Concept of a basis. Based on this treatment we can assign the basis to one of the irreducible representations of the point group.
|
|
|
- Betty Flowers
- 6 years ago
- Views:
Transcription
1 Concept of a basis A basis refers to a type of function that is transformed by the symmetry operations of a point group. Examples include the spherical harmonics, vectors, internal coordinates (e..g bonds, angles, torsions), translations, rotations and any other function needed to describe the electronic or nuclear properties of a molecule. The spherical harmonics include the orbitals, s, p, d etc. and can have more than one dimension. Thus, we need to examine how those functions are changed by the operations. Based on this treatment we can assign the basis to one of the irreducible representations of the point group.
2 Orbital basis
3 Oxygen s-orbitals in water, I C 2v E C 2 σ v σ v ' Γ O(s) Γ O(s) = a 1 s-orbitals on central elements will always transform as the totally symmetric representation but are not included in character tables
4 p z E C 2 σ v(xz) σ v(yz)
5 p z E C 2 σ v(xz) σ v(yz)
6 p x E C 2 σ v(xz) σ v(yz)
7 p x E C 2 σ v(xz) σ v(yz)
8 Oxygen p-orbitals in water, p x C 2v E C 2 σ v (xz) σ v '(yz) p z a 1 p x b 1 p y b 2 Γ p Thus, Γ p = a 1 + b 1 + b 2. The px orbital is said to form the basis for the b 1 representation, have b 1 symmetry, or transform as b 1 p z p y
9 S-orbitals as a basis I II III E each atom has 1 and not 3 labels so each operation is a 3x3 matrix as opposed to the 9x9 matrices of the Cartesian basis. In addition, there can be no sign change for an s- orbital. The resulting representation is, E = ; C 2 = ; σ = v ; σ ' v =
10 s-orbitals as a basis I II III E each atom has 1 and not 3 labels so each operation is a 3x3 matrix as opposed to the 9x9 matrices of the Cartesian basis. In addition, there can be no sign change for an s- orbital. The resulting representation is, E = ; C 2 = ; σ v = ; σ ' v = Γ
11 Intuitive approach to finding the basis vectors in the s-orbital space Consider the symmetry adapted linear combinations (SALC's) A C for water s-orbitals I II III II III A = I B = II + III C = II - III A B = C I II = III I II + III II III
12 In this basis, no basis vector is changed into another by a symmetry operation, i.e., this basis is symmetry adapted. Old 3x3 is now three 1x1 matrices and reducible representation Γ S is now three irreducible representations, Γ A, Γ B and Γ C. I II III Γ S = i.e., Γ S = Γ A + Γ B + Γ C E C 2 σ σ' Γ A Γ B Γ C Γ S I A Γ A = II III Γ B = II III Γ C = B C
13 Rotation basis
14 R z E C 2 σ v(xz) σ v(yz)
15 R z E C 2 σ v(xz) σ v(yz)
16 R y E C 2 σ v(xz) σ v(yz)
17 R y E C 2 σ v(xz) σ v(yz)
18 Rotation of the water molecule, H z O x y O H H H ΓR(z) = C 2v E C 2 σ v (xz) σ v '(yz) R z a 2 R x b 2 R y b 1 Γ rot Thus, Γ rot = a 2 + b 1 + b 2
19 Translation basis Cartesian basis
20 Translations along the x, y and z directions (x, y, z) transform in the same way as px, py and pz. z O x yx y O ΓT(x) = H H H H C 2v E C 2 σ v (xz) σ v '(yz) T z a 1 T y b 2 T x b 1 Γ trans Thus, Γ trans = a 1 + b 1 + b 2
21 Motions of H 2 O The basis vectors are shown for the three atoms of water. Also shown is the result of the C 2 rotation: Using analysis of the 3N Cartesian vectors we can determine a reducible representation. We have already seen that the method involves counting the characters of the unmoved atoms. The important realization is that all atoms that are unmoved by a particular symmetry operation must have the same character.
22 We showed previously that C 2 has a character of -1 and σ v (yz) has a character of 3. For a reflection through the plane bisecting the H-O-H bond angle, χ(σv') = +1 since only the O is unshifted and a plane contributes +1 for each unshifted atom. The character for the identity element will always be the dimension of the basis since all labels are unchanged. For water then, χ(e) = 9. The representation (Γ) for water in this Cartesian basis is: E C 2 σ v (yz) σ v '(xz) Γ
23 Reducible and irreducible representations We call the representation Γ a reducible representation. Here we write the reducible representation as: E C 2 σ v (yz) σ v '(xz) Γ The irreducible representations form the basis of the point group in the same way that the vectors along x, y and z form the basis for three dimensional space. H 2 O belongs the point group C 2v. In this point group There are 4 irreducible representations, A 1, B 1, A 2, B 2. The decomposition of the reducible representation is a Unique determination of the irreducible reps (or irreps)z spanned by Γ.
24 Internal coordinates as a basis The internal coordinates are Stretch r Bend θ Torsion τ Wag ω The advantages of this coordinate system are: Translation and rotation are eliminated. Force constant are defined in terms of bond stretches, valence angle bends, torsions, and wags. These quantities can be related to bond strengths and barriers for internal rotation.
25 Example of H 2 O For example, for H 2 O we have the following internal coordinates. r 1 r 2 θ 1 The bond coordinates r 1 and r 2 transform as: C 2v E C 2 σ v(xz) σ v(yz) Γ This is a reducible representation.
Construction of the C 2v character table
 Construction of the C 2v character table The character table C 2v has the following form: C 2v E C 2 σ v (xz) σ v '(yz) Α 1 1 1 1 1 z x 2, y 2, z 2 Α 2 1 1-1 -1 R z xy Β 1 1-1 1-1 x, R y xz Β 2 1-1 -1
Construction of the C 2v character table The character table C 2v has the following form: C 2v E C 2 σ v (xz) σ v '(yz) Α 1 1 1 1 1 z x 2, y 2, z 2 Α 2 1 1-1 -1 R z xy Β 1 1-1 1-1 x, R y xz Β 2 1-1 -1
Chemistry 431. Lecture 14. Wave functions as a basis Diatomic molecules Polyatomic molecules Huckel theory. NC State University
 Chemistry 431 Lecture 14 Wave functions as a basis Diatomic molecules Polyatomic molecules Huckel theory NC State University Wave functions as the basis for irreducible representations The energy of the
Chemistry 431 Lecture 14 Wave functions as a basis Diatomic molecules Polyatomic molecules Huckel theory NC State University Wave functions as the basis for irreducible representations The energy of the
Molecular orbitals, potential energy surfaces and symmetry
 Molecular orbitals, potential energy surfaces and symmetry mathematical presentation of molecular symmetry group theory spectroscopy valence theory molecular orbitals Wave functions Hamiltonian: electronic,
Molecular orbitals, potential energy surfaces and symmetry mathematical presentation of molecular symmetry group theory spectroscopy valence theory molecular orbitals Wave functions Hamiltonian: electronic,
Also interested only in internal energies Uel (R) only internal forces, has symmetry of molecule--that is source of potential.
