Name. Pre-Test. Directions: Circle the T if a statement is true or F if it is false (F). 1. Matter always stays the same.
|
|
|
- Christina Robinson
- 6 years ago
- Views:
Transcription
1 1 Pre-est Directions: Circle the if a statement is true or if it is false (). 1. Matter always stays the same. 2. A physical change is a change in size, shape or phase of matter, without the matter changing chemically.. 3. emperature can play a big role in physical changes. 4. In a physical change, matter cannot change its shape. 5. he ability to burn is a chemical property called flammability. 6. A chemical change is a process by which a certain substance becomes a different substance. 7. In a chemical change, the same substance is formed. 8. Chemical changes occur at the same speeds. 9. Chemical reactions occur when two or more substances combine to form a new substance. 10. Photosynthesis is an important chemical reaction on earth.
2 2 Video Quiz Directions: ill in the blank with the correct word from the list at the bottom of the page. Not all words from the list will be used. 1. Ice being broken into small bits is an example of a change. 2. In a physical change the matter does not change. 3. In a chemical change a substance is formed. 4. A chemical is a way to describe how matter reacts with other matter. 5. lammability is the chemical property describing how a substance. 6. A clue that a chemical change is occurring in a burning flame is that is given off. 7. Chemical changes occur at speeds. 8. is the substance produced by iron reacting with oxygen in the air. 9. A chemical is the chemical change in which one or more substances change. 10. is a chemical reaction in green plants that produces energy and oxygen. acetic acid burns chemically decomposition different energy flammability freezes heat new old photosynthesis physical property reaction rust sodium bicarbonate solid
3 3 Discussion Questions Directions: Answer the following questions in the space provided or out loud. 1. What is a physical change? 2. What are some examples of physical changes in matter? 3. What is a chemical change? 4. What are some examples of chemical changes in matter? 5. What is a chemical reaction? 6. Write out an example of a chemical reaction. 7. What are some physical changes that occur at school? At home? In nature? 8. What are some chemical changes that occur at school? At home? In nature?
4 4 Vocabulary Exercise Direction: Match the vocabulary word with its correct definition by drawing a line from the word to the definition. physical change A. A chemical property describing how a substance burns. chemical property B. A change in the size, shape, or phase of matter, without the matter changing chemically. chemical change C. Process of plants producing energy, oxygen, and water by taking in carbon dioxide, water, and the sun s energy. rusting D. A way to describe how matter reacts with other matter. chemical reactions E. A chemical change in which one or more substances is changed. flammability. When iron reacts with oxygen, it corrodes to form a new substance, and is an example of a slow chemical change. photosynthesis G. he process by which a certain substance becomes a different substance.
5 5a Observing Changes in Matter Objective In this activity, you will create different changes, and identify them as physical or chemical. Materials Data able safety glasses paper baking soda vinegar dropper limestone salt container of water pencil pencil sharpener ph paper rusty piece of iron non-rusty piece of iron clay ice cube ant-acid tablets Procedure 1. In this activity, you will create both physical and chemical changes. Before you begin the activity, put on the safety glasses provided by your teacher, and wear them during the entire activity. 2. Your teacher has set up several stations throughout the classroom. At each station, you will observe or create a change. Perform the task, and decide if the change is a physical change or a chemical change, then fill in your answer in the data table. Station 1 - A piece of paper is provided in this station. ear the paper into many smaller pieces. Is this a physical or chemical change? Station 2 - A small amount of baking soda and a dropper with vinegar is at this station. Place small drops of vinegar onto the baking soda. Record your observations. Is this a physical or chemical change? Station 3 - At this station, a small white rock, called limestone, is provided, as well as a dropper and vinegar. Place a drop of vinegar on the limestone and record your observation. Is this a physical or chemical change? Station 4 - A teaspoon of salt, a glass of water, and a spoon are provided. Stir the salt into the water. Is this a physical or chemical change?
6 5b Observing Changes in Matter Station 5 - ake the pencil provided and sharpen it at the pencil sharpener. Is this a physical or chemical change? Station 6 - A small piece of ph paper is provided. his is special paper used to determine acidic or basic a substance is. Place the tip of the paper in the vinegar solution. Observe any color change on the paper. Is this a physical or chemical change? Station 7 - wo pieces of iron are at this station. One is a silvery color; the other piece of iron has been left in the air for a long period of time, and is an orange color. Has this piece of iron undergone a physical or chemical reaction? Station 8 - Mold the piece into the shape of a snake. Is this a physical or chemical change? Station 9 - Sitting in a cup are several ice cubes. As these ice cubes are exposed to the air in your classroom, are they undergoing a physical or chemical change? Station 10 - A white ant-acid tablet is provided, along with a cup of water. Place the table in the water, and observe the reaction. Is this a physical or chemical change? Data able (next page) Conclusion As a class, discuss any problems you had identifying the different types of changes. Discuss any other common changes that take place in the classroom on a daily basis. Discuss the clues you used to decide whether the change was a physical change or a chemical change.
