6-5. H 2 O 200 kpa 200 C Q. Entropy Changes of Pure Substances
|
|
|
- Chad Carroll
- 5 years ago
- Views:
Transcription
1 Canges f ure Substances 6-0C Yes, because an ternally reversible, adiabatic prcess vlves n irreversibilities r eat transfer. 6- e radiatr f a steam eatg system is itially filled wit supereated steam. e valves are clsed, and steam is allwed t cl until te temperature drps t a specified value by transferrg eat t te rm. e entrpy cange f te steam durg tis prcess is t be determed. Analysis Frm te steam tables, 00ka v.080m /kg 00 C s kJ/kg K 80 C v v s x e mass f te steam is v v f v fg s f + xs fg kj/kg K V 0.00m m kg v.080m /kg ( 0.7)( 6.569) en te entrpy cange f te steam durg tis prcess becmes S m s ( s ) ( kg)( ) kj/kg K kj/k H O 00 ka 00 C Q 6-5
2 6-8 An sulated cylder is itially filled wit saturated liquid water at a specified pressure. e water is eated electrically at cnstant pressure. e entrpy cange f te water durg tis prcess is t be determed. Assumpts e ketic and ptential energy canges are negligible. e cylder is well-sulated and tus eat transfer is negligible. e termal energy stred te cylder itself is negligible. 4 e cmpress r expans prcess is quasi-equilibrium. Analysis Frm te steam tables, v v m /kg 50ka H O 467.kJ/kg sat. liquid 50 ka s s K 00 kj Sat. liquid Als, m V m kg v m / kg We take te cntents f te cylder as te system. is is a clsed system sce n mass enters r leaves. e energy balance fr tis statary clsed system can be expressed as E E E 4 4 system 4 4 Net energy transfer by eat, wrk, and mass Cange ternal, ketic, ptential, etc. energies We, Wb, U We, m( ) sce U + W b H durg a cnstant pressure quasi-equilibrium prcess. Slvg fr, We, 00 kj kj / kg m kg us, f ka x 0.08 fg kJ/kg s s + x s kJ/kg en te entrpy cange f te water becmes f fg ( )( ) K ( ) ( 4.748kg)(.68.46) kj/kg K 5.7 kj/k S m s s 6-9
3 6-6 Oxygen gas is cmpressed frm a specified itial state t a specified fal state. e entrpy cange f xygen durg tis prcess is t be determed fr te case f cnstant specific eats. Assumpts At specified cndits, xygen can be treated as an ideal gas. rperties e gas cnstant and mlar mass f xygen are R kj/kg.k and M kg/kml (able A-). Analysis e cnstant vlume specific eat f xygen at te average temperature is (able A-) ave 49 K Cv, ave kj / kg K us, V s s Cv, ave + R V O 560K 0.m /kg 0.8 m ( 0.690kJ/kg K) + ( 0.598kJ/kg K) /kg 98K 5 C 0.8m /kg 0.05kJ/kg K 6-6 An sulated tank cntas CO gas at a specified pressure and vlume. A paddle-weel te tank stirs te gas, and te pressure and temperature f CO rises. e entrpy cange f CO durg tis prcess is t be determed usg cnstant specific eats. Assumpts At specified cndits, CO can be treated as an ideal gas wit cnstant specific eats at rm temperature. rperties e specific eat f CO is C v kj/kg.k (able A-). Analysis Usg te ideal gas relat, te entrpy cange is determed t be us, V V S m( s ) s m C 0. kj/k v, ave 0ka. 00ka (.7 kg)( 0.657kJ/kg K) (.) V + R V mc v, ave CO.5 m 00 ka. kg 6-0
4 6-67 Air is cmpressed a pistn-cylder device a reversible and istermal manner. e entrpy cange f air and te wrk dne are t be determed. Assumpts At specified cndits, air can be treated as an ideal gas. e prcess is specified t be reversible. rperties e gas cnstant f air is R 0.87 kj/kg.k (able A-). Analysis (a) Ntg tat te temperature remas cnstant, te entrpy cange f air is determed frm S air C p, ave R R 400ka 90ka ( 0.87kJ/kg K) 0.48kJ/kg K Als, fr a reversible istermal prcess, ( 9 K)( 0.48kJ/kg K) 5.4kJ/kg q 5.4 kj/kg q s (b) e wrk dne durg tis prcess is determed frm te clsed system energy balance, E E 44 Net energy transfer by eat, wrk, and mass W Q w U mc q Esystem 44 Cange ternal, ketic, ptential, etc.energies v ( ) kj/kg AIR cnst Q 6-68 Air is cmpressed steadily by a 5-kW cmpressr frm ne specified state t anter specified state. e rate f entrpy cange f air is t be determed. Assumpts At specified cndits, air can be treated as an ideal gas. Air as variable specific eats. rperties e gas cnstant f air is R 0.87 kj/kg.k (able A-). Analysis Frm te air table (able A-7), 90 K s 00 ka 440 K s 600 ka.6680 kj/kg K.0887 kj/kg K en te rate f entrpy cange f air becmes & S sys m& s s R kw/k (.6/60kg/s) ( 0.87kJ/kg K) 600ka 00ka 600 ka 440 K AIR COMRESSOR 00 ka 90 K 5 kw 6-
5 Reversible Steady-Flw Wrk 6-78C e wrk assciated wit steady-flw devices is prprtal t te specific vlume f te gas. Clg a gas durg cmpress will reduce its specific vlume, and tus te pwer cnsumed by te cmpressr. 6-79C Clg te steam as it expands a turbe will reduce its specific vlume, and tus te wrk put f te turbe. erefre, tis is nt a gd prpsal. 6-80C We wuld nt supprt tis prpsal sce te steady-flw wrk put t te pump is prprtal t te specific vlume f te liquid, and clg will nt affect te specific vlume f a liquid significantly. 6-0
6 6-84 A steam pwer plant perates between te pressure limits f 0 Ma and 0 ka. e rati f te turbe wrk t te pump wrk is t be determed. Assumpts Liquid water is an cmpressible substance. Ketic and ptential energy canges are negligible. e prcess is reversible. 4 e pump and te turbe are adiabatic. rperties e specific vlume f saturated liquid water at 0 ka is v v 0 ka m /kg (able A-5). Analysis Bt te cmpress and expans prcesses are reversible and adiabatic, and tus isentrpic, s s and s s 4. en te prperties f te steam are 4 0 ka 4 sat. vapr s4 s 0 Ma s s kj/kg Als, v v 0 ka m /kg kj/kg kj/kg K e wrk put t tis isentrpic turbe is determed frm te steady-flw energy balance t be 0 (steady) E E Esystem 0 & 44 & & Rate f net energy transfer by eat, wrk, and mass Rate f cange ternal, ketic, ptential, etc.energies E& E& m & m & 4 + m& ( 4 ) Substitutg, wturb, kj / kg e pump wrk put is determed frm te steady-flw wrk relat t be H O H O 4 us, w w w pump, turb, pump, ( ) vd + ke + pe v kj (0.0007m /kg)(0,000 0)ka ka m kJ/kg 6-85 EES slut f tis (and ter cmpreensive prblems designated wit te cmputer icn) is available t structrs at te Instructr Manual sect f te Onle Learng Center (OLC) at See te reface fr access frmat. 6-
7 6-97 Steam enters an adiabatic turbe at a specified state, and leaves at a specified state. e mass flw rate f te steam and te isentrpic efficiency are t be determed. Assumpts is is a steady-flw prcess sce tere is n cange wit time. tential energy canges are negligible. e device is adiabatic and tus eat transfer is negligible. Analysis (a) Frm te steam tables (ables A-4 and A-6), 6Ma 658.4kJ/kg 600 C 7.677kJ/kg K s 50ka 00 C a 68.5kJ/kg ere is nly ne let and ne exit, and tus m& m& m&. We take te actual turbe as te system, wic is a cntrl vlume sce mass crsses te bundary. e energy balance fr tis steady-flw system can be expressed te rate frm as E E E & 44 & & (steady) system Rate f net energy transfer by eat, wrk, and mass m& ( + V E& E& / ) + m& ( a, m& Rate f cange ternal, ketic, ptential, etc.energies a, + V V + V /) (sce Q& pe 0) Substitutg, te mass flw rate f te steam is determed t be (40 m/s) (80 m/s) kj/kg 8000 kj/s m& m /s m& 8.5 kg/s (b) e isentrpic exit entalpy f te steam and te pwer put f te isentrpic turbe are and s s, s s 50ka s m& s f x s + x s s ( + {( V V )/ } ) s fg s s f s fg (40 m/s) (80 m/s) ( 8.5 kg/s) s, kw en te isentrpic efficiency f te turbe becmes η a 8000 kw % 9554 kw s ( 0.94)( 05.4) 49.7 kj/kg kj/kg 000 m /s H O 8 MW 6-40
