Frieder Mugele. Physics of Complex Fluids. University of Twente. Jacco Snoeier Physics of Fluids / UT
|
|
|
- Eileen Johns
- 5 years ago
- Views:
Transcription
1 coorganizers: Frieder Mugele Physics of Comple Fluids Jacco Snoeier Physics of Fluids / UT University of Twente Anton Darhuber Mesoscopic Transport Phenomena / Tu/e speakers: José Bico (ESPCI Paris) Daniel Bonn (UvA) Michiel Kreutzer (TUD) Ralph Lindken (TUD)
2 program Monday: :00 3:00h registration + lunch 3:00h welcome: Frieder Mugele 3:5h 4:00h Frieder Mugele: Wetting basics (Young-Laplace equation; Young equation; eamples) 4:0-5:5h Jacco Snoeijer: Wetting flows: the lubrication approimation 5:5-5:50h coffee break 5:50-6:35h Jacco Snoeijer: Coating flows: the Landau-Levich problem and its solution using asymptotic matching 6:45-7:30h Anton Darhuber: Surface tension, capillary forces and disjoining pressure I Tuesday: 9:00h-9:45h Frieder Mugele: Dewetting 9:550:40 Anton Darhuber: Surface tension, capillary forces and disjoining pressure II 0:40-:05h coffee break :05h-:50h Anton Darhuber: Surface tension-gradient-driven flows :00h-:45h Daniel Bonn: Evaporating drops :45-4:00h lunch 4:00h-4:45h Daniel Bonn: Drop impact 5:55h-5:40h José Bico: Elastocapillarity (I) 5:40-6:05h coffee break 6:05h 6:50h José Bico: Elasticity & Capillarity (II) 8: joint dinner & get together
3 program Wednesday: 9:00h-9:45h Michiel Kreutzer: Two-phase flow in microchannels: the Bretherton problem 9:55h-0:40h Michiel Kreutzer: Drop generation& emulsification in microchannels 0:40h-:05h coffee break :05h-:50h Michiel Kreutzer: Jet instabilities in microchannels :00h-:45h Ralph Lindken: PiV characterization of capillarity-driven flows :45-4:00h lunch 4:00h-5:00h: occasion for ecercises 5:00h-7:00h lab tour (Physics of Comple Fluids / Physics of Fluids) Thursday: 9:00h-9:45h Jacco Snoeijer: Contact line dynamics(i) 9:55h-0:40h Jacco Snoeijer: Contact line dynamics (II) 0:40h-:05h coffee break :05h-:50h Frieder Mugele: Wetting of heterogeneous surfaces: Wenzel, Cassie-Bater :00h-:45h: Jacco Snoeijer: Contact angle hysteresis :45-4:00h lunch 4:00h-4:45h José Bico: Sperhydrophobicity 4:55h-5:40h Anton Darhuber: Thermocapillary flows 5:40h-6:05h coffee break 6:05h-6:50h Anton Darhuber: Surfactant-driven and solutocapillary flows Friday: 9:00h-9:45h Frieder Mugele: Electrowetting: basic principles 9:55h-0:40h Frieder Mugele: Eectrowetting applications. 0:40h-:05h coffee break :05-:00h round up highlights / short summaries by students :00h closure 3
4 principles of wetting and capillarity p + R R κ cosθ Y sv sl capillary (Laplace) equation Young equation 4
5 capillarity-induced instabilities driving force: minimization of surface energy time Rayleigh-Plateau instability 5
6 drops in microchannels drop generation drop dynamics Anna et al. APL 003 6
7 wetting and dewetting flows coating technology dewetting of paint e.g. heating Landau-Levich films 7
8 fundamental flow properties v lubrication flows contact line motion 8
9 wetting & molecular interactions nanoscale drop θ Y 0 vertical scale: 00 nm disjoining pressure 9
10 capillary forces capillary bridges eert mechanical forces 0
11 wetting of comple surfaces superhydrophobic surfaces: the Lotus effect θ
12 switching wettability voltage electrowetting & thermocapillarity
13 lecture : basics of wetting 3
14 wetting & liquid microdroplets 50 µm capillary equation p p L κ Young equation cosθ Y sv sl H. Gau et al. Science 999 4
15 origin of interfacial energy O(Å) width 0: sharp interface model (will be handled throughout this course) range of interactions (O(nm)) surface tension is ecess energy w.r.t. bulk cohesive energy U coh unhappy molecules at interfaces a 5
16 interfacial tension liquid A liquid B AB : interfacial tension interfacial tensions (of immiscible fluids) are always positive 6
17 interfacial tensions matter at small scales fraction of molecules close to the surface: A dr V 3dr r for r cm for r µm r capillarity is crucial for micro- and nanofluidics 7
18 mechanical definition of surface tension definition A: The mechanical work δ W required to create an additional surface area da (e.g. by deforming a drop) is given by the surface tension δ W da thermodynamically: F A T, N, V energy dimension and units: [ ] ; J/m (typically: mj/m ) area 8
19 mechanical definition of surface tension l soap film d e f i n i t i o n definition B: is a force per unit length acting along the liquid-vapor interface aiming to shrink the interfacial area force dimension and units: [ ] ; N/m J/m length (typically: mn/m) connection to definition A work required to move the rod: force per unit length per interface: δw f lδ δw l δ 9
20 surface tension of selected liquids material water (5 C) water (00 C) ethanol decanol heane decane headecane glycerol acetone mercury water/oil surface tension [mj/m ] T-coefficient: ( ) mj / m K 0
21 consequences: the Laplace pressure spherical drop R δr variation of internal energy: P drop P et δ U p dv p dv + mechanical equilibrium: δu ( p p ) dv + da 0 p L p et drop drop drop p et drop drop et da dv drop et! dv da et dv drop Laplace pressure: p L R
22 generalization to arbitrary surfaces upon crossing an interface between two fluids with an interfacial tension s, the pressure increases by Young-Laplace law κ + R R p L κ: mean curvature + R R κ R, R : principal radii of curvature (sphere: R R )
23 principle radii of curvature sign convention: air ϕ n r R > 0 R < 0 n r mean curvature: κ R + R liquid (κ is independent of azimuthal angle φ) 3
24 generalization to arbitrary surfaces upon crossing an interface between two fluids with an interfacial tension s, the pressure increases by p L κ R + R Young-Laplace law κ: mean curvature κ + R R R, R : principal radii of curvature (sphere: R R ) consequence: liquid surfaces in mechanical equilibrium have a constant mean curvature (n the absence of other forces) 50 µm H. Gau et al. Science 999 4
25 variational derivation of Laplace equation equilibrium surface profile minimum of Gibbs free energy (at constant volume) G ( F p ) min V surf! pressure: Lagrange multiplier F surf : functional of surface profile A: F surf [ A] da eplicit representation of surface: z z(, y) da r da r r s s y + ( z) + ( yz) y F surf [ A] da + volume: V z (, y) d dy ( z) + ( z) d dy y s r 0 z r s y 0 y z y y 5
