823 4(4"5) # 67- 8* +!"!C ABSTRACT:
|
|
|
- Joel McDaniel
- 5 years ago
- Views:
Transcription
1 %&'() * +$!#!" 1 2 " $ %!. /-0#)$,-,'( 823 4(4"5) # 67- =' ; - 3!9 :; 9.-<+#6 "$@ A /., 43 BA :$>? - ABSTRACT: 8* +!"!C -D3 D=' Expression of emotional experience that is illustrated in an inspired image is a literary work, thus it is difficult to frame the literary work in accurate manner or put it in ready-made frames. Poetic experience hasn't relied only on old heritage as it borrowed new images and experiences that contributed to the literary structure. These challenges and questions about the literary image, accompanied with traditional vision, controversial term, and contribution of veteran and modern critics, do not put themselves out of accountability and the addendum and guidance, which words of human are exposed to. $@!3!"C$- 'F@.$)-* E I ;J$!-? 4 $G->E H 5:? 1 / 9 : 5$= 4 4 : $A 85KCL $@ C;!"$ "F0#)-- E :,->,' ' -9 "?6+# 8@ (#CL $45K9 "6$I ; J> N,-,,H" D D? L 3,M "!" 6 QPO 8&G 5A 63
2 I!""A ",>,-#>!" $"# =A!<+#!H5$? C3"=A <+# 35! ;&5>$"<+#>? %B4! -0 $,' / BA $4 - )" ) >" L ' 6 8H5'<+#9 J N $)" H!- T $GS -,NH, T $L?EH': QVO 8->U4 3'- +-I 4,,: -? #-@(' " +#!5) " 84 #4 "!R9 E<+40#)4A $3 :!!>" W?6! 3#9 $3 'E?4A 85XC4 A +.:;#! >? --.M -? 6$3 ='? 6<+#9 # Y# S - = Z 6>$3 43! 8'3 '6A - )3 ; "# 1 --.K9 A 1 $ 3 5T49 8.K<+4=A! B$4GMG"#9 3 > $ $%&'( 9 " [H' : J3\ =]' C3 =' H,2,43C3.!J3 *#@8@ :7) #+%RJN43? :,-C3+#"+6! H 5:? 6! Q_O 8 43?6 `56#$!%@A!-.?J3 [H' : 8C3#845M?6?C3 64
3 ::6 $-9 ' >(('9 Z $1 =' ^3 ^B A -!""$)-NH1 >N <+4=' QaO 8!L -'9 356$1 4'=' #> 4C9 C3%<+5 +3!"#3 9 ',5 - ) ) & J QbO 8@ -T ":$#+&5E6 $4 " 7,2,2NH4cccd\e3 cbca ffce+fgw.? dhy) "i!" 672% 2NH -[<2 7@H" cchjc* k+!e+l 74mee? Y@cH"c.? d QnO '7G0 ( -@2 7A " " G0?- +"# !" C3 B7" 2! "7# 8J J' 4 H > 4' 0! +# " 7&5 4 84" -;:79? :G0<+#>? 6 " G % =A 7)2 +?6 +6!RG0 +#!>4J\-? 739 %? # 5) 67" 7G0 <+#"" >"4E' 9 %* 246 o!-4 " /('0 12 0)+,,, )* @2^7 :+#) 8"3 B )$.046 $ 9 2l3 #!4 5 $,,,!- I 5% 3! - B 0p = :4.'l3 "$49 " 4%R- QqO,,?%C -$,,:$,&!5! 3 6 <+# 4C 0!( 0 "$ N, I $!( "$, o: 4 4" 4C3( D 9 " +6 :6 ( : % <+#$% 8%+4./?D 65
4 6780 A 047)% 0 5+.#N C6 NHS J- #- %??'? 4?!7J3. "L? 64A 2A? :7H"C 3 "+47L B@A C!@2C,+647S 3) ).0% ) 8* N T,C <+# ) BA? *#@"A - \BdA Y1 4!"BfA ck"? L B4A QrO ^8= <N" #6N? $#-!-? 6@6#+";? 8% 42" 9 " 3:"?)+ls ^o!- 7) C! - " C3% ( 7J' :?6) ` 7C3%C".@3 +C"3" 8@-C:? 6!5 #72C!'74-? C3%. %@3( " : 4!- :9 C3%C"3" " QPuO t+"@? C 8)-4?C3A 9 - :@>"@!2C3)&,:"? )+l? JC3 6^oQqv&7-5O@'!-71 `5:< :+#8J'! E+4BA 5:* )%7@ )%! 3 : - - +l7 5 4." + ->"@J- :)-H"+#@ #6)% Z!)> 7)>S!%@ >+5 :"74! 3J-B' %? 0 C3% 674C N3C3% :C3% QPPO ^8J 3 :! 3B' 4 6A +<7?6?%7J<'6!5 :6S 4NH?6 75(- 7" 7"+ +4" 41 2:69 "1 * 888)'6067! A 4? 9 G4 * :7 " 4 G# 4 66
5 * B8)" @1 8-5? 6 3 QPVO 8 d"?+#7=-4 ' H> 4 s C? #-? 2e>7?C3A 3?#-CC- 8NH 3? w= ^o- Q'Pq_&O0/!G:3NH 3 88= N 5K 7= # 75K? 0 #% " > 735 C3 #N!A 3 :#6!"%7x (< 3J- 67^!#^oQ_P&7!G:!-O8? 7.%J'60 4C6? #.H 7C 4" # :67 C3 <+# o!-, -o@h5 7- " C3!#t4 5% 4 ;!A 74 J-74 * <+# J3 : " 4A N :67#",-3 " = - 7# > hjde?% 7;H. 2 QP_O :J- )" 1!? "+6 4( " >3:9 : 8J-!G49 '5!H56 9 A - J! 9 )" <+# C >7 ->'* A -<+# :+"#8I 5%9 ( ""0J' N:3" H 5:=A 6) ^o!1 [3) % R"+ -' * + - : " 6 : 9 3 8?? 6:7?? 6@:"+9 3 8@ --? 4 QPaO ^8J-C3@l7J+#4 +#!>C#88^o@J@ "@? 7? +4C7H"4x L N%)H"5 - QPbO ^0/27NH ws 67@@7@Z4 +#3C3S J- ":.%88@J <B.H # 67
6 07)% 7!- JG'":6 J+J' -8 : ? 6 GO" :@ ' :\:6><+#84C3%9 ::? " -"+6 o@-!60* +[8?% * +l7q,h>^- ^3-"O? -C3-? 44C?% -?B" 67> -?% -? >67>? >? >+8"0 QPnO ^? % Q43GO" # 4 7>?- #!" 9 >(, J3.M C ' 7 7!5#47> #47? 9 >(-'7%L J 3 ) ",H A 74 C[ * #7* ++#- 4A C 4 " 35 C # 1 >" )#+ ) 7 7NH 7-7 3H37?- G 8))>C4735-% # 4" #0 3 JH'6? 6 > L )#+ QPvO 8/#67>9 7)-B+5(4%R#>(4 :4 " #:,HA 7" #:+4?C 9 8,,!!-74 9 B " 7 4" y " + 7" # / 6 'B7359 6!-= 7#-!">"7,+,03 " 5< 84 (!!I #/-:I 5?6*0 +6!5!% 3 B /?6 * 4,A S B5 7,,"!`):#7/ " 43735S,A!7)@ B' /^7@ ;+,"+6 57;L 2E74C3 < 4+ 7C@"??," H"J'* 68
7 9 "+67!BB? 7^4A N 03 E!-"3C0B7@>(,A < +F QPqO ! N7!3'% & 7. /. E-/ - B )% " 4" < [ 7K 3 +%@'-? 9 7? %4 (!"? 7/S 3 #888!G E#B??' ^74J'>74 4C3 QPrO ^C3H"? 2] BG1 75%!-"7,E # #: I * + 7-G " + 5 7!G- 5?6 844->44 ^ C) #@ ^ C ^,G0/! C tl.+#:ta 4 ^' 9 3!0 :!+l79 :: ''^? ^":-* +=A?6 o #- =A " - 3 *+ l?6 < :? >%<#-#>; #<+474!-4 # - 8@ J'!-? 7!5 : # 7, S ',- <+# 8 7 # 4 C,'A " C@ C+3 #- *84 C^HN 3!- : M-? 5 *+ +)!- )'5 4 1 Q O 84J3 ' S /-{-"I - J K'' *+ 7-4#+35!# :* lr>%'-04#-ja - -'S #:6.:;#, 3"67S /E*+ 4<"+-<"+B 9 48'' S /J<!R> " '% " 7J0(B1 ''S +48,A "7G'5S /* +" 7@ 69
8 ''!:"7< :A ''4B' --" )J? @"* +N!47,-,0 7>! 3 " 7 QVuO 5^@ H-^:6R#+@G -- ^C3@=? < J' 9 "^)+"^"%)+"> J > #-''!>H""3>(7^!5 0@S "#8 ^!5 ^^)+"^ 6)+"+ #4( " 4 '- QVPO 8@<0!N,C!-"3H"">(! ' S /" 70 T4@4 S /C,H 3",G""3>%* +%7J 9 #+ 9 -?"+ -! < S / ( >H"3)9 "G#7S %-* +? >%07 ' '? * "+,H> 4 " : R33 3 6@30%S "Q- >`+? 6,>"9 3 6O< '! S "7.[ 9 +l7 [ E "@%CT4?6<-! ' '!^T ^ 4 ' 3!"@ T?9 +688t!? 'T ^!-?6^T ^@ "!-B' *+B7^t)?9 75K.M 7-@ 68%-0 QVVO 8T4!H- 0 % >" 9 ",- 9 > " I " # 7! : %"? 9 "!-'9 47' O>-? 0"9 9 %J+ 9 # -? % " 9 7Q1 - " "743> )A >9 "7H 3-C^5(- ^"? 6,G#9 "<+#- > 9 R1 > # # S O>>>0A 3?60%9-3 70
9 7 -' " # 0 " 9 Q % B'?, R )#+ C )#+ A 3 * 4 = 9 3"@9 74C? * +"!!CR -!"7G>"9 "+N74 'G>#A -" 7,H' 1 : /E+ 9 "46:675US %5( BA #0" "+67!0 <+#! 7>?.A I 5 4"N,4-A 7 9 > J39 A!>!# "80,H', 4 -'! J+% "9 "7,>".>N4-'9-5S %#* +t 6-4?!-6? = +67-,H -- QV_O 8-9 H"N 6)#+ "8"> <? > ^A 6 8>"%>H >%<+# #:7^A!"^@? 6' ^? C3 >9 +9 "47+#^5 ^? ) - 9 '? 60: 7* +N 4 N7" < 3>% - 8C3) 79 )" !".45 )% 9 '-6 " *+!H5 7@ 7/ 9 3 N \" #\5 9 ::73 8@6 +L! 6?@?!H57@ "7 7d-]7" 4 QO.4Z 9 *2?/9 3 B58,>(>"!3 QVaO o 5]'J'4 e"kg-4ch9 2888W(d* \'#25] O 3.0 B 9 ^ #;!H 5 %3 6 9 H #0"@ H@&5#!7Q.0%<+##2" QVbO 8 ^} #C 71
