1. (18) Multiple choice questions. Please place your answer on the line preceding each question.
|
|
|
- Myrtle Grant
- 5 years ago
- Views:
Transcription
1 CEM 5720 ame KEY Exam 2 ctober 21, 2015 Read all questions carefully and attempt those questions you are sure of first. Remember to proof your work; art work carries as much importance as written responses. Look over the entire exam before you begin. GD LUCK!! 1. (18) Multiple choice questions. Please place your answer on the line preceding each question. C Which of the following statements is true? a. A positive value of π indicates that a substituent is larger than hydrogen b. A positive value of π indicates that a substituent is more electron donating than hydrogen c. A positive value of π indicates that a substituent is more hydrophobic than hydrogen d. A negative value of π indicates that a substituent is more hydrophobic than hydrogen C Which of the following defines a structure which can be used as the starting point for drug design and development? a. Active principle b. Pharmacophore c. Lead compound d. rphan drug D From which source did the lead compound for the antiulcer agent cimetidine come from? a. Marine sources b. Venoms and toxins c. Combinatorial chemistry d. An endogenous compound B ydrogen is often replaced in drugs with a fluorine atom for a variety of reasons. What term is used to describe the fluorine atom when it is used to replace hydrogen? a. Isotope b. Isostere c. Isomer d. Isobar C Which of the following physicochemical properties is least commonly considered in QSAR studies? a. ydrophobicity b. Electronic influence of substituents c. Dipole moment d. Size of substituents C The simplification strategy is commonly used in drug design. What must be retained in a molecule during the simplification process to keep the biological activity? a. The molecular weight b. The stereochemistry c. The pharmacophore d. The hydrophobic character
2 2. (24) Three of the best selling drugs for 2010 in the United States are shown below. Determine which of the compounds, if any, violate Lipinski s rules. Log P values are given from online at oxycodone (xycontin) analgesic C 3 M.W. = Log P = 1.04 & = 2 's&'s=5 Violates Lipinski's Rules YES S M.W. = Log P = 1.66 & = 1 esomeprazole (exium) antiacid reflux 's&'s=6 Violates Lipinski's Rules YES 3 C Ac C 3 C 3 C 3 Bz Ac M.W. = Log P = 3.54 & = 4 's&'s=14 Taxol Violates Lipinski's Rules YES f the top 10 best selling drugs of 2010, GSK s Advair, an inhaled asthma medication, could not be used in the above question. Why was this drug left out of the Lipinski analysis? Because the Lipinski analysis describes characteristics that are for oral medications and Advair would be absorbed through the lungs and not in the gastrointestinal tract.
3 3. (20) In a recent article (J. d. Chem. 2009, 52, ) the authors explored the development of a potent agonist for the histamine 4 receptor subtype. The 4 receptor subtype has been shown to play a critical role in immunological and inflammatory processes. The goal of the research was to develop a compound that was selective for the 4 subtype over the 2 and 3 receptors. A. Define agonist. A molecule that produces a biological response or action, B. The following scheme depicts the molecules that were produced in the development process. For each indicated step suggest what common modification process was used to arrive at the shown structure. 1 2 C C 3 4 C S 1. omologation 2. Isosteric replacement 3. omologation 4. Isosteric replacement C. From the table of isosteres suggest another possible replacement for the sulfur atom in the final compound shown above Se-
4 4. (12) Captopril is an ACE inhibitor used in the treatment of high blood pressure. Based on the data in the following table, suggest two additional analogs that should be synthesized and screened. ote the larger the K i value the better the analog. STRUCTURE RELATIVE K i S captopril C 2 1 analog 1 S S 12,500 S C 2 S C C 2 S S C 2 12, analog 2 2 C C 2 S C 2 C C S C 2 2 C 1,100 C 2 C 2 5. (12) Use the partial list of Log P values at the end of the exam to calculate the log P ibuprofen. For full credit show all your work. branching C =1.41 C 3 C C 3 C Ibuprofen Log P = (2 x C3 )+ C + C2 + (branching) + PhC2C + C3 + (branching) Log P = (2 x 0.50) (-0.20) (-0.20) Log P = 3.51
5 6. (16) Carbonic anhydrases are ubiquitous zinc enzymes present in biological systems. These enzymes catalyze the interconversion between carbon dioxide and bicarbonate. f the approximately thirty compounds in clinical development as carbonic anhydrase inhibitors each compound contains a sulfonamide functional group (-S 2 2 ) that is responsible for binding to the zinc atom in the enzyme. Substituted sulfonamides exist in equilibrium between the protonated and deprotonated state shown below. A model of the zinc binding site for carbonic anhydrase is shown below. It contains a zinc (II) atom coordinated to three histidine residues on the protein chain and a fourth site that binds the natural substrates (carbon dioxide or bicarbonate) as well as the sulfonamide inhibitors. Which of the two structures in the above equilibria should be the better inhibitor of carbonic anhydrase? (Draw the chemical structure) Binding site is 94 is119 Zn 2+ is 96 X S 2 2 The binding site is a Lewis acid (positively charged) and would bindtothelonepaironthe sulfonamide nitrogen. The protonated sulfonamide would be repelled by the zinc. f the sixteen carbonic anhydrase isozymes (enzyme forms) found in mammals four of them are membrane bound. Using the table of sigma values ( ) what single group would you incorporate on the aromatic ring of the sulfonamide in order to enhance both binding to the zinc atom and penetration of the lipophilic membrane? (Draw the chemical structure) In mammals there are also five isozymes that are cytosolic existing in the aqueous portion of cellular fluids. Using the table of sigma values ( ) draw the structure of a disubstituted sulfonamide that would strongly bind the zinc atom at the sulfonamide, not enter the membrane, and minimize promiscuous binding at the zinc atom by the substituent. Using the table of sigma values ( ) what single group would you incorporate on the aromatic ring of the sulfonamide to produce a compound that was inactive as a carbonic anhydrase inhibitor? (Draw the chemical structure) S 2 2
MEDICINAL CHEMISTRY Homework 2 - ANSWERS
 MEDICIAL CEMISTY omework 2 - ASWES 1. eceptor binding curves. Make a plot of %drug-bound versus -log[drug] for a drug whose K D is 5 x 10-6 M. Make a plot of %drug-bound vs [drug] for this same system.
MEDICIAL CEMISTY omework 2 - ASWES 1. eceptor binding curves. Make a plot of %drug-bound versus -log[drug] for a drug whose K D is 5 x 10-6 M. Make a plot of %drug-bound vs [drug] for this same system.
