Overexpression of type-a rice response regulators, OsRR3 and OsRR5, results in lower sensitivity to cytokinins
|
|
|
- Nelson Hall
- 5 years ago
- Views:
Transcription
1 Overexpression of type-a rice response regulators, OsRR3 and OsRR5, results in lower sensitivity to cytokinins X. Cheng*, H. Jiang*, J. Zhang, Y. Qian, S. Zhu and B. Cheng School of Life Science, Anhui Agricultural University, Hefei, Anhui, China *These authors contributed equally to this study. Corresponding author: B. Cheng Genet. Mol. Res. 9 (1): (2010) Received November 13, 2009 Accepted December 8, 2009 Published March 2, 2010 ABSTRACT. Response regulators are part of a two-component regulatory system. The type-a Arabidopsis response regulators act as negative regulators. To understand the function of type-a response regulators in the model monocot plant, rice (Japonica cultivar-group: Zhonghua11), we overexpressed two type-a OsRR genes, OsRR3 and OsRR5 (pact1:osrr3 and pact1:osrr5). We hoped to gain insight into their molecular function in cytokinin-signaling pathways. Both OsRR3 and OsRR5 overexpressors had longer roots and more lateral roots compared with Zhonghua11, when treated with exogenous cytokinin. Using callus formation and chlorophyll content assays, we found that Zhonghua11 was more sensitive to cytokinin compared with other cultivars of rice, expressing high transcript levels of OsRR3 and OsRR5. The expression of most type-a OsRR genes was repressed by OsRR3 and OsRR5 overexpression. However, semi-quantitative RT- PCR showed that three type-a OsRR genes showed increased expression. Our results suggest that both OsRR3 and OsRR5 mainly act as negative regulators of cytokinin signaling, as indicated by the reduced sensitivity of OsRR3 and OsRR5 overexpressors to exogenous cytokinins. Key words: Cytokinin; Response regulator; Rice; RT-PCR
2 Overexpression of type-a rice response regulators 349 INTRODUCTION Cytokinins are plant hormones that regulate various growth and physiological processes, such as cell division, apical dominance, cell proliferation, chloroplast biogenesis, vascular differentiation, root and leaf differentiation, photomorphogenic development, and inhibition of leaf senescence (Mok and Mok, 2001). Genetic and molecular research in plants has suggested the involvement of a two-component signaling system in cytokinin signal perception and transduction, which are used by prokaryotic and eukaryotic organisms (Stock et al., 2000). The simple two-component system involves a His kinase (HK) sensor that perceives the signal and a response regulator (RR) that mediates the output of the pathway. The HK sensor autophosphorylates a His residue in response to an output signal, and the phosphate group is transferred to a conserved Asp residue in the receiver domain of the response regulator. The downstream signaling of the pathway is then modulated by the phosphorylated response regulator. The cytokinin multistep two-component signaling system comprises sensor histidine kinase proteins, histidine phosphotransfer proteins (HPt), and response regulator proteins. In this pathway, signals are transferred from a His in the receptor kinase to an Asp in the response regulator via His-Asp-His-Asp phosphotransfer reactions between alternating His and Asp residues (Kakimoto, 2003; Muller and Sheen, 2007). In Arabidopsis thaliana, cytokinin independent 1 (CKI1) was first isolated in an activation-tagging screen for hypocotyls that produce calli in the absence of exogenous cytokinin. As CKI1 did not require cytokinin for its activity, it was not thought to be a cytokinin receptor (Kakimoto, 1996). The cytokinin receptors cytokinin response 1 (CRE1), wooden leg 1 (WOL), Arabidopsis histidine kinase 4 (AHK4), and its homologs AHK2 and AHK3 are similar to bacterial His sensor hybrid kinases, which include a ligandbinding site, a His kinase domain, and a receiver domain, and were suggested to be receptors for cytokinins by yeast and Escherichia coli systems (Inoue et al., 2001; Suzuki et al., 2001; Ueguchi et al., 2001a,b). Loss-of-function mutations of the cytokinin receptor CRE1 exhibited reduced cytokinin sensitivity, and overexpression of AHK2 in Arabidopsis protoplasts enhanced the cytokinin-induced expression from the ARR6 promoter (Inoue et al., 2001; Hwang and Sheen, 2001). Analysis of ahk2, ahk3, and ahk4 with double mutants showed reduced response to cytokinin, and triple mutants where all three cytokinin receptors were disrupted indicated that they possess distinct overlapping functions in shoot, root, and seed growth (Higuchi et al., 2004; Nishimura et al., 2004). All plant HKs contain a C-terminal receiver domain and a conserved transmitter domain, which includes characteristic sequence motifs and a conserved His residue (West and Stock, 2001). In a recent study, three Arabidopsis HK genes, three maize HK genes, and five rice HK genes have been identified (Inoue et al., 2001; Yonekura-Sakakibara et al., 2004). In response to cytokinin binding, HKs autophosphorylate on a conserved His residue and then transfer the phosphate group to response regulators via an intermediate set of histidine phosphotransfer proteins (Suzuki et al., 1998; Hutchison et al., 2006). The majority of HPs act as redundant, positive regulators of cytokinin signaling and affect many aspects of plant development (Hutchison et al., 2006). In addition, physiological and biochemical analyses have shown that most AHPt proteins are capable of interacting with the hybrid kinase and response regulator (Urao et al., 1998).
3 X. Cheng et al. 350 The typical response regulators in Arabidopsis are classified into type-a and type- B (Hwang et al., 2002). The type-a response regulators are relatively small, containing a receiver domain along with short N- and C-terminal extensions (Imamura et al., 1998; Brandstatter and Kieber, 1998). The type-b response regulators have a receiver domain, an extensive C-terminal extension involved in transcription regulation (Sakai et al., 1998; Lohrmann et al., 1999), and a type-b signature GARP motif of about 60 amino acids that is related to the Myb repeat of transcription factors (Sakai et al., 2000). Expression is rapidly induced in response to exogenous cytokinin in the type-a ARR genes. In contrast, the steady-state levels of type-b ARR transcripts are not affected by cytokinin or other plant hormones (Brandstatter and Kieber, 1998; Taniguchi et al., 1998; Lohrmann and Harter, 2002). Overexpression of various type-a ARRs in Arabidopsis protoplasts repressed the ability of cytokinin to induce the ARR6 promoter, indicating that type-a ARRs are transcriptional repressors (Hwang and Sheen, 2001). Overexpression of ARR8 repressed shoot regeneration and greening of calli, and overexpression of ARR15 resulted in reduced sensitivity to cytokinin (Osakabe et al., 2002; Kiba et al., 2003). Overexpression of ARR7 showed that it is a negative regulator of early cytokinin-regulated genes and mainly acts as a transcriptional repressor (Lee et al., 2007). In rice, type-a genes have been isolated and characterized (Sakakibara et al., 1998). Overexpression of OsRR6 displayed altered morphologies and changes in cytokinin metabolism (Hirose et al., 2008). The response regulators involved in cytokinin signaling have been reported in the Gramineae, but knowledge of type-a response regulators in rice is very limited compared with Arabidopsis. To better understand the role of type-a OsRRs in cytokinin signaling, we overexpressed OsRR3 and OsRR5, two type-a OsRRs, in rice and investigated which genes are regulated by OsRR3 and OsRR5 in response to cytokinin. MATERIAL AND METHODS Plasmid construction and rice transformation Total RNA was extracted from the Japonica cultivar-group: Zhonghua11 (ZH11) using the RNAiso reagent (TaKaRa Biotechnology, Japan). After RNase-free DNase (TaKaRa Biotechnology) treatment, purified RNA was reverse transcribed using the Access RT-PCR (reverse transcription-polymerase chain reaction) system (Promega, USA) to obtain first-strand cdna. The OsRR3 and OsRR5 sequences were amplified from cdna with the following primers: 5-GCTCTAGATGTCGACGAAGACAGTGCC-3 and 5-AACTGCAGCAAAGT CATTTCATGATGACGC-3 for OsRR3, and 5-GCTCTAGATCTGATGGCCACCTGC-3 and 5-GCTCTAGATCACCGGAGGACGCGGC-3 for OsRR5. The PCR products were subcloned into the pmd18-t vector (TaKaRa Biotechnology). The fragments with the coding regions of OsRR3 and OsRR5 were inserted into the pxqact vector, after removing the GUS gene, under the control of the pact1 promoter. The resulting pact1:osrr3 and pact1:osrr5 constructs were introduced into ZH11 by Agrobacterium-mediated transformation. Rice plants were cultivated in the experimental field at Anhui Agricultural University in the natural growing season. For the analysis of cytokinin-responsive genes in rice, we screened T1-generation transgenic rice using the selective marker NPTII, which has resistance to G418. The haploid T2-generation transgenic rice plants were then selected by DNA hybridization to eliminate
4 Overexpression of type-a rice response regulators 351 polyploidy transgenic rice (data not shown). Finally, progeny from identified T3 homozygous transformants for OsRR3 and OsRR5 overexpression was produced and used in subsequent experiments. ZH11 was used as the wild-type line for all experiments. Seedling hormone response assays Rice seeds were sterilized with 0.1% HgCl 2 for 1 h and washed with distilled water before soaking overnight in distilled water. The seeds were transferred to a nylon net floating on sterilized water. For hormone sensitivity assay, seeds were grown on plates at 23 C in a culture room with a 16-h photoperiod for 6 days, then transferred to plates containing either different concentrations of 2,4- D (2,4-dichlorophenoxy acetic acid), 6-BA (6-benzyladenine acid), and abscisic acid (ABA) in 0.1% DMSO (dimethylsulfoxide), respectively, or an equal volume of DMSO as a control for another 10 days. Hormone treatment Rice seeds were grown on germination agar plates containing N6 media with vitamins, 3% sucrose, 0.03% casein enzymatic hydrolysate, 0.3% agar, ph 5.9, at 28 C in a culture room with constant light. To test the hormone response of rice calli, seeds were grown on N6 media for 7 days and then transferred to sterilized N6 media agar plates containing different concentrations of 2,4-D and 6-BA for callus induction. After 3 weeks under constant light, rice calli were photographed with a digital camera. Chlorophyll content assays Seeds were grown on plates at 23 C with a 16-h photoperiod for 30 days. For chlorophyll assays, seedlings were transferred to new plates supplemented with different concentrations of 6-BA and incubated in the dark at 23 C for either 2 or 4 days. To assess dark-induced leaf senescence, full leaves were detached from the plants and the chlorophylls were extracted with methanol. Chlorophyll content was then measured spectrophotometrically (Porra et al., 1989). Semiquantitative RT-PCR analysis Seeds were germinated on plates at 37 C for 2 days and then transferred to new plants in a nutrient solution at 28 C in a culture room under a 16-h light and a 8-h dark photoperiod for an additional 15 days. For cytokinin assays, the seedlings were transferred to a nutrient solution supplemented with 1 μm 6-BA in 0.1% DMSO or an equal volume of DMSO as a control for 2 h and then harvested. Total RNAs were extracted from roots and leaves of seedlings with the RNAiso reagent (TaKaRa Biotechnology) according to manufacturer instructions. cdnas were generated from the RNA with the Access RT-PCR system (Promega, USA). To examine the expression patterns of OsRR genes, sets of specific primers for the corresponding genes were designed. The rice actin1 gene was used as an internal control to normalize for variance in the quality of RNA and the amount of input cdna. Reactions were performed using Taq polymerase (TaKaRa Biotechnology) on a thermal cycler (Tpersonal 48; Biometra, Germany). A 3-μL aliquot of the reaction mix was separated on 1% (w/v) agarose gels and stained with ethidium bromide. Two independent experiments were performed with consistent results.
