Secondary Cell Wall Deposition Causes Radial Growth of Fibre Cells in the Maturation Zone of Elongating Tall Fescue Leaf Blades
|
|
|
- Marybeth Harrison
- 5 years ago
- Views:
Transcription
1 Annals of Botany 89: 89±96, 2002 doi: /aob.2002.mcf010, available online at Secondary Cell Wall Deposition Causes Radial Growth of Fibre Cells in the Maturation Zone of Elongating Tall Fescue Leaf Blades JENNIFER W. MACADAM* and CURTIS J. NELSON Department of Agronomy, University of Missouri, Columbia, Missouri 65211, USA Received: 5 June 2001 Returned for revision: 26 August 2001 Accepted: 21 September 2001 A gradient of development consisting of successive zones of cell division, cell elongation and cell maturation occurs along the longitudinal axis of elongating leaf blades of tall fescue (Festuca arundinacea Schreb.), a C 3 grass. An increase in speci c leaf weight (SLW; dry weight per unit leaf area) in the maturation region has been hypothesized to result from deposition of secondary cell walls in structural tissues. Our objective was to measure the transverse cell wall area (CWA) associated with the increase in SLW, which occurs following the cessation of leaf blade elongation at about 25 mm distal to the ligule. Digital image analysis of transverse sections at 5, 15, 45, 75 and 105 mm distal to the ligule was used to determine cell number, cell area and protoplast area of structural tissues, namely bre bundles, mestome sheaths and xylem vessel elements, along the developmental gradient. Cell diameter, protoplast diameter and area, and cell wall thickness and area of bre bundle cells were calculated from these data. CWA of structural tissues increased in sections up to 75 mm distal to the ligule, con- rming the role of cell wall deposition in the increase in SLW (r 2 = 0 924; P < 0 01). However, protoplast diameter of bre cells did not decrease signi cantly as CWA increased, although mean thickness of bre cell walls increased by 95 % between 15 and 105 mm distal to the ligule. Therefore, secondary cell wall deposition in bre bundles of tall fescue leaf blades resulted in continued radial expansion of bre cells rather than in a decrease in protoplast diameter. ã 2002 Annals of Botany Company Key words: Festuca arundinacea Schreb., tall fescue, secondary cell wall deposition, speci c leaf weight (SLW), structural cell wall area (CWA), bre cell protoplast diameter, digital image analysis, sclerenchyma bre bundle development. INTRODUCTION During elongation growth of grass leaf blades, tissue is displaced from the leaf base by cell division and elongation. Progressive cell differentiation can be observed in cell les along the developmental gradient from the base of the leaf blade to the tip. In tall fescue (Festuca arundinacea Schreb.), speci c leaf weight (SLW; dry weight per unit leaf area) rst decreases along this gradient in the basal portion of the leaf blade as newly divided cells are displaced through the epidermal cell elongation zone, then increases following the cessation of elongation (MacAdam and Nelson, 1987). The decrease in SLW associated with elongation is due to a decrease in the density of transverse cell walls per unit leaf area. The increase in SLW following cessation of cell elongation is thought to result from the deposition of secondary cell walls in structural tissues (MacAdam and Nelson, 1987). In standard descriptions of the development of sclerenchyma bre cells, the secondary wall is laid down after cell expansion has ceased, and secondary wall deposition occurs at the expense of protoplast transverse area (Brett and Waldron, 1996). However, in the maturation region of tall fescue, where increase in SLW is most likely due to deposition of secondary cell walls, leaf width also continues to increase (MacAdam and Nelson, 1987). This suggests * For correspondence: Department of Plants, Soils, and Biometeorology, Utah State University, Logan, UT , USA. jenmac@ cc.usu.edu that all cells, including those with secondary walls, must still be capable of radial expansion. There is evidence that differentiation of relatively mature bre cells contributes to leaf curvature of maize (Zea mays L.) via shortening of cells (Hay et al., 2000). In sorghum [Sorghum bicolor (L.) Moench], both proto- and metaxylem elements in leaves affected by salinity were narrower than those in controls. This reduced water deposition in expanding cells and could thus reduce the rate of cell growth (Baum et al., 2000). These studies suggest that secondary growth in herbaceous plants may contribute to both form and function in ways not generally recognized. In this study we sought to document the correlation between secondary cell wall deposition and the increase in SLW in the maturation region of tall fescue by direct measurement of the transverse cell wall area of structural tissue. We used these data to determine the relationship between wall thickness and protoplast diameter of bre bundle cells along the developmental gradient of elongating leaf blades. MATERIALS AND METHODS Plant growth conditions Leaf blades used in this study were from vegetatively propagated plants of a single genotype of tall fescue (Festuca arundinacea Schreb.), designated low yield per tiller (LYT) in previous studies (Vassey, 1986; MacAdam ã 2002 Annals of Botany Company
2 90 MacAdam and Nelson Ð Secondary Cell Wall Deposition During Grass Leaf Development and Nelson, 1987). Three vegetative tillers were transplanted into plastic pots (11 cm diameter, 15 cm deep) containing a sphagnum peat moss±vermiculite potting mixture (Terra-Lite Redi-Earth Peat-Lite; Scotts-Sierra Horticultural Products Co., Marysville, OH, USA). Plants were grown for 15 weeks in a controlled-environment chamber. Mean photosynthetic photon ux density at canopy height was 490 mmol m ±2 s ±1, photoperiod was 14 h, day/night temperature was a constant 22 C and relative humidity was 70 %. Plants were watered daily to eld capacity, and fertilized once a week with 100 ml per pot of Hoagland's No. 2 nutrient solution (Epstein, 1972). Leaf growth measurements The tips of elongating leaves used in this study had emerged from the sheaths of older leaves; the length of these sheaths was about 60 mm. The overall length of the elongating leaves was less than half that of the blade of the next-oldest leaf. At this stage of development cell division and cell elongation are both continuing at the base of the leaf blade and a ligule has differentiated approx. 1 mm distal to the point of attachment of the leaf to the shoot apex. The leaf elongation rate is nearly constant (Schnyder and Nelson, 1988), and the sheath of the elongating leaf has not begun to elongate. The abaxial epidermis is exposed in rolled, elongating leaves of tall fescue as they emerge from the sheaths of older leaves. Epidermal cell lengths of leaves used in this study were determined from replicas of the abaxial epidermis of eight elongating leaves selected for uniform length. The full length of elongating leaves was exposed by removing older leaf sheaths, and leaves were excised from the plant at the point of attachment to the shoot apex. The ligule was marked using the tip of a needle dipped in a 1 % (w/v) aqueous solution of acid fuchsin. To form the replica, a thin coating of a 4 % (w/v) solution of polyvinylformaldehyde (Formvar; Ted Pella Co., Redding, CA, USA) in chloroform was spread along the leaf using a glass rod and allowed to dry for approx. 5 s. A strip of clear Cellophane adhesive tape was applied along the length of the leaf, the replica was lifted off, and the Cellophane tape and replica were applied to a glass microscope slide (Schnyder et al., 1990). Formvar is routinely used to make replicas for electron microscopy, and accurately reproduces surface features without distortion (Schaefer and Harker, 1942). The lengths of ve intercostal epidermal cells were measured at successive 5 mm intervals distal to the ligule on each replica. The pattern of change in SLW of elongating tall fescue leaf blades had been established in earlier replicated studies (MacAdam and Nelson, 1987; Schnyder and Nelson, 1988). For this study, SLW of segments along the elongating leaf blade was determined using just six leaves selected for uniform length to verify previous data for SLW. Each leaf blade was cut into 10-mm-long segments from the ligule to the tip, and the width at the middle of each segment was determined with an ocular micrometer. Segments were pooled by location along the leaf blade, dried for 48 h at 60 C, allowed to cool under desiccation, and dry weights were determined. Measurement of structural cell wall area Transverse segments 1 mm long and the full width of the leaf blade were excised from three elongating leaves at successive 5 mm intervals from the ligule to 105 mm distal to the ligule. Segments were xed overnight in a solution of 4 % (v/v) formaldehyde and 1 % (v/v) glutaraldehyde in 0 2 M Na-phosphate buffer, ph 7 2 (Burns and Bretschneider, 1981). Segments were dehydrated in an ethanol series and in ltrated for two 3 h periods with catalysed glycol methacrylate (JB-4; Polysciences, Warrington, PA, USA). Segments were in ltrated for an additional 10 min in one change of catalysed JB-4 saturated with acid fuchsin to aid orientation. Segments were rinsed in clear, catalysed JB-4, then transferred to fresh, catalysed, activated JB-4 and allowed to polymerize at room temperature. After hardening, blocks were xed to wooden dowels with epoxy resin and sectioned. Differential shrinking and swelling of tissues within a gradient of development is a potential problem in a study such as this. More recent observations of tissue from the same genotype of tall fescue xed in glutaraldehyde and osmium tetroxide and embedded in Spurr's (1969) resin show the same relative dimensions of bre and other cells as found in this study (see photomicrographs in MacAdam et al., 1992). Transverse sections 5 mm thick were cut using a rotary microtome equipped with a glass knife. Sections were stained for 1 min in a 1 % (w/v) aqueous solution of acid fuchsin, then rinsed, dried and stained for 1 min in a 0 05 % (w/v) solution of toluidine blue O in benzoate buffer, ph 4 4 (O'Brien and McCully, 1981). Segments at 5, 15, 45, 75 and 105 mm were chosen for measurements of structural cell wall area (CWA). There is no distinct midrib in tall fescue leaf blades, and ridges containing major (with xylem) or minor (without xylem) veins alternate across the width of the leaf blade, with a minor vein usually located at each margin. After a preliminary determination of ridge number and type (major vs. minor vein) across the leaf blade at each location, the same two adjacent ridges from a given leaf were photographed at each location and used to measure structural CWA. Selected ridges were located at least two ridges away from the margin of each leaf. In a C 3 grass such as tall fescue, secondary cell walls are deposited in various tissues, including bre cells, xylem elements, along the inner wall of the mestome (inner bundle) sheath, and along the outer wall of the abaxial (lower) epidermis (Hanna et al., 1973). Individual micrographs were made of each sclerenchyma bre bundle in adjacent major and minor veins at each location. Sections were photographed using a Zeiss Photomicroscope III with a 633 objective, and nal magni cation was determined from photomicrographs of a stage micrometer taken using the same objective. Micrographs were printed at a nal magni cation of 9943 for image analysis, and Digital Paintbrush (The Computer Colorworks, Sausalito, CA, USA) was used as a planimeter and digitizer.
