Chemistry and Atoms! 8 th grade history information to help you understand the background of how our knowledge grew through the years.
|
|
|
- Ronald Murphy
- 5 years ago
- Views:
Transcription
1 Chemistry and Atoms! 8 th grade history information to help you understand the background of how our knowledge grew through the years.
2 Chemistry lies at the roots of civilization! Chemical reactions are the strange transformations that reveal matter s hidden properties. The first chemical reaction observed by early man was FIRE!
3 This (fire) was important because it revealed hidden properties of matter. Experiments with fire led to other chemical reactions such as cooking. Besides food, early man also began to cook other things. One material he cooked was rocks!
4 A strange thing happened when green crumbly rocks were melted They turned into an orange liquid that cooled into shiny metallic copper.
5 This discovery of cooking rocks led to other discoveries. Early man also was able to melt red rocks into iron, bake mud into bricks, and saute fat and ashes into soap.
6 Different theories have been put forth about the secrets of matter. Greek intellectuals came up with different theories. The Atomists, led by Democritus thought matter was made of tiny INDIVISIBLE particles.
7 Aristotle believed there were really four elements, air, earth, fire and water from which all else was composed.
8 The most influential idea was Aristotle s. If everything was a mixture of 4 elements.... then it must be possible to turn anything into anything by just tweaking the ingredients.
9 Chemistry spread across Medieval Europe under the name ALCHEMY. Early chemists tried to make gold from other substances including urine!!
10 In medieval times, alchemists used to believe that a certain substance held special magical powers. These special magical powers gave the ability to turn normal metals into gold or silver. This certain substance was known as the philosopher s stone.
11 Some looked for the universal solvent failing to realize that water was it. Others looked for the Elixir Vitae, a substance to extend life. Although alchemists did not discover the philosopher s stone, they did accomplish a lot. They perfected distillation, filtration, and titration.
12 They also advanced glassmaking, metallurgy, explosives and corrosives:
13 Alchemist lab techniques did miss one big thing they failed to collect gases! This meant they could never account for the ingredients or products of chemical reactions!!
14 The study of gases began in the 1600 s with investigations into the effects of air pressure. Otto Von Guericke performed an experiment where he sealed two metal hemispheres together. He then inserted a valve and pumped the air out of the interior. When the sphere closed it formed a vacuum which two horses could not even pull apart!
15 However, when Otto Von Guericke let the air back in, the two hemispheres separated easily In England during the 1770 s, while Joseph Priestly experimented with airs, in France, Antoine-Laurent Lavoisier was also experimenting with gases. His experiment proved that air was a mixture of two different gases which meant air was NOT an element. The French Chemist named it oxygen. He was also known as the Father of modern chemistry.
16 Lavoisier made chemistry an independent science which earned him the title of Father of Modern Chemistry. Such discoveries led John Dalton to revive matter s atomic theory. He reasoned that each element was made of tiny indivisible atoms.
17 Dalton thought that the atoms of any one element were all alike, but different from the atoms of other elements. Compound substances were composed of fixed groupings of atoms called molecules. A molecule is the smallest amount of a chemical substance that can exist. Essentially, a compound is a type of molecule. A molecule can be made up of two or more atoms of the same element or two or more atoms of different elements. However, compounds are molecules that are made up of atoms of different molecule.
18 By the 1860 s many new elements had been discovered. Chemists made an important observation. It appeared that some elements were more alike than others. It was almost as if elements had families like people!
19 In 1869, A Russian chemist named Mendeleev decided to arrange the elements in order of increasing atomic mass. The result was a table with the elements arranged in rows.
20 The elements showed a periodic or repeating pattern. Each column contained chemically similar elements. With this table Mendeleev was able to successfully predict new elements that would fill gaps on his table. However, an important question still lingered, how to explain chemistry. It seemed the secret to chemistry required understanding of the: ATOM!
21 Now we will watch three clips that have to do with our learning in the chemistry and atom world. 1. Hennig Brand and the discovery of the element phosphorus. 2. A cartoon about the Atomic Theory TED Ed 3. Bill Nye and the greatest discoveries in chemistry All great stuff. Sit up and enjoy your learning!! Remember no notes BUT listen well.
Chemistry and Atoms! 8 th grade history information to help you understand the background of how our knowledge grew through the years.
 Chemistry and Atoms! 8 th grade history information to help you understand the background of how our knowledge grew through the years. Chemistry lies at the roots of civilization! Chemical reactions are
Chemistry and Atoms! 8 th grade history information to help you understand the background of how our knowledge grew through the years. Chemistry lies at the roots of civilization! Chemical reactions are
HISTORY OF CHEMISTRY BY SARA C. MIGUEL M. JESSICA S.
 HISTORY OF CHEMISTRY BY SARA C. MIGUEL M. JESSICA S. A Little Info! Chemistry is a branch of science Dates back to prehistoric times ( looong time ago ) Is separated into four different time periods Prehistoric
HISTORY OF CHEMISTRY BY SARA C. MIGUEL M. JESSICA S. A Little Info! Chemistry is a branch of science Dates back to prehistoric times ( looong time ago ) Is separated into four different time periods Prehistoric
Atoms, Elements, and the Periodic Table
 chapter 00 3 3 Atoms, Elements, and the Periodic Table section 1 Structure of Matter Before You Read Take a deep breath. What fills your lungs? Can you see it or hold it in your hand? What You ll Learn
chapter 00 3 3 Atoms, Elements, and the Periodic Table section 1 Structure of Matter Before You Read Take a deep breath. What fills your lungs? Can you see it or hold it in your hand? What You ll Learn
The Story of the Atom. A history of atomic theory over many years
 The Story of the Atom A history of atomic theory over many years Democritus Many years ago, between 460BC and 370BC the Greek philosophers wondered what we were made of. Leucippus and Democritus came up
The Story of the Atom A history of atomic theory over many years Democritus Many years ago, between 460BC and 370BC the Greek philosophers wondered what we were made of. Leucippus and Democritus came up
Vocabulary atom atomos Dalton's atomic theory law of constant composition law of definite proportions law of multiple proportions matter.
 1.3 Early Atomic Theory Lesson Objectives The student will: define matter and explain how it is composed of building blocks known as atoms. give a short history of how the concept of the atom developed.
