MPIKG Public Access Author Manuscript
|
|
|
- Solomon Newman
- 5 years ago
- Views:
Transcription
1 MPIKG Public Access Author Manuscript Max Planck Institute of of Colloids and Interfaces Author Manuscript Published in final edited form as: Stocco, A., & Möhwald, H. (2015). The influence of long-range surface forces on the contact angle of nanometric droplets and bubbles. Langmuir, 31(43), doi: /acs.langmuir.5b Abstract The influence of long-range surface forces on the contact angle of nanometric droplets and bubbles For a droplet or a bubble of dimensions below 100 nm, long-range surface forces such as longrange van der Waals forces can compete with capillarity, which leads to a size dependence of the contact angle. This is discussed in this work, where we also show that the effect cannot simply be described by a normalized line tension. We calculate interfacial profiles for typical values of van der Waals forces and discuss the role of long-range surface forces on the contact angle of nanobubbles and nanodrops.
2 The influence of long-range surface forces on the contact angle of nanometric droplets and bubbles Antonio Stocco, 1,2* Helmuth Möhwald 3 1 Laboratoire Charles Coulomb (L2C), UMR 5221 CNRS-University of Montpellier, Montpellier F-34095, France. 2 DWI Leibniz-Institut fu r Interaktive Materialien, Aachen 52056, Germany. 3 Max Planck Institute of Colloids and Interfaces, Potsdam 14476, Germany *corresponding author: antonio.stocco@umontpellier.fr KEYWORDS Nanodrops, nanobubbles, contact angle, long-range surface forces, line tension 1
3 ABSTRACT. For a droplet or a bubble of dimensions below 100 nm long-range surface forces Max Planck Institute of Colloids and Interfaces Author Manuscript such as long range van der Waals forces can compete with capillarity, which leads to a size dependence of the contact angle. This is discussed in this work, where we also show that the effect cannot simply be described by a normalized line tension. We calculate interfacial profiles for typical values of van der Waals forces and discuss the role of long-range surface forces on the contact angle of nanobubbles and nanodrops. height, h (nm) L,2 L,1 VdW local profile L,1 L,2 1 2 horizontal coordinate, x (nm) 1. INTRODUCTION Wetting at the nanoscale is a most relevant field of fundamental research and applications. Nanobubbles play key roles for the control of interfacial friction and for the propulsion 2
4 mechanism of some artificial microswimmers. 1,2 Nanosized liquid drops are also important in microelectronics, microfluidics and are relevant for biomedical coatings and textile processes. 3 At length scales between a nanometer and a micron, long-range surface forces play a key role for the equilibrium states of a given multiphase system. Such forces include long range Van der Waals, electrostatic, hydrophobic and hydration interactions. Hydrophobic interactions refer to the attraction manifested between two hydrophobic media in water. 4 Hydration interactions (1-2 nm range) are repulsive solvation forces observed, when two hydrophilic interfaces approach. The nature of the observed repulsion is connected to the strong binding of water at the interface and the formation of hydrogen bonding networks, which act against the approach of the two interfaces. 5 Only Van der Waals, electrostatic and structural interactions are usually included in the Derjaguin disjoining pressure, 6 even if other interactions such as double layer electrostatic and long range hydrophobic interactions act on the same length scales. 4 If the system under consideration is composed of two fluids and a solid, an equilibrium state can be described by partial wetting, where a drop or a bubble adopts a shape which minimizes the interactions at play. If the solid is a planar substrate, a drop tends to minimize its interfacial energy which leads to a spherical cap shape, if the drop dimension is smaller than few millimeters and gravity can be neglected. The spherical cap shape is found in many experiments for drops and bubbles sitting on smooth and clean substrates even when their dimensions become micrometric or even smaller. 7,8,9,10 In any case, for millimetric, micrometric or submicrometric drops, the interfacial profile deviates from the spherical cap shape close to the triple line at the nanoscale. This is expected since capillarity competes with long-range surface forces, which eventually dominate for distances close to few nanometers. 11 This deviation of the profile may be neglected for macroscopic bubbles or drops, since it represents only a negligible part of the 3
5 profile. However, this is not the case for nanometric drops and bubbles, for which long-range surface forces play a central role in the statics and dynamics of wetting. Some recent investigations have been focused on the formation and stability of nanobubbles, which are observed mostly on hydrophobic surfaces immersed in a liquid. Nanobubbles possess heights lower than 100 nm and in many experiments they are found to be very stable. 2,9,10,12,13 This long time stability is somewhat surprising and many theoretical models attempted to quantify the experimental observations. Recently, an explanation based on contact line pinning and gas oversaturation of the liquid, which balance the gas diffusion driven by the Laplace pressure, has been proposed. 14,15 Line tension, collective diffusion effects have been also discussed in the literature to explain some aspects of the stability and the contact angle of nanobubbles. 2,16,17 One remarkable feature is also represented by the difference in contact angle between those nanobubbles and the corresponding macroscopic air bubbles. However, some questions such as the value of the capillary pressure inside the nanobubbles and the role of surface forces on the contact angle remain open, 2,10,16,18 and the desirable control on the formation and stability of nanobubbles is not yet fully achieved, which represents a major drawback for applications. Many issues related to the physics of surface nanobubbles are common to the physics of surface nanodrops. 14 Also for nano-sized drops, line tension is usually invoked to describe the contact angle of droplets of decreasing sizes. In many experiments the droplet height can be as small as few nanometers. Negative values of the line tension have been found in most of the experiments. 6,7,16 Negative line tension values would imply that drops are not stable, since the increase of the contact line perimeter results in a decrease of the total energy of the system. This 4
6 is in contrast to the experimental observations which attest a long time stability of those nanodrops. In this contribution, we recall the importance of long-range surface forces (which are usually neglected in the recent literature) on the shape of nanometric droplets and bubbles. The focus is on Van der Waals interactions and on the interfacial profiles and contact angles of nanometric drops and bubbles. We expect that the long range nature of the forces affects the domain shape especially near the triple line and consider this in a quantitative way. Section 2 and 3 provide a theoretical background on contact angle and line tension. Motivation of this work and related key problems are presented in section 4. Interfacial profile results and discussion (section 5 and 6) are followed by the summary and perspectives. 2. CONTACT ANGLE The equilibrium contact angle of a sessile drop on a solid substrate in partial wetting is defined in the far-field, where the minimum energetic state leads to a spherical cap profile of the drop. Zooming on the triple line region where the three phases meet, one finds that the droplet profile deviates from a spherical cap due to long-range surface forces originating from the physicochemical properties of the media, see Fig. 1. Despite the fact that the local contact angle L in the colloidal region of a drop is different from a macroscopic contact angle, an equilibrium contact angle E can be always defined by considering that in the far field any local change (down to the molecular scale) will not affect the calculation of a free energy balance, if the triple line is simply shifted by an infinitesimal distance. 11 5
7 The equilibrium of interfacial energies for a solid (S)-vapour (V)- liquid (L) system can be expressed in terms of the work associated with an incremental area change ldx on the solid substrate, see Fig. 1: 11 W II-I = SV ldx SL ldx ldx cos E = 0 Eq. 1 where SV, SL and are the solid-vapour, solid-liquid and liquid-vapour interfacial tensions, respectively. In Eq. 1 it is also assumed that local contact angles in the colloidal region simply translate when shifted by dx. Note that we define x = 0 at the place where the spherical cap profile touches the solid substrate. The Young-Laplace equation is obtained from eq. 1 : cos E = ( SV SL ) /. Eq. 2 Hence, without accounting for a line tension contribution, it is possible to describe an equilibrium contact angle in the far-field, being aware that in the colloidal region the local profile may be very different from the far field profile, i.e. for distances < 1 µm. The latter distance describes the limit, for which long-range surface forces become negligible compared to capillary forces. 6
8 Figure 1. Sketch of a droplet profile in the colloidal region for characteristic length scales lower than a micrometer and in the far field region. Upon a shift dx of the triple line, one can always define an equilibrium contact angle accounting only for interfacial energy contributions. x = 0 at the place where the spherical cap profile touches the solid substrate. Different theoretical approaches aimed at describing the interface profile at small scales. 20 Longrange surface forces, line tension and contact angle hysteresis have been discussed for the interface profile of macroscopic drops. 20,21 3. LINE TENSION Considering that in partial wetting three bulk phases form three interfaces and a common contact line, a thermodynamic equilibrium can be described in the far field accounting both for three interfacial tension and a line tension terms. The latter is associated to the energy contribution of the triple line. Line tension can be regarded as the leading term of the total macroscopic free energy of the system after subtracting contributions proportional to volume and interface. 22 Two 7