8 6-00 [Als slved by EES n enclsed CD] Refrigerant-4a enters an adiabatic cmpressr wit an isentrpic efficiency f 0.80 at a specified state wit a specified vlume flw rate, and leaves at a specified pressure. e cmpressr exit temperature and pwer put t te cmpressr are t be determed. Assumpts is is a steady-flw prcess sce tere is n cange wit time. Ketic and ptential energy canges are negligible. e device is adiabatic and tus eat transfer is negligible. Analysis (a) Frm te refrigerant tables, 0ka s s g sat. vapr v v Ma ss s s g@0ka 77.84kJ/kg Frm te isentrpic efficiency relat, us, η C a a s a Ma 88.84kJ/kg a a C.86kJ/kg 0.954kJ/kg K 0.64m /kg ( )/η.86 + ( ) s C / kJ/kg (b) e mass flw rate f te refrigerant is determed frm & m& V 0./60 m / s 0.0 kg / s v 0.64 m / kg ere is nly ne let and ne exit, and tus m& m& m&. We take te actual cmpressr as te system, wic is a cntrl vlume sce mass crsses te bundary. e energy balance fr tis steady-flw system can be expressed as E E & 44 & Rate f net energy transfer by eat, wrk, and mass E & (steady) system a, E& Rate f cange ternal, ketic, ptential, etc.energies E& a, m& ( ) Substitutg, te pwer put t te cmpressr becmes, W & a, 0.0kg/s kj/kg.70 + m & m & (sce Q& ke pe 0) ( )( ) kw R-4a η C 80% 0. m /m 6-0 EES slut f tis (and ter cmpreensive prblems designated wit te cmputer icn) is available t structrs at te Instructr Manual sect f te Onle Learng Center (OLC) at See te reface fr access frmat. 6-4
Department of Civil Engineering & Applied Mechanics McGill University, Montreal, Quebec Canada
 Department f Ciil ngeerg Applied Mechanics McGill Uniersity, Mntreal, Quebec Canada CI 90 THRMODYNAMICS HAT TRANSFR Assignment #4 SOLUTIONS. A 68-kg man whse aerage bdy temperature is 9 C drks L f cld
Department f Ciil ngeerg Applied Mechanics McGill Uniersity, Mntreal, Quebec Canada CI 90 THRMODYNAMICS HAT TRANSFR Assignment #4 SOLUTIONS. A 68-kg man whse aerage bdy temperature is 9 C drks L f cld
2-18. (a) For mercury, (b) For water,
 -8-5 CD EES Bt a gage and a manmeter are attaced t a gas t measure its pressure. Fr a specified reading f gage pressure, te difference between te fluid levels f te tw arms f te manmeter is t be determined
-8-5 CD EES Bt a gage and a manmeter are attaced t a gas t measure its pressure. Fr a specified reading f gage pressure, te difference between te fluid levels f te tw arms f te manmeter is t be determined
8-4 P 2. = 12 kw. AIR T = const. Therefore, Q &
 8-4 8-4 Air i compreed teadily by a compreor. e air temperature i mataed contant by eat rejection to te urroundg. e rate o entropy cange o air i to be determed. Aumption i i a teady-low proce ce tere i
8-4 8-4 Air i compreed teadily by a compreor. e air temperature i mataed contant by eat rejection to te urroundg. e rate o entropy cange o air i to be determed. Aumption i i a teady-low proce ce tere i
Thermodynamics EAS 204 Spring 2004 Class Month Day Chapter Topic Reading Due 1 January 12 M Introduction 2 14 W Chapter 1 Concepts Chapter 1 19 M MLK
 Thermdynamics EAS 204 Spring 2004 Class Mnth Day Chapter Tpic Reading Due 1 January 12 M Intrductin 2 14 W Chapter 1 Cncepts Chapter 1 19 M MLK Hliday n class 3 21 W Chapter 2 Prperties Chapter 2 PS1 4
Thermdynamics EAS 204 Spring 2004 Class Mnth Day Chapter Tpic Reading Due 1 January 12 M Intrductin 2 14 W Chapter 1 Cncepts Chapter 1 19 M MLK Hliday n class 3 21 W Chapter 2 Prperties Chapter 2 PS1 4
Instructions: Show all work for complete credit. Work in symbols first, plugging in numbers and performing calculations last. / 26.
 CM ROSE-HULMAN INSTITUTE OF TECHNOLOGY Name Circle sectin: 01 [4 th Lui] 02 [5 th Lui] 03 [4 th Thm] 04 [5 th Thm] 05 [4 th Mech] ME301 Applicatins f Thermdynamics Exam 1 Sep 29, 2017 Rules: Clsed bk/ntes
CM ROSE-HULMAN INSTITUTE OF TECHNOLOGY Name Circle sectin: 01 [4 th Lui] 02 [5 th Lui] 03 [4 th Thm] 04 [5 th Thm] 05 [4 th Mech] ME301 Applicatins f Thermdynamics Exam 1 Sep 29, 2017 Rules: Clsed bk/ntes
300 kpa 77 C. (d) If we neglect kinetic energy in the calculation of energy transport by mass
 6-6- Air flows steadily a ie at a secified state. The diameter of the ie, the rate of flow energy, and the rate of energy transort by mass are to be determed. Also, the error oled the determation of energy
6-6- Air flows steadily a ie at a secified state. The diameter of the ie, the rate of flow energy, and the rate of energy transort by mass are to be determed. Also, the error oled the determation of energy
Delft University of Technology DEPARTMENT OF AEROSPACE ENGINEERING
 Delft University of Technology DEPRTMENT OF EROSPCE ENGINEERING Course: Physics I (E-04) Course year: Date: 7-0-0 Time: 4:00-7:00 Student name and itials (capital letters): Student number:. You have attended
Delft University of Technology DEPRTMENT OF EROSPCE ENGINEERING Course: Physics I (E-04) Course year: Date: 7-0-0 Time: 4:00-7:00 Student name and itials (capital letters): Student number:. You have attended
th th th The air-fuel ratio is determined by taking the ratio of the mass of the air to the mass of the fuel,
 Cheical Reactins 14-14 rpane is burned wi 75 percent excess during a cbustin prcess. The AF rati is t be deterined. Assuptins 1 Cbustin is cplete. The cbustin prducts cntain CO, H O, O, and N nly. rperties
Cheical Reactins 14-14 rpane is burned wi 75 percent excess during a cbustin prcess. The AF rati is t be deterined. Assuptins 1 Cbustin is cplete. The cbustin prducts cntain CO, H O, O, and N nly. rperties
Chapter 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES
 5- Chapter 5 MASS AND ENERGY ANALYSIS OF CONTROL OLUMES Conseration of Mass 5-C Mass, energy, momentum, and electric charge are consered, and olume and entropy are not consered durg a process. 5-C Mass
5- Chapter 5 MASS AND ENERGY ANALYSIS OF CONTROL OLUMES Conseration of Mass 5-C Mass, energy, momentum, and electric charge are consered, and olume and entropy are not consered durg a process. 5-C Mass
ME 200 Thermodynamics 1 Fall 2017 Exam 3
 ME 200 hermodynamics 1 Fall 2017 Exam Circle your structor s last name Division 1: Naik Division : Wassgren Division 6: Braun Division 2: Sojka Division 4: Goldenste Division 7: Buckius Division 8: Meyer
ME 200 hermodynamics 1 Fall 2017 Exam Circle your structor s last name Division 1: Naik Division : Wassgren Division 6: Braun Division 2: Sojka Division 4: Goldenste Division 7: Buckius Division 8: Meyer
& out. R-134a 34 C
 5-9 5-76 Saturated refrigerant-4a vapor at a saturation temperature of T sat 4 C condenses side a tube. Te rate of eat transfer from te refrigerant for te condensate exit temperatures of 4 C and 0 C are
5-9 5-76 Saturated refrigerant-4a vapor at a saturation temperature of T sat 4 C condenses side a tube. Te rate of eat transfer from te refrigerant for te condensate exit temperatures of 4 C and 0 C are
Fill in your name and ID No. in the space above. There should be 11 pages (including this page and the last page which is a formula page).
