Atomic Force Microscope Imaging of the Aggregation of Mouse Immunoglobulin G Molecules
|
|
|
- Bryce Mitchell
- 6 years ago
- Views:
Transcription
1 Molecules 2003, 8, molecules ISSN Atomic Force Microscope Imaging of the Aggregation of Mouse Immunoglobulin G Molecules Jiye Cai*, Yao Chen, Qingcai Xu, Yong Chen, Tao Zhao, Xiaoyan Wang and Ke Xia Department of Chemistry, Jinan University, Guangzhou, , China; Tel. (+86) (020) , Fax (+86) (020) *Author to whom correspondence should be addressed; tjycai@jnu.edu.cn Abstract: Mouse immunoglobulin G (Ig G1 and the mixture of Ig G1 and Ig G2) deposited on mica were imaged with an atomic force microscope at room temperature and ambient pressure. At a concentration around 1.0mg/L, the molecules were well dispersed. 2~3 days after sample preparation, both Ig G1 and the mixture could self-assemble into different shapes and further form some types of local-ordered toroidal aggregations (monotoroidal, intercrossed toroidal, concentric toroidal, etc.). The number of monomers was not identical in the different toroidal aggregations but in a same circle, the shapes of polymer self-assembled by several monomolecules were found to be almost the same. There was difference between the aggregation behavior of Ig G1 and the mixture. The mechanism of Ig G molecule aggregation was ascribed to the Y shape and loops structure of Ig G molecule. Keywords: Mouse immunoglobulin G, self-assembly, aggregation, atomic force microscope.
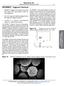
2 Molecules 2003, 8 87 Introduction The atomic force microscope (AFM), with its high resolution and continuous images of cells and biomacromolecules, has recently become a powerful tool for the study of biomolecular shape s, conformations, behavior and the relationship between function and molecular conformation. As an antibody, the immunoglobulin G (Ig G) molecule is used as a model in immunology studies and has been studied by many methods, including with AFM [1], due to its important biological function. Its characteristic molecular structure shape, composed of two identical light chains and two identical heavy chains with many loops, makes the Ig G molecule very active and able to bind tightly to almost any other molecule, such as those on the surface of an invading microorganism, therefore the monomolecular behavior of Ig G deserves our attention. Its self-assembled monolayer was studied as a method for immobilizing the interfacial-recognition layer of a surface plasmon resonance immunosensor [2]. In order to further understand the molecular behavior of Ig G itself, AFM was used to probe the aggregation of Ig G molecules deposited on mica in air. The phenomena of further aggregation of Ig G molecules, which have not been reported previously, are reported in this paper and the mechanisms of their aggregation are discussed based on the Y shape and loop structures of the Ig G molecule. This study provides valuable data for immunology and clinical medicine. Results Figure 1 shows the monomolecular image of Ig G1 and Ig G1+Ig G2 (sample concentrations: 0.1µg/mL; scanning scope: 0.5µm) when they were just put on the mica. Ig G1 and Ig G1+Ig G2 were well dispersed as monomers, which in the case of Ig G1 was a 17.0~21.1nm globe, while that of Ig G1+Ig G2 was 20.4~28.8nm. Slight bigger monomer images were obtained here than with X-ray crystallography [3] due to the fact that the Ig G molecule was covered with a water membrane [4] and combined with marker protein PE. The image mechanism of AFM also affected the results [5]. (a) (b) Figure 1. Monomolecular images of Ig G1 and Ig G1+Ig G2 (a. Ig G1; b. Ig G1+Ig G2).
3 Molecules 2003, or 3 days after sample preparation, as the water in sample solution evaporated gradually, Ig G1 monomers displayed self-assembly and aggregation as shown in Figure 2. (a) (b) (c) (d) (e) (f) Figure 2. Some aggregation images of Ig G1 Scanning scopes: a. 18µm; b. 10µm; c. 5µm; d. 5µm; e. 3.5µm; f. 3.31µm. Obviously, these regular aggregations were formed not only by the surface tension as the water evaporated, but also by the interaction among those monomers or polymers, because of the existence of intercrossed toroidal, concentric or nonconcentric circles and so on. These phenomena also appeared in Ig G1+Ig G2, shown in Figure 3, and their aggregation was quicker. From the figures, we find that the monomers self-assembled first into polymers before their further aggregation. It was interesting that the shapes of polymer self-assembled by several monomolecules were found to be almost same in a concentric circle, and several kinds of them are shown in Figure 4. These results explained also the existence of interaction among Ig G molecules or among their polymers. Those aggregation circles of Ig G finally disappeared naturally.
4 Molecules 2003, 8 89 a) (b) Figure 3. Aggregation images of Ig G1+Ig G2 Scanning scope: a. 7µm; b. 1µm. (a) (b) (c) (d) Figure 4. Shapes of polymer self-assembled by several monomolecules Scanning scopes: a. 2µm; b. 800nm; c. 600nm; d. 250nm. Discussion Although the self-assembly of Ig G molecules had been studied, further aggregation had not been reported in the literature. Their further aggregation phenomena in aqueous solution were observed in this study as water evaporated and the ir solutions became more concentrated.the dispersed Ig G
5 Molecules 2003, 8 90 molecules deposited on mica in solution moved closer and formed polymers. The self-assembly and aggregation of Ig G molecules can be ascribed to monomolecular polarity and affinity, for example Van der Waals attraction forces caused by those chemical groups on the molecular surface, as well as hydrogen bonds, salt bridges, etc., which make the Ig G molecules aggregate closely and therefore the protein becomes more stable. On the other hand, the Y shape and loop structure [6] of Ig G should be main reason to make their self-assembly and aggregation easy. Ig G molecules can bind to their antigens extremely tightly, and in this process the loops allow a large number of chemical groups to surround a ligand (antigen) so that the protein can link to it with many weak bonds. Therefore, it is the loop structure that is mainly responsible for making Ig G very active and become an attack molecule. When Ig G molecules (including polymers) moved closer, they contact each other widely due to the Y shape of molecule instead of the globe conformation. Many chemical bonds between loops are formed, especially in the antigen binding site of Ig G, which may change its conformation to fit the ligand. The se inter-attractions make monomolecules combine tightly to form several kinds of regular shapes as shown in Figure 4. The phenomena seen in this study suggest that Ig G may attack the other molecule (antigen) easily. Polymers became more active and unstable because their molecular volume increases, so these polymers tend to aggregate to balancing the energy or to reach the lowest energy combination in the system. The aggregation behavior of Ig G polymers helps their stability to avoid the decomposition of the polymer structure, which is in agreement of our understanding of the process of the protein's denaturing, precipitation, crystallization and so on. The circle is a system of lower energy, as we know, so this kind of aggregation appears in the Ig G polymers. In addition, these aggregations of protein molecules may be explained by the dissipative structure theory. The unstable macromolecules always tend to make themselves more stable. We present experimental results to show the interaction among Ig G molecules as seen in the study, and although we assume the Y shape and loops structure of Ig G to explain the aggregation of Ig G molecules, we don t know whether new covalent linkages have formed between monomolecules or polymers. For that, we need further study to better understand the mechanism of aggregation of Ig G molecules. Conclusions Ig G molecules self-assemble in aqueous solution and further aggregations of these Ig G molecules are presented. Furthermore, the mechanism of Ig G molecule aggregation was explained by using the Y shape and loops structure of Ig G molecule.