CHEM 4170 Problem Set #1
 CHEM 4170 Problem Set #1 0. Work problems 1-7 at the end of Chapter ne and problems 1, 3, 4, 5, 8, 10, 12, 17, 18, 19, 22, 24, and 25 at the end of Chapter Two and problem 1 at the end of Chapter Three
CHEM 4170 Problem Set #1 0. Work problems 1-7 at the end of Chapter ne and problems 1, 3, 4, 5, 8, 10, 12, 17, 18, 19, 22, 24, and 25 at the end of Chapter Two and problem 1 at the end of Chapter Three
Lecture 12. Metalloproteins - II
 Lecture 12 Metalloproteins - II Metalloenzymes Metalloproteins with one labile coordination site around the metal centre are known as metalloenzyme. As with all enzymes, the shape of the active site is
Lecture 12 Metalloproteins - II Metalloenzymes Metalloproteins with one labile coordination site around the metal centre are known as metalloenzyme. As with all enzymes, the shape of the active site is
Ping-Chiang Lyu. Institute of Bioinformatics and Structural Biology, Department of Life Science, National Tsing Hua University.
 Pharmacophore-based Drug design Ping-Chiang Lyu Institute of Bioinformatics and Structural Biology, Department of Life Science, National Tsing Hua University 96/08/07 Outline Part I: Analysis The analytical
Pharmacophore-based Drug design Ping-Chiang Lyu Institute of Bioinformatics and Structural Biology, Department of Life Science, National Tsing Hua University 96/08/07 Outline Part I: Analysis The analytical
Plan. Day 2: Exercise on MHC molecules.
 Plan Day 1: What is Chemoinformatics and Drug Design? Methods and Algorithms used in Chemoinformatics including SVM. Cross validation and sequence encoding Example and exercise with herg potassium channel:
Plan Day 1: What is Chemoinformatics and Drug Design? Methods and Algorithms used in Chemoinformatics including SVM. Cross validation and sequence encoding Example and exercise with herg potassium channel:
Structural biology and drug design: An overview
 Structural biology and drug design: An overview livier Taboureau Assitant professor Chemoinformatics group-cbs-dtu otab@cbs.dtu.dk Drug discovery Drug and drug design A drug is a key molecule involved
Structural biology and drug design: An overview livier Taboureau Assitant professor Chemoinformatics group-cbs-dtu otab@cbs.dtu.dk Drug discovery Drug and drug design A drug is a key molecule involved
PHYSIO-CHEMICAL PROPERTIES OF THE DRUG. It is the interaction of the drug with its environment
 PYSIO-CEMICAL PROPERTIES OF TE DRUG It is the interaction of the drug with its environment Reducing the complexity Biological process in drug action Dissolution of drug in gastrointestinal fluids Absorption
PYSIO-CEMICAL PROPERTIES OF TE DRUG It is the interaction of the drug with its environment Reducing the complexity Biological process in drug action Dissolution of drug in gastrointestinal fluids Absorption
COMPUTER AIDED DRUG DESIGN (CADD) AND DEVELOPMENT METHODS
 COMPUTER AIDED DRUG DESIGN (CADD) AND DEVELOPMENT METHODS DRUG DEVELOPMENT Drug development is a challenging path Today, the causes of many diseases (rheumatoid arthritis, cancer, mental diseases, etc.)
COMPUTER AIDED DRUG DESIGN (CADD) AND DEVELOPMENT METHODS DRUG DEVELOPMENT Drug development is a challenging path Today, the causes of many diseases (rheumatoid arthritis, cancer, mental diseases, etc.)
Practice Midterm Exam 200 points total 75 minutes Multiple Choice (3 pts each 30 pts total) Mark your answers in the space to the left:
 MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
Welcome to Week 5. Chapter 9 - Binding, Structure, and Diversity. 9.1 Intermolecular Forces. Starting week five video. Introduction to Chapter 9
 Welcome to Week 5 Starting week five video Please watch the online video (49 seconds). Chapter 9 - Binding, Structure, and Diversity Introduction to Chapter 9 Chapter 9 contains six subsections. Intermolecular
Welcome to Week 5 Starting week five video Please watch the online video (49 seconds). Chapter 9 - Binding, Structure, and Diversity Introduction to Chapter 9 Chapter 9 contains six subsections. Intermolecular
Chapter 3: Stereochemistry & Chirality
 Chapter 3: Stereochemistry & Chirality 1. Chiral & Achiral Compounds - Identifying Stereocenters 2. Assigning R & S configurations 3. Diastereomers - Molecules with two or more stereocenters 4. Properties
Chapter 3: Stereochemistry & Chirality 1. Chiral & Achiral Compounds - Identifying Stereocenters 2. Assigning R & S configurations 3. Diastereomers - Molecules with two or more stereocenters 4. Properties
Introduction to Chemoinformatics and Drug Discovery
 Introduction to Chemoinformatics and Drug Discovery Irene Kouskoumvekaki Associate Professor February 15 th, 2013 The Chemical Space There are atoms and space. Everything else is opinion. Democritus (ca.
Introduction to Chemoinformatics and Drug Discovery Irene Kouskoumvekaki Associate Professor February 15 th, 2013 The Chemical Space There are atoms and space. Everything else is opinion. Democritus (ca.
Learning Organic Chemistry
 Objective 1 Represent organic molecules with chemical formulas, expanded formulas, Lewis structures, skeletal structures. Determine shape (VSEPR), bond polarity, and molecule polarity. Identify functional
Objective 1 Represent organic molecules with chemical formulas, expanded formulas, Lewis structures, skeletal structures. Determine shape (VSEPR), bond polarity, and molecule polarity. Identify functional
Notes of Dr. Anil Mishra at 1
 Introduction Quantitative Structure-Activity Relationships QSPR Quantitative Structure-Property Relationships What is? is a mathematical relationship between a biological activity of a molecular system
Introduction Quantitative Structure-Activity Relationships QSPR Quantitative Structure-Property Relationships What is? is a mathematical relationship between a biological activity of a molecular system
Number 1 What is a chemical reaction?
 Chemical Reactions and Enzymes Number 1 What is a chemical reaction? A process that changes, or transforms, one set of chemicals into another by changing the chemical bonds that join atoms in compounds.
Chemical Reactions and Enzymes Number 1 What is a chemical reaction? A process that changes, or transforms, one set of chemicals into another by changing the chemical bonds that join atoms in compounds.