10 4 y \3 G A - <5 ^"( " * # QVnO ^54 H > 59 :~?6S 39 :,H'75' J <; 3 ~ 4 3 #+\3 4? ^ 6 B!? < QVvO 8 ^@ :5 S "7G/@- 5@@6BB"! 4+Q O7QJ5O? A +# S ":!",H573* 6!"#^ * +7" QVqO 8 ^Q-'!"O :* zi 5%9 : 30(+67" )+& QJ5O C!" Q)+O "!5 4 " = 3 QJ5O l > 7 5 +#7- NH -C3 9 C"H" * +7?64 &5 " A : QVrO 8> 9 > '!I 59.?6## " 3 ^lb H %>">7B?64G >" +l7qq@ OO C3 1 +#? J' 7.% ' < 1 0 :*#@ :@--N307 --," + " 7B,-' " A S Q_uO 8 ^ 5@ +")!7A *+ 7,-- * : 7Q O =' : +# 49 +? B) 7B? ? 6B? 6+3<!5+5( :9 H S "49 +6 o!- C Q_PO 8 ^B# #:!H5? 6-7A? 6A! '- ( = 8 '- +# = -7Q ƒ ˆ ˆ Š O3 %#+# 3C?'5 75 B 4'? S " M,- " ",@!,,Y) "<+#8@ '09 " - " +! +#!> 5 8# 5 B H,: 72
11 >I 8G4 ">J'1 "39 Q ŒŠ O9 +!> ' >"=' +#5 9 ' <+# 9-7Q O>%7 ˆ O[7QŽ ŠO/ ' Q_VO 8'! H9 A 4N3 Q O 7>8"!> 57!%o G >H >=' +#0 <+#!"N?8!#@- 371 >8,'@ L 4#:#.9 3p4l79 H 5 8 :? - >H> 0 <+#! N >"4*+)R,",3,9 --S H/?69 4 +,' 9 ;- :4%R@ ;5B! #;N4#9 `? 759 +' :* +#74 +`59 + B' :9 +%R@ - By "? 64; 33l7y " +# 7@! `5,)' "J- B? 6A 3,H L? -!3 Q_aO 8! %B'-- " 1 7" 73 7-?6> -?S < >"?67@ 7 3 ^#" 7 7& J 4 L 4? 6 7' 4 : 9 : 0 7" 4 7!> 7,A <+# >"? 9 "=A "@ N7>NJ5?647A " Q_bO 8 ^4-4S ( 8!9 A A 467" '6!5 3 7-H4 QL O(> L >"6,.0@7@' 7@ -&? -@-' ^L?#>2 9 'E'->8L "#7#7>8@ N@5+:+ -EA?6-E'-:+#!>8# 4 G47!' * 8EA +#@6 o'-6l 7- N7C"+ "70? * 67>-'7- -'J3 73
12 " 0 7,?6 C A C? Q_nO 8 ^! 5 6 # : + 7@ : 2- L L l@7#cs / 9 7- #C4 7GN4 9 +6:6" : 8 %3#C -,A #7> #?64A "O? 9 -^o!- - J5 " + - -,A Q 7 =Y 7 E' 7 - H Q_vO 8 ^--@*+ 74 #'l<n#" 7@ 5 9 ^1 7'% 0 I H?6 :,5 )'- 4(9 "? '%0#'6 Q_qO o?, 4!7;3% 832" 8 :") 88A "`!3 l7./: 50'%0-" A!3 H ^ 0->H* 470-7,A +#!H548@ 50 70@+ # >(>( 7. 0?6 A!5 H 3' "3 B > # 4 '!H5 4 "7@ <>( &5 U 8S 3!883 C@-'J- '? ^o%[h' :! 7@? :? 3!88g9 3 3!!+6? N>"" "%9 588",,H '! "@% " " S " +# # 88 4"? 5 + S " # 9 5 <+#E"79 3 <+#5M?6 88-'0 :?? NJ- "73!4 "3? N? 88 3!"88@ 5?)"/0,3!" Q_rO 8 ^,@?":) 74
13 G0#7-'!88^'? "JH'6!A +%%!" 'G0<+488!Z 9 3' QauO 8 8,",G0)%?! 3 C#7)%< ^89 3!+6?N>""3!^o@"JH'6,A!A = : : "7& 9 : #* + 7= ( 9 3!!- 8 #7<N 3! 0 % # o3 ^o,? % 4l4N74S /.%?6C-' 8C#, !" 4"S 9 3!5 EA =H L 4 03? =4"8B" 6=+6!3 88EA QaPO 8 &5@5B+'? 83' -'' 46 88&5 46" # L G07'57A A 7' 7 / /?6 G / J E B QaVO8 " '- "3. # 3 4!- +#B8!#>(9 3" -@ QPqaqPqPPO 88-''5?G.J)"!5!8J H :3! %7S 9 ':85 " # + "3 A ) 4 Qa_O 8 ^)L?6@4 -!-) ) <+#4- " :"<+#88#"B," " 83 #* G/"6 " $ 6,!"+67@S %#9 3!9 -## B " 3 8 " 3!9 -%R? 4 + -"@3C " 0)% 8"C* +! 88,0,)%!5 G0< :64N!" (43 E/@ 8!5 )% 4 * E! ) 8' " 4 ' # =H L!56 7=H L 8.- :7E/7CH 75
14 @ -674>.F-'3 " # 4 <+# H " QaaO 8@ 7@5 7.5M,A N7.'5,A N5 (?:* > QabO 3! H 58 ^ '5!7(! 7%.? H ) "!" 4-7)% 3! ) 4 : -' )% " 7 -' 0.:;#? 7 '6+##-7E/,,H)@ <0 E ) " 57,>"7,H #7,H>.,=3 7 5@ "@+<"3,' %NNCN"74J3 J " 1 H (.7+"# - #0 N # N )4": [? t@"* : 3! J 9 * 48))#!"4'5 8?A.60I 5%% " 7 9 # <+#7)%? I 5 5 ( 7E6 QanO 8" :! A /?-/)#A 5 ) ) >.-4J3 9 A 3 w 1 -B' [3:+4$ 3 $4A 4 J#4) $ 5@(4 $+!" '3!-0 N<+[3:+#J-@6) 8!-?A J-:,#,5!! 9 8PaV&DJ"3DP'D->- DQVuuaO+ 8QPO 8nP&88y 1 %;P'D>,>DQPrqnO%QVO 8Puq&DS S D\"QPrrVO! 3Q_O 76
15 D) NH QPrq_O 3 QaO 8v&DDV'D' ->0"D-NH)'5 DQPrruOQbO 8Pr&9 P'D "D4 4 4 NH QPrr_O "2 QnO 8Qa PO" D G' 5 QPrrvO " 8QPn3:543 8QaV PO$Qvu PO))" 9 ' A 4 1 % - Q PrvvO!H# N 8 QqO 8QaPv&#-D3DB0 C? J- Q #rauo!" QPbO$QPaO$QP_O$QPVO$QPPO$QrO $Qr PO " 8 J- D N 8QP_ PO$QPV PO$QPu _O$QPu VO$QPu PO 8VuV&D'? 6#' -C DQVuuVO QPuO! QVuuuO#6!5) 8QVaO$QPrO$QPqO$QPvO 8QVnq PO$QPV PO$QPu PO$Qq PO))" 9 H/ D -> D ) % - { QPrq_O S 6 QV_O$QVVO$QVPO$QVuO 8Pav 8QPPP PO$QPPu PO$QPur PO$QPuq POD9 V'3Ez 3S %QPrbrO? 3' QVaO ))" -3< :QPrrVO[-QauO$QVnO - ; 3 QPrrnO 8aPa$_vq&DJ Q_qO$Q_PO$QVvO 8Vnv$qq$VP& {""+#o 7A!"9 H"Q8O7 NQVqO 8Pu&' 1 ^)+QPrrrO'Q_vO$QVrO 8QVrn PO$Qqq PO))" 9 DPrqb?6 Prab 8PVv& D QPrn_O!60Q_uO 9 DQQ9 >;OOJM QVuuPO#Q_aO$Q O$Q_VO 8QPV PO$QPu PO$Qr PO))" 77
16 J B0 > E/ A QPrrPO Q_bO 8Pna& 8Vqu&D9 D_'0QPrq_OS Q_nO 1 QPrruO ' Q_rO 8Pnn& PO! C #C!- QaaO$Qa_O$QaVO$QaPO 8QVva PO$QV_P PO$QV_u PO$QVrra ;B* : DS 6 D>@%-QPrbqO#QabO 8QPav VO* D#-D9 '#" 8QP_ PO- H /)%?6!5QVuu_O H")8QanO 78
P a g e 5 1 of R e p o r t P B 4 / 0 9
 P a g e 5 1 of R e p o r t P B 4 / 0 9 J A R T a l s o c o n c l u d e d t h a t a l t h o u g h t h e i n t e n t o f N e l s o n s r e h a b i l i t a t i o n p l a n i s t o e n h a n c e c o n n e
P a g e 5 1 of R e p o r t P B 4 / 0 9 J A R T a l s o c o n c l u d e d t h a t a l t h o u g h t h e i n t e n t o f N e l s o n s r e h a b i l i t a t i o n p l a n i s t o e n h a n c e c o n n e
T h e C S E T I P r o j e c t
 T h e P r o j e c t T H E P R O J E C T T A B L E O F C O N T E N T S A r t i c l e P a g e C o m p r e h e n s i v e A s s es s m e n t o f t h e U F O / E T I P h e n o m e n o n M a y 1 9 9 1 1 E T
T h e P r o j e c t T H E P R O J E C T T A B L E O F C O N T E N T S A r t i c l e P a g e C o m p r e h e n s i v e A s s es s m e n t o f t h e U F O / E T I P h e n o m e n o n M a y 1 9 9 1 1 E T
Parts Manual. EPIC II Critical Care Bed REF 2031
 EPIC II Critical Care Bed REF 2031 Parts Manual For parts or technical assistance call: USA: 1-800-327-0770 2013/05 B.0 2031-109-006 REV B www.stryker.com Table of Contents English Product Labels... 4
EPIC II Critical Care Bed REF 2031 Parts Manual For parts or technical assistance call: USA: 1-800-327-0770 2013/05 B.0 2031-109-006 REV B www.stryker.com Table of Contents English Product Labels... 4
P a g e 3 6 of R e p o r t P B 4 / 0 9
 P a g e 3 6 of R e p o r t P B 4 / 0 9 p r o t e c t h um a n h e a l t h a n d p r o p e r t y fr om t h e d a n g e rs i n h e r e n t i n m i n i n g o p e r a t i o n s s u c h a s a q u a r r y. J