6 Molecules 2003, 8 91 Experimental General Mouse Ig G1 combined with the marker protein PE, and mixtures of Ig G1 and Ig G2 (Ig G1+Ig G2, the proportions of them being as same as in normal serum) were supplied by Becton, Dickinson and Company (both the same concentrations of 100mg/L). Ig G1 and Ig G1+Ig G2 were diluted respectively to 0.1mg/L with bacteria-free water. A sample of about 0.5µL was put on the surface of fresh mica, then the sample dispersed and dried gradually. The samples were imaged with AFM every day thereafter. AFM (Autoprobe CP Research, THERMO, USA) was performed at 25ºC, 50% humidity. Samples were measured with a 100µm scanner, high pressure NCM model and UL 20D silicon probe needle (microarm length 85µm, force constant 25N/m, needle tip r=10nm, data collection in constant force made). All images were treated with the Flatten Active software to only remove the lower-frequency noise in the scanning direction. References and Notes 1. Preininger, C.; Clausen-Schaumamn, H.; Ahluwalia, A.; De Rossi, D. Characterization of Ig G Langmuir-Blodgett films immobilized on funtionalized polymers. Talanta 2000, 52, Disley, D. M.; Cullen, D. C.; You, H-X.; Lowe, C. R. Covalent coupling of immunoglobulin G to self-assembled monolayers as a method for immobilizing the interfa cial-recognition layer of a surface plasmon resonance immunosensor. Biosensors & Bioelectronics 1998, 13, Hansma H. G.; Vesenka J.; Siegerist, C.; Kelderman G..; Morrett, H.; Shinsheimer, R. L.; Elings, V.; Bustamante, C.; Hansma, P.K. Reproducible Imaging and Dissection of Plasmid DNA Under Liquid With the Atomic Force Microscope. Science 1992, 256, Drake B.; Prater C. B.; Weisenhorn A. L. ; Gould, S.A.C.; Albrecht, T.R.; Quate, C.F.; Cannell, D.S.; Hansma, H.G.; Hansma, P. K. Imaging crystals, polymers, and processes in water with the atomic force microscope. Science 1989, 243, Keller D.; Reconstruction of STM and AFM images distorted by finite-size tips. Surf. Sci. 1991, 253, Alberts B.; Bray D.; Johnson A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. Essential Cell Biology; Garland Publishing, Inc.: New York & London, 1998, p by MDPI ( Reproduction is permitted for noncommercial purposes.
Lecture 12: Biomaterials Characterization in Aqueous Environments
 3.051J/20.340J 1 Lecture 12: Biomaterials Characterization in Aqueous Environments High vacuum techniques are important tools for characterizing surface composition, but do not yield information on surface
3.051J/20.340J 1 Lecture 12: Biomaterials Characterization in Aqueous Environments High vacuum techniques are important tools for characterizing surface composition, but do not yield information on surface
Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation
 Supplementary Information Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation Yu Liu, Shuping Xu, Haibo Li, Xiaoguang Jian, Weiqing Xu* State Key
Supplementary Information Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation Yu Liu, Shuping Xu, Haibo Li, Xiaoguang Jian, Weiqing Xu* State Key
Supporting information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supporting information Self-assembled nanopatch with peptide-organic multilayers and mechanical
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supporting information Self-assembled nanopatch with peptide-organic multilayers and mechanical
Atomic force microscopy study of polypropylene surfaces treated by UV and ozone exposure: modification of morphology and adhesion force
 Ž. Applied Surface Science 144 145 1999 627 632 Atomic force microscopy study of polypropylene surfaces treated by UV and ozone exposure: modification of morphology and adhesion force H.-Y. Nie ), M.J.
Ž. Applied Surface Science 144 145 1999 627 632 Atomic force microscopy study of polypropylene surfaces treated by UV and ozone exposure: modification of morphology and adhesion force H.-Y. Nie ), M.J.
Monolayers. Factors affecting the adsorption from solution. Adsorption of amphiphilic molecules on solid support
 Monolayers Adsorption as process Adsorption of gases on solids Adsorption of solutions on solids Factors affecting the adsorption from solution Adsorption of amphiphilic molecules on solid support Adsorption
Monolayers Adsorption as process Adsorption of gases on solids Adsorption of solutions on solids Factors affecting the adsorption from solution Adsorption of amphiphilic molecules on solid support Adsorption
Electonegativity, Polar Bonds, and Polar Molecules
 Electonegativity, Polar Bonds, and Polar Molecules Some Definitions Electronegativity: the ability of an atom to attract bonding electrons to itself. Intramolecular forces: the attractive force between
Electonegativity, Polar Bonds, and Polar Molecules Some Definitions Electronegativity: the ability of an atom to attract bonding electrons to itself. Intramolecular forces: the attractive force between
The four forces of nature. Intermolecular forces, surface forces & the Atomic Force Microscope (AFM) Force- and potential curves
 Intermolecular forces, surface forces & the Atomic Force Microscope (AFM) The four forces of nature Strong interaction Holds neutrons and protons together in atomic nuclei. Weak interaction β and elementary
Intermolecular forces, surface forces & the Atomic Force Microscope (AFM) The four forces of nature Strong interaction Holds neutrons and protons together in atomic nuclei. Weak interaction β and elementary
Preparation and Characterization of Double Metal Cyanide Complex Catalysts
 Molecules 2003, 8, 67-73 molecules ISSN 1420-3049 http://www.mdpi.org Preparation and Characterization of Double Metal Cyanide Complex Catalysts Hanxia Liu 1, Xikui Wang 1, *, Yao Gu 2 and Weilin Guo 1
Molecules 2003, 8, 67-73 molecules ISSN 1420-3049 http://www.mdpi.org Preparation and Characterization of Double Metal Cyanide Complex Catalysts Hanxia Liu 1, Xikui Wang 1, *, Yao Gu 2 and Weilin Guo 1
`1AP Biology Study Guide Chapter 2 v Atomic structure is the basis of life s chemistry Ø Living and non- living things are composed of atoms Ø
 `1AP Biology Study Guide Chapter 2 v Atomic structure is the basis of life s chemistry Ø Living and non- living things are composed of atoms Ø Element pure substance only one kind of atom Ø Living things
