Name Date CUMULATIVE TEST FOR LESSON CLUSTERS 1-4
|
|
|
- Susanna Adele Heath
- 5 years ago
- Views:
Transcription
1 Cumulative Test 1 Name Date CUMULATIVE TEST FOR LESSON CLUSTERS Why can you change ice into water but not into glass? 2. Why can't you see air? 3. Describe the ways in which ice, liquid water, and water vapor are different when you look at them through magic eyeglasses. Draw pictures if you want to, but also use words. 4. Describe, as best you can, what molecules are.
2 TEACHER'S GUIDE FOR SCORING CUMULATIVE TEST FOR LESSON CLUSTERS 1-4 Teacher's Guide Test 1 1. Students should say that ice and water are made up of the same kind of molecules, but ice and glass are not. 2. Students should say that the molecules that make up air are so small and so far apart (that is, not clumped together as they are in solids and liquids) and air cannot be seen. This is true for most gases. 3. Students should describe both the arrangement and motions of water molecules in the three states: ice: molecules are close together, in a rigid pattern, vibrating back and forth liquid water: molecules are also close together, but not in a pattern, sliding past each other as they constantly move around water vapor: molecules are very far apart, constantly moving, sometimes colliding with each other. 4. Sixth grade students should be able to say that molecules are the smallest pieces of a substance, or that molecules are the particles or bits that the substance is made up of. Some students say that molecules are very small things in substances. When they say that, it is not clear what they mean.
3 Cumulative Test 1, Page 2 5. Why is it that we can see trees, but we can't see the molecules that trees are made up of? 6. When you put your finger over the end of an empty syringe and push down on the plunger, you can push it in part of the way, but not all of the way. (a) We know that there is air in the syringe. Explain why you can push the plunger in with your finger over the end and push air together. (b) When you push it in almost to the end, it gets very hard to push it any farther. Explain why you can't push the plunger all the way in. 7. When you smell soup cooking on the stove, what does that tell you about the molecules in the air?
4 Teacher's Guide Test 1 5. The important part of a correct answer is that individual molecules are too small to be seen. (The situation is analogous to seeing a beach. We can see the beach even when we are much too far away to see individual grains of sand.) 6. (a) Students should talk about molecules in their answer, and not just say that air is compressible. The plunger can be pushed part of the way in because the molecules that make up air are far apart and they can be pushed closer together. (b) The plunger can't be pushed in all the way because the air molecules are pushed closer together. They hit back on the plunger more and more often, until it becomes too hard to push them in any farther. (This is a difficult question, and you may want to use it as an "extra credit" question.) 7. The important part of a correct answer is that there are soup molecules in the air if you can smell the soup. (Of course, soup is a mixture of different substances, and not all of them evaporate into the air, nor are all of them smelly. But if you can smell it, then some smelly molecules have left the soup and mixed in with the air.)
5 Cumulative Test 1, Page 3 8. How is it that molecules of smelly substances can move through the air and finally get to your nose? 9. Industries sometimes dump poisonous or harmful liquids that they want to get rid of into rivers and lakes. (a) If you took some water out of a river or lake that had been polluted in this way, would that water be a mixture or a pure substance? (b) Explain your answer. (c) Draw what you think you would see if you looked at polluted water with magic eyeglasses. 10. How are ice, water, and water vapor the same? Talk about molecules, if you can.
6 Teacher's Guide Test 1 8. Students should say that molecules are always moving. That is why some of them that are mixed in with the air eventually get to your nose. Some students say that the air carries the smell (or the molecules) to your nose. A better explanation includes the idea that the molecules of the smelly substance are always moving. 9. (a) The polluted water is a mixture. (b) It is a mixture because it is made up of two or more kinds of molecules, water molecules and the molecules that make up the poisonous or otherwise harmful liquids. (c) Drawings should show water molecules and at least one other kind of molecule. 10. Students should talk about water molecules in their answers. The molecules that make up ice, liquid water, and water vapor are the same--they are all water molecules.
7 Cumulative Test 1, Page Is air a pure substance or a mixtures? Explain your answer: 12. How is humid air different from air that is not so humid?
8 Teacher's Guide Test Air is a mixture. Students should say that it is a mixture because it is made of more than one kind of molecule. Whether they list the different components of air (nitrogen, oxygen, carbon dioxide, water vapor, and other substances) or not, is not important. 12. Humid air has more water vapor in it than air that is not so humid. Another acceptable way of saying this is that humid air has more water molecules mixed in with the other molecules.
Chapter 7.1. States of Matter
 Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
DEMONSTRATION 4.1 MOLECULES HITTING EACH OTHER
 DEMONSTRATION 4.1 MOLECULES HITTING EACH OTHER Directions for doing the demonstration are in the Science Book Teacher's Guide. 1. Students should include these ideas: The air moving out of the hair dryer
DEMONSTRATION 4.1 MOLECULES HITTING EACH OTHER Directions for doing the demonstration are in the Science Book Teacher's Guide. 1. Students should include these ideas: The air moving out of the hair dryer
INTRODUCTION TO LESSON CLUSTER 7
 INTRODUCTION TO LESSON CLUSTER 7 EXPLAINING MELTING AND SOLIDIFYING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain melting and solidifying, by reference to the molecular
INTRODUCTION TO LESSON CLUSTER 7 EXPLAINING MELTING AND SOLIDIFYING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain melting and solidifying, by reference to the molecular
CHM Solids, Liquids, and Phase Changes (r15) Charles Taylor 1/9
 CHM 111 - Solids, Liquids, and Phase Changes (r15) - 2015 Charles Taylor 1/9 Introduction In CHM 110, we used kinetic theory to explain the behavior of gases. Now, we will discuss solids and liquids. While
CHM 111 - Solids, Liquids, and Phase Changes (r15) - 2015 Charles Taylor 1/9 Introduction In CHM 110, we used kinetic theory to explain the behavior of gases. Now, we will discuss solids and liquids. While
Name Date Block LESSON CLUSTER 6: Expansion and Contraction
 LESSON CLUSTER 6: Expansion and Contraction Did you know that when you say that something is hot or cold, you are actually saying something about the molecules of that substance? Words like hot and cold
LESSON CLUSTER 6: Expansion and Contraction Did you know that when you say that something is hot or cold, you are actually saying something about the molecules of that substance? Words like hot and cold
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 2, LESSON 1 HEAT, TEMPERATURE, AND CONDUCTION MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state
The Next Generation Science Standards (NGSS) CHAPTER 2, LESSON 1 HEAT, TEMPERATURE, AND CONDUCTION MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state
How Does the Sun s Energy Cause Rain?
