Spatiotemporal dynamics of membrane remodeling and fusion proteins during endocytic transport
|
|
|
- Opal Rose
- 5 years ago
- Views:
Transcription
1 MBo ARTILE Spatiotemporal dynamics of membrane remodeling and fusion proteins during endocytic transport Henning Arlt a, Kathrin Auffarth a, Rainer Kurre b, ominik Lisse c, Jacob Piehler c, and hristian Ungermann a a Biochemistry Section, b enter of Advanced Light Microscopy, and c Biophysics Section, University of Osnabrück, 97 Osnabrück, Germany ABSTRAT Organelles of the endolysosomal system undergo multiple fission and fusion events to combine sorting of selected proteins to the vacuole with endosomal recycling. This sorting requires a consecutive remodeling of the organelle surface in the course of endosomal maturation. Here we dissect the remodeling and fusion machinery on endosomes during the process of endocytosis. We traced selected GFP-tagged endosomal proteins relative to exogenously added fluorescently labeled α-factor on its way from the plasma membrane to the vacuole. Our data reveal that the machinery of endosomal fusion and ESRT proteins has similar temporal localization on endosomes, whereas they precede the retromer cargo recognition complex. Neither deletion of retromer nor the fusion machinery with the vacuole affects this maturation process, although the kinetics seems to be delayed due to ESRT deletion. Of importance, in strains lacking the active Rab7-like Ypt7 or the vacuolar SNARE fusion machinery, α-factor still proceeds to late endosomes with the same kinetics. This indicates that endosomal maturation is mainly controlled by the early endosomal fusion and remodeling machinery but not the downstream Rab Ypt7 or the SNARE machinery. Our data thus provide important further understanding of endosomal biogenesis in the context of cargo sorting. Monitoring Editor Akihiko Nakano RIKEN Received: Aug, 1 Revised: ec, 1 Accepted: Jan, 15 INTROUTION Organelles in the endomembrane system dynamically associate with a variety of proteins to mediate membrane fission and fusion events during protein trafficking. The association of proteins with their particular target membrane depends on the lipid composition, phosphorylation of specific phosphoinositides, and other peripheral and integral membrane proteins. The surface of these organelles is, This article was published online ahead of print in MBo in Press ( on February 5, 15. Address correspondence to: hristian Ungermann (cu@uos.de). Abbreviations used:, bright field; MA, 7-amino--chloromethylcoumarin; ORVET, class core vacuole/endosome tethering complex; PY, carboxypeptidase Y; R, cargo recognition complex; ESRT, endosomal sorting complexes required for transport; GEF, guanine nucleotide exchange factor; GFP, green fluorescent protein; HOPS, homotypic fusion and vacuole protein sorting; ILV, intraluminal vesicle; PI3P, phosphoinositide-3-phosphate; SNARE, N-ethylmaleimide sensitive factor attachment protein receptor; SNX, sorting nexin. 15 Arlt et al. This article is distributed by The American Society for ell Biology under license from the author(s). Two months after publication it is available to the public under an Attribution Noncommercial Share Alike 3. Unported reative ommons License ( ASB, The American Society for ell Biology, and Molecular Biology of the ell are registered trademarks of The American Society for ell Biology. however, not static but undergoes remodeling to function in membrane tubulation or membrane fusion (Huotari and Helenius, 11). Within the endocytic pathway, such remodeling processes are particularly dramatic at the early endosome/late endosome transition. Early endosomes contain multiple tubular extensions, start to form intraluminal vesicles (ILVs), and eventually convert into late endosomes during a process termed endosomal maturation. The machinery of membrane remodeling is closely linked to the Rab GTPases Rab5 at the early endosome and Rab7 at the late endosome. Rabs are switch-like proteins with -terminal prenyl anchors. In their GP form, Rabs are kept soluble in the cytosol by binding to the GP dissociation inhibitor (GI). Membrane-bound guanine nucleotide exchange factors (GEFs) recruit the Rab-GP and trigger nucleotide exchange to the active Rab-GTP, which can then bind to effector proteins such as fusion factors or recycling protein complexes (Gerondopoulos et al., 1; Barr, 13; Blumer et al., 13; abrera and Ungermann, 13). The process of endocytosis is critical for the down-regulation of plasma membrane proteins, which is generally initiated by ligand binding or in response to quality control measures. Supplemental Material can be found at: Volume April 1,
2 Membrane proteins are subsequently packaged into endocytic vesicles, which fuse with early endosomes in a Rab5-dependent reaction (Zeigerer et al., 1). In yeast, this process requires the homologous Vps1 protein, the tethering protein Vac1, and the soluble N-ethylmaleimide sensitive factor attachment protein receptor (SNARE) fusion machinery, including the Pep1 syntaxinlike SNARE (Horazdovsky et al., 199; Gerrard et al., ; Peterson and Emr, 1; abrera et al., 13). In addition, Vps1- GTP interacts with the hexameric class core vacuole/endosome tethering (ORVET) complex to promote endosome endosome fusion (Peplowska et al., 7; Balderhaar et al., 13; abrera et al., 13). Early endosomes then mature to late endosomes. In this context, endosomal sorting complexes required for transport (ESRT) proteins promote the sorting of membrane proteins into ILVs (Henne et al., 11) and thus profit from multiple endosome endosome fusion events (Zeigerer et al., 1), by which additional surface is generated for this process. onsequently, endosomes are converted into more rounded structures. In parallel, cargo receptors that transport lysosomal hydrolase from the Golgi to the endosome release their cargo and are collected by the retromer complex. This complex consists of the cargo recognition complex (R), consisting of -Vps and Vps9, and the dimeric sorting nexin (SNX) complex, Vps5-Vps17 (Seaman, 1). It is believed that recruitment and activation of the Rab7-like Ypt7 then triggers the formation of endosomal tubules (Rojas et al., ; Seaman et al., 9; Balderhaar et al., 1; Liu et al., 1). The recruitment of Ypt7 depends on its GEF complex, Mon1-cz1 (Nordmann et al., 1; abrera and Ungermann, 13; abrera et al., 1), which functions similarly in higher eukaryotes (Kinchen and Ravichandran, 1; Poteryaev et al., 1; Yousefian et al., 13; ui et al., 1; Singh et al., 1). It is likely that activation of Ypt7 is connected to the amount of Vps1 on early endosomes, suggesting coordinated turnover from one Rab GTPase to the subsequent one (del onte-zerial et al., ; Barr, 13). Even though earlier studies analyzed the morphology of endosomes during cargo uptake (Prescianotto-Baschong and Riezman, 199, ; Griffith and Reggiori, 9), the relative function of the machinery involved in endosomal maturation has not been analyzed in detail. We therefore set out to correlate the spatiotemporal distribution of this machinery with actively transported cargo. Our colocalization data and mutant analyses reveal that endosomal protein complexes function in similar time windows on endosomes but do not sense the absence of the final Rab GTPase Ypt7 or the fusion machinery. RESULTS Trafficking of α-factor through the endocytic pathway To determine the relative localization of proteins involved in endosomal tubulation, fission, and fusion relative to incoming and transported cargo and thus monitor endosomal maturation during this process, we used the α-factor receptor Ste in yeast. This receptor resides in the plasma membrane and binds α-factor, a small peptide pheromone that is secreted by yeast cells with mating type α (Jenness et al., 193; Burkholder and Hartwell, 195; Blumer et al., 19). Binding of the peptide to the receptor triggers its uptake into endocytic vesicles and subsequent trafficking via endosomes to the lytic vacuole for degradation (Singer and Riezman, 199; Schandel and Jenness, 199). The ligand was labeled at a central lysine residue with a linker connected to y5/y7 (Figure 1A) to trace the molecule in the fluorescence microscope without losing its binding capacity toward the receptor (Toshima et al., ; Figure 1A). To analyze protein localization relative to incoming cargo, we first cooled cells to to block endocytosis, followed by addition of labeled α-factor. After removal of unbound ligand, cells were resuspended in medium at 3 to allow endocytosis and subsequent passage through the endocytic pathway (Figure 1B). When cells were heated to 3, the probe was readily taken up and was present in intracellular puncta after 5 min. Further incubation then resulted in labeling of the vacuolar lumen, which became first visible after 15 min (Figure 1, white arrows). After min, all α-factor was quantitatively localized to the vacuolar lumen (Figure 1), in agreement with previous studies (Toshima et al.,, 1). Uptake of α-factor required expression of the Ste receptor (Figure 1), which was endocytosed with its bound ligand (Figure 1E), leaving behind the likely unbound receptor at the plasma membrane (Figure 1E). Furthermore, we compared uptake of α-factor relative to another plasma membrane transporter, Mup1, which is endocytosed upon methionine addition (Menant et al., ), and observed colocalization of both during passage through the endocytic pathway (Figure 1F, white arrows). The stronger plasma membrane staining of Mup1 is likely due to a continuous uptake of this protein compared with the rapid uptake of Ste when α-factor is bound. Our combined data indicate that labeled α-factor migrates with the same kinetics and characteristics as other cargo through the endosomal pathway. Analysis of the spatiotemporal protein localization during endosomal transport To investigate the spatiotemporal membrane localization of proteins involved in endosomal biogenesis during maturation, we chromosomally tagged one gene at a time with green fluorescent protein (GFP) and followed colocalization with y5-labeled α-factor during uptake of the probe. To measure colocalization in a quantitative and unbiased manner, we developed a tool for ImageJ to measure colocalization (Figure A; for details, see Materials and Methods). To analyze all endosomes in a cell at a certain time, we took z-stacks of GFP and y5 in 5-min intervals after cells had been heated to 3 and then used our tool to determine the number of colocalizing structures per cell by segmentation of endosomes in each channel. Structures that showed substantial overlap between the channels were scored as colocalized. Structures that were > nm were removed to exclude vacuolar signals. For each endosomal protein, we counted on average 1 cells/time point to reduce effects due to experimental variations. For our analysis of endosomal maturation, we selected representative proteins for specific fusion and fission reactions at endosomes. These included the Rabs Vps1 and Ypt7 and respective GEFs, the interacting tethering proteins ORVET and Vac1, proteins involved in lipid signaling, such as the Vps3 subunit of the phosphoinositol- 3-kinase Vps3, a FYVE probe to label phosphoinositide-3-phosphate (PI3P) on endosomes, and subunits of the retromer complex, as well as ESRT proteins (Figure B). Furthermore, we verified that all of the tagged constructs remained functional by quantitative measurement of carboxypeptidase Y (PY) transport to the vacuole. Any vps mutant results in a block of vacuolar transport and subsequent secretion from the cells (Figure ). Our analysis results in three important pieces of information. First, we monitor the number of endosomes (as marked by the GFP-tagged protein) over time. Second, we observe the time it takes for α-factor to reach the vacuole, observed by decreasing number of α-factor positive structures at later time points. This may change in certain mutants or during stages of starvation. Finally, we can derive the degree of colocalization of α-factor endosomes with GFP-tagged proteins over time and thus determine the likely activity of the tagged protein relative to the monitored 135 H. Arlt et al. Molecular Biology of the ell