1.3 Early Atomic Theory Lesson Objectives The student will: define matter and explain how it is composed of building blocks known as atoms. give a short history of how the concept of the atom developed.
Atomic Structure and The Periodic Table. Unit 3
 Atomic Structure and The Periodic Table Unit 3 Lesson 1: Atoms Unit 5: Atomic Structure & The Periodic Table Atoms How small can things get? If you break a stone wall into smaller and smaller pieces, you
Atomic Structure and The Periodic Table Unit 3 Lesson 1: Atoms Unit 5: Atomic Structure & The Periodic Table Atoms How small can things get? If you break a stone wall into smaller and smaller pieces, you
CHAPTER 3. Chemical Foundations
 CHAPTER 3 Chemical Foundations 3.1 THE ELEMENTS 118 elements in 92 occur naturally, the rest are synthesized All matter in the universe can be chemically broken down into elements Compounds are made by
CHAPTER 3 Chemical Foundations 3.1 THE ELEMENTS 118 elements in 92 occur naturally, the rest are synthesized All matter in the universe can be chemically broken down into elements Compounds are made by
Scientist wanted to understand how the atom looked. It was known that matter was neutral. It was known that matter had mass
 Atom Models Scientist wanted to understand how the atom looked It was known that matter was neutral It was known that matter had mass They used these ideas to come up with their models, however science
Atom Models Scientist wanted to understand how the atom looked It was known that matter was neutral It was known that matter had mass They used these ideas to come up with their models, however science
Atomic Theory. Introducing the Atomic Theory:
 Atomic Theory Chemistry is the science of matter. Matter is made up of things called atoms, elements, and molecules. But have you ever wondered if atoms and molecules are real? Would you be surprised to
Atomic Theory Chemistry is the science of matter. Matter is made up of things called atoms, elements, and molecules. But have you ever wondered if atoms and molecules are real? Would you be surprised to
Review Worksheet. Symbol # protons # electrons #neutrons. Silver. 4. Draw a Bohr Diagram for: a) Carbon b) Chlorine c) Magnesuim
 Review Worksheet 1. List the 3 subatomic particles; the charge that each has; and the mass of each particle in the space below Subatomic Particle Charge Mass of Particle 2. Complete the following table
Review Worksheet 1. List the 3 subatomic particles; the charge that each has; and the mass of each particle in the space below Subatomic Particle Charge Mass of Particle 2. Complete the following table
Simple Schooling Atomic Theory 2011 The Simple Homeschool
 1 ATOMIC THEORY It sounds so mysterious doesn t it? Atomic theory conjures up images of secret science in the 1940 s, the atom bomb, and explosions! But the truth is, atomic theory is simply the internal
1 ATOMIC THEORY It sounds so mysterious doesn t it? Atomic theory conjures up images of secret science in the 1940 s, the atom bomb, and explosions! But the truth is, atomic theory is simply the internal
Lesson 1.2 Classifying Matter
 Lesson 1.2 Classifying Matter Vocabulary element atom chemical bond mixture molecule compound chemical formula What is Matter Made Of? What is matter? Why is one kind of matter different from another kind
Lesson 1.2 Classifying Matter Vocabulary element atom chemical bond mixture molecule compound chemical formula What is Matter Made Of? What is matter? Why is one kind of matter different from another kind
1.02 Scientific Method
 1.02 Scientific Method Dr. Fred O. Garces Chemistry 100 Miramar College 1 Science and the meaning of life Ever since humans had the ability to think and reason, they have been trying to answer some basic
1.02 Scientific Method Dr. Fred O. Garces Chemistry 100 Miramar College 1 Science and the meaning of life Ever since humans had the ability to think and reason, they have been trying to answer some basic
Properties of Atoms and The Periodic Table. Ch 16, pg
 Properties of Atoms and The Periodic Table Ch 16, pg. 488-506 Today s Learning Objectives Describe the 5 models of the atom. Be able to arrange the 5 models of the atom in order. Explain why the models
Properties of Atoms and The Periodic Table Ch 16, pg. 488-506 Today s Learning Objectives Describe the 5 models of the atom. Be able to arrange the 5 models of the atom in order. Explain why the models
A brief history of Chemistry. Science 9- Mr. Klasz
 A brief history of Chemistry Science 9- Mr. Klasz What is a pure substance? An element is a pure substance. A compound is made up of two or more elements which have been chemically compounded together.
A brief history of Chemistry Science 9- Mr. Klasz What is a pure substance? An element is a pure substance. A compound is made up of two or more elements which have been chemically compounded together.
The History of Atomic Theory Chapter 3--Chemistry
 The History of Atomic Theory Chapter 3--Chemistry In this lesson, we ll learn about the men whose quests for knowledge about the fundamental nature of the universe helped define our views. The atomic model
The History of Atomic Theory Chapter 3--Chemistry In this lesson, we ll learn about the men whose quests for knowledge about the fundamental nature of the universe helped define our views. The atomic model
4.1 Defining the Atom > Chapter 4 Atomic Structure. 4.1 Defining the Atom. 4.2 Structure of the Nuclear Atom. 4.3 Distinguishing Among Atoms
 Chapter 4 Atomic Structure 4.1 Defining the Atom 4.2 Structure of the Nuclear Atom 4.3 Distinguishing Among Atoms 1 Copyright Pearson Education, Inc., or its affiliates. All Rights Reserved. CHEMISTRY
Chapter 4 Atomic Structure 4.1 Defining the Atom 4.2 Structure of the Nuclear Atom 4.3 Distinguishing Among Atoms 1 Copyright Pearson Education, Inc., or its affiliates. All Rights Reserved. CHEMISTRY
Chemistry. Chemistry is. The Study of Matter Change Study Study of the Composition of Substances & the Changes They Undergo. Matter: Mass: Volume:
 Chemistry 9/4/2005 Chemistry: Chapters 1 & 2 1 Chemistry is The Study of Matter Change Study Study of the Composition of Substances & the Changes They Undergo 9/4/2005 Chemistry: Chapters 1 & 2 2 Matter:
Chemistry 9/4/2005 Chemistry: Chapters 1 & 2 1 Chemistry is The Study of Matter Change Study Study of the Composition of Substances & the Changes They Undergo 9/4/2005 Chemistry: Chapters 1 & 2 2 Matter:
Science In Action 9 - Unit 1 Matter and Chemical Change Summary of Key Concepts and Review Questions Booklet
 10. Matter can be described and organized by its physical and chemical properties Key Concepts Workplace Hazardous Materials Information System (WHMIS) and safety substances and their properties. Recognition
10. Matter can be described and organized by its physical and chemical properties Key Concepts Workplace Hazardous Materials Information System (WHMIS) and safety substances and their properties. Recognition
HIGH SCHOOL SCIENCE. Physical Science 9: Atomic Structure
 HIGH SCHOOL SCIENCE Physical Science 9: Atomic Structure WILLMAR PUBLIC SCHOOL 2013-2014 EDITION CHAPTER 9 Atomic Structure In this chapter you will: 1. Compare and contrast quarks, leptons, and bosons.