 ENGR -503 Name: Final Exam, Sem. 03C ID N.: /6/003 3:30 5:30 p.m. Rm N.: 7B Fill in yur name and ID N. in the space abve. There shuld be pages (including this page and the last page which is a frmula page).
ENGR -503 Name: Final Exam, Sem. 03C ID N.: /6/003 3:30 5:30 p.m. Rm N.: 7B Fill in yur name and ID N. in the space abve. There shuld be pages (including this page and the last page which is a frmula page).
1 = The rate at which the entropy of the high temperature reservoir changes, according to the definition of the entropy, is
 8-7 8-9 A reverible eat um wit eciied reervoir temerature i conidered. e entroy cange o two reervoir i to be calculated and it i to be determed i ti eat um atiie te creae entroy rcile. Aumtion e eat um
8-7 8-9 A reverible eat um wit eciied reervoir temerature i conidered. e entroy cange o two reervoir i to be calculated and it i to be determed i ti eat um atiie te creae entroy rcile. Aumtion e eat um
I. (20%) Answer the following True (T) or False (F). If false, explain why for full credit.
 I. (20%) Answer the following True (T) or False (F). If false, explain why for full credit. Both the Kelvin and Fahrenheit scales are absolute temperature scales. Specific volume, v, is an intensive property,
I. (20%) Answer the following True (T) or False (F). If false, explain why for full credit. Both the Kelvin and Fahrenheit scales are absolute temperature scales. Specific volume, v, is an intensive property,
In the next lecture...
 16 1 In the next lecture... Solve problems from Entropy Carnot cycle Exergy Second law efficiency 2 Problem 1 A heat engine receives reversibly 420 kj/cycle of heat from a source at 327 o C and rejects
16 1 In the next lecture... Solve problems from Entropy Carnot cycle Exergy Second law efficiency 2 Problem 1 A heat engine receives reversibly 420 kj/cycle of heat from a source at 327 o C and rejects
4F-5 : Performance of an Ideal Gas Cycle 10 pts
 4F-5 : Perfrmance f an Cycle 0 pts An ideal gas, initially at 0 C and 00 kpa, underges an internally reversible, cyclic prcess in a clsed system. The gas is first cmpressed adiabatically t 500 kpa, then
4F-5 : Perfrmance f an Cycle 0 pts An ideal gas, initially at 0 C and 00 kpa, underges an internally reversible, cyclic prcess in a clsed system. The gas is first cmpressed adiabatically t 500 kpa, then
ENERGY TRANSFER BY WORK: Electrical Work: When N Coulombs of electrical charge move through a potential difference V
 Weight, W = mg Where m=mass, g=gravitational acceleration ENERGY TRANSFER BY WOR: Sign convention: Work done on a system = (+) Work done by a system = (-) Density, ρ = m V kg m 3 Where m=mass, V =Volume
Weight, W = mg Where m=mass, g=gravitational acceleration ENERGY TRANSFER BY WOR: Sign convention: Work done on a system = (+) Work done by a system = (-) Density, ρ = m V kg m 3 Where m=mass, V =Volume
Number of extra papers used if any
 Last Name: First Name: Thermo no. ME 200 Thermodynamics 1 Fall 2018 Exam Circle your structor s last name Division 1 (7:0): Naik Division (1:0): Wassgren Division 6 (11:0): Sojka Division 2 (9:0): Choi
Last Name: First Name: Thermo no. ME 200 Thermodynamics 1 Fall 2018 Exam Circle your structor s last name Division 1 (7:0): Naik Division (1:0): Wassgren Division 6 (11:0): Sojka Division 2 (9:0): Choi
Find: a) Mass of the air, in kg, b) final temperature of the air, in K, and c) amount of entropy produced, in kj/k.
 PROBLEM 6.25 Three m 3 of air in a rigid, insulated container fitted with a paddle wheel is initially at 295 K, 200 kpa. The air receives 1546 kj of work from the paddle wheel. Assuming the ideal gas model,
PROBLEM 6.25 Three m 3 of air in a rigid, insulated container fitted with a paddle wheel is initially at 295 K, 200 kpa. The air receives 1546 kj of work from the paddle wheel. Assuming the ideal gas model,
( )( ) 7 MPa q in = = 10 kpa q out. 1 h. = s. Thus, and = 38.9% (b) (c) The rate of heat rejection to the cooling water and its temperature rise are
 . A team poer plant operate on a imple ideal Ranke cycle beteen te peciied preure limit. e termal eiciency o te cycle, te ma lo rate o te team, and te temperature rie o te coolg ater are to be determed.
. A team poer plant operate on a imple ideal Ranke cycle beteen te peciied preure limit. e termal eiciency o te cycle, te ma lo rate o te team, and te temperature rie o te coolg ater are to be determed.
Dishwasher. Heater. Homework Solutions ME Thermodynamics I Spring HW-1 (25 points)
 HW-1 (25 points) (a) Given: 1 for writing given, find, EFD, etc., Schematic of a household piping system Find: Identify system and location on the system boundary where the system interacts with the environment
HW-1 (25 points) (a) Given: 1 for writing given, find, EFD, etc., Schematic of a household piping system Find: Identify system and location on the system boundary where the system interacts with the environment
The First Law of Thermodynamics. By: Yidnekachew Messele
 The First Law of Thermodynamics By: Yidnekachew Messele It is the law that relates the various forms of energies for system of different types. It is simply the expression of the conservation of energy
The First Law of Thermodynamics By: Yidnekachew Messele It is the law that relates the various forms of energies for system of different types. It is simply the expression of the conservation of energy
ME Thermodynamics I
 Homework - Week 01 HW-01 (25 points) Given: 5 Schematic of the solar cell/solar panel Find: 5 Identify the system and the heat/work interactions associated with it. Show the direction of the interactions.
Homework - Week 01 HW-01 (25 points) Given: 5 Schematic of the solar cell/solar panel Find: 5 Identify the system and the heat/work interactions associated with it. Show the direction of the interactions.
Answer Key THERMODYNAMICS TEST (a) 33. (d) 17. (c) 1. (a) 25. (a) 2. (b) 10. (d) 34. (b) 26. (c) 18. (d) 11. (c) 3. (d) 35. (c) 4. (d) 19.
 HERMODYNAMICS ES Answer Key. (a) 9. (a) 7. (c) 5. (a). (d). (b) 0. (d) 8. (d) 6. (c) 4. (b). (d). (c) 9. (b) 7. (c) 5. (c) 4. (d). (a) 0. (b) 8. (b) 6. (b) 5. (b). (d). (a) 9. (a) 7. (b) 6. (a) 4. (d).
HERMODYNAMICS ES Answer Key. (a) 9. (a) 7. (c) 5. (a). (d). (b) 0. (d) 8. (d) 6. (c) 4. (b). (d). (c) 9. (b) 7. (c) 5. (c) 4. (d). (a) 0. (b) 8. (b) 6. (b) 5. (b). (d). (a) 9. (a) 7. (b) 6. (a) 4. (d).
