Activity 12: Energy from Nuclear Reactions
|
|
|
- Rosa Lucas
- 5 years ago
- Views:
Transcription
1 Name Section Activity 12: Energy from Nuclear Reactions 12.1 A Model of the Composition of Nucleons 1) Formation of Nucleons Nucleons consist of quark trios. a) Place orange or green quarks into the metal nucleon or gluon cloud to represent a proton. Draw a diagram showing the quarks in the proton. b) Flip one of the quarks in the metal gluon cloud to represent a neutron. Make sure the spin of the neutron points in the same direction as the spin of the proton. Draw a diagram showing the quarks in the neutron. c) Which of the four fundamental forces is involved in binding quarks into nucleons? d) The exchange of which type of gauge boson is responsible for binding quarks into nucleons? 77
2 12.2 A Model of Beta Decay 2) Feynman Diagram of a Beta Decay Beta decays, in which a neutron changes into a proton or a proton changes into a neutron, can be represented by a Feynman diagram. Time 2 d u d??? boson Time 1 d u u a) Place the appropriate orange and green quarks into the gluon cloud to form the nucleon shown at Time 1. Which type of nucleon is this? b) Flip the appropriate quark to form the nucleon at Time 2. Which type of nucleon is this? c) Open the black plastic box that correctly completes this reaction. Which box is this? d) Is an electron or an antielectron emitted? Why? e) Is a neutrino or antineutrino emitted? Why? f) Write the nuclear reaction illustrated by this Feynman diagram. g) Which of the four fundamental forces is responsible for beta decay? h) Which type of boson is responsible for beta plus decay? i) Group Discussion Question: What changes occur in the identity of the quark trio making up the nucleus when a proton becomes a neutron? Why? 78
3 3) Summary of beta decay Fill in the table below to summarize the discussion of beta decay. Decay Nucleon change Quark change Particles emitted Force involved Gauge boson beta-plus (β + ) beta-minus (β ) 12.3 How Are Nucleons Bound into Nuclei? 4) Pions Your instructor will discuss pions, which consist of two quarks. a) Which quark and antiquark can form a positive pion? Draw a diagram showing this pair and the directions of their spins. b) Which quark and antiquark can form a negative pion? Draw a diagram showing this pair and the directions of their spins. 79
4 c) Which quark and antiquark can form a neutral pion? Draw a diagram showing this pair and the directions of their spins. (There are two combinations of a quark and an antiquark that form a neutral pion. Draw diagrams of both combinations.) d) Which of the four fundamental forces is responsible for binding quarks and antiquarks into pions? 12.4 Nuclear Binding Energy 1) Releasing energy from nuclear decay a) Open the plastic boxes representing beta plus decay, beta minus decay, and gamma decay. What reaction product do each of these decays have in common? b) Name three other nuclear reactions that release energy. c) What is the one nucleus naturally occurring that fissions spontaneously (without the addition of activation energy)? 2) Releasing energy from fusing matter a) When two protons come together, one proton can change into a neutron. The proton and neutron can then bind together to form one deuterium nucleus. For each deuterium nucleus formed, 3.52 x joules of energy are given off. What is this energy called? b) What is the process of nucleons binding called? c) Why would you expect the process of forming a deuterium nucleus from two protons to require a large amount of activation energy? 80
5 9 Binding energy per nucleon (MeV) Fe Fusion Fission U Mass number A (Total number of nucleons) 12.5 Nuclear Reactions Produced in the Laboratory 3) Nuclear accelerators a) Are naturally-occurring nuclear reactions endothermic or exothermic? b) How can activation energy be supplied to produce nuclear reactions? c) In addition to providing activation energy, what other function do accelerators play in particle physics? d) What is the advantage of a circular particle accelerator? e) Which type of particle accelerator could provide the most energy to colliding particles: a linear accelerator, a circular accelerator, or two accelerators? f) Group Discussion Question: Nuclear fission occurs in nuclear reactors. Why don t we have nuclear fusion reactors? 81
6 12.6 Nuclear Reactions in Stars 4) Fusion reactions in stars 12/12/05 a) What is the source of energy in stars such as the Sun? b) Major challenges to controlling nuclear fusion reactions on Earth are providing the activation energy and containing the reactants. What force provides the activation energy and contains the reactants in fusion reactions in stars? c) What is the proton-proton fusion chain? d) Where does nuclear fusion occur in a star? e) How does the energy from fusion reach the star s surface? 5) Formation of chemical elements in stars a) What is the origin of most of the hydrogen, helium, and lithium in the Universe? b) What is the origin of carbon? c) How are heavier elements through iron formed? d) How are elements heavier than iron formed? e) Group Discussion Question: What is the origin of the heavy elements on the Earth? 12.5 Types of Stars 6) The Hertzsprung Russell (H-R) diagram of stars a) Place each metal disk, which represents a star, on the appropriate point of the grid of the H-R diagram that corresponds to the star s temperature and luminosity (brightness). Note that the temperature on the horizontal axis increases as one moves to the left. The axes are logarithmic scales, therefore the spacing between temperatures on the horizontal scale is not linear. 82
7 b) Indicate on the diagram below approximately where you have placed the disks. Include information on the color of the disk and its size. Luminosity Increasing surface temperature c) Explain the change in color of the stars in the main sequence. d) Sirius is one of the brightest stars in the northern hemisphere s sky. What color is Sirius? How does its temperature compare to the temperature of the Sun? e) What are the small stars in the lower left corner of the diagram? f) What are the large red stars in the upper right corner of the diagram? g) Group Discussion Question: Are any white dwarf stars visible from Earth? Are any red giant stars visible from earth? 83
Nuclear Physics and Nuclear Reactions
 Slide 1 / 33 Nuclear Physics and Nuclear Reactions The Nucleus Slide 2 / 33 Proton: The charge on a proton is +1.6x10-19 C. The mass of a proton is 1.6726x10-27 kg. Neutron: The neutron is neutral. The
Slide 1 / 33 Nuclear Physics and Nuclear Reactions The Nucleus Slide 2 / 33 Proton: The charge on a proton is +1.6x10-19 C. The mass of a proton is 1.6726x10-27 kg. Neutron: The neutron is neutral. The
[2] State in what form the energy is released in such a reaction.... [1]
![[2] State in what form the energy is released in such a reaction.... [1] [2] State in what form the energy is released in such a reaction.... [1]](/thumbs/87/96058923.jpg) (a) The following nuclear reaction occurs when a slow-moving neutron is absorbed by an isotope of uranium-35. 0n + 35 9 U 4 56 Ba + 9 36Kr + 3 0 n Explain how this reaction is able to produce energy....
(a) The following nuclear reaction occurs when a slow-moving neutron is absorbed by an isotope of uranium-35. 0n + 35 9 U 4 56 Ba + 9 36Kr + 3 0 n Explain how this reaction is able to produce energy....
Nuclear Physics 3 8 O+ B. always take place and the proton will be emitted with kinetic energy.
