Highwire Regulates Synaptic Growth in Drosophila
|
|
|
- Irma Wheeler
- 5 years ago
- Views:
Transcription
1 Neuron, Vol. 26, , May, 2000, Copyright 2000 by Cell Press Highwire Regulates Synaptic Growth in Drosophila Hong I. Wan,* Aaron DiAntonio,* Richard D. Fetter,* Kendra Bergstrom,* Roland Strauss, and Corey S. Goodman* * Howard Hughes Medical Institute Department of Molecular and Cell Biology University of California, Berkeley Berkeley, California Theodor-Boveri-Institut fuer Biowissenschaften LS Genetik Am Hubland D Würzburg Germany Summary The formation, stabilization, and growth of synaptic connections are dynamic and highly regulated pro- cesses. The glutamatergic neuromuscular junction (NMJ) in Drosophila grows new boutons and branches throughout larval development. A primary walking be- havior screen followed by a secondary anatomical screen led to the identification of the highwire (hiw) gene. In hiw mutants, the specificity of motor axon pathfinding and synapse formation appears normal. However, NMJ synapses grow exuberantly and are greatly expanded in both the number of boutons and the extent and length of branches. These synapses appear normal ultrastructurally but have reduced quantal content physiologically. hiw encodes a large protein found at presynaptic terminals. Within presyn- aptic terminals, HIW is localized to the periactive zone surrounding active zones; Fasciclin II (Fas II), which also controls synaptic growth, is found at the same location. Introduction Once synapses form, most of them are not static but continue to expand and retract throughout the animal s life. What is the molecular machinery that controls synaptic growth? How are synaptic activity and signaling translated into expansion or retraction of synaptic terminals? We reasoned that synaptic growth, like other de- velopmental processes, is likely to be under both posi- tive and negative control. To identify such positive and negative regulators, we decided to take a genetic approach in Drosophila, employing genetic screens to un- cover mutants with changes in the rate and extent of growth of the synapse. The most accessible synapse in Drosophila for such a genetic approach is the neuromuscular junction (NMJ). In contrast to the fairly static NMJ in mammals, the To whom correspondence should be addressed ( goodman@ uclink4.berkeley.edu). Present address: Department of Molecular Biology and Pharmacology, Washington University School of Medicine, St. Louis, Missouri Drosophila NMJ is a dynamic structure, growing new boutons and sprouting new branches during larval development. The Drosophila NMJ shares several impor- tant features with central excitatory synapses in the vertebrate brain. The Drosophila NMJ is a glutamatergic synapse and has ionotropic glutamate receptors homologous to those of humans (Petersen et al., 1997). This synapse does not have a conspicuous basal lamina separating the two sides, as does the vertebrate NMJ, but rather appears by ultrastructural analysis to consist of close membrane apposition of pre- and postsynaptic cells. Finally, this synapse is organized into a series of boutons and branches that can be added or eliminated during development and plastic changes. As the Drosophila larva develops from first to third instar over a period of several days, the surface area of the postsynaptic muscle increases as much as 100-fold. During this developmental period, there is a concomitant growth of the presynaptic nerve terminal, resulting in up to a 10-fold increase in the number of boutons and a 10-fold increase in the number of active zones per bouton (Schuster et al., 1996a). A number of activity-dependent and activity-indepen- dent mechanisms appear to control the dramatic larval growth of this synapse. Long-term activity-dependent synaptic plasticity was first demonstrated at this synapse by genetic manipulations that alter neuronal activ- ity and camp concentrations. K channel mutants, such as ether a go-go Shaker (eag Sh), increase neuronal activity, and synaptic structure and branching (Budnik et al., 1990). In addition, dunce (dnc) mutants, deficient in camp phosphodiesterase, increase synaptic structure (Zhong and Wu, 1991; Zhong et al., 1992). This activity- dependent synaptic growth is controlled in part by the regulation of the levels of the synaptic cell adhesion molecule (CAM) Fasciclin II (Fas II) (Schuster et al., 1996b). Genetic analysis showed that this downregulation of Fas II is both necessary and sufficient for structural plasticity at this synapse: FasII mutants that decrease Fas II levels by 50% lead to sprouting, similar to eag Sh and dnc, while transgenes that maintain synaptic Fas II levels suppress sprouting in eag Sh and dnc. A role for CAMs in the control of synaptic growth is not unique to Drosophila. In fact, CAMs of both the immunoglobulin and cadherin families are thought to play important roles in the regulation of synaptic growth in organisms as diverse as snails and mammals (Bailey et al., 1992; Mayford et al., 1992; Doherty et al., 1995; Fields and Itoh, 1996; Schachner, 1997). Although CAMs have been implicated in the control of synaptic growth, little is known about the molecular machinery that car- ries out this growth process. This lack of knowledge led us to undertake a genetic approach to identify genes and genetic pathways controlling synaptic growth. We took advantage of a sophisticated behavioral screen of the X chromosome conducted to detect walking mutants (Strauss, 1995a, 1995b). The X chromosome repre- sents about 20% of the Drosophila genome. We thus undertook a secondary anatomical screen of the larval NMJ of this preselected collection of walking
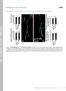
2 Neuron 314 Figure 1. hiw Mutants Have Dramatically Expanded Synaptic Structure at Third Instar NMJs Wild-type (A, C, E, and G) and hiw mutant (B, D, F, and H) third instar larvae were stained with anti-syt and anti Fas II to reveal presynaptic branches and boutons. (A D) Muscle 4: in wild-type larvae (A and C), muscle 4 is innervated by two motor neurons (here called MN4a and MN4b, [A]). MN4a has a type Ib and MN4b, a type Is, terminal arbor. Both motor terminals are present on muscle 4 in hiw mutants but contain many more branches and boutons (compare [B] and [D] with [A] and [C]). (E H) Muscles 12 and 13: the pattern of motor axon innervation of muscles 12 and 13 is also correct in hiw mutants, although the terminal branches are much longer and more numerous (compare [F] and [H] with [E] and [G]). Genotypes are wild-type (Berlin) (A, C, E, and G); hiw EMS (B and D); and hiw ND8 /RK4 (F and H). hiw mutants is dramatic; all NMJs grow exuberantly. hiw encodes a large protein that is localized at periac- tive zones which surround active zones at synaptic mutants. Using this genetic approach, we identified highwire (hiw) as a gene that controls synaptic growth at the larval NMJ. The increase in synaptic structure in
3 Highwire Regulates Synaptic Growth 315 terminals, the same region where Fas II is localized. Hiw appears to function as a negative regulator of synaptic growth. Results hiw Mutants Have Expanded Presynaptic Terminals at Neuromuscular Synapses in Third Instar Larvae Each abdominal hemisegment in the Drosophila embryo and larva has a stereotyped pattern of 30 muscles, each identifiable by its size, shape, body wall insertion position, and expression of molecular markers. About 45 motor neurons extend axons to innervate specific muscles in each hemisegment. At the larval NMJ, there are two major types of synaptic terminals (Johansen et al., 1989). Type I synapses have larger boutons and use glutamate as their neurotransmitter, while type II synapses have smaller boutons containing a variety of vesicles, including dense core vesicles filled with other transmitters, such as neuropeptides and biogenic amines (Atwood et al., 1993). Type I boutons are surrounded postsynaptically by a conspicuous subsynaptic reticulum (SSR) consisting of multiple folds and invaginations of the muscle cell membrane. Type I synapses can be further divided into type Ib (big) and type Is (small) based on the sizes of their boutons. These synapses undergo dynamic growth during larval development. As the muscle increases its volume, there is a parallel increase in the number of synaptic boutons and the number and complexity of branches (Schuster et al., 1996a; Zito et al., 1999). In a pilot effort to look for synaptic structural mutants, we screened a collection of viable mutants on the X chromosome, looking for structural defects at the NMJ of third instar larvae. These 230 mutant lines have various degrees of walking defects in adults (R. S., unpublished data). We used antibodies against synaptotagmin Figure 2. Growth of NMJ Synapses in Wild-Type and hiw Mutant (Syt) (Littleton et al., 1993) and Fas II (Van Vactor et al., Larvae 1993) as markers to label NMJ synaptic terminals of Quantification of the number of type I boutons at synapses on musthird instar larvae. We identified one mutant line, hiw, cles 6/7 (A) and muscle 4 (B), and the value of synaptic span (calcuwhich has a dramatic synaptic structure phenotype (Fig- lated by dividing the length of type I synapses by the length of the ure 1). This initial hiw EMS mutant allele and all 12 subse- muscle) of the synapses on muscle 12 (C). All measurements are quent hiw ND mutant alleles, as well as 3 P element alleles made in segment A3 at various larval stages in wild type (Berlin) (see below), behave as viable recessive alleles. They are and hiw EMS mutants labeled with anti-syt and anti Fas II. The results are plotted as a function of the rectangular muscle surface area of all viable as transheterozygotes with each other or with the corresponding muscles. The growth curve of the number of deficiencies that remove the gene. boutons in both wild type and hiw mutants can be fitted best with In hiw mutants, the presynaptic boutons visualized by power functions: hiw, y 2.7 x 0.39 (r 0.9); wild type, y 1.2 x 0.39 Syt staining are more numerous at all NMJs (Figure 2; (r 0.9) (A); hiw, y 2.5 x 0.38 (r 0.8); wild type, y 0.5 x 0.45 (r Table 1). Compared with wild-type synapses, hiw synsurface area, are indicated above the graph. Abbreviations: e-2nd, 0.8) (B). The developmental stages, which are relatively linear with apses have a greatly expanded branching pattern. The early second instar; l-2nd, late second instar; e-3rd, early third instar; presynaptic terminal arbors contain many more Fas II m-3rd, mid third instar. positive branches, the branches are longer, and the branch orders are increased (Table 2). The synaptic area, the muscle surface area occupied by presynaptic struc- in the number of boutons. After correcting for muscle tures, is also increased in hiw mutants (data not shown). area, hiw synapses also have a 2-fold increase in synap- This synaptic overgrowth phenotype is 100% penetrant tic size compared with wild type. The combination of in that we observe expanded presynaptic arborizations hiw EMS chromosome in trans over deficiencies showed and an increased number of boutons at every NMJ in essentially identical phenotypes. Developmental curves every segment of every mutant third instar animal. of both hiw and wild-type larvae (Figure 2) can be fitted We quantified several aspects of the mutant pheno- with similar types of power functions, but hiw synapses type (Figure 2; Tables 1 and 2). We counted the number have a larger synaptic size at first instar and grow at a of type I boutons on muscles 6/7 and on muscle 4 (Figure higher rate during further larval stages. 2; Table 1). hiw mutants have a 2-fold or greater increase On average, there is a decrease of bouton size in hiw