 IV. Molecular Vibrations IV-1 As discussed solutions, ψ, of the amiltonian, (Schrödinger Equation) must be representations of the group of the molecule i.e. energy cannot change due to a symmetry operation,
IV. Molecular Vibrations IV-1 As discussed solutions, ψ, of the amiltonian, (Schrödinger Equation) must be representations of the group of the molecule i.e. energy cannot change due to a symmetry operation,
PAPER No. : 8 (PHYSICAL SPECTROSCOPY) MODULE NO. : 23 (NORMAL MODES AND IRREDUCIBLE REPRESENTATIONS FOR POLYATOMIC MOLECULES)
 Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 23/ Normal modes and irreducible representations for polyatomic molecules CHE_P8_M23 TABLE OF CONTENTS 1. Learning
Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 23/ Normal modes and irreducible representations for polyatomic molecules CHE_P8_M23 TABLE OF CONTENTS 1. Learning
B F N O. Chemistry 6330 Problem Set 4 Answers. (1) (a) BF 4. tetrahedral (T d )
 hemistry 6330 Problem Set 4 Answers (1) (a) B 4 - tetrahedral (T d ) B T d E 8 3 3 2 6S 4 6s d G xyz 3 0-1 -1 1 G unmoved atoms 5 2 1 1 3 G total 15 0-1 -1 3 If we reduce G total we find that: G total
hemistry 6330 Problem Set 4 Answers (1) (a) B 4 - tetrahedral (T d ) B T d E 8 3 3 2 6S 4 6s d G xyz 3 0-1 -1 1 G unmoved atoms 5 2 1 1 3 G total 15 0-1 -1 3 If we reduce G total we find that: G total
Degrees of Freedom and Vibrational Modes
 Degrees of Freedom and Vibrational Modes 1. Every atom in a molecule can move in three possible directions relative to a Cartesian coordinate, so for a molecule of n atoms there are 3n degrees of freedom.
Degrees of Freedom and Vibrational Modes 1. Every atom in a molecule can move in three possible directions relative to a Cartesian coordinate, so for a molecule of n atoms there are 3n degrees of freedom.
The heart of group theory
 The heart of group theory. We can represent a molecule in a mathematical way e.g. with the coordinates of its atoms. This mathematical description of the molecule forms a basis for symmetry operation.
The heart of group theory. We can represent a molecule in a mathematical way e.g. with the coordinates of its atoms. This mathematical description of the molecule forms a basis for symmetry operation.
Symmetry. Chemistry 481(01) Spring 2017 Instructor: Dr. Upali Siriwardane Office: CTH 311 Phone Office Hours:
 Chemistry 481(01) Spring 2017 Instructor: Dr. Upali Siriwardane e-mail: upali@latech.edu Office: CT 311 Phone 257-4941 Office ours: M,W 8:00-9:00 & 11:00-12:00 am; Tu,Th, F 9:30-11:30 a.m. April 4, 2017:
Chemistry 481(01) Spring 2017 Instructor: Dr. Upali Siriwardane e-mail: upali@latech.edu Office: CT 311 Phone 257-4941 Office ours: M,W 8:00-9:00 & 11:00-12:00 am; Tu,Th, F 9:30-11:30 a.m. April 4, 2017:
Quiz 5 R = lit-atm/mol-k 1 (25) R = J/mol-K 2 (25) 3 (25) c = X 10 8 m/s 4 (25)
 ADVANCED INORGANIC CHEMISTRY QUIZ 5 and FINAL December 18, 2012 INSTRUCTIONS: PRINT YOUR NAME > NAME. QUIZ 5 : Work 4 of 1-5 (The lowest problem will be dropped) FINAL: #6 (10 points ) Work 6 of 7 to 14
ADVANCED INORGANIC CHEMISTRY QUIZ 5 and FINAL December 18, 2012 INSTRUCTIONS: PRINT YOUR NAME > NAME. QUIZ 5 : Work 4 of 1-5 (The lowest problem will be dropped) FINAL: #6 (10 points ) Work 6 of 7 to 14
Lecture 12. Symmetry Operations. NC State University
 Chemistry 431 Lecture 12 Group Theory Symmetry Operations NC State University Wave functions as the basis for irreducible representations The energy of the system will not change when symmetry Operations
Chemistry 431 Lecture 12 Group Theory Symmetry Operations NC State University Wave functions as the basis for irreducible representations The energy of the system will not change when symmetry Operations
LECTURE 3 DIRECT PRODUCTS AND SPECTROSCOPIC SELECTION RULES
 SYMMETRY II. J. M. GOICOECHEA. LECTURE 3 1 LECTURE 3 DIRECT PRODUCTS AND SPECTROSCOPIC SELECTION RULES 3.1 Direct products and many electron states Consider the problem of deciding upon the symmetry of
SYMMETRY II. J. M. GOICOECHEA. LECTURE 3 1 LECTURE 3 DIRECT PRODUCTS AND SPECTROSCOPIC SELECTION RULES 3.1 Direct products and many electron states Consider the problem of deciding upon the symmetry of
Symmetrical: implies the species possesses a number of indistinguishable configurations.
 Chapter 3 - Molecular Symmetry Symmetry helps us understand molecular structure, some chemical properties, and characteristics of physical properties (spectroscopy) used with group theory to predict vibrational
Chapter 3 - Molecular Symmetry Symmetry helps us understand molecular structure, some chemical properties, and characteristics of physical properties (spectroscopy) used with group theory to predict vibrational
Chemistry 431. NC State University. Lecture 17. Vibrational Spectroscopy
 Chemistry 43 Lecture 7 Vibrational Spectroscopy NC State University The Dipole Moment Expansion The permanent dipole moment of a molecule oscillates about an equilibrium value as the molecule vibrates.
Chemistry 43 Lecture 7 Vibrational Spectroscopy NC State University The Dipole Moment Expansion The permanent dipole moment of a molecule oscillates about an equilibrium value as the molecule vibrates.