 Name: Date: Nuclear Physics 3. A student suggests that the following transformation may take place. Measurement of rest masses shows that 7 7 N+ He 8 O+ total rest mass( N 7 + He ) < total rest mass( O
Name: Date: Nuclear Physics 3. A student suggests that the following transformation may take place. Measurement of rest masses shows that 7 7 N+ He 8 O+ total rest mass( N 7 + He ) < total rest mass( O
WELCOME TO PERIOD 20: RADIANT ENERGY FROM THE SUN
 WELCOME TO PERIOD 20: RADIANT ENERGY FROM THE SUN Homework #19 is due today. Midterm 2: Weds, Mar 27, 7:45 8:55 pm (Same room as your midterm 1 exam.) Covers periods 10 19 and videos 3 & 4 Review: Tues,
WELCOME TO PERIOD 20: RADIANT ENERGY FROM THE SUN Homework #19 is due today. Midterm 2: Weds, Mar 27, 7:45 8:55 pm (Same room as your midterm 1 exam.) Covers periods 10 19 and videos 3 & 4 Review: Tues,
CHAPTER 7 TEST REVIEW
 IB PHYSICS Name: Period: Date: # Marks: 94 Raw Score: IB Curve: DEVIL PHYSICS BADDEST CLASS ON CAMPUS CHAPTER 7 TEST REVIEW 1. An alpha particle is accelerated through a potential difference of 10 kv.
IB PHYSICS Name: Period: Date: # Marks: 94 Raw Score: IB Curve: DEVIL PHYSICS BADDEST CLASS ON CAMPUS CHAPTER 7 TEST REVIEW 1. An alpha particle is accelerated through a potential difference of 10 kv.
Nuclear Physics Questions. 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of?
 Nuclear Physics Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
Nuclear Physics Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
General Physics (PHY 2140)
 General Physics (PHY 2140) Lecture 20 Modern Physics Nuclear Energy and Elementary Particles Fission, Fusion and Reactors Elementary Particles Fundamental Forces Classification of Particles Conservation
General Physics (PHY 2140) Lecture 20 Modern Physics Nuclear Energy and Elementary Particles Fission, Fusion and Reactors Elementary Particles Fundamental Forces Classification of Particles Conservation
[1] (c) Some fruits, such as bananas, are naturally radioactive because they contain the unstable isotope of potassium-40 ( K.
![[1] (c) Some fruits, such as bananas, are naturally radioactive because they contain the unstable isotope of potassium-40 ( K. [1] (c) Some fruits, such as bananas, are naturally radioactive because they contain the unstable isotope of potassium-40 ( K.](/thumbs/82/85827188.jpg) (a) State, with a reason, whether or not protons and neutrons are fundamental particles....... [] (b) State two fundamental particles that can be classified as leptons.... [] (c) Some fruits, such as bananas,
(a) State, with a reason, whether or not protons and neutrons are fundamental particles....... [] (b) State two fundamental particles that can be classified as leptons.... [] (c) Some fruits, such as bananas,
Today. Homework Due. Stars. Properties (Recap) Nuclear Reactions. proton-proton chain. CNO cycle. Stellar Lifetimes
 Today Stars Properties (Recap) Nuclear Reactions proton-proton chain CNO cycle Stellar Lifetimes Homework Due Stellar Properties Luminosity Surface Temperature Size Mass Composition Stellar Properties
Today Stars Properties (Recap) Nuclear Reactions proton-proton chain CNO cycle Stellar Lifetimes Homework Due Stellar Properties Luminosity Surface Temperature Size Mass Composition Stellar Properties
LECTURE 25 NUCLEAR STRUCTURE AND STABILITY. Instructor: Kazumi Tolich
 LECTURE 25 NUCLEAR STRUCTURE AND STABILITY Instructor: Kazumi Tolich Lecture 25 2 30.1 Nuclear structure Isotopes Atomic mass 30.2 Nuclear stability Biding energy 30.3 Forces and energy in the nucleus
LECTURE 25 NUCLEAR STRUCTURE AND STABILITY Instructor: Kazumi Tolich Lecture 25 2 30.1 Nuclear structure Isotopes Atomic mass 30.2 Nuclear stability Biding energy 30.3 Forces and energy in the nucleus
Lecture 14, 8/9/2017. Nuclear Reactions and the Transmutation of Elements Nuclear Fission; Nuclear Reactors Nuclear Fusion
 Lecture 14, 8/9/2017 Nuclear Reactions and the Transmutation of Elements Nuclear Fission; Nuclear Reactors Nuclear Fusion Nuclear Reactions and the Transmutation of Elements A nuclear reaction takes place
Lecture 14, 8/9/2017 Nuclear Reactions and the Transmutation of Elements Nuclear Fission; Nuclear Reactors Nuclear Fusion Nuclear Reactions and the Transmutation of Elements A nuclear reaction takes place
Chapter 12: Nuclear Reaction
 Chapter 12: Nuclear Reaction A nuclear reaction occurs when a nucleus is unstable or is being bombarded by a nuclear particle. The product of a nuclear reaction is a new nuclide with an emission of a nuclear
Chapter 12: Nuclear Reaction A nuclear reaction occurs when a nucleus is unstable or is being bombarded by a nuclear particle. The product of a nuclear reaction is a new nuclide with an emission of a nuclear
Basic Nuclear Theory. Lecture 1 The Atom and Nuclear Stability
 Basic Nuclear Theory Lecture 1 The Atom and Nuclear Stability Introduction Nuclear power is made possible by energy emitted from either nuclear fission or nuclear fusion. Current nuclear power plants utilize
Basic Nuclear Theory Lecture 1 The Atom and Nuclear Stability Introduction Nuclear power is made possible by energy emitted from either nuclear fission or nuclear fusion. Current nuclear power plants utilize
PhysicsAndMathsTutor.com 1
 PhysicsAndMathsTutor.com 1 1. Describe briefly one scattering experiment to investigate the size of the nucleus of the atom. Include a description of the properties of the incident radiation which makes
PhysicsAndMathsTutor.com 1 1. Describe briefly one scattering experiment to investigate the size of the nucleus of the atom. Include a description of the properties of the incident radiation which makes
H 1. Nuclear Physics. Nuclear Physics. 1. Parts of Atom. A. Nuclear Structure. 2b. Nomenclature. 2. Isotopes. AstroPhysics Notes
 AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia Nuclear Physics A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions Updated: 0Feb07 Rough draft A. Nuclear Structure. Parts of Atom. Parts of
AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia Nuclear Physics A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions Updated: 0Feb07 Rough draft A. Nuclear Structure. Parts of Atom. Parts of
NJCTL.org 2015 AP Physics 2 Nuclear Physics
 AP Physics 2 Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
AP Physics 2 Questions 1. What particles make up the nucleus? What is the general term for them? What are those particles composed of? 2. What is the definition of the atomic number? What is its symbol?
H 1. Nuclear Physics. Nuclear Physics. 1. Parts of Atom. 2. Isotopes. AstroPhysics Notes. Dr. Bill Pezzaglia. Rough draft. A.