7 5c Observing Changes in Matter Data able Station Number Observation Physical or Chemical Change? 1 - Paper 2 - Baking soda and vinegar 3 - Limestone and vinegar 4 - Salt and water 5 - Pencil and sharpener 6 - ph paper and vinegar 7 - two pieces of iron 8 - clay 9 - ice cubes 10 - Ant-acid tablet and water
8 6a Observing Changes in Your Environment Objective In this activity, you will identify the many different physical and chemical changes that occur in your home, school, and neighborhood. Materials Data able pencil Procedure 1. Believe it or not, there are dozens, if not hundreds, of physical and chemical changes that occur around you everyday. or example, on your way to school you may have traveled on a bus or in a car. he engines in these vehicles used a chemical change involving gas or diesel fuel to make the vehicles run. Perhaps you cut a sandwich in half before eating your lunch; this was a physical change you created in the sandwich. 2. Using the Data able on the next page, record the physical and chemical changes in your environment. In the first column, record the change; in the second column, record your observations; and in the third column, state if the change is a physical change or a chemical change. ry to identify at least ten physical and chemical changes. he first one is done for you. Have fun! Data able (next page) Conclusion After completing the exercise, compile a class list of physical and chemical changes you observed. Discuss these observations as a class. Point out any errors in the list. Decide which changes are the most important, useful, and interesting.
9 6b Observing Changes in Your Environment Data able Change Observations Chemical or Physical Change? branch falling off tree branch made a cracking sound as it broke off main trunk of tree physical change
10 7 Writing Chemical Reactions We experience many chemical reactions everyday. A chemical reaction is a change in which two or more substances become new substances. Cooking our food often involves chemical changes, as does burning a candle or fire wood. We can observe chemical reactions by noting changes in color and/or temperature, the presence of bubbles, and the actual chemical change of a substance. Chemical reactions can be described in words and addition signs, the same addition signs you use in math to show that two numbers are added together. In a chemical reaction, the beginning compounds, or the reactants, that are present before the reaction occurs are written on the left side of the reaction. he new substances produced as a result of the reaction are written on the right side of the reaction. An arrow is written between the reactants and products. ollowing is an example of how to write the reaction between sodium and chlorine to produce sodium chloride (salt): sodium + chlorine sodium chloride Directions: Below are five chemical reactions. Describe the chemical reactions using words, addition signs, and arrows. 1. Photosynthesis involves plants taking in carbon dioxide, water, and the sun s energy to produce oxygen, water, and energy in the form of sugar. 2. When we breathe, we take in oxygen and water vapor, and give off carbon dioxide and water vapor. Directions: Using the information provided in the video, and hints from your teacher, symbolize the chemical reactions for the following, using words, addition signs, and arrows. 3. Symbolize the reaction of burning a piece of paper. 4. Symbolize the reaction of the rusting of a piece of iron. 5. Symbolize the he reaction that occurs between baking soda and vinegar.
11 8 Post - est Directions: ill in the blank with the correct word and or answer the questions if true or if false. Each question is worth 10 points. 1. Ice melting is an example of a change. 2. does not always stay the same, and can undergo physical and/ or chemical changes. 3. can play a large role in the physical change of matter. 4. In a chemical change, a substance if formed. 5. describes the ability of a substance to burn. 6. Heat is a clue that chemical change is occurring. 7. Rust is a substance produced by iron reacting with water. 8. Photosynthesis is a chemical reaction in green plants that produces energy and oxygen. 9. All chemical changes occur at the same speeds. 10. Every time you move, a chemical reaction occurs.
Physical and Chemical Changes
 Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
Physical & Chemical PROPERTIES
 Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
1 The Nature of Chemical Reactions
 CHAPTER 7 1 The Nature of Chemical Reactions SECTION Chemical Reactions KEY IDEAS As you read this section, keep these questions in mind: What happens in a chemical reaction? What is the role of energy
CHAPTER 7 1 The Nature of Chemical Reactions SECTION Chemical Reactions KEY IDEAS As you read this section, keep these questions in mind: What happens in a chemical reaction? What is the role of energy
Lesson 1: What are chemical changes?
 Lesson 1 Summary Lesson 1: What are chemical changes? Vocabulary Use with pp. 375 377 physical change a change in which the material keeps its identify chemical change a change in which one substance or
Lesson 1 Summary Lesson 1: What are chemical changes? Vocabulary Use with pp. 375 377 physical change a change in which the material keeps its identify chemical change a change in which one substance or
3.3. Physical and Chemical Changes. Before You Read. What is a physical change? What is a chemical change?
 Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment
 Science 24: Day 10 1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment https://www.youtube.com/watch?v=fofpjj7v414 Which reaction was
Science 24: Day 10 1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment https://www.youtube.com/watch?v=fofpjj7v414 Which reaction was
Classification of Matter
 Matter in Chemistry Classification of Matter Elements: substances that are made from one type of atom only. Compounds: substances that are made from more than one type of atom chemically bonded together.
Matter in Chemistry Classification of Matter Elements: substances that are made from one type of atom only. Compounds: substances that are made from more than one type of atom chemically bonded together.
Changes in Matter Study Guide
 Changes in Matter Study Guide Define the following and give a real world example: 1. : A change in matter that requires the substances to absorb energy Example: Photosynthesis (the plants absorb light
Changes in Matter Study Guide Define the following and give a real world example: 1. : A change in matter that requires the substances to absorb energy Example: Photosynthesis (the plants absorb light
CHEM1301. F2014 Chapter 1 and 3
 CHEM1301 F2014 Chapter 1 and 3 Chemistry:Why am I here? Chemistry The science that seeks to understand the behavior of matter by studying the BEHAVIOR of atoms and molecules. Not just a cause and effect
CHEM1301 F2014 Chapter 1 and 3 Chemistry:Why am I here? Chemistry The science that seeks to understand the behavior of matter by studying the BEHAVIOR of atoms and molecules. Not just a cause and effect
Name: 1. Which of the following is probably true about 300 ml of sand and 300 ml of water?
 Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Compound. Math Focus. What are compounds? What is a chemical reaction? How are compounds used in everyday life?
 CHAPTER 3 2 Compounds SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What are compounds? What is a chemical reaction?