3 MacAdam and Nelson Ð Secondary Cell Wall Deposition During Grass Leaf Development 91 F IG. 1. Epidermal cell length and speci c leaf weight (SLW) of elongating tall fescue leaf blades of the LYT genotype. Speci c leaf weight is dry weight per unit leaf area. For this study, SLW was calculated for a pooled sample of six leaves. Cell length LSD 0 05 = 127 mm, n =8. Total area was calculated for each of the three sclerenchyma bre bundles in each vein, namely the adaxial or upper bre bundle, the abaxial or lower bre bundle, and the vascular bre bundle, located between the metaxylem vessel elements. The area of each protoplast of all cells within each bre bundle was also measured and total protoplast area was subtracted from total bre bundle area to determine CWA. No air spaces were noted between cells of bre bundles. Secondary cell wall deposition has been determined by other means, such as by measuring the thickness of the double wall between two bre cells in internodes of reproductive tillers of darnel (Lolium temulentum L.). In this case, the thickness of secondary walls of bre cells increased linearly with distance distal to the node (Juniper et al., 1981). However, this approach would not have detected change in protoplast diameter or cell number during leaf development. More automated methods such as image skeletonization (Travis et al., 1993) have been used to estimate CWA, and such an approach would allow more samples to be examined. However, it is not readily adapted to measurement of irregular wall shapes such as the inner wall of the mestome sheath. F IG. 2. Photomicrographs of transverse sections of a major and minor ridge at 5 (A), 15 (B), 45 (C) and 75 (D) mm distal to the ligule of an elongating leaf blade. The arrow in A indicates a ligni ed protoxylem element; arrows in B indicate bre bundles with thickened cell wall junctions. Veins in minor ridges do not develop metaxylem vessel elements. b, Abaxial bre bundle; d, adaxial bre bundle; m, mestome sheath; v, vascular bre bundle; M, metaxylem element; P, protoxylem element. Bar = 50 mm.
4 92 MacAdam and Nelson Ð Secondary Cell Wall Deposition During Grass Leaf Development F IG. 3. Photomicrographs of transverse sections of the abaxial bre bundle and the lower half of the mestome sheath from the same major ridge of one elongating leaf blade at 45 (A) and 75 (B) mm distal to the ligule. Bar = 20 mm. To determine mean wall thickness for cells within a given bre bundle, we rst calculated mean bre cell area (total bre bundle area divided by the number of bre cells in that bundle). Cell area was used to calculate mean bre cell diameter, assuming a circular shape, which is generally representative. Similarly, protoplast diameter was calculated from mean protoplast area, again assuming a circular shape. Protoplast radius was subtracted from bre cell radius to give mean cell wall thickness. Dimensions of CWAs for proto- and metaxylem vessels were calculated similarly. The CWA of the thickened inner wall of the mestome sheath was measured directly. At the light microscope level, the thickened outer wall of the abaxial epidermis could not be distinguished from cuticular wax so the contribution of this tissue was not estimated. Of the cells that developed ligni ed secondary cell walls, those adaxial to the mestome sheath were included in the adaxial bre bundle and those abaxial to the mestome sheath were included in the abaxial bre bundle. The total of the measured structural CWAs for a leaf blade at a given location distal to the ligule was estimated by multiplying all measured structural CWAs for one major and one minor ridge by the number of ridges of that type occurring across the leaf blade at that location. RESULTS At 5 mm distal to the ligule, epidermal cells were approx. one-tenth of their nal length (Fig. 1). Ligni ed cell walls appear green when stained with toluidine blue (O'Brien et al., 1964), and at 5 mm distal to the ligule only walls of protoxylem vessel elements of major ridges were stained green (Fig. 2A). As epidermal cells elongated, tissue was also displaced through the basal 25 mm of the leaf blade by continuing basal leaf growth (Fig. 1). Near the centre of the elongation zone, at 15 mm distal to the ligule for this genotype, cell elongation is at its maximal rate (MacAdam FIG. 4. Using weighted means of cells in sclerenchyma bre bundles, bre cell area was 76 mm 2 and bre protoplast area was 47 mm 2 at 45 mm distal to the ligule (A) while bre cell area was 89 mm 2 and protoplast area was 50 mm 2 at 75 mm distal to the ligule (B). Increase in cell wall thickness occurred primarily by increase in the outer diameter of bre cells rather than by a decrease in diameter of bre protoplasts. et al., 1989). At this location, cells in bre bundles had thin walls but some thickening could be observed at cell junctions (Fig. 2B). Speci c leaf weight increased about 40 % between 25 mm, where epidermal cell elongation ceased, and 85 mm distal to the ligule (Fig. 1). When leaf blade tissue was displaced to 45 mm (Fig. 2C), well into the region of increasing SLW, green staining of secondary cell walls was visible throughout the vascular bundle and the bundle sheath extension that connects the mestome sheath with the adaxial and abaxial bundle bres in major veins (Metcalfe, 1960). At 75 mm distal to the ligule (Fig. 2D), metaxylem elements and all bre bundles had thickened, green-stained cell walls.
5 MacAdam and Nelson Ð Secondary Cell Wall Deposition During Grass Leaf Development 93 TABLE 1. Mean wall thickness (mm), protoplast diameter (mm), and number of cells in sclerenchyma bre bundles at ve locations along elongating leaf blades Location distal to the ligule (mm) Sclerenchyma bre bundle P LSD 0.05 (n =3) Major veinsðnumber across leaf: Fibre cell number Adaxial bundle ± Vascular bundle ± Abaxial bundle Wall thickness (mm) Adaxial bundle Vascular bundle Abaxial bundle Protoplast diameter (mm) Adaxial bundle ± Vascular bundle ± Abaxial bundle ± Minor veinsðnumber across leaf: Fibre cell number Adaxial bundle ± Vascular bundle ± Abaxial bundle ± Wall thickness (mm) Adaxial bundle Vascular bundle Abaxial bundle Protoplast diameter (mm) Adaxial bundle ± Vascular bundle ± Abaxial bundle ± Means of major and minor vein bre cells Mean bre cell number per leaf ± Weighted mean wall thickness (mm) Mean wall thickness (mm) Weighted mean protoplast diameter (mm) Mean protoplast diameter (mm) ± Mean cell number is a sum of all bre cells across the leaf (n = 3). Weighted means were calculated using the number of each type of cell per bre bundle, and were not substantially different from statistical means. Staining was consistent with the assumption that protoxylem elements have ligni ed cell wall thickenings in meristematic tissue, and that ligni cation occurs in secondary cell walls of bre cells as successive wall layers are deposited following cessation of cell elongation. The hexagonal appearance of bre cells in the abaxial bundle sheath at 45 mm distal to the ligule (Fig. 3A) resulted from wall thickening at cell junctions, and protoplast shape appeared similar to wall shape. As more secondary cell wall was deposited in bre cells as they were displaced to 75 mm distal to the ligule (Fig. 3B), some bre cell protoplasts appeared more circular, while the shared walls within bre bundles remained pentagonal or hexagonal. The overall change in appearance and mean increase in diameter of bre bundle cells from 45 to 75 mm is also illustrated in Fig. 4. Between 75 and 105 mm, there was little change in either SLW (Fig. 1) or in the appearance of bre cell walls, so a section at 105 mm was not included in Fig. 2. Total structural CWA in transverse sections (Fig. 5) increased between 5 and 75 mm distal to the ligule (P = 0 02), with the greatest incremental change occurring between 45 and 75 mm distal to the ligule. The regression coef cient (r 2 ) of SLW with structural CWA was (P < 0 01). The largest absolute increase in CWA was in abaxial bre bundles of major veins, while the largest percentage increase occurred in CWA of minor veins. Leaf width continued to increase beyond the distal margin of the elongation zone, which was probably due to continued radial cell growth. However, in a study of elongating leaf blades, the contribution of radial cell expansion to leaf width is confounded by the tapering of leaf width due to a decrease in vein number towards the leaf tip. From 75 to 105 mm the cell wall area of individual bundle sheaths remained constant or increased. The decrease in total structural CWA of major and minor abaxial bre bundle fractions (Fig. 5) resulted from a decrease in the number of cells in these bre bundles (Table 1). This decrease in cell number re ects the normal ontogeny of tall fescue leaf blades, which taper toward the leaf tip. Because the number of veins across the leaf decreases gradually towards the tip, data for wall thickness and protoplast diameter are presented on a per-cell basis (Table 1). The weighted mean thickness of sclerenchyma bre bundle cell walls, calculated using the number of each type of cell per bre bundle, nearly doubled between 15 and 105 mm distal to the ligule, from 0 80 to 1 44 mm (Table 1).