Nonisothermal Chemical Reactors
 he 471 Fall 2014 LEUE 7a Nnithermal hemical eactr S far we have dealt with ithermal chemical reactr and were able, by ug nly a many pecie ma balance a there are dependent react t relate reactr ize, let
he 471 Fall 2014 LEUE 7a Nnithermal hemical eactr S far we have dealt with ithermal chemical reactr and were able, by ug nly a many pecie ma balance a there are dependent react t relate reactr ize, let
ME 200 Thermodynamics 1 Spring Exam 2
 Last Name: First Name: Thermo no. ME 200 Thermodynamics 1 Sprg 2017 - Exam 2 Circle your structor s last name Ardekani Fisher Hess Naik Sojka (onle and on campus) INSTRUCTIONS This is a closed book and
Last Name: First Name: Thermo no. ME 200 Thermodynamics 1 Sprg 2017 - Exam 2 Circle your structor s last name Ardekani Fisher Hess Naik Sojka (onle and on campus) INSTRUCTIONS This is a closed book and
Exergy and the Dead State
 EXERGY The energy content of the universe is constant, just as its mass content is. Yet at times of crisis we are bombarded with speeches and articles on how to conserve energy. As engineers, we know that
EXERGY The energy content of the universe is constant, just as its mass content is. Yet at times of crisis we are bombarded with speeches and articles on how to conserve energy. As engineers, we know that
ME 200 Final Exam December 14, :00 a.m. to 10:00 a.m.
 CIRCLE YOUR LECTURE BELOW: First Name Last Name 7:30 a.m. 8:30 a.m. 10:30 a.m. 11:30 a.m. Boregowda Boregowda Braun Bae 2:30 p.m. 3:30 p.m. 4:30 p.m. Meyer Naik Hess ME 200 Final Exam December 14, 2015
CIRCLE YOUR LECTURE BELOW: First Name Last Name 7:30 a.m. 8:30 a.m. 10:30 a.m. 11:30 a.m. Boregowda Boregowda Braun Bae 2:30 p.m. 3:30 p.m. 4:30 p.m. Meyer Naik Hess ME 200 Final Exam December 14, 2015
Chapter Outline 4/28/2014. P-V Work. P-V Work. Isolated, Closed and Open Systems. Exothermic and Endothermic Processes. E = q + w
 Islated, Clsed and Open Systems 9.1 Energy as a Reactant r a Prduct 9.2 Transferring Heat and Ding Wrk 9.5 Heats f Reactin and Calrimetry 9.6 Hess s Law and Standard Heats f Reactin 9.7 Heats f Reactin
Islated, Clsed and Open Systems 9.1 Energy as a Reactant r a Prduct 9.2 Transferring Heat and Ding Wrk 9.5 Heats f Reactin and Calrimetry 9.6 Hess s Law and Standard Heats f Reactin 9.7 Heats f Reactin
5/6/ :41 PM. Chapter 6. Using Entropy. Dr. Mohammad Abuhaiba, PE
 Chapter 6 Using Entropy 1 2 Chapter Objective Means are introduced for analyzing systems from the 2 nd law perspective as they undergo processes that are not necessarily cycles. Objective: introduce entropy
Chapter 6 Using Entropy 1 2 Chapter Objective Means are introduced for analyzing systems from the 2 nd law perspective as they undergo processes that are not necessarily cycles. Objective: introduce entropy
Circle your instructor s last name
 ME 00 Thermodynamics Fall 07 Exam Circle your structor s last name Division : Naik Division : Wassgren Division 6: Braun Division : Sojka Division 4: Goldenste Division 7: Buckius Division 8: Meyer INSTRUCTIONS
ME 00 Thermodynamics Fall 07 Exam Circle your structor s last name Division : Naik Division : Wassgren Division 6: Braun Division : Sojka Division 4: Goldenste Division 7: Buckius Division 8: Meyer INSTRUCTIONS
Solutions to Homework #10
 Solution to Homeork #0 0-6 A teady-lo Carnot enge it ater a te orkg luid operate at peciied condition. e termal eiciency, te preure at te turbe let, and te ork put are to be determed. Aumption Steady operatg
Solution to Homeork #0 0-6 A teady-lo Carnot enge it ater a te orkg luid operate at peciied condition. e termal eiciency, te preure at te turbe let, and te ork put are to be determed. Aumption Steady operatg
Chapter 7. Entropy. by Asst.Prof. Dr.Woranee Paengjuntuek and Asst. Prof. Dr.Worarattana Pattaraprakorn
 Chapter 7 Entropy by Asst.Prof. Dr.Woranee Paengjuntuek and Asst. Prof. Dr.Worarattana Pattaraprakorn Reference: Cengel, Yunus A. and Michael A. Boles, Thermodynamics: An Engineering Approach, 5th ed.,
Chapter 7 Entropy by Asst.Prof. Dr.Woranee Paengjuntuek and Asst. Prof. Dr.Worarattana Pattaraprakorn Reference: Cengel, Yunus A. and Michael A. Boles, Thermodynamics: An Engineering Approach, 5th ed.,
CHAPTER 7 ENTROPY. Copyright Hany A. Al-Ansary and S. I. Abdel-Khalik (2014) 1
 CHAPTER 7 ENTROPY S. I. Abdel-Khalik (2014) 1 ENTROPY The Clausius Inequality The Clausius inequality states that for for all cycles, reversible or irreversible, engines or refrigerators: For internally-reversible
CHAPTER 7 ENTROPY S. I. Abdel-Khalik (2014) 1 ENTROPY The Clausius Inequality The Clausius inequality states that for for all cycles, reversible or irreversible, engines or refrigerators: For internally-reversible
(b) The heat transfer can be determined from an energy balance on the system
 8-5 Heat is transferred to a iston-cylinder device wit a set of stos. e work done, te eat transfer, te exergy destroyed, and te second-law efficiency are to be deterined. Assutions e device is stationary
8-5 Heat is transferred to a iston-cylinder device wit a set of stos. e work done, te eat transfer, te exergy destroyed, and te second-law efficiency are to be deterined. Assutions e device is stationary
Lecture 38: Vapor-compression refrigeration systems
 ME 200 Termodynamics I Lecture 38: Vapor-compression refrigeration systems Yong Li Sangai Jiao Tong University Institute of Refrigeration and Cryogenics 800 Dong Cuan Road Sangai, 200240, P. R. Cina Email
ME 200 Termodynamics I Lecture 38: Vapor-compression refrigeration systems Yong Li Sangai Jiao Tong University Institute of Refrigeration and Cryogenics 800 Dong Cuan Road Sangai, 200240, P. R. Cina Email
Thermodynamics Lecture Series
 Termodynamics Lecture Series Ideal Ranke Cycle Te Practical Cycle Applied Sciences Education Researc Group (ASERG) Faculty of Applied Sciences Universiti Teknologi MARA email: drjjlanita@otmail.com ttp://www5.uitm.edu.my/faculties/fsg/drjj1.tml
Termodynamics Lecture Series Ideal Ranke Cycle Te Practical Cycle Applied Sciences Education Researc Group (ASERG) Faculty of Applied Sciences Universiti Teknologi MARA email: drjjlanita@otmail.com ttp://www5.uitm.edu.my/faculties/fsg/drjj1.tml
Chapter 5. Mass and Energy Analysis of Control Volumes. by Asst. Prof. Dr.Woranee Paengjuntuek and Asst. Prof. Dr.Worarattana Pattaraprakorn
 Chapter 5 Mass and Energy Analysis of Control Volumes by Asst. Prof. Dr.Woranee Paengjuntuek and Asst. Prof. Dr.Worarattana Pattaraprakorn Reference: Cengel, Yunus A. and Michael A. Boles, Thermodynamics:
Chapter 5 Mass and Energy Analysis of Control Volumes by Asst. Prof. Dr.Woranee Paengjuntuek and Asst. Prof. Dr.Worarattana Pattaraprakorn Reference: Cengel, Yunus A. and Michael A. Boles, Thermodynamics:
Special Topic: Binary Vapor Cycles
 0- Special opic: Bary Vapor ycle 0- Bary poer cycle i a cycle ic i actually a combation o to cycle; one te ig temperature region, and te oter te lo temperature region. It purpoe i to creae termal eiciency.
0- Special opic: Bary Vapor ycle 0- Bary poer cycle i a cycle ic i actually a combation o to cycle; one te ig temperature region, and te oter te lo temperature region. It purpoe i to creae termal eiciency.