3 A B W-H-W-L-Q-L-K-P-G-Q-P-M-Y G-G-G-NHO(H ) S-y5 PM EE LE/MVB Golgi t Vacuole -y5 Ste 5 min 15 min 5 min 35 min 5 min cargo. This may allow us to monitor sequential localization of endosomal proteins. To test our tool in combination with the α-factor uptake assay, we monitored its colocalization with the endosomal ORVET subunit over time (Figure, F, and Supplemental Movie S1). - GFP is a reproducible endosomal marker and is found in 5 endosomal dots/cell during the entire experiment (Figure E). uring the course of the experiment, α-factor accumulated in dot-like structures already at 5 min, which we counted as endosomes. This amount decreased over time, in agreement with initial endosomal fusion and subsequent uptake into the vacuole (Figure, F). Figure F shows the degree of overlap between - and α-factor positive structures. This graph indicates the peak of colocalization at 15 min, which then steadily decreased as α-factor migrated to the vacuole. The peak is the results of multiple assays and is therefore a reliable measure of endosomal localization during cargo transport. To control specificity of our assay, we monitored α-factor relative to the Golgi protein Mnn9, which was found in at least 1 dots/cell but did not colocalize significantly with α-factor, unlike (Figure, G I). Furthermore, we did not observe any overlap between the Golgi marker Sec7, labeled with mherry, and -GFP, even over shorter time periods (Figure J). MATa min MAT min E Ste 5 min 15 min 5 min 35 min 5 min Ste F Mup1 5 min 15 min 5 min Early endosomal proteins behave similarly during cargo uptake We then began with our analysis of the endosomal pathway by focusing on putative early endosomal proteins. Vps3 is part of the phosphoinositide 3-kinase complex. Like, Vps3 was present in approximately six endosomes and had maximal colocalization at 15 min with the y5-labeled α-factor (Figure 3, A and B). espite some experimental variation, similar results were obtained for the Vac1 tethering protein (Figure 3), Vps1 (Figure 3), its GEF Muk1 (Figure 3E), the GFP-tagged FYVE domain, which monitors PI3P levels in cells (Figure 3F), and the Rab5 homologue Ypt5 (Figure 3G). These data indicate that these endosomal proteins reside on similar endosomal compartments at early stages of endocytic transport. ORVET mediates fusion of early endosomes We next asked whether we could dissect the role of early endosomal proteins and obtain insights into early endosomal fusion processes. For this, we monitored the formation of α-factor positive endosomes directly after endocytosis. After 1 min of the uptake assay, α- factor is completely associated with the plasma membrane, followed by an increase in internal vesicles per cell until 5 min (Figure, A and ) and increased colocalization with (Figure E). Beyond 5 min, these structures coalesce into fewer structures, probably due to fusion of endocytic vesicles to endosomes or endosome endosome Mup1 FIGURE 1: ynamics of α-factor trafficking through the endocytic pathway. (A) Schematic representation of α-factor peptide sequence including a linker at the central lysine for dye labeling. (B) Model of α-factor and Ste trafficking to analyze the endocytic pathway. () Wild-type cells were grown in synthetic medium supplemented with amino acids to logarithmic phase, cooled to to block endocytosis, treated with fluorescent α-factor, washed, and resuspended in medium at 3 to allow uptake of the probe. Indicated time points refer to the time interval after cells were heated to 3. White arrows indicate first appearance of vacuolar y5 staining. () Uptake of α-factor depends on cell mating type. ells were treated as in and analyzed in the microscope directly after washing at. (E) α-factor and its receptor Ste colocalize during passage through the endocytic pathway. ells expressing endogenously tagged Ste-GFP were treated as in and analyzed in the fluorescence microscope. (F) Mup1 and α-y5 are transported through the same endosomes. ells were grown in synthetic medium without methionine to accumulate endogenously expressed Mup1-3xmherry at the plasma membrane. ells were then treated as in and resuspended in medium containing 1 mm methionine at 3 to allow endocytosis and trigger Mup1 uptake. White arrows indicate Mup1 and α-y5 colocalizing endosomes., bright field; scale bar, 5 μm. Volume April 1, 15 ynamics of endosomal machinery 1359
4 A raw deconvolved brightfield B Rab GTPases / GEFs cz1(ypt7 GEF) Vps1(Rab5) Ypt7(Rab7) Muk1(Vps1 GEF) µm z-stack sum projection ell 1: split channels ch1 ch merge ch1 local maxima above threshold ch single cell analysis detect cells 1 colocalized dots dots/channel Tethering / Fusion (ORVET) Vac1(EEA1-homolog) Signal Lipids Vps3(PI3 Kinase omplex) FYVE-domain(PI3P reporter) Endosomal Recycling Vps5(Retromer) Vps1(argo) (Retromer) Snx1 Vps17(Retromer) MVB Sorting / ESRT Vfa1(Vps AAA-ATPase) Vps3(ESRT I) wt vps35 GFP-VPS1 MUK1-GFP Z1-GFP GFP-YPT5 VPS-GFP VA1-GFP VPS3-GFP GFP-FYVE VPS1-GFP VPS17-GFP SNX1-GFP VPS35-GFP VPS3-GFP VFA1-GFP PY Tom 5 min 15 min 5 min 35 min E F... - H 1 1 Mnn I... - Mnn G Mnn9 J Sec7 Mnn9 5 min 15 min 5 min 35 min Sec7 s s s s FIGURE : Spatiotemporal analysis of protein (co)localization on endosomes. (A) Workflow to measure colocalization of α-factor on endosomes. We acquired and deconvolved -μm z-stacks with -nm spacing of the fluorescence channels. A bright-field image of the same image section was used to segment single yeast cells. A sum projection of the deconvolved image stack was used to detect local intensity maxima in each channel in every cell to colocalize and quantify the respective dots per cell (see Materials and Methods). (B) Schematic representation of 1 proteins chosen 13 H. Arlt et al. Molecular Biology of the ell
5 Vps3 A B Vps3 5 min 15 min 5 min 35 min Vps3 Vac Vps1 E Muk1 F PI(3)P G Ypt FIGURE 3: Analysis of proteins involved in early endosomal fusion and PI3P generation. (A) α-factor uptake assay in cells expressing endogenously tagged Vps3-GFP. Experiment was carried out as described in Figure 1 (see Materials and Methods). (B) Quantification of uptake assay shown in A. ( G) Quantification of α-factor uptake assay in cells expressing GFP-tagged Vac1, Muk1, Vps1, Ypt5, and FYVE domain. Scale bars, 5 μm. fusion. To test this hypothesis, we asked whether loss of the endosomal ORVET complex would affect this early stage. ORVET has two Rab-specific subunits, Vps3 and (Peplowska et al., 7; Plemel et al., 11; Epp and Ungermann, 13). In vps3 cells, is still associated with the remaining four subunits and can thus be used as an endosomal marker (Markgraf et al., 9; Ostrowicz et al., 1). In vps3 strains, we observed a similar initial increase in α- factor positive endosomes as in wild type until min (Figure, B and ). However, unlike in wild type, the number of α-factor positive endosomes remained high in vps3 cells (Figure ). Moreover, for analysis of the endocytic pathway, categorized by function. () All tagged constructs are functional. Trafficking of PY was monitored by analysis of internal PY content using antibodies against PY and Tom as loading control on whole-cell extracts. () α-factor uptake assay in cells expressing endogenously tagged VPS-GFP. ells were analyzed as in Figure 1. (E, F) Quantification of α-y5 uptake assay shown in. Endosome number per cell (E) and colocalization of endosomes in the y5 and GFP channels (F) were quantified using the ImageJ plug-in described in A. represents the fraction of y5-labeled α-factor endosomes per cell that are colocalized with the GFP channel or (colocalized endosomes per cell)/(y5-positive endosomes per cell) at the respective time points of the assay. (G) α-factor uptake assay was carried out as in in cells expressing endogenous Mnn9-GFP. (H, I) Quantification of uptake assay shown in G as in E and F. (J) Analysis of dynamic colocalization of endosome and Golgi markers. ells expressing endogenous -GFP and Sec7-mherry were grown as described for the α-uptake assay and analyzed by time-lapse microscopy in -s intervals. Graphs show mean values and SEM., bright field; scale bars, 5 μm. Volume April 1, 15 ynamics of endosomal machinery 131
6 A B 1 min 1 min wt vps3 5 min 5 min 1 min 1 min FIGURE : Analysis of the first 1 min of the endocytic pathway. (A, B) α-factor uptake assay in cells expressing -GFP in the wild type (A) or vps3 mutant (B). Experiment was carried out as in Figure 1, but cells were directly spotted on cover slides for microscopy after washing in cold medium without incubation at 3. The same image section was analyzed for 1 min. ( E) Quantification of the experiment in A and B as in Figure. Graphs show mean values ± SEM. Scale bars, 5 μm. even though the number of -positive endosomes was higher in vps3 cells (Figure ), the colocalization of -GFP and α-factor increased over time in wild-type cells but remained low in vps3 cells (Figure E). These data indicate that ORVET is required to deliver endocytic cargo to -positive endosomes, probably by mediating specific fusion reactions. Analysis of endosomal recycling during maturation In a next set of experiments, we analyzed proteins involved in endosomal membrane recycling relative to α-factor trafficking to the vacuole. onsidering that recycling should follow the initial delivery of other hydrolases to the endosome, such as PY, we expected a delayed overall localization of these proteins relative to early fusion factors. We thus analyzed as a central subunit of the R part of the retromer complex. Unlike the previously analyzed fusion factors, had a clear colocalization peak that was shifted to min (Figure 5, A and B). In contrast, Vps17 as part of the SNX BAR complex of retromer and Snx1 both peaked at 15 min, indicating that their function may be required earlier (Figure 5, and ). We next analyzed Vps1, which transports cargo such as PY from the E endosomes wt vps3 -GFP endosomes vps3 wt wt vps Golgi to the endosome and is subsequently recycled by the retromer complex (ooper and Stevens, 199; Seaman et al., 1997). To reveal the cross-talk between this pathway and endocytosis, we sought the time at which the cargo receptor Vps1 would colocalize with α-factor. Vps1 was found in 1 dots/cell, in agreement with its dual localization on endosomes and the Golgi (hi et al., 1). Of importance, Vps1 colocalized early with α-factor, indicating that it is delivered to early endosomes (Figure 5E). Furthermore, Vps1 colocalization then decreased, probably due to constant removal of Vps1 by retromer and subsequent sorting of α-factor to vacuoles (Figure 5E). We next asked whether removal of retromer would affect the kinetics of α-factor delivery along the endocytic pathway and therefore traced and α-factor in vps cells. However, deletion of retromer did not affect the overall colocalization pattern, indicating that retromer has no major influence on processes controlling endosomal maturation and fusion (Figure 5F). ESRT function begins early in the yeast endocytic pathway ESRT proteins function in four distinct complexes and thus promote formation of ILVs (Henne et al., 11). To analyze ES- RT, we selected one of the few ESRT subunits that is not affected in function by tagging. Vps3, an ESRT-I subunit, was monitored relative to α-factor uptake as before (Figure A). To our surprise, although in agreement with data from metazoan cells (Raiborg et al., ), Vps3 colocalized very early with α-factor and was present in up to endosomal dots/cell (Figure, A and B). onsidering that the number of α-factor positive dots decreases over time (Figure B), such a function is consistent with an early role of ESRT proteins. Another ESRT protein, Vfa1, functions in the context of the Vps AAA-type ATPase (Arlt et al., 11). Vfa1 showed similar behavior in the assay, with a peak at 1 min, indicating that Vps also acts early in the pathway (Figure ). To address the role of ESRTs in endosomal maturation and endocytic trafficking, we deleted VPS from our tester strain (Figure ). In vps cells, endosomes collapse into an aggregated structure with lamellae-like morphology, termed the class E compartment (Babst et al., 1997), which seems to form due to increased Vps1 activity (Russell et al., 1). These structures also accumulate, which only colocalized with α-factor efficiently at later time points, in strong contrast to the wild-type situation (Figure E). However, the class E compartment was already stained with α-factor after 5 min of the assay (Figure, white arrows). We noticed that α-factor localized to the class E compartment even at later time points of the assay (35 min), which might indicate delayed transport through the pathway in this mutant. 13 H. Arlt et al. Molecular Biology of the ell
