Physics 240: Worksheet 30 Name:
|
|
|
- Douglas Morton
- 6 years ago
- Views:
Transcription
1 (1) One mole of an deal monatomc gas doubles ts temperature and doubles ts volume. What s the change n entropy of the gas? () 1 kg of ce at 0 0 C melts to become water at 0 0 C. What s the change n entropy of the ce? (3) Suppose you mx 1 kg of water at 0 0 C wth 1 kg of water at C. What s the change n entropy for ths mxng process? (4) You have just made your frst mllon. It also happens that the world has a terrble energy crunch at ths tme, and effcent engne development s all the rage. An nventor approaches you and suggests that you nvest n hs engne whch works between 30 0 C and C because t has an effcency of 70%. Do you nvest? Why or Why not? (5) Show that: (a) U s a functon of temperature only (b) C v =3/R and the other features of deal gases.
2 Entropy and the Carnot Cycle There are many ways to talk about entropy, rangng from nformaton theory, languages, computer scence to physcs. We ll here concentrate on the physcs formulaton, specfcally pertanng to thermodynamcs. We frst need to defne the state functon Entropy (whch s a measure of effcency of heat flow) s gven by: Q S= T Strctly sad, ths apples only to a small amount of heat added to a system at a constant temperature. In general, as temperature changes over a range, you would have to add up ndvdual changes as: S= Q T steps Snce entropy s a state functon, the change n entropy wll be zero for any closed reversble path. If the path s not reversble, then the change s greater than zero. I ll later show you examples of ths. f = dq T Calculus verson: S, where dq ndcates a small amount of Q. I ll later show you how to calculate some entropes. Now, t s mportant to realze that the area enclosed by a closed T-S plot represents work. Let s assume a small closed cyclc change occurs n a system at a temperature T. A small amount of heat wll be added to the system. From the frst law and the defnton of entropy, we then have = = = Q U+ W W S T T T U s zero because the cycle s closed. We thus have: T S = W ( ) It s not much harder to show ths n other ways but I thnk I ve shown my pont.
3 Calculaton of entropes for dfferent processes When we calculate the entropy changes for an deal gas, t s useful to use the frst law: U= Q W Q= U+ W Here, we have n general: U= nc T so long as we re talkng about an deal gas. ( ) We also have that the work s gven by: W= P = nrt Thus, f we wanted to calculate Q, we have: Q= nc T + nrt As t stands, ths can t be calculated n general for any process (because you don t know how T vares when t multples the last term). However, dvdng by T gves: Q T T= nc( T) + nr( ) You can now use the useful approxmaton: Tf f S= nc ln + nrln T Calculus verson: dq= nc dt + nrt d so, = nc + nr dq dt d T T and S = nc + nr dq dt d T T Thus Tf f S= nc ln + nrln T
4 completely ndependently of the process that the gas went though, so long as the process s reversble. Ths s nce although you probably want to work out specfc examples for the dfferent processes. Now, as promsed, I ll use entropy to show the Carnot cycle to be the most effcent cycle possble. How? Remember that for an adabatc process, Q=0 whch, n ths case, wll gve a vertcal lne on a TS plot. For an sothermal process, we end up needng to connect the two ends of these lnes whch then gves us a square for the Carnot cycle. It looks lke the one I ve shown before. Let s see where Q h and Q c become nvolved: What you want to do to make the most effcent engne possble s to maxmze the area (green stuff) for a gven length of red (and you must have Q c present). The shape that gves ths s the rectangle whch s the Carnot cycle. At least that s my smplfed verson of why the Carnot cycle s the most effcent cycle possble. Heat Engne dagram Heat engnes work lke the dagram shows. Another statement of the second law of thermo s you have got to reject some heat to a cold reservor (you can t convert heat drectly nto work wthout losng some heat n the process). It s perhaps less obvous why ths s true but I lke the analogy of a battery: the cold reservor s the termnal, the + termnal s the hot reservor and a lght bulb s connected between the two. Both
5 termnals must be connected n order for the bulb to lght. Here, f you do work you have extracted some heat from the gas that dd work and thus, you automatcally have a colder gas left or somethng lke that. It s also mpossble to have energy go from cold to hot wthout dong work. (ths s the mpossble refrgerator). Interestngly enough, although we don t have the tme to explore ths completely, another statement of the second law of thermodynamcs s that the entropy of the unverse ncreases n all processes. I ll show you ths n the context of mxng two fluds. (1) One mole of an deal monatomc gas doubles ts temperature and doubles ts volume. What s the change n entropy of the gas? Soluton: For a monatomc gas, C v =3/R. Thus, Tf f S= nc ln + nrln T ( ) S= 1 Rln + 1Rln = Rln = T 5 J T K Notce that I don t need to talk about the path (so long as t s reversble) to answer ths queston. () 1 kg of ce at 0 0 C melts to become water at 0 0 C. What s the change n entropy of the ce? Soluton: Start wth: Q S= But f you use T=0 0 C, you re just about as wrong as you can hope to be. You must work n K here. The soluton? ml f 333. x10 J S = = = K (3) Suppose you mx 1 kg of water at 0 0 C wth 1 kg of water at C. What s the change n entropy for ths mxng process? Tm ( ) ( ) 4186 S= + = mc + = ln + ln = Q1 Q T1 T T T T T T T T T ( ) ( ) 4186 ln + ln = = (4) You have just made your frst mllon. It also happens that the world has a terrble energy crunch at ths tme, and effcent engne development s all the rage. An nventor approaches you and suggests that you nvest n hs engne whch works between 30 0 C and C because t has an effcency of 70%. Do you nvest? Why or Why not? c H J K
6 Soluton: If you calculate ε = = = Tc 30 c % T H 100 and then nvest, you ve just lost your frst mllon to a scam artst. If you calculate: 303 ε c = 1 373= = % and then don t nvest, you re just seen the value of my physcs course. You wll probably want to be sure to recommend that ths guy vst your competton. Note: each year when I use ths problem, t gets closer and closer to beng true!
