Lecture 2 Grand Canonical Ensemble GCE
|
|
|
- Domenic Bruno Greene
- 5 years ago
- Views:
Transcription
1 Lecture 2 Grand Canoncal Ensemble GCE 2.1 hermodynamc Functons Contnung on from last day we also note that thus, dω = df dµ µd = Sd P dv dµ (2.1) P = V = S = From the expresson for the entropy, we therefore fnd E µ = Ω + S = Ω ( Ω But recall that,,µ V,µ (2.2) (2.3) (2.4) ) V,µ. whch therefore mples that, ( ) ln Ξ E µ = k 2 ( ) k ln Ξ = µ,v µ,v k ln Ξ Ω = k ln Ξ (2.5) or P V k = ln Ξ (2.6)
2 LECURE 2. GRAD CAOICAL ESEMBLE GCE Partcle Fluctuatons Mean-square devaton n ( ) 2 = 2 2 (2.7) On your assgnment you wll show that, ( ) 2 = k = z z ( ) 2 Ω = k 2 (2.8) and the relatve fluctuatons are then, = ( ) 1/2 k 2 Ω ( 2 ). (2.9) Ω ow the Grand Potental s an extensve quantty (recall that Ω Ω(, V, µ), but the chemcal potental s clearly an ntensve quantty, ( ) ( ) F G µ = = snce F and G are extensve. hus the relatve fluctuatons n partcle number must scale as: Gbbs-Duhem Equaton,P 1/2 1 1/2 We wll now do an alternatve dervaton of the above result 2.9 Start wth the Gbbs free energy, G = µ = µ More generally, for a multcomponent system, snce G s extensve we have, G(λ 1, λ 2,..., λ m,, P ) = λg( 1, 2,..., m,, P ) (2.10) where λ s an arbtrary scale factor.
3 LECURE 2. GRAD CAOICAL ESEMBLE GCE 8 Dfferentate 2.10 w.r.t. λ to fnd, L.H.S.: α λ α,p, j (α = λ ) = = = λ,p, j (forλ = 1),P, j µ R.H.S.: λg λ = G( 1,..., m,, P ) herefore, most generally for a multcomponent system G ( 1, 2,..., m,, P ) = ow take dfferental of the above expresson µ (2.11) dg = [µ d + dµ ] (2.12) But f G = G ( 1,..., m,, P ) then, dg = d + dp P, 1,..., m = Sd + V dp + + P, 1,..., m d,p µ d (2.13) Comparng 2.12 and 2.13 gves us the famous Gbbs-Duhem equaton: dµ = Sd + V dp (2.14) For a sngle component system, ths becomes: dµ = Sd + V dp (2.15)
4 LECURE 2. GRAD CAOICAL ESEMBLE GCE 9 At constant temperature, ths reduces to dµ = V dp dµ = V dp = vdp where v s the volume per molecule ( ) ( ) P = v = v ( P = v2 Invertng the above expresson gves us, = v 2 ) where κ s the Isothermal Compressblty. ( P = P (v, )) ( ) 2 = 2 2 = k κ v v κ (2.16) (2.17).B. ( ) 2 = 2 2 > 0, κ t > 0 Once agan, from 2.17 t s clear that the relatve fluctuatons n partcle number scale as, ( ) 1/2 k = κ 1! (2.18) v For a macroscopc system, where 10 11, the relatve fluctuatons are therefore extremely small. Pcture: P() <> where the probablty of fndng the system wth partcles s P () = z Q Ξ
5 LECURE 2. GRAD CAOICAL ESEMBLE GCE 10 Small fluctuatons means that P () should be sharply peaked around herefore, wth Ξ = =0 z Q, the sum wll be domnated by terms wth whch mples that, Ξ z Q whch agrees wth our earler result 2.6 k ln Ξ k ln z k ln Q = µ + F (, V, ) = P V hs demonstrates consstency between the Grand Canoncal and and Canoncal ensembles. 2.3 Comments about κ : Partcle fluctuatons here are exceptons when the scalng arguments 2.18 breakdown because the sothermal compressblty dverges and one obtans large partcle fluctuatons. hs can happen at a phase transton or when system s n a two-phase state (where system s a mxture of two phases wth dfferent v s, but exstng at the same pressure). For such cases, = 0
10.40 Appendix Connection to Thermodynamics and Derivation of Boltzmann Distribution
 10.40 Appendx Connecton to Thermodynamcs Dervaton of Boltzmann Dstrbuton Bernhardt L. Trout Outlne Cannoncal ensemble Maxmumtermmethod Most probable dstrbuton Ensembles contnued: Canoncal, Mcrocanoncal,
10.40 Appendx Connecton to Thermodynamcs Dervaton of Boltzmann Dstrbuton Bernhardt L. Trout Outlne Cannoncal ensemble Maxmumtermmethod Most probable dstrbuton Ensembles contnued: Canoncal, Mcrocanoncal,
Lecture. Polymer Thermodynamics 0331 L Chemical Potential
 Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
Introduction to Statistical Methods
 Introducton to Statstcal Methods Physcs 4362, Lecture #3 hermodynamcs Classcal Statstcal Knetc heory Classcal hermodynamcs Macroscopc approach General propertes of the system Macroscopc varables 1 hermodynamc
Introducton to Statstcal Methods Physcs 4362, Lecture #3 hermodynamcs Classcal Statstcal Knetc heory Classcal hermodynamcs Macroscopc approach General propertes of the system Macroscopc varables 1 hermodynamc
...Thermodynamics. If Clausius Clapeyron fails. l T (v 2 v 1 ) = 0/0 Second order phase transition ( S, v = 0)
 If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
Review of Classical Thermodynamics
 Revew of Classcal hermodynamcs Physcs 4362, Lecture #1, 2 Syllabus What s hermodynamcs? 1 [A law] s more mpressve the greater the smplcty of ts premses, the more dfferent are the knds of thngs t relates,
Revew of Classcal hermodynamcs Physcs 4362, Lecture #1, 2 Syllabus What s hermodynamcs? 1 [A law] s more mpressve the greater the smplcty of ts premses, the more dfferent are the knds of thngs t relates,
Thermodynamics and statistical mechanics in materials modelling II
 Course MP3 Lecture 8/11/006 (JAE) Course MP3 Lecture 8/11/006 Thermodynamcs and statstcal mechancs n materals modellng II A bref résumé of the physcal concepts used n materals modellng Dr James Ellott.1
Course MP3 Lecture 8/11/006 (JAE) Course MP3 Lecture 8/11/006 Thermodynamcs and statstcal mechancs n materals modellng II A bref résumé of the physcal concepts used n materals modellng Dr James Ellott.1
a for save as PDF Chemistry 163B Introduction to Multicomponent Systems and Partial Molar Quantities