7 A 5 min 15 min 5 min 35 min B Vps Snx1 E 1 Vps1 F vps FIGURE 5: Mapping of proteins involved in endosomal recycling. (A) α-factor uptake assay in cells expressing endogenous -GFP. Experiment was carried out as in Figure 1 (see Materials and Methods). (B) Quantification of assay shown in A as described in Figure. ( F) Quantification of α-factor uptake assay in cells expressing GFP-tagged Vps17, Snx1, and Vps1 in wild-type cells or -GFP in vps cells. (B F) Mean values ± SEM. Scale bar, 5 μm. A block in endosome vacuole fusion does not affect endosomal maturation Endosomal maturation concludes with the recruitment of the fusion machinery and the fusion of endosomes with vacuoles. uring this process, the Rab7-like Ypt7 protein is activated by the Mon1-cz1 GEF complex and can interact with both retromer and the homotypic fusion and vacuole protein sorting (HOPS) tethering complex (Balderhaar and Ungermann, 13). We thus expected that following the GEF complex or Ypt7 would be indicative of mature late endosomes. When we monitored cz1-gfp relative to endocytosed α-factor, we were surprised to observe a very similar localization pattern to that of, with a peak at 15 min (Figure 7, A and B). However, its substrate, Ypt7, was hardly found on endosomes and localized predominantly to vacuole membranes, which is also reflected by the low colocalization with α-factor over time and the low Ypt7 endosome number per cell (Figure 7). This suggests that Mon1- cz1 might depend on additional activating factors on endosomes to recruit and activate Ypt7. We then asked whether loss of the machinery required for terminal fusion would result in altered trafficking of α-factor through the endocytic pathway. In the absence of the vacuolar SNARE Vam3, uptake of fluorescent α-factor appeared normal in the first min, as reflected by normal decrease of α-factor endosomes/cell (Figure 7, and E). However, fusion was efficiently blocked, as indicated by a plateau of 7 α-factor endosomes/cell at time points later than min and no α-factor localizing to vacuolar structures even after min of the assay (Figure 7, and E). When we then analyzed the progression of endosomal maturation by monitoring colocalization Volume April 1, 15 ynamics of endosomal machinery 133
8 A Vps3 Vps3 5 min 15 min 5 min 35 min B 1 Vps3 Vfa vps 5 min 15 min 5 min 35 min E 1 vps FIGURE : Endosomal localization of proteins involved in ESRT sorting. (A) Uptake assay of labeled α-factor in cells expressing Vps3-GFP. Experiment was carried out as described in Figure 1. (B) Quantification of α-factor uptake assay shown in A as described in Figure. () α-factor uptake assay and quantification of cells expressing Vfa1-GFP. (, E) ells expressing GFP-tagged in vps cells were analyzed as in A and B. White arrows indicate class E compartments at early time points. Graphs show mean values ± SEM; scale bars, 5 μm. with, we observed a colocalization pattern like that in wildtype cells (Figure 7E). Similar results were obtained in mutants lacking the GEF subunit Mon1 (Figure 7E). Vacuoles are strongly fragmented in the vam3 mutant, which could be detected as endosomes with our tool. We thus tested whether there is overlap of vacuoles with α-factor at min of the assay by costaining of vacuoles using 7-amino--chloromethylcoumarin (MA) but did not detect any (Figure 7F). This confirms the block in endosome vacuole fusion, in that α-factor localizes to endosomes even at later time points in this mutant. In sum, our data show that a block in fusion has no effect on upstream maturation events in our assay. Finally, on the basis of our multiple assays, we compared the relative amounts of GFP-positive structures marked by the analyzed proteins, which we interpret as endosomes per cell (Figure A). Of interest, proteins that we scored as early, such as the ESRT-I subunit Vps3, are also present in more structures ( endosomes/cell), whereas proteins involved in late endosomal fusion, such as Ypt7 or cz1, are in comparably few structures ( 3). This indicates that the relative function of proteins in the endocytic pathway approximately correlates with the number of structures decorated by these proteins. Although we saw some variation in the colocalization pattern with endocytosed α-factor, most of the investigated proteins localized in a similar time frame on endosomes (Figure B). To this point, our analysis was based on average colocalization values with a time interval of 5 min. We wondered whether we might have missed faster protein localization dynamics on single endosomes. To investigate this, we tagged the early marker and the late marker 13 H. Arlt et al. Molecular Biology of the ell
9 A cz1 cz1 5 min 15 min 5 min 35 min B cz1 Ypt vam3 5 min 15 min 5 min 35 min E wildtype vam3 mon F MA MA 5 min min FIGURE 7: A block of endosome vacuole fusion does not affect endosomal maturation. (A) α-factor uptake assay in cells expressing cz1-gfp, a subunit of the Ypt7 GEF complex. (B) The experiment in A was quantified as described in Figure. () The assay described in A was carried out in cells expressing GFP-tagged Ypt7. () ells expressing -GFP in a vam3 background were analyzed as in A. (E) Quantification of α-factor uptake assays carried out in cells expressing -GFP in the wild type (left) and vam3 (middle) or mon1 (right) mutant. (F) Uptake assay as in with additional MA staining of the vacuolar lumen (see Materials and Methods). Graphs show mean values ± SEM. Scale bars, 5 μm. with GFP or mherry, respectively, and followed single endosomes over time with a 1- to 15-s time interval. On the basis of our colocalization assay with α-factor, we would predict that and would still substantially overlap on similar endosomal structures (Figure B). Following both endosomal markers over time, we indeed found high colocalization ( 5%) between these proteins (Figure, and ). As a control, we found a similar amount of colocalization between Vps3 and, in agreement with our uptake assay (Figure, B ). When we followed single endosomes, we detected endosomes containing both and over a time of > min (Figure E). This indicates that 1) both proteins can act simultaneously on the same structures and ) early and late endosomes are not separate entities with separate protein profiles, at least not at this level of resolution that our analysis can provide. In addition to these long colocalization events on a single endosome, we also detected homotypic fusion and fission of endosomes with both markers and also heterotypic fission events in which is separated from a - positive structure (Figure, E and F). Volume April 1, 15 ynamics of endosomal machinery 135
10 A 1 1 B GFP dots / cell Vps1 Muk1 cz1 Ypt5 Vps1 Vps3 Ypt5 Vps3 Muk1 Vfa1 Vps1 Vac1 Snx1 Vps17 FYVE cz1 Ypt7 Rab GTPases / GEFs Vac1 Tethering / Fusion Vps3 FYVE Lipid signalling Vps1 Vps17 Snx1 Endosomal Recycling Vps3 Vfa1 with MVB Sorting / ESRT Vps3,5 1 Vps3 Vps3 E F merge i) homotypic fusion 1 3 ii) homotypic fission 1 3 iii) heterotypic fission 15 1 merge 1 t (sec) merge t (sec) 13 H. Arlt et al. Molecular Biology of the ell
11 All of these observations were made on endosomes that were relatively immobile, since they could be traced with a time interval of 1 15 s. However, there was another population of endosomes included in our previous bulk analysis, which was much more mobile. Owing to its mobility, we were not able to follow these single endosomes even with a time interval of.5 s. ISUSSION Based on the trafficking of an endocytosed ligand through the endocytic pathway, our work provides insights into the consecutive function of early and late factors involved in endosomal remodeling and fusion. Surprisingly, almost all endosomal factors analyzed colocalize with the endocytosed probe within a similar time window (Figure B). The only notable exception is the R subcomplex of retromer, which overall seems to come to the endosome later. Our data further show that proteins involved in endosomal fusion and ILV formation such as ESRTs cannot be separated by our assay. This is in agreement with earlier findings on ESRT localization in mammalian cells (Sachse et al., ). It also fits the idea that endosomal fusion is required to generate enough membrane for the generation of intraluminal vesicles. Finally, we demonstrate that impaired fusion of endosomes with vacuoles does not block the kinetics of α-factor through the endocytic pathway. Thus endosomal maturation does not necessarily require activation of Ypt7 or recognition by the terminal vacuole, which agrees with observation on the Rab7 GEF protein Mon1 and cz1 mutants in plants, where endosomal maturation also seemed unperturbed by the mutation (Singh et al., 1). This is surprising, considering that both Mon1 and cz1 localized similarly to relative to endocytosed α-factor, suggesting that recruitment of Mon1-cz1 does not necessarily coincide with full GEF activity. Potentially, Mon1-cz1 is inhibited in its activity until endosomes have reached a degree of maturation that then triggers its activity. Of interest, the subunit of retromer is also a Ypt7 effector. According to our working model, activated Mon1-cz1 would recruit Ypt7, which then promotes recycling before engaging with the HOPS complex in fusion with the vacuole (Bröcker et al., 1; Gautreau et al., 1). How Mon1-cz1 activity is controlled and whether activated Ypt7 might affect the previous Rab Vps1 are then major questions that need to be addressed to understand the molecular basis of endosomal maturation. An intriguing observation of our analysis is the consecutive recruitment of retromer subcomplexes to the endosome. The SNX complex colocalized earlier with α-factor than with the R complex, as monitored by (Figure 5). This situation is similar to observations made in mammalian cells, where the SNX complex localizes to Rab5-positive endosomes and subsequently recruits the R together with Rab7 (Rojas et al., ; van Weering et al., 1). This further implies that also yeast retromer can act in separate subcomplexes, which would be in line with the R being part of several different recycling complexes (Kama et al., 7; Strochlic et al., 7; Harrison et al., 1). The analysis of protein localization relative to incoming labeled α-factor has been nicely used to dissect early steps in endocytosis (Toshima et al., ). It has also been used to monitor the localization of Vps1 relative to cargo, and these data suggested that the AP-3 pathway also intersects with endocytosis (Toshima et al., 1). We would like to note that the analysis of class mutants such as vps1, pep1, and vac1 generally interfere with the functionality of the endocytic pathway, and trafficking thus causes a very general shift in PI3P levels and protein sorting (owles et al., 199; Becherer et al., 199; Peterson et al., 1999; Tall et al., 1999; abrera et al., 13). In agreement, ORVET mutants strongly impaired endosomal number and cargo trafficking (Figure ), indicating that our assay is able to reproduce the previous proposal that ORVET acts in endosomal fusion (abrera et al., 13). One general challenge in our analysis was the consecutive action of endosomal proteins. This was not necessarily expected, as several complexes, such as ORVET and ESRTs, are expected to coincide with function. On the other hand, retromer was separated at least partially from ESRT-I and ORVET. However, at the single-endosome level, and were observed together over long time periods (Figure E). We also saw fusion and fission dynamics on single endosomes, which could not be further investigated. A next improvement of this analysis will therefore be single-endosome tracing, which has been hampered by the high mobility of a subpopulation of endosomes. Single-point analysis has been successfully used to investigate Golgi maturation and trans- Golgi function (Losev et al., ; Matsuura-Tokita et al., ), trans-golgi dynamics (aboussi et al., 