Efficient Agrobacterium-Mediated Transformation of Hybrid Poplar Populus davidiana Dode Populus bollena Lauche
|
|
|
- Esmond Dalton
- 6 years ago
- Views:
Transcription
1 Int. J. Mol. Sci. 2013, 14, ; doi: /ijms Article OPEN ACCESS International Journal of Molecular Sciences ISSN Efficient Agrobacterium-Mediated Transformation of Hybrid Poplar Populus davidiana Dode Populus bollena Lauche Xue Han 1, Shurong Ma 1, Xianghui Kong 1,2, Tetsuo Takano 3 and Shenkui Liu 1, * Key Laboratory of Saline-alkali Vegetation Ecology Restoration in Oil Field (SAVER), Ministry of Education, Alkali Soil Natural Environmental Science Center (ASNESC), Northeast Forestry University, Harbin Hexing Road, Harbin , China; s: hanxue_home@163.com (X.H.); msrbsc@163.com (S.M.); kxh29@126.com (X.K.) Institute of Microbiology, Heilongjiang Academy of Sciences, Harbin , China Asian Natural Environmental Science Center, University of Tokyo, Nishitokyo-shi, Tokyo , Japan; takano@anesc.u-tokyo.ac.jp * Author to whom correspondence should be addressed; shenkuiliu@nefu.edu.cn; Tel./Fax: Received: 7 November 2012; in revised form: 29 December 2012 / Accepted: 17 January 2013 / Published: 25 January 2013 Abstract: Poplar is a model organism for high in vitro regeneration in woody plants. We have chosen a hybrid poplar Populus davidiana Dode Populus bollena Lauche. By optimizing the Murashige and Skoog medium with (0.3 mg/l) 6-benzylaminopurine and (0.08 mg/l) naphthaleneacetic acid, we have achieved the highest frequency (90%) for shoot regeneration from poplar leaves. It was also important to improve the transformation efficiency of poplar for genetic breeding and other applications. In this study, we found a significant improvement of the transformation frequency by controlling the leaf age. Transformation efficiency was enhanced by optimizing the Agrobacterium concentration (OD 600 = ) and an infection time (20 30 min). According to transmission electron microscopy observations, there were more Agrobacterium invasions in the 30-day-old leaf explants than in 60-day-old and 90-day-old explants. Using the green fluorescent protein (GFP) marker, the expression of MD GFP fusion proteins in the leaf, shoot, and root of hybrid poplar P. davidiana Dode P. bollena Lauche was visualized for confirmation of transgene integration. Southern and Northern blot analysis also showed the integration of T-DNA into the genome and gene expression of transgenic plants. Our results suggest that younger leaves had higher transformation efficiency (~30%) than older leaves (10%).
2 Int. J. Mol. Sci. 2013, Keywords: regeneration; transformation; leaf age; poplar; Agrobacterium Abbreviations: AS, Acetosyringone; BA, 6-Benzylaminopurine; Cef, Cefotaxime sodium; GFP, Green fluorescent protein; Kan, Kanamycin sulfate; MS, Murashige and Skoog; NAA, Naphthaleneacetic acid; Rif, Rifampicin; TEM, Transmission electron microscopy; FOX, Full-length cdna overexpression. 1. Introduction Poplar has several exceptional qualities, such as a high capacity for vegetative propagation and a fast growth rate. It has been extensively used for the pulp and paper industry, reforestation of lowlands, and phytoremediation of contaminated soils. The micro-vegetative propagation and regeneration of poplar has been reported in recent decades. Different poplar genotypes have been used to perform regeneration experiments [1 10]. Growth and differentiation response is not only controlled by the cultural environment, but is also dependent upon the genotype [11]. In vitro regeneration has been attempted in several poplar species using different explants such as leaves [8,11 14], petioles [15], internodes [8,14], stems [5,16], roots [8,11], and shoot tips [17]. The leaf disc transformation method [18] has been widely used in plant genetic engineering. Parsons et al. [19] first reported the genetic transformation of poplar. This technology has been applied to various Populus species to improve their transformation efficiency [20 28]. Several factors were systematically analyzed to improve transformation efficiency, including poplar genotype, Agrobacterium tumefaciens strain for transformation, bacterial concentration, acetosyringone (AS) [20,29 32], and different explants including leaf discs and stems [23,33]. It is important to improve the transformation efficiency of poplar for genetic breeding and other applications. For example, the improvement of the transformation efficiency is crucial to build the full-length (fl) cdna overexpression (FOX) hunting system and T-DNA mutant system. FOX requires large numbers of dominant mutations that enable the comprehensive characterization of mutant phenotypes and the identification of functional genes. 15,000 transgenic lines were used for expressing Arabidopsis full-length (fl) cdnas [34], and >23,000 independent Arabidopsis transgenic lines for expressing rice fl-cdnas [35]; Nakamura et al. [36] generated approximately 12,000 FOX-rice lines. Busov et al. [37] also reported that from the 627 independent activation-tagged poplar lines, nine exhibited an obvious morphological phenotype that had never been seen among the thousands of transgenic poplars produced. We have chosen a hybrid poplar Populus davidiana Dode P. bollena Lauche to improve the transformation efficiency. Populus was planted in northeast China and has a well-characterized morphology with cold resistance features. We have optimized the Murashige and Skoog [38] medium with supplementary 6-Benzylaminopurine and Naphthaleneacetic acid to achieve a high regeneration efficiency of Populus. Subsequently, we attempted to maximize the transformation efficiency. Although several factors effecting transformation efficiency have been studied, there were no attention to explant age. In order to improve the transformation efficiency in this work, we optimized the conditions of the leaf explants age, as well as Agrobacterium concentration.
3 Int. J. Mol. Sci. 2013, Results and Discussion 2.1. Optimization of Poplar Shoot Regeneration Leaf explants of a hybrid poplar Populus davidiana Dode P. bollena Lauche were cultured in media with 0.1~0.5 mg/l BA and 0.01~0.1 mg/l NAA. There were no explants regenerated for 0.1 mg/l BA and mg/l NAA (Table 1). Low regeneration frequencies were induced using BA (0.1 mg/l) and NAA (0.05, 0.08, or 0.1 mg/l) and BA (0.5 mg/l) with NAA (0.01, 0.03, 0.05, 0.08, or 0.1 mg/l). Ninety percent of the explants regeneration shoots when BA (0.3 mg/l) and NAA (0.08 mg/l) were used. Regenerated shoots from leaf explants were shown in Figure 1a. Confalonieri et al. [20] reported the positive effect of BA and NAA on shoot regeneration in Populus nigra. To obtain shoot regeneration, leaf discs were cultivated in Petri dishes on MS medium with 0.5 mg/l BA and 0.2 mg/l NAA. The rooting experiments showed that new root formation and growth were promoted by NAA at any concentration, and root regeneration was 100%. However, NAA at 0.25 mg/l was the optimal rooting concentration because the growth status of roots was long and polyrhizal (Figure 1b). After the hardening process, 90% of the poplar plantlets survived acclimatization. Previously, various media were used to different genotypes and explants for shoot regeneration and rooting. IBA was used to induce roots for leaf of aspen and zeatin was used for shoot regeneration [24]. Adventitious shoots regenerated in a woody plant medium (WPM) supplemented with TDZ (Thidiazuron) from aspen stem explants [16]. Ferreira et al. [14] reported that adventitious shoot regeneration of P. euphratica from organogenic nodules of leaf explants was achieved within a range of concentrations of a-naphtalenacetic acid and 6-benzylaminopurine, at a fixed 2:1 ratio. Shoots of Populus ciliata Wall were regenerated at high frequencies from explants grown on Murashige and Skoog (MS) medium supplemented with 0.5 mg/l kinetin and 0.2 mg/l indole-3-acetic acid (IAA). Regenerated shoots developed roots in MS medium supplemented with 0.1 mg/l IAA [7]. Table 1. Effect of BA and NAA on shoot regeneration of leaf explants of Populus davidiana Dode Populus bollena Lauche. Medium BA (mg/l) NAA (mg/l) Mean No. shoots formed per explant Explant regeneration (%) ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± ± 3.85
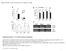
4 Int. J. Mol. Sci. 2013, Figure 1. High-frequency plant regeneration of Populus davidiana Dode Populus bollena Lauche. (a) Regenerated shoots from leaf explants; (b) Regenerated plantlet. Scale bar 1 cm Improvement of Genetic Transformation of Poplar The effect of Agrobacterium concentration on transformation frequency was shown in Figure 2. At OD 600 = 0.6, a lower transformation frequency was observed. As OD 600 ranging from 0.8 to 1.0, a higher transformation frequency was observed with little contamination. At OD 600 = 1.2, the transformation frequency was also reduced because of a higher contamination frequency. At OD 600 ranging from 0.8 to 1.0, the suitable incubation time of the explants in the bacterial suspension ranged from 20 min to 30 min (Figure 2). Previous study by Confalonieri et al. [20] optimized the production of transgenic plantlets from different P. nigra clones, Agrobacterium concentration ( ; cells/ml) were used. The results show that the optimal procedure involved dipping of leaf discs into a bacterial suspension ( cells/ml) for 20 min. Previous research has shown that the transformation efficiency increases in the presence of AS in the co-cultivation medium [32,39]. However, in the current experiment, the presence of AS during co-cultivation did not increase further the transformation efficiency (data not shown). Figure 2. Frequency of transformation of Populus davidiana Dode Populus bollena Lauche with different Agrobacterium concentrations and infection times.
