3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make?
|
|
|
- Christopher Aldous Johns
- 6 years ago
- Views:
Transcription
1 Chapter 4 Test Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make? packs
2 Work Space: 5. A kennel is moving 160 dogs to a new facility. Each dog has its own crate. Each truck holds 9 dogs in their crates. Part A Write a division problem that can be used to find the number of trucks needed to carry the dogs in their crates. Then solve. = r trucks Part B What does the remainder mean in the context of this problem? Teacher use only: Part C The facility manager rents 17 trucks. How can you use your answer to determine if the facility manager rented enough trucks? Explain. Teacher use only:
3 6. Which quotients are equal to 600? Mark Yes or No to tell if the quotient is equal to , , , ,000 3 Work Space: 7. Amanda and her four sisters divided 1,021 stickers equally. About how many stickers did each girl receive? stickers (Solve)
4 8. Part B 1 week = 7 days crickets
5 9. Use partial quotients to solve. 10. A. Teacher use only: B. Write a division problem to show how many are in each group. = blocks
6 11. Use Teacher use only: = ( ) + ( ) = ( ) = 12. Chad bought 8 dozen note pads for his office. The note pads were divided equally into 6 boxes. How many notepads are in each box? 1 dozen= 12 notepads
7
8 Chapter 5 Test 2.
9
10 7. Teacher use only:
11
12 14. Teacher use only: Teacher use only: Teacher use only:
13
14 Student directions for Learning Performance 1 Station #1 Iodine on a Potato Cut a SMALL fresh slice from the potato. Use a dropper to put 3-4 drops of iodine solution on the potato slice. After your observations are complete, throw away the used slice of potato into the trash can! Station #2 Baggie Experiment Place one small spoonful of calcium chloride into a plastic sealable bag and the same amount of BAKING SODA. Seal the bag, shake it. Observe. Measure 2 small teaspoons (about 10 ml) of indicator solution into the small cup (or beaker). Carefully put the small cup into the bag. Seal it. GENTLY tilt the bag to tip the small cup over. Observe. Hold the baggie in the palm of your hand while making your observations. Clean the small cup with water and throw the baggie away. Station #3 Coloring Paper Take a square of paper and choose a crayon. Color the square on your paper with your crayon. Station #4 Two Clear (or mostly) Liquids Take full dropper of the vinegar solution and squirt its contents into a 50 ml beaker. Take a full dropper of the ammonia and add it slowly, a drop at a time, into the 50 ml beaker. Make sure you don t mix up the droppers!! When your observations are completed, pour the contents down the drain and clean the beaker with water. Station #5 Water Baths Fill a test tube ¼ or less with tap water. Place it in the salt water ice bath and stir it around gently and constantly for 2-3 minutes (possibly more) without removing it. Take the test tube out. Record your observations. Rinse the inside AND outside of the test tube well with water and return to the test tube rack.
15 Unit 2 Assessment Chemical Reactions Learning Performance A: Students will interpret data to determine if a change is physical or chemical. Station # Physical/Chemical Evidence (List ALL signs of a physical or chemical change) 1. 1 Physical change 2. Chemical change 3. 2 Physical change 4. Chemical change 5. 3 Physical change 6. Chemical change 7. 4 Physical change 8. Chemical change 9. 5 Physical change 10. Chemical change
16 Learning Performance B: Students will use information to describe that synthetic materials come from natural resources and impact society. 11. The table provides information on three alternative fuels. Alternative Fuel Source Amount of time to produce Average Price Physical State Ethanol Corn 3-5 days $3.04/ gal Liquid Hydrogen Water 1-3 hours $1.50/ gal Gas Biodiesel Vegetable Oil 6-8 hours $4.02/ gal Liquid The data in this table can be used as evidence to support the claim that all of these fuels- Are very costly to produce Exist in the same state of matter Are made of natural materials 12. Information on the raw materials required to produce four synthetic materials is provided. Synthetic Material Produced Nylon fibers Glass Paper Ceramic Raw Material(s) Required Petroleum Silica (sand) Wood Clay The production of which of the listed materials would require growing plants? Nylon fibers Glass Paper
17 Learning Performance C: Students will evaluate if a model supports the law of conservation of mass. 13. Which of the following is true regarding the atoms involved in a chemical reaction? The same number of each type of atom will always be present before and after a chemical reaction takes place. Some of the atoms present before the reaction will always be lost during a chemical reaction. Some of the atoms will always be changed into a different type of atom by a chemical reaction. 14. Students want to gather evidence for the claim that the number of atoms present before a chemical reaction is equal to the number of atoms present after the chemical reaction. They decide to react vinegar and baking soda in a sealed plastic bag. Which of the following would provide the evidence the students need? The mass of the plastic bag, baking soda and vinegar before the reaction was equal to the mass after the reaction. Bubble were produced during the reaction which meant that a gas was being produced. The mass of the baking soda was exactly equal to the mass of the vinegar used to create the chemical reaction.
18 15. Which of the following atomic models of chemical reactions correctly demonstrates the Law of Conservation of Matter?