9 different sources of excess energy are related to the line tension. On a molecular level, the intermolecular interactions between two media will be affected by the presence of the third one on the triple line, leading to an unsaturation of interactions. 23 The second source of excess energy is related to the interface profile disturbed by long-range surface forces which tend to bend a spherical cap profile in order to minimize the local energy of the system. 23 However, it is non trivial to calculate and measure the line tension according to the previous definition. One way to evaluate the line tension contribution is to find the microscopic profile of a partially wetting drop and consider the energy associated with the deviation between the microscopic profile and the macroscopic spherical cap profile. 22 Within this approach, the line tension is a constant calculated by the effective interfacial potential for the solid liquid vapour system accounting for three terms: (i) the asymptote of the profile at the contact line (ii), the whole interfacial profile and the corresponding local effective interface potential, (iii) and the interactions between fluid molecules. 22 From these calculations one finds the theoretical line tension for a given system which can be compared to the experimental line tension measured from contact angle experiments performed on droplets or bubbles of different sizes. 7,18,24 Note that in this description the drop dimension is macroscopic, but only the microscopic profile (for characteristic length scale smaller than a micrometer) is relevant for the line tension calculation. 24 The line tension has also an effect on the equilibrium contact angle. This is apparently in contrast with the definition of the equilibrium contact angle described in the previous section where an equilibrium contact angle can be always defined in the far field (see Fig. 1). 8
10 A reconciliation between these descriptions could be done accounting for a change of the local interfacial profile upon an infinitesimal translation and introducing a line energy contribution kdl in Eq. 1: 23 SV ldx SL ldx ldx cos kdl= 0 Eq. 3 However, it is worth noting that even if the interfacial profile simply translates (Fig. 1), one could always write equation 3 and associate the line energy term to a cohesive excess energy of the three phase system analogously as one considers the interfacial tension for a two phase system. From equation 3, a far field equilibrium contact angle accounting for a line tension term could be defined: SV = SL cos k dl/(ldx) cos cos E k/ dl/(ldx) Eq. 4 is the equilibrium contact angle that accounts also for the line energy contribution; whilst, E accounts only for interfacial tensions. Note that for a drop wetting a planar solid, dl = 2 (R d +dx) 2 R d = 2 dx (where R d is the droplet contact radius) and dl/(ldx) = 1/ R d is the curvature of the contact line. Hence Eq.4 becomes: 23 cos cos E k/( R d Eq. 5 The modified Young-Laplace equation describes as well a far field balance of surface forces where an additional term, the line tension, is introduced. Note that depends on the droplet size R d. From eq. 5, the linear dependence of the cosine of the contact angle on the inverse of the 9
11 droplet size was used to calculate the line tension. Within the resolution of the experiments the drop/bubble shapes were found always spherical cap like even for drops/bubbles of nanometric sizes. Analysis of the experimental data was carried out within the modified Young-Laplace equation and in most of the cases a linear fit of the cosine of the contact angle versus the inverse of the drop size was imposed to evaluate the line tension. 7,8,18 The way equation 2 and 5 are derived implies that drop or bubble dimensions extend to the far field. 4. KEY PROBLEM OF THIS WORK Now we ask the following questions: Can we predict the interfacial profile when the drop/bubble dimensions extend only in the colloidal region (i.e. dimensions smaller than a micrometer)? Can the spherical cap profile of submicrometric drops/bubbles be set by the far field equilibrium contact angle accounting or not for a line tension? What is the role of the local contact angle defined in the colloidal region? We show in Fig 2 that for a 2 µm radius droplet, the spherical cap profile merges a local interfacial profile for a distance of the order of 0.1 µm (5 % of the radius). Hence, the droplet will adopt a spherical cap profile defined by a far field contact angle and only in the colloidal region (well below a distance of 0.1 µm), the interfacial profile will deviate because of a competition between the long-range surface and capillary forces, i.e. for radial distances < 100 nm. Keeping constant the contact angle ( E = 0.1), we plot spherical cap profiles for submicron sized droplets. Comparison with a calculated local interfacial profile (as described in the 10
12 following section) reveals that cap profiles cross the local profile for distances of the order of 40 Max Planck Institute of Colloids and Interfaces Author Manuscript nm, which is almost of the same order as the droplet contact radius. Now it becomes problematic to imagine a matching between a local profile and a spherical cap profile, as the slopes of the two profiles are very different at the crossing points. Moreover, if one would predict that the contact angle of submicron sized droplets can be described by the modified Young Laplace equation, it is interesting to notice that the calculation of the line tension may become length scale dependent. In fact, for large drops the line tension is calculated from the deviation of the spherical cap profile due to long-range surface forces, which does not change for a drop radius larger than a micron. On the other hand for submicron sized drops the spherical cap profile is strongly affected by the long-range surface interactions and the deviation of the local profile may be due to short-range molecular forces. Indeed, a dependence of the line tension on the wetting perimeter was described by Churaev et al.. 25 height, h (nm) E = 0.1 h(x) a=0.2 nm R d = 2 µm (a=0) R d = 200 nm (a=0) R d = 100 nm (a=0) horizontal coordinate, x (nm) Figure 2. Solid line is an interfacial profile accounting for a repulsive VdW force (a=0.2 nm, E = 0.1, see the text). Three spherical cap profiles for three decreasing droplet radii with E = 0.1 are also shown. At x=0, h=0 for spherical cap profiles (for null VdW forces, a=0). 11
13 5. INTERFACIAL PROFILES Here we focus our description of interfacial profiles for length scales larger than a nanometer and smaller than a micrometer, on cases where the interfacial tensions are not length scale dependent and where short-range molecular forces can be neglected. The profile of the interface can be calculated by assuming that at equilibrium the chemical potential should be equal along the interface profile. The following equation defines the equilibrium state of a liquid layer of height h : 25 + (h) = P 0 Eq. 6 Where is the surface tension of the liquid, = d 2 h/dx 2 [1+(dh/dx) 2 ] 3/2is the local interface curvature, (h) is the Derjaguin disjoining pressure and the constant P 0 is the capillary pressure of the meniscus. Note that, given the length scale of interest,gravity is neglected here. In the following we consider the case of nanodroplets, the case of nanobubbles being analogous. In the colloidal region, the interfacial profile of a droplet can show both positive and negative curvatures, since the Derjaguin disjoining pressure could show a maximum in the range from 1 nm to 1 µm. In fact the interaction between the solid-fluid and fluid-fluid interfaces can be repulsive and attractive depending on the interactions and distance Interfacial profiles of droplets accounting for attractive and repulsive VdW forces were calculated and analytical expressions are reported in literature. 27,28,29 12
14 Note that calculations of VdW forces are based on a frequency independent Hamaker constant Max Planck Institute of Colloids and Interfaces Author Manuscript term, which depends only on the refractive indexes of the media; and a frequency dependent term, which accounts for the spectral properties of the media. 30 Considering only the effect of VdW forces, the curvature of the interfacial profile is negative for repulsive VdW and positive for attractive VdW forces (see Fig. 3). Note that the sign of the curvature depends on the sign of the second derivative of h(x). Those profiles should be found in every drop and bubble. However, it is very challenging from an experimental view point to measure those profiles without introducing any artifacts. In Figure 3A we show the spherical cap profile for E = 0.1 rad and R d = 10 µm together with calculated profiles for repulsive VdW interactions. For the spherical cap profile, when zooming into the nm length scale, the drop shape looks as a straight line of tan E slope. Accounting for long-range repulsive VdW forces in equation 6, the drop profile h(x) becomes hyperbolic: 27 Z 2 = 1 + X 2, Eq. 7 where h = a θ E Z, x = a θ E 2 X. a 2 = A 6πγ and a is a characteristic length describing the balance between VdW and interfacial tension terms, A is the repulsive Hamaker constant and is the fluid interfacial tension. Note that equation 7 contains two free parameters, a that describes the strength of the VdW force, and E, which describes the far-field equilibrium. 13
15 The three profiles are plotted for typical values of a = 0.1, 0.2 and 0.3 nm (e.g. the repulsive Hamaker constant for glass-water-air 5 is J). 30 The deviation of the profile increases, if the characteristic length a increases. Note that the deviation of the profile is significant for heights lower than a/ E, i.e. 1, 2 and 3 nm. Hence, as we discussed in Fig. 2, the hyperbolic profile will match the spherical cap profile for heights of the order of a/ E, as one can expect for droplets of micron size or larger. Note that this will be not the case for nanodrops or nanobubbles. In fact for the latter systems the maximum height could be of the same order or even smaller than a/ E. 14
