MEDICINAL CHEMISTRY Homework 2 - ANSWERS
|
|
|
- Oswin Hicks
- 6 years ago
- Views:
Transcription
1 MEDICIAL CEMISTY omework 2 - ASWES 1. eceptor binding curves. Make a plot of %drug-bound versus -log[drug] for a drug whose K D is 5 x 10-6 M. Make a plot of %drug-bound vs [drug] for this same system. To do this calculate %drug-bound at drug concentrations of 5 x 10-8 M, 5 x 10-7 M, 5 x 10-6 M, 5 x 10-5 M, and 5 x 10-4 M. To calculate % drug bound, use the equation that we have used previously (shown below). Are there advantages that you see for the plot using -log[drug]? 1/( K eq D free 1) = T free / T total ere are the plots: % Bound (esponse) % Bound (esponse) e0 1.00e e e-4 Concentration 4.00e e Log[Drug] 7 8 Advantages of the Log plot: Midpoint of the plot is K D for drug. Easily identified by inspection. Data for several different drugs could be placed on one graph and it would be easy to tell which drug binds better. 2. In Panel A (below) a plot of the % effect (response) versus the log of the natural substrate concentration for this receptor is given. In Panel B we have a similar plot, except we see the response produced by drug binding at this receptor. What is the general term that is used for the natural ligand of a receptor? There is a generic name or term given to a compound which elicits a response such as this. What is it? ow well does the drug bind to the receptor compared to the natural substrate? Be quantitative (what is the K D for the drug and the natural substrate?)
2 atural ligand for receptor is call the "autocoid" "Agonist" gives same type of response as the autocoid. In this example it appears that drug and autocoid have identical K D values. The K D value looks like about 1 x 10-8 M (midpoint of the curve). 3. In Panel C we see the response of the same receptor when a second drug, Drug 2, is given by itself. In Panel D we see the results of a set of experiments with Drug 2 and the natural substrate for this receptor. In the first experiment drug 2 is absent and we see the dose response from the substrate. In the next experiment Drug 2 is given at one dose along with variable doses of the substrate so as to produce the dose response curve. Similar experiments are done to produce the other curves at 2 and 3 times the initial Drug 2 dose. A generic name or term is given to a compound such as Drug 2 which produces this type of effect. What is it? Drugs that give this type of response are called "competitive antagonists". 4. Explain the mechanism of action of Drug 2 and how it produces the phenomena exhibited in panel D above. ormally, the autocoid binds to the receptor and activates the receptor. In this case the drug binds to the same site that the autocoid normally binds and blocks the ability of the autocoid to bind and activate the receptor. 5. In the development of cimetidine, the early bioassays employed histamine at concentrations sufficient to cause 50% activation of the 2 receptor. This assay was not capable of detecting that α-guanyl histamine was binding to the receptor as a partial agonist. Why not? Use mock data plots to illustrate your answer. They changed the assay conditions to use a histamine concentration that causes 80% activation of the receptor. This allowed them to see that α-guanyl histamine is a partial agonist. In fact, this assay highlights the fact that a partial agonist can be viewed as a partial antagonist. Again, use mock data plots to illustrate your answer. At istidine 50% concentrations the presence of a partial agonist that activates the receptor to ~50% will be invisible. At istidine 80% concentrations this same partial agonist will show up as a weak antagonist. This reveals that a partial agonist also can be viewed as a partial antagonist. Which property of the molecule that is observed depends upon the way that the assay is conducted.
3 Partial agonist alone Partial agonist added in the presence of "histidine-50%" Partial agonist added in the presence of "histidine-80%" % response 6. In the development of the drug Cimetidine, which is used to decrease acid secretion into the stomach, and prevent the discomfort of heartburn, acid reflux and pre-ulcerative conditions, the molecules tested had an imidazole group at one end of the molecule, and, among others, one of the three groups shown below at the other end of the molecule. S C C 3 C 3 C a. What was the rationale for including the imidazole ring? The natural ligand, histidine,contains an imidazole ring. In this case the autocoid served as the lead compound in drug discovery. b. Of the structures 1-3 shown above which is the most basic and why? In answering this question use words, resonance forms, etc. in your explanation. Which structure will be more easily protonated (at any given p)? The most basic is 2. In each case, you can draw the same resonance structure. So, what determines the basicity of the molecules is the electronegativity of. The more electronegative will be less basic. = is the least electronegative, thus 2 is the most basic. =1 or 2 x C 3 x C 3 x C 3 c. Which of the structures 1-3 was used in Cimetidine and what was the rationale for its inclusion? Structure 2 was used in cimetidine. ammett plots identified a QSA suggesting that this functional group might be effective.
4 7. What are two key properties of receptors that allow small doses of an appropriate drug to elicit a significant biological response. (I discussed these key features in our first lecture on receptors). eceptors SELECTIVELYbind an autocoid and AMPLIFY this signal. So, a single molecule of drug can block signal amplification (or activate a receptor resulting in an amplified signal). Thus, small amount of drug gives large effect due to amplification. 8. Consider the following situation. The natural ligand for a receptor has a K B of 1 x 10 6 M -1. A competitive antagonist for this receptor has a K B of 1 x 10 9 M -1. Biological studies show that activation of the receptor occurs at natural ligand concentration of about 1 x 10-6 M (1 µm). Under these conditions, what concentration of drug will be required to place 90% of the receptor in the drug-bound form (i.e. 90% of the receptor is "blocked")? (We derived the necessary equations in class). We derived the following relationship in lecture [ec Drug]/[ec Autocoid] = (K Bdrug [Drug])/(K Bautocoid [Autocoid]) 90/10 = (1 x 10 9 M -1 [])/(1 x 10 6 M -1 [1 x 10-6 M]) =9 x 10-9 M (9 nm drug is required) 9. Does cimetidine adhere to Lipinki s rules? MW = 252.3; -bond donors = 3; -bond acceptors = 7; Log P = 0.4, free-rotating bonds = 7. one of these parameters violate Lipinskiʼs rules. Thus, the rules predict that the drug will be OK with respect to bioavailability. 10. Given information about the IC 50 of the drug and the concentrations and binding properties of the autocoid it is possible to calculate the K i for the drug. From the Ki is possible to calulcate the ligand efficiency. The equation relating K i to IC 50 can be written: Plugging in the numbers given in the problem (for K M insert the K D of histamine for binding at the 2 receptor) we can calculate that K i = 1.5 x 10-8 M. Using the equation DG = TlnK eq where, at 25 C, T = 0.59 kcal/mol, we can calculate that the free energy of cimetidine binding to the 2 receptor is about 10.6 kcal/mol (make sure you convert K D to K B for use in this calculation). Ligand efficiency, Δg = 10.6 kcal/mol / 17 atoms = 0.62 kcal/mol/atom.