Chemistry in Biology. Section 1. Atoms, Elements, and Compounds
 Section 1 Atoms, Elements, and Compounds Atoms! Chemistry is the study of matter.! Atoms are the building blocks of matter.! Neutrons and protons are located at the center of the atom.! Protons are positively
Section 1 Atoms, Elements, and Compounds Atoms! Chemistry is the study of matter.! Atoms are the building blocks of matter.! Neutrons and protons are located at the center of the atom.! Protons are positively
BIOLOGY LTF DIAGNOSTIC TEST CHEMISTRY OF LIFE
 016024 BILGY LTF DIAGNSTI TEST EMISTRY F LIFE TEST DE: 016024 Directions: Each of the questions or incomplete statements below is followed by five suggested answers or completions. Select the one that
016024 BILGY LTF DIAGNSTI TEST EMISTRY F LIFE TEST DE: 016024 Directions: Each of the questions or incomplete statements below is followed by five suggested answers or completions. Select the one that
Chemical Reactions and Enzymes. (Pages 49-59)
 Chemical Reactions and Enzymes (Pages 49-59) Chemical Reactions Chemistry of Life Not just what life is made of. What life does! Chemical Reactions Chemistry of Life Not just what life is made of. What
Chemical Reactions and Enzymes (Pages 49-59) Chemical Reactions Chemistry of Life Not just what life is made of. What life does! Chemical Reactions Chemistry of Life Not just what life is made of. What
Electronic Structure. Hammett Correlations. Linear Free Energy Relationships. Quantitative Structure Activity Relationships (QSAR)
 Electronic Structure ammett Correlations Quantitative Structure Activity Relationships (QSAR) Quantitative Structure Activity Relationships (QSAR) We have already seen that by changing groups in a drug
Electronic Structure ammett Correlations Quantitative Structure Activity Relationships (QSAR) Quantitative Structure Activity Relationships (QSAR) We have already seen that by changing groups in a drug
Medicinal Chemistry/ CHEM 458/658 Chapter 3- SAR and QSAR
 Medicinal Chemistry/ CHEM 458/658 Chapter 3- SAR and QSAR Bela Torok Department of Chemistry University of Massachusetts Boston Boston, MA 1 Introduction Structure-Activity Relationship (SAR) - similar
Medicinal Chemistry/ CHEM 458/658 Chapter 3- SAR and QSAR Bela Torok Department of Chemistry University of Massachusetts Boston Boston, MA 1 Introduction Structure-Activity Relationship (SAR) - similar
Chemical Reac+ons and Enzymes. Lesson Overview. Lesson Overview. 2.4 Chemical Reactions and Enzymes
 Lesson Overview Chemical Reac+ons and Enzymes Lesson Overview 2.4 Chemical Reactions and Enzymes THINK ABOUT IT Living things are made up of chemical compounds, but chemistry isn t just what life is made
Lesson Overview Chemical Reac+ons and Enzymes Lesson Overview 2.4 Chemical Reactions and Enzymes THINK ABOUT IT Living things are made up of chemical compounds, but chemistry isn t just what life is made
CHEM 341: Organic Chemistry I at North Dakota State University Midterm Exam 01 - Fri, Feb 10, 2012!! Name:!
 CEM 341: rganic Chemistry I at North Dakota State University Midterm Exam 01 - Fri, Feb 10, 2012!! Name:! Please read through each question carefully and answer in the spaces provided. A good strategy
CEM 341: rganic Chemistry I at North Dakota State University Midterm Exam 01 - Fri, Feb 10, 2012!! Name:! Please read through each question carefully and answer in the spaces provided. A good strategy
Chem 314. Problem Points Credit. 1. Nomenclature D Lewis structures D Structures, Formal Charge & Resonance 34
 alifornia State Polytechnic University, Pomona 1 Spring, 2013 Midterm Exam hem 314 Beauchamp hem 314 ame Problem Points redit 1. omenclature 30 2. 2D Lewis structures 20 3. 3D Structures, Formal harge
alifornia State Polytechnic University, Pomona 1 Spring, 2013 Midterm Exam hem 314 Beauchamp hem 314 ame Problem Points redit 1. omenclature 30 2. 2D Lewis structures 20 3. 3D Structures, Formal harge
NURS1004 Week 12 Lecture 1 Acid Base Balance Prepared by Didy Button
 NURS1004 Week 12 Lecture 1 Acid Base Balance Prepared by Didy Button The Role of Amino Acids in Protein Buffer Systems 2-7 ph and Homeostasis ph The concentration of hydrogen ions (H + ) in a solution
NURS1004 Week 12 Lecture 1 Acid Base Balance Prepared by Didy Button The Role of Amino Acids in Protein Buffer Systems 2-7 ph and Homeostasis ph The concentration of hydrogen ions (H + ) in a solution
A Brief Overview of Biochemistry. And I mean BRIEF!
 A Brief Overview of Biochemistry And I mean BRIEF! Introduction A. Chemistry deals with the composition of substances and how they change. B. A knowledge of chemistry is necessary for the understanding
A Brief Overview of Biochemistry And I mean BRIEF! Introduction A. Chemistry deals with the composition of substances and how they change. B. A knowledge of chemistry is necessary for the understanding
Statistical concepts in QSAR.
