Student Questions and Answers November 19, 2002
|
|
|
- Bernard Harmon
- 5 years ago
- Views:
Transcription
1 Student Questions and Answers November 19, 2002 Q 1. Why is the D-glycerol phosphate shuttle used? The malate-aspartate shuttle is better, because there is no energy lost. If there is a problem with the concentration of oxaloacetate, the cell could easily generate more oxaloacetate. Q 2. If the shuttle transport does not depend on energy it would be a perpetuum mobile. How is this possible? Answer: FK: The malate-aspartate shuttle can function effectively only in cells in which the NADH/NAD+ ratio is much higher in the cytosol than in the mitochondrial matrix (a situation usually found in liver, kidney, and heart muscle cells). This simply represents a transporter-facilitated downhill transport of NADH. In the phospho glycerol shuttle part of the free energy is sacrificed to allow even an uphill transport: in brain and skeletal muscle mainly this shuttle is operative, since here usually NADH levels in the cytosol are considerably lower than within the mitochondria. Q 3. Are both shuttle mechanisms present in all cells? Is the malate-aspartate shuttle also used for other purposes, e.g. to transport amino-groups out? Answer: FK: (see above) The shuttle also serves to transport ammonia (from glutamate) from the mitochondria to the cytosol, which, among others, is fed into the urea cycle ( aspartate-argininosuccinate-shunt of the TCA cycle. Q 4. Why is the radical (semiquinone) form of ubiquinone not harmful to the cell? Answer: FK: To some extent it actually is. It is estimated that 1 to 2% of all electrons traveling down the mitochondrial respiratory chain form superoxide, and ubisemiquinone is either directly or indirectly responsible for >80% of them. This value is kept relatively) low by the various binding sites for CoQ in complex I and III, which significantly stabilise the radical semiquinone form, so that most of them will be fully reduced within their lifetime. Q 5. How long does CoQ stay a radical? Wouldn t it react very quickly with the other molecules around? Answer: FK: Unfortunately I couldn t find numbers. Otherwise see above. Q 6. When the oxidised ubiquinone is reduced to the free radical, the electron and the proton are added at different sites of the molecule and therefore forming the radical? And when the semiquinone is further reduced, this addition is the other way round addition of the electron where the proton was added first, and vice versa? Answer: FK: e - 2 H + Q 7. The E for complex I is larger than that for complex III. So are there more protons translocated by complex I than by complex III?
2 Answer: FK: Please see the answer to Q21, Nov 14; the detailed mechanisms of the coupling of electron transfer and proton translocation are not known for complex I and for complex IV, whereas for complex III, the Q-cycle already suggested by Mitchell has been experimentally proven. For both complexes different models have been proposed; most suggest the translocation of 4 protons (per 2 electrons) for complex I, and 2 protons for complex IV (and another 2 protons whih are consumed on the matrix-side by formation of H2O, but this is balanced with 2 protons, which are released to the matrix when the original substrate is oxidised). Q 8. How many ATP synthases are located in the membrane? (How many electron transport chains? How large is the distance between them?) Q 9. Is there a stoichiometry between the different complexes of the e-transport chain? Q 10. Is there the same number of electron transport chains and ATP synthase complexes in the membrane? Answer: FK: (Part of the answers provided in the lecture, see also Q17/Nov 14) In mammalian mitochondria, the stoichiometry of complex I : III : IV is about 1 : 3 : 6. It s not so easy to find numbers, but average figures are ~ 1500 copies of cytochrome oxidase (complex IV) and about 500 copies of F 1 -ATPase (complex V) per µm 2 ; (roughly the same number is given for the corresponding ATPase in chloroplasts, CF 1, while about 1500 complexes of PSI (photosystem I) were detected per µm 2 of the thylakoid membrane). Q 11. Charge of mitochondrial membrane: are these charges just in a small area directly above the membrane or are these charges distributed the whole space of the compartment? Q 12. Do the negative charges within the matrix accumulate along the membrane, or do they diffuse within the matrix, so there is auniform distribution of ions within this compartment? Answer: FK: Membrane potentials arise from unequal charge distributions in the solvent layer immediately adjacent to the membrane. You have to take into account that a membrane potential of ~50 mv means a voltage gradient of >100,000 V/cm (across the inner mitochondrial membrane it is even 300,000 V/cm!), so the respective charges have to sit immediately on the surface of the insulating bilayer. Q 13. Are there numbers of how many protons that have been pumped into the intermembrane space are used for 1) ATP synthesis, or b) symport of pyruvate, Pi, etc.? Answer: FK: Phosphorylation of ADP requires the passage of 3 protons through the F 1 /F 0 -ATPase, but the transport of ATP (out) and ADP + Pi (in) is driven by the proton gradient, which requires the equivalent of another proton per ATP. The total amount of pmf or ATP necessary to drive the various active transport systems depends, of course, of the metabolic situation of the cell, but can amount up to 2 / 3 of the proton gradient formed by electron transport. Q 14. Cytochrome c is associated to the membrane at the inter membrane space-side because of its positively charged sise chains. But the electrochemical potential of the membrane makes this side also positively charged? Answer: FK: (answer provided in the lecture)
3 Q 15. ATP synthase: does the shaft have a special structure that by it s rotation causes the conformational changes in the α and β-subunits? Answer: FK: The γ-subunit of the F0-rotor is asymmetric and induces conformational changes in the F1-part of the ATP synthase: crystal and NMR-structure studies demonstrate that α3β3 alone has perfect 3-fold symmetry, the ATPase activity follows Michaelis-Menten kinetics, and there is no cooperativity; if the γ-subunit is included in this molecule, one of the three β-subunits clearly shows a different conformation than the other two (whereas the conformation of the 3 α-subunits still is nearly identical) and now the high affinity nucleotide binding of one of the 3 sites can be observed. Q 16. Does the proton flow really work like shown in the movie? How are they really handed over? Answer: FK: Sorry, I don t remember exactly how the proton flow was suggested in the video sequence. Typical models generally follow this scenario: protons from the intermembrane space have access to the carboxylate group of the essential aspartate in the transmembrane helix of one of the c-subunits. Protonation of this side chains makes the interaction of this helix with a transmembrane helix of the a-subunit unfavourable, and the c-subunit is therefore displaced by rotation of the entire rotor complex (consisting of e.g. 12 c-subunits). At its new position the proton now can escape to the matrix. Q 17. ATP synthase: what is the function of the α-subunit? (asked twice) Answer: FK: A couple of residues of the α-subunits contribute to substrate binding. Very recently it was also shown that the N-terminus of α-subunits, located at the top of the F 1 hexamer is essential for energy coupling between proton translocation and catalytic activity. This domain appears to be connected to F 0 by OSCP (F 0 subunit conferring sensitivity of the complex to oligomycin), which in turn contacts the central rotary shaft. There are also reports about a heat-shock protection-like function of this subunit (showing some homology to hsp-60 s). Q 18. Are more copies of ATP synthase built into the membrane if more energy is required, or is the number of mitochondria increased? Answer: FK: Obviously the composition of the inner mitochondrial membrane remains largely unchanged under different metabolic conditions. However, under extreme conditions F 1 F 0 -ATP synthase may function in a different way! In the absence of oxygen the most important ATP-dependent process in animal cells is ion pumping by Na/K-ATPase to maintain the membrane potential, and thus membrane integrity. Under these conditions ATP synthase will operate as ATPase, pumping protons from the matrix, in order to maintain the mitochondrial membrane potential. This action is, however, limited by an inhibitory subunit of F 0 (IF1), which blocks the F 1 -part in a phdependent manner. As you suggested in your question, mitochondria are dynamic structures that alter their organization and metabolic capacity in response to changing environmental conditions. Yeast growing on a fermentable carbon source such as glucose has highly reticulated mitochondria comprising ~3% of the cell volume. This fraction increases up to 12% when grown on nonfermentable carbon sources (e.g. ethanol). When cells have entered
