Volcanic ash as an oceanic iron source and sink. Nicholas Rogan 1,2 *, Eric P. Achterberg 1, Frederic A. C. Le Moigne 1,3, Chris M.
|
|
|
- Nelson Fox
- 5 years ago
- Views:
Transcription
1 Volcanic ash as an oceanic iron source and sink Nicholas Rogan 1,2 *, Eric P. Achterberg 1, Frederic A. C. Le Moigne 1,3, Chris M. Marsay 4, Alessandro Tagliabue 2 and Richard G. Williams 2. 1 GEOMAR Helmholtz Centre for Ocean Research Kiel, Wischhofstrasse 1-3, Kiel, Germany 2 Department of Earth Ocean and Ecological Sciences, School of Environmental Sciences, University of Liverpool, Liverpool, L69 3GP, UK 3 Ocean biogeochemistry and Ecosystems, European Way, National Oceanography Centre, Southampton, SO14 3ZH, UK 4 Skidaway Institute of Oceanography, University of Georgia, 10 Ocean Science Circle, Savannah, GA 31411, USA. *nrogan@geomar.de This article has been accepted for publication and undergone full peer review but has not been through the copyediting, typesetting, pagination and proofreading process which may lead to differences between this version and the Version of Record. Please cite this article as doi: /2016GL067905
2 Abstract Volcanic ash deposition to the ocean forms a natural source of iron (Fe) to surface water microbial communities. Inputs of lithogenic material may also facilitate Fe removal through scavenging. Combining dissolved Fe (dfe) and thorium-234 observations alongside modelling, we investigate scavenging of Fe in the North Atlantic following the Eyjafjallajökull volcanic eruption. Under typical conditions biogenic particles dominate scavenging, whereas ash particles dominate during the eruption. The size of particles is important as smaller scavenging particles can become saturated with surface-associated ions. Model simulations indicate that ash deposition associated with Eyjafjallajökull likely led to net Fe removal. Our model suggests a three-fold greater stimulation of biological activity if ash deposition had occurred later in the growing season when the region was Fe-limited. The implications of ash particle-scavenging, eruption timing and particle saturation need to be considered when assessing the impact of ash deposition on the ocean Fe cycle and productivity. Key points: 1. Volcanic ash deposition can lead to elevated Fe scavenging 2. Timing of an eruption determines magnitude of biological response to Fe 3. Biological control of particle scavenging breaks down during lithogenic particle deposition events
3 1. Introduction The Iceland and Irminger Basins of the high latitude North Atlantic (HLNA) typically exhibit a phytoplankton growth cycle with a pronounced spring bloom commencing in April, peaking in June [Sanders et al., 2005] and decaying into the autumn [Henson et al., 2013]. The Iceland Basin features residual macronutrient concentrations (2-4 µm) following the spring bloom [Sanders et al., 2005], which have been attributed to limitation of phytoplankton growth by iron (Fe) [Nielsdóttir et al., 2009]. Accordingly, primary production and subsequent carbon export are likely to be enhanced by an increased Fe supply [Le Moigne et al., 2014; Nielsdóttir et al., 2009; Ryan-Keogh et al., 2013]. The eruption of the Eyjafjallajökull volcano in Iceland started on April 14th 2010, and continued for five weeks, discharging ~270 ± 70 x 10 6 m 3 of ash particles into the atmosphere [Gudmundsson et al., 2012]. The eruption caused a major ash deposition event in the HLNA resulting in significant biogeochemical perturbations to the Iceland Basin [Achterberg et al., 2013] and sporadic ash supply to the adjacent Irminger Basin [Stohl et al., 2011]. This ash deposition is thought to have stimulated phytoplankton growth and increased nutrient drawdown of the Iceland Basin to very low nitrate concentrations, < 1 μm [Achterberg et al., 2013], among the lowest ever observed in the region. A study of the Irminger Basin also highlighted the role of anomalous winds, associated with an extremely negative state of the North Atlantic Oscillation in in driving the anomalous phytoplankton growth [Henson et al., 2013], but only for western and central parts of the basin. The fertilising effect of the ash was low compared to traditional bioassay additions of inorganic Fe (FeCl 3 ) [Ryan- Keogh et al., 2013; Achterberg et al., 2013], which produced a chlorophyll increase times greater per unit Fe. The relatively weak response to ash suggests that additional processes restrict the efficiency of ash-derived Fe to sustain phytoplankton growth.
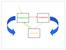
4 The removal of Fe through particle scavenging forms an important, poorly constrained, process in the oceanic Fe cycle [Balistrieri et al., 1981]. Control of trace metal scavenging by sinking biogenic particles [Siddall et al., 2005; Luo & Ku, 1999] and dust-derived lithogenic particles [Ye et al., 2011] has been demonstrated, thus ash deposition may act as an Fe sink. However, quantifying the Fe removal processes is challenging due to the inherent uncertainties of the scavenging processes [Cullen et al., 2006; Wu et al., 2001]. The scavenging of Fe is thought to be similar to that for thorium [Bruland & Lohan, 2004]. Thorium-234 ( 234 Th) is often used to derive particle export in aquatic systems [Buesseler et al., 1992], but can also provide information on the Fe cycle. Integrating thorium isotope observations ( 230 Th and 232 Th) reduces uncertainties related to steady-state assumptions and, when combined with Fe/Th ratios, permits the derivation of dfe residence times and fluxes [Hayes et al., 2015]. Following Honeyman et al. (1988), we use the combination of 234 Th and 238 U data to understand the scavenging of Fe, by evaluating (1) the net adsorption rate: rate of 234 Th transfer from the dissolved pool to the particulate pool, and (2) the net fallout rate: the vertical removal rate of particulate scavenged 234 Th. In addition, the size fraction of particles in the water column is important for scavenging, since smaller particles are likely to have a larger role in the adsorption of trace metals [Zhang et al., 2005]. At present, we do not clearly understand whether ash deposition acts as an Fe source or sink. Moreover, our ability to constrain how the prevailing biogeochemical conditions affect the adsorption and fallout rates of Fe is lacking. By combining observations of 234 Th and 238 U, we compare typical conditions and a volcanic ash deposition event in the HLNA to assess the relative roles of biogenic and lithogenic particles in controlling particle scavenging (Figure 1a) and the role of different particle size fractions. Using an idealised one-dimensional water
5 column model, we challenge the presumption that volcanic ash deposition to the surface ocean provides a net Fe supply by including the ash-derived particle scavenging and exploring the importance of eruption timing on the biogeochemical response. 2. Determining the drivers of scavenging 2.1. Data acquisition and analysis In order to assess the relative importance of biogenic particle and atmospheric mineral aerosol control on particle scavenging and explore the importance of particle size fractionation, a range of data were required. Size-fractionated (1-53 µm and >53 µm) 234 Th, particulate organic carbon (POC), biogenic silica (BSi) and calcite (CaCO 3 ) data from depths of 50 and 150 m, were obtained using in situ high volume pumps (Challenger Oceanic) [McDonnell et al., 2015] in spring and summer 2010 in the HLNA (RRS Discovery spring cruise, D350 from 26 April to 9 May, and summer cruise, D354 from 4 July to 11 August) (Figure 1b and 1c); methods described in Le Moigne et al. (2013) and Le Moigne et al. (2014). Details of sampling and analysis for dfe (< 0.2 µm), particulate Fe (pfe) species and lithogenic particles are described elsewhere [Achterberg et al., 2013; Marsay, 2012]. The rates of adsorption (k f ) and fallout (λ fal ) for 234 Th are diagnosed from the activities of dissolved and particulate 234 Th following Honeyman et al., (1988). Using the decay constant for thorium (λ Th ) and an assumption of chemical equilibrium we calculate rates from Here is the specific adsorption rate (d -1 ), λ fal is the specific fallout rate for 234 Th (d -1 ), is the activity of dissolved 234 Th, defined as the rate of change of dissolved 234 Th,
6 is the activity or rate of change of particleassociated 234 Th and A U is the rate of change of 238 U; the activities are measured in decays per day per litre (dpd l -1 ). The equilibrium relationships in (1) and (2) involve a balance between supply of dissolved 234 Th from 238 U decay represented by, the decay of dissolved 234 Th represented by, the desorption of 234 Th from particles represented by, the adsorption of dissolved 234 Th onto particles represented by and the decay of particle-associated 234 Th represented by ; all of these terms are in dpd l -1 d -1. The diagnostics of and fal for 234 Th are then taken to be appropriate for Fe, due to their similar behaviour in the surface ocean [Hayes et al., 2015]. The pfe samples were collected concurrently with particulate 234 Th using the in situ high volume pumps (Challenger Oceanic) and ash deposition fields were diagnosed from the characteristics of the source material together with an atmospheric Lagrangian dispersion model and satellite data [Stohl et al., 2011]. To take into account the timescale of particle sinking we use a lag in the ash deposition fields in order to compare scavenging rates with an appropriate ash supply. The relative importance of each size class is calculated by substituting activities for each particular size class into (1) and (2). Statistical analysis is carried out using non-parametric methods; the Wilcoxon Rank Sum test is used to determine whether datasets are significantly different, giving W, and Spearman s Rank Correlation coefficient is used to measure correlation, denoted by ρ How does volcanic ash affect fallout rates? The prevailing view is that removal of trace elements by scavenging is driven by biogenic particle production and leads to a particle fallout flux from the upper water column, at a specific fallout rate ( fal ) [Siddall et al., 2005; Luo & Ku, 1999]. This viewpoint is tested by