4 Neuron 316 Table 1. Synaptic Size Is Increased in hiw Mutants Bouton Number Bouton Number/Muscle Surface Area Genotype Muscle 6/7 Muscle 4 Muscle 6/7 Muscle 4 Wild-type (32) 64 3 (30) hiw EMS (35) (31) hiw EMS /Df (24) (25) Quantification of the number of boutons of synapses on muscle 6/7 and muscle 4 in wild-type (Berlin) and hiw mutant third instar larvae. The unit for bouton number/muscle surface area is 10 3 m 2. The numbers in parentheses indicate the number of hemisegments counted. mutants (Figure 1). Given the dramatic increase in the Although the morphology of NMJ synapses is dramatically number of boutons, this decrease in bouton size might expanded in hiw mutants, the specificity of connecnumber reflect some limitation of one or more structural compo- tions is not altered. The initial motor axon pathfinding nents as a result of oversprouting. Type Ib and type Is and target selection appear normal. Antibodies that la- boutons still can be distinguished from one another in bel both CNS and peripheral axon pathways (anti Fas hiw mutants, although both are smaller than their wildtype II and BP102 mabs) reveal a normal pattern of axon counterparts. Our impression is that type II boutons pathways and a normal pattern of muscle innervation and branches are also more numerous in hiw mutants. (data not shown). We did observe longer and larger However, this phenotype is less dramatic and more diffi- presynaptic terminal branches on the surfaces of muscles cult to score, and thus we did not quantitate it. in hiw mutants toward the end of embryogenesis, To compare branching, we measured three aspects but the specificity of contacts was normal. It appears of the branching pattern on muscle 12 (Figure 2; Table that hiw mutants display the synaptic overgrowth phe- 2). We chose muscle 12 for this analysis because its notype from the onset of synaptogenesis during embryogenesis. innervation is relatively planar and accessible, and the This is consistent with our observation that branching pattern of that innervation is relatively simple hiw first instar larvae, even 30 min after hatching, already in wild-type larvae, making it easier to quantitate branch display a dramatic increase in bouton number (34 3 number and complexity. First, we examined the total boutons in hiw [n 7] versus 17 1 in wild type [n length of this synapse (of only those branches containing 18] at muscles 6/7) and increase in synaptic span (56% type I boutons) as a function of the length of the 3% of muscle length in hiw versus 27% 2% in wild muscle (Figure 2C; Table 2). We find that from the first type at muscle 12). larval instar stage onward, the synaptic length in hiw mutants is greater than in wild type. This relationship is maintained throughout larval development. Several Synaptic Components Are Localized Second, we measured the length of the two longest Normally at hiw Mutant Synapses branches (Table 2). We find that these branches are We wanted to determine to what extent synaptic components more than 2-fold longer in hiw mutants compared with are localized normally in hiw mutant synapses. wild type. Postsynaptic glutamate receptors appear to be correctly Third, we examined the branching pattern of this synapse localized just opposite active zones as determined (Table 2). We divided the branches containing only by expression of the MHC-myc-DGluRIIA and MHCmyc-DGluRIIB type I boutons into different groups based on their transgenes (Petersen et al., 1997) in hiw branch order and counted the number of branches. mutant larvae (Figures 3C and 3D; data not shown). The There is a dramatic increase in the complexity of presynaptic protein Syt and the synaptic CAM Fas II are branches at hiw synapses; mutant synapses have many expressed normally at hiw mutant synapses (Syt shown more branches and higher order branches. The average in Figures 3A and 3B; Fas II not shown). Several other number of branches is (mean SEM, n synaptic proteins (Discs-large [DLG], Pak, and Dap160; 55) at hiw synapses on muscle 12, compared with 7.7 see below) are also localized correctly in hiw mutants 0.3 (n 79) at the same synapse in third instar wild- (data not shown). Thus, many of the molecular events type larvae. hiw synapses on muscle 12 often have several associated with synapse formation, including clustering fifth order branches, something that is rarely ob- of postsynaptic receptors and organization of presynap- served in wild type (Table 2). tic vesicles, appear normal in hiw mutants. This is con- Table 2. hiw Mutants Have More and Longer Presynaptic Branches Number of Branches Length of Branches ( m) Synaptic Span Second (Synapse Length/ Genotype First Second Third Fourth Fifth Longest Longest Muscle Length 100) Wild-type (79) hiw EMS (57) Quantification of branching pattern of the synapses on muscle 12. Wild-type (Berlin) and hiw EMS mutant third instar larvae were stained with anti-syt and anti Fas II. Tye type I branches were divided into different groups based on branch order. The number of branches in each group was counted. The lengths of synaptic branches and muscle were measured using an ocular micrometer (Zeiss).
5 Highwire Regulates Synaptic Growth 317 Figure 3. hiw Mutant Synapses Have Normal Components and Ultrastructure (A D) Confocal micrographs of wild-type (A and C) and hiw EMS mutant (B and D) synapses in third instar larvae showing normal presynaptic expression of synaptotagmin (Syt, red, [A and B]) and normal postsynaptic expression of glutamate receptor IIA subunit (GluR- IIA, green, [C and D]); Syt staining using anti- Syt antibody, GluRIIA staining usinganti-myc in flies carrying MHC-myc-GluRIIA transgene. (E) Conventional EM analysis was performed on hiw EMS /RK4 third instar larvae. A cross section of a bouton on muscle 6 is shown. This bouton looks identical to that of wild type. Asterisks mark the positions of active zones. SSR, the postsynaptic membrane infoldings, has normal thickness and morphology. Inset shows an active zone at higher magnification; note that the electron-dense T bar structure and clustered synaptic vesicles are present. Scale bar, 1 m; inset, 0.5 m. sistent with the observation that hiw mutants are viable examined serial sections of both hiw and wild-type bouand are not severely impaired in motor behaviors. tons at muscle 6 (segment A3) of third instar larvae in To further examine the relationship of FasII and hiw, the electron microscope (EM). We found that the hiw we tested whether overexpression of Fas II either pre- mutant synapses have active zones with T bars, clusters or postsynaptically could rescue the hiw mutant pheno- of synaptic vesicles, and other features that appear wildtype. We found that Fas II overexpression cannot rescue type (Figure 3E). The postsynaptic SSR also appeared the hiw mutant phenotype (data not shown). This sug- normal in size and shape (Figure 3E). We quantified gests that FasII is not downstream of hiw in the genetic some of these characteristics from serial sections of 9 control of synaptic growth. hiw boutons and compared them with similar data from 18 wild-type boutons. We find that although hiw boutons hiw Mutant Synapses Appear Normal Ultrastructurally are, on average, smaller than wild-type boutons, they Given the dramatic structural phenotype we observed nevertheless have the same relative bouton surface area at hiw NMJ synapses, we wondered whether the ultrastructure per active zone ( m 2 in hiw compared with of these mutant synapses was affected. We in wild type, p 0.22, Student s t test). In
6 Neuron 318 Figure 4. hiw Mutants Have Decreased Synaptic Transmisssion (A) Representative traces of spontaneous transmitter release recorded in 0.5 mm Ca 2 from muscle 6 of male wild-type (Berlin) and mutant (hiw EMS ) third instar larvae. Scale bar, 1 mv (vertical axis); 200 ms (horizontal axis). (B) Quantification of the average mejp amplitude. hiw mutants have reduced quantal size compared with that of wild type (p 0.001, Student s t test). n is the number of segments recorded. (C) Representative traces of evoked transmitter release recorded in 0.5 mm Ca 2 from muscle 6 of male wild-type (Berlin) and mutant (hiw EMS ) third instar larvae. Scale bar, 10 mv (vertical axis); 50 ms (horizontal axis). (D) The mean EJP amplitude is significantly reduced in hiw mutants compared with that of the wild-type control (p 0.001). n is the number of segments recorded. (E) Quantal content is significantly reduced in hiw mutants (p 0.001). The mean quantal content was determined for each recording by dividing the average superthreshold EJP amplitude by the average amplitude of the spontaneous miniature events. EJP amplitudes were corrected for nonlinear summation before calculation of quantal content. addition, the average maximum width of individual ac- responses to both spontaneous and evoked transmitter tive zones (as measured by membrane apposition, loca- release. The mean amplitude of spontaneous miniature tion of T bars, and clusters of synaptic vesicles) was junctional potentials (mejps), also called quantal size, nm (n 22) in hiw compared with is typically considered a measure of postsynaptic sensinm (n 18) in wild type. Finally, the number of T bars tivity to transmitter (in principle, it might also reflect the per active zone was statistically similar ( in amount of neurotransmitter packed into each synaptic hiw versus in wild type, p 0.15). We con- vesicle, although this is less likely). The mean amplitude clude from these data that the fundamental organiza- of the excitatory junctional potential (EJP), on the other tion, size, and spacing of active zones in hiw mutants hand, depends on both the postsynaptic receptor funcis relatively normal. tion and the amount of transmitter released from the presynaptic terminal. hiw Mutant Synapses Appear The average mejp amplitude is only slightly reduced Abnormal Physiologically in hiw mutants ( mv [n 30] versus 0.91 Given that hiw synapses, although greatly expanded, 0.04 mv [n 34] in wild type; Figure 4A). However, the appear normal ultrastructurally, we wondered whether evoked release is greatly reduced; the mutant shows a synaptic transmission is altered at these mutant synmv 66% decrease compared with wild type ( apses. One might imagine that the expanded structure [n 20] versus mv [n 19], respec- would lead to a stronger synapse. Much to our surprise, tively; Figure 4B). Thus, the quantal content (number of we found that these expanded synapses are actually vesicles released) of the mutant synapses ( weaker physiologically. [n 16]; Figure 4C), which is estimated by dividing the We performed intracellular recordings from muscle mean EJP amplitude by the mean mejp amplitude and 6 (segment A3) of third instar larvae. This muscle is adjusting for nonlinear summation, is about 34% of that innervated by type I glutamatergic boutons from motor of wild type ( [n 15]). Quantal content is neurons RP3 and 6/7b. We measured the postsynaptic an indication of presynaptic function. Since we did not