Section A 01. (12 M) (s 2 s 3 ) = 313 s 2 = s 1, h 3 = h 4 (s 1 s 3 ) = kj/kgk. = kj/kgk. 313 (s 3 s 4f ) = ln
 0. (a) Sol: Section A A refrigerator macine uses R- as te working fluid. Te temperature of R- in te evaporator coil is 5C, and te gas leaves te compressor as dry saturated at a temperature of 40C. Te mean
0. (a) Sol: Section A A refrigerator macine uses R- as te working fluid. Te temperature of R- in te evaporator coil is 5C, and te gas leaves te compressor as dry saturated at a temperature of 40C. Te mean
= T. (kj/k) (kj/k) 0 (kj/k) int rev. Chapter 6 SUMMARY
 Capter 6 SUMMARY e second la of termodynamics leads to te definition of a ne property called entropy ic is a quantitative measure of microscopic disorder for a system. e definition of entropy is based
Capter 6 SUMMARY e second la of termodynamics leads to te definition of a ne property called entropy ic is a quantitative measure of microscopic disorder for a system. e definition of entropy is based
A) 0.77 N B) 0.24 N C) 0.63 N D) 0.31 N E) 0.86 N. v = ω k = 80 = 32 m/s. Ans: (32) 2 = 0.77 N
 Q1. A transverse sinusidal wave travelling n a string is given by: y (x,t) = 0.20 sin (2.5 x 80 t) (SI units). The length f the string is 2.0 m and its mass is 1.5 g. What is the magnitude f the tensin
Q1. A transverse sinusidal wave travelling n a string is given by: y (x,t) = 0.20 sin (2.5 x 80 t) (SI units). The length f the string is 2.0 m and its mass is 1.5 g. What is the magnitude f the tensin
Name: I have observed the honor code and have neither given nor received aid on this exam.
 ME 235 FINAL EXAM, ecember 16, 2011 K. Kurabayashi and. Siegel, ME ept. Exam Rules: Open Book and one page of notes allowed. There are 4 problems. Solve each problem on a separate page. Name: I have observed
ME 235 FINAL EXAM, ecember 16, 2011 K. Kurabayashi and. Siegel, ME ept. Exam Rules: Open Book and one page of notes allowed. There are 4 problems. Solve each problem on a separate page. Name: I have observed
v v = = Mixing chamber: = 30 or, = s6 Then, and = 52.4% Turbine Boiler process heater Condenser 7 MPa Q in 0.6 MPa Q proces 10 kpa Q out
 0-0- A cogeneration plant i to generate poer and proce eat. art o te team extracted rom te turbe at a relatively ig preure i ued or proce eatg. e poer produced and te utilization actor o te plant are to
0-0- A cogeneration plant i to generate poer and proce eat. art o te team extracted rom te turbe at a relatively ig preure i ued or proce eatg. e poer produced and te utilization actor o te plant are to
Thermodynamics Lecture Series
 Thermodynamics Lecture Series Second Law uality of Energy Applied Sciences Education Research Group (ASERG) Faculty of Applied Sciences Universiti Teknologi MARA email: drjjlanita@hotmail.com http://www.uitm.edu.my/faculties/fsg/drjj.html
Thermodynamics Lecture Series Second Law uality of Energy Applied Sciences Education Research Group (ASERG) Faculty of Applied Sciences Universiti Teknologi MARA email: drjjlanita@hotmail.com http://www.uitm.edu.my/faculties/fsg/drjj.html
4-64. Review Problems
 4-64 Reiew Prblems 4-0 Heat is transferred t a pistn-cylinder deice cntaining air. The expansin wrk is t be determined. Assumptins There is n frictin between pistn and cylinder. Air is an ideal gas. Prperties
4-64 Reiew Prblems 4-0 Heat is transferred t a pistn-cylinder deice cntaining air. The expansin wrk is t be determined. Assumptins There is n frictin between pistn and cylinder. Air is an ideal gas. Prperties
REVIEW QUESTIONS Chapter 18. H = H (Products) - H (Reactants) H (Products) = (1 x -125) + (3 x -271) = -938 kj
 Chemistry 102 ANSWER KEY REVIEW QUESTIONS Chapter 18 1. Calculate the heat reactin ( H ) in kj/ml r the reactin shwn belw, given the H values r each substance: NH (g) + F 2 (g) NF (g) + HF (g) H (kj/ml)
Chemistry 102 ANSWER KEY REVIEW QUESTIONS Chapter 18 1. Calculate the heat reactin ( H ) in kj/ml r the reactin shwn belw, given the H values r each substance: NH (g) + F 2 (g) NF (g) + HF (g) H (kj/ml)
Kelvin Planck Statement of the Second Law. Clausius Statement of the Second Law
 Kelv Planck Statement of te Second aw It is imossible to construct an enge wic, oeratg a cycle, will roduce no oter effect tan te extraction of eat from a sgle reservoir and te erformance of an equivalent
Kelv Planck Statement of te Second aw It is imossible to construct an enge wic, oeratg a cycle, will roduce no oter effect tan te extraction of eat from a sgle reservoir and te erformance of an equivalent
" 1 = # $H vap. Chapter 3 Problems
 Chapter 3 rblems rblem At 1 atmsphere pure Ge melts at 1232 K and bils at 298 K. he triple pint ccurs at =8.4x1-8 atm. Estimate the heat f vaprizatin f Ge. he heat f vaprizatin is estimated frm the Clausius
Chapter 3 rblems rblem At 1 atmsphere pure Ge melts at 1232 K and bils at 298 K. he triple pint ccurs at =8.4x1-8 atm. Estimate the heat f vaprizatin f Ge. he heat f vaprizatin is estimated frm the Clausius
ECE309 THERMODYNAMICS & HEAT TRANSFER MIDTERM EXAMINATION. Instructor: R. Culham. Name: Student ID Number:
 ECE309 THERMODYNAMICS & HEAT TRANSFER MIDTERM EXAMINATION June 19, 2015 2:30 pm - 4:30 pm Instructor: R. Culham Name: Student ID Number: Instructions 1. This is a 2 hour, closed-book examination. 2. Permitted
ECE309 THERMODYNAMICS & HEAT TRANSFER MIDTERM EXAMINATION June 19, 2015 2:30 pm - 4:30 pm Instructor: R. Culham Name: Student ID Number: Instructions 1. This is a 2 hour, closed-book examination. 2. Permitted
first law of ThermodyNamics
 first law of ThermodyNamics First law of thermodynamics - Principle of conservation of energy - Energy can be neither created nor destroyed Basic statement When any closed system is taken through a cycle,
first law of ThermodyNamics First law of thermodynamics - Principle of conservation of energy - Energy can be neither created nor destroyed Basic statement When any closed system is taken through a cycle,
Chapter 5. Mass and Energy Analysis of Control Volumes
 Chapter 5 Mass and Energy Analysis of Control Volumes Conservation Principles for Control volumes The conservation of mass and the conservation of energy principles for open systems (or control volumes)
Chapter 5 Mass and Energy Analysis of Control Volumes Conservation Principles for Control volumes The conservation of mass and the conservation of energy principles for open systems (or control volumes)
Content. Entropy and principle of increasing entropy. Change of entropy in an ideal gas.
 Entropy Content Entropy and principle of increasing entropy. Change of entropy in an ideal gas. Entropy Entropy can be viewed as a measure of molecular disorder, or molecular randomness. As a system becomes
Entropy Content Entropy and principle of increasing entropy. Change of entropy in an ideal gas. Entropy Entropy can be viewed as a measure of molecular disorder, or molecular randomness. As a system becomes
P=1 atm. vapor. liquid
 P1 atm ar liqid Q Gien and P Ser Heat Regin i, P < P sat > sat Cmressed Liqid Regin i, P > P sat < sat SEAM PRESUE AND EMPERAURE ABLES Satrated Liqid Line Satrated Var Line s s g g g g ree ables emeratre
P1 atm ar liqid Q Gien and P Ser Heat Regin i, P < P sat > sat Cmressed Liqid Regin i, P > P sat < sat SEAM PRESUE AND EMPERAURE ABLES Satrated Liqid Line Satrated Var Line s s g g g g ree ables emeratre
Entropy and the Second Law of Thermodynamics
 Entropy and the Second Law of Thermodynamics Reading Problems 7-1 7-3 7-88, 7-131, 7-135 7-6 7-10 8-24, 8-44, 8-46, 8-60, 8-73, 8-99, 8-128, 8-132, 8-1 8-10, 8-13 8-135, 8-148, 8-152, 8-166, 8-168, 8-189
Entropy and the Second Law of Thermodynamics Reading Problems 7-1 7-3 7-88, 7-131, 7-135 7-6 7-10 8-24, 8-44, 8-46, 8-60, 8-73, 8-99, 8-128, 8-132, 8-1 8-10, 8-13 8-135, 8-148, 8-152, 8-166, 8-168, 8-189
T Turbine 8. Boiler fwh fwh I Condenser 4 3 P II P I P III. (a) From the steam tables (Tables A-4, A-5, and A-6), = = 10 MPa. = 0.
