CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED)
|
|
|
- Egbert Sherman
- 5 years ago
- Views:
Transcription
1 CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student will: identify that a chemical reaction has taken place; perform an experiment using variables to create chemical reactions; and identify chemical reactions that have taken place around them. Targeted Alaska Grade Level Expectations: Science [9] SB3.1 The student demonstrates an understanding of the interactions between matter and energy and the effects of these interactions on the systems by recognizing that a chemical reaction has taken place. [9] SA1.1 The student demonstrates an understanding of the process of science by asking questions, predicting, observing, describing, measuring, classifying, making generalizations, inferring, and communicating. [9] SA1.2 The student demonstrates an understanding of the processes of science by hypothesizing, designing a controlled experiment, making qualitative and quantitative observations, interpreting data, and using this information to communicate conclusions. Vocabulary: chemical relating to or produced by means of chemistry; a substance obtained by or used in a chemical process; a chemical compound chemical reaction a rearrangement of the atoms or molecules of two or more substances that come into contact with each other, resulting in the formation of one or more new substances chemistry the scientific study of the structure, properties, and reactions of the chemical elements and the compounds they form; the composition, structure, properties, and reactions of a substance products a substance produced during a natural, chemical or manufacturing process reactant a substance participating in a chemical reaction, especially one present at the start of the reaction Materials: Gear up (teacher demonstration): Plastic zipper type sandwich baggie Baking soda (sodium bicarbonate 2NaHCO 3 ) Dilute hydrochloric acid HCl or vinegar (approximately 2 milliliters) Plastic pipette Chart paper posted on walls (3-4 pieces) Triple beam balance or electronic milligram balance (one per group) Plastic zipper bag (three or more per group) Plastic pipette (one per group) Alka Seltzer tablets Antacid tablets White chalk Safety goggles (one per student) Chemical aprons (one per student) Science journal or blank piece of paper STUDENT LAB: Plop, Plop, Fizz, Fizz
2 CHEMICAL REACTION IN A BAGGY Activity Preparation: 1. Make sure all materials are available for demonstration and student activity. Small groups are asked to perform three separate trials using the materials, so make sure enough is available. 2. Students will wear safety gear: eye protection and chemical aprons. Be sure these items are available. 3. Students must properly dispose of materials. Have a procedure in place before starting. 4. Teacher should wear gear to set a good example. Activity Procedure: NOTE: The scoring guide and assessment task are located at the end of the lesson. Please share them with students before beginning the lesson. Gear Up: Process Skills: Observing, communicating 1. Begin by asking students the following questions about what they know about chemical reactions, and record their responses on chart paper. a. How can you tell if a chemical reaction has taken place? b. How do you know this is not a physical change? c. What are some examples of chemical reactions? 2. Perform the following demonstration: Place 5 grams of baking soda in a baggie. Fill pipette with approximately 2 milliliters of vinegar. Carefully place filled pipette in the bag and seal the baggie. Be careful not to allow any liquid to leak from the pipette. Direct the stem of the pipette toward the baking soda, and squeeze the contents of the pipette so that it reacts with the baking soda. Immediately remove the pipette and seal the baggie. Allow students to make observations of the contents of the bag, noting bubble formation and baggie expansion. Discuss observations. 3. Write the definition for chemical reaction on the board (see Vocabulary). Ask: What chemical reactions are going on around you every day? Record student responses on the chart paper. Explore: Process Skills: Predicting, observing, inferring 4. Divide students into small groups then hand out STUDENT LAB: Plop, Plop, Fizz, Fizz. Show students the materials listed under Reactants on the lab sheet. The lab sheet asks students, Which set of reactants will produce new substances when combined? List three possibilities. Ask students to think about different combinations that might produce a reaction. Have students record their predictions on the lab sheet. Ask groups to share predictions and record them on chart paper or the board. 5. Review lab procedures required in STUDENT LAB: Plop, Plop, Fizz, Fizz, including the importance following instructions step by step. Provide student groups with listed materials to use and explore. Students are asked to perform two trials, then make a list on the board of reactant combinations that produced a chemical reaction. Circulate to assess understanding. Ask students to complete the lab up to the conclusion, then clean up. 6. After clean up is complete, allow students to complete the lab, Part Three: Conclusion. This can be done in small groups or as an individual assignment. 7. Assessment Task: In their science journal or on a blank piece of paper students will write at least one paragraph using complete sentences and proper grammar. The paragraph must explain what a chemical reaction is and give at least two examples. The student will describe at least one chemical reaction that she/ he encounters in everyday life.
3 CHEMICAL REACTION IN A BAGGY Answers: STUDENT LAB: Plop, Plop, Fizz, Fizz Part One: Prediction A. and B. Answers will vary. Part Two: Investigation All sections of the Investigation are dependent on student investigation. Answers will vary, but should reflect that student groups recorded their observations. Students provide a new prediction for the third trial. Part Three: Conclusion 1. Any two of the following: color change, temperature change, precipitate has formed, an odor is produced, a texture change, weight change, a new substance has formed, gas bubbles form, it can no longer be separated into its original form, or other answers that are correct and appropriate. 2. You will not get an accurate assessment of the reaction that has occurred. 3. If the bag was not sealed there certainly could have been a mess, but more importantly some reactions produced gas. To accurately assess the reaction, the gas had to be captured and included in the weight. The gas (CO 2 ) was the result of the chemical reaction releasing the CO 2 from the solid material. (Note: Baking soda and vinegar yields sodium acetate, water, and carbon dioxide: NaHCO 3 + CH 3 COOH > NaCH 3 COO + H 2 CO 3 >NaCH 3 COO+H 2 O+CO 2.) 4. The gas is generated from the chemical reaction. 5. Answers will vary, but may include the combustion of fossil fuels to energy (light, heat and carbon dioxide); digestion of food is transformed into energy; sunlight, water, and carbon dioxide photosynthesized by plants; decomposition, where plant and animal material break down into water and carbon dioxide; and burning wood releases energy and carbon dioxide. Scoring Guide GLE/Standard Below Proficient Proficient Above Proficient GLE [9] SB3.1 Student explains what a chemical reaction is and gives one example Student explains what a chemical reaction is and gives two examples. Student explains what a chemical reaction is and gives three or more examples.