4 the stationary phase they show large numbers of separate, small mitochondrial compartments. Under conditions of starvation (or switching from a nonfermentable to a fermentable carbon source) most mitochondria are autophagised by the vacuole. Q 19. How conserved are the sequences of the subunits of ATP synthase in evolution? Answer: FK: The ATP synthases are conserved through evolution. One subfamily comprises the bacterial enzymes and those from mitochondria of animals, plants and fungi, as well as the chloroplast ATPase. There is a second family of closely related enzymes from Archaea, aslo comprising the H + -ATPase found in eukaryotic vacuoles. Surprisingly, the number of c-subunits of the rotary (F 0 ) unit differs between different systems: there are 12 copies in bacterial ATP synthase, 14 in CF 0 from chloroplasts of higher plants, but only 10 in the yeast enzyme. Accordingly, the stoichiometry of protons per ATP varies with system, and not necessarily is an integral number. Q 20. If you had other NDPs (like GDP) instead of ADP, could you synthesise GTP with the ATP synthase (maybe in vitro)? Answer: FK: The substrate specificity of F 1 is rather high; the order of binding strength is ADP GDP>IDP CDP. Q 21. When the concentration of ATP is too high, it is hydrolysed to ADP + Pi, while protons are pumped out. Is there any mechanism to prevent this? Wouldn t it be enough to just shut down ATP synthesis and wait until enough H + have been ejected via the electron transport chain? Answer: FK: Q 22. Is every catalytic centre of the ATP synthase filled with ADP + Pi, or could they also stay empty because no ADP + Pi reaches the centre in time for a new round? Or is a filled reaction centre necessary for rotation (the enzyme somehow recognises that one site is empty and waits until ADP and Pi have bound)? Answer: FK: In experiments with isolated F 1, at low ATP concentrations some of the binding sites remain empty, and the ATPase loses its subunit co-operativity, but the reaction still proceeds at a lower rate. Apparently, this cannot occur in vivo; proton flow requires substrates bound to F1 (a single-residue exchange in subunit b leads to uncoupling of proton flow and ATP synthesis in yeast). Recent studies suggest that the catalytic turnover of ATP synthase leads to a compression of the stator stalk; besides being a mechanism of energy conservation, this could be a condition for further rotation of the stalk. Anyway, due to the large affinity of F 1 for ADP+Pi, and ATP, respectively, empty active sites are very unlikely under normal substrate concentrations. Q 23. In the membrane potential the electrostatic interaction of the counterions acts over a distance of ~ 5 nm (the membrane). So why is it that interactions in polypeptides (e.g. between enzyme and substrate) need a very short distance (~ 0.5 nm) to occur? Answer: FK: The phospholipid bilayer acts as an insulator (low dielectricity constant), so the force attracting opposing charges are much larger than in solution (remember that the d.c. of water is abnormally high, so it is comparatively easy to separate charges in free solution).
5 Q 24. Where does an electrochemical gradient with the inside positively charged occur? Can it be useful for anything? Answer: FK: The electric potential built up by proton pumping across the thylakoid membrane has the opposite orientation: positive at the lumenal side, negative outside. The same is true with endosomes/lysosomes. AS far as cell membranes are concerned, I think the resting potential is always inside negative. Q 25. Which interactions of the rotating shaft with the α- and β-subunits cause the conformational changes (charged side chains, )? Answer: FK: Two of the β-subunits have almost identical conformation (β DP ), the third one (β E ) is clearly different (see Q15 above). In β DP there is strong hydrogen bonding between β-e381 and γ-r242, which is not possible for the β-subunit which faces the back of this part of the γ-subunit. This allows a conformational change of β E, which itself is stabilised by hydrogen bonding involving β-d302, T304, D305 and γ-r268, Q269, and T273 (all numbering for F 1 F 0 -ATP synthase from E. coli). Q 26. Doesn t the rotation of the shaft create a kind of tension in the membrane? Answer: FK: This should not lead to any tension, since membrane proteins and the phospholipid bilayer can move independently. Moving trans-membrane helices within the membrane certainly will require some energy (a number of weak interactions are broken and presumably the same number is formed, but there must be some activation barriere), but this should not be significant at ordinary temperatures. Q 27. Does complex V/ATPase age i.e. does its performance decline with time? Answer: FK: As with all other proteins, there will be some modifications with time (mostly oxidative), some of them detrimental for proper functioning. Unfortunately I can t give you any information about in vivo-turnover of the components of this complex, which you would have to know to judge whether this could be a significant problem or not. Q 28. You mentioned that yeast cells may live without functional electron transport chain. This means that they have to rely on passive transport (diffusion), right? Answer: FK: These mutants ( petite strains ) effectively live anaerobic irrespective of the availability of oxygen, and thus can only grow on fermententable carbon sources. The required amount of ATP will, of course, be spent for active transport. Q 29. How does ADP + Pi bind to the O-side of the ATPase? Answer: FK: The term O does not mean that this site does not bind the reactants at all, it only shows significantly lower binding affinity as compared with state L (see table below). Substrate binding induces the conformational change leading to state L, which by co-operativity facilitates tight substrate binding (state T) in the neighbouring subunit. The rotating shaft then promotes release of the product and state O is re-established.
6 Binding strength Kd (µm) T L O Mg.ATP Mg.ADP Mg.Pi 10,000 > 10,000 > 10,000
20. Electron Transport and Oxidative Phosphorylation
 20. Electron Transport and Oxidative Phosphorylation 20.1 What Role Does Electron Transport Play in Metabolism? Electron transport - Role of oxygen in metabolism as final acceptor of electrons - In inner
20. Electron Transport and Oxidative Phosphorylation 20.1 What Role Does Electron Transport Play in Metabolism? Electron transport - Role of oxygen in metabolism as final acceptor of electrons - In inner
Lectures by Kathleen Fitzpatrick
 Chapter 10 Chemotrophic Energy Metabolism: Aerobic Respiration Lectures by Kathleen Fitzpatrick Simon Fraser University Figure 10-1 Figure 10-6 Conversion of pyruvate The conversion of pyruvate to acetyl
Chapter 10 Chemotrophic Energy Metabolism: Aerobic Respiration Lectures by Kathleen Fitzpatrick Simon Fraser University Figure 10-1 Figure 10-6 Conversion of pyruvate The conversion of pyruvate to acetyl
The Proton Motive Force. Overview. Compartmentalization 11/6/2015. Chapter 21 Stryer Short Course. ATP synthesis Shuttles
 The Proton Motive Force Chapter 21 Stryer Short Course Redox reactions Electron transport chain Proton gradient Overview ATP synthesis Shuttles Analogy: How does burning coal put flour in the grocery store?
The Proton Motive Force Chapter 21 Stryer Short Course Redox reactions Electron transport chain Proton gradient Overview ATP synthesis Shuttles Analogy: How does burning coal put flour in the grocery store?
Life 21 - Aerobic respiration Raven & Johnson Chapter 9 (parts)
 1 Life 21 - Aerobic respiration Raven & Johnson Chapter 9 (parts) Objectives 1: Describe the overall action of the Krebs cycle in generating ATP, NADH and FADH 2 from acetyl-coa 2: Understand the generation
1 Life 21 - Aerobic respiration Raven & Johnson Chapter 9 (parts) Objectives 1: Describe the overall action of the Krebs cycle in generating ATP, NADH and FADH 2 from acetyl-coa 2: Understand the generation
TCA Cycle. Voet Biochemistry 3e John Wiley & Sons, Inc.