 1.2 Investigate 3.3 Read How Does the Sun s Energy Cause Rain? In the water-cycle simulation, you observed water change from a liquid to a gas, and then back to a liquid falling to the bottom of the container.
1.2 Investigate 3.3 Read How Does the Sun s Energy Cause Rain? In the water-cycle simulation, you observed water change from a liquid to a gas, and then back to a liquid falling to the bottom of the container.
Copyright 2015 Edmentum - All rights reserved. During which of the following phase changes is there a gain in energy? I.
 Study Island Copyright 2015 Edmentum - All rights reserved. Generation Date: 03/16/2015 Generated By: Kristina Brown 1. Examine the phase-change diagram below. During which of the following phase changes
Study Island Copyright 2015 Edmentum - All rights reserved. Generation Date: 03/16/2015 Generated By: Kristina Brown 1. Examine the phase-change diagram below. During which of the following phase changes
Atoms and molecules are in motion and have energy
 Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Solids, liquids and gases
 Solids, liquids and gases Duration 60 minutes Lesson overview Students share what they know about the three states of matter solid, liquid and gas and consider some of their scientific properties. They
Solids, liquids and gases Duration 60 minutes Lesson overview Students share what they know about the three states of matter solid, liquid and gas and consider some of their scientific properties. They
ACTIVITY 1.1 SOLID WATER AND LIQUID WATER
 TEACHING SUGGESTIONS: ACTIVITY 1.1 SOLID WATER AND LIQUID WATER 1. Separate the class into groups of about three students each and distribute the materials. Circulate around the room to monitor the activity
TEACHING SUGGESTIONS: ACTIVITY 1.1 SOLID WATER AND LIQUID WATER 1. Separate the class into groups of about three students each and distribute the materials. Circulate around the room to monitor the activity
What Is The Matter? Matter Concept Map
 What Is The Matter? Matter Concept Map Phases of Matter Solid Liquid Gas Use the selections below to complete the concept map for the phases of matter. Write the letter of each characteristic in the appropriate
What Is The Matter? Matter Concept Map Phases of Matter Solid Liquid Gas Use the selections below to complete the concept map for the phases of matter. Write the letter of each characteristic in the appropriate
SAM Teachers Guide Newton s Laws at the Atomic Scale Overview Learning Objectives Possible Student Pre/Misconceptions
 SAM Teachers Guide Newton s Laws at the Atomic Scale Overview Students explore how Newton s three laws apply to the world of atoms and molecules. Observations start with the originally contradictory observation
SAM Teachers Guide Newton s Laws at the Atomic Scale Overview Students explore how Newton s three laws apply to the world of atoms and molecules. Observations start with the originally contradictory observation
Matter combines to form different substances
 500 500 500 500 500 500 Matter has mass and volume What s the matter Matter combines to form different substances Matter exists in different physical states Matter is made of atoms Miscellaneous 100 100
500 500 500 500 500 500 Matter has mass and volume What s the matter Matter combines to form different substances Matter exists in different physical states Matter is made of atoms Miscellaneous 100 100
KINETIC PARTICLE THEORY
 KINETIC PARTICLE THEORY IMPORTANT DEFINITIONS: The mixing process in gases or solutions due to the random motion of particles is called Diffusion. The process by which a liquid changes into a vapour at
KINETIC PARTICLE THEORY IMPORTANT DEFINITIONS: The mixing process in gases or solutions due to the random motion of particles is called Diffusion. The process by which a liquid changes into a vapour at
States of Matter. Solids, Liquids, and Gases
 States of Matter Solids, Liquids, and Gases What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
States of Matter Solids, Liquids, and Gases What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with the statement
INTRODUCTION TO LESSON CLUSTER 8 Explaining Evaporation and Boiling
 INTRODUCTION TO LESSON CLUSTER 8 Explaining Evaporation and Boiling A. Lesson Cluster Goals and Lesson Objectives Goals: Students should be able to explain evaporation and boiling, both in macroscopic
INTRODUCTION TO LESSON CLUSTER 8 Explaining Evaporation and Boiling A. Lesson Cluster Goals and Lesson Objectives Goals: Students should be able to explain evaporation and boiling, both in macroscopic
TEACHER NOTES: ICE CUBE POSTER
 TEACHER NOTES: NATIONAL CURRICULUM LINKS THE PARTICULATE NATURE OF MATTER the properties of the different states of matter (solid, liquid and gas) in terms of the particle model, including gas pressure
TEACHER NOTES: NATIONAL CURRICULUM LINKS THE PARTICULATE NATURE OF MATTER the properties of the different states of matter (solid, liquid and gas) in terms of the particle model, including gas pressure
Chapter 6 Kinetic Particle Theory
 Chapter 6 Kinetic Particle Theory To understand that matter is made up of small particles which are in constant and random motion. To describe simple model of solids, liquids and gases, in terms of the
Chapter 6 Kinetic Particle Theory To understand that matter is made up of small particles which are in constant and random motion. To describe simple model of solids, liquids and gases, in terms of the
Foundations of Chemistry
 Name Foundations of Chemistry What is matter, and how does it change? Date Before You Read Before you read the chapter, think about what you know about matter and how it changes Record three things that
Name Foundations of Chemistry What is matter, and how does it change? Date Before You Read Before you read the chapter, think about what you know about matter and how it changes Record three things that
Supplemental Material
 Supplemental Material Chemical and Physical Change Assessment CPCA γ-version This assessment can be fashioned into a quiz booklet using the following cover page. At the end of the quiz is a customized
Supplemental Material Chemical and Physical Change Assessment CPCA γ-version This assessment can be fashioned into a quiz booklet using the following cover page. At the end of the quiz is a customized
Chapter 3. Preview. Section 1 Three States of Matter. Section 2 Behavior of Gases. Section 3 Changes of State. States of Matter.