5 Int. J. Mol. Sci. 2013, In addition to Agrobacterium concentration, and infection time, the transformation frequency was also influenced by leaf age. We selected three different leaf age groups, 30-, 60-, and 90-day-old (Figure 3). The transformation frequency of the 30-day-old leaves was more than that of the 60-day-old and 90-day-old leaves (Figure 4). As the age of the sample leaf increased, the transformation frequency decreased (Figure 4d). The transformation frequency was 10% for the 90-day-old samples, while ~30% for the 30-day-old samples. Although several factors affecting transformation efficiency have been studied, there was no focus on explant age. A previous study by Smith and McCown [40] reported that juvenile sources of explant tissues have superior growth and regeneration potential compared with other sources. Civinova and Sladky [41] reported that juvenile explants have superior regeneration capacity and they are less lignified. Han et al. [22] reported that incompletely lignified explant sections of hybrid cottonwoods have also been efficiently transformed with Agrobacterium. Figure 3. Leaf age definition diagram. Leaf age was defined as 30-, 60-, and 90-day-old growth from a 5 mm leaf in length. Figure 4. Transgenic shoot on regeneration-selection medium containing 50 mg/l Kan and 200 mg/l Cef. (a) 90-day-old leaf; (b) 60-day-old leaf; (c) 30-day-old leaf. Scale bar 1 cm. (d) Transformation percentage per leaf age from more than 100 samples. Different genotypes may respond differently to the optimal treatment conditions, and genetic transformation efficiency would be changed. According to published works, transformation frequency appeared to be different: 4% 6% for Populus trichocarpa, genotype Nisqually-1 [33], 16% for
6 Int. J. Mol. Sci. 2013, Populus tremuloides [27], and 20% for Populus nigra var. italica [26]. To improve its utility for functional genomics research, mutant library construction using the FOX hunting system was necessary for an efficient means for transformation and regeneration. Using hybrid poplar Populus davidiana Dode P. bollena Lauche for transformation, the transformation frequency exceeded 30% by controlling the age of the leaf. Figure 5. Transmission electron micrographs of the different leaf ages with Agrobacterium invasion in intercellular spaces. Leaf explants were infected with Agrobacterium at OD 600 ranging from 0.8 to 1.0 and infected for 20 min to 30 min. The leaf explants were cut transversely. (a) and (b) Uninfected leaf as the control; (c) and (d) 30-day-old leaf explant; (e) and (f) 60-day-old leaf explant; (g) and (h) 90-day-old leaf explant. In c h, Agrobacterium invasion into the intercellular space was indicated by white circles and arrows. The magnified images of the boxes in the left column (a, c, e, g) were placed in the right column (b, d, f, h), respectively. Scale bar 20 μm.
7 Int. J. Mol. Sci. 2013, In order to understand the higher transformation ability in the younger leaf explants, the Agrobacterium invasion of various leaves were analyzed to confirm that leaf age was an important factor in Agrobacterium-mediated transformation. Figure 5a,b showed the magnified TEM image of the intercellular spaces with no Agrobacterium invasion. For 30-day-old leaf explants, some bacteria were scattered throughout the intercellular space (Figure 5c,d). For the 60-day-old leaf explants, the bacteria decreased slightly in number, and some of the intercellular space appeared empty (Figure 5e,f). For the 90-day-old leaf explants, almost all the intercellular space appeared clean and just a few bacteria were observed in the intercellular space (Figure 5g,h). These results demonstrated that there were Agrobacterium-mediated transgenic activities in the leaf explants, as well as the 30-day-old leaf explants contained more Agrobacterium than the 60-day-old and 90-day-old explants. The highest transformation efficiency was generated by the 30-day-old leaf explants (Figure 4d). The transformation frequencies were influenced by the amount of invasive bacteria. Younger leaf explants may have a higher capability of shoot regeneration. However, in our TEM observations, the younger leaf explants had higher Agrobacterium infection. The invasion behavior of Agrobacterium in plant cells and their effects on genetic transformation is interesting. Agrobacterium invasion into plant cells can be affected by plant tissue age [42]. Younger poplar leaves were more susceptible to Agrobacterium-mediated transformation. As leaf age increased, the susceptibility to genetic transformation by Agrobacterium invasion decreased. A possible reason for the observed differential response of Agrobacterium invasion in different tissues was because of changes in the cell wall composition at different physiologic and developmental stages. As the plant cells mature, their cell wall composition changes so that Agrobacterium-binding sites were gradually lost [43] Observations of Genetic Transformation We observed the genetic transformation further using a GFP label and Southern and Northern blot analysis. A plant transformation vector designed to express the MD GFP fusion protein was used to detect the GFP signals in transgenic poplar. There were no GFP signals in the non-transgenic plants, shoots (Figure 6c,d), or roots (Figure 6g,h). However, the GFP signals from the shoots (Figure 6a,b), roots (Figure 6e,f), and leaf epidermis (Figure 6i,j) of the transgenic plants were detected. These results demonstrated that the pbi121-md-gfp plasmid was introduced into hybrid poplar P. davidiana Dode P. bollena Lauche. Genomic DNA from seven of the different independent transgenic lines was subjected to Southern blot analysis. In line 1, non-specific DNA bands appeared as a result of the continuous distribution (line 1, Figure 7a), but all other lines contained the distinct bands. The banded MD GFP gene was associated with high molecular weight invaded transgenic DNA(T-DNA), and produced the diverse copies as shown in line 2~8 (Figure 7a). The T-DNA appeared to be randomly integrated into the poplar genome. For Northern blot analysis, all transgenic lines displayed a distinct band consistent with the predicted MD GFP mrna (lines 2~8, Figure 7b), whereas no hybridization signal was detected from the non-transgenic wild-type plant (line 1, Figure 7b). These results indicated that the MD GFP gene was successfully integrated and expressed in the seven transgenic poplar lines 2~8 (Figure 7b).
8 Int. J. Mol. Sci. 2013, Figure 6. Expression of the MD GFP fusion protein in transgenic poplar. Non-transgenic with no GFP signal in poplar shoots (c) and (d) and roots (g) and (h). GFP fluorescence in the shoots (a) and (b), roots (e) and (f), and leaf epidermis (i) and (j) of transgenics. Left column was the GFP fluorescence images and right column was the optical images. Scale bar (a h) 0.2 cm; (i j) 10 μm. GFP Bright
9 Int. J. Mol. Sci. 2013, Figure 7 (a) Southern blot analysis of transgenic poplar. Poplar DNA was digested with Bam HI, electrophoresed, and probed with DIG-labeled MD GFP gene. Molecular weight DNA markers were shown on the left. Lane 1: non-transgenic poplar (control), and Lanes 2~8: transgenic poplar lines; (b) Detection of the transgene by Northern blot analysis. Analysis of the exogenous MD GFP expression of the transgenic lines by Northern blot. Lane 1: non-transgenic poplar (control), and Lanes 2~8: transgenic poplar lines. 3. Experimental Section 3.1. Plant Material, Agrobacterium Strain, Probe, and Plasmid Populus davidiana Dode P. bollena Lauche leaf explants were obtained randomly from in vitro shoot cultures and then used for regeneration and transformation. Populus davidiana Dode P. bollena Lauche was hybrid by Xinjiang poplar as the male parent and aspen as female parent. The plasmid pbi121-md-gfp and DIG-labeled MD gene probe were kindly provided by Prof. Che (Northeast Agricultural University) and used as a marker for plant transformation in our laboratory. The plasmid pbi121-md-gfp with the CaMV 35S promoter was transferred into Agrobacterium tumefaciens EHA105 using electroporation method (Bio-Rad, Micropluser, Hercules, CA, USA) Establishment of Regeneration System for Poplar Murashige and Skoog (MS) medium [38] was supplemented with 6-benzylaminopurine (BA) at 0.1 mg/l to 0.5 mg/l and naphthaleneacetic acid (NAA) at 0.01 mg/l to 0.1 mg/l (Table 1). The leaves were cut into 1 cm 1 cm blades and were cultured adaxial. The leaves were transferred to fresh regeneration medium at 10-day intervals. The regenerated shoots (3 cm to 5 cm in length) were excised and rooted on MS medium containing NAA at 0.1, 0.2, 0.25, 0.3, or 0.4 mg/l. All media included 3% (w/v) sucrose and 0.8% (w/v) agar. The ph of the media was adjusted to 5.8 after adding the plant growth regulation, and then autoclaved at 121 C for 20 min. After 1 to 2 months, the root formation was lignified, and the well-developed plantlets were removed from the media. The plantlets were gently washed under running tap water to remove the
10 Int. J. Mol. Sci. 2013, adhering media. For the hardening process, the plantlets were transferred into plastic cups containing a mixture of sterile compost. The regeneration experiments were designed with 10 Petri dishes for shoot regeneration and 10 triangular flasks for rooting per treatment, and each dish contained at least six leaf explants per treatment for shoot regeneration analysis, and each flask contained three shoots per treatment for rooting frequency analysis. Experiments were done in triplicate. The cultures were incubated at 25 C ± 2 C under a 16 h photoperiod (45 μmol m 2 s 1 ) Plant Transformation Sample Cultivation Leaves were cut into 0.5 cm 0.5 cm blades, immersed in the bacterial suspension in Petri dishes, and inoculated for different durations. Leaf explants were then dried on sterile filter paper and cultured on the shoot regeneration medium without Kanamycin sulfate (Kan) and cefotaxime sodium (Cef) in the dark. After 48 h of co-cultivation, leaf explants were washed five to six times (20 min to 30 min each) in liquid co-cultivation medium containing 200 mg/l Cef, and then blotted dry on sterile filter paper. The explants were then transferred to shoot regeneration medium containing Kan and Cef for selection, and cultured at 25 C ± 2 C under a 16-h photoperiod (45 μmol m 2 s 1 ) for 4 to 6 weeks. The cultures were transferred to fresh selection medium at 15-day intervals until shoot differentiation. The shoots were then transferred to rooting medium containing Kan and Cef after regeneration selection. Approximately 100 leaf explants were used for each transformation experiment and replicated thrice. The transformation frequencies were calculated by dividing the number of Kan-resistant regenerated shoots with the number of infected explants Effects of Agrobacterium Concentration and Infection Time A single Agrobacterium colony was inoculated into 3 ml of liquid YEP (Yeast extract 10 g/l; Tryptone 10 g/l; NaCl 5 g/l) medium with 100 mg/l rifampicin (Rif) and 50 mg/l Kan, and then incubated at 28 C for 24 h with constant agitation (175 rpm). An additional 30 ml of YEP was added and cultured overnight in the same culture conditions until OD 600 = 1.2. After centrifuging at 5000 rpm for 5 min, the pellet was diluted to OD 600 = 0.6, 0.8, 1.0, or 1.2 for transformation. The leaf explants were incubated in the bacterial suspension at 10, 20, 30, or 40 min. The experiment was repeated thrice with around 100~130 leaf explants per Agrobacterium treatment Effects of Leaf Ages Leaf explant age (leaf age) was timed since it was 5mm in length. The interval durations were 30-day-old leaf, 60-day-old leaf, or 90-day-old leaf (Figure 3). The experiment was repeated thrice with around 100 leaf explants per treatment. The leaf explants used for transformation were also examined under transmission electron microscopy (TEM).
