STATISTICAL MECHANICS
|
|
|
- Austin Walker
- 5 years ago
- Views:
Transcription
1 STATISTICAL MECHANICS
2 Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of all partcles n few eqns. In general unrealstc to descrbe KE of every atom Alternatve: descrbe a small sample of atoms nfer KE nternal Ths wll work only f the sample s statstcally sound Goal: fnd how thermal energy s statstcally dstrbuted Elastc collsons KE transfers from faster to slower partcle Equlbrum collsons occur, but dstrbuton of KE s constant
3 Dstrbuton Functons Let x = result of some measurement Spectrum of values for x can be ether dscrete or contnuous If many measurements are made: Descrbe results usng a probablty dstrbuton functon f(x): Dscrete x g x f x = 1 Normalzaton g x f x Expectaton Value Contnuous x f x dx = 1 g x g x Infnte precson: Prob(exact value) = 0 f x dx f(x ) 2 con flps (# heads) probablty densty p a b b = a f x dx x
4 State Space for Ideal Gas Each partcle has 6 degrees of freedom (D.O.F.): r, v Ideal gas of N partcles 6N-dmensonal state space Each pont n state space s called a mcrostate of system A macrostate descrbes possble values for a measurement Example: Many mcrostates produce the same pressure To calculate dstrbuton f(x) for some measurement x: 1) Fnd % of mcrostates whch yeld result x (or x x+dx) densty of states Ω(x) / degeneracy volume n state space 2) Fnd probablty of each mcrostate (ncludng constrants) e.g. (conservaton of energy) and (maxmzaton of entropy )
5 Densty of States / Degeneracy Goal: count % of mcrostates n a gven macrostate Example: Flp a con N tmes, let x = # of heads Degeneracy (# of mcrostates w/ value x): N x = N! x! N x! Densty of states Ω(E) de for an deal gas Volume of state space whch has energy n range E (E+dE) dv ss = 4 v 2 dv E = 1 2 m v 2 v = 2 E m dv = de 2 E m dv ss = 4 2 E m E de = A 1 E de de m 2 E (more mcrostates at hgher E)
6 Constrants Wthout constrants all mcrostates equally possble Constrants restrct the state space avalable to system Example: 6-sded de rolled 3 tmes (216 mcrostates) Constrant: <x> = 2 Calculate how many mcrostates Among these mcrostates, fnd dstrbuton of 1's, 2's, etc. Constrants on deal gas N partcles, total energy E 0 Goal: calculate dstrbuton of E for allowable mcrostates only To smplfy calculaton, use dscrete spectrum of energes E Dstrbuton: (n 1 wth energy E 1 ), (n 2 wth energy E 2 ),
7 Countng Mcrostates Consder measurement wth possble results x Perform measurement N tmes dstrbuton: n Countng the # of mcrostates for a partcular dstrbuton: = N n 1 N n 1 n 2 N n 1 n 2... n 3 p x = n N = N! N n 1! N n 1 n 2!... n 1! n 2! n 3!... N n 1! N n 1 n 2! N n 1 n 2 n 3!... = N! n 1! n 2! n 3!... Strlng's approxmaton (good for large X): ln X! X ln X X ln [ N ln N N ] [n 1 ln n 1 n 1 n 2 ln n 2 n 2 n 3 ln n 3 n 3...] ln n ln n N # of mcrostate arrangements wll be maxmzed for some partcular dstrbuton of n
8 Entropy ln n ln n N ln N n N ln n N Ths quantty s called entropy S Probablty dstrbuton p for system naturally fnds maxmal S Wthout extra constrant: maxmal S all p are equal (prove!) Wth constrants: non-unform dstrbuton of p p ln p Examples: calculate entropy for 1) con 2) 6-sded de Constrant: based 3-sded de (<x> = 2.5) Calculate p 1, p 2, p 3 such that S s maxmzed Image compresson: Each color pxel has (256) 3 possbltes How much can fle be compressed usng black/whte (256)?
9 Lagrange Multplers Goal: Maxmze functon S(x 1, x 2, x N ) wth constrants M constrants: C 1 (x 1, x 2, )=0, C 2 (x 1, x 2, )=0, C M (x 1, x 2,...)=0 Usual procedure: solve M constrant equatons for x j Maxmze S wth respect to remanng (N M) x 's Solve (N M) equatons for (N M) unknowns Problem: calculatons are often tedous and/or dffcult Alternatve method: Lagrange multplers Consder the functon Maxmzng S' also maxmzes S Solve (N+M) equatons for (N+M) unknowns S 1 C 1 2 C 2... S ' x 1, x 2,..., 1, 2,... S ' x S max = S ' j S max = 0
10 Example Mnmze the functon S x, y, z = x 2 y 2 z 2 Subject to the constrant C 1 x, y, z = 6 3 x 2 y z = 0 What does ths look lke n 3-D space? Method 1: Solve constrant for x and plug nto S Method 2: Lagrange multplers Now add another constrant C 2 x, y, z = 5 z = 0 Calculate the new x,y,z whch mnmzes S
11 Boltzmann Factor Constrants on p for system wth possble energes E : 1) Normalzaton: C 1 p 1, p 2,... = 1 p = 0 2) Total KE of system: C 2 p 1, p 2,... = E p E = 0 Maxmze S p 1, p 2,... = p ln p (usng Lagrange): S p 1 C 1 p 2 C 2 p = 0 ln p E = 0 p = e 1 e 1 e 2 E p = A e E (rename constants) p (E ) Boltzmann Factor descrbes statstcal dstrbutons of energy (gnores densty of states) E Normalze: 1 = p A = = A 1 e E e E normalzaton constant partton functon
12 Temperature Boltzmann factor contans unknown parameter β When β s large most partcles have low energy Hstorcal dea of temperature : average partcle moton Heat / cold felt by human senses Measured by thermal expanson of substances (arbtrary unts) β and temperature nversely related And measured n dfferent unts (temperature n ºK/ºC/ºF, β n J 1 ) Unts matched up by Boltzmann's constant: 1 k B T k B = J K