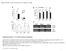
5 X. Cheng et al. 352 RESULTS Validation of T3 transgenic rice plants Transgenic plants overexpressing the OsRR3 and OsRR5 genes were generated, and the expression of the OsRR3 and OsRR5 genes was examined to determine the levels of OsRR3 and OsRR5 in the transgenic plants. RT-PCR analysis showed that plants harboring pact1:osrr3 and pact1:osrr5 had significantly increased levels of OsRR3 and OsRR5 transcripts. All T3 transgenic rice plants exhibited higher expression of OsRR3 or OsRR5 compared with ZH11 (Figure 1). Transgenic lines , , and , , and were used in all subsequent experiments based on their high OsRR3 or OsRR5 expression levels. Figure 1. Semi-quantitative RT-PCR identification of transgenic rice. Total RNAs were prepared from leaves of transgenic plants and ZH11 (labeled CK). OsAct1 was amplified as a control. A. PCR identification of OsRR3 overexpression in transgenic rice. B. PCR identification of OsRR5 overexpression in transgenic rice. Rice seedlings overexpressing OsRR3 and OsRR5 are less sensitive to cytokinin inhibition of root elongation and lateral root formation The two transgenic rice lines constitutively overexpressing OsRR3 and OsRR5 (pact1:osrr3 and pact1:osrr5) looked similar to cultivated species ZH11, in terms of leaf initiation, formation of leaves, roots, flowers, and seeds. To assess the roles of the two type-a OsRRs in the cytokinin response pathway, we examined root elongation and lateral root formation in response to exogenous hormone addition. We compared root elongation of ZH11 with pact1:osrr3 and pact1:osrr5 seedlings across a range of hormone concentrations between 0 and 10 μm (Figure 2). In the absence of exogenous hormone, the roots of ZH11, pact1:osrr3, and pact1:osrr5 were the same. The roots of all three kinds of rice were not affected until the hormone concentration reached 10 nm (Figure 2). With further increase in BA concentration, primary root elongation decreased sharply, with a half-maximal inhibition at 0.3
6 Overexpression of type-a rice response regulators 353 μm (Figure 2A). At higher concentrations (>5 μm), primary root elongations of the three kinds of rice were similar (Figure 2). At concentrations of BA of μm, pact1:osrr3 and pact1:osrr5 plants had longer roots compared to ZH11. This showed that pact1:osrr3 and pact1:osrr5 were less sensitive to cytokinin compared to ZH11 (Figure 2A). However, the root growth responses of pact1:osrr3 and pact1:osrr5 to varying concentrations of other plant hormones (2,4-D or ABA) were similar to that of ZH11, indicating that resistance to root elongation inhibition in both pact1:osrr3 and pact1:osrr5 are cytokinin dependent (Figure 2B and C). Formation of lateral roots is inhibited by cytokinin in plants (Werner et al., 2003). In the lateral root formation assay, we measured the number of lateral roots on ZH11, pact1:osrr3, and pact1:osrr5 with a range of BA concentrations between 0 and 10 μm. Lateral root formation decreased sharply between 0.01 and 1 μm in ZH11 compared with pact1:osrr3 and pact1:osrr5 (Figure 2D). Overall, the results were similar to those observed in the root elongation assay. These results showed that high levels of both OsRR3 and OsRR5 expression reduce sensitivity to exogenous application of cytokinin. Figure 2. Analysis of root elongation and lateral root formation during overexpression of OsRR3 and OsRR5 in transgenic rice and ZH11 (CK) in the presence of exogenous hormones. A., B., C. Seeds were grown on plates supplemented with the specified concentrations of BA, 2,4-D, ABA, or DMSO control, under a 16-h photoperiod at 23 C. Error bars represent SE (N = 30). D. Seeds were grown on plates supplemented with the specified concentrations of BA control under a 16-h photoperiod at 23 C. The total number of lateral roots was determined at 15 days. Error bars represent SE (N = 30). E. Phenotypes of roots for 16-day-old seedlings of (from left to right) ZH11, pact1:osrr3, and pact1:osrr5 treated with 0.5 μm BA under a 16-h photoperiod.
7 X. Cheng et al. 354 Callus formation of pact1:osrr3 and pact1:osrr5 is less sensitive to cytokinin Cytokinin promotes cell division and initiates shoots in concert with auxin in cultured plant tissues (Miller et al., 1955). To investigate the cytokinin responses of OsRR3 and OsRR5 transgenic plants, we examined callus formation in ZH11, pact1:osrr3, and pact1:osrr5 in response to various concentration of the cytokinin 6-BA and the auxin 2,4-D. ZH11 explants formed green shoots at high cytokinin:auxin ratios. At low cytokinin:auxin ratios, calli were observed. With increasing cytokinin:auxin ratios, larger calli were formed and green shoots were longer (Figure 3A). Both pact1:osrr3 and pact1:osrr5 formed smaller calli and shorter shoots with concentrations of hormones that were able to induce ZH11 calli (Figure 3B and C). The decrease in sensitivity to hormones of pact1:osrr3 and pact1:osrr5 in callus and shoot formation showed that both OsRR3 and OsRR5 act as negative regulators of the cytokinin-signaling system. Figure 3. Callus formation of ZH11, pact1:osrr3, and pact1:osrr5. Rice seeds were grown on N6 media containing specified concentrations of 2,4-D and 6-BA. After 4 weeks in culture, the induced calli were arranged and photographed. A. ZH11. B. pact1:osrr3. C. pact1:osrr5.