Chapter 6 Answers to Problems
 Chapter 6 Answers to Problems 6.1 (a) NH 3 C3v E 2C3 3 v 4 1 2 3 0 1 12 0 2 3n = 3A 1 A 2 4E trans = A 1 E rot = A 2 E = 2A 2E = 4 frequencies 3n-6 1 Infrared 4 (2A 1 2E) Raman 4 (2A 1 2E) Polarized 2
Chapter 6 Answers to Problems 6.1 (a) NH 3 C3v E 2C3 3 v 4 1 2 3 0 1 12 0 2 3n = 3A 1 A 2 4E trans = A 1 E rot = A 2 E = 2A 2E = 4 frequencies 3n-6 1 Infrared 4 (2A 1 2E) Raman 4 (2A 1 2E) Polarized 2
Types of Molecular Vibrations
 Important concepts in IR spectroscopy Vibrations that result in change of dipole moment give rise to IR absorptions. The oscillating electric field of the radiation couples with the molecular vibration
Important concepts in IR spectroscopy Vibrations that result in change of dipole moment give rise to IR absorptions. The oscillating electric field of the radiation couples with the molecular vibration
Symmetry: Translation and Rotation
 Symmetry: Translation and Rotation The sixth column of the C 2v character table indicates the symmetry species for translation along (T) and rotation about (R) the Cartesian axes. y y y C 2 F v (x) T x
Symmetry: Translation and Rotation The sixth column of the C 2v character table indicates the symmetry species for translation along (T) and rotation about (R) the Cartesian axes. y y y C 2 F v (x) T x
Ch120 - Study Guide 10
 Ch120 - Study Guide 10 Adam Griffith October 17, 2005 In this guide: Symmetry; Diatomic Term Symbols; Molecular Term Symbols Last updated October 27, 2005. 1 The Origin of m l States and Symmetry We are
Ch120 - Study Guide 10 Adam Griffith October 17, 2005 In this guide: Symmetry; Diatomic Term Symbols; Molecular Term Symbols Last updated October 27, 2005. 1 The Origin of m l States and Symmetry We are
5.4. Electronic structure of water
 5.4. Electronic structure of water Water belongs to C 2v point group, we have discussed the corresponding character table. Here it is again: C 2v E C 2 σ v (yz) σ v (xz) A 1 1 1 1 1 A 2 1 1-1 -1 B 1 1-1
5.4. Electronic structure of water Water belongs to C 2v point group, we have discussed the corresponding character table. Here it is again: C 2v E C 2 σ v (yz) σ v (xz) A 1 1 1 1 1 A 2 1 1-1 -1 B 1 1-1
26 Group Theory Basics
 26 Group Theory Basics 1. Reference: Group Theory and Quantum Mechanics by Michael Tinkham. 2. We said earlier that we will go looking for the set of operators that commute with the molecular Hamiltonian.
26 Group Theory Basics 1. Reference: Group Theory and Quantum Mechanics by Michael Tinkham. 2. We said earlier that we will go looking for the set of operators that commute with the molecular Hamiltonian.
Using Symmetry to Generate Molecular Orbital Diagrams
 Using Symmetry to Generate Molecular Orbital Diagrams review a few MO concepts generate MO for XH 2, H 2 O, SF 6 Formation of a bond occurs when electron density collects between the two bonded nuclei
Using Symmetry to Generate Molecular Orbital Diagrams review a few MO concepts generate MO for XH 2, H 2 O, SF 6 Formation of a bond occurs when electron density collects between the two bonded nuclei
Molecular Orbital Theory
 Molecular Orbital Theory 1. MO theory suggests that atomic orbitals of different atoms combine to create MOLECULAR ORBITALS 2. Electrons in these MOLECULAR ORBITALS belong to the molecule as whole 3. This
Molecular Orbital Theory 1. MO theory suggests that atomic orbitals of different atoms combine to create MOLECULAR ORBITALS 2. Electrons in these MOLECULAR ORBITALS belong to the molecule as whole 3. This
ChemWiki BioWiki GeoWiki StatWiki PhysWiki MathWiki SolarWiki
 Ashley Robison My Preferences Site Tools FAQ Sign Out If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki BioWiki GeoWiki StatWiki
Ashley Robison My Preferences Site Tools FAQ Sign Out If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki BioWiki GeoWiki StatWiki
Hybridization of Atomic Orbitals. (Chapter 1 in the Klein text)
 Hybridization of Atomic Orbitals (Chapter 1 in the Klein text) Basic Ideas The atomic structures, from the Periodic Table, of atoms such as C, N, and O do not adequately explain how these atoms use orbitals
Hybridization of Atomic Orbitals (Chapter 1 in the Klein text) Basic Ideas The atomic structures, from the Periodic Table, of atoms such as C, N, and O do not adequately explain how these atoms use orbitals
5 Irreducible representations
 Physics 129b Lecture 8 Caltech, 01/1/19 5 Irreducible representations 5.5 Regular representation and its decomposition into irreps To see that the inequality is saturated, we need to consider the so-called
Physics 129b Lecture 8 Caltech, 01/1/19 5 Irreducible representations 5.5 Regular representation and its decomposition into irreps To see that the inequality is saturated, we need to consider the so-called
Symmetry and Group Theory
 Symmetry and Group Theory Based on Inorganic Chemistry, Miessler and Tarr, 4 th edition, 2011, Pearson Prentice Hall Images from Miessler and Tarr Inorganic Chemistry 2011 obtained from Pearson Education,
Symmetry and Group Theory Based on Inorganic Chemistry, Miessler and Tarr, 4 th edition, 2011, Pearson Prentice Hall Images from Miessler and Tarr Inorganic Chemistry 2011 obtained from Pearson Education,
Molecular Symmetry. Symmetry is relevant to: spectroscopy, chirality, polarity, Group Theory, Molecular Orbitals
 Molecular Symmetry Symmetry is relevant to: spectroscopy, chirality, polarity, Group Theory, Molecular Orbitals - A molecule has a symmetry element if it is unchanged by a particular symmetry operation
Molecular Symmetry Symmetry is relevant to: spectroscopy, chirality, polarity, Group Theory, Molecular Orbitals - A molecule has a symmetry element if it is unchanged by a particular symmetry operation
Problem Set 2 Due Thursday, October 1, & & & & # % (b) Construct a representation using five d orbitals that sit on the origin as a basis:
 Problem Set 2 Due Thursday, October 1, 29 Problems from Cotton: Chapter 4: 4.6, 4.7; Chapter 6: 6.2, 6.4, 6.5 Additional problems: (1) Consider the D 3h point group and use a coordinate system wherein
Problem Set 2 Due Thursday, October 1, 29 Problems from Cotton: Chapter 4: 4.6, 4.7; Chapter 6: 6.2, 6.4, 6.5 Additional problems: (1) Consider the D 3h point group and use a coordinate system wherein
PAPER No.13 :Applications of molecular symmetry and group theory Module No.35: Symmetry and chemical bonding part-6 MO
 1 Subject Chemistry Paper No and Title 13: Applications of molecular symmetry and group theory Module No and Title 35: Symmetry and chemical bonding part-v MO Module Tag CHE_P13_M35 2 TABLE O CONTENTS
1 Subject Chemistry Paper No and Title 13: Applications of molecular symmetry and group theory Module No and Title 35: Symmetry and chemical bonding part-v MO Module Tag CHE_P13_M35 2 TABLE O CONTENTS
Problem Set 2 Due Tuesday, September 27, ; p : 0. (b) Construct a representation using five d orbitals that sit on the origin as a basis: 1
 Problem Set 2 Due Tuesday, September 27, 211 Problems from Carter: Chapter 2: 2a-d,g,h,j 2.6, 2.9; Chapter 3: 1a-d,f,g 3.3, 3.6, 3.7 Additional problems: (1) Consider the D 4 point group and use a coordinate
Problem Set 2 Due Tuesday, September 27, 211 Problems from Carter: Chapter 2: 2a-d,g,h,j 2.6, 2.9; Chapter 3: 1a-d,f,g 3.3, 3.6, 3.7 Additional problems: (1) Consider the D 4 point group and use a coordinate
Molecular Symmetry 10/25/2018
 Symmetry helps us understand molecular structure, some chemical properties, and characteristics of physical properties (spectroscopy). Predict IR spectra or Interpret UV-Vis spectra Predict optical activity
Symmetry helps us understand molecular structure, some chemical properties, and characteristics of physical properties (spectroscopy). Predict IR spectra or Interpret UV-Vis spectra Predict optical activity
The symmetry properties & relative energies of atomic orbitals determine how they react to form molecular orbitals. These molecular orbitals are then
 1 The symmetry properties & relative energies of atomic orbitals determine how they react to form molecular orbitals. These molecular orbitals are then filled with the available electrons according to
1 The symmetry properties & relative energies of atomic orbitals determine how they react to form molecular orbitals. These molecular orbitals are then filled with the available electrons according to
13 Applications of molecular symmetry and group theory
 Subject Chemistry Paper No and Title Module No and Title Module Tag 13 Applications of molecular symmetry and 26 and and vibrational spectroscopy part-iii CHE_P13_M26 TABLE OF CONTENTS 1. Learning Outcomes
Subject Chemistry Paper No and Title Module No and Title Module Tag 13 Applications of molecular symmetry and 26 and and vibrational spectroscopy part-iii CHE_P13_M26 TABLE OF CONTENTS 1. Learning Outcomes
