Chapter 1: Introduction...1 Chapter 2: Energy, Entropy and Fundamental Thermodynamic Concepts...24
|
|
|
- Myron Williams
- 5 years ago
- Views:
Transcription
1 Table of Contents Part I: The Geochemical Toolbox Chapter 1: Introduction...1 Geochemistry...1 This Book...2 The Philosophy of Science...4 Building Scientific Understanding...4 The Scientist as Skeptic...5 Elements, Atoms, and Chemical Bonds...6 The Periodic Table...6 Electrons and Orbits...7 Some Chemical Properties of the Elements...13 Chemical Bonding...16 A Brief Look at the Earth...18 Structure of the Earth...18 Plate Tectonics and the Hydrologic Cycle...19 Earth Materials...21 References and Suggestions for Further Reading...23 Chapter 2: Energy, Entropy and Fundamental Thermodynamic Concepts...24 The Thermodynamic Perspective...24 Thermodynamic Systems and Equilibrium...24 Fundamental Thermodynamic Variables...26 Equations of State...28 Ideal Gas Law...28 Equations of State for Real Gases...29 Critical phenomena...31 Equation of State for Other Substances...32 Temperature, Absolute Zero, and The Zeroth Law Of Thermodynamics...33 Energy and The First Law of Thermodynamics...34 Energy...34 Work...34 Path independence, Exact differentials, State functions, and the First Law...35 The Second Law and Entropy...37 Statement...37 Statistical Mechanics: A Microscopic Perspective of Entropy...38 Integrating Factors and Exact Differentials...44 Enthalpy...45 Heat Capacity...46 Constant Volume Heat Capacity...47 Constant Pressure Heat Capacity...47 Energy Associated with Volume Occupied and the Relationshp between C v and C p...47 Heat Capacity of Solids: A Problem in Quantum Physics...50 Relationship of Entropy to Other State Variables...57 Additive Nature of Silicate Heat Capacities...57 The Third Law and Absolute Entropy...58 Statement of the Third Law...58 Absolute Entropy...58 Calculating Enthalpy and Entropy Changes...59 Adiabatic Changes...59 Isobaric and Isothermal Changes in Enthalpy...59 Enthalpies of Formation, Vaporization and Fusion...60 Entropies of Reaction...61 i November 25, 1997
2 Free Energy...62 Helmholtz Free Energy...63 Gibbs Free Energy...64 Criteria for Equilibrium and Spontaneity...64 Temperature and Pressure Dependence of the Gibbs Free Energy...65 The Maxwell Relations...67 References and Suggestions for Further Reading...67 Problems...68 Chapter 3: Solutions and Thermodynamics of Multicomponent Systems...70 Introduction...70 Phase Equilibria...70 Some Definitions...70 The Gibbs Phase Rule...73 The Clapeyron Equation...75 Solutions...77 Raoult's Law...77 Henry's Law...77 The Chemical Potential...78 Partial Molar Quantities...78 Definition of Chemical Potential and Relationship to Gibbs Free Energy...79 Properties of the Chemical Potential...80 The Gibbs-Duhem Relation...80 Derivation of the Phase Rule...81 Ideal Solutions...82 Real solutions...84 Chemical Potential in Real Solutions...84 Fugacities...85 Activities and Activity Coefficients...86 Excess Functions...88 Electrolyte Solutions...90 The Nature of Water and Water-Electrolyte Interaction...90 Some Definitions and Conventions...92 Activities in Electrolytes...94 Solid Solutions and Their Activities Ideal Solutions in Crystalline Solids Equilibrium Constants Derivation and Definition K D Values, Apparent Equilibrium Constants and the Solubility Product HenryÕs Law and Gas Solubilities Temperature Dependence of Equilibrium Constant Pressure Dependence of Equilibrium Constant Practical Approach to Electrolyte Equilibrium Chosing Components and Species Mass Balance Electrical Neutrality Equilibrium Constant Expressions Oxidation and Reduction Redox in Aqueous Solutions Redox in Magmatic Systems References and Suggestions for Further Reading Problems Chapter 4: Applications of Thermodynamics to the Earth Introduction i i November 25, 1997
3 Activities in Non-Ideal Solid Solutions Mathematical Models of Real Solutions Exsolution Phenomena Thermodynamics and Phase Diagrams The Thermodynamics of Melting Thermodynamics of Phase Diagrams for Binary Systems Geothermometry and Geobarometry Theoretical considerations Practical Thermobarometers Thermodynamic Models of Magmas Structure of Silicate Melts Magma Solution Models Reprise: Thermodynamics of Electrolyte Solutions Mean Ionic Quantities Activities in High Ionic Strength Solutions References and Suggestions For Further Reading Problems Chapter 5: Kinetics: The Pace of Things Introduction Reaction Kinetics Elementary and Overall Reactions Reaction Mechanisms Reaction Rates Rates of Complex Reactions Steady-State and Equilibrium Relationships between Kinetics and Thermodynamics Principle of Detailed Balancing Enthalpy and Activation Energy Aspects of Transition State Theory Diffusion Diffusion Flux and Fick's Law Diffusion in Multicomponent Systems and The Diffusion Coefficient Diffusion in Solds and the Temperature Dependence of the Diffusion Coefficient Diffusion in Liquids Surfaces, Interfaces, and Inferface Processes The Surface Free Energy The Kelvin Effect Nucleation and Crystal Growth Adsorption Catalysis Kinetics of Dissolution and Leaching Diagenesis Compostional Gradients in Accumulating Sediment References and Suggestions for Further Reading Problems Chapter 6: Aquatic Chemistry Introduction Acid-Base Reactions Proton Accounting, Charge Balance, and Conservation Equations The Carbonate System Conservative and Non-Conservative Ions Total Alkalinity and Carbonate Alkalinity Buffer Intensity i i i November 25, 1997
4 Complexation Stability Constants Water-Related Complexes Other Complexes Complexation in Fresh Waters Dissolution and Precipitation Reactions Dissolution of Carbonates by Ground and Surface Waters Solubility of Mg Solubility of SiO Solubility of Al(OH) 3 and Other Hydroxides Solubility of Silicates and Related Minerals Clays and Their Properties Clay Mineralogy Ion-Exchange Properties of Clays Mineral Surfaces and Their Interaction With Solutions Adsorption Development of Surface Charge and the Electric Double Layer References and Suggestions for Further Reading Problems Chapter 7: Trace Elements in Igneous Processes Introduction What is a Trace Element? Goldschmidt's Classification of the Elements The Geochemical Periodic Table The Volatile Elements The Semi-Volatiles The Alkali and Alkaline Earth Elements The Rare Earth Elements and Y The HFS Elements The First Series Transition Metals The Noble Metals Other Elements Distribution of Trace Elements Between Co-existing Phases The Partition Coefficient Factors Governing the Value of Parition Coefficients Temperature and Pressure Dependence of the Partition Coefficient Ionic Size and Charge Compositional Dependency Mineral-Liquid Partition Coefficients for Mafic and Ultramafic Systems Crystal-Field Effects Crystal Field Influences on Transition Metal Partitioning Trace Element Distribution During Partial Melting Equilibrium or Batch Melting Fractional Melting Zone Refining Multiphase Solids Continuous Melting Constraints on Melting Models Trace Element Distribution during Crystallization Equilibrium Crystallization Fractional Crystallization In Situ Crystallization Crystallization in Open System Magma Chambers i v November 25, 1997