7 (5) Show that: (a) U s a functon of temperature only (b) C v =3/R and the other features of deal gases. The knetc theory of an deal gas envsons pont partcles bouncng off of a wall and not nteractng wth each other. The force exerted by one molecule on the wall s gven from Newton s law as: p F= t The tme between successve collsons for a box of length L s: L t= for a one dmensonal box. The change n momentum s gven by: p= mv x If we permt what I refer to as the great le to preval, we ll use that t and p n the force equaton to get: mvx F= 1 L / = L mv x x In general, the partcle has equal amounts of veloctes n the x, y and z drectons. Thus, 1 v = v = v = v x y z where v s the magntude of the velocty of the molecule. We can thus fnd that for a 3-d box, 1 F= 3 L mv Snce we re nterested n pressure, and snce the wall of nterest has an area LxL, we fnd: P F 1 mv 1 = = = mv 3 L 3 L 3 Multply by the volume to get: 1 P= mv 1 = mv = K.E. x 3 ( ) We n general have N atoms present to gve addtons as: P N K.E. Compare ths to the IDG: = 3 N K.E. = NkT T= 3 3 Ths means that the temperature and the average knetc energy of an atom are connected as shown. But, the knetc energy s ndeed the nternal energy of the deal K.E. k
8 gas so thus, the nternal energy of an deal gas depends only upon temperature. (N s constant). We can wrte ths as: U= N K.E. and thus, we fnd the exact functonal dependence: U NkT How does U change wth temperature? In terms of moles: so, Thus, we have the result: = 3 U T = 3 Nk U Nk = nr = 3 T nr = nc v C R = 3 Notce the 3.. t s very suggestve. Indeed, there are 3 degrees of freedom and we have 3 (1/R) contrbutons to C v. Suppose that we have a datomc molecule. The rotatonal modes have 3 degrees of freedom but only of these modes are capable of storng energy for a true pont-partcle system. Ths would add: / R to C v. If the molecule can also vbrate, we have addtonal consderatons. There are ways the molecule can vbrate and ths adds an addtonal / R to C v. Thus, we have potentally: 3 C = R+ R+ R+ v translatonal rotatonal vbratonal But some of the modes wll freeze out at low temperatures. Thus, we have the concluson: ½ R s added to C v for each degree of freedom whch s realzed n a system. Here s an mportant quote regardng the vbratonal contrbuton (From Modern Physcs for Scentsts and Engneers, 3 rd. Edton (006) (Brooks/Cole Publshers), page 301. How many degrees of freedom does ths add? One may be tempted to say just 1 one, because of the potental energy κ( r r) 0, where κ s the sprng s force constant, r the separaton between atoms, and r 0 the equlbrum separaton between the atoms. But another degree of freedom s assocated wth the dr vbratonal velocty ( ) m. 1 dr dt, because the vbratonal knetc energy s ( dt)
#64. ΔS for Isothermal Mixing of Ideal Gases
 #64 Carnot Heat Engne ΔS for Isothermal Mxng of Ideal Gases ds = S dt + S T V V S = P V T T V PV = nrt, P T ds = v T = nr V dv V nr V V = nrln V V = - nrln V V ΔS ΔS ΔS for Isothermal Mxng for Ideal Gases
#64 Carnot Heat Engne ΔS for Isothermal Mxng of Ideal Gases ds = S dt + S T V V S = P V T T V PV = nrt, P T ds = v T = nr V dv V nr V V = nrln V V = - nrln V V ΔS ΔS ΔS for Isothermal Mxng for Ideal Gases
STATISTICAL MECHANICS
 STATISTICAL MECHANICS Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of
STATISTICAL MECHANICS Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of
Thermodynamics Second Law Entropy
 Thermodynamcs Second Law Entropy Lana Sherdan De Anza College May 8, 2018 Last tme the Boltzmann dstrbuton (dstrbuton of energes) the Maxwell-Boltzmann dstrbuton (dstrbuton of speeds) the Second Law of
Thermodynamcs Second Law Entropy Lana Sherdan De Anza College May 8, 2018 Last tme the Boltzmann dstrbuton (dstrbuton of energes) the Maxwell-Boltzmann dstrbuton (dstrbuton of speeds) the Second Law of
Physics 207: Lecture 20. Today s Agenda Homework for Monday
 Physcs 207: Lecture 20 Today s Agenda Homework for Monday Recap: Systems of Partcles Center of mass Velocty and acceleraton of the center of mass Dynamcs of the center of mass Lnear Momentum Example problems
Physcs 207: Lecture 20 Today s Agenda Homework for Monday Recap: Systems of Partcles Center of mass Velocty and acceleraton of the center of mass Dynamcs of the center of mass Lnear Momentum Example problems
Physics 141. Lecture 14. Frank L. H. Wolfs Department of Physics and Astronomy, University of Rochester, Lecture 14, Page 1
 Physcs 141. Lecture 14. Frank L. H. Wolfs Department of Physcs and Astronomy, Unversty of Rochester, Lecture 14, Page 1 Physcs 141. Lecture 14. Course Informaton: Lab report # 3. Exam # 2. Mult-Partcle
Physcs 141. Lecture 14. Frank L. H. Wolfs Department of Physcs and Astronomy, Unversty of Rochester, Lecture 14, Page 1 Physcs 141. Lecture 14. Course Informaton: Lab report # 3. Exam # 2. Mult-Partcle
Homework Chapter 21 Solutions!!
 Homework Chapter 1 Solutons 1.7 1.13 1.17 1.19 1.6 1.33 1.45 1.51 1.71 page 1 Problem 1.7 A mole sample of oxygen gas s confned to a 5 lter vessel at a pressure of 8 atm. Fnd the average translatonal knetc
Homework Chapter 1 Solutons 1.7 1.13 1.17 1.19 1.6 1.33 1.45 1.51 1.71 page 1 Problem 1.7 A mole sample of oxygen gas s confned to a 5 lter vessel at a pressure of 8 atm. Fnd the average translatonal knetc
Outline. Unit Eight Calculations with Entropy. The Second Law. Second Law Notes. Uses of Entropy. Entropy is a Property.
 Unt Eght Calculatons wth Entropy Mechancal Engneerng 370 Thermodynamcs Larry Caretto October 6, 010 Outlne Quz Seven Solutons Second law revew Goals for unt eght Usng entropy to calculate the maxmum work
Unt Eght Calculatons wth Entropy Mechancal Engneerng 370 Thermodynamcs Larry Caretto October 6, 010 Outlne Quz Seven Solutons Second law revew Goals for unt eght Usng entropy to calculate the maxmum work
Chapter 21 - The Kinetic Theory of Gases
 hapter 1 - he Knetc heory o Gases 1. Δv 8.sn 4. 8.sn 4. m s F Nm. 1 kg.94 N Δ t. s F A 1.7 N m 1.7 a N mv 1.6 Use the equaton descrbng the knetc-theory account or pressure:. hen mv Kav where N nna NA N
hapter 1 - he Knetc heory o Gases 1. Δv 8.sn 4. 8.sn 4. m s F Nm. 1 kg.94 N Δ t. s F A 1.7 N m 1.7 a N mv 1.6 Use the equaton descrbng the knetc-theory account or pressure:. hen mv Kav where N nna NA N
Thermodynamics and Gases
 hermodynamcs and Gases Last tme Knetc heory o Gases or smple (monatomc) gases Atomc nature o matter Demonstrate deal gas law Atomc knetc energy nternal energy Mean ree path and velocty dstrbutons From
hermodynamcs and Gases Last tme Knetc heory o Gases or smple (monatomc) gases Atomc nature o matter Demonstrate deal gas law Atomc knetc energy nternal energy Mean ree path and velocty dstrbutons From
A quote of the week (or camel of the week): There is no expedience to which a man will not go to avoid the labor of thinking. Thomas A.