 TCA Cycle Voet Biochemistry 3e Voet Biochemistry 3e The Electron Transport System (ETS) and Oxidative Phosphorylation (OxPhos) We have seen that glycolysis, the linking step, and TCA generate a large number
TCA Cycle Voet Biochemistry 3e Voet Biochemistry 3e The Electron Transport System (ETS) and Oxidative Phosphorylation (OxPhos) We have seen that glycolysis, the linking step, and TCA generate a large number
Sara Khraim. Shaymaa Alnamos ... Dr. Nafeth
 10 Sara Khraim Shaymaa Alnamos... Dr. Nafeth *Requirement of oxidative phosphorylation: 1- Source and target for electrons(nadh+fadh2 >> O2). 2- Electron carriers. 3- Enzymes, like oxidoreductases and
10 Sara Khraim Shaymaa Alnamos... Dr. Nafeth *Requirement of oxidative phosphorylation: 1- Source and target for electrons(nadh+fadh2 >> O2). 2- Electron carriers. 3- Enzymes, like oxidoreductases and
Bio102 Problems Photosynthesis
 Bio102 Problems Photosynthesis 1. Why is it advantageous for chloroplasts to have a very large (in surface area) thylakoid membrane contained within the inner membrane? A. This limits the amount of stroma
Bio102 Problems Photosynthesis 1. Why is it advantageous for chloroplasts to have a very large (in surface area) thylakoid membrane contained within the inner membrane? A. This limits the amount of stroma
Cellular Respiration: Harvesting Chemical Energy. 9.1 Catabolic pathways yield energy by oxidizing organic fuels
 Cellular Respiration: Harvesting Chemical Energy 9.1 Catabolic pathways yield energy by oxidizing organic fuels 9.2 Glycolysis harvests chemical energy by oxidizing glucose to pyruvate 9.3 The citric acid
Cellular Respiration: Harvesting Chemical Energy 9.1 Catabolic pathways yield energy by oxidizing organic fuels 9.2 Glycolysis harvests chemical energy by oxidizing glucose to pyruvate 9.3 The citric acid
2015 AP Biology PRETEST Unit 3: Cellular Energetics Week of October
 Name: Class: _ Date: _ 2015 AP Biology PRETEST Unit 3: Cellular Energetics Week of 19-23 October Multiple Choice Identify the choice that best completes the statement or answers the question. 1) Which
Name: Class: _ Date: _ 2015 AP Biology PRETEST Unit 3: Cellular Energetics Week of 19-23 October Multiple Choice Identify the choice that best completes the statement or answers the question. 1) Which
Transporters and Membrane Motors Nov 15, 2007
 BtuB OM vitamin B12 transporter F O F 1 ATP synthase Human multiple drug resistance transporter P-glycoprotein Transporters and Membrane Motors Nov 15, 2007 Transport and membrane motors Concentrations
BtuB OM vitamin B12 transporter F O F 1 ATP synthase Human multiple drug resistance transporter P-glycoprotein Transporters and Membrane Motors Nov 15, 2007 Transport and membrane motors Concentrations
ACTIVE TRANSPORT AND GLUCOSE TRANSPORT. (Chapter 14 and 15, pp and pp )
 ACTIVE TRANSPORT AND GLUCOSE TRANSPORT (Chapter 14 and 15, pp 140-143 and pp 146-151) Overview Active transport is the movement of molecules across a cell membrane in the direction against their concentration
ACTIVE TRANSPORT AND GLUCOSE TRANSPORT (Chapter 14 and 15, pp 140-143 and pp 146-151) Overview Active transport is the movement of molecules across a cell membrane in the direction against their concentration
AP Bio-Ms.Bell Unit#3 Cellular Energies Name
 AP Bio-Ms.Bell Unit#3 Cellular Energies Name 1. Base your answer to the following question on the image below. 7. Base your answer to the following question on Which of the following choices correctly
AP Bio-Ms.Bell Unit#3 Cellular Energies Name 1. Base your answer to the following question on the image below. 7. Base your answer to the following question on Which of the following choices correctly
ETC/CHEMIOSIS. By: Leslie, Kelsey, Morgan
 ETC/CHEMIOSIS By: Leslie, Kelsey, Morgan WHY THIS IS IMPORTANT House Clip SO3E7 The Son of a Coma Guy- Time: 32:00 Patient was visiting his father who was in a vegetative state for 10 years, and his only
ETC/CHEMIOSIS By: Leslie, Kelsey, Morgan WHY THIS IS IMPORTANT House Clip SO3E7 The Son of a Coma Guy- Time: 32:00 Patient was visiting his father who was in a vegetative state for 10 years, and his only
Biochemical bases for energy transformations. Biochemical bases for energy transformations. Nutrition 202 Animal Energetics R. D.
 Biochemical bases for energy transformations Biochemical bases for energy transformations Nutrition 202 Animal Energetics R. D. Sainz Lecture 02 Energy originally from radiant sun energy Captured in chemical
Biochemical bases for energy transformations Biochemical bases for energy transformations Nutrition 202 Animal Energetics R. D. Sainz Lecture 02 Energy originally from radiant sun energy Captured in chemical
Photosynthesis and Cellular Respiration Note-taking Guide
 Photosynthesis and Cellular Respiration Note-taking Guide Preview to Photosynthesis glucose, reactions, light-dependent, Calvin cycle, thylakoid, photosystem II, oxygen, light-harvesting, two, chloroplasts,
Photosynthesis and Cellular Respiration Note-taking Guide Preview to Photosynthesis glucose, reactions, light-dependent, Calvin cycle, thylakoid, photosystem II, oxygen, light-harvesting, two, chloroplasts,
Lecture Series 9 Cellular Pathways That Harvest Chemical Energy
 Lecture Series 9 Cellular Pathways That Harvest Chemical Energy Reading Assignments Review Chapter 3 Energy, Catalysis, & Biosynthesis Read Chapter 13 How Cells obtain Energy from Food Read Chapter 14
Lecture Series 9 Cellular Pathways That Harvest Chemical Energy Reading Assignments Review Chapter 3 Energy, Catalysis, & Biosynthesis Read Chapter 13 How Cells obtain Energy from Food Read Chapter 14
Photosynthesis. Chapter 10. Active Lecture Questions for use with Classroom Response Systems Biology, Seventh Edition Neil Campbell and Jane Reece
 Chapter 10 Photosynthesis Active Lecture Questions for use with Classroom Response Systems Biology, Seventh Edition Neil Campbell and Jane Reece Edited by William Wischusen, Louisiana State University
Chapter 10 Photosynthesis Active Lecture Questions for use with Classroom Response Systems Biology, Seventh Edition Neil Campbell and Jane Reece Edited by William Wischusen, Louisiana State University
Photosynthesis and Cellular Respiration Note-taking Guide
 Photosynthesis and Cellular Respiration Note-taking Guide Preview to Photosynthesis glucose, reectlons, light-dependent, Calvin cycle, thylakoid, oxygen, light-harvesting, two, chloroplasts, photosynthesis,
Photosynthesis and Cellular Respiration Note-taking Guide Preview to Photosynthesis glucose, reectlons, light-dependent, Calvin cycle, thylakoid, oxygen, light-harvesting, two, chloroplasts, photosynthesis,
f) Adding an enzyme does not change the Gibbs free energy. It only increases the rate of the reaction by lowering the activation energy.