8 Overexpression of type-a rice response regulators 355 Reduced chlorophyll content in pact1:osrr3 and pact1:osrr5 Cytokinins take part in most aspects of plant growth and development, including leaf senescence. Cytokinins are known to delay leaf senescence (Gan and Amansino, 1995). For this reason, we measured chlorophyll content with various concentrations of BA to test the effects of exogenous cytokinin on the senescence process. Without dark-induced treatment, the chloroplast levels of ZH11, pact1:osrr3 and pact1:osrr5 were similar. After 2 days of dark-induced senescence, the leaf chloroplast levels of pact1:osrr3 and pact1:osrr5 plants were more reduced compared with ZH11 at the same concentrations of BA. A similar tendency was found after an additional 2 days of dark-induced senescence (Figure 4). Leaf senescence induced by the dark was delayed by increasing BA concentration, but the chlorophyll content was more reduced in pact1:osrr3 and pact1:osrr5 with increasing BA concentrations compared with ZH11. These data also confirm that overexpression of OsRR3 and OsRR5 reduces sensitivity to exogenous cytokinins. Figure 4. pact1:osrr3 and pact1:osrr5 show accelerated leaf senescence. Full leaves of 30-day-old ZH11 (CK), pact1:osrr3 and pact1:osrr5 plants were cultured in 0.1% DMSO containing given concentrations of BA for either 2 or 4 days in the dark. Chlorophyll content was determined spectrophotometrically as described in the Methods. Results are pooled from four independent experiments (N = 15). Expression of OsRR genes during cytokinin treatment The expression of the OsRR genes was examined by RT-PCR. The expression of most type-a and type-b OsRR genes was detected in leaves and roots at different levels, except for OsRR11, which was expressed only in roots. However, three type-a OsRR genes: OsRR8, OsRR12, and OsRR 13, and one type-b OsRR gene, OsRR19, were not detected in leaves and roots. The expression of type-b OsRR genes was higher in both leaves and roots compared with type-a OsRR genes (Figure 5A and C). The expressions of most type-a ARR genes increased
9 X. Cheng et al. 356 rapidly in response to exogenous cytokinin (D Agostino et al., 2000), and thus, we analyzed the expression of OsRR genes after cytokinin treatment for 2 h. The results showed different OsRR expression patterns in ZH11, pact1:osrr3, and pact1:osrr5. However, the results for pact1:osrr3 and pact1:osrr5 were similar (Figure 5B and D). After exogenous cytokinin 6-BA treatment, the expressions of most type-a OsRR genes increased in ZH11, but type-b OsRR genes were not affected. In contrast, the expression of most type-a OsRR genes decreased or did not increase in the two transgenic plants, with the exception of the OsRR2, OsRR4, and OsRR6; the transcripts of these three type-a OsRR genes accumulated. Similar to ZH11, type-b OsRR genes were not affected. The results suggest that OsRR3 and OsRR5 are primary cytokinin response genes, functioning as negative regulators of cytokinin signaling. Figure 5. Expression pattern of OsRR genes. RNAs were isolated from roots and leaves from ZH11, pact1:osrr3, and pact1:osrr5 growing in nutrient medium or with 1 μm 6-BA. OsACT1 was used as a template control. A. Expression of OsRR genes without hormone. Lane 1 = Leaf of ZH11; lane 2 = leaf of pact1:osrr3; lane 3 = root of ZH11; lane 4 = root of pact1:osrr3. B. Expression of OsRR genes treated with 1 μm 6-BA for 2 h. Lane 1 = Leaf of ZH11; lane 2 = leaf of pact1:osrr3; lane 3 = root of ZH11; lane 4 = root of pact1:osrr3. C. Expression of OsRR genes without hormone. Lane 1 = Leaf of ZH11; lane 2 = leaf of pact1:osrr5; lane 3 = root of ZH11; lane 4 = root of pact1:osrr5. D. Expression of OsRR genes treated with 1 μm 6-BA for 2 h. Lane 1 = Leaf of ZH11; lane 2 = leaf of pact1:osrr5; lane 3 = root of ZH11; lane 4 = root of pact1:osrr5. DISCUSSION The components of the cytokinin-signaling system comprise histidine kinases, histidine phosphotransferase proteins, and response regulators. Various genetic, molecular biological, and biochemical studies have suggested that type-a Arabidopsis response regulators are negative regulators in cytokinin signaling; however, studies of cytokinin signaling in rice are limited. To gain new insights into the function of type-a OsRRs in cytokinin signaling, OsRR3 and OsRR5, two type-a OsRRs, were overexpressed in rice under the control of the rice actin1
10 Overexpression of type-a rice response regulators 357 (pact1) promoter. This promoter produces much higher levels of constitutive expression in rice compared with the cauliflower mosaic virus 35S promoter (Sentoku et al., 2000). There are many reports of the expression of cytokinin-inducible type-a ARR genes in Arabidopsis. Overexpression of ARR4 in cultured stems of transgenic plants promoted shoot formation in the presence of cytokinin, and overexpression of ARR8 repressed shoot formation and greening of calli (Osakabe et al., 2002). Root elongation was enhanced by ARR15 overexpression (Kiba et al., 2003). Overexpression ARR7 led to reduced sensitivity to cytokinin (Lee et al., 2007). Our results showed that overexpression of OsRR3 and OsRR5 causes reduced sensitivity to cytokinin in terms of root elongation, lateral root formation, callus formation, and darkinduced leaf senescence. OsRRs are cytokinin response genes, but the two overexpressing plants were not affected by 2,4-D and ABA in the root elongation assays. In addition, overexpression of OsRR6 alters rice morphology (abnormal flowers and sterility) and cytokinin metabolism (Hirose et al., 2007). Overexpression of ARR4 and ARR8 alters cytokinin responses differentially, in terms of flowering time and longer stems, compared with control plant (Osakabe et al., 2002). However, in our experiments, transgenic rice showed normal growth compared with ZH11, and showed no changes in morphogenesis, such as formation of leaves, roots, stem, flowers, flowering time, and callus formation of adult plants. We hypothesize that type-a RRs may not exhibit the same cytokinin-signaling regulation in all conditions, and may use different pathways to affect upstream and downstream development in the cytokinin-signaling system. In our experiments, both pact1:osrr3 and pact1:osrr5 showed similar results in different experiments. According to protein sequence analysis, we found that the OsRR3 and OsRR5 proteins have high homology, with highly conserved Lys and Asp residues (D-D- K) in the receiver domain. By phylogenetic analysis, we know that OsRR3 and OsRR5 have a highly homologous phylogenetic relationship (Jain et al., 2006). Thus, we hypothesize that OsRR3 and OsRR5 have similar functions in the cytokinin-signaling system. The mrna of most type-a ARRs, ZmRRs, and OsRRs rapidly accumulate after cytokinin treatment (Sakakibara et al., 1998; Asakura et al., 2003; Giulini et al., 2004; Hirose et al., 2007). In our study, the expression of OsRR genes in the two overexpressing plants was the same as in the wild-type ZH11 in the absence of cytokinin. More than six RR genes are induced by cytokinin, yet only three of the induced genes are validated by the results of Hirose et al. (2008). This result may be due to different environmental factors and different tissues used for the expression of these genes in this study. We found that OsRRs expressions are different in leaves and roots, and that the expression of the same OsRR gene in leaf and root is different, indicating that cytokinin signaling in rice may have overlapping functions. After cytokinin treatment, the expression of three of the 16 type-a OsRR genes was increased, and that of nine type-a OsRR genes was reduced, while the remaining 3 type-a genes were not detected. Overexpression of OsRR3 and OsRR5 caused transcription repression but also activated type-a OsRRs, such as OsRR2, OsRR4, and OsRR6. One possible explanation is that overexpression of OsRR3 and OsRR5 mainly participates in the repression of the transcription of other type-a OsRRs, providing a negative feedback mechanism. Most of the OsRR genes were expressed at different levels in leaves and roots without exogenous cytokinin treatment. The transcript levels of type-b OsRRs are relatively high compared with type-a OsRR genes, and the expression of OsRR11 was not found in leaves. OsRR8, OsRR12 and OsRR13 transcripts were not found in either leaves or roots. In contrast, OsRR8, OsRR12, and OsRR13 were expressed in flowers and panicles (Ito and Kurata, 2006). These results suggest that OsRR genes exhibit organ-specific expression and differential expression patterns. The
11 X. Cheng et al. 358 type-b RRs are the transcription factors that respond to cytokinins (Sakai et al., 2000). We found that the expression levels of type-b OsRRs did not change in response to exogenous cytokinin treatment; thus, we think that type-a OsRRs may represent the primary cytokinin response genes. The data presented here indicate that the cytokinin-signaling system in rice is a complex-signaling pathway that involves a degree of cross-talk. ACKNOWLEDGMENTS Research supported by the National Natural Science Foundation of China (# ) and National High-Tech Research and Development Program (863 Program) (#2008AA10Z408). We thank members of the Key Laboratory of Crop Biotechnology of Anhui Province and Guo Wei for assistance in conducting these experiments. REFERENCES Asakura Y, Hagino T, Ohta Y, Aoki K, et al. (2003). Molecular characterization of His-Asp phosphorelay signaling factors in maize leaves: implications of the signal divergence by cytokinin-inducible response regulators in the cytosol and the nuclei. Plant Mol. Biol. 52: Brandstatter I and Kieber JJ (1998). Two genes with similarity to bacterial response regulators are rapidly and specifically induced by cytokinin in Arabidopsis. Plant Cell 10: Gan and Amansino (1995). Inhibition of leaf senescence by autoregulated production of cytokinin. Science 270: Giulini A, Wang J and Jackson D (2004). Control of phyllotaxy by the cytokinin-inducible response regulator homologue ABPHYL1. Nature 430: Higuchi M, Pischke MS, Mahonen AP, Miyawaki K, et al. (2004). In planta functions of the Arabidopsis cytokinin receptor family. Proc. Natl. Acad. Sci. U. S. A. 101: Hirose N, Makita N, Kojima M, Kamada-Nobusada T, et al. (2007). Overexpression of a type-a response regulator alters rice morphology and cytokinin metabolism. Plant Cell Physiol. 48: Hirose N, Takei K, Kuroha T, Kamada-Nobusada T, et al. (2008). Regulation of cytokinin biosynthesis, compartmentalization and translocation. J. Exp. Bot. 59: Hutchison CE, Li J, Argueso C, Gonzalez M, et al. (2006). The Arabidopsis histidine phosphotransfer proteins are redundant positive regulators of cytokinin signaling. Plant Cell 18: Hwang I and Sheen J (2001). Two-component circuitry in Arabidopsis cytokinin signal transduction. Nature 413: Hwang I, Chen HC and Sheen J (2002). Two-component signal transduction pathways in Arabidopsis. Plant Physiol. 129: Imamura A, Hanaki N, Umeda H, Nakamura A, et al. (1998). Response regulators implicated in His-to-Asp phosphotransfer signaling in Arabidopsis. Proc. Natl. Acad. Sci. U.S.A. 95: Inoue T, Higuchi M, Hashimoto Y, Seki M, et al. (2001). Identification of CRE1 as a cytokinin receptor from Arabidopsis. Nature 409: Ito Y and Kurata N (2006). Identification and characterization of cytokinin-signalling gene families in rice. Gene 382: Jain M, Tyagi AK and Khurana JP (2006). Molecular characterization and differential expression of cytokinin-responsive type-a response regulators in rice (Oryza sativa). BMC Plant Biol. 6: 1. Kakimoto T (1996). CKI1, a histidine kinase homolog implicated in cytokinin signal transduction. Science 274: Kakimoto T (2003). Perception and signal transduction of cytokinins. Annu. Rev. Plant Biol. 54: Kiba T, Yamada H, Sato S, Kato T, et al. (2003). The type-a response regulator, ARR15, acts as a negative regulator in the cytokinin-mediated signal transduction in Arabidopsis thaliana. Plant Cell Physiol. 44: Lee DJ, Park JY, Ku SJ, Ha YM, et al. (2007). Genome-wide expression profiling of Arabidopsis response regulator 7 (ARR7) overexpression in cytokinin response. Mol. Genet. Genomics 277: Lohrmann J and Harter K (2002). Plant two-component signaling systems and the role of response regulators. Plant Physiol. 128: Lohrmann J, Buchholz G, Keitel C, Sweere U, et al. (1999). Differential expression and nuclear localization of response regulator-like proteins from Arabidopsis thaliana. Plant Biol. 5: Miller CO, Skoog F, Von Saltza MH and Strong F (1955). Kinetin, a cell division factor from deoxyribonucleic acid. J.