26 functional minimization { ( ) ( ) }! + z + z p z d min G[ z(, y)] dy y f ( z, z, z) y Euler-Lagrange equation: d d f + d dy f ( z) ( z) y f z 0 f d d ( z) S z S z % z S z z ( z z + z z) S y y / S ( z ( + ( z) + ( z) ) z( z z + z z ) S 3 y y y d d d dy ( z) ( z ( + ( z) ) z z z) f 3 S y symmetrically: ( z) y ( z ( + ( z) ) z z z) f 3 S yy y y y y f z p 6
27 Young Laplace equation mean curvature κ R + R z( + ( y z) ) ( ( + ( z)( z) y z) ( + ( y y z) z) + ) 3/ yy z( + ( z) ) p non-linear second order partial differential equation z two-dimensional version: p 3 + ( z) 7
28 cylindrical coordinates surface parameterization: r r( ϕ, z) S + r + ( r) ϕ z r volume: V dv dz dr r dϕ dz dϕ r area: A da dz r dϕ S( r, ϕ) cylindrical symmetry: r 0 r r( z) ϕ p r S S r 3 zz S + ( zr) ordinary differential equation 8
29 an eample fiber immersed in water (complete wetting; no gravity) z0 radius R r 0 Sr r zz 3 S z BCs: r : κ 0 r R: r 0 S + ( r) z 0 r' ' 3 + ' r r' dz + r' r + r' d r r + r' const. R r' ( r / R) > 0 solution: z>> R r ( z) R cosh( z / R) ep( z / R) 9
30 three phase equilibrium: wetting θ π 0 < θ < π θ 0 θ sl sv non-wetting partial wetting complete wetting sl : solid-liquid interfacial energy; sv (solid-vapor); (liquid-vapor) 30
31 spreading parameter controls wetting behavior partial wetting complete wetting spreading parameter S [ ] ( + ) A F init F final sv sl S > 0 : complete wetting S < 0 : partial wetting 3
32 contact angle in partial wetting situation sv sl θ Y d cos θ θ Y d (horizontal) force balance energy minimization sv + cosθ δw { + cos θ } d 0 sl Y sl Y sv Young equation cosθ Y sv sl v : vapor or second immiscible liquid 3
33 connecting wetting behavior & surface properties S sv ( + sl ) > 0 : complete < 0 : partial wetting wetting high energy surfaces (metals, ionic crystals, covalent materials ) are usually wetted sv E coh a mj m low energy surfaces (polymers, molecular crystals) are usually partially wetted sv k B T mj a m How to relate wetting behavior to microscopic interaction energies? 33
34 Gedankeneperiment A A A A d 0 A A B B δw U final U init Av 0 VAA( ) VAA( d0) δ W + Av Bv AB (III) Av V AA ( d0) ( d ) ( ) Bv V BB 0 (I) (II) V AB V AB ( ) VAB( d0) ( d 0 ) 34
35 Gedankeneperiment (II) Av Av V AA ( d0) + Bv Bv V BB ( d0) AB V AB ( d 0 ) (I) (II) (III) A: solid; B: liquid (III)-(II) sv ( + sl ) S Vll ( d0) Vsl ( d0) binding energies: <0 V ll > V S sl < 0 partial wetting V sl > V S ll > 0 complete wetting van der Waals interaction: S α l ( α s αl ) Vsl α s α l Vll α complete wetting if solid more polarisable than liquid l 35
36 wetting and gravity hydrostatic pressure z g h 0 -z ( 0 p + ρ g( h z)) da κ da Young equation cosθ Y sv sl now κκ(z) capillary equation κ p + ρg( h 0 z) 36
37 non-dimensionalization dimensionless variables: z R ~ z R ~ p ~ p R κ κ ~ R κ ~ ~ p + ρ g R ~ ( h 0 ~ z ) ~ p + Bo ~ ( h 0 ~ z ) Bo: Bond number Bo << gravity negligible equivalently: capillary length λ c ρ g / R << λ c gravity negligible water in air: λ.7mm gravity is usually negligible in microfluidics 37
38 summary equilibrium shape of wetting structures is determined by minimum of surface energy variation of free energy functional results in p + R R κ cosθ Y sv sl capillary (Laplace) equation Young equation occurrence of complete vs. partial wetting is determined by relative strength of adhesive vs. cohesive forces gravity is negligible on length scales << capillary length λ c ρ g / O( mm) 38
Jacco Snoeijer PHYSICS OF FLUIDS
 Jacco Snoeijer PHYSICS OF FLUIDS dynamics dynamics freezing dynamics freezing microscopics of capillarity Menu 1. surface tension: thermodynamics & microscopics 2. wetting (statics): thermodynamics & microscopics
Jacco Snoeijer PHYSICS OF FLUIDS dynamics dynamics freezing dynamics freezing microscopics of capillarity Menu 1. surface tension: thermodynamics & microscopics 2. wetting (statics): thermodynamics & microscopics
Microfluidics 2 Surface tension, contact angle, capillary flow
 MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
Surface and Interfacial Tensions. Lecture 1
 Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
Capillarity and Wetting Phenomena
 ? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
DLVO interaction between the spheres
 DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
Lecture 7 Contact angle phenomena and wetting
 Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
emulsions, and foams March 21 22, 2009
 Wetting and adhesion Dispersions in liquids: suspensions, emulsions, and foams ACS National Meeting March 21 22, 2009 Salt Lake City Ian Morrison 2009 Ian Morrison 2009 Lecure 2 - Wetting and adhesion
Wetting and adhesion Dispersions in liquids: suspensions, emulsions, and foams ACS National Meeting March 21 22, 2009 Salt Lake City Ian Morrison 2009 Ian Morrison 2009 Lecure 2 - Wetting and adhesion
Interfaces and interfacial energy
 Interfaces and interfacial energy 1/14 kinds: l/g }{{ l/l } mobile s/g s/l s/s Example. Estimate the percetage of water molecules on the surface of a fog droplet of diameter (i) 0.1 mm (naked eye visibility
Interfaces and interfacial energy 1/14 kinds: l/g }{{ l/l } mobile s/g s/l s/s Example. Estimate the percetage of water molecules on the surface of a fog droplet of diameter (i) 0.1 mm (naked eye visibility
drops in motion Frieder Mugele the physics of electrowetting and its applications Physics of Complex Fluids University of Twente
 drops in motion the physics of electrowetting and its applications Frieder Mugele Physics of Complex Fluids niversity of Twente 1 electrowetting: the switch on the wettability voltage outline q q q q q
drops in motion the physics of electrowetting and its applications Frieder Mugele Physics of Complex Fluids niversity of Twente 1 electrowetting: the switch on the wettability voltage outline q q q q q
INTERFACIAL PHENOMENA GRADING SCHEME
 18.357 INTERFACIAL PHENOMENA Professor John W. M. Bush Fall 2010 Office 2-346 MW 2-3:30 Phone: 253-4387 (office) Room 2-135 email: bush@math.mit.edu Office hours: after class, available upon request GRADING
18.357 INTERFACIAL PHENOMENA Professor John W. M. Bush Fall 2010 Office 2-346 MW 2-3:30 Phone: 253-4387 (office) Room 2-135 email: bush@math.mit.edu Office hours: after class, available upon request GRADING
Praktikum zur. Materialanalytik
 Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
5.2 Surface Tension Capillary Pressure: The Young-Laplace Equation. Figure 5.1 Origin of surface tension at liquid-vapor interface.