16 A h (nm) E = 0.1 rad a= 0.1 nm a= 0.2 nm a= 0.3 nm R d = 10 µm (a=0) x (nm) B Figure 3. (A) Calculated interfacial profile accounting for repulsive VdW forces with three characteristic lengths a = 0.1, 0.2, 0.3 nm and a spherical cap droplet profiles of 10 µm contact radius. (B) Calculated interfacial profile accounting for attractive VdW forces with three characteristic lengths 0 /b = 0.1, 0.2, 0.3 nm and a spherical cap droplet profiles of 10 µm contact radius. At x=0, h=0 for spherical cap profiles (for null VdW forces, A=0). h (nm) 10 E = 0.51 rad We have also calculated interfacial profiles for attractive Van der Waals forces (Fig. 3B). In this /b = 0.1 nm 0 /b = 0.2 nm 0 /b = 0.3 nm R d =10 µm ( 0 =0) x (nm) case the square of the slope of the profile is given by: 29 ( dz dx )2 = [(1 2b) 1 + (2bZ 2 ) 1 ] 1, Eq. 8 where h = δ 0 Z, x x 0 = δ 0 X. Here 0 is a distance defined by: δ 2 0 = ba, and b is related to the equilibrium contact angle by: 6πγ cos E / (2b). x 0 is a constant, which depends on the boundary condition of the profile. Profiles are calculated subtracting a baseline of height δ 0, which corresponds to the height of the molecular wetting film coexisting with the meniscus. To calculate the profiles in Fig 3(B), Eq. 8 15
17 was solved numerically and in a good approximation can be represented by the analytical function: Z = tanθ E (X 2b arctan ( X 2b )) Eq. 9 In Fig. 3 we show the interfacial profiles for three typical values of δ 0 (e.g. the attractive Hamaker constant for glass-air-water 5 is J) together with a spherical cap profile for E = 0.51 rad and R d = 10 µm. As we pointed out, it must be noted that the height of nanodroplets and nanobubbles corresponds to a length, where the interfacial profile is strongly affected by VdW forces. The nanometric droplet or bubble actually exists only in the colloidal region (see Fig. 1), where equation 2 and 5 can not be strictly applied. Hence, it can be argued that if a droplet exists only in the colloidal region and it adopts a spherical-like cap profile, it does not imply that it will adopt the same contact angle as the one defined in the far field, where only capillarity matters. Here we suggest that the contact angle adopted by the droplet is closely related to the local angle of the interfacial profile, which describes the competition between long-range surface and capillary forces. Accounting for a disjoining pressure only due to VdW forces of the form: (h) = A/6 h 3 one might estimate the contribution of long-range surface and capillary forces on the interfacial profile. 5 Neglecting the gravity term, the droplet profile obeys to equation 6: 31 + (h)/ = P 0 / C 0. To find the constant C 0 it is convenient to look at the drop maximum, where the first derivative of the profile is zero. 16
18 When the drop maximum height is larger than 0.1 µm, the disjoining pressure decays almost to zero, (h MAX ) = 0.2 J/m 3 0 (A/6 h 3 MAX 1/(0.3 m)), and C 0 = P 0 / 1/R 0 is the curvature of the drop at the maximum height, i.e. assuming a spherical cap shape and L = 0.1 rad, 1/R 0 = 1/(20 µm) (see Fig. 4B). For small droplets the disjoining pressure does not decay close to zero anymore and C 0 = P 0 / 1/R 0 + A/6 h 3 MAX. Eq. 10 When h = 1 nm, A/6 h 3 MAX 1/(30 nm) showing that the contribution of the long-range surface force becomes as important as the capillary one for nanodrops and nanobubbles, i.e. assuming a spherical cap shape and L = 0.1 rad, 1/R 0 = 1/(200 nm) (see Fig. 4B).. 6. RESULTS AND DISCUSSION Here, we propose a simple criterion which states that a droplet, whose dimensions are in the colloidal domain, will adopt a spherical cap profile to minimize its area with a contact angle dictated by the local contact angle which accounts for both long-range surface forces and capillarity. Hence according to this criterion, the contact angle of a nano-sized droplet of height h MAX is equal to the local contact angle of the interfacial profile at the same height (Fig. 4A). The nanosized droplet and the interfacial profile share the same contact in x, and the droplet contact radius is (see Fig. 4B): 17
19 R d = h MAX sin L /(1 cos L ). In Fig. 4, two droplets of different heights are shown. Significant differences in contact angles can be observed for droplet radii R d smaller than 100 nm. Increasing the droplet height, the slope of the interfacial profile varies slowly and approaches the far-field value tan E. A h (nm) 2 L,2 1 L,1 L,1 L, x (nm) Figure 4. (A) Criterion for the definition of the contact angle for two nanometric droplets. The height of the droplet defines the contact angle in the interfacial profile calculated accounting for VdW forces. The droplet and the interfacial profile share the same contact at h = 0 (here at x=20 nm). (B) Sketch of the contact radius R d, radius of curvature R 0 and maximum height of a spherical cap shape drop or bubble. Using the same criterion represented in Fig. 4, in Fig. 5A and 5C we plot the local contact angle L as a function of R d = h MAX sin L /(1 cos L ) for all calculated profiles. Note that for droplet radius of 50 nm the local contact angle approaches the equilibrium contact angle, while local contact angles dramatically change around droplet radii of about 10 nm. At this point we would like to compare our calculated data shown in Fig. 5A and 5C with the modified Young-Laplace equation (equation 5), and the linear relation of the cosine of the contact angle with the inverse of the droplet radius. 18
20 A L (rad) B cos L cos E = a= 0.1 nm a= 0.2 nm a= 0.3 nm E = 0.1 rad a= 0.1 nm a= 0.2 nm a= 0.3 nm R d (nm) /R d (nm -1 ) C L (rad) D cos L E = 0.51 rad (b=4) /b = 0.1 nm /b = 0.2 nm /b = 0.3 nm R d (nm) cos E = 0.85 (b=4) /b = 0.1 nm /b = 0.2 nm /b = 0.3 nm /R d (nm -1 ) Figure 5. Local contact angles calculated for repulsive (A) and attractive (C) VdW forces as a function of a droplet contact radius defined as R d = h MAX sin L /(1 cos L ). Cosine of local contact angles calculated for repulsive (B) and attractive (D) VdW forces as a function of the inverse of R d. 19
21 Note that our calculations accounted only for repulsive and attractive VdW forces in the interfacial profile of drops in the mean field. The shapes of the interfacial profiles were calculated using equation 7 and 8. Equation 5 instead assumes that the interfacial profile could be described simply by a constant value line tension defined for macroscopic systems. Hence the underlying physics of our result and the line tension approach are completely different, and we argue, that interpreting the size-dependence of the contact angle in terms of a constant value line tension is not correct. Just for the sake of comparison, in Fig. 5B and 5D we replotted the data shown in Fig. 5A and 5C as for line tension measurements (equation 5). For repulsive VdW forces, a linear trend of cos L with the drop curvature can be observed. From these slopes one would find positive line tensions k = 6.5 x N (a = 0.1 nm), 1.2 x N (a = 0.2 nm), 1.7 x N (a = 0.3 nm) assuming an interfacial tension = 72 mn/m. For attractive VdW forces, the trend of cos L with the drop curvature is not linear. Actually, these curves recall the shape of experimental curves reported by Berg et al and Checco et al.. 7,8 The apparent slopes found fitting the data for attractive VdW forces correspond to negative line tensions k 6 x N ( b = 0.1 nm), 9 x N ( b = 0.2 nm), 1.4 x N ( b = 0.3 nm), assuming an interfacial tension = 72 mn/m. Those line tension values would agree with reported theoretical and experimental values. 24 Here, positive line tensions correspond to the case of repulsive VdW forces, whereas negative line tensions correspond to attractive VdW forces. In the Supporting Information, we show a comparison of our model with nanobubble and nanodroplet experiments. 20
22 As shown in Fig. 4D and in some experiments, the cosine of the droplet contact angle is not linear with the inverse of the droplet radius if the droplet size is nanometric. 7,8 The linear relation could be found only for the modified Young-Laplace equation if drops exist in the far-field. In this case the line tension k can be also calculated theoretically. k is a constant of the system, which represents the energy not accounted for in the bulk and surface terms, when far-field asymptotes can be defined. For nanometric droplets we can relate the contact angle to the local interfacial profile, which represents the competition of forces acting at the colloidal length scale. Thus, the relation between the droplet contact angle and the droplet size is dictated by the shape of the interfacial profile in the colloidal region. Hence in general, the cosine of the contact angle is not expected to be linear with the droplet curvature, since the form of the disjoining pressure may show local minima and maxima with the distance, and both positive and negative curvatures could be observed (see Figure 6). 25 Here we suggest an interpretation of experiments performed on droplets and bubbles of decreasing sizes (down to the nanometer scale) as an effect of local contact angle due to longrange surface forces such as long range VdW forces. 7. SUMMARY AND PERSPECTIVES Finally, for large drops whose sizes are larger than a micron, the contact angle is the equilibrium contact angle E defined by the Young-Laplace equations (eq. 2 and 5). Even for those drops, in the contact line region (at nanometric length scales) local contact angles deviate from E, but this 21
