Statistical approach to protein quantification
|
|
|
- Rudolf Dawson
- 5 years ago
- Views:
Transcription
1 MCP Papers in Press. Published on November 19, 2013 as Manuscript M Statistical approach to protein quantification Authors and affilitations Sarah Gerster a,g, Taejoon Kwon b, Christina Ludwig c, Mariette Matondo c, Christine Vogel d,b, Edward Marcotte b,e, Ruedi Aebersold c,f and Peter Bühlmann a,f a Seminar for Statistics, Eidgenössische Technische Hochschule (ETH) Zurich, Rämistrasse 101, 8092 Zurich, Switzerland. b c d e f g Center for Systems and Synthetic Biology, Institute for Cellular and Molecular Biology, University of Texas at Austin, Austin, Texas 78712, United States. Institute of Molecular Systems Biology, Eidgenössische Technische Hochschule (ETH) Zurich, Wolfgang-Pauli Strasse 16, 8093 Zurich, Switzerland. Center for Genomics and Systems Biology, New York University, New York 10003, United States. Department of Chemistry and Biochemistry, University of Texas at Austin, Austin, Texas 78712, United States. Competence Center for Systems Physiology and Metabolic Diseases, 8092 Zurich, Switzerland. current address: Bioinformatics Core Facility, Swiss Institute of Bioinformatics, Quartier Sorge, Genopode, 1015 Lausanne, Switzerland. Keywords: absolute and relative protein quantification, statistical modeling, shared peptides, bipartite graph, tandem mass spectrometry Corresponding Author: Sarah Gerster, Bioinformatics Core Facility, Swiss Institute of Bioinformatics, Quartier Sorge, Genopode, 1015 Lausanne, Switzerland tel: , fax: , sarah.gerster@isb-sib.ch 1 Copyright 2013 by The American Society for Biochemistry and Molecular Biology, Inc.
2 Abbreviations AQUA peptide Synthetic tryptic peptide incorporating a stable isotope labeled amino acid and corresponding to a peptide of interest in the analyzed mixture. Allows to absolutely quantify peptides/proteins.[1] CRAN The Comprehensive R Archive Network FDR False Discovery Rate i.i.d. Independent and identically distributed (random variables) ILSE Indirect Least Squares Estimation iqr inter-quartile range LSE Least Squares Estimation MLE Maximum Likelihood Estimation MS Mass Spectrometry MS/MS Tandem mass spectrometry PSM Peptide Spectrum Match SCAMPI StatistiCAl Model for Protein quantification SRM Selected Reaction Monitoring TOPn Protein quantification approach proposed by [2]. 2
3 Summary A major goal in proteomics is the comprehensive and accurate description of a proteome. This task does not only include the identification of proteins in a sample, but also the accurate quantification of their abundances. While mass spectrometry typically provides information on peptide identity and abundance in the sample, it does not directly measure the concentration of the corresponding proteins. Specifically, most mass spectrometry based approaches e.g. shotgun proteomics or selected reaction monitoring allow to quantify peptides using chromatographic peak intensities or spectral counting information. Ultimately, based on these measurements, one wants to infer the concentrations of the corresponding proteins. Inferring properties of the proteins based on experimental peptide evidence is often a complex problem due to ambiguities of peptide assignments and different chemical properties of the peptides that affect observed concentrations. We present SCAMPI, a novel generic and statistically sound framework to compute protein abundance scores based on quantified peptides. In contrast to most previous approaches, our model explicitly includes information from shared peptides to improve protein quantitation especially in eukaryotes with many homologous sequences. The model accounts for uncertainty in the input data, leading to statistical prediction intervals for the protein scores. Furthermore, peptides with extreme abundances can be reassessed and classified as either regular data points or actual outliers. We applied the proposed model on several datasets and compared its performance to other previously used approaches for protein quantification in bottom-up mass spectrometry. 3
4 Introduction The comprehensive and quantitative analysis of proteins expressed in various organisms, tissues or cell lines provides important insights to systems biology which cannot be inferred by genomics or transcriptomics approaches [3]. Although protein identification remains an important topic of ongoing research, the focus has moved to quantification in the past years. It is not only important to know which proteins are present in a sample, but the abundance of these molecules is also of major interest. For instance, one would like to be able to identify which are the most/least abundant proteins in a sample, or to compare the concentration of the same protein in two samples taken under different biological conditions. In medical sciences, for example, biomarkers can be used to distinguish healthy from ill patients or to monitor the efficiency of a treatment by comparing a molecule s concentration before and after therapy [4]. Most methods for analyzing mass spectrometry (MS) based proteomics data rely on a sequential approach: first identification and then quantification of the peptides and proteins in a sample [5, 6]. Peptide identification is based on finding the peptide sequences corresponding to the measured spectra and has been intensely studied. A wide range of solutions have been proposed for example in [7, 8, 9, 10]. A recent review of the main approaches can be found in [11]. Inferring abundances for proteins relies on quantitative information about the corresponding peptides, usually either chromatographic peak intensities (ion-currents) or spectral counts (number of recorded MS/MS spectra). Hence, the peptides are usually quantified first, and then this knowledge is transferred to the protein level. The protein inference problem [12] consists of deciding which protein sequences are present in a sample based on the set of identified peptide sequences 1. It has been addressed in many works including [13, 10, 14, 15, 16, 17, 18, 19]. The methods vary by the underlying model, the implied 1 In the remainder of this manuscript we use peptide and peptide sequence interchangeably. The same holds for protein and protein sequence. 4
5 independence assumptions and the way of handling shared peptides (which match to several protein sequences, also called degenerate (or degenerated) peptides). Many different approaches for protein quantification have been published in the past few years. Some of them work directly on raw data (e.g. SuperHirn [20], MaxQuant [10], Progenesis [21] and OpenMS [22]), primarily to obtain quantitative information in form of intensities or spectral counts for the peptides. Other tools are exclusively designed to combine/transform peptide abundances into quantitative data at protein level (e.g. empai [23], APEX [24], msci [25], TOPn [2], MSstats [26], SI N [27] and SRMstats [28]). Further differences between the approaches arise from their use of peak intensities or spectral counts as measures for the peptide abundance, whether they specialize on absolute or on relative quantification, which mass spectrometric technique is used (discovery-driven, directed or targeted MS [29]) and which, if any, isotopic labeling of the peptides is supported. Most publications proposing a procedure based on peptide intensities actually provide an elaborate solution to quantify peptides (allowing to combine replicates, normalize the data) but rely on a very simple averaging approach to combine these scores into estimates for protein concentrations. Especially, none of the methods above including methods based on spectral counts take full advantage of the information withheld in shared peptides. Instead, the degenerate peptides are either grouped, reassigned to single proteins or even discarded to derive a simple solution to the identification and quantification of proteins. Studies focusing on the inclusion of shared peptides in the protein quantification process include [30, 31, 32, 33]. While shared peptides make the protein quantification problem more difficult and can introduce errors in the estimates when not handled properly [30], they also hold essential information. Being able to use this additional knowledge is of great importance especially when working with higher eukaryotes. In such organisms it is difficult to observe enough unique peptides per protein (peptides matching to a single protein) for quantification [32]. Furthermore, a lot of valuable information is lost by discarding or grouping shared peptides or indistinguishable proteins. 5
6 Here we present SCAMPI a statistical model for protein quantification. In contrast to most existing approaches, SCAMPI includes quantitative information from shared peptides. Furthermore, it is generic in the sense that (i) input can come from various experiments (e.g. SRM or shotgun, isotope-labeled or label-free), and (ii) method used to compute the peptide abundances is left to the user s choice. In addition, an abundance score is computed for each protein matching to at least one experimentally observed peptide (no grouping). The implemented model holds several parameters which are trained on the dataset, allowing to adapt to different types of input/instrumentation/experiments. The underlying assumptions are clearly stated and a proper statistical framework is used. In comparison to models such as [30, 31, 32], SCAMPI offers a novel approach involving a probabilistic framework and generic formulation. In contrast to previous models handling shared peptides, SCAMPI readily provides a prediction interval for each protein abundance score and allows to reassess the peptide abundances. The Results section shows applications of SCAMPI to three data sets and compares its performance to some previously used protein quantification methods, namely TOPn [2] and MaxQuant [10] which are briefly discussed below. The examples shown rely on intensity-based peptide abundances. Applications to spectral count data might be possible, but would require further investigation. TOPn [2] This approach quantifies a protein based on its identified unique peptides exhibiting the most intense mass spectrometry response (best flyer peptides). TOPn is based on the assumption that the peptide specific MS response of the best-flyer peptides is approximately constant throughout the whole proteome. The validity of this assumption has been empirically tested and demonstrated for the first time by Silva et al. [2] and applied at a proteome wide-scale in [34]. Additional information contained in shared peptides is disregarded by TOPn. The approach suggests to predict the protein concentration by averaging the peptide concentration of its n most abundant peptides, with n often set to 3. If this requirement of having three unique peptides quantified for each protein was applied strictly, TOP3 would only be able to estimate concentrations for a small 6
7 fraction of the proteins in samples from higher eukaryotes (many similar protein sequences). In practice however, proteins are often quantified even if they have only one or two unique peptides. MaxQuant [10] This method was originally designed for relative protein quantification based on peptide intensities. MaxQuant allows to align, normalize and quantify spectra over multiple peptides, and then combines these results to compute relative protein abundances. The issue of shared peptides is avoided by combining indistinguishable proteins into groups. Besides relative comparisons of protein quantities, our method can also be used for absolute quantification by using carefully selected anchor proteins (see e.g. in [34, 35, 36]). Absolute values are required to determine stoichiometries of protein complexes and to facilitate mathematical modeling, for example of cell signaling. Absolute protein quantification is also important for many questions in molecular biology and medical sciences, for example when one would like to compare results obtained on different platforms, with different settings or across various species. An additional feature provided by our model is the possibility to use the computed protein abundance scores to reassess the peptide input scores. Dost et al. [32] also briefly mention this option as an advantage of using approaches including shared peptides. As mentioned above, shared peptides hold additional information and thus allow to discover discrepancies in the input data which might be missed when focusing only on unique peptides. The model is fully described in the section Method and some of its properties are further highlighted in the Discussion. The performance is evaluated on several datasets in the Results section and compared to the results obtained with other protein quantification approaches. 7
8 Method Our proposed model is designed to estimate protein abundance scores based on experimental quantitation knowledge gained at peptide level by MS-based approaches (e.g. shotgun proteomics or SRM). The model is applied to one sample (one biological replicate) at a time. The input data consists of n identified peptide sequences and their m matching protein sequences in a given sample. Choices such as how to handle charge states, modifications or semi-tryptic peptides are left to the user (see Supporting Information 1 Workflow and input data for details). For each peptide the model requires an input score U i (i = 1,..., n) which is assumed to be proportional to its abundance. As values for U i one can for example use a (log-transformed) measurement of the total ion-current (intensity) of a peptide. The aim of the model is to infer the abundance C j for each protein matching to at least one experimentally quantified peptide (j = 1,..., m). As underlying data structure the model uses a bipartite graph, one set of nodes representing the peptides, and the other one the proteins. There is an edge between two nodes if and only if the peptide sequence is part of the protein sequence (inclusion). The graph is composed of many connected components (also called subgraphs) which are referred to as cc r (r = 1,..., R). Each connected component holds n r peptides and m r proteins. Figure 1 exemplifies the notation. The data on peptide level, U i, is known from experiments. The protein abundance, C j, is a latent variable. The main goal of the presented approach is to compute estimates Ĉj. To address this aim we design a model for U i, from which we will later infer the estimates Ĉj: U i = α + β j Ne(i) C j + ɛ i, with (1) i.i.d. C 1, C 2,..., C m N ( µ, 1 ), ɛ 1, ɛ 2,..., ɛ n i.i.d. N ( 0, τ 2), where Ne(i) denotes the set of proteins having a common edge with peptide i (neighbors of i) 8
