1 The Nature of Chemical Reactions
|
|
|
- Brenda Evans
- 5 years ago
- Views:
Transcription
1 CHAPTER 7 1 The Nature of Chemical Reactions SECTION Chemical Reactions KEY IDEAS As you read this section, keep these questions in mind: What happens in a chemical reaction? What is the role of energy in a chemical reaction? What is the difference between an exothermic and an endothermic reaction? What Is a Chemical Reaction? Scientists often carry out chemical reactions in laboratories. However, chemical reactions also happen outside science laboratories. In fact, chemical reactions happen around you and inside you all the time. A tree growing, an apple ripening, a leaf decaying, and a log burning are all chemical reactions. Digestion and respiration are also chemical reactions. In a chemical reaction, substances go through chemical changes to form new substances. Consider what happens when you combine sugar, water, flour, and yeast to make bread. The yeast breaks down the sugar to form new substances. One of these substances is carbon dioxide gas, which causes the bread to rise. The formation of a gas is a clue that a chemical reaction has happened. Sometimes a gas from a chemical reaction forms bubbles that you can see. For example, if you mix baking soda and vinegar, gas bubbles will form. The gas bubbles indicate that a reaction has happened. Other clues that a chemical reaction has happened are shown below. READING TOOLBOX Organize After you read this section, make a Concept Map using the section vocabulary terms. Review your Concept Map with a partner. You and your partner may have organized the terms differently. 1. Describe What happens in a chemical reaction? Formation of a solid Formation of a gas Chemical reaction clues Release of sound energy Release of light energy Interactive Reader 135 Chemical Reactions
2 2. Explain How do new substances form in a chemical reaction? BREAKING BONDS AND REARRANGING ATOMS Recall that matter is made of atoms. Compounds consist of atoms of two or more different elements bonded to one another. During a chemical reaction, the bonds between some atoms break. The atoms rearrange and new bonds form. In other words, the atoms recombine to form new substances. Atoms are never destroyed in a chemical reaction. Consider what happens when gasoline burns in an engine. Gasoline contains isooctane (C 8 H 18 ). When gasoline burns, isooctane reacts with oxygen gas (O 2 ). These molecules are shown below. Oxygen, O Isooctane is made up of carbon and hydrogen atoms. Oxygen gas is made up of only oxygen atoms. Isooctane and oxygen react to form carbon dioxide (CO 2 ) and water (H 2 O). 3. Compare Examine the molecules in this figure and those in the figure above. Where did the oxygen and hydrogen atoms in H 2 O come from? A carbon dioxide molecule consists of carbon and oxygen atoms. Carbon, C Hydrogen, H A water molecule consists of hydrogen and oxygen atoms. Oxygen, O Oxygen, O A water molecule consists of hydrogen and oxygen atoms. If you compare the figures above, you will see that bonds between the carbon and hydrogen atoms in isooctane broke. Bonds also broke between the oxygen atoms in the oxygen molecule. The atoms recombined to form new substances. New bonds formed between carbon atoms and oxygen atoms to make carbon dioxide. New bonds also formed between hydrogen atoms and oxygen atoms to make water. Interactive Reader 136 Chemical Reactions
3 REACTANTS AND PRODUCTS When gasoline burns in an engine, isooctane and oxygen are the reactants. A reactant is a substance that participates in a chemical reaction. Isooctane and oxygen react to produce carbon dioxide and water, which are the products. A product is a substance that forms during a chemical reaction. You can use a word equation to show what happens in a chemical reaction. For example, the following word equation shows what happens when gasoline burns. isooctane + oxygen carbon dioxide + water Reactants The reactants are on the left side of the equation. An arrow separates the two sides of the equation. Products The products are on the right side of the equation. 4. Identify What do scientists call a substance that forms during a chemical reaction? 5. Identify On what side of the equation are reactants found? What Is the Role of Energy in Chemical Reactions? Chemical reactions always involve changes in energy. Most chemical reactions need added energy to start. Some chemical reactions produce, or release, energy. Other chemical reactions absorb energy. Chemical compounds store energy in the bonds between their atoms. Energy that is stored in the form of chemical bonds is called chemical energy. During a chemical reaction, chemical energy may change form. For example, it could become light or heat energy. However, the total amount of energy in the reactants must equal the total amount of energy in the products. In other words, energy in a chemical reaction can change form, but no energy is created or destroyed. FORMING BONDS Recall that new bonds form in chemical reactions. When new bonds form, energy is released. For example, when gasoline burns, energy is released as heat and light. 6. Identify Where is energy stored in chemical compounds? Interactive Reader 137 Chemical Reactions
4 BREAKING BONDS Recall that some bonds between atoms break in a chemical reaction. Breaking bonds requires energy. For example, energy is needed to break bonds between the atoms in isooctane and oxygen. A spark from a spark plug provides the energy needed to break these bonds. An open flame can also start this reaction. For this reason, sparks and flames are not allowed near a gas pump. LIGHT ENERGY AND PHOTOSYNTHESIS During photosynthesis, plants use light energy to change carbon dioxide and water into sugars and oxygen. As a result of photosynthesis, light energy is changed into chemical energy. This chemical energy is stored in the bonds of carbohydrate molecules. You gain some of this chemical energy when you eat fruits and vegetables. Carbon dioxide, CO 2 Oxygen, O 2 7. Infer The cells in your body get energy from the foods you eat. However, your cells must use energy to get this energy. Why? Water, H 2 O 8. Describe How does the energy of the reactants and products differ in an endothermic reaction? What Are Endothermic and Exothermic Reactions? Recall that during a chemical reaction, energy is needed to break bonds in the reactants. Energy is released as new bonds form in the products. In an endothermic reaction, more energy is needed to break the bonds in the reactants than is released when new bonds form in the products. In other words, endothermic reactions absorb more energy than they release. In an endothermic reaction, the products have more chemical energy than the reactants do. Interactive Reader 138 Chemical Reactions