5 Summary of Trace Element Variations during Melting and Crystallization References and Suggestions for Further Reading Problems Chapter 8: Radiogenic Isotope Geochemistry Introduction Physics of the Nucleus and the Structure of Nuclei Nuclear Structure and Energetics The Decay of Excited and Unstable Nuclei Basics of Radiogenic Isotope Geochemistry Geochronology Decay Systems and Their Applications Rb-Sr Sm-Nd Lu-Hf Re-Os La-Ce U-Th-Pb U and Th Decay Series Isotopes Isotopes of He and Other Rare Gases Cosmogenic and Fossil Isotopes C Cl in hydrology Be in Subduction Zone Studies Cosmic-Ray Exposure Ages of Meteorites Fossil Nuclides References and Suggestions for Further Reading Problems Chapter 9: Stable Isotope Geochemistry Introduction Scope of Stable Isotope Geochemistry Notation Theoretical Considerations Equilibrium Isotope Fractionations Kinetic Isotope Fractionations Isotope Geothermometry Isotope Fractionation in the Hydrologic System Isotope Fractionation in Biological Systems Carbon Isotope Fractionation During Photosynthesis Nitrogen Isotope Fractionation in Biological Processes Oxygen and Hydrogen Isotope Fractionation by Plants Biological Fractionation of Sulfur Isotopes Isotopes and Diet: You Are What You Eat Paleoclimatology The Marine Quaternary δ 18 O Record and Milankovitch Cycles The Record in Glacial Ice Soils and Paleosols Hydrothermal Systems and Ore Deposits Water-Rock Ratios Sulfur Isotopes and Ore Deposits Stable Isotopes in the Mantle and Magmatic Systems Stable Isotopic Composition of the Mantle v November 25, 1997
6 Stable Isotopes in Crystallizing Magmas Combined Fractional Crystallization and Assimilation Isotopes of Boron and Lithium References and Suggestions for Further Reading Problems Part II: Understanding the Earth Chapter 10: The Big Picture: Cosmochemistry Introduction In the Beginning...Nucleosynthesis Astronomical Background The Polygenetic Hypothesis of Burbidge, Burbidge, Fowler and Hoyle Cosmological Nucleosynthesis Nucleosynthesis in Stellar Interiors Explosive Nucleosynthesis Nucleosynthesis in Interstellar Space Summary Meteorites: Essential Clues to the Beginning Chondrites: The Most Primitive Objects Differentiated Meteorites Meteorite Mineralogy Relationships among Meteorites and Meteorite Parent-Bodies Time and the Isotopic composition of the Solar System Meteorite Ages Cosmic Ray Exposure Ages and the Origin of Meteorites Isotopic Anomalies in Meteorites Formation of the Solar System Astronomical Observations and Theoretical Models of Star Formation Formation of Chondritic Meteorites and Processes in the Solar Nebula Formation of the Planets The Moon: Its Chemistry and History The Giant Impact Hypothesis Geology and History of the Moon References and Suggestions for Further Reading Problems Chapter 11: Geochemistry of the Solid Earth I: The Mantle and Core Introduction Composition of the EarthÕs Mantle Cosmochemical Constraints on the EarthÕs Composition Upper Mantle Composition Phase Transitions in the Mantle The Transition Zone Composition of the Lower Mantle The Primitive Mantle Magma Ocean and Mantle Differentiation The Core and Core Formation Mantle Geochemical Reservoirs Evidence from Oceanic Basalts Evolution of the Depleted Upper Mantle Geochemical Evolution of Mantle Plume Reservoirs The Subcontinental Lithosphere References and Suggestions for Further Reading vi November 25, 1997
7 Problems Chapter 12: Geochemistry of the Solid Earth II: The Crust Introduction The Oceanic Crust Processes in Subduction Zones and Island Arc Geochemistry Major Element Composition Trace Element Composition Isotopic Composition and Sediment Subduction Magma Genesis in Subduction Zones CrustÑMantle Interaction: Assimilation Geochemistry of Two-Component Mixtures AssimilationÐFractional Crystallization Composition of the Continental Crust The Upper Crust The Middle and Lower Crust The Total Continental Crust Growth of the Continental Crust The Pace of Crustal Growth Mechanisms of Crustal Growth Crustal Growth in the Archean References and Suggestions for Further Reading Problems Chapter 13: Reactions at the EarthÕs Surface: Weathering, Soils, and Stream Chemistry Introduction Redox in Natural Waters Biogeochemical Reodox Reactions Redox Buffers and Transition Metal Chemistry Weathering, Soils, and Biogeochemical Cycling Soil Profiles Chemical Cycling in Soils Biogeochemical Cycling Weathering Rates and Reactions The Watershed Approach Thermodynamic and Kinetic Assessment of Stream Compositions Factors Controlling Weathering Rates The Composition of Rivers Continental Saline Waters References and Suggestions for Further Reading Problems Chapter 14: Organic Geochemistry Introduction A Brief Biological Background Organic Compounds and Their Nomenclature Hydrocarbons Functional Groups Biologically Important Organic Compounds The Chemistry of Life: Important Biochemical Processes Photosynthesis Respiration Organic Matter in Soils and Natural Waters Dissolved Organic Substances vii November 25, 1997
8 Organic Matter in Soils Chemical Properties of Organic Molecules Acid-Base Properties Complexation Adsorption Phenomena Sedimentary Organic Matter and Coal and Oil Formation Formation and Diagenesis of Organic-Rich Sediments Kerogen and Bitumen Thermal Evolution of Organic Matter and Petroleum Generation Compositional Evolution of Coal Isotopic Composition of Hydrocarbons References and Suggestions for Further Reading Problems Chapter 15: The Oceans as a Chemical System Introduction Some Background Oceanographic Concepts Salinity, Chlorinity, Density, and Temperature Circulation of the Ocean and the Structure of Ocean Water Composition of Seawater Conservative Elements Dissolved Gases Nutrient Elements Particle-Reactive Elements Sources and Sinks of Dissolved Matter in Seawater Residence Time The Riverine Source Ridge Hydrothermal Systems The Atmospheric Source Sedimentary Sinks and Sources References and Suggestions for Further Reading Problems vi i i November 25, 1997
AN INTRODUCTION TO COSMOCHEMISTRY
 AN INTRODUCTION TO COSMOCHEMISTRY CHARLES R. COWLEY Professor of Astronomy, University of Michigan CAMBRIDGE UNIVERSITY PRESS Foreword V a % e x i 1 Overview 1 1.1 The Scope of Cosmochemistry 1 1.2 Cosmochemistry
AN INTRODUCTION TO COSMOCHEMISTRY CHARLES R. COWLEY Professor of Astronomy, University of Michigan CAMBRIDGE UNIVERSITY PRESS Foreword V a % e x i 1 Overview 1 1.1 The Scope of Cosmochemistry 1 1.2 Cosmochemistry
HIGLEY UNIFIED SCHOOL DISTRICT INSTRUCTIONAL ALIGNMENT. Earth and Space Science Quarter 1. Earth and Space Science (Duration 1 Week)
 HIGLEY UNIFIED SCHOOL DISTRICT INSTRUCTIONAL ALIGNMENT Earth and Space Science Quarter 1 Earth and Space Science (Duration 1 Week) Big Idea: Essential Questions: 1. Describe how matter is classified by
HIGLEY UNIFIED SCHOOL DISTRICT INSTRUCTIONAL ALIGNMENT Earth and Space Science Quarter 1 Earth and Space Science (Duration 1 Week) Big Idea: Essential Questions: 1. Describe how matter is classified by
THE PLANETARY SCIENTIST'S COMPANION
 THE PLANETARY SCIENTIST'S COMPANION Katharina Lodders Bruce Fegley, Jr. New York Oxford Oxford University Press 1998 Contents 1 Technical data Table 1.1 The Greek alphabet 1 Table 1.2 Prefixes used with
THE PLANETARY SCIENTIST'S COMPANION Katharina Lodders Bruce Fegley, Jr. New York Oxford Oxford University Press 1998 Contents 1 Technical data Table 1.1 The Greek alphabet 1 Table 1.2 Prefixes used with
Chapter 5. The Biogeochemical Cycles. Botkin & Keller Environmental Science 5e
 Chapter 5 The Biogeochemical Cycles How Chemicals Cycle Biogeochemical Cycle The complete path a chemical takes through the four major components or reservoirs of Earth s systems 1. Atmosphere 2. Hydrosphere
Chapter 5 The Biogeochemical Cycles How Chemicals Cycle Biogeochemical Cycle The complete path a chemical takes through the four major components or reservoirs of Earth s systems 1. Atmosphere 2. Hydrosphere
Use models to describe the sun s place in space in relation to the Milky Way Galaxy and the distribution of galaxy clusters in the universe.
