4. The material balances for isothermal ideal reactor models
|
|
|
- John Foster
- 6 years ago
- Views:
Transcription
1 Summay Geneal mateal balane f eatng system Bath eat Cntnuus-flw eats: CST (Cntnuus Sted Tank eat) P (Plug lw eat) Steady state f CST and P Desgn tasks : utlet (fnal nvesn), gven vlume f eat x vlume f eat, gven utlet nvesn
2 4. The mateal balanes f sthemal deal eat mdels T=nst. Abtay vlume element Inlet nvetve mla flws (ml/s) A 0 k, k... 2 n ( t) ( t) Mla balane f spees 0 n d d k, k k t t Oveall mla balane k, k k, k k k Outlet nvetve mla flws (ml/s) d d... k 2 n t k
3 nst. T=nst. Pefet mxng Q Heat flux Bath eats Mxe Manmete P=f(t) Labaty Phamaeutal ndusty Speal hemals Plymes... A 0 k, k n ( t) ( t) lume f eatn mxtue Mla balane f spees n d t 0 k, k k dn k k, k.
4 eat vlume nstant ( =nst.) dn d n d k,,... k, k 2 eat pessue nstant (P = knst.) n. dn d. d d ln k k, k d k k, k d ln T alulate P( t) ( t) state equatn f(t,p,, m pstn) = 0 s needed
5 Example Cnstant vlume sthemal bath eat, deal gas mxtue P T n k dn dp T T T k, k k, k k k k Cnstant pessue sthemal bath eat, deal gas mxtue T P n d ln( ) T P d T dn T T P P P k, k k, k k k k k, k d T k, k k, k k P k y k, k k, k k k P y T *
6 Example Cnstant vlume bath eat, lqud mxtue nstant pessue Send de evesble eatn d d d d 2 3 k 2 k 2 k k 2 2 A (l) + A 2(l) A 3(l),, d d k k k, t 0,,, 0 ml/l t mle/l mle/l 0 t/mn 2 2 e 2 kt 2 kt 2 3
7 Cntnuus-flw eats Cntnuus Sted Tank eat CST A 0 k, k n ( t) nst lume f eat Mla balane f spees 0 k, k k dn Oveall mla balane k k, k dn We need suplementay nfmatn state behav f eatn mxtue stat-up (shut-dwn) mla flw ates f ndvdual spees
8 d Cnstant pessue sthemal CST, deal gas mxtue It ases fm veall mla balane (vlume f eat, s nstant) n dn k, k k, k k k dn k, k k and mla balane f spees bemes y k, k k, k k k dn P dy ( y y ) ( y ) T k k, k k dy T T = ( y y ) ( ), y k k, k P P k Inlet vlumet flw ate s T P Mean esdene tme f eatn mxtue (based n nlet flw ate) s gven by T n p 0 y P T
9 and mla balane f spees bemes fnally dy T = ( y y ) ( ), y k k, k P k If nly ne eatn takes plae: dy T = ( y y ) ( y ), P.00 Example Stat-up f an sthemal CST O / 2O 2 g 2 g 2 g = kp, k 2.028x0 m le.pa. m n.m y, y y y 0 (= O, 2=O, 3=, 4=H e) t 0, y 0 y y 0, y 0. l =0 m, T =320 C, P 0 kpa O umeal slutn f balane equatns y (-) O (fatnal) nvesn f 2 O* ) =( )/ He 2 O t (s) * ) The equatn s vald nly at steady state
10 Steady state,, k, k k If nly ne eatn takes plae:,, usng fatnal nvesn f key spees ( ) a) T he vlum e f eat f gven utlet nvesn ( ) Smple substtutn b) T he utlet nvesn f gven vlum e f eat ( ) ( ) 0 f Gaphal numeal (teatve) slutn
11 Gaphal assessment f the CST vlume ( ) ( ) Levenspel dagam usually v
12 Gaphal assessment f the utlet nvesn ( )
13 Casade f CST ( n ) ( n ) ( n ) ( n ) Steady state n-th membe f the asade ( n ) ( n ) ( n ) ( n ),, n, C ST ( n ) ( n) ( n ) ( n ) ( ) ( n ) ( n ) ( n ) ( n ) ( n ) ( n ) ( ), n, C ST
14 Gaphal assessment f the utlet nvesn n the asade f CST ( ) () ( 2 ) () Hmewk 6 Pve that In the lmt () ( n ) ( n) n ( k ), ( n ) k n lm e, n ( n ) (2) f fst de nstant-vlume eatn.
15 Plug lw eat P ( ) ( ) v Tubula eats Hgh pdutn apaty Catalyt eats (e.g. ammna synthess)... z z + z S z + Mla balane f spees ( t, z ) ( t, z ) = ( t, z), k, k t S z k
16 Steady state d d k, k, k If nly ne eatn takes plae: d d, usng fatnal nvesn f key spees d ( ) d ( 2 ) d ( ) () y y, m useful elatns: m m 3 m la vlum e (m m le )
17 BATCH, CST, P (PBC), ne eatn, fatnal nvesn f key mpnent BATCH n d ( ) 0 0 t t d ( ) 0 0 t t CST P CST ( ) ( ) PBC Paked Bed Catalyt eat P d d ( ) ( ) 0 0 W d d ( ) 0 M 0 M
18 Mean esdene tme BATCH, P d ( ) 0 CST ( ) vlumet flw ate (m 3 /s) f eatn mxtue at nlet nns
19 Gas Huly Spae elty G H S G as vlum et flw ate(m /h) lum e f eat (m ) 3 3 Lqud Huly Spae elty LH S Lqud vlum et flw ate (m /h) lum e f eat (m ) 3 3
20 Tasks. Calulate the vlume f eat (mass f atalyst) (CST, P, PBC) tme f eatn (BATCH) t btan gven nvesn. 2. Calulate the utlet nvesn f gven vlume f eat. 3. Calulate the eatn ate n labaty eat t btan knet law and estmate the knet paametes.