 a for save as PDF Chemstry 163B Introducton to Multcomponent Systems and Partal Molar Quanttes 1 the problem of partal mmolar quanttes mx: 10 moles ethanol C 2 H 5 OH (580 ml) wth 1 mole water H 2 O (18
a for save as PDF Chemstry 163B Introducton to Multcomponent Systems and Partal Molar Quanttes 1 the problem of partal mmolar quanttes mx: 10 moles ethanol C 2 H 5 OH (580 ml) wth 1 mole water H 2 O (18
Poisson brackets and canonical transformations
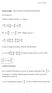 rof O B Wrght Mechancs Notes osson brackets and canoncal transformatons osson Brackets Consder an arbtrary functon f f ( qp t) df f f f q p q p t But q p p where ( qp ) pq q df f f f p q q p t In order
rof O B Wrght Mechancs Notes osson brackets and canoncal transformatons osson Brackets Consder an arbtrary functon f f ( qp t) df f f f q p q p t But q p p where ( qp ) pq q df f f f p q q p t In order
Statistical mechanics handout 4
 Statstcal mechancs handout 4 Explan dfference between phase space and an. Ensembles As dscussed n handout three atoms n any physcal system can adopt any one of a large number of mcorstates. For a quantum
Statstcal mechancs handout 4 Explan dfference between phase space and an. Ensembles As dscussed n handout three atoms n any physcal system can adopt any one of a large number of mcorstates. For a quantum
4.2 Chemical Driving Force
 4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
University of Washington Department of Chemistry Chemistry 453 Winter Quarter 2015
 Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
Open Systems: Chemical Potential and Partial Molar Quantities Chemical Potential
 Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Appendix II Summary of Important Equations
 W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
Chemistry 163B Free Energy and Equilibrium E&R ( ch 6)
 Chemstry 163B Free Energy and Equlbrum E&R ( ch 6) 1 ΔG reacton and equlbrum (frst pass) 1. ΔG < spontaneous ( natural, rreversble) ΔG = equlbrum (reversble) ΔG > spontaneous n reverse drecton. ΔG = ΔHΔS
Chemstry 163B Free Energy and Equlbrum E&R ( ch 6) 1 ΔG reacton and equlbrum (frst pass) 1. ΔG < spontaneous ( natural, rreversble) ΔG = equlbrum (reversble) ΔG > spontaneous n reverse drecton. ΔG = ΔHΔS
Supplementary Notes for Chapter 9 Mixture Thermodynamics
 Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Monte Carlo method II
 Course MP3 Lecture 5 14/11/2006 Monte Carlo method II How to put some real physcs nto the Monte Carlo method Dr James Ellott 5.1 Monte Carlo method revsted In lecture 4, we ntroduced the Monte Carlo (MC)
Course MP3 Lecture 5 14/11/2006 Monte Carlo method II How to put some real physcs nto the Monte Carlo method Dr James Ellott 5.1 Monte Carlo method revsted In lecture 4, we ntroduced the Monte Carlo (MC)
Fermi-Dirac statistics
 UCC/Physcs/MK/EM/October 8, 205 Fer-Drac statstcs Fer-Drac dstrbuton Matter partcles that are eleentary ostly have a type of angular oentu called spn. hese partcles are known to have a agnetc oent whch
UCC/Physcs/MK/EM/October 8, 205 Fer-Drac statstcs Fer-Drac dstrbuton Matter partcles that are eleentary ostly have a type of angular oentu called spn. hese partcles are known to have a agnetc oent whch
Solution Thermodynamics
 Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Physics 607 Exam 1. ( ) = 1, Γ( z +1) = zγ( z) x n e x2 dx = 1. e x2
 Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
NAME and Section No. it is found that 0.6 mol of O
 NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
Assignment 2. Tyler Shendruk February 19, 2010
 Assgnment yler Shendruk February 9, 00 Kadar Ch. Problem 8 We have an N N symmetrc matrx, M. he symmetry means M M and we ll say the elements of the matrx are m j. he elements are pulled from a probablty
Assgnment yler Shendruk February 9, 00 Kadar Ch. Problem 8 We have an N N symmetrc matrx, M. he symmetry means M M and we ll say the elements of the matrx are m j. he elements are pulled from a probablty
Chemical Equilibrium. Chapter 6 Spontaneity of Reactive Mixtures (gases) Taking into account there are many types of work that a sysem can perform
 Ths chapter deals wth chemcal reactons (system) wth lttle or no consderaton on the surroundngs. Chemcal Equlbrum Chapter 6 Spontanety of eactve Mxtures (gases) eactants generatng products would proceed
Ths chapter deals wth chemcal reactons (system) wth lttle or no consderaton on the surroundngs. Chemcal Equlbrum Chapter 6 Spontanety of eactve Mxtures (gases) eactants generatng products would proceed
2. Introduction to Thermodynamics
 . Introducton to hermodynamcs.a..b..c..d..e..f..g..h. Introductory Remarks State Varables and Exact Dfferentals Some Mechancal Equatons of State he Laws of hermodynamcs Fundamental Equaton of hermodynamcs
. Introducton to hermodynamcs.a..b..c..d..e..f..g..h. Introductory Remarks State Varables and Exact Dfferentals Some Mechancal Equatons of State he Laws of hermodynamcs Fundamental Equaton of hermodynamcs
Thermodynamics II. Department of Chemical Engineering. Prof. Kim, Jong Hak
 Thermodynamcs II Department of Chemcal Engneerng Prof. Km, Jong Hak Soluton Thermodynamcs : theory Obectve : lay the theoretcal foundaton for applcatons of thermodynamcs to gas mxture and lqud soluton
Thermodynamcs II Department of Chemcal Engneerng Prof. Km, Jong Hak Soluton Thermodynamcs : theory Obectve : lay the theoretcal foundaton for applcatons of thermodynamcs to gas mxture and lqud soluton
3. Be able to derive the chemical equilibrium constants from statistical mechanics.
 Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
NAME and Section No.