 The Milky Way Galaxy consists of more than two hundred billion stars, the sun being one of them, and is one of hundreds of billions of galaxies in the known universe. s of the formation and structure of
The Milky Way Galaxy consists of more than two hundred billion stars, the sun being one of them, and is one of hundreds of billions of galaxies in the known universe. s of the formation and structure of
Alfonso Mucci and Anthony Williams-Jones Frank Dawson Adams (FDA)- 201/317
 Department of Earth and Planetary Sciences McGill University EPSC-220 PRINCIPLES OF GEOCHEMISTRY Alfonso Mucci and Anthony Williams-Jones alfonso.mucci@mcgill.ca; Anthony.Williams-jones@mcgill.ca Frank
Department of Earth and Planetary Sciences McGill University EPSC-220 PRINCIPLES OF GEOCHEMISTRY Alfonso Mucci and Anthony Williams-Jones alfonso.mucci@mcgill.ca; Anthony.Williams-jones@mcgill.ca Frank
Trace Elements - Definitions
 Trace Elements - Definitions Elements that are not stoichiometric constituents in phases in the system of interest For example, IG/MET systems would have different trace elements than aqueous systems Do
Trace Elements - Definitions Elements that are not stoichiometric constituents in phases in the system of interest For example, IG/MET systems would have different trace elements than aqueous systems Do
Lecture 5. Introduction to Stable Isotopes
 Lecture 5 Introduction to Stable Isotopes Stable Isotope Geochemistry Primarily concerned with the isotope ratios of H, C, N, O, and S Si and B often included and new instrumentation has opened up others
Lecture 5 Introduction to Stable Isotopes Stable Isotope Geochemistry Primarily concerned with the isotope ratios of H, C, N, O, and S Si and B often included and new instrumentation has opened up others
Radiogenic Isotopes. W. F. McDonough 1 1 Department of Earth Sciences and Research Center for
 Radiogenic Isotopes W. F. McDonough 1 1 Department of Earth Sciences and Research Center for Neutrino Science, Tohoku University, Sendai 980-8578, Japan (Dated: May 17, 2018) I. SUMMRY Isotope systems
Radiogenic Isotopes W. F. McDonough 1 1 Department of Earth Sciences and Research Center for Neutrino Science, Tohoku University, Sendai 980-8578, Japan (Dated: May 17, 2018) I. SUMMRY Isotope systems
Lecture 4 What Controls the Composition of Seawater
 Lecture 4 What Controls the Composition of Seawater Seawater is salty! Why? What controls the composition of seawater? Do Chemical Equilibrium reactions control the composition of the Ocean? What is meant
Lecture 4 What Controls the Composition of Seawater Seawater is salty! Why? What controls the composition of seawater? Do Chemical Equilibrium reactions control the composition of the Ocean? What is meant
Trace Elements. Today s lecture
 Trace Elements 300 Ni 200 ppm 100 0 300 Zr 200 100 0 40 50 60 70 80 SiO 2 wt. % Updates: M&M due date: Tuesday Today s lecture Topics: Trace element compositions Trace element behavior Partitioning Spider(
Trace Elements 300 Ni 200 ppm 100 0 300 Zr 200 100 0 40 50 60 70 80 SiO 2 wt. % Updates: M&M due date: Tuesday Today s lecture Topics: Trace element compositions Trace element behavior Partitioning Spider(
RAYMOND SIEVER Harvard University
 E A R T H FOURTH EDITION FRANK PRESS National Academy of Sciences RAYMOND SIEVER Harvard University W. H. Freeman and Company New York Preface xiii Acknowledgments xviii PART I PROLOGUE CHAPTER 1 HISTORY
E A R T H FOURTH EDITION FRANK PRESS National Academy of Sciences RAYMOND SIEVER Harvard University W. H. Freeman and Company New York Preface xiii Acknowledgments xviii PART I PROLOGUE CHAPTER 1 HISTORY
Systems? Climate Systems. Earth Systems. Earth Interior Systems. Atmospheric/Biospheric Systems: Human Impact Hydrologic Cycle.
 Chapter 15 Climate Systems Systems? What is a system? Geologic phenomena are complex. All processes are related to, and interact with, other processes. So it is useful to think of geologic processes as
Chapter 15 Climate Systems Systems? What is a system? Geologic phenomena are complex. All processes are related to, and interact with, other processes. So it is useful to think of geologic processes as
Chapter 7: Environmental Systems and Ecosystem Ecology
 Chapter 7: Environmental Systems and Ecosystem Ecology Vocabulary words to know: Hypoxia Negative feedback Dynamic equilibrium Emergent properties Lithosphere Biosphere Gross primary production Nutrients
Chapter 7: Environmental Systems and Ecosystem Ecology Vocabulary words to know: Hypoxia Negative feedback Dynamic equilibrium Emergent properties Lithosphere Biosphere Gross primary production Nutrients
At the beginning. Matter + antimatter. Matter has the advantage. baryons quarks, leptons, electrons, photons (no protons or neutrons)
 At the beginning Matter + antimatter Matter has the advantage baryons quarks, leptons, electrons, photons (no protons or neutrons) Hadrons protons, neutrons Hydrogen, helium (:0 H:He) Origin of the Universe
At the beginning Matter + antimatter Matter has the advantage baryons quarks, leptons, electrons, photons (no protons or neutrons) Hadrons protons, neutrons Hydrogen, helium (:0 H:He) Origin of the Universe
OCE 290A (Topics in Chemical Oceanography) Application of Isotopes in Geological and Environmental Research
 OCE 290A (Topics in Chemical Oceanography) Application of Isotopes in Geological and Environmental Research Class participation 25%, Oral presentation 25%, Mid-term manuscript to review 25%, Final 3-5
OCE 290A (Topics in Chemical Oceanography) Application of Isotopes in Geological and Environmental Research Class participation 25%, Oral presentation 25%, Mid-term manuscript to review 25%, Final 3-5
Wednesday week 12. These ions move through the soil to streams and eventually to the ocean. In the ocean; CaCO 3 + H 2 O + CO 2 H 2 O + H 2 O
 Wednesday week 12 I. Control of CO 2 content of atmosphere by the ocean H 4 SiO 4 A. Consider a hypothetical planet with a crust made of single mineral (Wallastonite) CaSiO3. We could use the composition
Wednesday week 12 I. Control of CO 2 content of atmosphere by the ocean H 4 SiO 4 A. Consider a hypothetical planet with a crust made of single mineral (Wallastonite) CaSiO3. We could use the composition
 http://eps.mcgill.ca/~courses/c220/ Nucleosynthesis neutron electron + proton = é + H + t 1/2 = 12 minutes H + + neutron Deuterium (D) 2 H + + neutrons Helium (He) 3 H + + neutrons Lithium (Li) From: W.S.
http://eps.mcgill.ca/~courses/c220/ Nucleosynthesis neutron electron + proton = é + H + t 1/2 = 12 minutes H + + neutron Deuterium (D) 2 H + + neutrons Helium (He) 3 H + + neutrons Lithium (Li) From: W.S.