5 11. Why did addition of a -C 3 and a -OC 3 group make the pyridine ring in omeprazole more basic? Why does this structural feature help omeprazole accumulate inside acidic compartments of the stomach-acid producing parietal cells? (int: draw the rxn in question the protonation of the pyridine ring. Then use resonance structures to consider how substituents on the pyridine ring affect the stability of the compound on each side of the equation that is, how substituents alter the equilibrium of the reaction). The unprotonated pyridine is lipophilic and easily crosses membranes (into acidic compartments). Once inside an acid compartment the pyridine ring is protonated. This makes the molecule charged and very hydrophilic, thus unable to cross membranes and escape the acidic compartment. Any electron-donating substitutent will increase the basicity of the pyridine ring. Look at a table of ammett substituent constants (sigma, σ) to find other electron donating substituents. 12. Suggest two other substituents that would have a similar effect on the basicity of the pyridine ring found in omeprazole. Try - 2 and -C 2 C 3 ammett tables tell us that these are electron-donating. 13. Imagine that you have been placed in charge of a drug-development team with the goal of developing a receptor antagonist. There are no known lead compounds. What would you use as the lead compound? (ote: this is a general question you do not need to know anything about the structure of the natural ligand for the target receptor.). Could you use a similar strategy in the design of a reversible enzyme inhibitor? Use the structure of the natural ligand, the autocoid, as a starting point ("lead") for drug discovery.
1. (18) Multiple choice questions. Please place your answer on the line preceding each question.
 CEM 5720 ame KEY Exam 2 ctober 21, 2015 Read all questions carefully and attempt those questions you are sure of first. Remember to proof your work; art work carries as much importance as written responses.
CEM 5720 ame KEY Exam 2 ctober 21, 2015 Read all questions carefully and attempt those questions you are sure of first. Remember to proof your work; art work carries as much importance as written responses.
CHEM 4170 Problem Set #1
 CHEM 4170 Problem Set #1 0. Work problems 1-7 at the end of Chapter ne and problems 1, 3, 4, 5, 8, 10, 12, 17, 18, 19, 22, 24, and 25 at the end of Chapter Two and problem 1 at the end of Chapter Three
CHEM 4170 Problem Set #1 0. Work problems 1-7 at the end of Chapter ne and problems 1, 3, 4, 5, 8, 10, 12, 17, 18, 19, 22, 24, and 25 at the end of Chapter Two and problem 1 at the end of Chapter Three
Structural biology and drug design: An overview
 Structural biology and drug design: An overview livier Taboureau Assitant professor Chemoinformatics group-cbs-dtu otab@cbs.dtu.dk Drug discovery Drug and drug design A drug is a key molecule involved
Structural biology and drug design: An overview livier Taboureau Assitant professor Chemoinformatics group-cbs-dtu otab@cbs.dtu.dk Drug discovery Drug and drug design A drug is a key molecule involved
FST 123 Problem Set 3 Spring, V o at [S] 0 = 10 mm (mm/min)
![FST 123 Problem Set 3 Spring, V o at [S] 0 = 10 mm (mm/min) FST 123 Problem Set 3 Spring, V o at [S] 0 = 10 mm (mm/min)](/thumbs/86/93227301.jpg) FST 23 Problem Set 3 Spring, 2009 Name. A student needed to know the activity versus ph profile for an enzyme that he was going to use in a later application. He collected the appropriate data and determined
FST 23 Problem Set 3 Spring, 2009 Name. A student needed to know the activity versus ph profile for an enzyme that he was going to use in a later application. He collected the appropriate data and determined
Electronic Structure. Hammett Correlations. Linear Free Energy Relationships. Quantitative Structure Activity Relationships (QSAR)
 Electronic Structure ammett Correlations Quantitative Structure Activity Relationships (QSAR) Quantitative Structure Activity Relationships (QSAR) We have already seen that by changing groups in a drug
Electronic Structure ammett Correlations Quantitative Structure Activity Relationships (QSAR) Quantitative Structure Activity Relationships (QSAR) We have already seen that by changing groups in a drug
MOLECULAR DRUG TARGETS
 MOLECULAR DRUG TARGETS LEARNING OUTCOMES At the end of this session student shall be able to: List different types of druggable targets Describe forces involved in drug-receptor interactions Describe theories
MOLECULAR DRUG TARGETS LEARNING OUTCOMES At the end of this session student shall be able to: List different types of druggable targets Describe forces involved in drug-receptor interactions Describe theories
Chemistry 14C Winter 2017 Final Exam Part A Page 1
 Chemistry 14C Winter 2017 Final Exam Part A Page 1 Please use the backs of the exam pages for scratch space. Please do not use the exam margins for this purpose. The active site of an enzyme is a cleft
Chemistry 14C Winter 2017 Final Exam Part A Page 1 Please use the backs of the exam pages for scratch space. Please do not use the exam margins for this purpose. The active site of an enzyme is a cleft
SAM Teacher s Guide Protein Partnering and Function
 SAM Teacher s Guide Protein Partnering and Function Overview Students explore protein molecules physical and chemical characteristics and learn that these unique characteristics enable other molecules
SAM Teacher s Guide Protein Partnering and Function Overview Students explore protein molecules physical and chemical characteristics and learn that these unique characteristics enable other molecules
Notes of Dr. Anil Mishra at 1
 Introduction Quantitative Structure-Activity Relationships QSPR Quantitative Structure-Property Relationships What is? is a mathematical relationship between a biological activity of a molecular system
Introduction Quantitative Structure-Activity Relationships QSPR Quantitative Structure-Property Relationships What is? is a mathematical relationship between a biological activity of a molecular system
Statistical concepts in QSAR.
 Statistical concepts in QSAR. Computational chemistry represents molecular structures as a numerical models and simulates their behavior with the equations of quantum and classical physics. Available programs
Statistical concepts in QSAR. Computational chemistry represents molecular structures as a numerical models and simulates their behavior with the equations of quantum and classical physics. Available programs
Recommended Reading: 23, 29 (3rd edition); 22, 29 (4th edition) Ch 102 Problem Set 7 Due: Thursday, June 1 Before Class. Problem 1 (1 points) Part A
 Recommended Reading: 23, 29 (3rd edition); 22, 29 (4th edition) Ch 102 Problem Set 7 Due: Thursday, June 1 Before Class Problem 1 (1 points) Part A Kinetics experiments studying the above reaction determined
Recommended Reading: 23, 29 (3rd edition); 22, 29 (4th edition) Ch 102 Problem Set 7 Due: Thursday, June 1 Before Class Problem 1 (1 points) Part A Kinetics experiments studying the above reaction determined
Acid-Base Properties
 TAMMAR H. Ali Lecture 1 Course No. 326 Faculty of Pharmacy University Of Al-Muthanna 1 Physicochemical Principles of Drug Action Physicochemical Principles of Drug Action To design better drugs: Molecular
TAMMAR H. Ali Lecture 1 Course No. 326 Faculty of Pharmacy University Of Al-Muthanna 1 Physicochemical Principles of Drug Action Physicochemical Principles of Drug Action To design better drugs: Molecular
A primer on pharmacology pharmacodynamics
 A primer on pharmacology pharmacodynamics Drug binding & effect Universidade do Algarve Faro 2017 by Ferdi Engels, Ph.D. 1 Pharmacodynamics Relation with pharmacokinetics? dosage plasma concentration site
A primer on pharmacology pharmacodynamics Drug binding & effect Universidade do Algarve Faro 2017 by Ferdi Engels, Ph.D. 1 Pharmacodynamics Relation with pharmacokinetics? dosage plasma concentration site
Principles of Drug Design
 Advanced Medicinal Chemistry II Principles of Drug Design Tentative Course Outline Instructors: Longqin Hu and John Kerrigan Direct questions and enquiries to the Course Coordinator: Longqin Hu I. Introduction
Advanced Medicinal Chemistry II Principles of Drug Design Tentative Course Outline Instructors: Longqin Hu and John Kerrigan Direct questions and enquiries to the Course Coordinator: Longqin Hu I. Introduction
Name. Chapter 4 covers acid-base chemistry. That should help you get going.