 A quote of the week (or camel of the week): here s no expedence to whch a man wll not go to avod the labor of thnkng. homas A. Edson Hess law. Algorthm S Select a reacton, possbly contanng specfc compounds
A quote of the week (or camel of the week): here s no expedence to whch a man wll not go to avod the labor of thnkng. homas A. Edson Hess law. Algorthm S Select a reacton, possbly contanng specfc compounds
Review of Classical Thermodynamics
 Revew of Classcal hermodynamcs Physcs 4362, Lecture #1, 2 Syllabus What s hermodynamcs? 1 [A law] s more mpressve the greater the smplcty of ts premses, the more dfferent are the knds of thngs t relates,
Revew of Classcal hermodynamcs Physcs 4362, Lecture #1, 2 Syllabus What s hermodynamcs? 1 [A law] s more mpressve the greater the smplcty of ts premses, the more dfferent are the knds of thngs t relates,
Thermodynamics General
 Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
PHYS 705: Classical Mechanics. Newtonian Mechanics
 1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
Introduction to Statistical Methods
 Introducton to Statstcal Methods Physcs 4362, Lecture #3 hermodynamcs Classcal Statstcal Knetc heory Classcal hermodynamcs Macroscopc approach General propertes of the system Macroscopc varables 1 hermodynamc
Introducton to Statstcal Methods Physcs 4362, Lecture #3 hermodynamcs Classcal Statstcal Knetc heory Classcal hermodynamcs Macroscopc approach General propertes of the system Macroscopc varables 1 hermodynamc
and Statistical Mechanics Material Properties
 Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
NAME and Section No.
 Chemstry 391 Fall 2007 Exam I KEY (Monday September 17) 1. (25 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). Defne the terms: open system, closed system and solated system
Chemstry 391 Fall 2007 Exam I KEY (Monday September 17) 1. (25 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). Defne the terms: open system, closed system and solated system
Lecture 3 Examples and Problems
 Lecture 3 Examles and Problems Mechancs & thermodynamcs Equartton Frst Law of Thermodynamcs Ideal gases Isothermal and adabatc rocesses Readng: Elements Ch. 1-3 Lecture 3, 1 Wllam Thomson (1824 1907) a.k.a.
Lecture 3 Examles and Problems Mechancs & thermodynamcs Equartton Frst Law of Thermodynamcs Ideal gases Isothermal and adabatc rocesses Readng: Elements Ch. 1-3 Lecture 3, 1 Wllam Thomson (1824 1907) a.k.a.
Chapter 5 rd Law of Thermodynamics
 Entropy and the nd and 3 rd Chapter 5 rd Law o hermodynamcs homas Engel, hlp Red Objectves Introduce entropy. Derve the condtons or spontanety. Show how S vares wth the macroscopc varables,, and. Chapter
Entropy and the nd and 3 rd Chapter 5 rd Law o hermodynamcs homas Engel, hlp Red Objectves Introduce entropy. Derve the condtons or spontanety. Show how S vares wth the macroscopc varables,, and. Chapter
A particle in a state of uniform motion remain in that state of motion unless acted upon by external force.
 The fundamental prncples of classcal mechancs were lad down by Galleo and Newton n the 16th and 17th centures. In 1686, Newton wrote the Prncpa where he gave us three laws of moton, one law of gravty,
The fundamental prncples of classcal mechancs were lad down by Galleo and Newton n the 16th and 17th centures. In 1686, Newton wrote the Prncpa where he gave us three laws of moton, one law of gravty,
General Formulas applicable to ALL processes in an Ideal Gas:
 Calormetrc calculatons: dq mcd or dq ncd ( specc heat) Q ml ( latent heat) General Formulas applcable to ALL processes n an Ideal Gas: P nr du dq dw dw Pd du nc d C R ( monoatomc) C C R P Specc Processes:
Calormetrc calculatons: dq mcd or dq ncd ( specc heat) Q ml ( latent heat) General Formulas applcable to ALL processes n an Ideal Gas: P nr du dq dw dw Pd du nc d C R ( monoatomc) C C R P Specc Processes:
Physics 5153 Classical Mechanics. D Alembert s Principle and The Lagrangian-1
 P. Guterrez Physcs 5153 Classcal Mechancs D Alembert s Prncple and The Lagrangan 1 Introducton The prncple of vrtual work provdes a method of solvng problems of statc equlbrum wthout havng to consder the
P. Guterrez Physcs 5153 Classcal Mechancs D Alembert s Prncple and The Lagrangan 1 Introducton The prncple of vrtual work provdes a method of solvng problems of statc equlbrum wthout havng to consder the
Introduction to Vapor/Liquid Equilibrium, part 2. Raoult s Law:
 CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
Week 8: Chapter 9. Linear Momentum. Newton Law and Momentum. Linear Momentum, cont. Conservation of Linear Momentum. Conservation of Momentum, 2
 Lnear omentum Week 8: Chapter 9 Lnear omentum and Collsons The lnear momentum of a partcle, or an object that can be modeled as a partcle, of mass m movng wth a velocty v s defned to be the product of
Lnear omentum Week 8: Chapter 9 Lnear omentum and Collsons The lnear momentum of a partcle, or an object that can be modeled as a partcle, of mass m movng wth a velocty v s defned to be the product of
Chapter 7. Potential Energy and Conservation of Energy
 Chapter 7 Potental Energy and Conservaton o Energy 1 Forms o Energy There are many orms o energy, but they can all be put nto two categores Knetc Knetc energy s energy o moton Potental Potental energy
Chapter 7 Potental Energy and Conservaton o Energy 1 Forms o Energy There are many orms o energy, but they can all be put nto two categores Knetc Knetc energy s energy o moton Potental Potental energy
University of Washington Department of Chemistry Chemistry 453 Winter Quarter 2015
 Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
Physics 115. Molecular motion and temperature Phase equilibrium, evaporation
 Physcs 115 General Physcs II Sesson 9 Molecular moton and temperature Phase equlbrum, evaporaton R. J. Wlkes Emal: phy115a@u.washngton.edu Home page: http://courses.washngton.edu/phy115a/ 4/14/14 Physcs
