Chapter 14 9/21/15. Solids, Liquids & Gasses. Essential Questions! Kinetic Theory! Gas State! Gas State!
|
|
|
- Beverly Gregory
- 5 years ago
- Views:
Transcription
1 Chapter 14 Solids, Liquids & Gasses Essential Questions What is the kinetic theory of matter? How do particles move in the different states of matter? How do particles behave at the boiling and melting points? Kinetic Theory Gas State The kinetic theory is an explanation of how particles in matter behave. The three assumptions of the kinetic theory are as follows: All matter is composed of small particles (atoms, molecules, or ions). These particles are in constant, random motion. These particles are colliding with each other and the walls of their container. Particles lose some energy during collisions with other particles. But the amount of energy lost is very small and can be neglected in most cases. Gas State Gases do not have a fixed volume or shape. Therefore, they can spread far apart or contract to fill the container that they are in. 1
2 Liquid state Particles in the liquid state have less kinetic energy than in the gas state. Thus, the particles in the liquid state are less able to overcome their attractions to each other. The particles cling together, giving liquids a definite volume. The particles can slide past each other, allowing liquids to flow and take the shape of their container. Solid State The particles of a solid are closely packed together. Most solid materials have a specific type of geometric arrangement in which they form when cooled. Solid State The type of geometric arrangement formed by a solid is important. Chemical and physical properties of solids often can be attributed to the type of geometric arrangement that the solid forms. Thermal energy What happens to a solid when thermal energy or heat is added to it? The particles on the surface of the solid vibrate faster. These particles collide with and transfer energy to other particles. Soon the particles have enough kinetic energy to overcome the attractive forces. 2
3 Thermal energy Thermal energy is the total energy of a material s particles, including kinetic vibrations and movement within and between the particles and potential resulting from forces that act within or between particles. Temperature In science, temperature means the average kinetic energy of particles in the substance, or how fast the particles are moving. On average, molecules of frozen water at 0 C will move slower than molecules of water at 100 C. Water molecules at 0 C have lower average kinetic energy than the molecules at 100 C. Molecules have kinetic energy at all temperatures, including absolute zero. Changes of State Melting and freezing The particles gain enough kinetic energy to slip out of their ordered arrangement and the solid melts. This is known as the melting point, or the temperature at which a solid begins to liquefy. Energy is required for the particles to slip out of the ordered arrangement. Vaporization and condensation How does a liquid become a gas? The particles in a liquid are constantly moving. Some particles are moving faster and have more kinetic energy than others. The particles that are moving fast enough can escape the attractive forces of other particles and enter the gas state. This process is called vaporization. The amount of energy required to change a substance from the solid phase to the liquid phase at its melting point is known as the heat of fusion. 3
4 Vaporization and condensation Vaporization can occur in two ways evaporation and boiling. Evaporation is vaporization that occurs at the surface of a liquid and can occur at nearly any temperature. To evaporate, particles must have enough kinetic energy to escape the attractive forces of the liquid. The boiling point of a liquid is the temperature at which the pressure of the vapor in the liquid is equal to the external pressure acting on the surface of the liquid. Heat of vaporization is the amount of energy required for the liquid at its boiling point to become a gas. Sublimation is the process of a solid changing directly to a gas without forming a liquid. Heating curves This type of graph is called a heating curve because it shows the temperature change of water as thermal energy, or heat, is added. Notice the two areas on the graph where the temperature does not change. Heating curves At 0 C, ice is melting. The temperature remains constant during melting. After the attractive forces are overcome, particles move more freely and their average kinetic energy, or temperature, increases. At 100 C, water is boiling or vaporizing and the temperature remains constant again. Plasma State Plasma is matter that has enough energy to overcome not just the attractive forces between its particles but also the attractive forces within its atoms. The forces produced from high-energy collisions are so great that electrons from the atom are stripped off. All of the observed stars including the Sun consist of plasma. Plasma also is found in lightning bolts, neon and fluorescent tubes, and auroras. 4
5 Thermal Expansion Particles move faster and separate as the temperature rises. This separation of particles results in an expansion of the entire object, known as thermal expansion. Thermal expansion is an increase in the size of a substance when the temperature is increased. The kinetic theory can be used to explain the contraction in objects, too. When the temperature of an object is lowered, particles slow down. The attraction between the particles increases and the particles move closer together. The movements of the particles closer together result in an overall shrinking of the object, known as contraction. Thermometers A common example of expansion in liquids occurs in thermometers. The addition of energy causes the particles of the liquid in the thermometer to move faster. The particles in the liquid in the narrow thermometer tube start to move farther apart as their motion increases. The liquid has to expand only slightly to show a large change on the temperature scale. Hot-air balloons Hot-air balloons Hot-air balloons are able to rise due to thermal expansion of air. The air in the balloon is heated, causing the distance between the particles in the air to increase. As the hot-air balloon expands, the number of particles per cubic centimeter decreases. This expansion results in a decreased density of the hot air. The density of the air in the hot-air balloon is lower than the density of the cooler air outside, which allows the balloon will rise. 5
6 Water s strange behavior Water molecules are unusual in that they have highly positive and highly negative areas. These charged regions affect the behavior of water. As the temperature of water drops, the particles move closer together. Water s strange behavior The unlike charges will be attracted to each other and line up so that only positive and negative zones are near each other. Because the water molecules orient themselves according to charge, empty spaces occur in the structure. Solid or Liquid? Other substances also have unusual behavior when changing states. Amorphous solids and liquid crystals are two classes of materials that do not react as you would expect when they are changing states. Amorphous solids Not all solids have a definite temperature at which they change from solid to liquid. Some solids merely soften and gradually turn into a liquid over a temperature range. These solids lack the highly ordered structure found in crystals. They are known as amorphous solids from the Greek word for without form. 6
7 Liquid Crystals Liquid crystals are placed in classes depending upon the type of order they maintain when they liquefy. They are highly responsive to temperature changes and electric fields. Scientists use the unique properties of liquid crystals to make liquid crystal displays (LCDs). LCD is common display technology for cell phones, televisions, calculators, and netbooks. LCD screens are composed of individual crystal picture elements (pixels) varying the amount of electricity that passes through the pixel determines how the crystals are aligned and whether light is able to pass through them. 7
Matter and Thermal Energy
 Section States of Matter Can you identify the states of matter present in the photo shown? Kinetic Theory The kinetic theory is an explanation of how particles in matter behave. Kinetic Theory The three
Section States of Matter Can you identify the states of matter present in the photo shown? Kinetic Theory The kinetic theory is an explanation of how particles in matter behave. Kinetic Theory The three
Kinetic Theory of Matter
 1 Temperature and Thermal Energy Kinetic Theory of Matter The motion of the particles in matter is described by kinetic theory of matter. Matter is composed of particles that are atoms, molecules, or ions
1 Temperature and Thermal Energy Kinetic Theory of Matter The motion of the particles in matter is described by kinetic theory of matter. Matter is composed of particles that are atoms, molecules, or ions
Solids, Liquids, and Gases. Chapter 14
 Solids, Liquids, and Gases Chapter 14 Matter & Thermal Energy Matter can exist as a solid, a liquid, a gas or a plasma. The Molecular Kinetic Theory of Matter explains their differences and how they can
Solids, Liquids, and Gases Chapter 14 Matter & Thermal Energy Matter can exist as a solid, a liquid, a gas or a plasma. The Molecular Kinetic Theory of Matter explains their differences and how they can
Chapter 7.1. States of Matter
 Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Solids (cont.) Describe the movement of particles in a solid and the forces between them.
 Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
States of Matter. Essential Question: How does the movement of atoms and molecules relate to matter s different phases?
 States of Matter Essential Question: How does the movement of atoms and molecules relate to matter s different phases? These notes come from pages 60 to 73 in your Physical Science Textbook All Matter
States of Matter Essential Question: How does the movement of atoms and molecules relate to matter s different phases? These notes come from pages 60 to 73 in your Physical Science Textbook All Matter
4 Discuss and evaluate the 5th state of matter. 3 - Differentiate among the four states of matter in terms of energy,
 Goal: Differentiate among the four states of matter in terms of energy, particle motion, and phase transitions. 4 States of Mater Sections 3.1, 3.2 4 Discuss and evaluate the 5 th state of matter. 3 -
Goal: Differentiate among the four states of matter in terms of energy, particle motion, and phase transitions. 4 States of Mater Sections 3.1, 3.2 4 Discuss and evaluate the 5 th state of matter. 3 -
Unit 4: The Nature of Matter
 16 16 Table of Contents Unit 4: The Nature of Matter Chapter 16: Solids, Liquids, and Gases 16.1: Kinetic Theory 16.2: Properties and Fluids 16.3: Behavior of Gases 16.1 Kinetic Theory Kinetic Theory kinetic
16 16 Table of Contents Unit 4: The Nature of Matter Chapter 16: Solids, Liquids, and Gases 16.1: Kinetic Theory 16.2: Properties and Fluids 16.3: Behavior of Gases 16.1 Kinetic Theory Kinetic Theory kinetic
Chapter: Heat and States
 Table of Contents Chapter: Heat and States of Matter Section 1: Temperature and Thermal Energy Section 2: States of Matter Section 3: Transferring Thermal Energy Section 4: Using Thermal Energy 1 Temperature
Table of Contents Chapter: Heat and States of Matter Section 1: Temperature and Thermal Energy Section 2: States of Matter Section 3: Transferring Thermal Energy Section 4: Using Thermal Energy 1 Temperature
THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES
 THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES The particle model of a gas A gas has no fixed shape or volume, but always spreads out to fill any container. There are
THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES The particle model of a gas A gas has no fixed shape or volume, but always spreads out to fill any container. There are
Matter. Energy- which is a property of matter!! Matter: anything that takes up space and has mass
 Matter Matter: anything that takes up space and has mass Can you think of anything that is not made of matter? Energy- which is a property of matter!! Matter is made up of moving particles! Instead of
Matter Matter: anything that takes up space and has mass Can you think of anything that is not made of matter? Energy- which is a property of matter!! Matter is made up of moving particles! Instead of
Chapter 3. Preview. Section 1 Three States of Matter. Section 2 Behavior of Gases. Section 3 Changes of State. States of Matter.
 States of Matter Preview Section 1 Three States of Matter Section 2 Behavior of Gases Section 3 Changes of State Concept Mapping Section 1 Three States of Matter Bellringer In the kitchen, you might find
States of Matter Preview Section 1 Three States of Matter Section 2 Behavior of Gases Section 3 Changes of State Concept Mapping Section 1 Three States of Matter Bellringer In the kitchen, you might find
Physical Science Exam 3 Study Guide. Dr. Karoline Rostamiani. Chapter 3
 Chapter 3 Section 1 States of Matter What is matter made of? What are the three most common states of matter? How do particles behave in each state of matter? Solids, Liquids, and Gases Materials can be
Chapter 3 Section 1 States of Matter What is matter made of? What are the three most common states of matter? How do particles behave in each state of matter? Solids, Liquids, and Gases Materials can be
Chapter 8. Chapter 8. Preview. Bellringer. Chapter 8. Particles of Matter. Objectives. Chapter 8. Particles of Matter, continued
 States of Matter Preview Bellringer Section 2 Behavior of Gases In the kitchen, you might find three different forms of water. What are these three forms of water, and where exactly in the kitchen would
States of Matter Preview Bellringer Section 2 Behavior of Gases In the kitchen, you might find three different forms of water. What are these three forms of water, and where exactly in the kitchen would
Unit 1 Lesson 6 Changes of State. Copyright Houghton Mifflin Harcourt Publishing Company
 The Fact of the Matter What happens when matter changes state? The three most familiar states of matter are solid, liquid, and gas. A change of state is the change of a substance from one physical form
The Fact of the Matter What happens when matter changes state? The three most familiar states of matter are solid, liquid, and gas. A change of state is the change of a substance from one physical form
STATES OF MATTER. The Four States of Ma/er. Four States. Solid Liquid Gas Plasma
 STATES OF MATTER The Four States of Ma/er Solid Liquid Gas Plasma Four States STATES OF MATTER Ø What makes a substance a par:cular state of ma
STATES OF MATTER The Four States of Ma/er Solid Liquid Gas Plasma Four States STATES OF MATTER Ø What makes a substance a par:cular state of ma
Unit 4: Gas Laws. Matter and Phase Changes
 Unit 4: Gas Laws Matter and Phase Changes ENERGY and matter What is 에너지 A fundamental property of the universe that cannot be easily defined. Energy No one knows what energy is, only what it does or has
Unit 4: Gas Laws Matter and Phase Changes ENERGY and matter What is 에너지 A fundamental property of the universe that cannot be easily defined. Energy No one knows what energy is, only what it does or has
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 1, LESSON 1 MOLECULES MATTER MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state of a pure substance
The Next Generation Science Standards (NGSS) CHAPTER 1, LESSON 1 MOLECULES MATTER MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state of a pure substance
THE PHASES OF MATTER. Solid: holds its shape and does not flow. The molecules in a solid vibrate in place, but on average, don t move very far.