HIGH SCHOOL SCIENCE Physical Science 9: Atomic Structure WILLMAR PUBLIC SCHOOL 2013-2014 EDITION CHAPTER 9 Atomic Structure In this chapter you will: 1. Compare and contrast quarks, leptons, and bosons.
Time to develop a model
 ATOMIC THEORY ONCE UPON A TIME People have been fascinated with matter for a long time. What is matter? What is all this stuff around us made of? Can it be broken down? Are there different types of matter?
ATOMIC THEORY ONCE UPON A TIME People have been fascinated with matter for a long time. What is matter? What is all this stuff around us made of? Can it be broken down? Are there different types of matter?
Chapter 4. History of the atom. History of Atom Smallest possible piece? Atomos - not to be cut. Atoms and their structure
 Chapter 4 Atoms and their structure History of the atom Not the history of atom, but the idea of the atom. Original idea Ancient Greece (400 B.C.) Democritus and Leucippus Greek philosophers. Looked at
Chapter 4 Atoms and their structure History of the atom Not the history of atom, but the idea of the atom. Original idea Ancient Greece (400 B.C.) Democritus and Leucippus Greek philosophers. Looked at
Major upsetting discoveries: Today s Objectives/Agenda. Notice: New Unit: with Ms. V. after school Before Friday 9/22.
 Bellwork Monday 9 18 17 Tuesday 9 19 17 *This should be the last box on bellwork: 1. What are some major points in history where common knowledge was upset by a new discovery? 2. Draw what you think an
Bellwork Monday 9 18 17 Tuesday 9 19 17 *This should be the last box on bellwork: 1. What are some major points in history where common knowledge was upset by a new discovery? 2. Draw what you think an
What s the Matter? An in depth look at matter.
 What s the Matter? An in depth look at matter. What is a mixture? Examine the objects. Then sort them into at least three groups. Each item should be grouped with similar items. Think about each objects
What s the Matter? An in depth look at matter. What is a mixture? Examine the objects. Then sort them into at least three groups. Each item should be grouped with similar items. Think about each objects
UNIT 2 Atomic Structure
 UNIT 2 Atomic Structure Section 1: History & Development of Atomic Theory (Chapter 3) History of the Atom Video The Greeks Democritus World made of empty space and tiny particles ( atoms ) Thought there
UNIT 2 Atomic Structure Section 1: History & Development of Atomic Theory (Chapter 3) History of the Atom Video The Greeks Democritus World made of empty space and tiny particles ( atoms ) Thought there
Year 7 Science. 7C1: The Particle Model. PPA Challenge
 Year 7 Science 7C1: The Particle Model PPA Challenge Name: Form: Task Sheet 1 (Bronze Challenge): The Particle Model Use the words in the box to label the diagram below. This particle diagram shows the
Year 7 Science 7C1: The Particle Model PPA Challenge Name: Form: Task Sheet 1 (Bronze Challenge): The Particle Model Use the words in the box to label the diagram below. This particle diagram shows the
3.01 Understanding Atoms
 3.01 Understanding Atoms The Events Leading to the Discovery of the Building Block of Matter Dr. Fred Omega Garces Chemistry 111 Miramar College 1 3.02 Atomic Evolution Environmental Problems in our Lifetime
3.01 Understanding Atoms The Events Leading to the Discovery of the Building Block of Matter Dr. Fred Omega Garces Chemistry 111 Miramar College 1 3.02 Atomic Evolution Environmental Problems in our Lifetime
Outcome-Based Student Self Assessment
 Outcome-Based Student Self Assessment Course: Science 10F Unit: Chemistry Student Name: Outcomes & Examples List the Greek philosophers and their contribution to Chemistry. Include Democritus, Empedocles,
Outcome-Based Student Self Assessment Course: Science 10F Unit: Chemistry Student Name: Outcomes & Examples List the Greek philosophers and their contribution to Chemistry. Include Democritus, Empedocles,
Origins of the Atom. Atoms: The Building Blocks of Matter. Let s Get Ready to Rumble. Aristotle s Theory of the Atom CHAPTER 3
 Origins of the Atom CHAPTER 3 Atoms: The Building Blocks of Matter Let s Get Ready to Rumble The idea of the atom was met with great skepticism, especially among great thinkers. The most vocal critic of
Origins of the Atom CHAPTER 3 Atoms: The Building Blocks of Matter Let s Get Ready to Rumble The idea of the atom was met with great skepticism, especially among great thinkers. The most vocal critic of
Chemistry Day 7. Friday, September 7 th Monday, September 10 th, 2018
 Chemistry Day 7 Friday, September 7 th Monday, September 10 th, 2018 Do-Now Title: Ch. 13/14 Notes C 1. Write down today s FLT 2. List the three states of matter and list an example for each 3. List two
Chemistry Day 7 Friday, September 7 th Monday, September 10 th, 2018 Do-Now Title: Ch. 13/14 Notes C 1. Write down today s FLT 2. List the three states of matter and list an example for each 3. List two
4.1 Studying Atoms Ancient Greek Models of Atoms
 Studying the structure of atoms is a little like studying wind. Because you cannot see air, you must use indirect evidence to tell the direction of the wind. Atoms pose a similar problem because they are
Studying the structure of atoms is a little like studying wind. Because you cannot see air, you must use indirect evidence to tell the direction of the wind. Atoms pose a similar problem because they are
Atomic theory. Atoms: The Building Blocks of Matter
 Atomic theory Atoms: The Building Blocks of Matter First, there was Democritus Democritus was a Greek philosopher atomos He came up with the idea of the atom around 400BCE He had no evidence, he just thought
Atomic theory Atoms: The Building Blocks of Matter First, there was Democritus Democritus was a Greek philosopher atomos He came up with the idea of the atom around 400BCE He had no evidence, he just thought
Sub atomic Mass in a.m.u. Relative Position in the
 IDEAS ABOUT ATOMS In chapter one we looked briefly at the ideas of the Ancient Greeks about atoms. You will remember that the main idea involved tiny particles of matter that could not be broken down.