The Ocean as a stratified body of water
 The Ocean as a stratified body of water Density = f (salt content, T, pressure) Γ 35,25,1 = 1.02335 g cm -3 (= 1023 kg m -3 ) σ S,t,P = (Γ S,t,P -1) x 1000 σ 35,25,1 = 23.25 g cm -3 (= 23.25 kg m -3 )
The Ocean as a stratified body of water Density = f (salt content, T, pressure) Γ 35,25,1 = 1.02335 g cm -3 (= 1023 kg m -3 ) σ S,t,P = (Γ S,t,P -1) x 1000 σ 35,25,1 = 23.25 g cm -3 (= 23.25 kg m -3 )
I. Earth spheres A. Three major spheres 1. atmosphere, thin envelope 2. hydrosphere covers more than 71% of surface 3. geosphere from hydrosphere to
 I. Earth spheres A. Three major spheres 1. atmosphere, thin envelope 2. hydrosphere covers more than 71% of surface 3. geosphere from hydrosphere to center 4. Biosphere penetrates all three, a. only thin
I. Earth spheres A. Three major spheres 1. atmosphere, thin envelope 2. hydrosphere covers more than 71% of surface 3. geosphere from hydrosphere to center 4. Biosphere penetrates all three, a. only thin
Earth Science. Explain how Earth's biogeochemical cycles create a balance of materials. Examine the importance of biogeochemical cycles.
 Unit 1: DYNAMIC STRUCTURE OF EARTH Introduction to Understand that earth science is based upon the four sciences of astronomy, geology, meteorology, and oceanography. Identify topics studied within the
Unit 1: DYNAMIC STRUCTURE OF EARTH Introduction to Understand that earth science is based upon the four sciences of astronomy, geology, meteorology, and oceanography. Identify topics studied within the
Contents. 1 Introduction and guide for this text 1. 2 Equilibrium and entropy 6. 3 Energy and how the microscopic world works 21
 Preface Reference tables Table A Counting and combinatorics formulae Table B Useful integrals, expansions, and approximations Table C Extensive thermodynamic potentials Table D Intensive per-particle thermodynamic
Preface Reference tables Table A Counting and combinatorics formulae Table B Useful integrals, expansions, and approximations Table C Extensive thermodynamic potentials Table D Intensive per-particle thermodynamic
BIOGEOCHEMICAL CYCLES
 BIOGEOCHEMICAL CYCLES BASICS Biogeochemical Cycle: The complete path a chemical takes through the four major components, or reservoirs, of Earth s system (atmosphere, lithosphere, hydrosphere and biosphere)
BIOGEOCHEMICAL CYCLES BASICS Biogeochemical Cycle: The complete path a chemical takes through the four major components, or reservoirs, of Earth s system (atmosphere, lithosphere, hydrosphere and biosphere)
1) Radioactive Decay, Nucleosynthesis, and Basic Geochronology
 1) Radioactive Decay, Nucleosynthesis, and Basic Geochronology Reading (all from White s Notes) Lecture 1: Introduction And Physics Of The Nucleus: Skim Lecture 1: Radioactive Decay- Read all Lecture 3:
1) Radioactive Decay, Nucleosynthesis, and Basic Geochronology Reading (all from White s Notes) Lecture 1: Introduction And Physics Of The Nucleus: Skim Lecture 1: Radioactive Decay- Read all Lecture 3:
Jochen Hoefs Stable Isotope Geochemistry
 Jochen Hoefs Stable Isotope Geochemistry Springer-Verlag Berlin Heidelberg GmbH Jochen Hoefs Stable Isotope Geochemistry 4th, Completely Revised, Updated, and Enlarged Edition With 73 Figures and 22 Tables
Jochen Hoefs Stable Isotope Geochemistry Springer-Verlag Berlin Heidelberg GmbH Jochen Hoefs Stable Isotope Geochemistry 4th, Completely Revised, Updated, and Enlarged Edition With 73 Figures and 22 Tables
Contents. 1 Matter: Its Properties and Measurement 1. 2 Atoms and the Atomic Theory Chemical Compounds Chemical Reactions 111
 Ed: Pls provide art About the Authors Preface xvii xvi 1 Matter: Its Properties and Measurement 1 1-1 The Scientific Method 2 1-2 Properties of Matter 4 1-3 Classification of Matter 5 1-4 Measurement of
Ed: Pls provide art About the Authors Preface xvii xvi 1 Matter: Its Properties and Measurement 1 1-1 The Scientific Method 2 1-2 Properties of Matter 4 1-3 Classification of Matter 5 1-4 Measurement of
The Earth System Connections among the great spheres
 Why should we discuss the Earth System? The Earth System Connections among the great spheres Before we delve into the connection between geology, health, and forensics, we must gain an appreciation of
Why should we discuss the Earth System? The Earth System Connections among the great spheres Before we delve into the connection between geology, health, and forensics, we must gain an appreciation of
Lesson 3.1 Matter and the Environment. Water s abundance is a primary reason there is life on Earth.
 Lesson 3.1 Matter and the Environment Water s abundance is a primary reason there is life on Earth. Lesson 3.1 Matter and the Environment Atoms and Elements Atoms are the basic unit of matter. Nucleus:
Lesson 3.1 Matter and the Environment Water s abundance is a primary reason there is life on Earth. Lesson 3.1 Matter and the Environment Atoms and Elements Atoms are the basic unit of matter. Nucleus:
EPSS 15 Introduction to Oceanography Spring The Physical and Chemical Properties of Seawater
 EPSS 15 Introduction to Oceanography Spring 2017 The Physical and Chemical Properties of Seawater The focus of the Lab this week is seawater--its composition, physical and chemical properties. Seawater
EPSS 15 Introduction to Oceanography Spring 2017 The Physical and Chemical Properties of Seawater The focus of the Lab this week is seawater--its composition, physical and chemical properties. Seawater
N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0
 N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0 is initial number of parents, D* is number of radiogenic daughter atoms, and λ is the decay
N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0 is initial number of parents, D* is number of radiogenic daughter atoms, and λ is the decay
Chemistry: The Central Science Twelfth Edition, AP* Edition 2012
 A Correlation of The Central Science Twelfth Edition, AP* Edition 2012 to the AP* Chemistry Topics I. Structure of Matter A. Atomic theory and atomic structure 1. Evidence for the atomic theory SECTIONS:
A Correlation of The Central Science Twelfth Edition, AP* Edition 2012 to the AP* Chemistry Topics I. Structure of Matter A. Atomic theory and atomic structure 1. Evidence for the atomic theory SECTIONS:
Differentiation 2: mantle, crust OUTLINE
 Differentiation 2: mantle, crust OUTLINE Reading this week: Should have been White Ch 10 and 11!! 7- Nov Differentiation of the Earth, Core formation W 10.6.6, 11.4 9- Nov Moon, crust, mantle, atmosphere
Differentiation 2: mantle, crust OUTLINE Reading this week: Should have been White Ch 10 and 11!! 7- Nov Differentiation of the Earth, Core formation W 10.6.6, 11.4 9- Nov Moon, crust, mantle, atmosphere
XI. the natural carbon cycle. with materials from J. Kasting (Penn State)
 XI. the natural carbon cycle with materials from J. Kasting (Penn State) outline properties of carbon the terrestrial biological cycle of carbon the ocean cycle of carbon carbon in the rock cycle overview
XI. the natural carbon cycle with materials from J. Kasting (Penn State) outline properties of carbon the terrestrial biological cycle of carbon the ocean cycle of carbon carbon in the rock cycle overview
Geol. 655 Isotope Geochemistry
 ISOTOPIC GEOCHEMISTRY OF THE CONTINENTAL CRUST Isotope systems have particular value in studies of mantle geochemistry because of the difficulty of obtaining direct, representative samples of mantle. Because
ISOTOPIC GEOCHEMISTRY OF THE CONTINENTAL CRUST Isotope systems have particular value in studies of mantle geochemistry because of the difficulty of obtaining direct, representative samples of mantle. Because
Chapter 11. The Archean Era of Precambrian Time
 Chapter 11 The Archean Era of Precambrian Time 1 Guiding Questions When and how did Earth and its moon come into being? How did the core, mantle, crust form? Where did Archean rocks form, and what is their
Chapter 11 The Archean Era of Precambrian Time 1 Guiding Questions When and how did Earth and its moon come into being? How did the core, mantle, crust form? Where did Archean rocks form, and what is their
Chemical Oceanography Spring 2000 Final Exam (Use the back of the pages if necessary)(more than one answer may be correct.)