 THE QUESTIONS What are the phases of matter? What makes these phases different from each other? What is the difference between melting, freezing, boiling and condensation? How do you interpret a Temperature
THE QUESTIONS What are the phases of matter? What makes these phases different from each other? What is the difference between melting, freezing, boiling and condensation? How do you interpret a Temperature
Everything in the universe can be classified as either matter or energy. Kinetic Energy Theory: All particles of matter are in constant motion.
 Physical Science Everything in the universe can be classified as either matter or energy. Kinetic Energy Theory: All particles of matter are in constant motion. State of Matter Bose- Einstein Condensate
Physical Science Everything in the universe can be classified as either matter or energy. Kinetic Energy Theory: All particles of matter are in constant motion. State of Matter Bose- Einstein Condensate
S8P1C. STATES OF MATTER E.Q. WHAT ARE THE STATES OF MATTER? WHAT ARE THE CHARACTERISTICS OF EACH STATE OF MATTER?
 S8P1C. STATES OF MATTER E.Q. WHAT ARE THE STATES OF MATTER? WHAT ARE THE CHARACTERISTICS OF EACH STATE OF MATTER? ATOMIC MODEL PROJECT BE CREATIVE!!! MIXTURES VS. SUBSTANCES TASK DUE BY FRIDAY, NOVEMBER
S8P1C. STATES OF MATTER E.Q. WHAT ARE THE STATES OF MATTER? WHAT ARE THE CHARACTERISTICS OF EACH STATE OF MATTER? ATOMIC MODEL PROJECT BE CREATIVE!!! MIXTURES VS. SUBSTANCES TASK DUE BY FRIDAY, NOVEMBER
WARM-UP. 1. What are the four states of matter? 2. What is melting point? 3. How does water change from a liquid to a gas? 4. Define viscosity.
 WARM-UP 1. What are the four states of matter? 2. What is melting point? 3. How does water change from a liquid to a gas? 4. Define viscosity. STATES OF MATTER: WEB QUEST With your lab partner, you will
WARM-UP 1. What are the four states of matter? 2. What is melting point? 3. How does water change from a liquid to a gas? 4. Define viscosity. STATES OF MATTER: WEB QUEST With your lab partner, you will
Unit 3: States of Matter, Heat and Gas Laws
 Unit 3 - Stevens 1 Unit 3: States of Matter, Heat and Gas Laws Vocabulary: Solid Term Definition Example Liquid Gas No definite shape, but definite volume; Particles close together, but can move around
Unit 3 - Stevens 1 Unit 3: States of Matter, Heat and Gas Laws Vocabulary: Solid Term Definition Example Liquid Gas No definite shape, but definite volume; Particles close together, but can move around
Atoms and molecules are in motion and have energy
 Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Term Info Picture. Anything that has mass and takes up space; everything is made of matter.
 Characteristics, Changes, and States of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. B. Develop and use models to describe the movement of particles
Characteristics, Changes, and States of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. B. Develop and use models to describe the movement of particles
Chapter 3. States of Matter
 Chapter 3 States of Matter 1. Solid 2. Liquid 3. Gas States of Matter Two More (discuss later) Plasma Bose-Einstein condensate States of Matter Solid (definite shape and volume) Particles are tightly packed
Chapter 3 States of Matter 1. Solid 2. Liquid 3. Gas States of Matter Two More (discuss later) Plasma Bose-Einstein condensate States of Matter Solid (definite shape and volume) Particles are tightly packed
The States of Matter
 The States of Matter AZ State Standards Concept 1: Structure and Properties of Matter Understand physical, chemical, and atomic properties of matter. PO 1. Describe substances based on their physical properties.
The States of Matter AZ State Standards Concept 1: Structure and Properties of Matter Understand physical, chemical, and atomic properties of matter. PO 1. Describe substances based on their physical properties.
CIE Physics IGCSE. Topic 2: Thermal Physics
 CIE Physics IGCSE Topic 2: Thermal Physics Summary Notes Simple kinetic molecular model of matter Molecular model Solids Molecules close together in regular pattern Strong intermolecular forces of attraction
CIE Physics IGCSE Topic 2: Thermal Physics Summary Notes Simple kinetic molecular model of matter Molecular model Solids Molecules close together in regular pattern Strong intermolecular forces of attraction
Chapter 22 States of matter. Section 1 matter Section 2 Changes of State
 Chapter 22 States of matter Section 1 matter Section 2 Changes of State States of Matter is a physical property ***Matter is made of atoms Atoms form chemical bonds to make matter **** Atoms vibrate constantly
Chapter 22 States of matter Section 1 matter Section 2 Changes of State States of Matter is a physical property ***Matter is made of atoms Atoms form chemical bonds to make matter **** Atoms vibrate constantly
ENERGY IN CHEMISTRY. R. Ashby Duplication by permission only.