 - - A team poer plant operate on an ideal regenerative anke cycle it to open feedater eater. e poer put of te poer plant and te termal efficiency of te cycle are to be determed. Aumption Steady operatg
- - A team poer plant operate on an ideal regenerative anke cycle it to open feedater eater. e poer put of te poer plant and te termal efficiency of te cycle are to be determed. Aumption Steady operatg
c Dr. Md. Zahurul Haq (BUET) Thermodynamic Processes & Efficiency ME 6101 (2017) 2 / 25 T145 = Q + W cv + i h 2 = h (V2 1 V 2 2)
 Thermodynamic Processes & Isentropic Efficiency Dr. Md. Zahurul Haq Professor Department of Mechanical Engineering Bangladesh University of Engineering & Technology (BUET Dhaka-1000, Bangladesh zahurul@me.buet.ac.bd
Thermodynamic Processes & Isentropic Efficiency Dr. Md. Zahurul Haq Professor Department of Mechanical Engineering Bangladesh University of Engineering & Technology (BUET Dhaka-1000, Bangladesh zahurul@me.buet.ac.bd
Chapter 7. Entropy: A Measure of Disorder
 Chapter 7 Entropy: A Measure of Disorder Entropy and the Clausius Inequality The second law of thermodynamics leads to the definition of a new property called entropy, a quantitative measure of microscopic
Chapter 7 Entropy: A Measure of Disorder Entropy and the Clausius Inequality The second law of thermodynamics leads to the definition of a new property called entropy, a quantitative measure of microscopic
CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES
 Thermodynamics: An Engineering Approach 8th Edition in SI Units Yunus A. Çengel, Michael A. Boles McGraw-Hill, 2015 CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES Lecture slides by Dr. Fawzi Elfghi
Thermodynamics: An Engineering Approach 8th Edition in SI Units Yunus A. Çengel, Michael A. Boles McGraw-Hill, 2015 CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES Lecture slides by Dr. Fawzi Elfghi
MAE 320 THERODYNAMICS FINAL EXAM - Practice. Name: You are allowed three sheets of notes.
 50 MAE 320 THERODYNAMICS FINAL EXAM - Practice Name: You are allowed three sheets of notes. 1. Fill in the blanks for each of the two (Carnot) cycles below. (a) 5 a) Heat engine or Heat pump/refrigerator
50 MAE 320 THERODYNAMICS FINAL EXAM - Practice Name: You are allowed three sheets of notes. 1. Fill in the blanks for each of the two (Carnot) cycles below. (a) 5 a) Heat engine or Heat pump/refrigerator
Today lecture. 1. Entropy change in an isolated system 2. Exergy
 Today lecture 1. Entropy change in an isolated system. Exergy - What is exergy? - Reversible Work & Irreversibility - Second-Law Efficiency - Exergy change of a system For a fixed mass For a flow stream
Today lecture 1. Entropy change in an isolated system. Exergy - What is exergy? - Reversible Work & Irreversibility - Second-Law Efficiency - Exergy change of a system For a fixed mass For a flow stream
4-93 RT RT. He PV n = C = = Then the boundary work for this polytropic process can be determined from. n =
 - A cylder i itially filled it eliu ga at a ecified tate. Heliu i creed lytrically t a ecified teerature and reure. e eat tranfer durg te rce i t be detered. Autin Heliu i an ideal ga it cntant ecific
- A cylder i itially filled it eliu ga at a ecified tate. Heliu i creed lytrically t a ecified teerature and reure. e eat tranfer durg te rce i t be detered. Autin Heliu i an ideal ga it cntant ecific
(SP 1) DLLL. Given: In a closed rigid tank,
 (SP 1) Given: In a closed rigid tank, State 1: m 1,ice = 1, m 1,g = 0.05 P1= 0.0381 kpa, T1= -30 o C State 2: the liquid vapor equilibrium line, either saturated liquid or saturated vapor Find: (a) The
(SP 1) Given: In a closed rigid tank, State 1: m 1,ice = 1, m 1,g = 0.05 P1= 0.0381 kpa, T1= -30 o C State 2: the liquid vapor equilibrium line, either saturated liquid or saturated vapor Find: (a) The
Compressibility Effects
 Definitin f Cmpressibility All real substances are cmpressible t sme greater r lesser extent; that is, when yu squeeze r press n them, their density will change The amunt by which a substance can be cmpressed
Definitin f Cmpressibility All real substances are cmpressible t sme greater r lesser extent; that is, when yu squeeze r press n them, their density will change The amunt by which a substance can be cmpressed
Then the amount of water that flows through the pipe during a differential time interval dt is (1) 4
 5-98 Review Problems 5-45 A tank oen to te atmosere is itially filled wit. e tank discarges to te atmosere troug a long ie connected to a valve. e itial discarge velocity from te tank and te time required
5-98 Review Problems 5-45 A tank oen to te atmosere is itially filled wit. e tank discarges to te atmosere troug a long ie connected to a valve. e itial discarge velocity from te tank and te time required
CHAPTER 8 ENTROPY. Blank
 CHAPER 8 ENROPY Blank SONNAG/BORGNAKKE SUDY PROBLEM 8-8. A heat engine efficiency from the inequality of Clausius Consider an actual heat engine with efficiency of η working between reservoirs at and L.
CHAPER 8 ENROPY Blank SONNAG/BORGNAKKE SUDY PROBLEM 8-8. A heat engine efficiency from the inequality of Clausius Consider an actual heat engine with efficiency of η working between reservoirs at and L.
NEWCOMEN ATMOSPHERIC ENGINE
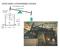 NECMEN AMSPERIC ENGINE atmoseric ressure Ford Museum, 760 strokes/m F 7 stroke 5 sia steam 8 50 F water NECMEN AMSPERIC ENGINE atmoseric ressure Ford Museum, 760 strokes/m 5 sia steam 8 F 50 F water 7
NECMEN AMSPERIC ENGINE atmoseric ressure Ford Museum, 760 strokes/m F 7 stroke 5 sia steam 8 50 F water NECMEN AMSPERIC ENGINE atmoseric ressure Ford Museum, 760 strokes/m 5 sia steam 8 F 50 F water 7
the first derivative with respect to time is obtained by carefully applying the chain rule ( surf init ) T Tinit
 .005 ermal Fluids Engineering I Fall`08 roblem Set 8 Solutions roblem ( ( a e -D eat equation is α t x d erfc( u du π x, 4αt te first derivative wit respect to time is obtained by carefully applying te
.005 ermal Fluids Engineering I Fall`08 roblem Set 8 Solutions roblem ( ( a e -D eat equation is α t x d erfc( u du π x, 4αt te first derivative wit respect to time is obtained by carefully applying te
 CHAPTER INTRODUCTION AND BASIC PRINCIPLES. (Tutorial). Determine if the following properties of the system are intensive or extensive properties: Property Intensive Extensive Volume Density Conductivity
CHAPTER INTRODUCTION AND BASIC PRINCIPLES. (Tutorial). Determine if the following properties of the system are intensive or extensive properties: Property Intensive Extensive Volume Density Conductivity
SOLUTION: Consider the system to be the refrigerator (shown in the following schematic), which operates over a cycle in normal operation.
 Soln_21 An ordinary household refrigerator operating in steady state receives electrical work while discharging net energy by heat transfer to its surroundings (e.g., the kitchen). a. Is this a violation
Soln_21 An ordinary household refrigerator operating in steady state receives electrical work while discharging net energy by heat transfer to its surroundings (e.g., the kitchen). a. Is this a violation
Lecture 29-30: Closed system entropy balance
 ME 200 Thermodynamics I Spring 2016 Lecture 29-30: Closed system entropy balance Yong Li Shanghai Jiao Tong University Institute of Refrigeration and Cryogenics 800 Dong Chuan Road Shanghai, 200240, P.