11 Int. J. Mol. Sci. 2013, Visualization of Genetic Transformation TEM Observations of Agrobacterium Infection The leaf explants used for transformation were washed five times for 15 s in 0.2 M phosphate buffer (ph = 6.8) containing 0.9% (w/v) NaCl to remove the Agrobacterium that were not firmly attached after co-cultivation. The leaf explants were then sampled for TEM (H-7650, Hitachi, Kyushu, Japan). The method was carried out following the reference of Williams et al. [44] Green Fluorescent Protein Fluorescence Observations in Transgenic Poplar The leaves, shoots, and roots were removed from the transgenic poplar and observed under the fluorescent microscope (Olympus, Nagano-ken, Japan) and using a laser-scanning confocal imaging system (Olympus Fluoview, FV500, Nagano-ken, Japan). The GFP fluorescent signals were detected at an emitting wavelength of 480 nm Southern and Northern Blot Analysis DNA and RNA were extracted from leaf samples of the non-transgenic poplar and 7 different independent transgenic lines poplar. Poplar genomic DNA was extracted using the procedure of Porebski et al. [45]. Total RNA were extracted using the RNeasy Mini Kit (Qiagen) according to the manufacturer s instructions. For Southern analysis, a total of 20 μg genomic DNA was digested with restriction enzyme BamHI at 37 C in a water bath overnight. The digested DNA fragments were then electrophoresed in 0.8% agarose gel. For Northern analysis, 10 μg total RNA was electrophoresed on a 1% formaldehyde agarose gel. DNA and RNA samples were hybridized with DIG-labeled MD gene probe, and samples blotted onto a Hybond N + - membrane (Amersham Pharmacia, Piscataway, NJ, USA). The membrane was hybridized and washed according to Sambrook et al. [46]. The signals were detected with CDP-Star detection reagent using a LAS-4000 plus image analyzer (Fuji Film, Tokyo, Japan). 4. Conclusions A hybrid poplar Populus davidiana Dode Populus bollena Lauche planted in northeast China with cold tolerance has been optimized for high regeneration efficiency (90%). Genetic transformation was improved in terms of the age of the leaf explants (30-day-old leaf), Agrobacterium concentration (OD 600 = ), and infection time (20 30 min). The transmission electron microscopy observations of Agrobacterium invasions in the leaf explants, the visualization of the green fluorescent protein marker, and Southern and Northern blot analyses supported that there was successful integration of T-DNA into the genome and gene expression of transgenic plants. Acknowledgments This work was supported by the State Forestry Administration 948 Program of China (No ) and Heilongjiang Provincial Program for Distinguished Young Scholars (JC200609) to S. Liu. We
12 Int. J. Mol. Sci. 2013, thank Daidi Che (Northeast Agricultural University) for kindly providing the plasmid pbi121-md-gfp and DIG-labeled MD gene probe. Conflict of Interest The authors declare no conflict of interest. References 1. Christie, C.B. Rapid propagation of aspens and silver poplars using tissue culture techniques. Proc. Int. Plant Prop. Soc. 1978, 28, De Block, M. Factors influencing the tissue culture and the Agrobacterium tumefaciens-mediated transformation of hybrid aspen and Poplar clones. Plant Physiol. 1990, 93, Son, S.H.; Hall, R.B. Multiple shoot regeneration from root organ cultures of Populus alba P. grandidentata. Plant Cell Tiss. Organ. Cult. 1990, 20, Tsai, C.J.; Podila, G.K.; Chiang, V.L. Agrobacterium-mediated transformation of quaking aspen (Populus tremuloides) and regeneration of transgenic plants. Plant Cell Rep. 1994, 14, Gozukirmizi, N.; Bajrovic, K.; Ipekqi, Z. Genotype differencies in direct plant regeneration from stem explants of Populus tremula in Turkey. J. For. Res. 1998, 3, Noel, N.; Leple, J.C.; Pilate, G. Optimization of in vitro micropropagation and regeneration for Populus interamericana and Populus euramericana hybrids (P. deltoides, P. trichocarpa, and P. nigra). Plant Cell Rep. 2002, 20, Thakur, D.K.; Srivastava, D.K. High-efficiency plant regeneration from leaf explants of male Himalayan poplar (Populus ciliata Wall). In Vitro Cell Dev. Biol. Plant 2006, 42, Yadav, R.; Arora, P.; Kumar, D.; Katyal, D.; Dilbaghi, N.; Chaudhury, A. High frequency direct plant regeneration from leaf, internode, and root segments of Eastern Cottonwood (Populus deltoides). Plant Biotechnol. Rep. 2009, 3, Huang, D.Q.; Dai, W.H. Direct regeneration from in vitro leaf and petiole tissues of Populus tremula Erecta. Plant Cell Tiss. Organ. Cult. 2011, 107, Harry, I.S.; Thrope, T.A. In vitro Culture of Forest Trees. In Plant Cell and Tissue Culture; Vasil, I.K., Thrope, T.A., Eds.; Kluwer Academic: Dordrecht, The Netherlands, 1994; pp Ahuja, M.R. Somatic cell differentiation and rapid clonal propagation of aspen. Silvae Genet. 1983, 32, Fillatti, J.J.; Sellmer J.; McCown, B.; Haissig, B.; Comai, L. Agrobacterium mediated transformation and regeneration of Populus. Mol. Gen. Genet. 1987, 206, Dai, W.H.; Cheng, Z.M.; Sargent, W. Plant regeneration and Agrobacterium-mediated transformation of two elite aspen hybrid clones from in vitro leaf tissues. In Vitro Cell Dev. Biol. Plant 2003, 39, Ferreira, S.; Batista, D.; Serrazina, S.; Pais, M.S. Morphogenesis induction and organogenic nodule differentiation in Populus euphratica Oliv. leaf explants. Plant Cell Tiss. Organ. Cult. 2009, 96, Thakur, A.K.; Sarita, S.; Srivastava, D.K. Plant regeneration and genetic transformation studies in petiole tissue of Himalayan poplar (Populus ciliata Wall). Curr. Sci. 2005, 89,
13 Int. J. Mol. Sci. 2013, Tzfira, T.; Jensen, C.S.; Vainstein, A.; Altman, A. Transformation and regeneration of transgenic aspen plants via shoot formation from stem explants. Physiol. Plant. 1997b, 99, Kang, B.; Osburn, L.; Kopsell, D.; Tuskan, G.A.; Cheng, Z.M. Micropropagation of Populus trichocarpa Nisqually-1 : The genotype deriving the Populus reference genome. Plant Cell Tiss. Organ. Cult. 2009, 99, Horsch, R.B.; Fry, J.E.; Hoffmann, N.L.; Eichholtz, D.; Rogers, S.G.; Fraley, R.T. A simple and general method for transferring genes into plants. Science 1985, 227, Parsons, T.J.; Sinkar, V.P.; Stettler, R.F.; Nester, E.W.; Gordon, M.P. Transformation of poplar by Agrobacterium tumefaciens. Nat. Biotech. 1986, 4, Confalonieri, M.; Balestrazzi, A.; Bisoffi, S. Genetic transformation of Populus nigra by Agrobacterium tumefaciens. Plant Cell Rep. 1994, 13, Tzfira, T.; Jensen, C.S.; Wang, W.X.; Zuker, A.; Vinocur, B.; Altman, A.; Vainstein, A. Transgenic Populus tremula: A step-by-step protocol for its Agrobacterium-mediated transformation. Plant Mol. Biol. Rep. 1997a, 15, Han, K.H.; Gordon, M.P.; Strauss, S.H. High-frequency transformation of cottonwoods (genus Populus) by Agrobacterium rhizogenes. Can. J. For. Res. 1997, 27, Han, K.H.; Meilan, R.; Ma, C.; Strauss, S.H. An Agrobacterium tumefaciens transformation protocol effective on a variety of cottonwood hybrids (genus Populus). Plant Cell Rep. 2000, 19, Zhan, X.C.; Kawai, S.; Katayama, Y.; Morohoshi, N. A new approach based on the leaf disc method for Agrobacterium mediated transformation and regeneration of aspen. Plant Sci. 1997, 123, Okumura, S.; Sawada, M.; Park, Y.W.; Takahisa, H.Y.S.; Shimamura, H.; Takase, H.; Tomizawa, K.I. Transformation of poplar (Populus alba) plastids and expression of foreign proteins in tree chloroplasts. Transgenic Res. 2006, 15, Nishiguchi, M.; Yoshida, K.; Mohri, T.; Igasaki, T.; Shinohara, K. An improved transformation system for Lombardy poplar (Populus nigra var. italica). J. For. Res. 2006, 11, Cseke, L.J.; Cseke, S.B.; Podila, G.K. High efficiency poplar transformation. Plant Cell Rep. 2007, 26, Yevtushenko, D.P.; Santosh, M. Efficient Agrobacterium-mediated transformation of commercial hybrid poplar Populus nigra L. P. maximowiczii A. Henry. Plant Cell Rep. 2010, 29, Meilan, R.; Ma, C. Poplar (Populus ssp.). In Methods in Molecular Biology, Agrobacterium Protocols, 2nd ed.; Wang, K., Ed.; Humana Press: Totowa, NJ, USA, 2006; Volume 344, pp Confalonieri, M.; Balestrazzi, A.; Bisoffi, S.; Cella, R. Factors affecting Agrobacterium tumefaciens-mediated transformation in several black poplar clones. Plant Cell Tiss. Organ. Cult. 1995, 43, Confalonieri, M.; Balestrazzi, A.; Cella, R. Genetic transformation of Populus deltoides and P.x euramericana clones using Agrobacterium tumefaciens. Plant Cell Tiss. Organ. Cult. 1997, 48, Li, X.Y.; Huang, F.H.; Gbur, E.E. Factors affecting transformation efficiency of poplar hybrid line NC5331 by Agrobacterium tumefaciens. J. Arkansas Acad. Sci. 1997, 51,
14 Int. J. Mol. Sci. 2013, Ma, C.P.; Strauss, S.H.; Meilan, R. Agrobacterium-mediated transformation of the genome sequenced poplar clone, Nisqually-1(Populus trichocarpa). Plant Mol. Biol. Rep. 2004, 22, Ichikawa, T.; Nakazawa, M.; Kawashima, M. The FOX hunting system: An alternative gain-of-function gene hunting technique. Plant J. 2006, 45, Kondou, Y.; Higuchi, M.; Takahashi, S. Systematic approaches to using the FOX hunting system to identify useful rice genes. Plant J. 2009, 57, Nakamura, H.; Hakata, M.; Amano, K.; Miyao, A. A genome-wide gain-of-function analysis of rice genes using the FOX-Hunting system. Plant Mol. Biol. 2007, 65, Busov, V.B.; Meilan, R.; Pearce, D.W.; Ma, C.; Rood, S.B.; Strauss, S.H. Activation tagging of a dominant gibberellin catabolism gene (GA 2-oxidase) from poplar that regulates tree stature. Plant Physiol. 2003, 132, Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant 1962, 15, Godwin, I.; Todd, G.; Brian, F.L.; Newbury, H.J. The effects of acetosyringone and ph on Agrobacterium-mediated transformation vary according to plant species. Plant Cell Rep. 1991, 9, Smith, M.A.L.; McCown, B.H. A comparison of source tissue for protoplast isolation from three woody plant species. Plant Sci. Lett. 1983, 28, Civinova, B.; Sladky, Z. Stimulation of the regeneration capacity of tree shoot segment explants in vitro. Biol. Plant 1990, 32, Graves, A.E.; Goldman, S.L.; Banks, S.W.; Graves, A.C.F. Scanning electron microscope studies of Agrobacterium tumefaciens attachment to Zea mays, Gladiolus sp., and Triticum aestivum. J. Bactriol. 1988, 170, Verma, A.; Nain, V.; Kumari, C.; Singh, S.K.; Lakshmi, N.M.; Ananda, K.P. Tissue specific response of Agrobacterium tumefaciens attachment to Sorghum bicolor (L) Moench. Physiol. Mol. Biol. Plants 2008, 14, Willians, D.; Carter, C.B. Transmission Electron Microscopy; Plenum. Press: New York, NY, USA, Porebski, L.; Bailey, L.; Baum, B. Modification of a CTAB DNA extraction protocol for plants containing high polysaccharide and polyphenol components. Plant Mol. Biol. Rep. 1997, 15, Sambrook, J.; Fritsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd ed.; Cold Spring Harbor Laboratory Press: New York, NY, USA, by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (
EXPRESSION AND STABILITY OF TRANSGENES IN ASPEN-POPULUS
 EXPRESSION AND STABILITY OF TRANSGENES IN ASPEN-POPULUS M.R. Ahuja and M. Fladung Abstract:-- We have employed Populus as a model system to investigate questions regarding stability and expression of foreign
EXPRESSION AND STABILITY OF TRANSGENES IN ASPEN-POPULUS M.R. Ahuja and M. Fladung Abstract:-- We have employed Populus as a model system to investigate questions regarding stability and expression of foreign
Expression of Viral Resistance in Transformed Petunia Plants Regenerated in Vitro
 Expression of Viral Resistance in Transformed Petunia Plants Regenerated in Vitro Meira Ziv and Mirit Gandelman The RH Smith Institute of Plant Science The Hebrew University of Jerusalem, Rehovot, PO Box
Expression of Viral Resistance in Transformed Petunia Plants Regenerated in Vitro Meira Ziv and Mirit Gandelman The RH Smith Institute of Plant Science The Hebrew University of Jerusalem, Rehovot, PO Box
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated
 Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
IPC TECHNICAL PAPER SERIES NUMBER 270 TRANSFORMATION AND FOREIGN GENE EXPRESSION IN MICROPROPAGATED SWEETGUM (LIQUIDAMBAR STYRACIFLUA L.