4 Part One: Prediction 1. Which set of reactants will produce a new substance when combined? Combine one liquid and one solid each time. A. Choose two reactants: B. Change out one of the above reactants: Part Two: Investigation TRIAL ONE A 1. List the two reactants from Prediction A. Include the quantity you will use to start. Recommended quantities are listed on the right. Materials Triple beam balance (one) Plastic zipper bag (up to nine) Plastic pipette (one) Reactants Baking Soda (sodium bicarbonate 2NaHCo 3 ) Vinegar Water (H 2 O) Alka Seltzer tablets Antacid tablets White chalk 2. What do you think will happen when these two reactants are combined? 3. Using a plastic zipper bag as your container, use the pipette to add the correct amount of any liquid listed as a reactant. Seal the bag. Place the bag (with liquid inside), and any solid reactant on the triple beam balance and weigh. Quantities at Start Baking soda (2NaHCO 3 ) start with 5 grams. Vinegar start with 2 milliliters Hydrochloric Acid (HCl) start with 2 milliliters Alka Seltzer start with one tablet Antacid start with one tablet White chalk start with 3/4 inch piece Weight prior to reaction: 4. Add the second reactant to the baggie and immediately seal. Describe the observed reaction. Did it match your prediction? 5. Weigh the sealed bag. Weight after reaction: Did the weight change? What is the difference, if any?
5 6. Open the baggie (without spilling the contents) and squeeze out excess gases. Reseal the baggie and weigh it again. Weight: 7. Discuss any difference in weight between steps 4 and 6. To what do you attribute any difference? TRIAL ONE B 1. List the two reactants from Prediction B on page one. Include the quantity you will use to start. Recommended quantities are listed on page one. 2. What do you think will happen when these two reactants are combined? 3. Using a plastic zipper bag as your container, use the pipette to add the correct amount of any liquid listed as a reactant. Seal the bag. Place the bag (with liquid inside), and any solid reactant on the triple beam balance and weigh. Weight prior to reaction: 4. Add the second reactant to the baggie and immediately seal. Describe the observed reaction. Did it match your prediction? 5. Weigh the sealed bag. Weight after reaction: 6. Open the baggie (without spilling the contents) and squeeze out excess gases. Reseal the baggie and weigh it again. Weight: 7. Discuss any different in weight between steps 4 and 6. To what so you attribute any difference?
6 8. Report results of Trials A and B to the class. On the board, make a list of combined reactants that produced a chemical reaction. TRIAL ONE C 1. From the list on the board, choose two of the reactants that produced a chemical reaction. Change the amount of one or both of the reactants. List the quantity you will use. 2. Make a new prediction. What do you think will happen when the amount of the reactant(s) has changed? 3. Using a plastic zipper bag as your container, use the pipette to add the correct amount of any liquid listed as a reactant. Seal the bag. Place the bag (with liquid inside), and all solid reactants on the triple beam balance and weigh. Weight prior to reaction: 4. Add all of the reactants to the baggie and immediately seal. Describe the observed reaction. Did it match your prediction? 5. Weigh the sealed bag. Weight after reaction: 6. Open the baggie (without spilling the contents) and squeeze out excess gases. Reseal the baggie and weigh it again. Weight: 7. Discuss any different in weight between steps 3 and 6. To what do you attribute any difference? Part Three: Conclusion Directions: Answer the following critical thinking questions. 1. List ways you can recognize that a chemical reaction has taken place. 2. Why is important that measurements are made accurately?
7 3. Why was it necessary for the plastic baggie to be sealed? 4. When the bag(s) expanded, gas had been released. Where did it come from? Part Four: Application 1. What chemical reactions are taking place in the environment around you right now? Name at least three. (Think about your community, school and home.)
Moles Lab Activity 1: PCU (Popcorn Counting Units)
 Moles Lab Activity 1: PCU (Popcorn Counting Units) Materials: A container of each of the following: Popcorn kernels Another type of beans A large unopened bag of popcorn Kernels Balance Safety goggles
Moles Lab Activity 1: PCU (Popcorn Counting Units) Materials: A container of each of the following: Popcorn kernels Another type of beans A large unopened bag of popcorn Kernels Balance Safety goggles
Limiting Reactants Lab
 Name: Teacher s Name: Class: Block: Date: Partners: Limiting Reactants Lab Purpose: Through experimentation, determine the limiting reactant and the percent yield in a chemical reaction that generates
Name: Teacher s Name: Class: Block: Date: Partners: Limiting Reactants Lab Purpose: Through experimentation, determine the limiting reactant and the percent yield in a chemical reaction that generates
Matter Lesson 2. Learning Goal 3: I can describe the differences between physical and chemical changes of matter.
 Matter Lesson 2 Learning Goal 2: I can describe the differences between intensive physical properties, extensive physical properties, and chemical properties of matter. Learning Goal 3: I can describe
Matter Lesson 2 Learning Goal 2: I can describe the differences between intensive physical properties, extensive physical properties, and chemical properties of matter. Learning Goal 3: I can describe
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic
 Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Bellevue College CHEM& 121 Experiment: Stoichiometric Analysis of an Antacid 1
 Experiment: Stoichiometric Analysis of an Antacid 1 Introduction In this lab, you will use the concept of stoichiometry to solve two sequential problems. First, you will try to determine the products of
Experiment: Stoichiometric Analysis of an Antacid 1 Introduction In this lab, you will use the concept of stoichiometry to solve two sequential problems. First, you will try to determine the products of
Chapter 6, Lesson 9: Neutralizing Acids and Bases
 Chapter 6, Lesson 9: Neutralizing Acids and Bases Key Concepts ph is a measure of the concentration of H 3 O + ions in a solution. Adding an acid increases the concentration of H 3 O + ions in the solution.