9 and ɛ 1,..., ɛ n are independent of C 1,..., C m. The parameters α, β, µ and τ are unknown and will be estimated from the data. Briefly, we assume that U i depends linearly on the abundance of the neighboring proteins. Furthermore, we include an intercept α which allows us to take into account a possible shift in the measured values for different platforms and/or organisms. Eventually, the model contains an error term to account for measurement and modeling errors. Note that i.i.d. C j N (µ, 1) provides the prior value for each protein abundance. The posterior distributions, used for statistical inference, are different for each protein. In other words, all proteins C j are, a priori, treated equally with the same prior distribution N (µ, 1). The posterior distribution is obtained by updating this prior assumption, using the data about peptides. Prior distributions with an i.i.d. structure (as here) are widely used in Bayesian statistics (and frequentist statistics in connection with random effects models). Further note that the whole dataset is used for the training of the parameters, even if abundance predictions are only required for a small subset of the proteins. Working on a bipartite graph naturally leads to making some Markovian-type assumptions [37]: Peptides belonging to the same connected component are assumed to be independent given their matching proteins. (2) Hence, dependencies among peptides are exclusively due to their common proteins. In addition, the model assumes that only neighboring proteins matter in the (conditional) distribution for the peptides (see also [16]). For better readability we introduce the following notations: U (r) is the vector of all U i for peptides belonging to the r th connected component (n r 1 vector). 1 (r) = (1,..., 1) (for the connected component r, this is an n r 1 vector). 9
10 The n r n r matrix D (r) gives information about the connectivity in the connected component r: D (r) ii = number of proteins sharing an edge with peptide i, D (r) ik = number of proteins sharing an edge with peptide i and peptide k. Given the peptide scores, an estimate of the abundance of a protein j in connected component r [ is Ĉj = E C j U (r)]. With multivariate analysis theory [38, p ] and using the Markovian assumption (2) this can be rewritten as Ĉ j [ ] = E C j U (r) = µ + ( ) U (r) α 1 (r) β µ diag(d (r) ) Σ 1 Γ. (3) U (r) (r) C j U The variance of the protein abundance estimates given the peptide scores U i can be computed as ( ) Var C j U (r) = 1 Γ C j U (r) Σ 1 U (r) Γ C j U (r). (4) Thereby, the covariances between the peptide scores ( Σ) and between the peptide and protein scores (Γ) are defined as follows (see Supporting Information 2 Covariances for details): ( ΣU (r) ) ik ( ) Γ C j U (r) i ( = Cov U (r) i ), U (r) k ( ) = Cov C j, U (r) i = = β 2 D (r) ik for i k, β 2 D (r) ii + τ 2 for i = k. 0 if there is no edge between i and j, β if there is an edge between i and j. (5) (6) The covariances between different connected components are all zero. Note the choice of variance 1 for the distribution for the protein scores in Equation 1. Using an i.i.d. additional parameter, say C j N ( µ, σ 2), would lead to a non-identifiable model. In fact, any value set for σ can be compensated by reparametrizing β and µ. Hence, the model as presented 10
11 in Equation 1 with the four parameters α, β, µ and τ is retained. Two approaches to estimate the parameters from data will be discussed in Parameter estimation. An important advantage of a model-based approach such as SCAMPI is the possibility to estimate the accuracy of the predicted scores: Ĉ j ± z Var (Ĉj ) U (r) (7) where z = 1.96 for a 95% prediction interval. In the presence of anchor proteins, one is interested in using this additional information to transform the computed scores Ĉj into estimates of the absolute protein concentrations C. We do this with a linear transformation C j = â +ˆb Ĉj for all proteins in the sample. The parameters â and ˆb are estimated on the subset of anchor proteins by using a linear regression model: C anchor = log 10 (concentration anchor ) = a + b Ĉanchor + ɛ, where ɛ is a mean zero error term. The 95% prediction intervals for C j are then given by C j ± 1.96 ˆb Var (Ĉj ) U (r). Note that the uncertainty of the parameter estimates is ignored to compute these intervals. Furthermore, note that the anchor proteins should be chosen to cover a broad dynamic range and one should only trust predictions lying in this interval. There is no reason to assume that the fitted linear model is suitable for predicting much lower or higher abundant proteins. Peptide reassessment Consider the peptide quantities which are explicitly modeled in Equation 1. Figure 2 describes a hypothetical example of a situation where some peptide abundances show, at a first glance, surprisingly high values. If a protein quantification model can handle shared peptides, it should be able to identify which of these high peptide abundances correspond to real outliers and which ones are due to the aggregation of several proteins. If the protein abundances were known, one could compute the peptide quantities with the model and check if the predicted and measured values match. In most cases the true protein abundances are 11
12 not known, but the predicted values Ĉ can be used to compute estimates of the peptide quantities (see Equation 8). Comparing these estimates to the original values allows us to identify outliers in the peptide measurements. Of course, one should not use a peptide quantity to estimate a protein abundance, and then reuse this estimate to predict this same peptide s abundance. This would lead to over-fitting and potentially over-optimistic results. Instead, the expected value for the abundance of peptide i given all other peptide measurements ({U k i }) is computed (see Supporting Information 3 Reassessing peptide abundances for details): Û i = E [U i {U k i }] = α + β E [C j {U k i }] (8) j Ne(i) Hence, to predict the quantity of peptide i, estimates of the protein abundances (see Equation 3, but adapted to {U k i }) which are computed by using all peptide intensities except the i th one are used. The estimates Ûi can then be compared with the measured values U i. For connected components holding a single peptide, the formula simplifies to Ûi = α+β µ D ii. Note that although there are no further peptides in the connected component, U i can still be approximated thanks to the parameters trained on the whole dataset and the number of neighboring proteins to peptide i. Supporting Information 4 Selecting outliers in the measured peptides provides details about the selection of the outliers. There can be several reasons to observe outliers in the peptide data, including measurement errors, incomplete search databases, missed cleavages and modified sequences. Being able to automatically compile a list of peptide outliers may help to gain a better understanding of the data. The selected outlying peptides can then be validated individually, for example by finding reasons for their peculiar abundance scores. 12
13 Parameter estimation A classical approach to estimate the unknown parameters (α, β, µ and τ) in model (1) is to use maximum likelihood estimation (MLE). From the model in Equation 1 it follows that U (r) N nr (m (r), ΣU (r) ) with m (r) = α 1 (r) + β µ diag(d (r) ), (9) and the density function of U (r) is given by ( f(u (r) ; α, β, µ, τ) = 2π ΣU (r) 1 2 exp 1 2 (U (r) m (r) ) Σ 1 (r) (U (r) U ) m (r) ). (10) Maximizing the likelihood is equivalent to minimizing the negative log-likelihood which is given by l(α, β, µ, τ U) = R r=1 ( ) log f(u (r) ; α, β, µ, τ), (11) where the independence among different connected components, see (2), has been invoked. Minimizing the function in Equation 11 with respect to the four parameters yields estimates for α, β, µ and τ. Positivity constraints are used for β, µ and τ. While MLE is typically optimal from an asymptotic point of view (see e.g. [39]), the computations are rather expensive because of the involved numerical optimization over a four dimensional space. Sample size, i.e. the number of peptides in the dataset, is typically large (compared to the number of parameters in the model) and thus, efficiency issues of the estimator are negligible in comparison to modeling errors. Hence, as alternative to the MLE, a method of moments approach which needs much less computation time and leads to similar results as MLE on the tested datasets (see Results) is suggested. This second method relies on least squares estimations on the elements of the covariance matrix Σ. Henceforth, we refer to this parameter estimation approach as ILSE (Indirect Least Squares Estimation). In detail, several steps are needed: 13
14 1. From Equation 1 it is known that E [U i ] = α + β µ D ii. Fitting a linear regression model with the peptide intensity U as response variable and the number of neighboring proteins diag(d) as predictor variable (U diag(d)) provides the estimates ˆα (intercept) and βµ (slope). 2. Compute the sample covariance matrix estimated for each connected component cc r : Σ U (r) = (U (r) m (r) ) (U (r) m (r) ) with m (r) defined in Equation 9 (12) 3. Compare the off-diagonal elements of the sample covariance matrix with the parametric version given in Equation 5. Minimize the sum of squared errors R r=1 i k i,k cc r (( ΣU (r) ) ik D (r) ik β2 ) 2 (13) with respect to β 2. This allows us to compute the estimate ˆβ, as well as ˆµ (from the previously computed βµ (step 1)). 4. Similarly, working on the diagonal elements of Σ and plugging in ˆβ (step 3) yields the estimate ˆτ by minimizing the sum of squared errors with respect to τ 2. R n r (( ) ΣU (r) r=1 i=1 ii ˆβ 2 D (r) ii τ 2 ) 2 (14) The solutions to the minimization problems in Equations 13 and 14 can be written in closed form (see Supporting Information 5 ILSE parameter estimates). Hence, no numerical optimization is required and the estimates can be computed quickly even for large datasets. The two approaches can also be combined by using the estimates computed by ILSE as starting values for the numerical optimization performed in the MLE approach. Note that if a dataset does not hold any shared peptides, the parameters are non-identifiable. The estimates Ĉj in Equation 3 and Ûi in Equation 8 are still well-defined though. While no adjustment 14
15 is required for the MLE approach, the ILSE is not applicable directly as described above. As a workaround, ˆµ is set to zero in such situations, ˆα is then the average of all input peptide abundance scores, and ˆβ and ˆτ are computed as described in Equations 13 and 14. Relative quantification It is often of interest to compare the abundance of a protein under two conditions. For such a comparison, SCAMPI can be used to compute the protein abundance scores on each sample separately. A Quantile-Quantile plot of the protein abundance scores can be used to assess if the two score distributions are comparable (e.g. comparable median and quartiles of the two conditions). The score differences can then be assessed to find proteins undergoing particularly high changes in abundance: D j = Ĉcondition2 j Ĉcondition1 j. Ideally, one would have several biological replicates in each condition to make sure one can distinguish the effect due to the condition from the variability within the condition. Typical workflow The model has been implemented in R [40] (see Implementation) and is available in the R package protiq [41] on CRAN (Comprehensive R Archive Network). The following steps are required to run SCAMPI: 1. Prepare the input data (three input tables, see Supporting Information 1 Workflow and input data for details): (a) Data frame of quantified peptides: each row corresponds to one peptide and includes the sequence, as well as a score related to the peptide s abundance (U). Note that each peptide should occur only once in this table. It is up to the user to decide how to 15
16 aggregate the scores in case of multiple features matching to the same peptide, or, for example, whether sequences with different charge states should be combined in a single peptide or treated as separate instances. The user also has to decide how to handle modifications and semi-tryptic peptides at this stage. (b) Data frame of matching proteins: each row corresponds to one protein and includes the identifier or sequence of the protein. Note that this table should not hold the same protein sequence several times. This needs particular attention when a sequence is described by several accession numbers. (c) Data frame providing information as to which peptide matches to which protein. Each row of this table defines one edge of the bipartite graph. 2. Estimate the model parameters (either with MLE or ILSE approach). 3. Compute the protein abundance scores Ĉ (Equation 3 with estimated parameters from step 2). 4. Optionally compute peptide intensities given the estimates Ĉ (Equation 8 with estimated parameters from step 2) and compare Û to the input values U. Identify outliers. 5. Optionally re-evaluate steps 3 4 after having removed the outliers and updated the bipartite graph. The workflow is depicted in Supporting Information 1 Workflow and input data. Note that the model does not provide methods to combine measurements from e.g. different technical replicates or charge states. The user is expected to perform these adjustments prior to applying the protein quantification model, for example by running one of the peptide quantification tools mentioned in the section Introduction. As examples, decriptions of how the datasets presented in the Results section have been prepared are provided in Supporting Information 6 Input data preparation for SCAMPI. 16
