department of Chemistry University of Massachusetts Boston Boston, MA 1 Computer Aided Drug Design - Introduction Development
Medicinal Chemistry/ CHEM 458/658 Chapter 4- Computer-Aided Drug Design Bela Torok Department of Chemistry University of Massachusetts Boston Boston, MA 1 Computer Aided Drug Design - Introduction Development
Practice Midterm Exam 200 points total 75 minutes Multiple Choice (3 pts each 30 pts total) Mark your answers in the space to the left:
 MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
Final Exam. Chem 3B, Fall 2016 Monday, Dec 12, pm. Name Answer Key. Student ID. If you are making up an incomplete, list the semester here:
 Final Exam Chem 3B, Fall 2016 Monday, Dec 12, 2016 3-6 pm ame Answer Key Student ID If you are making up an incomplete, list the semester here: You have 180 minutes to complete this exam. Please provide
Final Exam Chem 3B, Fall 2016 Monday, Dec 12, 2016 3-6 pm ame Answer Key Student ID If you are making up an incomplete, list the semester here: You have 180 minutes to complete this exam. Please provide
NAME. EXAM I I. / 36 September 25, 2000 Biochemistry I II. / 26 BICH421/621 III. / 38 TOTAL /100
 EXAM I I. / 6 September 25, 2000 Biochemistry I II. / 26 BIH421/621 III. / 8 TOTAL /100 I. MULTIPLE HOIE (6 points) hoose the BEST answer to the question by circling the appropriate letter. 1. An amino
EXAM I I. / 6 September 25, 2000 Biochemistry I II. / 26 BIH421/621 III. / 8 TOTAL /100 I. MULTIPLE HOIE (6 points) hoose the BEST answer to the question by circling the appropriate letter. 1. An amino