7 comparing data collected during summer 2010 (Figure 1b) with the Eyjafjallajökull volcanic eruption during spring 2010 (Figure 1c ). The mean fallout rates of 234 Th during the volcanic eruption in spring are double those seen for the summer, 0.25 ± 0.13 compared to 0.13 ± 0.07 d -1 respectively (W = 289, p < 0.02), indicating a faster removal of 234 Th coinciding with the ash deposition. During the ash deposition the fallout rates are not correlated with calcite concentrations, an indication of biogenic particle abundance, but the rates are positively correlated with calcite under typical conditions during the summer (ρ = 0.58, p < 0.01, Figure 2a). The observed fallout rates at 150 m (Figure 2b) in spring 2010 are related to ash deposition to the surface ocean (ρ = 0.89 p < 0.01). The fallout rates of small (1-53 µm, ρ = 0.86, p < 0.03) and large (>53 µm, ρ = 0.86, p < 0.03) particles are correlated with ash deposition (Figure 2c) on sinking timescales of 9 and 3 days respectively. The observations of fallout rates, the traditional measure of particle removal, thus support the hypothesis that biogenic particles are important for setting removal rates under typical conditions, but ash particle supply determines the removal rates during a volcanic eruption How does volcanic ash affect adsorption rates? A key loss process for the dissolved trace metals in the water column is specific net adsorption rate (k f ); the rate at which dissolved elements become associated with particulate material prior to vertical fallout. The specific net adsorption rates increased four fold from ± d -1 during the period of ash deposition in spring to ± d -1 in the summer after the eruption (W = 190, p < 0.03). There is a strong positive correlation between calcite concentration and specific net adsorption rate (ρ = 0.80, p <<0.01) under typical conditions (Figure 3a), but this relationship breaks down during the ash deposition. There is a moderate correlation between specific net adsorption rate and ash deposition (ρ = 0.43, p < 0.09) (Figure 3b). However, if lithogenic particle concentrations are used to normalise
8 specific net adsorption rate, a stronger correlation emerges between the normalised adsorption rates ((μg L -1 ) -1 d -1 ) and ash deposition (ρ = 1, p < 0.02) (Figure 3c). This latter correlation suggests that ash particles control water column adsorption rates during periods of ash deposition. At 150m this relationship is observed for large particle normalised adsorption rates (ρ = 1, p < 0.02) (Figure 3d), but is not significant for small particle adsorption rates (ρ = 0.9, p < 0.09). The specific net adsorption rates resulting from biogenic particles are higher than those for ash particles, and how this links to absolute scavenging removal fluxes is discussed in Section How does the scavenging response differ for small versus large particles? Our analyses find both small and large particles are effective in enhancing fallout, but only large particles are effective in adsorbing 234 Th at a depth of 150m. This challenges the expectation that smaller particles, with larger ratios of surface area to volume, should have a higher adsorption affinity for dissolved ions. These views can be reconciled if the slower sinking rates of small particles versus faster sinking rates of large particles are taken into account. Small particles can initially act as the strongest scavengers, but the surface sites of the small particles may become saturated with metals during their relatively slow descent to 150 m in our study system. Support for this view is derived from the strong decline in lithogenic particle normalised adsorption rates between 50m and 150m for the small particles (42 to 12 (g m -3 ) -1 d -1 ); the decline is smaller for large particles (17 to 10 (g m -3 ) -1 d -1 ) Derived Fe removal rates and fluxes As Fe and Th are both scavenged elements [Hayes et al., 2015; Whitfield & Turner, 1987; Clegg & Sarmiento, 1989; Anderson, 2014], we can use our results from 234 Th to make an appraisal of the likely changes in Fe adsorption and fallout between periods with and without
9 ash deposition. We assume adsorption rates of Fe can be quantified via the dfe pool, which actively associates with particles (>1 μm) through processes such as colloidal aggregation and surface association [Honeyman & Santschi, 1989]. The fallout rates of Fe are assessed using the leachable pfe (determined using an acetic acid-hydroxylamine leach on the particles collected from the high volume pumps), which may underestimate the total adsorbed metal [Rauschenberg & Twining, 2015]). Adsorption rates of Fe onto particles during the spring volcanic eruption and subsequent summer are 1.03 ±0.89 and 7.14 ±5.9 pm Fe d -1, respectively (W = 52, p < 0.005). The difference between specific net adsorption rates of ash particles and biogenic particles (Section 2.3) is further increased by higher observed dfe concentrations in summer. However, higher concentrations of particles and pfe in spring resulted in greater overall Fe fallout rates during the ash deposition period (26.9 ± 63 pm Fe d -1, compared to 1.3 ± 1.7 pm Fe d -1 during summer without ash deposition (W = 161, p < 0.001). Vertical integration between depths of 50m and 150m yields a pfe flux out of the upper 150 m of 2.69 ± 6.3 μmol Fe m -2 d -1 in spring (during the eruption), which is much greater than the summer flux of 0.13 ± 0.17 μmol Fe m -2 d -1. Our summer estimates for typical conditions compare reasonably well to independent estimates of Fe removal, thereby providing confidence in our estimates. For example, in the study region, fluxes of total pfe in summer were 0.56 ± 0.16 μmol Fe m -2 d -1, as measured by a neutrally buoyant sediment trap at 150 m [Marsay, 2012]. Additionally, in a different dynamical regime, dfe from dust fluxes at 150 m from the Hawaii Ocean Timeseries station ALOHA ranged from 0.06 to 0.22 μmol Fe m -2 d -1 derived from integrated 230 Th residence times, [Hayes et al., 2015]. Assuming the spring fallout rates are valid for the duration of ash deposition, integrating over
10 the surface area of the Iceland Basin (~5 x 10 5 km 2 ) and the period of significant volcanic activity (~21 days [Stohl et al., 2011]), yields a total removal of 28 ± 66 Mmol of Fe compared to a removal of 1.4 ± 1.8 Mmol Fe over the summer. Our estimate for the spring removal compares well with the total Fe supply by volcanic ash of 1.43 to 31.4 Mmol Fe [Achterberg et al., 2013] that is derived from Fe dissolution related to ash salt-layer thickness calculations. Our spring removal estimate is greater than the estimated bioavailable dfe supply, 0.55 to 1.80 Mmol Fe [Achterberg et al., 2013], that is derived from experimentally determined Fe solubility measurements. The instantaneous solubility of Fe is a parameter that takes into account any Fe that precipitates out of solution due to the Fe concentrations exceeding Fe solubility, as modified by organic Fe binding ligands, in seawater [Gledhill & Buck, 2012]. This precipitate is included in our pfe data, thus our net scavenging removal fluxes include the losses of insoluble ash Fe. The Fe losses are up to two orders of magnitude larger than the bioavailable Fe supply. Hence, our results suggest that the observed biological response to the Eyjafjallajökull eruption [Achterberg et al., 2013] was lower than might have been anticipated due to scavenging of Fe by ash particles. Therefore, an overall Fe fertilisation from an ash deposition event requires the Fe supply to be larger than its loss of Fe arising from the enhancement of particle scavenging. 3. Modelling sensitivity of biological production to ash deposition In order to test our hypothesis that ash particle scavenging forms an important process in determining oceanic biogeochemical responses to volcanic eruptions, a one-dimensional biogeochemical formulation of the Massachusetts Institute of Technology General Circulation Model [Dutkiewicz et al., 2005] is employed. A 1D version of the model is used as ash particle addition and residence time in the upper water column is on the timescale of a few days, thereby making horizontal processes relatively unimportant to our experiments.
11 The model has periodic boundaries and maintains a seasonally-repeating cycle of mixing, nutrient consumption and remineralisation by inclusion of advective supplies of heat, salt and nutrients [Williams, 1988]. An Eyjafjallajökull-analogue eruption was simulated using ash deposition fields [Stohl et al., 2011]. The ash deposition was implemented as a single supply of ash particles to the surface ocean with an instantaneous Fe solubility of 0.1% as determined by a rapid leach experiment on the ash [Achterberg et al., 2013]. In addition to Fe supply, the ash particles were explicitly represented using small (1-53 μm) and large (>53 μm) ash-particle size classes with specific particle concentration dependent scavenging rates, which were constrained by the observational data (supplementary information). Ash particle scavenging was added to the traditional scavenging closure, accounting for the role of biogenic particles, climatological lithogenic dust supply and colloidal aggregation in removing Fe from the dissolved pool. A control model run (without additional Fe supply or ash-derived processes) was compared to two model closures that resolved the volcanic ash deposition event: one including additional ash-derived scavenging (SCAV) and another without additional ash-derived scavenging (NO- SCAV). Further information on model formulation and diagnostics is included in the supplementary information. In our model, additional ash-derived scavenging is required in order to reproduce observed surface dfe concentrations (Figure 4a). Notably, removing ash scavenging yields dfe concentrations that are too high and persist for too long (Figures 4a and S2c). The ash scavenging simulation reveals that there is a strong increase in biological Fe uptake, although the ash scavenging removal of Fe is intermittently a more dominant process (Figure 4b). For the duration of the eruption the ash scavenging is responsible for 50% of the total Fe removed
12 from the surface Fe pool (the sum of net ash scavenging, net biological uptake and net traditional scavenging, Figure 4c) and peaks at almost 100% when deposition rates are high. For most of the eruption, Fe inputs (summed from dust deposition, ash deposition, advective supplies and ash particle desorption) were greater than Fe removal (Figure 4d), but when ash deposition was low the removal of Fe was greater than the supply. During this period of low ash deposition, the ash scavenging was as important for the removal of Fe from the surface water as biological uptake (Figure 4c). The Eyjafjallajökull eruption took place at the onset of the 2010 spring bloom, when macronutrients and dfe were replete. Our model suggests that the largest biological response to ash deposition would be observed when ash deposition occurs later in the season, coincident with seasonal Fe limitation and when there are residual macronutrients in the surface waters (Figure S3a and S3b). For example, the change in dfe concentration is much greater if an eruption is simulated in September (Figure 4a). A September ash deposition event leads to a greater increase in biological Fe uptake (Figure 4b), but a much lower level of ash scavenging, both in absolute and proportional terms (Figures 4b and S3c). Later in the season the ash deposition also sustains a higher proportion of Fe supply for a longer period (Figure S3d). The relative importance of the scavenging by ash particles is dependent on the timing of the ash deposition relative to the biogeochemical environment. For the September simulation, biological Fe uptake forms a larger sink for dfe as Fe stimulates biological activity during this period. Consequently, the amount of Fe that is removed by ash scavenging is reduced due to the increase in biological Fe uptake.