7. Arrange the molecular orbitals in order of increasing energy and add the electrons.
 Molecular Orbital Theory I. Introduction. A. Ideas. 1. Start with nuclei at their equilibrium positions. 2. onstruct a set of orbitals that cover the complete nuclear framework, called molecular orbitals
Molecular Orbital Theory I. Introduction. A. Ideas. 1. Start with nuclei at their equilibrium positions. 2. onstruct a set of orbitals that cover the complete nuclear framework, called molecular orbitals
SYMMETRY IN CHEMISTRY
 SYMMETRY IN CHEMISTRY Professor MANOJ K. MISHRA CHEMISTRY DEPARTMENT IIT BOMBAY ACKNOWLEGDEMENT: Professor David A. Micha Professor F. A. Cotton WHY SYMMETRY? An introduction to symmetry analysis For H
SYMMETRY IN CHEMISTRY Professor MANOJ K. MISHRA CHEMISTRY DEPARTMENT IIT BOMBAY ACKNOWLEGDEMENT: Professor David A. Micha Professor F. A. Cotton WHY SYMMETRY? An introduction to symmetry analysis For H
CH 612 Advanced Inorganic Chemistry Structure and Reactivity. Exam # 2 10/25/2010. Print name:
 CH 612 dvanced Inorganic Chemistry Structure and Reactivity Print name: 1. (25 points) a) Given the set of operations C 4, h determine the other operations that must be present to form a complete point
CH 612 dvanced Inorganic Chemistry Structure and Reactivity Print name: 1. (25 points) a) Given the set of operations C 4, h determine the other operations that must be present to form a complete point
130 points on 6 pages + a useful page Circle the element/compound most likely to have the desired property. Briefly explain your choice
 Name Chemistry 35 Spring 212 Exam #2, March 3, 212 5 minutes 13 points on 6 pages + a useful page 7 1. Circle the element/compound most likely to have the desired property. Briefly explain your choice
Name Chemistry 35 Spring 212 Exam #2, March 3, 212 5 minutes 13 points on 6 pages + a useful page 7 1. Circle the element/compound most likely to have the desired property. Briefly explain your choice
Molecular orbitals for σbonding in T d complexes
 Molecular orbitals for σbonding in T d complexes The set of n A B σ bonds in AB n (T d n = 4) molecules are often thought of as independent entities. The concept of MO s allows us to begin with a very
Molecular orbitals for σbonding in T d complexes The set of n A B σ bonds in AB n (T d n = 4) molecules are often thought of as independent entities. The concept of MO s allows us to begin with a very
Physical Chemistry I Fall 2016 Second Hour Exam (100 points) Name:
 Physical Chemistry I Fall 2016 Second Hour Exam (100 points) Name: (20 points) 1. Quantum calculations suggest that the molecule U 2 H 2 is planar and has symmetry D 2h. D 2h E C 2 (z) C 2 (y) C 2 (x)
Physical Chemistry I Fall 2016 Second Hour Exam (100 points) Name: (20 points) 1. Quantum calculations suggest that the molecule U 2 H 2 is planar and has symmetry D 2h. D 2h E C 2 (z) C 2 (y) C 2 (x)
Inorganic Chemistry with Doc M. Fall Semester, 2012 Day 9. Molecular Orbitals, Part 4. Beyond Diatomics, continued
 Inorganic Chemistry with Doc M. Fall Semester, 2012 Day 9. Molecular Orbitals, Part 4. Beyond Diatomics, continued Topics: Name(s): Element: 1. Using p-orbitals for σ-bonding: molecular orbital diagram
Inorganic Chemistry with Doc M. Fall Semester, 2012 Day 9. Molecular Orbitals, Part 4. Beyond Diatomics, continued Topics: Name(s): Element: 1. Using p-orbitals for σ-bonding: molecular orbital diagram
where, c is the speed of light, ν is the frequency in wave numbers (cm -1 ) and µ is the reduced mass (in amu) of A and B given by the equation: ma
 Vibrational Spectroscopy A rough definition of spectroscopy is the study of the interaction of matter with energy (radiation in the electromagnetic spectrum). A molecular vibration is a periodic distortion
Vibrational Spectroscopy A rough definition of spectroscopy is the study of the interaction of matter with energy (radiation in the electromagnetic spectrum). A molecular vibration is a periodic distortion
Group Theory: Matrix Representation & Consequences of Symmetry
 Group Theory: Matrix Representation & Consequences of Symmetry Matrix Representation of Group Theory Reducible and Irreducible Representations The Great Orthogonality Theorem The ive Rules The Standard
Group Theory: Matrix Representation & Consequences of Symmetry Matrix Representation of Group Theory Reducible and Irreducible Representations The Great Orthogonality Theorem The ive Rules The Standard
Determining the Normal Modes of Vibration
 Determining the ormal Modes of Vibration Introduction at the end of last lecture you determined the symmetry and activity of the vibrational modes of ammonia Γ vib 3 ) = A 1 IR, pol) + EIR,depol) the vibrational
Determining the ormal Modes of Vibration Introduction at the end of last lecture you determined the symmetry and activity of the vibrational modes of ammonia Γ vib 3 ) = A 1 IR, pol) + EIR,depol) the vibrational
ADVANCED INORGANIC CHEMISTRY QUIZ 4 November 29, 2012 INSTRUCTIONS: PRINT YOUR NAME > NAME.