6 94 MacAdam and Nelson Ð Secondary Cell Wall Deposition During Grass Leaf Development FIG. 5. Transverse structural cell wall area (CWA) per leaf at ve locations along the elongating leaf blade and CWA of the contributing structural tissues. Data for individual structural tissues from one major and one minor ridge from each leaf were multiplied by the number of ridges of that type occurring across the width of that leaf to estimate total structural CWA. LSD 0 05 = mm 2 to compare total structural CWA at different locations along the leaf blade (n = 3). F IG. 6. Development of vessel elements in the elongating leaf blade. Longitudinal sections of protoxylem elements at 6 (A) and 15 (B) mm distal to the ligule. Elongation increases the distance between annular wall thickenings of protoxylem vessels. Bar = 50 mm. C, Transverse CWA per leaf of xylem vessel elements at ve locations distal to the ligule. Mean metaxylem wall thickness at each location is indicated numerically on the gure; metaxylem cell wall thickness LSD 0 05 = 0 90 mm (n = 3).
7 MacAdam and Nelson Ð Secondary Cell Wall Deposition During Grass Leaf Development 95 Contrary to the expectation that secondary cell wall thickening would occur at the expense of the diameter of bre cell protoplasts, there was no consistent decrease in mean bre cell protoplast diameter as walls thickened following cessation of elongation at 25 mm distal to the ligule. As leaf tissue was displaced from 15 to 105 mm distal to the ligule, the 0 64 mm (80 %) increase in weighted mean wall thickness was primarily achieved by an increase in overall cell diameter rather than by a decrease in protoplast diameter: sclerenchyma bre cell diameter increased from 9 52 to mm (10 %), while protoplast diameter only decreased from 7 92 to 7 61 mm (4 %). The ligni ed annular wall thickenings of protoxylem vessel elements in tall fescue leaf blades resemble a coiled spring (Fig. 6A). As these cells were displaced through the elongation zone their longitudinal walls continued to elongate with adjacent tissues and distance between annular wall thickenings increased (Fig. 6B). There was progressively less probability of sectioning through a wall-thickening of a protoxylem element in transverse section as location distal to the ligule increased (Fig. 6C). In contrast, metaxylem cells gained thick secondary walls and walls increased in cross-sectional area between 5 (Fig. 2A) and 45 mm (Fig. 2C). Both xylem CWA and mestome sheath inner wall area are included in calculations of total transverse structural cell wall area. DISCUSSION These data demonstrate that synthesis of secondary cell walls in structural tissues of elongating tall fescue leaf blades takes place between 15 and 75 mm distal to the ligule, in the same region where increase in speci c leaf weight occurs. This work con rms, therefore, that structural cell wall deposition accounts for the majority of the increase in SLW. In addition, but more surprisingly, deposition of secondary cell walls was found to increase the transverse area of bre cells rather than to decrease signi cantly bre cell protoplast area. This increase in cell diameter probably contributes to the continuing increase in leaf width following cessation of longitudinal growth observed in tall fescue leaf blades by MacAdam and Nelson (1987) and Maurice et al. (1997). In rapidly expanding plant cells, the middle lamella and its adjacent primary cell wall must expand to accommodate both longitudinal and radial growth, while in the standard de nition, a secondary cell wall is deposited after expansion of the primary cell wall has ceased (Esau, 1977). However, the progress of secondary cell wall development in bre cells in our study resembled collenchyma cell development, in which cell wall thickening begins at cell junctions and results in radial expansion that continues following the cessation of cell elongation (Vian et al., 1993). Our results suggest that as cell elongation stops and wall component synthesis shifts, by de nition, from primary to secondary cell wall deposition, radial expansion of bre cells in tall fescue leaf blades continues despite evidence of ligni cation in these tissues. While it is often assumed that ligni cation occurs only after cell wall growth has ceased, this is apparently not the case. Using polyclonal antibodies that detect uncondensed lignin, MuÈsel et al. (1997) reported the presence of lignin throughout the primary walls of rapidly expanding maize coleoptile parenchyma cells. Furthermore, in elongating maize internodes, where cell wall carbohydrates were accumulating in the basal 40 % of internodes, lignin content increased from 35 to 59 mg g ±1, or to about 80 % of the maximum lignin content of 73 mg g ±1 in these tissues (calculations from data of Scobbie et al., 1993). Thus, neither deposition of secondary cell walls nor ligni cation should continue to be considered mutually exclusive with cell wall expansion. ACKNOWLEDGEMENTS The authors thank P. R. Beuselinck and J. P. Gustafson (USDA-ARS University of Missouri) for access to microscopes, D. W. Sisson (Utah State University) for statistical analyses, and R. J. Mueller (Utah State University) for helpful comments on the manuscript. Contribution of the Missouri Agriculture Experiment Station, Journal Series No LITERATURE CITED Baum SF, Tran PN, Silk WK Effects of salinity on xylem structure and water use in growing leaves of sorghum. New Phytologist 146: 119±197. Brett CT, Waldron KW Physiology and biochemistry of plant cell walls. 2nd edn. London: Chapman and Hall. Burns WA, Bretschneider A Thin is in: plastic embedding of tissue for light microscopy. Chicago: American Society of Clinical Pathologists. Epstein E Mineral nutrition of plants: principles and perspectives. New York: Wiley. Esau K Anatomy of seed plants. 2nd edn. New York: Wiley. Hanna WW, Monson WG, Burton GW Histological examination of fresh forage leaves after in vitro digestion. Crop Science 13: 90± 102. Hay JO, Moulia B, Lane B, Freeling M, Silk WK Biomechanical analysis of the Rolled (Rld) leaf phenotype of maize. American Journal of Botany 87: 625±633. Juniper BE, Lawton JR, Harris PJ Cellular organelles and cellwall formation in bres from the owering stem of Lolium temulentum L. New Phytologist 89: 609±619. MacAdam JW, Nelson CJ Speci c leaf weight in zones of cell division, elongation and maturation in tall fescue leaf blades. Annals of Botany 59: 369±376. MacAdam JW, Nelson CJ, Sharp RE Peroxidase activity in the leaf elongation zone of tall fescue. 1. Spatial distribution of ionically bound peroxidase activity in genotypes differing in length of the elongation zone. Plant Physiology 99: 872±878. MacAdam JW, Volenec JJ, Nelson CJ Effects of nitrogen on mesophyll cell division and epidermal cell elongation in tall fescue leaf blades. Plant Physiology 89: 549±556. Maurice I, Gastal F, Durand J-L Generation of form and associated mass deposition during leaf development in grasses: a kinematic approach for non-steady growth. Annals of Botany 80: 673±683. Metcalfe CR Anatomy of the monocotyledons. I. Gramineae. London and Oxford: Clarendon Press. MuÈsel G, Schindler T, Bergfeld R, Ruel K, Jacquet G, Lapierre C, Speth V, Schopfer P Structure and distribution of lignin in primary and secondary cell walls of maize coleoptiles analyzed by chemical and immunological probes. Planta 201: 146±159. O'Brien TP, McCully ME The study of plant structure. Principles and selected methods. Melbourne: Termarcarphi Pty Ltd.
8 96 MacAdam and Nelson Ð Secondary Cell Wall Deposition During Grass Leaf Development O'Brien TP, Feder N, McCully ME Polychromatic staining of plant cell walls by toluidine blue O. Protoplasma 59: 368±373. Schaefer VJ, Harker D Surface replicas for use in the electron microscope. Journal of Applied Physics 13: 427±433. Schnyder H, Nelson CJ Diurnal growth of tall fescue leaf blades. I. Spatial distribution of growth, deposition of water, and assimilate import in the elongation zone. Plant Physiology 86: 1070±1076. Schnyder H, Seo S, Rademacher IF, KuÈhbauch W Spatial distribution of growth rates and of epidermal cell lengths in the elongation zone during leaf development in Lolium perenne L. Planta 181: 423±431. Scobbie L, Russell W, Provan GJ, Chesson A The newly extended maize internode: a model for the study of secondary cell wall formation and consequences for digestibility. The Journal of the Science of Food and Agriculture 61: 217±225. Spurr AR A low-viscosity epoxy resin embedding medium for electron microscopy. Journal of Ultrastructure Research 26: 31±43. Travis AJ, Murison SD, Chesson A Estimation of plant cell wall thickness and cell size by image skeletonization. Journal of Agricultural Science, Cambridge 120: 279±287. Vassey TL Morphological, anatomical and cytohistological evaluations of terminal and axillary meristems of tall fescue. PhD Thesis, University of Missouri, Columbia, USA. Vian B, Roland J-C, Reis D Primary cell wall texture and its relation to surface expansion. International Journal of Plant Science 154: 1±9.