Chapter 6, Lesson 9: Neutralizing Acids and Bases Key Concepts ph is a measure of the concentration of H 3 O + ions in a solution. Adding an acid increases the concentration of H 3 O + ions in the solution.
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions
 Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic
 Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic Name Date DEMONSTRATION 1. Your teacher blew through a straw into a universal indicator solution until it changed color. Did
Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic Name Date DEMONSTRATION 1. Your teacher blew through a straw into a universal indicator solution until it changed color. Did
MiSP CHEMICAL REACTIONS, L3 Teacher Guide. Introduction
 MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
Chemical Reaction Lab Bagged Chemical Reactions
 Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
Chemistry: classifying chemical and physical changes in various materials/substances
 Chemistry: classifying chemical and physical changes in various materials/substances Nikki Schilling, Ames, St. Paul Mn Based on original activity from Cool Chemistry Concoctions by Joe Rhatigan and Veronika
Chemistry: classifying chemical and physical changes in various materials/substances Nikki Schilling, Ames, St. Paul Mn Based on original activity from Cool Chemistry Concoctions by Joe Rhatigan and Veronika
Chapter 6, Lesson 1: What is a Chemical Reaction?
 Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
CO 2. Lesson 1. Production of a gas
 Lesson 1 Production of a gas T E A C H E R G U I D E CO 2 Lesson summary Students meet volcanologist Victor Helguson, who is studying the gases released by volcanoes in Iceland. Students conduct a chemical
Lesson 1 Production of a gas T E A C H E R G U I D E CO 2 Lesson summary Students meet volcanologist Victor Helguson, who is studying the gases released by volcanoes in Iceland. Students conduct a chemical
MiSP Chemical Reactions Lab Temperature L2. Plop, Plop, Fizz, Fizz... Temperature and Rate of Reaction Activity
 MiSP Chemical Reactions Lab Temperature L2 Name Date Plop, Plop, Fizz, Fizz... Temperature and Rate of Reaction Activity Introduction: Problem: Alka-Seltzer in water produces carbon dioxide. It is the
MiSP Chemical Reactions Lab Temperature L2 Name Date Plop, Plop, Fizz, Fizz... Temperature and Rate of Reaction Activity Introduction: Problem: Alka-Seltzer in water produces carbon dioxide. It is the
Maintaining Mass Created By: Allyn Short
 Maintaining Mass Created By: Allyn Short Focus on Inquiry The student will demonstrate that mass is conserved when substances undergo chemical and/or physical changes through experimentation and evaluation
Maintaining Mass Created By: Allyn Short Focus on Inquiry The student will demonstrate that mass is conserved when substances undergo chemical and/or physical changes through experimentation and evaluation
What Do You Think? Investigate GOALS
 Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Stoichiometry: Baking Soda and Vinegar Reactions Student Version
 Stoichiometry: Baking Soda and Vinegar Reactions Student Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Stoichiometry: Baking Soda and Vinegar Reactions Student Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Virtual Library Lesson: Oobleck, Gloop, and Glurch
 Oobleck, Gloop, and Glurch Lesson Overview Throughout this lesson, students will use inquiry skills to identify states of matter, describe physical properties, and modify the recipe to change physical
Oobleck, Gloop, and Glurch Lesson Overview Throughout this lesson, students will use inquiry skills to identify states of matter, describe physical properties, and modify the recipe to change physical
Lesson 2. Color change
 Lesson 2 Color change T E A C H E R G U I D E Lesson summary Students meet marine chemist Sera Tuikabe, who is studying ocean acidification in the water surrounding the Republic of the Fiji Islands. Students
Lesson 2 Color change T E A C H E R G U I D E Lesson summary Students meet marine chemist Sera Tuikabe, who is studying ocean acidification in the water surrounding the Republic of the Fiji Islands. Students
Characteristics of Chemical Change
 Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version
 Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
VANDERBILT STUDENT VOLUNTEERS FOR SCIENCE
 VANDERBILT STUDENT VOLUNTEERS FOR SCIENCE http://studentorgs.vanderbilt.edu/vsvs Evidence of a Chemical Reaction Fall 2018 Goal: To show students evidence of a chemical change. Fits TN standards:7ps1.2,
VANDERBILT STUDENT VOLUNTEERS FOR SCIENCE http://studentorgs.vanderbilt.edu/vsvs Evidence of a Chemical Reaction Fall 2018 Goal: To show students evidence of a chemical change. Fits TN standards:7ps1.2,
1. Write date, topic and purpose in notebook. 2. Answer in your notebook: What are two signs that a chemical reaction is happening?