 CH 11 TOPIC 28 CHANGING STATES OF MATTER 1 You have mastered this topic when you can: 1) define or describe: ENERGY, POTENTIAL ENERGY, KINETIC ENERGY & KINETIC MOLECULAR THEORY 2) define or describe HEAT
CH 11 TOPIC 28 CHANGING STATES OF MATTER 1 You have mastered this topic when you can: 1) define or describe: ENERGY, POTENTIAL ENERGY, KINETIC ENERGY & KINETIC MOLECULAR THEORY 2) define or describe HEAT
Chemistry Joke. Once you ve seen 6.02 x You ve seen a mole!
 States of Matter Chemistry Joke Once you ve seen 6.02 x 10 23 atoms You ve seen a mole! Kinetic Theory Kinetic Theory explains the states of matter based on the concept that the particles in all forms
States of Matter Chemistry Joke Once you ve seen 6.02 x 10 23 atoms You ve seen a mole! Kinetic Theory Kinetic Theory explains the states of matter based on the concept that the particles in all forms
Physical Science Chapter 5 Cont3. Temperature & Heat
 Physical Science Chapter 5 Cont3 Temperature & Heat What are we going to study? Heat Transfer Phases of Matter The Kinetic Theory of Gases Thermodynamics Specific Heat (Capacity) Specific Heat Latent Heat
Physical Science Chapter 5 Cont3 Temperature & Heat What are we going to study? Heat Transfer Phases of Matter The Kinetic Theory of Gases Thermodynamics Specific Heat (Capacity) Specific Heat Latent Heat
IGCSE Double Award Extended Coordinated Science
 IGCSE Double Award Extended Coordinated Science Physics 5 - Thermal Properties of Matter Thermal Expansion You need to know thermal expansions for solids, liquids, and gases, and their applications. Thermal
IGCSE Double Award Extended Coordinated Science Physics 5 - Thermal Properties of Matter Thermal Expansion You need to know thermal expansions for solids, liquids, and gases, and their applications. Thermal
CHM Solids, Liquids, and Phase Changes (r15) Charles Taylor 1/9
 CHM 111 - Solids, Liquids, and Phase Changes (r15) - 2015 Charles Taylor 1/9 Introduction In CHM 110, we used kinetic theory to explain the behavior of gases. Now, we will discuss solids and liquids. While
CHM 111 - Solids, Liquids, and Phase Changes (r15) - 2015 Charles Taylor 1/9 Introduction In CHM 110, we used kinetic theory to explain the behavior of gases. Now, we will discuss solids and liquids. While
Name Date Class THE NATURE OF GASES
 13.1 THE NATURE OF GASES Section Review Objectives Describe the assumptions of the kinetic theory as it applies to gases Interpret gas pressure in terms of kinetic theory Define the relationship between
13.1 THE NATURE OF GASES Section Review Objectives Describe the assumptions of the kinetic theory as it applies to gases Interpret gas pressure in terms of kinetic theory Define the relationship between
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 2, LESSON 1 HEAT, TEMPERATURE, AND CONDUCTION MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state
The Next Generation Science Standards (NGSS) CHAPTER 2, LESSON 1 HEAT, TEMPERATURE, AND CONDUCTION MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state
Matter & Energy. Kinetic Theory of Matter. Kinetic Theory of Matter. Kinetic Theory of Matter. Kinetic Theory of Matter. Temperature.
 Matter & Energy 1) All matter is made up of atoms and molecules that act as tiny particles. 1 2 2) These tiny particles are always in motion. State of matter depends on its molecular motion as measured
Matter & Energy 1) All matter is made up of atoms and molecules that act as tiny particles. 1 2 2) These tiny particles are always in motion. State of matter depends on its molecular motion as measured
CHEMISTRY Matter and Change. Chapter 12: States of Matter
 CHEMISTRY Matter and Change Chapter 12: States of Matter CHAPTER 12 States of Matter Section 12.1 Section 12.2 Section 12.3 Section 12.4 Gases Forces of Attraction Liquids and Solids Phase Changes Click
CHEMISTRY Matter and Change Chapter 12: States of Matter CHAPTER 12 States of Matter Section 12.1 Section 12.2 Section 12.3 Section 12.4 Gases Forces of Attraction Liquids and Solids Phase Changes Click
How Does the Sun s Energy Cause Rain?
 1.2 Investigate 3.3 Read How Does the Sun s Energy Cause Rain? In the water-cycle simulation, you observed water change from a liquid to a gas, and then back to a liquid falling to the bottom of the container.
1.2 Investigate 3.3 Read How Does the Sun s Energy Cause Rain? In the water-cycle simulation, you observed water change from a liquid to a gas, and then back to a liquid falling to the bottom of the container.
Ch. 1 States of Matter
 Ch. 1 States of Matter Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The surface of water can act like a sort of skin due to a property of liquids called
Ch. 1 States of Matter Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The surface of water can act like a sort of skin due to a property of liquids called
Chapter 10. Lesson Starter. Why did you not smell the odor of the vapor immediately? Explain this event in terms of the motion of molecules.