12 Overexpression of type-a rice response regulators 359 Am. Chem. Soc. 77: Mok DW and Mok MC (2001). Cytokinin metabolism and action. Annu. Rev. Plant Physiol. Plant Mol. Biol. 52: Muller B and Sheen J (2007). Advances in cytokinin signaling. Science 318: Nishimura C, Ohashi Y, Sato S, Kato T, et al. (2004). Histidine kinase homologs that act as cytokinin receptors possess overlapping functions in the regulation of shoot and root growth in Arabidopsis. Plant Cell 16: Osakabe Y, Miyata S, Urao T, Seki M, et al. (2002). Overexpression of Arabidopsis response regulators, ARR4/ATRR1/ IBC7 and ARR8/ATRR3, alters cytokinin responses differentially in the shoot and in callus formation. Biochem. Biophys. Res. Commun. 293: Porra RJ, Thompson WA and Kriedemann PE (1989). Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. Biophys. Acta 975: Sakai H, Aoyama T, Bono H and Oka A (1998). Two-component response regulators from Arabidopsis thaliana contain a putative DNA-binding motif. Plant Cell Physiol. 39: Sakai H, Aoyama T and Oka A (2000). Arabidopsis ARR1 and ARR2 response regulators operate as transcriptional activators. Plant J. 24: Sakakibara H, Suzuki M, Takei K, Deji A, et al. (1998). A response-regulator homologue possibly involved in nitrogen signal transduction mediated by cytokinin in maize. Plant J. 14: Sentoku N, Sato Y and Matsuoka M (2000). Overexpression of rice OSH genes induces ectopic shoots on leaf sheaths of transgenic rice plants. Dev. Biol. 220: Stock AM, Robinson VL and Goudreau PN (2000). Two-component signal transduction. Annu. Rev. Biochem. 69: Suzuki T, Imamura A, Ueguchi C and Mizuno T (1998). Histidine-containing phosphotransfer (HPt) signal transducers implicated in His-to-Asp phosphorelay in Arabidopsis. Plant Cell Physiol 39: Suzuki T, Miwa K, Ishikawa K, Yamada H, et al. (2001). The Arabidopsis sensor His-kinase, AHk4, can respond to cytokinins. Plant Cell Physiol. 42: Taniguchi M, Kiba T, Sakakibara H, Ueguchi C, et al. (1998). Expression of Arabidopsis response regulator homologs is induced by cytokinins and nitrate. FEBS Lett. 429: Ueguchi C, Koizumi H, Suzuki T and Mizuno T (2001a). Novel family of sensor histidine kinase genes in Arabidopsis thaliana. Plant Cell Physiol. 42: Ueguchi C, Sato S, Kato T and Tabata S (2001b). The AHK4 gene involved in the cytokinin-signaling pathway as a direct receptor molecule in Arabidopsis thaliana. Plant Cell Physiol. 42: Urao T, Yakubov B, Yamaguchi-Shinozaki K and Shinozaki K (1998). Stress-responsive expression of genes for twocomponent response regulator-like proteins in Arabidopsis thaliana. FEBS Lett. 427: Werner T, Motyka V, Laucou V, Smets R, et al. (2003). Cytokinin-deficient transgenic Arabidopsis plants show multiple developmental alterations indicating opposite functions of cytokinins in the regulation of shoot and root meristem activity. Plant Cell 15: West AH and Stock AM (2001). Histidine kinases and response regulator proteins in two-component signaling systems. Trends Biochem. Sci. 26: Yonekura-Sakakibara K, Kojima M, Yamaya T and Sakakibara H (2004). Molecular characterization of cytokinin-responsive histidine kinases in maize. Differential ligand preferences and response to cis-zeatin. Plant Physiol. 134:
Cytokinin Signaling in Arabidopsis
 The Plant Cell, S47 S59, Supplement 2002, www.plantcell.org 2002 American Society of Plant Biologists Cytokinin Signaling in Arabidopsis Claire E. Hutchison and Joseph J. Kieber 1 Biology Department, University
The Plant Cell, S47 S59, Supplement 2002, www.plantcell.org 2002 American Society of Plant Biologists Cytokinin Signaling in Arabidopsis Claire E. Hutchison and Joseph J. Kieber 1 Biology Department, University
Cytokinin. Fig Cytokinin needed for growth of shoot apical meristem. F Cytokinin stimulates chloroplast development in the dark
 Cytokinin Abundant in young, dividing cells Shoot apical meristem Root apical meristem Synthesized in root tip, developing embryos, young leaves, fruits Transported passively via xylem into shoots from
Cytokinin Abundant in young, dividing cells Shoot apical meristem Root apical meristem Synthesized in root tip, developing embryos, young leaves, fruits Transported passively via xylem into shoots from
23-. Shoot and root development depend on ratio of IAA/CK
 Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL
 GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
Cytokinin signal perception and transduction Alexander Heyl and Thomas Schmülling
 480 Cytokinin signal perception and transduction Alexander eyl and Thomas Schmülling In the past few years, enormous progress has been made in understanding cytokinin perception and signalling. Three cytokinin
480 Cytokinin signal perception and transduction Alexander eyl and Thomas Schmülling In the past few years, enormous progress has been made in understanding cytokinin perception and signalling. Three cytokinin
Two-Component Signal Transduction Pathways in Arabidopsis 1
 Genome Analysis Two-Component Signal Transduction Pathways in Arabidopsis 1 Ildoo Hwang, Huei-Chi Chen, and Jen Sheen* Department of Genetics, Harvard Medical School, Boston, Massachusetts 02114; and Department
Genome Analysis Two-Component Signal Transduction Pathways in Arabidopsis 1 Ildoo Hwang, Huei-Chi Chen, and Jen Sheen* Department of Genetics, Harvard Medical School, Boston, Massachusetts 02114; and Department
Elisabeth J Chapman and Mark Estelle
 Minireview Cytokinin and auxin intersection in root meristems Elisabeth J Chapman and Mark Estelle Address: Division of Biology, 9500 Gilman Drive, La Jolla, CA 92093-0116, USA. Correspondence: Mark Estelle.
Minireview Cytokinin and auxin intersection in root meristems Elisabeth J Chapman and Mark Estelle Address: Division of Biology, 9500 Gilman Drive, La Jolla, CA 92093-0116, USA. Correspondence: Mark Estelle.