19 16. For science homework, a student was responsible for creating a model that accurately represented the Law of Conservation of Matter. The model below depicts this student s attempt at creating this model by using the chemical reaction that creates water. Student s homework: Write a scientific explanation that describes why the model pictured above does NOT follow the Law of Conservation of Matter. Use the following words in your response: reactant, product, Law of Conservation of Matter, hydrogen, oxygen
20 Learning Performance D: Students will evaluate data to determine if different substances release or absorb thermal energy by chemical processes. Students performed the following experiments where they combined various chemicals in plastic bags. Their results are listed in the table below. Experiment Number Reactants Starting Temperature Ending Temperature 1 Hydrogen Peroxide and Yeast 22 degrees Celsius 42 degrees Celsius 2 Baking Soda and Vinegar 22 degrees Celsius 18 degrees Celsius 3 Iodine and Liquid Starch 22 degrees Celsius 22 degrees Celsius 17. Which of the above chemical reactions shows an endothermic reaction? Hydrogen Peroxide and Yeast Baking Soda and Vinegar Iodine and Liquid Starch 18. How do you know the above reaction was endothermic? The temperature increases The temperature decreases The temperature stays the same 19. Which of the above chemical reactions shows an exothermic reaction? Hydrogen Peroxide and Yeast Baking Soda and Vinegar Iodine and Liquid Starch 20. How do you know the above reaction was exothermic? The temperature increases The temperature decreases The temperature stays the same
Physical & Chemical PROPERTIES
 Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Chemical Reaction Lab Bagged Chemical Reactions
 Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EXPERIMENTS
 LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EPERIMENTS PURPOSE: To practice the scientific method of observation, hypothesis, experimentation, & conclusion. SAFETY CONCERNS: Always wear
LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EPERIMENTS PURPOSE: To practice the scientific method of observation, hypothesis, experimentation, & conclusion. SAFETY CONCERNS: Always wear
Student Notes. Chemical Reactions LINK
 LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
Activity Sheet Transferring thermal energy by dissolving salts
 Student Name: Date: Activity Sheet Transferring thermal energy by dissolving salts 1) Define Thermal energy and temperature in the boxes below. Thermal Energy Temperature Practice Experiment: Aim: To practice
Student Name: Date: Activity Sheet Transferring thermal energy by dissolving salts 1) Define Thermal energy and temperature in the boxes below. Thermal Energy Temperature Practice Experiment: Aim: To practice
Today is: Monday, October 17th
 Today is: Monday, October 17th Get out your homework! 10/17/2016 #motivationmonday (This is Week 11 Warm Ups!) 1. It s a new quarter, which means a fresh start! What can you do to stay motivated this quarter?
Today is: Monday, October 17th Get out your homework! 10/17/2016 #motivationmonday (This is Week 11 Warm Ups!) 1. It s a new quarter, which means a fresh start! What can you do to stay motivated this quarter?
Are Chemical Reactions Closed Systems?
 Measuring a Chemical Reaction 1. Place about 100 ml of vinegar in a large plastic cup. Carefully weigh the cup AND the vinegar all together, and write down the total mass. (Don t worry about subtracting
Measuring a Chemical Reaction 1. Place about 100 ml of vinegar in a large plastic cup. Carefully weigh the cup AND the vinegar all together, and write down the total mass. (Don t worry about subtracting
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions
 Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
MiSP CHEMICAL REACTIONS, L3 Teacher Guide. Introduction
 MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
Unit 6M.3: Changing materials
 Unit 6M.3: Adding materials to water. Chemical reactions Skill you will use: Classifying Observing Predicting By the end of this unit you should: Know that when substances are added to water, some will
Unit 6M.3: Adding materials to water. Chemical reactions Skill you will use: Classifying Observing Predicting By the end of this unit you should: Know that when substances are added to water, some will
Chapter 6, Lesson 1: What is a Chemical Reaction?
 Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Sulfuric acid is hazardous: Safety glasses are REQUIRED during this experiment.
 CHEMICAL COMPOSITION Life exists on Earth because of the abundant presence of liquid water. While other planets have water, it may be primarily found as either a gas, as on Venus, or as a solid, such as
CHEMICAL COMPOSITION Life exists on Earth because of the abundant presence of liquid water. While other planets have water, it may be primarily found as either a gas, as on Venus, or as a solid, such as
Student Exploration: Chemical Changes
 Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
Lesson 2. Color change
 Lesson 2 Color change T E A C H E R G U I D E Lesson summary Students meet marine chemist Sera Tuikabe, who is studying ocean acidification in the water surrounding the Republic of the Fiji Islands. Students
Lesson 2 Color change T E A C H E R G U I D E Lesson summary Students meet marine chemist Sera Tuikabe, who is studying ocean acidification in the water surrounding the Republic of the Fiji Islands. Students
Physical and Chemical Properties and Changes Lab
 Name: Date: Period: Group Members Physical and Chemical Properties and Changes Lab Station 1 Color Station Instruction: Describe the color of the following substances. Substance Color 1. Sulfur 2. Ammonium
Name: Date: Period: Group Members Physical and Chemical Properties and Changes Lab Station 1 Color Station Instruction: Describe the color of the following substances. Substance Color 1. Sulfur 2. Ammonium
Name. Pre-Test. Directions: Circle the T if a statement is true or F if it is false (F). 1. Matter always stays the same.