 Chemstry 391 Fall 2007 Exam I KEY (Monday September 17) 1. (25 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). Defne the terms: open system, closed system and solated system
Chemstry 391 Fall 2007 Exam I KEY (Monday September 17) 1. (25 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). Defne the terms: open system, closed system and solated system
Thermodynamics Second Law Entropy
 Thermodynamcs Second Law Entropy Lana Sherdan De Anza College May 8, 2018 Last tme the Boltzmann dstrbuton (dstrbuton of energes) the Maxwell-Boltzmann dstrbuton (dstrbuton of speeds) the Second Law of
Thermodynamcs Second Law Entropy Lana Sherdan De Anza College May 8, 2018 Last tme the Boltzmann dstrbuton (dstrbuton of energes) the Maxwell-Boltzmann dstrbuton (dstrbuton of speeds) the Second Law of
If two volatile and miscible liquids are combined to form a solution, Raoult s law is not obeyed. Use the experimental data in Table 9.
 9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
STATISTICAL MECHANICAL ENSEMBLES 1 MICROSCOPIC AND MACROSCOPIC VARIABLES PHASE SPACE ENSEMBLES. CHE 524 A. Panagiotopoulos 1
 CHE 54 A. Panagotopoulos STATSTCAL MECHACAL ESEMBLES MCROSCOPC AD MACROSCOPC ARABLES The central queston n Statstcal Mechancs can be phrased as follows: f partcles (atoms, molecules, electrons, nucle,
CHE 54 A. Panagotopoulos STATSTCAL MECHACAL ESEMBLES MCROSCOPC AD MACROSCOPC ARABLES The central queston n Statstcal Mechancs can be phrased as follows: f partcles (atoms, molecules, electrons, nucle,
V T for n & P = constant
 Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2014
 Lecture 12 7/25/14 ERD: 7.1-7.5 Devoe: 8.1.1-8.1.2, 8.2.1-8.2.3, 8.4.1-8.4.3 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 2014 A. Free Energy and Changes n Composton: The
Lecture 12 7/25/14 ERD: 7.1-7.5 Devoe: 8.1.1-8.1.2, 8.2.1-8.2.3, 8.4.1-8.4.3 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 2014 A. Free Energy and Changes n Composton: The
Solution Thermodynamics
 CH2351 Chemcal Engneerng Thermodynamcs II Unt I, II www.msubbu.n Soluton Thermodynamcs www.msubbu.n Dr. M. Subramanan Assocate Professor Department of Chemcal Engneerng Sr Svasubramanya Nadar College of
CH2351 Chemcal Engneerng Thermodynamcs II Unt I, II www.msubbu.n Soluton Thermodynamcs www.msubbu.n Dr. M. Subramanan Assocate Professor Department of Chemcal Engneerng Sr Svasubramanya Nadar College of
8.592J: Solutions for Assignment 7 Spring 2005
 8.59J: Solutons for Assgnment 7 Sprng 5 Problem 1 (a) A flament of length l can be created by addton of a monomer to one of length l 1 (at rate a) or removal of a monomer from a flament of length l + 1
8.59J: Solutons for Assgnment 7 Sprng 5 Problem 1 (a) A flament of length l can be created by addton of a monomer to one of length l 1 (at rate a) or removal of a monomer from a flament of length l + 1
PHYS 705: Classical Mechanics. Calculus of Variations II
 1 PHYS 705: Classcal Mechancs Calculus of Varatons II 2 Calculus of Varatons: Generalzaton (no constrant yet) Suppose now that F depends on several dependent varables : We need to fnd such that has a statonary
1 PHYS 705: Classcal Mechancs Calculus of Varatons II 2 Calculus of Varatons: Generalzaton (no constrant yet) Suppose now that F depends on several dependent varables : We need to fnd such that has a statonary
Non-Ideality Through Fugacity and Activity
 Non-Idealty Through Fugacty and Actvty S. Patel Deartment of Chemstry and Bochemstry, Unversty of Delaware, Newark, Delaware 19716, USA Corresondng author. E-mal: saatel@udel.edu 1 I. FUGACITY In ths dscusson,
Non-Idealty Through Fugacty and Actvty S. Patel Deartment of Chemstry and Bochemstry, Unversty of Delaware, Newark, Delaware 19716, USA Corresondng author. E-mal: saatel@udel.edu 1 I. FUGACITY In ths dscusson,
PHY688, Statistical Mechanics
 Department of Physcs & Astronomy 449 ESS Bldg. Stony Brook Unversty January 31, 2017 Nuclear Astrophyscs James.Lattmer@Stonybrook.edu Thermodynamcs Internal Energy Densty and Frst Law: ε = E V = Ts P +
Department of Physcs & Astronomy 449 ESS Bldg. Stony Brook Unversty January 31, 2017 Nuclear Astrophyscs James.Lattmer@Stonybrook.edu Thermodynamcs Internal Energy Densty and Frst Law: ε = E V = Ts P +
Statistical mechanics canonical ensemble
 canoncal ensemble system n thermal equlbrum wth bath of system probablty of mcro state T = k/ bath system p = 1 Z e E Z = e E average energy of system he = p E = what about? P E E e P e E = @ ln Z @ replcate
canoncal ensemble system n thermal equlbrum wth bath of system probablty of mcro state T = k/ bath system p = 1 Z e E Z = e E average energy of system he = p E = what about? P E E e P e E = @ ln Z @ replcate
PHYS 215C: Quantum Mechanics (Spring 2017) Problem Set 3 Solutions
 PHYS 5C: Quantum Mechancs Sprng 07 Problem Set 3 Solutons Prof. Matthew Fsher Solutons prepared by: Chatanya Murthy and James Sully June 4, 07 Please let me know f you encounter any typos n the solutons.