 7 THE INSTITUTE OF PAPER CHEMISTRY, APPLETON, WISCONSIN IPC TECHNICAL PAPER SERIES NUMBER 270 TRANSFORMATION AND FOREIGN GENE EXPRESSION IN MICROPROPAGATED SWEETGUM (LIQUIDAMBAR STYRACIFLUA L.) R. P. FEIRER
7 THE INSTITUTE OF PAPER CHEMISTRY, APPLETON, WISCONSIN IPC TECHNICAL PAPER SERIES NUMBER 270 TRANSFORMATION AND FOREIGN GENE EXPRESSION IN MICROPROPAGATED SWEETGUM (LIQUIDAMBAR STYRACIFLUA L.) R. P. FEIRER
IN VITRO RHIZOGENESIS IN PAPAYA (CARICA PAPAYA L.)
 J. Plant Develop. 20(2013): 51 55 IN VITRO RHIZOGENESIS IN PAPAYA (CARICA PAPAYA L.) Jaime A. TEIXEIRA DA SILVA 1,2 Abstract: The seeds of two papaya (Carica papaya L.) cultivars ('Rainbow' and 'Sunrise
J. Plant Develop. 20(2013): 51 55 IN VITRO RHIZOGENESIS IN PAPAYA (CARICA PAPAYA L.) Jaime A. TEIXEIRA DA SILVA 1,2 Abstract: The seeds of two papaya (Carica papaya L.) cultivars ('Rainbow' and 'Sunrise
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL
 GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
CALLUS INDUCTION AND SOMATIC EMBRYOGENESIS FROM MAIZE MATURE EMBRYOS (ZEA MAYS L.)
 Journal of Cell and Tissue Research Vol. 13(1) 3565-3569 (2013) (Available online at www.tcrjournals.com) ISSN: 0973-0028; E-ISSN: 0974-0910 Original Article CALLUS INDUCTION AND SOMATIC EMBRYOGENESIS
Journal of Cell and Tissue Research Vol. 13(1) 3565-3569 (2013) (Available online at www.tcrjournals.com) ISSN: 0973-0028; E-ISSN: 0974-0910 Original Article CALLUS INDUCTION AND SOMATIC EMBRYOGENESIS
GFP GAL bp 3964 bp
 Supplemental Data. Møller et al. (2009) Shoot Na + exclusion and increased salinity tolerance engineered by cell type-specific alteration of Na + transport in Arabidopsis Supplemental Figure 1. Salt-sensitive
Supplemental Data. Møller et al. (2009) Shoot Na + exclusion and increased salinity tolerance engineered by cell type-specific alteration of Na + transport in Arabidopsis Supplemental Figure 1. Salt-sensitive
Effect of Acetosyringone on Agrobacterium-mediated Transformation of Eustoma grandiflorum Leaf Disks
 JARQ 51 (4), 351-355 (2017) https://www.jircas.go.jp Improvement in Agrobacterium-mediated Transformation of Eustoma grandiflorum by Acetosyringone Effect of Acetosyringone on Agrobacterium-mediated Transformation
JARQ 51 (4), 351-355 (2017) https://www.jircas.go.jp Improvement in Agrobacterium-mediated Transformation of Eustoma grandiflorum by Acetosyringone Effect of Acetosyringone on Agrobacterium-mediated Transformation
Development of a protocol for Agrobacterium mediated transformation of Brassica oleraceae L var botrytis cv Early kunwari
 204; (3): 34-38 ISSN 232-922 EJBB 204; (3): 34-38 Received 20-0-204 Accepted: **--204 Development of a protocol for Agrobacterium mediated transformation of Brassica oleraceae L var botrytis cv Early kunwari
204; (3): 34-38 ISSN 232-922 EJBB 204; (3): 34-38 Received 20-0-204 Accepted: **--204 Development of a protocol for Agrobacterium mediated transformation of Brassica oleraceae L var botrytis cv Early kunwari
Induction of Haploid Callus from Isolated Microspores of Peony in vitro
 Plant & Cell Physiol. 22(2): 337-34 (98) Short communication Induction of Haploid Callus from Isolated Microspores of Peony in vitro Kanji Ono and Shuichi Harashima Department of Biology, Faculty of Science,
Plant & Cell Physiol. 22(2): 337-34 (98) Short communication Induction of Haploid Callus from Isolated Microspores of Peony in vitro Kanji Ono and Shuichi Harashima Department of Biology, Faculty of Science,
Development of genetic transformation protocol for selected elite clone of Populus deltoides
 Development of genetic transformation protocol for selected elite clone of Populus deltoides A Dissertation Submitted in partial fulfillment of the requirement for the award of the degree of Master of
Development of genetic transformation protocol for selected elite clone of Populus deltoides A Dissertation Submitted in partial fulfillment of the requirement for the award of the degree of Master of
Useful Propagation Terms. Propagation The application of specific biological principles and concepts in the multiplication of plants.
 Useful Propagation Terms Propagation The application of specific biological principles and concepts in the multiplication of plants. Adventitious Typically describes new organs such as roots that develop
Useful Propagation Terms Propagation The application of specific biological principles and concepts in the multiplication of plants. Adventitious Typically describes new organs such as roots that develop
Last time: Obtaining information from a cloned gene
 Last time: Obtaining information from a cloned gene Objectives: 1. What is the biochemical role of the gene? 2. Where and when is the gene expressed (transcribed)? 3. Where and when is the protein made?
Last time: Obtaining information from a cloned gene Objectives: 1. What is the biochemical role of the gene? 2. Where and when is the gene expressed (transcribed)? 3. Where and when is the protein made?
FAMILY AND CLONAL VARIATION IN SUSCEPTIBILITY OF PINUS RADIATA TO AGROBACTERIUM TUMEFACIENS IN RELATION TO IN VITRO SHOOT GROWTH RATE
 3 FAMILY AND CLONAL VARIATION IN SUSCEPTIBILITY OF PINUS RADIATA TO AGROBACTERIUM TUMEFACIENS IN RELATION TO IN VITRO SHOOT GROWTH RATE B. A. BERGMANN and A.-M. STOMP Department of Forestry, North Carolina
3 FAMILY AND CLONAL VARIATION IN SUSCEPTIBILITY OF PINUS RADIATA TO AGROBACTERIUM TUMEFACIENS IN RELATION TO IN VITRO SHOOT GROWTH RATE B. A. BERGMANN and A.-M. STOMP Department of Forestry, North Carolina
Efficient plant regeneration via somatic embryogenesis from anthers of Datura stramonium L.