 Warm Up (5 min) Date: Monday, 1/4/16 Topic: Alka Seltzer Lab Purpose: To assess the Law of Conservation of Mass by designing an experiment to determine mass before and after a chemical reaction. 1. Write
Warm Up (5 min) Date: Monday, 1/4/16 Topic: Alka Seltzer Lab Purpose: To assess the Law of Conservation of Mass by designing an experiment to determine mass before and after a chemical reaction. 1. Write
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from
 Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
ELECTROMAGNETIC WAVES AND CLIMATE (MODIFIED FOR ADEED)
 (MODIFIED FOR ADEED) Science Concept: Different frequencies of electromagnetic radiation behave differently in the atmosphere. Objectives: The student will: explain interactions between different frequencies
(MODIFIED FOR ADEED) Science Concept: Different frequencies of electromagnetic radiation behave differently in the atmosphere. Objectives: The student will: explain interactions between different frequencies
The Eight Solution Problem Exploring Reactions of Aqueous Ionic Compounds
 The Eight Solution Problem Exploring Reactions of Aqueous Ionic Compounds About this Lesson This activity allows students to mix a variety of known ionic solutions while making careful observations. After
The Eight Solution Problem Exploring Reactions of Aqueous Ionic Compounds About this Lesson This activity allows students to mix a variety of known ionic solutions while making careful observations. After
MiSP Chemical Reactions Lab Temperature L3. Plop, Plop, Fizz, Fizz... Temperature and Rate of Reaction Activity
 MiSP Chemical Reactions Lab Temperature L3 Name Date Plop, Plop, Fizz, Fizz... Temperature and Rate of Reaction Activity Introduction: Problem: Alka-Seltzer in water produces carbon dioxide. It is the
MiSP Chemical Reactions Lab Temperature L3 Name Date Plop, Plop, Fizz, Fizz... Temperature and Rate of Reaction Activity Introduction: Problem: Alka-Seltzer in water produces carbon dioxide. It is the
Apply the ideal gas law (PV = nrt) to experimentally determine the number of moles of carbon dioxide gas generated
 Teacher Information Ideal Gas Law Objectives Determine the number of moles of carbon dioxide gas generated during a reaction between hydrochloric acid and sodium bicarbonate. Through this investigation,
Teacher Information Ideal Gas Law Objectives Determine the number of moles of carbon dioxide gas generated during a reaction between hydrochloric acid and sodium bicarbonate. Through this investigation,
Bags of Reactions Chemistry I Acc
 Introduction: Bags of Reactions Chemistry I Acc Plop, plop, fizz, fizz, oh, what a relief it is. claims the old TV ad for a popular antacid. Just what is in the tablet that is relieving the upset stomach?
Introduction: Bags of Reactions Chemistry I Acc Plop, plop, fizz, fizz, oh, what a relief it is. claims the old TV ad for a popular antacid. Just what is in the tablet that is relieving the upset stomach?
Safety: Safety goggles should be worn at all times. Students should hold the balloons on the test tubes tightly while the reaction takes place.
 LIMITING REAGENT LAB: THE REACTION BETWEEN VINEGAR AND BAKING SODA Goal: During this lab students will gain a quantitative understanding of limiting reagents. Safety: Safety goggles should be worn at all
LIMITING REAGENT LAB: THE REACTION BETWEEN VINEGAR AND BAKING SODA Goal: During this lab students will gain a quantitative understanding of limiting reagents. Safety: Safety goggles should be worn at all
Student Notes. Chemical Reactions LINK
 LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
Acid Rain Eats Stone!
 SIDE DISPLAY Acid Rain Eats Stone! Visitors drip sulfuric acid onto one piece of chalk and water onto another piece of chalk and observe the results. OBJECTIVES: Visitors learn the composition of acid
SIDE DISPLAY Acid Rain Eats Stone! Visitors drip sulfuric acid onto one piece of chalk and water onto another piece of chalk and observe the results. OBJECTIVES: Visitors learn the composition of acid
What Do You Think? Investigate GOALS
 Ideal Toy Activity 7 Moving Molecules GOALS In this activity you will: Determine the effect of molecular size on molecular motion. Predict quantities of gas produced in chemical reactions. What Do You
Ideal Toy Activity 7 Moving Molecules GOALS In this activity you will: Determine the effect of molecular size on molecular motion. Predict quantities of gas produced in chemical reactions. What Do You
Lab- Properties of Acids and Bases. Name. PSI Chemistry
 Lab- Properties of Acids and Bases PSI Chemistry Name Introduction Acids and bases are useful reagents in the chemistry laboratory and play an important role in biology and nature. What are acids and bases?
Lab- Properties of Acids and Bases PSI Chemistry Name Introduction Acids and bases are useful reagents in the chemistry laboratory and play an important role in biology and nature. What are acids and bases?
2/22/2019 NEW UNIT! Chemical Interactions. Atomic Basics #19
 NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
Physical and Chemical Changes Or How Do You Know When You ve Made Something New?
 Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Percentage of Acetic Acid in Vinegar
 Microscale Percentage of Acetic Acid in Vinegar When sweet apple cider is fermented in the absence of oxygen, the product is an acid, vinegar. Most commercial vinegars are made by fermentation, but some,
Microscale Percentage of Acetic Acid in Vinegar When sweet apple cider is fermented in the absence of oxygen, the product is an acid, vinegar. Most commercial vinegars are made by fermentation, but some,
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from
 Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
Stoichiometry: Baking Soda and Vinegar Reactions Student Advanced Version
 Stoichiometry: Baking Soda and Vinegar Reactions Student Advanced Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household
Stoichiometry: Baking Soda and Vinegar Reactions Student Advanced Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household
Conservation of Mass in Chemical Reactions Lab. Experiment Question: What happens to the total mass of substances when a chemical reaction occurs?
 Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
Jell-O Rocks. Science Concept: Objectives: GLEs Addressed: Vocabulary: Materials: Most rocks are made of different substances.
 Science Concept: Most rocks are made of different substances. Objectives: The student will: describe a variety of rocks; observe and communicate about rocks; and take a photograph of a rock and describe
Science Concept: Most rocks are made of different substances. Objectives: The student will: describe a variety of rocks; observe and communicate about rocks; and take a photograph of a rock and describe
KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator
 KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator By Darby Sloss and Marianne Smith Edited by Anne Starace Abstract Chemistry is an important part of our lives. Kitchen Chemistry
KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator By Darby Sloss and Marianne Smith Edited by Anne Starace Abstract Chemistry is an important part of our lives. Kitchen Chemistry
Name. Pre-Test. Directions: Circle the T if a statement is true or F if it is false (F). 1. Matter always stays the same.