Cytokinin signaling Fernando J Ferreira and Joseph J Kieber
 Cytokinin signaling Fernando J Ferreira and Joseph J Kieber Cytokinins influence many aspects of plant growth and development. The current model for cytokinin signaling is a multi-step phosphorelay similar
Cytokinin signaling Fernando J Ferreira and Joseph J Kieber Cytokinins influence many aspects of plant growth and development. The current model for cytokinin signaling is a multi-step phosphorelay similar
Plant Two-Component Signaling Systems and the Role of Response Regulators 1
 Update on Plant Signaling Plant Two-Component Signaling Systems and the Role of Response Regulators 1 Jens Lohrmann and Klaus Harter* Universität Freiburg, Institut für Biologie II/Botanik, Schänzlestrasse
Update on Plant Signaling Plant Two-Component Signaling Systems and the Role of Response Regulators 1 Jens Lohrmann and Klaus Harter* Universität Freiburg, Institut für Biologie II/Botanik, Schänzlestrasse
NATURAL VARIATION IN THE CYTOKININ METABOLIC NETWORK IN ARABIDOPSIS THALIANA
 NATURAL VARIATION IN THE CYTOKININ METABOLIC NETWORK IN ARABIDOPSIS THALIANA PŘÍRODNÍ VARIACE METABOLISMU CYTOKININŮ U ARABIDOPSIS THALIANA Samsonová Z. 1, 2, 3, Kuklová A. 1, 2, Mazura P. 1, 2, Rotková
NATURAL VARIATION IN THE CYTOKININ METABOLIC NETWORK IN ARABIDOPSIS THALIANA PŘÍRODNÍ VARIACE METABOLISMU CYTOKININŮ U ARABIDOPSIS THALIANA Samsonová Z. 1, 2, 3, Kuklová A. 1, 2, Mazura P. 1, 2, Rotková
Type B Response Regulators of Arabidopsis Play Key Roles in Cytokinin Signaling and Plant Development W
 The Plant Cell, Vol. 20: 2102 2116, August 2008, www.plantcell.org ã 2008 American Society of Plant Biologists Type B Response Regulators of Arabidopsis Play Key Roles in Cytokinin Signaling and Plant
The Plant Cell, Vol. 20: 2102 2116, August 2008, www.plantcell.org ã 2008 American Society of Plant Biologists Type B Response Regulators of Arabidopsis Play Key Roles in Cytokinin Signaling and Plant
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated
 Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Analysis of regulatory function of circadian clock. on photoreceptor gene expression
 Thesis of Ph.D. dissertation Analysis of regulatory function of circadian clock on photoreceptor gene expression Tóth Réka Supervisor: Dr. Ferenc Nagy Biological Research Center of the Hungarian Academy
Thesis of Ph.D. dissertation Analysis of regulatory function of circadian clock on photoreceptor gene expression Tóth Réka Supervisor: Dr. Ferenc Nagy Biological Research Center of the Hungarian Academy
Ectopic expression of the phosphomimic mutant version of Arabidopsis response regulator 1 promotes a constitutive cytokinin response phenotype
 University of Kentucky UKnowledge Plant and Soil Sciences Faculty Publications Plant and Soil Sciences 1-14-2014 Ectopic expression of the phosphomimic mutant version of Arabidopsis response regulator
University of Kentucky UKnowledge Plant and Soil Sciences Faculty Publications Plant and Soil Sciences 1-14-2014 Ectopic expression of the phosphomimic mutant version of Arabidopsis response regulator
The Role of Cytokinin in Female Gametophyte Development in Arabidopsis. Chia-Yi Cheng
 The Role of Cytokinin in Female Gametophyte Development in Arabidopsis Chia-Yi Cheng A dissertation submitted to the faculty of the University of North Carolina at Chapel Hill in partial fulfillment of
The Role of Cytokinin in Female Gametophyte Development in Arabidopsis Chia-Yi Cheng A dissertation submitted to the faculty of the University of North Carolina at Chapel Hill in partial fulfillment of
CONTROL OF PLANT GROWTH AND DEVELOPMENT BI-2232 RIZKITA R E
 CONTROL OF PLANT GROWTH AND DEVELOPMENT BI-2232 RIZKITA R E The development of a plant the series of progressive changes that take place throughout its life is regulated in complex ways. Factors take part
CONTROL OF PLANT GROWTH AND DEVELOPMENT BI-2232 RIZKITA R E The development of a plant the series of progressive changes that take place throughout its life is regulated in complex ways. Factors take part
Maria V. Yamburenko, Yan O. Zubo, Radomíra Vanková, Victor V. Kusnetsov, Olga N. Kulaeva, Thomas Börner
 ABA represses the transcription of chloroplast genes Maria V. Yamburenko, Yan O. Zubo, Radomíra Vanková, Victor V. Kusnetsov, Olga N. Kulaeva, Thomas Börner Supplementary data Supplementary tables Table
ABA represses the transcription of chloroplast genes Maria V. Yamburenko, Yan O. Zubo, Radomíra Vanková, Victor V. Kusnetsov, Olga N. Kulaeva, Thomas Börner Supplementary data Supplementary tables Table
RNA Synthesis and Processing
 RNA Synthesis and Processing Introduction Regulation of gene expression allows cells to adapt to environmental changes and is responsible for the distinct activities of the differentiated cell types that
RNA Synthesis and Processing Introduction Regulation of gene expression allows cells to adapt to environmental changes and is responsible for the distinct activities of the differentiated cell types that
Reproduction, Seeds and Propagation
 Reproduction, Seeds and Propagation Diploid (2n) somatic cell Two diploid (2n) somatic cells Telophase Anaphase Metaphase Prophase I One pair of homologous chromosomes (homologues) II Homologues condense
Reproduction, Seeds and Propagation Diploid (2n) somatic cell Two diploid (2n) somatic cells Telophase Anaphase Metaphase Prophase I One pair of homologous chromosomes (homologues) II Homologues condense
THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING. AnitaHajdu. Thesis of the Ph.D.
 THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING AnitaHajdu Thesis of the Ph.D. dissertation Supervisor: Dr. LászlóKozma-Bognár - senior research associate Doctoral
THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING AnitaHajdu Thesis of the Ph.D. dissertation Supervisor: Dr. LászlóKozma-Bognár - senior research associate Doctoral
Major Plant Hormones 1.Auxins 2.Cytokinins 3.Gibberelins 4.Ethylene 5.Abscisic acid
 Plant Hormones Lecture 9: Control Systems in Plants What is a Plant Hormone? Compound produced by one part of an organism that is translocated to other parts where it triggers a response in target cells
Plant Hormones Lecture 9: Control Systems in Plants What is a Plant Hormone? Compound produced by one part of an organism that is translocated to other parts where it triggers a response in target cells
Plant Growth and Development
 Plant Growth and Development Concept 26.1 Plants Develop in Response to the Environment Factors involved in regulating plant growth and development: 1. Environmental cues (e.g., day length) 2. Receptors
Plant Growth and Development Concept 26.1 Plants Develop in Response to the Environment Factors involved in regulating plant growth and development: 1. Environmental cues (e.g., day length) 2. Receptors
Biological Roles of Cytokinins
 Direct Control of Shoot Meristem Activity by a Cytokinin-Activating Enzyme By Kurakawa et. Al. Published in Nature Presented by Boyana Grigorova Biological Roles of Cytokinins Cytokinins are positive regulators
Direct Control of Shoot Meristem Activity by a Cytokinin-Activating Enzyme By Kurakawa et. Al. Published in Nature Presented by Boyana Grigorova Biological Roles of Cytokinins Cytokinins are positive regulators
Effect of temperature on endogenous hormone levels and opposite phyllotaxy in maize leaf primordial
 Effect of temperature on endogenous hormone levels and opposite phyllotaxy in maize leaf primordial H. Ye, G.M. Han, Q. Ma, Y.Q. Tan, H.Y. Jiang, S.W. Zhu and B.J. Cheng School of Life Sciences, Anhui
Effect of temperature on endogenous hormone levels and opposite phyllotaxy in maize leaf primordial H. Ye, G.M. Han, Q. Ma, Y.Q. Tan, H.Y. Jiang, S.W. Zhu and B.J. Cheng School of Life Sciences, Anhui
CHAPTER : Prokaryotic Genetics
 CHAPTER 13.3 13.5: Prokaryotic Genetics 1. Most bacteria are not pathogenic. Identify several important roles they play in the ecosystem and human culture. 2. How do variations arise in bacteria considering
CHAPTER 13.3 13.5: Prokaryotic Genetics 1. Most bacteria are not pathogenic. Identify several important roles they play in the ecosystem and human culture. 2. How do variations arise in bacteria considering
Histidine Kinase Homologs That Act as Cytokinin Receptors Possess Overlapping Functions in the Regulation of Shoot and Root Growth in Arabidopsis
 The Plant Cell, Vol. 16, 1365 1377, June 2004, www.plantcell.org ª 2004 American Society of Plant Biologists RESEARCH ARTICLES Histidine Kinase Homologs That Act as Cytokinin Receptors Possess Overlapping
The Plant Cell, Vol. 16, 1365 1377, June 2004, www.plantcell.org ª 2004 American Society of Plant Biologists RESEARCH ARTICLES Histidine Kinase Homologs That Act as Cytokinin Receptors Possess Overlapping
Two-Component Signaling Elements and Histidyl-Aspartyl Phosphorelays
 The Arabidopsis Book 2008 American Society of Plant Biologists First published on July 14, 2008; doi: 10.1199/tab.0112 This chapter is an updated version of a chapter originally published on March 27,
The Arabidopsis Book 2008 American Society of Plant Biologists First published on July 14, 2008; doi: 10.1199/tab.0112 This chapter is an updated version of a chapter originally published on March 27,
Plant Molecular and Cellular Biology Lecture 10: Plant Cell Cycle Gary Peter
 Plant Molecular and Cellular Biology Lecture 10: Plant Cell Cycle Gary Peter 9/10/2008 1 Learning Objectives Explain similarities and differences between fungal, mammalian and plant cell cycles Explain
Plant Molecular and Cellular Biology Lecture 10: Plant Cell Cycle Gary Peter 9/10/2008 1 Learning Objectives Explain similarities and differences between fungal, mammalian and plant cell cycles Explain
Figure 18.1 Blue-light stimulated phototropism Blue light Inhibits seedling hypocotyl elongation
 Blue Light and Photomorphogenesis Q: Figure 18.3 Blue light responses - phototropsim of growing Corn Coleoptile 1. How do we know plants respond to blue light? 2. What are the functions of multiple BL
Blue Light and Photomorphogenesis Q: Figure 18.3 Blue light responses - phototropsim of growing Corn Coleoptile 1. How do we know plants respond to blue light? 2. What are the functions of multiple BL
Supplemental Data. Perrella et al. (2013). Plant Cell /tpc
 Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Cytokinins. Downloaded From: on 07 Mar 2019 Terms of Use:
 Cytokinins Authors: Joseph J. Kieber, and G. Eric Schaller Source: The Arabidopsis Book, 2014(12) Published By: American Society of Plant Biologists URL: https://doi.org/10.1199/tab.0168 BioOne Complete
Cytokinins Authors: Joseph J. Kieber, and G. Eric Schaller Source: The Arabidopsis Book, 2014(12) Published By: American Society of Plant Biologists URL: https://doi.org/10.1199/tab.0168 BioOne Complete
Supplemental Data. Chen and Thelen (2010). Plant Cell /tpc
 Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach
 Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Regulation of Phosphate Homeostasis by microrna in Plants
 Regulation of Phosphate Homeostasis by microrna in Plants Tzyy-Jen Chiou 1 *, Kyaw Aung 1,2, Shu-I Lin 1,3, Chia-Chune Wu 1, Su-Fen Chiang 1, and Chun-Lin Su 1 Abstract Upon phosphate (Pi) starvation,
Regulation of Phosphate Homeostasis by microrna in Plants Tzyy-Jen Chiou 1 *, Kyaw Aung 1,2, Shu-I Lin 1,3, Chia-Chune Wu 1, Su-Fen Chiang 1, and Chun-Lin Su 1 Abstract Upon phosphate (Pi) starvation,
Nature Genetics: doi: /ng Supplementary Figure 1. The phenotypes of PI , BR121, and Harosoy under short-day conditions.
 Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
3.B.1 Gene Regulation. Gene regulation results in differential gene expression, leading to cell specialization.