 AstroPhysics Notes Tom Lehrer: Elements Dr. Bill Pezzaglia Nuclear Physics Updated: 0Feb Rough draft Nuclear Physics A. Nuclear Structure A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions. Parts
AstroPhysics Notes Tom Lehrer: Elements Dr. Bill Pezzaglia Nuclear Physics Updated: 0Feb Rough draft Nuclear Physics A. Nuclear Structure A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions. Parts
Nuclear Physics. Chapter 43. PowerPoint Lectures for University Physics, Thirteenth Edition Hugh D. Young and Roger A. Freedman
 Chapter 43 Nuclear Physics PowerPoint Lectures for University Physics, Thirteenth Edition Hugh D. Young and Roger A. Freedman Lectures by Wayne Anderson Goals for Chapter 43 To understand some key properties
Chapter 43 Nuclear Physics PowerPoint Lectures for University Physics, Thirteenth Edition Hugh D. Young and Roger A. Freedman Lectures by Wayne Anderson Goals for Chapter 43 To understand some key properties
Matter and Energy. Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law.
 Fission & Fusion Matter and Energy Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law. 2 H 2 O 2 H 2 + O 2 We now need to understand
Fission & Fusion Matter and Energy Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law. 2 H 2 O 2 H 2 + O 2 We now need to understand
32 IONIZING RADIATION, NUCLEAR ENERGY, AND ELEMENTARY PARTICLES
 32 IONIZING RADIATION, NUCLEAR ENERGY, AND ELEMENTARY PARTICLES 32.1 Biological Effects of Ionizing Radiation γ-rays (high-energy photons) can penetrate almost anything, but do comparatively little damage.
32 IONIZING RADIATION, NUCLEAR ENERGY, AND ELEMENTARY PARTICLES 32.1 Biological Effects of Ionizing Radiation γ-rays (high-energy photons) can penetrate almost anything, but do comparatively little damage.
Thursday, April 23, 15. Nuclear Physics
 Nuclear Physics Some Properties of Nuclei! All nuclei are composed of protons and neutrons! Exception is ordinary hydrogen with just a proton! The atomic number, Z, equals the number of protons in the
Nuclear Physics Some Properties of Nuclei! All nuclei are composed of protons and neutrons! Exception is ordinary hydrogen with just a proton! The atomic number, Z, equals the number of protons in the
Particle Physics Outline the concepts of particle production and annihilation and apply the conservation laws to these processes.
 Particle Physics 12.3.1 Outline the concept of antiparticles and give examples 12.3.2 Outline the concepts of particle production and annihilation and apply the conservation laws to these processes. Every
Particle Physics 12.3.1 Outline the concept of antiparticles and give examples 12.3.2 Outline the concepts of particle production and annihilation and apply the conservation laws to these processes. Every
A. Incorrect! Do not confuse Nucleus, Neutron and Nucleon. B. Incorrect! Nucleon is the name given to the two particles that make up the nucleus.
 AP Physics - Problem Drill 24: Nuclear Physics 1. Identify what is being described in each of these statements. Question 01 (1) It is held together by the extremely short range Strong force. (2) The magnitude
AP Physics - Problem Drill 24: Nuclear Physics 1. Identify what is being described in each of these statements. Question 01 (1) It is held together by the extremely short range Strong force. (2) The magnitude
cgrahamphysics.com Particles that mediate force Book pg Exchange particles
 Particles that mediate force Book pg 299-300 Exchange particles Review Baryon number B Total # of baryons must remain constant All baryons have the same number B = 1 (p, n, Λ, Σ, Ξ) All non baryons (leptons
Particles that mediate force Book pg 299-300 Exchange particles Review Baryon number B Total # of baryons must remain constant All baryons have the same number B = 1 (p, n, Λ, Σ, Ξ) All non baryons (leptons
Introduction to Modern Physics Problems from previous Exams 3
 Introduction to Modern Physics Problems from previous Exams 3 2007 An electron of mass 9 10 31 kg moves along the x axis at a velocity.9c. a. Calculate the rest energy of the electron. b. Calculate its
Introduction to Modern Physics Problems from previous Exams 3 2007 An electron of mass 9 10 31 kg moves along the x axis at a velocity.9c. a. Calculate the rest energy of the electron. b. Calculate its
Stars and their properties: (Chapters 11 and 12)
 Stars and their properties: (Chapters 11 and 12) To classify stars we determine the following properties for stars: 1. Distance : Needed to determine how much energy stars produce and radiate away by using
Stars and their properties: (Chapters 11 and 12) To classify stars we determine the following properties for stars: 1. Distance : Needed to determine how much energy stars produce and radiate away by using
Lecture Outlines Chapter 32. Physics, 3 rd Edition James S. Walker
 Lecture Outlines Chapter 32 Physics, 3 rd Edition James S. Walker 2007 Pearson Prentice Hall This work is protected by United States copyright laws and is provided solely for the use of instructors in
Lecture Outlines Chapter 32 Physics, 3 rd Edition James S. Walker 2007 Pearson Prentice Hall This work is protected by United States copyright laws and is provided solely for the use of instructors in
T7-1 [255 marks] The graph shows the relationship between binding energy per nucleon and nucleon number. In which region are nuclei most stable?
![T7-1 [255 marks] The graph shows the relationship between binding energy per nucleon and nucleon number. In which region are nuclei most stable? T7-1 [255 marks] The graph shows the relationship between binding energy per nucleon and nucleon number. In which region are nuclei most stable?](/thumbs/77/75398334.jpg) T7-1 [255 marks] 1. In the Geiger Marsden experiment alpha particles were directed at a thin gold foil. Which of the following shows how the majority of the alpha particles behaved after reaching the foil?
T7-1 [255 marks] 1. In the Geiger Marsden experiment alpha particles were directed at a thin gold foil. Which of the following shows how the majority of the alpha particles behaved after reaching the foil?
Alta Chemistry CHAPTER 25. Nuclear Chemistry: Radiation, Radioactivity & its Applications
 CHAPTER 25 Nuclear Chemistry: Radiation, Radioactivity & its Applications Nuclear Chemistry Nuclear Chemistry deals with changes in the nucleus The nucleus of an atom contains Protons Positively Charged
CHAPTER 25 Nuclear Chemistry: Radiation, Radioactivity & its Applications Nuclear Chemistry Nuclear Chemistry deals with changes in the nucleus The nucleus of an atom contains Protons Positively Charged
Fission & Fusion Movie
 Fission & Fusion Movie Matter and Energy Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law. 2 H 2 O 2 H 2 + O 2 We now need to
Fission & Fusion Movie Matter and Energy Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law. 2 H 2 O 2 H 2 + O 2 We now need to
High Mass Stars. Dr Ken Rice. Discovering Astronomy G
 High Mass Stars Dr Ken Rice High mass star formation High mass star formation is controversial! May form in the same way as low-mass stars Gravitational collapse in molecular clouds. May form via competitive
High Mass Stars Dr Ken Rice High mass star formation High mass star formation is controversial! May form in the same way as low-mass stars Gravitational collapse in molecular clouds. May form via competitive
Type II Supernovae Overwhelming observational evidence that Type II supernovae are associated with the endpoints of massive stars: Association with
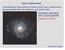 Type II Supernovae Overwhelming observational evidence that Type II supernovae are associated with the endpoints of massive stars: Association with spiral arms in spiral galaxies Supernova in M75 Type
Type II Supernovae Overwhelming observational evidence that Type II supernovae are associated with the endpoints of massive stars: Association with spiral arms in spiral galaxies Supernova in M75 Type
Atoms have two separate parts. The nucleus and the electron cloud.