 ADVANCED INORGANIC CHEMISTRY QIZ 4 November 29, 2012 INSTRCTIONS: PRINT YOR NAME > NAME. WORK all 4 problems SE THE CORRECT NMBER OF SIGNIFICANT FIGRES YOR SPPPLEMENTAL MATERIALS CONTAIN: A PERIODIC TABLE
ADVANCED INORGANIC CHEMISTRY QIZ 4 November 29, 2012 INSTRCTIONS: PRINT YOR NAME > NAME. WORK all 4 problems SE THE CORRECT NMBER OF SIGNIFICANT FIGRES YOR SPPPLEMENTAL MATERIALS CONTAIN: A PERIODIC TABLE
Tables for Group Theory
 Tables for Group Theory By P. W. ATKINS, M. S. CHILD, and C. S. G. PHILLIPS This provides the essential tables (character tables, direct products, descent in symmetry and subgroups) required for those
Tables for Group Theory By P. W. ATKINS, M. S. CHILD, and C. S. G. PHILLIPS This provides the essential tables (character tables, direct products, descent in symmetry and subgroups) required for those
13, Applications of molecular symmetry and group theory
 Subject Paper No and Title Module No and Title Module Tag Chemistry 13, Applications of molecular symmetry and group theory 27, Group theory and vibrational spectroscopy: Part-IV(Selection rules for IR
Subject Paper No and Title Module No and Title Module Tag Chemistry 13, Applications of molecular symmetry and group theory 27, Group theory and vibrational spectroscopy: Part-IV(Selection rules for IR
Tables for Group Theory
 Tables for Group Theory By P. W. ATKINS, M. S. CHILD, and C. S. G. PHILLIPS This provides the essential tables (character tables, direct products, descent in symmetry and subgroups) required for those
Tables for Group Theory By P. W. ATKINS, M. S. CHILD, and C. S. G. PHILLIPS This provides the essential tables (character tables, direct products, descent in symmetry and subgroups) required for those
A Quantum Mechanical Model for the Vibration and Rotation of Molecules. Rigid Rotor
 A Quantum Mechanical Model for the Vibration and Rotation of Molecules Harmonic Oscillator Rigid Rotor Degrees of Freedom Translation: quantum mechanical model is particle in box or free particle. A molecule
A Quantum Mechanical Model for the Vibration and Rotation of Molecules Harmonic Oscillator Rigid Rotor Degrees of Freedom Translation: quantum mechanical model is particle in box or free particle. A molecule
Chapter 5 Equations for Wave Function
 Chapter 5 Equations for Wave Function In very simple cases, the explicit expressions for the SALCs could be deduced by inspection, but not for complicated system. It would be useful for cases like these
Chapter 5 Equations for Wave Function In very simple cases, the explicit expressions for the SALCs could be deduced by inspection, but not for complicated system. It would be useful for cases like these
Chemistry 5325/5326. Angelo R. Rossi Department of Chemistry The University of Connecticut. January 17-24, 2012
 Symmetry and Group Theory for Computational Chemistry Applications Chemistry 5325/5326 Angelo R. Rossi Department of Chemistry The University of Connecticut angelo.rossi@uconn.edu January 17-24, 2012 Infrared
Symmetry and Group Theory for Computational Chemistry Applications Chemistry 5325/5326 Angelo R. Rossi Department of Chemistry The University of Connecticut angelo.rossi@uconn.edu January 17-24, 2012 Infrared
Notation. Irrep labels follow Mulliken s convention: A and B label nondegenerate E irreps, doubly T degenerate, triply degenerate
 Notation Irrep labels follow Mulliken s convention: A and B label nondegenerate E irreps, doubly T degenerate, triply degenerate Spectroscopists sometimes use F for triply degenerate; almost everyone G
Notation Irrep labels follow Mulliken s convention: A and B label nondegenerate E irreps, doubly T degenerate, triply degenerate Spectroscopists sometimes use F for triply degenerate; almost everyone G
In the fourth problem set, you derived the MO diagrams for two complexes containing Cr-Cr bonds:
 Problem 1 (2 points) Part 1 a. Consider the following V III complexes: V(H2O)6 3+, VF6 3-, and VCl6 3-. The table below contains the energies corresponding to the two lowest spin-allowed d-d transitions
Problem 1 (2 points) Part 1 a. Consider the following V III complexes: V(H2O)6 3+, VF6 3-, and VCl6 3-. The table below contains the energies corresponding to the two lowest spin-allowed d-d transitions
Figure 1: Transition State, Saddle Point, Reaction Pathway
 Computational Chemistry Workshops West Ridge Research Building-UAF Campus 9:00am-4:00pm, Room 009 Electronic Structure - July 19-21, 2016 Molecular Dynamics - July 26-28, 2016 Potential Energy Surfaces
Computational Chemistry Workshops West Ridge Research Building-UAF Campus 9:00am-4:00pm, Room 009 Electronic Structure - July 19-21, 2016 Molecular Dynamics - July 26-28, 2016 Potential Energy Surfaces
Molecular Orbitals in Inorganic Chemistry. Dr. P. Hunt Rm 167 (Chemistry)
 Molecular rbitals in Inorganic Chemistry Dr. P. unt p.hunt@imperial.ac.uk Rm 167 (Chemistry) http://www.ch.ic.ac.uk/hunt/ Lecture 2 utline L2 build a M diagram to show you the process quick revision stage
Molecular rbitals in Inorganic Chemistry Dr. P. unt p.hunt@imperial.ac.uk Rm 167 (Chemistry) http://www.ch.ic.ac.uk/hunt/ Lecture 2 utline L2 build a M diagram to show you the process quick revision stage
Complex Numbers; Digression. Handouts - Download and Read! Cyclic Groups. Character Tables for Cyclic Groups
 Handouts - Download and Read! http://www.chem.tamu.edu/rgroup/hughbanks/ courses/67/handouts/handouts.html translation_groups.pdf translation_groups.pdf transitions.pdf Provides background for selection
Handouts - Download and Read! http://www.chem.tamu.edu/rgroup/hughbanks/ courses/67/handouts/handouts.html translation_groups.pdf translation_groups.pdf transitions.pdf Provides background for selection
Name CHM 4610/5620 Fall 2016 November 15 EXAMINATION TWO SOLUTIONS
 Name CHM 4610/5620 Fall 2016 November 15 EXAMINATION TWO SOLUTIONS I II III IV V Total This exam consists of several problems. Rough point values are given to help you judge the value of problems. The
Name CHM 4610/5620 Fall 2016 November 15 EXAMINATION TWO SOLUTIONS I II III IV V Total This exam consists of several problems. Rough point values are given to help you judge the value of problems. The
130 points on 6 pages + a useful page 7
 Name KEY Chemistry 350 Spring 2012 Exam #2, March 30, 2012 50 minutes 130 points on 6 pages + a useful page 7 1. Circle the element/compound most likely to have the desired property. Briefly explain your