17 Overview of the assumptions It is important to keep the modeling assumptions in mind: 1. Peptide abundances are modeled as random quantities, allowing to account for measurement uncertainty and modeling errors. 2. Markovian-type assumption in (2) (see also [16]) (a) Connected components are independent. (b) Peptides are independent given the matching proteins. In other words, dependencies among peptides are exclusively due to their common proteins. (c) Only neighboring proteins influence the conditional distribution for the peptides. 3. The error terms ɛ i are i.i.d and follow a normal distribution. 4. Statistical prior distribution for the protein abundances C j : they are i.i.d and follow a normal distribution. 5. ɛ 1,..., ɛ n are independent of C 1,..., C m. In practice, only assumption 3 is easily verifiable, for example by using a normal plot for residuals. However, Markovian type assumptions for graphs are often used in similar problems and allow to account for some dependence (among and between peptides and proteins) while rendering the problem computable. Regarding the normality assumption for the protein abundances, E [C j U] is linear in the U i s in the Gaussian case. In the non-gaussian case, E [C j U] might be nonlinear, but the formula from the Gaussian case still leads to the best linear approximation for the estimates of Ĉj. 17
18 Results The proposed model has been tested on several datasets and compared to the previously used protein quantification approaches TOPn [2] and MaxQuant [10]. The presented datasets include (i) mixtures with added AQUA peptides [1] to experimentally quantify a subset of the proteins (see SRM experiment on Leptospira interrogans [36] and Directed MS human data [42]) and (ii) a Human SILAC labeled proteomics experiment without known ground truth (see SILAC labeled human shotgun proteomics data). The authors of each dataset provided the required input data to run SCAMPI. The data, prepared to be used for analysis with SCAMPI, is provided in the Supporting Information (SupportingInformation12 inputdata.zip). The protein concentration scores computed by SCAMPI were compared to the ground truth (if available) and to the results obtained by quantification tools used by the authors of the data. As performance measures for the models the Pearson correlation coefficient R and Spearman s rank correlation coefficient ρ are reported. A table summarizing the size of each dataset and the computation times is provided in Supporting Information 7 Computation times. The provided examples show that SCAMPI has a similar performance as other previously proposed quantification tools. In addition, they highlight some advantages of SCAMPI compared to other existing tools. SRM experiment on Leptospira interrogans [36] In a recent paper about label-free absolute protein abundance estimation using SRM, Ludwig et al. published experimental data from cellular protein lysates of 39 Leptospira interrogans proteins. The measurements are based on SRM and the best flyer methodology. Experimental details are provided in [36] and the data is published in the corresponding supplementary information. The sample contains 39 proteins, of which 16 were used as anchor proteins and their concentration has 18
19 been accurately determined with AQUA peptides [1]. The performance of SCAMPI is compared to TOP3 on the 16 anchor proteins for the control mixture. The complete dataset holds 151 peptides uniquely matching to one of the 39 proteins and the bipartite graph holds 39 connected components. Hence, the dataset does not contain any shared peptides. Although the new model is primarily designed to solve more complicated problems with (many) shared peptides, this first test dataset allows us to give a proof of principle that the presented model also works in simpler situations. Details about the preparation of the input data for SCAMPI is provided in Supporting Information 6 Input data preparation for SCAMPI. The estimated parameter values are provided in Supporting Information 8 Parameter estimates. SCAMPI s results are compared to the output of the TOP3 [2] approach. A TOP3 abundance score was computed for each protein with at least one matching unique peptide. The estimates for the protein concentrations are shown in Figure 3. Panel A shows the results for SCAMPI when using the ILSE parameter estimates. The error bars correspond to the 95% prediction intervals for the computed protein scores. Panel B shows the outcome for the TOP3 approach. The correlation coefficients obtained by the different approaches are very similar. The results obtained with SCAMPI by using the MLE parameter estimates are very similar to the ones presented above. For the sake of completeness, the resulting plots are provided in Supporting Information 9 Additional Figures, together with some Bland-Altman [43] and some diagnostic plots. Directed MS human data [42] Beck et al. provide a quantitative analysis for the human tissue culture cell line U2OS based on directed MS experiments. Some AQUA peptides [1] were spiked into the mixture to allow the absolute quantification of 53 proteins over a wide range of concentrations. See [42] for experimental 19
20 details. Beck et al. provided the progenesis [21] peptide quantification scores for the control mixture. Details about the preparation of the input data for SCAMPI is provided in Supporting Information 6 Input data preparation for SCAMPI. The dataset holds quantification information for peptides, about 6% of which are shared by at least two different protein sequences. The graph comprises 6257 proteins and edges, and can be split into 4984 connected components. The estimated parameter values for SCAMPI are reported in Supporting Information 8 Parameter estimates. The performance is assessed on the 42 anchor proteins for which experimental data is available on the peptide level (see Supporting Information 6 Input data preparation for SCAMPI for details). The performance of SCAMPI is compared to the TOP3 approach [2] in Figure 4. A TOP3 abundance score was computed for each protein with at least one matching unique peptide. The error bars in panel A correspond to the 95% prediction intervals for the computed protein scores. The reported correlation scores are similar between the different approaches. TOP3 slightly outperforms SCAMPI both, in terms of ρ and R. However, note that although the dataset holds about 7 peptides per protein on average, a strict TOP3 approach (quantifying only proteins with at least three quantified matching unique peptides) would not be able to predict the abundance of each protein in the dataset. Hence, some of the reported protein abundances computed by TOP3 relie on the values of one or two measured peptides only. In contrast, SCAMPI uses additional knowledge from shared peptides (if available) and from the whole data set (through the estimated parameters) to provide an abundance score for each protein. Moreover, SCAMPI, unlike TOP3, also provides the prediction error for each protein. The results obtained with SCAMPI by using the MLE parameter estimates are very similar to the ones presented above. For the sake of completeness, the resulting plots are provided in Supporting Information 9 Additional Figures, together with some Bland-Altman [43] and some diagnostic 20
21 plots. Finally, the predicted concentrations for all proteins in the dataset are compared to the results published in [42]. The latter predictions are computed with SSID/MW, a method based on spectral counting (see supplementary material to [42] for more information). Comparing the SSID/MW results to the protein concentrations predicted by SCAMPI yields a Pearson correlation coefficient of 0.81 and a Spearman s rank correlation coefficient of The comparison is based on 1741 proteins which have been quantified by both models and which have values in the range covered by the anchor proteins. SILAC labeled human shotgun proteomics data This dataset comes from a human acute myeloid leukaemia cell line (KG1a cells). Cells are grown in SILAC media containing either light or heavy isotope. The heavy isotope labeled cells were treated with proteasome inhibitor. The untreated cells (control) were grown in the presence of the light isotope. Details about the experimental procedure are provided in Supporting Information 10 Materials and Methods for the SILAC data set. The MaxQuant [10] peptide quantification results for the cytoplasmic fraction are used as input to SCAMPI. The measurements for the control and treated conditions were analyzed separately. Details about the preparation of the input data for SCAMPI is provided in Supporting Information 6 Input data preparation for SCAMPI. The underlying graph used for both samples is slightly different since there are a few peptides (less than 20) which could only be quantified in one of the two samples. For the control mixture, the graph holds peptides, 3892 proteins and edges organized in 2659 connected components. In the treated case, the graph holds peptides, 3890 proteins and edges organized in 2658 connected components. In both conditions about 17% of the peptides are shared, i.e. match 21
22 to more than one protein sequence. The estimated parameter values for SCAMPI are reported in Supporting Information 8 Parameter estimates. Although there is no known ground truth for this dataset, it is an interesting example (high percentage of shared peptides) to show how SCAMPI can be used to analyze relative protein abundance. Furthermore, it emphasizes SCAMPI s flexibility regarding the type of peptide level input it can handle, in this case peak intensities computed by MaxQuant. This dataset is primarily used to show how SCAMPI can be used to identify differentially abundant proteins and to explore a dataset by reassessing peptide scores. In addition, it illustrates how running SCAMPI recursively can be used to improve the predictions. The results for parameter estimates from ILSE and MLE are again very similar. The ILSE results are discussed here. The plots for the MLE estimations are provided in Supporting Information 9 Additional Figures. Figure 5 shows the obtained protein abundance score distribution for both conditions. The Quantile- Quantile plot in Figure 5 C confirms that direct comparisons between computed scores from the two conditions are reasonable. Hence, in order to investigate whether the abundance of some proteins changed between the two conditions, the score difference D j = Ĉtreated j Ĉcontrol j is analyzed. For this comparison, only proteins which could be quantified in both conditions are used (i.e. not being measured was not treated as being absent from the sample). The empirical distribution of the abundance changes D j is shown in Figure 6. Proteins with particularly high score differences are highlighted. An interquartile-based discrimination rule is used for this selection: proteins are highlighted if D j / [Q 1 k iqr, Q 3 + k iqr], where Q 1 and Q 3 correspond to the first and third quartile in the distribution of abundance differences, respectively, and iqr = Q 3 Q 1. A conservative choice of k = 4 was used. This leads to the selection of 59 differentially expressed proteins. These findings include for example some proteins in the heat-shock protein family (HSP) which were up-regulated upon proteasome inhibition in KG1a cells. This has been previously described in other cellular models [44, 45, 46, 47]. These changes likely reflect a stress response in agreement 22
23 with the recognized role of the chaperone proteins in the protection of cells against therapeutic agents. The fact that these proteins are detected with SCAMPI serves as proof of principle that the output of the presented model can be used to address biologically relevant questions. Note that one would typically require several biological replicates of each condition to test for significantly differentially abundant proteins. The scheme used above is a simplistic approach to illustrate what kind of problems can be tackled using SCAMPI. If one has several biological replicates, SCAMPI can be run on each of them. More general testing approaches (e.g. a standard two-sample test or versions such as the moderated t-test (see e.g. [48])) should then be used to assess differential expression. A direct comparison of SCAMPI s outcome with the results from MaxQuant [10] (normalized H/L ratios) is not straightforward. MaxQuant groups proteins, and gives abundance scores to these groups. It is not clear how such a group abundance should be compared to the abundance scores for single proteins obtained in SCAMPI. However, there is an overlap between the outcome of SCAMPI and MaxQuant: 15 of the 59 differentially expressed proteins (according to SCAMPI) also figure in one of the top scoring groups identified by MaxQuant. Another aspect of SCAMPI, namely the possibility to reassess peptide abundance scores is also demonstrated on the control condition of this last dataset. Based on the estimated parameter values and the graph structure, SCAMPI can be used to estimate peptide abundance scores (Ûi). These values can then be compared to the input data (U i ). The diagnostic plots (see e.g. [49, 50]) are shown in Figure 7. Panels A (residual plot) and B (normal Quantile-Quantile plot) show no major violation of the assumptions on the noise term ɛ for the bulk of the data and can be used to select outliers (see Supporting Information 4 Selecting outliers in the measured peptides for details). The 234 selected outliers are highlighted in panel B. On one hand, this assessment allows us to gain further insight in the data on the peptide level by checking which peptides tend to have particularly large negative or positive residuals. On the present data, it can be observed that a 23