Physcs 115 General Physcs II Sesson 9 Molecular moton and temperature Phase equlbrum, evaporaton R. J. Wlkes Emal: phy115a@u.washngton.edu Home page: http://courses.washngton.edu/phy115a/ 4/14/14 Physcs
Temperature. Chapter Heat Engine
 Chapter 3 Temperature In prevous chapters of these notes we ntroduced the Prncple of Maxmum ntropy as a technque for estmatng probablty dstrbutons consstent wth constrants. In Chapter 9 we dscussed the
Chapter 3 Temperature In prevous chapters of these notes we ntroduced the Prncple of Maxmum ntropy as a technque for estmatng probablty dstrbutons consstent wth constrants. In Chapter 9 we dscussed the
Lecture 16. Chapter 11. Energy Dissipation Linear Momentum. Physics I. Department of Physics and Applied Physics
 Lecture 16 Chapter 11 Physcs I Energy Dsspaton Lnear Momentum Course webste: http://aculty.uml.edu/andry_danylov/teachng/physcsi Department o Physcs and Appled Physcs IN IN THIS CHAPTER, you wll learn
Lecture 16 Chapter 11 Physcs I Energy Dsspaton Lnear Momentum Course webste: http://aculty.uml.edu/andry_danylov/teachng/physcsi Department o Physcs and Appled Physcs IN IN THIS CHAPTER, you wll learn
Problem Set #6 solution, Chem 340, Fall 2013 Due Friday, Oct 11, 2013 Please show all work for credit
 Problem Set #6 soluton, Chem 340, Fall 2013 Due Frday, Oct 11, 2013 Please show all work for credt To hand n: Atkns Chap 3 Exercses: 3.3(b), 3.8(b), 3.13(b), 3.15(b) Problems: 3.1, 3.12, 3.36, 3.43 Engel
Problem Set #6 soluton, Chem 340, Fall 2013 Due Frday, Oct 11, 2013 Please show all work for credt To hand n: Atkns Chap 3 Exercses: 3.3(b), 3.8(b), 3.13(b), 3.15(b) Problems: 3.1, 3.12, 3.36, 3.43 Engel
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructions
 THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
Linear Momentum. Center of Mass.
 Lecture 6 Chapter 9 Physcs I 03.3.04 Lnear omentum. Center of ass. Course webste: http://faculty.uml.edu/ndry_danylov/teachng/physcsi Lecture Capture: http://echo360.uml.edu/danylov03/physcssprng.html
Lecture 6 Chapter 9 Physcs I 03.3.04 Lnear omentum. Center of ass. Course webste: http://faculty.uml.edu/ndry_danylov/teachng/physcsi Lecture Capture: http://echo360.uml.edu/danylov03/physcssprng.html
Physics 207 Lecture 27
 hyscs 07 Lecture 7 hyscs 07, Lecture 7, Dec. 6 Agenda: h. 0, st Law o Thermodynamcs, h. st Law o thermodynamcs ( U Q + W du dq + dw ) Work done by an deal gas n a ston Introducton to thermodynamc cycles
hyscs 07 Lecture 7 hyscs 07, Lecture 7, Dec. 6 Agenda: h. 0, st Law o Thermodynamcs, h. st Law o thermodynamcs ( U Q + W du dq + dw ) Work done by an deal gas n a ston Introducton to thermodynamc cycles
5.60 Thermodynamics & Kinetics Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
Physics 114 Exam 2 Spring Name:
 Physcs 114 Exam Sprng 013 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem Answer each of the followng questons. Ponts for each queston are ndcated n red wth the amount beng
Physcs 114 Exam Sprng 013 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem Answer each of the followng questons. Ponts for each queston are ndcated n red wth the amount beng
Physics 114 Exam 2 Fall 2014 Solutions. Name:
 Physcs 114 Exam Fall 014 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem Answer each of the followng questons. Ponts for each queston are ndcated n red. Unless otherwse ndcated,
Physcs 114 Exam Fall 014 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem Answer each of the followng questons. Ponts for each queston are ndcated n red. Unless otherwse ndcated,
Conservation of Angular Momentum = "Spin"
 Page 1 of 6 Conservaton of Angular Momentum = "Spn" We can assgn a drecton to the angular velocty: drecton of = drecton of axs + rght hand rule (wth rght hand, curl fngers n drecton of rotaton, thumb ponts
Page 1 of 6 Conservaton of Angular Momentum = "Spn" We can assgn a drecton to the angular velocty: drecton of = drecton of axs + rght hand rule (wth rght hand, curl fngers n drecton of rotaton, thumb ponts
Lecture 20: Noether s Theorem
 Lecture 20: Noether s Theorem In our revew of Newtonan Mechancs, we were remnded that some quanttes (energy, lnear momentum, and angular momentum) are conserved That s, they are constant f no external
Lecture 20: Noether s Theorem In our revew of Newtonan Mechancs, we were remnded that some quanttes (energy, lnear momentum, and angular momentum) are conserved That s, they are constant f no external
Chapter 3 and Chapter 4
 Chapter 3 and Chapter 4 Chapter 3 Energy 3. Introducton:Work Work W s energy transerred to or rom an object by means o a orce actng on the object. Energy transerred to the object s postve work, and energy
Chapter 3 and Chapter 4 Chapter 3 Energy 3. Introducton:Work Work W s energy transerred to or rom an object by means o a orce actng on the object. Energy transerred to the object s postve work, and energy
G4023 Mid-Term Exam #1 Solutions
 Exam1Solutons.nb 1 G03 Md-Term Exam #1 Solutons 1-Oct-0, 1:10 p.m to :5 p.m n 1 Pupn Ths exam s open-book, open-notes. You may also use prnt-outs of the homework solutons and a calculator. 1 (30 ponts,
Exam1Solutons.nb 1 G03 Md-Term Exam #1 Solutons 1-Oct-0, 1:10 p.m to :5 p.m n 1 Pupn Ths exam s open-book, open-notes. You may also use prnt-outs of the homework solutons and a calculator. 1 (30 ponts,
Physics 5153 Classical Mechanics. Principle of Virtual Work-1
 P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
THERMAL DISTRIBUTION IN THE HCL SPECTRUM OBJECTIVE
 ame: THERMAL DISTRIBUTIO I THE HCL SPECTRUM OBJECTIVE To nvestgate a system s thermal dstrbuton n dscrete states; specfcally, determne HCl gas temperature from the relatve occupatons of ts rotatonal states.
ame: THERMAL DISTRIBUTIO I THE HCL SPECTRUM OBJECTIVE To nvestgate a system s thermal dstrbuton n dscrete states; specfcally, determne HCl gas temperature from the relatve occupatons of ts rotatonal states.