 Ocean 421 Your Name Chemical Oceanography Spring 2000 Final Exam (Use the back of the pages if necessary)(more than one answer may be correct.) 1. Due to the water molecule's (H 2 O) great abundance in
Ocean 421 Your Name Chemical Oceanography Spring 2000 Final Exam (Use the back of the pages if necessary)(more than one answer may be correct.) 1. Due to the water molecule's (H 2 O) great abundance in
Remote Sensing of the Earth s Interior
 Remote Sensing of the Earth s Interior Earth s interior is largely inaccessible Origin and Layering of the Earth: Geochemical Perspectives Composition of Earth cannot be understood in isolation Sun and
Remote Sensing of the Earth s Interior Earth s interior is largely inaccessible Origin and Layering of the Earth: Geochemical Perspectives Composition of Earth cannot be understood in isolation Sun and
Chemistry Class 11 Syllabus
 Chemistry Class 11 Syllabus Course Structure Unit Title Marks I Basic Concepts of Chemistry 11 II Structure of Atom III Classification of Elements & Periodicity in Properties 4 IV Chemical Bonding and
Chemistry Class 11 Syllabus Course Structure Unit Title Marks I Basic Concepts of Chemistry 11 II Structure of Atom III Classification of Elements & Periodicity in Properties 4 IV Chemical Bonding and
Igneous Rocks. Igneous Rocks. Genetic Classification of
 Igneous Rocks Fig. 5.1 Genetic Classification of Igneous Rocks Intrusive: crystallized from slowly cooling magma intruded within the Earth s crust; e.g. granite, gabbro 1 Fig. 5.2 Genetic Classification
Igneous Rocks Fig. 5.1 Genetic Classification of Igneous Rocks Intrusive: crystallized from slowly cooling magma intruded within the Earth s crust; e.g. granite, gabbro 1 Fig. 5.2 Genetic Classification
Lecture 3 Rocks and the Rock Cycle Dr. Shwan Omar
 Rocks A naturally occurring aggregate of one or more minerals (e.g., granite), or a body of non-crystalline material (e.g., obsidian glass), or of solid organic material (e.g., coal). Rock Cycle A sequence
Rocks A naturally occurring aggregate of one or more minerals (e.g., granite), or a body of non-crystalline material (e.g., obsidian glass), or of solid organic material (e.g., coal). Rock Cycle A sequence
High School Earth Science. High Science Strand 1: Earth s Place in the Universe
 High Science Strand 1: Earth s Place in the Universe Code Proposed Standards Existing GLES HS-ESS1-1. Develop a model based on evidence to illustrate the life span of the Sun and the role of nuclear fusion
High Science Strand 1: Earth s Place in the Universe Code Proposed Standards Existing GLES HS-ESS1-1. Develop a model based on evidence to illustrate the life span of the Sun and the role of nuclear fusion
Global Carbon Cycle - I Systematics: Reservoirs and Fluxes
 OCN 401-10 Nov. 16, 2010 KCR Global Carbon Cycle - I Systematics: Reservoirs and Fluxes The Global carbon cycle Reservoirs: biomass on land in the oceans, atmosphere, soil and rocks, waters Processes:
OCN 401-10 Nov. 16, 2010 KCR Global Carbon Cycle - I Systematics: Reservoirs and Fluxes The Global carbon cycle Reservoirs: biomass on land in the oceans, atmosphere, soil and rocks, waters Processes:
PTYS 214 Spring Announcements. Next midterm 3/1!
 PTYS 214 Spring 2018 Announcements Next midterm 3/1! 1 Previously Solar flux decreases as radiation spreads out away from the Sun Planets are exposed to some small amount of the total solar radiation A
PTYS 214 Spring 2018 Announcements Next midterm 3/1! 1 Previously Solar flux decreases as radiation spreads out away from the Sun Planets are exposed to some small amount of the total solar radiation A
SLO to PLO Alignment Report
 SLO to PLO Alignment Report CAN Program - Physical Sciences Use the scientific method and appreciate its importance to the development of scientific thought. CAN Dept - Astronomy CAN ASTR 100 : Introduction
SLO to PLO Alignment Report CAN Program - Physical Sciences Use the scientific method and appreciate its importance to the development of scientific thought. CAN Dept - Astronomy CAN ASTR 100 : Introduction
Miami Dade College CHM Second Semester General Chemistry
 Miami Dade College CHM 1046 - Second Semester General Chemistry Course Description: CHM 1046 is the second semester of a two-semester general chemistry course for science, premedical science and engineering
Miami Dade College CHM 1046 - Second Semester General Chemistry Course Description: CHM 1046 is the second semester of a two-semester general chemistry course for science, premedical science and engineering
Global phosphorus cycle
 Global phosphorus cycle OCN 623 Chemical Oceanography 11 April 2013 2013 Arisa Okazaki and Kathleen Ruttenberg Outline 1. Introduction on global phosphorus (P) cycle 2. Terrestrial environment 3. Atmospheric
Global phosphorus cycle OCN 623 Chemical Oceanography 11 April 2013 2013 Arisa Okazaki and Kathleen Ruttenberg Outline 1. Introduction on global phosphorus (P) cycle 2. Terrestrial environment 3. Atmospheric
Study guide for AP test on TOPIC 1 Matter & Measurement
 Study guide for AP test on IC 1 Matter & Measurement IC 1 Recall a definition of chemistry Understand the process and stages of scientific (logical) problem solving Recall the three states of matter, their
Study guide for AP test on IC 1 Matter & Measurement IC 1 Recall a definition of chemistry Understand the process and stages of scientific (logical) problem solving Recall the three states of matter, their
The Geochemistry of Natural Waters
 The Geochemistry of Natural Waters Surface and Groundwater Environments Third Edition James I. Drever University of Wyoming Prentice Hall Upper Saddle River. NJ 07458 Contents 3 Preface xi 1 The Hydrologie
The Geochemistry of Natural Waters Surface and Groundwater Environments Third Edition James I. Drever University of Wyoming Prentice Hall Upper Saddle River. NJ 07458 Contents 3 Preface xi 1 The Hydrologie
Seawater Chemistry Brooks/Cole, a division of Thomson Learning, Inc.
 Seawater Chemistry The ions of sodium and chloride in NaCl (table salt) are held together by ionic bonds, electrostatic attraction that exists between ions that have opposite charge. Sodium and chloride
Seawater Chemistry The ions of sodium and chloride in NaCl (table salt) are held together by ionic bonds, electrostatic attraction that exists between ions that have opposite charge. Sodium and chloride
Global Carbon Cycle - I
 Global Carbon Cycle - I OCN 401 - Biogeochemical Systems Reading: Schlesinger, Chapter 11 1. Overview of global C cycle 2. Global C reservoirs Outline 3. The contemporary global C cycle 4. Fluxes and residence
Global Carbon Cycle - I OCN 401 - Biogeochemical Systems Reading: Schlesinger, Chapter 11 1. Overview of global C cycle 2. Global C reservoirs Outline 3. The contemporary global C cycle 4. Fluxes and residence
KISS Resources for NSW Syllabuses & Australian Curriculum.