 Statistical concepts in QSAR. Computational chemistry represents molecular structures as a numerical models and simulates their behavior with the equations of quantum and classical physics. Available programs
Statistical concepts in QSAR. Computational chemistry represents molecular structures as a numerical models and simulates their behavior with the equations of quantum and classical physics. Available programs
2.1 Atoms, Ions, and Molecules. 2.1 Atoms, Ions, and Molecules. 2.1 Atoms, Ions, and Molecules. 2.1 Atoms, Ions, and Molecules
 All living things are based on atoms and their interactions. Living things consist of atoms of different elements. An atom is the smallest basic unit of matter. An element is one type of atom. ydrogen
All living things are based on atoms and their interactions. Living things consist of atoms of different elements. An atom is the smallest basic unit of matter. An element is one type of atom. ydrogen
10/16/17 ACIDS AND BASES, DEFINED WATER IS AMPHOTERIC OUTLINE. 9.1 Properties of Acids and Bases. 9.2 ph. 9.3 Buffers
 ACIDS AND BASES, DEFINED A hydrogen atom contains a proton and an electron, thus a hydrogen ion (H + ) is a proton: Acids: Proton (H + ) transfer between molecules is the basis of acid/base chemistry Ø
ACIDS AND BASES, DEFINED A hydrogen atom contains a proton and an electron, thus a hydrogen ion (H + ) is a proton: Acids: Proton (H + ) transfer between molecules is the basis of acid/base chemistry Ø
SAR. Structure - Activity Relationships (alkoholy, amíny, aldehydy, ketóny, estery, amidy, kyseliny, uhľovodíky) 2/28/2016
 Structure Activity elationships (SA) identifies which functional groups are important for binding and activity SA Structure - Activity elationships (alkoholy, amíny, aldehydy, ketóny, estery, amidy, kyseliny,
Structure Activity elationships (SA) identifies which functional groups are important for binding and activity SA Structure - Activity elationships (alkoholy, amíny, aldehydy, ketóny, estery, amidy, kyseliny,
Chapter 2 Polar Covalent Bonds; Acids and Bases SAMPLE. Chapter Outline
 Chapter 2 Polar Covalent Bonds; Acids and Bases Chapter utline I. Polar covalent bonds (Sections 2.1 2.3). A. Electronegativity (Section 2.1). 1. Although some bonds are totally ionic and some are totally
Chapter 2 Polar Covalent Bonds; Acids and Bases Chapter utline I. Polar covalent bonds (Sections 2.1 2.3). A. Electronegativity (Section 2.1). 1. Although some bonds are totally ionic and some are totally
Exam 1 (Monday, July 6, 2015)
 Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Hole s Human Anatomy and Physiology Eleventh Edition. Chapter 2
 Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 2 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. CHAPTER 2 CHEMICAL BASIS OF
Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 2 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. CHAPTER 2 CHEMICAL BASIS OF
Name (Print) Section # or TA name. 1. You may use a calculator and one 8 by 11-1/2 handwritten crib sheet during this
 1. You may use a calculator and one 8 by 11-1/2 handwritten crib sheet during this exam. Bring extra pencils, batteries, calculators, if necessary. 2. Please place all books, bags, hats, etc. under the
1. You may use a calculator and one 8 by 11-1/2 handwritten crib sheet during this exam. Bring extra pencils, batteries, calculators, if necessary. 2. Please place all books, bags, hats, etc. under the
Sample Questions for the Chemistry of Life Topic Test
 Sample Questions for the Chemistry of Life Topic Test 1. Enzymes play a crucial role in biology by serving as biological catalysts, increasing the rates of biochemical reactions by decreasing their activation
Sample Questions for the Chemistry of Life Topic Test 1. Enzymes play a crucial role in biology by serving as biological catalysts, increasing the rates of biochemical reactions by decreasing their activation
Problem Set 7: Stereochemistry-ANSWER KEY
 Problem Set 7: Stereochemistry-ANSWER KEY Chemistry 260 Organic Chemistry 1. The answer is (2). Circled isomers have a stereogenic carbon (*) and hence a stereogenic centre. C 2 C 2 C 2 C 2 C 2 C 2 C 2
Problem Set 7: Stereochemistry-ANSWER KEY Chemistry 260 Organic Chemistry 1. The answer is (2). Circled isomers have a stereogenic carbon (*) and hence a stereogenic centre. C 2 C 2 C 2 C 2 C 2 C 2 C 2
PLEASE DO NOT WRITE ON THE EXAM (EVEN YOUR NAME) UNTIL TOLD TO START! Student ID # CHEM 8A, Organic Chemistry EXAM 1 (300 points)
 PLEASE D T WRITE TE EXAM (EVE YUR AME) UTIL TLD T START! UCSC, Binder ame Student ID # CEM 8A, rganic Chemistry EXAM (300 points) In each of the following problems, use your knowledge of organic chemistry
PLEASE D T WRITE TE EXAM (EVE YUR AME) UTIL TLD T START! UCSC, Binder ame Student ID # CEM 8A, rganic Chemistry EXAM (300 points) In each of the following problems, use your knowledge of organic chemistry
OXFORD H i g h e r E d u c a t i o n Oxford University Press, All rights reserved.
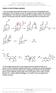 Patrick, An Introduction to dicinal hemistry 4e Answers to end-of-chapter questions 1) The mechanism below shows the release of one molecule of formaldehyde from methenamine. The mechanism can then be
Patrick, An Introduction to dicinal hemistry 4e Answers to end-of-chapter questions 1) The mechanism below shows the release of one molecule of formaldehyde from methenamine. The mechanism can then be
4 Examples of enzymes
 Catalysis 1 4 Examples of enzymes Adding water to a substrate: Serine proteases. Carbonic anhydrase. Restrictions Endonuclease. Transfer of a Phosphoryl group from ATP to a nucleotide. Nucleoside monophosphate
Catalysis 1 4 Examples of enzymes Adding water to a substrate: Serine proteases. Carbonic anhydrase. Restrictions Endonuclease. Transfer of a Phosphoryl group from ATP to a nucleotide. Nucleoside monophosphate
(1) Recall the different types of intermolecular interactions. (2) Look at the structure and determine the correct answer.
 MCAT rganic Chemistry - Problem Drill 11: Carboxylic Acids Question No. 1 of 10 Question 1. What kinds of interactions are holding together the carboxylic acid dimer shown? Question #01 3 C C 3 (A) Van
MCAT rganic Chemistry - Problem Drill 11: Carboxylic Acids Question No. 1 of 10 Question 1. What kinds of interactions are holding together the carboxylic acid dimer shown? Question #01 3 C C 3 (A) Van
CHEM J-9 June 2014
 CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
Chemistry 14C Fall 2015 Final Exam Part B Page 1
 Chemistry 14C Fall 2015 Final Exam Part B Page 1 Uric acid is a normal metabolic product derived from purine nucleosides. Gout is a painful arthritic condition in which excess uric acid precipitates as
Chemistry 14C Fall 2015 Final Exam Part B Page 1 Uric acid is a normal metabolic product derived from purine nucleosides. Gout is a painful arthritic condition in which excess uric acid precipitates as
Chapter 2 Polar Covalent Bonds; Acids and Bases. Chapter Outline
 rganic Chemistry 9th Edition McMurry SLUTINS MANUAL Full clear download at: https://testbankreal.com/download/organic-chemistry-9th-edition-mcmurrysolutions-manual/ rganic Chemistry 9th Edition McMurry
rganic Chemistry 9th Edition McMurry SLUTINS MANUAL Full clear download at: https://testbankreal.com/download/organic-chemistry-9th-edition-mcmurrysolutions-manual/ rganic Chemistry 9th Edition McMurry
Receptor Based Drug Design (1)
 Induced Fit Model For more than 100 years, the behaviour of enzymes had been explained by the "lock-and-key" mechanism developed by pioneering German chemist Emil Fischer. Fischer thought that the chemicals
Induced Fit Model For more than 100 years, the behaviour of enzymes had been explained by the "lock-and-key" mechanism developed by pioneering German chemist Emil Fischer. Fischer thought that the chemicals
Patrick: An Introduction to Medicinal Chemistry 5e Chapter 01
 Questions Patrick: An Introduction to Medicinal Chemistry 5e 01) Which of the following molecules is a phospholipid? a. i b. ii c. iii d. iv 02) Which of the following statements is false regarding the
Questions Patrick: An Introduction to Medicinal Chemistry 5e 01) Which of the following molecules is a phospholipid? a. i b. ii c. iii d. iv 02) Which of the following statements is false regarding the
BSc and MSc Degree Examinations
 Examination Candidate Number: Desk Number: BSc and MSc Degree Examinations 2018-9 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes Marking
Examination Candidate Number: Desk Number: BSc and MSc Degree Examinations 2018-9 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes Marking