IDEAS ABOUT ATOMS In chapter one we looked briefly at the ideas of the Ancient Greeks about atoms. You will remember that the main idea involved tiny particles of matter that could not be broken down.
How is the periodic table useful?
 1 3 4 11 12 Organizing the Elements Lesson 3:2 2 5 6 7 8 9 10 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 72 73
1 3 4 11 12 Organizing the Elements Lesson 3:2 2 5 6 7 8 9 10 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 72 73
Atomic Origins. Slide 1 / 20. Slide 2 / 20. Slide 3 / 20. Observing Chemical Reactions Lab. Thoughts on the Nature of Matter
 Slide 1 / 20 Slide 2 / 20 Atomic Origins Observing Chemical Reactions Lab 2015-10-27 www.njctl.org Thoughts on the Nature of Matter Slide 3 / 20 Since ancient times, humans have pondered the nature of
Slide 1 / 20 Slide 2 / 20 Atomic Origins Observing Chemical Reactions Lab 2015-10-27 www.njctl.org Thoughts on the Nature of Matter Slide 3 / 20 Since ancient times, humans have pondered the nature of
What Do You Think? Investigate GOALS
 Activity 3 Atoms and Their Masses GOALS In this activity you will: Explore the idea of atoms by trying to isolate a single atom. Measure how many times greater the mass of a copper atom is than a magnesium
Activity 3 Atoms and Their Masses GOALS In this activity you will: Explore the idea of atoms by trying to isolate a single atom. Measure how many times greater the mass of a copper atom is than a magnesium
Warm Up 9/17/12. How long have people been interested in understanding matter and its structure? A. Thousands of years. B.
 Warm Up 9/17/12 How long have people been interested in understanding matter and its structure? A. Thousands of years B. Hundreds of years C. A few years D. Never What is an atom? The smallest particle
Warm Up 9/17/12 How long have people been interested in understanding matter and its structure? A. Thousands of years B. Hundreds of years C. A few years D. Never What is an atom? The smallest particle
Atomic Structure. For thousands of years, people had many ideas about matter Ancient Greeks believed that everything was made up of the four elements
 An atom is the smallest particle of an element that retains its identity in a chemical reaction. Although early philosophers and scientists could not observe individual atoms, they were still able to propose
An atom is the smallest particle of an element that retains its identity in a chemical reaction. Although early philosophers and scientists could not observe individual atoms, they were still able to propose
Chapter 2: An Evolving Model
 Physical Science 4010 Nuclear Technology Chapter 2: An Evolving Model Introduction The universe is made up of two things: Matter (physical, tangible stuff) Energy (more than just the ability to do work)
Physical Science 4010 Nuclear Technology Chapter 2: An Evolving Model Introduction The universe is made up of two things: Matter (physical, tangible stuff) Energy (more than just the ability to do work)
Particle Theory of Matter. By the late 1700s, scientists had adopted the Particle Theory of Matter. This theory states that:
 Particle Theory of Matter By the late 1700s, scientists had adopted the Particle Theory of Matter. This theory states that: all matter is made up of very tiny particles each pure substance has its own
Particle Theory of Matter By the late 1700s, scientists had adopted the Particle Theory of Matter. This theory states that: all matter is made up of very tiny particles each pure substance has its own
ACTIVITY SHEETS PHYSICS AND CHEMISTRY 2 nd ESO) NAME:
 ACTIVITY SHEETS PHYSICS AND CHEMISTRY 2 nd ESO) NAME:. READING 1 The atom Lesson 4. JOURNEY TO THE INTERIOR OF MATTER Wise men and women of the great ancient civilisations thought a long time ago about
ACTIVITY SHEETS PHYSICS AND CHEMISTRY 2 nd ESO) NAME:. READING 1 The atom Lesson 4. JOURNEY TO THE INTERIOR OF MATTER Wise men and women of the great ancient civilisations thought a long time ago about
Topic 2 Atomic Structure. IB Chemistry SL Coral Gables Senior High School Ms. Kiely
 Topic 2 Atomic Structure IB Chemistry SL Coral Gables Senior High School Ms. Kiely Prepare for Lab Quiz: Put everything away except for a pen (no pencil) and a calculator. Bell-Ringer This is an example
Topic 2 Atomic Structure IB Chemistry SL Coral Gables Senior High School Ms. Kiely Prepare for Lab Quiz: Put everything away except for a pen (no pencil) and a calculator. Bell-Ringer This is an example
atomos is a Greek word which means indivisible
 The History of Atomic Theory i.e. the history of the development of thought about what an atom is. 1st timeframe: around 5 B.C. : This was the time of the Ancient Greeks (in Athens, Greece). During the
The History of Atomic Theory i.e. the history of the development of thought about what an atom is. 1st timeframe: around 5 B.C. : This was the time of the Ancient Greeks (in Athens, Greece). During the
Early Ideas about Matter
 Early Ideas about Matter atom The smallest piece of the element with all the chemical properties of the element an old and new idea Greeks Discontinuists Democritus Continuists Aristotle Discontinuists
Early Ideas about Matter atom The smallest piece of the element with all the chemical properties of the element an old and new idea Greeks Discontinuists Democritus Continuists Aristotle Discontinuists
3 FROM ALCHEMY TO CHEMISTRY 810L
 3 FROM ALCHEMY TO CHEMISTRY 810L FROM ALCHEMY TO CHEMISTRY THE ORIGINS OF TODAY S CENTRAL SCIENCE By Michelle Feder, adapted by Newsela Many of the first chemists, doctors, and philosophers were also alchemists.
3 FROM ALCHEMY TO CHEMISTRY 810L FROM ALCHEMY TO CHEMISTRY THE ORIGINS OF TODAY S CENTRAL SCIENCE By Michelle Feder, adapted by Newsela Many of the first chemists, doctors, and philosophers were also alchemists.