21 = A 0 = subspt f key mpnent BATCH LOW n n n n.. d. n n n n n... x n n. x x. n k n n n. x. x. x x.. x. n n x n. m m m x x. x. x. m m m x x. x. m - m la vlum e f eatn m xtue 3 (m /m l)
22 Gas phase - Ideal state behav x P P T T T P x x. x. m T P x x. P P T T T. x. P n n T P ( x ) T P n n.. T P T P T P T P x x T P x T P.. T P T P T P T P x x
23 Hmewk 7 Calulate vlume f P t pdue 50 kt ethylen/yea. eatn f the st de C 2 H 6(g) C 2 H 4(g) + H 2(g) takes plae at 00 K and 0,6 Mpa. nal nvesn f ethan s 80 %. eatn ate s gven by = k. A k (000 K) = 0,072 s - E = 343,6 kj/ml Pue ethan s fed nt the eat. Assumptns: Ideal gas, mla weght f ethylen s 28,054 kg/kml. K. J. Ladle and B. W. Wehwsk: Knets and Mehansms f the Themal Dempstn f Ethane. I. The Unnhbted eatn, Peedngs f the yal Sety f Lndn. Sees A, Mathematal and Physal Senes, l. 260,. 300 pp. 9-02
24 Hmewk 4 (due afte Chapte 4) Calulate vlumes f CST a P wkng at 50 C and 300 kpa t pdue t COCl 2 /day wth CO nvesn equal t 95 %. A mxtue f CO and Cl 2 (mla at :) s fed at 300 kpa and 50 C. Data k(423 K) = 0.07 (m 3 ml - ) 3/2.s - M COCl2 = kg/kml. Answe: CST = m 3 P = m 3 CO () Cl 2 (2) I COCl 2 (3) 0 CO (g) + Cl 2(g) COCl 2(g) 2 OUT steady state; - key m pnent
25 deal gas 3 3 C l 2 5 / 2 3 / 2 3 C O C l2 T m T P [m /m l] P P P C O 2 T T 2 T 2 k k m m P 5 / 2 0 / / [m l/m /s] m l/s [m l/m ] C ST 5 / 2 5 / 2 5 / / 2 ( ) P k 0.07 T
26 P / / 2 T 2 5 /2 5 / 2 ( ) 0 0 P k P 0 d d d k T / 2 3 m 3
27 Summay Geneal mateal balane f eatng system Bath eat Cntnuus-flw eats: CST (Cntnuus Sted Tank eat) P (Plug lw eat) Steady state f CST and P Desgn tasks : utlet (fnal nvesn), gven vlume f eat x vlume f eat, gven utlet nvesn
Tutorial Chemical Reaction Engineering:
 Dpl.-Ing. ndeas Jöke Tutoal Chemal eaton Engneeng:. eal eatos, esdene tme dstbuton and seletvty / yeld fo eaton netwoks Insttute of Poess Engneeng, G5-7, andeas.joeke@ovgu.de 8-Jun-6 Tutoal CE: esdene
Dpl.-Ing. ndeas Jöke Tutoal Chemal eaton Engneeng:. eal eatos, esdene tme dstbuton and seletvty / yeld fo eaton netwoks Insttute of Poess Engneeng, G5-7, andeas.joeke@ovgu.de 8-Jun-6 Tutoal CE: esdene
Cork Institute of Technology. Spring 2005 DCE 3.5 Thermodynamics & Heat Transfer (Time: 3 Hours) Section A
 Ck Insttute f echnlgy Bachel f Engneeng (Hnus) n Chemcal and Pcess Engneeng Stage 3 Bachel f Engneeng n Chemcal and Pcess Engneeng Stage 3 (NFQ Level 8) Spng 005 DCE 3.5 hemdynamcs & Heat ansfe (me: 3
Ck Insttute f echnlgy Bachel f Engneeng (Hnus) n Chemcal and Pcess Engneeng Stage 3 Bachel f Engneeng n Chemcal and Pcess Engneeng Stage 3 (NFQ Level 8) Spng 005 DCE 3.5 hemdynamcs & Heat ansfe (me: 3
Module 7: Solved Problems
 Mdule 7: Slved Prblems 1 A tn-walled nentr tube eat exanger f 019-m lengt s t be used t eat denzed water frm 40 t 60 at a flw rate f 5 kg/s te denzed water flws trug te nner tube f 30-mm dameter wle t
Mdule 7: Slved Prblems 1 A tn-walled nentr tube eat exanger f 019-m lengt s t be used t eat denzed water frm 40 t 60 at a flw rate f 5 kg/s te denzed water flws trug te nner tube f 30-mm dameter wle t
is needed and this can be established by multiplying A, obtained in step 3, by, resulting V = A x y =. = x, located in 1 st quadrant rotated about 2
 Ct Cllege f New Yk MATH (Calculus Ntes) Page 1 f 1 Essental Calculus, nd edtn (Stewat) Chapte 7 Sectn, and 6 auth: M. Pak Chapte 7 sectn : Vlume Suface f evlutn (Dsc methd) 1) Estalsh the tatn as and the
Ct Cllege f New Yk MATH (Calculus Ntes) Page 1 f 1 Essental Calculus, nd edtn (Stewat) Chapte 7 Sectn, and 6 auth: M. Pak Chapte 7 sectn : Vlume Suface f evlutn (Dsc methd) 1) Estalsh the tatn as and the
LEAP FROG TECHNIQUE. Operational Simulation of LC Ladder Filters ECEN 622 (ESS) TAMU-AMSC
 LEAP FOG TEHNQUE Opeatnal Smulatn f L Ladde Fltes L pttype lw senstvty One fm f ths technque s called Leapf Technque Fundamental Buldn Blcks ae - nteats - Secnd-de ealzatns Fltes cnsdeed - LP - BP - HP
LEAP FOG TEHNQUE Opeatnal Smulatn f L Ladde Fltes L pttype lw senstvty One fm f ths technque s called Leapf Technque Fundamental Buldn Blcks ae - nteats - Secnd-de ealzatns Fltes cnsdeed - LP - BP - HP
ChE 471: LECTURE 4 Fall 2003
 ChE 47: LECTURE 4 Fall 003 IDEL RECTORS One f the key gals f chemical reactin engineering is t quantify the relatinship between prductin rate, reactr size, reactin kinetics and selected perating cnditins.
ChE 47: LECTURE 4 Fall 003 IDEL RECTORS One f the key gals f chemical reactin engineering is t quantify the relatinship between prductin rate, reactr size, reactin kinetics and selected perating cnditins.
Approach: (Equilibrium) TD analysis, i.e., conservation eqns., state equations Issues: how to deal with
 Schl f Aerspace Chemcal D: Mtvatn Prevus D Analyss cnsdered systems where cmpstn f flud was frzen fxed chemcal cmpstn Chemcally eactng Flw but there are numerus stuatns n prpulsn systems where chemcal
Schl f Aerspace Chemcal D: Mtvatn Prevus D Analyss cnsdered systems where cmpstn f flud was frzen fxed chemcal cmpstn Chemcally eactng Flw but there are numerus stuatns n prpulsn systems where chemcal
Chapter 7. Systems 7.1 INTRODUCTION 7.2 MATHEMATICAL MODELING OF LIQUID LEVEL SYSTEMS. Steady State Flow. A. Bazoune
 Chapter 7 Flud Systems and Thermal Systems 7.1 INTODUCTION A. Bazune A flud system uses ne r mre fluds t acheve ts purpse. Dampers and shck absrbers are eamples f flud systems because they depend n the
Chapter 7 Flud Systems and Thermal Systems 7.1 INTODUCTION A. Bazune A flud system uses ne r mre fluds t acheve ts purpse. Dampers and shck absrbers are eamples f flud systems because they depend n the
V. Principles of Irreversible Thermodynamics. s = S - S 0 (7.3) s = = - g i, k. "Flux": = da i. "Force": = -Â g a ik k = X i. Â J i X i (7.
 Themodynamcs and Knetcs of Solds 71 V. Pncples of Ievesble Themodynamcs 5. Onsage s Teatment s = S - S 0 = s( a 1, a 2,...) a n = A g - A n (7.6) Equlbum themodynamcs detemnes the paametes of an equlbum
Themodynamcs and Knetcs of Solds 71 V. Pncples of Ievesble Themodynamcs 5. Onsage s Teatment s = S - S 0 = s( a 1, a 2,...) a n = A g - A n (7.6) Equlbum themodynamcs detemnes the paametes of an equlbum
_J _J J J J J J J J _. 7 particles in the blue state; 3 particles in the red state: 720 configurations _J J J _J J J J J J J J _
 Dsrder and Suppse I have 10 partcles that can be n ne f tw states ether the blue state r the red state. Hw many dfferent ways can we arrange thse partcles amng the states? All partcles n the blue state:
Dsrder and Suppse I have 10 partcles that can be n ne f tw states ether the blue state r the red state. Hw many dfferent ways can we arrange thse partcles amng the states? All partcles n the blue state:
Final Exam Spring 2014 SOLUTION
 Appled Opts H-464/564 C 594 rtland State nverst A. La Rsa Fnal am Sprng 14 SOLTION Name There are tw questns 1%) plus an ptnal bnus questn 1%) 1. Quarter wave plates and half wave plates The fgures belw
Appled Opts H-464/564 C 594 rtland State nverst A. La Rsa Fnal am Sprng 14 SOLTION Name There are tw questns 1%) plus an ptnal bnus questn 1%) 1. Quarter wave plates and half wave plates The fgures belw
Exercise 10: Theory of mass transfer coefficient at boundary
 Partle Tehnology Laboratory Prof. Sotrs E. Pratsns Sonneggstrasse, ML F, ETH Zentrum Tel.: +--6 5 http://www.ptl.ethz.h 5-97- U Stoffaustaush HS 7 Exerse : Theory of mass transfer oeffent at boundary Chapter,
Partle Tehnology Laboratory Prof. Sotrs E. Pratsns Sonneggstrasse, ML F, ETH Zentrum Tel.: +--6 5 http://www.ptl.ethz.h 5-97- U Stoffaustaush HS 7 Exerse : Theory of mass transfer oeffent at boundary Chapter,
Chapter 6 : Gibbs Free Energy