 5.2.1 Capillary Pressure: The Young-Laplace Equation Vapor Fo Fs Fs Fi Figure 5.1 Origin of surface tension at liquid-vapor interface. Liquid 1 5.2.1 Capillary Pressure: The Young-Laplace Equation Figure
5.2.1 Capillary Pressure: The Young-Laplace Equation Vapor Fo Fs Fs Fi Figure 5.1 Origin of surface tension at liquid-vapor interface. Liquid 1 5.2.1 Capillary Pressure: The Young-Laplace Equation Figure
Multiphase Flow and Heat Transfer
 Multiphase Flow and Heat Transfer ME546 -Sudheer Siddapureddy sudheer@iitp.ac.in Surface Tension The free surface between air and water at a molecular scale Molecules sitting at a free liquid surface against
Multiphase Flow and Heat Transfer ME546 -Sudheer Siddapureddy sudheer@iitp.ac.in Surface Tension The free surface between air and water at a molecular scale Molecules sitting at a free liquid surface against
Superhydrophobic surfaces. José Bico PMMH-ESPCI, Paris
 Superhydrophobic surfaces José Bico PMMH-ESPCI, Paris Superhydrophobic surfaces José Bico PMMH-ESPCI, Paris? Rain droplet on a window film pinning tear 180? mercury calefaction Leidenfrost point, T = 150
Superhydrophobic surfaces José Bico PMMH-ESPCI, Paris Superhydrophobic surfaces José Bico PMMH-ESPCI, Paris? Rain droplet on a window film pinning tear 180? mercury calefaction Leidenfrost point, T = 150
Capillarity. ESS5855 Lecture Fall 2010
 Capillarity ESS5855 Lecture Fall 2010 Capillarity: the tendency of a liquid in a narrow tube or pore to rise or fall as a result of surface tension (The concise Oxford Dictionary) Surface tension: the
Capillarity ESS5855 Lecture Fall 2010 Capillarity: the tendency of a liquid in a narrow tube or pore to rise or fall as a result of surface tension (The concise Oxford Dictionary) Surface tension: the
Fluid Mechanics Introduction
 Fluid Mechanics Introduction Fluid mechanics study the fluid under all conditions of rest and motion. Its approach is analytical, mathematical, and empirical (experimental and observation). Fluid can be
Fluid Mechanics Introduction Fluid mechanics study the fluid under all conditions of rest and motion. Its approach is analytical, mathematical, and empirical (experimental and observation). Fluid can be
Physics and Chemistry of Interfaces
 Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces.
 Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák & István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry http://kolloid.unideb.hu/~kolloid/
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák & István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry http://kolloid.unideb.hu/~kolloid/
Colloidal Particles at Liquid Interfaces: An Introduction
 1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
contact line dynamics
 contact line dynamics Jacco Snoeijer Physics of Fluids - University of Twente sliding drops flow near contact line static contact line Ingbrigtsen & Toxvaerd (2007) γ γ sv θ e γ sl molecular scales macroscopic
contact line dynamics Jacco Snoeijer Physics of Fluids - University of Twente sliding drops flow near contact line static contact line Ingbrigtsen & Toxvaerd (2007) γ γ sv θ e γ sl molecular scales macroscopic
Interfacial Flows of Contact Line Dynamics and Liquid Displacement in a Circular Microchannel
 Proceedings of the 3 rd World Congress on Mechanical, Chemical, and Material Engineering (MCM'17) Rome, Italy June 8 10, 2017 Paper No. HTFF 159 ISSN: 2369-8136 DOI: 10.11159/htff17.159 Interfacial Flows
Proceedings of the 3 rd World Congress on Mechanical, Chemical, and Material Engineering (MCM'17) Rome, Italy June 8 10, 2017 Paper No. HTFF 159 ISSN: 2369-8136 DOI: 10.11159/htff17.159 Interfacial Flows
Wetting of complex functional surfaces
 Wetting of complex functional surfaces Frieder Mugele Physics of Complex Fluids University of Twente Twente 2 Twente University the entrepreneurial university founded in 1961 part of 3TU federation of
Wetting of complex functional surfaces Frieder Mugele Physics of Complex Fluids University of Twente Twente 2 Twente University the entrepreneurial university founded in 1961 part of 3TU federation of
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa.