Chemistry Chapter 3. Atoms: The Building Blocks of Matter
 Chemistry Chapter 3 Atoms: The Building Blocks of Matter I. From Philosophical Idea to Scientific Theory History of the Atom The Ancient Greeks were the first to come up with the idea of the atom. Democritus
Chemistry Chapter 3 Atoms: The Building Blocks of Matter I. From Philosophical Idea to Scientific Theory History of the Atom The Ancient Greeks were the first to come up with the idea of the atom. Democritus
What is Chemistry? Some random thoughts from the Professor for what they re worth
 What is Chemistry? Some random thoughts from the Professor for what they re worth CHEMISTRY Chemistry is the science that describes matter: its properties, the changes it undergoes Important questions:
What is Chemistry? Some random thoughts from the Professor for what they re worth CHEMISTRY Chemistry is the science that describes matter: its properties, the changes it undergoes Important questions:
Chemistry for Changing Times, 13e (Hill) Chapter 2 Atoms. 2.1 Multiple Choice Questions
 Chemistry for Changing Times 13th Edition Hill Test Bank Full Download: http://testbanklive.com/download/chemistry-for-changing-times-13th-edition-hill-test-bank/ Chemistry for Changing Times, 13e (Hill)
Chemistry for Changing Times 13th Edition Hill Test Bank Full Download: http://testbanklive.com/download/chemistry-for-changing-times-13th-edition-hill-test-bank/ Chemistry for Changing Times, 13e (Hill)
Activity 2 Elements and Their Properties
 Activity 2 Elements and Their Properties Activity 2 Elements and Their Properties GOALS In this activity you will: Apply ancient definitions of elements to materials you believe are elements. Test some
Activity 2 Elements and Their Properties Activity 2 Elements and Their Properties GOALS In this activity you will: Apply ancient definitions of elements to materials you believe are elements. Test some
2 History of the Atomic
 www.ck12.org CHAPTER 2 History of the Atomic Theory Chapter Outline 2.1 DEMOCRITUS IDEA OF THE ATOM 2.2 DALTON S ATOMIC THEORY 2.3 THOMSON S ATOMIC MODEL 2.4 RUTHERFORD S ATOMIC MODEL 2.5 BOHR S ATOMIC
www.ck12.org CHAPTER 2 History of the Atomic Theory Chapter Outline 2.1 DEMOCRITUS IDEA OF THE ATOM 2.2 DALTON S ATOMIC THEORY 2.3 THOMSON S ATOMIC MODEL 2.4 RUTHERFORD S ATOMIC MODEL 2.5 BOHR S ATOMIC
Careful observations led to the discovery of the conservation of mass.
 Chapter 7, Section 2 Key Concept: The masses of reactants and products are equal. BEFORE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed
Chapter 7, Section 2 Key Concept: The masses of reactants and products are equal. BEFORE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed
Law of conservation of mass If a piece of magnesium is burnt, will there be a gain or a loss in mass? Why?
 1 Atomic Theory Law of conservation of mass If a piece of magnesium is burnt, will there be a gain or a loss in mass? Why? Activity Measure the mass of 500 cm 3 of your favorite drink. Then compare your
1 Atomic Theory Law of conservation of mass If a piece of magnesium is burnt, will there be a gain or a loss in mass? Why? Activity Measure the mass of 500 cm 3 of your favorite drink. Then compare your
1 The Development of Atomic Theory
 CHAPTER 4 1 The Development of Atomic Theory SECTION Atoms KEY IDEAS As you read this section, keep these questions in mind: What scientists helped to develop atomic theory? What part of atoms did Thomson
CHAPTER 4 1 The Development of Atomic Theory SECTION Atoms KEY IDEAS As you read this section, keep these questions in mind: What scientists helped to develop atomic theory? What part of atoms did Thomson
THE ATOM atoms. electrons nucleus, protons neutrons atomic number atomic weight isotopes
 1 THE ATOM All matter is made up of atoms. An atom is made up of two major parts: First, there is a cloud of electrons (very light, negatively charged particles filling most of the volume), and second,
1 THE ATOM All matter is made up of atoms. An atom is made up of two major parts: First, there is a cloud of electrons (very light, negatively charged particles filling most of the volume), and second,
Dissolving vs. phase change. What happens to the atoms and the substances?
 Dissolving vs. phase change What happens to the atoms and the substances? Atoms and molecules are the building blocks of matter! What did you learn about them from your readings? What happened in the Saltwater
Dissolving vs. phase change What happens to the atoms and the substances? Atoms and molecules are the building blocks of matter! What did you learn about them from your readings? What happened in the Saltwater
Early Ideas About Matter
 Early Ideas About Matter Democritus (460 370 BC) believed that matter is made of small, solid objects called atomos, from which the English word atom is derived. Early Ideas About Matter (cont.) Aristotle
Early Ideas About Matter Democritus (460 370 BC) believed that matter is made of small, solid objects called atomos, from which the English word atom is derived. Early Ideas About Matter (cont.) Aristotle
CHAPTER 3. Atoms: The Building Blocks of Matter
 CHAPTER 3 Atoms: The Building Blocks of Matter Origins of the Atom Democritus: Greek philosopher (460 BC - 370 BC) Coined the term atom from the Greek word atomos Democritus believes that atoms were indivisible
CHAPTER 3 Atoms: The Building Blocks of Matter Origins of the Atom Democritus: Greek philosopher (460 BC - 370 BC) Coined the term atom from the Greek word atomos Democritus believes that atoms were indivisible
Card 1 Chapter 18. Card 2. Chapter 18. Negative particles that surround the nucleus (like planets around the sun)
 Card 1 Card 2 Positive particles in the nucleus of the atom Negative particles that surround the nucleus (like planets around the sun) Card 3 Card 4 Neutral particles in the nucleus of the atom. They help
Card 1 Card 2 Positive particles in the nucleus of the atom Negative particles that surround the nucleus (like planets around the sun) Card 3 Card 4 Neutral particles in the nucleus of the atom. They help
Vocabulary QUIZ: 1. The total number of particles in the nucleus 2. 1 / 12
 Sep 29 11:29 AM Vocabulary QUIZ: 1. The total number of particles in the nucleus 2. 1 / 12 th of the mass of a carbon atom 3. The weighted average mass of all the isotopes of a particular element 4. A
Sep 29 11:29 AM Vocabulary QUIZ: 1. The total number of particles in the nucleus 2. 1 / 12 th of the mass of a carbon atom 3. The weighted average mass of all the isotopes of a particular element 4. A
Today is Thursday, September 7 th, 2017
 In This Lesson: Unit 1 Matter and Change (Lesson 1 of 6) Today is Thursday, September 7 th, 2017 Pre-Class: Identify the chemistry in this scene (and I don t mean between the people). Hint: There are chemical
In This Lesson: Unit 1 Matter and Change (Lesson 1 of 6) Today is Thursday, September 7 th, 2017 Pre-Class: Identify the chemistry in this scene (and I don t mean between the people). Hint: There are chemical
Passing an electric current makes a beam appear to move from the negative to the positive end.