 Wnter 01 Chem 54: ntrductry hermdynamcs Chapter 6 : Gbbs Free Energy... 64 Defntn f G, A... 64 Mawell Relatns... 65 Gbbs Free Energy G(,) (ure substances)... 67 Gbbs Free Energy fr Mtures... 68 ΔG f deal
Wnter 01 Chem 54: ntrductry hermdynamcs Chapter 6 : Gbbs Free Energy... 64 Defntn f G, A... 64 Mawell Relatns... 65 Gbbs Free Energy G(,) (ure substances)... 67 Gbbs Free Energy fr Mtures... 68 ΔG f deal
Physic 231 Lecture 33
 Physc 231 Lecture 33 Man pnts f tday s lecture: eat and heat capacty: Q cm Phase transtns and latent heat: Q Lm ( ) eat flw Q k 2 1 t L Examples f heat cnductvty, R values fr nsulatrs Cnvectn R L / k Radatn
Physc 231 Lecture 33 Man pnts f tday s lecture: eat and heat capacty: Q cm Phase transtns and latent heat: Q Lm ( ) eat flw Q k 2 1 t L Examples f heat cnductvty, R values fr nsulatrs Cnvectn R L / k Radatn
Lecture (10) Reactor Sizing and Design
 Lectue ( Rect Szng nd esgn. Genel Mle lnce Equtn Mle blnce n speces t ny nstnce n tme t ; lumn system te f flw te f genetn te f flw te f ccumultn f nt system f n systemby xn f ut f system f wthn system
Lectue ( Rect Szng nd esgn. Genel Mle lnce Equtn Mle blnce n speces t ny nstnce n tme t ; lumn system te f flw te f genetn te f flw te f ccumultn f nt system f n systemby xn f ut f system f wthn system
Mass Transfer (Stoffaustausch)
 Mass Tansfe (Stoffaustaush) Examination 3. August 3 Name: Legi-N.: Edition Diffusion by E. L. Cussle: none nd 3 d Test Duation: minutes The following mateials ae not pemitted at you table and have to be
Mass Tansfe (Stoffaustaush) Examination 3. August 3 Name: Legi-N.: Edition Diffusion by E. L. Cussle: none nd 3 d Test Duation: minutes The following mateials ae not pemitted at you table and have to be
Chapter 13 Experimental determination of kinetics of heterogeneous catalytic reactions and catalytic reactors design
 Chapte 3 Epemental detemnatn f net f hetegeneu atalyt eatn and atalyt eat degn OVEVIEW. teady tate tehnque. eat: ath, C, F. anpt gue at atle ale.3 net naly : ngle eatn, Cmple eatn, Etmatn Methd. anent
Chapte 3 Epemental detemnatn f net f hetegeneu atalyt eatn and atalyt eat degn OVEVIEW. teady tate tehnque. eat: ath, C, F. anpt gue at atle ale.3 net naly : ngle eatn, Cmple eatn, Etmatn Methd. anent
Transient Conduction: Spatial Effects and the Role of Analytical Solutions
 Transent Cnductn: Spatal Effects and the Rle f Analytcal Slutns Slutn t the Heat Equatn fr a Plane Wall wth Symmetrcal Cnvectn Cndtns If the lumped capactance apprxmatn can nt be made, cnsderatn must be
Transent Cnductn: Spatal Effects and the Rle f Analytcal Slutns Slutn t the Heat Equatn fr a Plane Wall wth Symmetrcal Cnvectn Cndtns If the lumped capactance apprxmatn can nt be made, cnsderatn must be
Analysis The characteristic length of the junction and the Biot number are
 -4 4 The temerature f a gas stream s t be measured by a thermule. The tme t taes t regster 99 erent f the ntal ΔT s t be determned. Assumtns The juntn s sheral n shae wth a dameter f D 0.00 m. The thermal
-4 4 The temerature f a gas stream s t be measured by a thermule. The tme t taes t regster 99 erent f the ntal ΔT s t be determned. Assumtns The juntn s sheral n shae wth a dameter f D 0.00 m. The thermal
Effects of high RH on α-pinene SOA phase partitioning
 Effets f hgh RH n α-pnene SA phase parttnng Götebrg summer shl 26/06/2008 Nenne Prsle PhD student Chemstry, CCAR, Unv. Cpenhagen, Denmark nlp@kem.ku.dk 26/06/2008 Götebrg, Nenne Prsle 1 Mtvatn Current
Effets f hgh RH n α-pnene SA phase parttnng Götebrg summer shl 26/06/2008 Nenne Prsle PhD student Chemstry, CCAR, Unv. Cpenhagen, Denmark nlp@kem.ku.dk 26/06/2008 Götebrg, Nenne Prsle 1 Mtvatn Current
Thermodynamics of solids 4. Statistical thermodynamics and the 3 rd law. Kwangheon Park Kyung Hee University Department of Nuclear Engineering
 Themodynamcs of solds 4. Statstcal themodynamcs and the 3 d law Kwangheon Pak Kyung Hee Unvesty Depatment of Nuclea Engneeng 4.1. Intoducton to statstcal themodynamcs Classcal themodynamcs Statstcal themodynamcs
Themodynamcs of solds 4. Statstcal themodynamcs and the 3 d law Kwangheon Pak Kyung Hee Unvesty Depatment of Nuclea Engneeng 4.1. Intoducton to statstcal themodynamcs Classcal themodynamcs Statstcal themodynamcs
Natural Circulation Systems
 Natural Crulatn Systems Natural rulatn lays an mrtant rle n the ln term ln f reatr systems durn adent ndtns, and n the strae f sent fuels. Cnsder the fllwn smle reresentatn f a reatr lant system eratn
Natural Crulatn Systems Natural rulatn lays an mrtant rle n the ln term ln f reatr systems durn adent ndtns, and n the strae f sent fuels. Cnsder the fllwn smle reresentatn f a reatr lant system eratn
Wp/Lmin. Wn/Lmin 2.5V
 UNIVERITY OF CALIFORNIA Cllege f Engneerng Department f Electrcal Engneerng and Cmputer cences Andre Vladmrescu Hmewrk #7 EEC Due Frday, Aprl 8 th, pm @ 0 Cry Prblem #.5V Wp/Lmn 0.0V Wp/Lmn n ut Wn/Lmn.5V
UNIVERITY OF CALIFORNIA Cllege f Engneerng Department f Electrcal Engneerng and Cmputer cences Andre Vladmrescu Hmewrk #7 EEC Due Frday, Aprl 8 th, pm @ 0 Cry Prblem #.5V Wp/Lmn 0.0V Wp/Lmn n ut Wn/Lmn.5V
WYSE Academic Challenge 2014 Sectional Physics Exam SOLUTION SET. [ F][ d] [ t] [ E]
![WYSE Academic Challenge 2014 Sectional Physics Exam SOLUTION SET. [ F][ d] [ t] [ E] WYSE Academic Challenge 2014 Sectional Physics Exam SOLUTION SET. [ F][ d] [ t] [ E]](/thumbs/93/114321029.jpg) WYSE Aaem Challenge 0 Setnal hss Exam SOLUTION SET. Crret answer: E Unts Trque / unts pwer: [ r ][ ] [ E] [ t] [ r ][ ][ t] [ E] [ r ][ ][ t] [ ][ ] [ r ][ t] [ ] m s m s. Crret answer: D The net external
WYSE Aaem Challenge 0 Setnal hss Exam SOLUTION SET. Crret answer: E Unts Trque / unts pwer: [ r ][ ] [ E] [ t] [ r ][ ][ t] [ E] [ r ][ ][ t] [ ][ ] [ r ][ t] [ ] m s m s. Crret answer: D The net external
Chem 116 POGIL Worksheet - Week 8 Equilibrium Continued - Solutions
 Chem 116 POGIL Wrksheet - Week 8 Equilibrium Cntinued - Slutins Key Questins 1. Cnsider the fllwing reatin At 425 C, an equilibrium mixture has the fllwing nentratins What is the value f K? -2 [HI] = 1.01
Chem 116 POGIL Wrksheet - Week 8 Equilibrium Cntinued - Slutins Key Questins 1. Cnsider the fllwing reatin At 425 C, an equilibrium mixture has the fllwing nentratins What is the value f K? -2 [HI] = 1.01
CSTR - PFR - PBR
 1. Mole Balances o The Rate of Reaction, - o The Geneal Mole Balance Equation o Continuous low Reactos - CSTR (Continuous-Stied Tank Reacto) - PR (Tubula Reacto) - PBR (Packed-Bed Reacto) o Industial Reactos
1. Mole Balances o The Rate of Reaction, - o The Geneal Mole Balance Equation o Continuous low Reactos - CSTR (Continuous-Stied Tank Reacto) - PR (Tubula Reacto) - PBR (Packed-Bed Reacto) o Industial Reactos
Electric potential energy Electrostatic force does work on a particle : Potential energy (: i initial state f : final state):
 Electc ptental enegy Electstatc fce des wk n a patcle : v v v v W = F s = E s. Ptental enegy (: ntal state f : fnal state): Δ U = U U = W. f ΔU Electc ptental : Δ : ptental enegy pe unt chag e. J ( Jule)
Electc ptental enegy Electstatc fce des wk n a patcle : v v v v W = F s = E s. Ptental enegy (: ntal state f : fnal state): Δ U = U U = W. f ΔU Electc ptental : Δ : ptental enegy pe unt chag e. J ( Jule)
Heat exchanger. Heat exchanger
 s are deves n w eat s transferred between tw fluds at dfferent teperatures wtut any xng f fluds. type. Dret eat transfer type 2. Strage type 3. Dret ntat type ttps://www.faebk./0000085304058/vdes/96230780490756/.
s are deves n w eat s transferred between tw fluds at dfferent teperatures wtut any xng f fluds. type. Dret eat transfer type 2. Strage type 3. Dret ntat type ttps://www.faebk./0000085304058/vdes/96230780490756/.