1), and early steps of endocytosis (Kaksonen et al., 3; Toshima et al., ; Kukulski et al., 1). Of note, these processes occur within short (1 5 min) time periods, whereas endosomal transport, including endosomal maturation, requires 15 min in yeast. Our data provide a starting point to also extend this analysis to substeps of endosomal maturation. In summary, our colocalization analysis of several endosomal proteins involved in membrane remodeling and fusion highlights the interconnection between these proteins in endosomal trafficking and the independence of endosomal maturation from Rab7/ Ypt7 activation. MATERIALS AN METHOS Yeast strains and plasmids Genetic manipulation in the yeast Saccharomyces cerevisiae was carried out by homologous recombination of PR-amplified cassettes as described (Janke et al., ). Yeast strains are listed in Table 1. The mating type of yeast strain SEY1 was changed to MATa by insertion of ps11, followed by looping out of the plasmid on 5-fluoroorotic acid plates, resulting in strain UY7. Plasmid pu375 (prs NOP1pr-GFP-FYVE) was integrated into strain UY7 to express a GFP-FYVE domain from human EEA1 under control of the weak NOP1 promoter. FIGURE : Model of endosomal maturation in yeast. (A) ata from α-factor uptake assays were used to calculate average number of endosomes per cell with respect to analyzed endosomal proteins. Graphs show mean values ± S. (B) pattern of analyzed endosomal proteins. All colocalization patterns obtained with the α-factor uptake assay were normalized, and a threshold was used to show the time points with most colocalization. () Localization of endogenous -GFP or Vps3-GFP with tagged with 3xmherry. () in was analyzed as in Figure A. Graph shows mean values ± SEM. (E, F) Tracking of single endosomes in cells expressing -GFP and -3xmherry. The z-stacks were acquired every 1 15 s over a time period of min, followed by deconvolution, sum projection, and bleach correction using ImageJ. Endosomes were tracked manually. Volume April 1, 15 ynamics of endosomal machinery 137
12 Strain Genotype Reference SEY1 MAT leu-311 ura3-5 his3- trp- 91 lys-1 suc- 9 Robinson et al. (19) UY7 MATa leu-311 ura3-5 his3- This study trp- 91 lys-1 suc- 9 UY79 UY7, MUP1::3xmherry-hphNT1 This study UY119 UY7, VA1::GFP-TRP1 This study UY1 UY7, YPT7::TRP1-PHO5pr-GFP This study UY11 UY7, VPS35::GFP-TRP1 This study UY13 UY7, MNN9::GFP-TRP1 This study UY1 UY7, VPS17::GFP-HIS3 This study UY15 UY7, SNX1::GFP-TRP1 This study UY17 UY7, VPS3::GFP-TRP1 This study UY13 UY7, VPS::GFP-TRP1 This study UY133 UY7, VFA1::GFP-TRP1 This study UY13 UY7, Z1::GFP-HIS3 This study UY511 UY7, prs NOP1pr-GFP- This study FYVE(hEEA1)::URA3 UY51 UY7, YPT5::URA3-PHO5pr-GFP This study UY5 UY7, VPS1::GFP-TRP1 This study UY57 UY7, STE::GFP-TRP1 This study UY77 UY7, VPS1::URA3-PHO5pr-GFP This study UY11 UY7, VPS35::3xmherry-HIS3 This study VPS::GFP-TRP1 UY1 UY7, VPS35::GFP-hphNT1 This study VPS::3xmherry-HIS3 UY13 UY7, VPS3::GFP-TRP1 This study UY1 UY7, MUK1::GFP-TRP1 This study UY35 UY7, VPS35::GFP-hphNT1 This study vam3 ::kanmx UY37 UY7, VPS::GFP-TRP1 This study vam3 ::kanmx UY5 UY7, VPS::GFP-TRP1 This study vps3 ::natnt UY95 UY7, SE7::GFP-TRP1 This study UY91 UY7, VPS::GFP-TRP1 This study SE7::mherry-hphNT1 UY9337 UY7, VPS3::GFP-TRP1 This study vps3 ::hphnt1 UY91 UY7, VPS3::GFP-TRP1 This study vam3 ::kanmx UY9 UY7, VPS::GFP-TRP1 This study vps ::hphnt1 UY939 UY7, VPS::GFP-TRP1 This study mon1 ::LEU UY955 UY7, vps ::TRP1 VPS::GFP- This study HIS3 UY973 UY7, SLA1::GFP-TRP1 This study UY977 UY7, VPS35::3xmherry-HIS3 VPS3::GFP-TRP1 This study TABLE 1: Strains used in this study. Fluorescence labeling of α-factor and uptake assays The α-factor peptide, including a propionyl-g3 linker at the central lysine residue (LifeTein, Hillsborough, NJ), was labeled with the y5 derivative maleimide-y7 (yomics, Jena, Germany) in HBS buffer (5 mm Nal, 5 mm -(-hydroxyethyl)-1-piperazineethanesulfonic acid/naoh,.1 mm ETA, ph ) for 3 h at room temperature, followed by purification via HPL on a 1 reversed-phase column and analysis by mass spectrometry. For uptake assays, cells were grown in synthetic medium supplemented with amino acids and % glucose at 3 for 1 h to logarithmic phase and an O of... The cells were then washed with cold medium and kept on ice for 15 min, followed by application of.5 μm labeled α-factor for 15 min on ice. The cells were washed extensively with cold medium and then resuspended in medium at 3, which marked the starting point of the assay. The cultures were incubated at 3 on a shaker, and different aliquots of cells were imaged at the respective time points up to min after heating the cells. To analyze early steps of the endocytic pathway, cells were prepared as described and then directly applied without resuspension in warm medium to a glass slide at 3. To analyze Mup1-GFP and α-factor trafficking simultaneously, cells were grown in synthetic medium without methionine as described. After addition of fluorescent α-factor for 15 min, cells were washed with cold synthetic medium with 1 mm methionine and then resuspended in the same medium at 3 to allow endocytosis. Fluorescence microscopy The vacuolar membrane was stained by adding 3 μm FM- for 3 min, followed by washing and incubation in medium without dye for 1 h as described (Vida and Emr, 1995). Staining of vacuoles was done by the addition of.1 MA for 1 min at 3 and subsequent washing with medium. ells were imaged on an Olympus IX-71 inverted microscope equipped with 1 NA 1.9 and NA 1. objectives, a smos camera (PO, Kelheim, Germany), an InsightSSI illumination system,,-diamidino--phenylindole, GFP, mherry, and y5 filters, and SoftWoRx software (Applied Precision, Issaquah, WA). We used -μm z-stacks with -nm spacing for constrained-iterative deconvolution (SoftWoRx) and quantification. Quantification of colocalization in yeast cells Quantification of colocalization was performed in ImageJ (National Institutes of Health, Bethesda, M) by a self-written graphical user interface based on ImageJ built-in routines. Briefly, single cells were segmented from bright-field images, followed by detection of endosome signals on sum projections of deconvolved image stacks. After removal of background signals, a binary image of the endosome signals was obtained by thresholding all pixels within the cell mask with the MaxEntropy threshold (Kapur et al., 195). Signals smaller than one-third of singleendosome signals, as well as signals > nm, were removed from the binary mask to remove noisy background signals or large background signals from the vacuole. Afterward, signal detection and counting were conducted by calculating local maxima for each channel within the binary endosome mask (Figure ). The output was an image containing the local maxima, which were then convolved with an endosome kernel (Airy disk of the reporter channel) to obtain a binary representation of the two channels. After logical conjunction of both representations, which results in the overlapping area, the number of colocalizing endosomes was obtained by counting the number of particles with an area >5% of the reporter Airy disk. 13 H. Arlt et al. Molecular Biology of the ell
13 AKNOWLEGMENTS We thank Frederik Sündermann, Markus Babst, and Lois Weisman for helpful advice and Angela Perz for expert technical support. This work was funded by the Sonderforschungsbereich/SFB 9, Projects P (J.P.) and P11 (.U.)..U. received additional support from the Hans-Mühlenhoff Foundation. REFERENES Arlt H, Perz A, Ungermann (11). An overexpression screen in Saccharomyces cerevisiae identifies novel genes that affect endocytic protein trafficking. Traffic 1, Babst M, Sato TK, Banta LM, Emr S (1997). Endosomal transport function in yeast requires a novel AAA-type ATPase, Vpsp. EMBO J 1, Balderhaar HJK, Arlt H, Ostrowicz W, Bröcker, Sündermann F, Brandt R, Babst M, Ungermann (1). The Rab GTPase Ypt7 is linked to retromer-mediated receptor recycling and fusion at the yeast late endosome. J ell Sci 13, 5 9. Balderhaar HJK, Lachmann J, Yavavli E, Bröcker, Lürick A, Ungermann (13). The ORVET complex promotes tethering and fusion of Rab5/ Vps1-positive membranes. Proc Natl Acad Sci USA 11, Balderhaar HJK, Ungermann (13). ORVET and HOPS tethering complexes coordinators of endosome and lysosome fusion. J ell Sci 1, Barr FA (13). Rab GTPases and membrane identity: causal or inconsequential? J ell Biol, Becherer KA, Rieder SE, Emr S, Jones EW (199). Novel syntaxin homologue, Pep1p, required for the sorting of lumenal hydrolases to the lysosome-like vacuole in yeast. Mol Biol ell 7, Blumer J, Rey J, ehmelt L, Mazel T, Wu YW, Bastiaens P, Goody RS, Itzen A (13). RabGEFs are a major determinant for specific Rab membrane targeting. J ell Biol, 7 3. Blumer KJ, Reneke JE, Thorner J (19). The STE gene product is the ligand-binding component of the -factor receptor of Saccharomyces cerevisiae. J Biol hem 3, Bröcker, Kuhlee A, Gatsogiannis, Kleine Balderhaar HJ, Hönscher, Engelbrecht-Vandré S, Ungermann, Raunser S (1). Molecular architecture of the multisubunit homotypic fusion and vacuole protein sorting (HOPS) tethering complex. Proc Natl Acad Sci USA 19, Burkholder A, Hartwell LH (195). The yeast -factor receptor: structural properties deduced from the sequence of the STE gene. Nucleic Acids Res 13, abrera M, Arlt H, Epp N, Lachmann J, Griffith J, Perz A, Reggiori F, Ungermann (13). Functional separation of endosomal fusion factors and the class core vacuole/endosome tethering (ORVET) complex in endosome biogenesis. J Biol hem, abrera M, Nordmann M, Perz A, Schmedt, Gerondopoulos A, Barr F, Piehler J, Engelbrecht-Vandré S, Ungermann (1). The Mon1- cz1 GEF activates the Rab7 GTPase Ypt7 via a longin-fold-rab interface and association with PI3P-positive membranes. J ell Sci 17, abrera M, Ungermann (13). Guanine nucleotide exchange factors (GEFs) have a critical but not exclusive role in organelle localization of Rab GTPases. J Biol hem, hi RJ, Liu J, West M, Wang J, Odorizzi G, Burd G (1). Fission of SNX- BAR-coated endosomal retrograde transport carriers is promoted by the dynamin-related protein Vps1. J ell Biol, 57. ooper A, Stevens T (199). Vps1p cycles between the late-golgi and prevacuolar compartments in its function as the sorting receptor for multiple yeast vacuolar hydrolases. J ell Biol 133, owles R, Emr S, Horazdovsky (199). Mutations in the VPS5 gene, a SE1 homologue, result in vacuolar protein sorting defects and accumulation of membrane vesicles. J ell Sci 17, ui Y, Zhao Q, Gao, ing Y, Zeng Y, Ueda T, Nakano A, Jiang L (1). Activation of the Rab7 GTPase by the MON1-Z1 complex is essential for PV-to-vacuole trafficking and plant growth in Arabidopsis. Plant ell, 97. aboussi L, ostaguta G, Payne GS (1). Phosphoinositide-mediated clathrin adaptor progression at the trans-golgi network. Nat ell Biol 1, 39. del onte-zerial P, Brusch L, Rink J, ollinet, Kalaidzidis Y, Zerial M, eutsch A (). Membrane identity and GTPase cascades regulated by toggle and cut-out switches. Mol Syst Biol,. Epp N, Ungermann (13). The N-terminal domains of Vps3 and are critical for localization and function of the ORVET tethering complex on endosomes. PLoS One, e737. Gautreau A, Oguievetskaia K, Ungermann (1). Function and regulation of the endosomal fusion and fission machineries. old Spring Harb Perspect Biol, a13. Gerondopoulos A, Langemeyer L, Liang JR, Linford A, Barr FA (1). BLO-3 mutated in Hermansky-Pudlak syndrome is a Rab3/3 guanine nucleotide exchange factor. urr Biol, Gerrard S, Bryant N, Stevens T (). VPS1 controls entry of endocytosed and biosynthetic proteins into the yeast prevacuolar compartment. Mol Biol ell 11, 13. Griffith J, Reggiori F (9). Ultrastructural analysis of nanogold-labeled endocytic compartments of yeast Saccharomyces cerevisiae using a cryosectioning procedure. J Histochem ytochem 57, 1 9. Harrison MS, Hung -S, Liu T-T, hristiano R, Walther T, Burd G (1). A mechanism for retromer endosomal coat complex assembly with cargo. Proc Natl Acad Sci USA 111, 7 7. Henne WM, Buchkovich NJ, Emr S (11). The ESRT pathway. ev ell 1, Horazdovsky B, Busch G, Emr S (199). VPS1 encodes a rab5-like GTP binding protein that is required for the sorting of yeast vacuolar proteins. EMBO J 13, Huotari J, Helenius A (11). Endosome