5 This bump represents the energy needed to start the reaction. In an endothermic reaction, energy is stored in the products as chemical energy. Thus, the products have more energy than the reactants. In an exothermic reaction, more energy is released when new bonds form than is needed to break bonds. In other words, exothermic reactions release more energy than they absorb. In an exothermic reaction, the products have less chemical energy than the reactants do. All combustion reactions are exothermic. Graphing Skills 9. Interpret How does the energy of the products differ from the energy of the reactants in an exothermic reaction? In an exothermic reaction, chemical energy is released, often as heat. CONSERVATION OF ENERGY All chemical reactions obey the law of conservation of energy. The law of conservation of energy states that energy cannot be created or destroyed. In both endothermic and exothermic reactions, energy changes from one form into another form. However, no energy is created or destroyed. If no energy is created or destroyed, how can the products of an exothermic reaction have less energy than the reactants? How can the products of an endothermic reaction have more energy than the reactants? 10. Explain How can products and reactants have different amounts of energy without violating the law of conservation of energy? Endothermic reaction: Energy of reactants + energy absorbed = energy of products Exothermic reaction: Energy of reactants = energy of products + energy released Interactive Reader 139 Chemical Reactions
6 Section 1 Review SECTION VOCABULARY chemical energy the energy released when a chemical compound reacts to produce new compounds endothermic reaction a chemical reaction that requires energy input exothermic reaction a chemical reaction in which energy is released to the surroundings as heat product a substance that forms in a chemical reaction reactant a substance or molecule that participates in a chemical reaction 1. List What are three clues that may tell you a chemical reaction is happening? 2. Predict Caramel forms when table sugar, C 12 H 22 O 11, reacts with oxygen. What elements make up caramel? 3. Apply Concepts Identify whether or not each of the following is a chemical reaction. Explain your answers. a. melting ice b. burning a candle c. rusting iron 4. Describe What is the role of energy in forming bonds? What is the role of energy in breaking bonds? 5. Apply Concepts Is a firecracker exploding an example of an endothermic reaction or an exothermic reaction? Explain your answer. Interactive Reader 140 Chemical Reactions
1 Forming New Substances
 CHAPTER 9 1 Forming New Substances SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a What are exothermic reactions? What are
CHAPTER 9 1 Forming New Substances SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a What are exothermic reactions? What are
The elements or compounds that enter into a chemical reaction are known as reactants.
 Chemical Reactions Chemical Reactions A chemical reaction is a process that changes, or transforms, one set of chemicals into another by changing the chemical bonds that join atoms in compounds. The elements
Chemical Reactions Chemical Reactions A chemical reaction is a process that changes, or transforms, one set of chemicals into another by changing the chemical bonds that join atoms in compounds. The elements
Section 1: What is a Chemical Reaction
 Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Physical and Chemical Changes
 Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment
 Science 24: Day 10 1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment https://www.youtube.com/watch?v=fofpjj7v414 Which reaction was
Science 24: Day 10 1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment https://www.youtube.com/watch?v=fofpjj7v414 Which reaction was
Observing Chemical Change - 5.1
 Observing Chemical Change - 5.1 Vocabulary: Physical change - Chemical change - Reactant Product Precipitate Exothermic reaction - Endothermic reaction - Properties of matter: Two kinds of properties of
Observing Chemical Change - 5.1 Vocabulary: Physical change - Chemical change - Reactant Product Precipitate Exothermic reaction - Endothermic reaction - Properties of matter: Two kinds of properties of
SCIENCE 9 CONCEPT 4 CHEMICAL REACTIONS
 SCIENCE 9 CONCEPT 4 CHEMICAL REACTIONS VOCABULARY TERMS CONCEPT 4 Chemical reaction Reactants Products Exothermic Endothermic Combustion Corrosion Cellular respiration Conservation of mass Closed systems
SCIENCE 9 CONCEPT 4 CHEMICAL REACTIONS VOCABULARY TERMS CONCEPT 4 Chemical reaction Reactants Products Exothermic Endothermic Combustion Corrosion Cellular respiration Conservation of mass Closed systems
Reactants and Products
 Biological Chemical Reactions Chemical reactions allow living things to grow, develop, reproduce, and adapt. Real-World Reading Link When you lie down for the night, you might think that your body is completely
Biological Chemical Reactions Chemical reactions allow living things to grow, develop, reproduce, and adapt. Real-World Reading Link When you lie down for the night, you might think that your body is completely
Exothermic and Endothermic Reactions
 Energy in hemical Reactions hemical Reactions Exothermic and Endothermic Reactions 1 hemical reactions occur all around us. They happen every second of every day. hemical reactions are more than just things
Energy in hemical Reactions hemical Reactions Exothermic and Endothermic Reactions 1 hemical reactions occur all around us. They happen every second of every day. hemical reactions are more than just things
Chapter Introduction Lesson 1 Understanding Chemical Reactions Lesson 2 Types of Chemical Reactions Lesson 3 Energy Changes and Chemical Reactions
 Chapter Introduction Lesson 1 Understanding Chemical Reactions Lesson 2 Types of Chemical Reactions Lesson 3 Energy Changes and Chemical Reactions Chapter Wrap-Up Changes in Matter A physical change does
Chapter Introduction Lesson 1 Understanding Chemical Reactions Lesson 2 Types of Chemical Reactions Lesson 3 Energy Changes and Chemical Reactions Chapter Wrap-Up Changes in Matter A physical change does
Section 1 Forming New Substances
 Section 1 Forming New Substances Key Concept During chemical reactions, atoms rearrange to form new substances that have different properties than the original substances had. What You Will Learn Four
Section 1 Forming New Substances Key Concept During chemical reactions, atoms rearrange to form new substances that have different properties than the original substances had. What You Will Learn Four
Chapter 7 TEST CHEMICAL REACTIONS PART 1: MULTIPLE CHOICE.
