Lesson 2. Color change
|
|
|
- Jack Goodwin
- 5 years ago
- Views:
Transcription
1 Lesson 2 Color change T E A C H E R G U I D E Lesson summary Students meet marine chemist Sera Tuikabe, who is studying ocean acidification in the water surrounding the Republic of the Fiji Islands. Students combine different amounts of acids and bases to explore the range of color changes of universal indicator solution. Then students make as many different-colored solutions as they can. Key concepts + A change in color is a clue that a chemical reaction may have occurred. + Universal indicator solution is green when it has a ph around 7. + Acidic solutions have a ph less than 7. + Basic solutions have a ph greater than 7. + Adding a base to an acidic solution makes the solution less acidic. + Adding an acid to a basic solution makes the solution less basic. + Carbon dioxide gas reacts with water to form carbonic acid. Safety Be sure you and the students wear properly fitting goggles. Read and follow all safety warnings on the labels of the citric acid and sodium carbonate containers. Have students wash their hands after the activity. Bromthymol blue and universal indicators are both alcohol-based and flammable. Proper disposal At the end of the lesson, have students pour their used solutions in a waste container. Dispose of this waste down the drain or according to local regulations. The leftover citric acid and sodium carbonate powders can be disposed of with the classroom trash. Wipe up spills with paper towels and dispose of them with the classroom trash. 1
2 Introduction 1. Introduce students to a scientist and her work. Distribute the student activity sheets for Lesson 2. In this lesson, students meet marine chemist Sera Tuikabe. She explains that carbon dioxide gas can dissolve in water and chemically change the water, making it more acidic. The harmful effects of acidic water on marine life are especially noticeable in corals, clams, starfish, and other creatures that have shells. Teacher demonstration 2. Bubble carbon dioxide gas from two different chemical reactions through a color-changing indicator solution. Tell students that you will produce carbon dioxide gas in two different ways. Then you will add the gas to a special solution called bromthymol blue indicator to see if the gas reacts with the solution. If a chemical reaction does occur, this blue solution will change color. A color change is a clue that a chemical reaction may have occurred. You will need + Goggles + Bromthymol blue indicator + Sodium bicarbonate + Vinegar (bring this from home or use from school) + Water + Small metric measuring cup + Small plastic spoon + Plastic disposable water bottle, 8 ounces (collect from a student) + Balloon + 3 clear plastic cups + 2 straws Spoon Preparation instructions + Place 20 ml of water into two clean, unused, clear plastic cups. + Add about drops of bromthymol blue indicator to each cup and swirl so that the solutions are blue. Place a clean straw in each cup. + Put about 30 ml of vinegar into the bottom of a small disposable plastic water bottle. + With the help of an assistant, place about 2 small spoonfuls of sodium bicarbonate into a deflated balloon. 1. Place the balloon over the top of the bottle. 2. Lift the balloon so that the baking soda drops into the vinegar and gently swirl the bottle. 2
3 The balloon will stand up over the top of the bottle. The balloon will not fully inflate, but it will contain enough carbon dioxide gas for the next part of the demonstration. 3. Twist the balloon so that you can remove it from the bottle without allowing the gas to escape. 4. Carefully remove the balloon and place the end of a straw into the opening while pinching the opening so that not too much gas escapes. 5. Place the other end of the straw into one of your cups of bromthymol blue indicator solution and gently squeeze the balloon to bubble the gas into the solution. The solution should change from blue to yellow. Tell students that the carbon dioxide gas reacted with the water in the solution and formed an acid called carbonic acid. This acidic solution reacted with the indicator and caused the color to change. Ask students + Next, I am going to gently blow my own breath into bromthymol blue indicator solution. Do you expect a color change? Why or why not? Students may know that we exhale carbon dioxide gas. Because carbon dioxide produced from the vinegar and baking soda reaction changed the color of the indicator solution, carbon dioxide from another source should do the same. 6. Place a straw in the cup of bromthymol blue indicator solution that you have not yet used. 7. Push the straw to the bottom of the cup and gently blow until the solution turns greenish-yellow. The indicator solution will turn a greenish yellow and may even turn completely yellow after about a minute of blowing through the solution. Remind students that a change in color is a clue that a chemical reaction may have occurred. In a chemical reaction the reactants that we start with change to become different chemicals, or products. In this reaction, carbon dioxide gas reacts with water to make the product carbonic acid, which causes the color change. Tell students that oceanographer Sera Tuikabe uses the color changes from different indicators to help her measure the health of the water surrounding coral reefs. 3
4 Teacher demonstration 3. Do a demonstration to introduce students to their chemistry challenge. Tell students that they will use a type of indicator called universal indicator. This indicator solution starts out green, but changes color as it reacts with different substances. Its color changes can help you tell whether the solution is acidic, neutral, or basic. And if you are familiar with the range of color changes, you can not only tell whether a solution is acidic or not, you can tell how acidic it is. You will need + Goggles + Universal indicator + Citric acid + Sodium carbonate + Water + Universal indicator ph color chart + Small metric measuring cup + 2 small plastic scoops + 3 small clear plastic cups Scoop Preparation instructions + Add about 1 small scoop of citric acid to one clear plastic cup and 1 small scoop of sodium carbonate to another. Tell students that you have added a substance to each cup but do not tell them what is in each cup. + In a clean, clear plastic cup, combine 60 ml water and 2.5 ml universal indicator solution. + Line the three cups up as shown. 1. While holding the cups up so that students can see, slowly add about 1 /3 of the universal indicator to the cup with the citric acid. 2. Then slowly add about the same amount to the cup with the sodium carbonate. Try to leave about one third of the indicator solution in its cup. The citric acid turns the indicator from green to reddish. The sodium carbonate turns the indicator from green to purple. Distribute one universal indicator ph color chart to each group. Ask students + What do you know about the powders in each cup? By looking at their ph color chart, students will know that the cup in which the solution turned reddish contained the acid and the solution that turned purple contained the base. 4