13 4. Conclusions Volcanic ash deposition provides both a source and a sink of Fe in the surface ocean. The prevailing biogeochemical conditions of the receiving waters determine the biological response to ash supply. Our approach of using 234 Th radioisotopes to quantify particle scavenging rates confirms that biogenic particles are important for Fe removal during typical conditions and that the specific net adsorption rates are stronger for biogenic particles relative to ash particles. However, removal fluxes of Fe out of the upper 150m during the volcanic eruption were twenty times higher than under typical conditions. The size of particles is important as smaller scavenging particles can become saturated with surface-associated ions in highly loaded systems. This analysis, supported by an idealised modelling study, suggests that the overall impact of a volcanic ash deposition event cannot be measured solely by the net supply of bioavailable Fe, instead the timing of the event relative to the biogeochemical environment is crucial. The biogeochemical response in the HLNA to ash deposition is likely to vary from being almost negligible during winter to a high sensitivity towards the end of the growing season when macronutrients are replete and the system is Fe-limited. This analysis implies that the observed biological response to the Eyjafjallajökull volcanic eruption was tempered by ash scavenging of Fe. Therefore, an ash deposition event need not automatically lead to a net increase in biological activity. 5. Acknowledgments We acknowledge support by UK NERC NE/E003818/1 Fe Biogeochemistry in the High latitude North Atlantic Ocean and the German BMBF SOPRAN project thank Maria Villa-Alfageme (University of Seville, Spain) for her
14 assistance. We are also grateful for constructive comments from two anonymous reviewers. 6. References Achterberg, E.P. et al., Natural iron fertilization by the Eyjafjallaj{ö}kull volcanic eruption. Geophys. Res. Lett., 40(5), pp Anderson, R.F., Chemical tracers of particle transport. In Treatise on Geochemistry (Second Edition). pp Balistrieri, L., Brewer, P. & Murray, J., Scavenging residence times of trace metals and surface chemistry of sinking particles in the deep ocean. Deep Sea Research Part A., 28, pp Bruland, K.W. & Lohan, M.C., Controls of Trace Metals in Seawater. Treatise Geochemistry, 6, pp Buesseler, K.O. et al., Carbon and Nitrogen Export during the Jgofs North-Atlantic Bloom Experiment Estimated from Th-234u-238 Disequilibria. Deep Sea Research Part A. Oceanographic Research Papers, 39(7-8), pp Clegg, S.L. & Sarmiento, J.L., The hydrolytic scavenging of metal ions by marine particulate matter. Progress in Oceanography, 23, pp Cullen, J.T., Bergquist, B.A. & Moffett, J.W., An assessment of particulate organic carbon to thorium-234 ratios in the ocean and their impact on the application of Th-234 as a POC flux proxy. Marine Chemistry, 98, pp Dutkiewicz, S. et al., A three-dimensional ocean-seaice-carbon cycle model and its coupling to a two-dimensional atmospheric model: uses in climate change studies, Gledhill, M. & Buck, K.N., The Organic Complexation of Iron in the Marine Environment: A Review. Front. Microbio., 3(69). Gudmundsson, M.T. et al., Ash generation and distribution from the April-May 2010 eruption of Eyjafjallajökull, Iceland. Scientific Reports, 2(572). Hayes, C.T. et al., Thorium isotopes tracing the iron cycle at the Hawaii Ocean Timeseries station ALOHA. Geochimica et Cosmochimica Acta, 169, pp Henson, S.A. et al., Unusual subpolar north Atlantic phytoplantkon bloom in 2010: Volcanic fertilization or North Atlantic Oscillation? Journal of Geophysical Research: Oceans, 118, pp Honeyman, B., Balistrieri, L. & Murray, J., Oceanic trace metal scavenging: the importance of particle concentration. Deep Sea Research Part A., 35(2), pp Honeyman, B. & Santschi, P., A Brownian-pumping model for oceanic trace metal scavenging: Evidence from Th isotopes. Journal of marine research, 47(42), pp.951
15 992. Luo, S. & Ku, T.-L., Oceanic 231Pa=230Th ratio influenced by particle composition and remineralization. Earth and Planetary Science Letters, 167, pp Marsay, C.M., Particulate trace metals, Carbon and Nitrogen in the mesopelagic. School of Ocean and Earth Science, University of Southampton, UK. McDonnell, A.M.P. et al., The oceanographic toolbox for the collection of sinking and suspended marine particles. Progress in Oceanography, 133, pp Le Moigne, F. a C. et al., Export of organic carbon and biominerals derived from 234Th and 210Po at the Porcupine Abyssal Plain. Deep-Sea Research Part I, 72, pp Le Moigne, F. a C. et al., Sequestration efficiency in the iron-limited North Atlantic: Implications for iron supply mode to fertilized blooms. Geophysical Research Letters, 41(13), pp Nielsdóttir, M.C. et al., Iron limitation of the postbloom phytoplankton communities in the Iceland Basin. Global Biogeochem. Cycles, 23(3), p.gb3001. Rauschenberg, S. & Twining, B.S., Evaluation of approaches to estimate biogenic particulate trace metals in the ocean. Marine Chemistry, 171, pp Ryan-Keogh, T.J. et al., Spatial and temporal development of phytoplankton iron stress in relation to bloom dynamics in the high-latitude North Atlantic Ocean. Limnology and Oceanography, 58(2), pp Sanders, R. et al., New production in the Irminger Basin during Journal of Marine Systems, 55(3-4), pp Siddall, M. et al., Pa/230Th fractionation by ocean transport, biogenic particle flux and particle type. Earth and Planetary Science Letters, 237, pp Stohl, A. et al., Determination of time-and height-resolved volcanic ash emissions and their use for quantitative ash dispersion modeling: the 2010 Eyjafjallajökull eruption. Atmospheric Chemistry and Physics, 11, pp Whitfield, M. & Turner, D.R., The role of particles in regulating the composition of seawater. In W. Stumm, ed. Aquatic Surface Chemistry. New York: Wiley, pp Williams, R.G., Modification of ocean eddies by air-sea interaction. Journal of Geophysical Research, 93(C12), pp Wu, J. et al., Soluble and Colloidal Iron in the Oligotrophic North Atlantic and North Pacific. Science, 293, pp Ye, Y. et al., Dust deposition: Iron source or sink? A case study. Biogeosciences, 8(8), pp
16 Zhang, L. et al., Size-fractionated thorium isotopes (228Th, 230Th, 232Th) in surface waters in the Jiulong River estuary, China. Journal of Environmental Radioactivity, 78, pp American Geophysical Union. All rights reserved.
17 Figure 1. a) Schematic illustrating how scavenging operates under typical circumstances (green arrows) and during an eruption (grey arrows), b) Contour plot of MODIS/AQUA 4km monthly mean satellite chlorophyll-a concentration (mg m -3 ) for July 2010 overlaid with calculated adsorption rates (d -1 ) for the same period, c) contour plot of ash deposition (log g m -2 d -1 ) on 25 th April 2010 overlaid with calculated adsorption rates (d -1 ) between 1 st and 7 th May 2010.
18 Figure 2 a) Relationship between calcite concentration (CaCO 3 in μmol L -1 ) and fallout rates (d -1 ) in periods with and without ash deposition (ρ = 0.58, p < 0.01), b), relationship between log ash deposition (g m -2 d -1 ) and fallout rates (d -1 ) during the volcanic eruption (ρ = 0.89, p < 0.01), c) relationship between fallout rates (d -1 ) of small (1-53 μm) and large (>53 μm) particles and ash deposition rate (g m -2 d -1 ) (both, ρ = 0.86, p < 0.03) with lags of 9 and 3 days respectively, note the second y-axis (For clarity we do not plot the full range on panel c); for full figure see Figure S1b).
19 Figure 3 a) Relationship between calcite concentration (CaCO 3 in μmol L -1 ) and adsorption rates (d -1 ) in periods with and without ash deposition (ρ = 0.80, p < <0.01), b) relationship between adsorption rate (d -1 ) and ash deposition rate (g m -2 d -1 ) (ρ = 0.43, not significant), c) relationship between lithogenic particle normalised adsorption rate ((μg L -1 ) -1 d -1 ) and ash deposition rate (g m -2 d -1 ) (ρ = 1, p < 0.02), d) relationship between lithogenic particle normalised adsorption rate ((μg L -1 ) -1 d -1 ) of small (1-53 μm) (ρ = 0.9, not significant p < 0.09) and large (>53 μm) (ρ = 1, p < 0.02) particles and ash deposition rate (g m -2 d -1 ) with lags of 9 and 3 days respectively.
20 Figure 4. a) Modelled surface dfe concentrations, from simulations with (SCAV, red lines) and without (NO-SCAV, green lines) ash-derived scavenging and from the Eyjafjallajökullanalogue (solid line) and September eruption (dashed line) simulations, with dfe data (both integrated down to 50 m) over one year, b) surface mixed-layer Fe removal fluxes (note ash scavenging on second y-axis) over one year for the Eyjafjallajökull-analogue (solid line) and September-eruption (dashed line) simulations (both using the SCAV closure), c) area plot showing relative contributions to dfe removal processes in the surface mixed-layer over a year for the Eyjafjallajökull-analogue, d) gross Fe supply and Fe removal terms in the surface mixed-layer for the Eyjafjallajökull-analogue (solid line) and September eruption (dashed line) simulations (both using SCAV closure). The timing of the two simulations examined are indicated with dashed lines and labelled in the top panel.