 1 Pre-est Directions: Circle the if a statement is true or if it is false (). 1. Matter always stays the same. 2. A physical change is a change in size, shape or phase of matter, without the matter changing
1 Pre-est Directions: Circle the if a statement is true or if it is false (). 1. Matter always stays the same. 2. A physical change is a change in size, shape or phase of matter, without the matter changing
JHS Regents Chemistry Department Rates of Chemical Reactions Inquiry Investigation
 Name: Date: Partner: Period: JHS Regents Chemistry Department Rates of Chemical Reactions Inquiry Investigation Introduction: According to the collision theory, the rate of a reaction depends on the frequency
Name: Date: Partner: Period: JHS Regents Chemistry Department Rates of Chemical Reactions Inquiry Investigation Introduction: According to the collision theory, the rate of a reaction depends on the frequency
Pre-Lab Exercises Lab 3: Chemical Properties
 Pre-Lab Exercises Lab 3: Chemical Properties 1. How is a chemical property different from a physical property? Name Date Section 2. How is a chemical change different from a physical change? 3. Give two
Pre-Lab Exercises Lab 3: Chemical Properties 1. How is a chemical property different from a physical property? Name Date Section 2. How is a chemical change different from a physical change? 3. Give two
Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise.
 Experiment 10 Stoichiometry- Gravimetric Analysis Pre-lab Assignment Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise. Purpose The purpose this experiment
Experiment 10 Stoichiometry- Gravimetric Analysis Pre-lab Assignment Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise. Purpose The purpose this experiment
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
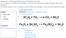 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Alternative Reaction Pathways
 Section 1 Energy and Entropy: Alternative Reaction Pathways What Do You See? Learning Outcomes In this section you will Apply the engineering-design process to scientific and everyday situations. Generate
Section 1 Energy and Entropy: Alternative Reaction Pathways What Do You See? Learning Outcomes In this section you will Apply the engineering-design process to scientific and everyday situations. Generate
Name: Hour: Photosynthesis in Leaf Disks
 Name: Hour: Photosynthesis in Leaf Disks Safety Information: While the solutions may be handled without gloves and may be disposed of in the sink drains, goggles must be worn during the experiment. Background
Name: Hour: Photosynthesis in Leaf Disks Safety Information: While the solutions may be handled without gloves and may be disposed of in the sink drains, goggles must be worn during the experiment. Background
Are Chemical Reactions Closed Systems?
 Measuring a Chemical Reaction 1. Place about 100 ml of vinegar in a large plastic cup. Carefully weigh the cup AND the vinegar all together, and write down the total mass. (Don t worry about subtracting
Measuring a Chemical Reaction 1. Place about 100 ml of vinegar in a large plastic cup. Carefully weigh the cup AND the vinegar all together, and write down the total mass. (Don t worry about subtracting
Physical & Chemical PROPERTIES
 Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
PSI Chemistry. Table spoons, disposable weighing dishes, scale, pencil and instruction hand out.
 Chemistry Honors Lab PSI Chemistry Names Measuring Mass As A Means Of Counting - Chemical quantities- Mole and Particles Purpose To determine the mass of given chemicals and use the data to count atoms.
Chemistry Honors Lab PSI Chemistry Names Measuring Mass As A Means Of Counting - Chemical quantities- Mole and Particles Purpose To determine the mass of given chemicals and use the data to count atoms.
Name Period Date. Lab 1: Mass of Ice Materials: beaker, ice and balance.
 Name Period Date Testing the Law of Conservation of MASS! Introduction: Does mass change in a chemical or physical reaction? In this series of experiments you will find the answer to this question. Lab
Name Period Date Testing the Law of Conservation of MASS! Introduction: Does mass change in a chemical or physical reaction? In this series of experiments you will find the answer to this question. Lab
Unit 2: Chemistry. Unit Overview:
 Unit 2: Chemistry Unit Overview: This unit will focus on basic chemistry and some of the major process of organic chemistry (dehydration synthesis, hydrolysis, and enzyme action) that help form carbon
Unit 2: Chemistry Unit Overview: This unit will focus on basic chemistry and some of the major process of organic chemistry (dehydration synthesis, hydrolysis, and enzyme action) that help form carbon
Ocean Acidification in a Cup Materials
 Ocean Acidification in a Cup Ocean acidification is a problem that humans will have to deal with as we release more and more carbon dioxide into the atmosphere. This activity demonstrates how water can
Ocean Acidification in a Cup Ocean acidification is a problem that humans will have to deal with as we release more and more carbon dioxide into the atmosphere. This activity demonstrates how water can
LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EXPERIMENTS
 LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EPERIMENTS PURPOSE: To practice the scientific method of observation, hypothesis, experimentation, & conclusion. SAFETY CONCERNS: Always wear
LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EPERIMENTS PURPOSE: To practice the scientific method of observation, hypothesis, experimentation, & conclusion. SAFETY CONCERNS: Always wear
More Chemical Changes
 Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Chem 2115 Experiment #7. Volumetric Analysis & Consumer Chemistry Standardization of an unknown solution, analysis of vinegar & antacid tablets
 Chem 2115 Experiment #7 Volumetric Analysis & Consumer Chemistry Standardization of an unknown solution, analysis of vinegar & antacid tablets OBJECTIVE: The goals of this experiment are to learn titration
Chem 2115 Experiment #7 Volumetric Analysis & Consumer Chemistry Standardization of an unknown solution, analysis of vinegar & antacid tablets OBJECTIVE: The goals of this experiment are to learn titration
Titration with an Acid and a Base
 Skills Practice Titration with an Acid and a Base Titration is a process in which you determine the concentration of a solution by measuring what volume of that solution is needed to react completely with
Skills Practice Titration with an Acid and a Base Titration is a process in which you determine the concentration of a solution by measuring what volume of that solution is needed to react completely with
HOW DO PLANTS MEET THEIR NEEDS?