Isothermal vs. adiabatic compression
 Isothermal vs. adabatc comresson 1. One and a half moles of a datomc gas at temerature 5 C are comressed sothermally from a volume of 0.015 m to a volume of 0.0015 m. a. Sketch the rocess on a dagram and
Isothermal vs. adabatc comresson 1. One and a half moles of a datomc gas at temerature 5 C are comressed sothermally from a volume of 0.015 m to a volume of 0.0015 m. a. Sketch the rocess on a dagram and
8.1 Arc Length. What is the length of a curve? How can we approximate it? We could do it following the pattern we ve used before
 .1 Arc Length hat s the length of a curve? How can we approxmate t? e could do t followng the pattern we ve used before Use a sequence of ncreasngly short segments to approxmate the curve: As the segments
.1 Arc Length hat s the length of a curve? How can we approxmate t? e could do t followng the pattern we ve used before Use a sequence of ncreasngly short segments to approxmate the curve: As the segments
Lagrange Multipliers. A Somewhat Silly Example. Monday, 25 September 2013
 Lagrange Multplers Monday, 5 September 013 Sometmes t s convenent to use redundant coordnates, and to effect the varaton of the acton consstent wth the constrants va the method of Lagrange undetermned
Lagrange Multplers Monday, 5 September 013 Sometmes t s convenent to use redundant coordnates, and to effect the varaton of the acton consstent wth the constrants va the method of Lagrange undetermned
1.3 Hence, calculate a formula for the force required to break the bond (i.e. the maximum value of F)
 EN40: Dynacs and Vbratons Hoework 4: Work, Energy and Lnear Moentu Due Frday March 6 th School of Engneerng Brown Unversty 1. The Rydberg potental s a sple odel of atoc nteractons. It specfes the potental
EN40: Dynacs and Vbratons Hoework 4: Work, Energy and Lnear Moentu Due Frday March 6 th School of Engneerng Brown Unversty 1. The Rydberg potental s a sple odel of atoc nteractons. It specfes the potental
Week 11: Chapter 11. The Vector Product. The Vector Product Defined. The Vector Product and Torque. More About the Vector Product
 The Vector Product Week 11: Chapter 11 Angular Momentum There are nstances where the product of two vectors s another vector Earler we saw where the product of two vectors was a scalar Ths was called the
The Vector Product Week 11: Chapter 11 Angular Momentum There are nstances where the product of two vectors s another vector Earler we saw where the product of two vectors was a scalar Ths was called the
Problem Points Score Total 100
 Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
Chapter 11: Angular Momentum
 Chapter 11: ngular Momentum Statc Equlbrum In Chap. 4 we studed the equlbrum of pontobjects (mass m) wth the applcaton of Newton s aws F 0 F x y, 0 Therefore, no lnear (translatonal) acceleraton, a0 For
Chapter 11: ngular Momentum Statc Equlbrum In Chap. 4 we studed the equlbrum of pontobjects (mass m) wth the applcaton of Newton s aws F 0 F x y, 0 Therefore, no lnear (translatonal) acceleraton, a0 For
...Thermodynamics. If Clausius Clapeyron fails. l T (v 2 v 1 ) = 0/0 Second order phase transition ( S, v = 0)
 If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
Study Guide For Exam Two
 Study Gude For Exam Two Physcs 2210 Albretsen Updated: 08/02/2018 All Other Prevous Study Gudes Modules 01-06 Module 07 Work Work done by a constant force F over a dstance s : Work done by varyng force
Study Gude For Exam Two Physcs 2210 Albretsen Updated: 08/02/2018 All Other Prevous Study Gudes Modules 01-06 Module 07 Work Work done by a constant force F over a dstance s : Work done by varyng force
EPR Paradox and the Physical Meaning of an Experiment in Quantum Mechanics. Vesselin C. Noninski
 EPR Paradox and the Physcal Meanng of an Experment n Quantum Mechancs Vesseln C Nonnsk vesselnnonnsk@verzonnet Abstract It s shown that there s one purely determnstc outcome when measurement s made on
EPR Paradox and the Physcal Meanng of an Experment n Quantum Mechancs Vesseln C Nonnsk vesselnnonnsk@verzonnet Abstract It s shown that there s one purely determnstc outcome when measurement s made on
Moments of Inertia. and reminds us of the analogous equation for linear momentum p= mv, which is of the form. The kinetic energy of the body is.
 Moments of Inerta Suppose a body s movng on a crcular path wth constant speed Let s consder two quanttes: the body s angular momentum L about the center of the crcle, and ts knetc energy T How are these
Moments of Inerta Suppose a body s movng on a crcular path wth constant speed Let s consder two quanttes: the body s angular momentum L about the center of the crcle, and ts knetc energy T How are these
At zero K: All atoms frozen at fixed positions on a periodic lattice.