 3.B.1 Gene Regulation Gene regulation results in differential gene expression, leading to cell specialization. We will focus on gene regulation in prokaryotes first. Gene regulation accounts for some of
3.B.1 Gene Regulation Gene regulation results in differential gene expression, leading to cell specialization. We will focus on gene regulation in prokaryotes first. Gene regulation accounts for some of
Plant Propagation PLS 3221/5222
 Plant Propagation PLS 3221/5222 Dr. Sandra Wilson Dr. Mack Thetford Chapter 2 Introduction to the Biology of Plant Propagation -A review- 1 5. Plant Hormones and Plant development Phytohormones Nt Naturally
Plant Propagation PLS 3221/5222 Dr. Sandra Wilson Dr. Mack Thetford Chapter 2 Introduction to the Biology of Plant Propagation -A review- 1 5. Plant Hormones and Plant development Phytohormones Nt Naturally
The Plant Cell, November. 2017, American Society of Plant Biologists. All rights reserved
 The Genetics of Floral Development Teaching Guide Overview The development of flowers in angiosperm plants provided a critical evolutionary advantage, allowing more options for pollen dispersal and seed
The Genetics of Floral Development Teaching Guide Overview The development of flowers in angiosperm plants provided a critical evolutionary advantage, allowing more options for pollen dispersal and seed
Regulation of Transcription in Eukaryotes. Nelson Saibo
 Regulation of Transcription in Eukaryotes Nelson Saibo saibo@itqb.unl.pt In eukaryotes gene expression is regulated at different levels 1 - Transcription 2 Post-transcriptional modifications 3 RNA transport
Regulation of Transcription in Eukaryotes Nelson Saibo saibo@itqb.unl.pt In eukaryotes gene expression is regulated at different levels 1 - Transcription 2 Post-transcriptional modifications 3 RNA transport
Computational Cell Biology Lecture 4
 Computational Cell Biology Lecture 4 Case Study: Basic Modeling in Gene Expression Yang Cao Department of Computer Science DNA Structure and Base Pair Gene Expression Gene is just a small part of DNA.
Computational Cell Biology Lecture 4 Case Study: Basic Modeling in Gene Expression Yang Cao Department of Computer Science DNA Structure and Base Pair Gene Expression Gene is just a small part of DNA.
LECTURE 4: PHOTOTROPISM
 http://smtom.lecture.ub.ac.id/ Password: https://syukur16tom.wordpress.com/ LECTURE 4: PHOTOTROPISM LECTURE FLOW 1. 2. 3. 4. 5. INTRODUCTION DEFINITION INITIAL STUDY PHOTROPISM MECHANISM PHOTORECEPTORS
http://smtom.lecture.ub.ac.id/ Password: https://syukur16tom.wordpress.com/ LECTURE 4: PHOTOTROPISM LECTURE FLOW 1. 2. 3. 4. 5. INTRODUCTION DEFINITION INITIAL STUDY PHOTROPISM MECHANISM PHOTORECEPTORS
PERCEPTION AND SIGNAL TRANSDUCTION
 Annu. Rev. Plant Biol. 2003. 54:605 27 doi: 10.1146/annurev.arplant.54.031902.134802 Copyright c 2003 by Annual Reviews. All rights reserved First published online as a Review in Advance on January 31,
Annu. Rev. Plant Biol. 2003. 54:605 27 doi: 10.1146/annurev.arplant.54.031902.134802 Copyright c 2003 by Annual Reviews. All rights reserved First published online as a Review in Advance on January 31,
Ph.D. thesis. Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana
 Ph.D. thesis Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana Written by: Edit Ábrahám Temesváriné Supervisors: Dr. László Szabados
Ph.D. thesis Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana Written by: Edit Ábrahám Temesváriné Supervisors: Dr. László Szabados
Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family
 Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family GENES & DEVELOPMENT (2000) 14: 108 117 INTRODUCTION Flower Diagram INTRODUCTION Abscission In plant, the process by which a plant
Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family GENES & DEVELOPMENT (2000) 14: 108 117 INTRODUCTION Flower Diagram INTRODUCTION Abscission In plant, the process by which a plant
Plant transformation
 Plant transformation Objectives: 1. What is plant transformation? 2. What is Agrobacterium? How and why does it transform plant cells? 3. How is Agrobacterium used as a tool in molecular genetics? References:
Plant transformation Objectives: 1. What is plant transformation? 2. What is Agrobacterium? How and why does it transform plant cells? 3. How is Agrobacterium used as a tool in molecular genetics? References:
Arabidopsis thaliana. Lucia Strader. Assistant Professor, Biology
 Arabidopsis thaliana Lucia Strader Assistant Professor, Biology Arabidopsis as a genetic model Easy to grow Small genome Short life cycle Self fertile Produces many progeny Easily transformed HIV E. coli
Arabidopsis thaliana Lucia Strader Assistant Professor, Biology Arabidopsis as a genetic model Easy to grow Small genome Short life cycle Self fertile Produces many progeny Easily transformed HIV E. coli
Questions for Biology IIB (SS 2006) Wilhelm Gruissem
 Questions for Biology IIB (SS 2006) Plant biology Wilhelm Gruissem The questions for my part of Biology IIB, Plant Biology, are provided for self-study and as material for the exam. Please note that the
Questions for Biology IIB (SS 2006) Plant biology Wilhelm Gruissem The questions for my part of Biology IIB, Plant Biology, are provided for self-study and as material for the exam. Please note that the
Expression Patterns of OsPIL11, a Phytochrome-Interacting Factor in Rice, and Preliminary Analysis of Its Roles in Light Signal Transduction
 Rice Science, 2012, 19(4): 263 268 Copyright 2012, China National Rice Research Institute Published by Elsevier BV. All rights reserved Expression Patterns of OsPIL11, a Phytochrome-Interacting Factor
Rice Science, 2012, 19(4): 263 268 Copyright 2012, China National Rice Research Institute Published by Elsevier BV. All rights reserved Expression Patterns of OsPIL11, a Phytochrome-Interacting Factor
Chapter 15 Active Reading Guide Regulation of Gene Expression
 Name: AP Biology Mr. Croft Chapter 15 Active Reading Guide Regulation of Gene Expression The overview for Chapter 15 introduces the idea that while all cells of an organism have all genes in the genome,
Name: AP Biology Mr. Croft Chapter 15 Active Reading Guide Regulation of Gene Expression The overview for Chapter 15 introduces the idea that while all cells of an organism have all genes in the genome,
Electromagenetic spectrum
 Light Controls of Plant Development 1 Electromagenetic spectrum 2 Light It is vital for photosynthesis and is also necessary to direct plant growth and development. It acts as a signal to initiate and
Light Controls of Plant Development 1 Electromagenetic spectrum 2 Light It is vital for photosynthesis and is also necessary to direct plant growth and development. It acts as a signal to initiate and
CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-15 PLANT GROWTH AND DEVELOPMENT
 CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-15 PLANT GROWTH AND DEVELOPMENT Root, stem leaves, flower, fruits and seeds arise in orderly manner in plants. The sequence of growth is as follows-
CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-15 PLANT GROWTH AND DEVELOPMENT Root, stem leaves, flower, fruits and seeds arise in orderly manner in plants. The sequence of growth is as follows-
Useful Propagation Terms. Propagation The application of specific biological principles and concepts in the multiplication of plants.
 Useful Propagation Terms Propagation The application of specific biological principles and concepts in the multiplication of plants. Adventitious Typically describes new organs such as roots that develop
Useful Propagation Terms Propagation The application of specific biological principles and concepts in the multiplication of plants. Adventitious Typically describes new organs such as roots that develop
CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON
 PROKARYOTE GENES: E. COLI LAC OPERON CHAPTER 13 CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON Figure 1. Electron micrograph of growing E. coli. Some show the constriction at the location where daughter
PROKARYOTE GENES: E. COLI LAC OPERON CHAPTER 13 CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON Figure 1. Electron micrograph of growing E. coli. Some show the constriction at the location where daughter
High nitrogen conditions enhance the cooling damage to pollen in rice plants : proteome analysis of mature anthers
 High nitrogen conditions enhance the cooling damage to pollen in rice plants : proteome analysis of mature anthers Takami Hayashi 1, Tomoya Yamaguchi 1, Katsuhiro Nakayama 1, Setsuko Komatsu 2 and Setsuo
High nitrogen conditions enhance the cooling damage to pollen in rice plants : proteome analysis of mature anthers Takami Hayashi 1, Tomoya Yamaguchi 1, Katsuhiro Nakayama 1, Setsuko Komatsu 2 and Setsuo
Supplementary Figure S1. Amino acid alignment of selected monocot FT-like and TFL-like sequences. Sequences were aligned using ClustalW and analyzed
 Supplementary Figure S1. Amino acid alignment of selected monocot FT-like and TFL-like sequences. Sequences were aligned using ClustalW and analyzed using the Geneious software. Accession numbers of the
Supplementary Figure S1. Amino acid alignment of selected monocot FT-like and TFL-like sequences. Sequences were aligned using ClustalW and analyzed using the Geneious software. Accession numbers of the
A MicroRNA as a Translational Repressor of APETALA2 in Arabidopsis Flower Development
 A MicroRNA as a Translational Repressor of APETALA2 in Arabidopsis Flower Development Xuemei Chen Waksman Institute, Rutgers University, Piscataway, NJ 08854, USA. E-mail: xuemei@waksman.rutgers.edu Plant
A MicroRNA as a Translational Repressor of APETALA2 in Arabidopsis Flower Development Xuemei Chen Waksman Institute, Rutgers University, Piscataway, NJ 08854, USA. E-mail: xuemei@waksman.rutgers.edu Plant
Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function
 Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function Brian D. Gregory Department of Biology Penn Genome Frontiers Institute University of Pennsylvania
Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function Brian D. Gregory Department of Biology Penn Genome Frontiers Institute University of Pennsylvania
** * * * Col-0 cau1 CAU1. Actin2 CAS. Actin2. Supplemental Figure 1. CAU1 affects calcium accumulation.
 Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Gene regulation I Biochemistry 302. Bob Kelm February 25, 2005
 Gene regulation I Biochemistry 302 Bob Kelm February 25, 2005 Principles of gene regulation (cellular versus molecular level) Extracellular signals Chemical (e.g. hormones, growth factors) Environmental
Gene regulation I Biochemistry 302 Bob Kelm February 25, 2005 Principles of gene regulation (cellular versus molecular level) Extracellular signals Chemical (e.g. hormones, growth factors) Environmental
Effect of Acetosyringone on Agrobacterium-mediated Transformation of Eustoma grandiflorum Leaf Disks
 JARQ 51 (4), 351-355 (2017) https://www.jircas.go.jp Improvement in Agrobacterium-mediated Transformation of Eustoma grandiflorum by Acetosyringone Effect of Acetosyringone on Agrobacterium-mediated Transformation
JARQ 51 (4), 351-355 (2017) https://www.jircas.go.jp Improvement in Agrobacterium-mediated Transformation of Eustoma grandiflorum by Acetosyringone Effect of Acetosyringone on Agrobacterium-mediated Transformation
Welcome to Class 21!