13 Dstrbutons n an Ideal Gas Each possble measurement has ts own dstrbuton: Velocty Components v x v y v z : Energy: Densty of states: v x = A 1 Densty of states: E = A 1 E Boltzmann factor: e m 2 v x 2 k T Boltzmann factor: e E k T Normalzed dstrbuton: f v x = m m 2 v x 2 k T e 2 k T 2 v x = 0 v x = k T m Normalzed dstrbuton: f E 4 = k T 3 E = 3 2 k T E e E k T Maxwell-Boltzmann dstrbuton of energes Note: E = 1 2 m v x 2 v y 2 v z2 = 3 2 m v x2 = 3 2 k T
14 Standard Devaton Useful statstcal quanttes for a dstrbuton f(x): Expectaton value <x> descrbes center of dstrbuton Standard devaton σ descrbes spread of dstrbuton x x 2 = x 2 x 2 Standard devaton for deal gas velocty component: v x = 0 = v x2 v x, rms v x,rms for deal gas depends on temperature: v x, rms = k T m
15 Examples Consder a 3-state system: E 1 / 3E 1 / 5E 1 Wth no degeneracy n the states If kt = 2E 1, calculate P(E ) Repeat calculaton f 5E 1 level has 2-fold degeneracy Calculate velocty dstrbuton f(v x, v y, v z ) for deal gas Includng overall normalzaton constant Calculate σ E for Maxwell-Boltzmann dstrbuton
16 Equpartton of Energy Energy of mcrostate depends of values of DOF: Common stuaton: E = C x 2 (where x = DOF) C x 2 E = C x 2 k T e dx C x 2 e k T dx = C 2 k T 3 C k T C = 1 2 k T Result holds for any value of C (thus any DOF) Examples: translaton / rotaton / vbraton KE of molecules Systems wth energy whch s quadratc n several DOF: Equally dstrbutes the energy among DOF (½ kt each) Explans heat capacty of deal gases
17 Heat Capacty of an Ideal Gas Heat Capacty heat energy requred for ΔT: Usually whle holdng volume or pressure constant (C V vs. C P ) C P = C V + Nk some heat s used as work Equpartton Theorem: predcts C V = 3 2 N k T T C = Q T = 3 2 N k Matches expermental results for argon / helum (monatomc) Experments on datomc gases (O 2, N 2, etc.): C V = 5 2 N k C V ncreases due to molecular vbraton / rotaton But these DOF requre a mnmum T to turn on C V becomes a functon of T:
18 Pressure n an Ideal Gas L Usng knowledge of f(v x, v y, v z ) Can relate gas pressure to statstcal quanttes Consder an atom wth a gven velocty v x, v y, v z Makes one collson wth rght-hand wall every t = 2 L v x Each collson delvers mpulse p x = 2 m v x Average force ths atom exerts on rght wall: Total pressure on rght wall: P = N F x A F x = p x t = m v 2 x L = N V m v x2 = N V k T Ideal Gas Law
19 Thermodynamc Cycles Thermal energy of enclosed gas can do work And have work from external force done on t W = P dv Energy can be added to system by heat source To replensh energy lost due to work Thus creatng a repeatng cycle of work on external object Examples: burnng fuel, solar energy, etc. Gas expands whle hot, compresses whle cold Thermodynamc engnes have theoretcal lmts on effcency Because not all of the thermal energy can be used
20 PV Dagrams Vsualzaton of thermodynamc cycles Common paths n PV space: Isothermal: temperature = constant P = C V Adabatc: total energy = constant P = C V = C P C V Isobarc: pressure = constant Constant-volume: vertcal lne on PV dagram
Physics 240: Worksheet 30 Name:
 (1) One mole of an deal monatomc gas doubles ts temperature and doubles ts volume. What s the change n entropy of the gas? () 1 kg of ce at 0 0 C melts to become water at 0 0 C. What s the change n entropy
(1) One mole of an deal monatomc gas doubles ts temperature and doubles ts volume. What s the change n entropy of the gas? () 1 kg of ce at 0 0 C melts to become water at 0 0 C. What s the change n entropy
and Statistical Mechanics Material Properties
 Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Lecture 4. Macrostates and Microstates (Ch. 2 )
 Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
University of Washington Department of Chemistry Chemistry 453 Winter Quarter 2015
 Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
Introduction to Statistical Methods
 Introducton to Statstcal Methods Physcs 4362, Lecture #3 hermodynamcs Classcal Statstcal Knetc heory Classcal hermodynamcs Macroscopc approach General propertes of the system Macroscopc varables 1 hermodynamc
Introducton to Statstcal Methods Physcs 4362, Lecture #3 hermodynamcs Classcal Statstcal Knetc heory Classcal hermodynamcs Macroscopc approach General propertes of the system Macroscopc varables 1 hermodynamc
Chapter 21 - The Kinetic Theory of Gases
 hapter 1 - he Knetc heory o Gases 1. Δv 8.sn 4. 8.sn 4. m s F Nm. 1 kg.94 N Δ t. s F A 1.7 N m 1.7 a N mv 1.6 Use the equaton descrbng the knetc-theory account or pressure:. hen mv Kav where N nna NA N
hapter 1 - he Knetc heory o Gases 1. Δv 8.sn 4. 8.sn 4. m s F Nm. 1 kg.94 N Δ t. s F A 1.7 N m 1.7 a N mv 1.6 Use the equaton descrbng the knetc-theory account or pressure:. hen mv Kav where N nna NA N
Physics 607 Exam 1. ( ) = 1, Γ( z +1) = zγ( z) x n e x2 dx = 1. e x2
 Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
Lecture 3 Examples and Problems
 Lecture 3 Examles and Problems Mechancs & thermodynamcs Equartton Frst Law of Thermodynamcs Ideal gases Isothermal and adabatc rocesses Readng: Elements Ch. 1-3 Lecture 3, 1 Wllam Thomson (1824 1907) a.k.a.
Lecture 3 Examles and Problems Mechancs & thermodynamcs Equartton Frst Law of Thermodynamcs Ideal gases Isothermal and adabatc rocesses Readng: Elements Ch. 1-3 Lecture 3, 1 Wllam Thomson (1824 1907) a.k.a.