 September, 00 Readng: Chapter Four Homework: None Entropy and The Degree of Dsorder: Consder a sold crystallne materal: At zero K: All atoms frozen at fxed postons on a perodc lattce. Add heat to a fnte
September, 00 Readng: Chapter Four Homework: None Entropy and The Degree of Dsorder: Consder a sold crystallne materal: At zero K: All atoms frozen at fxed postons on a perodc lattce. Add heat to a fnte
So far: simple (planar) geometries
 Physcs 06 ecture 5 Torque and Angular Momentum as Vectors SJ 7thEd.: Chap. to 3 Rotatonal quanttes as vectors Cross product Torque epressed as a vector Angular momentum defned Angular momentum as a vector
Physcs 06 ecture 5 Torque and Angular Momentum as Vectors SJ 7thEd.: Chap. to 3 Rotatonal quanttes as vectors Cross product Torque epressed as a vector Angular momentum defned Angular momentum as a vector
FREQUENCY DISTRIBUTIONS Page 1 of The idea of a frequency distribution for sets of observations will be introduced,
 FREQUENCY DISTRIBUTIONS Page 1 of 6 I. Introducton 1. The dea of a frequency dstrbuton for sets of observatons wll be ntroduced, together wth some of the mechancs for constructng dstrbutons of data. Then
FREQUENCY DISTRIBUTIONS Page 1 of 6 I. Introducton 1. The dea of a frequency dstrbuton for sets of observatons wll be ntroduced, together wth some of the mechancs for constructng dstrbutons of data. Then
Lectures - Week 4 Matrix norms, Conditioning, Vector Spaces, Linear Independence, Spanning sets and Basis, Null space and Range of a Matrix
 Lectures - Week 4 Matrx norms, Condtonng, Vector Spaces, Lnear Independence, Spannng sets and Bass, Null space and Range of a Matrx Matrx Norms Now we turn to assocatng a number to each matrx. We could
Lectures - Week 4 Matrx norms, Condtonng, Vector Spaces, Lnear Independence, Spannng sets and Bass, Null space and Range of a Matrx Matrx Norms Now we turn to assocatng a number to each matrx. We could
NAME and Section No. it is found that 0.6 mol of O
 NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
Open Systems: Chemical Potential and Partial Molar Quantities Chemical Potential
 Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
π e ax2 dx = x 2 e ax2 dx or x 3 e ax2 dx = 1 x 4 e ax2 dx = 3 π 8a 5/2 (a) We are considering the Maxwell velocity distribution function: 2πτ/m
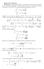 Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Molecular structure: Diatomic molecules in the rigid rotor and harmonic oscillator approximations Notes on Quantum Mechanics
 Molecular structure: Datomc molecules n the rgd rotor and harmonc oscllator approxmatons Notes on Quantum Mechancs http://quantum.bu.edu/notes/quantummechancs/molecularstructuredatomc.pdf Last updated
Molecular structure: Datomc molecules n the rgd rotor and harmonc oscllator approxmatons Notes on Quantum Mechancs http://quantum.bu.edu/notes/quantummechancs/molecularstructuredatomc.pdf Last updated
5.62 Physical Chemistry II Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Sprng 2008 Lecture 34 Page Transton
MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Sprng 2008 Lecture 34 Page Transton
Physics 53. Rotational Motion 3. Sir, I have found you an argument, but I am not obliged to find you an understanding.
 Physcs 53 Rotatonal Moton 3 Sr, I have found you an argument, but I am not oblged to fnd you an understandng. Samuel Johnson Angular momentum Wth respect to rotatonal moton of a body, moment of nerta plays
Physcs 53 Rotatonal Moton 3 Sr, I have found you an argument, but I am not oblged to fnd you an understandng. Samuel Johnson Angular momentum Wth respect to rotatonal moton of a body, moment of nerta plays
Chapter Twelve. Integration. We now turn our attention to the idea of an integral in dimensions higher than one. Consider a real-valued function f : D
 Chapter Twelve Integraton 12.1 Introducton We now turn our attenton to the dea of an ntegral n dmensons hgher than one. Consder a real-valued functon f : R, where the doman s a nce closed subset of Eucldean
Chapter Twelve Integraton 12.1 Introducton We now turn our attenton to the dea of an ntegral n dmensons hgher than one. Consder a real-valued functon f : R, where the doman s a nce closed subset of Eucldean
Section 8.3 Polar Form of Complex Numbers
 80 Chapter 8 Secton 8 Polar Form of Complex Numbers From prevous classes, you may have encountered magnary numbers the square roots of negatve numbers and, more generally, complex numbers whch are the
80 Chapter 8 Secton 8 Polar Form of Complex Numbers From prevous classes, you may have encountered magnary numbers the square roots of negatve numbers and, more generally, complex numbers whch are the
Physics for Scientists and Engineers. Chapter 9 Impulse and Momentum
 Physcs or Scentsts and Engneers Chapter 9 Impulse and Momentum Sprng, 008 Ho Jung Pak Lnear Momentum Lnear momentum o an object o mass m movng wth a velocty v s dened to be p mv Momentum and lnear momentum
Physcs or Scentsts and Engneers Chapter 9 Impulse and Momentum Sprng, 008 Ho Jung Pak Lnear Momentum Lnear momentum o an object o mass m movng wth a velocty v s dened to be p mv Momentum and lnear momentum
Lecture 4. Macrostates and Microstates (Ch. 2 )
 Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
PART I: MULTIPLE CHOICE (32 questions, each multiple choice question has a 2-point value, 64 points total).
 CHEMISTRY 123-07 Mdterm #2 answer key November 04, 2010 Statstcs: Average: 68 p (68%); Hghest: 91 p (91%); Lowest: 37 p (37%) Number of students performng at or above average: 58 (53%) Number of students
CHEMISTRY 123-07 Mdterm #2 answer key November 04, 2010 Statstcs: Average: 68 p (68%); Hghest: 91 p (91%); Lowest: 37 p (37%) Number of students performng at or above average: 58 (53%) Number of students
Q e E i /k B. i i i i
 Water and Aqueous Solutons 3. Lattce Model of a Flud Lattce Models Lattce models provde a mnmalst, or coarse-graned, framework for descrbng the translatonal, rotatonal, and conformatonal degrees of freedom
Water and Aqueous Solutons 3. Lattce Model of a Flud Lattce Models Lattce models provde a mnmalst, or coarse-graned, framework for descrbng the translatonal, rotatonal, and conformatonal degrees of freedom
Week3, Chapter 4. Position and Displacement. Motion in Two Dimensions. Instantaneous Velocity. Average Velocity
 Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
Rotational Dynamics. Physics 1425 Lecture 19. Michael Fowler, UVa
 Rotatonal Dynamcs Physcs 1425 Lecture 19 Mchael Fowler, UVa Rotatonal Dynamcs Newton s Frst Law: a rotatng body wll contnue to rotate at constant angular velocty as long as there s no torque actng on t.
Rotatonal Dynamcs Physcs 1425 Lecture 19 Mchael Fowler, UVa Rotatonal Dynamcs Newton s Frst Law: a rotatng body wll contnue to rotate at constant angular velocty as long as there s no torque actng on t.