24 large percentage of the peptides with a large negative residual (over-estimated abundance) contain at least one missed cleavage. On the other hand, the assessment can also be used to prune the graph and rerun SCAMPI on this modified dataset to try to improve the protein abundance estimates. The output of such an iterative approach is provided in Supporting Information 9 Additional Figures. Finally, a major aim of SCAMPI is to accurately model highly abundant shared peptides. Figure 8 shows that this aim is reached: SCAMPI can indeed over-proportionally well explain high abundant shared peptides, and thus affirm that these measurements are correct and should not be discarded as outliers. Discussion SCAMPI is a rigorous statistical approach for protein quantification on the basis of LC-MS/MS based experiments. Our model explicitly accounts for dependencies amongst and between peptides and proteins using a Markovian type assumption for graphs. In contrast to most other protein quantification approaches, SCAMPI s modeling framework (i) propagates the uncertainty from the peptide identification to the protein level, leading to prediction intervals for proteins, (ii) can be used to reassess the peptide measurements based on the predicted protein concentration scores, (iii) can handle different types of peptide abundance input scores, and (iv) makes use of all peptides in the input data, in particular also including the shared peptides. 24
25 Model assumptions Regarding the assumption of independent and identically distributed error terms, the residual plots for the analyzed datasets did not show any major violations. The other model assumptions cannot easily be checked. However, the Markovian type assumptions for graphs are often used in similar problems and allow to account for at least some dependence (among and between peptides and proteins) while rendering the problem computationally tractable. Regarding the normality assumption for the protein abundances, if this is not fulfilled, the result presented in Equation 3 still provides the best linear approximation for the estimate of Ĉj. Using log-transformed input data For all examples presented here, the input peptide intensities have been (base-10) log transformed. Applying a log transformation makes the data more symmetric and Gaussian. When working with peptide abundance scores, it typically makes sense to apply a log transformation before using them to infer protein abundances. After the transformation, the features are spread more evenly across the intensity range and the variability becomes constant at all intensity levels. Uncertainty of computed scores An important advantage of a model-based approach such as SCAMPI is the possibility to estimate the accuracy of the predicted scores. SCAMPI readily provides the variance of the abundance scores, which allows us to compute a prediction interval for each protein. Parameter estimation The four model parameters (α, β, µ and τ) are estimated based on the whole dataset. Hence, even though protein scores (and the variances of these) are computed locally on the corresponding connected component, knowledge gained on the whole dataset contributes via these parameter estimates. In particular, a protein matching to a single quantified peptide will still benefit from the total knowledge about the dataset thanks to the global model parameters. 25
26 MLE versus ILSE parameter estimates The results show that the parameter estimates obtained with the MLE or the ILSE approach (see Parameter estimation) are often similar. Generally, when comparing the estimated protein scores with the ILSE and MLE estimated parameter sets, the differences become almost imperceptible. However, the computational bottleneck when analyzing a dataset is not in the choice of the parameter estimation method. The computationally expensive part is the preprocessing of the connected components, peptide and protein data frames, which is necessary for both parameter estimation approaches. Nevertheless, the ILSE approach seems to be more robust. MLE uses the R function dmvnorm, which works with the inverse of the covariance matrices. Depending on the choice of the starting parameters, the optimization procedure reaches a state where some of the connected components covariance matrices become singular. A workaround is to restart the optimization procedure several times with different (random) starting values until success. This showed to be a feasible solution for all datasets presented in this manuscript, however, it can increase the computation time substantially. Performance comparison to the TOP3 approach When comparing a TOP3 approach to SCAMPI, the two models actually operate on different input data. The question arises as to what would happen if SCAMPI were run on the exact same input data as TOP3. This issue is discussed in Supporting Information 11 SCAMPI on TOP3 input. Briefly, if one is willing to discard all data except what is required for a strict TOP3, and if no model is required in order to be able to reassess peptide scores, then running TOP3 is the easier and faster solution to get protein abundance estimates. If not, SCAMPI is the better approach, since it makes use of all information in the data set (including the shared peptides). Absolute quantification For datasets with anchor proteins (e.g. SRM experiment on Leptospira interrogans [36] and Directed MS human data [42]) the linear fit between the computed abundance scores and the experimentally determined concentrations for these sequences can be used to predict the absolute concentrations of other proteins in the sample. The provided prediction intervals can 26
27 be adapted accordingly. Relative quantification Samples under different conditions (e.g. control and treatment) can also be compared with SCAMPI. As a general procedure, we suggest to run SCAMPI on each replicate/condition separately. Typically, several (biological) replicates of each condition are required to test for differentially abundant proteins. Running SCAMPI iteratively SCAMPI allows us to identify outliers in the input peptide abundance scores. Removing these outliers from the dataset and re-running SCAMPI can lead to improved protein abundance estimates. An automatic iterative outlier removal, as it is available in the protiq R package, is suitable for a first analysis. It is, however, important to go back to the list of rejected peptides, and to try to understand why outliers occur. This can lead to further insight about the dataset, for example by hinting at incomplete databases or by indicating potentially modified peptides that can be further investigated to possibly gain new biological information. General conclusion In summary, SCAMPI is well-suited to address the protein quantification problem on the basis of various types of LC-MS/MS based experiments to compute absolute abundances as well as for relative quantification. In contrast to many other approaches, it provides an estimate of the abundance for each single protein having some experimental peptide evidence. Proteins are neither discarded nor grouped, but the user could perform such selection/grouping operations prior to running SCAMPI. Prediction intervals for the scores allow to get an idea about the confidence of the computed abundances. Finally, providing a method which allows for a feedback loop to reassess the quantification on the peptide level has the potential to gain new insight in LC-MS/MS datasets. SCAMPI is implemented in the R package protiq [41] which is available on CRAN. It can be used to predict protein abundances, detect true outliers in the peptide measurements and can potentially be used for designing future experiments. 27
Protein Quantitation II: Multiple Reaction Monitoring. Kelly Ruggles New York University
 Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot RPPA Immunohistochemistry
Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot RPPA Immunohistochemistry
Overview - MS Proteomics in One Slide. MS masses of peptides. MS/MS fragments of a peptide. Results! Match to sequence database
 Overview - MS Proteomics in One Slide Obtain protein Digest into peptides Acquire spectra in mass spectrometer MS masses of peptides MS/MS fragments of a peptide Results! Match to sequence database 2 But
Overview - MS Proteomics in One Slide Obtain protein Digest into peptides Acquire spectra in mass spectrometer MS masses of peptides MS/MS fragments of a peptide Results! Match to sequence database 2 But
Protein Quantitation II: Multiple Reaction Monitoring. Kelly Ruggles New York University
 Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot Immunohistochemistry
Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot Immunohistochemistry
ECE521 week 3: 23/26 January 2017
 ECE521 week 3: 23/26 January 2017 Outline Probabilistic interpretation of linear regression - Maximum likelihood estimation (MLE) - Maximum a posteriori (MAP) estimation Bias-variance trade-off Linear
ECE521 week 3: 23/26 January 2017 Outline Probabilistic interpretation of linear regression - Maximum likelihood estimation (MLE) - Maximum a posteriori (MAP) estimation Bias-variance trade-off Linear
profileanalysis Innovation with Integrity Quickly pinpointing and identifying potential biomarkers in Proteomics and Metabolomics research
 profileanalysis Quickly pinpointing and identifying potential biomarkers in Proteomics and Metabolomics research Innovation with Integrity Omics Research Biomarker Discovery Made Easy by ProfileAnalysis
profileanalysis Quickly pinpointing and identifying potential biomarkers in Proteomics and Metabolomics research Innovation with Integrity Omics Research Biomarker Discovery Made Easy by ProfileAnalysis
Comprehensive support for quantitation
 Comprehensive support for quantitation One of the major new features in the current release of Mascot is support for quantitation. This is still work in progress. Our goal is to support all of the popular
Comprehensive support for quantitation One of the major new features in the current release of Mascot is support for quantitation. This is still work in progress. Our goal is to support all of the popular
DIA-Umpire: comprehensive computational framework for data independent acquisition proteomics
 DIA-Umpire: comprehensive computational framework for data independent acquisition proteomics Chih-Chiang Tsou 1,2, Dmitry Avtonomov 2, Brett Larsen 3, Monika Tucholska 3, Hyungwon Choi 4 Anne-Claude Gingras
DIA-Umpire: comprehensive computational framework for data independent acquisition proteomics Chih-Chiang Tsou 1,2, Dmitry Avtonomov 2, Brett Larsen 3, Monika Tucholska 3, Hyungwon Choi 4 Anne-Claude Gingras
Parametric Techniques
 Parametric Techniques Jason J. Corso SUNY at Buffalo J. Corso (SUNY at Buffalo) Parametric Techniques 1 / 39 Introduction When covering Bayesian Decision Theory, we assumed the full probabilistic structure
Parametric Techniques Jason J. Corso SUNY at Buffalo J. Corso (SUNY at Buffalo) Parametric Techniques 1 / 39 Introduction When covering Bayesian Decision Theory, we assumed the full probabilistic structure
Towards the Prediction of Protein Abundance from Tandem Mass Spectrometry Data
 Towards the Prediction of Protein Abundance from Tandem Mass Spectrometry Data Anthony J Bonner Han Liu Abstract This paper addresses a central problem of Proteomics: estimating the amounts of each of
Towards the Prediction of Protein Abundance from Tandem Mass Spectrometry Data Anthony J Bonner Han Liu Abstract This paper addresses a central problem of Proteomics: estimating the amounts of each of
Designed for Accuracy. Innovation with Integrity. High resolution quantitative proteomics LC-MS
 Designed for Accuracy High resolution quantitative proteomics Innovation with Integrity LC-MS Setting New Standards in Accuracy The development of mass spectrometry based proteomics approaches has dramatically
Designed for Accuracy High resolution quantitative proteomics Innovation with Integrity LC-MS Setting New Standards in Accuracy The development of mass spectrometry based proteomics approaches has dramatically
Parametric Techniques Lecture 3
 Parametric Techniques Lecture 3 Jason Corso SUNY at Buffalo 22 January 2009 J. Corso (SUNY at Buffalo) Parametric Techniques Lecture 3 22 January 2009 1 / 39 Introduction In Lecture 2, we learned how to
Parametric Techniques Lecture 3 Jason Corso SUNY at Buffalo 22 January 2009 J. Corso (SUNY at Buffalo) Parametric Techniques Lecture 3 22 January 2009 1 / 39 Introduction In Lecture 2, we learned how to
Tandem Mass Spectrometry: Generating function, alignment and assembly
 Tandem Mass Spectrometry: Generating function, alignment and assembly With slides from Sangtae Kim and from Jones & Pevzner 2004 Determining reliability of identifications Can we use Target/Decoy to estimate
Tandem Mass Spectrometry: Generating function, alignment and assembly With slides from Sangtae Kim and from Jones & Pevzner 2004 Determining reliability of identifications Can we use Target/Decoy to estimate
TUTORIAL EXERCISES WITH ANSWERS
 TUTORIAL EXERCISES WITH ANSWERS Tutorial 1 Settings 1. What is the exact monoisotopic mass difference for peptides carrying a 13 C (and NO additional 15 N) labelled C-terminal lysine residue? a. 6.020129
TUTORIAL EXERCISES WITH ANSWERS Tutorial 1 Settings 1. What is the exact monoisotopic mass difference for peptides carrying a 13 C (and NO additional 15 N) labelled C-terminal lysine residue? a. 6.020129
Development and Evaluation of Methods for Predicting Protein Levels from Tandem Mass Spectrometry Data. Han Liu
 Development and Evaluation of Methods for Predicting Protein Levels from Tandem Mass Spectrometry Data by Han Liu A thesis submitted in conformity with the requirements for the degree of Master of Science
Development and Evaluation of Methods for Predicting Protein Levels from Tandem Mass Spectrometry Data by Han Liu A thesis submitted in conformity with the requirements for the degree of Master of Science