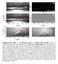 Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Instabilities in the Flow of Thin Liquid Films
 Instabilities in the Flow of Thin Liquid Films Lou Kondic Department of Mathematical Sciences Center for Applied Mathematics and Statistics New Jersey Institute of Technology Presented at Annual Meeting
Instabilities in the Flow of Thin Liquid Films Lou Kondic Department of Mathematical Sciences Center for Applied Mathematics and Statistics New Jersey Institute of Technology Presented at Annual Meeting
Four-phase merging in sessile compound drops
 J. Fluid Mech. (00), vol. 45, pp. 4 40. c 00 Cambridge University Press DOI: 0.07/S000000708 Printed in the United Kingdom 4 Four-phase merging in sessile compound drops By L. M A H A D E V A N, M. A D
J. Fluid Mech. (00), vol. 45, pp. 4 40. c 00 Cambridge University Press DOI: 0.07/S000000708 Printed in the United Kingdom 4 Four-phase merging in sessile compound drops By L. M A H A D E V A N, M. A D
8.2 Surface phenomenon of liquid. Out-class reading: Levine p Curved interfaces
 Out-class reading: Levine p. 387-390 13.2 Curved interfaces https://news.cnblogs.com/n/559867/ 8.2.1 Some interesting phenomena 8.2.1 Some interesting phenomena Provided by Prof. Yu-Peng GUO of Jilin
Out-class reading: Levine p. 387-390 13.2 Curved interfaces https://news.cnblogs.com/n/559867/ 8.2.1 Some interesting phenomena 8.2.1 Some interesting phenomena Provided by Prof. Yu-Peng GUO of Jilin
Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen
 Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen Department of Chemical and Biomolecular Engineering Clarkson University Outline
Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen Department of Chemical and Biomolecular Engineering Clarkson University Outline
dewetting driving forces dewetting mechanism? dewetting dynamics? final equilibrium state: drops with θ = θ Y
 dewetting initial state: continuous film of partially wetting liquid final equilibrium state: drops wit θ = θ Y driving forces dewetting mecanism? dewetting dynamics? 1 free energy vs. film tickness water
dewetting initial state: continuous film of partially wetting liquid final equilibrium state: drops wit θ = θ Y driving forces dewetting mecanism? dewetting dynamics? 1 free energy vs. film tickness water
Thermodynamics of a symmetric toroidal section and break up of a fluid torus
 Thermodynamics of a symmetric toroidal section and break up of a fluid torus Philip Lee March 1, 2016 1 Coordinate systems, and notation The toroidal section makes contact angle θ with the substrate, the
Thermodynamics of a symmetric toroidal section and break up of a fluid torus Philip Lee March 1, 2016 1 Coordinate systems, and notation The toroidal section makes contact angle θ with the substrate, the
Wetting & Adhesion on Soft Surfaces Young s Law is dead long live Young s Law. Eric Dufresne
 Wetting & Adhesion on Soft Surfaces Young s Law is dead long live Young s Law Eric Dufresne KITP 2014 Wetting Adhesion 3mm 30 um Young s Law relates contact line geometry and material properties in equilibrium
Wetting & Adhesion on Soft Surfaces Young s Law is dead long live Young s Law Eric Dufresne KITP 2014 Wetting Adhesion 3mm 30 um Young s Law relates contact line geometry and material properties in equilibrium
Module17: Intermolecular Force between Surfaces and Particles. Lecture 23: Intermolecular Force between Surfaces and Particles
 Module17: Intermolecular Force between Surfaces and Particles Lecture 23: Intermolecular Force between Surfaces and Particles 1 We now try to understand the nature of spontaneous instability in a confined
Module17: Intermolecular Force between Surfaces and Particles Lecture 23: Intermolecular Force between Surfaces and Particles 1 We now try to understand the nature of spontaneous instability in a confined
The Origins of Surface and Interfacial Tension
 The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
Mohamed Daoud Claudine E.Williams Editors. Soft Matter Physics. With 177 Figures, 16 of them in colour
 Mohamed Daoud Claudine E.Williams Editors Soft Matter Physics With 177 Figures, 16 of them in colour Contents 1. Droplets: CapiUarity and Wetting 1 By F. Brochard-Wyart (With 35 figures) 1.1 Introduction
Mohamed Daoud Claudine E.Williams Editors Soft Matter Physics With 177 Figures, 16 of them in colour Contents 1. Droplets: CapiUarity and Wetting 1 By F. Brochard-Wyart (With 35 figures) 1.1 Introduction
2. Exploiting wetting phenomena to tailor 1D nano- and microstructures
 2. Exploiting wetting phenomena to tailor 1D nano- and microstructures 2.1 Wetting on a macroscopic scale As a large part of the work in this thesis deals with wetting of porous materials, the phenomena
2. Exploiting wetting phenomena to tailor 1D nano- and microstructures 2.1 Wetting on a macroscopic scale As a large part of the work in this thesis deals with wetting of porous materials, the phenomena
Chapter -6(Section-1) Surface Tension
 Chapter -6(Section-1) Surface Tension Free surface of the liquid tends to minimize the surface area. e.g.(1)if the small quantity of mercury is allowed to fall on the floor, it converted in to small spherical
Chapter -6(Section-1) Surface Tension Free surface of the liquid tends to minimize the surface area. e.g.(1)if the small quantity of mercury is allowed to fall on the floor, it converted in to small spherical
Wetting contact angle
 Wetting contact angle Minh Do-Quang www.flow.kth.se Outline Statics; capillarity and wetting Dynamics; models describing dynamic wetting Hydrodynamics (Tanner-Cox-Voinov law) Molecular kinetics theory
Wetting contact angle Minh Do-Quang www.flow.kth.se Outline Statics; capillarity and wetting Dynamics; models describing dynamic wetting Hydrodynamics (Tanner-Cox-Voinov law) Molecular kinetics theory
Shape of the Interfaces
 NPTEL Chemical Engineering Interfacial Engineering Module : Lecture 3 Shape of the Interfaces Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati 781039 India
NPTEL Chemical Engineering Interfacial Engineering Module : Lecture 3 Shape of the Interfaces Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati 781039 India
NUMERICAL INVESTIGATION OF THERMOCAPILLARY INDUCED MOTION OF A LIQUID SLUG IN A CAPILLARY TUBE
 Proceedings of the Asian Conference on Thermal Sciences 2017, 1st ACTS March 26-30, 2017, Jeju Island, Korea ACTS-P00786 NUMERICAL INVESTIGATION OF THERMOCAPILLARY INDUCED MOTION OF A LIQUID SLUG IN A
Proceedings of the Asian Conference on Thermal Sciences 2017, 1st ACTS March 26-30, 2017, Jeju Island, Korea ACTS-P00786 NUMERICAL INVESTIGATION OF THERMOCAPILLARY INDUCED MOTION OF A LIQUID SLUG IN A
VIII. Phase Transformations. Lecture 38: Nucleation and Spinodal Decomposition
 VIII. Phase Transformations Lecture 38: Nucleation and Spinodal Decomposition MIT Student In this lecture we will study the onset of phase transformation for phases that differ only in their equilibrium