 Chapter 7 TEST CHEMICAL REACTIONS PART 1: MULTIPLE CHOICE. 1. Which of these is not a sign of a chemical reaction? a. A gas is given off. c. Heat is released. b. The material dissolves. d. A color change
Chapter 7 TEST CHEMICAL REACTIONS PART 1: MULTIPLE CHOICE. 1. Which of these is not a sign of a chemical reaction? a. A gas is given off. c. Heat is released. b. The material dissolves. d. A color change
3.3. Physical and Chemical Changes. Before You Read. What is a physical change? What is a chemical change?
 Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
2 Changes of State KEY IDEAS READING TOOLBOX ADDING AND REMOVING ENERGY. States of Matter. As you read this section, keep these questions in mind:
 CHAPTER 3 States of Matter 2 Changes of State SECTION KEY IDEAS As you read this section, keep these questions in mind: What happens when a substance changes from one state of matter to another? What happens
CHAPTER 3 States of Matter 2 Changes of State SECTION KEY IDEAS As you read this section, keep these questions in mind: What happens when a substance changes from one state of matter to another? What happens
Physical or Chemical Change?
 Chemical Formulas and Equations Physical or Chemical Change? Matter can change physically or chemically; a process that produces a chemical change is a chemical reaction. Chemical Formulas and Equations
Chemical Formulas and Equations Physical or Chemical Change? Matter can change physically or chemically; a process that produces a chemical change is a chemical reaction. Chemical Formulas and Equations
Class X. Exercises solution
 Exercises solution Question 1: Which of the statements about the reaction below are incorrect? Lead is getting reduced. Carbon dioxide is getting oxidised. Carbon is getting oxidised. Lead oxide is getting
Exercises solution Question 1: Which of the statements about the reaction below are incorrect? Lead is getting reduced. Carbon dioxide is getting oxidised. Carbon is getting oxidised. Lead oxide is getting
Chemical Reactions. Chapter Test A. Multiple Choice. 1 Pearson Education, Inc., or its affiliates. All rights reserved.
 Name Date Class Chemical Reactions Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. 1. Chemistry is a. a characteristic of a substance that can be observed
Name Date Class Chemical Reactions Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. 1. Chemistry is a. a characteristic of a substance that can be observed
Name. Pre-Test. Directions: Circle the T if a statement is true or F if it is false (F). 1. Matter always stays the same.
 1 Pre-est Directions: Circle the if a statement is true or if it is false (). 1. Matter always stays the same. 2. A physical change is a change in size, shape or phase of matter, without the matter changing
1 Pre-est Directions: Circle the if a statement is true or if it is false (). 1. Matter always stays the same. 2. A physical change is a change in size, shape or phase of matter, without the matter changing
Student Notes. Chemical Reactions LINK
 LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
Science 1206 Chemistry Unit #10
 Science 1206 Chemistry Unit #10 Introduction to Chemical Equations Communicating Chemical Changes Chemical change occurs when matter changes into a new and different form. The new form has a new make up
Science 1206 Chemistry Unit #10 Introduction to Chemical Equations Communicating Chemical Changes Chemical change occurs when matter changes into a new and different form. The new form has a new make up
8.1 Chemical Properties and Changes. chemical property chemical change dissolving
 8.1 Chemical Properties and Changes chemical property chemical change dissolving Ability to Change 8.1 Chemical Properties and Changes In a chemical change, the properties that give a substance its identity
8.1 Chemical Properties and Changes chemical property chemical change dissolving Ability to Change 8.1 Chemical Properties and Changes In a chemical change, the properties that give a substance its identity
Lesson 5: Oxygen, Oxidation, and Combustion
 Lesson 5: Oxidation, and Students are introduced to exothermic reactions that involve oxidation. Main Concept: Oxygen is a highly reactive element involved in chemical reactions that release heat energy.
Lesson 5: Oxidation, and Students are introduced to exothermic reactions that involve oxidation. Main Concept: Oxygen is a highly reactive element involved in chemical reactions that release heat energy.
Website: Page 1. Page 14»Exercise» Page 15» Question 1:
 Page 14»Exercise» Question 1: Which of the statements about the reaction below are incorrect? (a) Lead is getting reduced. (b) Carbon dioxide is getting oxidised. (c) Carbon is getting oxidised. (d) Lead
Page 14»Exercise» Question 1: Which of the statements about the reaction below are incorrect? (a) Lead is getting reduced. (b) Carbon dioxide is getting oxidised. (c) Carbon is getting oxidised. (d) Lead
The Question (Learning Goals)
 The Question (Learning Goals) What is the Law of Conservation of Mass (or atoms)? What is a balanced Chemical Reaction? How do we balance a chemical reaction?? (A Level) Review: Chemical Reactions Chemical
The Question (Learning Goals) What is the Law of Conservation of Mass (or atoms)? What is a balanced Chemical Reaction? How do we balance a chemical reaction?? (A Level) Review: Chemical Reactions Chemical
Changes in Matter Study Guide
 Changes in Matter Study Guide Define the following and give a real world example: 1. : A change in matter that requires the substances to absorb energy Example: Photosynthesis (the plants absorb light
Changes in Matter Study Guide Define the following and give a real world example: 1. : A change in matter that requires the substances to absorb energy Example: Photosynthesis (the plants absorb light
Physical and Chemical Changes & Properties of Matter
 Physical and Chemical Changes & Properties of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. 8P1CD Physical And Chemical Properties And Changes
Physical and Chemical Changes & Properties of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. 8P1CD Physical And Chemical Properties And Changes
Page 1 / 12. Chemistry Exam. Name: Matter Properties, Structure. Question 1 (1 point) The atomic number of an atom is. A. The mass of the atom.
 Chemistry Exam Matter Properties, Structure Name: Question 1 (1 point) The atomic number of an atom is A. The mass of the atom. B. The number of protons added to the number of neutrons in the nucleus.
Chemistry Exam Matter Properties, Structure Name: Question 1 (1 point) The atomic number of an atom is A. The mass of the atom. B. The number of protons added to the number of neutrons in the nucleus.