 Welcome to Class 21! Introductory Biochemistry! Lecture 21: Outline and Objectives l Regulation of Gene Expression in Prokaryotes! l transcriptional regulation! l principles! l lac operon! l trp attenuation!
Welcome to Class 21! Introductory Biochemistry! Lecture 21: Outline and Objectives l Regulation of Gene Expression in Prokaryotes! l transcriptional regulation! l principles! l lac operon! l trp attenuation!
DEVELOPMENTAL GENETICS OF ARABIDOPSIS THALIANA
 DEVELOPMENTAL GENETICS OF ARABIDOPSIS THALIANA CHASE BALLARD LINDA EAN HECTOR LOPEZ DR. JOANNA WERNER-FRACZEK IN COLLABORATION WITH DR. PATRICIA SPRINGER S LAB AT UCR AND ROBERT KOBLE PURPOSE OF RESEARCH
DEVELOPMENTAL GENETICS OF ARABIDOPSIS THALIANA CHASE BALLARD LINDA EAN HECTOR LOPEZ DR. JOANNA WERNER-FRACZEK IN COLLABORATION WITH DR. PATRICIA SPRINGER S LAB AT UCR AND ROBERT KOBLE PURPOSE OF RESEARCH
2. Yeast two-hybrid system
 2. Yeast two-hybrid system I. Process workflow a. Mating of haploid two-hybrid strains on YPD plates b. Replica-plating of diploids on selective plates c. Two-hydrid experiment plating on selective plates
2. Yeast two-hybrid system I. Process workflow a. Mating of haploid two-hybrid strains on YPD plates b. Replica-plating of diploids on selective plates c. Two-hydrid experiment plating on selective plates
EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA
 EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA Item Type text; Electronic Thesis Authors Bergstrand, Lauren Janel Publisher The University of Arizona. Rights Copyright is held by the author. Digital
EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA Item Type text; Electronic Thesis Authors Bergstrand, Lauren Janel Publisher The University of Arizona. Rights Copyright is held by the author. Digital
Control of Gene Expression in Prokaryotes
 Why? Control of Expression in Prokaryotes How do prokaryotes use operons to control gene expression? Houses usually have a light source in every room, but it would be a waste of energy to leave every light
Why? Control of Expression in Prokaryotes How do prokaryotes use operons to control gene expression? Houses usually have a light source in every room, but it would be a waste of energy to leave every light
Lecture 10: Cyclins, cyclin kinases and cell division
 Chem*3560 Lecture 10: Cyclins, cyclin kinases and cell division The eukaryotic cell cycle Actively growing mammalian cells divide roughly every 24 hours, and follow a precise sequence of events know as
Chem*3560 Lecture 10: Cyclins, cyclin kinases and cell division The eukaryotic cell cycle Actively growing mammalian cells divide roughly every 24 hours, and follow a precise sequence of events know as
10/4/2017. Chapter 39
 Chapter 39 1 Reception 1 Reception 2 Transduction CYTOPLASM CYTOPLASM Cell wall Plasma membrane Phytochrome activated by light Cell wall Plasma membrane Phytochrome activated by light cgmp Second messenger
Chapter 39 1 Reception 1 Reception 2 Transduction CYTOPLASM CYTOPLASM Cell wall Plasma membrane Phytochrome activated by light Cell wall Plasma membrane Phytochrome activated by light cgmp Second messenger
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/9/452/ra106/dc1 Supplementary Materials for Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots Hyo-Jun Lee, Jun-Ho
www.sciencesignaling.org/cgi/content/full/9/452/ra106/dc1 Supplementary Materials for Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots Hyo-Jun Lee, Jun-Ho
BIO1PS 2012 Plant Science Lecture 4 Hormones Pt. I
 BIO1PS 2012 Plant Science Lecture 4 Hormones Pt. I Dr. Michael Emmerling Department of Botany Room 410 m.emmerling@latrobe.edu.au Hormones and Ghost gum Eucalyptus papuana Coordination ~3 Lectures Leaves
BIO1PS 2012 Plant Science Lecture 4 Hormones Pt. I Dr. Michael Emmerling Department of Botany Room 410 m.emmerling@latrobe.edu.au Hormones and Ghost gum Eucalyptus papuana Coordination ~3 Lectures Leaves
Prokaryotic Regulation
 Prokaryotic Regulation Control of transcription initiation can be: Positive control increases transcription when activators bind DNA Negative control reduces transcription when repressors bind to DNA regulatory
Prokaryotic Regulation Control of transcription initiation can be: Positive control increases transcription when activators bind DNA Negative control reduces transcription when repressors bind to DNA regulatory
Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering
 Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering by Valverde et. Al Published in Science 2004 Presented by Boyana Grigorova CBMG 688R Feb. 12, 2007 Circadian Rhythms: The Clock Within
Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering by Valverde et. Al Published in Science 2004 Presented by Boyana Grigorova CBMG 688R Feb. 12, 2007 Circadian Rhythms: The Clock Within
CONTROL SYSTEMS IN PLANTS
 AP BIOLOGY PLANTS FORM & FUNCTION ACTIVITY #5 NAME DATE HOUR CONTROL SYSTEMS IN PLANTS HORMONES MECHANISM FOR HORMONE ACTION Plant Form and Function Activity #5 page 1 CONTROL OF CELL ELONGATION Plant
AP BIOLOGY PLANTS FORM & FUNCTION ACTIVITY #5 NAME DATE HOUR CONTROL SYSTEMS IN PLANTS HORMONES MECHANISM FOR HORMONE ACTION Plant Form and Function Activity #5 page 1 CONTROL OF CELL ELONGATION Plant
Supplemental Data. Wang et al. (2014). Plant Cell /tpc
 Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
Big Idea 3: Living systems store, retrieve, transmit and respond to information essential to life processes. Tuesday, December 27, 16
 Big Idea 3: Living systems store, retrieve, transmit and respond to information essential to life processes. Enduring understanding 3.B: Expression of genetic information involves cellular and molecular
Big Idea 3: Living systems store, retrieve, transmit and respond to information essential to life processes. Enduring understanding 3.B: Expression of genetic information involves cellular and molecular
AMADEPA Association Martiniquaise pour le Developpement des Plantes Alimentaires
 AMADEPA Association Martiniquaise pour le Developpement des Plantes Alimentaires 29eme CONGRES ANNUEL ANNUAL MEETING REUNION ANNUAL Agriculture Intensive dans les Iles de la Caraibe : enjeux, contraintes
AMADEPA Association Martiniquaise pour le Developpement des Plantes Alimentaires 29eme CONGRES ANNUEL ANNUAL MEETING REUNION ANNUAL Agriculture Intensive dans les Iles de la Caraibe : enjeux, contraintes
Last time: Obtaining information from a cloned gene
 Last time: Obtaining information from a cloned gene Objectives: 1. What is the biochemical role of the gene? 2. Where and when is the gene expressed (transcribed)? 3. Where and when is the protein made?
Last time: Obtaining information from a cloned gene Objectives: 1. What is the biochemical role of the gene? 2. Where and when is the gene expressed (transcribed)? 3. Where and when is the protein made?