VIII. Phase Transformations Lecture 38: Nucleation and Spinodal Decomposition MIT Student In this lecture we will study the onset of phase transformation for phases that differ only in their equilibrium
8.2 Surface phenomena of liquid. Out-class reading: Levine p Curved interfaces
 Out-class reading: Levine p. 387-390 13.2 Curved interfaces 8.2.1 Some interesting phenomena Evolution of bubbles on porous surface. 8.2.1 Some interesting phenomena Addition of a seed in Supersaturated
Out-class reading: Levine p. 387-390 13.2 Curved interfaces 8.2.1 Some interesting phenomena Evolution of bubbles on porous surface. 8.2.1 Some interesting phenomena Addition of a seed in Supersaturated
Capillary surfaces and complex analysis: new opportunities to study menisci singularities. Mars Alimov, Kazan Federal University, Russia
 Capillary surfaces and complex analysis: new opportunities to study menisci singularities Mars limov Kazan Federal University Russia Kostya Kornev Clemson University SC Outline Intro to wetting and capillarity
Capillary surfaces and complex analysis: new opportunities to study menisci singularities Mars limov Kazan Federal University Russia Kostya Kornev Clemson University SC Outline Intro to wetting and capillarity
Surface Tension and its measurements
 Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
CHAPTER 1 Fluids and their Properties
 FLUID MECHANICS Gaza CHAPTER 1 Fluids and their Properties Dr. Khalil Mahmoud ALASTAL Objectives of this Chapter: Define the nature of a fluid. Show where fluid mechanics concepts are common with those
FLUID MECHANICS Gaza CHAPTER 1 Fluids and their Properties Dr. Khalil Mahmoud ALASTAL Objectives of this Chapter: Define the nature of a fluid. Show where fluid mechanics concepts are common with those
Compound pendant drop tensiometry for. surface tension measurement at zero Bond number
 Compound pendant drop tensiometry for surface tension measurement at zero Bond number Michael J. Neeson, Derek Y. C. Chan,,, and Rico F. Tabor, Department of Mathematics and Statistics, University of Melbourne,
Compound pendant drop tensiometry for surface tension measurement at zero Bond number Michael J. Neeson, Derek Y. C. Chan,,, and Rico F. Tabor, Department of Mathematics and Statistics, University of Melbourne,
PHYSICS OF FLUID SPREADING ON ROUGH SURFACES
 INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
Fluid Mechanics-61341
 An-Najah National University College of Engineering Fluid Mechanics-61341 Chapter [1] Fundamentals 1 The Book (Elementary Fluid Mechanics by Street, Watters and Vennard) Each chapter includes: Concepts
An-Najah National University College of Engineering Fluid Mechanics-61341 Chapter [1] Fundamentals 1 The Book (Elementary Fluid Mechanics by Street, Watters and Vennard) Each chapter includes: Concepts
Generalized Wenzel equation for contact angle of droplets on spherical rough solid substrates
 Science Front Publishers Journal for Foundations and Applications of Physics, 3 (2), (2016) (sciencefront.org) ISSN 2394-3688 Generalized Wenzel equation for contact angle of droplets on spherical rough
Science Front Publishers Journal for Foundations and Applications of Physics, 3 (2), (2016) (sciencefront.org) ISSN 2394-3688 Generalized Wenzel equation for contact angle of droplets on spherical rough
We may have a general idea that a solid is hard and a fluid is soft. This is not satisfactory from
 Chapter 1. Introduction 1.1 Some Characteristics of Fluids We may have a general idea that a solid is hard and a fluid is soft. This is not satisfactory from scientific or engineering point of view. In
Chapter 1. Introduction 1.1 Some Characteristics of Fluids We may have a general idea that a solid is hard and a fluid is soft. This is not satisfactory from scientific or engineering point of view. In
Generalized Cassie-Baxter equation for wetting of a spherical droplet within a smooth and heterogeneous conical cavity
 Science Front Publishers Journal for Foundations and pplications of Physics, vol. 4, No. (017) (sciencefront.org) ISSN 394-3688 Generalized Cassie-axter equation for wetting of a spherical droplet within
Science Front Publishers Journal for Foundations and pplications of Physics, vol. 4, No. (017) (sciencefront.org) ISSN 394-3688 Generalized Cassie-axter equation for wetting of a spherical droplet within
Chemical Potential. Combining the First and Second Laws for a closed system, Considering (extensive properties)
 Chemical Potential Combining the First and Second Laws for a closed system, Considering (extensive properties) du = TdS pdv Hence For an open system, that is, one that can gain or lose mass, U will also
Chemical Potential Combining the First and Second Laws for a closed system, Considering (extensive properties) du = TdS pdv Hence For an open system, that is, one that can gain or lose mass, U will also
Phase Transitions. Ehrenfest Classification of Phase Transitions. Curved Surfaces Bubbles, cavities and droplets Nucleation
 Phase Transitions Chapter 6 of Atkins Sections 6.7-6.10 Ehrenfest Classification of Phase Transitions Physical Liquid Surface Surface Tension Curved Surfaces Bubbles, cavities and droplets Nucleation Capillary
Phase Transitions Chapter 6 of Atkins Sections 6.7-6.10 Ehrenfest Classification of Phase Transitions Physical Liquid Surface Surface Tension Curved Surfaces Bubbles, cavities and droplets Nucleation Capillary
Thickness and Shape of Films Driven by a Marangoni Flow
 Langmuir 1996, 12, 5875-5880 5875 Thickness and Shape of Films Driven by a Marangoni Flow X. Fanton, A. M. Cazabat,* and D. Quéré Laboratoire de Physique de la Matière Condensée, Collège de France, 11
Langmuir 1996, 12, 5875-5880 5875 Thickness and Shape of Films Driven by a Marangoni Flow X. Fanton, A. M. Cazabat,* and D. Quéré Laboratoire de Physique de la Matière Condensée, Collège de France, 11
THE MODIFIED YOUNG S EQUATION FOR THE CONTACT ANGLE OF A SMALL SESSILE DROP FROM AN INTERFACE DISPLACEMENT MODEL
 International Journal of Modern Physics B, Vol. 13, No. 7 (1999) 355 359 c World Scientific Publishing Company THE MODIFIED YOUNG S EQUATION FOR THE CONTACT ANGLE OF A SMALL SESSILE DROP FROM AN INTERFACE
International Journal of Modern Physics B, Vol. 13, No. 7 (1999) 355 359 c World Scientific Publishing Company THE MODIFIED YOUNG S EQUATION FOR THE CONTACT ANGLE OF A SMALL SESSILE DROP FROM AN INTERFACE
of Nebraska - Lincoln
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Xiao Cheng Zeng Publications Published Research - Department of Chemistry 10-1-2006 Homogeneous nucleation at high supersaturation
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Xiao Cheng Zeng Publications Published Research - Department of Chemistry 10-1-2006 Homogeneous nucleation at high supersaturation
IMFA s. intermolecular forces of attraction Chez Chem, LLC All rights reserved.