7 Highwire Regulates Synaptic Growth 319 Figure 5. HIW Is a Large Protein with Homologs in C. elegans and Human (A) The genomic organization of the hiw locus. Exons are indicated by boxes and introns, by lines. Conserved protein domains are indicated by the same colors as in (B). Locations of P element alleles and the nature of molecular lesions in EMS alleles are indicated. Alleles ND8, -9, -42, and -69 have premature stop codons. ND16 (a strong allele) has an amino acid substitution of a conserved residue (D4068), and ND51 (a weak allele) has an amino acid change of a nonconserved residue (G2054). The exon intron junctions were deduced by comparing cdna and genomic sequences. Gene A (encoded by LD10566) is located in the large intron, which spans 34 kb. This region corresponds approximately to cytological location 13A5-8. Centromere is to the right. (B) Schematics of HIW protein and HIW homologs. Conserved protein domains are as marked, with percent amino acid identity between domains. The yellow box above HIW protein indicates the fragment that is expressed in puas-hiw C1-LD. The small green box indicates the fragment used to generate anti-hiw mab 6H4. (C) Phylogenetic tree of HIW and HIW homologs. Relationships between HIW, Pam, and RPM-1 were determined by CLUSTAL analysis. The scale beneath the tree measures the distance between sequences. observe an obvious reduction in synaptic vesicle distribution in hiw mutants, the reduction in quantal content suggests more subtle defects in the release mechanism. Mapping the hiw Locus and Identification of the hiw Transcript The hiw locus was mapped by complementation analysis to lie between 12F and 13A on the X chromosome. Deficiency (Df) RK2 (12D2 E1; 13A2 5) and Df RK4 (12F5 6; 13A9 B1) (Drysdale et al., 1991) both uncover hiw. Screening of the P element insertions in the region revealed that the EP(X)1305 insert produces a weak hiw phenotype on its own. The phenotype is enhanced in EP/Df animals. Chromosome in situ hybridization placed the hiw EP insertion site at 13A5 6 (Berkeley Drosophila Genome Project [BDGP]), consistent with the recombination mapping of the hiw EMS allele, which placed hiw EMS at , between 13A and 13B (data not shown). Using inverse PCR and genomic rescue techniques, we obtained genomic sequences flanking the hiw EP insert. The 5 flanking sequence matched an expressed sequence tag (EST) from the BDGP database (cdna clone LD10566) that encodes a protein of 825 amino acids. Using 3 genomic DNA fragments as probes, we screened a Drosophila embryonic cdna library and identified an incomplete cdna clone encoding a different transcript. Using this analysis, we determined that hiw EP was inserted between two genes, which we refer to as gene A (5 transcript) and gene B (3 transcript) in the following section (Figure 5A). To determine which of the two transcripts is hiw, we performed RNA in situ hybridization with both tran- scripts. In wild-type embryos, gene A is expressed in mesodermal tissues, including the body wall muscles and the visceral mesoderm (data not shown). Gene B, on the other hand, has a nervous system specific pattern of expression (Figure 6A). Because the mutant phenotype is observed at the NMJ, we could not eliminate either
8 Neuron 320 Figure 6. HIW Is Expressed by CNS and PNS Neurons and Is Localized to CNS Neuropil and Synaptic Boutons at the Larval NMJ (A) Whole-mount stage 13 wild-type embryo labeled with mab BP102 (brown; this stains all CNS axons), as well as for hiw mrna (purple). hiw is strongly expressed by all CNS neurons, as well as all PNS neurons. No nonneuronal expression is seen. (B) Two abdominal segments of a wild-type stage 17 embryo in a fillet preparation. Arrows mark the localization of HIW protein at nascent NMJ synapses. (C) CNS of a stage 17 wild-type embryo stained for hiw mrna. hiw mrna is present uniformly within the CNS at low level and is localized to CNS axon tracts (open arrows) at high level. (D) CNS of a stage 17 wild-type embryo stained with anti-hiw mab. HIW is localized to CNS longitudinal axon tracts (open arrows). (This is where the synaptic neuropil forms.) Since we observed no HIW staining of CNS axons in the nerve roots, we presume this staining of the longitudinal axon tracts corresponds to staining of the synaptic neuropil. (E) Muscle 4 in a wild-type third instar larva stained with anti-hiw mab. HIW is localized to synaptic boutons of both type Ib (MN4a) and type Is (MN4b). Note that HIW is absent from the motor axons within the intersegmental nerve (open arrows). (F) Muscle 4 in a hiw ND8 third instar larvae stained with anti-hiw mab. HIW synaptic staining is absent. transcript as the candidate for hiw based on the expres- synapses on muscle 5 and muscles were missing, sion pattern. n 516 hemisegments). In contrast, injection of the We employed the RNA interference technique (Fire dsrna encoded by gene B produced expanded synapses et al., 1998; Kennerdell and Carthew, 1998) to test the identical to those seen in hiw mutants (92.5% of candidacy of both transcripts. Double stranded RNA the larval segments examined had hiw-like expanded (dsrna) fragments from both transcripts were generated synapses, n 384 hemisegments). and injected into embryos carrying the CD8-GFP- The results from the RNA interference experiments Sh marker (see Experimental Procedures). The CD8- suggest that hiw is encoded by the neurally expressed GFP-Sh marker allows us to visualize NMJ synapses gene B. In an independent screen for synaptic morphology in third instar larvae (Zito et al., 1999). Two different mutants on the X chromosome (A. D., unpublished fragments from each gene were used and produced data), we isolated 12 additional hiw alleles. Of the 12 similar results (data not shown). Injections of dsrna mutant alleles, 10 (ND8, -9, -16, -17, -36, -42, -45, -48, encoded by gene A produced animals with largely nor- -59, and -69) have the same phenotype as the original mal synapses (in about 5% of the segments examined, hiw EMS allele, and all display the same phenotype over
9 Highwire Regulates Synaptic Growth 321 Df RK2 or Df RK4, suggesting that all are null mutants rpm-1, the C. elegans homolog of hiw, was identified or very strong hypomorphs; 2 of the 12 (ND51 and -55) in two different genetic screens for synaptic mutants. have somewhat weaker phenotypes and have enhanced In one screen (Zhen et al., 2000), mutants were recovphenotypes when placed over deficiencies, indicating ered that showed abnormal distribution of a presynaptic that they are hypomorphs. Sequencing of all 13 hiw EMS marker at synapses from GABAergic motor neurons. alleles reveals that at least 6 of these alleles have single EM analysis reveals that rpm-1 mutant GABAergic and nucleotide changes within the coding sequence of gene cholinergic synapses have abnormal ultrastructure, such B that result in early stop codons or amino acid substitu- as a single presynaptic terminal containing multiple actions (ND8, -9, -16, -42, -51, and -69; Figure 5A). We tive zones. subsequently found two additional P element insertions, In the other screen (Schaefer et al., 2000), rpm-1 mu- PF253 and EP(X)1308 (Figure 5A; see Experimental Pro- tants were recovered that showed abnormal distribution cedures), that have strong and weak hiw phenotypes, of a presynaptic marker at synapses from posterior latrespectively. The PF253 insertion site is 13 nucleotides eral mechanosensory neurons. These are most likely away from EP(X)1305, both locating within the large in- glutamatergic synapses. This study subsequently examtron between gene A and gene B. EP(X)1308 is inserted ined cholinergic motor neurons and found similar synapin the 5 UTR region of gene B. We also could not rescue tic defects. In addition, the axonal branching pattern of the hiw mutant phenotype with either muscle or neuronal the mechanosensory neurons was found to be abnormal expression of a cdna encoded by gene A and did not in the mutants. detect any changes in gene A s coding sequence in the The homology among these three proteins (Drosophhiw EMS allele (data not shown). Taken together with the ila HIW, C. elegans RPM-1, and human Pam) is distribprotein localization data and the dominant-negative re- uted throughout the entire ORF (Figure 5B). All three sult obtained with a partial hiw cdna transgene (see proteins contain the RCC1-like domain, PHR repeats below), we conclude that hiw is encoded by gene B, the (named for Pam/HIW/RPM-1), and the zinc finger clusneuronal transcript. ter. Sequences outside the obvious structural domains also share similarities. From an evolutionary perspec- HIW Is a Large Protein with Homologs tive, HIW is more closely related to Pam than is RPM-1 in both C. elegans and Human (Figure 5C). The most conserved regions are the C-ter- We obtained overlapping hiw cdna clones encom- minal zinc finger cluster domain of 430 amino acids as passing a single complete open reading frame (ORF) well as the internal PHR repeats. HIW is the largest of by screening several cdna libraries made from various the three proteins: 593 amino acids larger than Pam developmental stages. We inferred the gene structure and 1468 amino acids larger than RPM-1. These extra of hiw by comparing the cdna sequence with genomic sequences are distributed throughout the protein. Sesequence generated by the BDGP and our own se- quences unique to HIW have similarity to coiled-coil quencing effort (Figure 5A). The full-length cdna is domains. One feature common to only HIW and RPM-1 16,179 bp in length and encodes an enormous protein is that they both partially lack a 300 amino acid region of 5233 amino acids (for details of cloning, see Experi- in Pam that was shown to be required for binding to mental Procedures; complete cdna sequence and de- Myc (Guo et al., 1998). duced protein ORF can be found in the public database as accession number TBD). The predicted molecular HIW Is Expressed at Synaptic Boutons weight is 566 kda. How does this large cytoplasmic protein function to There are several conspicuous protein motifs present control synaptic growth in Drosophila? In a first attempt in HIW (Figure 5B). These motifs include at the N termito understand HIW function, we examined both its nus, a region of seven tandem repeats similar to those first described in a protein known as RCC1, for regulator mrna and protein distribution. For protein localization, of chromosome condensation (Bischoff and Ponstingl, we generated a monoclonal antibody against HIW (see 1991). The C terminus contains a cysteine-rich domain Experimental Procedures). that may have multiple zinc finger motifs, two of which Analysis by in situ hybridization shows that hiw mrna can be clearly defined as a RING-type and a B-box-type is largely, if not exclusively, neural specific (Figure 6A). zinc finger. Toward the middle of the protein are two Antisense probes labeled the ventral neurogenic region direct repeats of 90 amino acid (PHR repeats, see below) as early as stage 5. The staining persists in the neuro- and a coiled-coil domain. We did not detect any signal genic region at later stages and reaches its peak around peptide or transmembrane domains. Thus, we infer that stage 13, when both CNS and PNS neurons clearly ex- HIW is a putative cytoplasmic protein. press this transcript (Figure 6A). After embryonic stage When we searched the NCBI database, we found that 14, the strength of CNS and PNS cell body expression HIW is homologous to a human protein called Pam, for starts to decrease. By stages 16 and 17, we observe protein associated with Myc (Guo et al., 1998), as well weak staining of neuronal cell bodies throughout the as to a C. elegans protein (Figure 5B) shown by two CNS. However, by this stage, we also observe darker other groups to be encoded by the rpm-1 gene (Schaefer staining in a pattern within the CNS that coincides with et al., 2000 [this issue of Neuron]; Zhen et al., 2000 [this the longitudinal axon tracts found where the synaptic issue of Neuron). neuropil forms (Figure 6C; we observed the same pattern Pam was identified in an expression screen for prosion with four independent RNA in situ probes). This expresteins that bind to Myc, the product of c-myc protooncoand of mrna is not associated with specific cell bodies, gene. Pam was shown to be located in the nucleus and thus we conclude that it represents mrna in axonal associated with Myc in tissue culture cells. processes or synaptic region.