Prokaryotic Gene Expression (Learning Objectives)
 Prokaryotic Gene Expression (Learning Objectives) 1. Learn how bacteria respond to changes of metabolites in their environment: short-term and longer-term. 2. Compare and contrast transcriptional control
Prokaryotic Gene Expression (Learning Objectives) 1. Learn how bacteria respond to changes of metabolites in their environment: short-term and longer-term. 2. Compare and contrast transcriptional control
Life Science Journal 2014;11(9) Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis
 Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis Sung-Il Kim 1, Sang Ik Song 3, Hak Soo Seo 1, 2, 4 * 1 Department of Plant Science and Research Institute of Agriculture
Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis Sung-Il Kim 1, Sang Ik Song 3, Hak Soo Seo 1, 2, 4 * 1 Department of Plant Science and Research Institute of Agriculture
Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport
 Ph.D. thesis Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport Zsigmond Laura Supervisor: Dr. Szabados László Arabidopsis Molecular Genetic Group Institute of Plant
Ph.D. thesis Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport Zsigmond Laura Supervisor: Dr. Szabados László Arabidopsis Molecular Genetic Group Institute of Plant
CONTROL OF GROWTH BY HORMONES
 CONTROL OF GROWTH BY HORMONES Growth and organogenesis are controlled......by genes (independent of environment): e.g., number of primary vascular bundles, general shape of a leaf or flower...by genes
CONTROL OF GROWTH BY HORMONES Growth and organogenesis are controlled......by genes (independent of environment): e.g., number of primary vascular bundles, general shape of a leaf or flower...by genes
The sucrose non-fermenting-1-related protein kinases SAPK1 and SAPK2 function collaboratively as positive regulators of salt stress tolerance in rice
 Lou et al. BMC Plant Biology (2018) 18:203 https://doi.org/10.1186/s12870-018-1408-0 RESEARCH ARTICLE Open Access The sucrose non-fermenting-1-related protein kinases SAPK1 and SAPK2 function collaboratively
Lou et al. BMC Plant Biology (2018) 18:203 https://doi.org/10.1186/s12870-018-1408-0 RESEARCH ARTICLE Open Access The sucrose non-fermenting-1-related protein kinases SAPK1 and SAPK2 function collaboratively
The Role of Inorganic Carbon Transport and Accumulation in the CO 2 -Concentrating Mechanism and CO 2 Assimilation in Chlamydomonas
 The Role of Inorganic Carbon Transport and Accumulation in the CO 2 -Concentrating Mechanism and CO 2 Assimilation in Chlamydomonas Is there a Role for the CCM in Increasing Biological CO 2 Capture? Generalized
The Role of Inorganic Carbon Transport and Accumulation in the CO 2 -Concentrating Mechanism and CO 2 Assimilation in Chlamydomonas Is there a Role for the CCM in Increasing Biological CO 2 Capture? Generalized
Simon Scofield, Walter Dewitte, and James AH Murray* School of Biosciences; Cardiff University; Cardiff, UK
 Short Communication Plant Signaling & Behavior 9, e28934; April; 2014 Landes Bioscience Short Communication STM sustains stem cell function in the Arabidopsis shoot apical meristem and controls KNOX gene
Short Communication Plant Signaling & Behavior 9, e28934; April; 2014 Landes Bioscience Short Communication STM sustains stem cell function in the Arabidopsis shoot apical meristem and controls KNOX gene
Chromosome variations in protoplast-derived calli and in plants regenerated from the calli of
 Jpn. J. Genet. (1989) 64, pp. 355-361 Chromosome variations in protoplast-derived calli and in plants regenerated from the calli of cultivated rice (Oryza sativa L.) Soryu NISHIBAYASHI*, Yasuyuki HAYASHI,
Jpn. J. Genet. (1989) 64, pp. 355-361 Chromosome variations in protoplast-derived calli and in plants regenerated from the calli of cultivated rice (Oryza sativa L.) Soryu NISHIBAYASHI*, Yasuyuki HAYASHI,
Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids
 Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids Plant growth conditions The soil was a 1:1 v/v mixture of loamy soil and organic compost. Initial soil water content was determined
Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids Plant growth conditions The soil was a 1:1 v/v mixture of loamy soil and organic compost. Initial soil water content was determined
Analysis of cytokinin-induced maize leaf developmental changes and interacting genetic modifiers
 Graduate Theses and Dissertations Iowa State University Capstones, Theses and Dissertations 2015 Analysis of cytokinin-induced maize leaf developmental changes and interacting genetic modifiers James Francis
Graduate Theses and Dissertations Iowa State University Capstones, Theses and Dissertations 2015 Analysis of cytokinin-induced maize leaf developmental changes and interacting genetic modifiers James Francis
Three types of RNA polymerase in eukaryotic nuclei
 Three types of RNA polymerase in eukaryotic nuclei Type Location RNA synthesized Effect of α-amanitin I Nucleolus Pre-rRNA for 18,.8 and 8S rrnas Insensitive II Nucleoplasm Pre-mRNA, some snrnas Sensitive
Three types of RNA polymerase in eukaryotic nuclei Type Location RNA synthesized Effect of α-amanitin I Nucleolus Pre-rRNA for 18,.8 and 8S rrnas Insensitive II Nucleoplasm Pre-mRNA, some snrnas Sensitive
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. HSP21 expression in 35S:HSP21 and hsp21 knockdown plants. (a) Since no T- DNA insertion line for HSP21 is available in the publicly available T-DNA collections,
Supplementary Figure 1 Supplementary Figure 1. HSP21 expression in 35S:HSP21 and hsp21 knockdown plants. (a) Since no T- DNA insertion line for HSP21 is available in the publicly available T-DNA collections,
16 CONTROL OF GENE EXPRESSION
 16 CONTROL OF GENE EXPRESSION Chapter Outline 16.1 REGULATION OF GENE EXPRESSION IN PROKARYOTES The operon is the unit of transcription in prokaryotes The lac operon for lactose metabolism is transcribed
16 CONTROL OF GENE EXPRESSION Chapter Outline 16.1 REGULATION OF GENE EXPRESSION IN PROKARYOTES The operon is the unit of transcription in prokaryotes The lac operon for lactose metabolism is transcribed
15.2 Prokaryotic Transcription *
 OpenStax-CNX module: m52697 1 15.2 Prokaryotic Transcription * Shannon McDermott Based on Prokaryotic Transcription by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons
OpenStax-CNX module: m52697 1 15.2 Prokaryotic Transcription * Shannon McDermott Based on Prokaryotic Transcription by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons
Plant Growth Regulators(NCERT)
 Plant Growth Regulators(NCERT) Promoters: 1. Auxins: -first isolated from urine, contains Zinc. -Natural: Indole Acetic Acid (IAA) Indole Butyric Acid (IBA) -Synthetic: Naphthalene Acetic Acid (NAA) 2-4
Plant Growth Regulators(NCERT) Promoters: 1. Auxins: -first isolated from urine, contains Zinc. -Natural: Indole Acetic Acid (IAA) Indole Butyric Acid (IBA) -Synthetic: Naphthalene Acetic Acid (NAA) 2-4
Prokaryotic Gene Expression (Learning Objectives)
 Prokaryotic Gene Expression (Learning Objectives) 1. Learn how bacteria respond to changes of metabolites in their environment: short-term and longer-term. 2. Compare and contrast transcriptional control
Prokaryotic Gene Expression (Learning Objectives) 1. Learn how bacteria respond to changes of metabolites in their environment: short-term and longer-term. 2. Compare and contrast transcriptional control
PLANT HORMONES-Introduction
 PLANT HORMONES-Introduction By convention hormone are said to be a substances whose site of synthesis and site of action are different; the two events are separated by space and time. Hormones are known
PLANT HORMONES-Introduction By convention hormone are said to be a substances whose site of synthesis and site of action are different; the two events are separated by space and time. Hormones are known
Development of genotype independent cotton transformation protocol
 Development of genotype independent cotton transformation protocol Shyam Barampuram and Sergei Krasnyanski Department of Horticultural Science North Carolina State University What are our goals? Robust
Development of genotype independent cotton transformation protocol Shyam Barampuram and Sergei Krasnyanski Department of Horticultural Science North Carolina State University What are our goals? Robust
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature10534 Supplementary Fig. 1. Diagrammatic representation of the N-end rule pathway of targeted proteolysis (after Graciet and Wellmer 2010 9 ). Tertiary, secondary
SUPPLEMENTARY INFORMATION doi:10.1038/nature10534 Supplementary Fig. 1. Diagrammatic representation of the N-end rule pathway of targeted proteolysis (after Graciet and Wellmer 2010 9 ). Tertiary, secondary
Bio 100 Guide 27.
 Bio 100 Guide 27 http://www.offthemarkcartoons.com/cartoons/1994-11-09.gif http://www.cneccc.edu.hk/subjects/bio/album/chapter20/images/plant_growth.jpg http://pgjennielove.files.wordpress.com/2008/06/apical_meristem.png
Bio 100 Guide 27 http://www.offthemarkcartoons.com/cartoons/1994-11-09.gif http://www.cneccc.edu.hk/subjects/bio/album/chapter20/images/plant_growth.jpg http://pgjennielove.files.wordpress.com/2008/06/apical_meristem.png
can affect division, elongation, & differentiation of cells to another region of plant where they have an effect
 Note that the following is a rudimentary outline of the class lecture; it does not contain everything discussed in class. Plant Hormones Plant Hormones compounds regulators growth or can affect division,
Note that the following is a rudimentary outline of the class lecture; it does not contain everything discussed in class. Plant Hormones Plant Hormones compounds regulators growth or can affect division,
Involvement of two-component signalling systems in the regulation of stomatal aperture by light in Arabidopsis thaliana
 Research Involvement of two-component signalling systems in the regulation of stomatal aperture by light in Arabidopsis thaliana Elodie Marchadier 1,2 and Alistair M. Hetherington 1 1 Department of Biological
Research Involvement of two-component signalling systems in the regulation of stomatal aperture by light in Arabidopsis thaliana Elodie Marchadier 1,2 and Alistair M. Hetherington 1 1 Department of Biological
Name: SBI 4U. Gene Expression Quiz. Overall Expectation:
 Gene Expression Quiz Overall Expectation: - Demonstrate an understanding of concepts related to molecular genetics, and how genetic modification is applied in industry and agriculture Specific Expectation(s):
Gene Expression Quiz Overall Expectation: - Demonstrate an understanding of concepts related to molecular genetics, and how genetic modification is applied in industry and agriculture Specific Expectation(s):
Complete all warm up questions Focus on operon functioning we will be creating operon models on Monday
 Complete all warm up questions Focus on operon functioning we will be creating operon models on Monday 1. What is the Central Dogma? 2. How does prokaryotic DNA compare to eukaryotic DNA? 3. How is DNA
Complete all warm up questions Focus on operon functioning we will be creating operon models on Monday 1. What is the Central Dogma? 2. How does prokaryotic DNA compare to eukaryotic DNA? 3. How is DNA