 Problem Set 2-Answer Key BILD1 SP16 1) How does an enzyme catalyze a chemical reaction? Define the terms and substrate and active site. An enzyme lowers the energy of activation so the reaction proceeds
Problem Set 2-Answer Key BILD1 SP16 1) How does an enzyme catalyze a chemical reaction? Define the terms and substrate and active site. An enzyme lowers the energy of activation so the reaction proceeds
Biophysics 490M Project
 Biophysics 490M Project Dan Han Department of Biochemistry Structure Exploration of aa 3 -type Cytochrome c Oxidase from Rhodobacter sphaeroides I. Introduction: All organisms need energy to live. They
Biophysics 490M Project Dan Han Department of Biochemistry Structure Exploration of aa 3 -type Cytochrome c Oxidase from Rhodobacter sphaeroides I. Introduction: All organisms need energy to live. They
Membranes 2: Transportation
 Membranes 2: Transportation Steven E. Massey, Ph.D. Associate Professor Bioinformatics Department of Biology University of Puerto Rico Río Piedras Office & Lab: NCN#343B Tel: 787-764-0000 ext. 7798 E-mail:
Membranes 2: Transportation Steven E. Massey, Ph.D. Associate Professor Bioinformatics Department of Biology University of Puerto Rico Río Piedras Office & Lab: NCN#343B Tel: 787-764-0000 ext. 7798 E-mail:
Edexcel (B) Biology A-level
 Edexcel (B) Biology A-level Topic 5: Energy for Biological Processes Notes Aerobic Respiration Aerobic respiration as splitting of the respiratory substrate, to release carbon dioxide as a waste product
Edexcel (B) Biology A-level Topic 5: Energy for Biological Processes Notes Aerobic Respiration Aerobic respiration as splitting of the respiratory substrate, to release carbon dioxide as a waste product
Scale in the biological world
 Scale in the biological world 2 A cell seen by TEM 3 4 From living cells to atoms 5 Compartmentalisation in the cell: internal membranes and the cytosol 6 The Origin of mitochondria: The endosymbion hypothesis
Scale in the biological world 2 A cell seen by TEM 3 4 From living cells to atoms 5 Compartmentalisation in the cell: internal membranes and the cytosol 6 The Origin of mitochondria: The endosymbion hypothesis
Biological Chemistry and Metabolic Pathways
 Biological Chemistry and Metabolic Pathways 1. Reaction a. Thermodynamics b. Kinetics 2. Enzyme a. Structure and Function b. Regulation of Activity c. Kinetics d. Inhibition 3. Metabolic Pathways a. REDOX
Biological Chemistry and Metabolic Pathways 1. Reaction a. Thermodynamics b. Kinetics 2. Enzyme a. Structure and Function b. Regulation of Activity c. Kinetics d. Inhibition 3. Metabolic Pathways a. REDOX
All organisms require a constant expenditure of energy to maintain the living state - "LIFE".
 CELLULAR RESPIRATION All organisms require a constant expenditure of energy to maintain the living state - "LIFE". Where does the energy come from and how is it made available for life? With rare exception,
CELLULAR RESPIRATION All organisms require a constant expenditure of energy to maintain the living state - "LIFE". Where does the energy come from and how is it made available for life? With rare exception,
Energy Exchanges Exam: What to Study
 Energy Exchanges Exam: What to Study Here s what you will need to make sure you understand in order to prepare for our exam: Free Energy Conceptual understanding of free energy as available energy in a
Energy Exchanges Exam: What to Study Here s what you will need to make sure you understand in order to prepare for our exam: Free Energy Conceptual understanding of free energy as available energy in a
Cellular Respiration Stage 4: Electron Transport Chain
 Cellular Respiration Stage 4: Electron Transport Chain 2006-2007 Cellular respiration What s the point? The point is to make ATP! ATP 2006-2007 ATP accounting so far Glycolysis 2 ATP Kreb s cycle 2 ATP
Cellular Respiration Stage 4: Electron Transport Chain 2006-2007 Cellular respiration What s the point? The point is to make ATP! ATP 2006-2007 ATP accounting so far Glycolysis 2 ATP Kreb s cycle 2 ATP
Metabolism. Fermentation vs. Respiration. End products of fermentations are waste products and not fully.
 Outline: Metabolism Part I: Fermentations Part II: Respiration Part III: Metabolic Diversity Learning objectives are: Learn about respiratory metabolism, ATP generation by respiration linked (oxidative)
Outline: Metabolism Part I: Fermentations Part II: Respiration Part III: Metabolic Diversity Learning objectives are: Learn about respiratory metabolism, ATP generation by respiration linked (oxidative)
Photosynthesis and Cellular Respiration Practice Test Name
 Photosynthesis and Cellular Respiration Practice Test Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which H+ has just passed through the
Photosynthesis and Cellular Respiration Practice Test Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which H+ has just passed through the
b) What is the gradient at room temperature? Du = J/molK * 298 K * ln (1/1000) = kj/mol
 Chem350 Practice Problems Membranes 1. a) What is the chemical potential generated by the movement of glucose by passive diffusion established by a 1000 fold concentration gradient at physiological temperature?
Chem350 Practice Problems Membranes 1. a) What is the chemical potential generated by the movement of glucose by passive diffusion established by a 1000 fold concentration gradient at physiological temperature?
Energy in the World of Life
 Cellular Energy Energy in the World of Life Sustaining life s organization requires ongoing energy inputs Assembly of the molecules of life starts with energy input into living cells Energy Conversion
Cellular Energy Energy in the World of Life Sustaining life s organization requires ongoing energy inputs Assembly of the molecules of life starts with energy input into living cells Energy Conversion
MITOCW watch?v=0xajihttcns
 MITOCW watch?v=0xajihttcns The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high quality educational resources for free. To
MITOCW watch?v=0xajihttcns The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high quality educational resources for free. To
Giving you the energy you need!
 Giving you the energy you need! Use your dominant hand Open and close the pin (with your thumb and forefinger) as many times as you can for 20 seconds while holding the other fingers straight out! Repeat
Giving you the energy you need! Use your dominant hand Open and close the pin (with your thumb and forefinger) as many times as you can for 20 seconds while holding the other fingers straight out! Repeat
2. In regards to the fluid mosaic model, which of the following is TRUE?
 General Biology: Exam I Sample Questions 1. How many electrons are required to fill the valence shell of a neutral atom with an atomic number of 24? a. 0 the atom is inert b. 1 c. 2 d. 4 e. 6 2. In regards
General Biology: Exam I Sample Questions 1. How many electrons are required to fill the valence shell of a neutral atom with an atomic number of 24? a. 0 the atom is inert b. 1 c. 2 d. 4 e. 6 2. In regards
Energy and Cells. Appendix 1. The two primary energy transformations in plants are photosynthesis and respiration.