`1AP Biology Study Guide Chapter 2 v Atomic structure is the basis of life s chemistry Ø Living and non- living things are composed of atoms Ø Element pure substance only one kind of atom Ø Living things
Specific ion effects on the interaction of. hydrophobic and hydrophilic self assembled
 Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Scanning Tunneling Microscopy and its Application
 Chunli Bai Scanning Tunneling Microscopy and its Application With 181 Figures SHANGHAI SCIENTIFIC & TECHNICAL PUBLISHERS Jpl Springer Contents 1. Introduction 1 1.1 Advantages of STM Compared with Other
Chunli Bai Scanning Tunneling Microscopy and its Application With 181 Figures SHANGHAI SCIENTIFIC & TECHNICAL PUBLISHERS Jpl Springer Contents 1. Introduction 1 1.1 Advantages of STM Compared with Other
AFM Imaging In Liquids. W. Travis Johnson PhD Agilent Technologies Nanomeasurements Division
 AFM Imaging In Liquids W. Travis Johnson PhD Agilent Technologies Nanomeasurements Division Imaging Techniques: Scales Proteins 10 nm Bacteria 1μm Red Blood Cell 5μm Human Hair 75μm Si Atom Spacing 0.4nm
AFM Imaging In Liquids W. Travis Johnson PhD Agilent Technologies Nanomeasurements Division Imaging Techniques: Scales Proteins 10 nm Bacteria 1μm Red Blood Cell 5μm Human Hair 75μm Si Atom Spacing 0.4nm
Interaction of Gold Nanoparticle with Proteins
 Chapter 7 Interaction of Gold Nanoparticle with Proteins 7.1. Introduction The interfacing of nanoparticle with biomolecules such as protein is useful for applications ranging from nano-biotechnology (molecular
Chapter 7 Interaction of Gold Nanoparticle with Proteins 7.1. Introduction The interfacing of nanoparticle with biomolecules such as protein is useful for applications ranging from nano-biotechnology (molecular
Saba Al Fayoumi. Tamer Barakat. Dr. Mamoun Ahram + Dr. Diala Abu-Hassan
 1 Saba Al Fayoumi Tamer Barakat Dr. Mamoun Ahram + Dr. Diala Abu-Hassan What is BIOCHEMISTRY??? Biochemistry = understanding life Chemical reactions are what makes an organism (An organism is simply atoms
1 Saba Al Fayoumi Tamer Barakat Dr. Mamoun Ahram + Dr. Diala Abu-Hassan What is BIOCHEMISTRY??? Biochemistry = understanding life Chemical reactions are what makes an organism (An organism is simply atoms
Effect of Non-Ionic Surfactants on Dispersion and. Polar Interactions in the Adsorption of Cellulases. onto Lignin
 Supporting Information Effect of Non-Ionic Surfactants on Dispersion and Polar Interactions in the Adsorption of Cellulases onto Lignin Feng Jiang, Chen Qian, Alan R. Esker and Maren Roman, * Macromolecules
Supporting Information Effect of Non-Ionic Surfactants on Dispersion and Polar Interactions in the Adsorption of Cellulases onto Lignin Feng Jiang, Chen Qian, Alan R. Esker and Maren Roman, * Macromolecules
Physics and Chemistry of Interfaces
 Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Spectrum-resolved Dual-color Electrochemiluminescence Immunoassay for Simultaneous Detection of Two Targets with Nanocrystals as Tags
 Supporting Information Spectrum-resolved Dual-color Electrochemiluminescence Immunoassay for Simultaneous Detection of Two Targets with Nanocrystals as Tags Guizheng Zou, *, Xiao Tan, Xiaoyan Long, Yupeng
Supporting Information Spectrum-resolved Dual-color Electrochemiluminescence Immunoassay for Simultaneous Detection of Two Targets with Nanocrystals as Tags Guizheng Zou, *, Xiao Tan, Xiaoyan Long, Yupeng
Patrick: An Introduction to Medicinal Chemistry 5e Chapter 01
 Questions Patrick: An Introduction to Medicinal Chemistry 5e 01) Which of the following molecules is a phospholipid? a. i b. ii c. iii d. iv 02) Which of the following statements is false regarding the
Questions Patrick: An Introduction to Medicinal Chemistry 5e 01) Which of the following molecules is a phospholipid? a. i b. ii c. iii d. iv 02) Which of the following statements is false regarding the
Basic Laboratory. Materials Science and Engineering. Atomic Force Microscopy (AFM)
 Basic Laboratory Materials Science and Engineering Atomic Force Microscopy (AFM) M108 Stand: 20.10.2015 Aim: Presentation of an application of the AFM for studying surface morphology. Inhalt 1.Introduction...
Basic Laboratory Materials Science and Engineering Atomic Force Microscopy (AFM) M108 Stand: 20.10.2015 Aim: Presentation of an application of the AFM for studying surface morphology. Inhalt 1.Introduction...
Supporting Information
 Supporting Information Gold Nanoparticle Dimers for Plasmon Sensing Yunan Cheng, 1,2,3 Mang Wang, 1 Gustaaf Borghs, * 2,3 Hongzheng Chen * 1 1. MOE Key Laboratory of Macromolecule Synthesis and Functionalization,
Supporting Information Gold Nanoparticle Dimers for Plasmon Sensing Yunan Cheng, 1,2,3 Mang Wang, 1 Gustaaf Borghs, * 2,3 Hongzheng Chen * 1 1. MOE Key Laboratory of Macromolecule Synthesis and Functionalization,
Supplementary Information
 Supplementary Information Facile preparation of superhydrophobic coating by spraying a fluorinated acrylic random copolymer micelle solution Hui Li, a,b Yunhui Zhao a and Xiaoyan Yuan* a a School of Materials
Supplementary Information Facile preparation of superhydrophobic coating by spraying a fluorinated acrylic random copolymer micelle solution Hui Li, a,b Yunhui Zhao a and Xiaoyan Yuan* a a School of Materials
CORE MOLIT ACTIVITIES at a glance
 CORE MOLIT ACTIVITIES at a glance 1. Amplification of Biochemical Signals: The ELISA Test http://molit.concord.org/database/activities/248.html The shape of molecules affects the way they function. A test
CORE MOLIT ACTIVITIES at a glance 1. Amplification of Biochemical Signals: The ELISA Test http://molit.concord.org/database/activities/248.html The shape of molecules affects the way they function. A test
Chemistry in Biology Section 1 Atoms, Elements, and Compounds
 Name Chemistry in Biology Section 1 Atoms, Elements, and Compounds Date Main Idea Details Scan the headings and boldfaced words in Section 1 of the chapter. Predict two things that you think might be discussed.
Name Chemistry in Biology Section 1 Atoms, Elements, and Compounds Date Main Idea Details Scan the headings and boldfaced words in Section 1 of the chapter. Predict two things that you think might be discussed.