The role of dust in the cycling of iron in the ocean
 The role of dust in the cycling of iron in the ocean Christoph Völker, Ying Ye Alfred Wegener Institut für Polar- und Meeresforschung Meteorologisches Kolloquium Leipzig, 3. November 2016 THE OCEAN IS
The role of dust in the cycling of iron in the ocean Christoph Völker, Ying Ye Alfred Wegener Institut für Polar- und Meeresforschung Meteorologisches Kolloquium Leipzig, 3. November 2016 THE OCEAN IS
Substantial energy input to the mesopelagic ecosystem from the seasonal mixed-layer pump
 SUPPLEMENTARY INFORMATION DOI: 10.1038/NGEO2818 Substantial energy input to the mesopelagic ecosystem from the seasonal mixed-layer pump Giorgio Dall Olmo, James Dingle, Luca Polimene, Robert J. W. Brewin
SUPPLEMENTARY INFORMATION DOI: 10.1038/NGEO2818 Substantial energy input to the mesopelagic ecosystem from the seasonal mixed-layer pump Giorgio Dall Olmo, James Dingle, Luca Polimene, Robert J. W. Brewin
Australia report
 Australia 2014-2015 report AUS GEOTRACES meetings and workshops: AGU Fall Meeting (San Francisco, USA, Dec 2014) ACCOMC-CGASM annual meeting (Aspendale, Victoria, Nov 2014) APICS (Melbourne, Australia),
Australia 2014-2015 report AUS GEOTRACES meetings and workshops: AGU Fall Meeting (San Francisco, USA, Dec 2014) ACCOMC-CGASM annual meeting (Aspendale, Victoria, Nov 2014) APICS (Melbourne, Australia),
Natural iron fertilization by the Eyjafjallajökull volcanic eruption
 GEOPHYSICAL RESEARCH LETTERS, VOL. 40, 921 926, doi:10.1002/grl.50221, 2013 Natural iron fertilization by the Eyjafjallajökull volcanic eruption Eric P. Achterberg, 1 C. Mark Moore, 1 Stephanie A. Henson,
GEOPHYSICAL RESEARCH LETTERS, VOL. 40, 921 926, doi:10.1002/grl.50221, 2013 Natural iron fertilization by the Eyjafjallajökull volcanic eruption Eric P. Achterberg, 1 C. Mark Moore, 1 Stephanie A. Henson,
INSIGHTS INTO PARTICLE FORMATION AND REMINERALIZATION USING THE SHORT-LIVED RADIONUCLIDE, THORUIM-234 LA Woods Hole, MA 02543
 1 1 2 3 4 INSIGHTS INTO PARTICLE FORMATION AND REMINERALIZATION USING THE SHORT-LIVED RADIONUCLIDE, THORUIM-234 Kanchan Maiti 1,2, Claudia R. Benitez-Nelson 3 and Ken O. Buesseler 2 5 6 7 8 9 10 1 Department
1 1 2 3 4 INSIGHTS INTO PARTICLE FORMATION AND REMINERALIZATION USING THE SHORT-LIVED RADIONUCLIDE, THORUIM-234 Kanchan Maiti 1,2, Claudia R. Benitez-Nelson 3 and Ken O. Buesseler 2 5 6 7 8 9 10 1 Department
Climate Variability Studies in the Ocean
 Climate Variability Studies in the Ocean Topic 1. Long-term variations of vertical profiles of nutrients in the western North Pacific Topic 2. Biogeochemical processes related to ocean carbon cycling:
Climate Variability Studies in the Ocean Topic 1. Long-term variations of vertical profiles of nutrients in the western North Pacific Topic 2. Biogeochemical processes related to ocean carbon cycling:
Biogeochemistry of trace elements and isotopes in the Indian Ocean
 Biogeochemistry of trace elements and isotopes in the Indian Ocean Sunil Kumar Singh Geosciences Division Physical Research Laboratory Ahmedabad 380009 Ministry of Earth Sciences Government of India 2
Biogeochemistry of trace elements and isotopes in the Indian Ocean Sunil Kumar Singh Geosciences Division Physical Research Laboratory Ahmedabad 380009 Ministry of Earth Sciences Government of India 2
Global phosphorus cycle
 Global phosphorus cycle OCN 623 Chemical Oceanography 11 April 2013 2013 Arisa Okazaki and Kathleen Ruttenberg Outline 1. Introduction on global phosphorus (P) cycle 2. Terrestrial environment 3. Atmospheric
Global phosphorus cycle OCN 623 Chemical Oceanography 11 April 2013 2013 Arisa Okazaki and Kathleen Ruttenberg Outline 1. Introduction on global phosphorus (P) cycle 2. Terrestrial environment 3. Atmospheric
Iron biogeochemistry & the HNLC condition. Philip Boyd Institute for Marine & Antarctic Studies
 Iron biogeochemistry & the HNLC condition Philip Boyd Institute for Marine & Antarctic Studies 2014 Outline HNLC waters definition and implications What causes the HNLC condition? Everyday life in HNLC
Iron biogeochemistry & the HNLC condition Philip Boyd Institute for Marine & Antarctic Studies 2014 Outline HNLC waters definition and implications What causes the HNLC condition? Everyday life in HNLC
Liverpool NEMO Shelf Arctic Ocean modelling
 St. Andrews 22 July 2013 ROAM @ Liverpool NEMO Shelf Arctic Ocean modelling Maria Luneva, Jason Holt, Sarah Wakelin NEMO shelf Arctic Ocean model About 50% of the Arctic is shelf sea (
St. Andrews 22 July 2013 ROAM @ Liverpool NEMO Shelf Arctic Ocean modelling Maria Luneva, Jason Holt, Sarah Wakelin NEMO shelf Arctic Ocean model About 50% of the Arctic is shelf sea (
Time-series observations in the Northern Indian Ocean V.V.S.S. Sarma National Institute of Oceanography Visakhapatnam, India
 The Second GEOSS Asia-Pacific Symposium, Tokyo, 14-16 th April 28 Time-series observations in the Northern Indian Ocean V.V.S.S. Sarma National Institute of Oceanography Visakhapatnam, India Seasonal variations
The Second GEOSS Asia-Pacific Symposium, Tokyo, 14-16 th April 28 Time-series observations in the Northern Indian Ocean V.V.S.S. Sarma National Institute of Oceanography Visakhapatnam, India Seasonal variations
Chemical Oceanography Spring 2000 Final Exam (Use the back of the pages if necessary)(more than one answer may be correct.)
 Ocean 421 Your Name Chemical Oceanography Spring 2000 Final Exam (Use the back of the pages if necessary)(more than one answer may be correct.) 1. Due to the water molecule's (H 2 O) great abundance in
Ocean 421 Your Name Chemical Oceanography Spring 2000 Final Exam (Use the back of the pages if necessary)(more than one answer may be correct.) 1. Due to the water molecule's (H 2 O) great abundance in
Jeffrey Polovina 1, John Dunne 2, Phoebe Woodworth 1, and Evan Howell 1
 Projected expansion of the subtropical biome and contraction of the temperate and equatorial upwelling biomes in the North Pacific under global warming Jeffrey Polovina 1, John Dunne 2, Phoebe Woodworth
Projected expansion of the subtropical biome and contraction of the temperate and equatorial upwelling biomes in the North Pacific under global warming Jeffrey Polovina 1, John Dunne 2, Phoebe Woodworth
1. Introduction 2. Ocean circulation a) Temperature, salinity, density b) Thermohaline circulation c) Wind-driven surface currents d) Circulation and
 1. Introduction 2. Ocean circulation a) Temperature, salinity, density b) Thermohaline circulation c) Wind-driven surface currents d) Circulation and climate change e) Oceanic water residence times 3.
1. Introduction 2. Ocean circulation a) Temperature, salinity, density b) Thermohaline circulation c) Wind-driven surface currents d) Circulation and climate change e) Oceanic water residence times 3.
Classification of Parameters by Priority
 Classification of Parameters by Priority Parameters (variables) to be studied along the section are divided into three categories: key, essential and of interest. Key parameters are designated in Table
Classification of Parameters by Priority Parameters (variables) to be studied along the section are divided into three categories: key, essential and of interest. Key parameters are designated in Table
Dynamics of particulate organic carbon flux in a global ocean model
 Biogeosciences, 11, 1177 1198, 2014 doi:10.5194/bg-11-1177-2014 Authors 2014. CC Attribution 3.0 License. Biogeosciences Open Access Dynamics of particulate organic carbon flux in a global ocean model
Biogeosciences, 11, 1177 1198, 2014 doi:10.5194/bg-11-1177-2014 Authors 2014. CC Attribution 3.0 License. Biogeosciences Open Access Dynamics of particulate organic carbon flux in a global ocean model
Does the Iron Cycle Regulate Atmospheric CO2?
 Does the Iron Cycle Regulate Atmospheric CO2? Mick Follows, Dec 2005 http://ocean.mit.edu/~mick What regulates atmospheric CO2 on glacial-interglacial timescales? Role of ocean biology? Does the iron cycle
Does the Iron Cycle Regulate Atmospheric CO2? Mick Follows, Dec 2005 http://ocean.mit.edu/~mick What regulates atmospheric CO2 on glacial-interglacial timescales? Role of ocean biology? Does the iron cycle
Nutrients; Aerobic Carbon Production and Consumption
 Nutrients; Aerobic Carbon Production and Consumption OCN 623 Chemical Oceanography Reading: Libes, Chapters 8 and 9 Formation and respiration of organic matter DINutrients POM Primary Producers Autotrophs
Nutrients; Aerobic Carbon Production and Consumption OCN 623 Chemical Oceanography Reading: Libes, Chapters 8 and 9 Formation and respiration of organic matter DINutrients POM Primary Producers Autotrophs
IPCC AR5 WG1 - Climate Change 2013: The Physical Science Basis. Nandini Ramesh
 IPCC AR5 WG1 - Climate Change 2013: The Physical Science Basis Nandini Ramesh Seminar in Atmospheric Science 21 st February, 2014 1. Introduc,on The ocean exchanges heat, freshwater, and C with the atmosphere.
IPCC AR5 WG1 - Climate Change 2013: The Physical Science Basis Nandini Ramesh Seminar in Atmospheric Science 21 st February, 2014 1. Introduc,on The ocean exchanges heat, freshwater, and C with the atmosphere.