 Energy and Cells Appendix 1 Energy transformations play a key role in all physical and chemical processes that occur in plants. Energy by itself is insufficient to drive plant growth and development. Enzymes
Energy and Cells Appendix 1 Energy transformations play a key role in all physical and chemical processes that occur in plants. Energy by itself is insufficient to drive plant growth and development. Enzymes
number Done by Corrected by Doctor Nafeth Abu Tarboush
 number 8 Done by Ali Yaghi Corrected by Mamoon Mohamad Alqtamin Doctor Nafeth Abu Tarboush 0 P a g e Oxidative phosphorylation Oxidative phosphorylation has 3 major aspects: 1. It involves flow of electrons
number 8 Done by Ali Yaghi Corrected by Mamoon Mohamad Alqtamin Doctor Nafeth Abu Tarboush 0 P a g e Oxidative phosphorylation Oxidative phosphorylation has 3 major aspects: 1. It involves flow of electrons
A + B = C C + D = E E + F = A
 Photosynthesis - Plants obtain energy directly from the sun - Organisms that do this are autotrophs (make their own food from inorganic forms) - Photosynthesis is a series of chemical reactions where the
Photosynthesis - Plants obtain energy directly from the sun - Organisms that do this are autotrophs (make their own food from inorganic forms) - Photosynthesis is a series of chemical reactions where the
Cellular Energetics. Photosynthesis, Cellular Respiration and Fermentation
 Cellular Energetics Photosynthesis, Cellular Respiration and Fermentation TEKS B.4 Science concepts. The student knows that cells are the basic structures of all living things with specialized parts that
Cellular Energetics Photosynthesis, Cellular Respiration and Fermentation TEKS B.4 Science concepts. The student knows that cells are the basic structures of all living things with specialized parts that
Metabolism Test D [50 marks]
![Metabolism Test D [50 marks] Metabolism Test D [50 marks]](/thumbs/95/123302288.jpg) Metabolism Test D [50 marks] 1. A cricket was placed in a respirometer at constant temperature for ten minutes. The soap bubble moved along the pipette. [Source: International Baccalaureate Organization
Metabolism Test D [50 marks] 1. A cricket was placed in a respirometer at constant temperature for ten minutes. The soap bubble moved along the pipette. [Source: International Baccalaureate Organization
Unit 5 Cellular Energy
 Unit 5 Cellular Energy I. Enzymes (159) 1.Are CATALYSTS: Speed up chemical reactions that would otherwise happen too slowly to support life. Catalysts DO NOT make reactions happen that couldn t happen
Unit 5 Cellular Energy I. Enzymes (159) 1.Are CATALYSTS: Speed up chemical reactions that would otherwise happen too slowly to support life. Catalysts DO NOT make reactions happen that couldn t happen
Cellular respiration. How do living things stay alive? Cellular Respiration Burning. Photosynthesis. Cellular Respiration
 How do living things stay alive? Cellular Respiration Burning Happens in ALL living things inside cells and has the main goal of producing ATP the fuel of life It does not matter whether the organisms
How do living things stay alive? Cellular Respiration Burning Happens in ALL living things inside cells and has the main goal of producing ATP the fuel of life It does not matter whether the organisms
State state describe
 Warm-Up State the products of the light-dependent reaction of photosynthesis, state which product has chemical energy, and describe how that product is made. KREBS ETC FADH 2 Glucose Pyruvate H 2 O NADH
Warm-Up State the products of the light-dependent reaction of photosynthesis, state which product has chemical energy, and describe how that product is made. KREBS ETC FADH 2 Glucose Pyruvate H 2 O NADH
Cellular Respiration. Mitochondria Rule! Mr. Kurt Kristensen
 Cellular Respiration Mitochondria Rule! Mr. Kurt Kristensen Harvard Biovisions Mitochondria Summer Session Week 1: Cellular Respiration Students should. 1) Understand the locations, and functions of the
Cellular Respiration Mitochondria Rule! Mr. Kurt Kristensen Harvard Biovisions Mitochondria Summer Session Week 1: Cellular Respiration Students should. 1) Understand the locations, and functions of the
Lecture 7 Cell Biolog y ٢٢٢ ١
 Lecture 7 ١ Mitochondria ٢ Mitochondria Mitochondria are the energy factories of the cells. The energy currency for the work that animals must do is the energy-rich molecule adenosine triphosphate (ATP).
Lecture 7 ١ Mitochondria ٢ Mitochondria Mitochondria are the energy factories of the cells. The energy currency for the work that animals must do is the energy-rich molecule adenosine triphosphate (ATP).
MitoSeminar II: Some calculations in bioenergetics
 MitoSeminar II: Some calculations in bioenergetics MUDr. Jan Pláteník, PhD. Ústav lékařské biochemie 1.LF UK Helpful comments of Prof. MUDr. Jiří Kraml, DrSc., are acknowledged. 1 Respiratory chain and
MitoSeminar II: Some calculations in bioenergetics MUDr. Jan Pláteník, PhD. Ústav lékařské biochemie 1.LF UK Helpful comments of Prof. MUDr. Jiří Kraml, DrSc., are acknowledged. 1 Respiratory chain and
2.A.2- Capture and Storage of Free Energy
 2.A.2- Capture and Storage of Free Energy Big Idea 2: Biological systems utilize free energy and molecular building blocks to grow, to reproduce, and to maintain dynamic homeostasis. EU 2.A- Growth, reproduction
2.A.2- Capture and Storage of Free Energy Big Idea 2: Biological systems utilize free energy and molecular building blocks to grow, to reproduce, and to maintain dynamic homeostasis. EU 2.A- Growth, reproduction
Advanced Higher Biology. Unit 1- Cells and Proteins 2c) Membrane Proteins
 Advanced Higher Biology Unit 1- Cells and Proteins 2c) Membrane Proteins Membrane Structure Phospholipid bilayer Transmembrane protein Integral protein Movement of Molecules Across Membranes Phospholipid
Advanced Higher Biology Unit 1- Cells and Proteins 2c) Membrane Proteins Membrane Structure Phospholipid bilayer Transmembrane protein Integral protein Movement of Molecules Across Membranes Phospholipid
CHLOROPLASTS, CALVIN CYCLE, PHOTOSYNTHETIC ELECTRON TRANSFER AND PHOTOPHOSPHORYLATION (based on Chapter 19 and 20 of Stryer )
 CHLOROPLASTS, CALVIN CYCLE, PHOTOSYNTHETIC ELECTRON TRANSFER AND PHOTOPHOSPHORYLATION (based on Chapter 19 and 20 of Stryer ) Photosynthesis Photosynthesis Light driven transfer of electron across a membrane
CHLOROPLASTS, CALVIN CYCLE, PHOTOSYNTHETIC ELECTRON TRANSFER AND PHOTOPHOSPHORYLATION (based on Chapter 19 and 20 of Stryer ) Photosynthesis Photosynthesis Light driven transfer of electron across a membrane
Unit 3: Cell Energy Guided Notes
 Enzymes Unit 3: Cell Energy Guided Notes 1 We get energy from the food we eat by breaking apart the chemical bonds where food is stored. energy is in the bonds, energy is the energy we use to do things.
Enzymes Unit 3: Cell Energy Guided Notes 1 We get energy from the food we eat by breaking apart the chemical bonds where food is stored. energy is in the bonds, energy is the energy we use to do things.
AHL Topic 8 IB Biology Miss Werba
 CELL RESPIRATION & PHOTOSYNTHESIS AHL Topic 8 IB Biology Miss Werba TOPIC 8 CELL RESPIRATION & PHOTOSYNTHESIS 8.1 CELL RESPIRATION 1. STATE that oxidation involves the loss of electrons from an element,
CELL RESPIRATION & PHOTOSYNTHESIS AHL Topic 8 IB Biology Miss Werba TOPIC 8 CELL RESPIRATION & PHOTOSYNTHESIS 8.1 CELL RESPIRATION 1. STATE that oxidation involves the loss of electrons from an element,
METABOLISM. What is metabolism? Categories of metabolic reactions. Total of all chemical reactions occurring within the body
 METABOLISM What is metabolism? METABOLISM Total of all chemical reactions occurring within the body Categories of metabolic reactions Catabolic reactions Degradation pathways Anabolic reactions Synthesis
METABOLISM What is metabolism? METABOLISM Total of all chemical reactions occurring within the body Categories of metabolic reactions Catabolic reactions Degradation pathways Anabolic reactions Synthesis
Aerobic Cellular Respiration
 Aerobic Cellular Respiration Under aerobic conditions (oxygen gas is available), cells will undergo aerobic cellular respiration. The end products of aerobic cellular respiration are carbon dioxide gas,
Aerobic Cellular Respiration Under aerobic conditions (oxygen gas is available), cells will undergo aerobic cellular respiration. The end products of aerobic cellular respiration are carbon dioxide gas,
Photosynthesis and Cellular Respiration
 Photosynthesis and Cellular Respiration Photosynthesis and Cellular Respiration All cellular activities require energy. Directly or indirectly nearly all energy for life comes from the sun. Autotrophs:
Photosynthesis and Cellular Respiration Photosynthesis and Cellular Respiration All cellular activities require energy. Directly or indirectly nearly all energy for life comes from the sun. Autotrophs:
AP Biology Cellular Respiration
 AP Biology Cellular Respiration The bonds between H and C represents a shared pair of electrons These are high-energy electrons This represents chemical potential energy Hydro-carbons posses a lot of chemical
AP Biology Cellular Respiration The bonds between H and C represents a shared pair of electrons These are high-energy electrons This represents chemical potential energy Hydro-carbons posses a lot of chemical
Chapter 10. Thermodynamics of Transport. Thermodynamics of Transport, con t. BCH 4053 Summer 2001 Chapter 10 Lecture Notes. Slide 1.