Chapter 10: Liquids, Solids, and Phase Changes
 Chapter 10: Liquids, Solids, and Phase Changes In-chapter exercises: 10.1 10.6, 10.11; End-of-chapter Problems: 10.26, 10.31, 10.32, 10.33, 10.34, 10.35, 10.36, 10.39, 10.40, 10.42, 10.44, 10.45, 10.66,
Chapter 10: Liquids, Solids, and Phase Changes In-chapter exercises: 10.1 10.6, 10.11; End-of-chapter Problems: 10.26, 10.31, 10.32, 10.33, 10.34, 10.35, 10.36, 10.39, 10.40, 10.42, 10.44, 10.45, 10.66,
Single-Molecule Recognition and Manipulation Studied by Scanning Probe Microscopy
 Single-Molecule Recognition and Manipulation Studied by Scanning Probe Microscopy Byung Kim Department of Physics Boise State University Langmuir (in press, 2006) swollen collapsed Hydrophilic non-sticky
Single-Molecule Recognition and Manipulation Studied by Scanning Probe Microscopy Byung Kim Department of Physics Boise State University Langmuir (in press, 2006) swollen collapsed Hydrophilic non-sticky
Surface Plasmon Resonance for Immunoassays. Sadagopan Krishnan Chem 395 Instructor: Prof.Rusling
 Surface Plasmon Resonance for Immunoassays Sadagopan Krishnan Chem 395 Instructor: Prof.Rusling 1 Outline Introduction Understanding the Basics SPR- Instrumental components Applications in Immunoassays
Surface Plasmon Resonance for Immunoassays Sadagopan Krishnan Chem 395 Instructor: Prof.Rusling 1 Outline Introduction Understanding the Basics SPR- Instrumental components Applications in Immunoassays
Chapter 2: Chemistry. What does chemistry have to do with biology? Vocabulary BIO 105
 Chapter 2: Chemistry What does chemistry have to do with biology? BIO 105 Vocabulary 1. Matter anything that takes up space and has mass Atoms are the smallest units of matter that can participate in chemical
Chapter 2: Chemistry What does chemistry have to do with biology? BIO 105 Vocabulary 1. Matter anything that takes up space and has mass Atoms are the smallest units of matter that can participate in chemical
Highly Sensitive and Selective Colorimetric Visualization of Streptomycin in Raw Milk Using Au Nanoparticles Supramolecular Assembly
 SUPPORTING INFORMATION Highly Sensitive and Selective Colorimetric Visualization of Streptomycin in Raw Milk Using Au Nanoparticles Supramolecular Assembly Jiayu Sun, Jiechao Ge, Weimin Liu, Zhiyuan Fan,
SUPPORTING INFORMATION Highly Sensitive and Selective Colorimetric Visualization of Streptomycin in Raw Milk Using Au Nanoparticles Supramolecular Assembly Jiayu Sun, Jiechao Ge, Weimin Liu, Zhiyuan Fan,
The Effect of Water and Confinement on Self-Assembly of
 Supporting Information: The Effect of Water and Confinement on Self-Assembly of Imidazolium Based Ionic Liquids at Mica Interface H.-W. Cheng, J.-N. Dienemann, P. Stock, C. Merola, Y.-J. Chen and M. Valtiner*
Supporting Information: The Effect of Water and Confinement on Self-Assembly of Imidazolium Based Ionic Liquids at Mica Interface H.-W. Cheng, J.-N. Dienemann, P. Stock, C. Merola, Y.-J. Chen and M. Valtiner*
Kinetic Molecular Theory of Gases used to account for Ideal Gas Behavior when gases approach high temperatures and low pressures
 LIQUIDS AND SOLIDS Kinetic Molecular Theory of Gases used to account for Ideal Gas Behavior when gases approach high temperatures and low pressures GASES are very different from solids and liquids. We
LIQUIDS AND SOLIDS Kinetic Molecular Theory of Gases used to account for Ideal Gas Behavior when gases approach high temperatures and low pressures GASES are very different from solids and liquids. We
Chapter 10. Dipole Moments. Intermolecular Forces (IMF) Polar Bonds and Polar Molecules. Polar or Nonpolar Molecules?
 Polar Bonds and Polar Molecules Chapter 10 Liquids, Solids, and Phase Changes Draw Lewis Structures for CCl 4 and CH 3 Cl. What s the same? What s different? 1 Polar Covalent Bonds and Dipole Moments Bonds
Polar Bonds and Polar Molecules Chapter 10 Liquids, Solids, and Phase Changes Draw Lewis Structures for CCl 4 and CH 3 Cl. What s the same? What s different? 1 Polar Covalent Bonds and Dipole Moments Bonds
Synthesis and Structure of the Cadmium (II) Complex: [Cd(C 5 H 5 N) 2 (S 2 CO-n-C 4 H 9 ) 2 ]
![Synthesis and Structure of the Cadmium (II) Complex: [Cd(C 5 H 5 N) 2 (S 2 CO-n-C 4 H 9 ) 2 ] Synthesis and Structure of the Cadmium (II) Complex: [Cd(C 5 H 5 N) 2 (S 2 CO-n-C 4 H 9 ) 2 ]](/thumbs/77/75259656.jpg) Molecules 2002, 7, 549-553 molecules ISSN 1420-3049 http://www.mdpi.org Synthesis and Structure of the Cadmium (II) Complex: [Cd(C 5 H 5 N) 2 (S 2 CO-n-C 4 H 9 ) 2 ] Xu Hong Jiang 1,2, Wei Guang Zhang
Molecules 2002, 7, 549-553 molecules ISSN 1420-3049 http://www.mdpi.org Synthesis and Structure of the Cadmium (II) Complex: [Cd(C 5 H 5 N) 2 (S 2 CO-n-C 4 H 9 ) 2 ] Xu Hong Jiang 1,2, Wei Guang Zhang
The Logic of Biological Phenomena
 The Logic of Biological Phenomena molecules are lifeless yet, in appropriate complexity and number, molecules compose living things What makes living things distinct? they can grow they can move they can
The Logic of Biological Phenomena molecules are lifeless yet, in appropriate complexity and number, molecules compose living things What makes living things distinct? they can grow they can move they can
Fabrication of ordered array at a nanoscopic level: context
 Fabrication of ordered array at a nanoscopic level: context Top-down method Bottom-up method Classical lithography techniques Fast processes Size limitations it ti E-beam techniques Small sizes Slow processes
Fabrication of ordered array at a nanoscopic level: context Top-down method Bottom-up method Classical lithography techniques Fast processes Size limitations it ti E-beam techniques Small sizes Slow processes
6 Hydrophobic interactions
 The Physics and Chemistry of Water 6 Hydrophobic interactions A non-polar molecule in water disrupts the H- bond structure by forcing some water molecules to give up their hydrogen bonds. As a result,
The Physics and Chemistry of Water 6 Hydrophobic interactions A non-polar molecule in water disrupts the H- bond structure by forcing some water molecules to give up their hydrogen bonds. As a result,
2015 AP Biology Unit 2 PRETEST- Introduction to the Cell and Biochemistry
 Name: Class: _ Date: _ 2015 AP Biology Unit 2 PRETEST- Introduction to the Cell and Biochemistry Multiple Choice Identify the choice that best completes the statement or answers the question. 1) In what
Name: Class: _ Date: _ 2015 AP Biology Unit 2 PRETEST- Introduction to the Cell and Biochemistry Multiple Choice Identify the choice that best completes the statement or answers the question. 1) In what
Thin film techniques: the layer-by-layer self assembly technique
 Thin film techniques: the layer-by-layer self assembly technique Carmelina Ruggiero University of Genoa Overview Thin films Thin film techniques Langmuir-Blodgett technique Chemical self-assembling Layer-by-Layer