3 Chemical Properties
 CHAPTER 7 3 Chemical Properties SECTION The Properties of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What are chemical properties of matter? What
CHAPTER 7 3 Chemical Properties SECTION The Properties of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What are chemical properties of matter? What
Lesson 1: What are chemical changes?
 Lesson 1 Summary Lesson 1: What are chemical changes? Vocabulary Use with pp. 375 377 physical change a change in which the material keeps its identify chemical change a change in which one substance or
Lesson 1 Summary Lesson 1: What are chemical changes? Vocabulary Use with pp. 375 377 physical change a change in which the material keeps its identify chemical change a change in which one substance or
The Chemistry of Respiration and Photosynthesis
 The Chemistry of Respiration and Photosynthesis Objective- You should be able to write balanced equations for respiration and photosynthesis and explain how the two equations are related. Directions :
The Chemistry of Respiration and Photosynthesis Objective- You should be able to write balanced equations for respiration and photosynthesis and explain how the two equations are related. Directions :
Chapter 14 Study Questions Name: Sci Class:
 Chapter 14 Study Questions Name: Sci Class: Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. Balancing a chemical equation so that the same
Chapter 14 Study Questions Name: Sci Class: Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. Balancing a chemical equation so that the same
Chapter 17: Chemical Reactions
 Chapter 17: Chemical Reactions Section 1: Chemical Formulas and Equations Chemical Equations Balancing Chemical Equations Law of Conservation of Mass Evidences of Chemical Reactions Symbols to represent
Chapter 17: Chemical Reactions Section 1: Chemical Formulas and Equations Chemical Equations Balancing Chemical Equations Law of Conservation of Mass Evidences of Chemical Reactions Symbols to represent
Activation Energy is the minimum amount of energy needed to start a chemical reaction.
 5.3 Controlling Chemical Reactions Vocabulary: Activation energy Concentration Catalyst Enzyme Inhibitor - How do reactions get started? Chemical reactions won t begin until the reactants have enough energy.
5.3 Controlling Chemical Reactions Vocabulary: Activation energy Concentration Catalyst Enzyme Inhibitor - How do reactions get started? Chemical reactions won t begin until the reactants have enough energy.
Physical Changes can be observed without changing the identity of the substance (often states of matter changes).
 Physical Changes can be observed without changing the identity of the substance (often states of matter changes). Remember: States of matter changes are physical changes. The identify of the substance
Physical Changes can be observed without changing the identity of the substance (often states of matter changes). Remember: States of matter changes are physical changes. The identify of the substance
Chemical Reactions. Chemical Reaction occur when atoms collide. Reaction rates change from increasing or decreasing collisions
 Chemical Reactions Questions 1. What is a chemical reaction 2. What evidence suggests that a chemical reaction has taken place? 3. What factors can affect the rate of a chemical reaction (how fast the
Chemical Reactions Questions 1. What is a chemical reaction 2. What evidence suggests that a chemical reaction has taken place? 3. What factors can affect the rate of a chemical reaction (how fast the
Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds.
 Chapter 11 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Chapter 11 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Classification of Matter
 Matter in Chemistry Classification of Matter Elements: substances that are made from one type of atom only. Compounds: substances that are made from more than one type of atom chemically bonded together.
Matter in Chemistry Classification of Matter Elements: substances that are made from one type of atom only. Compounds: substances that are made from more than one type of atom chemically bonded together.
2-4 Chemical Reactions and Enzymes
 2-4 Chemical Reactions and Enzymes Living things, as you have seen, are made up of chemical compounds-some simple and some complex. But chemistry isn t just what life is made of-chemistry is also what
2-4 Chemical Reactions and Enzymes Living things, as you have seen, are made up of chemical compounds-some simple and some complex. But chemistry isn t just what life is made of-chemistry is also what
Matter. Anything that has mass and takes up space. Solids Liquids Gases
 Introduction to Matter Matter Anything that has mass and takes up space Solids Liquids Gases Weight A measure of the gravitational force exerted on an object What is the difference between weight and mass?
Introduction to Matter Matter Anything that has mass and takes up space Solids Liquids Gases Weight A measure of the gravitational force exerted on an object What is the difference between weight and mass?
Chemistry Part 2: We re Not Done Yet!
 Chemistry Part 2: We re Not Done Yet! WOW! Learning about atoms sure was fun! Let s continue our fascinating exploration of chemicals. What is a Chemical? Chemical: A substance having a constant composition
Chemistry Part 2: We re Not Done Yet! WOW! Learning about atoms sure was fun! Let s continue our fascinating exploration of chemicals. What is a Chemical? Chemical: A substance having a constant composition
Plants Lesson 2, Activity 2: Using Molecular Models to Explain Photosynthesis
 Environmental Literacy Project Michigan State University Plants Lesson 2, Activity 2: Using Molecular Models to Explain Photosynthesis Answering the Three Questions for plants in the light The Movement
Environmental Literacy Project Michigan State University Plants Lesson 2, Activity 2: Using Molecular Models to Explain Photosynthesis Answering the Three Questions for plants in the light The Movement
Cellular Respiration and Photosynthesis Test
 Cellular Respiration and Photosynthesis Test 1. When bonds are made energy is, when bonds are broken energy is. A. stored / released C. released / stored B. used / not used D. created / destroyed 2. Aerobic
Cellular Respiration and Photosynthesis Test 1. When bonds are made energy is, when bonds are broken energy is. A. stored / released C. released / stored B. used / not used D. created / destroyed 2. Aerobic
by Cyndee Crawford October 2014
 7-5 Chemical Nature of Matter PART 2 by Cyndee Crawford October 2014 7-5 The student will demonstrate an understanding of the classifications and properties of matter and the changes that matter undergoes.
7-5 Chemical Nature of Matter PART 2 by Cyndee Crawford October 2014 7-5 The student will demonstrate an understanding of the classifications and properties of matter and the changes that matter undergoes.