CHAPTER 3 2 Compounds SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What are compounds? What is a chemical reaction?
Observing Chemical Change - 5.1
 Observing Chemical Change - 5.1 Vocabulary: Physical change - Chemical change - Reactant Product Precipitate Exothermic reaction - Endothermic reaction - Properties of matter: Two kinds of properties of
Observing Chemical Change - 5.1 Vocabulary: Physical change - Chemical change - Reactant Product Precipitate Exothermic reaction - Endothermic reaction - Properties of matter: Two kinds of properties of
Grade 8 Science Unit 2 Test» Form A (Master Copy) Directions: Please choose the best answer choice for each of the following questions.
 Directions: Please choose the best answer choice for each of the following questions. 1. Which of the following could be a molecule BUT not a compound? a pair of atoms of the same element a collection
Directions: Please choose the best answer choice for each of the following questions. 1. Which of the following could be a molecule BUT not a compound? a pair of atoms of the same element a collection
Section 2: Properties of Matter
 Section 2: A physical property can be observed without changing the identity of the material; a chemical property can be observed when one or more new substances are formed. K What I Know W What I Want
Section 2: A physical property can be observed without changing the identity of the material; a chemical property can be observed when one or more new substances are formed. K What I Know W What I Want
1º ESO UNIT 4: Chemical and physical changes. Susana Morales Bernal
 1º ESO UNIT 4: Chemical and physical changes Objectives 1. To know the basic characteristics of chemical reactions. 2. To know the differences between physical changes and chemical changes. 3. To know
1º ESO UNIT 4: Chemical and physical changes Objectives 1. To know the basic characteristics of chemical reactions. 2. To know the differences between physical changes and chemical changes. 3. To know
Chapter 2. Section 1
 Chapter 2 Section 1 Describing Matter Properties used to describe matter can be classified as extensive or intensive Extensive property - depends on the amount of matter in a sample. Ex. Mass and Volume
Chapter 2 Section 1 Describing Matter Properties used to describe matter can be classified as extensive or intensive Extensive property - depends on the amount of matter in a sample. Ex. Mass and Volume
Physical Changes can be observed without changing the identity of the substance (often states of matter changes).
 Physical Changes can be observed without changing the identity of the substance (often states of matter changes). Remember: States of matter changes are physical changes. The identify of the substance
Physical Changes can be observed without changing the identity of the substance (often states of matter changes). Remember: States of matter changes are physical changes. The identify of the substance
Compounds. Elements. Elements in a Compound. Matter. Pure Substances. Basic Chemistry. Chapter 3 Lecture. 3.1 Classification of Matter
 Chapter 3 Lecture Chapter 3 and Energy 3.1 Classification of Fifth Edition is the material that makes up all things is anything that has mass and occupies space Learning Goal Classify examples of matter
Chapter 3 Lecture Chapter 3 and Energy 3.1 Classification of Fifth Edition is the material that makes up all things is anything that has mass and occupies space Learning Goal Classify examples of matter
Matter and Change. Monday Tuesday Wednesday Thursday Friday. Textbook Activity. Meet in B122. Hw: pg 7 Hw: pg 4. Matter Review
 Matter and Change Monday Tuesday Wednesday Thursday Friday SEPTEMBER 1 2 3 4 5 NO SCHOOL Matter & Change Classifying Matter Textbook Activity Meet in B122 Physical and Chemical Properties and Changes Indiana
Matter and Change Monday Tuesday Wednesday Thursday Friday SEPTEMBER 1 2 3 4 5 NO SCHOOL Matter & Change Classifying Matter Textbook Activity Meet in B122 Physical and Chemical Properties and Changes Indiana
Matter and Its Changes
 Lesson 2 Matter and Its Changes 6.NS.3, 6.NS.5, 6.NS.6, 6.NS.7, 6.NS.10, 6.1.1, 6.1.2, 6.1.3 Skim or scan the heading, boldfaced words, and pictures in the lesson. Identify or predict three facts you will
Lesson 2 Matter and Its Changes 6.NS.3, 6.NS.5, 6.NS.6, 6.NS.7, 6.NS.10, 6.1.1, 6.1.2, 6.1.3 Skim or scan the heading, boldfaced words, and pictures in the lesson. Identify or predict three facts you will
Page 1 / 12. Chemistry Exam. Name: Matter Properties, Structure. Question 1 (1 point) The atomic number of an atom is. A. The mass of the atom.
 Chemistry Exam Matter Properties, Structure Name: Question 1 (1 point) The atomic number of an atom is A. The mass of the atom. B. The number of protons added to the number of neutrons in the nucleus.
Chemistry Exam Matter Properties, Structure Name: Question 1 (1 point) The atomic number of an atom is A. The mass of the atom. B. The number of protons added to the number of neutrons in the nucleus.
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED)
 CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
3 Chemical Properties
 CHAPTER 7 3 Chemical Properties SECTION The Properties of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What are chemical properties of matter? What
CHAPTER 7 3 Chemical Properties SECTION The Properties of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What are chemical properties of matter? What
2.3 CHEMICAL PROPERTIES
 2.3 CHEMICAL PROPERTIES n Vocabulary: n chemical property n flammability n reactivity n chemical change n precipitate n Objectives: n Describe chemical properties of matter. n Describe clues that indicate
2.3 CHEMICAL PROPERTIES n Vocabulary: n chemical property n flammability n reactivity n chemical change n precipitate n Objectives: n Describe chemical properties of matter. n Describe clues that indicate
Post-Show. Chemistry. Periodic Table of the Elements. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
STEMscopedia: PHYSICAL AND CHEMICAL PROPERTIES AND CHANGES 8P1CD
 Reflect STEMscopedia: PHYSICAL AND CHEMICAL 8P1CD What do ice cream, root beer, and carbon dioxide gas have in common? Not only do these ingredients combine to make a good treat on a hot summer day, but
Reflect STEMscopedia: PHYSICAL AND CHEMICAL 8P1CD What do ice cream, root beer, and carbon dioxide gas have in common? Not only do these ingredients combine to make a good treat on a hot summer day, but
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions
 Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Section 1: What is a Chemical Reaction
 Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Matter, mass, and volume are related.