THE ROLE OF CELL WALL PEROXIDASE IN THE INHIBITION OF LEAF AND FRUIT GROWTH
 264 BULG. J. PLANT PHYSIOL., SPECIAL ISSUE 2003, 264 272 THE ROLE OF CELL WALL PEROXIDASE IN THE INHIBITION OF LEAF AND FRUIT GROWTH T. Djaković 1, Z. Jovanović 2 1 Maize Research Institute, Slobodana
264 BULG. J. PLANT PHYSIOL., SPECIAL ISSUE 2003, 264 272 THE ROLE OF CELL WALL PEROXIDASE IN THE INHIBITION OF LEAF AND FRUIT GROWTH T. Djaković 1, Z. Jovanović 2 1 Maize Research Institute, Slobodana
Kinematic analysis of leaf growth in grasses a comment on "Spatial and Temporal
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 TITLE Kinematic analysis of leaf growth in grasses a comment on "Spatial and Temporal Quantitative Analysis of Cell Division and Elongation Rate in
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 TITLE Kinematic analysis of leaf growth in grasses a comment on "Spatial and Temporal Quantitative Analysis of Cell Division and Elongation Rate in
Lab Exercise 4: Primary Growth and Tissues in Stems
 Lab Exercise 4: Primary Growth and Tissues in Stems Tissues of the plant body can be classified in a variety of ways: functionally (based on the tissue function, e.g. vascular tissue ), morphologically
Lab Exercise 4: Primary Growth and Tissues in Stems Tissues of the plant body can be classified in a variety of ways: functionally (based on the tissue function, e.g. vascular tissue ), morphologically
 Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark the regions where active cell division and rapid division
Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark the regions where active cell division and rapid division
Bring Your Text to Lab!!!
 Bring Your Text to Lab!!! Vascular Plant Anatomy: Flowering Plants Objectives: 1. To observe what the basic structure of vascular plants is, and how and where this form originates. 2. To begin to understand
Bring Your Text to Lab!!! Vascular Plant Anatomy: Flowering Plants Objectives: 1. To observe what the basic structure of vascular plants is, and how and where this form originates. 2. To begin to understand
Plant Structure. Lab Exercise 24. Objectives. Introduction
 Lab Exercise Plant Structure Objectives - Be able to identify plant organs and give their functions. - Learn distinguishing characteristics between monocot and dicot plants. - Understand the anatomy of
Lab Exercise Plant Structure Objectives - Be able to identify plant organs and give their functions. - Learn distinguishing characteristics between monocot and dicot plants. - Understand the anatomy of
Class XI Chapter 6 Anatomy of Flowering Plants Biology
 Class XI Chapter 6 Anatomy of Flowering Plants Biology Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark
Class XI Chapter 6 Anatomy of Flowering Plants Biology Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark
MORPHOLOGICAL EXAMINATION OF PRAIRIE TURNIP (PSORALEA ESCULENTA PURSH) ROOT
 Proceedings of the South Dakota Academy of Science, Vol. 82 (2003) 113 MORPHOLOGICAL EXAMINATION OF PRAIRIE TURNIP (PSORALEA ESCULENTA PURSH) ROOT April L. Stahnke and R. Neil Reese Biology & Microbiology
Proceedings of the South Dakota Academy of Science, Vol. 82 (2003) 113 MORPHOLOGICAL EXAMINATION OF PRAIRIE TURNIP (PSORALEA ESCULENTA PURSH) ROOT April L. Stahnke and R. Neil Reese Biology & Microbiology
Plant Anatomy and Tissue Structures
 Plant Anatomy and Tissue Structures The Two Major Plant Systems Reproductive shoot (flower) Terminal bud Node Internode Angiosperm plants have threse major organs: Roots Stems Leaves & Flowers Terminal
Plant Anatomy and Tissue Structures The Two Major Plant Systems Reproductive shoot (flower) Terminal bud Node Internode Angiosperm plants have threse major organs: Roots Stems Leaves & Flowers Terminal
Anatomy of Flowering Plants. K C Meena PGT Biology
 Anatomy of Flowering Plants K C Meena PGT Biology Tissues A group of similar cells performing same function. Types of plant tissues - Meristematic tissues and permanent tissues. Meristematic tissues Have
Anatomy of Flowering Plants K C Meena PGT Biology Tissues A group of similar cells performing same function. Types of plant tissues - Meristematic tissues and permanent tissues. Meristematic tissues Have
Plants. Tissues, Organs, and Systems
 Plants Tissues, Organs, and Systems Meristematic cells Specialized cells that are responsible for producing specialized cells, they produce three types of tissue in the body of a plant. Meristematic Cells
Plants Tissues, Organs, and Systems Meristematic cells Specialized cells that are responsible for producing specialized cells, they produce three types of tissue in the body of a plant. Meristematic Cells
The plant body has a hierarchy of organs, tissues, and cells. Plants, like multicellular animals:
 Chapter 28 The plant body has a hierarchy of organs, tissues, and cells Plants, like multicellular animals: o Have organs composed of different tissues, which are in turn composed of cells 3 basic organs:
Chapter 28 The plant body has a hierarchy of organs, tissues, and cells Plants, like multicellular animals: o Have organs composed of different tissues, which are in turn composed of cells 3 basic organs:
Plant Anatomy: roots, stems and leaves
 Plant Anatomy: roots, stems and leaves The plant body has a hierarchy of organs, tissues and cells Plants, like animals, have organs composed of different tissues, which are composed of cells. Tissue is
Plant Anatomy: roots, stems and leaves The plant body has a hierarchy of organs, tissues and cells Plants, like animals, have organs composed of different tissues, which are composed of cells. Tissue is
Plant Anatomy: roots, stems and leaves
 Plant Anatomy: roots, stems and leaves The plant body has a hierarchy of organs, tissues and cells Plants, like animals, have organs composed of different tissues, which are composed of cells. Tissue is
Plant Anatomy: roots, stems and leaves The plant body has a hierarchy of organs, tissues and cells Plants, like animals, have organs composed of different tissues, which are composed of cells. Tissue is
NOTES: CH 35 - Plant Structure & Growth
 NOTES: CH 35 - Plant Structure & Growth In their evolutionary journey, plants adapted to the problems of a terrestrial existence as they moved from water to land ANGIOSPERMS (flowering plants) -most diverse
NOTES: CH 35 - Plant Structure & Growth In their evolutionary journey, plants adapted to the problems of a terrestrial existence as they moved from water to land ANGIOSPERMS (flowering plants) -most diverse
Topic 14. The Root System. II. Anatomy of an Actively Growing Root Tip
 Topic 14. The Root System Introduction. This is the first of two lab topics that focus on the three plant organs (root, stem, leaf). In these labs we want you to recognize how tissues are organized in
Topic 14. The Root System Introduction. This is the first of two lab topics that focus on the three plant organs (root, stem, leaf). In these labs we want you to recognize how tissues are organized in
The three principal organs of seed plants are roots, stems, and leaves.
 23 1 Specialized Tissues in Plants Seed Plant Structure The three principal organs of seed plants are roots, stems, and leaves. 1 of 34 23 1 Specialized Tissues in Plants Seed Plant Structure Roots: absorb
23 1 Specialized Tissues in Plants Seed Plant Structure The three principal organs of seed plants are roots, stems, and leaves. 1 of 34 23 1 Specialized Tissues in Plants Seed Plant Structure Roots: absorb
BIOL 305L Laboratory One
 Please print Full name clearly: BIOL 305L Laboratory One General plant anatomy a great place to start! Introduction Botany is the science of plant life. Traditionally, the science included the study of
Please print Full name clearly: BIOL 305L Laboratory One General plant anatomy a great place to start! Introduction Botany is the science of plant life. Traditionally, the science included the study of
Downloaded from
 POINTS TO REMEMBER : 6. Anatomy of Flowering Plants Study of internal structure of plant is called anatomy. In plants cells are the basic unit. Cells organized into tissues and tissues organized into organs.
POINTS TO REMEMBER : 6. Anatomy of Flowering Plants Study of internal structure of plant is called anatomy. In plants cells are the basic unit. Cells organized into tissues and tissues organized into organs.
Plant Tissues and Organs. Topic 13 Plant Science Subtopics , ,
 Plant Tissues and Organs Topic 13 Plant Science Subtopics 13.1.2, 13.1.3, 13.1.4 Objectives: List and describe the major plant organs their structure and function List and describe the major types of plant
Plant Tissues and Organs Topic 13 Plant Science Subtopics 13.1.2, 13.1.3, 13.1.4 Objectives: List and describe the major plant organs their structure and function List and describe the major types of plant
Leaf Growth in Dactylis glomerata following Defoliation J. L. DAVIDSON' AND F. L. MILTHORPE
 Leaf Growth in Dactylis glomerata following Defoliation BY J. L. DAVIDSON' AND F. L. MILTHORPE Unwertity of Nottingham School of Agriculture, Sutton Bonmgton, Loughborovgh ABSTRACT Defoliation to a height
Leaf Growth in Dactylis glomerata following Defoliation BY J. L. DAVIDSON' AND F. L. MILTHORPE Unwertity of Nottingham School of Agriculture, Sutton Bonmgton, Loughborovgh ABSTRACT Defoliation to a height
The Vascular Plant Body
 The Vascular Plant Body Like animals, plants are made up of specialized cells that are organized into tissues, which are themselves organized into systems of organs. The various parts of plants are adapted
The Vascular Plant Body Like animals, plants are made up of specialized cells that are organized into tissues, which are themselves organized into systems of organs. The various parts of plants are adapted
Visit For All NCERT solutions, CBSE sample papers, Question papers, Notes for Class 6 to 12. Chapter-6 ANATOMY OF FLOWERING PLANTS
 Chapter-6 ANATOMY OF FLOWERING PLANTS POINTS TO REMEMBER Anatomy : Anatomy is the study of internal structure of organisms. Plant anatomy includes organisation and structure of tissues. Tissue : A group
Chapter-6 ANATOMY OF FLOWERING PLANTS POINTS TO REMEMBER Anatomy : Anatomy is the study of internal structure of organisms. Plant anatomy includes organisation and structure of tissues. Tissue : A group
Topic 15. The Shoot System
 Topic 15. The Shoot System Introduction. This is the second of two lab topics that focus on the three plant organs (root, stem, leaf). In these labs we want you to recognize how tissues are organized in
Topic 15. The Shoot System Introduction. This is the second of two lab topics that focus on the three plant organs (root, stem, leaf). In these labs we want you to recognize how tissues are organized in
Topic 2: Plant Structure & Growth Ch. 35 Angiosperms are the most complex plants. They are composed of cells, tissues, organs and organ systems.