Chemical Engineering Department University of Washington
 Chemcal Engneerng Department Unversty of Washngton ChemE 60 - Exam I July 4, 003 - Mass Flow Rate of Steam Through a Turbne (5 onts) Steam enters a turbne at 70 o C and.8 Ma and leaves at 00 ka wth a qualty
Chemcal Engneerng Department Unversty of Washngton ChemE 60 - Exam I July 4, 003 - Mass Flow Rate of Steam Through a Turbne (5 onts) Steam enters a turbne at 70 o C and.8 Ma and leaves at 00 ka wth a qualty
6. ELUTRIATION OF PARTICLES FROM FLUIDIZED BEDS
 6. ELUTRIATION OF PARTICLES FROM FLUIDIZED BEDS Elutratn s the prcess n whch fne partcles are carred ut f a fludzed bed due t the flud flw rate passng thrugh the bed. Typcally, fne partcles are elutrated
6. ELUTRIATION OF PARTICLES FROM FLUIDIZED BEDS Elutratn s the prcess n whch fne partcles are carred ut f a fludzed bed due t the flud flw rate passng thrugh the bed. Typcally, fne partcles are elutrated
Design Equations. ν ij r i V R. ν ij r i. Q n components. = Q f c jf Qc j + Continuous Stirred Tank Reactor (steady-state and constant phase)
 Desgn Equatons Batch Reactor d(v R c j ) dt = ν j r V R n dt dt = UA(T a T) r H R V R ncomponents V R c j C pj j Plug Flow Reactor d(qc j ) dv = ν j r 2 dt dv = R U(T a T) n r H R Q n components j c j
Desgn Equatons Batch Reactor d(v R c j ) dt = ν j r V R n dt dt = UA(T a T) r H R V R ncomponents V R c j C pj j Plug Flow Reactor d(qc j ) dv = ν j r 2 dt dv = R U(T a T) n r H R Q n components j c j
HCB-3 Edition. Solutions Chapter 12 Problems. SOLUTION: Refer to saturated steam table (Table A3-SI) and superheated steam table (Table A4-SI)
 HCB- Editin 12.1 Slutins Chapter 12 Prbles GIVEN: Fllwing table fr water: T (C p (kpa v ( /kg Phase 60 (1.25 (2 ( 175 (4 Saturated vapr 00 00 (5 (6 100 10 (7 (8 (9 (10 0.001097 Saturated vapr 1000 10 (11
HCB- Editin 12.1 Slutins Chapter 12 Prbles GIVEN: Fllwing table fr water: T (C p (kpa v ( /kg Phase 60 (1.25 (2 ( 175 (4 Saturated vapr 00 00 (5 (6 100 10 (7 (8 (9 (10 0.001097 Saturated vapr 1000 10 (11
Chapter 3, Solution 1C.
 COSMOS: Cmplete Onlne Slutns Manual Organzatn System Chapter 3, Slutn C. (a If the lateral surfaces f the rd are nsulated, the heat transfer surface area f the cylndrcal rd s the bttm r the tp surface
COSMOS: Cmplete Onlne Slutns Manual Organzatn System Chapter 3, Slutn C. (a If the lateral surfaces f the rd are nsulated, the heat transfer surface area f the cylndrcal rd s the bttm r the tp surface
Section 3: Detailed Solutions of Word Problems Unit 1: Solving Word Problems by Modeling with Formulas
 Sectn : Detaled Slutns f Wrd Prblems Unt : Slvng Wrd Prblems by Mdelng wth Frmulas Example : The factry nvce fr a mnvan shws that the dealer pad $,5 fr the vehcle. If the stcker prce f the van s $5,, hw
Sectn : Detaled Slutns f Wrd Prblems Unt : Slvng Wrd Prblems by Mdelng wth Frmulas Example : The factry nvce fr a mnvan shws that the dealer pad $,5 fr the vehcle. If the stcker prce f the van s $5,, hw
CIRCLE YOUR DIVISION: Div. 1 (9:30 am) Div. 2 (11:30 am) Div. 3 (2:30 pm) Prof. Ruan Prof. Naik Mr. Singh
 Frst CIRCLE YOUR DIVISION: Dv. 1 (9:30 am) Dv. (11:30 am) Dv. 3 (:30 m) Prf. Ruan Prf. Na Mr. Sngh Schl f Mechancal Engneerng Purdue Unversty ME315 Heat and Mass ransfer Eam #3 Wednesday Nvember 17 010
Frst CIRCLE YOUR DIVISION: Dv. 1 (9:30 am) Dv. (11:30 am) Dv. 3 (:30 m) Prf. Ruan Prf. Na Mr. Sngh Schl f Mechancal Engneerng Purdue Unversty ME315 Heat and Mass ransfer Eam #3 Wednesday Nvember 17 010
Combustion Chamber. (0.1 MPa)
 ME 354 Tutial #10 Winte 001 Reacting Mixtues Pblem 1: Detemine the mle actins the pducts cmbustin when ctane, C 8 18, is buned with 00% theetical ai. Als, detemine the dew-pint tempeatue the pducts i the
ME 354 Tutial #10 Winte 001 Reacting Mixtues Pblem 1: Detemine the mle actins the pducts cmbustin when ctane, C 8 18, is buned with 00% theetical ai. Als, detemine the dew-pint tempeatue the pducts i the
Contact, information, consultations
 ontact, nfomaton, consultatons hemsty A Bldg; oom 07 phone: 058-347-769 cellula: 664 66 97 E-mal: wojtek_c@pg.gda.pl Offce hous: Fday, 9-0 a.m. A quote of the week (o camel of the week): hee s no expedence
ontact, nfomaton, consultatons hemsty A Bldg; oom 07 phone: 058-347-769 cellula: 664 66 97 E-mal: wojtek_c@pg.gda.pl Offce hous: Fday, 9-0 a.m. A quote of the week (o camel of the week): hee s no expedence
Dynamic simulation of multi-effect falling-film evaporator: Milk powder production plant
 Dynam smulatn f mult-effet fallng-flm evapratr: Mlk pwder prdutn plant Fateme Medhat Bjnurd, Mhammad Al Fanae *, Had Zhree Department f Chem. Eng., Faulty f Eng., Ferdws Unversty f Mashhad, I. R. Iran
Dynam smulatn f mult-effet fallng-flm evapratr: Mlk pwder prdutn plant Fateme Medhat Bjnurd, Mhammad Al Fanae *, Had Zhree Department f Chem. Eng., Faulty f Eng., Ferdws Unversty f Mashhad, I. R. Iran
Statistical mechanics canonical ensemble
 canoncal ensemble system n thermal equlbrum wth bath of system probablty of mcro state T = k/ bath system p = 1 Z e E Z = e E average energy of system he = p E = what about? P E E e P e E = @ ln Z @ replcate
canoncal ensemble system n thermal equlbrum wth bath of system probablty of mcro state T = k/ bath system p = 1 Z e E Z = e E average energy of system he = p E = what about? P E E e P e E = @ ln Z @ replcate
Introduction of Two Port Network Negative Feedback (Uni lateral Case) Feedback Topology Analysis of feedback applications
 Lectue Feedback mple ntductn w Pt Netwk Negatve Feedback Un lateal Case Feedback plg nalss eedback applcatns Clse Lp Gan nput/output esstances e:83h 3 Feedback w-pt Netwk z-paametes Open-Ccut mpedance
Lectue Feedback mple ntductn w Pt Netwk Negatve Feedback Un lateal Case Feedback plg nalss eedback applcatns Clse Lp Gan nput/output esstances e:83h 3 Feedback w-pt Netwk z-paametes Open-Ccut mpedance
Ph.D. Qualifying Examination in Kinetics and Reactor Design
 Knetcs and Reactor Desgn Ph.D.Qualfyng Examnaton January 2006 Instructons Ph.D. Qualfyng Examnaton n Knetcs and Reactor Desgn January 2006 Unversty of Texas at Austn Department of Chemcal Engneerng 1.