 Discusssion / Activity 1 Structure of the Earth Student Name... 1. Outline how we think the Sun & planets formed. The solar system formed from a cloud of gas & dust. Part of the cloud collapsed under gravity
Discusssion / Activity 1 Structure of the Earth Student Name... 1. Outline how we think the Sun & planets formed. The solar system formed from a cloud of gas & dust. Part of the cloud collapsed under gravity
Evidences for geochemically distinct mantle components
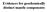 Evidences for geochemically distinct mantle components 1 Mantle Array Oceanic basalts, including seamounts, oceanic islands and middle ocean ridge basalts, were used. 2 Binary All analyses fall between
Evidences for geochemically distinct mantle components 1 Mantle Array Oceanic basalts, including seamounts, oceanic islands and middle ocean ridge basalts, were used. 2 Binary All analyses fall between
Lecture 38. Igneous geochemistry. Read White Chapter 7 if you haven t already
 Lecture 38 Igneous geochemistry Read White Chapter 7 if you haven t already Today. Magma mixing/afc 2. Spot light on using the Rare Earth Elements (REE) to constrain mantle sources and conditions of petrogenesis
Lecture 38 Igneous geochemistry Read White Chapter 7 if you haven t already Today. Magma mixing/afc 2. Spot light on using the Rare Earth Elements (REE) to constrain mantle sources and conditions of petrogenesis
Composition and the Early History of the Earth
 Composition and the Early History of the Earth Sujoy Mukhopadhyay CIDER 2006 What we will cover in this lecture Composition of Earth Short lived nuclides and differentiation of the Earth Atmosphere and
Composition and the Early History of the Earth Sujoy Mukhopadhyay CIDER 2006 What we will cover in this lecture Composition of Earth Short lived nuclides and differentiation of the Earth Atmosphere and
Today we begin with. Water is everywhere on and in Earth It is the only substance that exists in all 3 phases (solid, liquid, gas) on the surface!
 Water is everywhere on and in Earth It is the only substance that exists in all 3 phases (solid, liquid, gas) on the surface! Today we begin with Water Origin of Earth s water Properties of water Water
Water is everywhere on and in Earth It is the only substance that exists in all 3 phases (solid, liquid, gas) on the surface! Today we begin with Water Origin of Earth s water Properties of water Water
Chemistry: Molecules, Matter, and Change, Fourth Edition Loretta Jones and Peter Atkins Correlated with AP Chemistry, May 2002, May 2003
 Chemistry: Molecules, Matter, and Change, Fourth Edition Loretta Jones and Peter Atkins Correlated with AP Chemistry, May 2002, May 2003 ST=Student textbook I. Structure of Matter A. Atomic theory and
Chemistry: Molecules, Matter, and Change, Fourth Edition Loretta Jones and Peter Atkins Correlated with AP Chemistry, May 2002, May 2003 ST=Student textbook I. Structure of Matter A. Atomic theory and
1. Introduction 2. Ocean circulation a) Temperature, salinity, density b) Thermohaline circulation c) Wind-driven surface currents d) Circulation and
 1. Introduction 2. Ocean circulation a) Temperature, salinity, density b) Thermohaline circulation c) Wind-driven surface currents d) Circulation and climate change e) Oceanic water residence times 3.
1. Introduction 2. Ocean circulation a) Temperature, salinity, density b) Thermohaline circulation c) Wind-driven surface currents d) Circulation and climate change e) Oceanic water residence times 3.
GCSE Additional Science
 GCSE Additional Science Module C5 Chemicals of the Natural Environment: What you should know Name: Science Group: Teacher: each of the statements to help focus your revision: R = Red: I don t know this
GCSE Additional Science Module C5 Chemicals of the Natural Environment: What you should know Name: Science Group: Teacher: each of the statements to help focus your revision: R = Red: I don t know this
S= 95.02% S= 4.21% 35. S=radioactive 36 S=0.02% S= 0.75% 34 VI V IV III II I 0 -I -II SO 4 S 2 O 6 H 2 SO 3 HS 2 O 4- S 2 O 3
 SULFUR ISOTOPES 32 S= 95.02% 33 S= 0.75% 34 S= 4.21% 35 S=radioactive 36 S=0.02% S-H S-C S=C S-O S=O S-F S-Cl S-S VI V IV III II I 0 -I -II SO 4 2- S 2 O 6 2- H 2 SO 3 HS 2 O 4- S 2 O 3 2- S 2 F 2 S H
SULFUR ISOTOPES 32 S= 95.02% 33 S= 0.75% 34 S= 4.21% 35 S=radioactive 36 S=0.02% S-H S-C S=C S-O S=O S-F S-Cl S-S VI V IV III II I 0 -I -II SO 4 2- S 2 O 6 2- H 2 SO 3 HS 2 O 4- S 2 O 3 2- S 2 F 2 S H
Lecture 13. Hydrothermal Circulation
 Lecture 13. Hydrothermal Circulation The discovery of hot springs on the ocean floor during the 1970s was one of the most exciting events in the history of oceanography. Although hydrothermal activity
Lecture 13. Hydrothermal Circulation The discovery of hot springs on the ocean floor during the 1970s was one of the most exciting events in the history of oceanography. Although hydrothermal activity
Activity 1-2: Origin of the Earth
 Earth Science 11 Name: Block: Activity 1-2: Origin of the Earth Read the following passage, and then answer the questions at the end: Where Earth Science Begins: The Solar System Where shall the study
Earth Science 11 Name: Block: Activity 1-2: Origin of the Earth Read the following passage, and then answer the questions at the end: Where Earth Science Begins: The Solar System Where shall the study
California Science Content Standards Chemistry Grades 9-12
 California Science Content Standards Chemistry Grades 9-12 Standards that all students are expected to achieve in the course of their studies are unmarked. Standards that all students should have the opportunity
California Science Content Standards Chemistry Grades 9-12 Standards that all students are expected to achieve in the course of their studies are unmarked. Standards that all students should have the opportunity
PhET Interactive Chemistry Simulations Aligned to an Example General Chemistry Curriculum
 PhET Interactive Chemistry Simulations Aligned to an Example General Chemistry Curriculum Alignment is based on the topics and subtopics addressed by each sim. Sims that directly address the topic area
PhET Interactive Chemistry Simulations Aligned to an Example General Chemistry Curriculum Alignment is based on the topics and subtopics addressed by each sim. Sims that directly address the topic area
Edexcel Chemistry Checklist
 Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
What is Earth Science? Earth science is the branch of science dealing with the constitution of the earth and its atmosphere.
 What is Earth Science? Earth science is the branch of science dealing with the constitution of the earth and its atmosphere. 1 2 The Branches of Earth Science Major Astronomy (celestial objects, space,
What is Earth Science? Earth science is the branch of science dealing with the constitution of the earth and its atmosphere. 1 2 The Branches of Earth Science Major Astronomy (celestial objects, space,
Lecture 6 - Determinants of Seawater Composition. Sets up electric dipole because O is more electronegative A o. Figure 3.