401 Review. 6. Power analysis for one/two-sample hypothesis tests and for correlation analysis.
 401 Review Major topics of the course 1. Univariate analysis 2. Bivariate analysis 3. Simple linear regression 4. Linear algebra 5. Multiple regression analysis Major analysis methods 1. Graphical analysis
401 Review Major topics of the course 1. Univariate analysis 2. Bivariate analysis 3. Simple linear regression 4. Linear algebra 5. Multiple regression analysis Major analysis methods 1. Graphical analysis
Outline Challenges of Massive Data Combining approaches Application: Event Detection for Astronomical Data Conclusion. Abstract
 Abstract The analysis of extremely large, complex datasets is becoming an increasingly important task in the analysis of scientific data. This trend is especially prevalent in astronomy, as large-scale
Abstract The analysis of extremely large, complex datasets is becoming an increasingly important task in the analysis of scientific data. This trend is especially prevalent in astronomy, as large-scale
Data pre-processing in liquid chromatography mass spectrometry-based proteomics
 BIOINFORMATICS ORIGINAL PAPER Vol. 21 no. 21 25, pages 454 459 doi:1.193/bioinformatics/bti66 Data and text mining Data pre-processing in liquid chromatography mass spectrometry-based proteomics Xiang
BIOINFORMATICS ORIGINAL PAPER Vol. 21 no. 21 25, pages 454 459 doi:1.193/bioinformatics/bti66 Data and text mining Data pre-processing in liquid chromatography mass spectrometry-based proteomics Xiang
Computational Methods for Mass Spectrometry Proteomics
 Computational Methods for Mass Spectrometry Proteomics Eidhammer, Ingvar ISBN-13: 9780470512975 Table of Contents Preface. Acknowledgements. 1 Protein, Proteome, and Proteomics. 1.1 Primary goals for studying
Computational Methods for Mass Spectrometry Proteomics Eidhammer, Ingvar ISBN-13: 9780470512975 Table of Contents Preface. Acknowledgements. 1 Protein, Proteome, and Proteomics. 1.1 Primary goals for studying
Classification. Chapter Introduction. 6.2 The Bayes classifier
 Chapter 6 Classification 6.1 Introduction Often encountered in applications is the situation where the response variable Y takes values in a finite set of labels. For example, the response Y could encode
Chapter 6 Classification 6.1 Introduction Often encountered in applications is the situation where the response variable Y takes values in a finite set of labels. For example, the response Y could encode
SeqAn and OpenMS Integration Workshop. Temesgen Dadi, Julianus Pfeuffer, Alexander Fillbrunn The Center for Integrative Bioinformatics (CIBI)
 SeqAn and OpenMS Integration Workshop Temesgen Dadi, Julianus Pfeuffer, Alexander Fillbrunn The Center for Integrative Bioinformatics (CIBI) Mass-spectrometry data analysis in KNIME Julianus Pfeuffer,
SeqAn and OpenMS Integration Workshop Temesgen Dadi, Julianus Pfeuffer, Alexander Fillbrunn The Center for Integrative Bioinformatics (CIBI) Mass-spectrometry data analysis in KNIME Julianus Pfeuffer,
Analysing data: regression and correlation S6 and S7
 Basic medical statistics for clinical and experimental research Analysing data: regression and correlation S6 and S7 K. Jozwiak k.jozwiak@nki.nl 2 / 49 Correlation So far we have looked at the association
Basic medical statistics for clinical and experimental research Analysing data: regression and correlation S6 and S7 K. Jozwiak k.jozwiak@nki.nl 2 / 49 Correlation So far we have looked at the association
How To Use CORREP to Estimate Multivariate Correlation and Statistical Inference Procedures
 How To Use CORREP to Estimate Multivariate Correlation and Statistical Inference Procedures Dongxiao Zhu June 13, 2018 1 Introduction OMICS data are increasingly available to biomedical researchers, and
How To Use CORREP to Estimate Multivariate Correlation and Statistical Inference Procedures Dongxiao Zhu June 13, 2018 1 Introduction OMICS data are increasingly available to biomedical researchers, and
Tutorial 1: Setting up your Skyline document
 Tutorial 1: Setting up your Skyline document Caution! For using Skyline the number formats of your computer have to be set to English (United States). Open the Control Panel Clock, Language, and Region
Tutorial 1: Setting up your Skyline document Caution! For using Skyline the number formats of your computer have to be set to English (United States). Open the Control Panel Clock, Language, and Region
STA 4273H: Statistical Machine Learning
 STA 4273H: Statistical Machine Learning Russ Salakhutdinov Department of Statistics! rsalakhu@utstat.toronto.edu! http://www.utstat.utoronto.ca/~rsalakhu/ Sidney Smith Hall, Room 6002 Lecture 7 Approximate
STA 4273H: Statistical Machine Learning Russ Salakhutdinov Department of Statistics! rsalakhu@utstat.toronto.edu! http://www.utstat.utoronto.ca/~rsalakhu/ Sidney Smith Hall, Room 6002 Lecture 7 Approximate
Logistic Regression: Regression with a Binary Dependent Variable
 Logistic Regression: Regression with a Binary Dependent Variable LEARNING OBJECTIVES Upon completing this chapter, you should be able to do the following: State the circumstances under which logistic regression
Logistic Regression: Regression with a Binary Dependent Variable LEARNING OBJECTIVES Upon completing this chapter, you should be able to do the following: State the circumstances under which logistic regression
Correlation Networks
 QuickTime decompressor and a are needed to see this picture. Correlation Networks Analysis of Biological Networks April 24, 2010 Correlation Networks - Analysis of Biological Networks 1 Review We have
QuickTime decompressor and a are needed to see this picture. Correlation Networks Analysis of Biological Networks April 24, 2010 Correlation Networks - Analysis of Biological Networks 1 Review We have
Linear Regression (9/11/13)
 STA561: Probabilistic machine learning Linear Regression (9/11/13) Lecturer: Barbara Engelhardt Scribes: Zachary Abzug, Mike Gloudemans, Zhuosheng Gu, Zhao Song 1 Why use linear regression? Figure 1: Scatter
STA561: Probabilistic machine learning Linear Regression (9/11/13) Lecturer: Barbara Engelhardt Scribes: Zachary Abzug, Mike Gloudemans, Zhuosheng Gu, Zhao Song 1 Why use linear regression? Figure 1: Scatter
Bayesian Methods for Machine Learning
 Bayesian Methods for Machine Learning CS 584: Big Data Analytics Material adapted from Radford Neal s tutorial (http://ftp.cs.utoronto.ca/pub/radford/bayes-tut.pdf), Zoubin Ghahramni (http://hunch.net/~coms-4771/zoubin_ghahramani_bayesian_learning.pdf),
Bayesian Methods for Machine Learning CS 584: Big Data Analytics Material adapted from Radford Neal s tutorial (http://ftp.cs.utoronto.ca/pub/radford/bayes-tut.pdf), Zoubin Ghahramni (http://hunch.net/~coms-4771/zoubin_ghahramani_bayesian_learning.pdf),
Towards the Prediction of Protein Abundance from Tandem Mass Spectrometry Data
 Towards the Prediction of Protein Abundance from Tandem Mass Spectrometry Data Anthony J Bonner Han Liu Abstract This paper addresses a central problem of Proteomics: estimating the amounts of each of
Towards the Prediction of Protein Abundance from Tandem Mass Spectrometry Data Anthony J Bonner Han Liu Abstract This paper addresses a central problem of Proteomics: estimating the amounts of each of
Linear Regression Models
 Linear Regression Models Model Description and Model Parameters Modelling is a central theme in these notes. The idea is to develop and continuously improve a library of predictive models for hazards,
Linear Regression Models Model Description and Model Parameters Modelling is a central theme in these notes. The idea is to develop and continuously improve a library of predictive models for hazards,
Canonical Correlation, an Approximation, and the Prediction of Protein Abundance
 Canonical Correlation, an Approximation, and the Prediction of Protein Abundance Anthony Bonner Han Liu Abstract This paper addresses a central problem of Bioinformatics and Proteomics: estimating the
Canonical Correlation, an Approximation, and the Prediction of Protein Abundance Anthony Bonner Han Liu Abstract This paper addresses a central problem of Bioinformatics and Proteomics: estimating the
Part 6: Multivariate Normal and Linear Models
 Part 6: Multivariate Normal and Linear Models 1 Multiple measurements Up until now all of our statistical models have been univariate models models for a single measurement on each member of a sample of
Part 6: Multivariate Normal and Linear Models 1 Multiple measurements Up until now all of our statistical models have been univariate models models for a single measurement on each member of a sample of
Lecture 21: Spectral Learning for Graphical Models
 10-708: Probabilistic Graphical Models 10-708, Spring 2016 Lecture 21: Spectral Learning for Graphical Models Lecturer: Eric P. Xing Scribes: Maruan Al-Shedivat, Wei-Cheng Chang, Frederick Liu 1 Motivation
10-708: Probabilistic Graphical Models 10-708, Spring 2016 Lecture 21: Spectral Learning for Graphical Models Lecturer: Eric P. Xing Scribes: Maruan Al-Shedivat, Wei-Cheng Chang, Frederick Liu 1 Motivation
Chemical Labeling Strategy for Generation of Internal Standards for Targeted Quantitative Proteomics