maturation. EMBO J 3, Janke, Magiera M, Rathfelder N, Taxis, Reber S, Maekawa H, Moreno- Borchart A, oenges G, Schwob E, Schiebel E, et al. (). A versatile toolbox for PR-based tagging of yeast genes: new fluorescent proteins, more markers and promoter substitution cassettes. Yeast 1, Jenness, Burkholder A, Hartwell LH (193). Binding of -factor pheromone to yeast a cells: chemical and genetic evidence for an factor receptor. ell 35, Kaksonen M, Sun Y, rubin (3). A pathway for association of receptors, adaptors, and actin during endocytic internalization. ell 115, Kama R, Robinson M, Gerst JE (7). Btn, a Hook1 ortholog and potential Batten disease-related protein, mediates late endosome-golgi protein sorting in yeast. Mol ell Biol 7, 5 1. Kapur JN, Sahoo PK, Wong A (195). A new method for gray-level picture thresholding using the entropy of the histogram. omput Vis Graph Image Process 9, Kinchen JM, Ravichandran KS (1). Identification of two evolutionarily conserved genes regulating processing of engulfed apoptotic cells. Nature, Kukulski W, Schorb M, Kaksonen M, Briggs JAG (1). Plasma membrane reshaping during endocytosis is revealed by time-resolved electron tomography. ell 15, 5 5. Liu T-T, Gomez TS, Sackey BK, Billadeau, Burd G (1). Rab GTPase regulation of retromer-mediated cargo export during endosome maturation. Mol Biol ell 3, Losev E, Reinke, Jellen J, Strongin, Bevis B, Glick B (). Golgi maturation visualized in living yeast. Nature 1, 1 1. Markgraf F, Ahnert F, Arlt H, Mari M, Peplowska K, Epp N, Griffith J, Reggiori F, Ungermann (9). The ORVET subunit cooperates with the Rab5 homolog Vps1 to induce clustering of late endosomal compartments. Mol Biol ell, Matsuura-Tokita K, Takeuchi M, Ichihara A, Mikuriya K, Nakano A (). Live imaging of yeast Golgi cisternal maturation. Nature 1, Menant A, Barbey R, Thomas (). Substrate-mediated remodeling of methionine transport by multiple ubiquitin-dependent mechanisms in yeast cells. EMBO J 5, 3 7. Nordmann M, abrera M, Perz A, Bröcker, Ostrowicz W, Engelbrecht- Vandré S, Ungermann (1). The Mon1-cz1 complex is the GEF of the late endosomal Rab7 homolog Ypt7. urr Biol, Ostrowicz W, Bröcker, Ahnert F, Nordmann M, Lachmann J, Peplowska K, Perz A, Auffarth K, Engelbrecht-Vandré S, Ungermann (1). efined subunit arrangement and rab interactions are required for functionality of the HOPS tethering complex. Traffic 11, Peplowska K, Markgraf F, Ostrowicz W, Bange G, Ungermann (7). The ORVET tethering complex interacts with the yeast Rab5 homolog Vps1 and is involved in endo-lysosomal biogenesis. ev ell 1, Peterson MR, Burd G, Emr S (1999). Vac1p coordinates Rab and phosphatidylinositol 3-kinase signaling in Vps5p-dependent vesicle docking/fusion at the endosome. urr Biol 9, Volume April 1, 15 ynamics of endosomal machinery 139
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins
 13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
Protein Sorting, Intracellular Trafficking, and Vesicular Transport
 Protein Sorting, Intracellular Trafficking, and Vesicular Transport Noemi Polgar, Ph.D. Department of Anatomy, Biochemistry and Physiology Email: polgar@hawaii.edu Phone: 692-1422 Outline Part 1- Trafficking
Protein Sorting, Intracellular Trafficking, and Vesicular Transport Noemi Polgar, Ph.D. Department of Anatomy, Biochemistry and Physiology Email: polgar@hawaii.edu Phone: 692-1422 Outline Part 1- Trafficking
CELB40060 Membrane Trafficking in Animal Cells. Prof. Jeremy C. Simpson. Lecture 2 COPII and export from the ER
 CELB40060 Membrane Trafficking in Animal Cells Prof. Jeremy C. Simpson Lecture 2 COPII and export from the ER Today s lecture... The COPII coat - localisation and subunits Formation of the COPII coat at
CELB40060 Membrane Trafficking in Animal Cells Prof. Jeremy C. Simpson Lecture 2 COPII and export from the ER Today s lecture... The COPII coat - localisation and subunits Formation of the COPII coat at
JBC Papers in Press. Published on December 21, 2012 as Manuscript M
 JBC Papers in Press. Published on December 21, 2012 as Manuscript M112.431536 The latest version is at http://www.jbc.org/cgi/doi/10.1074/jbc.m112.431536 FUNCTIONAL SEPARATION OF ENDOSOMAL FUSION FACTORS
JBC Papers in Press. Published on December 21, 2012 as Manuscript M112.431536 The latest version is at http://www.jbc.org/cgi/doi/10.1074/jbc.m112.431536 FUNCTIONAL SEPARATION OF ENDOSOMAL FUSION FACTORS
Tracking of the dynamic localization of the Rabspecific HOPS subunits reveal their distinct interaction with Ypt7 and vacuoles
 RESEARCH PAPER Cellular Logistics 4, e29191; May 2014; 2014 Landes Bioscience RESEARCH PAPER Tracking of the dynamic localization of the Rabspecific HOPS subunits reveal their distinct interaction with
RESEARCH PAPER Cellular Logistics 4, e29191; May 2014; 2014 Landes Bioscience RESEARCH PAPER Tracking of the dynamic localization of the Rabspecific HOPS subunits reveal their distinct interaction with
Rab function. Introduction. Mini review. Johannes Numrich and Christian Ungermann * Endocytic Rabs in membrane trafficking and signaling
 DOI 10.1515/hsz-2013-0258 Biol. Chem. 2013; x(x): xxx xxx Mini review Johannes Numrich and Christian Ungermann * Endocytic Rabs in membrane trafficking and signaling Abstract: T h e e n d o lys o s o m
DOI 10.1515/hsz-2013-0258 Biol. Chem. 2013; x(x): xxx xxx Mini review Johannes Numrich and Christian Ungermann * Endocytic Rabs in membrane trafficking and signaling Abstract: T h e e n d o lys o s o m
The Rab GTPase Ypt7 is linked to retromer-mediated receptor recycling and fusion at the yeast late endosome
 Research Article 4085 The Rab GTPase Ypt7 is linked to retromer-mediated receptor recycling and fusion at the yeast late endosome Henning J. kleine Balderhaar 1, *, Henning Arlt 1, *, Clemens Ostrowicz
Research Article 4085 The Rab GTPase Ypt7 is linked to retromer-mediated receptor recycling and fusion at the yeast late endosome Henning J. kleine Balderhaar 1, *, Henning Arlt 1, *, Clemens Ostrowicz
Vacuole Biogenesis in Saccharomyces cerevisiae: Protein Transport Pathways to the Yeast Vacuole
 MICROBIOLOGY AND MOLECULAR BIOLOGY REVIEWS, Mar. 1998, p. 230 247 Vol. 62, No. 1 1092-2172/98/$04.00 0 Copyright 1998, American Society for Microbiology Vacuole Biogenesis in Saccharomyces cerevisiae:
MICROBIOLOGY AND MOLECULAR BIOLOGY REVIEWS, Mar. 1998, p. 230 247 Vol. 62, No. 1 1092-2172/98/$04.00 0 Copyright 1998, American Society for Microbiology Vacuole Biogenesis in Saccharomyces cerevisiae:
7.06 Problem Set #4, Spring 2005
 7.06 Problem Set #4, Spring 2005 1. You re doing a mutant hunt in S. cerevisiae (budding yeast), looking for temperaturesensitive mutants that are defective in the cell cycle. You discover a mutant strain
7.06 Problem Set #4, Spring 2005 1. You re doing a mutant hunt in S. cerevisiae (budding yeast), looking for temperaturesensitive mutants that are defective in the cell cycle. You discover a mutant strain
Rab GTPases and tethering in the yeast endocytic pathway
 Extra view Small GTPases 2:3, 182-186; May/June 2011; 2011 Landes Bioscience Rab GTPases and tethering in the yeast endocytic pathway Jens Lachmann, Christian Ungermann and Siegfried Engelbrecht-Vandré*
Extra view Small GTPases 2:3, 182-186; May/June 2011; 2011 Landes Bioscience Rab GTPases and tethering in the yeast endocytic pathway Jens Lachmann, Christian Ungermann and Siegfried Engelbrecht-Vandré*
Heather Currinn, Benjamin Guscott, Zita Balklava, Alice Rothnie and Thomas Wassmer*
 Online Resources APP controls the formation of PI(3,5)P 2 vesicles through its binding of the PIKfyve complex. Cellular and Molecular Life Sciences Heather Currinn, Benjamin Guscott, Zita Balklava, Alice
Online Resources APP controls the formation of PI(3,5)P 2 vesicles through its binding of the PIKfyve complex. Cellular and Molecular Life Sciences Heather Currinn, Benjamin Guscott, Zita Balklava, Alice
Under the Radar Screen: How Bugs Trick Our Immune Defenses
 Under the Radar Screen: How Bugs Trick Our Immune Defenses Session 2: Phagocytosis Marie-Eve Paquet and Gijsbert Grotenbreg Whitehead Institute for Biomedical Research Salmonella Gram negative bacteria
Under the Radar Screen: How Bugs Trick Our Immune Defenses Session 2: Phagocytosis Marie-Eve Paquet and Gijsbert Grotenbreg Whitehead Institute for Biomedical Research Salmonella Gram negative bacteria
!"#$%&'%()*%+*,,%-&,./*%01%02%/*/3452*%3&.26%&4752*,,*1%%
 !"#$%&'%()*%+*,,%-&,./*%01%02%/*/3452*%3&.26%&4752*,,*1%% !"#$%&'(")*++*%,*'-&'./%/,*#01#%-2)#3&)/% 4'(")*++*% % %5"0)%-2)#3&) %%% %67'2#72'*%%%%%%%%%%%%%%%%%%%%%%%4'(")0/./% % 8$+&'&,+"/7 % %,$&7&/9)7$*/0/%%%%%%%%%%
!"#$%&'%()*%+*,,%-&,./*%01%02%/*/3452*%3&.26%&4752*,,*1%% !"#$%&'(")*++*%,*'-&'./%/,*#01#%-2)#3&)/% 4'(")*++*% % %5"0)%-2)#3&) %%% %67'2#72'*%%%%%%%%%%%%%%%%%%%%%%%4'(")0/./% % 8$+&'&,+"/7 % %,$&7&/9)7$*/0/%%%%%%%%%%
Involvement of Specific COPI Subunits in Protein Sorting from the Late Endosome to the Vacuole in Yeast
 MOLECULAR AND CELLULAR BIOLOGY, Jan. 2007, p. 526 540 Vol. 27, No. 2 0270-7306/07/$08.00 0 doi:10.1128/mcb.00577-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Involvement of
MOLECULAR AND CELLULAR BIOLOGY, Jan. 2007, p. 526 540 Vol. 27, No. 2 0270-7306/07/$08.00 0 doi:10.1128/mcb.00577-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Involvement of
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2647 Figure S1 Other Rab GTPases do not co-localize with the ER. a, Cos-7 cells cotransfected with an ER luminal marker (either KDEL-venus or mch-kdel) and mch-tagged human Rab5 (mch-rab5,
DOI: 10.1038/ncb2647 Figure S1 Other Rab GTPases do not co-localize with the ER. a, Cos-7 cells cotransfected with an ER luminal marker (either KDEL-venus or mch-kdel) and mch-tagged human Rab5 (mch-rab5,
Signal Transduction. Dr. Chaidir, Apt
 Signal Transduction Dr. Chaidir, Apt Background Complex unicellular organisms existed on Earth for approximately 2.5 billion years before the first multicellular organisms appeared.this long period for
Signal Transduction Dr. Chaidir, Apt Background Complex unicellular organisms existed on Earth for approximately 2.5 billion years before the first multicellular organisms appeared.this long period for
Importance of Protein sorting. A clue from plastid development
 Importance of Protein sorting Cell organization depend on sorting proteins to their right destination. Cell functions depend on sorting proteins to their right destination. Examples: A. Energy production
Importance of Protein sorting Cell organization depend on sorting proteins to their right destination. Cell functions depend on sorting proteins to their right destination. Examples: A. Energy production
DISCOVERIES OF MACHINERY REGULATING VESICLE TRAFFIC, A MAJOR TRANSPORT SYSTEM IN OUR CELLS. Scientific Background on the Nobel Prize in Medicine 2013
 DISCOVERIES OF MACHINERY REGULATING VESICLE TRAFFIC, A MAJOR TRANSPORT SYSTEM IN OUR CELLS Scientific Background on the Nobel Prize in Medicine 2013 Daniela Scalet 6/12/2013 The Nobel Prize in Medicine
DISCOVERIES OF MACHINERY REGULATING VESICLE TRAFFIC, A MAJOR TRANSPORT SYSTEM IN OUR CELLS Scientific Background on the Nobel Prize in Medicine 2013 Daniela Scalet 6/12/2013 The Nobel Prize in Medicine
Regulation and signaling. Overview. Control of gene expression. Cells need to regulate the amounts of different proteins they express, depending on
 Regulation and signaling Overview Cells need to regulate the amounts of different proteins they express, depending on cell development (skin vs liver cell) cell stage environmental conditions (food, temperature,
Regulation and signaling Overview Cells need to regulate the amounts of different proteins they express, depending on cell development (skin vs liver cell) cell stage environmental conditions (food, temperature,
Subunit organization and Rab interactions of Vps-C protein complexes that control endolysosomal membrane traffic
 MBoC ARTICLE Subunit organization and Rab interactions of Vps-C protein complexes that control endolysosomal membrane traffic Rachael L. Plemel*, Braden T. Lobingier*, Christopher L. Brett, Cortney G.
MBoC ARTICLE Subunit organization and Rab interactions of Vps-C protein complexes that control endolysosomal membrane traffic Rachael L. Plemel*, Braden T. Lobingier*, Christopher L. Brett, Cortney G.