ME 200 Thermodynamics I Spring 2016 Lecture 29-30: Closed system entropy balance Yong Li Shanghai Jiao Tong University Institute of Refrigeration and Cryogenics 800 Dong Chuan Road Shanghai, 200240, P.
Consequences of Second Law of Thermodynamics. Entropy. Clausius Inequity
 onsequences of Second Law of hermodynamics Dr. Md. Zahurul Haq Professor Department of Mechanical Engineering Bangladesh University of Engineering & echnology BUE Dhaka-000, Bangladesh zahurul@me.buet.ac.bd
onsequences of Second Law of hermodynamics Dr. Md. Zahurul Haq Professor Department of Mechanical Engineering Bangladesh University of Engineering & echnology BUE Dhaka-000, Bangladesh zahurul@me.buet.ac.bd
Find this material useful? You can help our team to keep this site up and bring you even more content consider donating via the link on our site.
 Find this material useful? Yu can help ur team t keep this site up and bring yu even mre cntent cnsider dnating via the link n ur site. Still having truble understanding the material? Check ut ur Tutring
Find this material useful? Yu can help ur team t keep this site up and bring yu even mre cntent cnsider dnating via the link n ur site. Still having truble understanding the material? Check ut ur Tutring
Find this material useful? You can help our team to keep this site up and bring you even more content consider donating via the link on our site.
 Find this material useful? Yu can help ur team t keep this site up and bring yu even mre cntent cnsider dnating via the link n ur site. Still having truble understanding the material? Check ut ur Tutring
Find this material useful? Yu can help ur team t keep this site up and bring yu even mre cntent cnsider dnating via the link n ur site. Still having truble understanding the material? Check ut ur Tutring
ECE309 INTRODUCTION TO THERMODYNAMICS & HEAT TRANSFER. 20 June 2005
 ECE309 INTRODUCTION TO THERMODYNAMICS & HEAT TRANSFER 20 June 2005 Midterm Examination R. Culham & M. Bahrami This is a 90 minute, closed-book examination. You are permitted to use one 8.5 in. 11 in. crib
ECE309 INTRODUCTION TO THERMODYNAMICS & HEAT TRANSFER 20 June 2005 Midterm Examination R. Culham & M. Bahrami This is a 90 minute, closed-book examination. You are permitted to use one 8.5 in. 11 in. crib
Phys102 Final-061 Zero Version Coordinator: Nasser Wednesday, January 24, 2007 Page: 1
 Crdinatr: Nasser Wednesday, January 4, 007 Page: 1 Q1. Tw transmitters, S 1 and S shwn in the figure, emit identical sund waves f wavelength λ. The transmitters are separated by a distance λ /. Cnsider
Crdinatr: Nasser Wednesday, January 4, 007 Page: 1 Q1. Tw transmitters, S 1 and S shwn in the figure, emit identical sund waves f wavelength λ. The transmitters are separated by a distance λ /. Cnsider
ENT 254: Applied Thermodynamics
 ENT 54: Applied Thermodynamics Mr. Azizul bin Mohamad Mechanical Engineering Program School of Mechatronic Engineering Universiti Malaysia Perlis (UniMAP) azizul@unimap.edu.my 019-4747351 04-9798679 Chapter
ENT 54: Applied Thermodynamics Mr. Azizul bin Mohamad Mechanical Engineering Program School of Mechatronic Engineering Universiti Malaysia Perlis (UniMAP) azizul@unimap.edu.my 019-4747351 04-9798679 Chapter
Q1. A) 48 m/s B) 17 m/s C) 22 m/s D) 66 m/s E) 53 m/s. Ans: = 84.0 Q2.
 Phys10 Final-133 Zer Versin Crdinatr: A.A.Naqvi Wednesday, August 13, 014 Page: 1 Q1. A string, f length 0.75 m and fixed at bth ends, is vibrating in its fundamental mde. The maximum transverse speed
Phys10 Final-133 Zer Versin Crdinatr: A.A.Naqvi Wednesday, August 13, 014 Page: 1 Q1. A string, f length 0.75 m and fixed at bth ends, is vibrating in its fundamental mde. The maximum transverse speed
AP CHEMISTRY CHAPTER 6 NOTES THERMOCHEMISTRY
 AP CHEMISTRY CHAPTER 6 NOTES THERMOCHEMISTRY Energy- the capacity t d wrk r t prduce heat 1 st Law f Thermdynamics: Law f Cnservatin f Energy- energy can be cnverted frm ne frm t anther but it can be neither
AP CHEMISTRY CHAPTER 6 NOTES THERMOCHEMISTRY Energy- the capacity t d wrk r t prduce heat 1 st Law f Thermdynamics: Law f Cnservatin f Energy- energy can be cnverted frm ne frm t anther but it can be neither
Part One: Heat Changes and Thermochemistry. This aspect of Thermodynamics was dealt with in Chapter 6. (Review)
 CHAPTER 18: THERMODYNAMICS AND EQUILIBRIUM Part One: Heat Changes and Thermchemistry This aspect f Thermdynamics was dealt with in Chapter 6. (Review) A. Statement f First Law. (Sectin 18.1) 1. U ttal
CHAPTER 18: THERMODYNAMICS AND EQUILIBRIUM Part One: Heat Changes and Thermchemistry This aspect f Thermdynamics was dealt with in Chapter 6. (Review) A. Statement f First Law. (Sectin 18.1) 1. U ttal
å Q d = 0 T dq =0 ò T reversible processes 1
 δ reeribe ree d Caiu Inequaity fr an Irreeribe Cye eeribe Cye fr a CaiuInequaity fr a yemed f a reeribe andan irreeribe re irre re Entry Definitin and Cange DEFINE A POPEY S S re irre S S S ENOPY reeribe
δ reeribe ree d Caiu Inequaity fr an Irreeribe Cye eeribe Cye fr a CaiuInequaity fr a yemed f a reeribe andan irreeribe re irre re Entry Definitin and Cange DEFINE A POPEY S S re irre S S S ENOPY reeribe
1 st Law Analysis of Control Volume (open system) Chapter 6
 1 st Law Analysis of Control Volume (open system) Chapter 6 In chapter 5, we did 1st law analysis for a control mass (closed system). In this chapter the analysis of the 1st law will be on a control volume
1 st Law Analysis of Control Volume (open system) Chapter 6 In chapter 5, we did 1st law analysis for a control mass (closed system). In this chapter the analysis of the 1st law will be on a control volume
2-21. for gage pressure, the high and low pressures are expressed as. Noting that 1 psi = kpa,
 - -58E The systolic and diastolic pressures of a healthy person are given in mmhg. These pressures are to be expressed in kpa, psi, and meter water column. Assumptions Both mercury and water are incompressible
- -58E The systolic and diastolic pressures of a healthy person are given in mmhg. These pressures are to be expressed in kpa, psi, and meter water column. Assumptions Both mercury and water are incompressible
Consequences of Second Law of Thermodynamics. Entropy. Clausius Inequity
 onsequences of Second Law of hermodynamics Dr. Md. Zahurul Haq Professor Department of Mechanical Engineering Bangladesh University of Engineering & echnology BUE Dhaka-000, Bangladesh zahurul@me.buet.ac.bd
onsequences of Second Law of hermodynamics Dr. Md. Zahurul Haq Professor Department of Mechanical Engineering Bangladesh University of Engineering & echnology BUE Dhaka-000, Bangladesh zahurul@me.buet.ac.bd
The average velocity of water in the tube and the Reynolds number are Hot R-134a
 hater 0:, 8, 4, 47, 50, 5, 55, 7, 75, 77, 8 and 85. 0- Refrigerant-4a is cooled by water a double-ie heat exchanger. he overall heat transfer coefficient is to be determed. Assumtions he thermal resistance
hater 0:, 8, 4, 47, 50, 5, 55, 7, 75, 77, 8 and 85. 0- Refrigerant-4a is cooled by water a double-ie heat exchanger. he overall heat transfer coefficient is to be determed. Assumtions he thermal resistance
ES201 - Examination 2 Winter Adams and Richards NAME BOX NUMBER
 ES201 - Examinatin 2 Winter 2003-2004 Adams and Richards NAME BOX NUMBER Please Circle One : Richards (Perid 4) ES201-01 Adams (Perid 4) ES201-02 Adams (Perid 6) ES201-03 Prblem 1 ( 12 ) Prblem 2 ( 24
ES201 - Examinatin 2 Winter 2003-2004 Adams and Richards NAME BOX NUMBER Please Circle One : Richards (Perid 4) ES201-01 Adams (Perid 4) ES201-02 Adams (Perid 6) ES201-03 Prblem 1 ( 12 ) Prblem 2 ( 24
Compressor 1. Evaporator. Condenser. Expansion valve. CHE 323, October 8, Chemical Engineering Thermodynamics. Tutorial problem 5.