 Available online http://www.ijat-rmutto.com Journal of Agricultural Technology 2010 Vol. ISSN 6(4): 1686-9141 741-745 Efficient plant regeneration via somatic embryogenesis from anthers of Datura stramonium
Available online http://www.ijat-rmutto.com Journal of Agricultural Technology 2010 Vol. ISSN 6(4): 1686-9141 741-745 Efficient plant regeneration via somatic embryogenesis from anthers of Datura stramonium
PEROXIDASE ACTIVITY IN EUSTOM GRANDIFLOR UM PLANTS TRANSFORMED BY AGROBACTERIUM RHIZOGENES
 PEROXIDASE ACTIVITY IN EUSTOM GRANDIFLOR UM PLANTS TRANSFORMED BY AGROBACTERIUM RHIZOGENES GABRIELA POPA', AURELIA BREZEANU~, C. PETRUTA CORNEA', J.P. BOE The effects of inoculation with wild type of Agrobacterium
PEROXIDASE ACTIVITY IN EUSTOM GRANDIFLOR UM PLANTS TRANSFORMED BY AGROBACTERIUM RHIZOGENES GABRIELA POPA', AURELIA BREZEANU~, C. PETRUTA CORNEA', J.P. BOE The effects of inoculation with wild type of Agrobacterium
23-. Shoot and root development depend on ratio of IAA/CK
 Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
The involvement of photosynthesis in inducing bud formation on excised leaf segments of Heloniopsis orientalis (Liliaceae)
 Plant & Cell Physiol. 19(5): 791-799 (1978) The involvement of photosynthesis in inducing bud formation on excised leaf of Heloniopsis orientalis (Liliaceae) Yukio Kato Biological Laboratory, Fukui University,
Plant & Cell Physiol. 19(5): 791-799 (1978) The involvement of photosynthesis in inducing bud formation on excised leaf of Heloniopsis orientalis (Liliaceae) Yukio Kato Biological Laboratory, Fukui University,
Received 23 July 2014; received in revised form 25 October 2014; accepted 14 November 2014.
 Journal of Tropical Agriculture 52 (2) : 149-153, 2014 149 Short Communication Genetic transformation in Artemesia annua L. for hairy root induction and enhancement of secondary metabolites V.M. Shaneeja*,
Journal of Tropical Agriculture 52 (2) : 149-153, 2014 149 Short Communication Genetic transformation in Artemesia annua L. for hairy root induction and enhancement of secondary metabolites V.M. Shaneeja*,
Towards the Ultimate Solution: Genetic Resistance to HLB in Commercial Citrus. Greening Summit Florida Citrus Growers Institute 2008
 Towards the Ultimate Solution: Genetic Resistance to HLB in Commercial Citrus Greening Summit Florida Citrus Growers Institute 2008 Jude Grosser University of Florida, Citrus Research and Education Center,
Towards the Ultimate Solution: Genetic Resistance to HLB in Commercial Citrus Greening Summit Florida Citrus Growers Institute 2008 Jude Grosser University of Florida, Citrus Research and Education Center,
Plant transformation
 Plant transformation Objectives: 1. What is plant transformation? 2. What is Agrobacterium? How and why does it transform plant cells? 3. How is Agrobacterium used as a tool in molecular genetics? References:
Plant transformation Objectives: 1. What is plant transformation? 2. What is Agrobacterium? How and why does it transform plant cells? 3. How is Agrobacterium used as a tool in molecular genetics? References:
THE DEVELOPMENT OF PLANT REGENERATION SYSTEMS FOR THE GENETIC IMPROVEMENT OF WALNUT. Walt Tu1ecke and Gale McGranahan
 THE DEVELOPMENT OF PLANT REGENERATION SYSTEMS FOR THE GENETIC IMPROVEMENT OF WALNUT Walt Tu1ecke and Gale McGranahan ABSTRACT The techniques and capability to regenerate asexual embryos from walnut cotyledon
THE DEVELOPMENT OF PLANT REGENERATION SYSTEMS FOR THE GENETIC IMPROVEMENT OF WALNUT Walt Tu1ecke and Gale McGranahan ABSTRACT The techniques and capability to regenerate asexual embryos from walnut cotyledon
Supplemental Data. Perrella et al. (2013). Plant Cell /tpc
 Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Horizontal gene transfer from trees to ectomycorrhizal fungi: Lessons from laboratory and host plant liberation experiments
 Horizontal gene transfer from trees to ectomycorrhizal fungi: Lessons from laboratory and host plant liberation experiments Dr. Uwe Nehls 1,2, Dr. Chi Zhang 1, Dr. Mika Tarkka 1, Andrea Bock 1 1: University
Horizontal gene transfer from trees to ectomycorrhizal fungi: Lessons from laboratory and host plant liberation experiments Dr. Uwe Nehls 1,2, Dr. Chi Zhang 1, Dr. Mika Tarkka 1, Andrea Bock 1 1: University
TrioMol Isolation Reagent
 TrioMol Isolation Reagent Technical Manual No. 0242 Version 06142007 I Description... 1 II Key Features... 1 III Storage..... 1 IV General Protocol Using Triomol Isolation Reagent 1 V Troubleshooting.
TrioMol Isolation Reagent Technical Manual No. 0242 Version 06142007 I Description... 1 II Key Features... 1 III Storage..... 1 IV General Protocol Using Triomol Isolation Reagent 1 V Troubleshooting.
TrioMol Isolation Reagent
 TrioMol Isolation Reagent Technical Manual No. 0242 Version 06142007 I Description... 1 II Key Features... 1 III Storage..... 1 IV General Protocol Using Triomol Isolation Reagent 1 V Troubleshooting.
TrioMol Isolation Reagent Technical Manual No. 0242 Version 06142007 I Description... 1 II Key Features... 1 III Storage..... 1 IV General Protocol Using Triomol Isolation Reagent 1 V Troubleshooting.
Supplemental Data. Wang et al. (2014). Plant Cell /tpc
 Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
Leafy head formation of the progenies of transgenic plants of Chinese cabbage with exogenous auxin genes
 Cell Research (2000),10, 151-160 Leafy head formation of the progenies of transgenic plants of Chinese cabbage with exogenous auxin genes HE YU KE*, WAN XIN XUE, YU DONG SUN, XU HONG YU, PING LIN LIU National
Cell Research (2000),10, 151-160 Leafy head formation of the progenies of transgenic plants of Chinese cabbage with exogenous auxin genes HE YU KE*, WAN XIN XUE, YU DONG SUN, XU HONG YU, PING LIN LIU National
Identification of factors involved in Xylem Cell Differentiation Aarush Mohit Mittal 1, 2
 Identification of factors involved in Xylem Cell Differentiation Aarush Mohit Mittal 1, 2 1 Department of Biological Sciences and Bio-Engineering, Indian Institute of Technology, Kanpur, India 2 Department
Identification of factors involved in Xylem Cell Differentiation Aarush Mohit Mittal 1, 2 1 Department of Biological Sciences and Bio-Engineering, Indian Institute of Technology, Kanpur, India 2 Department
Establishment of rapid propagation cultures of Agrobacterium rhizogenes mediated transgenic hairy roots in sugarbeet
 EUROPEAN ACADEMIC RESEARCH Vol. III, Issue 12/ March 2016 ISSN 2286-4822 www.euacademic.org Impact Factor: 3.4546 (UIF) DRJI Value: 5.9 (B+) Establishment of rapid propagation cultures of Agrobacterium
EUROPEAN ACADEMIC RESEARCH Vol. III, Issue 12/ March 2016 ISSN 2286-4822 www.euacademic.org Impact Factor: 3.4546 (UIF) DRJI Value: 5.9 (B+) Establishment of rapid propagation cultures of Agrobacterium
Transgenic plants over-expressing HbCuZnSOD cytosolic isoform are more tolerant to a water deficit
 Transgenic plants over-expressing HbCuZnSOD cytosolic isoform are more tolerant to a water deficit J. Leclercq, F. Martin, C. Sanier, A. Clément-Vidal, D. Fabre, G. Oliver, L. Lardet, A. Ayar, M. Peyramard
Transgenic plants over-expressing HbCuZnSOD cytosolic isoform are more tolerant to a water deficit J. Leclercq, F. Martin, C. Sanier, A. Clément-Vidal, D. Fabre, G. Oliver, L. Lardet, A. Ayar, M. Peyramard
POPLAR PLANTS THROUGH ANTHER CULTURE
 POPLAR PLANTS THROUGH ANTHER CULTURE Rong H. Ho, A. Yesoda Raj and Louis Zsuffa Ontario Ministry of Natural Resources Ontario Tree Improvement and Forest Biomass Institute Maple, Ontario LOJ 1E0 Abstract.--
POPLAR PLANTS THROUGH ANTHER CULTURE Rong H. Ho, A. Yesoda Raj and Louis Zsuffa Ontario Ministry of Natural Resources Ontario Tree Improvement and Forest Biomass Institute Maple, Ontario LOJ 1E0 Abstract.--
In vitro flowering and in vitro pollination: methods that will benefit the orchid industry
 In vitro flowering and in vitro pollination: methods that will benefit the orchid industry Kim Hor HEE, Hock Hin YEOH, Chiang Shiong LOH Department of Biological Sciences, National University of Singapore
In vitro flowering and in vitro pollination: methods that will benefit the orchid industry Kim Hor HEE, Hock Hin YEOH, Chiang Shiong LOH Department of Biological Sciences, National University of Singapore
Rapid, Efficient Production of Homozygous Transgenic Tobacco Plants with Agrobacterium tumefaciens: A Seed to Seed Protocol.
 1 Rapid, Efficient Production of Homozygous Transgenic Tobacco Plants with Agrobacterium tumefaciens: A Seed to Seed Protocol. Dane K. Fisher and Mark J. Guiltinan* Department of Horticulture, Intercollegiate
1 Rapid, Efficient Production of Homozygous Transgenic Tobacco Plants with Agrobacterium tumefaciens: A Seed to Seed Protocol. Dane K. Fisher and Mark J. Guiltinan* Department of Horticulture, Intercollegiate
Part II. Agrobacterium rhizogenes-mediated Gene Transfer
 Part II Agrobacterium rhizogenes-mediated Gene Transfer Introduction II Agrobacterium rhizogenes, a Natural Transformation System D. TEPFER Plant-microorganism interactions are based on exchanges of nutritional
Part II Agrobacterium rhizogenes-mediated Gene Transfer Introduction II Agrobacterium rhizogenes, a Natural Transformation System D. TEPFER Plant-microorganism interactions are based on exchanges of nutritional
Methods of isolation of Cucumis sativus and C. melo pollen grains and their utilization in in vitro pollination 1
 Methods of isolation of Cucumis sativus and C. melo pollen grains and their utilization in in vitro pollination 1 D. Skálová *, B. Navrátilová, and A. Lebeda * Palacký University, Faculty of Science, Department
Methods of isolation of Cucumis sativus and C. melo pollen grains and their utilization in in vitro pollination 1 D. Skálová *, B. Navrátilová, and A. Lebeda * Palacký University, Faculty of Science, Department
Callus induction and plant regeneration on optimization of the culture conditions in Jow Haw rice (Oryza sativa L.)