 IMFA s intermolecular forces of attraction 2014 Chez Chem, LLC All rights reserved. **London Dispersion Forces Also know as Van der Waals forces A momentary non symmetrical electron distribution that can
IMFA s intermolecular forces of attraction 2014 Chez Chem, LLC All rights reserved. **London Dispersion Forces Also know as Van der Waals forces A momentary non symmetrical electron distribution that can
Thin Film Behavior after Ink Transfer in Printing Processes N. Bornemann, H. M. Sauer, E. Dörsam
 Thin Film Behavior after Ink Transfer in Printing Processes N. Bornemann, H. M. Sauer, E. Dörsam 15.04.2010 Institute of Printing Science and Technology Thin Film Behavior N. Bornemann Overview Thin Film
Thin Film Behavior after Ink Transfer in Printing Processes N. Bornemann, H. M. Sauer, E. Dörsam 15.04.2010 Institute of Printing Science and Technology Thin Film Behavior N. Bornemann Overview Thin Film
MPIKG Public Access Author Manuscript
 MPIKG Public Access Author Manuscript Max Planck Institute of of Colloids and Interfaces Author Manuscript Published in final edited form as: Stocco, A., & Möhwald, H. (2015). The influence of long-range
MPIKG Public Access Author Manuscript Max Planck Institute of of Colloids and Interfaces Author Manuscript Published in final edited form as: Stocco, A., & Möhwald, H. (2015). The influence of long-range
Contents. Preface XI Symbols and Abbreviations XIII. 1 Introduction 1
 V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
Chapter 9: Differential Analysis of Fluid Flow
 of Fluid Flow Objectives 1. Understand how the differential equations of mass and momentum conservation are derived. 2. Calculate the stream function and pressure field, and plot streamlines for a known
of Fluid Flow Objectives 1. Understand how the differential equations of mass and momentum conservation are derived. 2. Calculate the stream function and pressure field, and plot streamlines for a known
INTERMOLECULAR AND SURFACE FORCES
 INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
Thermocapillary Migration of a Drop
 Thermocapillary Migration of a Drop An Exact Solution with Newtonian Interfacial Rheology and Stretching/Shrinkage of Interfacial Area Elements for Small Marangoni Numbers R. BALASUBRAMANIAM a AND R. SHANKAR
Thermocapillary Migration of a Drop An Exact Solution with Newtonian Interfacial Rheology and Stretching/Shrinkage of Interfacial Area Elements for Small Marangoni Numbers R. BALASUBRAMANIAM a AND R. SHANKAR
Fluid Mechanics Theory I
 Fluid Mechanics Theory I Last Class: 1. Introduction 2. MicroTAS or Lab on a Chip 3. Microfluidics Length Scale 4. Fundamentals 5. Different Aspects of Microfluidcs Today s Contents: 1. Introduction to
Fluid Mechanics Theory I Last Class: 1. Introduction 2. MicroTAS or Lab on a Chip 3. Microfluidics Length Scale 4. Fundamentals 5. Different Aspects of Microfluidcs Today s Contents: 1. Introduction to
Interfacial forces and friction on the nanometer scale: A tutorial
 Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Electrowetting. space and ε l the liquid dielectric constant, Eq. (1) can be written as. γ = ε 0ε l 2d V2. (2)
 600 Electrowetting and hence a reduced flow rate in the pressure-drop direction. This reduced flow rate seems to suggest that the liquid have an apparent higher viscosity. The apparent viscosity is called
600 Electrowetting and hence a reduced flow rate in the pressure-drop direction. This reduced flow rate seems to suggest that the liquid have an apparent higher viscosity. The apparent viscosity is called
*blood and bones contain colloids. *milk is a good example of a colloidal dispersion.
 Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
The effects of gravity on the capillary instability in tubes
 J. Fluid Mech. (2006), vol. 556, pp. 217 226. c 2006 Cambridge University Press doi:10.1017/s0022112006009505 Printed in the United Kingdom 217 The effects of gravity on the capillary instability in tubes
J. Fluid Mech. (2006), vol. 556, pp. 217 226. c 2006 Cambridge University Press doi:10.1017/s0022112006009505 Printed in the United Kingdom 217 The effects of gravity on the capillary instability in tubes
3.10. Capillary Condensation and Adsorption Hysteresis
 3.10. Capillary Condensation and Adsorption Hysteresis We shall restrict our attention to the adsorption behavior of porous solids. Hysteresis: two quantities of adsorbed material for each equilibrium
3.10. Capillary Condensation and Adsorption Hysteresis We shall restrict our attention to the adsorption behavior of porous solids. Hysteresis: two quantities of adsorbed material for each equilibrium
Introduction to Micro/Nanofluidics. Date: 2015/03/13. Dr. Yi-Chung Tung. Outline
 Introduction to Micro/Nanofluidics Date: 2015/03/13 Dr. Yi-Chung Tung Outline Introduction to Microfluidics Basic Fluid Mechanics Concepts Equivalent Fluidic Circuit Model Conclusion What is Microfluidics
Introduction to Micro/Nanofluidics Date: 2015/03/13 Dr. Yi-Chung Tung Outline Introduction to Microfluidics Basic Fluid Mechanics Concepts Equivalent Fluidic Circuit Model Conclusion What is Microfluidics
Hydrodynamics of wetting phenomena. Jacco Snoeijer PHYSICS OF FLUIDS
 Hydrodynamics of wetting phenomena Jacco Snoeijer PHYSICS OF FLUIDS Outline 1. Creeping flow: hydrodynamics at low Reynolds numbers (2 hrs) 2. Thin films and lubrication flows (3 hrs + problem session
Hydrodynamics of wetting phenomena Jacco Snoeijer PHYSICS OF FLUIDS Outline 1. Creeping flow: hydrodynamics at low Reynolds numbers (2 hrs) 2. Thin films and lubrication flows (3 hrs + problem session
Line Tension Effect upon Static Wetting
 Line Tension Effect upon Static Wetting Pierre SEPPECHER Université de Toulon et du Var, BP 132 La Garde Cedex seppecher@univ tln.fr Abstract. Adding simply, in the classical capillary model, a constant
Line Tension Effect upon Static Wetting Pierre SEPPECHER Université de Toulon et du Var, BP 132 La Garde Cedex seppecher@univ tln.fr Abstract. Adding simply, in the classical capillary model, a constant
Reaction at the Interfaces
 Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Lotus leaf -Traditional, but smart pack from nature- Weon-Sun SHIN DEPT of FOOD & NUTRITION HANYANG UNIVERSITY
 Lotus leaf -Traditional, but smart pack from nature- Weon-Sun SHIN DEPT of FOOD & NUTRITION HANYANG UNIVERSITY Learning from nature & tradition Rice culture Buhdism Traditional lunch-box The way of cooking
Lotus leaf -Traditional, but smart pack from nature- Weon-Sun SHIN DEPT of FOOD & NUTRITION HANYANG UNIVERSITY Learning from nature & tradition Rice culture Buhdism Traditional lunch-box The way of cooking