 States of Matter Preview Section 1 Three States of Matter Section 2 Behavior of Gases Section 3 Changes of State Concept Mapping Section 1 Three States of Matter Bellringer In the kitchen, you might find
States of Matter Preview Section 1 Three States of Matter Section 2 Behavior of Gases Section 3 Changes of State Concept Mapping Section 1 Three States of Matter Bellringer In the kitchen, you might find
Matter, Atoms & Molecules
 Matter, Atoms & Molecules Matter is anything that has mass and takes up space. All matter is made of tiny particles called atoms, which are too small to see with the naked eye. Matter Matter is anything
Matter, Atoms & Molecules Matter is anything that has mass and takes up space. All matter is made of tiny particles called atoms, which are too small to see with the naked eye. Matter Matter is anything
S8P All of the substances on the periodic table are classified as elements because they
 S8P1-2 1. Putting sand and salt together makes A. a compound. B. an element. C. a mixture. D. a solution. 2. All of the substances on the periodic table are classified as elements because they A. are pure
S8P1-2 1. Putting sand and salt together makes A. a compound. B. an element. C. a mixture. D. a solution. 2. All of the substances on the periodic table are classified as elements because they A. are pure
1 Characteristics of the Atmosphere
 CHAPTER 15 1 Characteristics of the Atmosphere SECTION The Atmosphere BEFORE YOU READ After you read this section, you should be able to answer these questions: What is Earth s atmosphere made of? How
CHAPTER 15 1 Characteristics of the Atmosphere SECTION The Atmosphere BEFORE YOU READ After you read this section, you should be able to answer these questions: What is Earth s atmosphere made of? How
Name: 1. Which of the following is probably true about 300 ml of sand and 300 ml of water?
 Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Most substances can be in three states: solid, liquid, and gas.
 States of Matter Most substances can be in three states: solid, liquid, and gas. Solid Particles Have Fixed Positions The particles in a solid are very close together and have an orderly, fixed arrangement.
States of Matter Most substances can be in three states: solid, liquid, and gas. Solid Particles Have Fixed Positions The particles in a solid are very close together and have an orderly, fixed arrangement.
S8P1C. STATES OF MATTER E.Q. WHAT ARE THE STATES OF MATTER? WHAT ARE THE CHARACTERISTICS OF EACH STATE OF MATTER?
 S8P1C. STATES OF MATTER E.Q. WHAT ARE THE STATES OF MATTER? WHAT ARE THE CHARACTERISTICS OF EACH STATE OF MATTER? ATOMIC MODEL PROJECT BE CREATIVE!!! MIXTURES VS. SUBSTANCES TASK DUE BY FRIDAY, NOVEMBER
S8P1C. STATES OF MATTER E.Q. WHAT ARE THE STATES OF MATTER? WHAT ARE THE CHARACTERISTICS OF EACH STATE OF MATTER? ATOMIC MODEL PROJECT BE CREATIVE!!! MIXTURES VS. SUBSTANCES TASK DUE BY FRIDAY, NOVEMBER
Science 8 Chapter 7 Section 1
 Science 8 Chapter 7 Section 1 Describing Fluids (pp. 268-277) What is a fluid? Fluid: any thing that flows; a liquid or a gas While it would seem that some solids flow (sugar, salt, etc), they are not
Science 8 Chapter 7 Section 1 Describing Fluids (pp. 268-277) What is a fluid? Fluid: any thing that flows; a liquid or a gas While it would seem that some solids flow (sugar, salt, etc), they are not
Solids (cont.) Describe the movement of particles in a solid and the forces between them.
 Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
States of Matter. What physical changes and energy changes occur as matter goes from one state to another?
 Name States of Matter Date What physical changes and energy changes occur as matter goes from one state to another? Before You Read Before you read the chapter, think about what you know about states of
Name States of Matter Date What physical changes and energy changes occur as matter goes from one state to another? Before You Read Before you read the chapter, think about what you know about states of
Solids, Liquids, and Gases. Chapter 14
 Solids, Liquids, and Gases Chapter 14 Matter & Thermal Energy Matter can exist as a solid, a liquid, a gas or a plasma. The Molecular Kinetic Theory of Matter explains their differences and how they can
Solids, Liquids, and Gases Chapter 14 Matter & Thermal Energy Matter can exist as a solid, a liquid, a gas or a plasma. The Molecular Kinetic Theory of Matter explains their differences and how they can
SOLIDS, LIQUIDS, AND GASES
 CHAPTER 2 SOLIDS, LIQUIDS, AND GASES SECTION 2 1 States of Matter (pages 56-60) This section explains how shape, volume, and the motion of particles are useful in describing solids, liquids, and gases.