Knetcs and Reactor Desgn Ph.D.Qualfyng Examnaton January 2006 Instructons Ph.D. Qualfyng Examnaton n Knetcs and Reactor Desgn January 2006 Unversty of Texas at Austn Department of Chemcal Engneerng 1.
6.3.7 Example with Runga Kutta 4 th order method
 6.3.7 Example wth Runga Kutta 4 th order method Agan, as an example, 3 machne, 9 bus system shown n Fg. 6.4 s agan consdered. Intally, the dampng of the generators are neglected (.e. d = 0 for = 1, 2,
6.3.7 Example wth Runga Kutta 4 th order method Agan, as an example, 3 machne, 9 bus system shown n Fg. 6.4 s agan consdered. Intally, the dampng of the generators are neglected (.e. d = 0 for = 1, 2,
ELABORATING AND ANALYSING THE REAL BALANCE OF HEAT FOR THE STEAM GENERATOR RGL10/D-D
 Annals f te Unversty f Petrşan, Mecancal Engneerng, 10 (2008), 155-160 155 ELABORATING AND ANALYSING THE REAL BALANCE OF HEAT FOR THE STEAM GENERATOR RGL10/D-D DAN CODRUŢ PETRILEAN 1 Abstract: Te real
Annals f te Unversty f Petrşan, Mecancal Engneerng, 10 (2008), 155-160 155 ELABORATING AND ANALYSING THE REAL BALANCE OF HEAT FOR THE STEAM GENERATOR RGL10/D-D DAN CODRUŢ PETRILEAN 1 Abstract: Te real
Conduction Heat Transfer
 Cnductn Heat Transfer Practce prblems A steel ppe f cnductvty 5 W/m-K has nsde and utsde surface temperature f C and 6 C respectvely Fnd the heat flw rate per unt ppe length and flux per unt nsde and per
Cnductn Heat Transfer Practce prblems A steel ppe f cnductvty 5 W/m-K has nsde and utsde surface temperature f C and 6 C respectvely Fnd the heat flw rate per unt ppe length and flux per unt nsde and per
Analytical Modeling of Natural Convection in Horizontal Annuli
 Analytcal Mdelng f Natural Cnvectn n Hrzntal Annul Peter Teertstra, M. Mchael Yvanvch, J. Rchard Culham Mcrelectrncs Heat Transfer Labratry Department f Mechancal Engneerng Unversty f Waterl Waterl, Ontar,
Analytcal Mdelng f Natural Cnvectn n Hrzntal Annul Peter Teertstra, M. Mchael Yvanvch, J. Rchard Culham Mcrelectrncs Heat Transfer Labratry Department f Mechancal Engneerng Unversty f Waterl Waterl, Ontar,
Transistors. Lesson #10 Chapter 4. BME 372 Electronics I J.Schesser
 Tanssts essn #10 Chapte 4 BM 372 lectncs 154 Hmewk Ps. 4.40, 4.42, 4.43, 4.45, 4.46, 4.51, 4.53, 4.54, 4.56 BM 372 lectncs 155 Hmewk Answes #20 Ps. 4.40 See fgue 4.33 BM 372 lectncs 156 Ps. 4.42 Hmewk
Tanssts essn #10 Chapte 4 BM 372 lectncs 154 Hmewk Ps. 4.40, 4.42, 4.43, 4.45, 4.46, 4.51, 4.53, 4.54, 4.56 BM 372 lectncs 155 Hmewk Answes #20 Ps. 4.40 See fgue 4.33 BM 372 lectncs 156 Ps. 4.42 Hmewk
Applied CFD Project 1. Christopher Light MAE 598
 Applied CFD Project 1 Christopher Light MAE 598 October 5, 2017 Task 1 The hot water tank shown in Fig 1 is used for analysis of cool water flow with the heat from a hot plate at the bottom. For all tasks,
Applied CFD Project 1 Christopher Light MAE 598 October 5, 2017 Task 1 The hot water tank shown in Fig 1 is used for analysis of cool water flow with the heat from a hot plate at the bottom. For all tasks,
WYSE Academic Challenge Sectional Mathematics 2006 Solution Set
 WYSE Academic Challenge Sectinal 006 Slutin Set. Cect answe: e. mph is 76 feet pe minute, and 4 mph is 35 feet pe minute. The tip up the hill takes 600/76, 3.4 minutes, and the tip dwn takes 600/35,.70
WYSE Academic Challenge Sectinal 006 Slutin Set. Cect answe: e. mph is 76 feet pe minute, and 4 mph is 35 feet pe minute. The tip up the hill takes 600/76, 3.4 minutes, and the tip dwn takes 600/35,.70
Lecture 2 Feedback Amplifier
 Lectue Feedback mple ntductn w-pt Netwk Negatve Feedback Un-lateal Case Feedback plg nalss eedback applcatns Clse-Lp Gan nput/output esstances e:83hkn 3 Feedback mples w-pt Netwk z-paametes Open-Ccut mpedance
Lectue Feedback mple ntductn w-pt Netwk Negatve Feedback Un-lateal Case Feedback plg nalss eedback applcatns Clse-Lp Gan nput/output esstances e:83hkn 3 Feedback mples w-pt Netwk z-paametes Open-Ccut mpedance
ME306 Dynamics, Spring HW1 Solution Key. AB, where θ is the angle between the vectors A and B, the proof
 ME6 Dnms, Spng HW Slutn Ke - Pve, gemetll.e. usng wngs sethes n nltll.e. usng equtns n nequltes, tht V then V. Nte: qunttes n l tpee e vets n n egul tpee e sls. Slutn: Let, Then V V V We wnt t pve tht:
ME6 Dnms, Spng HW Slutn Ke - Pve, gemetll.e. usng wngs sethes n nltll.e. usng equtns n nequltes, tht V then V. Nte: qunttes n l tpee e vets n n egul tpee e sls. Slutn: Let, Then V V V We wnt t pve tht:
CHEM 1413 Chapter 6 Homework Questions TEXTBOOK HOMEWORK
 CHEM 1413 Chapter 6 Hmewrk Questins TEXTBOOK HOMEWORK 6.25 A 27.7-g sample f the radiatr clant ethylene glycl releases 688 J f heat. What was the initial temperature f the sample if the final temperature
CHEM 1413 Chapter 6 Hmewrk Questins TEXTBOOK HOMEWORK 6.25 A 27.7-g sample f the radiatr clant ethylene glycl releases 688 J f heat. What was the initial temperature f the sample if the final temperature
Multi-dimensional Central Limit Theorem
 Mult-dmensonal Central Lmt heorem Outlne ( ( ( t as ( + ( + + ( ( ( Consder a sequence of ndependent random proceses t, t, dentcal to some ( t. Assume t = 0. Defne the sum process t t t t = ( t = (; t
Mult-dmensonal Central Lmt heorem Outlne ( ( ( t as ( + ( + + ( ( ( Consder a sequence of ndependent random proceses t, t, dentcal to some ( t. Assume t = 0. Defne the sum process t t t t = ( t = (; t
Equation of State Modeling of Phase Equilibrium in the Low-Density Polyethylene Process
 Equaton of State Modelng of Phase Equlbrum n the Low-Densty Polyethylene Process H. Orbey, C. P. Boks, and C. C. Chen Ind. Eng. Chem. Res. 1998, 37, 4481-4491 Yong Soo Km Thermodynamcs & Propertes Lab.
Equaton of State Modelng of Phase Equlbrum n the Low-Densty Polyethylene Process H. Orbey, C. P. Boks, and C. C. Chen Ind. Eng. Chem. Res. 1998, 37, 4481-4491 Yong Soo Km Thermodynamcs & Propertes Lab.