 Suppose you had all of the parts needed to make an at home aquarium: a tank, water, rocks, plants, and some fish. To put the aquarium together, you would need to arrange the rocks and plants in the tank.
Suppose you had all of the parts needed to make an at home aquarium: a tank, water, rocks, plants, and some fish. To put the aquarium together, you would need to arrange the rocks and plants in the tank.
by Cyndee Crawford October 2014
 7-5 Chemical Nature of Matter PART 2 by Cyndee Crawford October 2014 7-5 The student will demonstrate an understanding of the classifications and properties of matter and the changes that matter undergoes.
7-5 Chemical Nature of Matter PART 2 by Cyndee Crawford October 2014 7-5 The student will demonstrate an understanding of the classifications and properties of matter and the changes that matter undergoes.
3. Which of the following would create a chemical change when it is added to a glass of warm milk?
 Test Name: Science 8 Assessment 6 ReviewScan Test Code: 9668632 Date: Your Name: Teacher: Class Name: 1. Which step in these instructions for making scrambled eggs results in a chemical change? First,
Test Name: Science 8 Assessment 6 ReviewScan Test Code: 9668632 Date: Your Name: Teacher: Class Name: 1. Which step in these instructions for making scrambled eggs results in a chemical change? First,
8.5E: Chemical Reactions
 Reflect Have you ever seen fireworks explode in the sky? If so, you may have observed a variety of different colors and shapes in the sky and watched with amazement as the colors changed overhead. These
Reflect Have you ever seen fireworks explode in the sky? If so, you may have observed a variety of different colors and shapes in the sky and watched with amazement as the colors changed overhead. These
Matter. Anything that has mass and takes up space. Solids Liquids Gases
 Introduction to Matter Matter Anything that has mass and takes up space Solids Liquids Gases Weight A measure of the gravitational force exerted on an object What is the difference between weight and mass?
Introduction to Matter Matter Anything that has mass and takes up space Solids Liquids Gases Weight A measure of the gravitational force exerted on an object What is the difference between weight and mass?
composition of matter, and the changes that matter undergoes. Examples of Uses of Chemistry in Everyday Life
 Name Matter and Change: Unit Objective Study Guide Date Due Directions: Write your answers to the following questions in the space provided. For problem solving, all of the work leading up to the final
Name Matter and Change: Unit Objective Study Guide Date Due Directions: Write your answers to the following questions in the space provided. For problem solving, all of the work leading up to the final
Name Date. Physical and Chemical Changes
 Name Date Physical and Chemical Changes Physical Changes Physical Change: A change in which no new substances form Do not change the type of matter an object is made of You change the shape, size or more
Name Date Physical and Chemical Changes Physical Changes Physical Change: A change in which no new substances form Do not change the type of matter an object is made of You change the shape, size or more
How are physical and chemical properties different?
 How are physical and chemical properties different? S8P1d. Distinguish between physical and chemical properties of matter as physical (i.e., density, melting point, boiling point) or chemical (i.e., reactivity,
How are physical and chemical properties different? S8P1d. Distinguish between physical and chemical properties of matter as physical (i.e., density, melting point, boiling point) or chemical (i.e., reactivity,
Unit 6M.3: Changing materials
 Unit 6M.3: Adding materials to water. Chemical reactions Skill you will use: Classifying Observing Predicting By the end of this unit you should: Know that when substances are added to water, some will
Unit 6M.3: Adding materials to water. Chemical reactions Skill you will use: Classifying Observing Predicting By the end of this unit you should: Know that when substances are added to water, some will
PROPERTIES OF MATTER STATION 1. Where did the water droplets on the outside of this cup come from? What phase change is this?
 PROPERTIES OF MATTER STATION 1 Where did the water droplets on the outside of this cup come from? What phase change is this? PROPERTIES OF MATTER STATION 2 Make a copy of this graph on your paper. Label
PROPERTIES OF MATTER STATION 1 Where did the water droplets on the outside of this cup come from? What phase change is this? PROPERTIES OF MATTER STATION 2 Make a copy of this graph on your paper. Label
Student Exploration: Chemical Changes
 Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
What Do You Think? Investigate GOALS
 Activity 1 Chemical and Physical Changes GOALS In this activity you will: Learn to differentiate between chemical and physical changes. Make observations and cite evidence to identify changes as chemical
Activity 1 Chemical and Physical Changes GOALS In this activity you will: Learn to differentiate between chemical and physical changes. Make observations and cite evidence to identify changes as chemical
Ashley Unit Conversions and Matter Chem 101
 1. Part 1: Unit Conversions Complete each of the following conversions to the proper number of significant figures : clearly show your set up with units in the set up and the answer. 1) 0.30 m = mm 2)
1. Part 1: Unit Conversions Complete each of the following conversions to the proper number of significant figures : clearly show your set up with units in the set up and the answer. 1) 0.30 m = mm 2)
Chemistry Review- Packet 11, Page 1
 Chemistry Review- Packet 11, Page 1 Atom- smallest particle of an element with the same properties as that element In size the entire atom has been thought to be approximately four-billionths of an inch,
Chemistry Review- Packet 11, Page 1 Atom- smallest particle of an element with the same properties as that element In size the entire atom has been thought to be approximately four-billionths of an inch,
Volume. measures how much space matter takes up. solubility. The amount of mass for an object is called. matter
 measures how much space matter takes up. Volume is the resistance of an object to sink, and it depends on. The ability of a substance to dissolve into another substance is called. The amount of mass for
measures how much space matter takes up. Volume is the resistance of an object to sink, and it depends on. The ability of a substance to dissolve into another substance is called. The amount of mass for
Matter has observable properties.