 Name Ch. 5 - Atomic Structure Pre-AP Modern Atomic Theory All atoms are made of three subatomic (smaller than the atom) particles: the protons, the electrons and the neutrons. (P.E.N. s) There are particles
Name Ch. 5 - Atomic Structure Pre-AP Modern Atomic Theory All atoms are made of three subatomic (smaller than the atom) particles: the protons, the electrons and the neutrons. (P.E.N. s) There are particles
How Do Stars Appear from Earth?
 How Do Stars Appear from Earth? Magnitude: the brightness a star appears to have from Earth Apparent Magnitude depends on 2 things: (actual intrinsic brightness) The color of a star is related to its temperature:
How Do Stars Appear from Earth? Magnitude: the brightness a star appears to have from Earth Apparent Magnitude depends on 2 things: (actual intrinsic brightness) The color of a star is related to its temperature:
Nuclear & Particle Physics
 AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia A. Nuclear Structure Nuclear & Particle Physics B. Nuclear Decay C. Nuclear Reactions D. Particle Physics Updated: 0Aug8 Rough draft A. Nuclear Structure
AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia A. Nuclear Structure Nuclear & Particle Physics B. Nuclear Decay C. Nuclear Reactions D. Particle Physics Updated: 0Aug8 Rough draft A. Nuclear Structure
Radioactivity. Nuclear Physics. # neutrons vs# protons Where does the energy released in the nuclear 11/29/2010 A=N+Z. Nuclear Binding, Radioactivity
 Physics 1161: Lecture 25 Nuclear Binding, Radioactivity Sections 32-1 32-9 Marie Curie 1867-1934 Radioactivity Spontaneous emission of radiation from the nucleus of an unstable isotope. Antoine Henri Becquerel
Physics 1161: Lecture 25 Nuclear Binding, Radioactivity Sections 32-1 32-9 Marie Curie 1867-1934 Radioactivity Spontaneous emission of radiation from the nucleus of an unstable isotope. Antoine Henri Becquerel
Nuclear & Particle Physics
 AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia A. Nuclear Structure Nuclear & Particle Physics B. Nuclear Decay C. Nuclear Reactions D. Particle Physics Updated: 03Aug9 (for physics 700) A. Nuclear
AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia A. Nuclear Structure Nuclear & Particle Physics B. Nuclear Decay C. Nuclear Reactions D. Particle Physics Updated: 03Aug9 (for physics 700) A. Nuclear
The Stars. Chapter 14
 The Stars Chapter 14 Great Idea: The Sun and other stars use nuclear fusion reactions to convert mass into energy. Eventually, when a star s nuclear fuel is depleted, the star must burn out. Chapter Outline
The Stars Chapter 14 Great Idea: The Sun and other stars use nuclear fusion reactions to convert mass into energy. Eventually, when a star s nuclear fuel is depleted, the star must burn out. Chapter Outline
Lecture PowerPoints. Chapter 31 Physics: Principles with Applications, 7th edition Giancoli
 Lecture PowerPoints Chapter 31 Physics: Principles with Applications, 7th edition Giancoli This work is protected by United States copyright laws and is provided solely for the use of instructors in teaching
Lecture PowerPoints Chapter 31 Physics: Principles with Applications, 7th edition Giancoli This work is protected by United States copyright laws and is provided solely for the use of instructors in teaching
Nuclear Reactions A Z. Radioactivity, Spontaneous Decay: Nuclear Reaction, Induced Process: x + X Y + y + Q Q > 0. Exothermic Endothermic
 Radioactivity, Spontaneous Decay: Nuclear Reactions A Z 4 P D+ He + Q A 4 Z 2 Q > 0 Nuclear Reaction, Induced Process: x + X Y + y + Q Q = ( m + m m m ) c 2 x X Y y Q > 0 Q < 0 Exothermic Endothermic 2
Radioactivity, Spontaneous Decay: Nuclear Reactions A Z 4 P D+ He + Q A 4 Z 2 Q > 0 Nuclear Reaction, Induced Process: x + X Y + y + Q Q = ( m + m m m ) c 2 x X Y y Q > 0 Q < 0 Exothermic Endothermic 2
Nucleosynthesis. W. F. McDonough 1. Neutrino Science, Tohoku University, Sendai , Japan. (Dated: Tuesday 24 th April, 2018)
 Nucleosynthesis W. F. McDonough 1 1 Department of Earth Sciences and Research Center for Neutrino Science, Tohoku University, Sendai 980-8578, Japan (Dated: Tuesday 24 th April, 2018) Goals Where were
Nucleosynthesis W. F. McDonough 1 1 Department of Earth Sciences and Research Center for Neutrino Science, Tohoku University, Sendai 980-8578, Japan (Dated: Tuesday 24 th April, 2018) Goals Where were
Nuclear Physics. Slide 1 / 87. Slide 2 / 87. Slide 3 / 87. Table of Contents.
 Slide 1 / 87 Slide 2 / 87 Nuclear Physics www.njctl.org Table of Contents Slide 3 / 87 Click on the topic to go to that section Nuclear Structure Binding Energy and Mass Defect Radioactivity Nuclear Half-life
Slide 1 / 87 Slide 2 / 87 Nuclear Physics www.njctl.org Table of Contents Slide 3 / 87 Click on the topic to go to that section Nuclear Structure Binding Energy and Mass Defect Radioactivity Nuclear Half-life
Nuclear Physics
 Slide 1 / 87 Slide 2 / 87 Nuclear Physics www.njctl.org Slide 3 / 87 Table of Contents Click on the topic to go to that section Nuclear Structure Binding Energy and Mass Defect Radioactivity Nuclear Half-life
Slide 1 / 87 Slide 2 / 87 Nuclear Physics www.njctl.org Slide 3 / 87 Table of Contents Click on the topic to go to that section Nuclear Structure Binding Energy and Mass Defect Radioactivity Nuclear Half-life
Chapter 22. Preview. Objectives Properties of the Nucleus Nuclear Stability Binding Energy Sample Problem. Section 1 The Nucleus
 Section 1 The Nucleus Preview Objectives Properties of the Nucleus Nuclear Stability Binding Energy Sample Problem Section 1 The Nucleus Objectives Identify the properties of the nucleus of an atom. Explain
Section 1 The Nucleus Preview Objectives Properties of the Nucleus Nuclear Stability Binding Energy Sample Problem Section 1 The Nucleus Objectives Identify the properties of the nucleus of an atom. Explain
Nuclear Physics. Radioactivity. # protons = # neutrons. Strong Nuclear Force. Checkpoint 4/17/2013. A Z Nucleus = Protons+ Neutrons
 Marie Curie 1867-1934 Radioactivity Spontaneous emission of radiation from the nucleus of an unstable isotope. Antoine Henri Becquerel 1852-1908 Wilhelm Roentgen 1845-1923 Nuclear Physics A Z Nucleus =
Marie Curie 1867-1934 Radioactivity Spontaneous emission of radiation from the nucleus of an unstable isotope. Antoine Henri Becquerel 1852-1908 Wilhelm Roentgen 1845-1923 Nuclear Physics A Z Nucleus =
NUCLEI 1. The nuclei having the same atomic number (Z), but different mass numbers (A) are called isotopes.