PHYS 5C: Quantum Mechancs Sprng 07 Problem Set 3 Solutons Prof. Matthew Fsher Solutons prepared by: Chatanya Murthy and James Sully June 4, 07 Please let me know f you encounter any typos n the solutons.
Lecture Notes 7: The Unruh Effect
 Quantum Feld Theory for Leg Spnners 17/1/11 Lecture Notes 7: The Unruh Effect Lecturer: Prakash Panangaden Scrbe: Shane Mansfeld 1 Defnng the Vacuum Recall from the last lecture that choosng a complex
Quantum Feld Theory for Leg Spnners 17/1/11 Lecture Notes 7: The Unruh Effect Lecturer: Prakash Panangaden Scrbe: Shane Mansfeld 1 Defnng the Vacuum Recall from the last lecture that choosng a complex
5.60 Thermodynamics & Kinetics Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
Introduction to Vapor/Liquid Equilibrium, part 2. Raoult s Law:
 CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CHEMICAL REACTIONS AND DIFFUSION
 CHEMICAL REACTIONS AND DIFFUSION A.K.A. NETWORK THERMODYNAMICS BACKGROUND Classcal thermodynamcs descrbes equlbrum states. Non-equlbrum thermodynamcs descrbes steady states. Network thermodynamcs descrbes
CHEMICAL REACTIONS AND DIFFUSION A.K.A. NETWORK THERMODYNAMICS BACKGROUND Classcal thermodynamcs descrbes equlbrum states. Non-equlbrum thermodynamcs descrbes steady states. Network thermodynamcs descrbes
10.34 Fall 2015 Metropolis Monte Carlo Algorithm
 10.34 Fall 2015 Metropols Monte Carlo Algorthm The Metropols Monte Carlo method s very useful for calculatng manydmensonal ntegraton. For e.g. n statstcal mechancs n order to calculate the prospertes of
10.34 Fall 2015 Metropols Monte Carlo Algorthm The Metropols Monte Carlo method s very useful for calculatng manydmensonal ntegraton. For e.g. n statstcal mechancs n order to calculate the prospertes of
y i x P vap 10 A T SOLUTION TO HOMEWORK #7 #Problem
 SOLUTION TO HOMEWORK #7 #roblem 1 10.1-1 a. In order to solve ths problem, we need to know what happens at the bubble pont; at ths pont, the frst bubble s formed, so we can assume that all of the number
SOLUTION TO HOMEWORK #7 #roblem 1 10.1-1 a. In order to solve ths problem, we need to know what happens at the bubble pont; at ths pont, the frst bubble s formed, so we can assume that all of the number
Lecture 4 Hypothesis Testing
 Lecture 4 Hypothess Testng We may wsh to test pror hypotheses about the coeffcents we estmate. We can use the estmates to test whether the data rejects our hypothess. An example mght be that we wsh to
Lecture 4 Hypothess Testng We may wsh to test pror hypotheses about the coeffcents we estmate. We can use the estmates to test whether the data rejects our hypothess. An example mght be that we wsh to
Rate of Absorption and Stimulated Emission
 MIT Department of Chemstry 5.74, Sprng 005: Introductory Quantum Mechancs II Instructor: Professor Andre Tokmakoff p. 81 Rate of Absorpton and Stmulated Emsson The rate of absorpton nduced by the feld
MIT Department of Chemstry 5.74, Sprng 005: Introductory Quantum Mechancs II Instructor: Professor Andre Tokmakoff p. 81 Rate of Absorpton and Stmulated Emsson The rate of absorpton nduced by the feld
x = , so that calculated
 Stat 4, secton Sngle Factor ANOVA notes by Tm Plachowsk n chapter 8 we conducted hypothess tests n whch we compared a sngle sample s mean or proporton to some hypotheszed value Chapter 9 expanded ths to
Stat 4, secton Sngle Factor ANOVA notes by Tm Plachowsk n chapter 8 we conducted hypothess tests n whch we compared a sngle sample s mean or proporton to some hypotheszed value Chapter 9 expanded ths to
In this section is given an overview of the common elasticity models.
 Secton 4.1 4.1 Elastc Solds In ths secton s gven an overvew of the common elastcty models. 4.1.1 The Lnear Elastc Sold The classcal Lnear Elastc model, or Hooean model, has the followng lnear relatonshp
Secton 4.1 4.1 Elastc Solds In ths secton s gven an overvew of the common elastcty models. 4.1.1 The Lnear Elastc Sold The classcal Lnear Elastc model, or Hooean model, has the followng lnear relatonshp
Electrochemical Equilibrium Electromotive Force
 CHM465/865, 24-3, Lecture 5-7, 2 th Sep., 24 lectrochemcal qulbrum lectromotve Force Relaton between chemcal and electrc drvng forces lectrochemcal system at constant T and p: consder Gbbs free energy
CHM465/865, 24-3, Lecture 5-7, 2 th Sep., 24 lectrochemcal qulbrum lectromotve Force Relaton between chemcal and electrc drvng forces lectrochemcal system at constant T and p: consder Gbbs free energy
STATISTICAL MECHANICS
 STATISTICAL MECHANICS Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of
STATISTICAL MECHANICS Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of
PHYS 705: Classical Mechanics. Newtonian Mechanics
 1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
COS 511: Theoretical Machine Learning. Lecturer: Rob Schapire Lecture # 15 Scribe: Jieming Mao April 1, 2013
 COS 511: heoretcal Machne Learnng Lecturer: Rob Schapre Lecture # 15 Scrbe: Jemng Mao Aprl 1, 013 1 Bref revew 1.1 Learnng wth expert advce Last tme, we started to talk about learnng wth expert advce.