 Overview INSTRUCTIONS In this lesson students will germinate radish seeds and observe the root hairs on the root. Objectives On successful completion of this unit, students will be able to: germinate seeds;
Overview INSTRUCTIONS In this lesson students will germinate radish seeds and observe the root hairs on the root. Objectives On successful completion of this unit, students will be able to: germinate seeds;
Matter [6th grade] Digital Trinity. Trinity University. Anne Cowell Trinity University
![Matter [6th grade] Digital Trinity. Trinity University. Anne Cowell Trinity University Matter [6th grade] Digital Trinity. Trinity University. Anne Cowell Trinity University](/thumbs/78/76800104.jpg) Trinity University Digital Commons @ Trinity Understanding by Design: Complete Collection Understanding by Design 6-14-2006 Matter [6th grade] Anne Cowell Trinity University Follow this and additional
Trinity University Digital Commons @ Trinity Understanding by Design: Complete Collection Understanding by Design 6-14-2006 Matter [6th grade] Anne Cowell Trinity University Follow this and additional
Stoichiometry Decomposition of Baking Soda For our soul is humbled down to the dust. Psalms 43:25
 Stoichiometry Decomposition of Baking Soda For our soul is humbled down to the dust. Psalms 43:25 Introduction A balanced chemical reaction shows products forming reactants such that the Law of Conservation
Stoichiometry Decomposition of Baking Soda For our soul is humbled down to the dust. Psalms 43:25 Introduction A balanced chemical reaction shows products forming reactants such that the Law of Conservation
Section 1: What is a Chemical Reaction
 Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Moles Lab Activity 2: Elements Copper
 Materials Sample of copper Balance Pre-1982 penny Moles Lab Activity 2: Elements Copper Procedure Take the necessary measurements, and record them with units. Show all your calculations, rounding your
Materials Sample of copper Balance Pre-1982 penny Moles Lab Activity 2: Elements Copper Procedure Take the necessary measurements, and record them with units. Show all your calculations, rounding your
Mystery Substance Laboratory Experiment
 Mystery Substance Laboratory Experiment Name: 5 th Grade PSI Science Score: / 5 Experiment Question: How effectively can you determine what a mystery substance is by testing its observable properties?
Mystery Substance Laboratory Experiment Name: 5 th Grade PSI Science Score: / 5 Experiment Question: How effectively can you determine what a mystery substance is by testing its observable properties?
Name: Period: Date: UNIT 1: Introduction to Matter Lesson 8: Baby, you look good
 Name: Period: Date: UNIT 1: Introduction to Matter Lesson 8: Baby, you look good Do Now: By the end of today, you will have an answer to: How do we describe and identify matter? Draw a particle diagram
Name: Period: Date: UNIT 1: Introduction to Matter Lesson 8: Baby, you look good Do Now: By the end of today, you will have an answer to: How do we describe and identify matter? Draw a particle diagram
Chapter Introduction Lesson 1 Understanding Chemical Reactions Lesson 2 Types of Chemical Reactions Lesson 3 Energy Changes and Chemical Reactions
 Chapter Introduction Lesson 1 Understanding Chemical Reactions Lesson 2 Types of Chemical Reactions Lesson 3 Energy Changes and Chemical Reactions Chapter Wrap-Up Changes in Matter A physical change does
Chapter Introduction Lesson 1 Understanding Chemical Reactions Lesson 2 Types of Chemical Reactions Lesson 3 Energy Changes and Chemical Reactions Chapter Wrap-Up Changes in Matter A physical change does
SOIL SHAKE-UP (MODIFIED FOR ADEED)
 SOIL SHAKE-UP (MODIFIED FOR ADEED) Science Concept: Soil is made of many different parts, including small rocks. Objectives: The student will: describe the parts in a sample of soil; observe and communicate
SOIL SHAKE-UP (MODIFIED FOR ADEED) Science Concept: Soil is made of many different parts, including small rocks. Objectives: The student will: describe the parts in a sample of soil; observe and communicate
Lab 2-Investigating the Law of Conservation of Mass
 Name: Period: Lab 2-Investigating the Law of Conservation of Mass Objective: To corroborate the law of conservation of mass through laboratory experimentation. Background: When wood burns or water evaporates,
Name: Period: Lab 2-Investigating the Law of Conservation of Mass Objective: To corroborate the law of conservation of mass through laboratory experimentation. Background: When wood burns or water evaporates,
Chemical Change. Day 1: 70 minutes; Day 2: minutes
 Chemical Change Lesson Concept When one substance interacts with another substance, a chemical change may occur. There are five indicators that a chemical change has occurred: gas production (bubbles),
Chemical Change Lesson Concept When one substance interacts with another substance, a chemical change may occur. There are five indicators that a chemical change has occurred: gas production (bubbles),
LAB: Photosynthesis in Leaf Disks
 Name Date Period LAB: Photosynthesis in Leaf Disks H O N O R S B I O L O G Y : U N I T 3 Introduction: Photosynthesis is a process in which plants convert light energy (sunlight) into usable chemical energy
Name Date Period LAB: Photosynthesis in Leaf Disks H O N O R S B I O L O G Y : U N I T 3 Introduction: Photosynthesis is a process in which plants convert light energy (sunlight) into usable chemical energy
What Do You Think? Investigate GOALS
 Activity 1 Chemical and Physical Changes GOALS In this activity you will: Learn to differentiate between chemical and physical changes. Make observations and cite evidence to identify changes as chemical
Activity 1 Chemical and Physical Changes GOALS In this activity you will: Learn to differentiate between chemical and physical changes. Make observations and cite evidence to identify changes as chemical