 Preview Lesson Starter Objectives The Kinetic-Molecular Theory of Gases The Kinetic-Molecular Theory and the Nature of Gases Deviations of Real Gases from Ideal Behavior Section 1 The Kinetic-Molecular
Preview Lesson Starter Objectives The Kinetic-Molecular Theory of Gases The Kinetic-Molecular Theory and the Nature of Gases Deviations of Real Gases from Ideal Behavior Section 1 The Kinetic-Molecular
NAME DATE CLASS TEST DATE:
 1 TEST DATE: 2 Vocabulary Chapter 8 Solids, liquids, and gases Condensation Crystals Evaporation Heat of fusion Heat of vaporization Kinetic theory of matter Plasma States of matter Thermal expansion Chapter
1 TEST DATE: 2 Vocabulary Chapter 8 Solids, liquids, and gases Condensation Crystals Evaporation Heat of fusion Heat of vaporization Kinetic theory of matter Plasma States of matter Thermal expansion Chapter
STATES OF MATTER STATES OF MATTER. The Four States of Matter 3/5/2015. Solid. Liquid Commonly found on Gas Earth Plasma
 Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) STATES OF MATTER The Four States of Matter Solid } Liquid Commonly found on Gas Earth Plasma STATES OF MATTER Based upon particle
Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) STATES OF MATTER The Four States of Matter Solid } Liquid Commonly found on Gas Earth Plasma STATES OF MATTER Based upon particle
STATES OF MATTER STATES OF MATTER. The Four States of Matter 3/5/2015
 The Four States of Matter Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) Solid } Liquid Commonly found on Gas Earth Plasma Based upon particle arrangement Based upon energy of
The Four States of Matter Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) Solid } Liquid Commonly found on Gas Earth Plasma Based upon particle arrangement Based upon energy of
Lesson 2 Changes in State
 Lesson 2 Changes in State Student Labs and Activities Page Launch Lab 25 Content Vocabulary 26 Lesson Outline 27 MiniLab 29 Content Practice A 30 Content Practice B 31 Language Arts Support 32 School to
Lesson 2 Changes in State Student Labs and Activities Page Launch Lab 25 Content Vocabulary 26 Lesson Outline 27 MiniLab 29 Content Practice A 30 Content Practice B 31 Language Arts Support 32 School to
States of Matter. What physical changes and energy changes occur as matter goes from one state to another?
 Name States of Matter Date What physical changes and energy changes occur as matter goes from one state to another? Before You Read Before you read the chapter, think about what you know about states of
Name States of Matter Date What physical changes and energy changes occur as matter goes from one state to another? Before You Read Before you read the chapter, think about what you know about states of
STATES OF MATTER. Chapter 3
 STATES OF MATTER Chapter 3 Labs done so far for ch. 3 sections 1 and 2: 1. Distilled wood and related read of temperatures with plateaus for substances produced 2. Distilling solution X (BP/CP evaporation/condensation)
STATES OF MATTER Chapter 3 Labs done so far for ch. 3 sections 1 and 2: 1. Distilled wood and related read of temperatures with plateaus for substances produced 2. Distilling solution X (BP/CP evaporation/condensation)
Changing States of Matter By Cindy Grigg
 By Cindy Grigg 1 On Earth, almost all matter exists in just three states. Matter is usually a solid, a liquid, or a gas. Plasma, the fourth state of matter, is rare on Earth. It sometimes can be found
By Cindy Grigg 1 On Earth, almost all matter exists in just three states. Matter is usually a solid, a liquid, or a gas. Plasma, the fourth state of matter, is rare on Earth. It sometimes can be found
The lower the energy of a substance, the interaction between its atoms and molecules.
 PHYSICAL STATES OF MATTER Kinetic Molecular Theory To understand the different states in which matter can exist, we need to understand something called the Kinetic Molecular Theory of Matter. Kinetic Molecular
PHYSICAL STATES OF MATTER Kinetic Molecular Theory To understand the different states in which matter can exist, we need to understand something called the Kinetic Molecular Theory of Matter. Kinetic Molecular
Chemistry A: States of Matter Packet Name: Hour: Page!1. Chemistry A States of Matter Packet
 Chemistry A: States of Matter Packet Name: Hour: Page!1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page!2 Worksheet #1: States of Matter In this packet we will
Chemistry A: States of Matter Packet Name: Hour: Page!1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page!2 Worksheet #1: States of Matter In this packet we will
Liquids & Solids: Section 12.3
 Liquids & Solids: Section 12.3 MAIN IDEA: The particles in and have a range of motion and are not easily. Why is it more difficult to pour syrup that is stored in the refrigerator than in the cabinet?
Liquids & Solids: Section 12.3 MAIN IDEA: The particles in and have a range of motion and are not easily. Why is it more difficult to pour syrup that is stored in the refrigerator than in the cabinet?
When liquid water crystallizes it has six sides. Create a snowflake with six sides.
 When liquid water crystallizes it has six sides. Create a snowflake with six sides. Purpose: To create a supersaturated solution and observe the crystal lattice of borax snowflakes. The state of matter
When liquid water crystallizes it has six sides. Create a snowflake with six sides. Purpose: To create a supersaturated solution and observe the crystal lattice of borax snowflakes. The state of matter
21) PHASE CHANGE: a change from one state (solid or liquid or gas) to another without a change in chemical composition.
 21) PHASE CHANGE: a change from one state (solid or liquid or gas) to another without a change in chemical composition. 22) Show that you understand how phase changes occur by writing an explanation for
21) PHASE CHANGE: a change from one state (solid or liquid or gas) to another without a change in chemical composition. 22) Show that you understand how phase changes occur by writing an explanation for
Section 16.3 Phase Changes
 Section 16.3 Phase Changes Solid Liquid Gas 3 Phases of Matter Density of Matter How packed matter is (The amount of matter in a given space) Solid: Liquid: Gas: High Density Medium Density Low Density
Section 16.3 Phase Changes Solid Liquid Gas 3 Phases of Matter Density of Matter How packed matter is (The amount of matter in a given space) Solid: Liquid: Gas: High Density Medium Density Low Density
Copyright 2015 Edmentum - All rights reserved. During which of the following phase changes is there a gain in energy? I.
 Study Island Copyright 2015 Edmentum - All rights reserved. Generation Date: 03/16/2015 Generated By: Kristina Brown 1. Examine the phase-change diagram below. During which of the following phase changes
Study Island Copyright 2015 Edmentum - All rights reserved. Generation Date: 03/16/2015 Generated By: Kristina Brown 1. Examine the phase-change diagram below. During which of the following phase changes
Matter & Energy. Objectives: properties and structures of the different states of matter.