COS 511: heoretcal Machne Learnng Lecturer: Rob Schapre Lecture # 15 Scrbe: Jemng Mao Aprl 1, 013 1 Bref revew 1.1 Learnng wth expert advce Last tme, we started to talk about learnng wth expert advce.
Lecture 7: Boltzmann distribution & Thermodynamics of mixing
 Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
TP A SOLUTION. For an ideal monatomic gas U=3/2nRT, Since the process is at constant pressure Q = C. giving ) =1000/(5/2*8.31*10)
 T A SOLUTION For an deal monatomc gas U/nRT, Snce the process s at constant pressure Q C pn T gvng a: n Q /( 5 / R T ) /(5/*8.*) C V / R and C / R + R 5 / R. U U / nr T (/ ) R T ( Q / 5 / R T ) Q / 5 Q
T A SOLUTION For an deal monatomc gas U/nRT, Snce the process s at constant pressure Q C pn T gvng a: n Q /( 5 / R T ) /(5/*8.*) C V / R and C / R + R 5 / R. U U / nr T (/ ) R T ( Q / 5 / R T ) Q / 5 Q
EPR Paradox and the Physical Meaning of an Experiment in Quantum Mechanics. Vesselin C. Noninski
 EPR Paradox and the Physcal Meanng of an Experment n Quantum Mechancs Vesseln C Nonnsk vesselnnonnsk@verzonnet Abstract It s shown that there s one purely determnstc outcome when measurement s made on
EPR Paradox and the Physcal Meanng of an Experment n Quantum Mechancs Vesseln C Nonnsk vesselnnonnsk@verzonnet Abstract It s shown that there s one purely determnstc outcome when measurement s made on
THERMAL DISTRIBUTION IN THE HCL SPECTRUM OBJECTIVE
 ame: THERMAL DISTRIBUTIO I THE HCL SPECTRUM OBJECTIVE To nvestgate a system s thermal dstrbuton n dscrete states; specfcally, determne HCl gas temperature from the relatve occupatons of ts rotatonal states.
ame: THERMAL DISTRIBUTIO I THE HCL SPECTRUM OBJECTIVE To nvestgate a system s thermal dstrbuton n dscrete states; specfcally, determne HCl gas temperature from the relatve occupatons of ts rotatonal states.
Osmotic pressure and protein binding
 Osmotc pressure and proten bndng Igor R. Kuznetsov, KochLab Symposum talk 5/15/09 Today we take a closer look at one of the soluton thermodynamcs key ponts from Steve s presentaton. Here t s: d[ln(k off
Osmotc pressure and proten bndng Igor R. Kuznetsov, KochLab Symposum talk 5/15/09 Today we take a closer look at one of the soluton thermodynamcs key ponts from Steve s presentaton. Here t s: d[ln(k off
A quote of the week (or camel of the week): There is no expedience to which a man will not go to avoid the labor of thinking. Thomas A.
 A quote of the week (or camel of the week): here s no expedence to whch a man wll not go to avod the labor of thnkng. homas A. Edson Hess law. Algorthm S Select a reacton, possbly contanng specfc compounds
A quote of the week (or camel of the week): here s no expedence to whch a man wll not go to avod the labor of thnkng. homas A. Edson Hess law. Algorthm S Select a reacton, possbly contanng specfc compounds
Assignment 4. Adsorption Isotherms
 Insttute of Process Engneerng Assgnment 4. Adsorpton Isotherms Part A: Compettve adsorpton of methane and ethane In large scale adsorpton processes, more than one compound from a mxture of gases get adsorbed,
Insttute of Process Engneerng Assgnment 4. Adsorpton Isotherms Part A: Compettve adsorpton of methane and ethane In large scale adsorpton processes, more than one compound from a mxture of gases get adsorbed,
Thermodynamics General
 Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Math 426: Probability MWF 1pm, Gasson 310 Homework 4 Selected Solutions
 Exercses from Ross, 3, : Math 26: Probablty MWF pm, Gasson 30 Homework Selected Solutons 3, p. 05 Problems 76, 86 3, p. 06 Theoretcal exercses 3, 6, p. 63 Problems 5, 0, 20, p. 69 Theoretcal exercses 2,
Exercses from Ross, 3, : Math 26: Probablty MWF pm, Gasson 30 Homework Selected Solutons 3, p. 05 Problems 76, 86 3, p. 06 Theoretcal exercses 3, 6, p. 63 Problems 5, 0, 20, p. 69 Theoretcal exercses 2,
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2014
 Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
5.62 Physical Chemistry II Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Lecture #8: Boltzmann, Ferm-Drac,
MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Lecture #8: Boltzmann, Ferm-Drac,
Chapter 18, Part 1. Fundamentals of Atmospheric Modeling
 Overhead Sldes for Chapter 18, Part 1 of Fundamentals of Atmospherc Modelng by Mark Z. Jacobson Department of Cvl & Envronmental Engneerng Stanford Unversty Stanford, CA 94305-4020 January 30, 2002 Types
Overhead Sldes for Chapter 18, Part 1 of Fundamentals of Atmospherc Modelng by Mark Z. Jacobson Department of Cvl & Envronmental Engneerng Stanford Unversty Stanford, CA 94305-4020 January 30, 2002 Types
Physics 607 Exam 2. ( ) = 1, Γ( z +1) = zγ( z) x n e x2 dx = 1. e x2
 Physics 607 Exam Please be well-organized, and show all significant steps clearly in all problems. You are graded on your work, so please do not just write down answers with no explanation! Do all your
Physics 607 Exam Please be well-organized, and show all significant steps clearly in all problems. You are graded on your work, so please do not just write down answers with no explanation! Do all your