5 3. Tell students to watch as you slowly pour the acidic (pink) universal indicator into the basic (purple) universal indicator solution. The basic solution changes color and goes toward neutral on the ph scale. Tell students that acids and bases are like chemical opposites: If you add an acid to a basic solution, the solution becomes less basic. If you add a base to an acidic solution, the solution becomes less acidic. Student activity 4. Have students make at least 6 different colors by adding drops of acid and base to universal indicator solution. Depending on students experience using droppers, you may need to have students practice using them. Be sure to demonstrate the difference between drops and squirts and, if necessary, give students a chance to practice releasing single drops of water. In this activity, students will add a certain number of drops of each of the required solutions to each well. Activity summary Students will begin by preparing their acid and base solutions and adding universal indicator solution to 12 different wells on a spot plate. Then they will combine drops of citric acid and sodium carbonate (a base) in each well to make as many different colors as they can. This activity is described in detail on the student activity sheet. Preparation instructions + Reuse the portion cup labeled citric acid from the previous activity. Then label one clean, unused, clear plastic cup and one dropper citric acid solution for each group. + Label one portion cup sodium carbonate. Then label one clean, unused, clear plastic cup and one dropper sodium carbonate solution for each group. + Place about ¼ teaspoon of citric acid and sodium carbonate in their labeled portion cups. Each group will make their own citric acid and sodium carbonate solutions in the clear plastic cups. The wells in the first row will be shades of green, blue, and purple. The wells in the second row will be shades of yellow, orange, and pink. And the wells in the third row will be varying shades of the colors in the first and second row. 5
6 Teacher demonstration 5. Do a demonstration to review the two clues of chemical change introduced in the first two lessons. You will need + Goggles + Bromthymol blue indicator + Sodium bicarbonate + Citric acid + Water + Small metric measuring cup + 2 small scoops + 2 clear plastic cups Scoop 1. Place 10 ml of water in a clear plastic cup. 2. Add 1 scoop of sodium bicarbonate (baking soda) and swirl to dissolve. 3. Add 10 drops of bromthymol blue indicator to the baking soda solution. 4. Place 10 ml of water in a separate clear plastic cup. 5. Add 2 scoops of citric acid to the water and swirl to dissolve. Ask students to predict + What do you think will happen if you pour the citric acid solution into the sodium bicarbonate solution? 6. Pour the citric acid solution into the sodium bicarbonate solution. The blue color will turn yellowish and there will be some bubbling. Ask students + What two clues of chemical change did you observe? A color change and production of a gas Application 6 Have students read the real life application on the student activity sheet. Explain that people with diabetes can use a color-changing chemical reaction to help them keep track of the glucose levels in their bodies. A small strip of paper with different chemicals on it reacts with a person s urine; the darker the color, the more glucose in the urine. This amount of glucose is a reflection of the level of glucose in a person s blood.
CO 2. Lesson 1. Production of a gas
 Lesson 1 Production of a gas T E A C H E R G U I D E CO 2 Lesson summary Students meet volcanologist Victor Helguson, who is studying the gases released by volcanoes in Iceland. Students conduct a chemical
Lesson 1 Production of a gas T E A C H E R G U I D E CO 2 Lesson summary Students meet volcanologist Victor Helguson, who is studying the gases released by volcanoes in Iceland. Students conduct a chemical
Chapter 6, Lesson 9: Neutralizing Acids and Bases
 Chapter 6, Lesson 9: Neutralizing Acids and Bases Key Concepts ph is a measure of the concentration of H 3 O + ions in a solution. Adding an acid increases the concentration of H 3 O + ions in the solution.
Chapter 6, Lesson 9: Neutralizing Acids and Bases Key Concepts ph is a measure of the concentration of H 3 O + ions in a solution. Adding an acid increases the concentration of H 3 O + ions in the solution.
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic
 Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Lesson 4. Temperature change
 Lesson 4 Temperature change T E A C H E R G U I D E Lesson summary Students meet scientist Jason Williams, an industrial chemist who designs the materials and processes for making solar cells. He explains
Lesson 4 Temperature change T E A C H E R G U I D E Lesson summary Students meet scientist Jason Williams, an industrial chemist who designs the materials and processes for making solar cells. He explains
PREPARE FOR THE ACTIVITY. Activity Sheet Chapter 6, Lesson 8 ph and Color Change
 Activity Sheet Chapter 6, Lesson 8 ph and Color Change Name Date DEMONSTRATION 1. Your teacher poured green universal indicator into each of two cups. What does the change in color of the indicator solution
Activity Sheet Chapter 6, Lesson 8 ph and Color Change Name Date DEMONSTRATION 1. Your teacher poured green universal indicator into each of two cups. What does the change in color of the indicator solution
Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic
 Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic Name Date DEMONSTRATION 1. Your teacher blew through a straw into a universal indicator solution until it changed color. Did
Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic Name Date DEMONSTRATION 1. Your teacher blew through a straw into a universal indicator solution until it changed color. Did
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions
 Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Physical and Chemical Changes Or How Do You Know When You ve Made Something New?
 Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
In your activity, you will use a chemical reaction and a filter to remove some very small dissolved particles.