10 Neuron 322 Figure 7. HIW and Fas II Are Localized to Periactive Zones Confocal fluorescence microscopy of synaptic boutons of wild-type (Canton S) third instar larvae stained with different antibodies. Scale bar, 0.5 m. (A) Fas II (green) is localized to subboutonic areas. Note that the staining is not uniform and that it often surrounds nonstaining circles. (B) Fas II (green) and Pak (red) protein localizations are complementary. Since Pak is at active zones (see [C]), this shows that Fas II is in boutons but is localized outside active zones. (C) Pak (red) and glutamate receptor IIB subunit (GluRIIB, green) proteins colocalize to active zones. Anti-Myc mab was used to detect Myc- GluRIIB localization in flies carrying MHC-myc-GluRIIB transgene. (D) HIW (green) protein distribution at synaptic boutons is not uniform, similar to that of Fas II (A). (E and F) HIW (green) and Pak (red) also have complementary and nonoverlapping protein localization, indicating that HIW, just like Fas II, is localized to periactive zones outside active zone areas. HIW Expression at Synaptic Boutons Is Localized to Periactive Zones To study the localization of HIW at individual boutons, we employed confocal microscopy. We first examined HIW alone and observed that the staining is not uniform in boutons but rather is confined to patches (Figure 7D). We next performed double labeling experiments with HIW and other synaptic markers. Antibodies against Syt and DLG (Budnik et al., 1996) label synaptic terminals in a rather uniform fashion. Double labeling with anti- HIW and anti-syt, as well as anti-dlg, confirms that HIW is not uniform within synaptic boutons (data not shown). To further identify the position and significance of these HIW patches, we looked for antibodies that label active zones versus the areas around active zones, HIW protein is detectable at early embryonic stages in the CNS (data not shown). In contrast to the mrna expression, we did not see a peak of protein expression around stage 13. Rather, antibody staining reveals that HIW protein expression in the CNS is, by and large, constant from stage 13 through the end of embryogenesis (data not shown). Within the CNS, most of the protein is expressed in the region of the longitudinal axon tracts around which the synaptic neuropil forms (Figure 6D). We see no protein in the CNS nerve roots, none in the peripheral motor and sensory nerves, and very little in the commissural axon pathways. Thus, its expression correlates with where the synaptic neuropil forms in the CNS. Outside the CNS, the predominant location of HIW protein expression is at NMJ synapses in the embyro (Figure 6B). We first observed protein expression during stage 16, just as these synapses are forming. In third instar larvae, we observe protein expression at the NMJ and at the CNS neuropil (Figure 6E; data not shown). Type I synaptic boutons are clearly stained at all NMJs on all muscles. We also detect weaker staining of type II boutons. The synaptic staining is largely absent at the NMJ in hiw mutant alleles ND8 (Figure 6F), -9, and -69, and is greatly reduced in hiw mutant alleles hiw EMS, ND16, and ND51. In addition, we generated a transgenic line of flies (puas-hiw C1-LD ) that ectopically expresses a fragment of HIW protein containing the epitope used for generating the antibody (Figure 5B; see Experimental Procedures). The HIW mab detects this HIW protein fragment ectopically expressed in muscles or in tracheal branches (data not shown), further confirming its specificity. When the horseradish peroxidase (HRP) product is overdeveloped, the antibody also faintly stains the surface of muscles and each of their attachment sites (data not shown). However, we believe that this staining is due to the recognition of an unrelated protein(s) since this staining is still present at about the same level in all of the hiw mutants.
11 Highwire Regulates Synaptic Growth 323 Figure 8. Immuno-EM Microscopy Analysis Confirms that HIW Is Localized to Periactive Zones (A E) Immuno-EMs show serial sections of a wild-type (Canton S) third instar larva stained with anti-hiw mab and HRP immuno-em. Asterisk marks the position of a T bar at the center of an active zone. Arrowheads point to the beginning of HIW staining areas outside the active zone. (F) A flattened reconstruction from serial immuno-em sections, showing that the HIW expression domain (gray) surrounds nonstaining areas, which are the active zones. These active zones have T bars (black). Scale bar, 0.5 m (A E); 1 m (F). called periactive zones (M. Sone et al., submitted). The To examine the periactive zone localization of HIW postsynaptic membrane just opposite the presynaptic protein in greater detail, we performed immuno-em active zones can be labeled with antibodies against an analysis (Figure 8). We found that HIW protein is associepitope tag on transgenic glutamate receptor subunits ated with both the plasma membrane of boutons in (MHC-myc-DGluRIIB) (Petersen et al., 1997). For double all periactive zone areas and with clusters of vesicles labeling with anti-hiw, however, we needed another outside active zones. However, active zones, T bars, active zone marker that was not a mouse monoclonal and vesicles clustered at active zones are devoid of antibody. For such a probe, we turned to the Drosophila staining (Figure 8). We also observed some HIW staining homolog of the Pak cytoplasmic kinase (Hing et al., associated with postsynaptic membranes opposite peri- 1999; C. Hama, personal communication). We found that active zones, even though in situ hybridization analysis the glutamate receptor subunit and Pak are precisely did not reveal muscle expression of hiw mrna. In our colocalized (Figure 7C). By double labeling HIW and immuno-em serial section reconstruction of boutons Pak, we found that most HIW patches are nonoverlapcontaining 20 active zones, the nonstaining active zones ping with Pak staining spots and form a pattern comple- (which contained T bars and clusters of clear synaptic mentary to that of Pak (Figures 7E and 7F). These data vesicles) measured nm, while the areas beindicate that HIW is localized to the periactive zones tween them (the periactive zones) were all stained by that surround the active zones. the anti-hiw antibody and measured nm. Interestingly, we observed a similar pattern of expres- Given the variability of these measurements, the HIW sion of the CAM Fas II. Fas II staining is seen in nonoverexclusion zone width of nm in the immunolapping areas around Pak staining (Figure 7B; this result EM is in the same range as, but is a bit wider than, was first described to us by C. Hama, personal commuour independent assessment of active zone width in nication; see also M. Sone et al., submitted). The staining with both Fas II and HIW fills the space outside the conventional EM (see above) of nm. We do Pak staining areas, suggesting that both are localized not know whether this 50 nm ( 10%) difference on around but not in active zones. each side represents a real difference or whether we We used one additional antibody to confirm the distriby classic ultrastructural criteria. have simply underestimated the width of the active zone bution of HIW protein outside active zones. Dap160 is a presynaptic membrane-associated protein that physically interacts with dynamin and is organized into an A Transgene Expressing a HIW Fragment Behaves endocytic honeycomb that surrounds active zones in a Dominant-Negative Manner When (Roos and Kelly, 1998, 1999). Double labeling between Expressed Presynaptically HIW and Dap160 reveals that the two proteins have As a first step to gaining insight into the mechanisms largely overlapping distributions (data not shown), furdifferent of HIW function, we generated transgenic flies carrying ther supporting the idea that HIW protein is localized to HIW fragments. One such transgene (puas- subboutonic regions outside the active zones. HIW C1-LD ) contains a HIW fragment between amino acids
12 Neuron and 3461, fused to enhanced green fluorescent However, although greatly expanded, these synapses protein (EGFP) (Figure 5B; see Experimental Procedures). are not stronger, as one might have expected, but rather When this transgene was expressed panneurally have a reduced quantal content. One possibility is that using elav-gal4 (Luo et al., 1994) in an otherwise wildtype the reduction in quantal content is itself the primary background, we observed expanded synaptic defect and that this physiological defect leads to the structures at the NMJ of third instar larvae (data not increased growth in an attempt to compensate and re- shown). This phenotype is qualitatively similar to that of turn the strength back to normal. But this is unlikely, hiw loss-of-function mutants, although quantitatively it since mutant conditions that lead to an even greater is not as severe. When we expressed the same trans- reduction in quantal content (e.g., mutations in synaptic gene in muscles using the panmuscle 24B-Gal4 enhancer vesicle protein synaptotagmin) do not lead to presynap- trap line (Luo et al., 1994), we did not observe tic sprouting at this synapse (DiAntonio and Schwarz, any phenotype at the NMJ. This result further suggests 1994). Thus, the overgrowth of the synapse appears to that HIW functions on the presynaptic side to control be a primary defect. The data suggest that the physiological synaptic growth. defect is either secondary to the overgrowth or an independent primary defect. Discussion hiw encodes a large protein of 5233 amino acids (predicted to be 566 kda) found at presynaptic terminals. Using a genetic approach in Drosophila, we have identisurrounding Within presynaptic terminals, HIW is localized to areas fied the hiw gene and shown that it functions as a negaspicuously active zones, the periactive zone. It is con- tive regulator of synaptic growth. Our phenotypic analysis absent from active zones. At the immunotive has focused on the glutamatergic NMJ, but since all EM level, HIW is associated with the plasma membrane neurons appear to express both hiw mrna and HIW and with vesicles near the membrane but outside the protein, it is reasonable to suggest that hiw functions active zone. Fas II, a CAM that also controls synaptic at most, if not all, synapses. growth (see Introduction), is found at the same location The original ethyl methanesulfonate (EMS) allele was as HIW at the periactive zones (M. Sone et al., submitted; isolated in a behavioral screen that selected for viable Figure 7B). mutations. We subsequently isolated 12 additional EMS Our results suggest that HIW functions largely on the alleles. All 13 EMS alleles and 3 P element alleles are presynaptic side of the synapse. Certainly, at the NMJ, viable, either alone or in trans over one another, or over we detect high levels of hiw mrna in motor neurons and either of two deficiencies that remove the gene. Several undetectable levels in muscles. We have been unable to of these alleles are likely to be true nulls based on their perform transgenic rescue experiments of this gene in phenotype over deficiencies and the presence of premathe Drosophila due to the enormous size of the ORF and ture stop codons. Thus, the hiw mutant phenotype detranscript. complexity of the genomic region containing the hiw scribed here viable flies with dramatic synaptic overpartial Nevertheless, we have been able to use a growth is likely to represent the true null phenotype. hiw cdna transgene to generate a dominantgrowth is The adult walking defect described for the original negative phenotype in which the NMJ overgrows, just as hiw EMS allele was mapped within the same deficiencies in hiw loss-of-function mutants. This dominant-negative that remove the hiw gene (R. S., unpublished data). We phenotype is only observed when the HIW fragment is also observed similarly expanded synaptic structures expressed on the presynaptic rather than postsynaptic at adult NMJs (data not shown). We have not tested any side of this synapse, further suggesting that HIW protein of the new hiw alleles for walking defects, and so we normally functions on the presynaptic side. cannot say with certainty that the behavioral defect seen The homologous gene in the nematode C. elegans, in the original allele is due to the loss of hiw function. rpm-1 also seems to function cell autonomously in pre- In hiw mutants, motor axon pathfinding and target synaptic neurons. The expression of RPM-1 on the prerecognition appear normal. Synapses initially form at synaptic side of the synapse completely rescues func- their normal locations on the appropriate muscles and tion, and rpm-1 is required only in presynaptic neurons induce appropriate postsynaptic glutamate receptor in genetic mosaic analysis (Schaefer et al., 2000; Zhen clustering. These synapses normally grow and sprout et al., 2000). Thus, taken together with the nematode during larval development. But in hiw mutants, they are data, we conclude that HIW regulates synaptic growth greatly expanded in both the number of boutons, and by functioning on the presynaptic side of the synapse. the extent and length of branches. The expanded synapses in hiw mutants have smaller-than-normal boutons HIW May Function at Many Synapses and Not Just but appear normal ultrastructurally in terms of active at the Drosophila NMJ zones, T bars, clusters of synaptic vesicles, and folds Our phenotypic analysis has been focused on the NMJ, of the subsynaptic reticulum. Occasionally, the overgrowth where we can visualize the branching pattern of individ- is so extensive that motor axons wander over ual presynaptic terminal arbors, count the number of to adjacent muscles that they would not innervate normally. boutons, use confocal microscopy to image subcellular This aspect of the hiw phenotype is similar to localization of components, and use conventional and what is seen when the CAM Fas II is overexpressed on immuno-em to analyze identified synapses. These tech- target muscles (Davis et al., 1997). niques are much more difficult to apply to identified Physiologically, hiw synapses are functional, as is evident terminal arbors and synapses in the CNS. We have been from the viable nature of the null mutants and the unable to find single cell probes that would allow this fact that they are capable of many motor behaviors. kind of quantitative analysis in the fruit fly CNS.