Physics 115. Molecular motion and temperature Phase equilibrium, evaporation
 Physcs 115 General Physcs II Sesson 9 Molecular moton and temperature Phase equlbrum, evaporaton R. J. Wlkes Emal: phy115a@u.washngton.edu Home page: http://courses.washngton.edu/phy115a/ 4/14/14 Physcs
Physcs 115 General Physcs II Sesson 9 Molecular moton and temperature Phase equlbrum, evaporaton R. J. Wlkes Emal: phy115a@u.washngton.edu Home page: http://courses.washngton.edu/phy115a/ 4/14/14 Physcs
Lecture 7: Boltzmann distribution & Thermodynamics of mixing
 Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
#64. ΔS for Isothermal Mixing of Ideal Gases
 #64 Carnot Heat Engne ΔS for Isothermal Mxng of Ideal Gases ds = S dt + S T V V S = P V T T V PV = nrt, P T ds = v T = nr V dv V nr V V = nrln V V = - nrln V V ΔS ΔS ΔS for Isothermal Mxng for Ideal Gases
#64 Carnot Heat Engne ΔS for Isothermal Mxng of Ideal Gases ds = S dt + S T V V S = P V T T V PV = nrt, P T ds = v T = nr V dv V nr V V = nrln V V = - nrln V V ΔS ΔS ΔS for Isothermal Mxng for Ideal Gases
Thermodynamics Second Law Entropy
 Thermodynamcs Second Law Entropy Lana Sherdan De Anza College May 8, 2018 Last tme the Boltzmann dstrbuton (dstrbuton of energes) the Maxwell-Boltzmann dstrbuton (dstrbuton of speeds) the Second Law of
Thermodynamcs Second Law Entropy Lana Sherdan De Anza College May 8, 2018 Last tme the Boltzmann dstrbuton (dstrbuton of energes) the Maxwell-Boltzmann dstrbuton (dstrbuton of speeds) the Second Law of
Problem Points Score Total 100
 Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
10.40 Appendix Connection to Thermodynamics and Derivation of Boltzmann Distribution
 10.40 Appendx Connecton to Thermodynamcs Dervaton of Boltzmann Dstrbuton Bernhardt L. Trout Outlne Cannoncal ensemble Maxmumtermmethod Most probable dstrbuton Ensembles contnued: Canoncal, Mcrocanoncal,
10.40 Appendx Connecton to Thermodynamcs Dervaton of Boltzmann Dstrbuton Bernhardt L. Trout Outlne Cannoncal ensemble Maxmumtermmethod Most probable dstrbuton Ensembles contnued: Canoncal, Mcrocanoncal,
Review of Classical Thermodynamics
 Revew of Classcal hermodynamcs Physcs 4362, Lecture #1, 2 Syllabus What s hermodynamcs? 1 [A law] s more mpressve the greater the smplcty of ts premses, the more dfferent are the knds of thngs t relates,
Revew of Classcal hermodynamcs Physcs 4362, Lecture #1, 2 Syllabus What s hermodynamcs? 1 [A law] s more mpressve the greater the smplcty of ts premses, the more dfferent are the knds of thngs t relates,
Chapter 1. Probability
 Chapter. Probablty Mcroscopc propertes of matter: quantum mechancs, atomc and molecular propertes Macroscopc propertes of matter: thermodynamcs, E, H, C V, C p, S, A, G How do we relate these two propertes?
Chapter. Probablty Mcroscopc propertes of matter: quantum mechancs, atomc and molecular propertes Macroscopc propertes of matter: thermodynamcs, E, H, C V, C p, S, A, G How do we relate these two propertes?
Thermodynamics and Gases
 hermodynamcs and Gases Last tme Knetc heory o Gases or smple (monatomc) gases Atomc nature o matter Demonstrate deal gas law Atomc knetc energy nternal energy Mean ree path and velocty dstrbutons From
hermodynamcs and Gases Last tme Knetc heory o Gases or smple (monatomc) gases Atomc nature o matter Demonstrate deal gas law Atomc knetc energy nternal energy Mean ree path and velocty dstrbutons From
Thermodynamics and statistical mechanics in materials modelling II
 Course MP3 Lecture 8/11/006 (JAE) Course MP3 Lecture 8/11/006 Thermodynamcs and statstcal mechancs n materals modellng II A bref résumé of the physcal concepts used n materals modellng Dr James Ellott.1
Course MP3 Lecture 8/11/006 (JAE) Course MP3 Lecture 8/11/006 Thermodynamcs and statstcal mechancs n materals modellng II A bref résumé of the physcal concepts used n materals modellng Dr James Ellott.1
Lecture 20: Hypothesis testing
 Lecture : Hpothess testng Much of statstcs nvolves hpothess testng compare a new nterestng hpothess, H (the Alternatve hpothess to the borng, old, well-known case, H (the Null Hpothess or, decde whether
Lecture : Hpothess testng Much of statstcs nvolves hpothess testng compare a new nterestng hpothess, H (the Alternatve hpothess to the borng, old, well-known case, H (the Null Hpothess or, decde whether
Physics 181. Particle Systems
 Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
5.04, Principles of Inorganic Chemistry II MIT Department of Chemistry Lecture 32: Vibrational Spectroscopy and the IR
 5.0, Prncples of Inorganc Chemstry II MIT Department of Chemstry Lecture 3: Vbratonal Spectroscopy and the IR Vbratonal spectroscopy s confned to the 00-5000 cm - spectral regon. The absorpton of a photon
5.0, Prncples of Inorganc Chemstry II MIT Department of Chemstry Lecture 3: Vbratonal Spectroscopy and the IR Vbratonal spectroscopy s confned to the 00-5000 cm - spectral regon. The absorpton of a photon
5.60 Thermodynamics & Kinetics Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
Week 9 Chapter 10 Section 1-5
 Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Physics 141. Lecture 14. Frank L. H. Wolfs Department of Physics and Astronomy, University of Rochester, Lecture 14, Page 1
 Physcs 141. Lecture 14. Frank L. H. Wolfs Department of Physcs and Astronomy, Unversty of Rochester, Lecture 14, Page 1 Physcs 141. Lecture 14. Course Informaton: Lab report # 3. Exam # 2. Mult-Partcle
Physcs 141. Lecture 14. Frank L. H. Wolfs Department of Physcs and Astronomy, Unversty of Rochester, Lecture 14, Page 1 Physcs 141. Lecture 14. Course Informaton: Lab report # 3. Exam # 2. Mult-Partcle
Chapters 18 & 19: Themodynamics review. All macroscopic (i.e., human scale) quantities must ultimately be explained on the microscopic scale.
 Chapters 18 & 19: Themodynamcs revew ll macroscopc (.e., human scale) quanttes must ultmately be explaned on the mcroscopc scale. Chapter 18: Thermodynamcs Thermodynamcs s the study o the thermal energy
Chapters 18 & 19: Themodynamcs revew ll macroscopc (.e., human scale) quanttes must ultmately be explaned on the mcroscopc scale. Chapter 18: Thermodynamcs Thermodynamcs s the study o the thermal energy
ESCI 341 Atmospheric Thermodynamics Lesson 10 The Physical Meaning of Entropy
 ESCI 341 Atmospherc Thermodynamcs Lesson 10 The Physcal Meanng of Entropy References: An Introducton to Statstcal Thermodynamcs, T.L. Hll An Introducton to Thermodynamcs and Thermostatstcs, H.B. Callen
ESCI 341 Atmospherc Thermodynamcs Lesson 10 The Physcal Meanng of Entropy References: An Introducton to Statstcal Thermodynamcs, T.L. Hll An Introducton to Thermodynamcs and Thermostatstcs, H.B. Callen
5.62 Physical Chemistry II Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Sprng 2008 Lecture 34 Page Transton
MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Sprng 2008 Lecture 34 Page Transton
Temperature. Chapter Heat Engine
 Chapter 3 Temperature In prevous chapters of these notes we ntroduced the Prncple of Maxmum ntropy as a technque for estmatng probablty dstrbutons consstent wth constrants. In Chapter 9 we dscussed the
Chapter 3 Temperature In prevous chapters of these notes we ntroduced the Prncple of Maxmum ntropy as a technque for estmatng probablty dstrbutons consstent wth constrants. In Chapter 9 we dscussed the
PHY688, Statistical Mechanics
 Department of Physcs & Astronomy 449 ESS Bldg. Stony Brook Unversty January 31, 2017 Nuclear Astrophyscs James.Lattmer@Stonybrook.edu Thermodynamcs Internal Energy Densty and Frst Law: ε = E V = Ts P +
Department of Physcs & Astronomy 449 ESS Bldg. Stony Brook Unversty January 31, 2017 Nuclear Astrophyscs James.Lattmer@Stonybrook.edu Thermodynamcs Internal Energy Densty and Frst Law: ε = E V = Ts P +
G4023 Mid-Term Exam #1 Solutions
 Exam1Solutons.nb 1 G03 Md-Term Exam #1 Solutons 1-Oct-0, 1:10 p.m to :5 p.m n 1 Pupn Ths exam s open-book, open-notes. You may also use prnt-outs of the homework solutons and a calculator. 1 (30 ponts,
Exam1Solutons.nb 1 G03 Md-Term Exam #1 Solutons 1-Oct-0, 1:10 p.m to :5 p.m n 1 Pupn Ths exam s open-book, open-notes. You may also use prnt-outs of the homework solutons and a calculator. 1 (30 ponts,
LAGRANGIAN MECHANICS
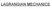 LAGRANGIAN MECHANICS Generalzed Coordnates State of system of N partcles (Newtonan vew): PE, KE, Momentum, L calculated from m, r, ṙ Subscrpt covers: 1) partcles N 2) dmensons 2, 3, etc. PE U r = U x 1,
LAGRANGIAN MECHANICS Generalzed Coordnates State of system of N partcles (Newtonan vew): PE, KE, Momentum, L calculated from m, r, ṙ Subscrpt covers: 1) partcles N 2) dmensons 2, 3, etc. PE U r = U x 1,
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM
 ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
Thermodynamics and Kinetics of Solids 33. III. Statistical Thermodynamics. Â N i = N (5.3) N i. i =0. Â e i = E (5.4) has a maximum.