The Gradient and Applications This unit is based on Sections 9.5 and 9.6, Chapter 9. All assigned readings and exercises are from the textbook
 The Gadient and Applicatins This unit is based n Sectins 9.5 and 9.6 Chapte 9. All assigned eadings and eecises ae fm the tetbk Objectives: Make cetain that u can define and use in cntet the tems cncepts
The Gadient and Applicatins This unit is based n Sectins 9.5 and 9.6 Chapte 9. All assigned eadings and eecises ae fm the tetbk Objectives: Make cetain that u can define and use in cntet the tems cncepts
SCHMIDT THEORY FOR STIRLING ENGINES
 SHMIDT THOY FO STILING NGINS KOIHI HIATA Musashin-jjutaku 6-10, Gakuen -6-1, Musashimurayama, Tky 08, Japan Phne & Fax: +81-45-67-0086 e-mail: khirata@gem.bekkame.ne.jp url: http://www.bekkame.ne.jp/~khirata
SHMIDT THOY FO STILING NGINS KOIHI HIATA Musashin-jjutaku 6-10, Gakuen -6-1, Musashimurayama, Tky 08, Japan Phne & Fax: +81-45-67-0086 e-mail: khirata@gem.bekkame.ne.jp url: http://www.bekkame.ne.jp/~khirata
Question 1. Cold air standard assumptions:
 Qestn (a A 4-stke 8-yde 3. lte tb-aged spak gntn ang engne as a mpessn at 0, and s n at 6000 pm. e a dawn nt te engne bee mpessn as a tempeate 5 C, and a pesse 40 000 kpa. e tempeate te a at te end te
Qestn (a A 4-stke 8-yde 3. lte tb-aged spak gntn ang engne as a mpessn at 0, and s n at 6000 pm. e a dawn nt te engne bee mpessn as a tempeate 5 C, and a pesse 40 000 kpa. e tempeate te a at te end te
Randomness and Computation
 Randomness and Computaton or, Randomzed Algorthms Mary Cryan School of Informatcs Unversty of Ednburgh RC 208/9) Lecture 0 slde Balls n Bns m balls, n bns, and balls thrown unformly at random nto bns usually
Randomness and Computaton or, Randomzed Algorthms Mary Cryan School of Informatcs Unversty of Ednburgh RC 208/9) Lecture 0 slde Balls n Bns m balls, n bns, and balls thrown unformly at random nto bns usually
Int. J. of Applied Mechanics and Engineering, 2014, vol.19, No.3, pp DOI: /ijame
 Int. J. f Appled Mechancs and Engneerng, 2014, vl.19, N.3, pp.539-548 DOI: 10.2478/jame-2014-0036 APPLICATION OF MULTI-VALUED WEIGHTING LOGICAL FUNCTIONS IN THE ANALYSIS OF A DEGREE OF IMPORTANCE OF CONSTRUCTION
Int. J. f Appled Mechancs and Engneerng, 2014, vl.19, N.3, pp.539-548 DOI: 10.2478/jame-2014-0036 APPLICATION OF MULTI-VALUED WEIGHTING LOGICAL FUNCTIONS IN THE ANALYSIS OF A DEGREE OF IMPORTANCE OF CONSTRUCTION
How can standard heats of formation be used to calculate the heat of a reaction?
 Answer Key ALE 28. ess s Law and Standard Enthalpies Frmatin (Reerence: Chapter 6 - Silberberg 4 th editin) Imprtant!! Fr answers that invlve a calculatin yu must shw yur wrk neatly using dimensinal analysis
Answer Key ALE 28. ess s Law and Standard Enthalpies Frmatin (Reerence: Chapter 6 - Silberberg 4 th editin) Imprtant!! Fr answers that invlve a calculatin yu must shw yur wrk neatly using dimensinal analysis
Compressibility Effects
 Definitin f Cmpressibility All real substances are cmpressible t sme greater r lesser extent; that is, when yu squeeze r press n them, their density will change The amunt by which a substance can be cmpressed
Definitin f Cmpressibility All real substances are cmpressible t sme greater r lesser extent; that is, when yu squeeze r press n them, their density will change The amunt by which a substance can be cmpressed
Applied Stochastic Processes
 STAT455/855 Fall 23 Appled Stochastc Processes Fnal Exam, Bref Solutons 1. (15 marks) (a) (7 marks) The dstrbuton of Y s gven by ( ) ( ) y 2 1 5 P (Y y) for y 2, 3,... The above follows because each of
STAT455/855 Fall 23 Appled Stochastc Processes Fnal Exam, Bref Solutons 1. (15 marks) (a) (7 marks) The dstrbuton of Y s gven by ( ) ( ) y 2 1 5 P (Y y) for y 2, 3,... The above follows because each of
Rotating Disk Electrode -a hydrodynamic method
 Rotatng Dsk Electode -a hdodnamc method Fe Lu Ma 3, 0 ente fo Electochemcal Engneeng Reseach Depatment of hemcal and Bomolecula Engneeng Rotatng Dsk Electode A otatng dsk electode RDE s a hdodnamc wokng
Rotatng Dsk Electode -a hdodnamc method Fe Lu Ma 3, 0 ente fo Electochemcal Engneeng Reseach Depatment of hemcal and Bomolecula Engneeng Rotatng Dsk Electode A otatng dsk electode RDE s a hdodnamc wokng
Review for the Mid-Term Exam
 Revew f the Md-Tem am A54/MA4/M 59/ Spg 8 Depatmet f Mechacal & Aespace geeg Chapte Md-Tem Revew-6 Date: Mach (Thusda), 8 Tme: :pm-:pm Place: Rm, Neddema Hall Smple devat Md-Tem am Pat : 4 pblems Smple
Revew f the Md-Tem am A54/MA4/M 59/ Spg 8 Depatmet f Mechacal & Aespace geeg Chapte Md-Tem Revew-6 Date: Mach (Thusda), 8 Tme: :pm-:pm Place: Rm, Neddema Hall Smple devat Md-Tem am Pat : 4 pblems Smple
5. Collection and Analysis of. Rate Data
 5. Collection and nalysis of o Objectives Rate Data - Determine the reaction order and specific reaction rate from experimental data obtained from either batch or flow reactors - Describe how to analyze
5. Collection and nalysis of o Objectives Rate Data - Determine the reaction order and specific reaction rate from experimental data obtained from either batch or flow reactors - Describe how to analyze
Youn-Woo Lee School of Chemical and Biological Engineering Seoul National University , 599 Gwanangro, Gwanak-gu, Seoul, Korea
 hemical Reacto esign Y W L Youn-Woo Lee School of hemical and iological Engineeing 55-74, 599 Gwanango, Gwana-gu, Seoul, Koea ywlee@snu.ac. http://sfpl.snu.ac. hapte 6 Multiple Reactions hemical Reaction
hemical Reacto esign Y W L Youn-Woo Lee School of hemical and iological Engineeing 55-74, 599 Gwanango, Gwana-gu, Seoul, Koea ywlee@snu.ac. http://sfpl.snu.ac. hapte 6 Multiple Reactions hemical Reaction
Chemical Reaction Engineering Lecture 5
 Chemical Reaction Engineering g Lecture 5 The Scope The im of the Course: To learn how to describe a system where a (bio)chemical reaction takes place (further called reactor) Reactors Pharmacokinetics
Chemical Reaction Engineering g Lecture 5 The Scope The im of the Course: To learn how to describe a system where a (bio)chemical reaction takes place (further called reactor) Reactors Pharmacokinetics
UNIT10 PLANE OF REGRESSION
 UIT0 PLAE OF REGRESSIO Plane of Regesson Stuctue 0. Intoducton Ojectves 0. Yule s otaton 0. Plane of Regesson fo thee Vaales 0.4 Popetes of Resduals 0.5 Vaance of the Resduals 0.6 Summay 0.7 Solutons /
UIT0 PLAE OF REGRESSIO Plane of Regesson Stuctue 0. Intoducton Ojectves 0. Yule s otaton 0. Plane of Regesson fo thee Vaales 0.4 Popetes of Resduals 0.5 Vaance of the Resduals 0.6 Summay 0.7 Solutons /
Outline. Unit Eight Calculations with Entropy. The Second Law. Second Law Notes. Uses of Entropy. Entropy is a Property.