Reaction Rates and Equilibrium
 CHAPTER 7 14 SECTION Chemical Reactions Reaction Rates and Equilibrium KEY IDEAS As you read this section, keep these questions in mind: How can you increase the rate of a reaction? What does a catalyst
CHAPTER 7 14 SECTION Chemical Reactions Reaction Rates and Equilibrium KEY IDEAS As you read this section, keep these questions in mind: How can you increase the rate of a reaction? What does a catalyst
Matter: Properties and Changes
 Matter: Properties and Changes Matter and Its Changes What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree
Matter: Properties and Changes Matter and Its Changes What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree
Vocabulary: Matter: has mass and takes up space (pure substances and mixtures) Pure Substances: composition definite, elements and compounds.
 Unit 2: Composition and Properties of Matter Review Elements, Compounds, Mixtures and Physical/Chemical Properties and Changes, Water Properties and Biogeochemical Cycles Vocabulary: Matter: has mass and
Unit 2: Composition and Properties of Matter Review Elements, Compounds, Mixtures and Physical/Chemical Properties and Changes, Water Properties and Biogeochemical Cycles Vocabulary: Matter: has mass and
Chemical Reactions and Equations
 Chemical Reactions and Equations Question 1: Why should a magnesium ribbon be cleaned before burning in air? Magnesium is very reactive metal. When stored it reacts with oxygen to form a layer magnesium
Chemical Reactions and Equations Question 1: Why should a magnesium ribbon be cleaned before burning in air? Magnesium is very reactive metal. When stored it reacts with oxygen to form a layer magnesium
7.1 Describing Reactions
 Chapter 7 Chemical Reactions 7.1 Describing Reactions Chemical Equations Equation states what a reaction starts with, and what it ends with. Reactants the starting materials that undergo change. (On the
Chapter 7 Chemical Reactions 7.1 Describing Reactions Chemical Equations Equation states what a reaction starts with, and what it ends with. Reactants the starting materials that undergo change. (On the
Chapter 7 Chemical Reactions
 Chapter 7 Chemical Reactions Chemical Equation --> is a representation of a chemical reaction in which the reactants and products are expressed as formulas Reactants --> substances that undergo change
Chapter 7 Chemical Reactions Chemical Equation --> is a representation of a chemical reaction in which the reactants and products are expressed as formulas Reactants --> substances that undergo change
Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds.
 Chapter 9 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Chapter 9 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
4 Energy and Rates of Chemical Reactions
 CHAPTER 14 4 and Rates of Chemical Reactions SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: How is energy involved in a chemical reaction?
CHAPTER 14 4 and Rates of Chemical Reactions SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: How is energy involved in a chemical reaction?
Physical Change - alters the form or appearance of a substance but does not change it into a new, different substance
 Chemical Reactions Physical Change - alters the form or appearance of a substance but does not change it into a new, different substance Chemical Change - forms one or more new substances with properties
Chemical Reactions Physical Change - alters the form or appearance of a substance but does not change it into a new, different substance Chemical Change - forms one or more new substances with properties
Lesson #6: Chemical Reaction Types
 Lesson #6: Chemical Types Type #1 The Synthesis In this type of reaction 2 or more elements or compounds combine to form a more complex compound Two or more things become one bigger thing (ex) 2Mg + O
Lesson #6: Chemical Types Type #1 The Synthesis In this type of reaction 2 or more elements or compounds combine to form a more complex compound Two or more things become one bigger thing (ex) 2Mg + O
INTRODUCTION TO CHEMICAL REACTIONS THE COLLISION-REACTION THEORY. R. Ashby Duplication by permission only.
 CH 11 TOPIC 28 EVIDENCE OF CHEMICAL REACTIONS 1 You have mastered this topic when you can: 1) describe or define these terms: chemical reaction, chemical equation, reactants, and products 2) describe a
CH 11 TOPIC 28 EVIDENCE OF CHEMICAL REACTIONS 1 You have mastered this topic when you can: 1) describe or define these terms: chemical reaction, chemical equation, reactants, and products 2) describe a
Chemical Reactions. 1. What is the difference between a physical change and a chemical change
 What is a Chemical Reaction Chemical Reactions 1. What is the difference between a physical change and a chemical change 2. Give an example of a physical change and a chemical change using paper. 3. What
What is a Chemical Reaction Chemical Reactions 1. What is the difference between a physical change and a chemical change 2. Give an example of a physical change and a chemical change using paper. 3. What
Standards 8.5.c. I know chemical reactions usually liberate or absorbs heat.
 6 March 2013 Give an example of a physical change and a chemical change, and then describe how they are different from the other. Explain your answer in 2-3 sentences. Standards 8.5.c. I know chemical
6 March 2013 Give an example of a physical change and a chemical change, and then describe how they are different from the other. Explain your answer in 2-3 sentences. Standards 8.5.c. I know chemical
Name: 1. Which of the following is probably true about 300 ml of sand and 300 ml of water?
 Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name Class Date. Complete each of the following sentences by choosing the correct term from the word bank.
 Skills Worksheet Chapter Review USING KEY TERMS Complete each of the following sentences by choosing the correct term from the word bank. subscript exothermic reaction inhibitor synthesis reaction coefficient
Skills Worksheet Chapter Review USING KEY TERMS Complete each of the following sentences by choosing the correct term from the word bank. subscript exothermic reaction inhibitor synthesis reaction coefficient
Number 1 What is a chemical reaction?
 Chemical Reactions and Enzymes Number 1 What is a chemical reaction? A process that changes, or transforms, one set of chemicals into another by changing the chemical bonds that join atoms in compounds.
Chemical Reactions and Enzymes Number 1 What is a chemical reaction? A process that changes, or transforms, one set of chemicals into another by changing the chemical bonds that join atoms in compounds.