 Topic 2: Plant Structure & Growth Ch. 35 Angiosperms are the most complex plants. They are composed of cells, tissues, organs and organ systems. Fig. 35.8 Plant Cells pp.798-802 Types of plant cells Include:
Topic 2: Plant Structure & Growth Ch. 35 Angiosperms are the most complex plants. They are composed of cells, tissues, organs and organ systems. Fig. 35.8 Plant Cells pp.798-802 Types of plant cells Include:
Chapter 29: Plant Tissues
 Chapter 29: Plant Tissues Shoots and Roots Shoots (Leaves and Stem) Produce food by photosynthesis Carry out reproductive functions Roots Anchor the plant Penetrate the soil and absorb water and dissolved
Chapter 29: Plant Tissues Shoots and Roots Shoots (Leaves and Stem) Produce food by photosynthesis Carry out reproductive functions Roots Anchor the plant Penetrate the soil and absorb water and dissolved
The Shoot System of the Primary Plant Body
 BIOL 221 Concepts of Botany Topic 03: The Shoot System of the Primary Plant Body A. Introduction The shoot consists of stems and leaves. It is quite modular in its construction. A shoot is made up of repeated
BIOL 221 Concepts of Botany Topic 03: The Shoot System of the Primary Plant Body A. Introduction The shoot consists of stems and leaves. It is quite modular in its construction. A shoot is made up of repeated
Chapter 35~ Plant Structure and Growth
 Chapter 35~ Plant Structure and Growth Plant Organization Plant morphology is based on plant s evolutionary history Need to draw in nutrients from the ground and the air Plant Organs Root system = roots
Chapter 35~ Plant Structure and Growth Plant Organization Plant morphology is based on plant s evolutionary history Need to draw in nutrients from the ground and the air Plant Organs Root system = roots
Lecture 4 Root Put line under your answer! There is only one correct answer in the multiple choice questions
 Lecture 4 Root Put line under your answer! There is only one correct answer in the multiple choice questions 1. The perception of gravity by a root is thought to take place in a) root hairs b) the region
Lecture 4 Root Put line under your answer! There is only one correct answer in the multiple choice questions 1. The perception of gravity by a root is thought to take place in a) root hairs b) the region
Overview of Plant Tissues
 Plant Tissue Growth Key Concepts Overview of Plant Tissues Seed-bearing vascular plants have a shoot system with stems, leaves, and reproductive parts Most also have a root system These systems consist
Plant Tissue Growth Key Concepts Overview of Plant Tissues Seed-bearing vascular plants have a shoot system with stems, leaves, and reproductive parts Most also have a root system These systems consist
AP Biology. Basic anatomy. Chapter 35. Plant Anatomy. Shoots. Expanded anatomy. Roots. Modified shoots root shoot (stem) leaves
 Chapter 35. Basic anatomy root shoot (stem) leaves Plant Anatomy Expanded anatomy root root tip root hairs shoot (stem) nodes internodes apical buds axillary buds flowers leaves veins Shoots Shoots consist
Chapter 35. Basic anatomy root shoot (stem) leaves Plant Anatomy Expanded anatomy root root tip root hairs shoot (stem) nodes internodes apical buds axillary buds flowers leaves veins Shoots Shoots consist
2.5 : Cells are grouped into tissue
 2.5 : Cells are grouped into tissue 1 CELL STRUCTURE AND FUNCTIONS Prokaryotic and eukaryotic cells Structures & functions: Cell membrane and organelles Animal Cells are grouped into tissue Plant Cell
2.5 : Cells are grouped into tissue 1 CELL STRUCTURE AND FUNCTIONS Prokaryotic and eukaryotic cells Structures & functions: Cell membrane and organelles Animal Cells are grouped into tissue Plant Cell
Outline. Leaf Development. Leaf Structure - Morphology. Leaf Structure - Morphology
 Outline 1. Leaf Structure: Morphology & Anatomy 2. Leaf Development A. Anatomy B. Sector analysis C. Leaf Development Leaf Structure - Morphology Leaf Structure - Morphology 1 Leaf Structure - Morphology
Outline 1. Leaf Structure: Morphology & Anatomy 2. Leaf Development A. Anatomy B. Sector analysis C. Leaf Development Leaf Structure - Morphology Leaf Structure - Morphology 1 Leaf Structure - Morphology
Plant Structure and Function (Ch. 23)
 Plant Structure and Function (Ch. 23) Basic plant anatomy 1 root root tip root hairs Roots Roots anchor plant in soil, absorb minerals & water, & store food fibrous roots (1) mat of thin roots that spread
Plant Structure and Function (Ch. 23) Basic plant anatomy 1 root root tip root hairs Roots Roots anchor plant in soil, absorb minerals & water, & store food fibrous roots (1) mat of thin roots that spread
Exercise 12. Procedure. Aim: To study anatomy of stem and root of monocots and dicots.
 Aim: To study anatomy of stem and root of monocots and dicots. Principle: The study of internal morphology, i.e., cells of various tissues in an organ of a living body is called Anatomy. Tissue, which
Aim: To study anatomy of stem and root of monocots and dicots. Principle: The study of internal morphology, i.e., cells of various tissues in an organ of a living body is called Anatomy. Tissue, which
II. SIMPLE TISSUES Bot 404--Fall A. Introduction to Tissues (DIAGRAM allow a full page)
 II. SIMPLE TISSUES Bot 404--Fall 2004 A. Introduction to Tissues (DIAGRAM allow a full page) B. Definitions Adaxial = facing the axil; upper surface of leaf Abaxial = facing away from the axil; lower surface
II. SIMPLE TISSUES Bot 404--Fall 2004 A. Introduction to Tissues (DIAGRAM allow a full page) B. Definitions Adaxial = facing the axil; upper surface of leaf Abaxial = facing away from the axil; lower surface
Plant Structure. Objectives At the end of this sub section students should be able to:
 Name: 3.2 Organisation and the Vascular Structures 3.2.1 Flowering plant structure and root structure Objectives At the end of this sub section students should be able to: 1. Label a diagram of the external
Name: 3.2 Organisation and the Vascular Structures 3.2.1 Flowering plant structure and root structure Objectives At the end of this sub section students should be able to: 1. Label a diagram of the external
2/25/2013. o Plants take up water and minerals from below ground o Plants take up CO2 and light from above ground THREE BASIC PLANT ORGANS ROOTS
 o Plants take up water and minerals from below ground o Plants take up CO2 and light from above ground THREE BASIC PLANT ORGANS o Roots o Stems o Leaves ROOTS o Anchor plant o Absorb water and minerals
o Plants take up water and minerals from below ground o Plants take up CO2 and light from above ground THREE BASIC PLANT ORGANS o Roots o Stems o Leaves ROOTS o Anchor plant o Absorb water and minerals
Chapter #35~ Plant Structure and Growth
 Chapter #35~ Plant Structure and Growth What part of a plant is represented by each of these: Carrot Celery Red Pepper Tomato Lettuce Garbanzo Bean Angiosperm structure Three basic organs: Roots (root
Chapter #35~ Plant Structure and Growth What part of a plant is represented by each of these: Carrot Celery Red Pepper Tomato Lettuce Garbanzo Bean Angiosperm structure Three basic organs: Roots (root
Answer Key. Vocabulary Practice. 1. guard cell 2. parenchyma cell 3. sclerenchyma cell 4. collenchyma cell 5. All are types of plant cells
 Answer Key Vocabulary Practice A. Choose the Right Word 1. guard cell 2. parenchyma cell 3. sclerenchyma cell 4. collenchyma cell 5. All are types of cells 6. meristem 7. ground tissue 8. dermal tissue
Answer Key Vocabulary Practice A. Choose the Right Word 1. guard cell 2. parenchyma cell 3. sclerenchyma cell 4. collenchyma cell 5. All are types of cells 6. meristem 7. ground tissue 8. dermal tissue
The Science of Plants in Agriculture Pl.Sci 102. Getting to Know Plants
 The Science of Plants in Agriculture Pl.Sci 102 Getting to Know Plants Growth and Development of Plants Growth and Development of Plants Why it s important to have knowledge about plant development. What
The Science of Plants in Agriculture Pl.Sci 102 Getting to Know Plants Growth and Development of Plants Growth and Development of Plants Why it s important to have knowledge about plant development. What
In vitro digestibility and neutral detergent fibre and lignin contents of plant parts of nine forage species
 Journal of Agricultural Science, Cambridge (1998), 131, 51 58. 1998 Cambridge University Press Printed in the United Kingdom 51 In vitro digestibility and neutral detergent fibre and lignin contents of
Journal of Agricultural Science, Cambridge (1998), 131, 51 58. 1998 Cambridge University Press Printed in the United Kingdom 51 In vitro digestibility and neutral detergent fibre and lignin contents of
Seed Development and Yield Components. Thomas G Chastain CROP 460/560 Seed Production
 Seed Development and Yield Components Thomas G Chastain CROP 460/560 Seed Production The Seed The zygote develops into the embryo which contains a shoot (covered by the coleoptile) and a root (radicle).
Seed Development and Yield Components Thomas G Chastain CROP 460/560 Seed Production The Seed The zygote develops into the embryo which contains a shoot (covered by the coleoptile) and a root (radicle).