 12.742 - Marine Chemistry Fall 2004 Lecture 6 - Determinants of Seawater Composition Prof. Scott Doney What is seawater? Water Dissolved inorganic salts (major ions) Trace species, organics, colloids,
12.742 - Marine Chemistry Fall 2004 Lecture 6 - Determinants of Seawater Composition Prof. Scott Doney What is seawater? Water Dissolved inorganic salts (major ions) Trace species, organics, colloids,
Chemistry. Atomic and Molecular Structure
 Chemistry Atomic and Molecular Structure 1. The periodic table displays the elements in increasing atomic number and shows how periodicity of the physical and chemical properties of the elements relates
Chemistry Atomic and Molecular Structure 1. The periodic table displays the elements in increasing atomic number and shows how periodicity of the physical and chemical properties of the elements relates
CHAPTER. 3 Earth s Environmental Systems
 CHAPTER 3 Earth s Environmental Systems The Gulf of Mexico s Dead Zone Nutrient-rich runoff causes plankton blooms and hypoxia low oxygen levels in the Gulf of Mexico. Hypoxia kills or displaces marine
CHAPTER 3 Earth s Environmental Systems The Gulf of Mexico s Dead Zone Nutrient-rich runoff causes plankton blooms and hypoxia low oxygen levels in the Gulf of Mexico. Hypoxia kills or displaces marine
Worked Example of Batch Melting: Rb and Sr
 Worked Example of Batch Melting: Rb and Sr Basalt with the mode: Table 9.2. Conversion from mode to weight percent Mineral Mode Density Wt prop Wt% ol 15 3.6 54 0.18 cpx 33 3.4 112.2 0.37 plag 51 2.7 137.7
Worked Example of Batch Melting: Rb and Sr Basalt with the mode: Table 9.2. Conversion from mode to weight percent Mineral Mode Density Wt prop Wt% ol 15 3.6 54 0.18 cpx 33 3.4 112.2 0.37 plag 51 2.7 137.7
Ideal Gas Laws Empirical Gas Laws The Mole Equations of State Dalton's Law The Mole Fraction Extensive and Intensive Variables Graham's Law of
 Ideal Gas Laws Empirical Gas Laws The Mole Equations of State Dalton's Law The Mole Fraction Extensive and Intensive Variables Graham's Law of Effusion The Maxwell-Boltzmann Distribution A Digression on
Ideal Gas Laws Empirical Gas Laws The Mole Equations of State Dalton's Law The Mole Fraction Extensive and Intensive Variables Graham's Law of Effusion The Maxwell-Boltzmann Distribution A Digression on
Course Title: Academic chemistry Topic/Concept: Chapter 1 Time Allotment: 11 day Unit Sequence: 1 Major Concepts to be learned:
 Course Title: Academic chemistry Topic/Concept: Chapter 1 Time Allotment: 11 day Unit Sequence: 1 1. Nature of chemistry 2. Nature of measurement 1. Identify laboratory equipment found in the lab drawer
Course Title: Academic chemistry Topic/Concept: Chapter 1 Time Allotment: 11 day Unit Sequence: 1 1. Nature of chemistry 2. Nature of measurement 1. Identify laboratory equipment found in the lab drawer
Environmental Isotopes in Hydrology. Woocay substituting for Walton
 Environmental Isotopes in Hydrology Oct 7, 2010 1 What is an Isotope? An element is defined by the number of protons (Z) in the nucleus The number of neutrons (N) defines the isotope(s) of that element
Environmental Isotopes in Hydrology Oct 7, 2010 1 What is an Isotope? An element is defined by the number of protons (Z) in the nucleus The number of neutrons (N) defines the isotope(s) of that element
Carbon Cycling Internal
 Carbon Cycling Internal The 4 subcycles Atmosphere The Earth s Atmosphere The Earth has a radius of some 6400 km. Ninety-nine percent of the earth's atmosphere is contained within a layer approximately
Carbon Cycling Internal The 4 subcycles Atmosphere The Earth s Atmosphere The Earth has a radius of some 6400 km. Ninety-nine percent of the earth's atmosphere is contained within a layer approximately
Cherokee High School. Class Syllabus
 Cherokee High School Class Syllabus 2017-2018 Teacher: Lynne Styke lynne.styke@hck12.net Course Title: AP Chemistry Course Description This AP Chemistry course is designed to be the equivalent of the general
Cherokee High School Class Syllabus 2017-2018 Teacher: Lynne Styke lynne.styke@hck12.net Course Title: AP Chemistry Course Description This AP Chemistry course is designed to be the equivalent of the general
CHAPTER GENERAL CONCEPTS READING from text HOMEWORK from text OTHER ASSIGNMENTS LABORATORIES. R1: Sections 1,2 Set 1: CR 15; PP 1
 MONTPELIER HIGH SCHOOL teacher: Toni Ceckler / Room 220, phone: 225-8182, email: toni@mpsvt.org CHEMISTRY 2018-2019 GENERAL SYLLABUS Text: Chemistry: Connections to Our Changing World (Prentice Hall, 2000)
MONTPELIER HIGH SCHOOL teacher: Toni Ceckler / Room 220, phone: 225-8182, email: toni@mpsvt.org CHEMISTRY 2018-2019 GENERAL SYLLABUS Text: Chemistry: Connections to Our Changing World (Prentice Hall, 2000)
Halogen and argon evidence of Martian hydrous fluids in nakhlite meteorites Ray Burgess
 Halogen and argon evidence of Martian hydrous fluids in nakhlite meteorites Ray Burgess School of Earth, Atmospheric and Environmental Sciences University of Manchester, UK Topics Halogens and noble gases
Halogen and argon evidence of Martian hydrous fluids in nakhlite meteorites Ray Burgess School of Earth, Atmospheric and Environmental Sciences University of Manchester, UK Topics Halogens and noble gases
Origin of the Solar System
 Origin of the Solar System Look for General Properties Dynamical Regularities Orbits in plane, nearly circular Orbit sun in same direction (CCW from N.P.) Rotation Axes to orbit plane (Sun & most planets;
Origin of the Solar System Look for General Properties Dynamical Regularities Orbits in plane, nearly circular Orbit sun in same direction (CCW from N.P.) Rotation Axes to orbit plane (Sun & most planets;
b. atomic mass H What is the density of an object with a volume of 15cm 3 and a mass of 45g?
 Name Period Date Earth Science Midterm Review 2015-2016 Quarter 1 Review Assign #1 Basic Chemistry An atom is a basic chemical building block of matter. An atom consists of protons, neutrons, and electrons.
Name Period Date Earth Science Midterm Review 2015-2016 Quarter 1 Review Assign #1 Basic Chemistry An atom is a basic chemical building block of matter. An atom consists of protons, neutrons, and electrons.
Sedimentary Geology. Strat and Sed, Ch. 1 1
 Sedimentary Geology Strat and Sed, Ch. 1 1 Sedimentology vs. Stratigraphy Sedimentology is the study of the origin and classification of sediments and sedimentary rocks Mostly the physical and chemical
Sedimentary Geology Strat and Sed, Ch. 1 1 Sedimentology vs. Stratigraphy Sedimentology is the study of the origin and classification of sediments and sedimentary rocks Mostly the physical and chemical
Describe how the inter-conversion of solids, liquids and gases are achieved and recall names used for these inter-conversions
 Understand the arrangements, movements and energy of the particle in each of the 3 states of matter : solid, liquid and gas Describe how the inter-conversion of solids, liquids and gases are achieved and
Understand the arrangements, movements and energy of the particle in each of the 3 states of matter : solid, liquid and gas Describe how the inter-conversion of solids, liquids and gases are achieved and
Chapter 11 Properties of Solutions
 Chapter 11 Properties of Solutions Solutions Homogeneous mixtures of two or more substances Composition is uniform throughout the sample No chemical reaction between the components of the mixture Solvents
Chapter 11 Properties of Solutions Solutions Homogeneous mixtures of two or more substances Composition is uniform throughout the sample No chemical reaction between the components of the mixture Solvents
OUTCOMES BASED LEARNING MATRIX. Course: Physical Geology Department: _Physical Science. Study the text and lecture material
 OUTCOMES BASED LEARNING MATRIX Course: Physical Geology Department: _Physical Science Physical Geology This course is intended to acquaint students with the physical structure of the Earth, the nature
OUTCOMES BASED LEARNING MATRIX Course: Physical Geology Department: _Physical Science Physical Geology This course is intended to acquaint students with the physical structure of the Earth, the nature
Drawing from:
 California State Science Standards Adopted 1997 Grades 6- Drawing from: http://www.cde.ca.gov/ci/sc/cf/documents/scienceframework.pdf ===================== 6 th Grade Standard Set 1 Plate Tectonics and
California State Science Standards Adopted 1997 Grades 6- Drawing from: http://www.cde.ca.gov/ci/sc/cf/documents/scienceframework.pdf ===================== 6 th Grade Standard Set 1 Plate Tectonics and
Norwich City Schools AP Chemistry
 Topic: Structure of Matter Students will use inquiry based methods to investigate scientific concepts Students will examine and investigate the structure, properties, and interactions of matter. Students
Topic: Structure of Matter Students will use inquiry based methods to investigate scientific concepts Students will examine and investigate the structure, properties, and interactions of matter. Students
Dating. AST111 Lecture 8a. Isotopic composition Radioactive dating
 Dating Martian Lafayette Asteroid with patterns caused by the passaged through the atmosphere. Line on the fusion crust were caused by beads of molten rock. AST111 Lecture 8a Isotopic composition Radioactive
Dating Martian Lafayette Asteroid with patterns caused by the passaged through the atmosphere. Line on the fusion crust were caused by beads of molten rock. AST111 Lecture 8a Isotopic composition Radioactive
(4) Give an example of important reactions that are responsible for the composition of river water.