Chemical Reactions. Unit 4
 Chemical Reactions Unit 4 Lesson 1: Chemical Bonds Unit 4: Reactions Compounds Most substances around you are NOT elements. There are around 100 elements, but millions of different substances. Most substances
Chemical Reactions Unit 4 Lesson 1: Chemical Bonds Unit 4: Reactions Compounds Most substances around you are NOT elements. There are around 100 elements, but millions of different substances. Most substances
Chapter 9 Practice Test
 Chapter 9 Practice Test Multiple Choice Identify the choice that best completes the statement or answers the question. 1) Which of the following describes a chemical reaction? a) A gas is given off when
Chapter 9 Practice Test Multiple Choice Identify the choice that best completes the statement or answers the question. 1) Which of the following describes a chemical reaction? a) A gas is given off when
What is a chemical property of matter?
 What is a chemical property of matter? A. Chemical properties of matter involve a chemical change. 1. Observations of matter that involve a chemical change cause new matter to be formed. a. Chemical changes
What is a chemical property of matter? A. Chemical properties of matter involve a chemical change. 1. Observations of matter that involve a chemical change cause new matter to be formed. a. Chemical changes
Do Now. What is happening in the pictures below? How do you know? What evidence do you have to support your answer?
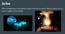 Do Now What is happening in the pictures below? How do you know? What evidence do you have to support your answer? Energy and Enzymes 5.10-5.16 Chemical Reactions Lab Clean Up Procedure Sample Data Do
Do Now What is happening in the pictures below? How do you know? What evidence do you have to support your answer? Energy and Enzymes 5.10-5.16 Chemical Reactions Lab Clean Up Procedure Sample Data Do
LESSON 1: DESCRIBING MATTER pg.5. Chemistry = Is the study of matter & how matter changes. Liquid/Solid/Gas
 Chemistry..CHAPTER 1: INTRO TO MATTER LESSON 1: DESCRIBING MATTER pg.5 Chemistry = Is the study of matter & how matter changes A. Matter = anything that has mass & takes up space à You, air, plastic, metal,
Chemistry..CHAPTER 1: INTRO TO MATTER LESSON 1: DESCRIBING MATTER pg.5 Chemistry = Is the study of matter & how matter changes A. Matter = anything that has mass & takes up space à You, air, plastic, metal,
CLASSIFYING MATTER. What is matter? -Anything that has mass and takes up space You are matter. The wall is matter. Light and sound are NOT matter
 MATTER What is it? CLASSIFYING MATTER What is matter? -Anything that has mass and takes up space You are matter The wall is matter Light and sound are NOT matter CLASSIFYING MATTER There are 3 types of
MATTER What is it? CLASSIFYING MATTER What is matter? -Anything that has mass and takes up space You are matter The wall is matter Light and sound are NOT matter CLASSIFYING MATTER There are 3 types of
Good morning, please do the following:
 Good morning, please do the following: Get out your What is Cellular Respiration? reading for a stamp Get out your Assignment Sheet Get out your concept maps for a check-in If you were absent on Monday,
Good morning, please do the following: Get out your What is Cellular Respiration? reading for a stamp Get out your Assignment Sheet Get out your concept maps for a check-in If you were absent on Monday,
Chemical Reactions: The Law of Conservation of Mass
 Chemical Reactions: The Law of Conservation of Mass What happens in a Chemical Reaction? Chemical bonds in the reactants are broken, then atoms are rearranged to form new substances (products). Reactants
Chemical Reactions: The Law of Conservation of Mass What happens in a Chemical Reaction? Chemical bonds in the reactants are broken, then atoms are rearranged to form new substances (products). Reactants
Chemical reactions. C2- Topic 5
 Chemical reactions C2- Topic 5 What is a chemical reaction? A chemical reaction is a change that takes place when one or more substances (called reactants) form one or more new substances (called products)
Chemical reactions C2- Topic 5 What is a chemical reaction? A chemical reaction is a change that takes place when one or more substances (called reactants) form one or more new substances (called products)
2 4 Chemical Reactions and Enzymes
 2 4 Chemical Reactions and Enzymes THINK ABOUT IT Living things are made up of chemical compounds, but chemistry isn t just what life is made of chemistry is also what life does. Everything that happens
2 4 Chemical Reactions and Enzymes THINK ABOUT IT Living things are made up of chemical compounds, but chemistry isn t just what life is made of chemistry is also what life does. Everything that happens
Properties of Matter. Mrs. Lapierre Chemistry
 Properties of Matter. Mrs. Lapierre Chemistry What is a property? In chemistry, a property is what we use to distinguish between different types of matter. There are two types: Chemical. Physical. Why
Properties of Matter. Mrs. Lapierre Chemistry What is a property? In chemistry, a property is what we use to distinguish between different types of matter. There are two types: Chemical. Physical. Why
Chemical Reactions and Equations
 Name Date Chemical Reactions and Equations What happens to atoms and energy during a chemical reaction? Before You Read Before you read the chapter, think about what you know about chemical reactions Record
Name Date Chemical Reactions and Equations What happens to atoms and energy during a chemical reaction? Before You Read Before you read the chapter, think about what you know about chemical reactions Record
8.5E: Chemical Reactions
 Reflect Have you ever seen fireworks explode in the sky? If so, you may have observed a variety of different colors and shapes in the sky and watched with amazement as the colors changed overhead. These
Reflect Have you ever seen fireworks explode in the sky? If so, you may have observed a variety of different colors and shapes in the sky and watched with amazement as the colors changed overhead. These
4. Every CHANGE in matter includes a change in, which is conserved in a chemical reaction and. TRANSFORMED from one form to another.