Name KEY Chemistry 350 Spring 2012 Exam #2, March 30, 2012 50 minutes 130 points on 6 pages + a useful page 7 1. Circle the element/compound most likely to have the desired property. Briefly explain your
The Unshifted Atom-A Simpler Method of Deriving Vibrational Modes of Molecular Symmetries
 Est. 1984 ORIENTAL JOURNAL OF CHEMISTRY An International Open Free Access, Peer Reviewed Research Journal www.orientjchem.org ISSN: 0970-020 X CODEN: OJCHEG 2012, Vol. 28, No. (1): Pg. 189-202 The Unshifted
Est. 1984 ORIENTAL JOURNAL OF CHEMISTRY An International Open Free Access, Peer Reviewed Research Journal www.orientjchem.org ISSN: 0970-020 X CODEN: OJCHEG 2012, Vol. 28, No. (1): Pg. 189-202 The Unshifted
5.80 Small-Molecule Spectroscopy and Dynamics
 MIT OpenCourseWare http://ocw.mit.edu 5.80 Small-Molecule Spectroscopy and Dynamics Fall 008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. Fall, 008
MIT OpenCourseWare http://ocw.mit.edu 5.80 Small-Molecule Spectroscopy and Dynamics Fall 008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. Fall, 008
5.80 Small-Molecule Spectroscopy and Dynamics
 MIT OpenCourseWare http://ocw.mit.edu 5.80 Small-Molecule Spectroscopy and Dynamics Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.80 Lecture
MIT OpenCourseWare http://ocw.mit.edu 5.80 Small-Molecule Spectroscopy and Dynamics Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.80 Lecture
Advanced Spectroscopy. Dr. P. Hunt Rm 167 (Chemistry) web-site:
 Advanced Spectroscopy Dr. P. Hunt p.hunt@imperial.ac.uk Rm 167 (Chemistry) web-site: http://www.ch.ic.ac.uk/hunt Maths! Coordinate transformations rotations! example 18.1 p501 whole chapter on Matrices
Advanced Spectroscopy Dr. P. Hunt p.hunt@imperial.ac.uk Rm 167 (Chemistry) web-site: http://www.ch.ic.ac.uk/hunt Maths! Coordinate transformations rotations! example 18.1 p501 whole chapter on Matrices
Chapter 12. Linear Molecules
 Chapter 1. Linear Molecules Notes: Most of the material presented in this chapter is taken from Bunker and Jensen (1998), Chap. 17. 1.1 Rotational Degrees of Freedom For a linear molecule, it is customary
Chapter 1. Linear Molecules Notes: Most of the material presented in this chapter is taken from Bunker and Jensen (1998), Chap. 17. 1.1 Rotational Degrees of Freedom For a linear molecule, it is customary
Chem Spring, 2017 Assignment 5 - Solutions
 Page 1 of 10 Chem 370 - Spring, 2017 Assignment 5 - Solutions 5.1 Additional combinations are p z ± d z 2, p x ±d xz, and p y ±d yz. p z ± d z 2 p x ±d xz or p y ±d yz 5.2 a. Li 2 has the configuration
Page 1 of 10 Chem 370 - Spring, 2017 Assignment 5 - Solutions 5.1 Additional combinations are p z ± d z 2, p x ±d xz, and p y ±d yz. p z ± d z 2 p x ±d xz or p y ±d yz 5.2 a. Li 2 has the configuration
b) For this ground state, obtain all possible J values and order them from lowest to highest in energy.
 Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
b) For this ground state, obtain all possible J values and order them from lowest to highest in energy.
 Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
Chem Symmetry and Introduction to Group Theory. Symmetry is all around us and is a fundamental property of nature.
 Chem 59-65 Symmetry and Introduction to Group Theory Symmetry is all around us and is a fundamental property of nature. Chem 59-65 Symmetry and Introduction to Group Theory The term symmetry is derived
Chem 59-65 Symmetry and Introduction to Group Theory Symmetry is all around us and is a fundamental property of nature. Chem 59-65 Symmetry and Introduction to Group Theory The term symmetry is derived
Review of Matrices. L A matrix is a rectangular array of numbers that combines with other such arrays according to specific rules.
 Review of Matrices L A matrix is a rectangular array of numbers that combines with other such arrays according to specific rules. T The dimension of a matrix is given as rows x columns; i.e., m x n. Matrix
Review of Matrices L A matrix is a rectangular array of numbers that combines with other such arrays according to specific rules. T The dimension of a matrix is given as rows x columns; i.e., m x n. Matrix
C 2 '' σ v ' C 2 ' "side on" "in-plane" 2S determine how the MO transforms under each symmetry operation, Figure 3.
 Lecture Model nswers to Problems Self-study Problems / Exam Preparation determine the point group of o use VESPR (from st year) to determine that is planar, then use the flow chart. is the molecule linear?
Lecture Model nswers to Problems Self-study Problems / Exam Preparation determine the point group of o use VESPR (from st year) to determine that is planar, then use the flow chart. is the molecule linear?
LECTURE 2 DEGENERACY AND DESCENT IN SYMMETRY: LIGAND FIELD SPLITTINGS AND RELATED MATTERS
 SYMMETRY II. J. M. GOICOECHEA. LECTURE 2. 1 LECTURE 2 DEGENERACY AND DESCENT IN SYMMETRY: LIGAND FIELD SPLITTINGS AND RELATED MATTERS 2.1 Degeneracy When dealing with non-degenerate symmetry adapted wavefunctions
SYMMETRY II. J. M. GOICOECHEA. LECTURE 2. 1 LECTURE 2 DEGENERACY AND DESCENT IN SYMMETRY: LIGAND FIELD SPLITTINGS AND RELATED MATTERS 2.1 Degeneracy When dealing with non-degenerate symmetry adapted wavefunctions
Chapter 6 Vibrational Spectroscopy
 Chapter 6 Vibrational Spectroscopy As with other applications of symmetry and group theory, these techniques reach their greatest utility when applied to the analysis of relatively small molecules in either
Chapter 6 Vibrational Spectroscopy As with other applications of symmetry and group theory, these techniques reach their greatest utility when applied to the analysis of relatively small molecules in either
Homework Problem Set 1 Solutions
 Chemistry 380.37 Dr. Jean M. Standard omework Problem Set 1 Solutions 1. A student investigates a bond between atoms A and B in a molecule using a software package for molecular mechanics. The student
Chemistry 380.37 Dr. Jean M. Standard omework Problem Set 1 Solutions 1. A student investigates a bond between atoms A and B in a molecule using a software package for molecular mechanics. The student
6.2 Polyatomic Molecules
 6.2 Polyatomic Molecules 6.2.1 Group Vibrations An N-atom molecule has 3N - 5 normal modes of vibrations if it is linear and 3N 6 if it is non-linear. Lissajous motion A polyatomic molecule undergoes a
6.2 Polyatomic Molecules 6.2.1 Group Vibrations An N-atom molecule has 3N - 5 normal modes of vibrations if it is linear and 3N 6 if it is non-linear. Lissajous motion A polyatomic molecule undergoes a
Mo 2+, Mo 2+, Cr electrons. Mo-Mo quadruple bond.