 hermodynamcs and Knetcs of Solds 33 III. Statstcal hermodynamcs 5. Statstcal reatment of hermodynamcs 5.1. Statstcs and Phenomenologcal hermodynamcs. Calculaton of the energetc state of each atomc or molecular
hermodynamcs and Knetcs of Solds 33 III. Statstcal hermodynamcs 5. Statstcal reatment of hermodynamcs 5.1. Statstcs and Phenomenologcal hermodynamcs. Calculaton of the energetc state of each atomc or molecular
Lecture 10. Reading: Notes and Brennan Chapter 5
 Lecture tatstcal Mechancs and Densty of tates Concepts Readng: otes and Brennan Chapter 5 Georga Tech C 645 - Dr. Alan Doolttle C 645 - Dr. Alan Doolttle Georga Tech How do electrons and holes populate
Lecture tatstcal Mechancs and Densty of tates Concepts Readng: otes and Brennan Chapter 5 Georga Tech C 645 - Dr. Alan Doolttle C 645 - Dr. Alan Doolttle Georga Tech How do electrons and holes populate
THERMAL DISTRIBUTION IN THE HCL SPECTRUM OBJECTIVE
 ame: THERMAL DISTRIBUTIO I THE HCL SPECTRUM OBJECTIVE To nvestgate a system s thermal dstrbuton n dscrete states; specfcally, determne HCl gas temperature from the relatve occupatons of ts rotatonal states.
ame: THERMAL DISTRIBUTIO I THE HCL SPECTRUM OBJECTIVE To nvestgate a system s thermal dstrbuton n dscrete states; specfcally, determne HCl gas temperature from the relatve occupatons of ts rotatonal states.
Homework Chapter 21 Solutions!!
 Homework Chapter 1 Solutons 1.7 1.13 1.17 1.19 1.6 1.33 1.45 1.51 1.71 page 1 Problem 1.7 A mole sample of oxygen gas s confned to a 5 lter vessel at a pressure of 8 atm. Fnd the average translatonal knetc
Homework Chapter 1 Solutons 1.7 1.13 1.17 1.19 1.6 1.33 1.45 1.51 1.71 page 1 Problem 1.7 A mole sample of oxygen gas s confned to a 5 lter vessel at a pressure of 8 atm. Fnd the average translatonal knetc
Chapter 11: Angular Momentum
 Chapter 11: ngular Momentum Statc Equlbrum In Chap. 4 we studed the equlbrum of pontobjects (mass m) wth the applcaton of Newton s aws F 0 F x y, 0 Therefore, no lnear (translatonal) acceleraton, a0 For
Chapter 11: ngular Momentum Statc Equlbrum In Chap. 4 we studed the equlbrum of pontobjects (mass m) wth the applcaton of Newton s aws F 0 F x y, 0 Therefore, no lnear (translatonal) acceleraton, a0 For
Q e E i /k B. i i i i
 Water and Aqueous Solutons 3. Lattce Model of a Flud Lattce Models Lattce models provde a mnmalst, or coarse-graned, framework for descrbng the translatonal, rotatonal, and conformatonal degrees of freedom
Water and Aqueous Solutons 3. Lattce Model of a Flud Lattce Models Lattce models provde a mnmalst, or coarse-graned, framework for descrbng the translatonal, rotatonal, and conformatonal degrees of freedom
Lossy Compression. Compromise accuracy of reconstruction for increased compression.