CHAPTER 2 SOLIDS, LIQUIDS, AND GASES SECTION 2 1 States of Matter (pages 56-60) This section explains how shape, volume, and the motion of particles are useful in describing solids, liquids, and gases.
Properties and Structure of Matter
 Properties and Structure of Matter Chapter 10 You can use a spider map to organize the main ideas and supporting details of a topic such as properties of matter. Look at the example shown below. The central
Properties and Structure of Matter Chapter 10 You can use a spider map to organize the main ideas and supporting details of a topic such as properties of matter. Look at the example shown below. The central
States of Matter: A Solid Lesson where Liquids Can be a Gas!
 TEACHER GUIDE STATES OF MATTER 60 Minute Physical Science Lesson Science- to- Go! Program Grades: 1-3 States of Matter: A Solid Lesson where Liquids Can be a Gas! Description Your classroom will be converted
TEACHER GUIDE STATES OF MATTER 60 Minute Physical Science Lesson Science- to- Go! Program Grades: 1-3 States of Matter: A Solid Lesson where Liquids Can be a Gas! Description Your classroom will be converted
Lesson 9: States of Matter
 Lesson 9: States of Matter Do Now 6O, 6S 11.8.18 Take out HW 6.14 to be checked. Copy info into CJ keep CJ out and open on desk throughout class. On Do Now Page #5, copy and answer: 1. If you use a magnet
Lesson 9: States of Matter Do Now 6O, 6S 11.8.18 Take out HW 6.14 to be checked. Copy info into CJ keep CJ out and open on desk throughout class. On Do Now Page #5, copy and answer: 1. If you use a magnet
1 Characteristics of the Atmosphere
 CHAPTER 1 1 Characteristics of the Atmosphere SECTION The Atmosphere BEFORE YOU READ After you read this section, you should be able to answer these questions: What is Earth s atmosphere made of? How do
CHAPTER 1 1 Characteristics of the Atmosphere SECTION The Atmosphere BEFORE YOU READ After you read this section, you should be able to answer these questions: What is Earth s atmosphere made of? How do
OBJECTIVES: By the end of today s lesson, you will be able to. SWBAT list the layers of the atmosphere and describe the characteristics of each one.
 7 th Grade Science Unit: Water s Cycles and Patterns Lesson: WCP 10 Name: Date: Monday, September 12, 2016 Homeroom: OBJECTIVES: By the end of today s lesson, you will be able to SWBAT list the layers
7 th Grade Science Unit: Water s Cycles and Patterns Lesson: WCP 10 Name: Date: Monday, September 12, 2016 Homeroom: OBJECTIVES: By the end of today s lesson, you will be able to SWBAT list the layers
Unsaved Test, Version: 1 1
 Name: Key Concepts Select the term that best completes the statement. A. beam balance B. graduated cylinder C. scale D. matter E. mass F. newton G. volume H. height I. weight J. kilogram 1. Anything that
Name: Key Concepts Select the term that best completes the statement. A. beam balance B. graduated cylinder C. scale D. matter E. mass F. newton G. volume H. height I. weight J. kilogram 1. Anything that
SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES
 30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
CHEMISTRY Matter and Change. Chapter 12: States of Matter
 CHEMISTRY Matter and Change Chapter 12: States of Matter CHAPTER 12 States of Matter Section 12.1 Section 12.2 Section 12.3 Section 12.4 Gases Forces of Attraction Liquids and Solids Phase Changes Click
CHEMISTRY Matter and Change Chapter 12: States of Matter CHAPTER 12 States of Matter Section 12.1 Section 12.2 Section 12.3 Section 12.4 Gases Forces of Attraction Liquids and Solids Phase Changes Click
Matter, mass, and volume are related.
 Suppose you had all of the parts needed to make an at home aquarium: a tank, water, rocks, plants, and some fish. To put the aquarium together, you would need to arrange the rocks and plants in the tank.
Suppose you had all of the parts needed to make an at home aquarium: a tank, water, rocks, plants, and some fish. To put the aquarium together, you would need to arrange the rocks and plants in the tank.
STATES OF MATTER NOTES..