 Name Chapter 4 covers acid-base chemistry. That should help you get going. 1 Use curved arrows to illustrate the transfer of a proton (i.e. an + ) from benzoic acid to phenoxide, and draw the products.
Name Chapter 4 covers acid-base chemistry. That should help you get going. 1 Use curved arrows to illustrate the transfer of a proton (i.e. an + ) from benzoic acid to phenoxide, and draw the products.
CHAPTER 3. Vibrational Characteristics of PTP-1B Inhibitors
 CHAPTER 3 Vibrational Characteristics of PTP-1B Inhibitors 3.1 Preface Theoretical Frequency calculations were performed on the molecules chosen in this study. Initially all the geometries were optimized
CHAPTER 3 Vibrational Characteristics of PTP-1B Inhibitors 3.1 Preface Theoretical Frequency calculations were performed on the molecules chosen in this study. Initially all the geometries were optimized
The wise does at once what the fool does at last.
 hem 201 Midterm all, 2018 Beauchamp ame Problems Points redit 1. unctional Group omenclature (1 large structure) 30 2. esonance, ormal harge, Arrows 18 3. yclohexane onformations, ewman Projections 30
hem 201 Midterm all, 2018 Beauchamp ame Problems Points redit 1. unctional Group omenclature (1 large structure) 30 2. esonance, ormal harge, Arrows 18 3. yclohexane onformations, ewman Projections 30
Plan. Day 2: Exercise on MHC molecules.
 Plan Day 1: What is Chemoinformatics and Drug Design? Methods and Algorithms used in Chemoinformatics including SVM. Cross validation and sequence encoding Example and exercise with herg potassium channel:
Plan Day 1: What is Chemoinformatics and Drug Design? Methods and Algorithms used in Chemoinformatics including SVM. Cross validation and sequence encoding Example and exercise with herg potassium channel:
CHEM 109A Organic Chemistry
 CHEM 109A Organic Chemistry https://labs.chem.ucsb.edu/zakarian/armen/courses.html Chapter 2 Acids and Bases Central to Understanding Organic Chemistry Draw the conjugate acid of each of the following:
CHEM 109A Organic Chemistry https://labs.chem.ucsb.edu/zakarian/armen/courses.html Chapter 2 Acids and Bases Central to Understanding Organic Chemistry Draw the conjugate acid of each of the following:
Introduction to Chemoinformatics and Drug Discovery
 Introduction to Chemoinformatics and Drug Discovery Irene Kouskoumvekaki Associate Professor February 15 th, 2013 The Chemical Space There are atoms and space. Everything else is opinion. Democritus (ca.
Introduction to Chemoinformatics and Drug Discovery Irene Kouskoumvekaki Associate Professor February 15 th, 2013 The Chemical Space There are atoms and space. Everything else is opinion. Democritus (ca.
Patrick, An Introduction to Medicinal Chemistry 5e Chapter 7 Enzymes as drug targets. 1) The structures of isoleucine and valine are as follows.
 Answers to end-of-chapter questions 1) The structures of isoleucine and valine are as follows. 2 C 2 2 C 2 3 C C 3 3 C C 3 Isoleucine Valine Isoleucine has a larger side chain than valine, and so there
Answers to end-of-chapter questions 1) The structures of isoleucine and valine are as follows. 2 C 2 2 C 2 3 C C 3 3 C C 3 Isoleucine Valine Isoleucine has a larger side chain than valine, and so there
5. Stereochemical Analysis. 7. Dipole Moments and Inductive versus Resonance Effects. 8. Types of isomers from a given formula. 9. Physical Properties
 hem 201 Sample Midterm Beauchamp ame Problems Points redit 1. Functional Group omenclature (1 large structure) 2. Lewis Structures, Resonance, Formal harge 3. yclohexane onformations, 2 substituents, ewman
hem 201 Sample Midterm Beauchamp ame Problems Points redit 1. Functional Group omenclature (1 large structure) 2. Lewis Structures, Resonance, Formal harge 3. yclohexane onformations, 2 substituents, ewman
Lecture 14 (10/18/17) Lecture 14 (10/18/17)
 Lecture 14 (10/18/17) Reading: Ch6; 190-191, 194-195, 197-198 Problems: Ch6 (text); 7, 24 Ch6 (study guide-facts); 4, 13 NEXT Reading: Ch6; 198-203 Ch6; Box 6-1 Problems: Ch6 (text); 8, 9, 10, 11, 12,
Lecture 14 (10/18/17) Reading: Ch6; 190-191, 194-195, 197-198 Problems: Ch6 (text); 7, 24 Ch6 (study guide-facts); 4, 13 NEXT Reading: Ch6; 198-203 Ch6; Box 6-1 Problems: Ch6 (text); 8, 9, 10, 11, 12,
Energy and Cells. Appendix 1. The two primary energy transformations in plants are photosynthesis and respiration.
 Energy and Cells Appendix 1 Energy transformations play a key role in all physical and chemical processes that occur in plants. Energy by itself is insufficient to drive plant growth and development. Enzymes
Energy and Cells Appendix 1 Energy transformations play a key role in all physical and chemical processes that occur in plants. Energy by itself is insufficient to drive plant growth and development. Enzymes
Welcome to Week 5. Chapter 9 - Binding, Structure, and Diversity. 9.1 Intermolecular Forces. Starting week five video. Introduction to Chapter 9
 Welcome to Week 5 Starting week five video Please watch the online video (49 seconds). Chapter 9 - Binding, Structure, and Diversity Introduction to Chapter 9 Chapter 9 contains six subsections. Intermolecular
Welcome to Week 5 Starting week five video Please watch the online video (49 seconds). Chapter 9 - Binding, Structure, and Diversity Introduction to Chapter 9 Chapter 9 contains six subsections. Intermolecular
EMPIRICAL VS. RATIONAL METHODS OF DISCOVERING NEW DRUGS
 EMPIRICAL VS. RATIONAL METHODS OF DISCOVERING NEW DRUGS PETER GUND Pharmacopeia Inc., CN 5350 Princeton, NJ 08543, USA pgund@pharmacop.com Empirical and theoretical approaches to drug discovery have often
EMPIRICAL VS. RATIONAL METHODS OF DISCOVERING NEW DRUGS PETER GUND Pharmacopeia Inc., CN 5350 Princeton, NJ 08543, USA pgund@pharmacop.com Empirical and theoretical approaches to drug discovery have often
Acid-Base Chemistry. Chapter Brønsted Acid-Base Chemistry R P
 Chapter 5 Acid-Base Chemistry 5.1 Brønsted Acid-Base Chemistry R P The equilibrium constant (K eq ) gives quick insight into whether the reactant or product is more stable, and the extent to which the
Chapter 5 Acid-Base Chemistry 5.1 Brønsted Acid-Base Chemistry R P The equilibrium constant (K eq ) gives quick insight into whether the reactant or product is more stable, and the extent to which the
1. methyl 2. methylene 3. methine 4. primary 5. secondary 6. tertiary 7. quarternary 8. isopropyl
 hem 201 Sample Midterm Beauchamp Exams are designed so that no one question will make or break you. The best strategy is to work steadily, starting with those problems you understand best. Make sure you
hem 201 Sample Midterm Beauchamp Exams are designed so that no one question will make or break you. The best strategy is to work steadily, starting with those problems you understand best. Make sure you
Periodic Table. 8/3/2006 MEDC 501 Fall
 Periodic Table 8/3/2006 MEDC 501 Fall 2006 1 rbitals Shapes of rbitals s - orbital p -orbital 8/3/2006 MEDC 501 Fall 2006 2 Ionic Bond - acl Electronic Structure 11 a :: 1s 2 2s 2 2p x2 2p y2 2p z2 3s
Periodic Table 8/3/2006 MEDC 501 Fall 2006 1 rbitals Shapes of rbitals s - orbital p -orbital 8/3/2006 MEDC 501 Fall 2006 2 Ionic Bond - acl Electronic Structure 11 a :: 1s 2 2s 2 2p x2 2p y2 2p z2 3s
Prof. Jason D. Kahn Your Signature: Exams written in pencil or erasable ink will not be re-graded under any circumstances.