 Lossy Compresson Compromse accuracy of reconstructon for ncreased compresson. The reconstructon s usually vsbly ndstngushable from the orgnal mage. Typcally, one can get up to 0:1 compresson wth almost
Lossy Compresson Compromse accuracy of reconstructon for ncreased compresson. The reconstructon s usually vsbly ndstngushable from the orgnal mage. Typcally, one can get up to 0:1 compresson wth almost
STATISTICAL MECHANICAL ENSEMBLES 1 MICROSCOPIC AND MACROSCOPIC VARIABLES PHASE SPACE ENSEMBLES. CHE 524 A. Panagiotopoulos 1
 CHE 54 A. Panagotopoulos STATSTCAL MECHACAL ESEMBLES MCROSCOPC AD MACROSCOPC ARABLES The central queston n Statstcal Mechancs can be phrased as follows: f partcles (atoms, molecules, electrons, nucle,
CHE 54 A. Panagotopoulos STATSTCAL MECHACAL ESEMBLES MCROSCOPC AD MACROSCOPC ARABLES The central queston n Statstcal Mechancs can be phrased as follows: f partcles (atoms, molecules, electrons, nucle,
Lecture 3: Probability Distributions
 Lecture 3: Probablty Dstrbutons Random Varables Let us begn by defnng a sample space as a set of outcomes from an experment. We denote ths by S. A random varable s a functon whch maps outcomes nto the
Lecture 3: Probablty Dstrbutons Random Varables Let us begn by defnng a sample space as a set of outcomes from an experment. We denote ths by S. A random varable s a functon whch maps outcomes nto the
PES 2130 Fall 2014, Spendier Lecture 7/Page 1
 PES 2130 Fall 2014, Spender Lecture 7/Page 1 Lecture today: Chapter 20 (ncluded n exam 1) 1) Entropy 2) Second Law o hermodynamcs 3) Statstcal Vew o Entropy Announcements: Next week Wednesday Exam 1! -
PES 2130 Fall 2014, Spender Lecture 7/Page 1 Lecture today: Chapter 20 (ncluded n exam 1) 1) Entropy 2) Second Law o hermodynamcs 3) Statstcal Vew o Entropy Announcements: Next week Wednesday Exam 1! -
Physics 207: Lecture 20. Today s Agenda Homework for Monday
 Physcs 207: Lecture 20 Today s Agenda Homework for Monday Recap: Systems of Partcles Center of mass Velocty and acceleraton of the center of mass Dynamcs of the center of mass Lnear Momentum Example problems
Physcs 207: Lecture 20 Today s Agenda Homework for Monday Recap: Systems of Partcles Center of mass Velocty and acceleraton of the center of mass Dynamcs of the center of mass Lnear Momentum Example problems
Quantum Statistical Mechanics
 Chapter 8 Quantum Statstcal Mechancs 8.1 Mcrostates For a collecton of N partcles the classcal mcrostate of the system s unquely specfed by a vector (q, p) (q 1...q N, p 1...p 3N )(q 1...q 3N,p 1...p 3N
Chapter 8 Quantum Statstcal Mechancs 8.1 Mcrostates For a collecton of N partcles the classcal mcrostate of the system s unquely specfed by a vector (q, p) (q 1...q N, p 1...p 3N )(q 1...q 3N,p 1...p 3N
Thermodynamics General
 Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Robert Eisberg Second edition CH 09 Multielectron atoms ground states and x-ray excitations
 Quantum Physcs 量 理 Robert Esberg Second edton CH 09 Multelectron atoms ground states and x-ray exctatons 9-01 By gong through the procedure ndcated n the text, develop the tme-ndependent Schroednger equaton
Quantum Physcs 量 理 Robert Esberg Second edton CH 09 Multelectron atoms ground states and x-ray exctatons 9-01 By gong through the procedure ndcated n the text, develop the tme-ndependent Schroednger equaton
π e ax2 dx = x 2 e ax2 dx or x 3 e ax2 dx = 1 x 4 e ax2 dx = 3 π 8a 5/2 (a) We are considering the Maxwell velocity distribution function: 2πτ/m
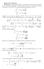 Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
5.62 Physical Chemistry II Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Lecture #8: Boltzmann, Ferm-Drac,
MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Lecture #8: Boltzmann, Ferm-Drac,
Markov Chain Monte Carlo (MCMC), Gibbs Sampling, Metropolis Algorithms, and Simulated Annealing Bioinformatics Course Supplement
 Markov Chan Monte Carlo MCMC, Gbbs Samplng, Metropols Algorthms, and Smulated Annealng 2001 Bonformatcs Course Supplement SNU Bontellgence Lab http://bsnuackr/ Outlne! Markov Chan Monte Carlo MCMC! Metropols-Hastngs
Markov Chan Monte Carlo MCMC, Gbbs Samplng, Metropols Algorthms, and Smulated Annealng 2001 Bonformatcs Course Supplement SNU Bontellgence Lab http://bsnuackr/ Outlne! Markov Chan Monte Carlo MCMC! Metropols-Hastngs
Problem Set #6 solution, Chem 340, Fall 2013 Due Friday, Oct 11, 2013 Please show all work for credit
 Problem Set #6 soluton, Chem 340, Fall 2013 Due Frday, Oct 11, 2013 Please show all work for credt To hand n: Atkns Chap 3 Exercses: 3.3(b), 3.8(b), 3.13(b), 3.15(b) Problems: 3.1, 3.12, 3.36, 3.43 Engel
Problem Set #6 soluton, Chem 340, Fall 2013 Due Frday, Oct 11, 2013 Please show all work for credt To hand n: Atkns Chap 3 Exercses: 3.3(b), 3.8(b), 3.13(b), 3.15(b) Problems: 3.1, 3.12, 3.36, 3.43 Engel
Probability Theory. The nth coefficient of the Taylor series of f(k), expanded around k = 0, gives the nth moment of x as ( ik) n n!
 8333: Statstcal Mechancs I Problem Set # 3 Solutons Fall 3 Characterstc Functons: Probablty Theory The characterstc functon s defned by fk ep k = ep kpd The nth coeffcent of the Taylor seres of fk epanded
8333: Statstcal Mechancs I Problem Set # 3 Solutons Fall 3 Characterstc Functons: Probablty Theory The characterstc functon s defned by fk ep k = ep kpd The nth coeffcent of the Taylor seres of fk epanded
Supplemental Material: Causal Entropic Forces
 Supplemental Materal: Causal Entropc Forces A. D. Wssner-Gross 1, 2, and C. E. Freer 3 1 Insttute for Appled Computatonal Scence, Harvard Unversty, Cambrdge, Massachusetts 02138, USA 2 The Meda Laboratory,
Supplemental Materal: Causal Entropc Forces A. D. Wssner-Gross 1, 2, and C. E. Freer 3 1 Insttute for Appled Computatonal Scence, Harvard Unversty, Cambrdge, Massachusetts 02138, USA 2 The Meda Laboratory,
Physics 207 Lecture 27
 hyscs 07 Lecture 7 hyscs 07, Lecture 7, Dec. 6 Agenda: h. 0, st Law o Thermodynamcs, h. st Law o thermodynamcs ( U Q + W du dq + dw ) Work done by an deal gas n a ston Introducton to thermodynamc cycles
hyscs 07 Lecture 7 hyscs 07, Lecture 7, Dec. 6 Agenda: h. 0, st Law o Thermodynamcs, h. st Law o thermodynamcs ( U Q + W du dq + dw ) Work done by an deal gas n a ston Introducton to thermodynamc cycles
SIO 224. m(r) =(ρ(r),k s (r),µ(r))
 SIO 224 1. A bref look at resoluton analyss Here s some background for the Masters and Gubbns resoluton paper. Global Earth models are usually found teratvely by assumng a startng model and fndng small
SIO 224 1. A bref look at resoluton analyss Here s some background for the Masters and Gubbns resoluton paper. Global Earth models are usually found teratvely by assumng a startng model and fndng small
Lecture 10 Support Vector Machines II
 Lecture 10 Support Vector Machnes II 22 February 2016 Taylor B. Arnold Yale Statstcs STAT 365/665 1/28 Notes: Problem 3 s posted and due ths upcomng Frday There was an early bug n the fake-test data; fxed
Lecture 10 Support Vector Machnes II 22 February 2016 Taylor B. Arnold Yale Statstcs STAT 365/665 1/28 Notes: Problem 3 s posted and due ths upcomng Frday There was an early bug n the fake-test data; fxed
Linear Momentum. Center of Mass.