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10923 Supplementary Figure 1 Ten-a and Ten-m antibody and cell type specificities. a c, Representative single confocal sections of a Drosophila NMJ stained with antibodies to Ten-a (red),
doi:10.1038/nature10923 Supplementary Figure 1 Ten-a and Ten-m antibody and cell type specificities. a c, Representative single confocal sections of a Drosophila NMJ stained with antibodies to Ten-a (red),
Chapter 4 Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays.
 Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays. The data described in chapter 3 presented evidence that endogenous
Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays. The data described in chapter 3 presented evidence that endogenous
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
Nature Neuroscience: doi: /nn.2662
 Supplementary Figure 1 Atlastin phylogeny and homology. (a) Maximum likelihood phylogenetic tree based on 18 Atlastin-1 sequences using the program Quicktree. Numbers at internal nodes correspond to bootstrap
Supplementary Figure 1 Atlastin phylogeny and homology. (a) Maximum likelihood phylogenetic tree based on 18 Atlastin-1 sequences using the program Quicktree. Numbers at internal nodes correspond to bootstrap
Open Syntaxin Docks Synaptic Vesicles
 Marc Hammarlund 1,2[, Mark T. Palfreyman 1,2[, Shigeki Watanabe 1,2[, Shawn Olsen 1,2, Erik M. Jorgensen 1,2* PLoS BIOLOGY 1 Department of Biology, University of Utah, Salt Lake City, Utah, United States
Marc Hammarlund 1,2[, Mark T. Palfreyman 1,2[, Shigeki Watanabe 1,2[, Shawn Olsen 1,2, Erik M. Jorgensen 1,2* PLoS BIOLOGY 1 Department of Biology, University of Utah, Salt Lake City, Utah, United States
Cells. Steven McLoon Department of Neuroscience University of Minnesota
 Cells Steven McLoon Department of Neuroscience University of Minnesota 1 Microscopy Methods of histology: Treat the tissue with a preservative (e.g. formaldehyde). Dissect the region of interest. Embed
Cells Steven McLoon Department of Neuroscience University of Minnesota 1 Microscopy Methods of histology: Treat the tissue with a preservative (e.g. formaldehyde). Dissect the region of interest. Embed
Midterm 1. Average score: 74.4 Median score: 77
 Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
MCN. Complex Genetic Interactions among Four Receptor Tyrosine Phosphatases Regulate Axon Guidance in Drosophila
 MCN Molecular and Cellular Neuroscience 17, 274 291 (2001) doi:10.1006/mcne.2000.0939, available online at http://www.idealibrary.com on Complex Genetic Interactions among Four Receptor Tyrosine Phosphatases
MCN Molecular and Cellular Neuroscience 17, 274 291 (2001) doi:10.1006/mcne.2000.0939, available online at http://www.idealibrary.com on Complex Genetic Interactions among Four Receptor Tyrosine Phosphatases
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Kasprowicz et al., http://www.jcb.org/cgi/content/full/jcb.201310090/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. NMJ morphology of shi 12-12B ; shi-4c not treated
Supplemental material Kasprowicz et al., http://www.jcb.org/cgi/content/full/jcb.201310090/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. NMJ morphology of shi 12-12B ; shi-4c not treated
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
Neuronal Differentiation: Synapse Formation NMJ
 Neuronal Differentiation: Synapse Formation NMJ Approaches to study synapse formation Rodent NMJ (cholinergic) Drosophila NMJ (glutamatergic) Rodent CNS synapse (glutamatergic or GABAergic) C. elegans
Neuronal Differentiation: Synapse Formation NMJ Approaches to study synapse formation Rodent NMJ (cholinergic) Drosophila NMJ (glutamatergic) Rodent CNS synapse (glutamatergic or GABAergic) C. elegans
Anterograde Activin Signaling Regulates Postsynaptic Membrane Potential and GluRIIA/B Abundance at the Drosophila Neuromuscular Junction
 Anterograde Activin Signaling Regulates Postsynaptic Membrane Potential and GluRIIA/B Abundance at the Drosophila Neuromuscular Junction Myung-Jun Kim, Michael B. O Connor* Department of Genetics, Cell
Anterograde Activin Signaling Regulates Postsynaptic Membrane Potential and GluRIIA/B Abundance at the Drosophila Neuromuscular Junction Myung-Jun Kim, Michael B. O Connor* Department of Genetics, Cell
Homeostatic Control of Presynaptic Release Is Triggered by Postsynaptic Membrane Depolarization
 Neuron, Vol. 30, 737 749, June, 2001, Copyright 2001 by Cell Press Homeostatic Control of Presynaptic Release Is Triggered by Postsynaptic Membrane Depolarization Suzanne Paradis, Sean T. Sweeney, and
Neuron, Vol. 30, 737 749, June, 2001, Copyright 2001 by Cell Press Homeostatic Control of Presynaptic Release Is Triggered by Postsynaptic Membrane Depolarization Suzanne Paradis, Sean T. Sweeney, and
A complementation test would be done by crossing the haploid strains and scoring the phenotype in the diploids.
 Problem set H answers 1. To study DNA repair mechanisms, geneticists isolated yeast mutants that were sensitive to various types of radiation; for example, mutants that were more sensitive to UV light.
Problem set H answers 1. To study DNA repair mechanisms, geneticists isolated yeast mutants that were sensitive to various types of radiation; for example, mutants that were more sensitive to UV light.
The Transmembrane Semaphorin Sema I Is Required in Drosophila for Embryonic Motor and CNS Axon Guidance
 Neuron, Vol. 20, 207 220, February, 1998, Copyright 1998 by Cell Press The Transmembrane Semaphorin Sema I Is Required in Drosophila for Embryonic Motor and CNS Axon Guidance Hung-Hsiang Yu, Houmam H.
Neuron, Vol. 20, 207 220, February, 1998, Copyright 1998 by Cell Press The Transmembrane Semaphorin Sema I Is Required in Drosophila for Embryonic Motor and CNS Axon Guidance Hung-Hsiang Yu, Houmam H.
Developmental genetics: finding the genes that regulate development
 Developmental Biology BY1101 P. Murphy Lecture 9 Developmental genetics: finding the genes that regulate development Introduction The application of genetic analysis and DNA technology to the study of
Developmental Biology BY1101 P. Murphy Lecture 9 Developmental genetics: finding the genes that regulate development Introduction The application of genetic analysis and DNA technology to the study of
Introduction Principles of Signaling and Organization p. 3 Signaling in Simple Neuronal Circuits p. 4 Organization of the Retina p.
 Introduction Principles of Signaling and Organization p. 3 Signaling in Simple Neuronal Circuits p. 4 Organization of the Retina p. 5 Signaling in Nerve Cells p. 9 Cellular and Molecular Biology of Neurons
Introduction Principles of Signaling and Organization p. 3 Signaling in Simple Neuronal Circuits p. 4 Organization of the Retina p. 5 Signaling in Nerve Cells p. 9 Cellular and Molecular Biology of Neurons
A role for PS integrins in morphological growth and synaptic function at the postembryonic neuromuscular junction of Drosophila
 Development 126, 5833-5846 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV8655 5833 A role for PS integrins in morphological growth and synaptic function at the postembryonic
Development 126, 5833-5846 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV8655 5833 A role for PS integrins in morphological growth and synaptic function at the postembryonic
Supporting Information
 Supporting Information Cao et al. 10.1073/pnas.1306220110 Gram - bacteria Hemolymph Cytoplasm PGRP-LC TAK1 signalosome Imd dfadd Dredd Dnr1 Ikk signalosome P Relish Nucleus AMP and effector genes Fig.
Supporting Information Cao et al. 10.1073/pnas.1306220110 Gram - bacteria Hemolymph Cytoplasm PGRP-LC TAK1 signalosome Imd dfadd Dredd Dnr1 Ikk signalosome P Relish Nucleus AMP and effector genes Fig.
Drosophila Spastin Regulates Synaptic Microtubule Networks and Is Required for Normal Motor Function
 Drosophila Spastin Regulates Synaptic Microtubule Networks and Is Required for Normal Motor Function Nina Tang Sherwood 1, Qi Sun 1 1, Mingshan Xue 2 2, Bing Zhang 2, Kai Zinn 1 * Open access, freely available
Drosophila Spastin Regulates Synaptic Microtubule Networks and Is Required for Normal Motor Function Nina Tang Sherwood 1, Qi Sun 1 1, Mingshan Xue 2 2, Bing Zhang 2, Kai Zinn 1 * Open access, freely available
Illegitimate translation causes unexpected gene expression from on-target out-of-frame alleles
 Illegitimate translation causes unexpected gene expression from on-target out-of-frame alleles created by CRISPR-Cas9 Shigeru Makino, Ryutaro Fukumura, Yoichi Gondo* Mutagenesis and Genomics Team, RIKEN
Illegitimate translation causes unexpected gene expression from on-target out-of-frame alleles created by CRISPR-Cas9 Shigeru Makino, Ryutaro Fukumura, Yoichi Gondo* Mutagenesis and Genomics Team, RIKEN
Reading: Chapter 5, pp ; Reference chapter D, pp Problem set F
 Mosaic Analysis Reading: Chapter 5, pp140-141; Reference chapter D, pp820-823 Problem set F Twin spots in Drosophila Although segregation and recombination in mitosis do not occur at the same frequency
Mosaic Analysis Reading: Chapter 5, pp140-141; Reference chapter D, pp820-823 Problem set F Twin spots in Drosophila Although segregation and recombination in mitosis do not occur at the same frequency
Control and Integration. Nervous System Organization: Bilateral Symmetric Animals. Nervous System Organization: Radial Symmetric Animals
 Control and Integration Neurophysiology Chapters 10-12 Nervous system composed of nervous tissue cells designed to conduct electrical impulses rapid communication to specific cells or groups of cells Endocrine
Control and Integration Neurophysiology Chapters 10-12 Nervous system composed of nervous tissue cells designed to conduct electrical impulses rapid communication to specific cells or groups of cells Endocrine
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
Chapter 11. Development: Differentiation and Determination
 KAP Biology Dept Kenyon College Differential gene expression and development Mechanisms of cellular determination Induction Pattern formation Chapter 11. Development: Differentiation and Determination
KAP Biology Dept Kenyon College Differential gene expression and development Mechanisms of cellular determination Induction Pattern formation Chapter 11. Development: Differentiation and Determination
Chapter 9. Nerve Signals and Homeostasis
 Chapter 9 Nerve Signals and Homeostasis A neuron is a specialized nerve cell that is the functional unit of the nervous system. Neural signaling communication by neurons is the process by which an animal
Chapter 9 Nerve Signals and Homeostasis A neuron is a specialized nerve cell that is the functional unit of the nervous system. Neural signaling communication by neurons is the process by which an animal
Systematic Screening of Drosophila Deficiency Mutations for Embryonic Phenotypes and Orphan Receptor Ligands
 Systematic Screening of Drosophila Deficiency Mutations for Embryonic Phenotypes and Orphan Receptor Ligands Ashley P. Wright 1, A. Nicole Fox 1, Karl G. Johnson 2, Kai Zinn 1 * 1 Division of Biology,
Systematic Screening of Drosophila Deficiency Mutations for Embryonic Phenotypes and Orphan Receptor Ligands Ashley P. Wright 1, A. Nicole Fox 1, Karl G. Johnson 2, Kai Zinn 1 * 1 Division of Biology,
Supplementary Figures
 Supplementary Figures Supplementary Fig. S1: Normal development and organization of the embryonic ventral nerve cord in Platynereis. (A) Life cycle of Platynereis dumerilii. (B-F) Axonal scaffolds and
Supplementary Figures Supplementary Fig. S1: Normal development and organization of the embryonic ventral nerve cord in Platynereis. (A) Life cycle of Platynereis dumerilii. (B-F) Axonal scaffolds and
Nature Biotechnology: doi: /nbt Supplementary Figure 1. Overexpression of YFP::GPR-1 in the germline.