 Chemical Labeling Strategy for Generation of Internal Standards for Targeted Quantitative Proteomics mtraq Reagents Triplex Christie Hunter, Brian Williamson, Marjorie Minkoff AB SCIEX, USA The utility
Chemical Labeling Strategy for Generation of Internal Standards for Targeted Quantitative Proteomics mtraq Reagents Triplex Christie Hunter, Brian Williamson, Marjorie Minkoff AB SCIEX, USA The utility
A Course in Applied Econometrics Lecture 18: Missing Data. Jeff Wooldridge IRP Lectures, UW Madison, August Linear model with IVs: y i x i u i,
 A Course in Applied Econometrics Lecture 18: Missing Data Jeff Wooldridge IRP Lectures, UW Madison, August 2008 1. When Can Missing Data be Ignored? 2. Inverse Probability Weighting 3. Imputation 4. Heckman-Type
A Course in Applied Econometrics Lecture 18: Missing Data Jeff Wooldridge IRP Lectures, UW Madison, August 2008 1. When Can Missing Data be Ignored? 2. Inverse Probability Weighting 3. Imputation 4. Heckman-Type
Proteomics and Variable Selection
 Proteomics and Variable Selection p. 1/55 Proteomics and Variable Selection Alex Lewin With thanks to Paul Kirk for some graphs Department of Epidemiology and Biostatistics, School of Public Health, Imperial
Proteomics and Variable Selection p. 1/55 Proteomics and Variable Selection Alex Lewin With thanks to Paul Kirk for some graphs Department of Epidemiology and Biostatistics, School of Public Health, Imperial
Quantitative Proteomics
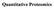 Quantitative Proteomics Quantitation AND Mass Spectrometry Condition A Condition B Identify and quantify differently expressed proteins resulting from a change in the environment (stimulus, disease) Lyse
Quantitative Proteomics Quantitation AND Mass Spectrometry Condition A Condition B Identify and quantify differently expressed proteins resulting from a change in the environment (stimulus, disease) Lyse
Introduction. Chapter 1
 Chapter 1 Introduction In this book we will be concerned with supervised learning, which is the problem of learning input-output mappings from empirical data (the training dataset). Depending on the characteristics
Chapter 1 Introduction In this book we will be concerned with supervised learning, which is the problem of learning input-output mappings from empirical data (the training dataset). Depending on the characteristics
Key questions of proteomics. Bioinformatics 2. Proteomics. Foundation of proteomics. What proteins are there? Protein digestion
 s s Key questions of proteomics What proteins are there? Bioinformatics 2 Lecture 2 roteomics How much is there of each of the proteins? - Absolute quantitation - Stoichiometry What (modification/splice)
s s Key questions of proteomics What proteins are there? Bioinformatics 2 Lecture 2 roteomics How much is there of each of the proteins? - Absolute quantitation - Stoichiometry What (modification/splice)
Bayesian linear regression
 Bayesian linear regression Linear regression is the basis of most statistical modeling. The model is Y i = X T i β + ε i, where Y i is the continuous response X i = (X i1,..., X ip ) T is the corresponding
Bayesian linear regression Linear regression is the basis of most statistical modeling. The model is Y i = X T i β + ε i, where Y i is the continuous response X i = (X i1,..., X ip ) T is the corresponding
TECHNICAL REPORT # 59 MAY Interim sample size recalculation for linear and logistic regression models: a comprehensive Monte-Carlo study
 TECHNICAL REPORT # 59 MAY 2013 Interim sample size recalculation for linear and logistic regression models: a comprehensive Monte-Carlo study Sergey Tarima, Peng He, Tao Wang, Aniko Szabo Division of Biostatistics,
TECHNICAL REPORT # 59 MAY 2013 Interim sample size recalculation for linear and logistic regression models: a comprehensive Monte-Carlo study Sergey Tarima, Peng He, Tao Wang, Aniko Szabo Division of Biostatistics,
K. Model Diagnostics. residuals ˆɛ ij = Y ij ˆµ i N = Y ij Ȳ i semi-studentized residuals ω ij = ˆɛ ij. studentized deleted residuals ɛ ij =
 K. Model Diagnostics We ve already seen how to check model assumptions prior to fitting a one-way ANOVA. Diagnostics carried out after model fitting by using residuals are more informative for assessing
K. Model Diagnostics We ve already seen how to check model assumptions prior to fitting a one-way ANOVA. Diagnostics carried out after model fitting by using residuals are more informative for assessing
PeptideProphet: Validation of Peptide Assignments to MS/MS Spectra
 PeptideProphet: Validation of Peptide Assignments to MS/MS Spectra Andrew Keller Day 2 October 17, 2006 Andrew Keller Rosetta Bioinformatics, Seattle Outline Need to validate peptide assignments to MS/MS
PeptideProphet: Validation of Peptide Assignments to MS/MS Spectra Andrew Keller Day 2 October 17, 2006 Andrew Keller Rosetta Bioinformatics, Seattle Outline Need to validate peptide assignments to MS/MS
Developing Algorithms for the Determination of Relative Abundances of Peptides from LC/MS Data
 Developing Algorithms for the Determination of Relative Abundances of Peptides from LC/MS Data RIPS Team Jake Marcus (Project Manager) Anne Eaton Melanie Kanter Aru Ray Faculty Mentors Shawn Cokus Matteo
Developing Algorithms for the Determination of Relative Abundances of Peptides from LC/MS Data RIPS Team Jake Marcus (Project Manager) Anne Eaton Melanie Kanter Aru Ray Faculty Mentors Shawn Cokus Matteo
9/12/17. Types of learning. Modeling data. Supervised learning: Classification. Supervised learning: Regression. Unsupervised learning: Clustering
 Types of learning Modeling data Supervised: we know input and targets Goal is to learn a model that, given input data, accurately predicts target data Unsupervised: we know the input only and want to make
Types of learning Modeling data Supervised: we know input and targets Goal is to learn a model that, given input data, accurately predicts target data Unsupervised: we know the input only and want to make
STAT 518 Intro Student Presentation
 STAT 518 Intro Student Presentation Wen Wei Loh April 11, 2013 Title of paper Radford M. Neal [1999] Bayesian Statistics, 6: 475-501, 1999 What the paper is about Regression and Classification Flexible
STAT 518 Intro Student Presentation Wen Wei Loh April 11, 2013 Title of paper Radford M. Neal [1999] Bayesian Statistics, 6: 475-501, 1999 What the paper is about Regression and Classification Flexible
What s New in Econometrics? Lecture 14 Quantile Methods
 What s New in Econometrics? Lecture 14 Quantile Methods Jeff Wooldridge NBER Summer Institute, 2007 1. Reminders About Means, Medians, and Quantiles 2. Some Useful Asymptotic Results 3. Quantile Regression
What s New in Econometrics? Lecture 14 Quantile Methods Jeff Wooldridge NBER Summer Institute, 2007 1. Reminders About Means, Medians, and Quantiles 2. Some Useful Asymptotic Results 3. Quantile Regression
Homework 5. Convex Optimization /36-725
 Homework 5 Convex Optimization 10-725/36-725 Due Tuesday November 22 at 5:30pm submitted to Christoph Dann in Gates 8013 (Remember to a submit separate writeup for each problem, with your name at the top)
Homework 5 Convex Optimization 10-725/36-725 Due Tuesday November 22 at 5:30pm submitted to Christoph Dann in Gates 8013 (Remember to a submit separate writeup for each problem, with your name at the top)
Supplementary material to Structure Learning of Linear Gaussian Structural Equation Models with Weak Edges
 Supplementary material to Structure Learning of Linear Gaussian Structural Equation Models with Weak Edges 1 PRELIMINARIES Two vertices X i and X j are adjacent if there is an edge between them. A path
Supplementary material to Structure Learning of Linear Gaussian Structural Equation Models with Weak Edges 1 PRELIMINARIES Two vertices X i and X j are adjacent if there is an edge between them. A path
1 Using standard errors when comparing estimated values
 MLPR Assignment Part : General comments Below are comments on some recurring issues I came across when marking the second part of the assignment, which I thought it would help to explain in more detail
MLPR Assignment Part : General comments Below are comments on some recurring issues I came across when marking the second part of the assignment, which I thought it would help to explain in more detail
Empirical Bayes Moderation of Asymptotically Linear Parameters
 Empirical Bayes Moderation of Asymptotically Linear Parameters Nima Hejazi Division of Biostatistics University of California, Berkeley stat.berkeley.edu/~nhejazi nimahejazi.org twitter/@nshejazi github/nhejazi
Empirical Bayes Moderation of Asymptotically Linear Parameters Nima Hejazi Division of Biostatistics University of California, Berkeley stat.berkeley.edu/~nhejazi nimahejazi.org twitter/@nshejazi github/nhejazi
Regression, Ridge Regression, Lasso
 Regression, Ridge Regression, Lasso Fabio G. Cozman - fgcozman@usp.br October 2, 2018 A general definition Regression studies the relationship between a response variable Y and covariates X 1,..., X n.
Regression, Ridge Regression, Lasso Fabio G. Cozman - fgcozman@usp.br October 2, 2018 A general definition Regression studies the relationship between a response variable Y and covariates X 1,..., X n.
Model-based assignment and inference of protein backbone nuclear magnetic resonances.