 Chapter 4 Atoms and their structure History of the atom Not the history of atom, but the idea of the atom. Original idea Ancient Greece (400 B.C.) Democritus and Leucippus Greek philosophers. Smallest
Chapter 4 Atoms and their structure History of the atom Not the history of atom, but the idea of the atom. Original idea Ancient Greece (400 B.C.) Democritus and Leucippus Greek philosophers. Smallest
Early Atomic Theory. Alchemy. The atom
 Early Atomic Theory Chapter 3 Democritus 460 BC- ~ 370 BC Nothing exists except atoms and empty space; everything else is opinion. Matter is composed of small indivisible particles, atomos meaning Indivisible
Early Atomic Theory Chapter 3 Democritus 460 BC- ~ 370 BC Nothing exists except atoms and empty space; everything else is opinion. Matter is composed of small indivisible particles, atomos meaning Indivisible
Everything is a chemical!!!
 Why Take Chemistry? 1.Guidance 2. Career 3. How does the world work? Ice skating Sweat Water bugs Atomic bombs Fossil fuels Fireworks Why doesn t a gas tank explode? Everything is a chemical!!! 1 Chemistry
Why Take Chemistry? 1.Guidance 2. Career 3. How does the world work? Ice skating Sweat Water bugs Atomic bombs Fossil fuels Fireworks Why doesn t a gas tank explode? Everything is a chemical!!! 1 Chemistry
Name Date Class DEFINING THE ATOM
 4.1 DEFINING THE ATOM Section Review Objectives Describe Democritus s ideas about atoms Explain Dalton s atomic theory Describe the size of an atom Vocabulary atom Dalton s atomic theory Part A Completion
4.1 DEFINING THE ATOM Section Review Objectives Describe Democritus s ideas about atoms Explain Dalton s atomic theory Describe the size of an atom Vocabulary atom Dalton s atomic theory Part A Completion
Chapter 4: Atomic Structure Section 4.1 Defining the Atom
 Chapter 4: Atomic Structure Section 4.1 Defining the Atom Early Models of the Atom atom the smallest particle of an element that retains its identity in a chemical reaction Democritus s Atomic Philosophy
Chapter 4: Atomic Structure Section 4.1 Defining the Atom Early Models of the Atom atom the smallest particle of an element that retains its identity in a chemical reaction Democritus s Atomic Philosophy
Atoms, Elements and the Periodic Table. By: Mrs. Herman 2013
 Atoms, Elements and the Periodic Table By: Mrs. Herman 2013 Made of A t o m s Protons (positive charge) Neutrons (neutral charge) Electrons (negative charge) Each atom contains a specific number of protons
Atoms, Elements and the Periodic Table By: Mrs. Herman 2013 Made of A t o m s Protons (positive charge) Neutrons (neutral charge) Electrons (negative charge) Each atom contains a specific number of protons
Ancient Greek Models of Atoms
 Atomic Theory Ancient Greek Models of Atoms The philosopher Democritus believed that all matter consisted of extremely small particles that could not be divided. He called these particles atoms from the
Atomic Theory Ancient Greek Models of Atoms The philosopher Democritus believed that all matter consisted of extremely small particles that could not be divided. He called these particles atoms from the
Scientists to Know CHADWICK THOMSON RUTHERFORD DEMOCRITUS BOHR HEISENBERG DALTON
 Unit 1: Atoms Scientists to Know CHADWICK THOMSON RUTHERFORD DEMOCRITUS BOHR HEISENBERG DALTON The History of Discovering the Atom The Timeline of Discovery 1. Philosophical Era 2. Alchemical Era 3. Classical
Unit 1: Atoms Scientists to Know CHADWICK THOMSON RUTHERFORD DEMOCRITUS BOHR HEISENBERG DALTON The History of Discovering the Atom The Timeline of Discovery 1. Philosophical Era 2. Alchemical Era 3. Classical
Foundations of Chemistry
 Foundations of Chemistry Physical Properties Physical Properties As you read in Lesson 1, the arrangement of atoms determines whether matter is a substance or a mixture. The arrangement of atoms also determines
Foundations of Chemistry Physical Properties Physical Properties As you read in Lesson 1, the arrangement of atoms determines whether matter is a substance or a mixture. The arrangement of atoms also determines
Molecules, Compounds, and Crystals
 Matter: Properties and Change Atoms and Elements 1. Matter is anything that has mass and volume. All substances are made up of different types of matter. 2. Mass is the amount of matter in a substance.
Matter: Properties and Change Atoms and Elements 1. Matter is anything that has mass and volume. All substances are made up of different types of matter. 2. Mass is the amount of matter in a substance.