 KEY CONCEPT Matter has observable properties. BEFORE, you learned Matter has mass and volume Matter is made of atoms Matter exists in different states NOW, you will learn About physical and chemical properties
KEY CONCEPT Matter has observable properties. BEFORE, you learned Matter has mass and volume Matter is made of atoms Matter exists in different states NOW, you will learn About physical and chemical properties
Rainbow Ice. 3 small bowls or containers 3 small plastic spoons Science Journal Rainbow Ice (page 40) Word Cards Rainbow Ice (page 41)
 Physical Science Demonstration Materials clear, shoebox-size plastic container food coloring (red, blue, and yellow) water 3 clear, plastic cups (9 12 oz) 3 pipettes or eye droppers various types of salt
Physical Science Demonstration Materials clear, shoebox-size plastic container food coloring (red, blue, and yellow) water 3 clear, plastic cups (9 12 oz) 3 pipettes or eye droppers various types of salt
Solutions: Chemical or Physical Change?
 Section 1 Solutions: Chemical or Physical Change? What Do You See? Learning Outcomes In this section you will Learn to differentiate between chemical and physical changes. Make observations and cite evidence
Section 1 Solutions: Chemical or Physical Change? What Do You See? Learning Outcomes In this section you will Learn to differentiate between chemical and physical changes. Make observations and cite evidence
Chapter 2: Properties of Matter Student Outline 2.1 Classifying Matter A. Pure Substances
 Name: Date: Physical Science Period: Chapter 2: Properties of Matter Student Outline GA Performance Standards SPS1. Students will investigate our current understanding of the atom. SPS2. Students will
Name: Date: Physical Science Period: Chapter 2: Properties of Matter Student Outline GA Performance Standards SPS1. Students will investigate our current understanding of the atom. SPS2. Students will
Period 11: Chemical Energy and Fossil Fuels
 Name Section Period 11: Chemical Energy and Fossil Fuels 11.1 What is the Composition of Matter? 1) Atoms a) What are the building blocks of an atom? b) Which nucleons make up an atomic nucleus? c) What
Name Section Period 11: Chemical Energy and Fossil Fuels 11.1 What is the Composition of Matter? 1) Atoms a) What are the building blocks of an atom? b) Which nucleons make up an atomic nucleus? c) What
2 Properties of Matter
 CHAPTER 2 2 Properties of Matter SECTION Matter KEY IDEAS As you read this section, keep these questions in mind: Why are color, volume, and density physical properties? Why are flammability and reactivity
CHAPTER 2 2 Properties of Matter SECTION Matter KEY IDEAS As you read this section, keep these questions in mind: Why are color, volume, and density physical properties? Why are flammability and reactivity
PROPERTIES OF MATTER Review Stations
 PROPERTIES OF MATTER Review Stations PROPERTIES OF MATTER STATION 1 Where did the water droplets on the outside of this cup come from? Answer: The droplets came from the air surrounding the cup. What phase
PROPERTIES OF MATTER Review Stations PROPERTIES OF MATTER STATION 1 Where did the water droplets on the outside of this cup come from? Answer: The droplets came from the air surrounding the cup. What phase
Properties of Matter
 Properties of Matter OBJECTIVES: Identify properties of matter as extensive or intensive. Define physical property, and list several common physical properties of substances. Differentiate among three
Properties of Matter OBJECTIVES: Identify properties of matter as extensive or intensive. Define physical property, and list several common physical properties of substances. Differentiate among three
Physical Changes vs Chemical Changes Lab
 Physical Changes vs Chemical Changes Lab What was done? What can you observe? Is it a physical or chemical change and why? What else have you learned during the demonstration? #1) Crumpling Paper Notice
Physical Changes vs Chemical Changes Lab What was done? What can you observe? Is it a physical or chemical change and why? What else have you learned during the demonstration? #1) Crumpling Paper Notice
bending. Special types of physical changes where any object changes state, such as when water freezes or evaporates, are sometimes called change of st
 Physical and Chemical Properties and Changes The properties of a substance are those characteristics that are used to identify or describe it. When we say that water is "wet", or that silver is "shiny",
Physical and Chemical Properties and Changes The properties of a substance are those characteristics that are used to identify or describe it. When we say that water is "wet", or that silver is "shiny",
Chapter 6, Lesson 1: What is a Chemical Reaction?
 Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Introduction to Weathering
 Name: Date: Period: Unit 9: Earth s Destructive Forces A. Kinds of Weathering Introduction to Weathering Distinguish between two major processes that change the Earth surface. Identify two types of weathering.
Name: Date: Period: Unit 9: Earth s Destructive Forces A. Kinds of Weathering Introduction to Weathering Distinguish between two major processes that change the Earth surface. Identify two types of weathering.