 1 Biochemistry 461, Section I February 27, 1997 Exam #1 Prof. Jason D. Kahn Your Printed ame: Your SS#: Your Signature: You have 80 minutes for this exam. Exams written in pencil or erasable ink will not
1 Biochemistry 461, Section I February 27, 1997 Exam #1 Prof. Jason D. Kahn Your Printed ame: Your SS#: Your Signature: You have 80 minutes for this exam. Exams written in pencil or erasable ink will not
Chapter 3 Acids and Bases. The Curved-Arrow Notation
 Organic Chemistry, 5th ed. Marc Loudon Chapter 3 Acids and Bases. The Curved-Arrow Notation Eric J. Kantorows ki California Polytechnic State University San Luis Obispo, CA Chapter 3 Overview 3.1 Lewis
Organic Chemistry, 5th ed. Marc Loudon Chapter 3 Acids and Bases. The Curved-Arrow Notation Eric J. Kantorows ki California Polytechnic State University San Luis Obispo, CA Chapter 3 Overview 3.1 Lewis
Full file at Chapter 2 Water: The Solvent for Biochemical Reactions
 Chapter 2 Water: The Solvent for Biochemical Reactions SUMMARY Section 2.1 Summary Water is a polar molecule, with a partial negative charge on the oxygen and partial positive charges on the hydrogens.
Chapter 2 Water: The Solvent for Biochemical Reactions SUMMARY Section 2.1 Summary Water is a polar molecule, with a partial negative charge on the oxygen and partial positive charges on the hydrogens.
Organic Chem Chapter 3: Acids and Bases
 Organic Chem Chapter 3: Acids and Bases Title and Highlight Right side: NOTES! Topic: EQ: Date NOTES: Write out the notes from my website. Use different types of note-taking methods to help you recall
Organic Chem Chapter 3: Acids and Bases Title and Highlight Right side: NOTES! Topic: EQ: Date NOTES: Write out the notes from my website. Use different types of note-taking methods to help you recall
2. Which of the following are nucleophiles and which are electrophiles?
 Life Sciences 1a ractice roblems 7 1. a) ow many intermediates are there in the reaction? b) ow many transition states are there? c) What is the fastest step in the reaction? d) Which is more stable, A
Life Sciences 1a ractice roblems 7 1. a) ow many intermediates are there in the reaction? b) ow many transition states are there? c) What is the fastest step in the reaction? d) Which is more stable, A
An Introduction to Metabolism
 Chapter 8 An Introduction to Metabolism oweroint Lectures for Biology, Seventh Edition Neil Campbell and Jane Reece Concept 8.1: An organism s metabolism transforms matter and energy, subject to the laws
Chapter 8 An Introduction to Metabolism oweroint Lectures for Biology, Seventh Edition Neil Campbell and Jane Reece Concept 8.1: An organism s metabolism transforms matter and energy, subject to the laws
Concept review: Binding equilibria
 Concept review: Binding equilibria 1 Binding equilibria and association/dissociation constants 2 The binding of a protein to a ligand at equilibrium can be written as: P + L PL And so the equilibrium constant
Concept review: Binding equilibria 1 Binding equilibria and association/dissociation constants 2 The binding of a protein to a ligand at equilibrium can be written as: P + L PL And so the equilibrium constant
7.012 Problem Set 1. i) What are two main differences between prokaryotic cells and eukaryotic cells?
 ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
PHYSIO-CHEMICAL PROPERTIES OF THE DRUG. It is the interaction of the drug with its environment
 PYSIO-CEMICAL PROPERTIES OF TE DRUG It is the interaction of the drug with its environment Reducing the complexity Biological process in drug action Dissolution of drug in gastrointestinal fluids Absorption
PYSIO-CEMICAL PROPERTIES OF TE DRUG It is the interaction of the drug with its environment Reducing the complexity Biological process in drug action Dissolution of drug in gastrointestinal fluids Absorption
Amines Chemical / Biological / Neurological Activity
 Amines hemical / Biological / eurological Activity Measures of Basicity The basicity of amines may be measured by: 1) K b 2) pk b 3) K a of conjugate acid 4) pk a of conjugate acid Basicity onstant (K
Amines hemical / Biological / eurological Activity Measures of Basicity The basicity of amines may be measured by: 1) K b 2) pk b 3) K a of conjugate acid 4) pk a of conjugate acid Basicity onstant (K
Chemistry 217 Problem Set 1
 hemistry 217 Problem Set 1 Recommended Problems from the Book (1 st ed. in parentheses): 1.1, 1.3-1.9, 1.36-1.42, 1.57-1.62 (1.1-1.8, 1.34-1.40, 1.54-1.58) Supplemental Problems: Lewis structures: Weeks,
hemistry 217 Problem Set 1 Recommended Problems from the Book (1 st ed. in parentheses): 1.1, 1.3-1.9, 1.36-1.42, 1.57-1.62 (1.1-1.8, 1.34-1.40, 1.54-1.58) Supplemental Problems: Lewis structures: Weeks,
Other Cells. Hormones. Viruses. Toxins. Cell. Bacteria
 Other Cells Hormones Viruses Toxins Cell Bacteria ΔH < 0 reaction is exothermic, tells us nothing about the spontaneity of the reaction Δ H > 0 reaction is endothermic, tells us nothing about the spontaneity
Other Cells Hormones Viruses Toxins Cell Bacteria ΔH < 0 reaction is exothermic, tells us nothing about the spontaneity of the reaction Δ H > 0 reaction is endothermic, tells us nothing about the spontaneity
BI/CH421 Biochemistry I Exam 1 09/29/2014
 Part I: Multiple choice for each question, circle the choice that best answers the question. Do not write the letter in the margin to indicate your answer, circle it. 3 points each. 1. For a reaction with
Part I: Multiple choice for each question, circle the choice that best answers the question. Do not write the letter in the margin to indicate your answer, circle it. 3 points each. 1. For a reaction with
Energy, Enzymes, and Metabolism. Energy, Enzymes, and Metabolism. A. Energy and Energy Conversions. A. Energy and Energy Conversions
 Energy, Enzymes, and Metabolism Lecture Series 6 Energy, Enzymes, and Metabolism B. ATP: Transferring Energy in Cells D. Molecular Structure Determines Enzyme Fxn Energy is the capacity to do work (cause