 Lecture 12 Global Biogeochemical Cycles (1) If rivers are the chief source of the dissolved salts in seawater, why is seawater not simply a concentrated version of average composition of all rivers? The
Lecture 12 Global Biogeochemical Cycles (1) If rivers are the chief source of the dissolved salts in seawater, why is seawater not simply a concentrated version of average composition of all rivers? The
Figure 65: Reservoir in a steady state condition where the input flux is equal to the output flux and the reservoir size remains constant.
 7. The carbon cycle 7.1. Box model of the carbon cycle Without the greenhouse effect, our planet would experience a permanent ice age and life as we know it would not be possible. The main contributors
7. The carbon cycle 7.1. Box model of the carbon cycle Without the greenhouse effect, our planet would experience a permanent ice age and life as we know it would not be possible. The main contributors
Chapter Chemical Elements Matter solid, liquid, and gas elements atoms. atomic symbol protons, neutrons, electrons. atomic mass atomic number
 Chapter 2 2.1 Chemical Elements 1. Matter is defined as anything that takes up space and has mass. 2. Matter exists in three states: solid, liquid, and gas. A. Elements 1. All matter (both living and non-living)
Chapter 2 2.1 Chemical Elements 1. Matter is defined as anything that takes up space and has mass. 2. Matter exists in three states: solid, liquid, and gas. A. Elements 1. All matter (both living and non-living)
Lecture 6 - Igneous Rocks and Volcanoes
 Lecture 6 - Igneous Rocks and Volcanoes Learning objectives Understand and be able to predict where and why magma will be forming at different tectonic settings Understand the factors controlling magma
Lecture 6 - Igneous Rocks and Volcanoes Learning objectives Understand and be able to predict where and why magma will be forming at different tectonic settings Understand the factors controlling magma
Evolution of the Earth
 Evolution of the Earth http://static.newworldencyclopedia.org/f/fe/geologic_clock.jpg Evolution of the Earth Solar system, 4.6 byr Collapse of a nebula Star forms as gravity concentrates material at center
Evolution of the Earth http://static.newworldencyclopedia.org/f/fe/geologic_clock.jpg Evolution of the Earth Solar system, 4.6 byr Collapse of a nebula Star forms as gravity concentrates material at center
REFERENCE: The Blue Planet An Introduction to Earth System Science. Brian J. Skinner and Barbara W. Murck (2011) Third Edition. John Wiley and Sons
 REFERENCE: The Blue Planet An Introduction to Earth System Science. Brian J. Skinner and Barbara W. Murck (2011) Third Edition. John Wiley and Sons Inc. is substance It has mass and occupies space. The
REFERENCE: The Blue Planet An Introduction to Earth System Science. Brian J. Skinner and Barbara W. Murck (2011) Third Edition. John Wiley and Sons Inc. is substance It has mass and occupies space. The
Solapur University, Solapur. Syllabus for B.Sc. II- Geochemistry - (IDS) Semester System - CGPA To be implemented from Academic Year
 Solapur University, Solapur Syllabus for B.Sc. II- Geochemistry - (IDS) Semester System - CGPA To be implemented from Academic Year- 2015-16 Course Structure Total Credit 16 - (Theory (4 x 3) = 12+Practical
Solapur University, Solapur Syllabus for B.Sc. II- Geochemistry - (IDS) Semester System - CGPA To be implemented from Academic Year- 2015-16 Course Structure Total Credit 16 - (Theory (4 x 3) = 12+Practical
2016 PLED 576 Earth and Space Science Correlation to Next Generation Science Standards (NGSS) High School Earth and Space Science
 2016 PLED 576 Earth and Space Science Correlation to Next Generation Science Standards () High School Earth and Space Science Course Objectives 1. Evaluate the differing theories that explain the structure,
2016 PLED 576 Earth and Space Science Correlation to Next Generation Science Standards () High School Earth and Space Science Course Objectives 1. Evaluate the differing theories that explain the structure,
GEOCHEMISTRY, GROUNDWATER AND POLLUTION,
 GEOCHEMISTRY, GROUNDWATER AND POLLUTION, 2 ND EDITION C.A.J. APPELO Hydrochemical Consultant, Amsterdam, the Netherlands D. POSTMA Environment & Resources DTU, Technical University of Denmark, Kgs. Lyngby,
GEOCHEMISTRY, GROUNDWATER AND POLLUTION, 2 ND EDITION C.A.J. APPELO Hydrochemical Consultant, Amsterdam, the Netherlands D. POSTMA Environment & Resources DTU, Technical University of Denmark, Kgs. Lyngby,
EOC Study Guide Honors
 Name Date Science. SC.912.E.5.1 Cite evidence used to develop and verify the scientific theory of the Big Bang (also known as the Big Bang Theory) of the origin of the universe. 1. What is the approximate
Name Date Science. SC.912.E.5.1 Cite evidence used to develop and verify the scientific theory of the Big Bang (also known as the Big Bang Theory) of the origin of the universe. 1. What is the approximate
TOPIC 1: RELATIVE DATING ESSENTIAL QUESTION: HOW DO WE DETERMINE A ROCK S AGE BY THE SURROUNDING ROCKS?
 TOPIC 1: RELATIVE DATING ESSENTIAL QUESTION: HOW DO WE DETERMINE A ROCK S AGE BY THE SURROUNDING ROCKS? TOPIC 1: RELATIVE DATING UNIFORMITARIANISM: THE IDEA THAT THE SAME FORCES HAVE BEEN AND CONTINUE
TOPIC 1: RELATIVE DATING ESSENTIAL QUESTION: HOW DO WE DETERMINE A ROCK S AGE BY THE SURROUNDING ROCKS? TOPIC 1: RELATIVE DATING UNIFORMITARIANISM: THE IDEA THAT THE SAME FORCES HAVE BEEN AND CONTINUE
EASC100: EXAM1 (07) Lithosphere and Hydrosphere
 EASC100: EXAM1 (07) Lithosphere and Hydrosphere Name: 1. Why do Mercury and the Moon lack an atmosphere? A) They formed after all the gas had been used up. B) They are so cold that all their gases have
EASC100: EXAM1 (07) Lithosphere and Hydrosphere Name: 1. Why do Mercury and the Moon lack an atmosphere? A) They formed after all the gas had been used up. B) They are so cold that all their gases have
The Biogeochemical Carbon Cycle: CO 2,the greenhouse effect, & climate feedbacks. Assigned Reading: Kump et al. (1999) The Earth System, Chap. 7.
 The Biogeochemical Carbon Cycle: CO 2,the greenhouse effect, & climate feedbacks Assigned Reading: Kump et al. (1999) The Earth System, Chap. 7. Overhead Transparencies Faint Faint Young Sun Paradox Young
The Biogeochemical Carbon Cycle: CO 2,the greenhouse effect, & climate feedbacks Assigned Reading: Kump et al. (1999) The Earth System, Chap. 7. Overhead Transparencies Faint Faint Young Sun Paradox Young
Phys 214. Planets and Life
 Phys 214. Planets and Life Dr. Cristina Buzea Department of Physics Room 259 E-mail: cristi@physics.queensu.ca (Please use PHYS214 in e-mail subject) Lecture 10. Geology and life. Part 1 (Page 99-123)
Phys 214. Planets and Life Dr. Cristina Buzea Department of Physics Room 259 E-mail: cristi@physics.queensu.ca (Please use PHYS214 in e-mail subject) Lecture 10. Geology and life. Part 1 (Page 99-123)
Unit 3. Atoms and molecules
 Unit 3. Atoms and molecules Index. s and compounds...2.. Dalton's Atomic theory...2 2.-The atom...2 3.-Atomic number and mass number...2 4.-Isotopes, atomic mass unit and atomic mass...3 5.- configuration...3
Unit 3. Atoms and molecules Index. s and compounds...2.. Dalton's Atomic theory...2 2.-The atom...2 3.-Atomic number and mass number...2 4.-Isotopes, atomic mass unit and atomic mass...3 5.- configuration...3