7.012 Problem Set 1. i) What are two main differences between prokaryotic cells and eukaryotic cells?
 ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
Resonance and M.O. View of Butadiene. Super-Conjugated or Aromatic p e - Systems
 Resonance and M.. View of Butadiene The different resonance forms of butadiene suggest p bonding character between the two central carbon atoms. 2 2 2 2 carbanion 2 2 carbocation The M.. view of butadiene
Resonance and M.. View of Butadiene The different resonance forms of butadiene suggest p bonding character between the two central carbon atoms. 2 2 2 2 carbanion 2 2 carbocation The M.. view of butadiene
CH MEDICINAL CHEMISTRY
 CH 458 - MEDICINAL CHEMISTRY SPRING 2011 M: 5:15pm-8 pm Sci-1-089 Prerequisite: Organic Chemistry II (Chem 254 or Chem 252, or equivalent transfer course) Instructor: Dr. Bela Torok Room S-1-132, Science
CH 458 - MEDICINAL CHEMISTRY SPRING 2011 M: 5:15pm-8 pm Sci-1-089 Prerequisite: Organic Chemistry II (Chem 254 or Chem 252, or equivalent transfer course) Instructor: Dr. Bela Torok Room S-1-132, Science
EXAMINATION 1 Chemistry 3A SID #:
 EXAMINATION hemistry A Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt February, 08 GSI (if you are taking hem AL): Please provide the following information if applicable.
EXAMINATION hemistry A Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt February, 08 GSI (if you are taking hem AL): Please provide the following information if applicable.
"Friendship is one mind in two bodies." Mencius
 California State Polytechnic University, Pomona 1 Fall, 2014 Midterm Exam Chem 314 Beauchamp Chem 314 Name Problem Points Credit 1. Nomenclature 30 2. 2D Lewis structures 20 3. 3D Structures, Formal Charge
California State Polytechnic University, Pomona 1 Fall, 2014 Midterm Exam Chem 314 Beauchamp Chem 314 Name Problem Points Credit 1. Nomenclature 30 2. 2D Lewis structures 20 3. 3D Structures, Formal Charge
Organic Chemistry FINAL EXAM A (250 points)
 UCSC, Binder ame Student ID # Section Day/Time rganic Chemistry FIAL EXAM A (250 points) D T BEGI TE EXAM R TUR TE PAGE UTIL ISTRUCTED T D S. In the meantime, please read the instructions below. In each
UCSC, Binder ame Student ID # Section Day/Time rganic Chemistry FIAL EXAM A (250 points) D T BEGI TE EXAM R TUR TE PAGE UTIL ISTRUCTED T D S. In the meantime, please read the instructions below. In each
Organic Chemistry FINAL EXAM A (250 points)
 UCSC, Binder ame Student ID # Section Day/Time rganic Chemistry FIAL EXAM A (250 points) D T BEGI TE EXAM R TUR TE PAGE UTIL ISTRUCTED T D S. In the meantime, please read the instructions below. In each
UCSC, Binder ame Student ID # Section Day/Time rganic Chemistry FIAL EXAM A (250 points) D T BEGI TE EXAM R TUR TE PAGE UTIL ISTRUCTED T D S. In the meantime, please read the instructions below. In each
Atoms. Smallest particles that retain properties of an element. Made up of subatomic particles: Protons (+) Electrons (-) Neutrons (no charge)
 Basic Chemistry Atoms Smallest particles that retain properties of an element Made up of subatomic particles: Protons (+) Electrons (-) Neutrons (no charge) Examples of Atoms electron proton neutron Hydrogen
Basic Chemistry Atoms Smallest particles that retain properties of an element Made up of subatomic particles: Protons (+) Electrons (-) Neutrons (no charge) Examples of Atoms electron proton neutron Hydrogen
Chapter 2. Chemical Basis of Life
 hapter 2 hemical Basis of Life opyright The McGrawill ompanies, Inc. Permission required for reproduction or display. Introduction: A. hemistry deals with the composition of matter and how it changes.
hapter 2 hemical Basis of Life opyright The McGrawill ompanies, Inc. Permission required for reproduction or display. Introduction: A. hemistry deals with the composition of matter and how it changes.
Chem 204. Mid-Term Exam I. July 21, There are 3 sections to this exam: Answer ALL questions
 Chem 204 Mid-Term Exam I July 21, 2009 Name: Answer Key Student ID: There are 3 sections to this exam: Answer ALL questions Section I: Multiple-Choice 20 questions, 2 pts each Section II: Fill-in-the-Blank
Chem 204 Mid-Term Exam I July 21, 2009 Name: Answer Key Student ID: There are 3 sections to this exam: Answer ALL questions Section I: Multiple-Choice 20 questions, 2 pts each Section II: Fill-in-the-Blank
Chemistry in Biology Section 1 Atoms, Elements, and Compounds
 Name Chemistry in Biology Section 1 Atoms, Elements, and Compounds Date Main Idea Details Scan the headings and boldfaced words in Section 1 of the chapter. Predict two things that you think might be discussed.
Name Chemistry in Biology Section 1 Atoms, Elements, and Compounds Date Main Idea Details Scan the headings and boldfaced words in Section 1 of the chapter. Predict two things that you think might be discussed.
Problems Points Credit
 Chem 201 Midterm Winter, 2018 Beauchamp ame Problems Points Credit 1. Functional Group omenclature (1 large structure) 30 2. Resonance, Formal Charge, Arrows 18 3. Properties of Atoms, Logic Arguments
Chem 201 Midterm Winter, 2018 Beauchamp ame Problems Points Credit 1. Functional Group omenclature (1 large structure) 30 2. Resonance, Formal Charge, Arrows 18 3. Properties of Atoms, Logic Arguments
1.5 h; 100 points please print clearly Dr. Kathleen Nolta Signature UM ID # Problem Points Score GSI I 20 II 20 III 20 IV 17 V 23 Total 100
 hemistry 0 irst ame irst Examination Last ame. h; 00 points please print clearly Dr. athleen olta ignature UM ID # PRIT TE IRT LETTER YUR LAT AME ERE: Problem Points core GI I 0 II 0 III 0 IV 7 V Total
hemistry 0 irst ame irst Examination Last ame. h; 00 points please print clearly Dr. athleen olta ignature UM ID # PRIT TE IRT LETTER YUR LAT AME ERE: Problem Points core GI I 0 II 0 III 0 IV 7 V Total
Exam III. Please read through each question carefully, and make sure you provide all of the requested information.