 Lesson 3 Forming a precipitate S T U D E N T A C T I V I T Y S H E E T Name: Meet the scientist Country: Republic of Peru Scientist: Adriana Garcia Sera, from Fiji, told you about the problems of ocean
Lesson 3 Forming a precipitate S T U D E N T A C T I V I T Y S H E E T Name: Meet the scientist Country: Republic of Peru Scientist: Adriana Garcia Sera, from Fiji, told you about the problems of ocean
Ocean Acidification in a Cup Materials
 Ocean Acidification in a Cup Ocean acidification is a problem that humans will have to deal with as we release more and more carbon dioxide into the atmosphere. This activity demonstrates how water can
Ocean Acidification in a Cup Ocean acidification is a problem that humans will have to deal with as we release more and more carbon dioxide into the atmosphere. This activity demonstrates how water can
Chapter 1, Lesson 3: The Ups and Downs of Thermometers
 Chapter 1, Lesson 3: The Ups and Downs of Thermometers Key Concepts The way a thermometer works is an example of heating and cooling a liquid. When heated, the molecules of the liquid in the thermometer
Chapter 1, Lesson 3: The Ups and Downs of Thermometers Key Concepts The way a thermometer works is an example of heating and cooling a liquid. When heated, the molecules of the liquid in the thermometer
What Do You Think? Investigate GOALS
 Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Chapter 6, Lesson 1: What is a Chemical Reaction?
 Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Percentage of Acetic Acid in Vinegar
 Microscale Percentage of Acetic Acid in Vinegar When sweet apple cider is fermented in the absence of oxygen, the product is an acid, vinegar. Most commercial vinegars are made by fermentation, but some,
Microscale Percentage of Acetic Acid in Vinegar When sweet apple cider is fermented in the absence of oxygen, the product is an acid, vinegar. Most commercial vinegars are made by fermentation, but some,
Chapter 5, Lesson 2 Surface Tension
 Chapter 5, Lesson 2 Surface Tension Key Concepts The attraction of molecules at the surface of a liquid is called surface tension. The polarity of water molecules can help explain why water has a strong
Chapter 5, Lesson 2 Surface Tension Key Concepts The attraction of molecules at the surface of a liquid is called surface tension. The polarity of water molecules can help explain why water has a strong
Titration with an Acid and a Base
 Skills Practice Titration with an Acid and a Base Titration is a process in which you determine the concentration of a solution by measuring what volume of that solution is needed to react completely with
Skills Practice Titration with an Acid and a Base Titration is a process in which you determine the concentration of a solution by measuring what volume of that solution is needed to react completely with
Characteristics of Chemical Change
 Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Exploring Acids & Bases
 Food Explorations Lab: Exploring Acids & Bases STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will use cabbage juice indicator to determine if two unknown samples are acids or
Food Explorations Lab: Exploring Acids & Bases STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will use cabbage juice indicator to determine if two unknown samples are acids or
Chapter 5, Lesson 1: Water is a Polar Molecule
 Chapter 5, Lesson 1: Water is a Polar Molecule Key Concepts The water molecule, as a whole, has 10 protons and 10 electrons, so it is neutral. In a water molecule, the oxygen atom and hydrogen atoms share
Chapter 5, Lesson 1: Water is a Polar Molecule Key Concepts The water molecule, as a whole, has 10 protons and 10 electrons, so it is neutral. In a water molecule, the oxygen atom and hydrogen atoms share
Photosynthesis-Cellular Respiration Cycle
 Photosynthesis-Cellular Respiration Cycle Lesson Concept Link Photosynthesis and cellular respiration are reverse processes. Plants use photosynthesis to make food and release oxygen and plants and animals
Photosynthesis-Cellular Respiration Cycle Lesson Concept Link Photosynthesis and cellular respiration are reverse processes. Plants use photosynthesis to make food and release oxygen and plants and animals
More Chemical Changes
 Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Post-Show. Chemistry. Periodic Table of the Elements. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED)
 CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
2/22/2019 NEW UNIT! Chemical Interactions. Atomic Basics #19
 NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
Chapter 5, Lesson 1: Water is a Polar Molecule
 Chapter 5, Lesson 1: Water is a Polar Molecule Key Concepts The water molecule, as a whole, has 10 protons and 10 electrons, so it is neutral. In a water molecule, the oxygen atom and hydrogen atoms share
Chapter 5, Lesson 1: Water is a Polar Molecule Key Concepts The water molecule, as a whole, has 10 protons and 10 electrons, so it is neutral. In a water molecule, the oxygen atom and hydrogen atoms share
Chemistry: classifying chemical and physical changes in various materials/substances
 Chemistry: classifying chemical and physical changes in various materials/substances Nikki Schilling, Ames, St. Paul Mn Based on original activity from Cool Chemistry Concoctions by Joe Rhatigan and Veronika
Chemistry: classifying chemical and physical changes in various materials/substances Nikki Schilling, Ames, St. Paul Mn Based on original activity from Cool Chemistry Concoctions by Joe Rhatigan and Veronika
Limiting Reactants Lab
 Name: Teacher s Name: Class: Block: Date: Partners: Limiting Reactants Lab Purpose: Through experimentation, determine the limiting reactant and the percent yield in a chemical reaction that generates
Name: Teacher s Name: Class: Block: Date: Partners: Limiting Reactants Lab Purpose: Through experimentation, determine the limiting reactant and the percent yield in a chemical reaction that generates
Bellevue College CHEM& 121 Experiment: Stoichiometric Analysis of an Antacid 1
 Experiment: Stoichiometric Analysis of an Antacid 1 Introduction In this lab, you will use the concept of stoichiometry to solve two sequential problems. First, you will try to determine the products of
Experiment: Stoichiometric Analysis of an Antacid 1 Introduction In this lab, you will use the concept of stoichiometry to solve two sequential problems. First, you will try to determine the products of
Name Date Class. Chapter 19 Acids, Bases, and Salts EXPERIMENT. ESTIMATION OF ph PURPOSE BACKGROUND MATERIALS (PER PAIR)
 Chapter 19 Acids, Bases, and Salts EXPERIMENT ESTIMATION OF Text Reference Sections 19.2 PURPOSE To estimate the of solutions by using acid base indicators. BACKGROUND If you are interested in gardening,
Chapter 19 Acids, Bases, and Salts EXPERIMENT ESTIMATION OF Text Reference Sections 19.2 PURPOSE To estimate the of solutions by using acid base indicators. BACKGROUND If you are interested in gardening,
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
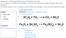 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
PDFMAILER.COM Print and send PDF files as s with any application, ad-sponsored and free of charge Activity # 14.