Modeling Indian Ocean Biogeochemistry Iron Limitation and Dipole-Zonal Mode Impacts
 Modeling Indian Ocean Biogeochemistry Iron Limitation and Dipole-Zonal Mode Impacts Jerry Wiggert jwiggert@ccpo.odu.edu Funded by the NASA Oceanography Program Outline 1) Coupled 3-D Bio-physical Model
Modeling Indian Ocean Biogeochemistry Iron Limitation and Dipole-Zonal Mode Impacts Jerry Wiggert jwiggert@ccpo.odu.edu Funded by the NASA Oceanography Program Outline 1) Coupled 3-D Bio-physical Model
Distributions of dissolved inorganic carbon and total alkalinity in the Western Arctic Ocean
 Article Advances in Polar Science doi:10.3724/sp.j.1085.2011.00246 December 2011 Vol.22 No.4 246 252 Distributions of dissolved inorganic carbon and total alkalinity in the Western Arctic Ocean SUN Heng
Article Advances in Polar Science doi:10.3724/sp.j.1085.2011.00246 December 2011 Vol.22 No.4 246 252 Distributions of dissolved inorganic carbon and total alkalinity in the Western Arctic Ocean SUN Heng
1 Carbon - Motivation
 1 Carbon - Motivation Figure 1: Atmospheric pco 2 over the past 400 thousand years as recorded in the ice core from Vostok, Antarctica (Petit et al., 1999). Figure 2: Air-sea flux of CO 2 (mol m 2 yr 1
1 Carbon - Motivation Figure 1: Atmospheric pco 2 over the past 400 thousand years as recorded in the ice core from Vostok, Antarctica (Petit et al., 1999). Figure 2: Air-sea flux of CO 2 (mol m 2 yr 1
ROLES OF THE OCEAN MESOSCALE IN THE LATERAL SUPPLY OF MASS, HEAT, CARBON AND NUTRIENTS TO THE NORTHERN HEMISPHERE SUBTROPICAL GYRE
 ROLES OF THE OCEAN MESOSCALE IN THE LATERAL SUPPLY OF MASS, HEAT, CARBON AND NUTRIENTS TO THE NORTHERN HEMISPHERE SUBTROPICAL GYRE AYAKO YAMAMOTO 1*, JAIME B. PALTER 1,2, CAROLINA O. DUFOUR 1,3, STEPHEN
ROLES OF THE OCEAN MESOSCALE IN THE LATERAL SUPPLY OF MASS, HEAT, CARBON AND NUTRIENTS TO THE NORTHERN HEMISPHERE SUBTROPICAL GYRE AYAKO YAMAMOTO 1*, JAIME B. PALTER 1,2, CAROLINA O. DUFOUR 1,3, STEPHEN
Iron Biogeochemistry in the High Latitude North Atlantic Ocean
 www.nature.com/scientificreports Received: 3 July 2017 Accepted: 2 January 2018 Published: xx xx xxxx OPEN Iron Biogeochemistry in the High Latitude North Atlantic Ocean Eric P. Achterberg 1,2, Sebastian
www.nature.com/scientificreports Received: 3 July 2017 Accepted: 2 January 2018 Published: xx xx xxxx OPEN Iron Biogeochemistry in the High Latitude North Atlantic Ocean Eric P. Achterberg 1,2, Sebastian
The effects of iron fertilization on carbon sequestration in the Southern Ocean
 The effects of iron fertilization on carbon sequestration in the Southern Ocean Ken O. Buesseler, John E. Andrews, Steven M. Pike and Matthew A. Charette Department of Marine Chemistry and Geochemistry
The effects of iron fertilization on carbon sequestration in the Southern Ocean Ken O. Buesseler, John E. Andrews, Steven M. Pike and Matthew A. Charette Department of Marine Chemistry and Geochemistry
Sulfur Biogeochemical Cycle
 Sulfur Biogeochemical Cycle Chris Moore 11/16/2015 http://www.inorganicventures.com/element/sulfur 1 Sulfur Why is it important? 14 th most abundant element in Earth s crust Sulfate is second most abundant
Sulfur Biogeochemical Cycle Chris Moore 11/16/2015 http://www.inorganicventures.com/element/sulfur 1 Sulfur Why is it important? 14 th most abundant element in Earth s crust Sulfate is second most abundant
Supplementary Figure 1. New downcore data from this study. Triangles represent the depth of radiocarbon dates. Error bars represent 2 standard error
 Supplementary Figure 1. New downcore data from this study. Triangles represent the depth of radiocarbon dates. Error bars represent 2 standard error of measurement (s.e.m.). 1 Supplementary Figure 2. Particle
Supplementary Figure 1. New downcore data from this study. Triangles represent the depth of radiocarbon dates. Error bars represent 2 standard error of measurement (s.e.m.). 1 Supplementary Figure 2. Particle
Nutrients; Aerobic Carbon Production and Consumption
 Nutrients; Aerobic Carbon Production and Consumption OCN 623 Chemical Oceanography Reading: Libes, Chapters 8 and 9 Formation and respiration of organic matter DINutrients POM Primary Producers Autotrophs
Nutrients; Aerobic Carbon Production and Consumption OCN 623 Chemical Oceanography Reading: Libes, Chapters 8 and 9 Formation and respiration of organic matter DINutrients POM Primary Producers Autotrophs
CGSN Overview. GSN Sites CSN Sites Shore Facilities
 GSN Sites CSN Sites Shore Facilities CGSN Overview Coastal Pioneer Array Endurance Array Global Irminger Sea Southern Ocean Station Papa Fixed assets Surface mooring Subsurface mooring Mobile assets Ocean
GSN Sites CSN Sites Shore Facilities CGSN Overview Coastal Pioneer Array Endurance Array Global Irminger Sea Southern Ocean Station Papa Fixed assets Surface mooring Subsurface mooring Mobile assets Ocean
Radioisotope Tracers
 Radioisotope Tracers OCN 623 Chemical Oceanography 23 March 2017 Reading: Emerson and Hedges, Chapter 5, p.153-169 2017 Frank Sansone Student Learning Outcomes At the completion of this class, students
Radioisotope Tracers OCN 623 Chemical Oceanography 23 March 2017 Reading: Emerson and Hedges, Chapter 5, p.153-169 2017 Frank Sansone Student Learning Outcomes At the completion of this class, students
Where is all the water?
 Where is all the water? The distribution of water at the Earth's surface % of total Oceans 97.25 Ice caps and glaciers 2.05 Groundwater 0.68 Lakes 0.01 Soils 0.005 Atmosphere (as vapour) 0.001 Rivers 0.0001
Where is all the water? The distribution of water at the Earth's surface % of total Oceans 97.25 Ice caps and glaciers 2.05 Groundwater 0.68 Lakes 0.01 Soils 0.005 Atmosphere (as vapour) 0.001 Rivers 0.0001
Iron flux induced by Haida eddies in the Gulf of Alaska
 GEOPHYSICAL RESEARCH LETTERS, VOL. 38,, doi:10.1029/2011gl047946, 2011 Iron flux induced by Haida eddies in the Gulf of Alaska Peng Xiu, 1 Artur P. Palacz, 1 Fei Chai, 1 Eric G. Roy, 1 and Mark L. Wells
GEOPHYSICAL RESEARCH LETTERS, VOL. 38,, doi:10.1029/2011gl047946, 2011 Iron flux induced by Haida eddies in the Gulf of Alaska Peng Xiu, 1 Artur P. Palacz, 1 Fei Chai, 1 Eric G. Roy, 1 and Mark L. Wells
Acceleration of oxygen decline in the tropical Pacific over the past decades by aerosol pollutants
 Acceleration of oxygen decline in the tropical Pacific over the past decades by aerosol pollutants T. Ito 1 *, A. Nenes 1,2,3,4, M. S. Johnson 5, N. Meskhidze 6 and C. Deutsch 7 Dissolved oxygen in the
Acceleration of oxygen decline in the tropical Pacific over the past decades by aerosol pollutants T. Ito 1 *, A. Nenes 1,2,3,4, M. S. Johnson 5, N. Meskhidze 6 and C. Deutsch 7 Dissolved oxygen in the
Nutrients; Aerobic Carbon Production and Consumption
 Nutrients; Aerobic Carbon Production and Consumption OCN 623 Chemical Oceanography Reading: Libes, Chapters 8 and 9 Why is organic matter such a good electron donor? Every (other) breath you take is a
Nutrients; Aerobic Carbon Production and Consumption OCN 623 Chemical Oceanography Reading: Libes, Chapters 8 and 9 Why is organic matter such a good electron donor? Every (other) breath you take is a
Radioisotope Tracers
 Radioisotope Tracers OCN 623 Chemical Oceanography 31 March 2016 Reading: Emerson and Hedges, Chapter 5, p.153-169 2016 Frank Sansone and David Ho Student Learning Outcomes At the completion of this module,
Radioisotope Tracers OCN 623 Chemical Oceanography 31 March 2016 Reading: Emerson and Hedges, Chapter 5, p.153-169 2016 Frank Sansone and David Ho Student Learning Outcomes At the completion of this module,
Anthropogenic aerosol deposition reduces the sensitivity of oceanic productivity to warming
 INI2016 in Melbourne Dec 4-8, 2016 Anthropogenic aerosol deposition reduces the sensitivity of oceanic productivity to warming Feng Zhou (Peking University, zhouf@pku.edu.cn) Rong Wang, Yves Balkanski,
INI2016 in Melbourne Dec 4-8, 2016 Anthropogenic aerosol deposition reduces the sensitivity of oceanic productivity to warming Feng Zhou (Peking University, zhouf@pku.edu.cn) Rong Wang, Yves Balkanski,
SCOPE 35 Scales and Global Change (1988)
 1. Types and origins of marine sediments 2. Distribution of sediments: controls and patterns 3. Sedimentary diagenesis: (a) Sedimentary and organic matter burial (b) Aerobic and anaerobic decomposition
1. Types and origins of marine sediments 2. Distribution of sediments: controls and patterns 3. Sedimentary diagenesis: (a) Sedimentary and organic matter burial (b) Aerobic and anaerobic decomposition
SCOPE 57 Particle Flux in the Ocean
 SCOPE 57 Particle Flux in the Ocean Edited by VENUGOPALAN ITTEKKOT PETRA SCHAFER SCOPEIUNEP International Carbon Unit Universitat Hamburg, Germany SUSUMU HON JO Woods Hole Oceanographic Institution Woods
SCOPE 57 Particle Flux in the Ocean Edited by VENUGOPALAN ITTEKKOT PETRA SCHAFER SCOPEIUNEP International Carbon Unit Universitat Hamburg, Germany SUSUMU HON JO Woods Hole Oceanographic Institution Woods
Catastrophic reduction of seaice in the Arctic Ocean - its impact on the marine ecosystems in the polar region-
 1/12 Catastrophic reduction of seaice in the Arctic Ocean - its impact on the marine ecosystems in the polar region- KAKENHI No.22221003 Naomi Harada (JAMSTEC) J. Onodera, E. Watanabe, K. Matsuno, K. Kimoto,
1/12 Catastrophic reduction of seaice in the Arctic Ocean - its impact on the marine ecosystems in the polar region- KAKENHI No.22221003 Naomi Harada (JAMSTEC) J. Onodera, E. Watanabe, K. Matsuno, K. Kimoto,
The Transport Matrix Method (TMM) (for fast, offline simulation of passive tracers in the ocean) Samar Khatiwala
 The Transport Matrix Method (TMM) (for fast, offline simulation of passive tracers in the ocean) Samar Khatiwala Department of Earth Sciences University of Oxford Why do we need alternatives to GCMs? Ocean
The Transport Matrix Method (TMM) (for fast, offline simulation of passive tracers in the ocean) Samar Khatiwala Department of Earth Sciences University of Oxford Why do we need alternatives to GCMs? Ocean
Actual bathymetry (with vertical exaggeration) Geometry of the ocean 1/17/2018. Patterns and observations? Patterns and observations?