 UCLEI Important Points: 1. The nuclei having the same atomic number (Z), but different mass numbers (A) are called isotopes. Ex: 1 H, 2 H, 3 1 1 1H are the isotopes of hydrogen atom. 2. The nuclei having
UCLEI Important Points: 1. The nuclei having the same atomic number (Z), but different mass numbers (A) are called isotopes. Ex: 1 H, 2 H, 3 1 1 1H are the isotopes of hydrogen atom. 2. The nuclei having
Nuclear Physics and Radioactivity
 Nuclear Physics and Radioactivity Structure and Properties of the Nucleus Nucleus is made of protons and neutrons Proton has positive charge: Neutron is electrically neutral: Neutrons and protons are collectively
Nuclear Physics and Radioactivity Structure and Properties of the Nucleus Nucleus is made of protons and neutrons Proton has positive charge: Neutron is electrically neutral: Neutrons and protons are collectively
Astronomy 104: Second Exam
 Astronomy 104: Second Exam Stephen Lepp October 29, 2014 Each question is worth 2 points. Write your name on this exam and on the scantron. Short Answer A The Sun is powered by converting hydrogen to what?
Astronomy 104: Second Exam Stephen Lepp October 29, 2014 Each question is worth 2 points. Write your name on this exam and on the scantron. Short Answer A The Sun is powered by converting hydrogen to what?
= : K A
 Atoms and Nuclei. State two limitations of JJ Thomson s model of atom. 2. Write the SI unit for activity of a radioactive substance. 3. What observations led JJ Thomson to conclusion that all atoms have
Atoms and Nuclei. State two limitations of JJ Thomson s model of atom. 2. Write the SI unit for activity of a radioactive substance. 3. What observations led JJ Thomson to conclusion that all atoms have
THE NUCLEUS: A CHEMIST S VIEW Chapter 20
 THE NUCLEUS: A CHEMIST S VIEW Chapter 20 "For a long time I have considered even the craziest ideas about [the] atom[ic] nucleus... and suddenly discovered the truth." [shell model of the nucleus]. Maria
THE NUCLEUS: A CHEMIST S VIEW Chapter 20 "For a long time I have considered even the craziest ideas about [the] atom[ic] nucleus... and suddenly discovered the truth." [shell model of the nucleus]. Maria
Lecture PowerPoint. Chapter 31 Physics: Principles with Applications, 6 th edition Giancoli
 Lecture PowerPoint Chapter 31 Physics: Principles with Applications, 6 th edition Giancoli 2005 Pearson Prentice Hall This work is protected by United States copyright laws and is provided solely for the
Lecture PowerPoint Chapter 31 Physics: Principles with Applications, 6 th edition Giancoli 2005 Pearson Prentice Hall This work is protected by United States copyright laws and is provided solely for the
Nuclear Physics. Slide 1 / 87. Slide 2 / 87. Slide 3 / 87. Table of Contents.
 Slide 1 / 87 Slide 2 / 87 Nuclear Physics www.njctl.org Table of Contents Slide 3 / 87 Click on the topic to go to that section Nuclear Structure Binding Energy and Mass Defect Radioactivity Nuclear Half-life
Slide 1 / 87 Slide 2 / 87 Nuclear Physics www.njctl.org Table of Contents Slide 3 / 87 Click on the topic to go to that section Nuclear Structure Binding Energy and Mass Defect Radioactivity Nuclear Half-life
Nuclear Physics. Nuclear Structure. Slide 1 / 87 Slide 2 / 87. Slide 4 / 87. Slide 3 / 87. Slide 6 / 87. Slide 5 / 87. Table of Contents.
 Slide 1 / 87 Slide 2 / 87 Nuclear Physics www.njctl.org Slide 3 / 87 Slide 4 / 87 Table of Contents Click on the topic to go to that section Nuclear Structure Binding Energy and Mass Defect Radioactivity
Slide 1 / 87 Slide 2 / 87 Nuclear Physics www.njctl.org Slide 3 / 87 Slide 4 / 87 Table of Contents Click on the topic to go to that section Nuclear Structure Binding Energy and Mass Defect Radioactivity
Nuclear Physics
 Slide 1 / 87 Slide 2 / 87 Nuclear Physics www.njctl.org Slide 3 / 87 Table of Contents Click on the topic to go to that section Nuclear Structure Binding Energy and Mass Defect Radioactivity Nuclear Half-life
Slide 1 / 87 Slide 2 / 87 Nuclear Physics www.njctl.org Slide 3 / 87 Table of Contents Click on the topic to go to that section Nuclear Structure Binding Energy and Mass Defect Radioactivity Nuclear Half-life
9.2.E - Particle Physics. Year 12 Physics 9.8 Quanta to Quarks
 + 9.2.E - Particle Physics Year 12 Physics 9.8 Quanta to Quarks + Atomic Size n While an atom is tiny, the nucleus is ten thousand times smaller than the atom and the quarks and electrons are at least
+ 9.2.E - Particle Physics Year 12 Physics 9.8 Quanta to Quarks + Atomic Size n While an atom is tiny, the nucleus is ten thousand times smaller than the atom and the quarks and electrons are at least
 Atoms and Nuclei 1. The radioactivity of a sample is X at a time t 1 and Y at a time t 2. If the mean life time of the specimen isτ, the number of atoms that have disintegrated in the time interval (t
Atoms and Nuclei 1. The radioactivity of a sample is X at a time t 1 and Y at a time t 2. If the mean life time of the specimen isτ, the number of atoms that have disintegrated in the time interval (t
Class XII Chapter 13 - Nuclei Physics
 Question 13.1: (a) Two stable isotopes of lithium and have respective abundances of 7.5% and 92.5%. These isotopes have masses 6.01512 u and 7.01600 u, respectively. Find the atomic mass of lithium. (b)
Question 13.1: (a) Two stable isotopes of lithium and have respective abundances of 7.5% and 92.5%. These isotopes have masses 6.01512 u and 7.01600 u, respectively. Find the atomic mass of lithium. (b)
Star Formation A cloud of gas and dust, called a nebula, begins spinning & heating up. Eventually, it gets hot enough for fusion to take place, and a
 Stars Star- large ball of gas held together by gravity that produces tremendous amounts of energy and shines Sun- our closest star Star Formation A cloud of gas and dust, called a nebula, begins spinning
Stars Star- large ball of gas held together by gravity that produces tremendous amounts of energy and shines Sun- our closest star Star Formation A cloud of gas and dust, called a nebula, begins spinning
Atomic and Nuclear Physics. Topic 7.3 Nuclear Reactions
 Atomic and Nuclear Physics Topic 7.3 Nuclear Reactions Nuclear Reactions Rutherford conducted experiments bombarding nitrogen gas with alpha particles from bismuth-214. He discovered that fast-moving particles
Atomic and Nuclear Physics Topic 7.3 Nuclear Reactions Nuclear Reactions Rutherford conducted experiments bombarding nitrogen gas with alpha particles from bismuth-214. He discovered that fast-moving particles
Brock University. Test 1, February, 2017 Number of pages: 9 Course: ASTR 1P02 Number of Students: 480 Date of Examination: February 6, 2017
 Brock University Test 1, February, 2017 Number of pages: 9 Course: ASTR 1P02 Number of Students: 480 Date of Examination: February 6, 2017 Number of hours: 50 min Time of Examination: 18:00 18:50 Instructor:
Brock University Test 1, February, 2017 Number of pages: 9 Course: ASTR 1P02 Number of Students: 480 Date of Examination: February 6, 2017 Number of hours: 50 min Time of Examination: 18:00 18:50 Instructor:
Chapter 30. Nuclear Energy and Elementary Particles
 Chapter 30 Nuclear Energy and Elementary Particles Processes of Nuclear Energy Fission A nucleus of large mass number splits into two smaller nuclei Fusion Two light nuclei fuse to form a heavier nucleus