LAB TEST Physical and Chemical Changes
 NAME: DATE: STATION: LAB TEST Physical and Chemical Changes PURPOSE: To observe physical and chemical changes in matter MATERIALS: 3 medium test tubes 1 small test tube test tube rack test tube holder
NAME: DATE: STATION: LAB TEST Physical and Chemical Changes PURPOSE: To observe physical and chemical changes in matter MATERIALS: 3 medium test tubes 1 small test tube test tube rack test tube holder
SNC2D CHEMISTRY 3/12/2013. CHEMICAL REACTIONS L Neutralization Reactions (P ) ph & Plants. ph & Plants
 SNC2D CHEMISTRY CHEMICAL REACTIONS L Reactions (P.204-212) ph & Plants Growing plants for a living can be risky. Many factors can affect the success of a crop, from weather conditions to the nutrient content
SNC2D CHEMISTRY CHEMICAL REACTIONS L Reactions (P.204-212) ph & Plants Growing plants for a living can be risky. Many factors can affect the success of a crop, from weather conditions to the nutrient content
Post-Show. Chemistry. Periodic Table of the Elements. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
General Stoichiometry Notes STOICHIOMETRY: tells relative amts of reactants & products in a chemical reaction
 General Stoichiometry Notes STOICHIOMETRY: tells relative amts of reactants & products in a chemical reaction Given an amount of a substance involved in a chemical reaction, we can figure out the amount
General Stoichiometry Notes STOICHIOMETRY: tells relative amts of reactants & products in a chemical reaction Given an amount of a substance involved in a chemical reaction, we can figure out the amount
Safety and Types of Fires
 Cookin' Chem Activity 2 Safety and Types of Fires CHEM POETRY Oliver learned all those Don t s and Do s, The ones that get passed down through moles of O 2 s; Each new generation did soon learn to trust
Cookin' Chem Activity 2 Safety and Types of Fires CHEM POETRY Oliver learned all those Don t s and Do s, The ones that get passed down through moles of O 2 s; Each new generation did soon learn to trust
Lab #9 Stoichiometric Relationships & Theoretical Yield
 Stoichiometric Relationships & Theoretical Yield Name: Nov. 28, 2016 Background and Purpose: Carbonates when reacted with acids will produce carbon dioxide, water and an ionic salt. In this lab, we will
Stoichiometric Relationships & Theoretical Yield Name: Nov. 28, 2016 Background and Purpose: Carbonates when reacted with acids will produce carbon dioxide, water and an ionic salt. In this lab, we will
OBJECTIVES: By the end of class, students will be able to
 7 th Grade Science Unit: Matter and Periodic Table Lesson: Matter 11 Law of Conservation of Mass Review Name: Date: Monday, December 12, 2016 Homeroom: OBJECTIVES: By the end of class, students will be
7 th Grade Science Unit: Matter and Periodic Table Lesson: Matter 11 Law of Conservation of Mass Review Name: Date: Monday, December 12, 2016 Homeroom: OBJECTIVES: By the end of class, students will be
Name Date Class. Chapter 19 Acids, Bases, and Salts EXPERIMENT. ESTIMATION OF ph PURPOSE BACKGROUND MATERIALS (PER PAIR)
 Chapter 19 Acids, Bases, and Salts EXPERIMENT ESTIMATION OF Text Reference Sections 19.2 PURPOSE To estimate the of solutions by using acid base indicators. BACKGROUND If you are interested in gardening,
Chapter 19 Acids, Bases, and Salts EXPERIMENT ESTIMATION OF Text Reference Sections 19.2 PURPOSE To estimate the of solutions by using acid base indicators. BACKGROUND If you are interested in gardening,
8.5E: Chemical Reactions
 Reflect Have you ever seen fireworks explode in the sky? If so, you may have observed a variety of different colors and shapes in the sky and watched with amazement as the colors changed overhead. These
Reflect Have you ever seen fireworks explode in the sky? If so, you may have observed a variety of different colors and shapes in the sky and watched with amazement as the colors changed overhead. These
MONDAY (12/12) TUESDAY (12/13) WEDNESDAY (12/14) THURSDAY (12/15) FRIDAY (12/16) Making Acid Rain (a lab) Quiz
 Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
SNC2D CHEMISTRY 2/24/2013. CHEMICAL REACTIONS L Acids & Bases (P ) Activity: Introduction to (2DCHEM-ASG3) Introduction to Acids & Bases
 SNC2D CHEMISTRY CHEMICAL REACTIONS L Acids & Bases (P.198-201) Activity: Introduction to (2DCHEM-ASG3) INSTRUCTIONS A. Read the activity 2DCHEM - ASG3 (Introduction to Acids & Bases). B. Follow the instructions
SNC2D CHEMISTRY CHEMICAL REACTIONS L Acids & Bases (P.198-201) Activity: Introduction to (2DCHEM-ASG3) INSTRUCTIONS A. Read the activity 2DCHEM - ASG3 (Introduction to Acids & Bases). B. Follow the instructions
Chemistry Foundations of Chemistry Test. This is due:
 Chemistry Foundations of Chemistry Test This is due: Directions: Answer the following questions on a separate sheet of paper (or on this paper if you have room), staple to this paper (if you used a separate
Chemistry Foundations of Chemistry Test This is due: Directions: Answer the following questions on a separate sheet of paper (or on this paper if you have room), staple to this paper (if you used a separate
By All INdICATIONS (2 Hours)
 By All INdICATIONS (2 Hours) Addresses NGSS Level of Difficulty: 5 Grade Range: 6-8 OVERVIEW In this activity, students create an acid-base indicator using red cabbage extract. Students then use this indicator
By All INdICATIONS (2 Hours) Addresses NGSS Level of Difficulty: 5 Grade Range: 6-8 OVERVIEW In this activity, students create an acid-base indicator using red cabbage extract. Students then use this indicator
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
Stoichiometry Lab Report
 Stoichiometry Lab Report Touche G10 Variables Variable Dependent Controlled: Amount of NaHCO3 Theoretical Yield Vinegar Evaporation Manipulation We measure the remaining NaC 2 H 3 O 2 and compare it with