 Supplementary Figure 1 Overexpression of YFP::GPR-1 in the germline. The pie-1 promoter and 3 utr were used to express yfp::gpr-1 in the germline. Expression levels from the yfp::gpr-1(cai 1.0)-expressing
Supplementary Figure 1 Overexpression of YFP::GPR-1 in the germline. The pie-1 promoter and 3 utr were used to express yfp::gpr-1 in the germline. Expression levels from the yfp::gpr-1(cai 1.0)-expressing
Cellular Neuroanatomy I The Prototypical Neuron: Soma. Reading: BCP Chapter 2
 Cellular Neuroanatomy I The Prototypical Neuron: Soma Reading: BCP Chapter 2 Functional Unit of the Nervous System The functional unit of the nervous system is the neuron. Neurons are cells specialized
Cellular Neuroanatomy I The Prototypical Neuron: Soma Reading: BCP Chapter 2 Functional Unit of the Nervous System The functional unit of the nervous system is the neuron. Neurons are cells specialized
Positional Cues in the Drosophila Nerve Cord: Semaphorins Pattern the Dorso-Ventral Axis
 Positional Cues in the Drosophila Nerve Cord: Semaphorins Pattern the Dorso-Ventral Axis Marta Zlatic 1,2,3 *, Feng Li 1, Maura Strigini 4, Wesley Grueber 2, Michael Bate 1 * 1 Department of Zoology, University
Positional Cues in the Drosophila Nerve Cord: Semaphorins Pattern the Dorso-Ventral Axis Marta Zlatic 1,2,3 *, Feng Li 1, Maura Strigini 4, Wesley Grueber 2, Michael Bate 1 * 1 Department of Zoology, University
Nervous Tissue. Neurons Neural communication Nervous Systems
 Nervous Tissue Neurons Neural communication Nervous Systems What is the function of nervous tissue? Maintain homeostasis & respond to stimuli Sense & transmit information rapidly, to specific cells and
Nervous Tissue Neurons Neural communication Nervous Systems What is the function of nervous tissue? Maintain homeostasis & respond to stimuli Sense & transmit information rapidly, to specific cells and
Chapter 18 Lecture. Concepts of Genetics. Tenth Edition. Developmental Genetics
 Chapter 18 Lecture Concepts of Genetics Tenth Edition Developmental Genetics Chapter Contents 18.1 Differentiated States Develop from Coordinated Programs of Gene Expression 18.2 Evolutionary Conservation
Chapter 18 Lecture Concepts of Genetics Tenth Edition Developmental Genetics Chapter Contents 18.1 Differentiated States Develop from Coordinated Programs of Gene Expression 18.2 Evolutionary Conservation
SUPPLEMENTARY INFORMATION
 med!1,2 Wild-type (N2) end!3 elt!2 5 1 15 Time (minutes) 5 1 15 Time (minutes) med!1,2 end!3 5 1 15 Time (minutes) elt!2 5 1 15 Time (minutes) Supplementary Figure 1: Number of med-1,2, end-3, end-1 and
med!1,2 Wild-type (N2) end!3 elt!2 5 1 15 Time (minutes) 5 1 15 Time (minutes) med!1,2 end!3 5 1 15 Time (minutes) elt!2 5 1 15 Time (minutes) Supplementary Figure 1: Number of med-1,2, end-3, end-1 and
Lecture 7. Development of the Fruit Fly Drosophila
 BIOLOGY 205/SECTION 7 DEVELOPMENT- LILJEGREN Lecture 7 Development of the Fruit Fly Drosophila 1. The fruit fly- a highly successful, specialized organism a. Quick life cycle includes three larval stages
BIOLOGY 205/SECTION 7 DEVELOPMENT- LILJEGREN Lecture 7 Development of the Fruit Fly Drosophila 1. The fruit fly- a highly successful, specialized organism a. Quick life cycle includes three larval stages
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo
 Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
CHAPTER 3. Cell Structure and Genetic Control. Chapter 3 Outline
 CHAPTER 3 Cell Structure and Genetic Control Chapter 3 Outline Plasma Membrane Cytoplasm and Its Organelles Cell Nucleus and Gene Expression Protein Synthesis and Secretion DNA Synthesis and Cell Division
CHAPTER 3 Cell Structure and Genetic Control Chapter 3 Outline Plasma Membrane Cytoplasm and Its Organelles Cell Nucleus and Gene Expression Protein Synthesis and Secretion DNA Synthesis and Cell Division
Honors Biology Reading Guide Chapter 11
 Honors Biology Reading Guide Chapter 11 v Promoter a specific nucleotide sequence in DNA located near the start of a gene that is the binding site for RNA polymerase and the place where transcription begins
Honors Biology Reading Guide Chapter 11 v Promoter a specific nucleotide sequence in DNA located near the start of a gene that is the binding site for RNA polymerase and the place where transcription begins
Nervous System Organization
 The Nervous System Nervous System Organization Receptors respond to stimuli Sensory receptors detect the stimulus Motor effectors respond to stimulus Nervous system divisions Central nervous system Command
The Nervous System Nervous System Organization Receptors respond to stimuli Sensory receptors detect the stimulus Motor effectors respond to stimulus Nervous system divisions Central nervous system Command
The BMP Ligand Gbb Gates the Expression of Synaptic Homeostasis Independent of Synaptic Growth Control
 Article The BMP Ligand Gbb Gates the Expression of Synaptic Homeostasis Independent of Synaptic Growth Control Carleton P. Goold 1 and Graeme W. Davis 1, * 1 Department of Biochemistry and Biophysics,
Article The BMP Ligand Gbb Gates the Expression of Synaptic Homeostasis Independent of Synaptic Growth Control Carleton P. Goold 1 and Graeme W. Davis 1, * 1 Department of Biochemistry and Biophysics,
Student Handout Fruit Fly Ethomics & Genomics
 Student Handout Fruit Fly Ethomics & Genomics Summary of Laboratory Exercise In this laboratory unit, students will connect behavioral phenotypes to their underlying genes and molecules in the model genetic
Student Handout Fruit Fly Ethomics & Genomics Summary of Laboratory Exercise In this laboratory unit, students will connect behavioral phenotypes to their underlying genes and molecules in the model genetic
Nature Neuroscience: doi: /nn.2717
 Supplementary Fig. 1. Dendrite length is not secondary to body length. Dendrite growth proceeds independently of the rate of body growth and decreases in rate in adults. n 20 on dendrite measurement, n
Supplementary Fig. 1. Dendrite length is not secondary to body length. Dendrite growth proceeds independently of the rate of body growth and decreases in rate in adults. n 20 on dendrite measurement, n
Shaggy, the Homolog of Glycogen Synthase Kinase 3, Controls Neuromuscular Junction Growth in Drosophila
 The Journal of Neuroscience, July 21, 2004 24(29):6573 6577 6573 Brief Communication Shaggy, the Homolog of Glycogen Synthase Kinase 3, Controls Neuromuscular Junction Growth in Drosophila Bénédicte Franco,
The Journal of Neuroscience, July 21, 2004 24(29):6573 6577 6573 Brief Communication Shaggy, the Homolog of Glycogen Synthase Kinase 3, Controls Neuromuscular Junction Growth in Drosophila Bénédicte Franco,
Modulation of central pattern generator output by peripheral sensory cells in Drosophila larvae. BioNB4910 Cornell University.
 Modulation of central pattern generator output by peripheral sensory cells in Drosophila larvae BioNB4910 Cornell University Goals 1) Observe the behavioral effects of remotely activating different populations
Modulation of central pattern generator output by peripheral sensory cells in Drosophila larvae BioNB4910 Cornell University Goals 1) Observe the behavioral effects of remotely activating different populations
Chapter 37 Active Reading Guide Neurons, Synapses, and Signaling
 Name: AP Biology Mr. Croft Section 1 1. What is a neuron? Chapter 37 Active Reading Guide Neurons, Synapses, and Signaling 2. Neurons can be placed into three groups, based on their location and function.
Name: AP Biology Mr. Croft Section 1 1. What is a neuron? Chapter 37 Active Reading Guide Neurons, Synapses, and Signaling 2. Neurons can be placed into three groups, based on their location and function.