 Matter & Energy Objectives: 1. Use the kinetic theory to describe the properties and structures of the different states of matter. 2. Describe energy transfers involved in changes of state. 3. Describe
Matter & Energy Objectives: 1. Use the kinetic theory to describe the properties and structures of the different states of matter. 2. Describe energy transfers involved in changes of state. 3. Describe
Chapter: States of Matter
 Table of Contents Chapter: States of Matter Section 1: Matter Section 2: Changes of State Section 3: Behavior of Fluids 1 What is matter? Matter is anything that takes up space and has mass. Matter Matter
Table of Contents Chapter: States of Matter Section 1: Matter Section 2: Changes of State Section 3: Behavior of Fluids 1 What is matter? Matter is anything that takes up space and has mass. Matter Matter
Name Date Class STATES OF MATTER. Match the correct state of matter with each description of water by writing a letter on each line.
 10 STATES OF MATTER SECTION 10.1 THE NATURE OF GASES (pages 267 272) This section describes how the kinetic theory applies to gases. It defines gas pressure and explains how temperature is related to the
10 STATES OF MATTER SECTION 10.1 THE NATURE OF GASES (pages 267 272) This section describes how the kinetic theory applies to gases. It defines gas pressure and explains how temperature is related to the
Distillation & Pressure. States of Matter. Kinetic Theory. Phase Change H2O & D=M/V
 Kinetic Theory States of Matter Phase Change Distillation & Pressure H2O & D=M/V 100 100 100 100 100 200 200 200 200 200 300 300 300 300 300 400 400 400 400 400 500 500 500 500 500 Question 1-100 When
Kinetic Theory States of Matter Phase Change Distillation & Pressure H2O & D=M/V 100 100 100 100 100 200 200 200 200 200 300 300 300 300 300 400 400 400 400 400 500 500 500 500 500 Question 1-100 When
Quiz Review Topical Questions
 Quiz Review Topical Questions Kinetic Theory of Matter Expansion and Contraction Solids, Liquids, Gases States of Matter Phase Changes Distillation Water Properties Kinetic Theory 1. The kinetic theory
Quiz Review Topical Questions Kinetic Theory of Matter Expansion and Contraction Solids, Liquids, Gases States of Matter Phase Changes Distillation Water Properties Kinetic Theory 1. The kinetic theory
Chemistry Day 5. Friday, August 31 st Tuesday, September 4 th, 2018
 Chemistry Day 5 Friday, August 31 st Tuesday, September 4 th, 2018 Do-Now Title: BrainPOP: States of Matter 1. Write down today s FLT 2. List two examples of gases 3. List two examples of things that are
Chemistry Day 5 Friday, August 31 st Tuesday, September 4 th, 2018 Do-Now Title: BrainPOP: States of Matter 1. Write down today s FLT 2. List two examples of gases 3. List two examples of things that are
Most substances can be in three states: solid, liquid, and gas.
 States of Matter Most substances can be in three states: solid, liquid, and gas. Solid Particles Have Fixed Positions The particles in a solid are very close together and have an orderly, fixed arrangement.
States of Matter Most substances can be in three states: solid, liquid, and gas. Solid Particles Have Fixed Positions The particles in a solid are very close together and have an orderly, fixed arrangement.
* Defining Temperature * Temperature is proportional to the kinetic energy of atoms and molecules. * Temperature * Internal energy
 * Defining Temperature * We associate temperature with how hot or cold an object feels. * Our sense of touch serves as a qualitative indicator of temperature. * Energy must be either added or removed from
* Defining Temperature * We associate temperature with how hot or cold an object feels. * Our sense of touch serves as a qualitative indicator of temperature. * Energy must be either added or removed from
INTRODUCTION TO LESSON CLUSTER 7
 INTRODUCTION TO LESSON CLUSTER 7 EXPLAINING MELTING AND SOLIDIFYING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain melting and solidifying, by reference to the molecular
INTRODUCTION TO LESSON CLUSTER 7 EXPLAINING MELTING AND SOLIDIFYING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain melting and solidifying, by reference to the molecular
Science 8 Chapter 7 Section 1
 Science 8 Chapter 7 Section 1 Describing Fluids (pp. 268-277) What is a fluid? Fluid: any thing that flows; a liquid or a gas While it would seem that some solids flow (sugar, salt, etc), they are not
Science 8 Chapter 7 Section 1 Describing Fluids (pp. 268-277) What is a fluid? Fluid: any thing that flows; a liquid or a gas While it would seem that some solids flow (sugar, salt, etc), they are not
Chapter 2. States of Matter
 Chapter 2 States of Matter 2-1 Matter Matter Matter Anything that takes up space and has mass. Is air matter? Yes. It takes up space and has mass. It has atoms. All matter is made up of atoms. ( Dalton
Chapter 2 States of Matter 2-1 Matter Matter Matter Anything that takes up space and has mass. Is air matter? Yes. It takes up space and has mass. It has atoms. All matter is made up of atoms. ( Dalton
KINETIC PARTICLE THEORY
 KINETIC PARTICLE THEORY IMPORTANT DEFINITIONS: The mixing process in gases or solutions due to the random motion of particles is called Diffusion. The process by which a liquid changes into a vapour at
KINETIC PARTICLE THEORY IMPORTANT DEFINITIONS: The mixing process in gases or solutions due to the random motion of particles is called Diffusion. The process by which a liquid changes into a vapour at
Kinetic Theory of Matter notes 2012
 Kinetic Theory of Matter notes 2012 Kinetic Theory of Matter 3 parts: 1) All matter is made up of and that act as tiny 2) These tiny particles are always in. State of matter depends on its molecular motion
Kinetic Theory of Matter notes 2012 Kinetic Theory of Matter 3 parts: 1) All matter is made up of and that act as tiny 2) These tiny particles are always in. State of matter depends on its molecular motion
The OTHER TWO states of matter
 ` The OTHER TWO states of matter LIQUIDS A decrease in the average kinetic energy of gas particles causes the temperature to decrease. As it cools, the particles tend to move more slowly if they slow down
` The OTHER TWO states of matter LIQUIDS A decrease in the average kinetic energy of gas particles causes the temperature to decrease. As it cools, the particles tend to move more slowly if they slow down
Name Date Class STATES OF MATTER
 13 STATES OF MATTER Chapter Test A A. Matching Match each description in Column B with the correct term in Column A. Write the letter of the correct description on the line. Column A Column B 1. amorphous