 1 Pre-est Directions: Circle the if a statement is true or if it is false (). 1. Matter always stays the same. 2. A physical change is a change in size, shape or phase of matter, without the matter changing
1 Pre-est Directions: Circle the if a statement is true or if it is false (). 1. Matter always stays the same. 2. A physical change is a change in size, shape or phase of matter, without the matter changing
Observations. Direct Observations Those observations we make ourselves
 Observations Direct Observations Those observations we make ourselves Indirect Observations Those observations that we figure out from direct observations Observing Milk Consumption The cafeteria is considering
Observations Direct Observations Those observations we make ourselves Indirect Observations Those observations that we figure out from direct observations Observing Milk Consumption The cafeteria is considering
MONDAY (12/12) TUESDAY (12/13) WEDNESDAY (12/14) THURSDAY (12/15) FRIDAY (12/16) Making Acid Rain (a lab) Quiz
 Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Chemical Reactions. Teachers Guide.
 Chemical Reactions. Teachers Guide. Introduction: Almost everything you will ever see in this universe is made of atoms. Each different type of atom is known as a chemical element there are currently 118
Chemical Reactions. Teachers Guide. Introduction: Almost everything you will ever see in this universe is made of atoms. Each different type of atom is known as a chemical element there are currently 118
Exploring Acids & Bases
 Food Explorations Lab: Exploring Acids & Bases STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will use cabbage juice indicator to determine if two unknown samples are acids or
Food Explorations Lab: Exploring Acids & Bases STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will use cabbage juice indicator to determine if two unknown samples are acids or
Station 1: Ionic versus Covalent Bonds / Electrolytes
 Station 1: Ionic versus Covalent Bonds / Electrolytes The salt and sugar on your kitchen table are both white crystals that dissolve easily in water, but the solutions they form have an important difference.
Station 1: Ionic versus Covalent Bonds / Electrolytes The salt and sugar on your kitchen table are both white crystals that dissolve easily in water, but the solutions they form have an important difference.
What Do You Think? Investigate GOALS
 Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Bags of Reactions Chemistry I Acc
 Introduction: Bags of Reactions Chemistry I Acc Plop, plop, fizz, fizz, oh, what a relief it is. claims the old TV ad for a popular antacid. Just what is in the tablet that is relieving the upset stomach?
Introduction: Bags of Reactions Chemistry I Acc Plop, plop, fizz, fizz, oh, what a relief it is. claims the old TV ad for a popular antacid. Just what is in the tablet that is relieving the upset stomach?
Name Period Date. Lab 1: Mass of Ice Materials: beaker, ice and balance.
 Name Period Date Testing the Law of Conservation of MASS! Introduction: Does mass change in a chemical or physical reaction? In this series of experiments you will find the answer to this question. Lab
Name Period Date Testing the Law of Conservation of MASS! Introduction: Does mass change in a chemical or physical reaction? In this series of experiments you will find the answer to this question. Lab
Experiment 17 It s A Gas and More!
 Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
Experiment 8 - Chemical Changes
 Experiment 8 - Chemical Changes When a chemical change occurs, the chemicals that you start with are changed into different chemicals. We know when this happens because the new chemicals have different
Experiment 8 - Chemical Changes When a chemical change occurs, the chemicals that you start with are changed into different chemicals. We know when this happens because the new chemicals have different
Physical and Chemical Properties of Matter Lab
 Physical and Chemical Properties of Matter Lab Purpose To introduce the student to physical and chemical properties of matter and their use for the identification and separation of compounds. Each student
Physical and Chemical Properties of Matter Lab Purpose To introduce the student to physical and chemical properties of matter and their use for the identification and separation of compounds. Each student
Photosynthesis in Leaf Disks Teacher Preparation and Background Information
 AP Biology Name: Date: Photosynthesis in Leaf Disks Teacher Preparation and Background Information General Information: Solutions may be handled without gloves and may be disposed of in sink drains. In
AP Biology Name: Date: Photosynthesis in Leaf Disks Teacher Preparation and Background Information General Information: Solutions may be handled without gloves and may be disposed of in sink drains. In
Physical and Chemical Changes Or How Do You Know When You ve Made Something New?
 Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Lesson 4. Temperature change
 Lesson 4 Temperature change T E A C H E R G U I D E Lesson summary Students meet scientist Jason Williams, an industrial chemist who designs the materials and processes for making solar cells. He explains
Lesson 4 Temperature change T E A C H E R G U I D E Lesson summary Students meet scientist Jason Williams, an industrial chemist who designs the materials and processes for making solar cells. He explains
Matter Lesson 2. Learning Goal 3: I can describe the differences between physical and chemical changes of matter.