Primary Plant Body: Embryogenesis and the Seedling
 BIOL 221 Concepts of Botany Primary Plant Body: Embryogenesis and the Seedling (Photo Atlas: Figures 1.29, 9.147, 9.148, 9.149, 9.150, 9.1, 9.2) A. Introduction Plants are composed of fewer cell types,
BIOL 221 Concepts of Botany Primary Plant Body: Embryogenesis and the Seedling (Photo Atlas: Figures 1.29, 9.147, 9.148, 9.149, 9.150, 9.1, 9.2) A. Introduction Plants are composed of fewer cell types,
Plant Anatomy Lab 7 - Stems II
 Plant Anatomy Lab 7 - Stems II This exercise continues the previous lab in studying primary growth in the stem. We will be looking at stems from a number of different plant species, and emphasize (1) the
Plant Anatomy Lab 7 - Stems II This exercise continues the previous lab in studying primary growth in the stem. We will be looking at stems from a number of different plant species, and emphasize (1) the
Plant Structure, Growth, and Development
 Plant Structure, Growth, and Development Plant hierarchy: Cells Tissue: group of similar cells with similar function: Dermal, Ground, Vascular Organs: multiple kinds of tissue, very diverse function Organ
Plant Structure, Growth, and Development Plant hierarchy: Cells Tissue: group of similar cells with similar function: Dermal, Ground, Vascular Organs: multiple kinds of tissue, very diverse function Organ
Plant Structure and Function Extension
 Plant Structure and Function Extension NGSSS: SC.912.L.14.7 Relate the structure of each of the major plant organs and tissues to physiological processes. (AA) Part 1A: Leaves The leaf of a plant serves
Plant Structure and Function Extension NGSSS: SC.912.L.14.7 Relate the structure of each of the major plant organs and tissues to physiological processes. (AA) Part 1A: Leaves The leaf of a plant serves
UNIT 6 - STRUCTURES OF FLOWERING PLANTS & THEIR FUNCTIONS
 6.1 Plant Tissues A tissue is a group of cells with common function, structures or both. In plants we can find 2 types of tissues: Meristem Permanent tissues Meristem is found in regions with continuous
6.1 Plant Tissues A tissue is a group of cells with common function, structures or both. In plants we can find 2 types of tissues: Meristem Permanent tissues Meristem is found in regions with continuous
STEMS Anytime you use something made of wood, you re using something made from the stem of a plant. Stems are linear structures with attached leaves
 STEMS OUTLINE External Form of a Woody Twig Stem Origin and Development Stem Tissue Patterns Herbaceous Dicotyledonous Stems Woody Dicotyledonous Stems Monocotyledonous Stems Specialized Stems Wood and
STEMS OUTLINE External Form of a Woody Twig Stem Origin and Development Stem Tissue Patterns Herbaceous Dicotyledonous Stems Woody Dicotyledonous Stems Monocotyledonous Stems Specialized Stems Wood and
Chapter 23 Notes Roots Stems Leaves
 Chapter 23 Notes Roots Stems Leaves I. Specialized tissue in plants - effective way to ensure the plant s survival A. Seed plant structure 1. Roots - a. Absorbs water and dissolves nutrients b. anchors
Chapter 23 Notes Roots Stems Leaves I. Specialized tissue in plants - effective way to ensure the plant s survival A. Seed plant structure 1. Roots - a. Absorbs water and dissolves nutrients b. anchors
Plant Structure and Growth
 Plant Structure and Growth A. Flowering Plant Parts: The flowering plants or are the most diverse group of plants. They are divided into 2 classes and. Examples of monocots: Examples of dicots: The morphology
Plant Structure and Growth A. Flowering Plant Parts: The flowering plants or are the most diverse group of plants. They are divided into 2 classes and. Examples of monocots: Examples of dicots: The morphology
TARGET STUDY MATERIAL
 TARGET STUDY MATERIAL Plus-1 Botany VOL I TARGET EDUCATIONAL INSTITUTION Target Educational institution is the one and only Entrance coaching and CBSE 10 th coaching centre at Mukkam with advanced technologies
TARGET STUDY MATERIAL Plus-1 Botany VOL I TARGET EDUCATIONAL INSTITUTION Target Educational institution is the one and only Entrance coaching and CBSE 10 th coaching centre at Mukkam with advanced technologies
SESSION 6: SUPPORT AND TRANSPORT SYSTEMS IN PLANTS PART 1
 SESSION 6: SUPPORT AND TRANSPORT SYSTEMS IN PLANTS PART 1 KEY CONCEPTS In this session we will focus on summarising what you need to know about: - Anatomy of dicotyledonous plants Root and stem: distribution
SESSION 6: SUPPORT AND TRANSPORT SYSTEMS IN PLANTS PART 1 KEY CONCEPTS In this session we will focus on summarising what you need to know about: - Anatomy of dicotyledonous plants Root and stem: distribution
Analysis of the temperature effect on the components of plant digestibility in two populations of perennial ryegrass
 Journal of the Science of Food and Agriculture J Sci Food Agric 83:320 329 (online: 2003) DOI: 10.1002/jsfa.1315 Analysis of the temperature effect on the components of plant digestibility in two populations
Journal of the Science of Food and Agriculture J Sci Food Agric 83:320 329 (online: 2003) DOI: 10.1002/jsfa.1315 Analysis of the temperature effect on the components of plant digestibility in two populations
PLANT TISSUES 12 MARCH 2014
 PLANT TISSUES 12 MARCH 2014 Lesson Description In this lesson we: Identify the different types of plant tissue Be able to relate the different structures with the different functions Plant Tissue Summary
PLANT TISSUES 12 MARCH 2014 Lesson Description In this lesson we: Identify the different types of plant tissue Be able to relate the different structures with the different functions Plant Tissue Summary
Plant Anatomy AP Biology
 Plant Anatomy 2006-2007 Basic plant anatomy 1 root root tip root hairs Roots 1 Roots anchor plant in soil, absorb minerals & water, & store food fibrous roots (1) mat of thin roots that spread out monocots
Plant Anatomy 2006-2007 Basic plant anatomy 1 root root tip root hairs Roots 1 Roots anchor plant in soil, absorb minerals & water, & store food fibrous roots (1) mat of thin roots that spread out monocots
Botany Physiology. Due Date Code Period Earned Points
 Botany Physiology Name C/By Due Date Code Period Earned Points Bot Phys 5N5 Stem Forms Bot Phys 5-05 Identify the major forms of stems in plants I. Identify the major forms of stems in plants A. internal
Botany Physiology Name C/By Due Date Code Period Earned Points Bot Phys 5N5 Stem Forms Bot Phys 5-05 Identify the major forms of stems in plants I. Identify the major forms of stems in plants A. internal
13.2 The Vascular Plant Body (textbook p )
 13.2 The Vascular Plant Body (textbook p544 550) Learning Goal: Label and explain the anatomy of the Vascular Plant and it's Tissue Types Plants are classified into two main groups: and. Vascular plants
13.2 The Vascular Plant Body (textbook p544 550) Learning Goal: Label and explain the anatomy of the Vascular Plant and it's Tissue Types Plants are classified into two main groups: and. Vascular plants
Non Permanent Tissues - Meristematic Tissue
 PLANT TISSUES Non Permanent Tissues - Meristematic Tissue Undifferentiated plant cells that are continually dividing by mitosis Large thin walled cells No vacuole Dense cytoplasm Large nucleus Found at
PLANT TISSUES Non Permanent Tissues - Meristematic Tissue Undifferentiated plant cells that are continually dividing by mitosis Large thin walled cells No vacuole Dense cytoplasm Large nucleus Found at
IN the following account the development of a monocotyledon shoot is
 Developmental Anatomy of the Shoot of Zea mays L. BY B. C. SHARMAN (Department of Botany, University of Leeds) With Plate VII and twenty-three Figures in the Text INTRODUCTION IN the following account
Developmental Anatomy of the Shoot of Zea mays L. BY B. C. SHARMAN (Department of Botany, University of Leeds) With Plate VII and twenty-three Figures in the Text INTRODUCTION IN the following account
(A) Buds (B) Lateral meristem (C) Apical meristem (D) Stem (E) Trichomes
 AP Biology - Problem Drill 17: Plant Structure Question No. 1 of 10 1. What are hair-like outgrowths that protect and absorb nutrients? Question #01 (A) Buds (B) Lateral meristem (C) Apical meristem (D)
AP Biology - Problem Drill 17: Plant Structure Question No. 1 of 10 1. What are hair-like outgrowths that protect and absorb nutrients? Question #01 (A) Buds (B) Lateral meristem (C) Apical meristem (D)
Turf Growth and Development
 Turf Growth and Development Germination and Seedling Development Spikelet borne in Inflorescence Germination and Seedling Development Leaf and Stem Formation Inflorescence Roots Spikelet s Apex Caryopsis
Turf Growth and Development Germination and Seedling Development Spikelet borne in Inflorescence Germination and Seedling Development Leaf and Stem Formation Inflorescence Roots Spikelet s Apex Caryopsis
(Photo Atlas: Figures 9.147, 9.148, 9.150, 9.1, 9.2, )
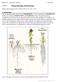 BIOL 221 Concepts of Botany Fall 2007 Topic 07: Primary Plant Body: The Root System (Photo Atlas: Figures 9.147, 9.148, 9.150, 9.1, 9.2, 9.5 9.23) A. Introduction The root has the primary functions of
BIOL 221 Concepts of Botany Fall 2007 Topic 07: Primary Plant Body: The Root System (Photo Atlas: Figures 9.147, 9.148, 9.150, 9.1, 9.2, 9.5 9.23) A. Introduction The root has the primary functions of
Leaf and Internode. Introduction. Parts of the Monocot and Dicot Leaf. Introductory article
 Andrew Hudson, University of Edinburgh, Edinburgh, UK Christopher Jeffree, University of Edinburgh, Edinburgh, UK Leaves of different species show wide variation in morphology and anatomy, usually associated
Andrew Hudson, University of Edinburgh, Edinburgh, UK Christopher Jeffree, University of Edinburgh, Edinburgh, UK Leaves of different species show wide variation in morphology and anatomy, usually associated
IX. PRIMARY STEM STRUCTURE AND DEVELOPMENT Bot 404 Fall 2004
 IX. PRIMARY STEM STRUCTURE AND DEVELOPMENT Bot 404 Fall 2004 A. Shoot apex -plants have an open system of growth, therefore the ability (at least potentially) to continue growth because there is a meristem
IX. PRIMARY STEM STRUCTURE AND DEVELOPMENT Bot 404 Fall 2004 A. Shoot apex -plants have an open system of growth, therefore the ability (at least potentially) to continue growth because there is a meristem
Plant Anatomy. By Umanga Chapagain
 Plant Anatomy By Umanga Chapagain PLANT ANATOMY The science of the structure of the organized plant body learned by dissection is called Plant Anatomy. In general, Plant Anatomy refers to study of internal
Plant Anatomy By Umanga Chapagain PLANT ANATOMY The science of the structure of the organized plant body learned by dissection is called Plant Anatomy. In general, Plant Anatomy refers to study of internal
The Shoot System: Primary Stem Structure - 1
 The Shoot System: Primary Stem Structure - 1 Shoot System The shoot system comprises the leaves and stems of plants. Leaves are located at nodes on the stem; the distance along the stem between nodes is
The Shoot System: Primary Stem Structure - 1 Shoot System The shoot system comprises the leaves and stems of plants. Leaves are located at nodes on the stem; the distance along the stem between nodes is
ROOTS. Syllabus Theme A Plant Structure and Function. Root systems. Primary Growth of Roots. Taproot system. Fibrous root system.