General Chemistry Standard : Identify the significance of the various outcomes of Thomson s and Rutherford s experiments
 Not the history of the atom, but the idea of the atom The atom was not discovered until recently Original Idea Ancient Greece (400 BC) Proposed by lesser-known scientists They looked at a beach made of
Not the history of the atom, but the idea of the atom The atom was not discovered until recently Original Idea Ancient Greece (400 BC) Proposed by lesser-known scientists They looked at a beach made of
1 Development of the Atomic Theory
 CHAPTER 4 1 Development of the Atomic Theory SECTION Introduction to Atoms BEFORE YOU READ After you read this section, you should be able to answer these questions: What is the atomic theory? How has
CHAPTER 4 1 Development of the Atomic Theory SECTION Introduction to Atoms BEFORE YOU READ After you read this section, you should be able to answer these questions: What is the atomic theory? How has
Chemical Reactions: The Law of Conservation of Mass
 Chemical Reactions: The Law of Conservation of Mass What happens in a Chemical Reaction? Chemical bonds in the reactants are broken, then atoms are rearranged to form new substances (products). Reactants
Chemical Reactions: The Law of Conservation of Mass What happens in a Chemical Reaction? Chemical bonds in the reactants are broken, then atoms are rearranged to form new substances (products). Reactants
4-1 Notes. Defining the Atom
 4-1 Notes Defining the Atom Early Models of the Atom All matter is composed of atoms Atoms are the smallest particles of an element that retains their identity in a chemical reaction Greek philosopher
4-1 Notes Defining the Atom Early Models of the Atom All matter is composed of atoms Atoms are the smallest particles of an element that retains their identity in a chemical reaction Greek philosopher
5.1. Developing the Atomic Theory. Science, Art, and Atoms
 5.1 Developing the Atomic Theory Here is a summary of what you will learn in this section: An element is a pure substance that cannot be broken down into other substances by chemical reactions. The smallest
5.1 Developing the Atomic Theory Here is a summary of what you will learn in this section: An element is a pure substance that cannot be broken down into other substances by chemical reactions. The smallest
Atom s Structure. Chemistry is the study of the structure, characteristics and behavior of matter. The basic
 AlMarzooqi 1 Hamad AlMarzooqi Professor Kafle ESL 015 26 February 2013 Atom s Structure Chemistry is the study of the structure, characteristics and behavior of matter. The basic unit of matter is an atom,
AlMarzooqi 1 Hamad AlMarzooqi Professor Kafle ESL 015 26 February 2013 Atom s Structure Chemistry is the study of the structure, characteristics and behavior of matter. The basic unit of matter is an atom,
1. 2. Differentiate states of matter. Arrangement of Particles
 Lesson 2 Predict three facts that will be discussed in Lesson 2 after reading the headings. Record your predictions in your Science Journal. Definition: Define physical property, and give two examples.
Lesson 2 Predict three facts that will be discussed in Lesson 2 after reading the headings. Record your predictions in your Science Journal. Definition: Define physical property, and give two examples.
Atomic Structure. A model uses familiar ideas to explain unfamiliar facts observed in nature.
 Atomic Structure 1 2 This model of the atom may look familiar to you. This is the Bohr model. In this model, the nucleus is orbited by electrons, which are in different energy levels. A model uses familiar
Atomic Structure 1 2 This model of the atom may look familiar to you. This is the Bohr model. In this model, the nucleus is orbited by electrons, which are in different energy levels. A model uses familiar
Name: Block Unit 3- The Atom
 Name: Block Unit 3- The Atom DEMOCRITUS 1. Was Democritus a scientist? Notes 2. In what time of history did he live? 3. Describe Democritus thoughts about gold. 4. What was Democritus word for something
Name: Block Unit 3- The Atom DEMOCRITUS 1. Was Democritus a scientist? Notes 2. In what time of history did he live? 3. Describe Democritus thoughts about gold. 4. What was Democritus word for something
1 Classifying Matter KEY IDEAS READING TOOLBOX MATTER AND CHEMISTRY. Matter. As you read this section, keep these questions in mind: Name Class Date
 CHAPTER 2 1 Classifying Matter SECTION Matter KEY IDEAS As you read this section, keep these questions in mind: How can matter be classified? Why are elements and compounds considered pure substances?
CHAPTER 2 1 Classifying Matter SECTION Matter KEY IDEAS As you read this section, keep these questions in mind: How can matter be classified? Why are elements and compounds considered pure substances?
What is matter? Matter is anything that has mass and takes up space. Matter is made up of atoms.
 Matter What is matter? Matter is anything that has mass and takes up space. Matter is made up of atoms. Is it matter? Can you measure the object? Does it take up space? Does the object have a mass? Come
Matter What is matter? Matter is anything that has mass and takes up space. Matter is made up of atoms. Is it matter? Can you measure the object? Does it take up space? Does the object have a mass? Come
CHAPTER 4: Matter is Made up of Atoms
 CHAPTER 4: Matter is Made up of Atoms ATOMS & THEIR STRUCTURE Aristotle thought matter was made of air, earth, fire and water. Democritus (250 B.C.)- Said the world is made of empty space & tiny particles
CHAPTER 4: Matter is Made up of Atoms ATOMS & THEIR STRUCTURE Aristotle thought matter was made of air, earth, fire and water. Democritus (250 B.C.)- Said the world is made of empty space & tiny particles
Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons Electrons Neutrons Atomic # Mass # 24 Na
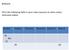 Bellwork: Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons Electrons Neutrons Atomic # Mass # 24 Na + 11 29 63 Bellwork: Fill in the Following Table in
Bellwork: Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons Electrons Neutrons Atomic # Mass # 24 Na + 11 29 63 Bellwork: Fill in the Following Table in
Chapter 11 Study Questions Name: Class:
 Chapter 11 Study Questions Name: Class: Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. The discovery of which particle proved that the atom
Chapter 11 Study Questions Name: Class: Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. The discovery of which particle proved that the atom
5th Grade. Slide 1 / 67. Slide 2 / 67. Slide 3 / 67. Matter and Its Interactions. Table of Contents: Matter and Its Interactions
 Slide 1 / 67 Slide 2 / 67 5th Grade Matter and Its Interactions 2015-11-02 www.njctl.org Table of Contents: Matter and Its Interactions Slide 3 / 67 Click on the topic to go to that section What Is Matter?
Slide 1 / 67 Slide 2 / 67 5th Grade Matter and Its Interactions 2015-11-02 www.njctl.org Table of Contents: Matter and Its Interactions Slide 3 / 67 Click on the topic to go to that section What Is Matter?