23 is a local effect that can be considered negligible. If the droplet height is in the nanoscale the droplet profile will be strongly affected by long-range surface forces. In this context, we propose a simple criterion based on the height of the droplet to describe the change of contact angle. We believe that the work presented here can stimulate some new discussion in the field of nanodrops and nanobubbles. Theoretical work aiming at describing exact interfacial profiles of nanofluids accounting for different terms of disjoining pressure will surely improve the understanding of contact angle and stability results of nanobubbles and nanodrops. To conclude, we point out two major results presented here that could be used in future studies. Eq. 10 shows that for nanofluids the capillary pressure depends strongly on the distance and on the Hamaker constant of the system. The latter could possess both signs, which would lead to a strong decrease or increase of the capillary pressure for nanobubbles and nanodrops. This result may help improving theoretical modeling on the gas diffusion of nanobubbles. We also point out that the stability of nanobubbles may result from the onset of repulsive longrange surface interactions observed when the height of the nanobubble decreases (see Figure 6). This repulsive force prevents the shrinking of the fluid and thus enhances the stability. We can assume that when the height of drops or bubbles is larger than an onset distance h * (h > h * ), the resulting interaction is attractive. In this region, the local contact angle decreases with the decreasing distance (Fig. 5(c)), which agrees with the effective negative line tension values observed in experiments and predictable for attractive VdW forces as in nanobubble systems. 14 Below this onset distance, h < h *, a repulsive VdW or electrostatic interaction may stabilize the fluid against further shrinking (see Figure 6). Repulsive VdW interactions are expected for systems when the static and the frequency dependent terms of the Hamaker constant result in 22
24 repulsive values. Repulsive electrostatic interactions for nanobubbles may manifest because of the negative potential of the gas-water and solid-water interfaces. 27 A clear perspective of this work is to include the contributions of electrostatic, hydrophobic and hydration interactions (together with VdW forces) into the disjoining pressure in order to reinterpret experimental results obtained for nanodrops and nanobubbles. 14 Figure 6. Sketch of an interfacial profile defying the contact angle of two nanodroplets or nanobubbles according to the criterion shown in Fig.4 and the connection with the profile of the disjoining pressure (h). ASSOCIATED CONTENT Supporting Information. Comparison between the model and experimental results. This material is available free of charge via the Internet at AUTHOR INFORMATION Corresponding Author 23
25 Author Contributions The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. ACKNOWLEDGMENT We thank Davide Ruffoni, Peter Kralchevski, Ahmed Mourran, Maurizio Nobili, Martin In and Renaud Denoyel for discussion and Labex Chemisyst for financial support. A. S. gratefully acknowledges the Alexander von Humboldt foundation for a research fellowship. REFERENCES (1) Gibbs, J. G.; Zhao, Y.-P. P. Appl. Phys. Lett. 2009, 94, (2) Craig, V. S. J. Soft Matter 2011, 7, 40. (3) Méndez-Vilas, A.; Jódar-Reyes, A. B.; González-Martín, M. L. Small 2009, 5, (4) Meyer, E. E.; Rosenberg, K. J.; Israelachvili, J. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, (5) Israelachvili, J. N. Intermol. Surf. Forces 2011, (6) Kuchin, I. V.; Matar, O. K.; Craster, R. V.; Starov, V. M. Colloids Interface Sci. Commun. 2014, 1, (7) Berg, J. K.; Weber, C. M.; Riegler, H. Phys. Rev. Lett. 2010, 105, (8) Checco, A.; Guenoun, P.; Daillant, J. Phys. Rev. Lett. 2003, 91, (9) Zhao, B.; Song, Y.; Wang, S.; Dai, B.; Zhang, L.; Dong, Y.; Lü, J.; Hu, J. Soft Matter 2013, 9, (10) Borkent, B. M.; De Beer, S.; Mugele, F.; Lohse, D. Langmuir 2010, 26,
26 (11) De Gennes, P.-G.; Rev. Mod. Phys. 1985, 57. (12) Karpitschka, S.; Dietrich, E.; Seddon, J.; Zandvliet, H.; Lohse, D.; Riegler, H. Phys. Rev. Lett. 2012, 109, (13) Zhang, X. H.; Maeda, N.; Craig, V. S. J. Langmuir 2006, 22, (14) Lohse, D.; Zhang, X. Rev. Mod. Phys. 2015, 87, (15) Lohse, D.; Zhang, X. Phys. Rev. E 2015, 91, (16) Peng, H.; Birkett, G. R.; Nguyen, A. V. Adv. Colloid Interface Sci. 2014, (17) Weijs, J. H.; Lohse, D. Phys. Rev. Lett. 2013, 110, (18) Kameda, N.; Nakabayashi, S. Chem. Phys. Lett. 2008, 461, (19) Drelich, J.; Miller, J. Part. Sci. Technol. 1982, 10, (20) Snoeijer, J. H.; Andreotti, B. Phys. Fluids 2008, 20, (21) Snoeijer, J. H.; Andreotti, B. Annu. Rev. Fluid Mech. 2013, 45, (22) Schimmele, L.; Naplórkowski, M.; Dietrich, S. J. Chem. Phys. 2007, 127. (23) Drelich, J. In Colloids and Surfaces A: Physicochemical and Engineering Aspects; 1996; Vol. 116, pp (24) Pompe, T.; Herminghaus, S. Phys. Rev. Lett. 2000, 85, (25) Churaev, N..; Starov, V..; Derjaguin, B.. J. Colloid Interface Sci. 1982, 89, (26) Kuchin, I. V.; Matar, O. K.; Craster, R. V.; Starov, V. M. Colloids Interface Sci. Commun. 2014, 1, (27) De Gennes, P. G.; Hua, X.; Levinson, P. J. Fluid Mech. 1990, 212, (28) Wayner, P.. J. Colloid Interface Sci. 1980, 77, (29) Wayner, P.. J. Colloid Interface Sci. 1982, 88, 6 7. (30) Bergström, L. Adv. Colloid Interface Sci. 1997, 70, (31) Sharma, A. Langmuir 1993, (32) Chaplin, M. Water 2009, 1,
27 Insert Table of Contents Graphic and Synopsis Here height, h (nm) L,1 L,1 L,2 L,2 1 2 VdW local profile horizontal coordinate, x (nm) 26
Jacco Snoeijer PHYSICS OF FLUIDS
 Jacco Snoeijer PHYSICS OF FLUIDS dynamics dynamics freezing dynamics freezing microscopics of capillarity Menu 1. surface tension: thermodynamics & microscopics 2. wetting (statics): thermodynamics & microscopics
Jacco Snoeijer PHYSICS OF FLUIDS dynamics dynamics freezing dynamics freezing microscopics of capillarity Menu 1. surface tension: thermodynamics & microscopics 2. wetting (statics): thermodynamics & microscopics
THE MODIFIED YOUNG S EQUATION FOR THE CONTACT ANGLE OF A SMALL SESSILE DROP FROM AN INTERFACE DISPLACEMENT MODEL
 International Journal of Modern Physics B, Vol. 13, No. 7 (1999) 355 359 c World Scientific Publishing Company THE MODIFIED YOUNG S EQUATION FOR THE CONTACT ANGLE OF A SMALL SESSILE DROP FROM AN INTERFACE
International Journal of Modern Physics B, Vol. 13, No. 7 (1999) 355 359 c World Scientific Publishing Company THE MODIFIED YOUNG S EQUATION FOR THE CONTACT ANGLE OF A SMALL SESSILE DROP FROM AN INTERFACE
Four-phase merging in sessile compound drops
 J. Fluid Mech. (00), vol. 45, pp. 4 40. c 00 Cambridge University Press DOI: 0.07/S000000708 Printed in the United Kingdom 4 Four-phase merging in sessile compound drops By L. M A H A D E V A N, M. A D
J. Fluid Mech. (00), vol. 45, pp. 4 40. c 00 Cambridge University Press DOI: 0.07/S000000708 Printed in the United Kingdom 4 Four-phase merging in sessile compound drops By L. M A H A D E V A N, M. A D
Surface and Interfacial Tensions. Lecture 1
 Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
Dynamic Equilibrium Mechanism for Surface Nanobubble Stabilization
 Dynamic Equilibrium Mechanism for Surface Nanobubble Stabilization The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation
Dynamic Equilibrium Mechanism for Surface Nanobubble Stabilization The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation
Thermodynamically Stable Emulsions Using Janus Dumbbells as Colloid Surfactants
 Thermodynamically Stable Emulsions Using Janus Dumbbells as Colloid Surfactants Fuquan Tu, Bum Jun Park and Daeyeon Lee*. Description of the term notionally swollen droplets When particles are adsorbed
Thermodynamically Stable Emulsions Using Janus Dumbbells as Colloid Surfactants Fuquan Tu, Bum Jun Park and Daeyeon Lee*. Description of the term notionally swollen droplets When particles are adsorbed
U nderstanding on the line tension becomes particularly important because of its relevance to a number of
 OPEN SUBJECT AREAS: WETTING SURFACE CHEMISTRY NANOSCALE MATERIALS STATISTICAL PHYSICS Received 15 March 2013 Accepted 30 May 2013 Published 18 June 2013 Correspondence and requests for materials should
OPEN SUBJECT AREAS: WETTING SURFACE CHEMISTRY NANOSCALE MATERIALS STATISTICAL PHYSICS Received 15 March 2013 Accepted 30 May 2013 Published 18 June 2013 Correspondence and requests for materials should
INTERMOLECULAR AND SURFACE FORCES
 INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
Microfluidics 2 Surface tension, contact angle, capillary flow
 MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
DLVO interaction between the spheres
 DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
Solvability condition for the moving contact line
 PHYSICAL REVIEW E 78, 564 28 Solvability condition for the moving contact line L. M. Pismen 1 and Jens Eggers 2 1 Department of Chemical Engineering and Minerva Center for Nonlinear Physics of Complex
PHYSICAL REVIEW E 78, 564 28 Solvability condition for the moving contact line L. M. Pismen 1 and Jens Eggers 2 1 Department of Chemical Engineering and Minerva Center for Nonlinear Physics of Complex
PHYSICS OF FLUID SPREADING ON ROUGH SURFACES
 INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
Colloidal Particles at Liquid Interfaces: An Introduction
 1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading
 Loughborough University Institutional Repository Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading This item was submitted
Loughborough University Institutional Repository Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading This item was submitted
Frieder Mugele. Physics of Complex Fluids. University of Twente. Jacco Snoeier Physics of Fluids / UT
 coorganizers: Frieder Mugele Physics of Comple Fluids Jacco Snoeier Physics of Fluids / UT University of Twente Anton Darhuber Mesoscopic Transport Phenomena / Tu/e speakers: José Bico (ESPCI Paris) Daniel
coorganizers: Frieder Mugele Physics of Comple Fluids Jacco Snoeier Physics of Fluids / UT University of Twente Anton Darhuber Mesoscopic Transport Phenomena / Tu/e speakers: José Bico (ESPCI Paris) Daniel
Lecture 7 Contact angle phenomena and wetting
 Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Molecular dynamics study of the lifetime of nanobubbles on the substrate
 Molecular dynamics study of the lifetime of nanobubbles on the substrate - Links of Hierarchies KOHNO Shunsuke Division of Physics and Astronomy, Graduate School of Science, Kyoto University Outline Introduction
Molecular dynamics study of the lifetime of nanobubbles on the substrate - Links of Hierarchies KOHNO Shunsuke Division of Physics and Astronomy, Graduate School of Science, Kyoto University Outline Introduction
Interfacial forces and friction on the nanometer scale: A tutorial
 Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Generalized Cassie-Baxter equation for wetting of a spherical droplet within a smooth and heterogeneous conical cavity
 Science Front Publishers Journal for Foundations and pplications of Physics, vol. 4, No. (017) (sciencefront.org) ISSN 394-3688 Generalized Cassie-axter equation for wetting of a spherical droplet within
Science Front Publishers Journal for Foundations and pplications of Physics, vol. 4, No. (017) (sciencefront.org) ISSN 394-3688 Generalized Cassie-axter equation for wetting of a spherical droplet within
INFLUENCE OF THE SURFACE FORCES ON THE APPARENT CONTACT ANGLE AT PARTIAL WETTING AND IN THE PRESENCE OF HEAT AND MASS TRANSFER
 Proceedings of the 2 nd European Conference on Microfluidics - Microfluidics 21 - Toulouse, December 8-1, 21 µflu1-212 INFLUENCE OF THE SURFACE FORCES ON THE APPARENT CONTACT ANGLE AT PARTIAL WETTING AND
Proceedings of the 2 nd European Conference on Microfluidics - Microfluidics 21 - Toulouse, December 8-1, 21 µflu1-212 INFLUENCE OF THE SURFACE FORCES ON THE APPARENT CONTACT ANGLE AT PARTIAL WETTING AND
Multiphase Flow and Heat Transfer
 Multiphase Flow and Heat Transfer ME546 -Sudheer Siddapureddy sudheer@iitp.ac.in Surface Tension The free surface between air and water at a molecular scale Molecules sitting at a free liquid surface against
Multiphase Flow and Heat Transfer ME546 -Sudheer Siddapureddy sudheer@iitp.ac.in Surface Tension The free surface between air and water at a molecular scale Molecules sitting at a free liquid surface against
J. Bico, C. Tordeux and D. Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS Collège de France Paris Cedex 05, France
 EUROPHYSICS LETTERS 15 July 2001 Europhys. Lett., 55 (2), pp. 214 220 (2001) Rough wetting J. Bico, C. Tordeux and D. Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS Collège de France
EUROPHYSICS LETTERS 15 July 2001 Europhys. Lett., 55 (2), pp. 214 220 (2001) Rough wetting J. Bico, C. Tordeux and D. Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS Collège de France
Specific ion effects on the interaction of. hydrophobic and hydrophilic self assembled
 Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Capillarity and Wetting Phenomena
 ? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
Contents. Preface XI Symbols and Abbreviations XIII. 1 Introduction 1
 V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
Physics and Chemistry of Interfaces
 Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
drops in motion Frieder Mugele the physics of electrowetting and its applications Physics of Complex Fluids University of Twente
 drops in motion the physics of electrowetting and its applications Frieder Mugele Physics of Complex Fluids niversity of Twente 1 electrowetting: the switch on the wettability voltage outline q q q q q
drops in motion the physics of electrowetting and its applications Frieder Mugele Physics of Complex Fluids niversity of Twente 1 electrowetting: the switch on the wettability voltage outline q q q q q
The Wilhelmy balance. How can we measure surface tension? Surface tension, contact angles and wettability. Measuring surface tension.
 ow can we measure surface tension? Surface tension, contact angles and wettability www.wikihow.com/measure-surface-tension Measuring surface tension The Wilhelmy balance F Some methods: Wilhelmy plate
ow can we measure surface tension? Surface tension, contact angles and wettability www.wikihow.com/measure-surface-tension Measuring surface tension The Wilhelmy balance F Some methods: Wilhelmy plate
ISCST shall not be responsible for statements or opinions contained in papers or printed in its publications.
 Modeling of Drop Motion on Solid Surfaces with Wettability Gradients J. B. McLaughlin, Sp. S. Saravanan, N. Moumen, and R. S. Subramanian Department of Chemical Engineering Clarkson University Potsdam,
Modeling of Drop Motion on Solid Surfaces with Wettability Gradients J. B. McLaughlin, Sp. S. Saravanan, N. Moumen, and R. S. Subramanian Department of Chemical Engineering Clarkson University Potsdam,
Derivation of continuum models for the moving contact line problem based on thermodynamic principles. Abstract
 Derivation of continuum models for the moving contact line problem based on thermodynamic principles Weiqing Ren Courant Institute of Mathematical Sciences, New York University, New York, NY 002, USA Weinan
Derivation of continuum models for the moving contact line problem based on thermodynamic principles Weiqing Ren Courant Institute of Mathematical Sciences, New York University, New York, NY 002, USA Weinan
Praktikum zur. Materialanalytik
 Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
of Nebraska - Lincoln
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Xiao Cheng Zeng Publications Published Research - Department of Chemistry 10-1-2006 Homogeneous nucleation at high supersaturation
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Xiao Cheng Zeng Publications Published Research - Department of Chemistry 10-1-2006 Homogeneous nucleation at high supersaturation
The Origins of Surface and Interfacial Tension
 The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
Generalized Wenzel equation for contact angle of droplets on spherical rough solid substrates
 Science Front Publishers Journal for Foundations and Applications of Physics, 3 (2), (2016) (sciencefront.org) ISSN 2394-3688 Generalized Wenzel equation for contact angle of droplets on spherical rough
Science Front Publishers Journal for Foundations and Applications of Physics, 3 (2), (2016) (sciencefront.org) ISSN 2394-3688 Generalized Wenzel equation for contact angle of droplets on spherical rough
Transition de séchage dans des nanopores et la tension de ligne de l eau
 Transition de séchage dans des nanopores et la tension de ligne de l eau L. Guillemot, T. Biben, A. Gallarneau, G. Vigier, E. Charlaix Laboratoire LiPhy Université Joseph Fourier Laboratoire MATEIS INSA-Lyon
Transition de séchage dans des nanopores et la tension de ligne de l eau L. Guillemot, T. Biben, A. Gallarneau, G. Vigier, E. Charlaix Laboratoire LiPhy Université Joseph Fourier Laboratoire MATEIS INSA-Lyon
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa.
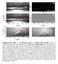 Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
+ S/y. The wetted portion of the surface is then delimited by a certain contact line L (here a
 EUROPHYSICS LETTERS Europhys. Lett., 21 (4), pp. 483-488 (1993) 1 February 1993 Contact Line Elasticity of a Completely Wetting Liquid Rising on a Wall. E. RAPHAEL(*)( ) and J. F. JoA"Y(**) (*) Institute
EUROPHYSICS LETTERS Europhys. Lett., 21 (4), pp. 483-488 (1993) 1 February 1993 Contact Line Elasticity of a Completely Wetting Liquid Rising on a Wall. E. RAPHAEL(*)( ) and J. F. JoA"Y(**) (*) Institute
Reaction at the Interfaces
 Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Line Tension Effect upon Static Wetting
 Line Tension Effect upon Static Wetting Pierre SEPPECHER Université de Toulon et du Var, BP 132 La Garde Cedex seppecher@univ tln.fr Abstract. Adding simply, in the classical capillary model, a constant
Line Tension Effect upon Static Wetting Pierre SEPPECHER Université de Toulon et du Var, BP 132 La Garde Cedex seppecher@univ tln.fr Abstract. Adding simply, in the classical capillary model, a constant
On the Landau-Levich Transition
 10116 Langmuir 2007, 23, 10116-10122 On the Landau-Levich Transition Maniya Maleki Institute for AdVanced Studies in Basic Sciences (IASBS), Zanjan 45195, P.O. Box 45195-1159, Iran Etienne Reyssat and
10116 Langmuir 2007, 23, 10116-10122 On the Landau-Levich Transition Maniya Maleki Institute for AdVanced Studies in Basic Sciences (IASBS), Zanjan 45195, P.O. Box 45195-1159, Iran Etienne Reyssat and
Module17: Intermolecular Force between Surfaces and Particles. Lecture 23: Intermolecular Force between Surfaces and Particles
 Module17: Intermolecular Force between Surfaces and Particles Lecture 23: Intermolecular Force between Surfaces and Particles 1 We now try to understand the nature of spontaneous instability in a confined
Module17: Intermolecular Force between Surfaces and Particles Lecture 23: Intermolecular Force between Surfaces and Particles 1 We now try to understand the nature of spontaneous instability in a confined
Brownian diffusion of a partially wetted colloid
 SUPPLEMENTARY INFORMATION DOI: 1.138/NMAT4348 Brownian diffusion of a partially wetted colloid Giuseppe Boniello, Christophe Blanc, Denys Fedorenko, Mayssa Medfai, Nadia Ben Mbarek, Martin In, Michel Gross,
SUPPLEMENTARY INFORMATION DOI: 1.138/NMAT4348 Brownian diffusion of a partially wetted colloid Giuseppe Boniello, Christophe Blanc, Denys Fedorenko, Mayssa Medfai, Nadia Ben Mbarek, Martin In, Michel Gross,
Theory of the dynamics of spreading of liquids on fibers
 Theory of the dynamics of spreading of liquids on fibers Françoise BrochardWyart, JeanMarc Di Meglio, David Quéré To cite this version: Françoise BrochardWyart, JeanMarc Di Meglio, David Quéré. Theory
Theory of the dynamics of spreading of liquids on fibers Françoise BrochardWyart, JeanMarc Di Meglio, David Quéré To cite this version: Françoise BrochardWyart, JeanMarc Di Meglio, David Quéré. Theory
Capillary surfaces and complex analysis: new opportunities to study menisci singularities. Mars Alimov, Kazan Federal University, Russia
 Capillary surfaces and complex analysis: new opportunities to study menisci singularities Mars limov Kazan Federal University Russia Kostya Kornev Clemson University SC Outline Intro to wetting and capillarity
Capillary surfaces and complex analysis: new opportunities to study menisci singularities Mars limov Kazan Federal University Russia Kostya Kornev Clemson University SC Outline Intro to wetting and capillarity
Instabilities in the Flow of Thin Liquid Films
 Instabilities in the Flow of Thin Liquid Films Lou Kondic Department of Mathematical Sciences Center for Applied Mathematics and Statistics New Jersey Institute of Technology Presented at Annual Meeting
Instabilities in the Flow of Thin Liquid Films Lou Kondic Department of Mathematical Sciences Center for Applied Mathematics and Statistics New Jersey Institute of Technology Presented at Annual Meeting