CHAPTER 6. LAGRANGE S EQUATIONS (Analytical Mechanics)
 CHAPTER 6 LAGRANGE S EQUATIONS (Analytcal Mechancs) 1 Ex. 1: Consder a partcle movng on a fxed horzontal surface. r P Let, be the poston and F be the total force on the partcle. The FBD s: -mgk F 1 x O
CHAPTER 6 LAGRANGE S EQUATIONS (Analytcal Mechancs) 1 Ex. 1: Consder a partcle movng on a fxed horzontal surface. r P Let, be the poston and F be the total force on the partcle. The FBD s: -mgk F 1 x O
EN40: Dynamics and Vibrations. Homework 4: Work, Energy and Linear Momentum Due Friday March 1 st
 EN40: Dynamcs and bratons Homework 4: Work, Energy and Lnear Momentum Due Frday March 1 st School of Engneerng Brown Unversty 1. The fgure (from ths publcaton) shows the energy per unt area requred to
EN40: Dynamcs and bratons Homework 4: Work, Energy and Lnear Momentum Due Frday March 1 st School of Engneerng Brown Unversty 1. The fgure (from ths publcaton) shows the energy per unt area requred to
Physics 114 Exam 3 Spring Name:
 Physcs 114 Exam 3 Sprng 015 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem 4. Answer each of the followng questons. Ponts for each queston are ndcated n red. Unless otherwse
Physcs 114 Exam 3 Sprng 015 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem 4. Answer each of the followng questons. Ponts for each queston are ndcated n red. Unless otherwse
In the figure below, the point d indicates the location of the consumer that is under competition. Transportation costs are given by td.
 UC Berkeley Economcs 11 Sprng 006 Prof. Joseph Farrell / GSI: Jenny Shanefelter Problem Set # - Suggested Solutons. 1.. In ths problem, we are extendng the usual Hotellng lne so that now t runs from [-a,
UC Berkeley Economcs 11 Sprng 006 Prof. Joseph Farrell / GSI: Jenny Shanefelter Problem Set # - Suggested Solutons. 1.. In ths problem, we are extendng the usual Hotellng lne so that now t runs from [-a,
Chapter 3. r r. Position, Velocity, and Acceleration Revisited
 Chapter 3 Poston, Velocty, and Acceleraton Revsted The poston vector of a partcle s a vector drawn from the orgn to the locaton of the partcle. In two dmensons: r = x ˆ+ yj ˆ (1) The dsplacement vector
Chapter 3 Poston, Velocty, and Acceleraton Revsted The poston vector of a partcle s a vector drawn from the orgn to the locaton of the partcle. In two dmensons: r = x ˆ+ yj ˆ (1) The dsplacement vector
Note on EM-training of IBM-model 1
 Note on EM-tranng of IBM-model INF58 Language Technologcal Applcatons, Fall The sldes on ths subject (nf58 6.pdf) ncludng the example seem nsuffcent to gve a good grasp of what s gong on. Hence here are
Note on EM-tranng of IBM-model INF58 Language Technologcal Applcatons, Fall The sldes on ths subject (nf58 6.pdf) ncludng the example seem nsuffcent to gve a good grasp of what s gong on. Hence here are
10/9/2003 PHY Lecture 11 1
 Announcements 1. Physc Colloquum today --The Physcs and Analyss of Non-nvasve Optcal Imagng. Today s lecture Bref revew of momentum & collsons Example HW problems Introducton to rotatons Defnton of angular
Announcements 1. Physc Colloquum today --The Physcs and Analyss of Non-nvasve Optcal Imagng. Today s lecture Bref revew of momentum & collsons Example HW problems Introducton to rotatons Defnton of angular
10. Canonical Transformations Michael Fowler
 10. Canoncal Transformatons Mchael Fowler Pont Transformatons It s clear that Lagrange s equatons are correct for any reasonable choce of parameters labelng the system confguraton. Let s call our frst
10. Canoncal Transformatons Mchael Fowler Pont Transformatons It s clear that Lagrange s equatons are correct for any reasonable choce of parameters labelng the system confguraton. Let s call our frst
Physics 181. Particle Systems
 Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
4.2 Chemical Driving Force
 4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
8.6 The Complex Number System
 8.6 The Complex Number System Earler n the chapter, we mentoned that we cannot have a negatve under a square root, snce the square of any postve or negatve number s always postve. In ths secton we want
8.6 The Complex Number System Earler n the chapter, we mentoned that we cannot have a negatve under a square root, snce the square of any postve or negatve number s always postve. In ths secton we want
Week 6, Chapter 7 Sect 1-5
 Week 6, Chapter 7 Sect 1-5 Work and Knetc Energy Lecture Quz The frctonal force of the floor on a large sutcase s least when the sutcase s A.pushed by a force parallel to the floor. B.dragged by a force
Week 6, Chapter 7 Sect 1-5 Work and Knetc Energy Lecture Quz The frctonal force of the floor on a large sutcase s least when the sutcase s A.pushed by a force parallel to the floor. B.dragged by a force
EMU Physics Department.
 Physcs 0 Lecture 9 Lnear Momentum and Collsons Assst. Pro. Dr. Al ÖVGÜN EMU Physcs Department www.aogun.com Lnear Momentum q Conseraton o Energy q Momentum q Impulse q Conseraton o Momentum q -D Collsons
Physcs 0 Lecture 9 Lnear Momentum and Collsons Assst. Pro. Dr. Al ÖVGÜN EMU Physcs Department www.aogun.com Lnear Momentum q Conseraton o Energy q Momentum q Impulse q Conseraton o Momentum q -D Collsons
Lecture 10 Support Vector Machines II
 Lecture 10 Support Vector Machnes II 22 February 2016 Taylor B. Arnold Yale Statstcs STAT 365/665 1/28 Notes: Problem 3 s posted and due ths upcomng Frday There was an early bug n the fake-test data; fxed
Lecture 10 Support Vector Machnes II 22 February 2016 Taylor B. Arnold Yale Statstcs STAT 365/665 1/28 Notes: Problem 3 s posted and due ths upcomng Frday There was an early bug n the fake-test data; fxed
Solution Thermodynamics
 Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
V T for n & P = constant
 Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
Period & Frequency. Work and Energy. Methods of Energy Transfer: Energy. Work-KE Theorem 3/4/16. Ranking: Which has the greatest kinetic energy?