Chapter 30 Nuclear Energy and Elementary Particles Processes of Nuclear Energy Fission A nucleus of large mass number splits into two smaller nuclei Fusion Two light nuclei fuse to form a heavier nucleus
DEVIL PHYSICS THE BADDEST CLASS ON CAMPUS IB PHYSICS
 DEVIL PHYSICS THE BADDEST CLASS ON CAMPUS IB PHYSICS TSOKOS LESSON 6-3 NUCLEAR REACTIONS Review Videos-Radioactivity2 Review Videos - Strong and Weak Nuclear Forces IB Assessment Statements, Topic 7.3
DEVIL PHYSICS THE BADDEST CLASS ON CAMPUS IB PHYSICS TSOKOS LESSON 6-3 NUCLEAR REACTIONS Review Videos-Radioactivity2 Review Videos - Strong and Weak Nuclear Forces IB Assessment Statements, Topic 7.3
AST 301 Introduction to Astronomy
 AST 301 Introduction to Astronomy John Lacy RLM 16.332 471-1469 lacy@astro.as.utexas.edu Myoungwon Jeon RLM 16.216 471-0445 myjeon@astro.as.utexas.edu Bohua Li RLM 16.212 471-8443 bohuali@astro.as.utexas.edu
AST 301 Introduction to Astronomy John Lacy RLM 16.332 471-1469 lacy@astro.as.utexas.edu Myoungwon Jeon RLM 16.216 471-0445 myjeon@astro.as.utexas.edu Bohua Li RLM 16.212 471-8443 bohuali@astro.as.utexas.edu
PHY-105: Introduction to Particle and Nuclear Physics
 M. Kruse, Spring 2011, Phy-105 PHY-105: Introduction to Particle and Nuclear Physics Up to 1900 indivisable atoms Early 20th century electrons, protons, neutrons Around 1945, other particles discovered.
M. Kruse, Spring 2011, Phy-105 PHY-105: Introduction to Particle and Nuclear Physics Up to 1900 indivisable atoms Early 20th century electrons, protons, neutrons Around 1945, other particles discovered.
Section 2: Nuclear Fission and Fusion. Preview Key Ideas Bellringer Nuclear Forces Nuclear Fission Chain Reaction Nuclear Fusion
 : Nuclear Fission and Fusion Preview Key Ideas Bellringer Nuclear Forces Nuclear Fission Chain Reaction Nuclear Fusion Key Ideas What holds the nuclei of atoms together? What is released when the nucleus
: Nuclear Fission and Fusion Preview Key Ideas Bellringer Nuclear Forces Nuclear Fission Chain Reaction Nuclear Fusion Key Ideas What holds the nuclei of atoms together? What is released when the nucleus
Slide 1 / 57. Nuclear Physics & Nuclear Reactions Practice Problems
 Slide 1 / 57 Nuclear Physics & Nuclear Reactions Practice Problems Slide 2 / 57 Multiple Choice Slide 3 / 57 1 The atomic nucleus consists of: A B C D E Electrons Protons Protons and electrons Protons
Slide 1 / 57 Nuclear Physics & Nuclear Reactions Practice Problems Slide 2 / 57 Multiple Choice Slide 3 / 57 1 The atomic nucleus consists of: A B C D E Electrons Protons Protons and electrons Protons
There are 82 protons in a lead nucleus. Why doesn t the lead nucleus burst apart?
 Question 32.1 The Nucleus There are 82 protons in a lead nucleus. Why doesn t the lead nucleus burst apart? a) Coulomb repulsive force doesn t act inside the nucleus b) gravity overpowers the Coulomb repulsive
Question 32.1 The Nucleus There are 82 protons in a lead nucleus. Why doesn t the lead nucleus burst apart? a) Coulomb repulsive force doesn t act inside the nucleus b) gravity overpowers the Coulomb repulsive
Fission and Fusion Book pg cgrahamphysics.com 2016
 Fission and Fusion Book pg 286-287 cgrahamphysics.com 2016 Review BE is the energy that holds a nucleus together. This is equal to the mass defect of the nucleus. Also called separation energy. The energy
Fission and Fusion Book pg 286-287 cgrahamphysics.com 2016 Review BE is the energy that holds a nucleus together. This is equal to the mass defect of the nucleus. Also called separation energy. The energy
CH 222 Chapter Twenty-one Concept Guide
 CH 222 Chapter Twenty-one Concept Guide 1. Terminology Alpha Radiation (α): Beta Radiation (β): Gamma Radiation (γ): Nuclear Reaction: Nucleons: Radioactive Decay Series: Positrons: Nuclear Binding Energy:
CH 222 Chapter Twenty-one Concept Guide 1. Terminology Alpha Radiation (α): Beta Radiation (β): Gamma Radiation (γ): Nuclear Reaction: Nucleons: Radioactive Decay Series: Positrons: Nuclear Binding Energy:
Brock University. Test 1, January, 2015 Number of pages: 9 Course: ASTR 1P02 Number of Students: 500 Date of Examination: January 29, 2015
 Brock University Test 1, January, 2015 Number of pages: 9 Course: ASTR 1P02 Number of Students: 500 Date of Examination: January 29, 2015 Number of hours: 50 min Time of Examination: 18:00 15:50 Instructor:
Brock University Test 1, January, 2015 Number of pages: 9 Course: ASTR 1P02 Number of Students: 500 Date of Examination: January 29, 2015 Number of hours: 50 min Time of Examination: 18:00 15:50 Instructor:
MockTime.com. Ans: (b) Q6. Curie is a unit of [1989] (a) energy of gamma-rays (b) half-life (c) radioactivity (d) intensity of gamma-rays Ans: (c)
![MockTime.com. Ans: (b) Q6. Curie is a unit of [1989] (a) energy of gamma-rays (b) half-life (c) radioactivity (d) intensity of gamma-rays Ans: (c) MockTime.com. Ans: (b) Q6. Curie is a unit of [1989] (a) energy of gamma-rays (b) half-life (c) radioactivity (d) intensity of gamma-rays Ans: (c)](/thumbs/95/125098222.jpg) Chapter Nuclei Q1. A radioactive sample with a half life of 1 month has the label: Activity = 2 micro curies on 1 8 1991. What would be its activity two months earlier? [1988] 1.0 micro curie 0.5 micro
Chapter Nuclei Q1. A radioactive sample with a half life of 1 month has the label: Activity = 2 micro curies on 1 8 1991. What would be its activity two months earlier? [1988] 1.0 micro curie 0.5 micro
Preview. Subatomic Physics Section 1. Section 1 The Nucleus. Section 2 Nuclear Decay. Section 3 Nuclear Reactions. Section 4 Particle Physics
 Subatomic Physics Section 1 Preview Section 1 The Nucleus Section 2 Nuclear Decay Section 3 Nuclear Reactions Section 4 Particle Physics Subatomic Physics Section 1 TEKS The student is expected to: 5A
Subatomic Physics Section 1 Preview Section 1 The Nucleus Section 2 Nuclear Decay Section 3 Nuclear Reactions Section 4 Particle Physics Subatomic Physics Section 1 TEKS The student is expected to: 5A
Card #1/28. Card #2/28. Science Revision P2. Science Revision P2. Science Revision P2. Card #4/28. Topic: F = ma. Topic: Resultant Forces
 Card #1/28 Card #2/28 Topic: Resultant Forces Topic: F = ma Topic: Distance-TIme Graphs Card #3/28 Card #4/28 Topic: Velocity-Time Graphs Card #2/28 Card #1/28 Card #4/28 Card #3/28 Card #5/28 Card #6/28
Card #1/28 Card #2/28 Topic: Resultant Forces Topic: F = ma Topic: Distance-TIme Graphs Card #3/28 Card #4/28 Topic: Velocity-Time Graphs Card #2/28 Card #1/28 Card #4/28 Card #3/28 Card #5/28 Card #6/28
Quantum Mechanics. Exam 3. Photon(or electron) interference? Photoelectric effect summary. Using Quantum Mechanics. Wavelengths of massive objects
 Exam 3 Hour Exam 3: Wednesday, November 29th In-class, Quantum Physics and Nuclear Physics Twenty multiple-choice questions Will cover:chapters 13, 14, 15 and 16 Lecture material You should bring 1 page
Exam 3 Hour Exam 3: Wednesday, November 29th In-class, Quantum Physics and Nuclear Physics Twenty multiple-choice questions Will cover:chapters 13, 14, 15 and 16 Lecture material You should bring 1 page
Every atom has a nucleus which contains protons and neutrons (both these particles are known nucleons). Orbiting the nucleus, are electrons.