DLVO Theory and Non-DLVO Forces
 NPTEL Chemical Engineering Interfacial Engineering Module 3: Lecture 5 DLVO Theory and Non-DLVO Forces Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati 781039
NPTEL Chemical Engineering Interfacial Engineering Module 3: Lecture 5 DLVO Theory and Non-DLVO Forces Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati 781039
Capillary interactions between particles bound to interfaces, liquid films and biomembranes
 Advances in Colloid and Interface Science 85 2000 145 192 Capillary interactions between particles bound to interfaces, liquid films and biomembranes Peter A. Kralchevsky a,b, Kuniaki Nagayama a, a Laboratory
Advances in Colloid and Interface Science 85 2000 145 192 Capillary interactions between particles bound to interfaces, liquid films and biomembranes Peter A. Kralchevsky a,b, Kuniaki Nagayama a, a Laboratory
Hysteresis of the contact angle of a meniscus inside a capillary with smooth, homogeneous solid walls
 Loughborough University Institutional Repository Hysteresis of the contact angle of a meniscus inside a capillary with smooth, homogeneous solid walls This item was submitted to Loughborough University's
Loughborough University Institutional Repository Hysteresis of the contact angle of a meniscus inside a capillary with smooth, homogeneous solid walls This item was submitted to Loughborough University's
Groovy Drops: Effect of Groove Curvature on Spontaneous Capillary Flow
 Groovy Drops: Effect of Groove Curvature on Spontaneous Capillary Flow Myra Kitron-Belinkov,, Abraham Marmur,*, Thomas Trabold, and Gayatri Vyas Dadheech Department of Chemical Engineering, Technion -
Groovy Drops: Effect of Groove Curvature on Spontaneous Capillary Flow Myra Kitron-Belinkov,, Abraham Marmur,*, Thomas Trabold, and Gayatri Vyas Dadheech Department of Chemical Engineering, Technion -
The evaporation of sessile droplets onto solid surfaces : experiments and simulations of the contact line pinning-depinning
 The evaporation of sessile droplets onto solid surfaces : experiments and simulations of the contact line pinning-depinning L.Kabeya-Mukeba, N.Vandewalle and S.Dorbolo GRASP, Institut de Physique B5a,
The evaporation of sessile droplets onto solid surfaces : experiments and simulations of the contact line pinning-depinning L.Kabeya-Mukeba, N.Vandewalle and S.Dorbolo GRASP, Institut de Physique B5a,
Surfactants role on the deformation of colliding small bubbles
 Colloids and Surfaces A: Physicochemical and Engineering Aspects 156 (1999) 547 566 www.elsevier.nl/locate/colsurfa Surfactants role on the deformation of colliding small bubbles D.S. Valkovska, K.D. Danov
Colloids and Surfaces A: Physicochemical and Engineering Aspects 156 (1999) 547 566 www.elsevier.nl/locate/colsurfa Surfactants role on the deformation of colliding small bubbles D.S. Valkovska, K.D. Danov
Surface forces and wetting features in drops and capillaries
 Advances in Fluid Mechanics VIII 345 Surface forces and wetting features in drops and capillaries M. G. Velarde1 & V. M. Starov2 1 2 Instituto Pluridisciplinar, Universidad Complutense de Madrid, Spain
Advances in Fluid Mechanics VIII 345 Surface forces and wetting features in drops and capillaries M. G. Velarde1 & V. M. Starov2 1 2 Instituto Pluridisciplinar, Universidad Complutense de Madrid, Spain
arxiv: v1 [physics.class-ph] 13 Sep 2008
![arxiv: v1 [physics.class-ph] 13 Sep 2008 arxiv: v1 [physics.class-ph] 13 Sep 2008](/thumbs/72/67619383.jpg) 1 arxiv:0809.2346v1 [physics.class-ph] 13 Sep 2008 14th Conference on Waves and Stability in Continuous Media, Baia Samuele, Sicily, Italy, 30 June - 7 July, 2007 Edited by N Manganaro, R Monaco and S
1 arxiv:0809.2346v1 [physics.class-ph] 13 Sep 2008 14th Conference on Waves and Stability in Continuous Media, Baia Samuele, Sicily, Italy, 30 June - 7 July, 2007 Edited by N Manganaro, R Monaco and S
Capillary rise between closely spaced plates : effect of Van der Waals forces
 Capillary rise between closely spaced plates : effect of Van der Waals forces B. Legait, P.G. De Gennes To cite this version: B. Legait, P.G. De Gennes. Capillary rise between closely spaced plates : effect
Capillary rise between closely spaced plates : effect of Van der Waals forces B. Legait, P.G. De Gennes To cite this version: B. Legait, P.G. De Gennes. Capillary rise between closely spaced plates : effect
characterize antiadhesive surfaces
 Capillary bridge formation and breakage: a test to characterize antiadhesive surfaces TITLE RUNNING HEAD: Capillary bridge for antiadhesive surfaces. Laurianne Vagharchakian 1, Frédéric Restagno 2, Liliane
Capillary bridge formation and breakage: a test to characterize antiadhesive surfaces TITLE RUNNING HEAD: Capillary bridge for antiadhesive surfaces. Laurianne Vagharchakian 1, Frédéric Restagno 2, Liliane
Supplementary table I. Table of contact angles of the different solutions on the surfaces used here. Supplementary Notes
 1 Supplementary Figure 1. Sketch of the experimental setup (not to scale) : it consists of a thin mylar sheet (0, 02 4 3cm 3 ) held fixed vertically. The spacing y 0 between the glass plate and the upper
1 Supplementary Figure 1. Sketch of the experimental setup (not to scale) : it consists of a thin mylar sheet (0, 02 4 3cm 3 ) held fixed vertically. The spacing y 0 between the glass plate and the upper
Supplementary Information. In colloidal drop drying processes, multi-ring depositions are formed due to the stick-slip
 Electronic Supplementary Material (ESI for Soft Matter. This journal is The Royal Society of Chemistry 14 Supplementary Information A1. Contact line receding velocity of an evaporating drop In colloidal
Electronic Supplementary Material (ESI for Soft Matter. This journal is The Royal Society of Chemistry 14 Supplementary Information A1. Contact line receding velocity of an evaporating drop In colloidal
Capillary Phenomena in Assemblies of Parallel Cylinders
 Capillary Phenomena in Assemblies of Parallel Cylinders I. Capillary Rise between Two Cylinders H. M. PRINCEN Lever Brothers Company, Research and Development Division, Edgewater, New Jersey 07020 Received
Capillary Phenomena in Assemblies of Parallel Cylinders I. Capillary Rise between Two Cylinders H. M. PRINCEN Lever Brothers Company, Research and Development Division, Edgewater, New Jersey 07020 Received
Self assembly of graphene oxide at the liquid-liquid interface: A new. rout to fabrication of graphene based composites
 Supporting Information for Self assembly of graphene oxide at the liquid-liquid interface: A new rout to fabrication of graphene based composites Mohsen Moazzami Gudarzi, Farhad Sharif * Department of
Supporting Information for Self assembly of graphene oxide at the liquid-liquid interface: A new rout to fabrication of graphene based composites Mohsen Moazzami Gudarzi, Farhad Sharif * Department of
Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
contact line dynamics
 contact line dynamics part 2: hydrodynamics dynamic contact angle? lubrication: Cox-Voinov theory maximum speed for instability corner shape? dimensional analysis: speed U position r viscosity η pressure
contact line dynamics part 2: hydrodynamics dynamic contact angle? lubrication: Cox-Voinov theory maximum speed for instability corner shape? dimensional analysis: speed U position r viscosity η pressure
Surface interactions part 1: Van der Waals Forces
 CHEM-E150 Interfacial Phenomena in Biobased Systems Surface interactions part 1: Van der Waals Forces Monika Österberg Spring 018 Content Colloidal stability van der Waals Forces Surface Forces and their
CHEM-E150 Interfacial Phenomena in Biobased Systems Surface interactions part 1: Van der Waals Forces Monika Österberg Spring 018 Content Colloidal stability van der Waals Forces Surface Forces and their
Capillarity. ESS5855 Lecture Fall 2010
 Capillarity ESS5855 Lecture Fall 2010 Capillarity: the tendency of a liquid in a narrow tube or pore to rise or fall as a result of surface tension (The concise Oxford Dictionary) Surface tension: the
Capillarity ESS5855 Lecture Fall 2010 Capillarity: the tendency of a liquid in a narrow tube or pore to rise or fall as a result of surface tension (The concise Oxford Dictionary) Surface tension: the
Thickness and Shape of Films Driven by a Marangoni Flow
 Langmuir 1996, 12, 5875-5880 5875 Thickness and Shape of Films Driven by a Marangoni Flow X. Fanton, A. M. Cazabat,* and D. Quéré Laboratoire de Physique de la Matière Condensée, Collège de France, 11
Langmuir 1996, 12, 5875-5880 5875 Thickness and Shape of Films Driven by a Marangoni Flow X. Fanton, A. M. Cazabat,* and D. Quéré Laboratoire de Physique de la Matière Condensée, Collège de France, 11
Faceted drops on heterogeneous surfaces
 EUROPHYSICS LETTERS 15 July 2001 Europhys. Lett., 55 (2), pp. 239 245 (2001) Faceted drops on heterogeneous surfaces T. Cubaud and M. Fermigier Laboratoire PMMH, CNRS UMR 7636, ESPCI 10 rue Vauquelin,
EUROPHYSICS LETTERS 15 July 2001 Europhys. Lett., 55 (2), pp. 239 245 (2001) Faceted drops on heterogeneous surfaces T. Cubaud and M. Fermigier Laboratoire PMMH, CNRS UMR 7636, ESPCI 10 rue Vauquelin,
Depinning of 2d and 3d droplets blocked by a hydrophobic defect
 Depinning of 2d and 3d droplets blocked by a hydrophobic defect P. Beltrame 1, P. Hänggi 1, E. Knobloch 2, and U. Thiele 3 1 Institut für Physik, Universität Augsburg, D-86135 Augsburg, Germany 2 Department
Depinning of 2d and 3d droplets blocked by a hydrophobic defect P. Beltrame 1, P. Hänggi 1, E. Knobloch 2, and U. Thiele 3 1 Institut für Physik, Universität Augsburg, D-86135 Augsburg, Germany 2 Department
Probing the Hydrophobic Interaction between Air Bubbles and Partially. Hydrophobic Surfaces Using Atomic Force Microscopy
 Supporting Information for Probing the Hydrophobic Interaction between Air Bubbles and Partially Hydrophobic Surfaces Using Atomic Force Microscopy Chen Shi, 1 Derek Y.C. Chan, 2.3 Qingxia Liu, 1 Hongbo
Supporting Information for Probing the Hydrophobic Interaction between Air Bubbles and Partially Hydrophobic Surfaces Using Atomic Force Microscopy Chen Shi, 1 Derek Y.C. Chan, 2.3 Qingxia Liu, 1 Hongbo
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces.
 Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák & István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry http://kolloid.unideb.hu/~kolloid/
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák & István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry http://kolloid.unideb.hu/~kolloid/
Line Tension Effects for Liquid Droplets on Circular Surface Domains
 Langmuir 2006, 22, 11041-11059 11041 Line Tension Effects for Liquid Droplets on Circular Surface Domains Pedro Blecua, Reinhard Lipowsky, and Jan Kierfeld* Max Planck Institute of Colloids and Interfaces,
Langmuir 2006, 22, 11041-11059 11041 Line Tension Effects for Liquid Droplets on Circular Surface Domains Pedro Blecua, Reinhard Lipowsky, and Jan Kierfeld* Max Planck Institute of Colloids and Interfaces,
Compound pendant drop tensiometry for. surface tension measurement at zero Bond number
 Compound pendant drop tensiometry for surface tension measurement at zero Bond number Michael J. Neeson, Derek Y. C. Chan,,, and Rico F. Tabor, Department of Mathematics and Statistics, University of Melbourne,
Compound pendant drop tensiometry for surface tension measurement at zero Bond number Michael J. Neeson, Derek Y. C. Chan,,, and Rico F. Tabor, Department of Mathematics and Statistics, University of Melbourne,
Capillary Contact Angle in a Completely Wet Groove
 Capillary Contact Angle in a Completely Wet Groove A.O. Parry, 1 A. Malijevský, 2 and C. Rascón 3 1 Department of Mathematics, Imperial College London, London SW7 2BZ, UK 2 Department of Physical Chemistry,
Capillary Contact Angle in a Completely Wet Groove A.O. Parry, 1 A. Malijevský, 2 and C. Rascón 3 1 Department of Mathematics, Imperial College London, London SW7 2BZ, UK 2 Department of Physical Chemistry,
Wetting and dewetting of structured and imprinted surfaces
 Colloids and Surfaces A: Physicochemical and Engineering Aspects 161 (2000) 3 22 www.elsevier.nl/locate/colsurfa Wetting and dewetting of structured and imprinted surfaces Reinhard Lipowsky *, Peter Lenz,
Colloids and Surfaces A: Physicochemical and Engineering Aspects 161 (2000) 3 22 www.elsevier.nl/locate/colsurfa Wetting and dewetting of structured and imprinted surfaces Reinhard Lipowsky *, Peter Lenz,
5.2 Surface Tension Capillary Pressure: The Young-Laplace Equation. Figure 5.1 Origin of surface tension at liquid-vapor interface.
 5.2.1 Capillary Pressure: The Young-Laplace Equation Vapor Fo Fs Fs Fi Figure 5.1 Origin of surface tension at liquid-vapor interface. Liquid 1 5.2.1 Capillary Pressure: The Young-Laplace Equation Figure
5.2.1 Capillary Pressure: The Young-Laplace Equation Vapor Fo Fs Fs Fi Figure 5.1 Origin of surface tension at liquid-vapor interface. Liquid 1 5.2.1 Capillary Pressure: The Young-Laplace Equation Figure
Electrowetting on dielectrics on lubricating fluid based slippery surfaces with negligible hysteresis
 Electrowetting on dielectrics on lubricating fluid based slippery surfaces with negligible hysteresis J. Barman a, A. K. Nagarajan b and K. Khare a* a Department of Physics, Indian Institute of Technology
Electrowetting on dielectrics on lubricating fluid based slippery surfaces with negligible hysteresis J. Barman a, A. K. Nagarajan b and K. Khare a* a Department of Physics, Indian Institute of Technology
Vibration of submillimeter-size supported droplets
 PHYSICAL REVIEW E 73, 041602 2006 Vibration of submillimeter-size supported droplets Franck Celestini* and Richard Kofman Laboratoire de Physique de la Matière Condensée, UMR 6622, CNRS, Université de
PHYSICAL REVIEW E 73, 041602 2006 Vibration of submillimeter-size supported droplets Franck Celestini* and Richard Kofman Laboratoire de Physique de la Matière Condensée, UMR 6622, CNRS, Université de
A shallow water type model to describe the dynamic. of thin partially wetting films
 A shallow water type model to describe the dynamic of thin partially wetting films J. LALLEMENT a, P. VILLEDIEU a, P. TRONTIN a, C. LAURENT a a. Onera - The French Aerospace Lab Toulouse) - ONERA - F-31055
A shallow water type model to describe the dynamic of thin partially wetting films J. LALLEMENT a, P. VILLEDIEU a, P. TRONTIN a, C. LAURENT a a. Onera - The French Aerospace Lab Toulouse) - ONERA - F-31055
arxiv: v1 [cond-mat.soft] 30 Aug 2016
![arxiv: v1 [cond-mat.soft] 30 Aug 2016 arxiv: v1 [cond-mat.soft] 30 Aug 2016](/thumbs/85/91746129.jpg) Free-energy barrier of filling a spherical cavity in arxiv:1608.08286v1 [cond-mat.soft] 30 Aug 2016 the presence of line tension: Implication to the energy barrier between the Cassie and Wenzel state on
Free-energy barrier of filling a spherical cavity in arxiv:1608.08286v1 [cond-mat.soft] 30 Aug 2016 the presence of line tension: Implication to the energy barrier between the Cassie and Wenzel state on
SYNTHESIS OF INORGANIC MATERIALS AND NANOMATERIALS. Pr. Charles Kappenstein LACCO, Laboratoire de Catalyse en Chimie Organique, Poitiers, France
 SYNTHESIS OF INORGANIC MATERIALS AND NANOMATERIALS Pr. Charles Kappenstein LACCO, Laboratoire de Catalyse en Chimie Organique, Poitiers, France Outline IV - FORMATION OF SOLIDS FROM SOLUTIONS 1) Glass
SYNTHESIS OF INORGANIC MATERIALS AND NANOMATERIALS Pr. Charles Kappenstein LACCO, Laboratoire de Catalyse en Chimie Organique, Poitiers, France Outline IV - FORMATION OF SOLIDS FROM SOLUTIONS 1) Glass
Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen
 Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen Department of Chemical and Biomolecular Engineering Clarkson University Outline
Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen Department of Chemical and Biomolecular Engineering Clarkson University Outline
Supplementary Information:
 Supplementary Information: Self assembly of tetrahedral CdSe nanocrystals: effective patchiness via anisotropic steric interaction Michael A. Boles and Dmitri V. Talapin Department of Chemistry and James
Supplementary Information: Self assembly of tetrahedral CdSe nanocrystals: effective patchiness via anisotropic steric interaction Michael A. Boles and Dmitri V. Talapin Department of Chemistry and James
Supporting Information
 Supporting Information On the Minimal Size of Coffee Ring Structure Xiaoying Shen, Chih-Ming Ho and Tak-Sing Wong * Mechanical and Aerospace Engineering Department, University of California, Los Angeles,
Supporting Information On the Minimal Size of Coffee Ring Structure Xiaoying Shen, Chih-Ming Ho and Tak-Sing Wong * Mechanical and Aerospace Engineering Department, University of California, Los Angeles,
Nanoemulsions obtained via bubble-bursting at a compound interface
 Nanoemulsions obtained via bubble-bursting at a compound interface 3 4 5 6 7 8 9 10 11 12 Jie Feng 1, Matthieu Roché 1, #, Daniele Vigolo 1, $, Luben N. Arnaudov 2, Simeon D. Stoyanov 2,3, Theodor D. Gurkov
Nanoemulsions obtained via bubble-bursting at a compound interface 3 4 5 6 7 8 9 10 11 12 Jie Feng 1, Matthieu Roché 1, #, Daniele Vigolo 1, $, Luben N. Arnaudov 2, Simeon D. Stoyanov 2,3, Theodor D. Gurkov
Multiphase Flow and Heat Transfer
 Multiphase Flow and Heat Transfer Liquid-Vapor Interface Sudheer Siddapuredddy sudheer@iitp.ac.in Department of Mechanical Engineering Indian Institution of Technology Patna Multiphase Flow and Heat Transfer
Multiphase Flow and Heat Transfer Liquid-Vapor Interface Sudheer Siddapuredddy sudheer@iitp.ac.in Department of Mechanical Engineering Indian Institution of Technology Patna Multiphase Flow and Heat Transfer