 BCH 4053 Summer 2001 Chapter 10 Lecture Notes 1 Chapter 10 Membrane Transport 2 3 Thermodynamics of Transport Free Energy change is given by difference in electrochemical potential and the quantity transported
BCH 4053 Summer 2001 Chapter 10 Lecture Notes 1 Chapter 10 Membrane Transport 2 3 Thermodynamics of Transport Free Energy change is given by difference in electrochemical potential and the quantity transported
Lecture 10. Proton Gradient-dependent ATP Synthesis. Oxidative. Photo-Phosphorylation
 Lecture 10 Proton Gradient-dependent ATP Synthesis Oxidative Phosphorylation Photo-Phosphorylation Model of the Electron Transport Chain (ETC) Glycerol-3-P Shuttle Outer Mitochondrial Membrane G3P DHAP
Lecture 10 Proton Gradient-dependent ATP Synthesis Oxidative Phosphorylation Photo-Phosphorylation Model of the Electron Transport Chain (ETC) Glycerol-3-P Shuttle Outer Mitochondrial Membrane G3P DHAP
Energy Transformation. Metabolism = total chemical reactions in cells.
 Energy Transformation Metabolism = total chemical reactions in cells. metabole = change Metabolism is concerned with managing the material and energy resources of the cell -Catabolism -Anabolism -Catabolism
Energy Transformation Metabolism = total chemical reactions in cells. metabole = change Metabolism is concerned with managing the material and energy resources of the cell -Catabolism -Anabolism -Catabolism
ATP. Division Ave. High School AP Biology. Cellular Respiration Stage 4: Electron Transport Chain. Cellular respiration. The point is to make ATP!
 ellular Respiration Stage 4: Electron Transport hain 2006-2007 ellular respiration What s the point? The point is to make! 2006-2007 1 accounting so far Glycolysis 2 Kreb s cycle 2 Life takes a lot of
ellular Respiration Stage 4: Electron Transport hain 2006-2007 ellular respiration What s the point? The point is to make! 2006-2007 1 accounting so far Glycolysis 2 Kreb s cycle 2 Life takes a lot of
BCH 4054 Spring 2001 Chapter 21 Lecture Notes
 BCH 4054 Spring 2001 Chapter 21 Lecture Notes 1 Chapter 21 Electron Transport and Oxidative Phosphorylation 2 Overview Oxidation of NADH and CoQH 2 produced in TCA cycle by O 2 is very exergonic. Some
BCH 4054 Spring 2001 Chapter 21 Lecture Notes 1 Chapter 21 Electron Transport and Oxidative Phosphorylation 2 Overview Oxidation of NADH and CoQH 2 produced in TCA cycle by O 2 is very exergonic. Some
Brain regions related to quantum coherence
 Brain regions related to quantum coherence Research since 2007 has shown that quantum coherence is utilised in increasing the efficiency of energy transfer in photosynthetic systems. What has not been
Brain regions related to quantum coherence Research since 2007 has shown that quantum coherence is utilised in increasing the efficiency of energy transfer in photosynthetic systems. What has not been
1.9 Practice Problems
 1.9 Practice Problems 1. Solution: B It s not only chlorophyll a but a combination of pigments. 2. Solution: D See at what wavelength rate of photosynthesis is the highest. 3. Solution: D It s a fact.
1.9 Practice Problems 1. Solution: B It s not only chlorophyll a but a combination of pigments. 2. Solution: D See at what wavelength rate of photosynthesis is the highest. 3. Solution: D It s a fact.
CP Biology Unit 5 Cell Energy Study Guide. Electron Carriers Electron Transport Chain Fermentation Glycolysis Krebs cycle Light-Dependent Reactions
 Name: KEY CP Biology Unit 5 Cell Energy Study Guide Vocabulary to know: ATP ADP Aerobic Anaerobic ATP Synthases Cellular Respiration Chlorophyll Chloroplast Electron Carriers Electron Transport Chain Fermentation
Name: KEY CP Biology Unit 5 Cell Energy Study Guide Vocabulary to know: ATP ADP Aerobic Anaerobic ATP Synthases Cellular Respiration Chlorophyll Chloroplast Electron Carriers Electron Transport Chain Fermentation
Energy for biological processes
 1 Energy transfer When you have finished revising this topic, you should: be able to explain the difference between catabolic and anabolic reactions be able to describe the part played by in cell metabolism
1 Energy transfer When you have finished revising this topic, you should: be able to explain the difference between catabolic and anabolic reactions be able to describe the part played by in cell metabolism
Energy for Life 12/11/14. Light Absorption in Chloroplasts
 Energy for Life Biochemical pathways A series of reactions where the products of one reaction is used in the next reaction Light Absorption in Chloroplasts Chloroplasts Two membranes Grana- layered stacks
Energy for Life Biochemical pathways A series of reactions where the products of one reaction is used in the next reaction Light Absorption in Chloroplasts Chloroplasts Two membranes Grana- layered stacks
Center for Academic Services & Advising
 March 2, 2017 Biology I CSI Worksheet 6 1. List the four components of cellular respiration, where it occurs in the cell, and list major products consumed and produced in each step. i. Hint: Think about
March 2, 2017 Biology I CSI Worksheet 6 1. List the four components of cellular respiration, where it occurs in the cell, and list major products consumed and produced in each step. i. Hint: Think about
2015 Biology Unit #3 Quiz 1 Photosynthesis, Cellular Respiration and Fermentation Week of November
 Name: Class: Date: 2015 Biology Unit #3 Quiz 1 Photosynthesis, Cellular Respiration and Fermentation Week of 02-09 November 1 Which of the following statements is true for all cells? a They use solar energy
Name: Class: Date: 2015 Biology Unit #3 Quiz 1 Photosynthesis, Cellular Respiration and Fermentation Week of 02-09 November 1 Which of the following statements is true for all cells? a They use solar energy
1/25/2018. Bio 1101 Lec. 5, Part A Chapter 6: Cellular Respiration
 1 2 3 4 5 Bio 1101 Lec. 5, Part A Chapter 6: Cellular Respiration Energy is needed by cells to do work Chemical energy, a form of potential energy, is stored in bonds of food molecules (such as glucose)
1 2 3 4 5 Bio 1101 Lec. 5, Part A Chapter 6: Cellular Respiration Energy is needed by cells to do work Chemical energy, a form of potential energy, is stored in bonds of food molecules (such as glucose)
Mitochondria Mitochondria were first seen by kollicker in 1850 in muscles and called them sarcosomes. Flemming (1882) described these organelles as
 Mitochondria Mitochondria were first seen by kollicker in 1850 in muscles and called them sarcosomes. Flemming (1882) described these organelles as filia Altmann (1890) observed these structures and named
Mitochondria Mitochondria were first seen by kollicker in 1850 in muscles and called them sarcosomes. Flemming (1882) described these organelles as filia Altmann (1890) observed these structures and named
Unit 8 Cell Metabolism. Foldable Notes
 Unit 8 Cell Metabolism Foldable Notes Silently read pages 94-96 of your biology textbook Middle Inside Top Vocabulary 1. ATP 2. ADP 3. Product 4. Reactant 5. Chloroplast 6. Mitochondria 7. Heterotroph
Unit 8 Cell Metabolism Foldable Notes Silently read pages 94-96 of your biology textbook Middle Inside Top Vocabulary 1. ATP 2. ADP 3. Product 4. Reactant 5. Chloroplast 6. Mitochondria 7. Heterotroph
In Cellular Respiration, are removed from sugar and transferred to
 1 2 3 4 5 Bio 1101 Lec. 5, Part A (Guided Notes) Chapter 6: Cellular Respiration Energy is needed by cells to do work Chemical energy, a form of potential energy, is stored in bonds of food molecules (such
1 2 3 4 5 Bio 1101 Lec. 5, Part A (Guided Notes) Chapter 6: Cellular Respiration Energy is needed by cells to do work Chemical energy, a form of potential energy, is stored in bonds of food molecules (such
UNIT 2: CELLS Chapter 4: Cells and Energy