 Syllabus Theme A lant Structure and Function A2: Structure and function of the basic plant organs ampbell & Reece hap. 35 Selected page numbers ROOTS Functions Anchors the vascular plant Absorbs minerals
Syllabus Theme A lant Structure and Function A2: Structure and function of the basic plant organs ampbell & Reece hap. 35 Selected page numbers ROOTS Functions Anchors the vascular plant Absorbs minerals
Roots and Soil Chapter 5
 Roots and Soil Chapter 5 Plant Organs Plant organs are groups of several types of tissues that together perform a particular function. Vegetative organs roots, stems, leaves make and use food, absorb water
Roots and Soil Chapter 5 Plant Organs Plant organs are groups of several types of tissues that together perform a particular function. Vegetative organs roots, stems, leaves make and use food, absorb water
EFFECTS OF GIBBERELLIC ACID ON INTERNODE GROWTH AND STARCH CONTENTS OF EUCALYPTUS CAMALDULENSIS SEEDLINGS
 New Phytol. {ig()) S, ioiyio22. EFFECTS OF GIBBERELLIC ACID ON INTERNODE GROWTH AND STARCH CONTENTS OF EUCALYPTUS CAMALDULENSIS SEEDLINGS BY E. P. BACHELARD Department of Forestry, Australian National
New Phytol. {ig()) S, ioiyio22. EFFECTS OF GIBBERELLIC ACID ON INTERNODE GROWTH AND STARCH CONTENTS OF EUCALYPTUS CAMALDULENSIS SEEDLINGS BY E. P. BACHELARD Department of Forestry, Australian National
Bio Factsheet. Transport in Plants. Number 342
 Number 342 Transport in Plants This Factsheet: Explains why plants need a transport system Describes what plants transport Describes the tissues which carry out transport Outlines the position of the xylem
Number 342 Transport in Plants This Factsheet: Explains why plants need a transport system Describes what plants transport Describes the tissues which carry out transport Outlines the position of the xylem
QUANTIFICATION OF EMBOLI BY VISUALIZATION OF AIR FILLED XYLEM VESSELS
 QUANTIFICATION OF EMBOLI BY VISUALIZATION OF AIR FILLED XYLEM VESSELS J. Nijsse and U. van Meeteren Wageningen University Plant Sciences Horticultural Production Chains Marijkeweg 22 6709 PG Wageningen
QUANTIFICATION OF EMBOLI BY VISUALIZATION OF AIR FILLED XYLEM VESSELS J. Nijsse and U. van Meeteren Wageningen University Plant Sciences Horticultural Production Chains Marijkeweg 22 6709 PG Wageningen
Useful Propagation Terms. Propagation The application of specific biological principles and concepts in the multiplication of plants.
 Useful Propagation Terms Propagation The application of specific biological principles and concepts in the multiplication of plants. Adventitious Typically describes new organs such as roots that develop
Useful Propagation Terms Propagation The application of specific biological principles and concepts in the multiplication of plants. Adventitious Typically describes new organs such as roots that develop
Division Ave. High School AP Biology
 Monocots & dicots Angiosperm are divide into 2 classes dicots (eudicot) 2 cotyledons (seed leaves) leaves with network of veins woody plants, trees, shrubs, beans monocots 1 cotyledon leaves with parallel
Monocots & dicots Angiosperm are divide into 2 classes dicots (eudicot) 2 cotyledons (seed leaves) leaves with network of veins woody plants, trees, shrubs, beans monocots 1 cotyledon leaves with parallel
Plant Structure And Growth
 Plant Structure And Growth The Plant Body is Composed of Cells and Tissues Tissue systems (Like Organs) made up of tissues Made up of cells Plant Tissue Systems Ground Tissue System Ø photosynthesis Ø
Plant Structure And Growth The Plant Body is Composed of Cells and Tissues Tissue systems (Like Organs) made up of tissues Made up of cells Plant Tissue Systems Ground Tissue System Ø photosynthesis Ø
BOTANY LAB #1 MITOSIS AND PLANT TISSUES
 Mitosis and cytokinesis in plants BOTANY LAB #1 MITOSIS AND PLANT TISSUES In plants the formation of new cells takes place in specialized regions of meristematic tissue. Meristematic tissues contain immature,
Mitosis and cytokinesis in plants BOTANY LAB #1 MITOSIS AND PLANT TISSUES In plants the formation of new cells takes place in specialized regions of meristematic tissue. Meristematic tissues contain immature,
Chapter 15 PLANT STRUCTURES AND TAXONOMY
 Chapter 15 PLANT STRUCTURES AND TAXONOMY Chapter 15: Parts of a plant Manufactures food by photosynthesis Attracts insects for pollination Contains seeds Supports branches and transports food and water
Chapter 15 PLANT STRUCTURES AND TAXONOMY Chapter 15: Parts of a plant Manufactures food by photosynthesis Attracts insects for pollination Contains seeds Supports branches and transports food and water
LOOKING AT PLANT STEMS
 Activity 4.17 Student Sheet LOOKING AT PLANT STEMS Purpose To look at the structure of xylem vessels, phloem sieve tubes and sclerenchyma fibres. To locate the position of these tissues within the stem.
Activity 4.17 Student Sheet LOOKING AT PLANT STEMS Purpose To look at the structure of xylem vessels, phloem sieve tubes and sclerenchyma fibres. To locate the position of these tissues within the stem.
Anatomy of dicotyledonous plants
 Anatomy of dicotyledonous plants Differences between Monocotyledons and Dicotyledons All plants are classified as producing seeds or not producing seeds. Those that produce seeds are divided into flowering
Anatomy of dicotyledonous plants Differences between Monocotyledons and Dicotyledons All plants are classified as producing seeds or not producing seeds. Those that produce seeds are divided into flowering
THE OHIO JOURNAL OF SCIENCE
 THE OHIO JOURNAL OF SCIENCE VOL. XXIV JULY, 1924 No.. 4 THE VASCULAR ANATOMY OF CALAMOVILFA LONGIFOLIA.* ERNEST LINCOLN STOVER Eastern Illinois State Teachers' College The present study of the anatomy
THE OHIO JOURNAL OF SCIENCE VOL. XXIV JULY, 1924 No.. 4 THE VASCULAR ANATOMY OF CALAMOVILFA LONGIFOLIA.* ERNEST LINCOLN STOVER Eastern Illinois State Teachers' College The present study of the anatomy
Chapter 6. Biology of Flowering Plants. Anatomy Seedlings, Meristems, Stems, and Roots
 BOT 3015L (Outlaw/Sherdan/Aghoram); Page 1 of 6 Chapter 6 Biology of Flowering Plants Anatomy Seedlings, Meristems, Stems, and Roots Objectives Seedling germination and anatomy. Understand meristem structure
BOT 3015L (Outlaw/Sherdan/Aghoram); Page 1 of 6 Chapter 6 Biology of Flowering Plants Anatomy Seedlings, Meristems, Stems, and Roots Objectives Seedling germination and anatomy. Understand meristem structure
Chapter. Transport in. Structure of. 1- Epidermis: 2- Cortex: All plants 2- a specialized. In higher moving by. hydra and. with cuticles) 1-2-
 Chapter 2 Transport in living organisms The concept of transport and the need for it: All plants need CO 2, water and mineral salts to perform photosynthesis In primitive plants such as algae these materials
Chapter 2 Transport in living organisms The concept of transport and the need for it: All plants need CO 2, water and mineral salts to perform photosynthesis In primitive plants such as algae these materials
Primary Internal structure & Normal Secondary growth in Sunflower stem
 Primary Internal structure & Normal Secondary growth in Sunflower stem B. Sc. II - Botany Dr. (Miss) Kalpana R. Datar Assistant Professor DEPARTMENT OF BOTANY Willingdon College, Sangli. kalpana_datar@yahoo.com.