On waiting time solutions, contact angles and stability of steady states in thin film models
 On waiting time solutions, contact angles and stability of steady states in thin film models Marina Chugunova Claremont Graduate University Institute of Mathematical Science in collaboration with: Roman
On waiting time solutions, contact angles and stability of steady states in thin film models Marina Chugunova Claremont Graduate University Institute of Mathematical Science in collaboration with: Roman
Sessile drops in microgravity
 ArXiv, April 24, 2013 Sessile drops in microgravity Amelia Carolina Sparavigna Dipartimento di Scienza Applicata e Tecnologia Politecnico di Torino, Torino, Italy Interfaces with a liquid are governing
ArXiv, April 24, 2013 Sessile drops in microgravity Amelia Carolina Sparavigna Dipartimento di Scienza Applicata e Tecnologia Politecnico di Torino, Torino, Italy Interfaces with a liquid are governing
Wetting and dewetting of structured and imprinted surfaces
 Colloids and Surfaces A: Physicochemical and Engineering Aspects 161 (2000) 3 22 www.elsevier.nl/locate/colsurfa Wetting and dewetting of structured and imprinted surfaces Reinhard Lipowsky *, Peter Lenz,
Colloids and Surfaces A: Physicochemical and Engineering Aspects 161 (2000) 3 22 www.elsevier.nl/locate/colsurfa Wetting and dewetting of structured and imprinted surfaces Reinhard Lipowsky *, Peter Lenz,
Capillary Phenomena in Assemblies of Parallel Cylinders
 Capillary Phenomena in Assemblies of Parallel Cylinders I. Capillary Rise between Two Cylinders H. M. PRINCEN Lever Brothers Company, Research and Development Division, Edgewater, New Jersey 07020 Received
Capillary Phenomena in Assemblies of Parallel Cylinders I. Capillary Rise between Two Cylinders H. M. PRINCEN Lever Brothers Company, Research and Development Division, Edgewater, New Jersey 07020 Received
Boiling and Condensation (ME742)
 Indian Institute of Technology Kanpur Department of Mechanical Engineering Boiling and Condensation (ME742) PG/Open Elective Credits: 3-0-0-9 Updated Syllabus: Introduction: Applications of boiling and
Indian Institute of Technology Kanpur Department of Mechanical Engineering Boiling and Condensation (ME742) PG/Open Elective Credits: 3-0-0-9 Updated Syllabus: Introduction: Applications of boiling and
Research strategy for Micro and complex fluids
 Research strategy for Micro and complex fluids Introduction Presently there is a strong trend towards miniaturizing equipment for chemical analysis and synthesis. This is made possible by development of
Research strategy for Micro and complex fluids Introduction Presently there is a strong trend towards miniaturizing equipment for chemical analysis and synthesis. This is made possible by development of
Chemical Reactions II
 Mass Transfer Chemical Reactions II Lecture, 6..7, r. K. Wegner . Homogeneous chemical reactions Chemical reactions can alter mass fluxes by orders of magnitude. Recall NH 3 absorption into water: log(c
Mass Transfer Chemical Reactions II Lecture, 6..7, r. K. Wegner . Homogeneous chemical reactions Chemical reactions can alter mass fluxes by orders of magnitude. Recall NH 3 absorption into water: log(c
when viewed from the top, the objects should move as if interacting gravitationally
 2 Elastic Space 2 Elastic Space The dynamics and apparent interactions of massive balls rolling on a stretched horizontal membrane are often used to illustrate gravitation. Investigate the system further.
2 Elastic Space 2 Elastic Space The dynamics and apparent interactions of massive balls rolling on a stretched horizontal membrane are often used to illustrate gravitation. Investigate the system further.
Chapter 9: Differential Analysis
 9-1 Introduction 9-2 Conservation of Mass 9-3 The Stream Function 9-4 Conservation of Linear Momentum 9-5 Navier Stokes Equation 9-6 Differential Analysis Problems Recall 9-1 Introduction (1) Chap 5: Control
9-1 Introduction 9-2 Conservation of Mass 9-3 The Stream Function 9-4 Conservation of Linear Momentum 9-5 Navier Stokes Equation 9-6 Differential Analysis Problems Recall 9-1 Introduction (1) Chap 5: Control
Nov : Lecture 20: Linear Homogeneous and Heterogeneous ODEs
 125 Nov. 09 2005: Lecture 20: Linear Homogeneous and Heterogeneous ODEs Reading: Kreyszig Sections: 1.4 (pp:19 22), 1.5 (pp:25 31), 1.6 (pp:33 38) Ordinary Differential Equations from Physical Models In
125 Nov. 09 2005: Lecture 20: Linear Homogeneous and Heterogeneous ODEs Reading: Kreyszig Sections: 1.4 (pp:19 22), 1.5 (pp:25 31), 1.6 (pp:33 38) Ordinary Differential Equations from Physical Models In
Diffuse Interface Field Approach (DIFA) to Modeling and Simulation of Particle-based Materials Processes
 Diffuse Interface Field Approach (DIFA) to Modeling and Simulation of Particle-based Materials Processes Yu U. Wang Department Michigan Technological University Motivation Extend phase field method to
Diffuse Interface Field Approach (DIFA) to Modeling and Simulation of Particle-based Materials Processes Yu U. Wang Department Michigan Technological University Motivation Extend phase field method to
MATHEMATICS 200 April 2010 Final Exam Solutions
 MATHEMATICS April Final Eam Solutions. (a) A surface z(, y) is defined by zy y + ln(yz). (i) Compute z, z y (ii) Evaluate z and z y in terms of, y, z. at (, y, z) (,, /). (b) A surface z f(, y) has derivatives
MATHEMATICS April Final Eam Solutions. (a) A surface z(, y) is defined by zy y + ln(yz). (i) Compute z, z y (ii) Evaluate z and z y in terms of, y, z. at (, y, z) (,, /). (b) A surface z f(, y) has derivatives
Lecture 6 Surface Diffusion Driven by Surface Energy
 Lecture 6 Surface Diffusion Driven by Surface Energy Examples Flattening a surface. Spherodizing. Rayleigh instability. Grain boundary grooving. Sintering Self-assembled quantum dots Atomic Flux and Surface
Lecture 6 Surface Diffusion Driven by Surface Energy Examples Flattening a surface. Spherodizing. Rayleigh instability. Grain boundary grooving. Sintering Self-assembled quantum dots Atomic Flux and Surface
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai
 Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Surfaces and Interfaces Defining of interfacial region Types of interfaces: surface vs interface Surface
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Surfaces and Interfaces Defining of interfacial region Types of interfaces: surface vs interface Surface
THE PHYSICS OF FOAM. Boulder School for Condensed Matter and Materials Physics. July 1-26, 2002: Physics of Soft Condensed Matter. 1.