Energy, Enzymes, and Metabolism Lecture Series 6 Energy, Enzymes, and Metabolism B. ATP: Transferring Energy in Cells D. Molecular Structure Determines Enzyme Fxn Energy is the capacity to do work (cause
Identifying Interaction Hot Spots with SuperStar
 Identifying Interaction Hot Spots with SuperStar Version 1.0 November 2017 Table of Contents Identifying Interaction Hot Spots with SuperStar... 2 Case Study... 3 Introduction... 3 Generate SuperStar Maps
Identifying Interaction Hot Spots with SuperStar Version 1.0 November 2017 Table of Contents Identifying Interaction Hot Spots with SuperStar... 2 Case Study... 3 Introduction... 3 Generate SuperStar Maps
Organic Resonance Systems
 rganic esonance Systems ommon, Possible Patterns of esonance There are four common resonance patterns that we encounter, using two donor sites and two acceptor sites. onor electrons () can come from lone
rganic esonance Systems ommon, Possible Patterns of esonance There are four common resonance patterns that we encounter, using two donor sites and two acceptor sites. onor electrons () can come from lone
Biochemistry. Lecture 8 Enzyme Kinetics
 Biochemistry Lecture 8 Enzyme Kinetics Why Enzymes? igher reaction rates Greater reaction specificity Milder reaction conditions Capacity for regulation C - - C N 2 - C N 2 - C - C Chorismate mutase -
Biochemistry Lecture 8 Enzyme Kinetics Why Enzymes? igher reaction rates Greater reaction specificity Milder reaction conditions Capacity for regulation C - - C N 2 - C N 2 - C - C Chorismate mutase -
Midterm Exam III. CHEM 181: Introduction to Chemical Principles November 25, 2014 Answer Key
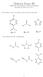 Midterm Exam III EM 181: Introduction to hemical Principles ovember 25, 2014 Answer Key 1. For reference, here are the pk a values for three weak acids: 3 pk a = 7.2 pk a = 4.8 pk a = 15 ow consider this
Midterm Exam III EM 181: Introduction to hemical Principles ovember 25, 2014 Answer Key 1. For reference, here are the pk a values for three weak acids: 3 pk a = 7.2 pk a = 4.8 pk a = 15 ow consider this
An Introduction to Metabolism
 An Introduction to Metabolism I. All of an organism=s chemical reactions taken together is called metabolism. A. Metabolic pathways begin with a specific molecule, which is then altered in a series of
An Introduction to Metabolism I. All of an organism=s chemical reactions taken together is called metabolism. A. Metabolic pathways begin with a specific molecule, which is then altered in a series of
Drug Discovery. Zainab Al Kharusi Office: 33-10
 Drug Discovery Zainab Al Kharusi Office: 33-10 Objectives: To understand the processes of Drug Development To be able to develop a plan for drug discovery Reference: Patrick G L. An Introduction to Medicinal
Drug Discovery Zainab Al Kharusi Office: 33-10 Objectives: To understand the processes of Drug Development To be able to develop a plan for drug discovery Reference: Patrick G L. An Introduction to Medicinal
2015 Ch112 problem set 4 Due: Thursday, Oct. 29 before class Remember to turn in your second paper critique with this problem set!
 Problem 1 (4 points) In 2004, a bimetallic compound exhibiting side-on 2 binding was reported b Chirik and coworkers (ature, 2004, 427, pp. 527-530). The crstal structure of this compound was obtained,
Problem 1 (4 points) In 2004, a bimetallic compound exhibiting side-on 2 binding was reported b Chirik and coworkers (ature, 2004, 427, pp. 527-530). The crstal structure of this compound was obtained,
Homework for Chapter 7 Chem 2310
 omework for Chapter 7 Chem 2310 Name I. Introduction to Reactions 1. Explain why the following fits the definition of a chemical reaction. C 3 Na C 3 Na 2. Using the chemical reaction above, give all compounds
omework for Chapter 7 Chem 2310 Name I. Introduction to Reactions 1. Explain why the following fits the definition of a chemical reaction. C 3 Na C 3 Na 2. Using the chemical reaction above, give all compounds
Introduction into Biochemistry. Dr. Mamoun Ahram Lecture 1
 Introduction into Biochemistry Dr. Mamoun Ahram Lecture 1 Course information Recommended textbooks Biochemistry; Mary K. Campbell and Shawn O. Farrell, Brooks Cole; 7 th edition Instructors Dr. Mamoun
Introduction into Biochemistry Dr. Mamoun Ahram Lecture 1 Course information Recommended textbooks Biochemistry; Mary K. Campbell and Shawn O. Farrell, Brooks Cole; 7 th edition Instructors Dr. Mamoun
2. Acids and Bases. Grossman, CHE Definitions.
 Grossman, CE 230 2. Acids and Bases. 2.1 Definitions. Brønsted acids are proton donors, and Brønsted bases are proton acceptors. Examples of Brønsted acids: Cl, Br, 2 S 4,, + 2, + 4, 3, C 3 C 2, C 2 CC
Grossman, CE 230 2. Acids and Bases. 2.1 Definitions. Brønsted acids are proton donors, and Brønsted bases are proton acceptors. Examples of Brønsted acids: Cl, Br, 2 S 4,, + 2, + 4, 3, C 3 C 2, C 2 CC
Lectures 13 & 14 Key, with contributions from Palleros CHEM 109
 Lectures & Key, with contributions from alleros CEM 09.Build the nucleosides (sugar and base) & nucleotides (sugar, base, and phosphate). ucleobase ucleoside (D) - deoxyribose ucleotide (R) - ribose denine
Lectures & Key, with contributions from alleros CEM 09.Build the nucleosides (sugar and base) & nucleotides (sugar, base, and phosphate). ucleobase ucleoside (D) - deoxyribose ucleotide (R) - ribose denine
Thermodynamics is the study of energy and its effects on matter
 00Note Set 3 1 THE ENERGETICS OF LIFE Thermodynamics and Bioenergetics: Thermodynamics is the study of energy and its effects on matter Bioenergetics is the quantitative analysis of how organisms gain
00Note Set 3 1 THE ENERGETICS OF LIFE Thermodynamics and Bioenergetics: Thermodynamics is the study of energy and its effects on matter Bioenergetics is the quantitative analysis of how organisms gain
Water, ph and pka. Lecture 2: Margaret A. Daugherty. Fall Water: What makes it so good for life? Solvent properties.