 Unt Eght Calculatons wth Entropy Mechancal Engneerng 370 Thermodynamcs Larry Caretto October 6, 010 Outlne Quz Seven Solutons Second law revew Goals for unt eght Usng entropy to calculate the maxmum work
Unt Eght Calculatons wth Entropy Mechancal Engneerng 370 Thermodynamcs Larry Caretto October 6, 010 Outlne Quz Seven Solutons Second law revew Goals for unt eght Usng entropy to calculate the maxmum work
Exploiting vector space properties for the global optimization of process networks
 Exptng vectr space prpertes fr the gbal ptmzatn f prcess netwrks Juan ab Ruz Ignac Grssmann Enterprse Wde Optmzatn Meetng March 00 Mtvatn - The ptmzatn f prcess netwrks s ne f the mst frequent prblems
Exptng vectr space prpertes fr the gbal ptmzatn f prcess netwrks Juan ab Ruz Ignac Grssmann Enterprse Wde Optmzatn Meetng March 00 Mtvatn - The ptmzatn f prcess netwrks s ne f the mst frequent prblems
State-Space Model Based Generalized Predictive Control for Networked Control Systems
 Prceedngs f the 7th Wrld Cngress he Internatnal Federatn f Autmatc Cntrl State-Space Mdel Based Generalzed Predctve Cntrl fr Netwred Cntrl Systems Bn ang* Gu-Png Lu** We-Hua Gu*** and Ya-Ln Wang**** *Schl
Prceedngs f the 7th Wrld Cngress he Internatnal Federatn f Autmatc Cntrl State-Space Mdel Based Generalzed Predctve Cntrl fr Netwred Cntrl Systems Bn ang* Gu-Png Lu** We-Hua Gu*** and Ya-Ln Wang**** *Schl
SELECTION OF MODEL PARAMETERS OF BIOGAS IC ENGINE. Karol Cupiał, Grzegorz Katolik
 TEKA Km. Mt. Energ. Rln., 2006, 6A, 32 38 SELECTION OF MODEL PARAMETERS OF BIOGAS IC ENGINE Karl Cupał, Grzegrz Katlk Insttute f Internal Cmbustn Engnes and Cntrl Engneerng Techncal Unversty f Częstchwa
TEKA Km. Mt. Energ. Rln., 2006, 6A, 32 38 SELECTION OF MODEL PARAMETERS OF BIOGAS IC ENGINE Karl Cupał, Grzegrz Katlk Insttute f Internal Cmbustn Engnes and Cntrl Engneerng Techncal Unversty f Częstchwa
Lecture 8. Chapter 7. - Thermodynamic Web - Departure Functions - Review Equations of state (chapter 4, briefly)
 Lecture 8 Chapter 5 - Thermodynamc Web - Departure Functons - Revew Equatons of state (chapter 4, brefly) Chapter 6 - Equlbrum (chemcal potental) * Pure Component * Mxtures Chapter 7 - Fugacty (chemcal
Lecture 8 Chapter 5 - Thermodynamc Web - Departure Functons - Revew Equatons of state (chapter 4, brefly) Chapter 6 - Equlbrum (chemcal potental) * Pure Component * Mxtures Chapter 7 - Fugacty (chemcal
6-5. H 2 O 200 kpa 200 C Q. Entropy Changes of Pure Substances
 Canges f ure Substances 6-0C Yes, because an ternally reversible, adiabatic prcess vlves n irreversibilities r eat transfer. 6- e radiatr f a steam eatg system is itially filled wit supereated steam. e
Canges f ure Substances 6-0C Yes, because an ternally reversible, adiabatic prcess vlves n irreversibilities r eat transfer. 6- e radiatr f a steam eatg system is itially filled wit supereated steam. e
A. Thicknesses and Densities
 10 Lab0 The Eath s Shells A. Thcknesses and Denstes Any theoy of the nteo of the Eath must be consstent wth the fact that ts aggegate densty s 5.5 g/cm (ecall we calculated ths densty last tme). In othe
10 Lab0 The Eath s Shells A. Thcknesses and Denstes Any theoy of the nteo of the Eath must be consstent wth the fact that ts aggegate densty s 5.5 g/cm (ecall we calculated ths densty last tme). In othe
(element) But, we do NOT know G!!! Correct, but not applicable! Free Energy Problems: o r. products. reactants. o f. reactants.
 SANDARD fr hemcal Rxns prducts G p G reactants r But, we d NO knw G!!! rrect, but nt applcable! prducts G f G reactants f f (element) 0 f standard Gbbs free energy f frmatn Free Energy Prblems: 5. Prf.
SANDARD fr hemcal Rxns prducts G p G reactants r But, we d NO knw G!!! rrect, but nt applcable! prducts G f G reactants f f (element) 0 f standard Gbbs free energy f frmatn Free Energy Prblems: 5. Prf.
Mass Transfer (Stoffaustausch) Fall 2012
 Mass Transfer (Stoffaustaush) Fall Examination 9. Januar Name: Legi-Nr.: Edition Diffusion by E. L. Cussler: none nd rd Test Duration: minutes The following materials are not permitted at your table and
Mass Transfer (Stoffaustaush) Fall Examination 9. Januar Name: Legi-Nr.: Edition Diffusion by E. L. Cussler: none nd rd Test Duration: minutes The following materials are not permitted at your table and
PHY2053 Summer 2012 Exam 2 Solutions N F o f k
 HY0 Suer 0 Ea Slutns. he ree-bdy dagra r the blck s N F 7 k F g Usng Newtn s secnd law r the -cnents F a F F cs7 k 0 k F F cs7 (0 N ( Ncs7 N he wrk dne by knetc rctn k r csθ ( N(6 cs80 0 N. Mechancal energy
HY0 Suer 0 Ea Slutns. he ree-bdy dagra r the blck s N F 7 k F g Usng Newtn s secnd law r the -cnents F a F F cs7 k 0 k F F cs7 (0 N ( Ncs7 N he wrk dne by knetc rctn k r csθ ( N(6 cs80 0 N. Mechancal energy
Gasometric Determination of NaHCO 3 in a Mixture
 60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
One-Dimensional, Steady-State. State Conduction with Thermal Energy Generation
 One-Dimensional, Steady-State State Conduction with Themal Enegy Geneation Implications of Enegy Geneation Involves a local (volumetic) souce of themal enegy due to convesion fom anothe fom of enegy in
One-Dimensional, Steady-State State Conduction with Themal Enegy Geneation Implications of Enegy Geneation Involves a local (volumetic) souce of themal enegy due to convesion fom anothe fom of enegy in
Chemical Reaction Engineering
 CHPTE 7 Chemical eaction Engineering (Gate 00). The conversion for a second order, irreversible reaction (constant volume) () k B, in batch mode is given by k C t o ( kcot) (C) k C t o + (D) kcot (B) k
CHPTE 7 Chemical eaction Engineering (Gate 00). The conversion for a second order, irreversible reaction (constant volume) () k B, in batch mode is given by k C t o ( kcot) (C) k C t o + (D) kcot (B) k
11/19/2013. PHY 113 C General Physics I 11 AM 12:15 PM MWF Olin 101
 PHY 113 C General Pyss I 11 AM 12:15 PM MWF Oln 101 Plan or Leture 23: Capter 22: Heat engnes 1. ermodynam yles; work and eat eeny 2. Carnot yle 3. Otto yle; desel yle 4. Bre omments on entropy 11/19/2013
PHY 113 C General Pyss I 11 AM 12:15 PM MWF Oln 101 Plan or Leture 23: Capter 22: Heat engnes 1. ermodynam yles; work and eat eeny 2. Carnot yle 3. Otto yle; desel yle 4. Bre omments on entropy 11/19/2013
Principles of Food and Bioprocess Engineering (FS 231) Solutions to Example Problems on Heat Transfer
 Prncples of Food and Boprocess Engneerng (FS 31) Solutons to Example Problems on Heat Transfer 1. We start wth Fourer s law of heat conducton: Q = k A ( T/ x) Rearrangng, we get: Q/A = k ( T/ x) Here,
Prncples of Food and Boprocess Engneerng (FS 31) Solutons to Example Problems on Heat Transfer 1. We start wth Fourer s law of heat conducton: Q = k A ( T/ x) Rearrangng, we get: Q/A = k ( T/ x) Here,
EE 221 Practice Problems for the Final Exam
 EE 1 Practce Prblems fr the Fnal Exam 1. The netwrk functn f a crcut s 1.5 H. ω 1+ j 500 Ths table recrds frequency respnse data fr ths crcut. Fll n the blanks n the table:. The netwrk functn f a crcut