 CORNELL NOTES Directions: You must create a minimum of 5 questions in this column per page (average). Use these to study your notes and prepare for tests and quizzes. Notes will be stamped after each assigned
CORNELL NOTES Directions: You must create a minimum of 5 questions in this column per page (average). Use these to study your notes and prepare for tests and quizzes. Notes will be stamped after each assigned
Pre-AP Biology Energy Unit Study Guide Part I
 Pre-AP Biology Energy Unit Study Guide Part I The Law of conservation of matter/mass : matter can not be created or destroyed However, matter may be rearranged in space In chemical reactions, the mass
Pre-AP Biology Energy Unit Study Guide Part I The Law of conservation of matter/mass : matter can not be created or destroyed However, matter may be rearranged in space In chemical reactions, the mass
Pathways that Harvest and Store Chemical Energy
 6 Pathways that Harvest and Store Chemical Energy Energy is stored in chemical bonds and can be released and transformed by metabolic pathways. Chemical energy available to do work is termed free energy
6 Pathways that Harvest and Store Chemical Energy Energy is stored in chemical bonds and can be released and transformed by metabolic pathways. Chemical energy available to do work is termed free energy
Cell Energy Notes ATP THE ENDOSYMBIOTIC THEORY. CELL ENERGY Cells usable source of is called ATP stands for. Name Per
 Cell Energy Notes Name Per THE ENDOSYMBIOTIC THEORY The Endosymbiotic theory is the idea that a long time ago, engulfed other prokaryotic cells by. This resulted in the first First proposed by Explains
Cell Energy Notes Name Per THE ENDOSYMBIOTIC THEORY The Endosymbiotic theory is the idea that a long time ago, engulfed other prokaryotic cells by. This resulted in the first First proposed by Explains
Scale in the biological world
 Scale in the biological world From living cells to atoms A cell seen by TEM Compartmentalisation in the cell: internal membranes and the cytosol The cytosol: more than just H2O RNAs Proteins Ribosomes
Scale in the biological world From living cells to atoms A cell seen by TEM Compartmentalisation in the cell: internal membranes and the cytosol The cytosol: more than just H2O RNAs Proteins Ribosomes
8.2 Photosynthesis Draw and label a diagram showing the structure of a chloroplast as seen in electron micrographs
 8.2 Photosynthesis 8.2.1 - Draw and label a diagram showing the structure of a chloroplast as seen in electron micrographs double membrane starch grain grana thylakoid internal membrane - location of the
8.2 Photosynthesis 8.2.1 - Draw and label a diagram showing the structure of a chloroplast as seen in electron micrographs double membrane starch grain grana thylakoid internal membrane - location of the
Located in the thylakoid membranes. Chlorophyll have Mg + in the center. Chlorophyll pigments harvest energy (photons) by absorbing certain
 a review Located in the thylakoid membranes. Chlorophyll have Mg + in the center. Chlorophyll pigments harvest energy (photons) by absorbing certain wavelengths (blue-420 nm and red-660 nm are most important).
a review Located in the thylakoid membranes. Chlorophyll have Mg + in the center. Chlorophyll pigments harvest energy (photons) by absorbing certain wavelengths (blue-420 nm and red-660 nm are most important).
The Electron-Transfer Chain and Oxidative Phosphorylation
 The Electron-Transfer Chain and Oxidative Phosphorylation The Mitochondrion - Scene of the Action 1.1 The inner mitochondrial membrane compartmentalizes metabolic functions. Although mitochondria in different
The Electron-Transfer Chain and Oxidative Phosphorylation The Mitochondrion - Scene of the Action 1.1 The inner mitochondrial membrane compartmentalizes metabolic functions. Although mitochondria in different
Lecture #8 9/21/01 Dr. Hirsh
 Lecture #8 9/21/01 Dr. Hirsh Types of Energy Kinetic = energy of motion - force x distance Potential = stored energy In bonds, concentration gradients, electrical potential gradients, torsional tension
Lecture #8 9/21/01 Dr. Hirsh Types of Energy Kinetic = energy of motion - force x distance Potential = stored energy In bonds, concentration gradients, electrical potential gradients, torsional tension
Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Outer Glycolysis mitochondrial membrane Glucose ATP
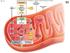 Fig. 7.5 uter Glycolysis mitochondrial membrane Glucose Intermembrane space xidation Mitochondrial matrix Acetyl-oA Krebs FAD e NAD + FAD Inner mitochondrial membrane e Electron e Transport hain hemiosmosis
Fig. 7.5 uter Glycolysis mitochondrial membrane Glucose Intermembrane space xidation Mitochondrial matrix Acetyl-oA Krebs FAD e NAD + FAD Inner mitochondrial membrane e Electron e Transport hain hemiosmosis
Cellular respiration ATP. Cellular Respiration Stage 4: Electron Transport Chain. AP Biology. The point is to make ATP! What s the point?
 ellular respiration ellular Respiration Stage 4: Electron Transport hain What s the point? The point is to make! accounting so far Glycolysis 2 Kreb s cycle 2 Life takes a lot of energy to run, need to
ellular respiration ellular Respiration Stage 4: Electron Transport hain What s the point? The point is to make! accounting so far Glycolysis 2 Kreb s cycle 2 Life takes a lot of energy to run, need to
Life Depends on Photosynthesis
 Photosynthesis Life Depends on Photosynthesis Most energy Comes from the Sun Life Depends on Photosynthesis Most energy Comes from the Sun Life Depends on Photosynthesis Most energy Comes from the Sun
Photosynthesis Life Depends on Photosynthesis Most energy Comes from the Sun Life Depends on Photosynthesis Most energy Comes from the Sun Life Depends on Photosynthesis Most energy Comes from the Sun
Electron Transport Chain (Respiratory Chain) - exercise - Vladimíra Kvasnicová
 Electron Transport Chain (Respiratory Chain) - exercise - Vladimíra Kvasnicová Respiratory chain (RCH) a) is found in all cells b) is located in a mitochondrion c) includes enzymes integrated in the inner
Electron Transport Chain (Respiratory Chain) - exercise - Vladimíra Kvasnicová Respiratory chain (RCH) a) is found in all cells b) is located in a mitochondrion c) includes enzymes integrated in the inner
4 GETTING READY TO LEARN Preview Key Concepts 4.1 Chemical Energy and ATP All cells need chemical energy.
 CHAPTER 4 Cells and Energy GETTING READY TO LEARN Preview Key Concepts 4.1 Chemical Energy and ATP All cells need chemical energy. 4.2 Overview of Photosynthesis The overall process of photosynthesis produces
CHAPTER 4 Cells and Energy GETTING READY TO LEARN Preview Key Concepts 4.1 Chemical Energy and ATP All cells need chemical energy. 4.2 Overview of Photosynthesis The overall process of photosynthesis produces
Lecture 9: Photosynthesis
 Lecture 9: Photosynthesis I. Characteristics of Light A. Light is composed of particles that travel as waves 1. Comprises a small part of the electromagnetic spectrum B. Radiation varies in wavelength
Lecture 9: Photosynthesis I. Characteristics of Light A. Light is composed of particles that travel as waves 1. Comprises a small part of the electromagnetic spectrum B. Radiation varies in wavelength
Change to Office Hours this Friday and next Monday. Tomorrow (Abel): 8:30 10:30 am. Monday (Katrina): Cancelled (05/04)
 Change to Office Hours this Friday and next Monday Tomorrow (Abel): 8:30 10:30 am Monday (Katrina): Cancelled (05/04) Lecture 10 Proton Gradient-dependent ATP Synthesis Oxidative Phosphorylation Photo-Phosphorylation
Change to Office Hours this Friday and next Monday Tomorrow (Abel): 8:30 10:30 am Monday (Katrina): Cancelled (05/04) Lecture 10 Proton Gradient-dependent ATP Synthesis Oxidative Phosphorylation Photo-Phosphorylation
Energy Converion: Mitochondria and Chloroplasts. Pınar Tulay, Ph.D.