 Activity # 14 Name Purpose Date Date due Activities 10c and 10d - Performing More Examples of Chemical Reactions To perform a number of different chemical reactions, determine what the reactants and products
Activity # 14 Name Purpose Date Date due Activities 10c and 10d - Performing More Examples of Chemical Reactions To perform a number of different chemical reactions, determine what the reactants and products
Examples of Strong Acids: Strong Acid Formula Common Source Hydrochloric Acid HCl Stomach Acid
 ACIDS AND BASES: PH AND BUFFERS PURPOSE: To determine the ph of common acids and bases using a ph meter, ph paper, and red cabbage indicator. To test the effect of adding an acid or base to a buffer solution.
ACIDS AND BASES: PH AND BUFFERS PURPOSE: To determine the ph of common acids and bases using a ph meter, ph paper, and red cabbage indicator. To test the effect of adding an acid or base to a buffer solution.
Chemical Reaction Lab Bagged Chemical Reactions
 Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
LAB: Photosynthesis in Leaf Disks
 Name Date Period LAB: Photosynthesis in Leaf Disks H O N O R S B I O L O G Y : U N I T 3 Introduction: Photosynthesis is a process in which plants convert light energy (sunlight) into usable chemical energy
Name Date Period LAB: Photosynthesis in Leaf Disks H O N O R S B I O L O G Y : U N I T 3 Introduction: Photosynthesis is a process in which plants convert light energy (sunlight) into usable chemical energy
Lab- Properties of Acids and Bases. Name. PSI Chemistry
 Lab- Properties of Acids and Bases PSI Chemistry Name Introduction Acids and bases are useful reagents in the chemistry laboratory and play an important role in biology and nature. What are acids and bases?
Lab- Properties of Acids and Bases PSI Chemistry Name Introduction Acids and bases are useful reagents in the chemistry laboratory and play an important role in biology and nature. What are acids and bases?
Lab: Detecting ph of Commonly Used Acids and Bases
 Lab: Detecting ph of Commonly Used Acids and Bases FOR THE TEACHER Summary In this lab, students will use their knowledge of acids and bases to determine the acidity and basicity of every day items by
Lab: Detecting ph of Commonly Used Acids and Bases FOR THE TEACHER Summary In this lab, students will use their knowledge of acids and bases to determine the acidity and basicity of every day items by
Physical & Chemical PROPERTIES
 Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
MONDAY (12/12) TUESDAY (12/13) WEDNESDAY (12/14) THURSDAY (12/15) FRIDAY (12/16) Making Acid Rain (a lab) Quiz
 Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Experiment 8 - Chemical Changes
 Experiment 8 - Chemical Changes When a chemical change occurs, the chemicals that you start with are changed into different chemicals. We know when this happens because the new chemicals have different
Experiment 8 - Chemical Changes When a chemical change occurs, the chemicals that you start with are changed into different chemicals. We know when this happens because the new chemicals have different
Chapter 5, Lesson 5 Using Dissolving to Identify an Unknown
 Chapter 5, Lesson 5 Using Dissolving to Identify an Unknown Key Concepts Different substances are made from different atoms, ions, or molecules, which interact with water in different ways. Since dissolving
Chapter 5, Lesson 5 Using Dissolving to Identify an Unknown Key Concepts Different substances are made from different atoms, ions, or molecules, which interact with water in different ways. Since dissolving
Liquid X Lab. Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties?
 Names Block Date Liquid X Lab Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette to carefully place drops of each liquid
Names Block Date Liquid X Lab Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette to carefully place drops of each liquid
MiSP CHEMICAL REACTIONS, L3 Teacher Guide. Introduction
 MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
By: Michael Wild, Matt Huber, Jasmine Gilbert and Dr. Faith Yarberry
 Acid Chemistry By: Michael Wild, Matt Huber, Jasmine Gilbert and Dr. Faith Yarberry In this module the student will: Understand the concept of an Acid. Discover the differences between strong acids and
Acid Chemistry By: Michael Wild, Matt Huber, Jasmine Gilbert and Dr. Faith Yarberry In this module the student will: Understand the concept of an Acid. Discover the differences between strong acids and
EXPERIMENT. for a Weak Acid. Determination of K a
 EXPERIMENT Determination of K a for a Weak Acid Hands-On Labs, Inc. Version 42-0151-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin.
EXPERIMENT Determination of K a for a Weak Acid Hands-On Labs, Inc. Version 42-0151-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin.
Lyniece McKim Biology Instructor Star Valley High Afton, WY.