 Lecture 6 Chapter 9 Physcs I 03.3.04 Lnear omentum. Center of ass. Course webste: http://faculty.uml.edu/ndry_danylov/teachng/physcsi Lecture Capture: http://echo360.uml.edu/danylov03/physcssprng.html
Lecture 6 Chapter 9 Physcs I 03.3.04 Lnear omentum. Center of ass. Course webste: http://faculty.uml.edu/ndry_danylov/teachng/physcsi Lecture Capture: http://echo360.uml.edu/danylov03/physcssprng.html
CS286r Assign One. Answer Key
 CS286r Assgn One Answer Key 1 Game theory 1.1 1.1.1 Let off-equlbrum strateges also be that people contnue to play n Nash equlbrum. Devatng from any Nash equlbrum s a weakly domnated strategy. That s,
CS286r Assgn One Answer Key 1 Game theory 1.1 1.1.1 Let off-equlbrum strateges also be that people contnue to play n Nash equlbrum. Devatng from any Nash equlbrum s a weakly domnated strategy. That s,
Chapter 9: Statistical Inference and the Relationship between Two Variables
 Chapter 9: Statstcal Inference and the Relatonshp between Two Varables Key Words The Regresson Model The Sample Regresson Equaton The Pearson Correlaton Coeffcent Learnng Outcomes After studyng ths chapter,
Chapter 9: Statstcal Inference and the Relatonshp between Two Varables Key Words The Regresson Model The Sample Regresson Equaton The Pearson Correlaton Coeffcent Learnng Outcomes After studyng ths chapter,
Study Guide For Exam Two
 Study Gude For Exam Two Physcs 2210 Albretsen Updated: 08/02/2018 All Other Prevous Study Gudes Modules 01-06 Module 07 Work Work done by a constant force F over a dstance s : Work done by varyng force
Study Gude For Exam Two Physcs 2210 Albretsen Updated: 08/02/2018 All Other Prevous Study Gudes Modules 01-06 Module 07 Work Work done by a constant force F over a dstance s : Work done by varyng force
PHYSICS 391 FALL 2017 QUANTUM INFORMATION THEORY I
 PHYSICS 391 FALL 2017 QUANTUM INFORMATION THEORY I Jean Well INDEPENDENT STUDY PAPER Professor S.G. Rajeev 10 th December 2017 1 Introducton A method s more mportant than a dscovery, snce the rght method
PHYSICS 391 FALL 2017 QUANTUM INFORMATION THEORY I Jean Well INDEPENDENT STUDY PAPER Professor S.G. Rajeev 10 th December 2017 1 Introducton A method s more mportant than a dscovery, snce the rght method
Week 8: Chapter 9. Linear Momentum. Newton Law and Momentum. Linear Momentum, cont. Conservation of Linear Momentum. Conservation of Momentum, 2
 Lnear omentum Week 8: Chapter 9 Lnear omentum and Collsons The lnear momentum of a partcle, or an object that can be modeled as a partcle, of mass m movng wth a velocty v s defned to be the product of
Lnear omentum Week 8: Chapter 9 Lnear omentum and Collsons The lnear momentum of a partcle, or an object that can be modeled as a partcle, of mass m movng wth a velocty v s defned to be the product of
Statistical mechanics handout 4
 Statstcal mechancs handout 4 Explan dfference between phase space and an. Ensembles As dscussed n handout three atoms n any physcal system can adopt any one of a large number of mcorstates. For a quantum
Statstcal mechancs handout 4 Explan dfference between phase space and an. Ensembles As dscussed n handout three atoms n any physcal system can adopt any one of a large number of mcorstates. For a quantum
7 Stellar Structure III. introduc)on to Astrophysics, C. Bertulani, Texas A&M-Commerce 1
 7 Stellar Structure III ntroduc)on to Astrophyscs, C. Bertulan, Texas A&M-Commerce 1 Fundamental physcal constants a radaton densty constant 7.55 10-16 J m -3 K -4 c velocty of lght 3.00 10 8 m s -1 G
7 Stellar Structure III ntroduc)on to Astrophyscs, C. Bertulan, Texas A&M-Commerce 1 Fundamental physcal constants a radaton densty constant 7.55 10-16 J m -3 K -4 c velocty of lght 3.00 10 8 m s -1 G
NUMERICAL DIFFERENTIATION
 NUMERICAL DIFFERENTIATION 1 Introducton Dfferentaton s a method to compute the rate at whch a dependent output y changes wth respect to the change n the ndependent nput x. Ths rate of change s called the
NUMERICAL DIFFERENTIATION 1 Introducton Dfferentaton s a method to compute the rate at whch a dependent output y changes wth respect to the change n the ndependent nput x. Ths rate of change s called the
Lecture 5: Ideal monatomic gas
 Lecture 5: Ideal monatomc gas Statstcal mechancs of the perfect gas Ams: Key new concepts and methods: Countng states Waves n a box. Demonstraton that β / kt Heat, work and Entropy n statstcal mechancs
Lecture 5: Ideal monatomc gas Statstcal mechancs of the perfect gas Ams: Key new concepts and methods: Countng states Waves n a box. Demonstraton that β / kt Heat, work and Entropy n statstcal mechancs
First Law: A body at rest remains at rest, a body in motion continues to move at constant velocity, unless acted upon by an external force.
 Secton 1. Dynamcs (Newton s Laws of Moton) Two approaches: 1) Gven all the forces actng on a body, predct the subsequent (changes n) moton. 2) Gven the (changes n) moton of a body, nfer what forces act
Secton 1. Dynamcs (Newton s Laws of Moton) Two approaches: 1) Gven all the forces actng on a body, predct the subsequent (changes n) moton. 2) Gven the (changes n) moton of a body, nfer what forces act
PHYS 705: Classical Mechanics. Newtonian Mechanics
 1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
Period & Frequency. Work and Energy. Methods of Energy Transfer: Energy. Work-KE Theorem 3/4/16. Ranking: Which has the greatest kinetic energy?