 Model-based assignment and inference of protein backbone nuclear magnetic resonances. Olga Vitek Jan Vitek Bruce Craig Chris Bailey-Kellogg Abstract Nuclear Magnetic Resonance (NMR) spectroscopy is a key
Model-based assignment and inference of protein backbone nuclear magnetic resonances. Olga Vitek Jan Vitek Bruce Craig Chris Bailey-Kellogg Abstract Nuclear Magnetic Resonance (NMR) spectroscopy is a key
Algorithm-Independent Learning Issues
 Algorithm-Independent Learning Issues Selim Aksoy Department of Computer Engineering Bilkent University saksoy@cs.bilkent.edu.tr CS 551, Spring 2007 c 2007, Selim Aksoy Introduction We have seen many learning
Algorithm-Independent Learning Issues Selim Aksoy Department of Computer Engineering Bilkent University saksoy@cs.bilkent.edu.tr CS 551, Spring 2007 c 2007, Selim Aksoy Introduction We have seen many learning
Analysis of Labeled and Non-Labeled Proteomic Data Using Progenesis QI for Proteomics
 Analysis of Labeled and Non-Labeled Proteomic Data Using Progenesis QI for Proteomics Lee Gethings, Gushinder Atwal, Martin Palmer, Chris Hughes, Hans Vissers, and James Langridge Waters Corporation, Wilmslow,
Analysis of Labeled and Non-Labeled Proteomic Data Using Progenesis QI for Proteomics Lee Gethings, Gushinder Atwal, Martin Palmer, Chris Hughes, Hans Vissers, and James Langridge Waters Corporation, Wilmslow,
Sparse Linear Models (10/7/13)
 STA56: Probabilistic machine learning Sparse Linear Models (0/7/) Lecturer: Barbara Engelhardt Scribes: Jiaji Huang, Xin Jiang, Albert Oh Sparsity Sparsity has been a hot topic in statistics and machine
STA56: Probabilistic machine learning Sparse Linear Models (0/7/) Lecturer: Barbara Engelhardt Scribes: Jiaji Huang, Xin Jiang, Albert Oh Sparsity Sparsity has been a hot topic in statistics and machine
Probabilistic Time Series Classification
 Probabilistic Time Series Classification Y. Cem Sübakan Boğaziçi University 25.06.2013 Y. Cem Sübakan (Boğaziçi University) M.Sc. Thesis Defense 25.06.2013 1 / 54 Problem Statement The goal is to assign
Probabilistic Time Series Classification Y. Cem Sübakan Boğaziçi University 25.06.2013 Y. Cem Sübakan (Boğaziçi University) M.Sc. Thesis Defense 25.06.2013 1 / 54 Problem Statement The goal is to assign
Using Mplus individual residual plots for. diagnostics and model evaluation in SEM
 Using Mplus individual residual plots for diagnostics and model evaluation in SEM Tihomir Asparouhov and Bengt Muthén Mplus Web Notes: No. 20 October 31, 2017 1 Introduction A variety of plots are available
Using Mplus individual residual plots for diagnostics and model evaluation in SEM Tihomir Asparouhov and Bengt Muthén Mplus Web Notes: No. 20 October 31, 2017 1 Introduction A variety of plots are available
Machine Learning Linear Classification. Prof. Matteo Matteucci
 Machine Learning Linear Classification Prof. Matteo Matteucci Recall from the first lecture 2 X R p Regression Y R Continuous Output X R p Y {Ω 0, Ω 1,, Ω K } Classification Discrete Output X R p Y (X)
Machine Learning Linear Classification Prof. Matteo Matteucci Recall from the first lecture 2 X R p Regression Y R Continuous Output X R p Y {Ω 0, Ω 1,, Ω K } Classification Discrete Output X R p Y (X)
Lecture 5: LDA and Logistic Regression
 Lecture 5: and Logistic Regression Hao Helen Zhang Hao Helen Zhang Lecture 5: and Logistic Regression 1 / 39 Outline Linear Classification Methods Two Popular Linear Models for Classification Linear Discriminant
Lecture 5: and Logistic Regression Hao Helen Zhang Hao Helen Zhang Lecture 5: and Logistic Regression 1 / 39 Outline Linear Classification Methods Two Popular Linear Models for Classification Linear Discriminant
Model Accuracy Measures
 Model Accuracy Measures Master in Bioinformatics UPF 2017-2018 Eduardo Eyras Computational Genomics Pompeu Fabra University - ICREA Barcelona, Spain Variables What we can measure (attributes) Hypotheses
Model Accuracy Measures Master in Bioinformatics UPF 2017-2018 Eduardo Eyras Computational Genomics Pompeu Fabra University - ICREA Barcelona, Spain Variables What we can measure (attributes) Hypotheses
iprophet: Multi-level integrative analysis of shotgun proteomic data improves peptide and protein identification rates and error estimates
 MCP Papers in Press. Published on August 29, 2011 as Manuscript M111.007690 This is the Pre-Published Version iprophet: Multi-level integrative analysis of shotgun proteomic data improves peptide and protein
MCP Papers in Press. Published on August 29, 2011 as Manuscript M111.007690 This is the Pre-Published Version iprophet: Multi-level integrative analysis of shotgun proteomic data improves peptide and protein
Fundamentals to Biostatistics. Prof. Chandan Chakraborty Associate Professor School of Medical Science & Technology IIT Kharagpur
 Fundamentals to Biostatistics Prof. Chandan Chakraborty Associate Professor School of Medical Science & Technology IIT Kharagpur Statistics collection, analysis, interpretation of data development of new
Fundamentals to Biostatistics Prof. Chandan Chakraborty Associate Professor School of Medical Science & Technology IIT Kharagpur Statistics collection, analysis, interpretation of data development of new
Nature Methods: doi: /nmeth Supplementary Figure 1. Fragment indexing allows efficient spectra similarity comparisons.
 Supplementary Figure 1 Fragment indexing allows efficient spectra similarity comparisons. The cost and efficiency of spectra similarity calculations can be approximated by the number of fragment comparisons
Supplementary Figure 1 Fragment indexing allows efficient spectra similarity comparisons. The cost and efficiency of spectra similarity calculations can be approximated by the number of fragment comparisons
6 Pattern Mixture Models
 6 Pattern Mixture Models A common theme underlying the methods we have discussed so far is that interest focuses on making inference on parameters in a parametric or semiparametric model for the full data
6 Pattern Mixture Models A common theme underlying the methods we have discussed so far is that interest focuses on making inference on parameters in a parametric or semiparametric model for the full data
SRM assay generation and data analysis in Skyline
 in Skyline Preparation 1. Download the example data from www.srmcourse.ch/eupa.html (3 raw files, 1 csv file, 1 sptxt file). 2. The number formats of your computer have to be set to English (United States).
in Skyline Preparation 1. Download the example data from www.srmcourse.ch/eupa.html (3 raw files, 1 csv file, 1 sptxt file). 2. The number formats of your computer have to be set to English (United States).
Performance Comparison of K-Means and Expectation Maximization with Gaussian Mixture Models for Clustering EE6540 Final Project
 Performance Comparison of K-Means and Expectation Maximization with Gaussian Mixture Models for Clustering EE6540 Final Project Devin Cornell & Sushruth Sastry May 2015 1 Abstract In this article, we explore
Performance Comparison of K-Means and Expectation Maximization with Gaussian Mixture Models for Clustering EE6540 Final Project Devin Cornell & Sushruth Sastry May 2015 1 Abstract In this article, we explore
Measures of hydroxymethylation
 Measures of hydroxymethylation Alla Slynko Axel Benner July 22, 2018 arxiv:1708.04819v2 [q-bio.qm] 17 Aug 2017 Abstract Hydroxymethylcytosine (5hmC) methylation is well-known epigenetic mark impacting
Measures of hydroxymethylation Alla Slynko Axel Benner July 22, 2018 arxiv:1708.04819v2 [q-bio.qm] 17 Aug 2017 Abstract Hydroxymethylcytosine (5hmC) methylation is well-known epigenetic mark impacting
HOWTO, example workflow and data files. (Version )
 HOWTO, example workflow and data files. (Version 20 09 2017) 1 Introduction: SugarQb is a collection of software tools (Nodes) which enable the automated identification of intact glycopeptides from HCD
HOWTO, example workflow and data files. (Version 20 09 2017) 1 Introduction: SugarQb is a collection of software tools (Nodes) which enable the automated identification of intact glycopeptides from HCD
Differentially Private Linear Regression
 Differentially Private Linear Regression Christian Baehr August 5, 2017 Your abstract. Abstract 1 Introduction My research involved testing and implementing tools into the Harvard Privacy Tools Project
Differentially Private Linear Regression Christian Baehr August 5, 2017 Your abstract. Abstract 1 Introduction My research involved testing and implementing tools into the Harvard Privacy Tools Project
Machine Learning Linear Regression. Prof. Matteo Matteucci
 Machine Learning Linear Regression Prof. Matteo Matteucci Outline 2 o Simple Linear Regression Model Least Squares Fit Measures of Fit Inference in Regression o Multi Variate Regession Model Least Squares
Machine Learning Linear Regression Prof. Matteo Matteucci Outline 2 o Simple Linear Regression Model Least Squares Fit Measures of Fit Inference in Regression o Multi Variate Regession Model Least Squares
Sound Recognition in Mixtures
 Sound Recognition in Mixtures Juhan Nam, Gautham J. Mysore 2, and Paris Smaragdis 2,3 Center for Computer Research in Music and Acoustics, Stanford University, 2 Advanced Technology Labs, Adobe Systems
Sound Recognition in Mixtures Juhan Nam, Gautham J. Mysore 2, and Paris Smaragdis 2,3 Center for Computer Research in Music and Acoustics, Stanford University, 2 Advanced Technology Labs, Adobe Systems
TIME SERIES ANALYSIS. Forecasting and Control. Wiley. Fifth Edition GWILYM M. JENKINS GEORGE E. P. BOX GREGORY C. REINSEL GRETA M.
 TIME SERIES ANALYSIS Forecasting and Control Fifth Edition GEORGE E. P. BOX GWILYM M. JENKINS GREGORY C. REINSEL GRETA M. LJUNG Wiley CONTENTS PREFACE TO THE FIFTH EDITION PREFACE TO THE FOURTH EDITION
TIME SERIES ANALYSIS Forecasting and Control Fifth Edition GEORGE E. P. BOX GWILYM M. JENKINS GREGORY C. REINSEL GRETA M. LJUNG Wiley CONTENTS PREFACE TO THE FIFTH EDITION PREFACE TO THE FOURTH EDITION
Classification and Regression Trees
 Classification and Regression Trees Ryan P Adams So far, we have primarily examined linear classifiers and regressors, and considered several different ways to train them When we ve found the linearity
Classification and Regression Trees Ryan P Adams So far, we have primarily examined linear classifiers and regressors, and considered several different ways to train them When we ve found the linearity
hsnim: Hyper Scalable Network Inference Machine for Scale-Free Protein-Protein Interaction Networks Inference
 CS 229 Project Report (TR# MSB2010) Submitted 12/10/2010 hsnim: Hyper Scalable Network Inference Machine for Scale-Free Protein-Protein Interaction Networks Inference Muhammad Shoaib Sehgal Computer Science
CS 229 Project Report (TR# MSB2010) Submitted 12/10/2010 hsnim: Hyper Scalable Network Inference Machine for Scale-Free Protein-Protein Interaction Networks Inference Muhammad Shoaib Sehgal Computer Science
Mass Spectrometry and Proteomics - Lecture 5 - Matthias Trost Newcastle University
 Mass Spectrometry and Proteomics - Lecture 5 - Matthias Trost Newcastle University matthias.trost@ncl.ac.uk Previously Proteomics Sample prep 144 Lecture 5 Quantitation techniques Search Algorithms Proteomics
Mass Spectrometry and Proteomics - Lecture 5 - Matthias Trost Newcastle University matthias.trost@ncl.ac.uk Previously Proteomics Sample prep 144 Lecture 5 Quantitation techniques Search Algorithms Proteomics
Sequential Procedure for Testing Hypothesis about Mean of Latent Gaussian Process
 Applied Mathematical Sciences, Vol. 4, 2010, no. 62, 3083-3093 Sequential Procedure for Testing Hypothesis about Mean of Latent Gaussian Process Julia Bondarenko Helmut-Schmidt University Hamburg University
Applied Mathematical Sciences, Vol. 4, 2010, no. 62, 3083-3093 Sequential Procedure for Testing Hypothesis about Mean of Latent Gaussian Process Julia Bondarenko Helmut-Schmidt University Hamburg University
Workflow concept. Data goes through the workflow. A Node contains an operation An edge represents data flow The results are brought together in tables
 PROTEOME DISCOVERER Workflow concept Data goes through the workflow Spectra Peptides Quantitation A Node contains an operation An edge represents data flow The results are brought together in tables Protein
PROTEOME DISCOVERER Workflow concept Data goes through the workflow Spectra Peptides Quantitation A Node contains an operation An edge represents data flow The results are brought together in tables Protein
SP Experimental Designs - Theoretical Background and Case Study
 SP Experimental Designs - Theoretical Background and Case Study Basil Schmid IVT ETH Zurich Measurement and Modeling FS2016 Outline 1. Introduction 2. Orthogonal and fractional factorial designs 3. Efficient
SP Experimental Designs - Theoretical Background and Case Study Basil Schmid IVT ETH Zurich Measurement and Modeling FS2016 Outline 1. Introduction 2. Orthogonal and fractional factorial designs 3. Efficient
Modeling Mass Spectrometry-Based Protein Analysis
 Chapter 8 Jan Eriksson and David Fenyö Abstract The success of mass spectrometry based proteomics depends on efficient methods for data analysis. These methods require a detailed understanding of the information
Chapter 8 Jan Eriksson and David Fenyö Abstract The success of mass spectrometry based proteomics depends on efficient methods for data analysis. These methods require a detailed understanding of the information
Massive Event Detection. Abstract
 Abstract The detection and analysis of events within massive collections of time-series has become an extremely important task for timedomain astronomy. In particular, many scientific investigations (e.g.