8. Superfluid to Mott-insulator transition
 8. Superflud to Mott-nsulator transton Overvew Optcal lattce potentals Soluton of the Schrödnger equaton for perodc potentals Band structure Bloch oscllaton of bosonc and fermonc atoms n optcal lattces
8. Superflud to Mott-nsulator transton Overvew Optcal lattce potentals Soluton of the Schrödnger equaton for perodc potentals Band structure Bloch oscllaton of bosonc and fermonc atoms n optcal lattces
PETE 310 Lectures # 24 & 25 Chapter 12 Gas Liquid Equilibrium
 ETE 30 Lectures # 24 & 25 Chapter 2 Gas Lqud Equlbrum Thermal Equlbrum Object A hgh T, Object B low T Intal contact tme Intermedate tme. Later tme Mechancal Equlbrum ressure essels Vale Closed Vale Open
ETE 30 Lectures # 24 & 25 Chapter 2 Gas Lqud Equlbrum Thermal Equlbrum Object A hgh T, Object B low T Intal contact tme Intermedate tme. Later tme Mechancal Equlbrum ressure essels Vale Closed Vale Open
Q e E i /k B. i i i i
 Water and Aqueous Solutons 3. Lattce Model of a Flud Lattce Models Lattce models provde a mnmalst, or coarse-graned, framework for descrbng the translatonal, rotatonal, and conformatonal degrees of freedom
Water and Aqueous Solutons 3. Lattce Model of a Flud Lattce Models Lattce models provde a mnmalst, or coarse-graned, framework for descrbng the translatonal, rotatonal, and conformatonal degrees of freedom
DECOUPLING THEORY HW2
 8.8 DECOUPLIG THEORY HW2 DOGHAO WAG DATE:OCT. 3 207 Problem We shall start by reformulatng the problem. Denote by δ S n the delta functon that s evenly dstrbuted at the n ) dmensonal unt sphere. As a temporal
8.8 DECOUPLIG THEORY HW2 DOGHAO WAG DATE:OCT. 3 207 Problem We shall start by reformulatng the problem. Denote by δ S n the delta functon that s evenly dstrbuted at the n ) dmensonal unt sphere. As a temporal
THE ARIMOTO-BLAHUT ALGORITHM FOR COMPUTATION OF CHANNEL CAPACITY. William A. Pearlman. References: S. Arimoto - IEEE Trans. Inform. Thy., Jan.
 THE ARIMOTO-BLAHUT ALGORITHM FOR COMPUTATION OF CHANNEL CAPACITY Wllam A. Pearlman 2002 References: S. Armoto - IEEE Trans. Inform. Thy., Jan. 1972 R. Blahut - IEEE Trans. Inform. Thy., July 1972 Recall
THE ARIMOTO-BLAHUT ALGORITHM FOR COMPUTATION OF CHANNEL CAPACITY Wllam A. Pearlman 2002 References: S. Armoto - IEEE Trans. Inform. Thy., Jan. 1972 R. Blahut - IEEE Trans. Inform. Thy., July 1972 Recall
Econ107 Applied Econometrics Topic 3: Classical Model (Studenmund, Chapter 4)
 I. Classcal Assumptons Econ7 Appled Econometrcs Topc 3: Classcal Model (Studenmund, Chapter 4) We have defned OLS and studed some algebrac propertes of OLS. In ths topc we wll study statstcal propertes
I. Classcal Assumptons Econ7 Appled Econometrcs Topc 3: Classcal Model (Studenmund, Chapter 4) We have defned OLS and studed some algebrac propertes of OLS. In ths topc we wll study statstcal propertes
Hidden Markov Models & The Multivariate Gaussian (10/26/04)
 CS281A/Stat241A: Statstcal Learnng Theory Hdden Markov Models & The Multvarate Gaussan (10/26/04) Lecturer: Mchael I. Jordan Scrbes: Jonathan W. Hu 1 Hdden Markov Models As a bref revew, hdden Markov models
CS281A/Stat241A: Statstcal Learnng Theory Hdden Markov Models & The Multvarate Gaussan (10/26/04) Lecturer: Mchael I. Jordan Scrbes: Jonathan W. Hu 1 Hdden Markov Models As a bref revew, hdden Markov models
For now, let us focus on a specific model of neurons. These are simplified from reality but can achieve remarkable results.
 Neural Networks : Dervaton compled by Alvn Wan from Professor Jtendra Malk s lecture Ths type of computaton s called deep learnng and s the most popular method for many problems, such as computer vson
Neural Networks : Dervaton compled by Alvn Wan from Professor Jtendra Malk s lecture Ths type of computaton s called deep learnng and s the most popular method for many problems, such as computer vson
Causal Diamonds. M. Aghili, L. Bombelli, B. Pilgrim
 Causal Damonds M. Aghl, L. Bombell, B. Plgrm Introducton The correcton to volume of a causal nterval due to curvature of spacetme has been done by Myrhem [] and recently by Gbbons & Solodukhn [] and later
Causal Damonds M. Aghl, L. Bombell, B. Plgrm Introducton The correcton to volume of a causal nterval due to curvature of spacetme has been done by Myrhem [] and recently by Gbbons & Solodukhn [] and later
1 Matrix representations of canonical matrices
 1 Matrx representatons of canoncal matrces 2-d rotaton around the orgn: ( ) cos θ sn θ R 0 = sn θ cos θ 3-d rotaton around the x-axs: R x = 1 0 0 0 cos θ sn θ 0 sn θ cos θ 3-d rotaton around the y-axs:
1 Matrx representatons of canoncal matrces 2-d rotaton around the orgn: ( ) cos θ sn θ R 0 = sn θ cos θ 3-d rotaton around the x-axs: R x = 1 0 0 0 cos θ sn θ 0 sn θ cos θ 3-d rotaton around the y-axs:
Physics 5153 Classical Mechanics. Principle of Virtual Work-1
 P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
Envr 210, Chapter 3, Intermolecular forces and partitioning Free energies and equilibrium partitioning chemical potential fugacity activity coef.