Primary Internal structure & Normal Secondary growth in Sunflower stem B. Sc. II - Botany Dr. (Miss) Kalpana R. Datar Assistant Professor DEPARTMENT OF BOTANY Willingdon College, Sangli. kalpana_datar@yahoo.com.
2.1 PLANT TISSUE HALIMAHTUN SAEDIAH BT ABU BAKAR KOLEJ TEKNOLOGI TIMUR
 2.1 PLANT TISSUE HALIMAHTUN SAEDIAH BT ABU BAKAR KOLEJ TEKNOLOGI TIMUR GENERAL Plant cell are differentiated possessing structural adaptations that make specific functions possible. Modifications of cell
2.1 PLANT TISSUE HALIMAHTUN SAEDIAH BT ABU BAKAR KOLEJ TEKNOLOGI TIMUR GENERAL Plant cell are differentiated possessing structural adaptations that make specific functions possible. Modifications of cell
A group of cells with common origin is called a tissue. The cells of a tissue usually perform a common function.
 Anatomy of Flowering Plants Tissues A group of cells with common origin is called a tissue. The cells of a tissue usually perform a common function. Types of Tissue: There are two main types of plant tissues,
Anatomy of Flowering Plants Tissues A group of cells with common origin is called a tissue. The cells of a tissue usually perform a common function. Types of Tissue: There are two main types of plant tissues,
TREES. Functions, structure, physiology
 TREES Functions, structure, physiology Trees in Agroecosystems - 1 Microclimate effects lower soil temperature alter soil moisture reduce temperature fluctuations Maintain or increase soil fertility biological
TREES Functions, structure, physiology Trees in Agroecosystems - 1 Microclimate effects lower soil temperature alter soil moisture reduce temperature fluctuations Maintain or increase soil fertility biological
23 1 Specialized Tissues in Plants Slide 1 of 34
 23 1 Specialized Tissues in Plants 1 of 34 Seed Plant Structure The three principal organs of seed plants are roots, stems, and leaves. These organs perform functions such as the transport of nutrients,
23 1 Specialized Tissues in Plants 1 of 34 Seed Plant Structure The three principal organs of seed plants are roots, stems, and leaves. These organs perform functions such as the transport of nutrients,
* School of Biological Sciences, Carslaw Building, University of Sydney, Sydney, N.S.W By VERONICA H. K. Low*
 Aust. J. biol. Sci., 1971, 24, 187-95 * School of Biological Sciences, Carslaw Building, University of Sydney, Sydney, N.S.W. 2006.. NTRODUCTON A detailed survey of the morphological and anatomical effects
Aust. J. biol. Sci., 1971, 24, 187-95 * School of Biological Sciences, Carslaw Building, University of Sydney, Sydney, N.S.W. 2006.. NTRODUCTON A detailed survey of the morphological and anatomical effects
Chapter 29. Table of Contents. Section 1 Plant Cells and Tissues. Section 2 Roots. Section 3 Stems. Section 4 Leaves. Plant Structure and Function
 Plant Structure and Function Table of Contents Section 1 Plant Cells and Tissues Section 2 Roots Section 3 Stems Section 4 Leaves Section 1 Plant Cells and Tissues Objectives Describe the three basic types
Plant Structure and Function Table of Contents Section 1 Plant Cells and Tissues Section 2 Roots Section 3 Stems Section 4 Leaves Section 1 Plant Cells and Tissues Objectives Describe the three basic types
Tissue architecture changes of expanding barley (Hordeum vulgare L.) leaf under salt stress
 AJCS 8(10):1373-1379 (2014) ISSN:1835-2707 Tissue architecture changes of expanding barley (Hordeum vulgare L.) leaf under salt stress Ehsan Bijanzadeh 1*, Seyed Abdolreza Kazemeini 2 1 Deperment of Plant
AJCS 8(10):1373-1379 (2014) ISSN:1835-2707 Tissue architecture changes of expanding barley (Hordeum vulgare L.) leaf under salt stress Ehsan Bijanzadeh 1*, Seyed Abdolreza Kazemeini 2 1 Deperment of Plant
Journal of Chemical and Pharmaceutical Research
 Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research J. Chem. Pharm. Res., 2010, 2(4):766-771 ISSN No: 0975-7384 CODEN(USA): JCPRC5 Leaf and stem anatomy of Cymbopogon citratus
Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research J. Chem. Pharm. Res., 2010, 2(4):766-771 ISSN No: 0975-7384 CODEN(USA): JCPRC5 Leaf and stem anatomy of Cymbopogon citratus
Secondary growth in stems
 Secondary growth in stems Secondary growth Some of the meristematic cells in plants with secondary growth keep their meristematic state and become cells of the cambium. The addition of secondary vascular
Secondary growth in stems Secondary growth Some of the meristematic cells in plants with secondary growth keep their meristematic state and become cells of the cambium. The addition of secondary vascular
Plants. Plant Form and Function. Tissue Systems 6/4/2012. Chapter 17. Herbaceous (nonwoody) Woody. Flowering plants can be divided into two groups:
 Monocots Dicots 6/4/2012 Plants Plant Form and Function Chapter 17 Herbaceous (nonwoody) In temperate climates, aerial parts die back Woody In temperate climates, aerial parts persist The Plant Body Functions
Monocots Dicots 6/4/2012 Plants Plant Form and Function Chapter 17 Herbaceous (nonwoody) In temperate climates, aerial parts die back Woody In temperate climates, aerial parts persist The Plant Body Functions
Germinating sunflowers, turgor and nutation. From:
 Germinating sunflowers, turgor and nutation From: http://sunflower.bio.indiana.edu/~rhangart/plantmotion Nutation is Sunflower due to unequal Germination rates of growth in that continuous is dependent
Germinating sunflowers, turgor and nutation From: http://sunflower.bio.indiana.edu/~rhangart/plantmotion Nutation is Sunflower due to unequal Germination rates of growth in that continuous is dependent
Two major categories. BIOLOGY 189 Fundamentals of Life Sciences. Spring 2004 Plant Structure and Function. Plant Structure and Function
 BIOLOGY 189 Fundamentals of Life Sciences Spring 2004 Plant Structure and Function 18 16 14 12 10 8 6 Examination #1 Class Average: 33/60 for 55% 4 Chapters 31-32 32 2 0 6 10 15 20 25 30 35 40 45 50 55
BIOLOGY 189 Fundamentals of Life Sciences Spring 2004 Plant Structure and Function 18 16 14 12 10 8 6 Examination #1 Class Average: 33/60 for 55% 4 Chapters 31-32 32 2 0 6 10 15 20 25 30 35 40 45 50 55
PLANT STRUCTURE: PARTS (ORGANS) Roots Leaves Stems
 PLANT STRUCTURE: PARTS (ORGANS) Roots Leaves Stems ROOTS El Hiquieron. Strangulating Plant Ficusjimenezii The trees you see growing on the wall are the Higueron. The Higueronsare plants that can grow in
PLANT STRUCTURE: PARTS (ORGANS) Roots Leaves Stems ROOTS El Hiquieron. Strangulating Plant Ficusjimenezii The trees you see growing on the wall are the Higueron. The Higueronsare plants that can grow in
Today: Plant Structure Exam II is on F March 31
 Next few lectures are on plant form and function Today: Plant Structure Exam II is on F March 31 Outline Plant structure I. Plant Cells structure & different types II. Types of meristems Apical meristems:
Next few lectures are on plant form and function Today: Plant Structure Exam II is on F March 31 Outline Plant structure I. Plant Cells structure & different types II. Types of meristems Apical meristems:
CHAPTER 6 ANATOMY OF FLOWERING PLANTS MULTIPLE CHOICE QUESTIONS
 ANATOMY OF FLOWERING PLANTS 27 27 CHAPTER 6 ANATOMY OF FLOWERING PLANTS MULTIPLE CHOICE QUESTIONS 1. A transverse section of stem is stained first with safranin and then with fast green following the usual
ANATOMY OF FLOWERING PLANTS 27 27 CHAPTER 6 ANATOMY OF FLOWERING PLANTS MULTIPLE CHOICE QUESTIONS 1. A transverse section of stem is stained first with safranin and then with fast green following the usual
ANATOMY OF PLANTS Introduction: The study of gross internal structure of plant organs by the technique of section cutting is called plant anatomy.
 ANATOMY OF PLANTS Introduction: The study of gross internal structure of plant organs by the technique of section cutting is called plant anatomy. (Pandey, 2002). Various plant organ viz. root, stem, leaves,
ANATOMY OF PLANTS Introduction: The study of gross internal structure of plant organs by the technique of section cutting is called plant anatomy. (Pandey, 2002). Various plant organ viz. root, stem, leaves,
-Each asexual organs. -Anchors the plant -Absorbs water and minerals -Stores sugars and starches
 Plants are made up of: -organs, tissues, and cells The three major plant organs are: -Roots, stems, and leaves -Each asexual organs Plants have a Root System beneath the ground that us a multicellular
Plants are made up of: -organs, tissues, and cells The three major plant organs are: -Roots, stems, and leaves -Each asexual organs Plants have a Root System beneath the ground that us a multicellular