 Problem 1 (2 points) 1. Consider the MoMoCr heterotrimetallic complex shown below (Berry, et. al. Inorganica Chimica Acta 2015, p. 241). Metal-metal bonds are not drawn. The ligand framework distorts this
Problem 1 (2 points) 1. Consider the MoMoCr heterotrimetallic complex shown below (Berry, et. al. Inorganica Chimica Acta 2015, p. 241). Metal-metal bonds are not drawn. The ligand framework distorts this
Chem 673, Problem Set 5 Due Thursday, November 29, 2007
 Chem 673, Problem Set 5 Due Thursday, November 29, 2007 (1) Trigonal prismatic coordination is fairly common in solid-state inorganic chemistry. In most cases the geometry of the trigonal prism is such
Chem 673, Problem Set 5 Due Thursday, November 29, 2007 (1) Trigonal prismatic coordination is fairly common in solid-state inorganic chemistry. In most cases the geometry of the trigonal prism is such
B7 Symmetry : Questions
 B7 Symmetry 009-10: Questions 1. Using the definition of a group, prove the Rearrangement Theorem, that the set of h products RS obtained for a fixed element S, when R ranges over the h elements of the
B7 Symmetry 009-10: Questions 1. Using the definition of a group, prove the Rearrangement Theorem, that the set of h products RS obtained for a fixed element S, when R ranges over the h elements of the
A. General (10 points) 2 Points Each
 Chem 104A - Midterm II Total Exam Score closed text, closed notes, no calculators There are 100 total points. General advice - if you are stumped by one problem, move on to finish other problems and come
Chem 104A - Midterm II Total Exam Score closed text, closed notes, no calculators There are 100 total points. General advice - if you are stumped by one problem, move on to finish other problems and come
Little Orthogonality Theorem (LOT)
 Little Orthogonality Theorem (LOT) Take diagonal elements of D matrices in RG * D R D R i j G ij mi N * D R D R N i j G G ij ij RG mi mi ( ) By definition, D j j j R TrD R ( R). Sum GOT over β: * * ( )
Little Orthogonality Theorem (LOT) Take diagonal elements of D matrices in RG * D R D R i j G ij mi N * D R D R N i j G G ij ij RG mi mi ( ) By definition, D j j j R TrD R ( R). Sum GOT over β: * * ( )
Chem 673, Problem Set 5 Due Thursday, December 1, 2005
 otton, Problem 9.3 (assume D 4h symmetry) Additional Problems: hem 673, Problem Set 5 Due Thursday, December 1, 2005 (1) Infrared and Raman spectra of Benzene (a) Determine the symmetries (irreducible
otton, Problem 9.3 (assume D 4h symmetry) Additional Problems: hem 673, Problem Set 5 Due Thursday, December 1, 2005 (1) Infrared and Raman spectra of Benzene (a) Determine the symmetries (irreducible
Symmetry and states of self stress in triangulated toroidal frames
 Symmetry and states of self stress in triangulated toroidal frames P.W. Fowler School of Chemistry, University of Exeter Stocker Road, Exeter EX4 4QD, UK S.D. Guest Department of Engineering, University
Symmetry and states of self stress in triangulated toroidal frames P.W. Fowler School of Chemistry, University of Exeter Stocker Road, Exeter EX4 4QD, UK S.D. Guest Department of Engineering, University
Problem Set 5 Solutions
 Chemistry 362 Dr Jean M Standard Problem Set 5 Solutions ow many vibrational modes do the following molecules or ions possess? [int: Drawing Lewis structures may be useful in some cases] In all of the
Chemistry 362 Dr Jean M Standard Problem Set 5 Solutions ow many vibrational modes do the following molecules or ions possess? [int: Drawing Lewis structures may be useful in some cases] In all of the
Determining the Normal Modes of Vibration
 Determining the ormal Modes of Vibration Introduction vibrational modes of ammonia are shown below! 1 A 1 ) symmetric stretch! A 1 ) symmetric bend! 3a E) degenerate stretch Figure 1 Vibrational modes!
Determining the ormal Modes of Vibration Introduction vibrational modes of ammonia are shown below! 1 A 1 ) symmetric stretch! A 1 ) symmetric bend! 3a E) degenerate stretch Figure 1 Vibrational modes!
Midterm Exam I. CHEM 181: Introduction to Chemical Principles September 24, 2015 Key
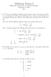 Midterm Exam I CHEM 8: Introduction to Chemical Principles September 24, 205 Key. A Li 2+ ion in an unknown, highly excited electronic state, first emits a photon at a wavelength of 4.36 µm, and following
Midterm Exam I CHEM 8: Introduction to Chemical Principles September 24, 205 Key. A Li 2+ ion in an unknown, highly excited electronic state, first emits a photon at a wavelength of 4.36 µm, and following
5.111 Principles of Chemical Science
 MIT OpenCourseWare http://ocw.mit.edu 5.111 Principles of Chemical Science Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.111 Lecture Summary
MIT OpenCourseWare http://ocw.mit.edu 5.111 Principles of Chemical Science Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.111 Lecture Summary
Lecture 4 Model Answers to Problems
 Self-Study Problems / Exam Preparation Construct and annotate a valence MO diagram for H 2 CN -. Use your diagram to explain why the neutral radical is more stable than the anion. (this is an old exam
Self-Study Problems / Exam Preparation Construct and annotate a valence MO diagram for H 2 CN -. Use your diagram to explain why the neutral radical is more stable than the anion. (this is an old exam
Theoretical Concepts of Spin-Orbit Splitting
 Chapter 9 Theoretical Concepts of Spin-Orbit Splitting 9.1 Free-electron model In order to understand the basic origin of spin-orbit coupling at the surface of a crystal, it is a natural starting point
Chapter 9 Theoretical Concepts of Spin-Orbit Splitting 9.1 Free-electron model In order to understand the basic origin of spin-orbit coupling at the surface of a crystal, it is a natural starting point
Degrees of Freedom and Vibrational Modes
 Degrees of Freedom and Vibrational Modes 1. Every atom in a molecule can move in three possible directions relative to a Cartesian coordinate, so for a molecule of n atoms there are 3n degrees of freedom.
Degrees of Freedom and Vibrational Modes 1. Every atom in a molecule can move in three possible directions relative to a Cartesian coordinate, so for a molecule of n atoms there are 3n degrees of freedom.