 STATES OF MATTER NOTES.. While you are reading, answer the following which will help you with the States of Matter Project. What is matter (definition): What are the states of matter and what are the characteristics/properties
STATES OF MATTER NOTES.. While you are reading, answer the following which will help you with the States of Matter Project. What is matter (definition): What are the states of matter and what are the characteristics/properties
ASSESSMENT CHART FOR INVESTIGATIONS 1 AND 2 STUDENT NAME
 ASSESSMENT CHART FOR INVESTIGATIONS 1 AND 2 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. STUDENT NAME Weather Measurements (TO)
ASSESSMENT CHART FOR INVESTIGATIONS 1 AND 2 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. STUDENT NAME Weather Measurements (TO)
Chapter 8. Chapter 8. Preview. Bellringer. Chapter 8. Particles of Matter. Objectives. Chapter 8. Particles of Matter, continued
 States of Matter Preview Bellringer Section 2 Behavior of Gases In the kitchen, you might find three different forms of water. What are these three forms of water, and where exactly in the kitchen would
States of Matter Preview Bellringer Section 2 Behavior of Gases In the kitchen, you might find three different forms of water. What are these three forms of water, and where exactly in the kitchen would
Classification of Matter. Chapter 10 Classification of Matter
 Chapter 10 Classification of Matter Grade 7 Classification of Matter Matter is anything that has mass and occupies space. We can classify matter based on whether it s solid, liquid, or gas. 2 1 Understanding
Chapter 10 Classification of Matter Grade 7 Classification of Matter Matter is anything that has mass and occupies space. We can classify matter based on whether it s solid, liquid, or gas. 2 1 Understanding
SAM Teachers Guide Newton s Laws at the Atomic Scale Overview Learning Objectives Possible Student Pre/Misconceptions
 SAM Teachers Guide Newton s Laws at the Atomic Scale Overview Students explore how Newton s three laws apply to the world of atoms and molecules. Observations start with the originally contradictory observation
SAM Teachers Guide Newton s Laws at the Atomic Scale Overview Students explore how Newton s three laws apply to the world of atoms and molecules. Observations start with the originally contradictory observation
The of that surrounds the Earth. Atmosphere. A greenhouse that has produced the most global. Carbon Dioxide
 Name: Date: # Weather and Climate Unit Review Directions: Complete this packet to help you prepare for your unit test by filling in the blanks to complete the definitions. Then if no picture is provided,
Name: Date: # Weather and Climate Unit Review Directions: Complete this packet to help you prepare for your unit test by filling in the blanks to complete the definitions. Then if no picture is provided,
Gases and States of Matter: Unit 8
 Gases and States of Matter: Unit 8 States of Matter There are three states (also called phases) of matter. The picture represents the same chemical substance, just in different states. There are three
Gases and States of Matter: Unit 8 States of Matter There are three states (also called phases) of matter. The picture represents the same chemical substance, just in different states. There are three
Chapter 22 States of matter. Section 1 matter Section 2 Changes of State
 Chapter 22 States of matter Section 1 matter Section 2 Changes of State States of Matter is a physical property ***Matter is made of atoms Atoms form chemical bonds to make matter **** Atoms vibrate constantly
Chapter 22 States of matter Section 1 matter Section 2 Changes of State States of Matter is a physical property ***Matter is made of atoms Atoms form chemical bonds to make matter **** Atoms vibrate constantly
OBJECTIVES: By the end of class, students will be able to DO NOW
 7 th Grade Science Unit: Matter and Periodic Table Lesson: Matter 9 States of Matter Demonstrations Name: Date: Thursday, December 8, 2016 OBJECTIVES: By the end of class, students will be able to SWBAT
7 th Grade Science Unit: Matter and Periodic Table Lesson: Matter 9 States of Matter Demonstrations Name: Date: Thursday, December 8, 2016 OBJECTIVES: By the end of class, students will be able to SWBAT
Notes: Phases of Matter and Phase Changes
 Name: Date: IP 670 Notes: Phases of Matter and Phase Changes There are four main phases of matter: We are only going to talk about the first three today. Solids Liquids Gases Molecular Molecules Wiggle
Name: Date: IP 670 Notes: Phases of Matter and Phase Changes There are four main phases of matter: We are only going to talk about the first three today. Solids Liquids Gases Molecular Molecules Wiggle
1 Characteristics of the Atmosphere
 CHAPTER 15 1 Characteristics of the Atmosphere SECTION The Atmosphere BEFORE YOU READ After you read this section, you should be able to answer these questions: What is Earth s atmosphere made of? How
CHAPTER 15 1 Characteristics of the Atmosphere SECTION The Atmosphere BEFORE YOU READ After you read this section, you should be able to answer these questions: What is Earth s atmosphere made of? How
States of matter. 22 Science Alive for VELS Level 5
 States of matter E verything around you is made of matter. Anything that has mass and takes up space is matter. The air we breathe, the water we drink and the food we eat are all different types of matter.
States of matter E verything around you is made of matter. Anything that has mass and takes up space is matter. The air we breathe, the water we drink and the food we eat are all different types of matter.
Chapter 14 9/21/15. Solids, Liquids & Gasses. Essential Questions! Kinetic Theory! Gas State! Gas State!
 Chapter 14 Solids, Liquids & Gasses Essential Questions What is the kinetic theory of matter? How do particles move in the different states of matter? How do particles behave at the boiling and melting
Chapter 14 Solids, Liquids & Gasses Essential Questions What is the kinetic theory of matter? How do particles move in the different states of matter? How do particles behave at the boiling and melting
Matter and Its Properties. Unit 2
 Matter and Its Properties Unit 2 Lesson 1: Physical & Chemical Properties & Changes Unit 2: Matter and Its Properties Section 1: Physical Properties & Change Lesson 1: Physical & Chemical Properties &
Matter and Its Properties Unit 2 Lesson 1: Physical & Chemical Properties & Changes Unit 2: Matter and Its Properties Section 1: Physical Properties & Change Lesson 1: Physical & Chemical Properties &
IES LAURETUM SCIENCE NAME.
 IES LAURETUM SCIENCE NAME. 1 CONTENTS 1. STATES 6F 0ATTER 2. STATES 6F 0ATTER AND THE5R *R6*ERT5ES 3. 25NET5C THE6RY 4. CHANGE 6F STATE 6F 0ATTER Break the code: Find the letter for each number * = 0 =
IES LAURETUM SCIENCE NAME. 1 CONTENTS 1. STATES 6F 0ATTER 2. STATES 6F 0ATTER AND THE5R *R6*ERT5ES 3. 25NET5C THE6RY 4. CHANGE 6F STATE 6F 0ATTER Break the code: Find the letter for each number * = 0 =
6th Grade: Great Salt Lake is Salty
 Curriculum written by Megan Black in partnership with The Great Salt Lake Institute at Westminster College. 6th Grade: Great Salt Lake is Salty Lesson Description: In this lesson students will compare
Curriculum written by Megan Black in partnership with The Great Salt Lake Institute at Westminster College. 6th Grade: Great Salt Lake is Salty Lesson Description: In this lesson students will compare
STATES OF MATTER. The Four States of Ma/er. Four States. Solid Liquid Gas Plasma
 STATES OF MATTER The Four States of Ma/er Solid Liquid Gas Plasma Four States STATES OF MATTER Ø What makes a substance a par:cular state of ma
STATES OF MATTER The Four States of Ma/er Solid Liquid Gas Plasma Four States STATES OF MATTER Ø What makes a substance a par:cular state of ma