 Energy Converion: Mitochondria and Chloroplasts Pınar Tulay, Ph.D. pintulay@gmail.com Energy Conversion Prokaryotes use plasma membrane to produce adenosine triphosphate (ATP) used in the cell function
Energy Converion: Mitochondria and Chloroplasts Pınar Tulay, Ph.D. pintulay@gmail.com Energy Conversion Prokaryotes use plasma membrane to produce adenosine triphosphate (ATP) used in the cell function
Forms of stored energy in cells
 Forms of stored energy in cells Electrochemical gradients Covalent bonds (ATP) Reducing power (NADH) During photosynthesis, respiration and glycolysis these forms of energy are converted from one to another
Forms of stored energy in cells Electrochemical gradients Covalent bonds (ATP) Reducing power (NADH) During photosynthesis, respiration and glycolysis these forms of energy are converted from one to another
4.1 Chemical Energy and ATP. KEY CONCEPT All cells need chemical energy.
 4.1 Chemical Energy and ATP KEY CONCEPT All cells need chemical energy. 4.1 Chemical Energy and ATP The chemical energy used for most cell processes is carried by ATP. Molecules in food store chemical
4.1 Chemical Energy and ATP KEY CONCEPT All cells need chemical energy. 4.1 Chemical Energy and ATP The chemical energy used for most cell processes is carried by ATP. Molecules in food store chemical
PHOTOSYNTHESIS. Light Reaction Calvin Cycle
 PHOTOSYNTHESIS Light Reaction Calvin Cycle Photosynthesis Purpose: use energy from light to convert inorganic compounds into organic fuels that have stored potential energy in their carbon bonds Carbon
PHOTOSYNTHESIS Light Reaction Calvin Cycle Photosynthesis Purpose: use energy from light to convert inorganic compounds into organic fuels that have stored potential energy in their carbon bonds Carbon
6.3 Overview of Photosynthesis
 6.3 Overview of Photosynthesis Chloroplast location of photosynthesis in plants and protists 3 membranes 2 make up the stroma Semifluid matrix Location of sugar production 1 makes up the thylakoid membrane
6.3 Overview of Photosynthesis Chloroplast location of photosynthesis in plants and protists 3 membranes 2 make up the stroma Semifluid matrix Location of sugar production 1 makes up the thylakoid membrane
The Life of a Cell. The Chemistry of Life. A View of the Cell. Cellular Transport and the Cell Cycle. Energy in a Cell
 The Life of a Cell The Chemistry of Life A View of the Cell Cellular Transport and the Cell Cycle Energy in a Cell Chapter 9 Energy in a Cell 9.1: The Need for Energy 9.1: Section Check 9.2: Photosynthesis:
The Life of a Cell The Chemistry of Life A View of the Cell Cellular Transport and the Cell Cycle Energy in a Cell Chapter 9 Energy in a Cell 9.1: The Need for Energy 9.1: Section Check 9.2: Photosynthesis:
6CO 2 + 6H 2 O C 6 H 12 O 6 + 6O 2. sun. Occurs in chloroplasts ATP. enzymes CO 2 O 2 H 2 O. sugars
 4.2 8.2 Overview Photosynthesis: of Photosynthesis An Overview Photosynthesis process by which plants make food using energy from the sun Plants are autotrophs that make their own source of chemical energy.
4.2 8.2 Overview Photosynthesis: of Photosynthesis An Overview Photosynthesis process by which plants make food using energy from the sun Plants are autotrophs that make their own source of chemical energy.
Bis2A 5.6: Oxidative Phosphorylation and the Electron Transport Chain *
 OpenStax-CNX module: m59707 1 Bis2A 5.6: Oxidative Phosphorylation and the Electron Transport Chain * The BIS2A Team This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution
OpenStax-CNX module: m59707 1 Bis2A 5.6: Oxidative Phosphorylation and the Electron Transport Chain * The BIS2A Team This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution
Active Transport * OpenStax. 1 Electrochemical Gradient
 OpenStax-CNX module: m44418 1 Active Transport * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0 By the end of this section, you will be able
OpenStax-CNX module: m44418 1 Active Transport * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0 By the end of this section, you will be able
CELL BIOLOGY - CLUTCH CH. 9 - TRANSPORT ACROSS MEMBRANES.
 !! www.clutchprep.com K + K + K + K + CELL BIOLOGY - CLUTCH CONCEPT: PRINCIPLES OF TRANSMEMBRANE TRANSPORT Membranes and Gradients Cells must be able to communicate across their membrane barriers to materials
!! www.clutchprep.com K + K + K + K + CELL BIOLOGY - CLUTCH CONCEPT: PRINCIPLES OF TRANSMEMBRANE TRANSPORT Membranes and Gradients Cells must be able to communicate across their membrane barriers to materials
METABOLISM CHAPTER 04 BIO 211: ANATOMY & PHYSIOLOGY I. Dr. Lawrence G. Altman Some illustrations are courtesy of McGraw-Hill.
 BIO 211: ANATOMY & PHYSIOLOGY I CHAPTER 04 1 Please wait 20 seconds before starting slide show. Mouse click or Arrow keys to navigate. Hit ESCAPE Key to exit. CELLULAR METABOLISM Dr. Lawrence G. Altman
BIO 211: ANATOMY & PHYSIOLOGY I CHAPTER 04 1 Please wait 20 seconds before starting slide show. Mouse click or Arrow keys to navigate. Hit ESCAPE Key to exit. CELLULAR METABOLISM Dr. Lawrence G. Altman
CELL METABOLISM OVERVIEW Keep the big picture in mind as we discuss the particulars!
 BIO 211: ANATOMY & PHYSIOLOGY I CHAPTER 04 CELLULAR METABOLISM 1 Please wait 20 seconds before starting slide show. Mouse click or Arrow keys to navigate. Hit ESCAPE Key to exit. Dr. Lawrence G. Altman
BIO 211: ANATOMY & PHYSIOLOGY I CHAPTER 04 CELLULAR METABOLISM 1 Please wait 20 seconds before starting slide show. Mouse click or Arrow keys to navigate. Hit ESCAPE Key to exit. Dr. Lawrence G. Altman
Energy Metabolism exergonic reaction endergonic reaction Energy of activation
 Metabolism Energy Living things require energy to grow and reproduce Most energy used originates from the sun Plants capture 2% of solar energy Some captured energy is lost as metabolic heat All energy
Metabolism Energy Living things require energy to grow and reproduce Most energy used originates from the sun Plants capture 2% of solar energy Some captured energy is lost as metabolic heat All energy
AQA Biology A-level Topic 5: Energy transfers in and between organisms
 AQA Biology A-level Topic 5: Energy transfers in and between organisms Notes Photosynthesis Photosynthesis is a reaction in which light energy is used to produce glucose in plants. The process requires
AQA Biology A-level Topic 5: Energy transfers in and between organisms Notes Photosynthesis Photosynthesis is a reaction in which light energy is used to produce glucose in plants. The process requires