Thin film techniques: the layer-by-layer self assembly technique Carmelina Ruggiero University of Genoa Overview Thin films Thin film techniques Langmuir-Blodgett technique Chemical self-assembling Layer-by-Layer
BIOCHEMISTRY GUIDED NOTES - AP BIOLOGY-
 BIOCHEMISTRY GUIDED NOTES - AP BIOLOGY- ELEMENTS AND COMPOUNDS - anything that has mass and takes up space. - cannot be broken down to other substances. - substance containing two or more different elements
BIOCHEMISTRY GUIDED NOTES - AP BIOLOGY- ELEMENTS AND COMPOUNDS - anything that has mass and takes up space. - cannot be broken down to other substances. - substance containing two or more different elements
Review Topic 8: Phases of Matter and Mixtures
 Name: Score: 24 / 24 points (100%) Review Topic 8: Phases of Matter and Mixtures Multiple Choice Identify the choice that best completes the statement or answers the question. C 1. Soda water is a solution
Name: Score: 24 / 24 points (100%) Review Topic 8: Phases of Matter and Mixtures Multiple Choice Identify the choice that best completes the statement or answers the question. C 1. Soda water is a solution
Elements and Isotopes
 Section 2-1 Notes Atoms Life depends on chemistry. The basic unit of matter is the atom. Atoms are incredibly small The subatomic particles that make up atoms are protons, neutrons, and electrons. Parts
Section 2-1 Notes Atoms Life depends on chemistry. The basic unit of matter is the atom. Atoms are incredibly small The subatomic particles that make up atoms are protons, neutrons, and electrons. Parts
Biophysics II. Hydrophobic Bio-molecules. Key points to be covered. Molecular Interactions in Bio-molecular Structures - van der Waals Interaction
 Biophysics II Key points to be covered By A/Prof. Xiang Yang Liu Biophysics & Micro/nanostructures Lab Department of Physics, NUS 1. van der Waals Interaction 2. Hydrogen bond 3. Hydrophilic vs hydrophobic
Biophysics II Key points to be covered By A/Prof. Xiang Yang Liu Biophysics & Micro/nanostructures Lab Department of Physics, NUS 1. van der Waals Interaction 2. Hydrogen bond 3. Hydrophilic vs hydrophobic
Chemistry of Life: Water and Solutions
 Chemistry of Life: Water and Solutions Unit Objective I can describe the role of organic and inorganic chemicals important to living things. During this unit, we will answer the following very important
Chemistry of Life: Water and Solutions Unit Objective I can describe the role of organic and inorganic chemicals important to living things. During this unit, we will answer the following very important
Paper No. 1: ORGANIC CHEMISTRY- I (Nature of Bonding and Stereochemistry)
 Subject Chemistry Paper No and Title Paper 1: ORGANIC - I (Nature of Bonding Module No and Title Module Tag CHE_P1_M10 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. Non-Covalent Interactions
Subject Chemistry Paper No and Title Paper 1: ORGANIC - I (Nature of Bonding Module No and Title Module Tag CHE_P1_M10 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. Non-Covalent Interactions
Properties of Solutions
 Properties of Solutions The States of Matter The state a substance is in at a particular temperature and pressure depends on two antagonistic entities: The kinetic energy of the particles The strength
Properties of Solutions The States of Matter The state a substance is in at a particular temperature and pressure depends on two antagonistic entities: The kinetic energy of the particles The strength
INTRODUCTION TO SCA\ \I\G TUNNELING MICROSCOPY
 INTRODUCTION TO SCA\ \I\G TUNNELING MICROSCOPY SECOND EDITION C. JULIAN CHEN Department of Applied Physics and Applied Mathematics, Columbia University, New York OXFORD UNIVERSITY PRESS Contents Preface
INTRODUCTION TO SCA\ \I\G TUNNELING MICROSCOPY SECOND EDITION C. JULIAN CHEN Department of Applied Physics and Applied Mathematics, Columbia University, New York OXFORD UNIVERSITY PRESS Contents Preface
Concept review: Binding equilibria
 Concept review: Binding equilibria 1 Binding equilibria and association/dissociation constants 2 The binding of a protein to a ligand at equilibrium can be written as: P + L PL And so the equilibrium constant
Concept review: Binding equilibria 1 Binding equilibria and association/dissociation constants 2 The binding of a protein to a ligand at equilibrium can be written as: P + L PL And so the equilibrium constant
Foundations in Microbiology Seventh Edition
 Lecture PowerPoint to accompany Foundations in Microbiology Seventh Edition Talaro Chapter 2 The Chemistry of Biology Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
Lecture PowerPoint to accompany Foundations in Microbiology Seventh Edition Talaro Chapter 2 The Chemistry of Biology Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
 For info and ordering all the 4 versions / languages of this book please visit: http://trl.lab.uic.edu/pon Contents Preface vii Chapter 1 Advances in Atomic and Molecular Nanotechnology Introduction 1
For info and ordering all the 4 versions / languages of this book please visit: http://trl.lab.uic.edu/pon Contents Preface vii Chapter 1 Advances in Atomic and Molecular Nanotechnology Introduction 1
Pre-seeding -assisted synthesis of high performance polyamide-zeolite nanocomposie membrane for water purification
 Electronic Supporting Information: Pre-seeding -assisted synthesis of high performance polyamide-zeolite nanocomposie membrane for water purification Chunlong Kong, a Takuji Shintani b and Toshinori Tsuru*
Electronic Supporting Information: Pre-seeding -assisted synthesis of high performance polyamide-zeolite nanocomposie membrane for water purification Chunlong Kong, a Takuji Shintani b and Toshinori Tsuru*
Atomic and molecular interactions. Scanning probe microscopy.
 Atomic and molecular interactions. Scanning probe microscopy. Balázs Kiss Nanobiotechnology and Single Molecule Research Group, Department of Biophysics and Radiation Biology 27. November 2013. 2 Atomic
Atomic and molecular interactions. Scanning probe microscopy. Balázs Kiss Nanobiotechnology and Single Molecule Research Group, Department of Biophysics and Radiation Biology 27. November 2013. 2 Atomic
POGIL 7 KEY Intermolecular Forces
 Honors Chem Block Name POGIL 7 KEY Intermolecular Forces In chemistry we talk a lot about properties of substances, since the object of chemistry is substances and their properties. After learning different
Honors Chem Block Name POGIL 7 KEY Intermolecular Forces In chemistry we talk a lot about properties of substances, since the object of chemistry is substances and their properties. After learning different
Supplementary Figure 1 a) Scheme of microfluidic device fabrication by photo and soft lithography,
 a b 1 mm Supplementary Figure 1 a) Scheme of microfluidic device fabrication by photo and soft lithography, (a1, a2) 50nm Pd evaporated on Si wafer with 100 nm Si 2 insulating layer and 5nm Cr as an adhesion