 Lyniece McKim Biology Instructor Star Valley High Afton, WY lmckm@lcsd2.org TEACHER SHARE-A-THON NABT 2010 Biology: Chemistry of Life Lab: Testing Mystery Substances Inquiry activity Using indicators;
Lyniece McKim Biology Instructor Star Valley High Afton, WY lmckm@lcsd2.org TEACHER SHARE-A-THON NABT 2010 Biology: Chemistry of Life Lab: Testing Mystery Substances Inquiry activity Using indicators;
Most people think of volcanoes as destructive. The high temperature
 37 Volcanic Landforms M O D E L I N G Most people think of volcanoes as destructive. The high temperature of volcanic lava can burn almost everything in its path. Volcanoes also release large amounts of
37 Volcanic Landforms M O D E L I N G Most people think of volcanoes as destructive. The high temperature of volcanic lava can burn almost everything in its path. Volcanoes also release large amounts of
Acids and Bases. How does ph affect biological solutions? Introduction. Prelab Preparation Review Section 2.3 on acids and bases in your textbook.
 Acids and Bases How does ph affect biological solutions? Learning Objectives To relate the ph scale to how acidic or basic a solution is. To explain how a buffer affects the ph of a solution. Process Objectives
Acids and Bases How does ph affect biological solutions? Learning Objectives To relate the ph scale to how acidic or basic a solution is. To explain how a buffer affects the ph of a solution. Process Objectives
White Before Your Eyes
 Name Class Date You have learned how to describe matter based on its physical and chemical properties. You have also learned some signs that can help you determine whether a change in matter is a physical
Name Class Date You have learned how to describe matter based on its physical and chemical properties. You have also learned some signs that can help you determine whether a change in matter is a physical
H 2 SO 4 HCN H 3 PO 4 HNO 3 HCl. Ca(OH) 2 C 3 H 5 (OH) 3 NaOH NH 4 OH. form LOTS of ions and. 2PbCO 3 Pb(OH) 2. VERY LITTLE ions
 2-15-17 WARM UP: INTRO TO ACIDS AND BASES.. Acids & Bases 14.2 14.3, p. 422-431 1) A few common Acids What do you notice about the formulas?? sulphuric acid hydrogen cyanide (prussic acid) phosphoric acid
2-15-17 WARM UP: INTRO TO ACIDS AND BASES.. Acids & Bases 14.2 14.3, p. 422-431 1) A few common Acids What do you notice about the formulas?? sulphuric acid hydrogen cyanide (prussic acid) phosphoric acid
Section I: Synthesis reactions Synthesis reactions occur when two or more substances come together to form a single new substance.
 TYPES OF CHEMICAL REACTIONS A Laboratory Investigation Purpose: Observe the five major types of reactions. Record observations for these reactions. Complete balanced equations for these reactions. Introduction:
TYPES OF CHEMICAL REACTIONS A Laboratory Investigation Purpose: Observe the five major types of reactions. Record observations for these reactions. Complete balanced equations for these reactions. Introduction:
Separating the Mixture
 Separating the Mixture 40- to 1 50-minute session ACTIVITY OVERVIEW I N V E S T 5 I O N I G AT Students perform their procedures written in Activity 3, A Plan to Separate the Mixture, to physically separate
Separating the Mixture 40- to 1 50-minute session ACTIVITY OVERVIEW I N V E S T 5 I O N I G AT Students perform their procedures written in Activity 3, A Plan to Separate the Mixture, to physically separate
T h e M yster y Pow der s L ab: Ph ysical an d Ch em ical Ch an ges
 T h e M yster y Pow der s L ab: Ph ysical an d Ch em ical Ch an ges Background Information: You have learned how to describe matter based on its physical and chemical properties. You have also learned
T h e M yster y Pow der s L ab: Ph ysical an d Ch em ical Ch an ges Background Information: You have learned how to describe matter based on its physical and chemical properties. You have also learned
Virtual Library Lesson: Oobleck, Gloop, and Glurch
 Oobleck, Gloop, and Glurch Lesson Overview Throughout this lesson, students will use inquiry skills to identify states of matter, describe physical properties, and modify the recipe to change physical
Oobleck, Gloop, and Glurch Lesson Overview Throughout this lesson, students will use inquiry skills to identify states of matter, describe physical properties, and modify the recipe to change physical
Learn to do quantitative titration reactions. Observe the mole ratios of several simple chemical reactions.
 CHAPTER 6 Stoichiometry of Reactions in Solution Objectives The objectives of this laboratory are to: Learn to do quantitative titration reactions. Observe the mole ratios of several simple chemical reactions.
CHAPTER 6 Stoichiometry of Reactions in Solution Objectives The objectives of this laboratory are to: Learn to do quantitative titration reactions. Observe the mole ratios of several simple chemical reactions.
Student Notes. Chemical Reactions LINK
 LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
1. Write date, topic and purpose in notebook. 2. Answer in your notebook: What are two signs that a chemical reaction is happening?
 Warm Up (5 min) Date: Monday, 1/4/16 Topic: Alka Seltzer Lab Purpose: To assess the Law of Conservation of Mass by designing an experiment to determine mass before and after a chemical reaction. 1. Write
Warm Up (5 min) Date: Monday, 1/4/16 Topic: Alka Seltzer Lab Purpose: To assess the Law of Conservation of Mass by designing an experiment to determine mass before and after a chemical reaction. 1. Write
Conservation of Mass in Chemical Reactions Lab. Experiment Question: What happens to the total mass of substances when a chemical reaction occurs?
 Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
11. Introduction to Acids, Bases, ph, and Buffers
 11. Introduction to Acids, Bases, ph, and Buffers What you will accomplish in this experiment You ll use an acid-base indicating paper to: Determine the acidity or basicity of some common household substances
11. Introduction to Acids, Bases, ph, and Buffers What you will accomplish in this experiment You ll use an acid-base indicating paper to: Determine the acidity or basicity of some common household substances
Acid Rain Drops Keep Falling on My Head Investigating Acid Rain
 Investigating Acid Rain Carbonic acid is produced when carbon dioxide gas dissolves in rain droplets of unpolluted air: (1) CO 2(g) + H 2 O (l) H 2 CO 3(aq) CO 2 Nitrous acid and nitric acid result from
Investigating Acid Rain Carbonic acid is produced when carbon dioxide gas dissolves in rain droplets of unpolluted air: (1) CO 2(g) + H 2 O (l) H 2 CO 3(aq) CO 2 Nitrous acid and nitric acid result from
Name Period Date. Lab: Introduction to Stoichiometry
 Name Period Date Lab: Introduction to Stoichiometry Introduction: Reactants are not always present in the exact ratio required by a balanced chemical equation. In planning any cost-effective production
Name Period Date Lab: Introduction to Stoichiometry Introduction: Reactants are not always present in the exact ratio required by a balanced chemical equation. In planning any cost-effective production
Liquid X Lab. Number of Drops Before Spilling Trial 1 Trial 2 Trial 3. Write a conclusion: How do your results for Liquid X compare to water?
 Names Block Date BIG QUESTION: Liquid X Lab Station 1 Surface Tension, Cohesion, and Adhesion Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette
Names Block Date BIG QUESTION: Liquid X Lab Station 1 Surface Tension, Cohesion, and Adhesion Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette
KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator
 KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator By Darby Sloss and Marianne Smith Edited by Anne Starace Abstract Chemistry is an important part of our lives. Kitchen Chemistry
KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator By Darby Sloss and Marianne Smith Edited by Anne Starace Abstract Chemistry is an important part of our lives. Kitchen Chemistry
Photosynthesis in Leaf Disks Teacher Preparation and Background Information
 AP Biology Name: Date: Photosynthesis in Leaf Disks Teacher Preparation and Background Information General Information: Solutions may be handled without gloves and may be disposed of in sink drains. In
AP Biology Name: Date: Photosynthesis in Leaf Disks Teacher Preparation and Background Information General Information: Solutions may be handled without gloves and may be disposed of in sink drains. In
Experiment#1 Beer s Law: Absorption Spectroscopy of Cobalt(II)
 : Absorption Spectroscopy of Cobalt(II) OBJECTIVES In successfully completing this lab you will: prepare a stock solution using a volumetric flask; use a UV/Visible spectrometer to measure an absorption
: Absorption Spectroscopy of Cobalt(II) OBJECTIVES In successfully completing this lab you will: prepare a stock solution using a volumetric flask; use a UV/Visible spectrometer to measure an absorption
By All INdICATIONS (2 Hours)
 By All INdICATIONS (2 Hours) Addresses NGSS Level of Difficulty: 5 Grade Range: 6-8 OVERVIEW In this activity, students create an acid-base indicator using red cabbage extract. Students then use this indicator
By All INdICATIONS (2 Hours) Addresses NGSS Level of Difficulty: 5 Grade Range: 6-8 OVERVIEW In this activity, students create an acid-base indicator using red cabbage extract. Students then use this indicator
Chemical Bonds. MATERIALS 24-well microplate calcium chloride candle citric acid conductivity tester ethanol gloves iron ring lab apron
 Microscale Chemical Bonds Chemical compounds are combinations of atoms held together by chemical bonds. These chemical bonds are of two basic types ionic and covalent. Ionic bonds result when one or more
Microscale Chemical Bonds Chemical compounds are combinations of atoms held together by chemical bonds. These chemical bonds are of two basic types ionic and covalent. Ionic bonds result when one or more
3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make?
 Chapter 4 Test 1. 2. 3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make? packs Work Space: 5. A kennel is moving 160
Chapter 4 Test 1. 2. 3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make? packs Work Space: 5. A kennel is moving 160
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version
 Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
By contrast, solubility equilibrium reactions are written from the perspective of the solid reactant dissolving into ions
 LAD F.2 (pg 1 of 8) Ksp Solubility Product for Calcium Hydroxide Name Per Introduction Most solubility equilibrium investigated in this course involve ionic compounds as opposed to molecular compounds.
LAD F.2 (pg 1 of 8) Ksp Solubility Product for Calcium Hydroxide Name Per Introduction Most solubility equilibrium investigated in this course involve ionic compounds as opposed to molecular compounds.