EE 1 Practce Prblems fr the Fnal Exam 1. The netwrk functn f a crcut s 1.5 H. ω 1+ j 500 Ths table recrds frequency respnse data fr ths crcut. Fll n the blanks n the table:. The netwrk functn f a crcut
Examiner: Dr. Mohamed Elsharnoby Time: 180 min. Attempt all the following questions Solve the following five questions, and assume any missing data
 Benha University Cllege f Engineering at Banha Department f Mechanical Eng. First Year Mechanical Subject : Fluid Mechanics M111 Date:4/5/016 Questins Fr Final Crrective Examinatin Examiner: Dr. Mhamed
Benha University Cllege f Engineering at Banha Department f Mechanical Eng. First Year Mechanical Subject : Fluid Mechanics M111 Date:4/5/016 Questins Fr Final Crrective Examinatin Examiner: Dr. Mhamed
Phys101 Final Code: 1 Term: 132 Wednesday, May 21, 2014 Page: 1
 Phys101 Final Cde: 1 Term: 1 Wednesday, May 1, 014 Page: 1 Q1. A car accelerates at.0 m/s alng a straight rad. It passes tw marks that are 0 m apart at times t = 4.0 s and t = 5.0 s. Find the car s velcity
Phys101 Final Cde: 1 Term: 1 Wednesday, May 1, 014 Page: 1 Q1. A car accelerates at.0 m/s alng a straight rad. It passes tw marks that are 0 m apart at times t = 4.0 s and t = 5.0 s. Find the car s velcity
Design of Analog Integrated Circuits
 Desgn f Analg Integrated Crcuts I. Amplfers Desgn f Analg Integrated Crcuts Fall 2012, Dr. Guxng Wang 1 Oerew Basc MOS amplfer structures Cmmn-Surce Amplfer Surce Fllwer Cmmn-Gate Amplfer Desgn f Analg
Desgn f Analg Integrated Crcuts I. Amplfers Desgn f Analg Integrated Crcuts Fall 2012, Dr. Guxng Wang 1 Oerew Basc MOS amplfer structures Cmmn-Surce Amplfer Surce Fllwer Cmmn-Gate Amplfer Desgn f Analg
Physics 231 Ch 9 Day
 Physs Ch 9 Day 0 0 Wed., /6 ab F, /8 n., / Tues. / 9. Rtatnal Enegy Quz 8 8 Enegy Quantzatn Reew Exam (Ch 5-8) Exam (Ch 5-8) RE 9.b bng laptp, smatphne, pad, Pate Exam (due begnnng lass) 9.-.5 (.9) The
Physs Ch 9 Day 0 0 Wed., /6 ab F, /8 n., / Tues. / 9. Rtatnal Enegy Quz 8 8 Enegy Quantzatn Reew Exam (Ch 5-8) Exam (Ch 5-8) RE 9.b bng laptp, smatphne, pad, Pate Exam (due begnnng lass) 9.-.5 (.9) The
EEE 241: Linear Systems
 EEE : Lnear Systems Summary #: Backpropagaton BACKPROPAGATION The perceptron rule as well as the Wdrow Hoff learnng were desgned to tran sngle layer networks. They suffer from the same dsadvantage: they
EEE : Lnear Systems Summary #: Backpropagaton BACKPROPAGATION The perceptron rule as well as the Wdrow Hoff learnng were desgned to tran sngle layer networks. They suffer from the same dsadvantage: they
IGEE 401 Power Electronic Systems. Solution to Midterm Examination Fall 2004
 Jós, G GEE 401 wer Electrnc Systems Slutn t Mdterm Examnatn Fall 2004 Specal nstructns: - Duratn: 75 mnutes. - Materal allwed: a crb sheet (duble sded 8.5 x 11), calculatr. - Attempt all questns. Make
Jós, G GEE 401 wer Electrnc Systems Slutn t Mdterm Examnatn Fall 2004 Specal nstructns: - Duratn: 75 mnutes. - Materal allwed: a crb sheet (duble sded 8.5 x 11), calculatr. - Attempt all questns. Make
Thermodynamics of Materials
 Thermdynamcs f Materals 14th Lecture 007. 4. 8 (Mnday) FUGACITY dg = Vd SdT dg = Vd at cnstant T Fr an deal gas dg = (RT/)d = RT dln Ths s true fr deal gases nly, but t wuld be nce t have a smlar frm fr
Thermdynamcs f Materals 14th Lecture 007. 4. 8 (Mnday) FUGACITY dg = Vd SdT dg = Vd at cnstant T Fr an deal gas dg = (RT/)d = RT dln Ths s true fr deal gases nly, but t wuld be nce t have a smlar frm fr
FINAL REPORT NO. Ill February 1, 1964
 O FNAL REPORT NO. ll Febuay, 964 MASS. MOMENTUM, AND HEAT TRANSFER WTHN A TURBULENT BOUNDARY LAYER WTH FOREGN GAS MASS TRANSFER AT THE SURFACE PART l-constant FLUD PROPERTES :J by Rbet M. Kendall Ms W.
O FNAL REPORT NO. ll Febuay, 964 MASS. MOMENTUM, AND HEAT TRANSFER WTHN A TURBULENT BOUNDARY LAYER WTH FOREGN GAS MASS TRANSFER AT THE SURFACE PART l-constant FLUD PROPERTES :J by Rbet M. Kendall Ms W.
3. Be able to derive the chemical equilibrium constants from statistical mechanics.
 Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
π e ax2 dx = x 2 e ax2 dx or x 3 e ax2 dx = 1 x 4 e ax2 dx = 3 π 8a 5/2 (a) We are considering the Maxwell velocity distribution function: 2πτ/m
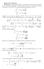 Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Chapter 1: Mole Balances
 CHEMICL RECTION ENGINEERING (SK3223) Chapte : Mole Balances WN NORHRYTI WN SLLEH hayati@petoleum.utm.my RIZIN MD. KSMNI afiziana@petoleum.utm.my Rate of Reaction The eaction ate is the ate at which a species
CHEMICL RECTION ENGINEERING (SK3223) Chapte : Mole Balances WN NORHRYTI WN SLLEH hayati@petoleum.utm.my RIZIN MD. KSMNI afiziana@petoleum.utm.my Rate of Reaction The eaction ate is the ate at which a species
Review of Vector Algebra and Vector Calculus Operations
 Revew of Vecto Algeba and Vecto Calculus Opeatons Tpes of vaables n Flud Mechancs Repesentaton of vectos Dffeent coodnate sstems Base vecto elatons Scala and vecto poducts Stess Newton s law of vscost
Revew of Vecto Algeba and Vecto Calculus Opeatons Tpes of vaables n Flud Mechancs Repesentaton of vectos Dffeent coodnate sstems Base vecto elatons Scala and vecto poducts Stess Newton s law of vscost
Thermodynamics II. Department of Chemical Engineering. Prof. Kim, Jong Hak
 Thermodynamcs II Department of Chemcal Engneerng Prof. Km, Jong Hak Soluton Thermodynamcs : theory Obectve : lay the theoretcal foundaton for applcatons of thermodynamcs to gas mxture and lqud soluton
Thermodynamcs II Department of Chemcal Engneerng Prof. Km, Jong Hak Soluton Thermodynamcs : theory Obectve : lay the theoretcal foundaton for applcatons of thermodynamcs to gas mxture and lqud soluton
Open Systems: Chemical Potential and Partial Molar Quantities Chemical Potential
 Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Lucas Imperfect Information Model
 Lucas Imerfect Infrmatn Mdel 93 Lucas Imerfect Infrmatn Mdel The Lucas mdel was the frst f the mdern, mcrfundatns mdels f aggregate suly and macrecnmcs It bult drectly n the Fredman-Phels analyss f the
Lucas Imerfect Infrmatn Mdel 93 Lucas Imerfect Infrmatn Mdel The Lucas mdel was the frst f the mdern, mcrfundatns mdels f aggregate suly and macrecnmcs It bult drectly n the Fredman-Phels analyss f the
Molecular Energy Changes During a Reaction
 Reation Kinetis Moleula Enegy Changes Duing a Reation Chemial Enegy of Speies E xn E* +BP E* P+B Moleules above this enegy level (defined somewhat abitaily) ae alled ativated omplexes Poduts Reatants Pogession
Reation Kinetis Moleula Enegy Changes Duing a Reation Chemial Enegy of Speies E xn E* +BP E* P+B Moleules above this enegy level (defined somewhat abitaily) ae alled ativated omplexes Poduts Reatants Pogession
Changing Topology and Communication Delays
 Prepared by F.L. Lews Updated: Saturday, February 3, 00 Changng Topology and Communcaton Delays Changng Topology The graph connectvty or topology may change over tme. Let G { G, G,, G M } wth M fnte be
Prepared by F.L. Lews Updated: Saturday, February 3, 00 Changng Topology and Communcaton Delays Changng Topology The graph connectvty or topology may change over tme. Let G { G, G,, G M } wth M fnte be