 CHE 33, October 8, 014. Cemical Engineering Termodynamics. Tutorial problem 5. In a simple compression refrigeration process, an adiabatic reversible compressor is used to compress propane, used as a refrigerant.
CHE 33, October 8, 014. Cemical Engineering Termodynamics. Tutorial problem 5. In a simple compression refrigeration process, an adiabatic reversible compressor is used to compress propane, used as a refrigerant.
GAS. Outline. Experiments. Device for in-class thought experiments to prove 1 st law. First law of thermodynamics Closed systems (no mass flow)
 Outline First law of thermodynamics Closed systems (no mass flow) Device for in-class thought experiments to prove 1 st law Rubber stops GAS Features: Quasi-equlibrium expansion/compression Constant volume
Outline First law of thermodynamics Closed systems (no mass flow) Device for in-class thought experiments to prove 1 st law Rubber stops GAS Features: Quasi-equlibrium expansion/compression Constant volume
Fundamental concept of metal rolling
 Fundamental cncept metal rlling Assumptins 1) Te arc cntact between te rlls and te metal is a part a circle. v x x α L p y y R v 2) Te ceicient rictin, µ, is cnstant in tery, but in reality µ varies alng
Fundamental cncept metal rlling Assumptins 1) Te arc cntact between te rlls and te metal is a part a circle. v x x α L p y y R v 2) Te ceicient rictin, µ, is cnstant in tery, but in reality µ varies alng
Chapters 19 & 20 Heat and the First Law of Thermodynamics
 Capters 19 & 20 Heat and te First Law of Termodynamics Te Zerot Law of Termodynamics Te First Law of Termodynamics Termal Processes Te Second Law of Termodynamics Heat Engines and te Carnot Cycle Refrigerators,
Capters 19 & 20 Heat and te First Law of Termodynamics Te Zerot Law of Termodynamics Te First Law of Termodynamics Termal Processes Te Second Law of Termodynamics Heat Engines and te Carnot Cycle Refrigerators,
c Dr. Md. Zahurul Haq (BUET) Entropy ME 203 (2017) 2 / 27 T037
 onsequences of Second Law of hermodynamics Dr. Md. Zahurul Haq Professor Department of Mechanical Engineering Bangladesh University of Engineering & echnology BUE Dhaka-000, Bangladesh zahurul@me.buet.ac.bd
onsequences of Second Law of hermodynamics Dr. Md. Zahurul Haq Professor Department of Mechanical Engineering Bangladesh University of Engineering & echnology BUE Dhaka-000, Bangladesh zahurul@me.buet.ac.bd
MAE 110A. Homework 3: Solutions 10/20/2017
 MAE 110A Homework 3: Solutions 10/20/2017 3.10: For H 2O, determine the specified property at the indicated state. Locate the state on a sketch of the T-v diagram. Given a) T 140 C, v 0.5 m 3 kg b) p 30MPa,
MAE 110A Homework 3: Solutions 10/20/2017 3.10: For H 2O, determine the specified property at the indicated state. Locate the state on a sketch of the T-v diagram. Given a) T 140 C, v 0.5 m 3 kg b) p 30MPa,
MAE 11. Homework 8: Solutions 11/30/2018
 MAE 11 Homework 8: Solutions 11/30/2018 MAE 11 Fall 2018 HW #8 Due: Friday, November 30 (beginning of class at 12:00p) Requirements:: Include T s diagram for all cycles. Also include p v diagrams for Ch
MAE 11 Homework 8: Solutions 11/30/2018 MAE 11 Fall 2018 HW #8 Due: Friday, November 30 (beginning of class at 12:00p) Requirements:: Include T s diagram for all cycles. Also include p v diagrams for Ch
ME Thermodynamics I. Lecture Notes and Example Problems
 ME 227.3 Thermodynamics I Lecture Notes and Example Problems James D. Bugg September 2018 Department of Mechanical Engineering Introduction Part I: Lecture Notes This part contains handout versions of
ME 227.3 Thermodynamics I Lecture Notes and Example Problems James D. Bugg September 2018 Department of Mechanical Engineering Introduction Part I: Lecture Notes This part contains handout versions of
The Second Law implies:
 e Send Law ilie: ) Heat Engine η W in H H L H L H, H H ) Ablute eerature H H L L Sale, L L W ) Fr a yle H H L L H 4) Fr an Ideal Ga Cyle H H L L L δ reerible ree d Claiu Inequality δ eerible Cyle fr a
e Send Law ilie: ) Heat Engine η W in H H L H L H, H H ) Ablute eerature H H L L Sale, L L W ) Fr a yle H H L L H 4) Fr an Ideal Ga Cyle H H L L L δ reerible ree d Claiu Inequality δ eerible Cyle fr a
Q1. A string of length L is fixed at both ends. Which one of the following is NOT a possible wavelength for standing waves on this string?
 Term: 111 Thursday, January 05, 2012 Page: 1 Q1. A string f length L is fixed at bth ends. Which ne f the fllwing is NOT a pssible wavelength fr standing waves n this string? Q2. λ n = 2L n = A) 4L B)
Term: 111 Thursday, January 05, 2012 Page: 1 Q1. A string f length L is fixed at bth ends. Which ne f the fllwing is NOT a pssible wavelength fr standing waves n this string? Q2. λ n = 2L n = A) 4L B)
Thermodynamics II. Week 9
 hermodynamics II Week 9 Example Oxygen gas in a piston cylinder at 300K, 00 kpa with volume o. m 3 is compressed in a reversible adiabatic process to a final temperature of 700K. Find the final pressure
hermodynamics II Week 9 Example Oxygen gas in a piston cylinder at 300K, 00 kpa with volume o. m 3 is compressed in a reversible adiabatic process to a final temperature of 700K. Find the final pressure
Chapter 4 ENERGY ANALYSIS OF CLOSED SYSTEMS
 4- Chapter 4 ENERGY ANALYSIS OF CLOSED SYSTEMS Mog Bondary ork 4-C It represents the bondary work for qasi-eqilibrim processes. 4-C Yes. 4-C The area nder the process cre, and ths the bondary work done,
4- Chapter 4 ENERGY ANALYSIS OF CLOSED SYSTEMS Mog Bondary ork 4-C It represents the bondary work for qasi-eqilibrim processes. 4-C Yes. 4-C The area nder the process cre, and ths the bondary work done,
Assume that the water in the nozzle is accelerated at a rate such that the frictional effect can be neglected.
 1 HW #3: Cnservatin f Linear Mmentum, Cnservatin f Energy, Cnservatin f Angular Mmentum and Turbmachines, Bernulli s Equatin, Dimensinal Analysis, and Pipe Flws Prblem 1. Cnservatins f Mass and Linear
1 HW #3: Cnservatin f Linear Mmentum, Cnservatin f Energy, Cnservatin f Angular Mmentum and Turbmachines, Bernulli s Equatin, Dimensinal Analysis, and Pipe Flws Prblem 1. Cnservatins f Mass and Linear
ME 2322 Thermodynamics I PRE-LECTURE Lesson 23 Complete the items below Name:
 Lesson 23 1. (10 pt) Write the equation for the thermal efficiency of a Carnot heat engine below: T η = T 1 L H 2. (10 pt) Can the thermal efficiency of an actual engine ever exceed that of an equivalent
Lesson 23 1. (10 pt) Write the equation for the thermal efficiency of a Carnot heat engine below: T η = T 1 L H 2. (10 pt) Can the thermal efficiency of an actual engine ever exceed that of an equivalent