 Atomic Structure Every atom has a nucleus which contains protons and neutrons (both these particles are known nucleons). Orbiting the nucleus, are electrons. Proton Number (Atomic Number): Amount of protons
Atomic Structure Every atom has a nucleus which contains protons and neutrons (both these particles are known nucleons). Orbiting the nucleus, are electrons. Proton Number (Atomic Number): Amount of protons
u d Fig. 6.1 (i) Identify the anti-proton from the table of particles shown in Fig [1]
![u d Fig. 6.1 (i) Identify the anti-proton from the table of particles shown in Fig [1] u d Fig. 6.1 (i) Identify the anti-proton from the table of particles shown in Fig [1]](/thumbs/79/80122691.jpg) 1 (a) Fig. 6.1 shows the quark composition of some particles. proton neutron A B u u d u d d u d u u u u d Fig. 6.1 (i) Identify the anti-proton from the table of particles shown in Fig. 6.1. (ii) State
1 (a) Fig. 6.1 shows the quark composition of some particles. proton neutron A B u u d u d d u d u u u u d Fig. 6.1 (i) Identify the anti-proton from the table of particles shown in Fig. 6.1. (ii) State
Fundamental Forces. Range Carrier Observed? Strength. Gravity Infinite Graviton No. Weak 10-6 Nuclear W+ W- Z Yes (1983)
 Fundamental Forces Force Relative Strength Range Carrier Observed? Gravity 10-39 Infinite Graviton No Weak 10-6 Nuclear W+ W- Z Yes (1983) Electromagnetic 10-2 Infinite Photon Yes (1923) Strong 1 Nuclear
Fundamental Forces Force Relative Strength Range Carrier Observed? Gravity 10-39 Infinite Graviton No Weak 10-6 Nuclear W+ W- Z Yes (1983) Electromagnetic 10-2 Infinite Photon Yes (1923) Strong 1 Nuclear
 Question 13.1: Two stable isotopes of lithium and have respective abundances of 7.5% and 92.5%. These isotopes have masses 6.01512 u and 7.01600 u, respectively. Find the atomic mass of lithium. Boron
Question 13.1: Two stable isotopes of lithium and have respective abundances of 7.5% and 92.5%. These isotopes have masses 6.01512 u and 7.01600 u, respectively. Find the atomic mass of lithium. Boron
Chapter Three (Nuclear Radiation)
 Al-Mustansiriyah University College of Science Physics Department Fourth Grade Nuclear Physics Dr. Ali A. Ridha Chapter Three (Nuclear Radiation) (3-1) Nuclear Radiation Whenever a nucleus can attain a
Al-Mustansiriyah University College of Science Physics Department Fourth Grade Nuclear Physics Dr. Ali A. Ridha Chapter Three (Nuclear Radiation) (3-1) Nuclear Radiation Whenever a nucleus can attain a
Chapter 25. Nuclear Chemistry. Types of Radiation
 Chapter 25 Nuclear Chemistry Chemical Reactions 1. Bonds are broken and formed 2. Atoms may rearrange, but remain unchanged 3. Involve only valence electrons 4. Small energy changes 5. Reaction rate is
Chapter 25 Nuclear Chemistry Chemical Reactions 1. Bonds are broken and formed 2. Atoms may rearrange, but remain unchanged 3. Involve only valence electrons 4. Small energy changes 5. Reaction rate is
Name: New Document 1. Class: Date: 54 minutes. Time: 54 marks. Marks: Comments: Page 1 of 22
 New Document Name: Class: Date: Time: 54 minutes Marks: 54 marks Comments: Page of 22 (a) Uranium has two natural isotopes, uranium-235 and uranium-238. Use the correct answer from the box to complete
New Document Name: Class: Date: Time: 54 minutes Marks: 54 marks Comments: Page of 22 (a) Uranium has two natural isotopes, uranium-235 and uranium-238. Use the correct answer from the box to complete
Filling the intellectual Vacuum: Energy Production. Contenders: From early 1920s: probably fusion, but how?
 Life of Stars Filling the intellectual Vacuum: Contenders: Energy Production Gravitational contraction Radioactivity (1903) Annihilation (E=mc 2, 1905) of proton and electron Hydrogen to helium nuclear
Life of Stars Filling the intellectual Vacuum: Contenders: Energy Production Gravitational contraction Radioactivity (1903) Annihilation (E=mc 2, 1905) of proton and electron Hydrogen to helium nuclear
Neutron-to-proton ratio
 Neutron-to-proton ratio After one second, the Universe had cooled to 10 13 K. The Universe was filled with protons, neutrons, electrons, and neutrinos. The temperature was high enough that they interconverted
Neutron-to-proton ratio After one second, the Universe had cooled to 10 13 K. The Universe was filled with protons, neutrons, electrons, and neutrinos. The temperature was high enough that they interconverted
Recap I Lecture 41 Matthias Liepe, 2012
 Recap I Lecture 41 Matthias Liepe, 01 Recap II Nuclear Physics The nucleus Radioactive decay Fission Fusion Particle Physics: What is the Higgs? Today: Nuclear Physics: The Nucleus Positive charge and
Recap I Lecture 41 Matthias Liepe, 01 Recap II Nuclear Physics The nucleus Radioactive decay Fission Fusion Particle Physics: What is the Higgs? Today: Nuclear Physics: The Nucleus Positive charge and
Nuclear Chemistry. Decay Reactions The most common form of nuclear decay reactions are the following:
 Nuclear Chemistry Nuclear reactions are transmutation of the one element into another. We can describe nuclear reactions in a similar manner as regular chemical reactions using ideas of stoichiometry,
Nuclear Chemistry Nuclear reactions are transmutation of the one element into another. We can describe nuclear reactions in a similar manner as regular chemical reactions using ideas of stoichiometry,
Baryons, mesons and leptons are affected by particle interactions. Write an account of these interactions. Your account should:
 Baryons, mesons and leptons are affected by particle interactions. Write an account of these interactions. Your account should: include the names of the interactions identify the groups of particles that
Baryons, mesons and leptons are affected by particle interactions. Write an account of these interactions. Your account should: include the names of the interactions identify the groups of particles that
Forces and Nuclear Processes
 Forces and Nuclear Processes To understand how stars generate the enormous amounts of light they produce will require us to delve into a wee bit of physics. First we will examine the forces that act at
Forces and Nuclear Processes To understand how stars generate the enormous amounts of light they produce will require us to delve into a wee bit of physics. First we will examine the forces that act at