2. What is meant by Chemical State?. 3. Changing states of matter is about changing,,, and other.
 Name: Date: Period: Matter Mania! Online Computer Activity (3 pages) Part I: Go to http://www.chem4kids.com/ and answer the following questions in complete sentences. a. Click on MATTER (written in yellow)
Name: Date: Period: Matter Mania! Online Computer Activity (3 pages) Part I: Go to http://www.chem4kids.com/ and answer the following questions in complete sentences. a. Click on MATTER (written in yellow)
Matter. Gas. Solid Liquid. Both shape and volume are not fixed. It has a fixed shape and a fixed volume.
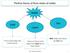 Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
EXTRA CREDIT PAGES K
 Mixtures (13K) EXTRA CREDIT PAGES 13-14 K Elements and compounds are pure substances, but most of the materials you see every day are not. Instead, they are mixtures. A mixture is made of two or more substances
Mixtures (13K) EXTRA CREDIT PAGES 13-14 K Elements and compounds are pure substances, but most of the materials you see every day are not. Instead, they are mixtures. A mixture is made of two or more substances
Leonie Boshoff-Mostert Edited by Anne Starace
 GASES, LIQUIDS AND SOLIDS Density Leonie Boshoff-Mostert Edited by Anne Starace Abstract Matter is sorted into three groups: solids, liquids and gases. Solids, liquids and gases each have characteristic
GASES, LIQUIDS AND SOLIDS Density Leonie Boshoff-Mostert Edited by Anne Starace Abstract Matter is sorted into three groups: solids, liquids and gases. Solids, liquids and gases each have characteristic
Close!Reading!and!Text!Dependent!Questions!in!Science! Particles!in!Motion!(Chemical!Interactions Grade!8)!
 CloseReadingandTextDependentQuestionsinScience ParticlesinMotion(ChemicalInteractions Grade8) Thetextselection,ParticlesinMotion,isfoundin FOSSStudentResourceBook,ChemicalInteractions,pgs.23A27. Science
CloseReadingandTextDependentQuestionsinScience ParticlesinMotion(ChemicalInteractions Grade8) Thetextselection,ParticlesinMotion,isfoundin FOSSStudentResourceBook,ChemicalInteractions,pgs.23A27. Science
What Is Air Temperature?
 2.2 Read What Is Air Temperature? In Learning Set 1, you used a thermometer to measure air temperature. But what exactly was the thermometer measuring? What is different about cold air and warm air that
2.2 Read What Is Air Temperature? In Learning Set 1, you used a thermometer to measure air temperature. But what exactly was the thermometer measuring? What is different about cold air and warm air that
Chemistry A: States of Matter Packet Name: Hour: Page 1. Chemistry A States of Matter Packet
 Chemistry A: States of Matter Packet Name: Hour: Page 1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page 2 Worksheet #1: States of Matter In this packet we will
Chemistry A: States of Matter Packet Name: Hour: Page 1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page 2 Worksheet #1: States of Matter In this packet we will
Second Grade: Unit 2: Properties of Matter. Matter solid liquid gas property
 Second Grade: Unit 2: Properties of Matter Matter solid liquid gas property Background: The universe is made of only two entities: matter and energy. Examples of energy are light, heat, and sound. Everything
Second Grade: Unit 2: Properties of Matter Matter solid liquid gas property Background: The universe is made of only two entities: matter and energy. Examples of energy are light, heat, and sound. Everything
SAM Teachers Guide Phase Change Overview Learning Objectives Possible Student Pre/Misconceptions
 SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion and observing and manipulating
SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion and observing and manipulating
composition of matter, and the changes that matter undergoes. Examples of Uses of Chemistry in Everyday Life
 Name Matter and Change: Unit Objective Study Guide Date Due Directions: Write your answers to the following questions in the space provided. For problem solving, all of the work leading up to the final
Name Matter and Change: Unit Objective Study Guide Date Due Directions: Write your answers to the following questions in the space provided. For problem solving, all of the work leading up to the final
Chemistry A: States of Matter Packet Name: Hour: Page!1. Chemistry A States of Matter Packet
 Chemistry A: States of Matter Packet Name: Hour: Page!1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page!2 Worksheet #1: States of Matter In this packet we will
Chemistry A: States of Matter Packet Name: Hour: Page!1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page!2 Worksheet #1: States of Matter In this packet we will
Foundations of Chemistry
 Foundations of Chemistry Physical Properties Physical Properties As you read in Lesson 1, the arrangement of atoms determines whether matter is a substance or a mixture. The arrangement of atoms also determines
Foundations of Chemistry Physical Properties Physical Properties As you read in Lesson 1, the arrangement of atoms determines whether matter is a substance or a mixture. The arrangement of atoms also determines
10 February 2009 Aim: How can I tell the difference between substances, compounds, and mixtures? Engagement: write three to four sentences describing
 10 February 2009 Aim: How can I tell the difference between substances, compounds, and mixtures? Engagement: write three to four sentences describing what you know about atoms. HW: Make note cards for
10 February 2009 Aim: How can I tell the difference between substances, compounds, and mixtures? Engagement: write three to four sentences describing what you know about atoms. HW: Make note cards for
NAME: ACTIVITY SHEETS PHYSICS AND CHEMISTRY (SECONDARY 3 rd YEAR)