 Journal of Agricultural Technology 2016 Vol. 12(2):241-248 Available online http://www.ijat-aatsea.com ISSN 1686-9141 Callus induction and plant regeneration on optimization of the culture conditions in
Journal of Agricultural Technology 2016 Vol. 12(2):241-248 Available online http://www.ijat-aatsea.com ISSN 1686-9141 Callus induction and plant regeneration on optimization of the culture conditions in
Supplemental Data. Perea-Resa et al. Plant Cell. (2012) /tpc
 Supplemental Data. Perea-Resa et al. Plant Cell. (22)..5/tpc.2.3697 Sm Sm2 Supplemental Figure. Sequence alignment of Arabidopsis LSM proteins. Alignment of the eleven Arabidopsis LSM proteins. Sm and
Supplemental Data. Perea-Resa et al. Plant Cell. (22)..5/tpc.2.3697 Sm Sm2 Supplemental Figure. Sequence alignment of Arabidopsis LSM proteins. Alignment of the eleven Arabidopsis LSM proteins. Sm and
WALNUT ROOTSTOCK TRANSFORMATION AND REGENERATION FROM VEGETATIVE TISSUE
 WALNUT ROOTSTOCK TRANSFORMATION AND REGENERATION FROM VEGETATIVE TISSUE David Tricoli, Donald Griffey, Monica T. Britton, Sandra L. Uratsu, Charles A. Leslie, Gale H. McGranahan, Wes Hackett, Edwin L.
WALNUT ROOTSTOCK TRANSFORMATION AND REGENERATION FROM VEGETATIVE TISSUE David Tricoli, Donald Griffey, Monica T. Britton, Sandra L. Uratsu, Charles A. Leslie, Gale H. McGranahan, Wes Hackett, Edwin L.
RNAi Suppression of AGAMOUS-like Genes Causes Field Sterility in Populus
 RNAi Suppression of AGAMOUS-like Genes Causes Field Sterility in Populus Haiwei Lu and Steven H. Strauss Oregon State University Forest Tree Workshop PAG XXVI, San Diego, CA, 2018 The containment issue
RNAi Suppression of AGAMOUS-like Genes Causes Field Sterility in Populus Haiwei Lu and Steven H. Strauss Oregon State University Forest Tree Workshop PAG XXVI, San Diego, CA, 2018 The containment issue
E.Z.N.A. MicroElute Clean-up Kits Table of Contents
 E.Z.N.A. MicroElute Clean-up Kits Table of Contents Introduction... 2 Kit Contents... 3 Preparing Reagents/Storage and Stability... 4 Guideline for Vacuum Manifold... 5 MicroElute Cycle-Pure - Spin Protocol...
E.Z.N.A. MicroElute Clean-up Kits Table of Contents Introduction... 2 Kit Contents... 3 Preparing Reagents/Storage and Stability... 4 Guideline for Vacuum Manifold... 5 MicroElute Cycle-Pure - Spin Protocol...
Changes of alkaloids in Tibetan medicine Przewalskia tangutica during tissue culture and differentiation of its stem rapid propagation system
 386 1, 2, 3 1 1, 2, 3 1, 2, 3* 4 4 1 1. 818 2. 818 3. 149 4. 8116 Przewalskia tangutica 3 d 2 3 cm HPLC 4 MS 6-BA 2. mg/l NAA.5 mg/l 96.67% MS NAA.5 mg/l 71.67% HPLC 4 141.25 μg/g R282.21 A 253-267(215)2-386
386 1, 2, 3 1 1, 2, 3 1, 2, 3* 4 4 1 1. 818 2. 818 3. 149 4. 8116 Przewalskia tangutica 3 d 2 3 cm HPLC 4 MS 6-BA 2. mg/l NAA.5 mg/l 96.67% MS NAA.5 mg/l 71.67% HPLC 4 141.25 μg/g R282.21 A 253-267(215)2-386
In Situ Gelation-Induced Death of Cancer Cells Based on Proteinosomes
 Supporting information for In Situ Gelation-Induced Death of Cancer Cells Based on Proteinosomes Yuting Zhou, Jianmin Song, Lei Wang*, Xuting Xue, Xiaoman Liu, Hui Xie*, and Xin Huang* MIIT Key Laboratory
Supporting information for In Situ Gelation-Induced Death of Cancer Cells Based on Proteinosomes Yuting Zhou, Jianmin Song, Lei Wang*, Xuting Xue, Xiaoman Liu, Hui Xie*, and Xin Huang* MIIT Key Laboratory
Maria V. Yamburenko, Yan O. Zubo, Radomíra Vanková, Victor V. Kusnetsov, Olga N. Kulaeva, Thomas Börner
 ABA represses the transcription of chloroplast genes Maria V. Yamburenko, Yan O. Zubo, Radomíra Vanková, Victor V. Kusnetsov, Olga N. Kulaeva, Thomas Börner Supplementary data Supplementary tables Table
ABA represses the transcription of chloroplast genes Maria V. Yamburenko, Yan O. Zubo, Radomíra Vanková, Victor V. Kusnetsov, Olga N. Kulaeva, Thomas Börner Supplementary data Supplementary tables Table
Penghui Li, Beibei Chen, Gaoyang Zhang, Longxiang Chen, Qiang Dong, Jiangqi Wen, Kirankumar S. Mysore and Jian Zhao
 New Phytologist Supporting Information Regulation of anthocyanin and proanthocyanidin biosynthesis by Medicago truncatula bhlh transcription factor MtTT8 Penghui Li, Beibei Chen, Gaoyang Zhang, Longxiang
New Phytologist Supporting Information Regulation of anthocyanin and proanthocyanidin biosynthesis by Medicago truncatula bhlh transcription factor MtTT8 Penghui Li, Beibei Chen, Gaoyang Zhang, Longxiang
Supplemental Data. Chen and Thelen (2010). Plant Cell /tpc
 Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
INDUCTION OF IN VITRO FLOWERING OF INDONESIAN WILD ORCHID, Phalaenopsis amabilis (L.) Blume
 ISSN 2413-0877 Volume 2 (2015) 398-404 The 3rd International Conference on Biological Science 2013 (The 3rd ICBS-2013) INDUCTION OF IN VITRO FLOWERING OF INDONESIAN WILD ORCHID, Phalaenopsis amabilis (L.)
ISSN 2413-0877 Volume 2 (2015) 398-404 The 3rd International Conference on Biological Science 2013 (The 3rd ICBS-2013) INDUCTION OF IN VITRO FLOWERING OF INDONESIAN WILD ORCHID, Phalaenopsis amabilis (L.)
Effect of temperature on endogenous hormone levels and opposite phyllotaxy in maize leaf primordial
 Effect of temperature on endogenous hormone levels and opposite phyllotaxy in maize leaf primordial H. Ye, G.M. Han, Q. Ma, Y.Q. Tan, H.Y. Jiang, S.W. Zhu and B.J. Cheng School of Life Sciences, Anhui
Effect of temperature on endogenous hormone levels and opposite phyllotaxy in maize leaf primordial H. Ye, G.M. Han, Q. Ma, Y.Q. Tan, H.Y. Jiang, S.W. Zhu and B.J. Cheng School of Life Sciences, Anhui
Isolation of Total RNA and mrna from Plant Tissues
 Promega Notes Magazine Number 54, 1995, p.02 Isolation of Total RNA and mrna from Plant Tissues By: Isabel Murillo, Dora Raventos, Estelle Jaeck, Blanca San Segundo* Centro de Investigacion y Desarrollo
Promega Notes Magazine Number 54, 1995, p.02 Isolation of Total RNA and mrna from Plant Tissues By: Isabel Murillo, Dora Raventos, Estelle Jaeck, Blanca San Segundo* Centro de Investigacion y Desarrollo
1( ) 5, dist. 4 5, dist. 3 5, dist. 5 5, dist
 and plant regeneration protocols for Brassica napus // International Journal of agriculture & Biology. 2011. Vol. 13. P. 83 88. 10. Gamborg O. L., Miller R. A, Ojima K. Nutrient requirements of suspension
and plant regeneration protocols for Brassica napus // International Journal of agriculture & Biology. 2011. Vol. 13. P. 83 88. 10. Gamborg O. L., Miller R. A, Ojima K. Nutrient requirements of suspension
Supplementary Information
 Supplementary Information Supplementary figures % Occupancy 90 80 70 60 50 40 30 20 Wt tol-1(nr2033) Figure S1. Avoidance behavior to S. enterica was not observed in wild-type or tol-1(nr2033) mutant nematodes.
Supplementary Information Supplementary figures % Occupancy 90 80 70 60 50 40 30 20 Wt tol-1(nr2033) Figure S1. Avoidance behavior to S. enterica was not observed in wild-type or tol-1(nr2033) mutant nematodes.
Doubled haploid ramets via embryogenesis of haploid tissue cultures
 Doubled haploid ramets via embryogenesis of haploid tissue cultures Harry E. Iswandar 1, J. M. Dunwell 2, Brian P. Forster 3, Stephen P. C. Nelson 1,4 and Peter D. S. Caligari,3,4,5 ABSTRACT Tissue culture
Doubled haploid ramets via embryogenesis of haploid tissue cultures Harry E. Iswandar 1, J. M. Dunwell 2, Brian P. Forster 3, Stephen P. C. Nelson 1,4 and Peter D. S. Caligari,3,4,5 ABSTRACT Tissue culture
Nature Genetics: doi: /ng Supplementary Figure 1. The phenotypes of PI , BR121, and Harosoy under short-day conditions.
 Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Ti plasmid derived plant vector systems: binary and co - integrative vectors transformation process; regeneration of the transformed lines
 Ti plasmid derived plant vector systems: binary and co - integrative vectors transformation process; regeneration of the transformed lines Mitesh Shrestha Constraints of Wild type Ti/Ri-plasmid Very large
Ti plasmid derived plant vector systems: binary and co - integrative vectors transformation process; regeneration of the transformed lines Mitesh Shrestha Constraints of Wild type Ti/Ri-plasmid Very large
Supporting Information
 Supporting Information A Low-Temperature Solid-Phase Method to Synthesize Highly Fluorescent Carbon Nitride Dots with Tunable Emission Juan Zhou, Yong Yang, and Chun-yang Zhang* Single-Molecule Detection
Supporting Information A Low-Temperature Solid-Phase Method to Synthesize Highly Fluorescent Carbon Nitride Dots with Tunable Emission Juan Zhou, Yong Yang, and Chun-yang Zhang* Single-Molecule Detection
Supplementary Figure 1. Phenotype of the HI strain.
 Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
EVALUATION OF WILD JUGLANS SPECIES FOR CROWN GALL RESISTANCE
 EVALUATION OF WILD JUGLANS SPECIES FOR CROWN GALL RESISTANCE Ed Stover, Malendia Maccree, Malli Aradhya, Ali E. McClean, and Daniel A. Kluepfel INTRODUCTION Crown Gall disease of walnut is caused by the
EVALUATION OF WILD JUGLANS SPECIES FOR CROWN GALL RESISTANCE Ed Stover, Malendia Maccree, Malli Aradhya, Ali E. McClean, and Daniel A. Kluepfel INTRODUCTION Crown Gall disease of walnut is caused by the
Development of genotype independent cotton transformation protocol
 Development of genotype independent cotton transformation protocol Shyam Barampuram and Sergei Krasnyanski Department of Horticultural Science North Carolina State University What are our goals? Robust
Development of genotype independent cotton transformation protocol Shyam Barampuram and Sergei Krasnyanski Department of Horticultural Science North Carolina State University What are our goals? Robust
State Forest Research Institute, Post Box No. 159, Itanagar , India 1 Department of Botany, Rajiv Gandhi University, Itanagar , India
 Indian Journal of Biotechnology Vol 6, April 2007, pp. 256-261 Effects of different culture media on seed germination and subsequent in vitro development of protocorms of Hygrochilus parishii (Veith &
Indian Journal of Biotechnology Vol 6, April 2007, pp. 256-261 Effects of different culture media on seed germination and subsequent in vitro development of protocorms of Hygrochilus parishii (Veith &
** * * * Col-0 cau1 CAU1. Actin2 CAS. Actin2. Supplemental Figure 1. CAU1 affects calcium accumulation.
 Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
Magnetic Janus Nanorods for Efficient Capture, Separation. and Elimination of Bacteria
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Magnetic Janus Nanorods for Efficient Capture, Separation and Elimination of Bacteria Zhi-min
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Magnetic Janus Nanorods for Efficient Capture, Separation and Elimination of Bacteria Zhi-min
ELECTRONIC SUPPLEMENTARY INFORMATION (ESI) variable light emission created via direct ultrasonic exfoliation of
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 ELECTRONIC SUPPLEMENTARY INFORMATION (ESI) High quantum-yield luminescent MoS 2 quantum dots
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 ELECTRONIC SUPPLEMENTARY INFORMATION (ESI) High quantum-yield luminescent MoS 2 quantum dots
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach
 Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Morphology and photosynthetic enzyme activity of maize phosphoenolpyruvate carboxylase transgenic rice
 Morphology and photosynthetic enzyme activity of maize phosphoenolpyruvate carboxylase transgenic rice W.C. Li 1, J. Wang 1, Y.L. Sun 1, S.D. Ji 1 and S.W. Guo 2 1 College of Life Sciences, Henan Normal
Morphology and photosynthetic enzyme activity of maize phosphoenolpyruvate carboxylase transgenic rice W.C. Li 1, J. Wang 1, Y.L. Sun 1, S.D. Ji 1 and S.W. Guo 2 1 College of Life Sciences, Henan Normal
AMADEPA Association Martiniquaise pour le Developpement des Plantes Alimentaires
 AMADEPA Association Martiniquaise pour le Developpement des Plantes Alimentaires 29eme CONGRES ANNUEL ANNUAL MEETING REUNION ANNUAL Agriculture Intensive dans les Iles de la Caraibe : enjeux, contraintes
AMADEPA Association Martiniquaise pour le Developpement des Plantes Alimentaires 29eme CONGRES ANNUEL ANNUAL MEETING REUNION ANNUAL Agriculture Intensive dans les Iles de la Caraibe : enjeux, contraintes
Agrobacterium rhizo genes-mediated Transformation and Regeneration of Vinca minor L.
 Original Papers Plant Tissue Culture Letters, 11(3), 191-198 (1994) Agrobacterium rhizo genes-mediated Transformation and Regeneration of Vinca minor L. Nobukazu TANAKA*, Misato TAKAO and Tekeshi MATSUMOTO
Original Papers Plant Tissue Culture Letters, 11(3), 191-198 (1994) Agrobacterium rhizo genes-mediated Transformation and Regeneration of Vinca minor L. Nobukazu TANAKA*, Misato TAKAO and Tekeshi MATSUMOTO
Microrhizome and minirhizome production in three high yielding cultivars of ginger (Zingiber officinale Rosc.)
 ISSN: 2319-7706 Volume 2 Number 10 (2013) pp. 477-484 http://www.ijcmas.com Original Research Article Microrhizome and minirhizome production in three high yielding cultivars of ginger (Zingiber officinale
ISSN: 2319-7706 Volume 2 Number 10 (2013) pp. 477-484 http://www.ijcmas.com Original Research Article Microrhizome and minirhizome production in three high yielding cultivars of ginger (Zingiber officinale
Transitivity-dependent and transitivity-independent cell-to-cell movement of RNA
 Himber et al. Transitivity-dependent and transitivity-independent cell-to-cell movement of RNA silencing SUPPLEMENTAL MATERIAL Supplemental material 1. Short-range movement of GFP silencing affects a nearly
Himber et al. Transitivity-dependent and transitivity-independent cell-to-cell movement of RNA silencing SUPPLEMENTAL MATERIAL Supplemental material 1. Short-range movement of GFP silencing affects a nearly
EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA
 EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA Item Type text; Electronic Thesis Authors Bergstrand, Lauren Janel Publisher The University of Arizona. Rights Copyright is held by the author. Digital
EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA Item Type text; Electronic Thesis Authors Bergstrand, Lauren Janel Publisher The University of Arizona. Rights Copyright is held by the author. Digital
Title Allantoin by Inosine in Nutrient So. Author(s) Toshihiro; Yokoi, Daisuke; Osaki, M
 Title Rice Root Growth with Increasing in Allantoin by Inosine in Nutrient So Author(s) Tokuhisa, Dai; Okazaki, Keiki; Shin Toshihiro; Yokoi, Daisuke; Osaki, M Citation The Proceedings of the Internationa
Title Rice Root Growth with Increasing in Allantoin by Inosine in Nutrient So Author(s) Tokuhisa, Dai; Okazaki, Keiki; Shin Toshihiro; Yokoi, Daisuke; Osaki, M Citation The Proceedings of the Internationa
Advances in tissue culture propagation of compact oil palm clones in Costa Rica
 Advances in tissue culture propagation of compact oil palm clones in Costa Rica Nidia Guzman 1 and Francisco Peralta After 20 years of research, ASD has developed a reliable protocol for cloning oil palm
Advances in tissue culture propagation of compact oil palm clones in Costa Rica Nidia Guzman 1 and Francisco Peralta After 20 years of research, ASD has developed a reliable protocol for cloning oil palm
CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-15 PLANT GROWTH AND DEVELOPMENT
 CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-15 PLANT GROWTH AND DEVELOPMENT Root, stem leaves, flower, fruits and seeds arise in orderly manner in plants. The sequence of growth is as follows-
CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-15 PLANT GROWTH AND DEVELOPMENT Root, stem leaves, flower, fruits and seeds arise in orderly manner in plants. The sequence of growth is as follows-
The Effect of Different levels and kinds of Cytokinins on Buds proliferation of Iraqian Date Palm Cultiver (Barhi) In vitro
 The Effect of Different levels and kinds of Cytokinins on Buds proliferation of Iraqian Date Palm Cultiver (Barhi) In vitro A. A. H. Al-Khalisi Department of Biology, College of Education Ibn Al-Haitham,
The Effect of Different levels and kinds of Cytokinins on Buds proliferation of Iraqian Date Palm Cultiver (Barhi) In vitro A. A. H. Al-Khalisi Department of Biology, College of Education Ibn Al-Haitham,
Gene expression in prokaryotic and eukaryotic cells, Plasmids: types, maintenance and functions. Mitesh Shrestha
 Gene expression in prokaryotic and eukaryotic cells, Plasmids: types, maintenance and functions. Mitesh Shrestha Plasmids 1. Extrachromosomal DNA, usually circular-parasite 2. Usually encode ancillary
Gene expression in prokaryotic and eukaryotic cells, Plasmids: types, maintenance and functions. Mitesh Shrestha Plasmids 1. Extrachromosomal DNA, usually circular-parasite 2. Usually encode ancillary
SCIENCE ROAD TO GOLD. Part 1- Biology Paper 1 Cell Biology Triple Science
 SCIENCE ROAD TO GOLD Part 1- Biology Paper 1 Cell Biology Triple Science 1 Below is a checklist for everything you need to know for this topic 2 A. Cell structure part 1 Eukaryotes, prokaryotes and animal
SCIENCE ROAD TO GOLD Part 1- Biology Paper 1 Cell Biology Triple Science 1 Below is a checklist for everything you need to know for this topic 2 A. Cell structure part 1 Eukaryotes, prokaryotes and animal
Optimization of Immunoblot Protocol for Use with a Yeast Strain Containing the CDC7 Gene Tagged with myc
 OPTIMIZATION OF IMMUNOBLOT PROTOCOL 121 Optimization of Immunoblot Protocol for Use with a Yeast Strain Containing the CDC7 Gene Tagged with myc Jacqueline Bjornton and John Wheeler Faculty Sponsor: Anne
OPTIMIZATION OF IMMUNOBLOT PROTOCOL 121 Optimization of Immunoblot Protocol for Use with a Yeast Strain Containing the CDC7 Gene Tagged with myc Jacqueline Bjornton and John Wheeler Faculty Sponsor: Anne
In Vitro Polyploid Induction of Ophiopogon planiscapus. Dominic A. Gillooly, Darren H. Touchell and Thomas G. Ranney
 In Vitro Polyploid Induction of Ophiopogon planiscapus Dominic A. Gillooly, Darren H. Touchell and Thomas G. Ranney North Carolina State University, Departement of Horticultural Science Mountain Crop Improvement