Abstract The detection and analysis of events within massive collections of time-series has become an extremely important task for timedomain astronomy. In particular, many scientific investigations (e.g.
Site-specific Identification of Lysine Acetylation Stoichiometries in Mammalian Cells
 Supplementary Information Site-specific Identification of Lysine Acetylation Stoichiometries in Mammalian Cells Tong Zhou 1, 2, Ying-hua Chung 1, 2, Jianji Chen 1, Yue Chen 1 1. Department of Biochemistry,
Supplementary Information Site-specific Identification of Lysine Acetylation Stoichiometries in Mammalian Cells Tong Zhou 1, 2, Ying-hua Chung 1, 2, Jianji Chen 1, Yue Chen 1 1. Department of Biochemistry,
Doing Right By Massive Data: How To Bring Probability Modeling To The Analysis Of Huge Datasets Without Taking Over The Datacenter
 Doing Right By Massive Data: How To Bring Probability Modeling To The Analysis Of Huge Datasets Without Taking Over The Datacenter Alexander W Blocker Pavlos Protopapas Xiao-Li Meng 9 February, 2010 Outline
Doing Right By Massive Data: How To Bring Probability Modeling To The Analysis Of Huge Datasets Without Taking Over The Datacenter Alexander W Blocker Pavlos Protopapas Xiao-Li Meng 9 February, 2010 Outline
1 Motivation for Instrumental Variable (IV) Regression
 ECON 370: IV & 2SLS 1 Instrumental Variables Estimation and Two Stage Least Squares Econometric Methods, ECON 370 Let s get back to the thiking in terms of cross sectional (or pooled cross sectional) data
ECON 370: IV & 2SLS 1 Instrumental Variables Estimation and Two Stage Least Squares Econometric Methods, ECON 370 Let s get back to the thiking in terms of cross sectional (or pooled cross sectional) data
Gaussian processes. Chuong B. Do (updated by Honglak Lee) November 22, 2008
 Gaussian processes Chuong B Do (updated by Honglak Lee) November 22, 2008 Many of the classical machine learning algorithms that we talked about during the first half of this course fit the following pattern:
Gaussian processes Chuong B Do (updated by Honglak Lee) November 22, 2008 Many of the classical machine learning algorithms that we talked about during the first half of this course fit the following pattern:
Ways to make neural networks generalize better
 Ways to make neural networks generalize better Seminar in Deep Learning University of Tartu 04 / 10 / 2014 Pihel Saatmann Topics Overview of ways to improve generalization Limiting the size of the weights
Ways to make neural networks generalize better Seminar in Deep Learning University of Tartu 04 / 10 / 2014 Pihel Saatmann Topics Overview of ways to improve generalization Limiting the size of the weights
STA 414/2104: Lecture 8
 STA 414/2104: Lecture 8 6-7 March 2017: Continuous Latent Variable Models, Neural networks With thanks to Russ Salakhutdinov, Jimmy Ba and others Outline Continuous latent variable models Background PCA
STA 414/2104: Lecture 8 6-7 March 2017: Continuous Latent Variable Models, Neural networks With thanks to Russ Salakhutdinov, Jimmy Ba and others Outline Continuous latent variable models Background PCA
Open Problems in Mixed Models
 xxiii Determining how to deal with a not positive definite covariance matrix of random effects, D during maximum likelihood estimation algorithms. Several strategies are discussed in Section 2.15. For
xxiii Determining how to deal with a not positive definite covariance matrix of random effects, D during maximum likelihood estimation algorithms. Several strategies are discussed in Section 2.15. For
CMPS 6630: Introduction to Computational Biology and Bioinformatics. Structure Comparison
 CMPS 6630: Introduction to Computational Biology and Bioinformatics Structure Comparison Protein Structure Comparison Motivation Understand sequence and structure variability Understand Domain architecture
CMPS 6630: Introduction to Computational Biology and Bioinformatics Structure Comparison Protein Structure Comparison Motivation Understand sequence and structure variability Understand Domain architecture
Glossary. The ISI glossary of statistical terms provides definitions in a number of different languages:
 Glossary The ISI glossary of statistical terms provides definitions in a number of different languages: http://isi.cbs.nl/glossary/index.htm Adjusted r 2 Adjusted R squared measures the proportion of the
Glossary The ISI glossary of statistical terms provides definitions in a number of different languages: http://isi.cbs.nl/glossary/index.htm Adjusted r 2 Adjusted R squared measures the proportion of the
Efficient Likelihood-Free Inference
 Efficient Likelihood-Free Inference Michael Gutmann http://homepages.inf.ed.ac.uk/mgutmann Institute for Adaptive and Neural Computation School of Informatics, University of Edinburgh 8th November 2017
Efficient Likelihood-Free Inference Michael Gutmann http://homepages.inf.ed.ac.uk/mgutmann Institute for Adaptive and Neural Computation School of Informatics, University of Edinburgh 8th November 2017
POLI 8501 Introduction to Maximum Likelihood Estimation
 POLI 8501 Introduction to Maximum Likelihood Estimation Maximum Likelihood Intuition Consider a model that looks like this: Y i N(µ, σ 2 ) So: E(Y ) = µ V ar(y ) = σ 2 Suppose you have some data on Y,
POLI 8501 Introduction to Maximum Likelihood Estimation Maximum Likelihood Intuition Consider a model that looks like this: Y i N(µ, σ 2 ) So: E(Y ) = µ V ar(y ) = σ 2 Suppose you have some data on Y,
When Should We Use Linear Fixed Effects Regression Models for Causal Inference with Panel Data?
 When Should We Use Linear Fixed Effects Regression Models for Causal Inference with Panel Data? Kosuke Imai Department of Politics Center for Statistics and Machine Learning Princeton University Joint
When Should We Use Linear Fixed Effects Regression Models for Causal Inference with Panel Data? Kosuke Imai Department of Politics Center for Statistics and Machine Learning Princeton University Joint
Experimental Design and Data Analysis for Biologists
 Experimental Design and Data Analysis for Biologists Gerry P. Quinn Monash University Michael J. Keough University of Melbourne CAMBRIDGE UNIVERSITY PRESS Contents Preface page xv I I Introduction 1 1.1
Experimental Design and Data Analysis for Biologists Gerry P. Quinn Monash University Michael J. Keough University of Melbourne CAMBRIDGE UNIVERSITY PRESS Contents Preface page xv I I Introduction 1 1.1
Tutorial on Approximate Bayesian Computation
 Tutorial on Approximate Bayesian Computation Michael Gutmann https://sites.google.com/site/michaelgutmann University of Helsinki Aalto University Helsinki Institute for Information Technology 16 May 2016
Tutorial on Approximate Bayesian Computation Michael Gutmann https://sites.google.com/site/michaelgutmann University of Helsinki Aalto University Helsinki Institute for Information Technology 16 May 2016
Lecture 6: Discrete Choice: Qualitative Response
 Lecture 6: Instructor: Department of Economics Stanford University 2011 Types of Discrete Choice Models Univariate Models Binary: Linear; Probit; Logit; Arctan, etc. Multinomial: Logit; Nested Logit; GEV;
Lecture 6: Instructor: Department of Economics Stanford University 2011 Types of Discrete Choice Models Univariate Models Binary: Linear; Probit; Logit; Arctan, etc. Multinomial: Logit; Nested Logit; GEV;
Linear Models 1. Isfahan University of Technology Fall Semester, 2014
 Linear Models 1 Isfahan University of Technology Fall Semester, 2014 References: [1] G. A. F., Seber and A. J. Lee (2003). Linear Regression Analysis (2nd ed.). Hoboken, NJ: Wiley. [2] A. C. Rencher and
Linear Models 1 Isfahan University of Technology Fall Semester, 2014 References: [1] G. A. F., Seber and A. J. Lee (2003). Linear Regression Analysis (2nd ed.). Hoboken, NJ: Wiley. [2] A. C. Rencher and
Empirical Bayes Moderation of Asymptotically Linear Parameters
 Empirical Bayes Moderation of Asymptotically Linear Parameters Nima Hejazi Division of Biostatistics University of California, Berkeley stat.berkeley.edu/~nhejazi nimahejazi.org twitter/@nshejazi github/nhejazi
Empirical Bayes Moderation of Asymptotically Linear Parameters Nima Hejazi Division of Biostatistics University of California, Berkeley stat.berkeley.edu/~nhejazi nimahejazi.org twitter/@nshejazi github/nhejazi
University of Cambridge. MPhil in Computer Speech Text & Internet Technology. Module: Speech Processing II. Lecture 2: Hidden Markov Models I
 University of Cambridge MPhil in Computer Speech Text & Internet Technology Module: Speech Processing II Lecture 2: Hidden Markov Models I o o o o o 1 2 3 4 T 1 b 2 () a 12 2 a 3 a 4 5 34 a 23 b () b ()
University of Cambridge MPhil in Computer Speech Text & Internet Technology Module: Speech Processing II Lecture 2: Hidden Markov Models I o o o o o 1 2 3 4 T 1 b 2 () a 12 2 a 3 a 4 5 34 a 23 b () b ()
STA 4273H: Statistical Machine Learning
 STA 4273H: Statistical Machine Learning Russ Salakhutdinov Department of Computer Science! Department of Statistical Sciences! rsalakhu@cs.toronto.edu! h0p://www.cs.utoronto.ca/~rsalakhu/ Lecture 7 Approximate
STA 4273H: Statistical Machine Learning Russ Salakhutdinov Department of Computer Science! Department of Statistical Sciences! rsalakhu@cs.toronto.edu! h0p://www.cs.utoronto.ca/~rsalakhu/ Lecture 7 Approximate