 NAME: ACTIVITY SHEETS PHYSICS AND CHEMISTRY (SECONDARY 3 rd YEAR) ACTIVITY 1: Matter Lesson 2 THE PARTICULATE NATURE OF MATTER 1-What is matter? 2-What is a particle (corpuscle)? Set some examples 3-What
NAME: ACTIVITY SHEETS PHYSICS AND CHEMISTRY (SECONDARY 3 rd YEAR) ACTIVITY 1: Matter Lesson 2 THE PARTICULATE NATURE OF MATTER 1-What is matter? 2-What is a particle (corpuscle)? Set some examples 3-What
1st Nine Weeks Midterm Study Guide
 Name: ate: 1. Which statement correctly describes a property of a type of matter?. ir is a mixture of gases.. Ice is a mixture of gases.. ir is a liquid.. Ice is a liquid. 4. Why is cobalt (o) placed before
Name: ate: 1. Which statement correctly describes a property of a type of matter?. ir is a mixture of gases.. Ice is a mixture of gases.. ir is a liquid.. Ice is a liquid. 4. Why is cobalt (o) placed before
Changing States of Matter By Cindy Grigg
 By Cindy Grigg 1 On Earth, almost all matter exists in just three states. Matter is usually a solid, a liquid, or a gas. Plasma, the fourth state of matter, is rare on Earth. It sometimes can be found
By Cindy Grigg 1 On Earth, almost all matter exists in just three states. Matter is usually a solid, a liquid, or a gas. Plasma, the fourth state of matter, is rare on Earth. It sometimes can be found
Leonie Boshoff-Mostert Edited by Anne Starace
 GASES, LIQUIDS AND SOLIDS: Condensation Leonie Boshoff-Mostert Edited by Anne Starace Abstract Matter is sorted into three groups: solids, liquids and gases. Solids, liquids and gases each have characteristic
GASES, LIQUIDS AND SOLIDS: Condensation Leonie Boshoff-Mostert Edited by Anne Starace Abstract Matter is sorted into three groups: solids, liquids and gases. Solids, liquids and gases each have characteristic
Pure Substances and Mixtures Picture Vocabulary. 8P1A Pure Substances and Mixtures
 Pure Substances and Mixtures Picture Vocabulary 8P1A Pure Substances and Mixtures T.I.P Chart for pure substances & mixtures terms Make a TIP chart to use for terms about pure substances & mixtures. Write
Pure Substances and Mixtures Picture Vocabulary 8P1A Pure Substances and Mixtures T.I.P Chart for pure substances & mixtures terms Make a TIP chart to use for terms about pure substances & mixtures. Write
Chemistry Final Study Guide KEY. 3. Define physical changes. A change in any physical property of a substance, not in the substance itself.
 Chemistry Final Study Guide KEY Unit 2: Matter & Its Properties, Lesson 1: Physical and Chemical Properties & Changes 1. Define physical properties. The characteristics of a substance that can be observed
Chemistry Final Study Guide KEY Unit 2: Matter & Its Properties, Lesson 1: Physical and Chemical Properties & Changes 1. Define physical properties. The characteristics of a substance that can be observed
MS-PS1 Matter and Its Interactions
 MS-PS1 Matter and Its Interactions Key Ideas and Common Student Misconceptions This document compiles Key Ideas and developed by AAAS Project 2061 and the National Science Foundation. For additional information
MS-PS1 Matter and Its Interactions Key Ideas and Common Student Misconceptions This document compiles Key Ideas and developed by AAAS Project 2061 and the National Science Foundation. For additional information
Name: Matter Review Packet 2017
 Name: Matter Review Packet 2017 1. At STP, which 2.0-gram sample of matter uniformly fills a 340-milliliter closed container? A) Br2(l) B) Fe(NO3)2(aq) C) KCl(s) D) Ne(g) 2. The particles in a crystalline
Name: Matter Review Packet 2017 1. At STP, which 2.0-gram sample of matter uniformly fills a 340-milliliter closed container? A) Br2(l) B) Fe(NO3)2(aq) C) KCl(s) D) Ne(g) 2. The particles in a crystalline
How are elements, compounds, and mixtures related?
 Essential Question: How are elements, compounds, and mixtures related? S8P1b. Describe the difference between pure substances (elements and compounds) and mixtures Matter is anything that has mass and
Essential Question: How are elements, compounds, and mixtures related? S8P1b. Describe the difference between pure substances (elements and compounds) and mixtures Matter is anything that has mass and
Lesson 1.2 Classifying Matter
 Lesson 1.2 Classifying Matter Vocabulary element atom chemical bond mixture molecule compound chemical formula What is Matter Made Of? What is matter? Why is one kind of matter different from another kind
Lesson 1.2 Classifying Matter Vocabulary element atom chemical bond mixture molecule compound chemical formula What is Matter Made Of? What is matter? Why is one kind of matter different from another kind
Limiting Reactants How do you know if there will be enough of each chemical to make your desired product?
 Limiting Reactants How do you know if there will be enough of each chemical to make your desired product? Why? If a factory runs out of tires while manufacturing a car, all production stops. No more cars
Limiting Reactants How do you know if there will be enough of each chemical to make your desired product? Why? If a factory runs out of tires while manufacturing a car, all production stops. No more cars
Matter & Energy. Objectives: properties and structures of the different states of matter.
 Matter & Energy Objectives: 1. Use the kinetic theory to describe the properties and structures of the different states of matter. 2. Describe energy transfers involved in changes of state. 3. Describe
Matter & Energy Objectives: 1. Use the kinetic theory to describe the properties and structures of the different states of matter. 2. Describe energy transfers involved in changes of state. 3. Describe
Everything in the universe can be classified as either matter or energy. Kinetic Energy Theory: All particles of matter are in constant motion.