arxiv: v1 [cond-mat.soft] 6 Oct 2015
![arxiv: v1 [cond-mat.soft] 6 Oct 2015 arxiv: v1 [cond-mat.soft] 6 Oct 2015](/thumbs/73/69466842.jpg) Reply to the Comments on Curvature capillary migration of microspheres by P. Galatola and A. Würger Nima Sharifi-Mood, 1 ris B. Liu, 1 and Kathleen J. Stebe 1, 1 Department of Chemical and Biomolecular
Reply to the Comments on Curvature capillary migration of microspheres by P. Galatola and A. Würger Nima Sharifi-Mood, 1 ris B. Liu, 1 and Kathleen J. Stebe 1, 1 Department of Chemical and Biomolecular
Capillary rise of a wetting fluid in a semi-circular groove
 68.45Gd L etalement The J. Phys. France 50 (1989) 485491 15 FÉVRIER 1989, 485 Classification Physics Abstracts 47.15 68.10Gw Capillary rise of a wetting fluid in a semicircular groove Elie Raphael Laboratoire
68.45Gd L etalement The J. Phys. France 50 (1989) 485491 15 FÉVRIER 1989, 485 Classification Physics Abstracts 47.15 68.10Gw Capillary rise of a wetting fluid in a semicircular groove Elie Raphael Laboratoire
RESEARCH STATEMENT BHAGYA ATHUKORALLAGE
 RESEARCH STATEENT BHAGYA ATHUKORALLAGE y research interests lie in several areas of applied mathematics. In particular, I recently focused my attention on the mathematical theory of capillary interfaces
RESEARCH STATEENT BHAGYA ATHUKORALLAGE y research interests lie in several areas of applied mathematics. In particular, I recently focused my attention on the mathematical theory of capillary interfaces
Supplementary Information. for
 Supplementary Information for Discrete Element Model for Suppression of Coffee-Ring Effect Ting Xu, 1 Miu Ling Lam, 2,3,4 and Ting-Hsuan Chen 1,2,3,4 1 Department of Mechanical and Biomedical Engineering,
Supplementary Information for Discrete Element Model for Suppression of Coffee-Ring Effect Ting Xu, 1 Miu Ling Lam, 2,3,4 and Ting-Hsuan Chen 1,2,3,4 1 Department of Mechanical and Biomedical Engineering,
Molecular dynamics investigation of thickness effect on liquid films
 JOURNAL OF CHEMICAL PHYSICS VOLUME 113, NUMBER 14 8 OCTOBER 2000 Molecular dynamics investigation of thicness effect on liquid films Jian-Gang Weng, Seungho Par, a) Jennifer R. Lues, and Chang-Lin Tien
JOURNAL OF CHEMICAL PHYSICS VOLUME 113, NUMBER 14 8 OCTOBER 2000 Molecular dynamics investigation of thicness effect on liquid films Jian-Gang Weng, Seungho Par, a) Jennifer R. Lues, and Chang-Lin Tien
COMPARISON OF WETTABILITY AND CAPILLARY EFFECT EVALUATED BY DIFFERENT CHARACTERIZING METHODS
 18 TH INTERNATIONAL CONFERENCE ON COMPOSITE MATERIALS COMPARISON OF WETTABILITY AND CAPILLARY EFFECT EVALUATED BY DIFFERENT CHARACTERIZING METHODS S.K. Wang*, M. Li*, Y.Z. Gu, Y.X. Li and Z.G. Zhang Key
18 TH INTERNATIONAL CONFERENCE ON COMPOSITE MATERIALS COMPARISON OF WETTABILITY AND CAPILLARY EFFECT EVALUATED BY DIFFERENT CHARACTERIZING METHODS S.K. Wang*, M. Li*, Y.Z. Gu, Y.X. Li and Z.G. Zhang Key
emulsions, and foams March 21 22, 2009
 Wetting and adhesion Dispersions in liquids: suspensions, emulsions, and foams ACS National Meeting March 21 22, 2009 Salt Lake City Ian Morrison 2009 Ian Morrison 2009 Lecure 2 - Wetting and adhesion
Wetting and adhesion Dispersions in liquids: suspensions, emulsions, and foams ACS National Meeting March 21 22, 2009 Salt Lake City Ian Morrison 2009 Ian Morrison 2009 Lecure 2 - Wetting and adhesion
Investigations on the Evaporation of Charged Droplets on Hydrophobic Surface
 Proceedings of the International Conference on Heat Transfer and Fluid Flow Prague, Czech Republic, August 11-12, 2014 Paper No. 15 Investigations on the Evaporation of Charged Droplets on Hydrophobic
Proceedings of the International Conference on Heat Transfer and Fluid Flow Prague, Czech Republic, August 11-12, 2014 Paper No. 15 Investigations on the Evaporation of Charged Droplets on Hydrophobic
*blood and bones contain colloids. *milk is a good example of a colloidal dispersion.
 Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
On the breakup of fluid films of finite and infinite extent
 PHYSICS OF FLUIDS 19, 072107 2007 On the breakup of fluid films of finite and infinite extent Javier A. Diez a Instituto de Física Arroyo Seco, Universidad Nacional del Centro de la Provincia de Buenos
PHYSICS OF FLUIDS 19, 072107 2007 On the breakup of fluid films of finite and infinite extent Javier A. Diez a Instituto de Física Arroyo Seco, Universidad Nacional del Centro de la Provincia de Buenos
dewetting driving forces dewetting mechanism? dewetting dynamics? final equilibrium state: drops with θ = θ Y
 dewetting initial state: continuous film of partially wetting liquid final equilibrium state: drops wit θ = θ Y driving forces dewetting mecanism? dewetting dynamics? 1 free energy vs. film tickness water
dewetting initial state: continuous film of partially wetting liquid final equilibrium state: drops wit θ = θ Y driving forces dewetting mecanism? dewetting dynamics? 1 free energy vs. film tickness water
Chemical Potential. Combining the First and Second Laws for a closed system, Considering (extensive properties)
 Chemical Potential Combining the First and Second Laws for a closed system, Considering (extensive properties) du = TdS pdv Hence For an open system, that is, one that can gain or lose mass, U will also
Chemical Potential Combining the First and Second Laws for a closed system, Considering (extensive properties) du = TdS pdv Hence For an open system, that is, one that can gain or lose mass, U will also
Thin films in partial wetting: stability, dewetting and coarsening
 Thin films in partial wetting: stability, dewetting and coarsening The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published
Thin films in partial wetting: stability, dewetting and coarsening The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published
Surface Tension and its measurements
 Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
Sessile drops in microgravity
 ArXiv, April 24, 2013 Sessile drops in microgravity Amelia Carolina Sparavigna Dipartimento di Scienza Applicata e Tecnologia Politecnico di Torino, Torino, Italy Interfaces with a liquid are governing
ArXiv, April 24, 2013 Sessile drops in microgravity Amelia Carolina Sparavigna Dipartimento di Scienza Applicata e Tecnologia Politecnico di Torino, Torino, Italy Interfaces with a liquid are governing
Part VII. Miscellaneous topics Module 3. Coarsening
 Part VII. Miscellaneous topics Module 3. Coarsening 3 Coarsening 3.1 Motivation Consider a precipitation hardenable alloy. As a function of aging time, after peak aging the strength reduces. It is found
Part VII. Miscellaneous topics Module 3. Coarsening 3 Coarsening 3.1 Motivation Consider a precipitation hardenable alloy. As a function of aging time, after peak aging the strength reduces. It is found
Proposed approximation for contact angles in Shan-and-Chen-type multicomponent multiphase lattice Boltzmann models
 PHYSICAL REVIEW E 7, 0701 007 Proposed approximation for contact angles in Shan-and-Chen-type multicomponent multiphase lattice Boltzmann models Haibo Huang, 1 Daniel T. Thorne, Jr., Marcel G. Schaap,
PHYSICAL REVIEW E 7, 0701 007 Proposed approximation for contact angles in Shan-and-Chen-type multicomponent multiphase lattice Boltzmann models Haibo Huang, 1 Daniel T. Thorne, Jr., Marcel G. Schaap,