13 STATES OF MATTER Chapter Test A A. Matching Match each description in Column B with the correct term in Column A. Write the letter of the correct description on the line. Column A Column B 1. amorphous
The Can Demonstration
 The Can Demonstration With your table, make a prediction as to what will happen to the can. Discuss why you think that the can imploded. What are some reasons? https://www.youtube.com/watch?v=xg5niowf_zw&t=28s
The Can Demonstration With your table, make a prediction as to what will happen to the can. Discuss why you think that the can imploded. What are some reasons? https://www.youtube.com/watch?v=xg5niowf_zw&t=28s
SAM Teachers Guide Phase Change Overview Learning Objectives Possible Student Pre/Misconceptions
 SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion and observing and manipulating
SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion and observing and manipulating
Chapter Preview. Improving Comprehension
 Chapter Preview Improving Comprehension Graphic Organizers are important visual tools that can help you organize information and improve your reading comprehension. The Graphic Organizer below is called
Chapter Preview Improving Comprehension Graphic Organizers are important visual tools that can help you organize information and improve your reading comprehension. The Graphic Organizer below is called
Chapter 13 - States of Matter. Section 13.1 The nature of Gases
 Chapter 13 - States of Matter Section 13.1 The nature of Gases Kinetic energy and gases Kinetic energy: the energy an object has because of its motion Kinetic theory: all matter is made if particles in
Chapter 13 - States of Matter Section 13.1 The nature of Gases Kinetic energy and gases Kinetic energy: the energy an object has because of its motion Kinetic theory: all matter is made if particles in
Chemistry A: States of Matter Packet Name: Hour: Page 1. Chemistry A States of Matter Packet
 Chemistry A: States of Matter Packet Name: Hour: Page 1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page 2 Worksheet #1: States of Matter In this packet we will
Chemistry A: States of Matter Packet Name: Hour: Page 1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page 2 Worksheet #1: States of Matter In this packet we will
PHASE CHANGE. Freezing Sublimation
 Melting Graphic Organizer Deposition PHASE CHANGE Freezing Sublimation Boiling Evaporation Condensation PHASE CHANGE Phase change happens as the temperature changes. All matter can move from one state
Melting Graphic Organizer Deposition PHASE CHANGE Freezing Sublimation Boiling Evaporation Condensation PHASE CHANGE Phase change happens as the temperature changes. All matter can move from one state
Kinetic Theory of Matter. Matter & Energy
 Kinetic Theory of Matter Matter & Energy 1 Kinetic Theory of Matter All matter is made up of atoms and molecules that act as tiny particles. 2 Kinetic Theory of Matter These tiny particles are always in
Kinetic Theory of Matter Matter & Energy 1 Kinetic Theory of Matter All matter is made up of atoms and molecules that act as tiny particles. 2 Kinetic Theory of Matter These tiny particles are always in
States of Matter. Changes in State
 CHAPTER 8 States of Matter LESSON 2 Changes in State What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
CHAPTER 8 States of Matter LESSON 2 Changes in State What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
Name: Class: Date: Figure 3-1
 Name: Class: Date: Chapter 3 test Multiple Choice Identify the choice that best completes the statement or answers the question. 1. A gas has a. a definite volume but no definite shape. b. a definite shape
Name: Class: Date: Chapter 3 test Multiple Choice Identify the choice that best completes the statement or answers the question. 1. A gas has a. a definite volume but no definite shape. b. a definite shape
Chapter 10 States of Matter
 Chapter 10 States of Matter 1 Section 10.1 The Nature of Gases Objectives: Describe the assumptions of the kinetic theory as it applies to gases. Interpret gas pressure in terms of kinetic theory. Define
Chapter 10 States of Matter 1 Section 10.1 The Nature of Gases Objectives: Describe the assumptions of the kinetic theory as it applies to gases. Interpret gas pressure in terms of kinetic theory. Define
Bell Ringer. What are the two types of mixtures? What is an element? What is a compound?
 Bell Ringer What are the two types of mixtures? What is an element? What is a compound? MATTER Solids, Liquids, & Gases States of Matter & Kinetic Molecular Theory Kinetic Molecular Theory KMT Tiny, constantly
Bell Ringer What are the two types of mixtures? What is an element? What is a compound? MATTER Solids, Liquids, & Gases States of Matter & Kinetic Molecular Theory Kinetic Molecular Theory KMT Tiny, constantly
The physical state of a substance can be changed by increasing or decreasing its temperature.
 Chemistry Lecture #63: Changes of State The physical state of a substance can be changed by increasing or decreasing its temperature. For example, a solid substance can be converted into a liquid by heating
Chemistry Lecture #63: Changes of State The physical state of a substance can be changed by increasing or decreasing its temperature. For example, a solid substance can be converted into a liquid by heating
SAM Teachers Guide Phase Change Overview Learning Objectives Possible Student Pre/Misconceptions
 SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion. Students observe and manipulate
SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion. Students observe and manipulate
Lesson 02: Physical Properties of Matter. 01 Matter
 Chemistry 11, Physical Properties, Unit 02 1 Lesson 02: Physical Properties of Matter 01 Matter Almost everything in the universe is made of matter matter has volume matter has mass matter is made up of
Chemistry 11, Physical Properties, Unit 02 1 Lesson 02: Physical Properties of Matter 01 Matter Almost everything in the universe is made of matter matter has volume matter has mass matter is made up of
CHAPTER 4 - STATES OF MATTER. Mr. Polard Physical Science Ingomar Middle School
 CHAPTER 4 - STATES OF MATTER Mr. Polard Physical Science Ingomar Middle School SECTION 1 MATTER VOCABULARY SECTION 1 Matter : anything that takes up space and has mass (pg 72, 102) Solid : Matter with
CHAPTER 4 - STATES OF MATTER Mr. Polard Physical Science Ingomar Middle School SECTION 1 MATTER VOCABULARY SECTION 1 Matter : anything that takes up space and has mass (pg 72, 102) Solid : Matter with
What Is Air Temperature?