 Matter Lesson 2 Learning Goal 2: I can describe the differences between intensive physical properties, extensive physical properties, and chemical properties of matter. Learning Goal 3: I can describe
Matter Lesson 2 Learning Goal 2: I can describe the differences between intensive physical properties, extensive physical properties, and chemical properties of matter. Learning Goal 3: I can describe
More Chemical Changes
 Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Chemical Energy Conversions. Vanderbilt Student Volunteers for Science VINSE/VSVS Rural
 Chemical Energy Conversions Vanderbilt Student Volunteers for Science 2018-2019 VINSE/VSVS Rural Important!!! Please use this resource to reinforce your understanding of the lesson! Make sure you have
Chemical Energy Conversions Vanderbilt Student Volunteers for Science 2018-2019 VINSE/VSVS Rural Important!!! Please use this resource to reinforce your understanding of the lesson! Make sure you have
Chemical Change. Day 1: 70 minutes; Day 2: minutes
 Chemical Change Lesson Concept When one substance interacts with another substance, a chemical change may occur. There are five indicators that a chemical change has occurred: gas production (bubbles),
Chemical Change Lesson Concept When one substance interacts with another substance, a chemical change may occur. There are five indicators that a chemical change has occurred: gas production (bubbles),
Liquid X Lab. Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties?
 Names Block Date Liquid X Lab Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette to carefully place drops of each liquid
Names Block Date Liquid X Lab Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette to carefully place drops of each liquid
Characteristics of Chemical Change
 Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
5 th GRADE PHYSICAL AND CHEMICAL TESTS
 5 th GRADE PHYSICAL AND CHEMICAL TESTS Summary: Students investigate 5 unknown white powders. They gather clues by observing the physical and chemical changes of the powders. At the end of the activity,
5 th GRADE PHYSICAL AND CHEMICAL TESTS Summary: Students investigate 5 unknown white powders. They gather clues by observing the physical and chemical changes of the powders. At the end of the activity,
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version
 Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Pre-Lab Exercises Lab 3: Chemical Properties
 Pre-Lab Exercises Lab 3: Chemical Properties 1. How is a chemical property different from a physical property? Name Date Section 2. How is a chemical change different from a physical change? 3. Give two
Pre-Lab Exercises Lab 3: Chemical Properties 1. How is a chemical property different from a physical property? Name Date Section 2. How is a chemical change different from a physical change? 3. Give two
Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic
 Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic Name Date DEMONSTRATION 1. Your teacher blew through a straw into a universal indicator solution until it changed color. Did
Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic Name Date DEMONSTRATION 1. Your teacher blew through a straw into a universal indicator solution until it changed color. Did
LAB: Photosynthesis in Leaf Disks
 Name Date Period LAB: Photosynthesis in Leaf Disks H O N O R S B I O L O G Y : U N I T 3 Introduction: Photosynthesis is a process in which plants convert light energy (sunlight) into usable chemical energy
Name Date Period LAB: Photosynthesis in Leaf Disks H O N O R S B I O L O G Y : U N I T 3 Introduction: Photosynthesis is a process in which plants convert light energy (sunlight) into usable chemical energy
Photosynthesis Lab. (adapted from: Photosynthesis.pdf )
 Photosynthesis Lab (adapted from: http://media.collegeboard.com/digitalservices/pdf/ap/bio-manual/bio_lab5- Photosynthesis.pdf ) Background and PreLab: Photosynthesis fuels ecosystems and replenishes the
Photosynthesis Lab (adapted from: http://media.collegeboard.com/digitalservices/pdf/ap/bio-manual/bio_lab5- Photosynthesis.pdf ) Background and PreLab: Photosynthesis fuels ecosystems and replenishes the
Liquid X Lab. Number of Drops Before Spilling Trial 1 Trial 2 Trial 3. Write a conclusion: How do your results for Liquid X compare to water?
 Names Block Date BIG QUESTION: Liquid X Lab Station 1 Surface Tension, Cohesion, and Adhesion Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette
Names Block Date BIG QUESTION: Liquid X Lab Station 1 Surface Tension, Cohesion, and Adhesion Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette
Bubbles and Colors and Smells Oh My! A lesson about physical and chemical changes By Glenda Kitts, Arlene Cuellar, and Maritza Aquino
 Glenda Kitts, Arlene Cuellar, Maritza Aquino Last Updated on 9/17/2017 Bubbles and Colors and Smells Oh My! A lesson about physical and chemical changes By Glenda Kitts, Arlene Cuellar, and Maritza Aquino
Glenda Kitts, Arlene Cuellar, Maritza Aquino Last Updated on 9/17/2017 Bubbles and Colors and Smells Oh My! A lesson about physical and chemical changes By Glenda Kitts, Arlene Cuellar, and Maritza Aquino
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
Lab #3 ph and Buffers
 Page1 Lab #3 ph and Objectives: Learn to construct a proper data table and line graph Understand how the ph scale works Use a ph meter to measure the ph of common household substances Understand the meaning
Page1 Lab #3 ph and Objectives: Learn to construct a proper data table and line graph Understand how the ph scale works Use a ph meter to measure the ph of common household substances Understand the meaning
Lab 16: Metals and Oxidation
 Concepts to explore: Observe an oxidation reduction reaction Use the properties of a reaction product to verify its identity Rank the reactivity of certain metals in a weak acid, and compare it to their
Concepts to explore: Observe an oxidation reduction reaction Use the properties of a reaction product to verify its identity Rank the reactivity of certain metals in a weak acid, and compare it to their
Describing Matter Laboratory
 Describing Matter Laboratory Name: 5 th Grade PSI Science Score: / 5 Experiment Question: How is matter identified? What are the observable properties of matter? Hypothesis Starters: 1. Your eyes help
Describing Matter Laboratory Name: 5 th Grade PSI Science Score: / 5 Experiment Question: How is matter identified? What are the observable properties of matter? Hypothesis Starters: 1. Your eyes help
Copyright 2016 Edmentum - All rights reserved.