 Part C: Lesson 1.4 - Read pages 20-29 to answer these questions. Add to notes - 1. A physical change alters the form or appearance of matter, such as Change in size, a change in SIZE or. SHAPE shape, or
Part C: Lesson 1.4 - Read pages 20-29 to answer these questions. Add to notes - 1. A physical change alters the form or appearance of matter, such as Change in size, a change in SIZE or. SHAPE shape, or
1.Matter and Organic Compounds Matter =
 The Chemistry of Life Notes Unit 2 1.Matter and Organic Compounds Matter = All things are made of matter Name Matter is made up of substances Chemical substance = definite composition throughout Either
The Chemistry of Life Notes Unit 2 1.Matter and Organic Compounds Matter = All things are made of matter Name Matter is made up of substances Chemical substance = definite composition throughout Either
A simple equation of what happens when you add baking soda to vinegar:
 What s the Matter? Matter is anything that takes up space and has mass. Mass is the stuff that matter is made of, or the amount of particles in a substance or object. Matter has physical and chemical properties
What s the Matter? Matter is anything that takes up space and has mass. Mass is the stuff that matter is made of, or the amount of particles in a substance or object. Matter has physical and chemical properties
Chemistry Review- Packet 11, Page 1
 Chemistry Review- Packet 11, Page 1 Atom- smallest particle of an element with the same properties as that element In size the entire atom has been thought to be approximately four-billionths of an inch,
Chemistry Review- Packet 11, Page 1 Atom- smallest particle of an element with the same properties as that element In size the entire atom has been thought to be approximately four-billionths of an inch,
Section 1 Chemical Changes
 Chemical Reactions Section 1 Chemical Changes What You ll Learn: How to identify the reactants & products in a chemical reaction How a chemical reaction follows the law of conservation of mass How chemists
Chemical Reactions Section 1 Chemical Changes What You ll Learn: How to identify the reactants & products in a chemical reaction How a chemical reaction follows the law of conservation of mass How chemists
Chemical Reactions Chapter 2 L book pages L44 - L73. examples?
 Name: Period: Chemical Reactions Chapter 2 L book pages L44 - L73 Vocabulary Word What is this? (definition) What are some examples? What does it look like? (draw a picture or diagram) Physical property
Name: Period: Chemical Reactions Chapter 2 L book pages L44 - L73 Vocabulary Word What is this? (definition) What are some examples? What does it look like? (draw a picture or diagram) Physical property
copper carbonate copper oxide carbon dioxide Is the reaction exothermic or endothermic? Use ideas about bonds to explain why.
 Endothermic and Exothermic reaction Worksheet Name date period 1 Exothermic and endothermic reactions Decide whether each of these reactions is exothermic or endothermic: a) When two chemicals mix their
Endothermic and Exothermic reaction Worksheet Name date period 1 Exothermic and endothermic reactions Decide whether each of these reactions is exothermic or endothermic: a) When two chemicals mix their
Name Date. Physical and Chemical Changes
 Name Date Physical and Chemical Changes Physical Changes Physical Change: A change in which no new substances form Do not change the type of matter an object is made of You change the shape, size or more
Name Date Physical and Chemical Changes Physical Changes Physical Change: A change in which no new substances form Do not change the type of matter an object is made of You change the shape, size or more
Chemical formula - tells you how many atoms of each element are in a compound example: CO 2 (carbon dioxide) has one carbon atom and two oxygen atoms
 Chemical Reactions Chemical formula - tells you how many atoms of each element are in a compound example: CO 2 (carbon dioxide) has one carbon atom and two oxygen atoms 2 points Chemical Equation - a short,
Chemical Reactions Chemical formula - tells you how many atoms of each element are in a compound example: CO 2 (carbon dioxide) has one carbon atom and two oxygen atoms 2 points Chemical Equation - a short,
Gummy Bear Demonstration:
 Name: Unit 8: Chemical Kinetics Date: Regents Chemistry Aim: _ Do Now: a) Using your glossary, define chemical kinetics: b) Sort the phrases on the SmartBoard into the two columns below. Endothermic Rxns
Name: Unit 8: Chemical Kinetics Date: Regents Chemistry Aim: _ Do Now: a) Using your glossary, define chemical kinetics: b) Sort the phrases on the SmartBoard into the two columns below. Endothermic Rxns
Chemical Reactions and Equations
 Chemical Reactions and Equations Changes and Chemical Reactions Key Concepts Why do chemical reactions always involve a change in? What is the difference between an endothermic reaction and an exothermic
Chemical Reactions and Equations Changes and Chemical Reactions Key Concepts Why do chemical reactions always involve a change in? What is the difference between an endothermic reaction and an exothermic
Name Date Class. This section explains how plants make food by using the energy from sunlight.