 2.2 Read What Is Air Temperature? In Learning Set 1, you used a thermometer to measure air temperature. But what exactly was the thermometer measuring? What is different about cold air and warm air that
2.2 Read What Is Air Temperature? In Learning Set 1, you used a thermometer to measure air temperature. But what exactly was the thermometer measuring? What is different about cold air and warm air that
SOLIDS, LIQUIDS, AND GASES
 CHAPTER 2 SOLIDS, LIQUIDS, AND GASES SECTION 2 1 States of Matter (pages 56-60) This section explains how shape, volume, and the motion of particles are useful in describing solids, liquids, and gases.
CHAPTER 2 SOLIDS, LIQUIDS, AND GASES SECTION 2 1 States of Matter (pages 56-60) This section explains how shape, volume, and the motion of particles are useful in describing solids, liquids, and gases.
When liquid water crystallizes it has six sides. Create a snowflake with six sides.
 When liquid water crystallizes it has six sides. Create a snowflake with six sides. Purpose: To create a supersaturated solution and observe the crystal lattice of borax snowflakes. The state of matter
When liquid water crystallizes it has six sides. Create a snowflake with six sides. Purpose: To create a supersaturated solution and observe the crystal lattice of borax snowflakes. The state of matter
S8P All of the substances on the periodic table are classified as elements because they
 S8P1-2 1. Putting sand and salt together makes A. a compound. B. an element. C. a mixture. D. a solution. 2. All of the substances on the periodic table are classified as elements because they A. are pure
S8P1-2 1. Putting sand and salt together makes A. a compound. B. an element. C. a mixture. D. a solution. 2. All of the substances on the periodic table are classified as elements because they A. are pure
Matter. Gas. Solid Liquid. Both shape and volume are not fixed. It has a fixed shape and a fixed volume.
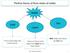 Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Chapter 7: Kinetic Molecular Theory. 7.1 States of Matter
 Chapter 7: Kinetic Molecular Theory 7.1 States of Matter 7.1 KMT and Changes in State Matter: anything with mass and volume Mass: quantity of matter that a substance or object contains (g or kg) Volume:
Chapter 7: Kinetic Molecular Theory 7.1 States of Matter 7.1 KMT and Changes in State Matter: anything with mass and volume Mass: quantity of matter that a substance or object contains (g or kg) Volume:
Name: Block: Date: Student Notes. OBJECTIVE Students will investigate the relationship between temperature and the change of the state of matter.
 Name: Block: Date: LCPS Core Experience Heat Transfer Student Notes OBJECTIVE Students will investigate the relationship between temperature and the change of the state of matter. LINK 1. Particles in
Name: Block: Date: LCPS Core Experience Heat Transfer Student Notes OBJECTIVE Students will investigate the relationship between temperature and the change of the state of matter. LINK 1. Particles in
Chemistry States of Matter Lesson 9 Lesson Plan David V. Fansler
 Chemistry States of Matter Lesson 9 Lesson Plan David V. Fansler States of Matter The Nature of Gases Objectives: Describe the motion of gas particles according to the kinetic theory; Interpret gas pressure
Chemistry States of Matter Lesson 9 Lesson Plan David V. Fansler States of Matter The Nature of Gases Objectives: Describe the motion of gas particles according to the kinetic theory; Interpret gas pressure
Solids, Liquids, and Gases
 Date Class _ Solids, Liquids, and Gases Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. _ 1. The surface of water can act like a sort of skin due to a property
Date Class _ Solids, Liquids, and Gases Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. _ 1. The surface of water can act like a sort of skin due to a property
SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES
 30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT.
 Chapter 11 THE NATURE OF GASES States of Matter Describe the motion of gas particles according to the kinetic theory Interpret gas pressure in terms of kinetic theory Key Terms: 1. kinetic energy 2. gas
Chapter 11 THE NATURE OF GASES States of Matter Describe the motion of gas particles according to the kinetic theory Interpret gas pressure in terms of kinetic theory Key Terms: 1. kinetic energy 2. gas
Calorimetry. A calorimeter is a device in which this energy transfer takes place
 Calorimetry One technique for measuring specific heat involves heating a material, adding it to a sample of water, and recording the final temperature This technique is known as calorimetry A calorimeter
Calorimetry One technique for measuring specific heat involves heating a material, adding it to a sample of water, and recording the final temperature This technique is known as calorimetry A calorimeter
Unit 13 Lesson 1 What Are Solids, Liquids, and Gases? Copyright Houghton Mifflin Harcourt Publishing Company
 Unit 13 Lesson 1 What Are Solids, Liquids, and Gases? Copyright Houghton Mifflin Harcourt Publishing Company What s the Matter? Matter has mass and volume. It cannot be created or destroyed. Mass is the
Unit 13 Lesson 1 What Are Solids, Liquids, and Gases? Copyright Houghton Mifflin Harcourt Publishing Company What s the Matter? Matter has mass and volume. It cannot be created or destroyed. Mass is the
Heating and Cooling Explained By The Particle Model. Notes: Part 2/4
 Heating and Cooling Explained By The Particle Model Notes: Part 2/4 Particles are the building blocks of all things. What are Particles? Some people call them molecules. Particles are NOT alive. How many
Heating and Cooling Explained By The Particle Model Notes: Part 2/4 Particles are the building blocks of all things. What are Particles? Some people call them molecules. Particles are NOT alive. How many
Post-Show HOT AND COLD. Gases. Liquids. Solids. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show HOT AND COLD After the Show We recently presented a Hot and Cold show at your school, and thought you and your students might like to continue investigating this topic.
Traveling Science Shows Post-Show HOT AND COLD After the Show We recently presented a Hot and Cold show at your school, and thought you and your students might like to continue investigating this topic.