a b 1 mm Supplementary Figure 1 a) Scheme of microfluidic device fabrication by photo and soft lithography, (a1, a2) 50nm Pd evaporated on Si wafer with 100 nm Si 2 insulating layer and 5nm Cr as an adhesion
SELF-ASSEMBLY AND NANOTECHNOLOGY A Force Balance Approach
 SELF-ASSEMBLY AND NANOTECHNOLOGY A Force Balance Approach Yoon S. Lee Scientific Information Analyst Chemical Abstracts Service A Division of the American Chemical Society Columbus, Ohio WILEY A JOHN WILEY
SELF-ASSEMBLY AND NANOTECHNOLOGY A Force Balance Approach Yoon S. Lee Scientific Information Analyst Chemical Abstracts Service A Division of the American Chemical Society Columbus, Ohio WILEY A JOHN WILEY
Lecture 5: Macromolecules, polymers and DNA
 1, polymers and DNA Introduction In this lecture, we focus on a subfield of soft matter: macromolecules and more particularly on polymers. As for the previous chapter about surfactants and electro kinetics,
1, polymers and DNA Introduction In this lecture, we focus on a subfield of soft matter: macromolecules and more particularly on polymers. As for the previous chapter about surfactants and electro kinetics,
Supporting Information s for
 Supporting Information s for # Self-assembling of DNA-templated Au Nanoparticles into Nanowires and their enhanced SERS and Catalytic Applications Subrata Kundu* and M. Jayachandran Electrochemical Materials
Supporting Information s for # Self-assembling of DNA-templated Au Nanoparticles into Nanowires and their enhanced SERS and Catalytic Applications Subrata Kundu* and M. Jayachandran Electrochemical Materials
BIOSENOSRS BIO 580. Optical Biosensors- theory part 3 WEEK-9 Fall Semester
 BIOSENOSRS BIO 580 Optical Biosensors- theory part 3 WEEK-9 Fall Semester Faculty: Dr. Javed H. Niazi KM Faculty of Engineering & Natural Sciences Sabanci University Topics that will be covered in the
BIOSENOSRS BIO 580 Optical Biosensors- theory part 3 WEEK-9 Fall Semester Faculty: Dr. Javed H. Niazi KM Faculty of Engineering & Natural Sciences Sabanci University Topics that will be covered in the
Measurements of interaction forces in (biological) model systems
 Measurements of interaction forces in (biological) model systems Marina Ruths Department of Chemistry, UMass Lowell What can force measurements tell us about a system? Depending on the technique, we might
Measurements of interaction forces in (biological) model systems Marina Ruths Department of Chemistry, UMass Lowell What can force measurements tell us about a system? Depending on the technique, we might
Contents. What is AFM? History Basic principles and devices Operating modes Application areas Advantages and disadvantages
 Contents What is AFM? History Basic principles and devices Operating modes Application areas Advantages and disadvantages Figure1: 2004 Seth Copen Goldstein What is AFM? A type of Scanning Probe Microscopy
Contents What is AFM? History Basic principles and devices Operating modes Application areas Advantages and disadvantages Figure1: 2004 Seth Copen Goldstein What is AFM? A type of Scanning Probe Microscopy
METHODS OF INVESTIGATING AT A NANO-LEVEL THE SURFACES OF METALLIC MATERIALS IN LIQUID ENVIRONMENTS
 METHODS OF INVESTIGATING AT A NANO-LEVEL THE SURFACES OF METALLIC MATERIALS IN LIQUID ENVIRONMENTS Marius Dan BENŢA*, Camelia GHIOCEL**, Lizica MODEL** * Transilvania University of Brasov, ** Transport
METHODS OF INVESTIGATING AT A NANO-LEVEL THE SURFACES OF METALLIC MATERIALS IN LIQUID ENVIRONMENTS Marius Dan BENŢA*, Camelia GHIOCEL**, Lizica MODEL** * Transilvania University of Brasov, ** Transport
Nanobiotechnology. Place: IOP 1 st Meeting Room Time: 9:30-12:00. Reference: Review Papers. Grade: 40% midterm, 60% final report (oral + written)
 Nanobiotechnology Place: IOP 1 st Meeting Room Time: 9:30-12:00 Reference: Review Papers Grade: 40% midterm, 60% final report (oral + written) Midterm: 5/18 Oral Presentation 1. 20 minutes each person
Nanobiotechnology Place: IOP 1 st Meeting Room Time: 9:30-12:00 Reference: Review Papers Grade: 40% midterm, 60% final report (oral + written) Midterm: 5/18 Oral Presentation 1. 20 minutes each person
schematic diagram; EGF binding, dimerization, phosphorylation, Grb2 binding, etc.
 Lecture 1: Noncovalent Biomolecular Interactions Bioengineering and Modeling of biological processes -e.g. tissue engineering, cancer, autoimmune disease Example: RTK signaling, e.g. EGFR Growth responses
Lecture 1: Noncovalent Biomolecular Interactions Bioengineering and Modeling of biological processes -e.g. tissue engineering, cancer, autoimmune disease Example: RTK signaling, e.g. EGFR Growth responses
ULTRATHIN ORGANIC FILMS
 An Introduction to ULTRATHIN ORGANIC FILMS From Langmuir-Blodgett to Self-Assembly Abraham Ulman Corporate Research Laboratories Eastman Kodak Company Rochester, New York Academic Press San Diego New York
An Introduction to ULTRATHIN ORGANIC FILMS From Langmuir-Blodgett to Self-Assembly Abraham Ulman Corporate Research Laboratories Eastman Kodak Company Rochester, New York Academic Press San Diego New York
AP Biology: Biochemistry Learning Targets (Ch. 2-5)
 Understand basic principles of chemistry. Distinguish between an element and a compound. Describe the structure of an atom. Compare the various types of chemical bonding. Describe what is meant by a covalent
Understand basic principles of chemistry. Distinguish between an element and a compound. Describe the structure of an atom. Compare the various types of chemical bonding. Describe what is meant by a covalent
Chapter 6 Chemistry in Biology
 Section 1: Atoms, Elements, and Compounds Section 2: Chemical Reactions Section 3: Water and Solutions Section 4: The Building Blocks of Life Click on a lesson name to select. 6.1 Atoms, Elements, and
Section 1: Atoms, Elements, and Compounds Section 2: Chemical Reactions Section 3: Water and Solutions Section 4: The Building Blocks of Life Click on a lesson name to select. 6.1 Atoms, Elements, and
SPHERO TM Magnetic Particles
 SPHER TM Magnetic Particles SPHER TM Magnetic Microparticles provide high quality and reproducible results for your application Allow for rapid and reliable binding between the target and magnetic particle
SPHER TM Magnetic Particles SPHER TM Magnetic Microparticles provide high quality and reproducible results for your application Allow for rapid and reliable binding between the target and magnetic particle
The Chemistry and Energy of Life
 2 The Chemistry and Energy of Life Chapter 2 The Chemistry and Energy of Life Key Concepts 2.1 Atomic Structure Is the Basis for Life s Chemistry 2.2 Atoms Interact and Form Molecules 2.3 Carbohydrates
2 The Chemistry and Energy of Life Chapter 2 The Chemistry and Energy of Life Key Concepts 2.1 Atomic Structure Is the Basis for Life s Chemistry 2.2 Atoms Interact and Form Molecules 2.3 Carbohydrates
Theory of knowledge: Aim 3: Use naming conventions to name ionic compounds.