 Patterns and observations? Patterns and observations? Observations? Patterns? Observations? Patterns? Geometry of the ocean Actual bathymetry (with vertical exaggeration) Continental Continental Basin
Patterns and observations? Patterns and observations? Observations? Patterns? Observations? Patterns? Geometry of the ocean Actual bathymetry (with vertical exaggeration) Continental Continental Basin
Carbon Exchanges between the Continental Margins and the Open Ocean
 Carbon Exchanges between the Continental Margins and the Open Ocean Outline: 1. Introduction to problem 2. Example of how circulation can export carbon to open ocean 3. Example of how particle transport
Carbon Exchanges between the Continental Margins and the Open Ocean Outline: 1. Introduction to problem 2. Example of how circulation can export carbon to open ocean 3. Example of how particle transport
Deep-Sea Research I 68 (2012) Contents lists available at SciVerse ScienceDirect. Deep-Sea Research I
 Deep-Sea Research I 68 (212) 24 45 Contents lists available at SciVerse ScienceDirect Deep-Sea Research I journal homepage: www.elsevier.com/locate/dsri How does dynamical spatial variability impact 234Th-derived
Deep-Sea Research I 68 (212) 24 45 Contents lists available at SciVerse ScienceDirect Deep-Sea Research I journal homepage: www.elsevier.com/locate/dsri How does dynamical spatial variability impact 234Th-derived
Tracking El Niño using optical indices of phytoplankton dynamics in the equatorial Pacific
 Abstract Tracking El Niño using optical indices of phytoplankton dynamics in the equatorial Pacific Joel Craig 1, Pete Strutton 2, Wiley Evans 2 1. College of Earth and Atmospheric Science, Georgia Institute
Abstract Tracking El Niño using optical indices of phytoplankton dynamics in the equatorial Pacific Joel Craig 1, Pete Strutton 2, Wiley Evans 2 1. College of Earth and Atmospheric Science, Georgia Institute
Problem Set #4 ANSWER KEY Fall 2009 Due: 9:30, Monday, Nov 30
 OCN 520 Problem Set #4 ANSWER KEY Fall 2009 Due: 9:30, Monday, Nov 30 1. Two-Box Ocean Model The B Flux Using a 2 box model like the one you have worked on in problem set #4 (question 1) assume the following
OCN 520 Problem Set #4 ANSWER KEY Fall 2009 Due: 9:30, Monday, Nov 30 1. Two-Box Ocean Model The B Flux Using a 2 box model like the one you have worked on in problem set #4 (question 1) assume the following
Seasonal and mesoscale variability of phytoplankton in the Arabian Sea. from satellite observations to models
 Seasonal and mesoscale variability of phytoplankton in the Arabian Sea from satellite observations to models Marina Lévy NIO Winter school, Feb 2015 LOCEAN-IPSL, France 1 Introduction Arabian Sea Chlorophyll
Seasonal and mesoscale variability of phytoplankton in the Arabian Sea from satellite observations to models Marina Lévy NIO Winter school, Feb 2015 LOCEAN-IPSL, France 1 Introduction Arabian Sea Chlorophyll
Making Sediments: Biogenic Production, Carbonate Saturation and Sediment Distributions
 Making Sediments: Biogenic Production, Carbonate Saturation and Sediment Distributions OCN 623 Chemical Oceanography Reading: Libes, Chapters 15 and 16 Outline I. Deep sea sedimentation Detrital sediments
Making Sediments: Biogenic Production, Carbonate Saturation and Sediment Distributions OCN 623 Chemical Oceanography Reading: Libes, Chapters 15 and 16 Outline I. Deep sea sedimentation Detrital sediments
APPENDIX B PHYSICAL BASELINE STUDY: NORTHEAST BAFFIN BAY 1
 APPENDIX B PHYSICAL BASELINE STUDY: NORTHEAST BAFFIN BAY 1 1 By David B. Fissel, Mar Martínez de Saavedra Álvarez, and Randy C. Kerr, ASL Environmental Sciences Inc. (Feb. 2012) West Greenland Seismic
APPENDIX B PHYSICAL BASELINE STUDY: NORTHEAST BAFFIN BAY 1 1 By David B. Fissel, Mar Martínez de Saavedra Álvarez, and Randy C. Kerr, ASL Environmental Sciences Inc. (Feb. 2012) West Greenland Seismic
Phytoplankton. Zooplankton. Nutrients
 Phytoplankton Zooplankton Nutrients Patterns of Productivity There is a large Spring Bloom in the North Atlantic (temperate latitudes remember the Gulf Stream!) What is a bloom? Analogy to terrestrial
Phytoplankton Zooplankton Nutrients Patterns of Productivity There is a large Spring Bloom in the North Atlantic (temperate latitudes remember the Gulf Stream!) What is a bloom? Analogy to terrestrial
Supplementary Figure 1. Observed Aragonite saturation variability and its drivers.
 Supplementary Figure 1. Observed Aragonite saturation variability and its drivers. Mean shift in aragonite saturation state from open ocean values, ΔΩ ocean-reef (left), due to freshwater fluxes, ΔΩ fresh
Supplementary Figure 1. Observed Aragonite saturation variability and its drivers. Mean shift in aragonite saturation state from open ocean values, ΔΩ ocean-reef (left), due to freshwater fluxes, ΔΩ fresh
Thermohaline and wind-driven circulation
 Thermohaline and wind-driven circulation Annalisa Bracco Georgia Institute of Technology School of Earth and Atmospheric Sciences NCAR ASP Colloquium: Carbon climate connections in the Earth System Tracer
Thermohaline and wind-driven circulation Annalisa Bracco Georgia Institute of Technology School of Earth and Atmospheric Sciences NCAR ASP Colloquium: Carbon climate connections in the Earth System Tracer
Chapter 17 Tritium, Carbon 14 and other "dyes" James Murray 5/15/01 Univ. Washington (note: Figures not included yet)
 Chapter 17 Tritium, Carbon 14 and other "dyes" James Murray 5/15/01 Univ. Washington (note: Figures not included yet) I. Cosmic Ray Production Cosmic ray interactions produce a wide range of nuclides in
Chapter 17 Tritium, Carbon 14 and other "dyes" James Murray 5/15/01 Univ. Washington (note: Figures not included yet) I. Cosmic Ray Production Cosmic ray interactions produce a wide range of nuclides in
Natural U-Th series radio-nuclides reveal important estuarine biogeochemical processes in the Delaware and Chesapeake Bays, USA
 Presentation to the 10th IEBC Meeting Xiamen, CHINA, 21 May 2008 Natural U-Th series radio-nuclides reveal important estuarine biogeochemical processes in the Delaware and Chesapeake Bays, USA Thomas M.
Presentation to the 10th IEBC Meeting Xiamen, CHINA, 21 May 2008 Natural U-Th series radio-nuclides reveal important estuarine biogeochemical processes in the Delaware and Chesapeake Bays, USA Thomas M.