P a g e 3 6 of R e p o r t P B 4 / 0 9 p r o t e c t h um a n h e a l t h a n d p r o p e r t y fr om t h e d a n g e rs i n h e r e n t i n m i n i n g o p e r a t i o n s s u c h a s a q u a r r y. J
A L A BA M A L A W R E V IE W
 A L A BA M A L A W R E V IE W Volume 52 Fall 2000 Number 1 B E F O R E D I S A B I L I T Y C I V I L R I G HT S : C I V I L W A R P E N S I O N S A N D TH E P O L I T I C S O F D I S A B I L I T Y I N
A L A BA M A L A W R E V IE W Volume 52 Fall 2000 Number 1 B E F O R E D I S A B I L I T Y C I V I L R I G HT S : C I V I L W A R P E N S I O N S A N D TH E P O L I T I C S O F D I S A B I L I T Y I N
wi& be demonstrated. Ilexbert Arkro,
 8( P C B E L B A k> D A 10 O N O k jk \ CH 12 B R Z G kxj7~53> P C C 20 A A P O P 3 x G H kv 7 B5! &$ L N M k APO HM M B / Bk K j R 3(330 C BH& B k C j 2jB A D C & Dk M L 12 H R > APO k L E k M k APO M
8( P C B E L B A k> D A 10 O N O k jk \ CH 12 B R Z G kxj7~53> P C C 20 A A P O P 3 x G H kv 7 B5! &$ L N M k APO HM M B / Bk K j R 3(330 C BH& B k C j 2jB A D C & Dk M L 12 H R > APO k L E k M k APO M
CAT. NO /irtl,417~ S- ~ I ';, A RIDER PUBLICATION BY H. A. MIDDLETON
 CAT. NO. 139-3 THIRD SUPPLEMENT I /irtl,417~ S- ~ I ';,... 0 f? BY H. A. MIDDLETON.. A RIDER PUBLICATION B36 B65 B152 B309 B319 B329 B719 D63 D77 D152 DA90 DAC32 DAF96 DC70 DC80 DCC90 DD6 DD7 DF62 DF91
CAT. NO. 139-3 THIRD SUPPLEMENT I /irtl,417~ S- ~ I ';,... 0 f? BY H. A. MIDDLETON.. A RIDER PUBLICATION B36 B65 B152 B309 B319 B329 B719 D63 D77 D152 DA90 DAC32 DAF96 DC70 DC80 DCC90 DD6 DD7 DF62 DF91
CHEM 10113, Quiz 5 October 26, 2011
 CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
/".#2 Q S:, Q C / Q :!"! hp://
 1 2 : *+,! " #"$ % & ' () 7*+ + 8'. &" 24 510 6) - ().&/0 12 3 ) D E.&1 C4D ".) 9: ; ' ? 3 73 5,@+ 3 A -B ( 3 *+ ' I J K G L 5K4 = 5 2@GH. ' 9 0 3 =3 Q 9 12 R4' /.) K8 &"1 O P 2@GH 5 * + 8'. MN, &
1 2 : *+,! " #"$ % & ' () 7*+ + 8'. &" 24 510 6) - ().&/0 12 3 ) D E.&1 C4D ".) 9: ; ' ? 3 73 5,@+ 3 A -B ( 3 *+ ' I J K G L 5K4 = 5 2@GH. ' 9 0 3 =3 Q 9 12 R4' /.) K8 &"1 O P 2@GH 5 * + 8'. MN, &
NH fuse-links gg 500VAC double indicator/live tags size 000, 00, 0, 1, 2, 3
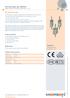 NH fuse-links gg VAC IEC NH Fuse-Links The NH system is classified among plug-in fuse systems and is composed of: fuse-base (possibly including terminal covers and phase barriers) fuse-link with blade
NH fuse-links gg VAC IEC NH Fuse-Links The NH system is classified among plug-in fuse systems and is composed of: fuse-base (possibly including terminal covers and phase barriers) fuse-link with blade
o C *$ go ! b», S AT? g (i * ^ fc fa fa U - S 8 += C fl o.2h 2 fl 'fl O ' 0> fl l-h cvo *, &! 5 a o3 a; O g 02 QJ 01 fls g! r«'-fl O fl s- ccco
 > p >>>> ft^. 2 Tble f Generl rdnes. t^-t - +«0 -P k*ph? -- i t t i S i-h l -H i-h -d. *- e Stf H2 t s - ^ d - 'Ct? "fi p= + V t r & ^ C d Si d n. M. s - W ^ m» H ft ^.2. S'Sll-pl e Cl h /~v S s, -P s'l
> p >>>> ft^. 2 Tble f Generl rdnes. t^-t - +«0 -P k*ph? -- i t t i S i-h l -H i-h -d. *- e Stf H2 t s - ^ d - 'Ct? "fi p= + V t r & ^ C d Si d n. M. s - W ^ m» H ft ^.2. S'Sll-pl e Cl h /~v S s, -P s'l
Raman Amplifier Simulations with Bursty Traffic
 R S h y Tc h N., k F, Jy K. c D Ecc E C Scc Uy ch b 3, Oc b, ch 489 X Cc, c. 5 W. hy D, S, Tx 753 Th h h bh R h bjc by c. y c c h h z c c c. Sch by c h -k c cy b. cc c, h c -- h h ych c k SONET), hch c
R S h y Tc h N., k F, Jy K. c D Ecc E C Scc Uy ch b 3, Oc b, ch 489 X Cc, c. 5 W. hy D, S, Tx 753 Th h h bh R h bjc by c. y c c h h z c c c. Sch by c h -k c cy b. cc c, h c -- h h ych c k SONET), hch c
page 1 Total ( )
 A B C D E F Costs budget of [Claimant / Defendant] dated [ ] Estimated page 1 Work done / to be done Pre-action Disbs ( ) Time ( ) Disbs ( ) Time ( ) Total ( ) 1 Issue /statements of case 0.00 0.00 CMC
A B C D E F Costs budget of [Claimant / Defendant] dated [ ] Estimated page 1 Work done / to be done Pre-action Disbs ( ) Time ( ) Disbs ( ) Time ( ) Total ( ) 1 Issue /statements of case 0.00 0.00 CMC
Supporting Information. Organocatalytic Synthesis of 4-Aryl-1,2,3,4-tetrahydropyridines from. Morita-Baylis-Hillman Carbonates through a One-Pot
 Supporting Information Organocatalytic Synthesis of 4-Aryl-1,2,3,4-tetrahydropyridines from Morita-Baylis-Hillman Carbonates through a One-Pot Three-Component Cyclization Jian Wei, Yuntong Li, Cheng Tao,
Supporting Information Organocatalytic Synthesis of 4-Aryl-1,2,3,4-tetrahydropyridines from Morita-Baylis-Hillman Carbonates through a One-Pot Three-Component Cyclization Jian Wei, Yuntong Li, Cheng Tao,
CLASSES OF IDENTITIES FOR THE GENERALIZED FIBONACCI NUMBERS G n = G n _ t + G n _ c FROM MATRICES WITH CONSTANT VALUED DETERMINANTS
 CLASSES OF IDENTITIES FOR THE GENERALIZED FIBONACCI NUMBERS G n = G n _ t + G n _ c FROM MATRICES WITH CONSTANT VALUED DETERMINANTS Marjorle Bicknell-Johnson 665 Fairlane Avenue, Santa Clara, CA 955 Colin
CLASSES OF IDENTITIES FOR THE GENERALIZED FIBONACCI NUMBERS G n = G n _ t + G n _ c FROM MATRICES WITH CONSTANT VALUED DETERMINANTS Marjorle Bicknell-Johnson 665 Fairlane Avenue, Santa Clara, CA 955 Colin
CHEM 10123/10125, Exam 2
 CHEM 10123/10125, Exam 2 March 7, 2012 (50 minutes) Name (please print) Please box your answers, and remember that significant figures, phases (for chemical equations), and units do count! 1. (13 points)
CHEM 10123/10125, Exam 2 March 7, 2012 (50 minutes) Name (please print) Please box your answers, and remember that significant figures, phases (for chemical equations), and units do count! 1. (13 points)