 09-107 onors Chemistry ame Exam III Please read through each question carefully, and make sure you provide all of the requested information. 1. A series of octahedral metal compounds are made from 1 mol
09-107 onors Chemistry ame Exam III Please read through each question carefully, and make sure you provide all of the requested information. 1. A series of octahedral metal compounds are made from 1 mol
Chapter 02 The Chemical Basis of Life I: Atoms, Molecules, and Water
 Chapter 02 The Chemical Basis of Life I: Atoms, Molecules, and Water Multiple Choice Questions 1. The atomic number of an atom is A. the number of protons in the atom. B. the number of neutrons in the
Chapter 02 The Chemical Basis of Life I: Atoms, Molecules, and Water Multiple Choice Questions 1. The atomic number of an atom is A. the number of protons in the atom. B. the number of neutrons in the
BIOCHEMISTRY BIOCHEMISTRY INTRODUCTION ORGANIZATION? MATTER. elements into the order and appearance we now
 BIOCHEMISTRY MR. HULSE BVHS BIOLOGY MATTER Matter - anything that occupies space and has mass Lacked clarity and flow BIOCHEMISTRY INTRODUCTION Biochemistry study of chemical and physiological process
BIOCHEMISTRY MR. HULSE BVHS BIOLOGY MATTER Matter - anything that occupies space and has mass Lacked clarity and flow BIOCHEMISTRY INTRODUCTION Biochemistry study of chemical and physiological process
H 3 C. biotin dependent O O. C O + Pi 2 + ADP 3 + H H 2 C C SCoA malonyl-scoa
 1 EXAM F SCIETIFIC CULTUE CEMISTY BLEM 1: BISYTESIS F FATTY ACIDS The biosynthesis of fatty acids proceeds by lengthening of a hydrocarbon chain by condensation of two-carbon units derived from acetyl-coenzyme
1 EXAM F SCIETIFIC CULTUE CEMISTY BLEM 1: BISYTESIS F FATTY ACIDS The biosynthesis of fatty acids proceeds by lengthening of a hydrocarbon chain by condensation of two-carbon units derived from acetyl-coenzyme
Please read and sign the Honor Code statement below:
 CHEM 3311 Exam #2 Name Dr. Minger June 22, 2015 Please read and sign the Honor Code statement below: I pledge that on my honor, as a University of Colorado at Boulder student, I have neither given nor
CHEM 3311 Exam #2 Name Dr. Minger June 22, 2015 Please read and sign the Honor Code statement below: I pledge that on my honor, as a University of Colorado at Boulder student, I have neither given nor
CHEMISTRY 1A Fall 2010 Final Exam Key
 CHEMISTRY 1A Fall 2010 Final Exam Key YOU MIGHT FIND THE FOLLOWING USEFUL; 0.008314 kj H E ( n)rt R = K mol 0.00418 kj q C cal m w T g C H rxn = H f (products) H f (reactants) Electronegativities H 2.2
CHEMISTRY 1A Fall 2010 Final Exam Key YOU MIGHT FIND THE FOLLOWING USEFUL; 0.008314 kj H E ( n)rt R = K mol 0.00418 kj q C cal m w T g C H rxn = H f (products) H f (reactants) Electronegativities H 2.2
Hole s Human Anatomy and Physiology Tenth Edition. Chapter 2
 PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Tenth Edition Shier w Butler w Lewis Chapter 2 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Tenth Edition Shier w Butler w Lewis Chapter 2 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
Chapter 2 The Chemical Context of Life
 Chapter 2 The Chemical Context of Life Information in this chapter establishes a foundation for later discussion and elaboration of molecular-level events and processes in biological systems. Ensuring
Chapter 2 The Chemical Context of Life Information in this chapter establishes a foundation for later discussion and elaboration of molecular-level events and processes in biological systems. Ensuring
RIO TINTO AUSTRALIAN CHEMISTRY OLYMPIAD
 RI TIT AUSTRALIA CEMISTRY LYMPIAD FIAL EXAMIATI PART B 2000 Please note that this answer book will be photocopied when returned and then split so that answers are sent to the appropriate markers. For this
RI TIT AUSTRALIA CEMISTRY LYMPIAD FIAL EXAMIATI PART B 2000 Please note that this answer book will be photocopied when returned and then split so that answers are sent to the appropriate markers. For this
CHEM 163 MIDTERM October 23, 2001 Dr. John C. Vederas
 CHEM 163 MIDTERM October 23, 2001 Dr. John C. Vederas I. Structure and Nomenclature - 56 Points - A. Draw structures for which names are given, or name the given structures by any correct (systematic or
CHEM 163 MIDTERM October 23, 2001 Dr. John C. Vederas I. Structure and Nomenclature - 56 Points - A. Draw structures for which names are given, or name the given structures by any correct (systematic or
2015 AP Biology Unit 2 PRETEST- Introduction to the Cell and Biochemistry
 Name: Class: _ Date: _ 2015 AP Biology Unit 2 PRETEST- Introduction to the Cell and Biochemistry Multiple Choice Identify the choice that best completes the statement or answers the question. 1) In what
Name: Class: _ Date: _ 2015 AP Biology Unit 2 PRETEST- Introduction to the Cell and Biochemistry Multiple Choice Identify the choice that best completes the statement or answers the question. 1) In what
CHEM 251 (4 credits): Description
 CHEM 251 (4 credits): Intermediate Reactions of Nucleophiles and Electrophiles (Reactivity 2) Description: An understanding of chemical reactivity, initiated in Reactivity 1, is further developed based
CHEM 251 (4 credits): Intermediate Reactions of Nucleophiles and Electrophiles (Reactivity 2) Description: An understanding of chemical reactivity, initiated in Reactivity 1, is further developed based
Enzymes are macromolecules (proteins) that act as a catalyst
 Chapter 8.4 Enzymes Enzymes speed up metabolic reactions by lowering energy barriers Even though a reaction is spontaneous (exergonic) it may be incredibly slow Enzymes cause hydrolysis to occur at a faster
Chapter 8.4 Enzymes Enzymes speed up metabolic reactions by lowering energy barriers Even though a reaction is spontaneous (exergonic) it may be incredibly slow Enzymes cause hydrolysis to occur at a faster
Principles of Drug Design
 Advanced Medicinal Chemistry II Principles of Drug Design Tentative Course Outline Instructors: Longqin Hu and John Kerrigan Direct questions and enquiries to the Course Coordinator: Longqin Hu I. Introduction
Advanced Medicinal Chemistry II Principles of Drug Design Tentative Course Outline Instructors: Longqin Hu and John Kerrigan Direct questions and enquiries to the Course Coordinator: Longqin Hu I. Introduction
Small-Molecule Kinetics
 Application Note No. 1 / September 1, 2014 Small-Molecule Kinetics Creoptix WAVE Small-Molecule Kinetics: Binding of Sulfonamides to Carbonic Anhydrase II Summary Label-free interaction analysis of biomolecules
Application Note No. 1 / September 1, 2014 Small-Molecule Kinetics Creoptix WAVE Small-Molecule Kinetics: Binding of Sulfonamides to Carbonic Anhydrase II Summary Label-free interaction analysis of biomolecules
An Introduction to Metabolism
 An Introduction to Metabolism I. All of an organism=s chemical reactions taken together is called metabolism. A. Metabolic pathways begin with a specific molecule, which is then altered in a series of
An Introduction to Metabolism I. All of an organism=s chemical reactions taken together is called metabolism. A. Metabolic pathways begin with a specific molecule, which is then altered in a series of