 Copyright 2016 Edmentum - All rights reserved. SI: Quiz 5 Question #1 Which of the following is true about the mass of the reactants in a chemical reaction? The total mass of the reactants in a chemical
Copyright 2016 Edmentum - All rights reserved. SI: Quiz 5 Question #1 Which of the following is true about the mass of the reactants in a chemical reaction? The total mass of the reactants in a chemical
Maintaining Mass Created By: Allyn Short
 Maintaining Mass Created By: Allyn Short Focus on Inquiry The student will demonstrate that mass is conserved when substances undergo chemical and/or physical changes through experimentation and evaluation
Maintaining Mass Created By: Allyn Short Focus on Inquiry The student will demonstrate that mass is conserved when substances undergo chemical and/or physical changes through experimentation and evaluation
Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds.
 Chapter 11 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Chapter 11 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Moles Lab Activity 2: Elements Copper
 Materials Sample of copper Balance Pre-1982 penny Moles Lab Activity 2: Elements Copper Procedure Take the necessary measurements, and record them with units. Show all your calculations, rounding your
Materials Sample of copper Balance Pre-1982 penny Moles Lab Activity 2: Elements Copper Procedure Take the necessary measurements, and record them with units. Show all your calculations, rounding your
Today is: Friday, October 21st
 Today is: Friday, October 21st 10/21/2016 #followyourdreamsfriday 1. What are some of your biggest goals in life? 2. Which of the following is an example of a chemical change? Why? Justify your answer.
Today is: Friday, October 21st 10/21/2016 #followyourdreamsfriday 1. What are some of your biggest goals in life? 2. Which of the following is an example of a chemical change? Why? Justify your answer.
Signs of Chemical Reactions
 Signs of Chemical Reactions Tell the difference between changes which are PHYSICAL and those which involve a CHEMICAL reaction Doing six different experiments and observing the changes which
Signs of Chemical Reactions Tell the difference between changes which are PHYSICAL and those which involve a CHEMICAL reaction Doing six different experiments and observing the changes which
Reactions that Produce Heat
 Chemical Dominoes Activity 7 Reactions that Produce Heat GOALS In this activity you will: Explain the effect of particle size and use of a catalyst on reaction rate. Represent energy changes graphically.
Chemical Dominoes Activity 7 Reactions that Produce Heat GOALS In this activity you will: Explain the effect of particle size and use of a catalyst on reaction rate. Represent energy changes graphically.
Lyniece McKim Biology Instructor Star Valley High Afton, WY.
 Lyniece McKim Biology Instructor Star Valley High Afton, WY lmckm@lcsd2.org TEACHER SHARE-A-THON NABT 2010 Biology: Chemistry of Life Lab: Testing Mystery Substances Inquiry activity Using indicators;
Lyniece McKim Biology Instructor Star Valley High Afton, WY lmckm@lcsd2.org TEACHER SHARE-A-THON NABT 2010 Biology: Chemistry of Life Lab: Testing Mystery Substances Inquiry activity Using indicators;
Grade 8 Science Unit 2 Test» Form A (Master Copy) Directions: Please choose the best answer choice for each of the following questions.
 Directions: Please choose the best answer choice for each of the following questions. 1. Which of the following could be a molecule BUT not a compound? a pair of atoms of the same element a collection
Directions: Please choose the best answer choice for each of the following questions. 1. Which of the following could be a molecule BUT not a compound? a pair of atoms of the same element a collection
Signs of Chemical Reactions
 Signs of Chemical Reactions Tell the difference between changes which are PHYSICAL and those which involve a CHEMICAL reaction Doing six different experiments and observing the changes which
Signs of Chemical Reactions Tell the difference between changes which are PHYSICAL and those which involve a CHEMICAL reaction Doing six different experiments and observing the changes which
General Chemistry I CHEM-1030 Laboratory Experiment No. 2 Physical Separation Techniques
 General Chemistry I CHEM-1030 Laboratory Experiment No. 2 Physical Separation Techniques Introduction When two or more substances that do not react chemically are blended together, the components of the
General Chemistry I CHEM-1030 Laboratory Experiment No. 2 Physical Separation Techniques Introduction When two or more substances that do not react chemically are blended together, the components of the
Conservation of Mass in Chemical Reactions Lab. Experiment Question: What happens to the total mass of substances when a chemical reaction occurs?
 Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from
 Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
CO 2. Lesson 1. Production of a gas
 Lesson 1 Production of a gas T E A C H E R G U I D E CO 2 Lesson summary Students meet volcanologist Victor Helguson, who is studying the gases released by volcanoes in Iceland. Students conduct a chemical
Lesson 1 Production of a gas T E A C H E R G U I D E CO 2 Lesson summary Students meet volcanologist Victor Helguson, who is studying the gases released by volcanoes in Iceland. Students conduct a chemical
Equilibrium/ Le Chatelier s Principle
 Equilibrium/ Le Chatelier s Principle Modified from an activity originally created by participating teachers 2008 Summer Green Chemistry workshop View video as demonstration guide: https://www.youtube.com/watch?v=fbdyl3hlbui#t=379
Equilibrium/ Le Chatelier s Principle Modified from an activity originally created by participating teachers 2008 Summer Green Chemistry workshop View video as demonstration guide: https://www.youtube.com/watch?v=fbdyl3hlbui#t=379
Chemical Reactions. Chapter Test A. Multiple Choice. 1 Pearson Education, Inc., or its affiliates. All rights reserved.
 Name Date Class Chemical Reactions Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. 1. Chemistry is a. a characteristic of a substance that can be observed
Name Date Class Chemical Reactions Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. 1. Chemistry is a. a characteristic of a substance that can be observed
Modeling Conservation of Matter
 Modeling Conservation of Matter Imagine that you and two of your classmates want to make a strawberry banana smoothie. You lay out the ingredients: one banana, five strawberries and two scoops of ice cream.
Modeling Conservation of Matter Imagine that you and two of your classmates want to make a strawberry banana smoothie. You lay out the ingredients: one banana, five strawberries and two scoops of ice cream.
Post-Show. Chemistry. Periodic Table of the Elements. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Science Supply List. General Science II Glynlyon, Inc.
 Science Supply List General Science II 2016 Glynlyon, Inc. Table of Contents UNIT 1: SCIENCE AND SOCIETY... 1 UNIT 2: STRUCTURE OF MATTER (PART 1)... 1 UNIT 3: STRUCTURE OF MATTER (PART 2)... 2 UNIT 4:
Science Supply List General Science II 2016 Glynlyon, Inc. Table of Contents UNIT 1: SCIENCE AND SOCIETY... 1 UNIT 2: STRUCTURE OF MATTER (PART 1)... 1 UNIT 3: STRUCTURE OF MATTER (PART 2)... 2 UNIT 4:
General Science II General Science II
 2017-2018 General Science II General Science II 2017 Glynlyon, Inc. Table of Contents UNIT 1: SCIENCE AND SOCIETY... 1 UNIT 2: STRUCTURE OF MATTER (PART 1)... 1 UNIT 3: STRUCTURE OF MATTER (PART 2)...
2017-2018 General Science II General Science II 2017 Glynlyon, Inc. Table of Contents UNIT 1: SCIENCE AND SOCIETY... 1 UNIT 2: STRUCTURE OF MATTER (PART 1)... 1 UNIT 3: STRUCTURE OF MATTER (PART 2)...
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from
 Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
Saturday Science Lesson Plan Fall 2008
 Saturday Science Lesson Plan Fall 2008 LEARNING OBJECTIVES STANDARDS 1.1.1 Observe, describe, draw, and sort objects carefully to learn about them. 1.2.6 Describe and compare objects in terms of number,
Saturday Science Lesson Plan Fall 2008 LEARNING OBJECTIVES STANDARDS 1.1.1 Observe, describe, draw, and sort objects carefully to learn about them. 1.2.6 Describe and compare objects in terms of number,
Unit 2- Chemistry: Matter/Periodic Table/Acids & Bases
 Unit 2 Chemistry LESSON OUTLINE: I. Matter a. Phases of Matter b. Conservation of Matter c. Physical and Chemical Change d. Phase Change and Energy e. Gas Laws II. III. IV. Elements and the Periodic Table
Unit 2 Chemistry LESSON OUTLINE: I. Matter a. Phases of Matter b. Conservation of Matter c. Physical and Chemical Change d. Phase Change and Energy e. Gas Laws II. III. IV. Elements and the Periodic Table
Period 11: Chemical Energy and Fossil Fuels
 Name Section Period 11: Chemical Energy and Fossil Fuels 11.1 What is the Composition of Matter? 1) Atoms a) What are the building blocks of an atom? b) Which nucleons make up an atomic nucleus? c) What
Name Section Period 11: Chemical Energy and Fossil Fuels 11.1 What is the Composition of Matter? 1) Atoms a) What are the building blocks of an atom? b) Which nucleons make up an atomic nucleus? c) What
Final Review Graphs and Charts TWO Page 1 of 35
 TEST NAME:Final Review Graphs and Charts TWO TEST ID:1086907 GRADE:11 Eleventh Grade 12 Twelfth Grade SUBJECT:Life and Physical Sciences TEST CATEGORY:School Assessment Final Review Graphs and Charts TWO
TEST NAME:Final Review Graphs and Charts TWO TEST ID:1086907 GRADE:11 Eleventh Grade 12 Twelfth Grade SUBJECT:Life and Physical Sciences TEST CATEGORY:School Assessment Final Review Graphs and Charts TWO
Po Kok Secondary School S.1 Integrated Science Chapter 1.1 Classwork What is Science? Class: S.1 ( ) Date: A. What is Science? P.