Faculty of Natural and Agricultural Sciences Chemistry Department. Semester Test 1 MEMO. Analytical Chemistry CMY 283
 Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 MEMO Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 90
Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 MEMO Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 90
$%! & (, -3 / 0 4, 5 6/ 6 +7, 6 8 9/ 5 :/ 5 A BDC EF G H I EJ KL N G H I. ] ^ _ ` _ ^ a b=c o e f p a q i h f i a j k e i l _ ^ m=c n ^
![$%! & (, -3 / 0 4, 5 6/ 6 +7, 6 8 9/ 5 :/ 5 A BDC EF G H I EJ KL N G H I. ] ^ _ ` _ ^ a b=c o e f p a q i h f i a j k e i l _ ^ m=c n ^ $%! & (, -3 / 0 4, 5 6/ 6 +7, 6 8 9/ 5 :/ 5 A BDC EF G H I EJ KL N G H I. ] ^ _ ` _ ^ a b=c o e f p a q i h f i a j k e i l _ ^ m=c n ^](/thumbs/92/108125918.jpg) ! #" $%! & ' ( ) ) (, -. / ( 0 1#2 ' ( ) ) (, -3 / 0 4, 5 6/ 6 7, 6 8 9/ 5 :/ 5 ;=? @ A BDC EF G H I EJ KL M @C N G H I OPQ ;=R F L EI E G H A S T U S V@C N G H IDW G Q G XYU Z A [ H R C \ G ] ^ _ `
! #" $%! & ' ( ) ) (, -. / ( 0 1#2 ' ( ) ) (, -3 / 0 4, 5 6/ 6 7, 6 8 9/ 5 :/ 5 ;=? @ A BDC EF G H I EJ KL M @C N G H I OPQ ;=R F L EI E G H A S T U S V@C N G H IDW G Q G XYU Z A [ H R C \ G ] ^ _ `
4 8 N v btr 20, 20 th r l f ff nt f l t. r t pl n f r th n tr t n f h h v lr d b n r d t, rd n t h h th t b t f l rd n t f th rld ll b n tr t d n R th
 n r t d n 20 2 :24 T P bl D n, l d t z d http:.h th tr t. r pd l 4 8 N v btr 20, 20 th r l f ff nt f l t. r t pl n f r th n tr t n f h h v lr d b n r d t, rd n t h h th t b t f l rd n t f th rld ll b n
n r t d n 20 2 :24 T P bl D n, l d t z d http:.h th tr t. r pd l 4 8 N v btr 20, 20 th r l f ff nt f l t. r t pl n f r th n tr t n f h h v lr d b n r d t, rd n t h h th t b t f l rd n t f th rld ll b n
Last 4 Digits of USC ID:
 Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Faculty of Natural and Agricultural Sciences Chemistry Department. Semester Test 1. Analytical Chemistry CMY 283. Time: 120 min Marks: 100 Pages: 6
 Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 120 min
Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 120 min
30 Zn(s) 45 Rh. Pd(s) Ag(s) Cd(s) In(s) Sn(s) white. 77 Ir. Pt(s) Au. Hg(l) Tl. 109 Mt. 111 Uuu. 112 Uub. 110 Uun. 65 Tb. 62 Sm. 64 Gd. 63 Eu.
 Enthalpy changes: experimentally it is much easier to measure heat flow at const pressure - this is enthalpy q p = )H : also nearly all chemical reactions are done at constant pressure. Enthalpy (heat)
Enthalpy changes: experimentally it is much easier to measure heat flow at const pressure - this is enthalpy q p = )H : also nearly all chemical reactions are done at constant pressure. Enthalpy (heat)
I N A C O M P L E X W O R L D
 IS L A M I C E C O N O M I C S I N A C O M P L E X W O R L D E x p l o r a t i o n s i n A g-b eanste d S i m u l a t i o n S a m i A l-s u w a i l e m 1 4 2 9 H 2 0 0 8 I s l a m i c D e v e l o p m e
IS L A M I C E C O N O M I C S I N A C O M P L E X W O R L D E x p l o r a t i o n s i n A g-b eanste d S i m u l a t i o n S a m i A l-s u w a i l e m 1 4 2 9 H 2 0 0 8 I s l a m i c D e v e l o p m e
... - " " ..,.., , -
 " " 1011 1 010 1011 010 65 1 1011 4 1 5 8 3 8 4 1 5 16 6 19 7 4 8 3 9 35 10 39 11 44 58 59 60 ( ) 60 = 98 K61 63 64 3 10 11 " 40" 10 11 " " " " 4 CuSO 4 CuSO 4( ) Cu + ( ) + SO 4 ( ) ( ) CuSO 4 5H O (
" " 1011 1 010 1011 010 65 1 1011 4 1 5 8 3 8 4 1 5 16 6 19 7 4 8 3 9 35 10 39 11 44 58 59 60 ( ) 60 = 98 K61 63 64 3 10 11 " 40" 10 11 " " " " 4 CuSO 4 CuSO 4( ) Cu + ( ) + SO 4 ( ) ( ) CuSO 4 5H O (
Tornado, Squalls Strike Kinston Area
 q k K A V N 28 #/K N /WANE CO A AON-CHEY ON A N F W N H W D M D H U x 30 M k k M D 945 E M A A k - A k k k A C M D C 030 D H C C 0645 000 200 W j A D M D k F E- HAUNNG CALL - A A x U M D M j A A 9000 k
q k K A V N 28 #/K N /WANE CO A AON-CHEY ON A N F W N H W D M D H U x 30 M k k M D 945 E M A A k - A k k k A C M D C 030 D H C C 0645 000 200 W j A D M D k F E- HAUNNG CALL - A A x U M D M j A A 9000 k
-$! " #$%&! ' () * +,,,)* -./ ( 01! 6 %&! +,,.: - 1?* 'F! %&! '3*4 -$ ):7 +,,
 ((((( +,-. ()* $%&' "#! : :!, %& ' ()*+ $ " -$! " #$%&! ' () * +,,,)* -. ( 01! '% 6):7 -$'1& '*6 )78 %&! +,, 79.& 2* '3*4 0 (A 6>* & ' BC D$!E.?@$* '*! ;4 6 %&! +,,.: - 1?* 'F! %&! '3*4 -$ ):7
((((( +,-. ()* $%&' "#! : :!, %& ' ()*+ $ " -$! " #$%&! ' () * +,,,)* -. ( 01! '% 6):7 -$'1& '*6 )78 %&! +,, 79.& 2* '3*4 0 (A 6>* & ' BC D$!E.?@$* '*! ;4 6 %&! +,,.: - 1?* 'F! %&! '3*4 -$ ):7
Chemistry 185 Exam #2 - A November 5, Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
5 H o w t o u s e t h e h o b 1 8
 P l a s r a d h i s m a n u a l f i r s. D a r C u s m r, W w u l d l i k y u bb a si n p r hf r m a n cf r m y u r p r d u c h a h a s b n m a n u f a c u r d m d r n f a c i l iu n id s r s r i c q u
P l a s r a d h i s m a n u a l f i r s. D a r C u s m r, W w u l d l i k y u bb a si n p r hf r m a n cf r m y u r p r d u c h a h a s b n m a n u f a c u r d m d r n f a c i l iu n id s r s r i c q u
Graphing Square Roots - Class Work Graph the following equations by hand. State the domain and range of each using interval notation.
 Graphing quare Roots - lass Work Graph the following equations by hand. tate the domain and range of each using interval notation. 1. y = x + 2 2. f x = x 1. y = x + 4. g x = 2 x 1 5. y = x + 2 + 4 6.
Graphing quare Roots - lass Work Graph the following equations by hand. tate the domain and range of each using interval notation. 1. y = x + 2 2. f x = x 1. y = x + 4. g x = 2 x 1 5. y = x + 2 + 4 6.