The neuron as a secretory cell
 The neuron as a secretory cell EXOCYTOSIS ENDOCYTOSIS The secretory pathway. Transport and sorting of proteins in the secretory pathway occur as they pass through the Golgi complex before reaching the
The neuron as a secretory cell EXOCYTOSIS ENDOCYTOSIS The secretory pathway. Transport and sorting of proteins in the secretory pathway occur as they pass through the Golgi complex before reaching the
Mapping of Vps21 and HOPS Binding Sites in Vps8 and Effect of Binding Site Mutants on Endocytic Trafficking
 EUKARYOTIC CELL, Apr. 2010, p. 602 610 Vol. 9, No. 4 1535-9778/10/$12.00 doi:10.1128/ec.00286-09 Copyright 2010, American Society for Microbiology. All Rights Reserved. Mapping of Vps21 and HOPS Binding
EUKARYOTIC CELL, Apr. 2010, p. 602 610 Vol. 9, No. 4 1535-9778/10/$12.00 doi:10.1128/ec.00286-09 Copyright 2010, American Society for Microbiology. All Rights Reserved. Mapping of Vps21 and HOPS Binding
Cell Biology Review. The key components of cells that concern us are as follows: 1. Nucleus
 Cell Biology Review Development involves the collective behavior and activities of cells, working together in a coordinated manner to construct an organism. As such, the regulation of development is intimately
Cell Biology Review Development involves the collective behavior and activities of cells, working together in a coordinated manner to construct an organism. As such, the regulation of development is intimately
Antagonistic Roles of ESCRT and Vps Class C/HOPS Complexes in the Recycling of Yeast Membrane Proteins
 Molecular Biology of the Cell Vol. 15, 4203 4214, September 2004 Antagonistic Roles of ESCRT and Vps Class C/HOPS Complexes in the Recycling of Yeast Membrane Proteins Amandine Bugnicourt,* Marine Froissard,*
Molecular Biology of the Cell Vol. 15, 4203 4214, September 2004 Antagonistic Roles of ESCRT and Vps Class C/HOPS Complexes in the Recycling of Yeast Membrane Proteins Amandine Bugnicourt,* Marine Froissard,*
Casein kinase I controls a late step in the endocytic trafficking of yeast uracil permease
 Research Article 217 Casein kinase I controls a late step in the endocytic trafficking of yeast uracil permease Christelle Marchal*, Sophie Dupré* and Daniele Urban-Grimal Institut Jacques Monod, CNRS-UMRC9922,
Research Article 217 Casein kinase I controls a late step in the endocytic trafficking of yeast uracil permease Christelle Marchal*, Sophie Dupré* and Daniele Urban-Grimal Institut Jacques Monod, CNRS-UMRC9922,
2. Yeast two-hybrid system
 2. Yeast two-hybrid system I. Process workflow a. Mating of haploid two-hybrid strains on YPD plates b. Replica-plating of diploids on selective plates c. Two-hydrid experiment plating on selective plates
2. Yeast two-hybrid system I. Process workflow a. Mating of haploid two-hybrid strains on YPD plates b. Replica-plating of diploids on selective plates c. Two-hydrid experiment plating on selective plates
Lecture 6 - Intracellular compartments and transport I
 01.26.11 Lecture 6 - Intracellular compartments and transport I Intracellular transport and compartments 1. Protein sorting: How proteins get to their appropriate destinations within the cell 2. Vesicular
01.26.11 Lecture 6 - Intracellular compartments and transport I Intracellular transport and compartments 1. Protein sorting: How proteins get to their appropriate destinations within the cell 2. Vesicular
Overexpression of the Arabidopsis Syntaxin PEP12/SYP21 Inhibits Transport from the Prevacuolar Compartment tothelyticvacuoleinvivo W
 The Plant Cell, Vol. 18, 2275 2293, September 2006, www.plantcell.org ª 2006 American Society of Plant Biologists Overexpression of the Arabidopsis Syntaxin PEP12/SYP21 Inhibits Transport from the Prevacuolar
The Plant Cell, Vol. 18, 2275 2293, September 2006, www.plantcell.org ª 2006 American Society of Plant Biologists Overexpression of the Arabidopsis Syntaxin PEP12/SYP21 Inhibits Transport from the Prevacuolar
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2215 Figure S1 Number of egfp-vps4a bursts versus cellular expression levels. The total number of egfp-vps4a bursts, counted at the end of each movie (frame 2000, after 1h 28 min) are plotted
DOI: 10.1038/ncb2215 Figure S1 Number of egfp-vps4a bursts versus cellular expression levels. The total number of egfp-vps4a bursts, counted at the end of each movie (frame 2000, after 1h 28 min) are plotted
Name: TF: Section Time: LS1a ICE 5. Practice ICE Version B
 Name: TF: Section Time: LS1a ICE 5 Practice ICE Version B 1. (8 points) In addition to ion channels, certain small molecules can modulate membrane potential. a. (4 points) DNP ( 2,4-dinitrophenol ), as
Name: TF: Section Time: LS1a ICE 5 Practice ICE Version B 1. (8 points) In addition to ion channels, certain small molecules can modulate membrane potential. a. (4 points) DNP ( 2,4-dinitrophenol ), as
Supporting Information
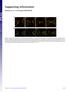 Supporting Information Fleissner et al. 10.1073/pnas.0907039106 Fig. S1. (A) MAK-2-GFP localized to CATs tips is not bound by membrane. his-3::pccg1 mak-2-gfp; mak-2 strain labeled with membrane dye FM4
Supporting Information Fleissner et al. 10.1073/pnas.0907039106 Fig. S1. (A) MAK-2-GFP localized to CATs tips is not bound by membrane. his-3::pccg1 mak-2-gfp; mak-2 strain labeled with membrane dye FM4
Actin-based Motility during Endocytosis in Budding Yeast V
 Molecular Biology of the Cell Vol. 17, 1354 1363, March 2006 Actin-based Motility during Endocytosis in Budding Yeast V Kyoungtae Kim,* Brian J. Galletta,* Kevin O. Schmidt, Fanny S. Chang, Kendall J.
Molecular Biology of the Cell Vol. 17, 1354 1363, March 2006 Actin-based Motility during Endocytosis in Budding Yeast V Kyoungtae Kim,* Brian J. Galletta,* Kevin O. Schmidt, Fanny S. Chang, Kendall J.
Vps41 Phosphorylation and the Rab Ypt7 Control the Targeting of the HOPS Complex to Endosome Vacuole Fusion Sites
 Molecular Biology of the Cell Vol. 20, 1937 1948, April 1, 2009 Vps41 Phosphorylation and the Rab Ypt7 Control the Targeting of the HOPS Complex to Endosome Vacuole Fusion Sites Margarita Cabrera,* Clemens
Molecular Biology of the Cell Vol. 20, 1937 1948, April 1, 2009 Vps41 Phosphorylation and the Rab Ypt7 Control the Targeting of the HOPS Complex to Endosome Vacuole Fusion Sites Margarita Cabrera,* Clemens
Ypt31/32 GTPases and their novel F-Box effector protein Rcy1 regulate protein recycling
 1 Ypt31/32 GTPases and their novel F-Box effector protein Rcy1 regulate protein recycling Shu Hui Chen 1, Shan Chen 1, Andrei A Tokarev, Fengli Liu, Gregory Jedd and Nava Segev 2 Department of Biological
1 Ypt31/32 GTPases and their novel F-Box effector protein Rcy1 regulate protein recycling Shu Hui Chen 1, Shan Chen 1, Andrei A Tokarev, Fengli Liu, Gregory Jedd and Nava Segev 2 Department of Biological
Supplemental Information. The Mitochondrial Fission Receptor MiD51. Requires ADP as a Cofactor
 Structure, Volume 22 Supplemental Information The Mitochondrial Fission Receptor MiD51 Requires ADP as a Cofactor Oliver C. Losón, Raymond Liu, Michael E. Rome, Shuxia Meng, Jens T. Kaiser, Shu-ou Shan,
Structure, Volume 22 Supplemental Information The Mitochondrial Fission Receptor MiD51 Requires ADP as a Cofactor Oliver C. Losón, Raymond Liu, Michael E. Rome, Shuxia Meng, Jens T. Kaiser, Shu-ou Shan,
7.06 Cell Biology EXAM #3 April 21, 2005
 7.06 Cell Biology EXAM #3 April 21, 2005 This is an open book exam, and you are allowed access to books, a calculator, and notes but not computers or any other types of electronic devices. Please write
7.06 Cell Biology EXAM #3 April 21, 2005 This is an open book exam, and you are allowed access to books, a calculator, and notes but not computers or any other types of electronic devices. Please write
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/41480 holds various files of this Leiden University dissertation Author: Tleis, Mohamed Title: Image analysis for gene expression based phenotype characterization
Cover Page The handle http://hdl.handle.net/1887/41480 holds various files of this Leiden University dissertation Author: Tleis, Mohamed Title: Image analysis for gene expression based phenotype characterization
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Breker et al., http://www.jcb.org/cgi/content/full/jcb.201301120/dc1 Figure S1. Single-cell proteomics of stress responses. (a) Using
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Breker et al., http://www.jcb.org/cgi/content/full/jcb.201301120/dc1 Figure S1. Single-cell proteomics of stress responses. (a) Using
downstream (0.8 kb) homologous sequences to the genomic locus of DIC. A DIC mutant strain (ro- 6
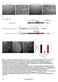 A B C D ts Figure S1 Generation of DIC- mcherry expressing N.crassa strain. A. N. crassa colony morphology. When a cot1 (top, left panel) strain is grown at permissive temperature (25 C), it exhibits straight
A B C D ts Figure S1 Generation of DIC- mcherry expressing N.crassa strain. A. N. crassa colony morphology. When a cot1 (top, left panel) strain is grown at permissive temperature (25 C), it exhibits straight
Lecture 10: Cyclins, cyclin kinases and cell division
 Chem*3560 Lecture 10: Cyclins, cyclin kinases and cell division The eukaryotic cell cycle Actively growing mammalian cells divide roughly every 24 hours, and follow a precise sequence of events know as
Chem*3560 Lecture 10: Cyclins, cyclin kinases and cell division The eukaryotic cell cycle Actively growing mammalian cells divide roughly every 24 hours, and follow a precise sequence of events know as
BE 159: Signal Transduction and Mechanics in Morphogenesis
 BE 159: Signal Transduction and Mechanics in Morphogenesis Justin Bois Caltech Winter, 2018 2018 Justin Bois. This work is licensed under a Creative Commons Attribution License CC-BY 4.0. 5 Delta-Notch
BE 159: Signal Transduction and Mechanics in Morphogenesis Justin Bois Caltech Winter, 2018 2018 Justin Bois. This work is licensed under a Creative Commons Attribution License CC-BY 4.0. 5 Delta-Notch
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. Purification of yeast CKM. (a) Silver-stained SDS-PAGE analysis of CKM purified through a TAP-tag engineered into the Cdk8 C-terminus. (b) Kinase activity
Supplementary Figures Supplementary Figure 1. Purification of yeast CKM. (a) Silver-stained SDS-PAGE analysis of CKM purified through a TAP-tag engineered into the Cdk8 C-terminus. (b) Kinase activity
Rho1 binding site PtdIns(4,5)P2 binding site Both sites
 localization Mutation site DMSO LatB WT F77A I115A I131A K134A Rho1 binding site PtdIns(4,5)P2 binding site Both sites E186A E199A N201A R84A-E186A-E199A L131A-K136A-E186A L131A-E186A-E199A K136A-E186A-E199A
localization Mutation site DMSO LatB WT F77A I115A I131A K134A Rho1 binding site PtdIns(4,5)P2 binding site Both sites E186A E199A N201A R84A-E186A-E199A L131A-K136A-E186A L131A-E186A-E199A K136A-E186A-E199A
Multivesicular Bodies Mature from the Trans-Golgi Network/Early Endosome in Arabidopsis W
 This article is a Plant Cell Advance Online Publication. The date of its first appearance online is the official date of publication. The article has been edited and the authors have corrected proofs,
This article is a Plant Cell Advance Online Publication. The date of its first appearance online is the official date of publication. The article has been edited and the authors have corrected proofs,
Richik N. Ghosh, Linnette Grove, and Oleg Lapets ASSAY and Drug Development Technologies 2004, 2:
 1 3/1/2005 A Quantitative Cell-Based High-Content Screening Assay for the Epidermal Growth Factor Receptor-Specific Activation of Mitogen-Activated Protein Kinase Richik N. Ghosh, Linnette Grove, and Oleg
1 3/1/2005 A Quantitative Cell-Based High-Content Screening Assay for the Epidermal Growth Factor Receptor-Specific Activation of Mitogen-Activated Protein Kinase Richik N. Ghosh, Linnette Grove, and Oleg
Serine-7 but not serine-5 phosphorylation primes RNA polymerase II CTD for P-TEFb recognition
 Supplementary Information to Serine-7 but not serine-5 phosphorylation primes RNA polymerase II CTD for P-TEFb recognition Nadine Czudnochowski 1,2, *, Christian A. Bösken 1, * & Matthias Geyer 1 1 Max-Planck-Institut
Supplementary Information to Serine-7 but not serine-5 phosphorylation primes RNA polymerase II CTD for P-TEFb recognition Nadine Czudnochowski 1,2, *, Christian A. Bösken 1, * & Matthias Geyer 1 1 Max-Planck-Institut