 Po Kok Secondary School S.1 Integrated Science Chapter 1.1 Classwork Name: What is Science? Class: S.1 ( ) Date: A. What is Science? P.3-4 The study of things and phenomena in nature and how they affect
Po Kok Secondary School S.1 Integrated Science Chapter 1.1 Classwork Name: What is Science? Class: S.1 ( ) Date: A. What is Science? P.3-4 The study of things and phenomena in nature and how they affect
Energy Transformations Activities Students engage in different activities to observe energy changing form!
 Energy Transformations Activities Students engage in different activities to observe energy changing form! Name: Energy Labs and Activities Activity 1: Static Electricity Glue this side down Procedure:
Energy Transformations Activities Students engage in different activities to observe energy changing form! Name: Energy Labs and Activities Activity 1: Static Electricity Glue this side down Procedure:
Stoichiometry World of Chemistry: Chapter 9
 Stoichiometry World of Chemistry: Chapter 9 Chocolate Chip Cookies!! 1 cup butter 1/2 cup white sugar 1 cup packed brown sugar 1 teaspoon vanilla extract 2 eggs 2 1/2 cups all-purpose flour 1 teaspoon
Stoichiometry World of Chemistry: Chapter 9 Chocolate Chip Cookies!! 1 cup butter 1/2 cup white sugar 1 cup packed brown sugar 1 teaspoon vanilla extract 2 eggs 2 1/2 cups all-purpose flour 1 teaspoon
#2: THE FLOATING PAPER CLIP
 Activity #1: PILE IT ON. Materials: 1 DRY penny, 1 eye dropper, water. Procedure: Make sure the penny is dry. Begin by estimating the number of drops of water that can be piled on the penny before it spills
Activity #1: PILE IT ON. Materials: 1 DRY penny, 1 eye dropper, water. Procedure: Make sure the penny is dry. Begin by estimating the number of drops of water that can be piled on the penny before it spills
C q T q C T. Heat is absorbed by the system H > 0 endothermic Heat is released by the system H < 0 exothermic
 PLEASE REORD ALL DATA DIRETLY INTO YOUR LAB NOTEBOOKS Introduction Heating a substance is one of the simplest processes carried out in the chemical laboratory, and is usually accompanied by a rise in the
PLEASE REORD ALL DATA DIRETLY INTO YOUR LAB NOTEBOOKS Introduction Heating a substance is one of the simplest processes carried out in the chemical laboratory, and is usually accompanied by a rise in the
To measure ph s in a variety of solutions and mixtures and to account for the results obtained.
 Acid-Base Studies PURPOSE To measure ph s in a variety of solutions and mixtures and to account for the results obtained. GOALS 1 To learn to use ph paper and a ph meter to measure the ph of a given solution.
Acid-Base Studies PURPOSE To measure ph s in a variety of solutions and mixtures and to account for the results obtained. GOALS 1 To learn to use ph paper and a ph meter to measure the ph of a given solution.
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic
 Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Name: 1. Which of the following is probably true about 300 ml of sand and 300 ml of water?
 Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
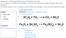 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Chapter 6, Lesson 9: Neutralizing Acids and Bases
 Chapter 6, Lesson 9: Neutralizing Acids and Bases Key Concepts ph is a measure of the concentration of H 3 O + ions in a solution. Adding an acid increases the concentration of H 3 O + ions in the solution.
Chapter 6, Lesson 9: Neutralizing Acids and Bases Key Concepts ph is a measure of the concentration of H 3 O + ions in a solution. Adding an acid increases the concentration of H 3 O + ions in the solution.
LESSON 1. Chemical Reactions. Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions.
 LESSON 1 Chemical Reactions Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions. By the end of this lesson... you will be able to explain ways
LESSON 1 Chemical Reactions Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions. By the end of this lesson... you will be able to explain ways
4. Every CHANGE in matter includes a change in, which is conserved in a chemical reaction and. TRANSFORMED from one form to another.
 Part C: Lesson 1.4 - Read pages 20-29 to answer these questions. Add to notes - 1. A physical change alters the form or appearance of matter, such as Change in size, a change in SIZE or. SHAPE shape, or
Part C: Lesson 1.4 - Read pages 20-29 to answer these questions. Add to notes - 1. A physical change alters the form or appearance of matter, such as Change in size, a change in SIZE or. SHAPE shape, or
Supernatant: The liquid layer lying above the solid layer after a precipitation reaction occurs.
 Limiting Reagent Introduction The quantities of substances involved in a chemical reaction represented by a balanced equation are often referred to as stoichiometric amounts. Solution stoichiometry is
Limiting Reagent Introduction The quantities of substances involved in a chemical reaction represented by a balanced equation are often referred to as stoichiometric amounts. Solution stoichiometry is
The grade 5 English science unit, Acids and Bases, meets the academic content standards set in the Korean curriculum, which state students should:
 This area addresses ph among different characteristics of solutions. It will be interesting for students to classify a variety of solutions into acids and bases by using the characteristics of the solutions.
This area addresses ph among different characteristics of solutions. It will be interesting for students to classify a variety of solutions into acids and bases by using the characteristics of the solutions.