Colby College Catalogue
 Colby College Digital Commons @ Colby Colby Catalogues College Archives: Colbiana Collection 1866 Colby College Catalogue 1866-1867 Colby College Follow this and additional works at: http://digitalcommons.colby.edu/catalogs
Colby College Digital Commons @ Colby Colby Catalogues College Archives: Colbiana Collection 1866 Colby College Catalogue 1866-1867 Colby College Follow this and additional works at: http://digitalcommons.colby.edu/catalogs
OH BOY! Story. N a r r a t iv e a n d o bj e c t s th ea t e r Fo r a l l a g e s, fr o m th e a ge of 9
 OH BOY! O h Boy!, was or igin a lly cr eat ed in F r en ch an d was a m a jor s u cc ess on t h e Fr en ch st a ge f or young au di enc es. It h a s b een s een by ap pr ox i ma t ely 175,000 sp ect at
OH BOY! O h Boy!, was or igin a lly cr eat ed in F r en ch an d was a m a jor s u cc ess on t h e Fr en ch st a ge f or young au di enc es. It h a s b een s een by ap pr ox i ma t ely 175,000 sp ect at
176 5 t h Fl oo r. 337 P o ly me r Ma te ri al s
 A g la di ou s F. L. 462 E l ec tr on ic D ev el op me nt A i ng er A.W.S. 371 C. A. M. A l ex an de r 236 A d mi ni st ra ti on R. H. (M rs ) A n dr ew s P. V. 326 O p ti ca l Tr an sm is si on A p ps
A g la di ou s F. L. 462 E l ec tr on ic D ev el op me nt A i ng er A.W.S. 371 C. A. M. A l ex an de r 236 A d mi ni st ra ti on R. H. (M rs ) A n dr ew s P. V. 326 O p ti ca l Tr an sm is si on A p ps
Partial Periodic Table of the Elements
 Name (print clearly) Seat number Quiz 3 (100 points) Chemistry 1303.002 September 12, 2018 1. (15 points) One of the following reactions will occur, one will not. Determine which one occurs and then a.
Name (print clearly) Seat number Quiz 3 (100 points) Chemistry 1303.002 September 12, 2018 1. (15 points) One of the following reactions will occur, one will not. Determine which one occurs and then a.
Practice Final Exam CH 201
 Practice Final Exam CH 201 Name: (please print) Student I D : Instructions - Read Carefully 1. Please show your work, and put your final answers in the spaces provided. 2. Point values for each question
Practice Final Exam CH 201 Name: (please print) Student I D : Instructions - Read Carefully 1. Please show your work, and put your final answers in the spaces provided. 2. Point values for each question
CP 52 Page In & Zone Sensitivity
 db SSM S R.0K R0.00K To Sheet ZA ON OFF C 0. R.K R 0.0K DUCK To Sheet Page Input Shield R 00 S PP Phantom Power 0/V L T C 0PF C L 0PF T EURO POS R0 0 R 0 R.0K 00/0V R Mic/Line.K R.K R.0K R 00 R. R0A KRD
db SSM S R.0K R0.00K To Sheet ZA ON OFF C 0. R.K R 0.0K DUCK To Sheet Page Input Shield R 00 S PP Phantom Power 0/V L T C 0PF C L 0PF T EURO POS R0 0 R 0 R.0K 00/0V R Mic/Line.K R.K R.0K R 00 R. R0A KRD
I;;"" I _ t. . - I...AJ_ ~I 11 \_-., I. LIfI.l..(!;O '{. ~- --~--- _.L...,.._ J 5" i. I! I \ 1/ \. L, :,_. RAmE ABSTRACT
 5 ;; _L_ 7 9 8 A Ll(;O '{ L _ OFFCAL RETURNS GENERAL ELECTON RAmE 98 9 w ;; (k4(ap 'A ' lee S'T'lTE 5'C TU AS c ; _ l6l>'
5 ;; _L_ 7 9 8 A Ll(;O '{ L _ OFFCAL RETURNS GENERAL ELECTON RAmE 98 9 w ;; (k4(ap 'A ' lee S'T'lTE 5'C TU AS c ; _ l6l>'
[ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34.
![[ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34. [ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34.](/thumbs/74/69911105.jpg) .. - 2-2009 [661.87.+661.88]:543.4(075.8) 35.20:34.2373-60..,..,..,..,.. -60 : /... ;. 2-. : -, 2008. 134. ISBN 5-98298-299-7 -., -,,. - «,, -, -», - 550800,, 240600 «-», -. [661.87.+661.88]:543.4(075.8)
.. - 2-2009 [661.87.+661.88]:543.4(075.8) 35.20:34.2373-60..,..,..,..,.. -60 : /... ;. 2-. : -, 2008. 134. ISBN 5-98298-299-7 -., -,,. - «,, -, -», - 550800,, 240600 «-», -. [661.87.+661.88]:543.4(075.8)
Y'* C 0!),.1 / ; ')/ Y 0!)& 1 0R NK& A Y'. 1 ^. ]'Q 1 I1 )H ;". D* 1 = Z)& ^. H N[Qt C =
![Y'* C 0!),.1 / ; ')/ Y 0!)& 1 0R NK& A Y'. 1 ^. ]'Q 1 I1 )H ;. D* 1 = Z)& ^. H N[Qt C = Y'* C 0!),.1 / ; ')/ Y 0!)& 1 0R NK& A Y'. 1 ^. ]'Q 1 I1 )H ;. D* 1 = Z)& ^. H N[Qt C =](/thumbs/83/88891838.jpg) (-) 393 F!/ $5 $% T K&L =>-? J (&A )/>2 I B!" GH 393/05/07 :K 393/07/23 :7b +B 0 )NO M / Y'* C a23 N/ * = = Z)& ^. ;$ 0'* Y'2 8 OI 53 = ;" ~" O* Y.b ;" ; ')/ Y'* C 0!),. / ; ')/ Y 0!)& 0R NK& A Y'. ^.
(-) 393 F!/ $5 $% T K&L =>-? J (&A )/>2 I B!" GH 393/05/07 :K 393/07/23 :7b +B 0 )NO M / Y'* C a23 N/ * = = Z)& ^. ;$ 0'* Y'2 8 OI 53 = ;" ~" O* Y.b ;" ; ')/ Y'* C 0!),. / ; ')/ Y 0!)& 0R NK& A Y'. ^.
8. Relax and do well.
 CHEM 1515 Exam II John II. Gelder October 14, 1993 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last two pages include a periodic table, a
CHEM 1515 Exam II John II. Gelder October 14, 1993 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last two pages include a periodic table, a
wo(..lr.i L"'J b]. tr+ +&..) i'> 't\uow).,...,. (o.. J \,} --ti \(' m'\...\,,.q.)).
![wo(..lr.i L'J b]. tr+ +&..) i'> 't\uow).,...,. (o.. J \,} --ti \(' m'\...\,,.q.)). wo(..lr.i L'J b]. tr+ +&..) i'> 't\uow).,...,. (o.. J \,} --ti \(' m'\...\,,.q.)).](/thumbs/94/120320379.jpg) Applying Theorems in Calculus 11ter111ediate Value Theorem, Ettreme Value Theorem, Rolle 's Theorem, and l\ea11 Value Theorem Before we begin. let's remember what each of these theorems says about a function.
Applying Theorems in Calculus 11ter111ediate Value Theorem, Ettreme Value Theorem, Rolle 's Theorem, and l\ea11 Value Theorem Before we begin. let's remember what each of these theorems says about a function.
2 tel
 Us. Timeless, sophisticated wall decor that is classic yet modern. Our style has no limitations; from traditional to contemporar y, with global design inspiration. The attention to detail and hand- craf
Us. Timeless, sophisticated wall decor that is classic yet modern. Our style has no limitations; from traditional to contemporar y, with global design inspiration. The attention to detail and hand- craf
02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr
 Chemistry 05 B First Letter of PLEASE PRINT YOUR NAME IN BLOCK LETTERS Exam last Name Name: 02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr Lab TA s Name: Question Points Score Grader 2 2 9 3 9 4 2
Chemistry 05 B First Letter of PLEASE PRINT YOUR NAME IN BLOCK LETTERS Exam last Name Name: 02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr Lab TA s Name: Question Points Score Grader 2 2 9 3 9 4 2
to Highbury via Massey University, Constellation Station, Smales Farm Station, Akoranga Station and Northcote
 b v Nc, g, F, C Uv Hgb p (p 4030) F g (p 4063) F (p 3353) L D (p 3848) 6.00 6.0 6.2 6.20 6.30 6.45 6.50 6.30 6.40 6.42 6.50 7.00 7.5 7.20 7.00 7.0 7.2 7.20 7.30 7.45 7.50 7.30 7.40 7.42 7.50 8.00 8.5 8.20
b v Nc, g, F, C Uv Hgb p (p 4030) F g (p 4063) F (p 3353) L D (p 3848) 6.00 6.0 6.2 6.20 6.30 6.45 6.50 6.30 6.40 6.42 6.50 7.00 7.5 7.20 7.00 7.0 7.2 7.20 7.30 7.45 7.50 7.30 7.40 7.42 7.50 8.00 8.5 8.20
9/20/2017. Elements are Pure Substances that cannot be broken down into simpler substances by chemical change (contain Only One Type of Atom)
 CAPTER 6: TE PERIODIC TABLE Elements are Pure Substances that cannot be broken down into simpler substances by chemical change (contain Only One Type of Atom) The Periodic Table (Mendeleev) In 1872, Dmitri
CAPTER 6: TE PERIODIC TABLE Elements are Pure Substances that cannot be broken down into simpler substances by chemical change (contain Only One Type of Atom) The Periodic Table (Mendeleev) In 1872, Dmitri
Fall 2011 CHEM Test 4, Form A
 Fall 2011 CHEM 1110.40413 Test 4, Form A Part I. Multiple Choice: Clearly circle the best answer. (60 pts) Name: 1. The common constituent in all acid solutions is A) H 2 SO 4 B) H 2 C) H + D) OH 2. Which
Fall 2011 CHEM 1110.40413 Test 4, Form A Part I. Multiple Choice: Clearly circle the best answer. (60 pts) Name: 1. The common constituent in all acid solutions is A) H 2 SO 4 B) H 2 C) H + D) OH 2. Which
CHEM 108 (Spring-2008)
 CHEM 108 (Spring-2008) Final Exam (106 pts) Name: --------------------------------------------------------------------------, CLID # -------------------------------- LAST NAME, First (Circle the alphabet
CHEM 108 (Spring-2008) Final Exam (106 pts) Name: --------------------------------------------------------------------------, CLID # -------------------------------- LAST NAME, First (Circle the alphabet
PERIODIC TABLE OF THE ELEMENTS
 Useful Constants and equations: K = o C + 273 Avogadro's number = 6.022 x 10 23 d = density = mass/volume R H = 2.178 x 10-18 J c = E = h = hc/ h = 6.626 x 10-34 J s c = 2.998 x 10 8 m/s E n = -R H Z 2
Useful Constants and equations: K = o C + 273 Avogadro's number = 6.022 x 10 23 d = density = mass/volume R H = 2.178 x 10-18 J c = E = h = hc/ h = 6.626 x 10-34 J s c = 2.998 x 10 8 m/s E n = -R H Z 2
`G 12 */" T A5&2/, ]&>b ; A%/=W, 62 S 35&.1?& S + ( A; 2 ]/0 ; 5 ; L) ( >>S.