Drug Discovery. Zainab Al Kharusi Office: 33-10
 Drug Discovery Zainab Al Kharusi Office: 33-10 Objectives: To understand the processes of Drug Development To be able to develop a plan for drug discovery Reference: Patrick G L. An Introduction to Medicinal
Drug Discovery Zainab Al Kharusi Office: 33-10 Objectives: To understand the processes of Drug Development To be able to develop a plan for drug discovery Reference: Patrick G L. An Introduction to Medicinal
5. Stereochemical Analysis. 7. Dipole Moments and Inductive versus Resonance Effects. 8. Types of isomers from a given formula. 9. Physical Properties
 hem 201 Sample Midterm Beauchamp ame Problems Points redit 1. Functional Group omenclature (1 large structure) 2. Lewis Structures, Resonance, Formal harge 3. yclohexane onformations, 2 substituents, ewman
hem 201 Sample Midterm Beauchamp ame Problems Points redit 1. Functional Group omenclature (1 large structure) 2. Lewis Structures, Resonance, Formal harge 3. yclohexane onformations, 2 substituents, ewman
Phys 102 Lecture 2 Coulomb s Law & Electric Dipoles
 Phys 102 Lecture 2 Coulomb s Law & Electric Dipoles 1 Today we will... Get practice using Coulomb s law & vector addition Learn about electric dipoles Apply these concepts! Molecular interactions Polar
Phys 102 Lecture 2 Coulomb s Law & Electric Dipoles 1 Today we will... Get practice using Coulomb s law & vector addition Learn about electric dipoles Apply these concepts! Molecular interactions Polar
SYSTEMATIC SEARCH AND ANGIOTENSIN CONVERTING ENZYME INHIBITORS Ligand-Based SAR
 SYSTEMATIC SEARCH AND ANGIOTENSIN CONVERTING ENZYME INHIBITORS Ligand-Based SAR Garland R. Marshall Sapienza Universita di Roma October 27, 2014 The Pharmacophore Concept (Active-site Mapping) MOST THERAPEUTIC
SYSTEMATIC SEARCH AND ANGIOTENSIN CONVERTING ENZYME INHIBITORS Ligand-Based SAR Garland R. Marshall Sapienza Universita di Roma October 27, 2014 The Pharmacophore Concept (Active-site Mapping) MOST THERAPEUTIC
Small-Molecule Kinetics
 Application Note No. 1 / February 4, 2015 Small-Molecule Kinetics Creoptix WAVE Small-Molecule Kinetics: Binding of Sulfonamides to Carbonic Anhydrase II Summary Label-free interaction analysis of biomolecules
Application Note No. 1 / February 4, 2015 Small-Molecule Kinetics Creoptix WAVE Small-Molecule Kinetics: Binding of Sulfonamides to Carbonic Anhydrase II Summary Label-free interaction analysis of biomolecules
Chapter 2: Chemical Basis of Life
 Chapter 2: Chemical Basis of Life Chemistry is the scientific study of the composition of matter and how composition changes. In order to understand human physiological processes, it is important to understand
Chapter 2: Chemical Basis of Life Chemistry is the scientific study of the composition of matter and how composition changes. In order to understand human physiological processes, it is important to understand
Topics in the November 2014 Exam Paper for CHEM1102
 November 2014 Topics in the November 2014 Exam Paper for CEM1102 Click on the links for resources on each topic. 2014-N-2: 2014-N-3: 2014-N-4: 2014-N-5: 2014-N-6: 2014-N-7: 2014-N-8: 2014-N-9: 2014-N-10:
November 2014 Topics in the November 2014 Exam Paper for CEM1102 Click on the links for resources on each topic. 2014-N-2: 2014-N-3: 2014-N-4: 2014-N-5: 2014-N-6: 2014-N-7: 2014-N-8: 2014-N-9: 2014-N-10:
5. (6 pts) Show how the following compound can be synthesized from the indicated starting material:
 Exam 2 Name CHEM 212 1. (36 pts) Complete the following chemical reactions showing all major organic products; illustrate proper stereochemistry where appropriate. If no reaction occurs, indicate NR :
Exam 2 Name CHEM 212 1. (36 pts) Complete the following chemical reactions showing all major organic products; illustrate proper stereochemistry where appropriate. If no reaction occurs, indicate NR :
UNIT 1: BIOCHEMISTRY
 UNIT 1: BIOCHEMISTRY UNIT 1: Biochemistry Chapter 6.1: Chemistry of Life I. Atoms, Ions, and Molecules A. Living things consist of atoms of different elements 1. An atom is the smallest basic unit of matter
UNIT 1: BIOCHEMISTRY UNIT 1: Biochemistry Chapter 6.1: Chemistry of Life I. Atoms, Ions, and Molecules A. Living things consist of atoms of different elements 1. An atom is the smallest basic unit of matter
1) Which of the following represents the breaking of a noncovalent interaction? Topic: The Nature of Noncovalent Interactions
 Multiple Choice Questions 1) Which of the following represents the breaking of a noncovalent interaction? A) hydrolysis of an ester B) dissolving of salt crystals C) ionization of water D) decomposition
Multiple Choice Questions 1) Which of the following represents the breaking of a noncovalent interaction? A) hydrolysis of an ester B) dissolving of salt crystals C) ionization of water D) decomposition
Chem ORGANIC CHEMISTRY I
 ORGANIC CHEMISTRY I CHEM 221 /4 02 Final Examination April 20, 2005 0900-1200 Dr. Cerrie ROGERS x x periodic table & pk a data table provided non-programmable calculators allowed molecular model kits allowed
ORGANIC CHEMISTRY I CHEM 221 /4 02 Final Examination April 20, 2005 0900-1200 Dr. Cerrie ROGERS x x periodic table & pk a data table provided non-programmable calculators allowed molecular model kits allowed
A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility
 (P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
(P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
CHEM 110 Exam 2 - Practice Test 1 - Solutions
 CHEM 110 Exam 2 - Practice Test 1 - Solutions 1D 1 has a triple bond. 2 has a double bond. 3 and 4 have single bonds. The stronger the bond, the shorter the length. 2A A 1:1 ratio means there must be the
CHEM 110 Exam 2 - Practice Test 1 - Solutions 1D 1 has a triple bond. 2 has a double bond. 3 and 4 have single bonds. The stronger the bond, the shorter the length. 2A A 1:1 ratio means there must be the
2-4 Chemical Reactions and Enzymes
 2-4 Chemical Reactions and Enzymes Living things, as you have seen, are made up of chemical compounds-some simple and some complex. But chemistry isn t just what life is made of-chemistry is also what
2-4 Chemical Reactions and Enzymes Living things, as you have seen, are made up of chemical compounds-some simple and some complex. But chemistry isn t just what life is made of-chemistry is also what
BA, BSc, and MSc Degree Examinations
 Examination Candidate Number: Desk Number: BA, BSc, and MSc Degree Examinations 2017-8 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes
Examination Candidate Number: Desk Number: BA, BSc, and MSc Degree Examinations 2017-8 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes
Exam 3 Chem 3045x Friday, December 5, 1997
 Exam 3 Chem 3045x Friday, December 5, 1997 Instructions: This is a closed book examination. You may not use any notes, books or external materials during the course of the examination. Please print your
Exam 3 Chem 3045x Friday, December 5, 1997 Instructions: This is a closed book examination. You may not use any notes, books or external materials during the course of the examination. Please print your
The following question(s) were incorrectly answered.