 Core 44 Essential idea: Ionic compounds consist of ions held together in lattice structures by ionic bonds. 4.1 Ionic bonding and structure Use theories to explain natural phenomena molten ionic compounds
Core 44 Essential idea: Ionic compounds consist of ions held together in lattice structures by ionic bonds. 4.1 Ionic bonding and structure Use theories to explain natural phenomena molten ionic compounds
Atomic weight = Number of protons + neutrons
 1 BIOLOGY Elements and Compounds Element is a substance that cannot be broken down to other substances by chemical reactions. Essential elements are chemical elements required for an organism to survive,
1 BIOLOGY Elements and Compounds Element is a substance that cannot be broken down to other substances by chemical reactions. Essential elements are chemical elements required for an organism to survive,
Valence Electrons, Bonds and Chemical Reactions 05 October 2006
 Valence Electrons, Bonds and Chemical Reactions 05 October 2006 Principles of Valence Electrons and Bonds Ionic Bonds Metallic Bonds Covalent Bonds Intermolecular Forces Common Chemical Reactions Atoms
Valence Electrons, Bonds and Chemical Reactions 05 October 2006 Principles of Valence Electrons and Bonds Ionic Bonds Metallic Bonds Covalent Bonds Intermolecular Forces Common Chemical Reactions Atoms
Unit 1: Chemistry of Life Guided Reading Questions (80 pts total)
 Name: AP Biology Biology, Campbell and Reece, 7th Edition Adapted from chapter reading guides originally created by Lynn Miriello Chapter 1 Exploring Life Unit 1: Chemistry of Life Guided Reading Questions
Name: AP Biology Biology, Campbell and Reece, 7th Edition Adapted from chapter reading guides originally created by Lynn Miriello Chapter 1 Exploring Life Unit 1: Chemistry of Life Guided Reading Questions
Salt vs. Sugar. 1. Ionic Compounds. 2. Molecular Compounds (Cont.) 12/18/2014. What is this Compound You Speak Of? Sodium Chloride Dissolving in Water
 Salt vs. Sugar Unit 7: Chemical Compounds & Formulas Lesson#7.1: Types of Compounds What is this Compound You Speak Of? Compound: Any substance that is formed by the chemical bonding of atoms. We classify
Salt vs. Sugar Unit 7: Chemical Compounds & Formulas Lesson#7.1: Types of Compounds What is this Compound You Speak Of? Compound: Any substance that is formed by the chemical bonding of atoms. We classify
Lecture 2. Fundamentals and Theories of Self-Assembly
 10.524 Lecture 2. Fundamentals and Theories of Self-Assembly Instructor: Prof. Zhiyong Gu (Chemical Engineering & UML CHN/NCOE Nanomanufacturing Center) Lecture 2: Fundamentals and Theories of Self-Assembly
10.524 Lecture 2. Fundamentals and Theories of Self-Assembly Instructor: Prof. Zhiyong Gu (Chemical Engineering & UML CHN/NCOE Nanomanufacturing Center) Lecture 2: Fundamentals and Theories of Self-Assembly
 There are two types of polysaccharides in cell: glycogen and starch Starch and glycogen are polysaccharides that function to store energy Glycogen Glucose obtained from primary sources either remains soluble
There are two types of polysaccharides in cell: glycogen and starch Starch and glycogen are polysaccharides that function to store energy Glycogen Glucose obtained from primary sources either remains soluble
Introduction to Binding Equilibrium Module
 IntroductionToBindingEquilibrium.docx Page 1 Introduction to Binding Equilibrium Module Specific information about the spectrophotometer below will change depending on the make and model used. Avidin and
IntroductionToBindingEquilibrium.docx Page 1 Introduction to Binding Equilibrium Module Specific information about the spectrophotometer below will change depending on the make and model used. Avidin and
Chapter 10. The Liquid and Solid States. Introduction. Chapter 10 Topics. Liquid-Gas Phase Changes. Physical State of a Substance
 Introduction Chapter 10 The Liquid and Solid States How do the properties of liquid and solid substances differ? How can we predict properties based on molecular- level structure? Glasses Wires Reshaping
Introduction Chapter 10 The Liquid and Solid States How do the properties of liquid and solid substances differ? How can we predict properties based on molecular- level structure? Glasses Wires Reshaping
Protein Structure Analysis and Verification. Course S Basics for Biosystems of the Cell exercise work. Maija Nevala, BIO, 67485U 16.1.
 Protein Structure Analysis and Verification Course S-114.2500 Basics for Biosystems of the Cell exercise work Maija Nevala, BIO, 67485U 16.1.2008 1. Preface When faced with an unknown protein, scientists
Protein Structure Analysis and Verification Course S-114.2500 Basics for Biosystems of the Cell exercise work Maija Nevala, BIO, 67485U 16.1.2008 1. Preface When faced with an unknown protein, scientists
Facile Synthesis of Gold Wavy Nanowires and Investigation of
 Supporting Information for Facile Synthesis of Gold Wavy Nanowires and Investigation of Their Growth Mechanism Cun Zhu,, Hsin-Chieh Peng, Jie Zeng, Jingyue Liu, Zhongze Gu and Younan Xia,,* The Wallace
Supporting Information for Facile Synthesis of Gold Wavy Nanowires and Investigation of Their Growth Mechanism Cun Zhu,, Hsin-Chieh Peng, Jie Zeng, Jingyue Liu, Zhongze Gu and Younan Xia,,* The Wallace
Essential Knowledge. 2.A.3 Organisms must exchange matter with the environment to grow, reproduce and maintain organization
 Ch3: Water Essential Knowledge 2.A.3 Organisms must exchange matter with the environment to grow, reproduce and maintain organization a. Molecules and atoms from the environment are necessary to build
Ch3: Water Essential Knowledge 2.A.3 Organisms must exchange matter with the environment to grow, reproduce and maintain organization a. Molecules and atoms from the environment are necessary to build
Hole s Human Anatomy and Physiology Eleventh Edition. Chapter 2
 Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 2 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. CHAPTER 2 CHEMICAL BASIS OF
Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 2 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. CHAPTER 2 CHEMICAL BASIS OF
What determines whether a substance will be a solid, liquid, or gas? Thursday, April 24, 14
 What determines whether a substance will be a solid, liquid, or gas? Answer: The attractive forces that exists between its particles. Answer: The attractive forces that exists between its particles. For
What determines whether a substance will be a solid, liquid, or gas? Answer: The attractive forces that exists between its particles. Answer: The attractive forces that exists between its particles. For
Chem 1075 Chapter 13 Liquids and Solids Lecture Outline
 Chem 1075 Chapter 13 Liquids and Solids Lecture Outline Slide 2-3 Properties of Liquids Unlike gases, liquids respond dramatically to temperature and pressure changes. We can study the liquid state and
Chem 1075 Chapter 13 Liquids and Solids Lecture Outline Slide 2-3 Properties of Liquids Unlike gases, liquids respond dramatically to temperature and pressure changes. We can study the liquid state and
Model Worksheet Teacher Key
 Introduction Despite the complexity of life on Earth, the most important large molecules found in all living things (biomolecules) can be classified into only four main categories: carbohydrates, lipids,
Introduction Despite the complexity of life on Earth, the most important large molecules found in all living things (biomolecules) can be classified into only four main categories: carbohydrates, lipids,
Water is one of the few compounds found in a liquid state over most of Earth s surface.
 The Water Molecule Water is one of the few compounds found in a liquid state over most of Earth s surface. Like other molecules, water (H2O) is neutral. The positive charges on its 10 protons balance out
The Water Molecule Water is one of the few compounds found in a liquid state over most of Earth s surface. Like other molecules, water (H2O) is neutral. The positive charges on its 10 protons balance out
Material Chemistry KJM 3100/4100. Synthetic Polymers (e.g., Polystyrene, Poly(vinyl chloride), Poly(ethylene oxide))
 Material Chemistry KJM 3100/4100 Lecture 1. Soft Materials: Synthetic Polymers (e.g., Polystyrene, Poly(vinyl chloride), Poly(ethylene oxide)) Biopolymers (e.g., Cellulose derivatives, Polysaccharides,
Material Chemistry KJM 3100/4100 Lecture 1. Soft Materials: Synthetic Polymers (e.g., Polystyrene, Poly(vinyl chloride), Poly(ethylene oxide)) Biopolymers (e.g., Cellulose derivatives, Polysaccharides,
Definition of Matter. Subatomic particles 8/20/2012
 Interplay of Biology and Chemistry Here is a link to the video these beetles are fairly common locally an amazing adaptation, and a good example of chemistry and physics in biology. Also look for creationist-evolutionist
Interplay of Biology and Chemistry Here is a link to the video these beetles are fairly common locally an amazing adaptation, and a good example of chemistry and physics in biology. Also look for creationist-evolutionist
Nanotechnology Fabrication Methods.