 THE PHYSICS OF FOAM Boulder School for Condensed Matter and Materials Physics July 1-26, 2002: Physics of Soft Condensed Matter 1. Introduction Formation Microscopics 2. Structure Experiment Simulation
THE PHYSICS OF FOAM Boulder School for Condensed Matter and Materials Physics July 1-26, 2002: Physics of Soft Condensed Matter 1. Introduction Formation Microscopics 2. Structure Experiment Simulation
Electrokinetic assembly and manipulation II Lecture by Chung, Jae-Hyun
 Electrokinetic assembly and manipulation II Lecture by Chung, Jae-Hyun Chung, Jae-Hyun, Mechanical Engineering, University of Washington Liu, Wing Kam, Mechanical Engineering, Northwestern University Liu,
Electrokinetic assembly and manipulation II Lecture by Chung, Jae-Hyun Chung, Jae-Hyun, Mechanical Engineering, University of Washington Liu, Wing Kam, Mechanical Engineering, Northwestern University Liu,
+ S/y. The wetted portion of the surface is then delimited by a certain contact line L (here a
 EUROPHYSICS LETTERS Europhys. Lett., 21 (4), pp. 483-488 (1993) 1 February 1993 Contact Line Elasticity of a Completely Wetting Liquid Rising on a Wall. E. RAPHAEL(*)( ) and J. F. JoA"Y(**) (*) Institute
EUROPHYSICS LETTERS Europhys. Lett., 21 (4), pp. 483-488 (1993) 1 February 1993 Contact Line Elasticity of a Completely Wetting Liquid Rising on a Wall. E. RAPHAEL(*)( ) and J. F. JoA"Y(**) (*) Institute
CHAPTER 2. Theory: Wetting Phenomena
 CHAPTER 2 2.1 Introduction Wetting phenomena are ubiquitous in nature and technology. Wetting phenomena are an area where chemistry, physics, and engineering intersect. Macroscopically the word Surface
CHAPTER 2 2.1 Introduction Wetting phenomena are ubiquitous in nature and technology. Wetting phenomena are an area where chemistry, physics, and engineering intersect. Macroscopically the word Surface
Note: Each problem is worth 14 points except numbers 5 and 6 which are 15 points. = 3 2
 Math Prelim II Solutions Spring Note: Each problem is worth points except numbers 5 and 6 which are 5 points. x. Compute x da where is the region in the second quadrant between the + y circles x + y and
Math Prelim II Solutions Spring Note: Each problem is worth points except numbers 5 and 6 which are 5 points. x. Compute x da where is the region in the second quadrant between the + y circles x + y and
Changes of polymer material wettability by surface discharge
 Changes of polymer material wettability by surface discharge Surface discharge and material treatment Surface treatment of materials in low temperature plasma belongs to the modern and very perspective
Changes of polymer material wettability by surface discharge Surface discharge and material treatment Surface treatment of materials in low temperature plasma belongs to the modern and very perspective
Measurement of the Liquid Film Thickness in. Micro Tube Slug Flow
 Measurement of the Liquid Film Thickness in Micro Tube Slug Flow Youngbae Han and Naoki Shikazono Department of Mechanical Engineering, The University of Tokyo Hongo 7-3-1, Bunkyo-ku, Tokyo, 113-8656,
Measurement of the Liquid Film Thickness in Micro Tube Slug Flow Youngbae Han and Naoki Shikazono Department of Mechanical Engineering, The University of Tokyo Hongo 7-3-1, Bunkyo-ku, Tokyo, 113-8656,
Experimental Studies of Liquid Marbles and Superhydrophobic Surfaces
 Experimental Studies of Liquid Marbles and Superhydrophobic Surfaces Stephen James Elliott A thesis submitted in partial fulfilment of the requirements of Nottingham Trent University for the degree of
Experimental Studies of Liquid Marbles and Superhydrophobic Surfaces Stephen James Elliott A thesis submitted in partial fulfilment of the requirements of Nottingham Trent University for the degree of
 Number of pages in the question paper : 05 Number of questions in the question paper : 48 Modeling Transport Phenomena of Micro-particles Note: Follow the notations used in the lectures. Symbols have their
Number of pages in the question paper : 05 Number of questions in the question paper : 48 Modeling Transport Phenomena of Micro-particles Note: Follow the notations used in the lectures. Symbols have their
Capillary rise between elastic sheets
 J. Fluid Mech. (26), vol. 548, pp. 141 15. c 26 Cambridge University Press doi:1.117/s2211257718 Printed in the United Kingdom 141 Capillary rise between elastic sheets By HO-YOUNG KIM 1 AND L. MAHADEVAN
J. Fluid Mech. (26), vol. 548, pp. 141 15. c 26 Cambridge University Press doi:1.117/s2211257718 Printed in the United Kingdom 141 Capillary rise between elastic sheets By HO-YOUNG KIM 1 AND L. MAHADEVAN
Derivation of continuum models for the moving contact line problem based on thermodynamic principles. Abstract
 Derivation of continuum models for the moving contact line problem based on thermodynamic principles Weiqing Ren Courant Institute of Mathematical Sciences, New York University, New York, NY 002, USA Weinan
Derivation of continuum models for the moving contact line problem based on thermodynamic principles Weiqing Ren Courant Institute of Mathematical Sciences, New York University, New York, NY 002, USA Weinan
Module 3: "Thin Film Hydrodynamics" Lecture 11: "" The Lecture Contains: Micro and Nano Scale Hydrodynamics with and without Free Surfaces
 The Lecture Contains: Micro and Nano Scale Hydrodynamics with and without Free Surfaces Order of Magnitude Analysis file:///e /courses/colloid_interface_science/lecture11/11_1.htm[6/16/2012 1:39:56 PM]
The Lecture Contains: Micro and Nano Scale Hydrodynamics with and without Free Surfaces Order of Magnitude Analysis file:///e /courses/colloid_interface_science/lecture11/11_1.htm[6/16/2012 1:39:56 PM]
Hydrodynamics of wetting phenomena. Jacco Snoeijer PHYSICS OF FLUIDS
 Hydrodynamics of wetting phenomena Jacco Snoeijer PHYSICS OF FLUIDS Outline 1. Creeping flow: hydrodynamics at low Reynolds numbers (2 hrs) 2. Thin films and lubrication flows (3 hrs + problem session
Hydrodynamics of wetting phenomena Jacco Snoeijer PHYSICS OF FLUIDS Outline 1. Creeping flow: hydrodynamics at low Reynolds numbers (2 hrs) 2. Thin films and lubrication flows (3 hrs + problem session
NUMERICAL SIMULATION OF MICRO-FILTRATION OF OIL-IN-WATER EMULSIONS. Tohid Darvishzadeh
 NUMERICAL SIMULATION OF MICRO-FILTRATION OF OIL-IN-WATER EMULSIONS By Tohid Darvishzadeh A DISSERTATION Submitted to Michigan State University in partial fulfillment of the requirements for the degree
NUMERICAL SIMULATION OF MICRO-FILTRATION OF OIL-IN-WATER EMULSIONS By Tohid Darvishzadeh A DISSERTATION Submitted to Michigan State University in partial fulfillment of the requirements for the degree