Activation of a receptor. Assembly of the complex
 Activation of a receptor ligand inactive, monomeric active, dimeric When activated by growth factor binding, the growth factor receptor tyrosine kinase phosphorylates the neighboring receptor. Assembly
Activation of a receptor ligand inactive, monomeric active, dimeric When activated by growth factor binding, the growth factor receptor tyrosine kinase phosphorylates the neighboring receptor. Assembly
Way to impose membrane curvature
 Way to impose membrane curvature Sar1 reticulons? clathrin and other vesicle coats dynamin BAR domains McMahon, 2005 The ER is continuous with the nuclear envelope and contains tubules and sheets Gia Voeltz
Way to impose membrane curvature Sar1 reticulons? clathrin and other vesicle coats dynamin BAR domains McMahon, 2005 The ER is continuous with the nuclear envelope and contains tubules and sheets Gia Voeltz
Molecular Cell Biology 5068 In Class Exam 1 September 30, Please print your name:
 Molecular Cell Biology 5068 In Class Exam 1 September 30, 2014 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Molecular Cell Biology 5068 In Class Exam 1 September 30, 2014 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
SUPPLEMENTARY INFORMATION
 med!1,2 Wild-type (N2) end!3 elt!2 5 1 15 Time (minutes) 5 1 15 Time (minutes) med!1,2 end!3 5 1 15 Time (minutes) elt!2 5 1 15 Time (minutes) Supplementary Figure 1: Number of med-1,2, end-3, end-1 and
med!1,2 Wild-type (N2) end!3 elt!2 5 1 15 Time (minutes) 5 1 15 Time (minutes) med!1,2 end!3 5 1 15 Time (minutes) elt!2 5 1 15 Time (minutes) Supplementary Figure 1: Number of med-1,2, end-3, end-1 and
The I-BAR protein Ivy1 is an effector of the Rab7 GTPase Ypt7 involved in vacuole membrane homeostasis
 2015. Published by The Company of Biologists Ltd. The I-BAR protein Ivy1 is an effector of the Rab7 GTPase Ypt7 involved in vacuole membrane homeostasis Johannes Numrich 1, Marie-Pierre Péli-Gulli 2, Henning
2015. Published by The Company of Biologists Ltd. The I-BAR protein Ivy1 is an effector of the Rab7 GTPase Ypt7 involved in vacuole membrane homeostasis Johannes Numrich 1, Marie-Pierre Péli-Gulli 2, Henning
Yeast Genome-wide Screens to Ascertain the Genetic Landscape for Barth Syndrome. Christopher R. McMaster, PhD Dalhousie University
 Yeast Genome-wide Screens to Ascertain the Genetic Landscape for Barth Syndrome Christopher R. McMaster, PhD Dalhousie University Using Systematic Genetics to Identify Modifies Genes that Affect Fitness
Yeast Genome-wide Screens to Ascertain the Genetic Landscape for Barth Syndrome Christopher R. McMaster, PhD Dalhousie University Using Systematic Genetics to Identify Modifies Genes that Affect Fitness
Protein transport from the late Golgi to the vacuole in the yeast Saccharomyces cerevisiae
 Biochimica et Biophysica Acta 1744 (2005) 438 454 Review Protein transport from the late Golgi to the vacuole in the yeast Saccharomyces cerevisiae Katherine Bowers a, Tom H. Stevens b, * a Cambridge Institute
Biochimica et Biophysica Acta 1744 (2005) 438 454 Review Protein transport from the late Golgi to the vacuole in the yeast Saccharomyces cerevisiae Katherine Bowers a, Tom H. Stevens b, * a Cambridge Institute
Chapter 6: A Tour of the Cell
 AP Biology Reading Guide Fred and Theresa Holtzclaw Chapter 6: A Tour of the Cell Name Period Chapter 6: A Tour of the Cell Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry
AP Biology Reading Guide Fred and Theresa Holtzclaw Chapter 6: A Tour of the Cell Name Period Chapter 6: A Tour of the Cell Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry
Supporting online material
 Supporting online material Materials and Methods Target proteins All predicted ORFs in the E. coli genome (1) were downloaded from the Colibri data base (2) (http://genolist.pasteur.fr/colibri/). 737 proteins
Supporting online material Materials and Methods Target proteins All predicted ORFs in the E. coli genome (1) were downloaded from the Colibri data base (2) (http://genolist.pasteur.fr/colibri/). 737 proteins
Translocation through the endoplasmic reticulum membrane. Institute of Biochemistry Benoît Kornmann
 Translocation through the endoplasmic reticulum membrane Institute of Biochemistry Benoît Kornmann Endomembrane system Protein sorting Permeable to proteins but not to ions IgG tetramer (16 nm) Fully hydrated
Translocation through the endoplasmic reticulum membrane Institute of Biochemistry Benoît Kornmann Endomembrane system Protein sorting Permeable to proteins but not to ions IgG tetramer (16 nm) Fully hydrated
Stochastic Model of Maturation and Vesicular Exchange in Cellular Organelles
 Stochastic Model of Maturation and Vesicular Exchange in Cellular Organelles arxiv:1606.01075v2 [physics.bio-ph] 19 Dec 2017 Running title: Maturation and Exchange in Organelles Quentin Vagne and Pierre
Stochastic Model of Maturation and Vesicular Exchange in Cellular Organelles arxiv:1606.01075v2 [physics.bio-ph] 19 Dec 2017 Running title: Maturation and Exchange in Organelles Quentin Vagne and Pierre
Transport between cytosol and nucleus
 of 60 3 Gated trans Lectures 9-15 MBLG 2071 The n GATED TRANSPORT transport between cytoplasm and nucleus (bidirectional) controlled by the nuclear pore complex active transport for macro molecules e.g.
of 60 3 Gated trans Lectures 9-15 MBLG 2071 The n GATED TRANSPORT transport between cytoplasm and nucleus (bidirectional) controlled by the nuclear pore complex active transport for macro molecules e.g.
Structural, Biochemical, and Cell Biological Characterization of Rab7 Mutants That Cause Peripheral Neuropathy
 University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations Spring 5-17-2010 Structural, Biochemical, and Cell Biological Characterization of Rab7 Mutants That Cause Peripheral Neuropathy
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations Spring 5-17-2010 Structural, Biochemical, and Cell Biological Characterization of Rab7 Mutants That Cause Peripheral Neuropathy
Report. Protein Delivery to Vacuole Requires SAND Protein-Dependent Rab GTPase Conversion for MVB-Vacuole Fusion
 Current Biology 24, 1383 1389, June 16, 2014 ª2014 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2014.05.005 Protein Delivery to Vacuole Requires SAND Protein-Dependent Rab GTPase Conversion
Current Biology 24, 1383 1389, June 16, 2014 ª2014 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2014.05.005 Protein Delivery to Vacuole Requires SAND Protein-Dependent Rab GTPase Conversion
HOPS Interacts with Apl5 at the Vacuole Membrane and Is Required for Consumption of AP-3 Transport Vesicles
 Molecular Biology of the Cell Vol. 20, 4563 4574, November 1, 2009 HOPS Interacts with Apl5 at the Vacuole Membrane and Is Required for Consumption of AP-3 Transport Vesicles Cortney G. Angers and Alexey
Molecular Biology of the Cell Vol. 20, 4563 4574, November 1, 2009 HOPS Interacts with Apl5 at the Vacuole Membrane and Is Required for Consumption of AP-3 Transport Vesicles Cortney G. Angers and Alexey
A redundant function for the N-terminal tail of Ndc80 in kinetochore-microtubule interaction in Saccharomyces cerevisiae.
 Genetics: Published Articles Ahead of Print, published on July 30, 2012 as 10.1534/genetics.112.143818 A redundant function for the N-terminal tail of Ndc80 in kinetochore-microtubule interaction in Saccharomyces
Genetics: Published Articles Ahead of Print, published on July 30, 2012 as 10.1534/genetics.112.143818 A redundant function for the N-terminal tail of Ndc80 in kinetochore-microtubule interaction in Saccharomyces
Three Myosins Contribute Uniquely to the Assembly and Constriction of the Fission Yeast Cytokinetic Contractile Ring
 Current Biology Supplemental Information Three Myosins Contribute Uniquely to the Assembly and Constriction of the Fission Yeast Cytokinetic Contractile Ring Caroline Laplante, Julien Berro, Erdem Karatekin,
Current Biology Supplemental Information Three Myosins Contribute Uniquely to the Assembly and Constriction of the Fission Yeast Cytokinetic Contractile Ring Caroline Laplante, Julien Berro, Erdem Karatekin,
Visual pigments. Neuroscience, Biochemistry Dr. Mamoun Ahram Third year, 2019
 Visual pigments Neuroscience, Biochemistry Dr. Mamoun Ahram Third year, 2019 References Webvision: The Organization of the Retina and Visual System (http://www.ncbi.nlm.nih.gov/books/nbk11522/#a 127) The
Visual pigments Neuroscience, Biochemistry Dr. Mamoun Ahram Third year, 2019 References Webvision: The Organization of the Retina and Visual System (http://www.ncbi.nlm.nih.gov/books/nbk11522/#a 127) The
Chapter 4 Active Reading Guide A Tour of the Cell
 Name: AP Biology Mr. Croft Chapter 4 Active Reading Guide A Tour of the Cell Section 1 1. The study of cells has been limited by their small size, and so they were not seen and described until 1665, when
Name: AP Biology Mr. Croft Chapter 4 Active Reading Guide A Tour of the Cell Section 1 1. The study of cells has been limited by their small size, and so they were not seen and described until 1665, when
Biological Process Term Enrichment
 Biological Process Term Enrichment cellular protein localization cellular macromolecule localization intracellular protein transport intracellular transport generation of precursor metabolites and energy
Biological Process Term Enrichment cellular protein localization cellular macromolecule localization intracellular protein transport intracellular transport generation of precursor metabolites and energy
Molecular Cell Biology 5068 In Class Exam 2 November 8, 2016
 Molecular Cell Biology 5068 In Class Exam 2 November 8, 2016 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Molecular Cell Biology 5068 In Class Exam 2 November 8, 2016 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Analysis of nucleotide binding to p97 reveals the properties of a tandem AAA hexameric ATPase
 SUPPLEMENTARY INFORMATION Analysis of nucleotide binding to p97 reveals the properties of a tandem AAA hexameric ATPase Louise C Briggs, Geoff S Baldwin, Non Miyata, Hisao Kondo, Xiaodong Zhang, Paul S
SUPPLEMENTARY INFORMATION Analysis of nucleotide binding to p97 reveals the properties of a tandem AAA hexameric ATPase Louise C Briggs, Geoff S Baldwin, Non Miyata, Hisao Kondo, Xiaodong Zhang, Paul S
Termination of Isoform-Selective Vps21/Rab5 Signaling at Endolysosomal Organelles by Msb3/Gyp3
 Traffic 212; 13: 1411 1428 212 John Wiley & Sons A/S doi:1.1111/j.16-854.212.139.x Termination of Isoform-Selective Vps21/Rab5 Signaling at Endolysosomal Organelles by Msb3/Gyp3 Daniel P. Nickerson 1,
Traffic 212; 13: 1411 1428 212 John Wiley & Sons A/S doi:1.1111/j.16-854.212.139.x Termination of Isoform-Selective Vps21/Rab5 Signaling at Endolysosomal Organelles by Msb3/Gyp3 Daniel P. Nickerson 1,
MOLECULAR CELL BIOLOGY
 1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
SUPPLEMENTARY INFORMATION
 GP2 Type I-piliated bacteria FAE M cell M cell pocket idc T cell mdc Generation of antigenspecific T cells Induction of antigen-specific mucosal immune response Supplementary Figure 1 Schematic diagram
GP2 Type I-piliated bacteria FAE M cell M cell pocket idc T cell mdc Generation of antigenspecific T cells Induction of antigen-specific mucosal immune response Supplementary Figure 1 Schematic diagram
Neurite formation & neuronal polarization. The cytoskeletal components of neurons have characteristic distributions and associations
 Mechanisms of neuronal migration & Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 The cytoskeletal
Mechanisms of neuronal migration & Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 The cytoskeletal
Plant Molecular and Cellular Biology Lecture 8: Mechanisms of Cell Cycle Control and DNA Synthesis Gary Peter
 Plant Molecular and Cellular Biology Lecture 8: Mechanisms of Cell Cycle Control and DNA Synthesis Gary Peter 9/10/2008 1 Learning Objectives Explain why a cell cycle was selected for during evolution
Plant Molecular and Cellular Biology Lecture 8: Mechanisms of Cell Cycle Control and DNA Synthesis Gary Peter 9/10/2008 1 Learning Objectives Explain why a cell cycle was selected for during evolution
4) Please cite Dagda et al J Biol Chem 284: , for any publications or presentations resulting from use or modification of the macro.