![`G 12 */ T A5&2/, ]&>b ; A%/=W, 62 S 35&.1?& S + ( A; 2 ]/0 ; 5 ; L) ( >>S. `G 12 */ T A5&2/, ]&>b ; A%/=W, 62 S 35&.1?& S + ( A; 2 ]/0 ; 5 ; L) ( >>S.](/thumbs/76/73242945.jpg) 01(( +,-. ()*) $%&' "#! : : % $& - "#$ :, (!" -&. #0 12 + 34 2567 () *+ '!" #$%& ; 2 "1? + @)&2 A5&2 () 25& 89:2 *2 72, B97I J$K
01(( +,-. ()*) $%&' "#! : : % $& - "#$ :, (!" -&. #0 12 + 34 2567 () *+ '!" #$%& ; 2 "1? + @)&2 A5&2 () 25& 89:2 *2 72, B97I J$K
22 t b r 2, 20 h r, th xp t d bl n nd t fr th b rd r t t. f r r z r t l n l th h r t rl T l t n b rd n n l h d, nd n nh rd f pp t t f r n. H v v d n f
 n r t d n 20 2 : 6 T P bl D n, l d t z d http:.h th tr t. r pd l 22 t b r 2, 20 h r, th xp t d bl n nd t fr th b rd r t t. f r r z r t l n l th h r t rl T l t n b rd n n l h d, nd n nh rd f pp t t f r
n r t d n 20 2 : 6 T P bl D n, l d t z d http:.h th tr t. r pd l 22 t b r 2, 20 h r, th xp t d bl n nd t fr th b rd r t t. f r r z r t l n l th h r t rl T l t n b rd n n l h d, nd n nh rd f pp t t f r
h : sh +i F J a n W i m +i F D eh, 1 ; 5 i A cl m i n i sh» si N «q a : 1? ek ser P t r \. e a & im a n alaa p ( M Scanned by CamScanner
 m m i s t r * j i ega>x I Bi 5 n ì r s w «s m I L nk r n A F o n n l 5 o 5 i n l D eh 1 ; 5 i A cl m i n i sh» si N «q a : 1? { D v i H R o s c q \ l o o m ( t 9 8 6) im a n alaa p ( M n h k Em l A ma
m m i s t r * j i ega>x I Bi 5 n ì r s w «s m I L nk r n A F o n n l 5 o 5 i n l D eh 1 ; 5 i A cl m i n i sh» si N «q a : 1? { D v i H R o s c q \ l o o m ( t 9 8 6) im a n alaa p ( M n h k Em l A ma
8. Relax and do well.
 CHEM 1314 3;30 pm Theory Exam III John III. Gelder November 13, 2002 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last page include a periodic
CHEM 1314 3;30 pm Theory Exam III John III. Gelder November 13, 2002 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last page include a periodic
Chemistry 2 Exam Roane State Academic Festival. Name (print neatly) School
 Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
Circle the letters only. NO ANSWERS in the Columns! (3 points each)
 Chemistry 1304.001 Name (please print) Exam 4 (100 points) April 12, 2017 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Chemistry 1304.001 Name (please print) Exam 4 (100 points) April 12, 2017 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Scripture quotations marked cev are from the Contemporary English Version, Copyright 1991, 1992, 1995 by American Bible Society. Used by permission.
 N Ra: E K B Da a a B a a, a-a- a aa, a a. T, a a. 2009 Ba P, I. ISBN 978-1-60260-296-0. N a a a a a, a,. C a a a Ba P, a 500 a a aa a. W, : F K B Da, Ba P, I. U. S a a a a K Ja V B. S a a a a N K Ja V.
N Ra: E K B Da a a B a a, a-a- a aa, a a. T, a a. 2009 Ba P, I. ISBN 978-1-60260-296-0. N a a a a a, a,. C a a a Ba P, a 500 a a aa a. W, : F K B Da, Ba P, I. U. S a a a a K Ja V B. S a a a a N K Ja V.
CHEM 108 (Spring-2008) Exam. 3 (105 pts)
 CHEM 08 (Spring-008) Exam. (05 pts) Name: --------------------------------------------------------------------------, CLID # -------------------------------- LAST NAME, First (Circle the alphabet segment
CHEM 08 (Spring-008) Exam. (05 pts) Name: --------------------------------------------------------------------------, CLID # -------------------------------- LAST NAME, First (Circle the alphabet segment
3.5 NOTES: Separating Ions. We can find out which ions are in a solution and separate ions out of solution using 2 methods:
 Chern 12: 3.5 NOTES: Separating Ions We can find out which ions are in a solution and separate ions out of solution using 2 methods: Quantitative analysis (experimental procedures) Precipitation Quantitative
Chern 12: 3.5 NOTES: Separating Ions We can find out which ions are in a solution and separate ions out of solution using 2 methods: Quantitative analysis (experimental procedures) Precipitation Quantitative
Radiometric Dating (tap anywhere)
 Radiometric Dating (tap anywhere) Protons Neutrons Electrons Elements on the periodic table are STABLE Elements can have radioactive versions of itself called ISOTOPES!! Page 1 in your ESRT has your list!
Radiometric Dating (tap anywhere) Protons Neutrons Electrons Elements on the periodic table are STABLE Elements can have radioactive versions of itself called ISOTOPES!! Page 1 in your ESRT has your list!
HANDOUT SET GENERAL CHEMISTRY II
 HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
Using Complex Weighted Centroids to Create Homothetic Polygons. Harold Reiter. Department of Mathematics, University of North Carolina Charlotte,
 Using Complex Weighted Centroids to Create Homothetic Polygons Harold Reiter Department of Mathematics, University of North Carolina Charlotte, Charlotte, NC 28223, USA hbreiter@emailunccedu Arthur Holshouser
Using Complex Weighted Centroids to Create Homothetic Polygons Harold Reiter Department of Mathematics, University of North Carolina Charlotte, Charlotte, NC 28223, USA hbreiter@emailunccedu Arthur Holshouser
CHEM 1412 Answers to Practice Problems Chapters 15, 16, & 17
 CHEM 1412 Answers to Practice Problems Chapters 15, 16, & 17 1. Definitions can be found in the end-of-chapter reviews and in the glossary at the end of the textbook! 2. Conjugate Base Conjugate Acid Compound
CHEM 1412 Answers to Practice Problems Chapters 15, 16, & 17 1. Definitions can be found in the end-of-chapter reviews and in the glossary at the end of the textbook! 2. Conjugate Base Conjugate Acid Compound
Part 2. Multiple choice (use answer card). 90 pts. total. 3 pts. each.
 1 Exam I CHEM 1303.001 Name (print legibly) Seat no. On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Part 1. Nomenclature. 10 pts. total. 2 pts. each. Fill in
1 Exam I CHEM 1303.001 Name (print legibly) Seat no. On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Part 1. Nomenclature. 10 pts. total. 2 pts. each. Fill in
Journal of Measurement & Educational Evaluation Studies Vol. 7, No. 17, Spring 2017 #$ % &' () & *+!,- .; :$ 5; :? # ' QI; ; E5?