In Vitro Polyploid Induction of Ophiopogon planiscapus Dominic A. Gillooly, Darren H. Touchell and Thomas G. Ranney North Carolina State University, Departement of Horticultural Science Mountain Crop Improvement
Construction of nanoantennas on the outer bacterial membrane
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Construction of nanoantennas on the outer bacterial membrane
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Construction of nanoantennas on the outer bacterial membrane
INFECTION OF PINUS RADIATA WITH AGROBACTERIUM RHIZOGENES AND LONG-TERM GROWTH OF DETACHED HAIRY ROOTS IN VITRO
 11 INFECTION OF PINUS RADIATA WITH AGROBACTERIUM RHIZOGENES AND LONG-TERM GROWTH OF DETACHED HAIRY ROOTS IN VITRO B. A. BERGMANN*, J. DUKES, and A-M. STOMP Department of Forestry, North Carolina State
11 INFECTION OF PINUS RADIATA WITH AGROBACTERIUM RHIZOGENES AND LONG-TERM GROWTH OF DETACHED HAIRY ROOTS IN VITRO B. A. BERGMANN*, J. DUKES, and A-M. STOMP Department of Forestry, North Carolina State
Reproduction, Seeds and Propagation
 Reproduction, Seeds and Propagation Diploid (2n) somatic cell Two diploid (2n) somatic cells Telophase Anaphase Metaphase Prophase I One pair of homologous chromosomes (homologues) II Homologues condense
Reproduction, Seeds and Propagation Diploid (2n) somatic cell Two diploid (2n) somatic cells Telophase Anaphase Metaphase Prophase I One pair of homologous chromosomes (homologues) II Homologues condense
Transient expression of β-glucuronidase reporter gene in Agrobacterium-inoculated shoots of various teak clones
 African Journal of Biotechnology Vol. 8 (10), pp. 2143-2150, 18 May, 2009 Available online at http://www.academicjournals.org/ajb ISSN 1684 5315 2009 Academic Journals Full Length Research Paper Transient
African Journal of Biotechnology Vol. 8 (10), pp. 2143-2150, 18 May, 2009 Available online at http://www.academicjournals.org/ajb ISSN 1684 5315 2009 Academic Journals Full Length Research Paper Transient
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/9/452/ra106/dc1 Supplementary Materials for Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots Hyo-Jun Lee, Jun-Ho
www.sciencesignaling.org/cgi/content/full/9/452/ra106/dc1 Supplementary Materials for Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots Hyo-Jun Lee, Jun-Ho
Horticulture 201H Spring, 2002 Exam 2 Name:
 Horticulture 201H Spring, 2002 Exam 2 Name: Section 1. In the space to the left of the statements below, write the word(s) that best fit the definition or description. (20 pts) Vegetative reproduction
Horticulture 201H Spring, 2002 Exam 2 Name: Section 1. In the space to the left of the statements below, write the word(s) that best fit the definition or description. (20 pts) Vegetative reproduction
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10244 a O07391_MYCAV/127-243 NLPC_HAEIN/80-181 SPR_SHIFL/79-183 P74160_SYNY3/112-245 O24914_HELPY/301-437 Q51835_PORGI/68-178 DPP6_BACSH/163-263 YKFC_BACSU/185-292 YDHO_ECOLI/153-263
doi:10.1038/nature10244 a O07391_MYCAV/127-243 NLPC_HAEIN/80-181 SPR_SHIFL/79-183 P74160_SYNY3/112-245 O24914_HELPY/301-437 Q51835_PORGI/68-178 DPP6_BACSH/163-263 YKFC_BACSU/185-292 YDHO_ECOLI/153-263
KnowIT Questions AQA GCSE Cell Biology
 A. Cell structure part 1 Eukaryotes, prokaryotes and animal and plant cells 1. Where is the genetic material in a prokaryotic cell? 2. Where is the genetic material in a eukaryotic cell? 3. Complete the
A. Cell structure part 1 Eukaryotes, prokaryotes and animal and plant cells 1. Where is the genetic material in a prokaryotic cell? 2. Where is the genetic material in a eukaryotic cell? 3. Complete the
tumefaciens Attachment to Zea mays, Gladiolus sp.,
 JOURNAL OF BACTERIOLOGY, May 1988, p. 2395-2400 Vol. 170, No. 5 0021-9193/88/052395-06$02.00/0 Copyright 1988, American Society for Microbiology Scanning Electron Microscope Studies of Agrobacterium tumefaciens
JOURNAL OF BACTERIOLOGY, May 1988, p. 2395-2400 Vol. 170, No. 5 0021-9193/88/052395-06$02.00/0 Copyright 1988, American Society for Microbiology Scanning Electron Microscope Studies of Agrobacterium tumefaciens
Production of Haploid and Doubled Haploid Plants from Anther-derived Callus of Lilium formosanum
 Production of Haploid and Doubled Haploid Plants from Anther-derived Callus of Lilium formosanum D.-S. Han and Y. Niimi Faculty of Agriculture, Niigata University 2-8050 Ikarashi, Niigata 950-2181 Japan
Production of Haploid and Doubled Haploid Plants from Anther-derived Callus of Lilium formosanum D.-S. Han and Y. Niimi Faculty of Agriculture, Niigata University 2-8050 Ikarashi, Niigata 950-2181 Japan
Plant Propagation PLS 3221/5222
 Plant Propagation PLS 3221/5222 Dr. Sandra Wilson Dr. Mack Thetford Chapter 2 Introduction to the Biology of Plant Propagation -A review- 1 5. Plant Hormones and Plant development Phytohormones Nt Naturally
Plant Propagation PLS 3221/5222 Dr. Sandra Wilson Dr. Mack Thetford Chapter 2 Introduction to the Biology of Plant Propagation -A review- 1 5. Plant Hormones and Plant development Phytohormones Nt Naturally
Plant and animal cells (eukaryotic cells) have a cell membrane, cytoplasm and genetic material enclosed in a nucleus.
 4.1 Cell biology Cells are the basic unit of all forms of life. In this section we explore how structural differences between types of cells enables them to perform specific functions within the organism.
4.1 Cell biology Cells are the basic unit of all forms of life. In this section we explore how structural differences between types of cells enables them to perform specific functions within the organism.
Mag-Bind Soil DNA Kit. M preps M preps M preps
 Mag-Bind Soil DNA Kit M5635-00 5 preps M5635-01 50 preps M5635-02 200 preps January 2013 Mag-Bind Soil DNA Kit Table of Contents Introduction and Overview...2 Kit Contents/Storage and Stability...3 Preparing
Mag-Bind Soil DNA Kit M5635-00 5 preps M5635-01 50 preps M5635-02 200 preps January 2013 Mag-Bind Soil DNA Kit Table of Contents Introduction and Overview...2 Kit Contents/Storage and Stability...3 Preparing
Journal of Agricultural Technology
 Study on the growth patterns of transformed carrot hairy roots in an optimized system Y.R. Danesh *, E. Mohammadi Goltapeh and A. Alizadeh Department of Plant Pathology, College of Agriculture, Tarbiat
Study on the growth patterns of transformed carrot hairy roots in an optimized system Y.R. Danesh *, E. Mohammadi Goltapeh and A. Alizadeh Department of Plant Pathology, College of Agriculture, Tarbiat
Growth and development of Arabidopsis thaliana under single-wavelength red
 1 Supplementary Information 2 3 4 Growth and development of Arabidopsis thaliana under single-wavelength red and blue laser light 5 6 7 8 Authors Amanda Ooi 1 *, Aloysius Wong 1 *, Tien Khee Ng 2, Claudius
1 Supplementary Information 2 3 4 Growth and development of Arabidopsis thaliana under single-wavelength red and blue laser light 5 6 7 8 Authors Amanda Ooi 1 *, Aloysius Wong 1 *, Tien Khee Ng 2, Claudius
In-gel digestion of immunoprecipitated proteins separated by SDS-PAGE
 In-gel digestion of immunoprecipitated proteins separated by SDS-PAGE (Lamond Lab / April 2008)! Perform all the pipetting steps in a laminar flow hood. We routinely do our digestions in our TC room hoods.
In-gel digestion of immunoprecipitated proteins separated by SDS-PAGE (Lamond Lab / April 2008)! Perform all the pipetting steps in a laminar flow hood. We routinely do our digestions in our TC room hoods.
Transcription and Translation involved in Uniparental Inheritance and Cell Nuclei Fusion in the Chlamydomonas reinhardtii Zygote
 1997 The Japan Mendel Society Cytologia 62: 427-433, 1997 Transcription and Translation involved in Uniparental Inheritance and Cell Nuclei Fusion in the Chlamydomonas reinhardtii Zygote Lena Suzuki 1,
1997 The Japan Mendel Society Cytologia 62: 427-433, 1997 Transcription and Translation involved in Uniparental Inheritance and Cell Nuclei Fusion in the Chlamydomonas reinhardtii Zygote Lena Suzuki 1,
ydci GTC TGT TTG AAC GCG GGC GAC TGG GCG CGC AAT TAA CGG TGT GTA GGC TGG AGC TGC TTC
 Table S1. DNA primers used in this study. Name ydci P1ydcIkd3 Sequence GTC TGT TTG AAC GCG GGC GAC TGG GCG CGC AAT TAA CGG TGT GTA GGC TGG AGC TGC TTC Kd3ydcIp2 lacz fusion YdcIendP1 YdcItrgP2 GAC AGC
Table S1. DNA primers used in this study. Name ydci P1ydcIkd3 Sequence GTC TGT TTG AAC GCG GGC GAC TGG GCG CGC AAT TAA CGG TGT GTA GGC TGG AGC TGC TTC Kd3ydcIp2 lacz fusion YdcIendP1 YdcItrgP2 GAC AGC
NORTHERN ILLINOIS UNIVERSITY. Screening of Chemical Libraries in Search of Inhibitors of Aflatoxin Biosynthesis. A Thesis Submitted to the
 NORTHERN ILLINOIS UNIVERSITY Screening of Chemical Libraries in Search of Inhibitors of Aflatoxin Biosynthesis A Thesis Submitted to the University Honors Program In Partial Fulfillment of the Requirements
NORTHERN ILLINOIS UNIVERSITY Screening of Chemical Libraries in Search of Inhibitors of Aflatoxin Biosynthesis A Thesis Submitted to the University Honors Program In Partial Fulfillment of the Requirements
AGROBACTERIUM MEDIATED GENETIC TRANSFORMATION OF THE RESURRECTION PLANT HABERLEA RHODOPENSIS FRIV. Abstract. Introduction
 10 Bulgarian Journal of Agricultural Science, 19 (2) 2013, 10 14 Agricultural Academy AGROBACTERIUM MEDIATED GENETIC TRANSFORMATION OF THE RESURRECTION PLANT HABERLEA RHODOPENSIS FRIV. G. PETROVA* and
10 Bulgarian Journal of Agricultural Science, 19 (2) 2013, 10 14 Agricultural Academy AGROBACTERIUM MEDIATED GENETIC TRANSFORMATION OF THE RESURRECTION PLANT HABERLEA RHODOPENSIS FRIV. G. PETROVA* and