 Nanotechnology Fabrication Methods. 10 / 05 / 2016 1 Summary: 1.Introduction to Nanotechnology:...3 2.Nanotechnology Fabrication Methods:...5 2.1.Top-down Methods:...7 2.2.Bottom-up Methods:...16 3.Conclusions:...19
Nanotechnology Fabrication Methods. 10 / 05 / 2016 1 Summary: 1.Introduction to Nanotechnology:...3 2.Nanotechnology Fabrication Methods:...5 2.1.Top-down Methods:...7 2.2.Bottom-up Methods:...16 3.Conclusions:...19
Mn 12 Single-Molecule Magnet Aggregates as Magnetic Resonance Imaging Contrast Agents
 Electronic Supplementary Information Mn 12 Single-Molecule Magnet Aggregates as Magnetic Resonance Imaging Contrast Agents Yinglin Wang, a Wen Li, a Shengyan Zhou, b Daliang Kong, b Haishan Yang, b and
Electronic Supplementary Information Mn 12 Single-Molecule Magnet Aggregates as Magnetic Resonance Imaging Contrast Agents Yinglin Wang, a Wen Li, a Shengyan Zhou, b Daliang Kong, b Haishan Yang, b and
BIOCHEMISTRY NOTES - UNIT 2-
 BIOCHEMISTRY NOTES - UNIT 2- ATOMS - the basic unit of matter. Contains subatomic particles o (+ charge) o (no charge/neutral) o (- charge) Protons and neutrons have about the same mass. Electrons are
BIOCHEMISTRY NOTES - UNIT 2- ATOMS - the basic unit of matter. Contains subatomic particles o (+ charge) o (no charge/neutral) o (- charge) Protons and neutrons have about the same mass. Electrons are
Controlled self-assembly of graphene oxide on a remote aluminum foil
 Supplementary Information Controlled self-assembly of graphene oxide on a remote aluminum foil Kai Feng, Yewen Cao and Peiyi Wu* State key Laboratory of Molecular Engineering of Polymers, Department of
Supplementary Information Controlled self-assembly of graphene oxide on a remote aluminum foil Kai Feng, Yewen Cao and Peiyi Wu* State key Laboratory of Molecular Engineering of Polymers, Department of
BIBC 100. Structural Biochemistry
 BIBC 100 Structural Biochemistry http://classes.biology.ucsd.edu/bibc100.wi14 Papers- Dialogue with Scientists Questions: Why? How? What? So What? Dialogue Structure to explain function Knowledge Food
BIBC 100 Structural Biochemistry http://classes.biology.ucsd.edu/bibc100.wi14 Papers- Dialogue with Scientists Questions: Why? How? What? So What? Dialogue Structure to explain function Knowledge Food
Name Biology Chapter 2 Note-taking worksheet
 Name Biology Chapter 2 Note-taking worksheet The Nature of Matter 1. Life depends on Atoms 1. The study of chemistry starts with the basic unit of matter, the. 2. The atom was first used by the Greek philosopher
Name Biology Chapter 2 Note-taking worksheet The Nature of Matter 1. Life depends on Atoms 1. The study of chemistry starts with the basic unit of matter, the. 2. The atom was first used by the Greek philosopher
Supporting Information
 Supporting Information Capping Agent-Free Gold Nanostars Show Greatly Increased Versatility And Sensitivity For Biosensing Debrina Jana, Carlos Matti, Jie He, and Laura Sagle* Department of Chemistry,
Supporting Information Capping Agent-Free Gold Nanostars Show Greatly Increased Versatility And Sensitivity For Biosensing Debrina Jana, Carlos Matti, Jie He, and Laura Sagle* Department of Chemistry,
The Chemical Context of Life
 CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 2 The Chemical Context of Life Questions prepared by Douglas Darnowski, Indiana University Southeast James Langeland, Kalamazoo College Murty
CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 2 The Chemical Context of Life Questions prepared by Douglas Darnowski, Indiana University Southeast James Langeland, Kalamazoo College Murty
- intermolecular forces forces that exist between molecules
 Chapter 11: Intermolecular Forces, Liquids, and Solids - intermolecular forces forces that exist between molecules 11.1 A Molecular Comparison of Liquids and Solids - gases - average kinetic energy of
Chapter 11: Intermolecular Forces, Liquids, and Solids - intermolecular forces forces that exist between molecules 11.1 A Molecular Comparison of Liquids and Solids - gases - average kinetic energy of
From Polymer Gel Nanoparticles to Nanostructured Bulk Gels
 From Polymer Gel Nanoparticles to Nanostructured Bulk Gels Zhibing Hu Departments of Physics and Chemistry, University of North Texas Denton, TX 76203, U. S. A. Phone: 940-565 -4583, FAX: 940-565-4824,
From Polymer Gel Nanoparticles to Nanostructured Bulk Gels Zhibing Hu Departments of Physics and Chemistry, University of North Texas Denton, TX 76203, U. S. A. Phone: 940-565 -4583, FAX: 940-565-4824,
Outline Scanning Probe Microscope (SPM)
 AFM Outline Scanning Probe Microscope (SPM) A family of microscopy forms where a sharp probe is scanned across a surface and some tip/sample interactions are monitored Scanning Tunneling Microscopy (STM)
AFM Outline Scanning Probe Microscope (SPM) A family of microscopy forms where a sharp probe is scanned across a surface and some tip/sample interactions are monitored Scanning Tunneling Microscopy (STM)
Study of monomolecular layers of azobenzene derivative by the scanning probe microscopy
 Materials Science and Engineering C 22 (2002) 453 458 www.elsevier.com/locate/msec Study of monomolecular layers of azobenzene derivative by the scanning probe microscopy A.M. Alexeev*, E.A. Kosobrodova,
Materials Science and Engineering C 22 (2002) 453 458 www.elsevier.com/locate/msec Study of monomolecular layers of azobenzene derivative by the scanning probe microscopy A.M. Alexeev*, E.A. Kosobrodova,
Water. Dr. Diala Abu-Hassan, DDS, PhD Lecture 2 MD summer Dr. Diala Abu-Hassan
 Water, DDS, PhD Dr.abuhassand@gmail.com Lecture 2 MD summer 2014 1 Lecture Content Importance of water in biological systems Noncovalent interactions Water structure Water properties Water as a solvent
Water, DDS, PhD Dr.abuhassand@gmail.com Lecture 2 MD summer 2014 1 Lecture Content Importance of water in biological systems Noncovalent interactions Water structure Water properties Water as a solvent
Presentation Microcalorimetry for Life Science Research
 Presentation Microcalorimetry for Life Science Research MicroCalorimetry The Universal Detector Heat is either generated or absorbed in every chemical process Capable of thermal measurements over a wide
Presentation Microcalorimetry for Life Science Research MicroCalorimetry The Universal Detector Heat is either generated or absorbed in every chemical process Capable of thermal measurements over a wide