 Perod & Frequency Perod (T): Tme to complete one ull rotaton Frequency (): Number o rotatons completed per second. = 1/T, T = 1/ v = πr/t Work and Energy Work: W = F!d (pcks out parallel components) F
Perod & Frequency Perod (T): Tme to complete one ull rotaton Frequency (): Number o rotatons completed per second. = 1/T, T = 1/ v = πr/t Work and Energy Work: W = F!d (pcks out parallel components) F
U-Pb Geochronology Practical: Background
 U-Pb Geochronology Practcal: Background Basc Concepts: accuracy: measure of the dfference between an expermental measurement and the true value precson: measure of the reproducblty of the expermental result
U-Pb Geochronology Practcal: Background Basc Concepts: accuracy: measure of the dfference between an expermental measurement and the true value precson: measure of the reproducblty of the expermental result
PES 1120 Spring 2014, Spendier Lecture 6/Page 1
 PES 110 Sprng 014, Spender Lecture 6/Page 1 Lecture today: Chapter 1) Electrc feld due to charge dstrbutons -> charged rod -> charged rng We ntroduced the electrc feld, E. I defned t as an nvsble aura
PES 110 Sprng 014, Spender Lecture 6/Page 1 Lecture today: Chapter 1) Electrc feld due to charge dstrbutons -> charged rod -> charged rng We ntroduced the electrc feld, E. I defned t as an nvsble aura
Axial Turbine Analysis
 Axal Turbne Analyss From Euler turbomachnery (conservaton) equatons need to Nole understand change n tangental velocty to relate to forces on blades and power m W m rc e rc uc uc e Analye flow n a plane
Axal Turbne Analyss From Euler turbomachnery (conservaton) equatons need to Nole understand change n tangental velocty to relate to forces on blades and power m W m rc e rc uc uc e Analye flow n a plane
CHAPTER 14 GENERAL PERTURBATION THEORY
 CHAPTER 4 GENERAL PERTURBATION THEORY 4 Introducton A partcle n orbt around a pont mass or a sphercally symmetrc mass dstrbuton s movng n a gravtatonal potental of the form GM / r In ths potental t moves
CHAPTER 4 GENERAL PERTURBATION THEORY 4 Introducton A partcle n orbt around a pont mass or a sphercally symmetrc mass dstrbuton s movng n a gravtatonal potental of the form GM / r In ths potental t moves
CSci 6974 and ECSE 6966 Math. Tech. for Vision, Graphics and Robotics Lecture 21, April 17, 2006 Estimating A Plane Homography
 CSc 6974 and ECSE 6966 Math. Tech. for Vson, Graphcs and Robotcs Lecture 21, Aprl 17, 2006 Estmatng A Plane Homography Overvew We contnue wth a dscusson of the major ssues, usng estmaton of plane projectve
CSc 6974 and ECSE 6966 Math. Tech. for Vson, Graphcs and Robotcs Lecture 21, Aprl 17, 2006 Estmatng A Plane Homography Overvew We contnue wth a dscusson of the major ssues, usng estmaton of plane projectve
The partition function is so important because everything else one ever wants (and can) know about the macroscopic system can be calculated from it:
 5. Thermochemstry 5.. Molecular partton functon and thermodynamc quanttes One of the man goals of computatonal chemstry s to calculate macroscopc chemcal quanttes that we observe n the laboratory (e.g.
5. Thermochemstry 5.. Molecular partton functon and thermodynamc quanttes One of the man goals of computatonal chemstry s to calculate macroscopc chemcal quanttes that we observe n the laboratory (e.g.
Introduction to Vapor/Liquid Equilibrium, part 2. Raoult s Law:
 CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
The Second Anti-Mathima on Game Theory
 The Second Ant-Mathma on Game Theory Ath. Kehagas December 1 2006 1 Introducton In ths note we wll examne the noton of game equlbrum for three types of games 1. 2-player 2-acton zero-sum games 2. 2-player
The Second Ant-Mathma on Game Theory Ath. Kehagas December 1 2006 1 Introducton In ths note we wll examne the noton of game equlbrum for three types of games 1. 2-player 2-acton zero-sum games 2. 2-player
Kernel Methods and SVMs Extension
 Kernel Methods and SVMs Extenson The purpose of ths document s to revew materal covered n Machne Learnng 1 Supervsed Learnng regardng support vector machnes (SVMs). Ths document also provdes a general
Kernel Methods and SVMs Extenson The purpose of ths document s to revew materal covered n Machne Learnng 1 Supervsed Learnng regardng support vector machnes (SVMs). Ths document also provdes a general
Physics 53. Rotational Motion 3. Sir, I have found you an argument, but I am not obliged to find you an understanding.
 Physcs 53 Rotatonal Moton 3 Sr, I have found you an argument, but I am not oblged to fnd you an understandng. Samuel Johnson Angular momentum Wth respect to rotatonal moton of a body, moment of nerta plays
Physcs 53 Rotatonal Moton 3 Sr, I have found you an argument, but I am not oblged to fnd you an understandng. Samuel Johnson Angular momentum Wth respect to rotatonal moton of a body, moment of nerta plays
Optimal information storage in noisy synapses under resource constraints
 Optmal nformaton storage n nosy synapses under resource constrants Lav arshney MIT Jesper Sjöström Brandes, UCL London Dmtr Mtya Chklovsk Cold Sprng Harbor Laboratory Modfcatons of synaptc connectons store
Optmal nformaton storage n nosy synapses under resource constrants Lav arshney MIT Jesper Sjöström Brandes, UCL London Dmtr Mtya Chklovsk Cold Sprng Harbor Laboratory Modfcatons of synaptc connectons store
Notes on Analytical Dynamics
 Notes on Analytcal Dynamcs Jan Peters & Mchael Mstry October 7, 004 Newtonan Mechancs Basc Asssumptons and Newtons Laws Lonely pontmasses wth postve mass Newtons st: Constant velocty v n an nertal frame
Notes on Analytcal Dynamcs Jan Peters & Mchael Mstry October 7, 004 Newtonan Mechancs Basc Asssumptons and Newtons Laws Lonely pontmasses wth postve mass Newtons st: Constant velocty v n an nertal frame
= z 20 z n. (k 20) + 4 z k = 4
 Problem Set #7 solutons 7.2.. (a Fnd the coeffcent of z k n (z + z 5 + z 6 + z 7 + 5, k 20. We use the known seres expanson ( n+l ( z l l z n below: (z + z 5 + z 6 + z 7 + 5 (z 5 ( + z + z 2 + z + 5 5
Problem Set #7 solutons 7.2.. (a Fnd the coeffcent of z k n (z + z 5 + z 6 + z 7 + 5, k 20. We use the known seres expanson ( n+l ( z l l z n below: (z + z 5 + z 6 + z 7 + 5 (z 5 ( + z + z 2 + z + 5 5
The Geometry of Logit and Probit
 The Geometry of Logt and Probt Ths short note s meant as a supplement to Chapters and 3 of Spatal Models of Parlamentary Votng and the notaton and reference to fgures n the text below s to those two chapters.
The Geometry of Logt and Probt Ths short note s meant as a supplement to Chapters and 3 of Spatal Models of Parlamentary Votng and the notaton and reference to fgures n the text below s to those two chapters.