Name Date Block. 3. Label a, b and c, of the atomic structure below with the proper terms (hint: #1 )
 Name Date Block CFA #1 Review You will have approximately 5 minutes at each station to record your answers. You may use your notebook as a reference. WORK EFFICIENTLY & STAY FOCUSED! YOU GOT THIS! Station
Name Date Block CFA #1 Review You will have approximately 5 minutes at each station to record your answers. You may use your notebook as a reference. WORK EFFICIENTLY & STAY FOCUSED! YOU GOT THIS! Station
Matter: Properties and Changes
 Matter: Properties and Changes Matter and Its Changes What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree
Matter: Properties and Changes Matter and Its Changes What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree
Characteristics of Chemical Change
 Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Vocabulary: Matter: has mass and takes up space (pure substances and mixtures) Pure Substances: composition definite, elements and compounds.
 Unit 2: Composition and Properties of Matter Review Elements, Compounds, Mixtures and Physical/Chemical Properties and Changes, Water Properties and Biogeochemical Cycles Vocabulary: Matter: has mass and
Unit 2: Composition and Properties of Matter Review Elements, Compounds, Mixtures and Physical/Chemical Properties and Changes, Water Properties and Biogeochemical Cycles Vocabulary: Matter: has mass and
Experiment 17 It s A Gas and More!
 Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
LESSON 1: DESCRIBING MATTER pg.5. Chemistry = Is the study of matter & how matter changes. Liquid/Solid/Gas
 Chemistry..CHAPTER 1: INTRO TO MATTER LESSON 1: DESCRIBING MATTER pg.5 Chemistry = Is the study of matter & how matter changes A. Matter = anything that has mass & takes up space à You, air, plastic, metal,
Chemistry..CHAPTER 1: INTRO TO MATTER LESSON 1: DESCRIBING MATTER pg.5 Chemistry = Is the study of matter & how matter changes A. Matter = anything that has mass & takes up space à You, air, plastic, metal,
2/22/2019 NEW UNIT! Chemical Interactions. Atomic Basics #19
 NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make?
 Chapter 4 Test 1. 2. 3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make? packs Work Space: 5. A kennel is moving 160
Chapter 4 Test 1. 2. 3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make? packs Work Space: 5. A kennel is moving 160
A simple equation of what happens when you add baking soda to vinegar:
 What s the Matter? Matter is anything that takes up space and has mass. Mass is the stuff that matter is made of, or the amount of particles in a substance or object. Matter has physical and chemical properties
What s the Matter? Matter is anything that takes up space and has mass. Mass is the stuff that matter is made of, or the amount of particles in a substance or object. Matter has physical and chemical properties
1 st Semester Exam Study Guide 1.) Which of the following is NOT a compound? Explain why. a. H2O b. O2
 1 st Semester Exam Study Guide 1.) Which of the following is NOT a compound? Explain why. a. H2O b. O2 2.) A chemist has discovered what she thinks is a new molecule. In order for it to be a molecule,
1 st Semester Exam Study Guide 1.) Which of the following is NOT a compound? Explain why. a. H2O b. O2 2.) A chemist has discovered what she thinks is a new molecule. In order for it to be a molecule,
Unit 3. Matter and Change
 Unit 3 Matter and Change 1 Matter Matter is anything that: a) has mass, and b) takes up space Mass = a measure of the amount of stuff (or material) the object contains (don t confuse this with weight,
Unit 3 Matter and Change 1 Matter Matter is anything that: a) has mass, and b) takes up space Mass = a measure of the amount of stuff (or material) the object contains (don t confuse this with weight,
ANSWER KEY. Introduction to Matter
 Chapter Project Worksheet 1 1. The object to be massed is placed on one side of the balance. Objects with known masses are placed on the other side of the arm until the balance arm is level. 2. Sample
Chapter Project Worksheet 1 1. The object to be massed is placed on one side of the balance. Objects with known masses are placed on the other side of the arm until the balance arm is level. 2. Sample
Chemistry is the study of matter, its properties and its changes or transformations. Feb 19 11:08 AM
 What is Chemistry? Chemistry is the study of matter, its properties and its changes or transformations. Matter is anything that has mass and takes up space. All matter has physical and chemical properties.
What is Chemistry? Chemistry is the study of matter, its properties and its changes or transformations. Matter is anything that has mass and takes up space. All matter has physical and chemical properties.
Insert the name of the element on the line below and insert the letters into the Periodic Table according to the clues.
 1. The periodic table Insert the name of the element on the line below and insert the letters into the Periodic Table according to the clues. (Just for fun) A: An element that is important in the development
1. The periodic table Insert the name of the element on the line below and insert the letters into the Periodic Table according to the clues. (Just for fun) A: An element that is important in the development
Matter: Properties and Changes
 CHAPTER 12 Matter: Properties and Changes Matter and Its Properties What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column
CHAPTER 12 Matter: Properties and Changes Matter and Its Properties What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column
Unit 3: Physical Science Classifying Matter in our Daily Lives
 Science 7 Unit 3: Physical Science Classifying Matter in our Daily Lives Name Period Purpose: I understand the relationship between atoms, molecules, elements, compounds, and mixtures and can also provide
Science 7 Unit 3: Physical Science Classifying Matter in our Daily Lives Name Period Purpose: I understand the relationship between atoms, molecules, elements, compounds, and mixtures and can also provide
2.1 Describing Matter Properties used to describe matter can be classified as extensive or intensive.
 2.1 Properties used to describe matter can be classified as extensive or intensive. Extensive Properties The mass of an object is a measure of the amount of matter the object contains. The volume of an
2.1 Properties used to describe matter can be classified as extensive or intensive. Extensive Properties The mass of an object is a measure of the amount of matter the object contains. The volume of an
Today is: Monday, October 17th
 Today is: Monday, October 17th Get out your homework! 10/17/2016 #motivationmonday (This is Week 11 Warm Ups!) 1. It s a new quarter, which means a fresh start! What can you do to stay motivated this quarter?