Experiment #7. Titration of Vinegar
 Experiment #7. Titration of Vinegar Goals 1. To determine the mass percent of acetic acid in a solution via titration. 2. To master the technique of titration. Introduction Vinegar is a common household
Experiment #7. Titration of Vinegar Goals 1. To determine the mass percent of acetic acid in a solution via titration. 2. To master the technique of titration. Introduction Vinegar is a common household
Name Period Date. Lab 9: Analysis of Commercial Bleach
 Name Period Date Lab 9: Analysis of Commercial Bleach Introduction Many common products are effective because they contain oxidizing agents. Some products, which contain oxidizing agents, are bleaches,
Name Period Date Lab 9: Analysis of Commercial Bleach Introduction Many common products are effective because they contain oxidizing agents. Some products, which contain oxidizing agents, are bleaches,
Global Experiment for the International Year of Chemistry. ph of the Planet
 Global Experiment for the International Year of Chemistry ph of the Planet This document contains a description for the ph of the Planet Activity that is part of the Global Experiment being conducted during
Global Experiment for the International Year of Chemistry ph of the Planet This document contains a description for the ph of the Planet Activity that is part of the Global Experiment being conducted during
Classifying Chemical Reactions: Lab Directions
 Classifying Chemical Reactions: Lab Directions Please Return Background: The power of chemical reactions to transform our lives is visible all around us in our homes, in our cars, even in our bodies. Chemists
Classifying Chemical Reactions: Lab Directions Please Return Background: The power of chemical reactions to transform our lives is visible all around us in our homes, in our cars, even in our bodies. Chemists
Activity Sheet Transferring thermal energy by dissolving salts
 Student Name: Date: Activity Sheet Transferring thermal energy by dissolving salts 1) Define Thermal energy and temperature in the boxes below. Thermal Energy Temperature Practice Experiment: Aim: To practice
Student Name: Date: Activity Sheet Transferring thermal energy by dissolving salts 1) Define Thermal energy and temperature in the boxes below. Thermal Energy Temperature Practice Experiment: Aim: To practice
Relative Solubility of Transition Elements
 Microscale Relative Solubility of Transition Elements The transition elements are found in periods 4, 5, and 6 between groups 2 and 13 of the periodic table. As the atomic number increases across a row
Microscale Relative Solubility of Transition Elements The transition elements are found in periods 4, 5, and 6 between groups 2 and 13 of the periodic table. As the atomic number increases across a row
Shifts in Equilibrium: Le Châtelier s Principle
 6 Shifts in Equilibrium: Le Châtelier s Principle Introduction Whenever a chemical reaction occurs, the reverse reaction can also occur. As the original reactants, on the left side of the equation, react
6 Shifts in Equilibrium: Le Châtelier s Principle Introduction Whenever a chemical reaction occurs, the reverse reaction can also occur. As the original reactants, on the left side of the equation, react
VANDERBILT STUDENT VOLUNTEERS FOR SCIENCE
 VANDERBILT STUDENT VOLUNTEERS FOR SCIENCE http://studentorgs.vanderbilt.edu/vsvs Evidence of a Chemical Reaction Fall 2018 Goal: To show students evidence of a chemical change. Fits TN standards:7ps1.2,
VANDERBILT STUDENT VOLUNTEERS FOR SCIENCE http://studentorgs.vanderbilt.edu/vsvs Evidence of a Chemical Reaction Fall 2018 Goal: To show students evidence of a chemical change. Fits TN standards:7ps1.2,
The grade 5 English science unit, Acids and Bases, meets the academic content standards set in the Korean curriculum, which state students should:
 This area addresses ph among different characteristics of solutions. It will be interesting for students to classify a variety of solutions into acids and bases by using the characteristics of the solutions.
This area addresses ph among different characteristics of solutions. It will be interesting for students to classify a variety of solutions into acids and bases by using the characteristics of the solutions.
CHM111 Lab Titration of Vinegar Grading Rubric
 Name Team Name CHM111 Lab Titration of Vinegar Grading Rubric Criteria Points possible Points earned Lab Performance Printed lab handout and rubric was brought to lab 3 Safety and proper waste disposal
Name Team Name CHM111 Lab Titration of Vinegar Grading Rubric Criteria Points possible Points earned Lab Performance Printed lab handout and rubric was brought to lab 3 Safety and proper waste disposal
EXPERIMENT. Titration for Acetic Acid in Vinegar
 EXPERIMENT Titration for Acetic Acid in Vinegar Hands-On Labs, Inc. Version 42-0208-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin.
EXPERIMENT Titration for Acetic Acid in Vinegar Hands-On Labs, Inc. Version 42-0208-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin.
Titration of HCl with Sodium Hydroxide
 Titration of HCl with Sodium Hydroxide Lab Report for the Subject of Advanced Chemistry Anon Durongpisitkul, Karis Katekovit, Varun Saketharam,Thanon Thamvorapol, Chanon Anektanasup- January 28, 2017 1
Titration of HCl with Sodium Hydroxide Lab Report for the Subject of Advanced Chemistry Anon Durongpisitkul, Karis Katekovit, Varun Saketharam,Thanon Thamvorapol, Chanon Anektanasup- January 28, 2017 1
Introduction. Objectives
 Experiment: Acids, Bases, and Buffers * Introduction Many common household solutions contain acids and bases. Acid-base indicators, such as litmus and red cabbage juice, turn different colors in acidic
Experiment: Acids, Bases, and Buffers * Introduction Many common household solutions contain acids and bases. Acid-base indicators, such as litmus and red cabbage juice, turn different colors in acidic
2 burets (50 ml) Standard solution of NaOH (0.600 M) Phenolphthalein indicator
 Name: \[-[L Percentage of Acetic Acid In Vinegar Lab 4-5 INTRODUCTION: The quality of acid in a sample of vinegar may be found by titrating the sample against a standard basic solution. ost commercial
Name: \[-[L Percentage of Acetic Acid In Vinegar Lab 4-5 INTRODUCTION: The quality of acid in a sample of vinegar may be found by titrating the sample against a standard basic solution. ost commercial
Chapter 2, Lesson 5: Changing State Melting
 Chapter 2, Lesson 5: Changing State Melting Key Concepts Melting is a process that causes a substance to change from a solid to a liquid. Melting occurs when the molecules of a solid speed up enough that