 Lecture 2: Water, ph and pka Margaret A. Daugherty Fall 2004 Water: What makes it so good for life? Structure ice vs. water or more technically solid vs. liquid Solvent properties High heat capacity High
Lecture 2: Water, ph and pka Margaret A. Daugherty Fall 2004 Water: What makes it so good for life? Structure ice vs. water or more technically solid vs. liquid Solvent properties High heat capacity High
Lecture 7. Acids. non-metals form anions. metals form cations H+ - Professor Hicks Inorganic Chemistry (CHE152) + anion. molecular compounds
 Lecture 7 Professor icks Inorganic Chemistry (CE152) Acids + + anion + - anion substances that release + ions when dissolved Strong acids Cl NO 3 2 SO 4 + Cl - + NO - 3 2 + SO 2-4 hydrochloric acid nitric
Lecture 7 Professor icks Inorganic Chemistry (CE152) Acids + + anion + - anion substances that release + ions when dissolved Strong acids Cl NO 3 2 SO 4 + Cl - + NO - 3 2 + SO 2-4 hydrochloric acid nitric
Chimica Farmaceutica
 Chimica Farmaceutica Drug Targets Why should chemicals, some of which have remarkably simple structures, have such an important effect «in such a complicated and large structure as a human being? The answer
Chimica Farmaceutica Drug Targets Why should chemicals, some of which have remarkably simple structures, have such an important effect «in such a complicated and large structure as a human being? The answer
Patrick: An Introduction to Medicinal Chemistry 5e Chapter 01
 Questions Patrick: An Introduction to Medicinal Chemistry 5e 01) Which of the following molecules is a phospholipid? a. i b. ii c. iii d. iv 02) Which of the following statements is false regarding the
Questions Patrick: An Introduction to Medicinal Chemistry 5e 01) Which of the following molecules is a phospholipid? a. i b. ii c. iii d. iv 02) Which of the following statements is false regarding the
Medicinal Chemistry/ CHEM 458/658 Chapter 8- Receptors and Messengers
 Medicinal Chemistry/ CHEM 458/658 Chapter 8- Receptors and Messengers Bela Torok Department of Chemistry University of Massachusetts Boston Boston, MA 1 Introduction Receptor specific areas of proteins
Medicinal Chemistry/ CHEM 458/658 Chapter 8- Receptors and Messengers Bela Torok Department of Chemistry University of Massachusetts Boston Boston, MA 1 Introduction Receptor specific areas of proteins
2054, Chap. 8, page 1
 2054, Chap. 8, page 1 I. Metabolism: Energetics, Enzymes, and Regulation (Chapter 8) A. Energetics and work 1. overview a. energy = ability to do work (1) chemical, transport, mechanical (2) ultimate source
2054, Chap. 8, page 1 I. Metabolism: Energetics, Enzymes, and Regulation (Chapter 8) A. Energetics and work 1. overview a. energy = ability to do work (1) chemical, transport, mechanical (2) ultimate source
Acids and Bases. Acids and Bases
 BrØnsted-Lowry A BrØnsted-Lowry acid is a proton donor. A BrØnsted-Lowry base is a proton acceptor. H + = proton BrØnsted-Lowry Some molecules contain both hydrogen atoms and lone pairs and thus, can act
BrØnsted-Lowry A BrØnsted-Lowry acid is a proton donor. A BrØnsted-Lowry base is a proton acceptor. H + = proton BrØnsted-Lowry Some molecules contain both hydrogen atoms and lone pairs and thus, can act
SIMPLE MODEL Direct Binding Analysis
 Neurochemistry, 56:120:575 Dr. Patrick J. McIlroy Supplementary Notes SIMPLE MODEL Direct Binding Analysis The interaction of a (radio)ligand, L, with its receptor, R, to form a non-covalent complex, RL,
Neurochemistry, 56:120:575 Dr. Patrick J. McIlroy Supplementary Notes SIMPLE MODEL Direct Binding Analysis The interaction of a (radio)ligand, L, with its receptor, R, to form a non-covalent complex, RL,
Covalent bonds can have ionic character These are polar covalent bonds
 Polar Covalent Bonds: Electronegativity Covalent bonds can have ionic character These are polar covalent bonds Bonding electrons attracted more strongly by one atom than by the other Electron distribution
Polar Covalent Bonds: Electronegativity Covalent bonds can have ionic character These are polar covalent bonds Bonding electrons attracted more strongly by one atom than by the other Electron distribution
Enzyme Enzymes are proteins that act as biological catalysts. Enzymes accelerate, or catalyze, chemical reactions. The molecules at the beginning of
 Enzyme Enzyme Enzymes are proteins that act as biological catalysts. Enzymes accelerate, or catalyze, chemical reactions. The molecules at the beginning of the process are called substrates and the enzyme
Enzyme Enzyme Enzymes are proteins that act as biological catalysts. Enzymes accelerate, or catalyze, chemical reactions. The molecules at the beginning of the process are called substrates and the enzyme
Problems Points Credit
 Chem 201 Midterm Winter, 2018 Beauchamp ame Problems Points Credit 1. Functional Group omenclature (1 large structure) 30 2. Resonance, Formal Charge, Arrows 18 3. Properties of Atoms, Logic Arguments
Chem 201 Midterm Winter, 2018 Beauchamp ame Problems Points Credit 1. Functional Group omenclature (1 large structure) 30 2. Resonance, Formal Charge, Arrows 18 3. Properties of Atoms, Logic Arguments
Structure based drug design and LIE models for GPCRs
 Structure based drug design and LIE models for GPCRs Peter Kolb kolb@docking.org Shoichet Lab ACS 237 th National Meeting, March 24, 2009 p.1/26 [Acknowledgements] Brian Shoichet John Irwin Mike Keiser
Structure based drug design and LIE models for GPCRs Peter Kolb kolb@docking.org Shoichet Lab ACS 237 th National Meeting, March 24, 2009 p.1/26 [Acknowledgements] Brian Shoichet John Irwin Mike Keiser
Physical Organic Chemistry (15 h)
 Course code : CEM 43244 Course title : Advanced rganic Chemistry I Physical rganic Chemistry (15 h) Dr. Dinesh Pandithavidana E-mail: dinesh@kln.ac.lk Mobile: 0777-745-720 ffice: B1 222/3 Stereochemical
Course code : CEM 43244 Course title : Advanced rganic Chemistry I Physical rganic Chemistry (15 h) Dr. Dinesh Pandithavidana E-mail: dinesh@kln.ac.lk Mobile: 0777-745-720 ffice: B1 222/3 Stereochemical
2. All of the following are accurate statements regarding the above graph EXCEPT:
 General Biology 115 Summer 2014: Exam I Form A Name: 1. Which of the following best explains why FADH2 yields less ATP than NADH during cell respiration? a. Energy captured by FADH2 contributes less to
General Biology 115 Summer 2014: Exam I Form A Name: 1. Which of the following best explains why FADH2 yields less ATP than NADH during cell respiration? a. Energy captured by FADH2 contributes less to
Biochemistry. Lecture 8
 Biochemistry Lecture 8 Why Enzymes? igher reaction rates Greater reaction specificity Milder reaction conditions Capacity for regulation C - - C N 2 - C N 2 - C - C Chorismate mutase - C - C - C Metabolites
Biochemistry Lecture 8 Why Enzymes? igher reaction rates Greater reaction specificity Milder reaction conditions Capacity for regulation C - - C N 2 - C N 2 - C - C Chorismate mutase - C - C - C Metabolites
Problem Set 5 Question 1
 2.32 Problem Set 5 Question As discussed in class, drug discovery often involves screening large libraries of small molecules to identify those that have favorable interactions with a certain druggable
2.32 Problem Set 5 Question As discussed in class, drug discovery often involves screening large libraries of small molecules to identify those that have favorable interactions with a certain druggable
COMPUTER AIDED DRUG DESIGN (CADD) AND DEVELOPMENT METHODS
 COMPUTER AIDED DRUG DESIGN (CADD) AND DEVELOPMENT METHODS DRUG DEVELOPMENT Drug development is a challenging path Today, the causes of many diseases (rheumatoid arthritis, cancer, mental diseases, etc.)
COMPUTER AIDED DRUG DESIGN (CADD) AND DEVELOPMENT METHODS DRUG DEVELOPMENT Drug development is a challenging path Today, the causes of many diseases (rheumatoid arthritis, cancer, mental diseases, etc.)