 Perod & Frequency Perod (T): Tme to complete one ull rotaton Frequency (): Number o rotatons completed per second. = 1/T, T = 1/ v = πr/t Work and Energy Work: W = F!d (pcks out parallel components) F
Perod & Frequency Perod (T): Tme to complete one ull rotaton Frequency (): Number o rotatons completed per second. = 1/T, T = 1/ v = πr/t Work and Energy Work: W = F!d (pcks out parallel components) F
TP A SOLUTION. For an ideal monatomic gas U=3/2nRT, Since the process is at constant pressure Q = C. giving ) =1000/(5/2*8.31*10)
 T A SOLUTION For an deal monatomc gas U/nRT, Snce the process s at constant pressure Q C pn T gvng a: n Q /( 5 / R T ) /(5/*8.*) C V / R and C / R + R 5 / R. U U / nr T (/ ) R T ( Q / 5 / R T ) Q / 5 Q
T A SOLUTION For an deal monatomc gas U/nRT, Snce the process s at constant pressure Q C pn T gvng a: n Q /( 5 / R T ) /(5/*8.*) C V / R and C / R + R 5 / R. U U / nr T (/ ) R T ( Q / 5 / R T ) Q / 5 Q
Physics 4B. A positive value is obtained, so the current is counterclockwise around the circuit.
 Physcs 4B Solutons to Chapter 7 HW Chapter 7: Questons:, 8, 0 Problems:,,, 45, 48,,, 7, 9 Queston 7- (a) no (b) yes (c) all te Queston 7-8 0 μc Queston 7-0, c;, a;, d; 4, b Problem 7- (a) Let be the current
Physcs 4B Solutons to Chapter 7 HW Chapter 7: Questons:, 8, 0 Problems:,,, 45, 48,,, 7, 9 Queston 7- (a) no (b) yes (c) all te Queston 7-8 0 μc Queston 7-0, c;, a;, d; 4, b Problem 7- (a) Let be the current
CHAPTER 8 Potential Energy and Conservation of Energy
 CHAPTER 8 Potental Energy and Conservaton o Energy One orm o energy can be converted nto another orm o energy. Conservatve and non-conservatve orces Physcs 1 Knetc energy: Potental energy: Energy assocated
CHAPTER 8 Potental Energy and Conservaton o Energy One orm o energy can be converted nto another orm o energy. Conservatve and non-conservatve orces Physcs 1 Knetc energy: Potental energy: Energy assocated
Force = F Piston area = A
 CHAPTER III Ths chapter s an mportant transton between the propertes o pure substances and the most mportant chapter whch s: the rst law o thermodynamcs In ths chapter, we wll ntroduce the notons o heat,
CHAPTER III Ths chapter s an mportant transton between the propertes o pure substances and the most mportant chapter whch s: the rst law o thermodynamcs In ths chapter, we wll ntroduce the notons o heat,
First Law: A body at rest remains at rest, a body in motion continues to move at constant velocity, unless acted upon by an external force.
 Secton 1. Dynamcs (Newton s Laws of Moton) Two approaches: 1) Gven all the forces actng on a body, predct the subsequent (changes n) moton. 2) Gven the (changes n) moton of a body, nfer what forces act
Secton 1. Dynamcs (Newton s Laws of Moton) Two approaches: 1) Gven all the forces actng on a body, predct the subsequent (changes n) moton. 2) Gven the (changes n) moton of a body, nfer what forces act
The Feynman path integral
 The Feynman path ntegral Aprl 3, 205 Hesenberg and Schrödnger pctures The Schrödnger wave functon places the tme dependence of a physcal system n the state, ψ, t, where the state s a vector n Hlbert space
The Feynman path ntegral Aprl 3, 205 Hesenberg and Schrödnger pctures The Schrödnger wave functon places the tme dependence of a physcal system n the state, ψ, t, where the state s a vector n Hlbert space
Lecture 10: May 6, 2013
 TTIC/CMSC 31150 Mathematcal Toolkt Sprng 013 Madhur Tulsan Lecture 10: May 6, 013 Scrbe: Wenje Luo In today s lecture, we manly talked about random walk on graphs and ntroduce the concept of graph expander,
TTIC/CMSC 31150 Mathematcal Toolkt Sprng 013 Madhur Tulsan Lecture 10: May 6, 013 Scrbe: Wenje Luo In today s lecture, we manly talked about random walk on graphs and ntroduce the concept of graph expander,
University Physics AI No. 10 The First Law of Thermodynamics
 Unversty hyscs I No he Frst Law o hermodynamcs lass Number Name Ihoose the orrect nswer Whch o the ollowng processes must volate the rst law o thermodynamcs? (here may be more than one answer!) (,B,D )
Unversty hyscs I No he Frst Law o hermodynamcs lass Number Name Ihoose the orrect nswer Whch o the ollowng processes must volate the rst law o thermodynamcs? (here may be more than one answer!) (,B,D )
Week 9 Chapter 10 Section 1-5
 Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Chapter 07: Kinetic Energy and Work
 Chapter 07: Knetc Energy and Work Conservaton o Energy s one o Nature s undamental laws that s not volated. Energy can take on derent orms n a gven system. Ths chapter we wll dscuss work and knetc energy.
Chapter 07: Knetc Energy and Work Conservaton o Energy s one o Nature s undamental laws that s not volated. Energy can take on derent orms n a gven system. Ths chapter we wll dscuss work and knetc energy.
Spin-rotation coupling of the angularly accelerated rigid body
 Spn-rotaton couplng of the angularly accelerated rgd body Loua Hassan Elzen Basher Khartoum, Sudan. Postal code:11123 E-mal: louaelzen@gmal.com November 1, 2017 All Rghts Reserved. Abstract Ths paper s
Spn-rotaton couplng of the angularly accelerated rgd body Loua Hassan Elzen Basher Khartoum, Sudan. Postal code:11123 E-mal: louaelzen@gmal.com November 1, 2017 All Rghts Reserved. Abstract Ths paper s
Physics 3 (PHYF144) Chap 2: Heat and the First Law of Thermodynamics System. Quantity Positive Negative
 Physcs (PHYF hap : Heat and the Frst aw of hermodynamcs -. Work and Heat n hermodynamc Processes A thermodynamc system s a system that may exchange energy wth ts surroundngs by means of heat and work.
Physcs (PHYF hap : Heat and the Frst aw of hermodynamcs -. Work and Heat n hermodynamc Processes A thermodynamc system s a system that may exchange energy wth ts surroundngs by means of heat and work.
Physics 101 Lecture 9 Linear Momentum and Collisions
 Physcs 0 Lecture 9 Lnear Momentum and Collsons Dr. Al ÖVGÜN EMU Physcs Department www.aogun.com Lnear Momentum and Collsons q q q q q q q Conseraton o Energy Momentum Impulse Conseraton o Momentum -D Collsons
Physcs 0 Lecture 9 Lnear Momentum and Collsons Dr. Al ÖVGÜN EMU Physcs Department www.aogun.com Lnear Momentum and Collsons q q q q q q q Conseraton o Energy Momentum Impulse Conseraton o Momentum -D Collsons