Symmetry Adapted Orbitals
 Symmetry Adapted Orbitals z B x y e a Figure Symmetry adapted fragment orbitals for 3 L= ML 3 M C is isolobal with P 3 P Figure 2 Isolobal relationship Introduction so far the fragments used in forming
Symmetry Adapted Orbitals z B x y e a Figure Symmetry adapted fragment orbitals for 3 L= ML 3 M C is isolobal with P 3 P Figure 2 Isolobal relationship Introduction so far the fragments used in forming
Chem 673, Problem Set 5 Due Tuesday, December 2, 2008
 Chem 673, Problem Set 5 Due Tuesday, December 2, 2008 (1) (a) Trigonal bipyramidal (tbp) coordination is fairly common. Calculate the group overlaps of the appropriate SALCs for a tbp with the 5 d-orbitals
Chem 673, Problem Set 5 Due Tuesday, December 2, 2008 (1) (a) Trigonal bipyramidal (tbp) coordination is fairly common. Calculate the group overlaps of the appropriate SALCs for a tbp with the 5 d-orbitals
Renner-Teller Effect in Tetra-Atomic Molecules
 Groupe de Chimie Théorique du MSME Renner-Teller Effect in Tetra-Atomic Molecules Laurent Jutier, G. Dhont, H. Khalil and C. Léonard jutier@univ-mlv.fr (non linear) Outline General Presentation Structure
Groupe de Chimie Théorique du MSME Renner-Teller Effect in Tetra-Atomic Molecules Laurent Jutier, G. Dhont, H. Khalil and C. Léonard jutier@univ-mlv.fr (non linear) Outline General Presentation Structure
5.04 Principles of Inorganic Chemistry II
 MIT OpenCourseWare http://ocw.mit.edu 5.04 Principles of Inorganic Chemistry II Fall 008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.04, Principles
MIT OpenCourseWare http://ocw.mit.edu 5.04 Principles of Inorganic Chemistry II Fall 008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.04, Principles
CHEMISTRY 4021/8021 MIDTERM EXAM 1 SPRING 2014
 CHEMISTRY 4021/8021 Q1) Propose a simple, united-atom molecular mechanics force-field needed to generate a potential energy surface for an isolated molecule of acetone (Me 2 CO). I.e., provide an energy
CHEMISTRY 4021/8021 Q1) Propose a simple, united-atom molecular mechanics force-field needed to generate a potential energy surface for an isolated molecule of acetone (Me 2 CO). I.e., provide an energy
1) For molecules A and B, list the symmetry operations and point groups.
 Problem 1 (2 points) The MO diagrams of complicated molecules can be constructed from the interactions of molecular fragments. The point groups of isolated fragments are often of higher symmetry that the
Problem 1 (2 points) The MO diagrams of complicated molecules can be constructed from the interactions of molecular fragments. The point groups of isolated fragments are often of higher symmetry that the
Inorganic Chemistry with Doc M. Day 18. Transition Metals Complexes IV: Ligand Field Theory continued
 Inorganic Chemistry with Doc M. Day 18. Transition Metals Complexes IV: Ligand Field Theory continued Topics: 1. The three scenarios 2. Scenario 3: π-back bonding 1. The three scenarios for the MO energy
Inorganic Chemistry with Doc M. Day 18. Transition Metals Complexes IV: Ligand Field Theory continued Topics: 1. The three scenarios 2. Scenario 3: π-back bonding 1. The three scenarios for the MO energy
Spectroscopic Selection Rules
 E 0 v = 0 v = 1 v = 2 v = 4 v = 3 For a vibrational fundamental (Δv = ±1), the transition will have nonzero intensity in either the infrared or Raman spectrum if the appropriate transition moment is nonzero.
E 0 v = 0 v = 1 v = 2 v = 4 v = 3 For a vibrational fundamental (Δv = ±1), the transition will have nonzero intensity in either the infrared or Raman spectrum if the appropriate transition moment is nonzero.
f lmn x m y n z l m n (1) 3/πz/r and Y 1 ±1 = 3/2π(x±iy)/r, and also gives a formula for Y
 Problem Set 6: Group representation theory and neutron stars Graduate Quantum I Physics 6572 James Sethna Due Monday Nov. 12 Last correction at November 14, 2012, 2:32 pm Reading Sakurai and Napolitano,
Problem Set 6: Group representation theory and neutron stars Graduate Quantum I Physics 6572 James Sethna Due Monday Nov. 12 Last correction at November 14, 2012, 2:32 pm Reading Sakurai and Napolitano,
Translation Groups; Introduction to Bands and Reciprocal Space. Timothy Hughbanks, Texas A& M University
 Translation Groups; Introduction to Bands and Reciprocal Space Timothy Hughbanks, Texas A& M University The One-dimensional Translation Group If chemists wish to understand the symmetry properties of a
Translation Groups; Introduction to Bands and Reciprocal Space Timothy Hughbanks, Texas A& M University The One-dimensional Translation Group If chemists wish to understand the symmetry properties of a
Quantum Numbers. principal quantum number: n. angular momentum quantum number: l (azimuthal) magnetic quantum number: m l
 Quantum Numbers Quantum Numbers principal quantum number: n angular momentum quantum number: l (azimuthal) magnetic quantum number: m l Principal quantum number: n related to size and energy of orbital
Quantum Numbers Quantum Numbers principal quantum number: n angular momentum quantum number: l (azimuthal) magnetic quantum number: m l Principal quantum number: n related to size and energy of orbital
EXPLAINING THE GEOMETRY OF SIMPLE MOLECULES USING MOLECULAR ORBITAL ENERGY-LEVEL DIAGRAMS BUILT BY USING SYMMETRY PRINCIPLES
 Quim. Nova, Vol. XY, No. 00, 17, 200_ http://dx.doi.org/10.21577/01004042.20170198 EXPLAINING THE GEOMETRY OF SIMPLE MOLECULES USING MOLECULAR ORBITAL ENERGYLEVEL DIAGRAMS BUILT BY USING SYMMETRY PRINCIPLES
Quim. Nova, Vol. XY, No. 00, 17, 200_ http://dx.doi.org/10.21577/01004042.20170198 EXPLAINING THE GEOMETRY OF SIMPLE MOLECULES USING MOLECULAR ORBITAL ENERGYLEVEL DIAGRAMS BUILT BY USING SYMMETRY PRINCIPLES
Rotational Motion. Chapter 4. P. J. Grandinetti. Sep. 1, Chem P. J. Grandinetti (Chem. 4300) Rotational Motion Sep.
 Rotational Motion Chapter 4 P. J. Grandinetti Chem. 4300 Sep. 1, 2017 P. J. Grandinetti (Chem. 4300) Rotational Motion Sep. 1, 2017 1 / 76 Angular Momentum The angular momentum of a particle with respect
Rotational Motion Chapter 4 P. J. Grandinetti Chem. 4300 Sep. 1, 2017 P. J. Grandinetti (Chem. 4300) Rotational Motion Sep. 1, 2017 1 / 76 Angular Momentum The angular momentum of a particle with respect
Wave Equations of Polyatomic Molecules
 Wave Equations of Polyatomic Molecules! Approximate wave functions are sought by combining atomic wave functions for the bonded atoms.! Several different approaches have been taken to constructing trial
Wave Equations of Polyatomic Molecules! Approximate wave functions are sought by combining atomic wave functions for the bonded atoms.! Several different approaches have been taken to constructing trial