MEDCHEM 562 Kent Kunze Lecture 2. Physicochemical Properties of Drugs and Drug Disposition. Stereochemistry (Double the trouble. or double the fun?
 12 MEDCHEM 562 Kent Kunze Lecture 2 Physicochemical Properties of Drugs and Drug Disposition Stereochemistry (Double the trouble. or double the fun?) The vast majority of drugs contain at least one stereo-center
12 MEDCHEM 562 Kent Kunze Lecture 2 Physicochemical Properties of Drugs and Drug Disposition Stereochemistry (Double the trouble. or double the fun?) The vast majority of drugs contain at least one stereo-center
Dr. Steven Pedersen October 3, Chemistry 3A. Midterm 1. No Calculators Allowed No Molecular Models Allowed Be Sure Your Exam has 9 Pages
 Dr. Steven Pedersen ctober 3, 2017 Chemistry 3A Midterm 1 Student name: ASWER KEY Student ID: (Also include your SID in the top left corner of each page) Student signature: Problem 1 Problem 2 Problem
Dr. Steven Pedersen ctober 3, 2017 Chemistry 3A Midterm 1 Student name: ASWER KEY Student ID: (Also include your SID in the top left corner of each page) Student signature: Problem 1 Problem 2 Problem
MEDCHEM 562 Kent Kunze Lecture 2. Physicochemical Properties of Drugs and Drug Disposition. Stereochemistry (Double the trouble. or double the fun?
 15 MEDCHEM 562 Kent Kunze Lecture 2 Physicochemical Properties of Drugs and Drug Disposition Stereochemistry (Double the trouble. or double the fun?) The vast majority of drugs contain at least one stereo-center
15 MEDCHEM 562 Kent Kunze Lecture 2 Physicochemical Properties of Drugs and Drug Disposition Stereochemistry (Double the trouble. or double the fun?) The vast majority of drugs contain at least one stereo-center
BCH Graduate Survey of Biochemistry
 BCH 5045 Graduate Survey of Biochemistry Instructor: Charles Guy Producer: Ron Thomas Director: Marsha Derosier Lecture 6 Slide sets available at: http://hort.ifas.ufl.edu/teach/guyweb/bch5045/index.html
BCH 5045 Graduate Survey of Biochemistry Instructor: Charles Guy Producer: Ron Thomas Director: Marsha Derosier Lecture 6 Slide sets available at: http://hort.ifas.ufl.edu/teach/guyweb/bch5045/index.html
Ligand Binding A. Binding to a Single Site:
 A. Binding to a Single Site: The uilibrium constant (also known as association constant or affinity constant) for the binding of a ligand to a protein is described by the following uation (note: A ): [
A. Binding to a Single Site: The uilibrium constant (also known as association constant or affinity constant) for the binding of a ligand to a protein is described by the following uation (note: A ): [
Final Exam. CHEM 181: Introduction to Chemical Principles Answer Key. 1. The major resonance structure of 1,4-diazepine is shown below:
 Final Exam EM 181: Introduction to hemical Principles Answer Key 1. The major resonance structure of 1,4-diazepine is shown below: or Show all reasonable resonance structures; label them as major (if equivalent
Final Exam EM 181: Introduction to hemical Principles Answer Key 1. The major resonance structure of 1,4-diazepine is shown below: or Show all reasonable resonance structures; label them as major (if equivalent
Chapter 2 Lecture Outline
 Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 2 Lecture Outline Prepared by Rabi Ann Musah State University of New York at Albany Copyright The McGraw-Hill Companies,
Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 2 Lecture Outline Prepared by Rabi Ann Musah State University of New York at Albany Copyright The McGraw-Hill Companies,
ENZYME KINETICS AND INHIBITION
 ENZYME KINETICS AND INHIBITION The kinetics of reactions involving enzymes are a little bit different from other reactions. First of all, there are sometimes lots of steps involved. Also, the reaction
ENZYME KINETICS AND INHIBITION The kinetics of reactions involving enzymes are a little bit different from other reactions. First of all, there are sometimes lots of steps involved. Also, the reaction
C h a p t e r T h r e e: Acids and Bases. 17, 21-Dimethylheptatriacontane, a sex attractant pheromone of the tsetse fly
 C h a p t e r T h r e e: Acids and Bases 17, 21-Dimethylheptatriacontane, a sex attractant pheromone of the tsetse fly CM 321: Summary of Important Concepts YConcepts for Chapter 3: Acids and Bases I.
C h a p t e r T h r e e: Acids and Bases 17, 21-Dimethylheptatriacontane, a sex attractant pheromone of the tsetse fly CM 321: Summary of Important Concepts YConcepts for Chapter 3: Acids and Bases I.
Ping-Chiang Lyu. Institute of Bioinformatics and Structural Biology, Department of Life Science, National Tsing Hua University.
 Pharmacophore-based Drug design Ping-Chiang Lyu Institute of Bioinformatics and Structural Biology, Department of Life Science, National Tsing Hua University 96/08/07 Outline Part I: Analysis The analytical
Pharmacophore-based Drug design Ping-Chiang Lyu Institute of Bioinformatics and Structural Biology, Department of Life Science, National Tsing Hua University 96/08/07 Outline Part I: Analysis The analytical
Chemistry 304B, Spring 99 Lecture 2 1
 Chemistry 04B, Spring 99 Lecture 2 1 Consider Acidity and Basicity Recall carboxylic acids and amines (base). Jones: p 27-8; 149, 291, 822-826, 1086-1090, 958-96, 878-882 General terms: Lewis acid: capable
Chemistry 04B, Spring 99 Lecture 2 1 Consider Acidity and Basicity Recall carboxylic acids and amines (base). Jones: p 27-8; 149, 291, 822-826, 1086-1090, 958-96, 878-882 General terms: Lewis acid: capable
I: Life and Energy. Lecture 2: Solutions and chemical potential; Osmotic pressure (B Lentz).