3. Midlatitude Storm Tracks and the North Atlantic Oscillation
 3. Midlatitude Storm Tracks and the North Atlantic Oscillation Copyright 2006 Emily Shuckburgh, University of Cambridge. Not to be quoted or reproduced without permission. EFS 3/1 Review of key results
3. Midlatitude Storm Tracks and the North Atlantic Oscillation Copyright 2006 Emily Shuckburgh, University of Cambridge. Not to be quoted or reproduced without permission. EFS 3/1 Review of key results
This Week: Biogeochemical Cycles. Hydrologic Cycle Carbon Cycle
 This Week: Biogeochemical Cycles Hydrologic Cycle Carbon Cycle Announcements Reading: Chapters 4 (p. 74 81) and 8 Another Problem Set (Due next Tuesday) Exam 2: Friday Feb 29 My office hours today and
This Week: Biogeochemical Cycles Hydrologic Cycle Carbon Cycle Announcements Reading: Chapters 4 (p. 74 81) and 8 Another Problem Set (Due next Tuesday) Exam 2: Friday Feb 29 My office hours today and
Term paper topics, due February 8
 Term paper topics, due February 8 ODV mini-projects, due March 13 (10% final grade) Individuals or teams of two Using any available datasets, put together a ~7-10 minute talk to present in class on March
Term paper topics, due February 8 ODV mini-projects, due March 13 (10% final grade) Individuals or teams of two Using any available datasets, put together a ~7-10 minute talk to present in class on March
Carbon and Nutrient Cycles on Continental Shelves - Current Advances and Thoughts for Future Research
 Carbon and Nutrient Cycles on Continental Shelves - Current Advances and Thoughts for Future Research 1. Brief summary of N-based productivity measures. 2. Thoughts for future studies of the ocean margin
Carbon and Nutrient Cycles on Continental Shelves - Current Advances and Thoughts for Future Research 1. Brief summary of N-based productivity measures. 2. Thoughts for future studies of the ocean margin
The role of sea ice algae in Arctic marine ecosystems and oceanic emissions of dimethylsulfide. Hakase Hayashida Candidacy Exam August 19, 2015
 The role of sea ice algae in Arctic marine ecosystems and oceanic emissions of dimethylsulfide Hakase Hayashida Candidacy Exam August 19, 2015 Introduction: Role of sea ice algae in Arctic marine ecosystems
The role of sea ice algae in Arctic marine ecosystems and oceanic emissions of dimethylsulfide Hakase Hayashida Candidacy Exam August 19, 2015 Introduction: Role of sea ice algae in Arctic marine ecosystems
Turbulence and the Spring Phytoplankton Bloom
 Turbulence and the Spring Phytoplankton Bloom Raffaele Ferrari Earth, Atmospheric and Planetary Sciences, MIT Collaborators: Sophia Merrifield and John Taylor Toronto, February 2, 2012 Phytoplankton Bloom
Turbulence and the Spring Phytoplankton Bloom Raffaele Ferrari Earth, Atmospheric and Planetary Sciences, MIT Collaborators: Sophia Merrifield and John Taylor Toronto, February 2, 2012 Phytoplankton Bloom
Overview of Dust in the Earth System
 AAAS Symposium 1 Overview of Dust in the Earth System Dr. Karen E. Kohfeld School of Resource and Environmental Management, Simon Fraser University, CANADA What is dust? Soil mineral fragments Quartz,
AAAS Symposium 1 Overview of Dust in the Earth System Dr. Karen E. Kohfeld School of Resource and Environmental Management, Simon Fraser University, CANADA What is dust? Soil mineral fragments Quartz,
Part 1. Ocean Composition & Circulation
 OCN 401 Biogeochemical Systems (10.19.17) (Schlesinger: Chapter 9) Part 1. Ocean Composition & Circulation 1. Introduction Lecture Outline 2. Ocean Circulation a) Global Patterns in T, S, ρ b) Thermohaline
OCN 401 Biogeochemical Systems (10.19.17) (Schlesinger: Chapter 9) Part 1. Ocean Composition & Circulation 1. Introduction Lecture Outline 2. Ocean Circulation a) Global Patterns in T, S, ρ b) Thermohaline
Chapter outline. Reference 12/13/2016
 Chapter 2. observation CC EST 5103 Climate Change Science Rezaul Karim Environmental Science & Technology Jessore University of science & Technology Chapter outline Temperature in the instrumental record
Chapter 2. observation CC EST 5103 Climate Change Science Rezaul Karim Environmental Science & Technology Jessore University of science & Technology Chapter outline Temperature in the instrumental record
What we know from VERTIGO VERtical Transport In the Global Ocean
 What we know from VERTIGO VERtical Transport In the Global Ocean What controls the efficiency of particle transport between the surface and deep ocean? Geochemistry (particle characteristics), Biology
What we know from VERTIGO VERtical Transport In the Global Ocean What controls the efficiency of particle transport between the surface and deep ocean? Geochemistry (particle characteristics), Biology
Workshop and Conference on Biogeochemical Impacts of Climate and Land-Use Changes on Marine Ecosystems November 2009
 2066-12 Workshop and Conference on Biogeochemical Impacts of Climate and Land-Use Changes on Marine Ecosystems 2-10 November 2009 Nutrient cycling in the Mekong River plume M. Voss IOW Germany Nutrient
2066-12 Workshop and Conference on Biogeochemical Impacts of Climate and Land-Use Changes on Marine Ecosystems 2-10 November 2009 Nutrient cycling in the Mekong River plume M. Voss IOW Germany Nutrient
Upper Ocean Circulation
 Upper Ocean Circulation C. Chen General Physical Oceanography MAR 555 School for Marine Sciences and Technology Umass-Dartmouth 1 MAR555 Lecture 4: The Upper Oceanic Circulation The Oceanic Circulation
Upper Ocean Circulation C. Chen General Physical Oceanography MAR 555 School for Marine Sciences and Technology Umass-Dartmouth 1 MAR555 Lecture 4: The Upper Oceanic Circulation The Oceanic Circulation
Carbon Dioxide, Alkalinity and ph
 Carbon Dioxide, Alkalinity and ph OCN 623 Chemical Oceanography 15 March 2018 Reading: Libes, Chapter 15, pp. 383 389 (Remainder of chapter will be used with the classes Global Carbon Dioxide and Biogenic
Carbon Dioxide, Alkalinity and ph OCN 623 Chemical Oceanography 15 March 2018 Reading: Libes, Chapter 15, pp. 383 389 (Remainder of chapter will be used with the classes Global Carbon Dioxide and Biogenic
CO2 in atmosphere is influenced by pco2 of surface water (partial pressure of water is the CO2 (gas) that would be in equilibrium with water).
 EART 254, Lecture on April 6 & 11, 2011 Introduction (skipped most of this) Will look at C and N (maybe) cycles with respect to how they influence CO2 levels in the atmosphere. Ocean chemistry controls
EART 254, Lecture on April 6 & 11, 2011 Introduction (skipped most of this) Will look at C and N (maybe) cycles with respect to how they influence CO2 levels in the atmosphere. Ocean chemistry controls
Investigating the active flux of carbon in the Southern Ocean: the contribution of a prominent euphausiid
 Investigating the active flux of carbon in the Southern Ocean: the contribution of a prominent euphausiid PhD research Cecilia Liszka, UEA and BAS Supervisors: Geraint Tarling 1, Clara Manno 1, Carol Robinson
Investigating the active flux of carbon in the Southern Ocean: the contribution of a prominent euphausiid PhD research Cecilia Liszka, UEA and BAS Supervisors: Geraint Tarling 1, Clara Manno 1, Carol Robinson
2013 ATLANTIC HURRICANE SEASON OUTLOOK. June RMS Cat Response
 2013 ATLANTIC HURRICANE SEASON OUTLOOK June 2013 - RMS Cat Response Season Outlook At the start of the 2013 Atlantic hurricane season, which officially runs from June 1 to November 30, seasonal forecasts
2013 ATLANTIC HURRICANE SEASON OUTLOOK June 2013 - RMS Cat Response Season Outlook At the start of the 2013 Atlantic hurricane season, which officially runs from June 1 to November 30, seasonal forecasts
Reminders: Week 14 Assessment closes tonight Watch for Week 15 Assessment (will close Wednesday, Dec. 13)
 Wednesday, December 6, 2017 The Pleistocene Glaciations, Continued (Chapter 14) Reminders: Week 14 Assessment closes tonight Watch for Week 15 Assessment (will close Wednesday, Dec. 13) Homework 5 due
Wednesday, December 6, 2017 The Pleistocene Glaciations, Continued (Chapter 14) Reminders: Week 14 Assessment closes tonight Watch for Week 15 Assessment (will close Wednesday, Dec. 13) Homework 5 due
Phosphorus storage in coastal sediments: Will sea-level rise mobilize P and elevate coastal fluxes?
 Phosphorus storage in coastal sediments: Will sea-level rise mobilize P and elevate coastal fluxes? Andrea Pain, Jonathan B Martin, Caitlin Young, Moutousi Roy University of Florida Lentein.com Relevance
Phosphorus storage in coastal sediments: Will sea-level rise mobilize P and elevate coastal fluxes? Andrea Pain, Jonathan B Martin, Caitlin Young, Moutousi Roy University of Florida Lentein.com Relevance
Chemical Oceanography 14 March 2012 Points are in parentheses (show all your work) Final Exam
 Ocean 400 Name: Chemical Oceanography 14 March 2012 Winter 2012 Points are in parentheses (show all your work) (give as much detail as you can) (use back if necessary) Final Exam 1. Sarmiento and Gruber
Ocean 400 Name: Chemical Oceanography 14 March 2012 Winter 2012 Points are in parentheses (show all your work) (give as much detail as you can) (use back if necessary) Final Exam 1. Sarmiento and Gruber
2001 State of the Ocean: Chemical and Biological Oceanographic Conditions in the Newfoundland Region
 Stock Status Report G2-2 (2) 1 State of the Ocean: Chemical and Biological Oceanographic Conditions in the Background The Altantic Zone Monitoring Program (AZMP) was implemented in 1998 with the aim of
Stock Status Report G2-2 (2) 1 State of the Ocean: Chemical and Biological Oceanographic Conditions in the Background The Altantic Zone Monitoring Program (AZMP) was implemented in 1998 with the aim of
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION DOI: 10.1038/NCLIMATE1857 Figure S1a shows significant inter-annual variability in seasonal SPA data with multi-decadal periods exhibiting positive and negative SPAs. A similar
SUPPLEMENTARY INFORMATION DOI: 10.1038/NCLIMATE1857 Figure S1a shows significant inter-annual variability in seasonal SPA data with multi-decadal periods exhibiting positive and negative SPAs. A similar
- vertical and horizontal segregation Univ. Washington - case studies (Fe and N) (10/29/01)
 Chapter 10: Biolimiting Elements James W. Murray - vertical and horizontal segregation Univ. Washington - case studies (Fe and N) (10/29/01) By definition, biolimiting elements are those: necessary to
Chapter 10: Biolimiting Elements James W. Murray - vertical and horizontal segregation Univ. Washington - case studies (Fe and N) (10/29/01) By definition, biolimiting elements are those: necessary to
Phytoplankton. The Biological Pump. Nutrient Cycling and the Marine Biological Pump. Phytoplankton and Zooplankton. CSU ATS Sco9 Denning 1
 Nutrient Cycling and the Marine Biological Pump Readings: SelecGons from Williams & Follows (2011) Sabine et al (2004): Ocean Sink for Anthropogenic CO 2 Phytoplankton Diameter: < 1 mm to over 100 mm ConcentraGon:
Nutrient Cycling and the Marine Biological Pump Readings: SelecGons from Williams & Follows (2011) Sabine et al (2004): Ocean Sink for Anthropogenic CO 2 Phytoplankton Diameter: < 1 mm to over 100 mm ConcentraGon:
Investigating the contribution of allochthonous subsidies to kelp forests in central California
 Investigating the contribution of allochthonous subsidies to kelp forests in central California melissa m foley UCSC Institute of Marine Science and Center for Ocean Solutions system connectivity rivers
Investigating the contribution of allochthonous subsidies to kelp forests in central California melissa m foley UCSC Institute of Marine Science and Center for Ocean Solutions system connectivity rivers
The North Atlantic Bloom: Species composition and vertical fluxes
 The North Atlantic Bloom: Species composition and vertical fluxes T. Rynearson Graduate School of Oceanography, University of Rhode Island North Atlantic-Arctic ecocsystems Develop a process-based understanding
The North Atlantic Bloom: Species composition and vertical fluxes T. Rynearson Graduate School of Oceanography, University of Rhode Island North Atlantic-Arctic ecocsystems Develop a process-based understanding
Research Programme Polar, Marine and Coastal Systems. Current and future Arctic research priorities of Germany Nicole Biebow, AWI
 Research Programme Polar, Marine and Coastal Systems Current and future Arctic research priorities of Germany Nicole Biebow, AWI Arctic Science and Technology (S&T) Collaboration and Engagement Workshop,
Research Programme Polar, Marine and Coastal Systems Current and future Arctic research priorities of Germany Nicole Biebow, AWI Arctic Science and Technology (S&T) Collaboration and Engagement Workshop,
Impact of Climate Change on Polar Ecology Focus on Arctic
 Impact of Climate Change on Polar Ecology Focus on Arctic Marcel Babin Canada Excellence Research Chair on Remote sensing of Canada s new Arctic Frontier Takuvik Joint International Laboratory CNRS & Université
Impact of Climate Change on Polar Ecology Focus on Arctic Marcel Babin Canada Excellence Research Chair on Remote sensing of Canada s new Arctic Frontier Takuvik Joint International Laboratory CNRS & Université
The role of eddies in the isopycnic transfer of nutrients and their impact on biological production
 Journal of Marine Research, 58, 895 917, 2000 The role of eddies in the isopycnic transfer of nutrients and their impact on biological production by Mei-Man Lee 1 and Richard G. Williams 2 ABSTRACT Eddies
Journal of Marine Research, 58, 895 917, 2000 The role of eddies in the isopycnic transfer of nutrients and their impact on biological production by Mei-Man Lee 1 and Richard G. Williams 2 ABSTRACT Eddies
Does a ballast effect occur in the surface ocean?