Nervous Systems: Neuron Structure and Function
 Nervous Systems: Neuron Structure and Function Integration An animal needs to function like a coherent organism, not like a loose collection of cells. Integration = refers to processes such as summation
Nervous Systems: Neuron Structure and Function Integration An animal needs to function like a coherent organism, not like a loose collection of cells. Integration = refers to processes such as summation
Genomics and bioinformatics summary. Finding genes -- computer searches
 Genomics and bioinformatics summary 1. Gene finding: computer searches, cdnas, ESTs, 2. Microarrays 3. Use BLAST to find homologous sequences 4. Multiple sequence alignments (MSAs) 5. Trees quantify sequence
Genomics and bioinformatics summary 1. Gene finding: computer searches, cdnas, ESTs, 2. Microarrays 3. Use BLAST to find homologous sequences 4. Multiple sequence alignments (MSAs) 5. Trees quantify sequence
Designer Genes C Test
 Northern Regional: January 19 th, 2019 Designer Genes C Test Name(s): Team Name: School Name: Team Number: Rank: Score: Directions: You will have 50 minutes to complete the test. You may not write on the
Northern Regional: January 19 th, 2019 Designer Genes C Test Name(s): Team Name: School Name: Team Number: Rank: Score: Directions: You will have 50 minutes to complete the test. You may not write on the
Drosophila VAP-33A Directs Bouton Formation at Neuromuscular Junctions in a Dosage-Dependent Manner
 Neuron, Vol. 35, 291 306, July 18, 2002, Copyright 2002 by Cell Press Drosophila VAP-33A Directs Bouton Formation at Neuromuscular Junctions in a Dosage-Dependent Manner Giuseppa Pennetta, 1,2 Peter Robin
Neuron, Vol. 35, 291 306, July 18, 2002, Copyright 2002 by Cell Press Drosophila VAP-33A Directs Bouton Formation at Neuromuscular Junctions in a Dosage-Dependent Manner Giuseppa Pennetta, 1,2 Peter Robin
Translation Part 2 of Protein Synthesis
 Translation Part 2 of Protein Synthesis IN: How is transcription like making a jello mold? (be specific) What process does this diagram represent? A. Mutation B. Replication C.Transcription D.Translation
Translation Part 2 of Protein Synthesis IN: How is transcription like making a jello mold? (be specific) What process does this diagram represent? A. Mutation B. Replication C.Transcription D.Translation
Nervous Tissue. Neurons Electrochemical Gradient Propagation & Transduction Neurotransmitters Temporal & Spatial Summation
 Nervous Tissue Neurons Electrochemical Gradient Propagation & Transduction Neurotransmitters Temporal & Spatial Summation What is the function of nervous tissue? Maintain homeostasis & respond to stimuli
Nervous Tissue Neurons Electrochemical Gradient Propagation & Transduction Neurotransmitters Temporal & Spatial Summation What is the function of nervous tissue? Maintain homeostasis & respond to stimuli
Neurons, Synapses, and Signaling
 Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Supplementary Figure 1. Phenotype of the HI strain.
 Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
RPM-1 is localized to distinct subcellular compartments and regulates axon length in GABAergic motor neurons
 Opperman and Grill Neural Development 14, 9:1 RESERCH RTICLE Open ccess RPM-1 is localized to distinct subcellular compartments and regulates axon length in Gergic motor neurons Karla J Opperman and rock
Opperman and Grill Neural Development 14, 9:1 RESERCH RTICLE Open ccess RPM-1 is localized to distinct subcellular compartments and regulates axon length in Gergic motor neurons Karla J Opperman and rock
Nature Genetics: doi: /ng Supplementary Figure 1. The phenotypes of PI , BR121, and Harosoy under short-day conditions.
 Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Neurons, Synapses, and Signaling
 Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lectures for Biology, Eighth Edition Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp and Janette Lewis Copyright
Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lectures for Biology, Eighth Edition Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp and Janette Lewis Copyright
7.06 Cell Biology EXAM #3 KEY
 7.06 Cell Biology EXAM #3 KEY May 2, 2006 This is an OPEN BOOK exam, and you are allowed access to books, a calculator, and notes BUT NOT computers or any other types of electronic devices. Please write
7.06 Cell Biology EXAM #3 KEY May 2, 2006 This is an OPEN BOOK exam, and you are allowed access to books, a calculator, and notes BUT NOT computers or any other types of electronic devices. Please write
Exam 1 ID#: October 4, 2007
 Biology 4361 Name: KEY Exam 1 ID#: October 4, 2007 Multiple choice (one point each) (1-25) 1. The process of cells forming tissues and organs is called a. morphogenesis. b. differentiation. c. allometry.
Biology 4361 Name: KEY Exam 1 ID#: October 4, 2007 Multiple choice (one point each) (1-25) 1. The process of cells forming tissues and organs is called a. morphogenesis. b. differentiation. c. allometry.
Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila
 Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila Melissa M. Rolls and Chris Q. Doe, Inst. Neurosci and Inst. Mol. Biol., HHMI, Univ. Oregon, Eugene, Oregon 97403 Correspondence
Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila Melissa M. Rolls and Chris Q. Doe, Inst. Neurosci and Inst. Mol. Biol., HHMI, Univ. Oregon, Eugene, Oregon 97403 Correspondence
Neurons, Synapses, and Signaling
 Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Neurons, Synapses, and Signaling
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 48 Neurons, Synapses, and Signaling
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 48 Neurons, Synapses, and Signaling
A Product of the Drosophila stoned Locus Regulates Neurotransmitter Release
 The Journal of Neuroscience, December 1, 1998, 18(23):9638 9649 A Product of the Drosophila stoned Locus Regulates Neurotransmitter Release Daniel T. Stimson, 1 Patricia S. Estes, 1 Michiko Smith, 2 Leonard
The Journal of Neuroscience, December 1, 1998, 18(23):9638 9649 A Product of the Drosophila stoned Locus Regulates Neurotransmitter Release Daniel T. Stimson, 1 Patricia S. Estes, 1 Michiko Smith, 2 Leonard
Synaptic Homeostasis Is Consolidated by the Cell Fate Gene gooseberry,adrosophila pax3/7 Homolog
 The Journal of Neuroscience, June 16, 2010 30(24):8071 8082 8071 Development/Plasticity/Repair Synaptic Homeostasis Is Consolidated by the Cell Fate Gene gooseberry,adrosophila pax3/7 Homolog Bruno Marie,
The Journal of Neuroscience, June 16, 2010 30(24):8071 8082 8071 Development/Plasticity/Repair Synaptic Homeostasis Is Consolidated by the Cell Fate Gene gooseberry,adrosophila pax3/7 Homolog Bruno Marie,
Developmental Biology
 Developmental Biology 366 (2012) 204 217 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/locate/developmentalbiology Developmental changes in
Developmental Biology 366 (2012) 204 217 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/locate/developmentalbiology Developmental changes in
A Hierarchy of Cell Intrinsic and Target-Derived Homeostatic Signaling
 Article A Hierarchy of Cell Intrinsic and Target-Derived Homeostatic Signaling Sharon Bergquist, 1 Dion K. Dickman, 1 and Graeme W. Davis 1, * 1 Department of Biochemistry and Biophysics, Program in Neuroscience,
Article A Hierarchy of Cell Intrinsic and Target-Derived Homeostatic Signaling Sharon Bergquist, 1 Dion K. Dickman, 1 and Graeme W. Davis 1, * 1 Department of Biochemistry and Biophysics, Program in Neuroscience,
Nervous System Organization
 The Nervous System Chapter 44 Nervous System Organization All animals must be able to respond to environmental stimuli -Sensory receptors = Detect stimulus -Motor effectors = Respond to it -The nervous
The Nervous System Chapter 44 Nervous System Organization All animals must be able to respond to environmental stimuli -Sensory receptors = Detect stimulus -Motor effectors = Respond to it -The nervous
Multiple Choice Review- Eukaryotic Gene Expression
 Multiple Choice Review- Eukaryotic Gene Expression 1. Which of the following is the Central Dogma of cell biology? a. DNA Nucleic Acid Protein Amino Acid b. Prokaryote Bacteria - Eukaryote c. Atom Molecule
Multiple Choice Review- Eukaryotic Gene Expression 1. Which of the following is the Central Dogma of cell biology? a. DNA Nucleic Acid Protein Amino Acid b. Prokaryote Bacteria - Eukaryote c. Atom Molecule
Neurons and Nervous Systems
 34 Neurons and Nervous Systems Concept 34.1 Nervous Systems Consist of Neurons and Glia Nervous systems have two categories of cells: Neurons, or nerve cells, are excitable they generate and transmit electrical
34 Neurons and Nervous Systems Concept 34.1 Nervous Systems Consist of Neurons and Glia Nervous systems have two categories of cells: Neurons, or nerve cells, are excitable they generate and transmit electrical
Full file at CHAPTER 2 Genetics
 CHAPTER 2 Genetics MULTIPLE CHOICE 1. Chromosomes are a. small linear bodies. b. contained in cells. c. replicated during cell division. 2. A cross between true-breeding plants bearing yellow seeds produces
CHAPTER 2 Genetics MULTIPLE CHOICE 1. Chromosomes are a. small linear bodies. b. contained in cells. c. replicated during cell division. 2. A cross between true-breeding plants bearing yellow seeds produces
Genomes and Their Evolution
 Chapter 21 Genomes and Their Evolution PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from
Chapter 21 Genomes and Their Evolution PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from
Dendrites - receives information from other neuron cells - input receivers.
 The Nerve Tissue Neuron - the nerve cell Dendrites - receives information from other neuron cells - input receivers. Cell body - includes usual parts of the organelles of a cell (nucleus, mitochondria)
The Nerve Tissue Neuron - the nerve cell Dendrites - receives information from other neuron cells - input receivers. Cell body - includes usual parts of the organelles of a cell (nucleus, mitochondria)
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1 Sns and Duf co-localise in embryonic nephrocytes a-c, Wild-type stage 11 (a),14 (b) and 16 (c) embryos stained with anti-duf (green) and anti-sns (red). Higher magnification images
Supplementary Figure 1 Sns and Duf co-localise in embryonic nephrocytes a-c, Wild-type stage 11 (a),14 (b) and 16 (c) embryos stained with anti-duf (green) and anti-sns (red). Higher magnification images
Drosophila Plexin B is a Sema-2a receptor required for axon guidance
 RESEARCH ARTICLE 2125 Development 133, 2125-2135 (2006) doi:10.1242/dev.02380 Drosophila Plexin B is a Sema-2a receptor required for axon guidance Joseph C. Ayoob 1, Jonathan R. Terman 1,2 and Alex L.
RESEARCH ARTICLE 2125 Development 133, 2125-2135 (2006) doi:10.1242/dev.02380 Drosophila Plexin B is a Sema-2a receptor required for axon guidance Joseph C. Ayoob 1, Jonathan R. Terman 1,2 and Alex L.