 Name: Marcie Joseph Module: Cells & chemistry Test topic/animation: My animations/all animations Test type: Multiple choice Score: 48/50 Percent correct: 96% The following question(s) were incorrectly
Name: Marcie Joseph Module: Cells & chemistry Test topic/animation: My animations/all animations Test type: Multiple choice Score: 48/50 Percent correct: 96% The following question(s) were incorrectly
2 4 Chemical Reactions
 SECTION 2 4 Chemical Reactions ^^^^^ ^^^ ^^ tff'\f f**^*.tffn*f i***i i i «i f.' KEY CONCEPT Life depends on chemical reactions. Student text pages 50-53 Bonds break and form during chemical reactions.
SECTION 2 4 Chemical Reactions ^^^^^ ^^^ ^^ tff'\f f**^*.tffn*f i***i i i «i f.' KEY CONCEPT Life depends on chemical reactions. Student text pages 50-53 Bonds break and form during chemical reactions.
Amides, Amino acids and Chirality
 R hemistry A 432 Amides, Amino Acids & hirality Amides, Amino acids and hirality aming of Amides The amide functional group consists of a carbonyl group bonded to the nitrogen of an amine. Like amines,
R hemistry A 432 Amides, Amino Acids & hirality Amides, Amino acids and hirality aming of Amides The amide functional group consists of a carbonyl group bonded to the nitrogen of an amine. Like amines,
Introduction into Biochemistry. Dr. Mamoun Ahram Lecture 1
 Introduction into Biochemistry Dr. Mamoun Ahram Lecture 1 Course information Recommended textbooks Biochemistry; Mary K. Campbell and Shawn O. Farrell, Brooks Cole; 7 th edition Instructors Dr. Mamoun
Introduction into Biochemistry Dr. Mamoun Ahram Lecture 1 Course information Recommended textbooks Biochemistry; Mary K. Campbell and Shawn O. Farrell, Brooks Cole; 7 th edition Instructors Dr. Mamoun
Upon successful completion of this course, the student will be able to:
 CHEM 244 PRINCIPLES OF ORGANIC CHEMISTRY I FOR CHEMICAL ENGINEERING STUDENTS, COLLEGE OF ENGINEERING PRE-REQUISITES COURSE; CHEM 101 CREDIT HOURS; 2 (2+0) Dr. Mohamed El-Newehy Chemistry Department, College
CHEM 244 PRINCIPLES OF ORGANIC CHEMISTRY I FOR CHEMICAL ENGINEERING STUDENTS, COLLEGE OF ENGINEERING PRE-REQUISITES COURSE; CHEM 101 CREDIT HOURS; 2 (2+0) Dr. Mohamed El-Newehy Chemistry Department, College
Exam I Review Solution Set
 Exam I Review Solution Set Paul Bracher hem 30 Fall 2004 Exam I Problem 1 (refer to the Evans pk a table and Solvents and Solvent Effects in rganic hemistry by. Reichardt). Explain the trend in relative
Exam I Review Solution Set Paul Bracher hem 30 Fall 2004 Exam I Problem 1 (refer to the Evans pk a table and Solvents and Solvent Effects in rganic hemistry by. Reichardt). Explain the trend in relative
Chemistry 14C Winter 2017 Final Exam Part A Page 1
 Chemistry 14C Winter 2017 Final Exam Part A Page 1 Please use the backs of the exam pages for scratch space. Please do not use the exam margins for this purpose. The active site of an enzyme is a cleft
Chemistry 14C Winter 2017 Final Exam Part A Page 1 Please use the backs of the exam pages for scratch space. Please do not use the exam margins for this purpose. The active site of an enzyme is a cleft
THE UNIVERSITY OF MANITOBA. PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR
 THE UNIVERSITY OF MANITOBA 1 November 1, 2016 Mid-Term EXAMINATION PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR EXAMINATION:
THE UNIVERSITY OF MANITOBA 1 November 1, 2016 Mid-Term EXAMINATION PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR EXAMINATION:
3. Two unknown samples are found to have the same R f value under identical TLC conditions. Are they the same compound? Explain.
 I. Techniques in Organic Lab and TLC Analysis a. Thin-Layer Chromatography 2. A TLC plate displays the compound spot approximately 3.2 cm above the base line upon visualization; the solvent ran 4.1 cm
I. Techniques in Organic Lab and TLC Analysis a. Thin-Layer Chromatography 2. A TLC plate displays the compound spot approximately 3.2 cm above the base line upon visualization; the solvent ran 4.1 cm
It s the amino acids!
 Catalytic Mechanisms HOW do enzymes do their job? Reducing activation energy sure, but HOW does an enzyme catalysis reduce the energy barrier ΔG? Remember: The rate of a chemical reaction of substrate
Catalytic Mechanisms HOW do enzymes do their job? Reducing activation energy sure, but HOW does an enzyme catalysis reduce the energy barrier ΔG? Remember: The rate of a chemical reaction of substrate