Chapter 3 and Chapter 4
 Chapter 3 and Chapter 4 Chapter 3 Energy 3. Introducton:Work Work W s energy transerred to or rom an object by means o a orce actng on the object. Energy transerred to the object s postve work, and energy
Chapter 3 and Chapter 4 Chapter 3 Energy 3. Introducton:Work Work W s energy transerred to or rom an object by means o a orce actng on the object. Energy transerred to the object s postve work, and energy
Lecture Notes on Linear Regression
 Lecture Notes on Lnear Regresson Feng L fl@sdueducn Shandong Unversty, Chna Lnear Regresson Problem In regresson problem, we am at predct a contnuous target value gven an nput feature vector We assume
Lecture Notes on Lnear Regresson Feng L fl@sdueducn Shandong Unversty, Chna Lnear Regresson Problem In regresson problem, we am at predct a contnuous target value gven an nput feature vector We assume
II.D Many Random Variables
 II.D Many Random Varables Wth more than one random varable, the set of outcomes s an -dmensonal space, S x = { < x 1, x 2,, x < }. For example, descrbng the locaton and velocty of a gas partcle requres
II.D Many Random Varables Wth more than one random varable, the set of outcomes s an -dmensonal space, S x = { < x 1, x 2,, x < }. For example, descrbng the locaton and velocty of a gas partcle requres
Chapter 7. Potential Energy and Conservation of Energy
 Chapter 7 Potental Energy and Conservaton o Energy 1 Forms o Energy There are many orms o energy, but they can all be put nto two categores Knetc Knetc energy s energy o moton Potental Potental energy
Chapter 7 Potental Energy and Conservaton o Energy 1 Forms o Energy There are many orms o energy, but they can all be put nto two categores Knetc Knetc energy s energy o moton Potental Potental energy
...Thermodynamics. If Clausius Clapeyron fails. l T (v 2 v 1 ) = 0/0 Second order phase transition ( S, v = 0)
 If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
Chapter 11: Simple Linear Regression and Correlation
 Chapter 11: Smple Lnear Regresson and Correlaton 11-1 Emprcal Models 11-2 Smple Lnear Regresson 11-3 Propertes of the Least Squares Estmators 11-4 Hypothess Test n Smple Lnear Regresson 11-4.1 Use of t-tests
Chapter 11: Smple Lnear Regresson and Correlaton 11-1 Emprcal Models 11-2 Smple Lnear Regresson 11-3 Propertes of the Least Squares Estmators 11-4 Hypothess Test n Smple Lnear Regresson 11-4.1 Use of t-tests
Gravitational Acceleration: A case of constant acceleration (approx. 2 hr.) (6/7/11)
 Gravtatonal Acceleraton: A case of constant acceleraton (approx. hr.) (6/7/11) Introducton The gravtatonal force s one of the fundamental forces of nature. Under the nfluence of ths force all objects havng
Gravtatonal Acceleraton: A case of constant acceleraton (approx. hr.) (6/7/11) Introducton The gravtatonal force s one of the fundamental forces of nature. Under the nfluence of ths force all objects havng
Physics 5153 Classical Mechanics. Principle of Virtual Work-1
 P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
Week3, Chapter 4. Position and Displacement. Motion in Two Dimensions. Instantaneous Velocity. Average Velocity
 Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructions
 THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
NP-Completeness : Proofs
 NP-Completeness : Proofs Proof Methods A method to show a decson problem Π NP-complete s as follows. (1) Show Π NP. (2) Choose an NP-complete problem Π. (3) Show Π Π. A method to show an optmzaton problem
NP-Completeness : Proofs Proof Methods A method to show a decson problem Π NP-complete s as follows. (1) Show Π NP. (2) Choose an NP-complete problem Π. (3) Show Π Π. A method to show an optmzaton problem
4 Analysis of Variance (ANOVA) 5 ANOVA. 5.1 Introduction. 5.2 Fixed Effects ANOVA
 4 Analyss of Varance (ANOVA) 5 ANOVA 51 Introducton ANOVA ANOVA s a way to estmate and test the means of multple populatons We wll start wth one-way ANOVA If the populatons ncluded n the study are selected
4 Analyss of Varance (ANOVA) 5 ANOVA 51 Introducton ANOVA ANOVA s a way to estmate and test the means of multple populatons We wll start wth one-way ANOVA If the populatons ncluded n the study are selected
Hidden Markov Models & The Multivariate Gaussian (10/26/04)
 CS281A/Stat241A: Statstcal Learnng Theory Hdden Markov Models & The Multvarate Gaussan (10/26/04) Lecturer: Mchael I. Jordan Scrbes: Jonathan W. Hu 1 Hdden Markov Models As a bref revew, hdden Markov models
CS281A/Stat241A: Statstcal Learnng Theory Hdden Markov Models & The Multvarate Gaussan (10/26/04) Lecturer: Mchael I. Jordan Scrbes: Jonathan W. Hu 1 Hdden Markov Models As a bref revew, hdden Markov models
Linear Approximation with Regularization and Moving Least Squares
 Lnear Approxmaton wth Regularzaton and Movng Least Squares Igor Grešovn May 007 Revson 4.6 (Revson : March 004). 5 4 3 0.5 3 3.5 4 Contents: Lnear Fttng...4. Weghted Least Squares n Functon Approxmaton...
Lnear Approxmaton wth Regularzaton and Movng Least Squares Igor Grešovn May 007 Revson 4.6 (Revson : March 004). 5 4 3 0.5 3 3.5 4 Contents: Lnear Fttng...4. Weghted Least Squares n Functon Approxmaton...
Maximum Likelihood Estimation
 Maxmum Lkelhood Estmaton INFO-2301: Quanttatve Reasonng 2 Mchael Paul and Jordan Boyd-Graber MARCH 7, 2017 INFO-2301: Quanttatve Reasonng 2 Paul and Boyd-Graber Maxmum Lkelhood Estmaton 1 of 9 Why MLE?
Maxmum Lkelhood Estmaton INFO-2301: Quanttatve Reasonng 2 Mchael Paul and Jordan Boyd-Graber MARCH 7, 2017 INFO-2301: Quanttatve Reasonng 2 Paul and Boyd-Graber Maxmum Lkelhood Estmaton 1 of 9 Why MLE?
Department of Statistics University of Toronto STA305H1S / 1004 HS Design and Analysis of Experiments Term Test - Winter Solution
 Department of Statstcs Unversty of Toronto STA35HS / HS Desgn and Analyss of Experments Term Test - Wnter - Soluton February, Last Name: Frst Name: Student Number: Instructons: Tme: hours. Ads: a non-programmable
Department of Statstcs Unversty of Toronto STA35HS / HS Desgn and Analyss of Experments Term Test - Wnter - Soluton February, Last Name: Frst Name: Student Number: Instructons: Tme: hours. Ads: a non-programmable
χ x B E (c) Figure 2.1.1: (a) a material particle in a body, (b) a place in space, (c) a configuration of the body
 Secton.. Moton.. The Materal Body and Moton hyscal materals n the real world are modeled usng an abstract mathematcal entty called a body. Ths body conssts of an nfnte number of materal partcles. Shown
Secton.. Moton.. The Materal Body and Moton hyscal materals n the real world are modeled usng an abstract mathematcal entty called a body. Ths body conssts of an nfnte number of materal partcles. Shown
6. Stochastic processes (2)
 Contents Markov processes Brth-death processes Lect6.ppt S-38.45 - Introducton to Teletraffc Theory Sprng 5 Markov process Consder a contnuous-tme and dscrete-state stochastc process X(t) wth state space
Contents Markov processes Brth-death processes Lect6.ppt S-38.45 - Introducton to Teletraffc Theory Sprng 5 Markov process Consder a contnuous-tme and dscrete-state stochastc process X(t) wth state space