Segment boundary formation in Drosophila embryos
 Segment boundary formation in Drosophila embryos Development 130, August 2003 Camilla W. Larsen, Elizabeth Hirst, Cyrille Alexandre and Jean Paul Vincent 1. Introduction: - Segment boundary formation:
Segment boundary formation in Drosophila embryos Development 130, August 2003 Camilla W. Larsen, Elizabeth Hirst, Cyrille Alexandre and Jean Paul Vincent 1. Introduction: - Segment boundary formation:
Synapses. Electrophysiology and Vesicle release
 Synapses Electrophysiology and Vesicle release Major point Cell theory (cells being separated) implies that cells must communicate with each other through extracellular connections most communication is
Synapses Electrophysiology and Vesicle release Major point Cell theory (cells being separated) implies that cells must communicate with each other through extracellular connections most communication is
IN the Drosophila ventral nerve cord, there are 20
 Copyright Ó 2007 by the Genetics Society of America DOI: 10.1534/genetics.107.075085 Regulation of Axon Guidance by Slit and Netrin Signaling in the Drosophila Ventral Nerve Cord Krishna Moorthi Bhat,
Copyright Ó 2007 by the Genetics Society of America DOI: 10.1534/genetics.107.075085 Regulation of Axon Guidance by Slit and Netrin Signaling in the Drosophila Ventral Nerve Cord Krishna Moorthi Bhat,
Fig. S1. Proliferation and cell cycle exit are affected by the med mutation. (A,B) M-phase nuclei are visualized by a-ph3 labeling in wild-type (A)
 Fig. S1. Proliferation and cell cycle exit are affected by the med mutation. (A,B) M-phase nuclei are visualized by a-ph3 labeling in wild-type (A) and mutant (B) 4 dpf retinae. The central retina of the
Fig. S1. Proliferation and cell cycle exit are affected by the med mutation. (A,B) M-phase nuclei are visualized by a-ph3 labeling in wild-type (A) and mutant (B) 4 dpf retinae. The central retina of the
Information processing. Divisions of nervous system. Neuron structure and function Synapse. Neurons, synapses, and signaling 11/3/2017
 Neurons, synapses, and signaling Chapter 48 Information processing Divisions of nervous system Central nervous system (CNS) Brain and a nerve cord Integration center Peripheral nervous system (PNS) Nerves
Neurons, synapses, and signaling Chapter 48 Information processing Divisions of nervous system Central nervous system (CNS) Brain and a nerve cord Integration center Peripheral nervous system (PNS) Nerves
Chapter 17. From Gene to Protein. Biology Kevin Dees
 Chapter 17 From Gene to Protein DNA The information molecule Sequences of bases is a code DNA organized in to chromosomes Chromosomes are organized into genes What do the genes actually say??? Reflecting
Chapter 17 From Gene to Protein DNA The information molecule Sequences of bases is a code DNA organized in to chromosomes Chromosomes are organized into genes What do the genes actually say??? Reflecting
Lesson Overview. Gene Regulation and Expression. Lesson Overview Gene Regulation and Expression
 13.4 Gene Regulation and Expression THINK ABOUT IT Think of a library filled with how-to books. Would you ever need to use all of those books at the same time? Of course not. Now picture a tiny bacterium
13.4 Gene Regulation and Expression THINK ABOUT IT Think of a library filled with how-to books. Would you ever need to use all of those books at the same time? Of course not. Now picture a tiny bacterium
Reading Assignments. A. Genes and the Synthesis of Polypeptides. Lecture Series 7 From DNA to Protein: Genotype to Phenotype
 Lecture Series 7 From DNA to Protein: Genotype to Phenotype Reading Assignments Read Chapter 7 From DNA to Protein A. Genes and the Synthesis of Polypeptides Genes are made up of DNA and are expressed
Lecture Series 7 From DNA to Protein: Genotype to Phenotype Reading Assignments Read Chapter 7 From DNA to Protein A. Genes and the Synthesis of Polypeptides Genes are made up of DNA and are expressed
Chapter 48 Neurons, Synapses, and Signaling
 Chapter 48 Neurons, Synapses, and Signaling Concept 48.1 Neuron organization and structure reflect function in information transfer Neurons are nerve cells that transfer information within the body Neurons
Chapter 48 Neurons, Synapses, and Signaling Concept 48.1 Neuron organization and structure reflect function in information transfer Neurons are nerve cells that transfer information within the body Neurons
Monitoring neurite morphology and synapse formation in primary neurons for neurotoxicity assessments and drug screening
 APPLICATION NOTE ArrayScan High Content Platform Monitoring neurite morphology and synapse formation in primary neurons for neurotoxicity assessments and drug screening Suk J. Hong and Richik N. Ghosh
APPLICATION NOTE ArrayScan High Content Platform Monitoring neurite morphology and synapse formation in primary neurons for neurotoxicity assessments and drug screening Suk J. Hong and Richik N. Ghosh
Attractive and repulsive functions of Slit are mediated by different receptors in the Drosophila trachea
 Development 129, 4941-4951 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV7966 4941 Attractive and repulsive functions of Slit are mediated by different receptors in the Drosophila
Development 129, 4941-4951 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV7966 4941 Attractive and repulsive functions of Slit are mediated by different receptors in the Drosophila
A Screen of Cell-Surface Molecules Identifies Leucine-Rich Repeat Proteins as Key Mediators of Synaptic Target Selection
 Article A Screen of Cell-Surface Molecules Identifies Leucine-Rich Repeat Proteins as Key Mediators of Synaptic Target Selection Mitsuhiko Kurusu, 1,2, * Amy Cording, 1 Misako Taniguchi, 2 Kaushiki Menon,
Article A Screen of Cell-Surface Molecules Identifies Leucine-Rich Repeat Proteins as Key Mediators of Synaptic Target Selection Mitsuhiko Kurusu, 1,2, * Amy Cording, 1 Misako Taniguchi, 2 Kaushiki Menon,
Math in systems neuroscience. Quan Wen
 Math in systems neuroscience Quan Wen Human brain is perhaps the most complex subject in the universe 1 kg brain 10 11 neurons 180,000 km nerve fiber 10 15 synapses 10 18 synaptic proteins Multiscale
Math in systems neuroscience Quan Wen Human brain is perhaps the most complex subject in the universe 1 kg brain 10 11 neurons 180,000 km nerve fiber 10 15 synapses 10 18 synaptic proteins Multiscale
10/2/2015. Chapter 4. Determination and Differentiation. Neuroanatomical Diversity
 Chapter 4 Determination and Differentiation Neuroanatomical Diversity 1 Neurochemical diversity: another important aspect of neuronal fate Neurotransmitters and their receptors Excitatory Glutamate Acetylcholine
Chapter 4 Determination and Differentiation Neuroanatomical Diversity 1 Neurochemical diversity: another important aspect of neuronal fate Neurotransmitters and their receptors Excitatory Glutamate Acetylcholine
Dimorphism and Homeostasis of Neuromuscular Synaptic Function
 Dimorphism and Homeostasis of Neuromuscular Synaptic Function - Epp m ε Release + + φ - q Sensitivity The nature of the problem Size, Strength, Resistance.. The Power of Drosophila The nature of the problem
Dimorphism and Homeostasis of Neuromuscular Synaptic Function - Epp m ε Release + + φ - q Sensitivity The nature of the problem Size, Strength, Resistance.. The Power of Drosophila The nature of the problem
Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila
 Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila Sangyun Jeong a,1, Da-som Yang a, Young Gi Hong a, Sarah P. Mitchell b,c, Matthew P. rown b,c, and
Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila Sangyun Jeong a,1, Da-som Yang a, Young Gi Hong a, Sarah P. Mitchell b,c, Matthew P. rown b,c, and
Conclusions. The experimental studies presented in this thesis provide the first molecular insights
 C h a p t e r 5 Conclusions 5.1 Summary The experimental studies presented in this thesis provide the first molecular insights into the cellular processes of assembly, and aggregation of neural crest and
C h a p t e r 5 Conclusions 5.1 Summary The experimental studies presented in this thesis provide the first molecular insights into the cellular processes of assembly, and aggregation of neural crest and
7.06 Cell Biology EXAM #3 April 21, 2005
 7.06 Cell Biology EXAM #3 April 21, 2005 This is an open book exam, and you are allowed access to books, a calculator, and notes but not computers or any other types of electronic devices. Please write
7.06 Cell Biology EXAM #3 April 21, 2005 This is an open book exam, and you are allowed access to books, a calculator, and notes but not computers or any other types of electronic devices. Please write
Biology I Level - 2nd Semester Final Review
 Biology I Level - 2nd Semester Final Review The 2 nd Semester Final encompasses all material that was discussed during second semester. It s important that you review ALL notes and worksheets from the
Biology I Level - 2nd Semester Final Review The 2 nd Semester Final encompasses all material that was discussed during second semester. It s important that you review ALL notes and worksheets from the
Supplementary Figures Supplementary Tables 1 and 2. Supplementary References
 The Nuclear Import of a Non-Canonical Frizzled2 Signal by Importins-β11 and α2 Promotes Postsynaptic Development Timothy J. Mosca and Thomas L. Schwarz * The F.M. Kirby Neurobiology Center, Children s
The Nuclear Import of a Non-Canonical Frizzled2 Signal by Importins-β11 and α2 Promotes Postsynaptic Development Timothy J. Mosca and Thomas L. Schwarz * The F.M. Kirby Neurobiology Center, Children s
NEURONS, SENSE ORGANS, AND NERVOUS SYSTEMS CHAPTER 34
 NEURONS, SENSE ORGANS, AND NERVOUS SYSTEMS CHAPTER 34 KEY CONCEPTS 34.1 Nervous Systems Are Composed of Neurons and Glial Cells 34.2 Neurons Generate Electric Signals by Controlling Ion Distributions 34.3
NEURONS, SENSE ORGANS, AND NERVOUS SYSTEMS CHAPTER 34 KEY CONCEPTS 34.1 Nervous Systems Are Composed of Neurons and Glial Cells 34.2 Neurons Generate Electric Signals by Controlling Ion Distributions 34.3
What Organelle Makes Proteins According To The Instructions Given By Dna
 What Organelle Makes Proteins According To The Instructions Given By Dna This is because it contains the information needed to make proteins. assemble enzymes and other proteins according to the directions
What Organelle Makes Proteins According To The Instructions Given By Dna This is because it contains the information needed to make proteins. assemble enzymes and other proteins according to the directions
Constructing a Pedigree
 Constructing a Pedigree Use the appropriate symbols: Unaffected Male Unaffected Female Affected Male Affected Female Male carrier of trait Mating of Offspring 2. Label each generation down the left hand
Constructing a Pedigree Use the appropriate symbols: Unaffected Male Unaffected Female Affected Male Affected Female Male carrier of trait Mating of Offspring 2. Label each generation down the left hand
CHROMOSOMAL BASIS OF INHERITANCE
 CHROMOSOMAL BASIS OF INHERITANCE The fruit fly (Drosophila melanogaster) has been widely used for the study of eukaryote genetics because of several features: short generation time (two weeks) prolific
CHROMOSOMAL BASIS OF INHERITANCE The fruit fly (Drosophila melanogaster) has been widely used for the study of eukaryote genetics because of several features: short generation time (two weeks) prolific
Bypass and interaction suppressors; pathway analysis
 Bypass and interaction suppressors; pathway analysis The isolation of extragenic suppressors is a powerful tool for identifying genes that encode proteins that function in the same process as a gene of
Bypass and interaction suppressors; pathway analysis The isolation of extragenic suppressors is a powerful tool for identifying genes that encode proteins that function in the same process as a gene of
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila November 2, 2006 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Developmental Biology Biology 4361 Axis Specification in Drosophila November 2, 2006 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Role of Neural Activity during Synaptogenesis in Drosophila
 The Journal of Neuroscience, December 1995, 15( 12): 8177-8190 Role of Neural Activity during Synaptogenesis in Drosophila Jill Jareckil and Haig Keshishian* Departments of Genetics and 2Biology, Yale
The Journal of Neuroscience, December 1995, 15( 12): 8177-8190 Role of Neural Activity during Synaptogenesis in Drosophila Jill Jareckil and Haig Keshishian* Departments of Genetics and 2Biology, Yale