Today is: Monday, October 17th Get out your homework! 10/17/2016 #motivationmonday (This is Week 11 Warm Ups!) 1. It s a new quarter, which means a fresh start! What can you do to stay motivated this quarter?
Name: Period: Date: UNIT 1: Introduction to Matter Lesson 8: Baby, you look good
 Name: Period: Date: UNIT 1: Introduction to Matter Lesson 8: Baby, you look good Do Now: By the end of today, you will have an answer to: How do we describe and identify matter? Draw a particle diagram
Name: Period: Date: UNIT 1: Introduction to Matter Lesson 8: Baby, you look good Do Now: By the end of today, you will have an answer to: How do we describe and identify matter? Draw a particle diagram
Name Date Class STUDY GUIDE FOR CONTENT MASTERY
 Chemical Reactions Section 10.1 Reactions and Equations In your textbook, read about evidence of chemical reactions. For each statement, write yes if evidence of a chemical reaction is present. Write no
Chemical Reactions Section 10.1 Reactions and Equations In your textbook, read about evidence of chemical reactions. For each statement, write yes if evidence of a chemical reaction is present. Write no
LESSON 1. Chemical Reactions. Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions.
 LESSON 1 Chemical Reactions Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions. By the end of this lesson... you will be able to explain ways
LESSON 1 Chemical Reactions Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions. By the end of this lesson... you will be able to explain ways
Supply List. Science 300. Released Glynlyon, Inc
 N Supply List Science 300 2012 Glynlyon, Inc Released 4-1-12 Table of Contents UNIT 1: YOU GROW AND CHANGE... 1 UNIT 2: PLANTS... 1 UNIT 3: ANIMALS: GROWTH AND CHANGE... 2 UNIT 4: YOU ARE WHAT YOU EAT...
N Supply List Science 300 2012 Glynlyon, Inc Released 4-1-12 Table of Contents UNIT 1: YOU GROW AND CHANGE... 1 UNIT 2: PLANTS... 1 UNIT 3: ANIMALS: GROWTH AND CHANGE... 2 UNIT 4: YOU ARE WHAT YOU EAT...
Matter Review Packet
 Matter Review Packet 1. A mixture (is/is not) a chemical combining of substances. 2. In a compound the (atoms/molecules) are (chemically/physically) combined so that the elements that make up the compound
Matter Review Packet 1. A mixture (is/is not) a chemical combining of substances. 2. In a compound the (atoms/molecules) are (chemically/physically) combined so that the elements that make up the compound
Elements and Compounds Power Point Questions
 Name period date assigned date due date returned 1. The is the smallest unit of an element. 2. An is the fundamental material of which all is composed. It only contains type of atom. 3. What is the symbol
Name period date assigned date due date returned 1. The is the smallest unit of an element. 2. An is the fundamental material of which all is composed. It only contains type of atom. 3. What is the symbol
Science Supply List. Science Glynlyon, Inc.
 Science Supply List Science 300 2016 Glynlyon, Inc. Table of Contents UNIT 1: YOU GROW AND CHANGE... 1 UNIT 2: PLANTS... 2 UNIT 3: ANIMALS: GROWTH AND CHANGE... 3 UNIT 4: YOU ARE WHAT YOU EAT... 4 UNIT
Science Supply List Science 300 2016 Glynlyon, Inc. Table of Contents UNIT 1: YOU GROW AND CHANGE... 1 UNIT 2: PLANTS... 2 UNIT 3: ANIMALS: GROWTH AND CHANGE... 3 UNIT 4: YOU ARE WHAT YOU EAT... 4 UNIT
1 Weathering. Critical Thinking 2. Infer Would ice wedging happen if water did not expand as it froze? Explain your answer.
 CHAPTER 10 1 Weathering SECTION Weathering and Soil Formation BEFORE YOU READ After you read this section, you should be able to answer these questions: What is weathering? What causes mechanical weathering?
CHAPTER 10 1 Weathering SECTION Weathering and Soil Formation BEFORE YOU READ After you read this section, you should be able to answer these questions: What is weathering? What causes mechanical weathering?