 Envr 20, Chapter 3, Intermolecular forces and parttonng Free energes and equlbrum parttonng chemcal potental fugacty actvty coef. phase transfer- actvty coef and fugactes more on free energes and equlbrum
Envr 20, Chapter 3, Intermolecular forces and parttonng Free energes and equlbrum parttonng chemcal potental fugacty actvty coef. phase transfer- actvty coef and fugactes more on free energes and equlbrum
Canonical transformations
 Canoncal transformatons November 23, 2014 Recall that we have defned a symplectc transformaton to be any lnear transformaton M A B leavng the symplectc form nvarant, Ω AB M A CM B DΩ CD Coordnate transformatons,
Canoncal transformatons November 23, 2014 Recall that we have defned a symplectc transformaton to be any lnear transformaton M A B leavng the symplectc form nvarant, Ω AB M A CM B DΩ CD Coordnate transformatons,
8.6 The Complex Number System
 8.6 The Complex Number System Earler n the chapter, we mentoned that we cannot have a negatve under a square root, snce the square of any postve or negatve number s always postve. In ths secton we want
8.6 The Complex Number System Earler n the chapter, we mentoned that we cannot have a negatve under a square root, snce the square of any postve or negatve number s always postve. In ths secton we want
Lecture 3: Boltzmann distribution
 Lecture 3: Boltzmann dstrbuton Statstcal mechancs: concepts Ams: Dervaton of Boltzmann dstrbuton: Basc postulate of statstcal mechancs. All states equally lkely. Equal a-pror probablty: Statstcal vew of
Lecture 3: Boltzmann dstrbuton Statstcal mechancs: concepts Ams: Dervaton of Boltzmann dstrbuton: Basc postulate of statstcal mechancs. All states equally lkely. Equal a-pror probablty: Statstcal vew of
Economics 130. Lecture 4 Simple Linear Regression Continued
 Economcs 130 Lecture 4 Contnued Readngs for Week 4 Text, Chapter and 3. We contnue wth addressng our second ssue + add n how we evaluate these relatonshps: Where do we get data to do ths analyss? How do
Economcs 130 Lecture 4 Contnued Readngs for Week 4 Text, Chapter and 3. We contnue wth addressng our second ssue + add n how we evaluate these relatonshps: Where do we get data to do ths analyss? How do
NMT EE 589 & UNM ME 482/582 ROBOT ENGINEERING. Dr. Stephen Bruder NMT EE 589 & UNM ME 482/582
 NMT EE 589 & UNM ME 482/582 ROBOT ENGINEERING Dr. Stephen Bruder NMT EE 589 & UNM ME 482/582 4. Moton Knematcs 4.2 Angular Velocty Knematcs Summary From the last lecture we concluded that: If the jonts
NMT EE 589 & UNM ME 482/582 ROBOT ENGINEERING Dr. Stephen Bruder NMT EE 589 & UNM ME 482/582 4. Moton Knematcs 4.2 Angular Velocty Knematcs Summary From the last lecture we concluded that: If the jonts
Name: SID: Discussion Session:
 Name: SID: Dscusson Sesson: Chemcal Engneerng Thermodynamcs 141 -- Fall 007 Thursday, November 15, 007 Mdterm II SOLUTIONS - 70 mnutes 110 Ponts Total Closed Book and Notes (0 ponts) 1. Evaluate whether
Name: SID: Dscusson Sesson: Chemcal Engneerng Thermodynamcs 141 -- Fall 007 Thursday, November 15, 007 Mdterm II SOLUTIONS - 70 mnutes 110 Ponts Total Closed Book and Notes (0 ponts) 1. Evaluate whether
Spring 2002 Lecture #13
 44-50 Sprng 00 ecture # Dr. Jaehoon Yu. Rotatonal Energy. Computaton of oments of nerta. Parallel-as Theorem 4. Torque & Angular Acceleraton 5. Work, Power, & Energy of Rotatonal otons Remember the md-term
44-50 Sprng 00 ecture # Dr. Jaehoon Yu. Rotatonal Energy. Computaton of oments of nerta. Parallel-as Theorem 4. Torque & Angular Acceleraton 5. Work, Power, & Energy of Rotatonal otons Remember the md-term
between standard Gibbs free energies of formation for products and reactants, ΔG! R = ν i ΔG f,i, we
 hermodynamcs, Statstcal hermodynamcs, and Knetcs 4 th Edton,. Engel & P. ed Ch. 6 Part Answers to Selected Problems Q6.. Q6.4. If ξ =0. mole at equlbrum, the reacton s not ery far along. hus, there would
hermodynamcs, Statstcal hermodynamcs, and Knetcs 4 th Edton,. Engel & P. ed Ch. 6 Part Answers to Selected Problems Q6.. Q6.4. If ξ =0. mole at equlbrum, the reacton s not ery far along. hus, there would
10. Canonical Transformations Michael Fowler
 10. Canoncal Transformatons Mchael Fowler Pont Transformatons It s clear that Lagrange s equatons are correct for any reasonable choce of parameters labelng the system confguraton. Let s call our frst
10. Canoncal Transformatons Mchael Fowler Pont Transformatons It s clear that Lagrange s equatons are correct for any reasonable choce of parameters labelng the system confguraton. Let s call our frst
The Feynman path integral
 The Feynman path ntegral Aprl 3, 205 Hesenberg and Schrödnger pctures The Schrödnger wave functon places the tme dependence of a physcal system n the state, ψ, t, where the state s a vector n Hlbert space
The Feynman path ntegral Aprl 3, 205 Hesenberg and Schrödnger pctures The Schrödnger wave functon places the tme dependence of a physcal system n the state, ψ, t, where the state s a vector n Hlbert space
763622S ADVANCED QUANTUM MECHANICS Solution Set 1 Spring c n a n. c n 2 = 1.