 Cell Processes and Energy Name Date Class Photosynthesis This section explains how plants make food by using the energy from sunlight. Use Target Reading Skills As you read, create a flowchart that shows
Cell Processes and Energy Name Date Class Photosynthesis This section explains how plants make food by using the energy from sunlight. Use Target Reading Skills As you read, create a flowchart that shows
IN THE PAST WEEKS WE SPOKE ABOUT THE 4 ORGANIC MOLECULES THAT ARE INSIDE THE CELL, SUCH AS PROTEINS, CARBOHYDRATES, LIPIDS AND NUCLEIC ACIDS
 IN THE PAST WEEKS WE SPOKE ABOUT THE 4 ORGANIC MOLECULES THAT ARE INSIDE THE CELL, SUCH AS PROTEINS, CARBOHYDRATES, LIPIDS AND NUCLEIC ACIDS CARBOHYDRATES ARE THE IMMEDIATE SOURCE OF ENERGY FOR THE CELL
IN THE PAST WEEKS WE SPOKE ABOUT THE 4 ORGANIC MOLECULES THAT ARE INSIDE THE CELL, SUCH AS PROTEINS, CARBOHYDRATES, LIPIDS AND NUCLEIC ACIDS CARBOHYDRATES ARE THE IMMEDIATE SOURCE OF ENERGY FOR THE CELL
WHAT IS CHEMISTRY? Chapter Preview Questions
 WHAT IS CHEMISTRY? 1. A piece of iron is made up of a. iron molecules. b. iron compounds. c. iron atoms. d. iron salts. 1 1. A piece of iron is made up of a. iron molecules. b. iron compounds. c. iron
WHAT IS CHEMISTRY? 1. A piece of iron is made up of a. iron molecules. b. iron compounds. c. iron atoms. d. iron salts. 1 1. A piece of iron is made up of a. iron molecules. b. iron compounds. c. iron
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
Notes: Balancing Chemical Equations
 Notes: Balancing Chemical Equations Effects of chemical reactions: Chemical reactions rearrange atoms in the reactants to form new products. The identities and properties of the products are completely
Notes: Balancing Chemical Equations Effects of chemical reactions: Chemical reactions rearrange atoms in the reactants to form new products. The identities and properties of the products are completely
Science 10. Unit 2: Chemistry. Book 6: energy changes in chemical reactions. Block: Name: Zukowski
 Science 10 Unit 2: Chemistry Book 6: energy changes in chemical reactions Name: Zukowski Block: 1 How is energy involved in chemical processes? and energy are continually interacting in the world around
Science 10 Unit 2: Chemistry Book 6: energy changes in chemical reactions Name: Zukowski Block: 1 How is energy involved in chemical processes? and energy are continually interacting in the world around
SNC2D CHEMISTRY 2/23/2013. CHEMICAL REACTIONS L Conservation of Mass (P ) Activity: Measuring Mass (Part 1) Activity: Measuring Mass (Part 1)
 SNC2D CHEMISTRY CHEMICAL REACTIONS L Conservation of Mass (P.176-177) Activity: Measuring Mass (Part 1) INSTRUCTIONS (CLOSED SYSTEM) A. Measure~ 30 ml of vinegar (acetic acid) into a small Erlenmeyer flask.
SNC2D CHEMISTRY CHEMICAL REACTIONS L Conservation of Mass (P.176-177) Activity: Measuring Mass (Part 1) INSTRUCTIONS (CLOSED SYSTEM) A. Measure~ 30 ml of vinegar (acetic acid) into a small Erlenmeyer flask.
Understanding Equations
 Chemical Reactions Chemical reaction: a process of chemically changing both the physical and chemical properties of a substance to a new substance with different physical and chemical properties. Video
Chemical Reactions Chemical reaction: a process of chemically changing both the physical and chemical properties of a substance to a new substance with different physical and chemical properties. Video
2 4 Chemical Reactions
 SECTION 2 4 Chemical Reactions ^^^^^ ^^^ ^^ tff'\f f**^*.tffn*f i***i i i «i f.' KEY CONCEPT Life depends on chemical reactions. Student text pages 50-53 Bonds break and form during chemical reactions.
SECTION 2 4 Chemical Reactions ^^^^^ ^^^ ^^ tff'\f f**^*.tffn*f i***i i i «i f.' KEY CONCEPT Life depends on chemical reactions. Student text pages 50-53 Bonds break and form during chemical reactions.
Law of Conservation of Mass
 Law of Conservation of Mass The Universe is made up of matter and energy. Stone, table and hydrogen gas are some examples of matter. Matter occupies as pacific space and has specific mass. Energy is the
Law of Conservation of Mass The Universe is made up of matter and energy. Stone, table and hydrogen gas are some examples of matter. Matter occupies as pacific space and has specific mass. Energy is the
Physical & Chemical PROPERTIES
 Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Material cycles and energy: photosynthesis
 7 Material cycles and energy: photosynthesis Remember: Plants are living organisms and can carry out all the life processes. Plants must be able to make foods. The foods provide raw materials for growth
7 Material cycles and energy: photosynthesis Remember: Plants are living organisms and can carry out all the life processes. Plants must be able to make foods. The foods provide raw materials for growth
Energy Changes in Chemical and Nuclear Reactions
 Energy Changes in Chemical and Nuclear Reactions What happens when matter undergoes change? Clearly, new substances or states are produced, but energy changes also occur. If chemistry is the study of matter
Energy Changes in Chemical and Nuclear Reactions What happens when matter undergoes change? Clearly, new substances or states are produced, but energy changes also occur. If chemistry is the study of matter
Characteristics & Types of Chemical Reactions
 Characteristics & Types of Chemical Reactions What is a chemical reaction? Another name for a chemical change Bonds break and atoms of one or more substances are rearranged and new bonds form New substance(s)
Characteristics & Types of Chemical Reactions What is a chemical reaction? Another name for a chemical change Bonds break and atoms of one or more substances are rearranged and new bonds form New substance(s)
Photosynthesis and Cellular Respiration Lapbook Pre-test. SC.912.L.18.7: Identify the reactants, products, and basic functions of photosynthesis.
 Photosynthesis and Cellular Respiration Lapbook Pre-test Covers Standards: SC.912.L.18.7: Identify the reactants, products, and basic functions of photosynthesis. SC.912.L.18.8: Identify the reactants,
Photosynthesis and Cellular Respiration Lapbook Pre-test Covers Standards: SC.912.L.18.7: Identify the reactants, products, and basic functions of photosynthesis. SC.912.L.18.8: Identify the reactants,
2-1: Describing Matter. 8 th Grade Physical Sciences
 8 th Grade Physical Sciences What is Matter? Matter is anything that has mass and takes up space. Properties of Matter Matter can be described in many ways; hard, soft, heavy, light, rough, smooth,
8 th Grade Physical Sciences What is Matter? Matter is anything that has mass and takes up space. Properties of Matter Matter can be described in many ways; hard, soft, heavy, light, rough, smooth,
Chemical Equations. Physical Science
 Chemical Equations Physical Science Chemical Equations A chemical equation is a shorthand way of expressing a chemical reaction. CHEMICAL REACTIONS Reactants: Zn + I 2 Product: Zn I 2 Chemical reactions
Chemical Equations Physical Science Chemical Equations A chemical equation is a shorthand way of expressing a chemical reaction. CHEMICAL REACTIONS Reactants: Zn + I 2 Product: Zn I 2 Chemical reactions