 I: Life and Energy Lecture 1: What is life? An attempt at definition. Energy, heat, and work: Temperature and thermal equilibrium. The First Law. Thermodynamic states and state functions. Reversible and
I: Life and Energy Lecture 1: What is life? An attempt at definition. Energy, heat, and work: Temperature and thermal equilibrium. The First Law. Thermodynamic states and state functions. Reversible and
Resonance and M.O. View of Butadiene. Super-Conjugated or Aromatic p e - Systems
 Resonance and M.. View of Butadiene The different resonance forms of butadiene suggest p bonding character between the two central carbon atoms. 2 2 2 2 carbanion 2 2 carbocation The M.. view of butadiene
Resonance and M.. View of Butadiene The different resonance forms of butadiene suggest p bonding character between the two central carbon atoms. 2 2 2 2 carbanion 2 2 carbocation The M.. view of butadiene
k -2 k 2 K F I-N k 3 k -1 K F U-I k 1 NSP
 Symposium on Protein Misfolding Diseases Biochemistry & Molecular Biology website May 1-2, 2007 University of Massachusetts Amherst Tuesday, May 1 1:00-1:30pm Registration 1:30-3:30pm Panel discussion:
Symposium on Protein Misfolding Diseases Biochemistry & Molecular Biology website May 1-2, 2007 University of Massachusetts Amherst Tuesday, May 1 1:00-1:30pm Registration 1:30-3:30pm Panel discussion:
Supplementary Material (ESI) for Natural Product Reports This journal is The Royal Society of Chemistry Effect on promastigotes, amastigotes of
 upplementary Material (EI) for atural Product Reports This journal is The Royal ociety of Chemistry 2010 Table 2. Biological activities of purely synthetic guanidines Entry Guanidine compound Biological
upplementary Material (EI) for atural Product Reports This journal is The Royal ociety of Chemistry 2010 Table 2. Biological activities of purely synthetic guanidines Entry Guanidine compound Biological
16 years ago TODAY (9/11) at 8:46, the first tower was hit at 9:03, the second tower was hit. Lecture 2 (9/11/17)
 16 years ago TODAY (9/11) at 8:46, the first tower was hit at 9:03, the second tower was hit By Anthony Quintano - https://www.flickr.com/photos/quintanomedia/15071865580, CC BY 2.0, https://commons.wikimedia.org/w/index.php?curid=38538291
16 years ago TODAY (9/11) at 8:46, the first tower was hit at 9:03, the second tower was hit By Anthony Quintano - https://www.flickr.com/photos/quintanomedia/15071865580, CC BY 2.0, https://commons.wikimedia.org/w/index.php?curid=38538291
Advanced Medicinal Chemistry SLIDES B
 Advanced Medicinal Chemistry Filippo Minutolo CFU 3 (21 hours) SLIDES B Drug likeness - ADME two contradictory physico-chemical parameters to balance: 1) aqueous solubility 2) lipid membrane permeability
Advanced Medicinal Chemistry Filippo Minutolo CFU 3 (21 hours) SLIDES B Drug likeness - ADME two contradictory physico-chemical parameters to balance: 1) aqueous solubility 2) lipid membrane permeability
CHM 251 Organic Chemistry 1
 CM 251 rganic Chemistry 1 NAME Fall 2008 omework #1 Due: Friday, September 26, 2008 5:00 pm Print out this assignment and complete all of your work on these pages. Use the back if necessary. 1. Given the
CM 251 rganic Chemistry 1 NAME Fall 2008 omework #1 Due: Friday, September 26, 2008 5:00 pm Print out this assignment and complete all of your work on these pages. Use the back if necessary. 1. Given the
3. Based on how energy is stored in the molecules, explain why ΔG is independent of the path of the reaction.
 B. Thermodynamics 1. What is "free energy"? 2. Where is this energy stored? We say that ΔG is a thermodynamic property, meaning that it is independent of the way that the conversion of reactants to products
B. Thermodynamics 1. What is "free energy"? 2. Where is this energy stored? We say that ΔG is a thermodynamic property, meaning that it is independent of the way that the conversion of reactants to products
Substituents already attached to an aromatic ring influence the preferred site of attachment of an incoming electrophile. NO2
 Lecture outline Directing effects of substituents Substituents already attached to an aromatic ring influence the preferred site of attachment of an incoming electrophile. e.g., nitration of toluene 3
Lecture outline Directing effects of substituents Substituents already attached to an aromatic ring influence the preferred site of attachment of an incoming electrophile. e.g., nitration of toluene 3
Acid Dissociation Constant
 CE 131 Lecture 37 Lewis Acids and Bases Chapter 16: pp. 800-802. Acid Dissociation Constant C 2 3 2 + 2 3 + + C 2 3-2 [ 3 + ][C 2 3-2 ] K = [ 2 ][C 2 3 2 ] [ 3 + ][C 2 3-2 ] K a = K [ 2 ] = [C 2 3 2 ]
CE 131 Lecture 37 Lewis Acids and Bases Chapter 16: pp. 800-802. Acid Dissociation Constant C 2 3 2 + 2 3 + + C 2 3-2 [ 3 + ][C 2 3-2 ] K = [ 2 ][C 2 3 2 ] [ 3 + ][C 2 3-2 ] K a = K [ 2 ] = [C 2 3 2 ]
Lewis Structure. Lewis Structures & VSEPR. Octet & Duet Rules. Steps for drawing Lewis Structures
 Lewis Structure Lewis Structures & VSEPR Lewis Structures shows how the are arranged among the atoms of a molecule There are rules for Lewis Structures that are based on the formation of a Atoms want to
Lewis Structure Lewis Structures & VSEPR Lewis Structures shows how the are arranged among the atoms of a molecule There are rules for Lewis Structures that are based on the formation of a Atoms want to
Name: (Print your name) Chem Spring, Midterm 1
 ame: (Print your name) hem 2010 Spring, 2019 Midterm 1 hem 2010 Midterm 1 Spring, 2019 Beauchamp ame Problems Points redit 1. Functional Group omenclature (1 large structure) 30 2. Lewis Structures, Resonance,
ame: (Print your name) hem 2010 Spring, 2019 Midterm 1 hem 2010 Midterm 1 Spring, 2019 Beauchamp ame Problems Points redit 1. Functional Group omenclature (1 large structure) 30 2. Lewis Structures, Resonance,
Nonlinear QSAR and 3D QSAR
 onlinear QSAR and 3D QSAR Hugo Kubinyi Germany E-Mail kubinyi@t-online.de HomePage www.kubinyi.de onlinear Lipophilicity-Activity Relationships drug receptor Possible Reasons for onlinear Lipophilicity-Activity
onlinear QSAR and 3D QSAR Hugo Kubinyi Germany E-Mail kubinyi@t-online.de HomePage www.kubinyi.de onlinear Lipophilicity-Activity Relationships drug receptor Possible Reasons for onlinear Lipophilicity-Activity
1.5 h; 120 points please print clearly Dr. Kathleen Nolta Dr. Jordan Walk. Problem Points Score GSI I 23 II 27 III 24 IV 22 V 24 Total 120
 hemistry 10 econd Examination First ame Last ame 1.5 h; 10 points please print clearly Dr. Kathleen olta Dr. Jordan Walk ignature UM ID # PRIT TE FIRT LETTER F YUR LAT AME ERE: Problem Points core GI I
hemistry 10 econd Examination First ame Last ame 1.5 h; 10 points please print clearly Dr. Kathleen olta Dr. Jordan Walk ignature UM ID # PRIT TE FIRT LETTER F YUR LAT AME ERE: Problem Points core GI I
Homework - Chapter 1 Chem 2310
 omework - hapter 1 hem 2310 ame I. Introduction to rganic hemistry 1. Explain in your own words what organic chemistry is, and what it is useful for. 2. Why do you think the field of study that you are
omework - hapter 1 hem 2310 ame I. Introduction to rganic hemistry 1. Explain in your own words what organic chemistry is, and what it is useful for. 2. Why do you think the field of study that you are
Receptor Based Drug Design (1)
 Induced Fit Model For more than 100 years, the behaviour of enzymes had been explained by the "lock-and-key" mechanism developed by pioneering German chemist Emil Fischer. Fischer thought that the chemicals
Induced Fit Model For more than 100 years, the behaviour of enzymes had been explained by the "lock-and-key" mechanism developed by pioneering German chemist Emil Fischer. Fischer thought that the chemicals
Supporting information
 Supporting information Fluorescent derivatives of AC-42 to probe bitopic orthosteric/allosteric binding mechanisms on muscarinic M1 receptors Sandrine B. Daval, Céline Valant, Dominique Bonnet, Esther
Supporting information Fluorescent derivatives of AC-42 to probe bitopic orthosteric/allosteric binding mechanisms on muscarinic M1 receptors Sandrine B. Daval, Céline Valant, Dominique Bonnet, Esther