1. This question is about the Rutherford model of the atom.
 1. This question is about the Rutherford model of the atom. (a) Most alpha particles used to bombard a thin gold foil pass through the foil without a significant change in direction. A few alpha particles
1. This question is about the Rutherford model of the atom. (a) Most alpha particles used to bombard a thin gold foil pass through the foil without a significant change in direction. A few alpha particles
Phys 100 Astronomy (Dr. Ilias Fernini) Review Questions for Chapter 9
 Phys 0 Astronomy (Dr. Ilias Fernini) Review Questions for Chapter 9 MULTIPLE CHOICE 1. We know that giant stars are larger in diameter than the sun because * a. they are more luminous but have about the
Phys 0 Astronomy (Dr. Ilias Fernini) Review Questions for Chapter 9 MULTIPLE CHOICE 1. We know that giant stars are larger in diameter than the sun because * a. they are more luminous but have about the
Unit 2 - Origin of Elements
 Unit 2 - Origin of Elements 1. What is the basic difference between ultraviolet, visible, and infrared radiation? A) temperature B) half-life C) wave velocity D) wavelength 2. The diagram below shows part
Unit 2 - Origin of Elements 1. What is the basic difference between ultraviolet, visible, and infrared radiation? A) temperature B) half-life C) wave velocity D) wavelength 2. The diagram below shows part
Nuclear Reactions. Fission Fusion
 Nuclear Reactions Fission Fusion Nuclear Reactions and the Transmutation of Elements A nuclear reaction takes place when a nucleus is struck by another nucleus or particle. Compare with chemical reactions!
Nuclear Reactions Fission Fusion Nuclear Reactions and the Transmutation of Elements A nuclear reaction takes place when a nucleus is struck by another nucleus or particle. Compare with chemical reactions!
Chapter 16: Ionizing Radiation
 Chapter 6: Ionizing Radiation Goals of Period 6 Section 6.: To discuss unstable nuclei and their detection Section 6.2: To describe the sources of ionizing radiation Section 6.3: To introduce three types
Chapter 6: Ionizing Radiation Goals of Period 6 Section 6.: To discuss unstable nuclei and their detection Section 6.2: To describe the sources of ionizing radiation Section 6.3: To introduce three types
State the main interaction when an alpha particle is scattered by a gold nucleus
 Q1.(a) Scattering experiments are used to investigate the nuclei of gold atoms. In one experiment, alpha particles, all of the same energy (monoenergetic), are incident on a foil made from a single isotope
Q1.(a) Scattering experiments are used to investigate the nuclei of gold atoms. In one experiment, alpha particles, all of the same energy (monoenergetic), are incident on a foil made from a single isotope
Abundance of Elements. Relative abundance of elements in the Solar System
 Abundance of Elements Relative abundance of elements in the Solar System What is the origin of elements in the universe? Three elements formed in the first minutes after the big bang (hydrogen, helium
Abundance of Elements Relative abundance of elements in the Solar System What is the origin of elements in the universe? Three elements formed in the first minutes after the big bang (hydrogen, helium
Unit 3: Chemistry in Society Nuclear Chemistry Summary Notes
 St Ninian s High School Chemistry Department National 5 Chemistry Unit 3: Chemistry in Society Nuclear Chemistry Summary Notes Name Learning Outcomes After completing this topic you should be able to :
St Ninian s High School Chemistry Department National 5 Chemistry Unit 3: Chemistry in Society Nuclear Chemistry Summary Notes Name Learning Outcomes After completing this topic you should be able to :
The Physics of Particles and Forces David Wilson
 The Physics of Particles and Forces David Wilson Particle Physics Masterclass 21st March 2018 Overview David Wilson (TCD) Particles & Forces 2/30 Overview of Hadron Spectrum Collaboration (HadSpec) scattering
The Physics of Particles and Forces David Wilson Particle Physics Masterclass 21st March 2018 Overview David Wilson (TCD) Particles & Forces 2/30 Overview of Hadron Spectrum Collaboration (HadSpec) scattering
Chapter 22 - Nuclear Chemistry
 Chapter - Nuclear Chemistry - The Nucleus I. Introduction A. Nucleons. Neutrons and protons B. Nuclides. Atoms identified by the number of protons and neutrons in the nucleus 8 a. radium-8 or 88 Ra II.
Chapter - Nuclear Chemistry - The Nucleus I. Introduction A. Nucleons. Neutrons and protons B. Nuclides. Atoms identified by the number of protons and neutrons in the nucleus 8 a. radium-8 or 88 Ra II.
Nuclear Chemistry Unit
 Nuclear Chemistry Unit January 28th HW Due Thurs. 1/30 Read pages 284 291 Define: Radioactivity Nuclear Radiation Alpha Particle Beta Particle Gamma Ray Half-Life Answer: -Questions 1-3 -Write the symbols
Nuclear Chemistry Unit January 28th HW Due Thurs. 1/30 Read pages 284 291 Define: Radioactivity Nuclear Radiation Alpha Particle Beta Particle Gamma Ray Half-Life Answer: -Questions 1-3 -Write the symbols
Nuclear Chemistry Lecture Notes: I Radioactive Decay A. Type of decay: See table. B. Predicting Atomic Stability
 Nuclear Chemistry Lecture Notes: I Radioactive Decay A. Type of decay: See table Type Symbol Charge Mass (AMU) Effect on Atomic # Alpha α +2 4 decrease by 2 Beta β- -1 0 increase electron by 1 Beta β+
Nuclear Chemistry Lecture Notes: I Radioactive Decay A. Type of decay: See table Type Symbol Charge Mass (AMU) Effect on Atomic # Alpha α +2 4 decrease by 2 Beta β- -1 0 increase electron by 1 Beta β+
General Physics (PHY 2140)
 General Physics (PHY 2140) Lecture 20 Modern Physics Nuclear Energy and Elementary Particles Fission, Fusion and Reactors Elementary Particles Fundamental Forces Classification of Particles Conservation
General Physics (PHY 2140) Lecture 20 Modern Physics Nuclear Energy and Elementary Particles Fission, Fusion and Reactors Elementary Particles Fundamental Forces Classification of Particles Conservation