Stoichiometry Lab Report Touche G10 Variables Variable Dependent Controlled: Amount of NaHCO3 Theoretical Yield Vinegar Evaporation Manipulation We measure the remaining NaC 2 H 3 O 2 and compare it with
Classifying Substances
 Classifying Substances There are other ways you can use to classify compounds. For example, you can classify some compounds as acids or bases. You are very familiar with acids and bases because you see
Classifying Substances There are other ways you can use to classify compounds. For example, you can classify some compounds as acids or bases. You are very familiar with acids and bases because you see
Classifying Chemical Reactions Analyzing and Predicting Products
 Classifying Chemical Reactions Analyzing and Predicting Products Background A chemical reaction is defined as any process in which one or more substances are converted into new substances with different
Classifying Chemical Reactions Analyzing and Predicting Products Background A chemical reaction is defined as any process in which one or more substances are converted into new substances with different
The Chemistry of Respiration and Photosynthesis
 The Chemistry of Respiration and Photosynthesis Objective- You should be able to write balanced equations for respiration and photosynthesis and explain how the two equations are related. Directions :
The Chemistry of Respiration and Photosynthesis Objective- You should be able to write balanced equations for respiration and photosynthesis and explain how the two equations are related. Directions :
Recognizing Chemical_Reactions By Dr. Kathleen Vandiver
 Recognizing Chemical_Reactions By Dr. Kathleen Vandiver Hello. My name is Dr. Kathy Vandiver. And we're here at the Edgerton Center at MIT, a university in the United States. And the Edgerton Center is
Recognizing Chemical_Reactions By Dr. Kathleen Vandiver Hello. My name is Dr. Kathy Vandiver. And we're here at the Edgerton Center at MIT, a university in the United States. And the Edgerton Center is
Matter. Anything that has mass and takes up space. Solids Liquids Gases
 Introduction to Matter Matter Anything that has mass and takes up space Solids Liquids Gases Weight A measure of the gravitational force exerted on an object What is the difference between weight and mass?
Introduction to Matter Matter Anything that has mass and takes up space Solids Liquids Gases Weight A measure of the gravitational force exerted on an object What is the difference between weight and mass?
Goal: During this lab students will gain a quantitative understanding of limiting reagents.
 LIMITING REAGENT LAB: THE REACTION BETWEEN VINEGAR AND BAKING SODA Goal: During this lab students will gain a quantitative understanding of limiting reagents. Safety: Safety goggles should be worn at all
LIMITING REAGENT LAB: THE REACTION BETWEEN VINEGAR AND BAKING SODA Goal: During this lab students will gain a quantitative understanding of limiting reagents. Safety: Safety goggles should be worn at all
Lab #5 - Limiting Reagent
 Objective Chesapeake Campus Chemistry 111 Laboratory Lab #5 - Limiting Reagent Use stoichiometry to determine the limiting reactant. Calculate the theoretical yield. Calculate the percent yield of a reaction.
Objective Chesapeake Campus Chemistry 111 Laboratory Lab #5 - Limiting Reagent Use stoichiometry to determine the limiting reactant. Calculate the theoretical yield. Calculate the percent yield of a reaction.
Chapter 6 Classification of Matter
 Chapter 6 Classification of Matter Pure substances can be classified into two types elements cannot be broken down into other pure substances are made up of only one type of atom only 115 known elements
Chapter 6 Classification of Matter Pure substances can be classified into two types elements cannot be broken down into other pure substances are made up of only one type of atom only 115 known elements
Unit 3: Physical Science Classifying Matter in our Daily Lives
 Science 7 Unit 3: Physical Science Classifying Matter in our Daily Lives Name Period Purpose: I understand the relationship between atoms, molecules, elements, compounds, and mixtures and can also provide
Science 7 Unit 3: Physical Science Classifying Matter in our Daily Lives Name Period Purpose: I understand the relationship between atoms, molecules, elements, compounds, and mixtures and can also provide
Lesson Plan Book-stacking Activity
 T o g o d i r e c t l y t o a l e s s o n, c l i c k o n e o f t h e f o l l o w i n g l i n k s : B o o k - s t a c k i n g A c t i v i t y B a l l o o n A c t i v i t y H y d r o g e n G a s L a b F
T o g o d i r e c t l y t o a l e s s o n, c l i c k o n e o f t h e f o l l o w i n g l i n k s : B o o k - s t a c k i n g A c t i v i t y B a l l o o n A c t i v i t y H y d r o g e n G a s L a b F
Objective: SWBAT identify a compound as either ionic, covalent or an acid after reviewing terminology at 85% accuracy.
 Mrs. Chausse s Chemistry Gifted Lesson Plan: Unit 11 Chemical Reactions January 4 18, 2018 Wednesday, January 3, 2018 Naming and writing formulas Objective: SWBAT identify a compound as either ionic, covalent
Mrs. Chausse s Chemistry Gifted Lesson Plan: Unit 11 Chemical Reactions January 4 18, 2018 Wednesday, January 3, 2018 Naming and writing formulas Objective: SWBAT identify a compound as either ionic, covalent
Photosynthesis-Cellular Respiration Cycle
 Photosynthesis-Cellular Respiration Cycle Lesson Concept Link Photosynthesis and cellular respiration are reverse processes. Plants use photosynthesis to make food and release oxygen and plants and animals
Photosynthesis-Cellular Respiration Cycle Lesson Concept Link Photosynthesis and cellular respiration are reverse processes. Plants use photosynthesis to make food and release oxygen and plants and animals