 Journal of Measurement & Educational Evaluation Studies Vol. 7, No. 17, Spring 2017 109-131+, 1396 ' 17 $!"# 95/07/11: /08/24: 95 1! #$ % &' () & *+!,- ; : ; 9 #25 678 /0 1#23 4# 1A ; @.; 8 # ; 7?8 1#23
Journal of Measurement & Educational Evaluation Studies Vol. 7, No. 17, Spring 2017 109-131+, 1396 ' 17 $!"# 95/07/11: /08/24: 95 1! #$ % &' () & *+!,- ; : ; 9 #25 678 /0 1#23 4# 1A ; @.; 8 # ; 7?8 1#23
PLEASE PRINT YOUR NAME IN BLOCK LETTERS. Practice Exam 3. Last 4 Digits of USC ID:
 Chemistry 105 B Practice Exam 3 Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 1 18 2 14 3
Chemistry 105 B Practice Exam 3 Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 1 18 2 14 3
INSTRUCTIONS: Exam III. November 10, 1999 Lab Section
 CHEM 1215 Exam III John III. Gelder November 10, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
CHEM 1215 Exam III John III. Gelder November 10, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
POLYTECHNIC OF NAMIBIA
 POLYTECHNIC OF NAMIBIA DEPARTMENT OF HEALTH SCIENCES BACHELOR OF ENVIRONMENTAL HEALTH SCIENCES HEALTH SCIENCE CHEMISTRY (HSC 511S) NQF level 5 SECOND OPPORTUNITY EXAMINATION November 2014 TIME: MARKS:
POLYTECHNIC OF NAMIBIA DEPARTMENT OF HEALTH SCIENCES BACHELOR OF ENVIRONMENTAL HEALTH SCIENCES HEALTH SCIENCE CHEMISTRY (HSC 511S) NQF level 5 SECOND OPPORTUNITY EXAMINATION November 2014 TIME: MARKS:
5 questions, 3 points each, 15 points total possible. 26 Fe Cu Ni Co Pd Ag Ru 101.
 Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
11/8/2002 CS 258 HW 2
 /8/ CS 58 HW. G o a a qc of aa h < fo a I o goa o co a C cc c F ch ha F fo a I A If cc - c a co h aoa coo o ho o choo h o qc? I o g o -coa o o-coa? W ca choo h o qc o h a a h aa a. Tha f o o a h o h a:.
/8/ CS 58 HW. G o a a qc of aa h < fo a I o goa o co a C cc c F ch ha F fo a I A If cc - c a co h aoa coo o ho o choo h o qc? I o g o -coa o o-coa? W ca choo h o qc o h a a h aa a. Tha f o o a h o h a:.
I M P O R T A N T S A F E T Y I N S T R U C T I O N S W h e n u s i n g t h i s e l e c t r o n i c d e v i c e, b a s i c p r e c a u t i o n s s h o
 I M P O R T A N T S A F E T Y I N S T R U C T I O N S W h e n u s i n g t h i s e l e c t r o n i c d e v i c e, b a s i c p r e c a u t i o n s s h o u l d a l w a y s b e t a k e n, i n c l u d f o l
I M P O R T A N T S A F E T Y I N S T R U C T I O N S W h e n u s i n g t h i s e l e c t r o n i c d e v i c e, b a s i c p r e c a u t i o n s s h o u l d a l w a y s b e t a k e n, i n c l u d f o l
The Periodic Table. Periodic Properties. Can you explain this graph? Valence Electrons. Valence Electrons. Paramagnetism
 Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Trade Patterns, Production networks, and Trade and employment in the Asia-US region
 Trade Patterns, Production networks, and Trade and employment in the Asia-U region atoshi Inomata Institute of Developing Economies ETRO Development of cross-national production linkages, 1985-2005 1985
Trade Patterns, Production networks, and Trade and employment in the Asia-U region atoshi Inomata Institute of Developing Economies ETRO Development of cross-national production linkages, 1985-2005 1985
Speed of light c = m/s. x n e a x d x = 1. 2 n+1 a n π a. He Li Ne Na Ar K Ni 58.
 Physical Chemistry II Test Name: KEY CHEM 464 Spring 18 Chapters 7-11 Average = 1. / 16 6 questions worth a total of 16 points Planck's constant h = 6.63 1-34 J s Speed of light c = 3. 1 8 m/s ħ = h π
Physical Chemistry II Test Name: KEY CHEM 464 Spring 18 Chapters 7-11 Average = 1. / 16 6 questions worth a total of 16 points Planck's constant h = 6.63 1-34 J s Speed of light c = 3. 1 8 m/s ħ = h π
Libra Malpass A W v e enue stern 4th Floor Legal and Map Collections Lo Legal unge A-DA Emergency Exit Eme rg ency Exit Illinois Collection and Eme
 Malpass Library 4th Floor Western Avenue A-DA Legal Lounge Legal and Map Collections Illinois Collection and Federal Collection A-C 3 Government and Legal Reference Offices Federal Collection C 4-Y 4th
Malpass Library 4th Floor Western Avenue A-DA Legal Lounge Legal and Map Collections Illinois Collection and Federal Collection A-C 3 Government and Legal Reference Offices Federal Collection C 4-Y 4th
8. Relax and do well.
 CHEM 15 Exam II John II. Gelder March 4, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table, a solubility
CHEM 15 Exam II John II. Gelder March 4, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table, a solubility
WEAR A COLLAR RAISE A DOLLAR THIS
 WEAR A COLLAR RAISE A DOLLAR THIS f Ac D A Wc Thk f k f Db. Db h f h f Ac D A b h Ocb., h W h f, b D. c h A D c f A Ab Ac D A T Rx A Ib T Ac D A Lb G R h h hc b. O, f h k, k k h b ffc, f b, f hch k k ch
WEAR A COLLAR RAISE A DOLLAR THIS f Ac D A Wc Thk f k f Db. Db h f h f Ac D A b h Ocb., h W h f, b D. c h A D c f A Ab Ac D A T Rx A Ib T Ac D A Lb G R h h hc b. O, f h k, k k h b ffc, f b, f hch k k ch
necessita d'interrogare il cielo
 gigi nei necessia d'inegae i cie cic pe sax span s inuie a dispiegaa fma dea uce < affeandi ves i cen dea uce isnane " sienzi dei padi sie veic dei' anima 5 J i f H 5 f AL J) i ) L '3 J J "' U J J ö'
gigi nei necessia d'inegae i cie cic pe sax span s inuie a dispiegaa fma dea uce < affeandi ves i cen dea uce isnane " sienzi dei padi sie veic dei' anima 5 J i f H 5 f AL J) i ) L '3 J J "' U J J ö'
EUROPEAN JOURNAL OF PHYSICAL AND REHABILITATION MEDICINE. An abridged version of the Cochrane review of exercise therapy for chronic fatigue syndrome
 EUROPEAN JOURNAL OF PHYSICAL AND REHABILITATION MEDICINE EDIZIONI MINERVA MEDICA T PDF. A. T j. A C L LARUN J ODGAARD-JENSEN J R PRICE Kj G BRURBERG E J P R M S 6 [E ] EUROPEAN JOURNAL OF PHYSICAL AND
EUROPEAN JOURNAL OF PHYSICAL AND REHABILITATION MEDICINE EDIZIONI MINERVA MEDICA T PDF. A. T j. A C L LARUN J ODGAARD-JENSEN J R PRICE Kj G BRURBERG E J P R M S 6 [E ] EUROPEAN JOURNAL OF PHYSICAL AND
u4 ~ ~~-Jk~ ~~.~h4t -Jk. ~ ~ 1:;tK.t ~..~~ ~.~~ ~~!t-o 0j~~ ~ wd/l ~ C0n~ 1~J4.(kvl ~. NAME:~ ~.. PERIOD: ~~~.&m~.. OVER CH 13 HOMEWORK.
 NAME:~ ~.. PERIOD: CH 13 HOMEWORK. a. Read CH 13 b. Review your notes. c. Answer the following: 1. Characterize a system at equilibrium with respect to each of the following: (a) The rates of the forward
NAME:~ ~.. PERIOD: CH 13 HOMEWORK. a. Read CH 13 b. Review your notes. c. Answer the following: 1. Characterize a system at equilibrium with respect to each of the following: (a) The rates of the forward
principles of f ta f a rt.
 DD H L L H PDG D BB PBLH L 20 D PP 32 C B P L s BDWY s BGG M W C WDM DLL P M DC GL CP F BW Y BBY PMB 5 855 C WHL X 6 s L Y F H 5 L & 5 zzzl s s zz z s s» z sk??» szz zz s L ~Lk Bz ZzY Z? ~ s s sgss s z«f
DD H L L H PDG D BB PBLH L 20 D PP 32 C B P L s BDWY s BGG M W C WDM DLL P M DC GL CP F BW Y BBY PMB 5 855 C WHL X 6 s L Y F H 5 L & 5 zzzl s s zz z s s» z sk??» szz zz s L ~Lk Bz ZzY Z? ~ s s sgss s z«f
Optimal Numerical Differentiation Using Three Function Evaluations
 mathematics of computation volume 37, number 155 JULY 1981 Optimal Numerical Differentiation Using Three Function Evaluations By J. Marshall Ash and Roger L. Jones Abstract. Approximation of f'(x) by a
mathematics of computation volume 37, number 155 JULY 1981 Optimal Numerical Differentiation Using Three Function Evaluations By J. Marshall Ash and Roger L. Jones Abstract. Approximation of f'(x) by a
NAME: SECOND EXAMINATION
 1 Chemistry 64 Winter 1994 NAME: SECOND EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
1 Chemistry 64 Winter 1994 NAME: SECOND EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Housing Market Monitor
 M O O D Y È S A N A L Y T I C S H o u s i n g M a r k e t M o n i t o r I N C O R P O R A T I N G D A T A A S O F N O V E M B E R İ Ī Ĭ Ĭ E x e c u t i v e S u m m a r y E x e c u t i v e S u m m a r y
M O O D Y È S A N A L Y T I C S H o u s i n g M a r k e t M o n i t o r I N C O R P O R A T I N G D A T A A S O F N O V E M B E R İ Ī Ĭ Ĭ E x e c u t i v e S u m m a r y E x e c u t i v e S u m m a r y