 7636S ADVANCED QUANTUM MECHANICS Soluton Set 1 Sprng 013 1 Warm-up Show that the egenvalues of a Hermtan operator  are real and that the egenkets correspondng to dfferent egenvalues are orthogonal (b)
7636S ADVANCED QUANTUM MECHANICS Soluton Set 1 Sprng 013 1 Warm-up Show that the egenvalues of a Hermtan operator  are real and that the egenkets correspondng to dfferent egenvalues are orthogonal (b)
NMT EE 589 & UNM ME 482/582 ROBOT ENGINEERING. Dr. Stephen Bruder NMT EE 589 & UNM ME 482/582
 NMT EE 589 & UNM ME 48/58 ROBOT ENGINEERING Dr. Stephen Bruder NMT EE 589 & UNM ME 48/58 7. Robot Dynamcs 7.5 The Equatons of Moton Gven that we wsh to fnd the path q(t (n jont space) whch mnmzes the energy
NMT EE 589 & UNM ME 48/58 ROBOT ENGINEERING Dr. Stephen Bruder NMT EE 589 & UNM ME 48/58 7. Robot Dynamcs 7.5 The Equatons of Moton Gven that we wsh to fnd the path q(t (n jont space) whch mnmzes the energy
Lecture 4. Instructor: Haipeng Luo
 Lecture 4 Instructor: Hapeng Luo In the followng lectures, we focus on the expert problem and study more adaptve algorthms. Although Hedge s proven to be worst-case optmal, one may wonder how well t would
Lecture 4 Instructor: Hapeng Luo In the followng lectures, we focus on the expert problem and study more adaptve algorthms. Although Hedge s proven to be worst-case optmal, one may wonder how well t would
AN EXTENDED CLASS OF TIME-CONTINUOUS BRANCHING PROCESSES. Rong-Rong Chen. ( University of Illinois at Urbana-Champaign)
 AN EXTENDED CLASS OF TIME-CONTINUOUS BRANCHING PROCESSES Rong-Rong Chen ( Unversty of Illnos at Urbana-Champagn Abstract. Ths paper s devoted to studyng an extended class of tme-contnuous branchng processes,
AN EXTENDED CLASS OF TIME-CONTINUOUS BRANCHING PROCESSES Rong-Rong Chen ( Unversty of Illnos at Urbana-Champagn Abstract. Ths paper s devoted to studyng an extended class of tme-contnuous branchng processes,
Limited Dependent Variables
 Lmted Dependent Varables. What f the left-hand sde varable s not a contnuous thng spread from mnus nfnty to plus nfnty? That s, gven a model = f (, β, ε, where a. s bounded below at zero, such as wages
Lmted Dependent Varables. What f the left-hand sde varable s not a contnuous thng spread from mnus nfnty to plus nfnty? That s, gven a model = f (, β, ε, where a. s bounded below at zero, such as wages
Lectures - Week 4 Matrix norms, Conditioning, Vector Spaces, Linear Independence, Spanning sets and Basis, Null space and Range of a Matrix
 Lectures - Week 4 Matrx norms, Condtonng, Vector Spaces, Lnear Independence, Spannng sets and Bass, Null space and Range of a Matrx Matrx Norms Now we turn to assocatng a number to each matrx. We could
Lectures - Week 4 Matrx norms, Condtonng, Vector Spaces, Lnear Independence, Spannng sets and Bass, Null space and Range of a Matrx Matrx Norms Now we turn to assocatng a number to each matrx. We could
Lecture 4. Macrostates and Microstates (Ch. 2 )
 Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
Physics 181. Particle Systems
 Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
π e ax2 dx = x 2 e ax2 dx or x 3 e ax2 dx = 1 x 4 e ax2 dx = 3 π 8a 5/2 (a) We are considering the Maxwell velocity distribution function: 2πτ/m
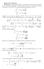 Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
MASSACHUSETTS INSTITUTE OF TECHNOLOGY 6.265/15.070J Fall 2013 Lecture 12 10/21/2013. Martingale Concentration Inequalities and Applications
 MASSACHUSETTS INSTITUTE OF TECHNOLOGY 6.65/15.070J Fall 013 Lecture 1 10/1/013 Martngale Concentraton Inequaltes and Applcatons Content. 1. Exponental concentraton for martngales wth bounded ncrements.
MASSACHUSETTS INSTITUTE OF TECHNOLOGY 6.65/15.070J Fall 013 Lecture 1 10/1/013 Martngale Concentraton Inequaltes and Applcatons Content. 1. Exponental concentraton for martngales wth bounded ncrements.
Module 1 : The equation of continuity. Lecture 1: Equation of Continuity
 1 Module 1 : The equaton of contnuty Lecture 1: Equaton of Contnuty 2 Advanced Heat and Mass Transfer: Modules 1. THE EQUATION OF CONTINUITY : Lectures 1-6 () () () (v) (v) Overall Mass Balance Momentum
1 Module 1 : The equaton of contnuty Lecture 1: Equaton of Contnuty 2 Advanced Heat and Mass Transfer: Modules 1. THE EQUATION OF CONTINUITY : Lectures 1-6 () () () (v) (v) Overall Mass Balance Momentum
Physics 240: Worksheet 30 Name:
 (1) One mole of an deal monatomc gas doubles ts temperature and doubles ts volume. What s the change n entropy of the gas? () 1 kg of ce at 0 0 C melts to become water at 0 0 C. What s the change n entropy
(1) One mole of an deal monatomc gas doubles ts temperature and doubles ts volume. What s the change n entropy of the gas? () 1 kg of ce at 0 0 C melts to become water at 0 0 C. What s the change n entropy
Mechanics Physics 151
 Mechancs Physcs 5 Lecture 0 Canoncal Transformatons (Chapter 9) What We Dd Last Tme Hamlton s Prncple n the Hamltonan formalsm Dervaton was smple δi δ p H(, p, t) = 0 Adonal end-pont constrants δ t ( )
Mechancs Physcs 5 Lecture 0 Canoncal Transformatons (Chapter 9) What We Dd Last Tme Hamlton s Prncple n the Hamltonan formalsm Dervaton was smple δi δ p H(, p, t) = 0 Adonal end-pont constrants δ t ( )