Cell Membranes and Permeability Laboratory
 Cell Membranes and Permeability Laboratory Do all chemical substances pass in and out of a cell membrane with equal ease? Do chemical substances move from areas of high concentration to areas of low concentration
Cell Membranes and Permeability Laboratory Do all chemical substances pass in and out of a cell membrane with equal ease? Do chemical substances move from areas of high concentration to areas of low concentration
Supply List. General Science II. Released Glynlyon, Inc
 N Supply List General Science II 2012 Glynlyon, Inc Released 4-1-12 Table of Contents UNIT 1: SCIENCE AND SOCIETY... 1 UNIT 2: STRUCTURE OF MATTER (PART 1)... 1 UNIT 3: STRUCTURE OF MATTER (PART 2)...
N Supply List General Science II 2012 Glynlyon, Inc Released 4-1-12 Table of Contents UNIT 1: SCIENCE AND SOCIETY... 1 UNIT 2: STRUCTURE OF MATTER (PART 1)... 1 UNIT 3: STRUCTURE OF MATTER (PART 2)...
Section I: Synthesis reactions Synthesis reactions occur when two or more substances come together to form a single new substance.
 TYPES OF CHEMICAL REACTIONS A Laboratory Investigation Purpose: Observe the five major types of reactions. Record observations for these reactions. Complete balanced equations for these reactions. Introduction:
TYPES OF CHEMICAL REACTIONS A Laboratory Investigation Purpose: Observe the five major types of reactions. Record observations for these reactions. Complete balanced equations for these reactions. Introduction:
o Test tube In this experiment, you ll be observing the signs of chemical reactions. These include the following:
 Experiment: Chemical Reactions & Chemical s Objective In this experiment, students perform a variety of chemical reactions. For each reaction, student identify the signs that a reaction has occurred, write
Experiment: Chemical Reactions & Chemical s Objective In this experiment, students perform a variety of chemical reactions. For each reaction, student identify the signs that a reaction has occurred, write
Unit 3.3 Test: Basic Chemistry
 Unit 3.3 Test: Basic Chemistry 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 1. A student placed 2 grams of baking soda in a balloon and
Unit 3.3 Test: Basic Chemistry 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 1. A student placed 2 grams of baking soda in a balloon and
The masses of reactants and products are equal.
 Page 1 of 7 KEY CNCEPT The masses of reactants and products are equal. BEFRE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed and identified
Page 1 of 7 KEY CNCEPT The masses of reactants and products are equal. BEFRE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed and identified
Pre-Lab Read the entire laboratory assignment. Answer all pre-lab questions before beginning the lab.
 Name: Date: Pd: Lab Partner: Lab # 13: Types of Reactions, Predicting Products of Chemical Reactions Lab Accelerated Chemistry 1 Introduction: If you examine your bicycle after it has been left out in
Name: Date: Pd: Lab Partner: Lab # 13: Types of Reactions, Predicting Products of Chemical Reactions Lab Accelerated Chemistry 1 Introduction: If you examine your bicycle after it has been left out in
5 th Grade Lesson Plan: Matter and Chemical Reactions
 5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
composition of matter, and the changes that matter undergoes. Examples of Uses of Chemistry in Everyday Life
 Name Matter and Change: Unit Objective Study Guide Date Due Directions: Write your answers to the following questions in the space provided. For problem solving, all of the work leading up to the final
Name Matter and Change: Unit Objective Study Guide Date Due Directions: Write your answers to the following questions in the space provided. For problem solving, all of the work leading up to the final
Chemistry. End of Year Cornerstone Assessment
 Chemistry End of Year Cornerstone Assessment The Cornerstone Assessments were developed with support through the VDOE Mathematics and Science Partnership Grant Program NCLB Title II, Part B program by
Chemistry End of Year Cornerstone Assessment The Cornerstone Assessments were developed with support through the VDOE Mathematics and Science Partnership Grant Program NCLB Title II, Part B program by
Supernatant: The liquid layer lying above the solid layer after a precipitation reaction occurs.
 Limiting Reagent Introduction The quantities of substances involved in a chemical reaction represented by a balanced equation are often referred to as stoichiometric amounts. Solution stoichiometry is
Limiting Reagent Introduction The quantities of substances involved in a chemical reaction represented by a balanced equation are often referred to as stoichiometric amounts. Solution stoichiometry is
Chemistry. Baseline Cornerstone Assessment
 Chemistry Baseline Cornerstone Assessment The Cornerstone Assessments were developed with support through the VDOE Mathematics and Science Partnership Grant Program NCLB Title II, Part B program by high
Chemistry Baseline Cornerstone Assessment The Cornerstone Assessments were developed with support through the VDOE Mathematics and Science Partnership Grant Program NCLB Title II, Part B program by high
Observing Chemical Change - 5.1
 Observing Chemical Change - 5.1 Vocabulary: Physical change - Chemical change - Reactant Product Precipitate Exothermic reaction - Endothermic reaction - Properties of matter: Two kinds of properties of
Observing Chemical Change - 5.1 Vocabulary: Physical change - Chemical change - Reactant Product Precipitate Exothermic reaction - Endothermic reaction - Properties of matter: Two kinds of properties of