8. Relax and do well.
 CHEM 1225 Exam III John III. Gelder April 8, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table and
CHEM 1225 Exam III John III. Gelder April 8, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table and
'NOTAS"CRITICAS PARA UNA TEDRIA DE M BUROCRACIA ESTATAL * Oscar Oszlak
 OVí "^Ox^ OqAÍ"^ Dcument SD-11 \ 'NOTAS"CRTCAS PARA UNA TEDRA DE M BUROCRACA ESTATAL * Oscr Oszlk * El presente dcument que se reprduce pr us exclusv de ls prtcpntes de curss de Prrms de Cpctcón, se h
OVí "^Ox^ OqAÍ"^ Dcument SD-11 \ 'NOTAS"CRTCAS PARA UNA TEDRA DE M BUROCRACA ESTATAL * Oscr Oszlk * El presente dcument que se reprduce pr us exclusv de ls prtcpntes de curss de Prrms de Cpctcón, se h
SECURITIES AND EXCHANGE COMMISSION FORM 10-D. Filing Date: Period of Report: SEC Accession No
 SECURITIES AND EXCHANGE COMMISSION FORM 10-D Periodic distribution reports by Asset-Backed issuers pursuant to Rule 13a-17 or 15d-17 Filing Date: 2007-12-06 Period of Report: 2007-11-26 SEC Accession No.
SECURITIES AND EXCHANGE COMMISSION FORM 10-D Periodic distribution reports by Asset-Backed issuers pursuant to Rule 13a-17 or 15d-17 Filing Date: 2007-12-06 Period of Report: 2007-11-26 SEC Accession No.
Chapter 12 The Atom & Periodic Table- part 2
 Chapter 12 The Atom & Periodic Table- part 2 Electrons found outside the nucleus; negatively charged Protons found in the nucleus; positive charge equal in magnitude to the electron s negative charge Neutrons
Chapter 12 The Atom & Periodic Table- part 2 Electrons found outside the nucleus; negatively charged Protons found in the nucleus; positive charge equal in magnitude to the electron s negative charge Neutrons
Secondary Support Pack. be introduced to some of the different elements within the periodic table;
 Secondary Support Pack INTRODUCTION The periodic table of the elements is central to chemistry as we know it today and the study of it is a key part of every student s chemical education. By playing the
Secondary Support Pack INTRODUCTION The periodic table of the elements is central to chemistry as we know it today and the study of it is a key part of every student s chemical education. By playing the
Problems and Solutions
 Problems and Solutions Problems and Solutions. The aim of this section is to encourage readers to participate in the intriguing process of problem solving in Mathematics. This section publishes problems
Problems and Solutions Problems and Solutions. The aim of this section is to encourage readers to participate in the intriguing process of problem solving in Mathematics. This section publishes problems
0 t b r 6, 20 t l nf r nt f th l t th t v t f th th lv, ntr t n t th l l l nd d p rt nt th t f ttr t n th p nt t th r f l nd d tr b t n. R v n n th r
 n r t d n 20 22 0: T P bl D n, l d t z d http:.h th tr t. r pd l 0 t b r 6, 20 t l nf r nt f th l t th t v t f th th lv, ntr t n t th l l l nd d p rt nt th t f ttr t n th p nt t th r f l nd d tr b t n.
n r t d n 20 22 0: T P bl D n, l d t z d http:.h th tr t. r pd l 0 t b r 6, 20 t l nf r nt f th l t th t v t f th th lv, ntr t n t th l l l nd d p rt nt th t f ttr t n th p nt t th r f l nd d tr b t n.
Atoms and the Periodic Table
 Atoms and the Periodic Table Parts of the Atom Proton Found in the nucleus Number of protons defines the element Charge +1, mass 1 Parts of the Atom Neutron Found in the nucleus Stabilizes the nucleus
Atoms and the Periodic Table Parts of the Atom Proton Found in the nucleus Number of protons defines the element Charge +1, mass 1 Parts of the Atom Neutron Found in the nucleus Stabilizes the nucleus
7. Relax and do well.
 CHEM 1215 Exam II John II. Gelder October 7, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 5 different pages. The last page includes a periodic table and a solubility
CHEM 1215 Exam II John II. Gelder October 7, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 5 different pages. The last page includes a periodic table and a solubility
UNIT IV PPT #3 Ka and Kb KEY.notebook. November 23, WHAT IS Ka? UNIT IV. CALCULATIONS USING Ka. WHAT IS Ka? Nov 10 9:42 PM.
 WHAT IS Ka? Recall: Find ph of 0.100 M HCl. But What is ph of 0.10 M HF? UNIT IV Ka and Kb CALCULATIONS USING Ka I. [H 3O + ] (or ph) from Ka Ex. Find the [H 3O + ] in 0.10 M HF. WHAT IS Ka? Look at equilibrium
WHAT IS Ka? Recall: Find ph of 0.100 M HCl. But What is ph of 0.10 M HF? UNIT IV Ka and Kb CALCULATIONS USING Ka I. [H 3O + ] (or ph) from Ka Ex. Find the [H 3O + ] in 0.10 M HF. WHAT IS Ka? Look at equilibrium
Upper plate. Electrode. Liquid crystal material. Under plate ) + ( )
 ! ) 6 $-.%A";22".%$$ #9! 9 7/B )./, )))())(!!() 7?&!"9 4 $ B ) B -7 ) 1 4:S?!R:bc4'+TIc6,/ T# K ( 4 b c 34K^4(+ 4.:. B4.OBII* ;% %?!B! O B R B(: 1% 4 M9 DF9 4 (: 1.: 23 1.467! )B!! +)!B!! 14--.1(1.1-$3-
! ) 6 $-.%A";22".%$$ #9! 9 7/B )./, )))())(!!() 7?&!"9 4 $ B ) B -7 ) 1 4:S?!R:bc4'+TIc6,/ T# K ( 4 b c 34K^4(+ 4.:. B4.OBII* ;% %?!B! O B R B(: 1% 4 M9 DF9 4 (: 1.: 23 1.467! )B!! +)!B!! 14--.1(1.1-$3-
THE TRANSLATION PLANES OF ORDER 49 AND THEIR AUTOMORPHISM GROUPS
 MATHEMATICS OF COMPUTATION Volume 67, Number 223, July 1998, Pages 1207 1224 S 0025-5718(98)00961-2 THE TRANSLATION PLANES OF ORDER 49 AND THEIR AUTOMORPHISM GROUPS C. CHARNES AND U. DEMPWOLFF Abstract.
MATHEMATICS OF COMPUTATION Volume 67, Number 223, July 1998, Pages 1207 1224 S 0025-5718(98)00961-2 THE TRANSLATION PLANES OF ORDER 49 AND THEIR AUTOMORPHISM GROUPS C. CHARNES AND U. DEMPWOLFF Abstract.
CLEARLY SHOW ALL WORK AND REASONING.
 Name (print clearly) Seat number Quiz 4 (100 points) Chemistry 1303.002 October 17, 2018 1. (25points) Using the information below, find the enthalpy of formation ( H f ) for gaseous hydrogen chloride.
Name (print clearly) Seat number Quiz 4 (100 points) Chemistry 1303.002 October 17, 2018 1. (25points) Using the information below, find the enthalpy of formation ( H f ) for gaseous hydrogen chloride.
Chem Exam 1. September 26, Dr. Susan E. Bates. Name 9:00 OR 10:00
 Chem 1711 Exam 1 September 26, 2013 Dr. Susan E. Bates Name 9:00 OR 10:00 N A = 6.022 x 10 23 mol 1 I A II A III B IV B V B VI B VII B VIII I B II B III A IV A V A VI A VII A inert gases 1 H 1.008 3 Li
Chem 1711 Exam 1 September 26, 2013 Dr. Susan E. Bates Name 9:00 OR 10:00 N A = 6.022 x 10 23 mol 1 I A II A III B IV B V B VI B VII B VIII I B II B III A IV A V A VI A VII A inert gases 1 H 1.008 3 Li
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
8. Relax and do well.
 CHEM 1225 Exam I John I. Gelder February 4, 1999 Name KEY TA's Name Lab Section Please sign your name below to give permission to post your course scores on homework, laboratories and exams. If you do
CHEM 1225 Exam I John I. Gelder February 4, 1999 Name KEY TA's Name Lab Section Please sign your name below to give permission to post your course scores on homework, laboratories and exams. If you do
Physical Chemistry I CHEM 4641 Final Exam 13 questions, 30 points
 Physical Chemistry I CHEM 4641 Final Exam 13 questions, 30 points Name: KEY Gas constant: R = 8.314 J mol -1 K -1 = 0.008314 kj mol -1 K -1. Boltzmann constant k = 1.381 10-23 J/K = 0.6950 cm -1 /K h =
Physical Chemistry I CHEM 4641 Final Exam 13 questions, 30 points Name: KEY Gas constant: R = 8.314 J mol -1 K -1 = 0.008314 kj mol -1 K -1. Boltzmann constant k = 1.381 10-23 J/K = 0.6950 cm -1 /K h =
Solutions and Ions. Pure Substances
 Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