 Physical Science Everything in the universe can be classified as either matter or energy. Kinetic Energy Theory: All particles of matter are in constant motion. State of Matter Bose- Einstein Condensate
Physical Science Everything in the universe can be classified as either matter or energy. Kinetic Energy Theory: All particles of matter are in constant motion. State of Matter Bose- Einstein Condensate
Unit 13 Lesson 1 What Are Solids, Liquids, and Gases? Copyright Houghton Mifflin Harcourt Publishing Company
 Unit 13 Lesson 1 What Are Solids, Liquids, and Gases? Copyright Houghton Mifflin Harcourt Publishing Company What s the Matter? Matter has mass and volume. It cannot be created or destroyed. Mass is the
Unit 13 Lesson 1 What Are Solids, Liquids, and Gases? Copyright Houghton Mifflin Harcourt Publishing Company What s the Matter? Matter has mass and volume. It cannot be created or destroyed. Mass is the
SAM Teachers Guide Phase Change Overview Learning Objectives Possible Student Pre/Misconceptions
 SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion. Students observe and manipulate
SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion. Students observe and manipulate
Section 1: What is a Chemical Reaction
 Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
STATES OF MATTER STATES OF MATTER. The Four States of Matter 3/5/2015. Solid. Liquid Commonly found on Gas Earth Plasma
 Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) STATES OF MATTER The Four States of Matter Solid } Liquid Commonly found on Gas Earth Plasma STATES OF MATTER Based upon particle
Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) STATES OF MATTER The Four States of Matter Solid } Liquid Commonly found on Gas Earth Plasma STATES OF MATTER Based upon particle
STATES OF MATTER STATES OF MATTER. The Four States of Matter 3/5/2015
 The Four States of Matter Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) Solid } Liquid Commonly found on Gas Earth Plasma Based upon particle arrangement Based upon energy of
The Four States of Matter Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) Solid } Liquid Commonly found on Gas Earth Plasma Based upon particle arrangement Based upon energy of
Dissolving vs. phase change. What happens to the atoms and the substances?
 Dissolving vs. phase change What happens to the atoms and the substances? Atoms and molecules are the building blocks of matter! What did you learn about them from your readings? What happened in the Saltwater
Dissolving vs. phase change What happens to the atoms and the substances? Atoms and molecules are the building blocks of matter! What did you learn about them from your readings? What happened in the Saltwater
Unit 4: Gas Laws. Matter and Phase Changes
 Unit 4: Gas Laws Matter and Phase Changes ENERGY and matter What is 에너지 A fundamental property of the universe that cannot be easily defined. Energy No one knows what energy is, only what it does or has
Unit 4: Gas Laws Matter and Phase Changes ENERGY and matter What is 에너지 A fundamental property of the universe that cannot be easily defined. Energy No one knows what energy is, only what it does or has
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 1, LESSON 1 MOLECULES MATTER MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state of a pure substance
The Next Generation Science Standards (NGSS) CHAPTER 1, LESSON 1 MOLECULES MATTER MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state of a pure substance
Objectives: 1. Define the atom and parts of the atom 2. Define the properties of an atom; molecules, compounds; states of matter
 Atomic Nature of Matter Objectives: 1. Define the atom and parts of the atom 2. Define the properties of an atom; molecules, compounds; states of matter Elements Elements class of identical atoms Living
Atomic Nature of Matter Objectives: 1. Define the atom and parts of the atom 2. Define the properties of an atom; molecules, compounds; states of matter Elements Elements class of identical atoms Living
5 th Grade Lesson Plan: Matter and Chemical Reactions
 5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
2º ESO UNIT 2: The physical states of matter. Susana Morales Bernal
 2º ESO UNIT 2: The physical states of matter Objectives 1. To know that in all the states of aggregation the matter has mass and takes a place although depending on the state, can have or not, form and
2º ESO UNIT 2: The physical states of matter Objectives 1. To know that in all the states of aggregation the matter has mass and takes a place although depending on the state, can have or not, form and
Classification of Matter. States of Matter Physical and Chemical Properties Physical and Chemical Changes
 1 Classification of Matter States of Matter Physical and Chemical Properties Physical and Chemical Changes 2 Classification of Matter Matter is anything that has mass and occupies space. We can classify
1 Classification of Matter States of Matter Physical and Chemical Properties Physical and Chemical Changes 2 Classification of Matter Matter is anything that has mass and occupies space. We can classify
Name Date Class STATES OF MATTER
 13 STATES OF MATTER Chapter Test A A. Matching Match each description in Column B with the correct term in Column A. Write the letter of the correct description on the line. Column A Column B 1. amorphous
13 STATES OF MATTER Chapter Test A A. Matching Match each description in Column B with the correct term in Column A. Write the letter of the correct description on the line. Column A Column B 1. amorphous
Science Wednesday - Friday September 21st - 23rd EQ: How is water important? What gives water its unique properties?
 Science Wednesday - Friday September 21st - 23rd EQ: How is water important? What gives water its unique properties? On your desk: Paper Pencil for notes Assignments: Thirsty Abe Climbing Water Water Metal
Science Wednesday - Friday September 21st - 23rd EQ: How is water important? What gives water its unique properties? On your desk: Paper Pencil for notes Assignments: Thirsty Abe Climbing Water Water Metal
Land and Water Study Guide
 Land and Water Study Guide Answer Key Part 1 States of Matter 1. What are the three states of matter for water? Give several examples for each. Solid Ice cube (non water examples = candy bar and a log).
Land and Water Study Guide Answer Key Part 1 States of Matter 1. What are the three states of matter for water? Give several examples for each. Solid Ice cube (non water examples = candy bar and a log).