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2010gl042574, 2010 Does a ballast effect occur in the surface ocean? Richard Sanders, 1 Paul J. Morris, 1,3 Alex J. Poulton,
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2010gl042574, 2010 Does a ballast effect occur in the surface ocean? Richard Sanders, 1 Paul J. Morris, 1,3 Alex J. Poulton,
OCN/ATM/ESS 587. Ocean circulation, dynamics and thermodynamics.
 OCN/ATM/ESS 587 Ocean circulation, dynamics and thermodynamics. Equation of state for seawater General T/S properties of the upper ocean Heat balance of the upper ocean Upper ocean circulation Deep circulation
OCN/ATM/ESS 587 Ocean circulation, dynamics and thermodynamics. Equation of state for seawater General T/S properties of the upper ocean Heat balance of the upper ocean Upper ocean circulation Deep circulation
PH YSIC A L PROPERT IE S TERC.UCDAVIS.EDU
 PH YSIC A L PROPERT IE S 8 Lake surface level Daily since 1900 Lake surface level varies throughout the year. Lake level rises due to high stream inflow, groundwater inflow and precipitation directly onto
PH YSIC A L PROPERT IE S 8 Lake surface level Daily since 1900 Lake surface level varies throughout the year. Lake level rises due to high stream inflow, groundwater inflow and precipitation directly onto
Ocean Mixing and Climate Change
 Ocean Mixing and Climate Change Factors inducing seawater mixing Different densities Wind stirring Internal waves breaking Tidal Bottom topography Biogenic Mixing (??) In general, any motion favoring turbulent
Ocean Mixing and Climate Change Factors inducing seawater mixing Different densities Wind stirring Internal waves breaking Tidal Bottom topography Biogenic Mixing (??) In general, any motion favoring turbulent
The North Atlantic Oscillation: Climatic Significance and Environmental Impact
 1 The North Atlantic Oscillation: Climatic Significance and Environmental Impact James W. Hurrell National Center for Atmospheric Research Climate and Global Dynamics Division, Climate Analysis Section
1 The North Atlantic Oscillation: Climatic Significance and Environmental Impact James W. Hurrell National Center for Atmospheric Research Climate and Global Dynamics Division, Climate Analysis Section
Project of Strategic Interest NEXTDATA. Deliverables D1.3.B and 1.3.C. Final Report on the quality of Reconstruction/Reanalysis products
 Project of Strategic Interest NEXTDATA Deliverables D1.3.B and 1.3.C Final Report on the quality of Reconstruction/Reanalysis products WP Coordinator: Nadia Pinardi INGV, Bologna Deliverable authors Claudia
Project of Strategic Interest NEXTDATA Deliverables D1.3.B and 1.3.C Final Report on the quality of Reconstruction/Reanalysis products WP Coordinator: Nadia Pinardi INGV, Bologna Deliverable authors Claudia
Ocean Sciences 101 The Marine Environment OCEA 101 THE MARINE ENVIRONMENT MID-TERM EXAM
 OCEA 101 THE MARINE ENVIRONMENT MID-TERM EXAM Part I. Multiple Choice Questions. Choose the one best answer from the list, and write the letter legibly in the blank to the left of the question. 2 points
OCEA 101 THE MARINE ENVIRONMENT MID-TERM EXAM Part I. Multiple Choice Questions. Choose the one best answer from the list, and write the letter legibly in the blank to the left of the question. 2 points
(4) Give an example of important reactions that are responsible for the composition of river water.
 Lecture 12 Global Biogeochemical Cycles (1) If rivers are the chief source of the dissolved salts in seawater, why is seawater not simply a concentrated version of average composition of all rivers? The
Lecture 12 Global Biogeochemical Cycles (1) If rivers are the chief source of the dissolved salts in seawater, why is seawater not simply a concentrated version of average composition of all rivers? The
Ocean Boundary Currents Guiding Question: How do western boundary currents influence climate and ocean productivity?
 Name: Date: TEACHER VERSION: Suggested Student Responses Included Ocean Boundary Currents Guiding Question: How do western boundary currents influence climate and ocean productivity? Introduction The circulation
Name: Date: TEACHER VERSION: Suggested Student Responses Included Ocean Boundary Currents Guiding Question: How do western boundary currents influence climate and ocean productivity? Introduction The circulation
National Oceanography Centre. Research & Consultancy Report No. 36
 National Oceanography Centre Research & Consultancy Report No. 36 State of the eastern North Atlantic subpolar gyre: The Extended Ellett Line Programme Annual Report No. 1 N P Holliday 1, S Cunningham
National Oceanography Centre Research & Consultancy Report No. 36 State of the eastern North Atlantic subpolar gyre: The Extended Ellett Line Programme Annual Report No. 1 N P Holliday 1, S Cunningham
Marine Sediments EPSS15 Spring 2017 Lab 4
 Marine Sediments EPSS15 Spring 2017 Lab 4 Why Sediments? Record of Earth s history - Tectonic plate movement - Past changes in climate - Ancient ocean circulation currents - Cataclysmic events 1 Classification
Marine Sediments EPSS15 Spring 2017 Lab 4 Why Sediments? Record of Earth s history - Tectonic plate movement - Past changes in climate - Ancient ocean circulation currents - Cataclysmic events 1 Classification
A twenty year reversal in water mass trends in the subtropical North Atlantic
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L12608, doi:10.1029/2007gl029957, 2007 A twenty year reversal in water mass trends in the subtropical North Atlantic S. J. Leadbetter,
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L12608, doi:10.1029/2007gl029957, 2007 A twenty year reversal in water mass trends in the subtropical North Atlantic S. J. Leadbetter,
PRMS WHITE PAPER 2014 NORTH ATLANTIC HURRICANE SEASON OUTLOOK. June RMS Event Response
 PRMS WHITE PAPER 2014 NORTH ATLANTIC HURRICANE SEASON OUTLOOK June 2014 - RMS Event Response 2014 SEASON OUTLOOK The 2013 North Atlantic hurricane season saw the fewest hurricanes in the Atlantic Basin
PRMS WHITE PAPER 2014 NORTH ATLANTIC HURRICANE SEASON OUTLOOK June 2014 - RMS Event Response 2014 SEASON OUTLOOK The 2013 North Atlantic hurricane season saw the fewest hurricanes in the Atlantic Basin
Nutrient streams and their induction into the mixed layer
 GLOBAL BIOGEOCHEMICAL CYCLES, VOL. 20,, doi:10.1029/2005gb002586, 2006 Nutrient streams and their induction into the mixed layer Richard G. Williams, 1 Vassil Roussenov, 1 and Michael J. Follows 2 Received
GLOBAL BIOGEOCHEMICAL CYCLES, VOL. 20,, doi:10.1029/2005gb002586, 2006 Nutrient streams and their induction into the mixed layer Richard G. Williams, 1 Vassil Roussenov, 1 and Michael J. Follows 2 Received
High initial time sensitivity of medium range forecasting observed for a stratospheric sudden warming
 GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2010gl044119, 2010 High initial time sensitivity of medium range forecasting observed for a stratospheric sudden warming Yuhji Kuroda 1 Received 27 May
GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2010gl044119, 2010 High initial time sensitivity of medium range forecasting observed for a stratospheric sudden warming Yuhji Kuroda 1 Received 27 May
Lecture 6 - Determinants of Seawater Composition. Sets up electric dipole because O is more electronegative A o. Figure 3.
 12.742 - Marine Chemistry Fall 2004 Lecture 6 - Determinants of Seawater Composition Prof. Scott Doney What is seawater? Water Dissolved inorganic salts (major ions) Trace species, organics, colloids,
12.742 - Marine Chemistry Fall 2004 Lecture 6 - Determinants of Seawater Composition Prof. Scott Doney What is seawater? Water Dissolved inorganic salts (major ions) Trace species, organics, colloids,
Seawater Chemistry and Chemical Oceanography. The Universal Solvent. Sphere of Hydration
 Seawater Chemistry and Chemical Oceanography The Universal Solvent Polarity of molecule makes water very effective at hydrating even weakly charged ions Sphere of Hydration Polarity of water molecules
Seawater Chemistry and Chemical Oceanography The Universal Solvent Polarity of molecule makes water very effective at hydrating even weakly charged ions Sphere of Hydration Polarity of water molecules
Climate/Ocean dynamics
 Interannual variations of the East-Kamchatka and East-Sakhalin Currents volume transports and their impact on the temperature and chemical parameters in the Okhotsk Sea Andrey G. Andreev V.I. Il ichev
Interannual variations of the East-Kamchatka and East-Sakhalin Currents volume transports and their impact on the temperature and chemical parameters in the Okhotsk Sea Andrey G. Andreev V.I. Il ichev
Carbon pathways in the South Atlantic
 Carbon pathways in the South Atlantic Olga T. Sato, Ph.D. Paulo Polito, Ph.D. olga.sato@usp.br - polito@usp.br Oceanographic Institute University of São Paulo Olga Sato and Paulo Polito (IOUSP) Carbon
Carbon pathways in the South Atlantic Olga T. Sato, Ph.D. Paulo Polito, Ph.D. olga.sato@usp.br - polito@usp.br Oceanographic Institute University of São Paulo Olga Sato and Paulo Polito (IOUSP) Carbon