Chapter 2 Matter and Change. Charles Page High School Pre-AP Chemistry Stephen L. Cotton
 Chapter 2 Matter and Change 1 Charles Page High School Pre-AP Chemistry Stephen L. Cotton Section 2.1 Properties of Matter OBJECTIVES: Identify properties of matter as extensive or intensive. 2 Section
Chapter 2 Matter and Change 1 Charles Page High School Pre-AP Chemistry Stephen L. Cotton Section 2.1 Properties of Matter OBJECTIVES: Identify properties of matter as extensive or intensive. 2 Section
Matter and Its Properties
 Section 2 4A, 4B, 4C, 4D Main Ideas Atoms are the building blocks of matter. All substances have characteristic properties. Matter can be a pure substance or a mixture. 4A differentiate between physical
Section 2 4A, 4B, 4C, 4D Main Ideas Atoms are the building blocks of matter. All substances have characteristic properties. Matter can be a pure substance or a mixture. 4A differentiate between physical
Pure Substances and Mixtures
 Pure Substances and Mixtures Unit 1 Lesson 4 Mix It Up! Make two mixtures in transparent containers with lids. 1. Find two transparent containers with tight lids. In one container, mix either salt or sugar
Pure Substances and Mixtures Unit 1 Lesson 4 Mix It Up! Make two mixtures in transparent containers with lids. 1. Find two transparent containers with tight lids. In one container, mix either salt or sugar
What is Oobleck? Can you use THE SCIENTIFIC METHOD AND your senses to solve the mystery of Oobleck? Problem
 What is Oobleck? Can you use THE SCIENTIFIC METHOD AND your senses to solve the mystery of Oobleck? Problem Three liquids are mixed together in a plastic bag. Using your senses (except for taste) can you
What is Oobleck? Can you use THE SCIENTIFIC METHOD AND your senses to solve the mystery of Oobleck? Problem Three liquids are mixed together in a plastic bag. Using your senses (except for taste) can you
CHAPTER 1: Chemistry, An Introduction
 CHAPTER 1: Chemistry, An Introduction science: the study of nature to explain what one observes 1.4 THE SCIENTIFIC METHOD: How Chemists Think Applying the Scientific Method 1. Make an observation, and
CHAPTER 1: Chemistry, An Introduction science: the study of nature to explain what one observes 1.4 THE SCIENTIFIC METHOD: How Chemists Think Applying the Scientific Method 1. Make an observation, and
Name: Broughton High School. Physical Science Work Book 2016
 Name: Broughton High School 1 Chapter 15 Classification of Matter Vocabulary Words Vocabulary Word Definition Chemical Change 2 Chemical Property Colloid Compound Distillation Element Heterogeneous Mixture
Name: Broughton High School 1 Chapter 15 Classification of Matter Vocabulary Words Vocabulary Word Definition Chemical Change 2 Chemical Property Colloid Compound Distillation Element Heterogeneous Mixture
Station 1: Atoms and Elements. positive neutral negative
 Name Chemistry Review You will have approximately 5 minutes at each station to record your answers. You may use your notebook as a reference. WORK EFFICIENTLY & STAY FOCUSED! YOU GOT THIS! Station 1: Atoms
Name Chemistry Review You will have approximately 5 minutes at each station to record your answers. You may use your notebook as a reference. WORK EFFICIENTLY & STAY FOCUSED! YOU GOT THIS! Station 1: Atoms
Physical and Chemical Changes Or How Do You Know When You ve Made Something New?
 Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Change Matter around us goes through many changes. E.g. Water evapourates, bread is toasted for your breakfast Changes are divided into two
 Change Matter around us goes through many changes. E.g. Water evapourates, bread is toasted for your breakfast Changes are divided into two categories: Physical changes Chemical changes Physical Changes
Change Matter around us goes through many changes. E.g. Water evapourates, bread is toasted for your breakfast Changes are divided into two categories: Physical changes Chemical changes Physical Changes
Chapter 14 Study Questions Name: Sci Class:
 Chapter 14 Study Questions Name: Sci Class: Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. Balancing a chemical equation so that the same
Chapter 14 Study Questions Name: Sci Class: Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. Balancing a chemical equation so that the same
1 Forming New Substances
 CHAPTER 9 1 Forming New Substances SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a What are exothermic reactions? What are
CHAPTER 9 1 Forming New Substances SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a What are exothermic reactions? What are
What Do You Think? Investigate GOALS
 Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Properties of Matter. Mrs. Lapierre Chemistry
 Properties of Matter. Mrs. Lapierre Chemistry What is a property? In chemistry, a property is what we use to distinguish between different types of matter. There are two types: Chemical. Physical. Why
Properties of Matter. Mrs. Lapierre Chemistry What is a property? In chemistry, a property is what we use to distinguish between different types of matter. There are two types: Chemical. Physical. Why
Physical and Chemical Properties and Changes Lab
 Name: Date: Period: Group Members Physical and Chemical Properties and Changes Lab Station 1 Color Station Instruction: Describe the color of the following substances. Substance Color 1. Sulfur 2. Ammonium
Name: Date: Period: Group Members Physical and Chemical Properties and Changes Lab Station 1 Color Station Instruction: Describe the color of the following substances. Substance Color 1. Sulfur 2. Ammonium
Is it possible to change your breath into food? Standard 1, Objective 2: Observe and evaluate evidence of chemical and physical change
 Vocabulary chemical change physical change combustion corrosion FIGURE 1: When glass breaks, its physical properties change. Instead of one solid sheet of glass, it now has holes and cracks. Is it possible
Vocabulary chemical change physical change combustion corrosion FIGURE 1: When glass breaks, its physical properties change. Instead of one solid sheet of glass, it now has holes and cracks. Is it possible
The masses of reactants and products are equal.
 Page 1 of 7 KEY CNCEPT The masses of reactants and products are equal. BEFRE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed and identified
Page 1 of 7 KEY CNCEPT The masses of reactants and products are equal. BEFRE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed and identified
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
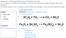 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Online Resource 10. December 2016
 Designing for the Next Generation Standards: Educative Curriculum Materials and Measures of Teacher Knowledge Online Resource 10 December 2016 Toward High School Biology: Tables 1-4 provide evidence that
Designing for the Next Generation Standards: Educative Curriculum Materials and Measures of Teacher Knowledge Online Resource 10 December 2016 Toward High School Biology: Tables 1-4 provide evidence that
Anything occupying space and having mass. Matter exists in three states.
 Chapter 3 Matter Section 3.1 Matter Matter Anything occupying space and having mass. Matter exists in three states. Solid Liquid Gas Section 3.1 Matter The Three States of Water Section 3.1 Matter Solid
Chapter 3 Matter Section 3.1 Matter Matter Anything occupying space and having mass. Matter exists in three states. Solid Liquid Gas Section 3.1 Matter The Three States of Water Section 3.1 Matter Solid