 Supplement Figure S1. Algorithmic quantification of mitochondrial morphology in SH- SY5Y cells treated with known fission/fusion mediators. Parental SH-SY5Y cells were transiently transfected with an empty
Supplement Figure S1. Algorithmic quantification of mitochondrial morphology in SH- SY5Y cells treated with known fission/fusion mediators. Parental SH-SY5Y cells were transiently transfected with an empty
Supplementary Figure 1. Biochemical and sequence alignment analyses the
 Supplementary Figure 1. Biochemical and sequence alignment analyses the interaction of OPTN and TBK1. (a) Analytical gel filtration chromatography analysis of the interaction between TBK1 CTD and OPTN(1-119).
Supplementary Figure 1. Biochemical and sequence alignment analyses the interaction of OPTN and TBK1. (a) Analytical gel filtration chromatography analysis of the interaction between TBK1 CTD and OPTN(1-119).
Chapter 12: Intracellular sorting
 Chapter 12: Intracellular sorting Principles of intracellular sorting Principles of intracellular sorting Cells have many distinct compartments (What are they? What do they do?) Specific mechanisms are
Chapter 12: Intracellular sorting Principles of intracellular sorting Principles of intracellular sorting Cells have many distinct compartments (What are they? What do they do?) Specific mechanisms are
Supplementary Figure 1. Phenotype of the HI strain.
 Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Supplementary Information
 Supplementary Information MAP2/Hoechst Hyp.-AP ph 6.5 Hyp.-SD ph 7.2 Norm.-SD ph 7.2 Supplementary Figure 1. Mitochondrial elongation in cortical neurons by acidosis. Representative images of neuronal
Supplementary Information MAP2/Hoechst Hyp.-AP ph 6.5 Hyp.-SD ph 7.2 Norm.-SD ph 7.2 Supplementary Figure 1. Mitochondrial elongation in cortical neurons by acidosis. Representative images of neuronal
Chapter 6: A Tour of the Cell
 Chapter 6: A Tour of the Cell 1. The study of cells has been limited by their small size, and so they were not seen and described until 1665, when Robert Hooke first looked at dead cells from an oak tree.
Chapter 6: A Tour of the Cell 1. The study of cells has been limited by their small size, and so they were not seen and described until 1665, when Robert Hooke first looked at dead cells from an oak tree.
targets. clustering show that different complex pathway
 Supplementary Figure 1. CLICR allows clustering and activation of cytoplasmic protein targets. (a, b) Upon light activation, the Cry2 (red) and LRP6c (green) components co-cluster due to the heterodimeric
Supplementary Figure 1. CLICR allows clustering and activation of cytoplasmic protein targets. (a, b) Upon light activation, the Cry2 (red) and LRP6c (green) components co-cluster due to the heterodimeric
Molecular Biology (9)
 Molecular Biology (9) Translation Mamoun Ahram, PhD Second semester, 2017-2018 1 Resources This lecture Cooper, Ch. 8 (297-319) 2 General information Protein synthesis involves interactions between three
Molecular Biology (9) Translation Mamoun Ahram, PhD Second semester, 2017-2018 1 Resources This lecture Cooper, Ch. 8 (297-319) 2 General information Protein synthesis involves interactions between three
A NOVEL ROLE FOR THE RGS PROTEIN SST2 IN YEAST SIGNALING REVEALED BY MATHEMATICAL MODELING AND LIVE CELL IMAGING. Sai Phanindra Venkatapurapu
 A NOVEL ROLE FOR THE RGS PROTEIN SST2 IN YEAST SIGNALING REVEALED BY MATHEMATICAL MODELING AND LIVE CELL IMAGING Sai Phanindra Venkatapurapu A dissertation submitted to the faculty of the University of
A NOVEL ROLE FOR THE RGS PROTEIN SST2 IN YEAST SIGNALING REVEALED BY MATHEMATICAL MODELING AND LIVE CELL IMAGING Sai Phanindra Venkatapurapu A dissertation submitted to the faculty of the University of
Newly made RNA is called primary transcript and is modified in three ways before leaving the nucleus:
 m Eukaryotic mrna processing Newly made RNA is called primary transcript and is modified in three ways before leaving the nucleus: Cap structure a modified guanine base is added to the 5 end. Poly-A tail
m Eukaryotic mrna processing Newly made RNA is called primary transcript and is modified in three ways before leaving the nucleus: Cap structure a modified guanine base is added to the 5 end. Poly-A tail
The Mon1 Ccz1 GEF activates the Rab7 GTPase Ypt7 via a longin-fold Rab interface and association with PI3Ppositive
 ß 2014. Published by The Company of Biologists Ltd (2014) 127, 1043 1051 doi:10.1242/jcs.140921 RESEARCH ARTICLE The Mon1 Ccz1 GEF activates the Rab7 GTPase Ypt7 via a longin-fold Rab interface and association
ß 2014. Published by The Company of Biologists Ltd (2014) 127, 1043 1051 doi:10.1242/jcs.140921 RESEARCH ARTICLE The Mon1 Ccz1 GEF activates the Rab7 GTPase Ypt7 via a longin-fold Rab interface and association
16 The Cell Cycle. Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization
 The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
7.06 Cell Biology EXAM #3 KEY
 7.06 Cell Biology EXAM #3 KEY May 2, 2006 This is an OPEN BOOK exam, and you are allowed access to books, a calculator, and notes BUT NOT computers or any other types of electronic devices. Please write
7.06 Cell Biology EXAM #3 KEY May 2, 2006 This is an OPEN BOOK exam, and you are allowed access to books, a calculator, and notes BUT NOT computers or any other types of electronic devices. Please write
Supplementary Figure 1. SDS-PAGE analysis of GFP oligomer variants with different linkers. Oligomer mixtures were applied to a PAGE gel containing
 Supplementary Figure 1. SDS-PAGE analysis of GFP oligomer variants with different linkers. Oligomer mixtures were applied to a PAGE gel containing 0.1% SDS without boiling. The gel was analyzed by a fluorescent
Supplementary Figure 1. SDS-PAGE analysis of GFP oligomer variants with different linkers. Oligomer mixtures were applied to a PAGE gel containing 0.1% SDS without boiling. The gel was analyzed by a fluorescent
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): The manuscript by Malli and coworkers reports the successful development and characterization of the first K+ specific FRET-based sensor protein.
Reviewers' comments: Reviewer #1 (Remarks to the Author): The manuscript by Malli and coworkers reports the successful development and characterization of the first K+ specific FRET-based sensor protein.
Tiffany Samaroo MB&B 452a December 8, Take Home Final. Topic 1
 Tiffany Samaroo MB&B 452a December 8, 2003 Take Home Final Topic 1 Prior to 1970, protein and DNA sequence alignment was limited to visual comparison. This was a very tedious process; even proteins with
Tiffany Samaroo MB&B 452a December 8, 2003 Take Home Final Topic 1 Prior to 1970, protein and DNA sequence alignment was limited to visual comparison. This was a very tedious process; even proteins with
Specific Aims: hypothesize
 Specific Aims: Defects in intracellular trafficking of membrane proteins (e.g. ion channels, receptors, transporters) can alter their surface expression and modulate their downstream signaling. Membrane
Specific Aims: Defects in intracellular trafficking of membrane proteins (e.g. ion channels, receptors, transporters) can alter their surface expression and modulate their downstream signaling. Membrane
Supplementary Figure 1 Schematic overview of ASTNs in neuronal migration. (a) Schematic of roles played by ASTNs 1 and 2. ASTN-1-mediated adhesions
 Supplementary Figure 1 Schematic overview of ASTNs in neuronal migration. (a) Schematic of roles played by ASTNs 1 and 2. ASTN-1-mediated adhesions undergo endocytosis into clathrin-coated vesicles dependent
Supplementary Figure 1 Schematic overview of ASTNs in neuronal migration. (a) Schematic of roles played by ASTNs 1 and 2. ASTN-1-mediated adhesions undergo endocytosis into clathrin-coated vesicles dependent
Correct Targeting of Plant ARF GTPases Relies on Distinct Protein Domains
 Traffic 2008; 9: 103 120 Blackwell Munksgaard # 2007 The Authors Journal compilation # 2007 Blackwell Publishing Ltd doi: 10.1111/j.1600-0854.2007.00671.x Correct Targeting of Plant ARF GTPases Relies
Traffic 2008; 9: 103 120 Blackwell Munksgaard # 2007 The Authors Journal compilation # 2007 Blackwell Publishing Ltd doi: 10.1111/j.1600-0854.2007.00671.x Correct Targeting of Plant ARF GTPases Relies
Structural and functional analysis of the HOPS tethering complex at the yeast vacuole
 Structural and functional analysis of the HOPS tethering complex at the yeast vacuole Dissertation zur Erlangung des Grades Doktor der Naturwissenschaften (Dr. rer. nat) eingereicht am Fachbereich Biologie/
Structural and functional analysis of the HOPS tethering complex at the yeast vacuole Dissertation zur Erlangung des Grades Doktor der Naturwissenschaften (Dr. rer. nat) eingereicht am Fachbereich Biologie/
Unit 2: Cells Guided Reading Questions (60 pts total)
 Name: AP Biology Biology, Campbell and Reece, 7th Edition Adapted from chapter reading guides originally created by Lynn Miriello Chapter 6 A Tour of the Cell Unit 2: Cells Guided Reading Questions (60
Name: AP Biology Biology, Campbell and Reece, 7th Edition Adapted from chapter reading guides originally created by Lynn Miriello Chapter 6 A Tour of the Cell Unit 2: Cells Guided Reading Questions (60
Supplementary Figure 1.
 Supplementary Figure 1. Characterisation of IHG-1 overexpressing and knockdown cell lines. (A) Total cellular RNA was prepared from HeLa cells stably overexpressing IHG-1 or mts-ihg-1. IHG-1 mrna was quantified
Supplementary Figure 1. Characterisation of IHG-1 overexpressing and knockdown cell lines. (A) Total cellular RNA was prepared from HeLa cells stably overexpressing IHG-1 or mts-ihg-1. IHG-1 mrna was quantified
Deubiquitination Step in the Endocytic Pathway of Yeast Plasma Membrane Proteins: Crucial Role of Doa4p Ubiquitin Isopeptidase
 MOLECULAR AND CELLULAR BIOLOGY, July 2001, p. 4482 4494 Vol. 21, No. 14 0270-7306/01/$04.00 0 DOI: 10.1128/MCB.21.14.4482 4494.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved.
MOLECULAR AND CELLULAR BIOLOGY, July 2001, p. 4482 4494 Vol. 21, No. 14 0270-7306/01/$04.00 0 DOI: 10.1128/MCB.21.14.4482 4494.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved.
Biology of Fungi. Fungal Structure and Function. Lecture: Structure/Function, Part A BIOL 4848/ Fall Overview of the Hypha
 Biology of Fungi Fungal Structure and Function Overview of the Hypha The hypha is a rigid tube containing cytoplasm Growth occurs at the tips of hyphae Behind the tip, the cell is aging Diagram of hyphal
Biology of Fungi Fungal Structure and Function Overview of the Hypha The hypha is a rigid tube containing cytoplasm Growth occurs at the tips of hyphae Behind the tip, the cell is aging Diagram of hyphal
Supplemental Data. Gao et al. (2012). Plant Cell /tpc
 Supplemental Figure 1. Plant EMP Proteins. (A) The Accession numbers of the 12 EMP members from Arabidopsis. (B) Phylogenetic analysis of EMP proteins from Arabidopsis, human and yeast using the Mac Vector
Supplemental Figure 1. Plant EMP Proteins. (A) The Accession numbers of the 12 EMP members from Arabidopsis. (B) Phylogenetic analysis of EMP proteins from Arabidopsis, human and yeast using the Mac Vector
Overview of Cells. Prokaryotes vs Eukaryotes The Cell Organelles The Endosymbiotic Theory
 Overview of Cells Prokaryotes vs Eukaryotes The Cell Organelles The Endosymbiotic Theory Prokaryotic Cells Archaea Bacteria Come in many different shapes and sizes.5 µm 2 µm, up to 60 µm long Have large
Overview of Cells Prokaryotes vs Eukaryotes The Cell Organelles The Endosymbiotic Theory Prokaryotic Cells Archaea Bacteria Come in many different shapes and sizes.5 µm 2 µm, up to 60 µm long Have large
Three different fusions led to three basic ideas: 1) If one fuses a cell in mitosis with a cell in any other stage of the cell cycle, the chromosomes
 Section Notes The cell division cycle presents an interesting system to study because growth and division must be carefully coordinated. For many cells it is important that it reaches the correct size
Section Notes The cell division cycle presents an interesting system to study because growth and division must be carefully coordinated. For many cells it is important that it reaches the correct size