Chapter 2, Lesson 5: Changing State Melting Key Concepts Melting is a process that causes a substance to change from a solid to a liquid. Melting occurs when the molecules of a solid speed up enough that
Name: Hour: Photosynthesis in Leaf Disks
 Name: Hour: Photosynthesis in Leaf Disks Safety Information: While the solutions may be handled without gloves and may be disposed of in the sink drains, goggles must be worn during the experiment. Background
Name: Hour: Photosynthesis in Leaf Disks Safety Information: While the solutions may be handled without gloves and may be disposed of in the sink drains, goggles must be worn during the experiment. Background
Experiment #10: Analysis of Antacids
 Experiment #10: Analysis of Antacids Purpose: In this experiment you will prepare one solution that is approximately 0.1 M NaOH. Then you will standardize this solution, which means that you will experimentally
Experiment #10: Analysis of Antacids Purpose: In this experiment you will prepare one solution that is approximately 0.1 M NaOH. Then you will standardize this solution, which means that you will experimentally
Identification of an Unknown Compound through Mass Correlations
 EXPERIMENT Identification of an Unknown Compound through Mass Correlations PURPOSE To carry out a series of decomposition reactions for five different unknown, and use stoichiometry in order to identify
EXPERIMENT Identification of an Unknown Compound through Mass Correlations PURPOSE To carry out a series of decomposition reactions for five different unknown, and use stoichiometry in order to identify
5 th GRADE PHYSICAL AND CHEMICAL TESTS
 5 th GRADE PHYSICAL AND CHEMICAL TESTS Summary: Students investigate 5 unknown white powders. They gather clues by observing the physical and chemical changes of the powders. At the end of the activity,
5 th GRADE PHYSICAL AND CHEMICAL TESTS Summary: Students investigate 5 unknown white powders. They gather clues by observing the physical and chemical changes of the powders. At the end of the activity,
Chapter 3 Cell Processes and Energy. Name: Teacher: Hour:
 Chapter 3 Cell Processes and Energy Name: Teacher: Hour: 1 Unit: Cell Processes and Energy Unit Completion Date: Vocabulary and Big Ideas Photosynthesis photosynthesis autotroph heterotroph chlorophyll
Chapter 3 Cell Processes and Energy Name: Teacher: Hour: 1 Unit: Cell Processes and Energy Unit Completion Date: Vocabulary and Big Ideas Photosynthesis photosynthesis autotroph heterotroph chlorophyll
Experiment 6 Shifts in Equilibrium: Le Châtelier s Principle
 Experiment 6 Shifts in Equilibrium: Le Châtelier s Principle Introduction Whenever a chemical reaction occurs, the reverse reaction can also occur. As the original reactants, on the left side of the equation,
Experiment 6 Shifts in Equilibrium: Le Châtelier s Principle Introduction Whenever a chemical reaction occurs, the reverse reaction can also occur. As the original reactants, on the left side of the equation,
Lab #3 ph and Buffers
 Page1 Lab #3 ph and Objectives: Learn to construct a proper data table and line graph Understand how the ph scale works Use a ph meter to measure the ph of common household substances Understand the meaning
Page1 Lab #3 ph and Objectives: Learn to construct a proper data table and line graph Understand how the ph scale works Use a ph meter to measure the ph of common household substances Understand the meaning
25. Qualitative Analysis 2
 25. Qualitative Analysis 2 This experiment uses a series of wet chemistry analytical tests to determine the functional group present in a series of known and an unknown compound. Each student receives
25. Qualitative Analysis 2 This experiment uses a series of wet chemistry analytical tests to determine the functional group present in a series of known and an unknown compound. Each student receives
Energy Transformations Activities Students engage in different activities to observe energy changing form!
 Energy Transformations Activities Students engage in different activities to observe energy changing form! Name: Energy Labs and Activities Activity 1: Static Electricity Glue this side down Procedure:
Energy Transformations Activities Students engage in different activities to observe energy changing form! Name: Energy Labs and Activities Activity 1: Static Electricity Glue this side down Procedure:
Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise.
 Experiment 10 Stoichiometry- Gravimetric Analysis Pre-lab Assignment Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise. Purpose The purpose this experiment
Experiment 10 Stoichiometry- Gravimetric Analysis Pre-lab Assignment Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise. Purpose The purpose this experiment
5 th Grade Lesson Plan: Matter and Chemical Reactions
 5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
Chemical Reactions: The Copper Cycle
 1 Chemical Reactions: The Copper Cycle ORGANIZATION Mode: pairs assigned by instructor Grading: lab notes, lab performance and post-lab report Safety: Goggles, closed-toe shoes, lab coat, long pants/skirts
1 Chemical Reactions: The Copper Cycle ORGANIZATION Mode: pairs assigned by instructor Grading: lab notes, lab performance and post-lab report Safety: Goggles, closed-toe shoes, lab coat, long pants/skirts
Chemical Reactions Lab PSI Chemistry
 Chemical Reactions Lab PSI Chemistry Name Purpose: Observe the different types of chemical reactions. Materials: - 6 test tubes - test tube rack - 0.3 M copper(ii)sulfate solution - 0.3 M sodium hydroxide
Chemical Reactions Lab PSI Chemistry Name Purpose: Observe the different types of chemical reactions. Materials: - 6 test tubes - test tube rack - 0.3 M copper(ii)sulfate solution - 0.3 M sodium hydroxide
Student Exploration: Chemical Changes
 Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
Chemistry Lab Define Acids and Bases
 Introduction Chemistry Lab Define Acids and Bases By the 1500 s chemists recognized that certain substances shared a common property a sour taste. These substances possessed other characteristic properties
Introduction Chemistry Lab Define Acids and Bases By the 1500 s chemists recognized that certain substances shared a common property a sour taste. These substances possessed other characteristic properties
Part II. Cu(OH)2(s) CuO(s)
 The Copper Cycle Introduction In this experiment, you will carry out a series of reactions starting with copper metal. This will give you practice handling chemical reagents and making observations. It
The Copper Cycle Introduction In this experiment, you will carry out a series of reactions starting with copper metal. This will give you practice handling chemical reagents and making observations. It