Understanding the Atom 1.3
 Understanding the Atom 1.3 Introduction to Atoms 1.What are some ways that scientists have described the atom? 2.What are the parts of the atom, and how are they arranged? 3.How are atoms of all elements
Understanding the Atom 1.3 Introduction to Atoms 1.What are some ways that scientists have described the atom? 2.What are the parts of the atom, and how are they arranged? 3.How are atoms of all elements
Unit 2 continued-chemical Foundations Atoms, Ions, &Elements
 Unit 2 continuedchemical Foundations Atoms, Ions, &Elements The Elements Most abundant elements in/on Earth: Oxygen 49.2% Silicon25.7% Most abundant in the human body: Oxygen65.0% Carbon18.0 % Hydrogen10.0%
Unit 2 continuedchemical Foundations Atoms, Ions, &Elements The Elements Most abundant elements in/on Earth: Oxygen 49.2% Silicon25.7% Most abundant in the human body: Oxygen65.0% Carbon18.0 % Hydrogen10.0%
Where it came from and what we know now
 Where it came from and what we know now History of the Atom The first mention of the atom came from Democritus in ancient Greece He suggested that the universe was made up of small, indivisible units called
Where it came from and what we know now History of the Atom The first mention of the atom came from Democritus in ancient Greece He suggested that the universe was made up of small, indivisible units called
Atomic and Molecular Physics Prof. Amal Kumar Das Department of Physics Indian Institute of Technology, Kharagpur
 Atomic and Molecular Physics Prof. Amal Kumar Das Department of Physics Indian Institute of Technology, Kharagpur Lecture 01 Experimental observations and theoretical development in discovery of constituents
Atomic and Molecular Physics Prof. Amal Kumar Das Department of Physics Indian Institute of Technology, Kharagpur Lecture 01 Experimental observations and theoretical development in discovery of constituents
Why Take Chemistry? 2. Career 3. How does the world work? 1.Guidance. Why doesn t a gas tank explode?
 Why Take Chemistry? 1.Guidance 2. Career 3. How does the world work? Ice skating Sweat Water bugs Computers Fossil fuels Atomic bombs Acid rain Fireworks Why doesn t a gas tank explode? 1 Chemistry What
Why Take Chemistry? 1.Guidance 2. Career 3. How does the world work? Ice skating Sweat Water bugs Computers Fossil fuels Atomic bombs Acid rain Fireworks Why doesn t a gas tank explode? 1 Chemistry What
In Atomic Theory, if we were to zoom in on a copper wire, it might look like the picture below. What is between the atoms?
 History of the Atom In Atomic Theory, if we were to zoom in on a copper wire, it might look like the picture below. What is between the atoms? What did be believe before we thought there were atoms? We
History of the Atom In Atomic Theory, if we were to zoom in on a copper wire, it might look like the picture below. What is between the atoms? What did be believe before we thought there were atoms? We
Chapter 02 Test Bank: Atoms, Ions, and the Periodic Table
 Introduction to Chemistry 4th Edition Bauer Test Bank Full Download: http://testbanklive.com/download/introduction-to-chemistry-4th-edition-bauer-test-bank/ Chapter 02 Test Bank: Atoms, Ions, and the Periodic
Introduction to Chemistry 4th Edition Bauer Test Bank Full Download: http://testbanklive.com/download/introduction-to-chemistry-4th-edition-bauer-test-bank/ Chapter 02 Test Bank: Atoms, Ions, and the Periodic
Unsaved Test, Version: 1 1
 Name: Key Concepts Select the term that best completes the statement. A. beam balance B. graduated cylinder C. scale D. matter E. mass F. newton G. volume H. height I. weight J. kilogram 1. Anything that
Name: Key Concepts Select the term that best completes the statement. A. beam balance B. graduated cylinder C. scale D. matter E. mass F. newton G. volume H. height I. weight J. kilogram 1. Anything that
The Periodic Table. Helium atom. So Many Elements!
 The Periodic Table So Many Elements! All things are made of atoms. There are more than 100 kinds of atoms. Something can be made with just one kind of atom. That is called an element. Silver is an element.
The Periodic Table So Many Elements! All things are made of atoms. There are more than 100 kinds of atoms. Something can be made with just one kind of atom. That is called an element. Silver is an element.
Dalton Thompson Rutherford Bohr Modern Model ("Wave. Models of the Atom
 Dalton Thompson Rutherford Bohr Modern Model ("Wave Models of the Atom Mechanical" Model) Aim: To discuss the scientists and their contributions to the current atomic model. Focus: Rutherford's Gold Foil
Dalton Thompson Rutherford Bohr Modern Model ("Wave Models of the Atom Mechanical" Model) Aim: To discuss the scientists and their contributions to the current atomic model. Focus: Rutherford's Gold Foil
Bellwork: 2/6/2013. atom is the. atom below. in an atom is found in the. mostly. 2. The smallest part of an. 1. Label the parts of the
 Bellwork: 2/6/2013 1. Label the parts of the atom below. B 2. The smallest part of an atom is the. 3. The majority of the mass in an atom is found in the. A C 4. An atom is made up of mostly. Bellwork:
Bellwork: 2/6/2013 1. Label the parts of the atom below. B 2. The smallest part of an atom is the. 3. The majority of the mass in an atom is found in the. A C 4. An atom is made up of mostly. Bellwork:
Chapter 4 (part 1) Chemical Foundations: Elements, Atoms, and Ions. Copyright Cengage Learning. All rights reserved 1
 Chapter 4 (part 1) Chemical Foundations: Elements, Atoms, and Ions Copyright Cengage Learning. All rights reserved 1 Section 4.1 The Elements 118 known: 88 found in nature, others are made in laboratories.
Chapter 4 (part 1) Chemical Foundations: Elements, Atoms, and Ions Copyright Cengage Learning. All rights reserved 1 Section 4.1 The Elements 118 known: 88 found in nature, others are made in laboratories.
Unit 7: The Periodic Table
 Unit 7: The Periodic Table Name Class Website: http://pilarz.weebly.com PS:7 TLW categorize elements of the periodic table according to common properties and explain how elements differ in structural parts
Unit 7: The Periodic Table Name Class Website: http://pilarz.weebly.com PS:7 TLW categorize elements of the periodic table according to common properties and explain how elements differ in structural parts
History of the Atom. Say Thanks to the Authors Click (No sign in required)
 History of the Atom Say Thanks to the Authors Click http://www.ck12.org/saythanks (No sign in required) To access a customizable version of this book, as well as other interactive content, visit www.ck12.org
History of the Atom Say Thanks to the Authors Click http://www.ck12.org/saythanks (No sign in required) To access a customizable version of this book, as well as other interactive content, visit www.ck12.org
Chemistry. Robert Taggart
 Chemistry Robert Taggart Table of Contents To the Student..................................................v Unit 1: Matter and Measurement Lesson 1: Chemistry and the Scientific Method...................3
Chemistry Robert Taggart Table of Contents To the Student..................................................v Unit 1: Matter and Measurement Lesson 1: Chemistry and the Scientific Method...................3
