(Electrostatics in Biology)
|
|
|
- Lindsay Chambers
- 5 years ago
- Views:
Transcription
1 Practice-oriented, student-friendly modernization of the biomedical education for strengthening the international competitiveness of the rural Hungarian universities TÁMOP C-13/1/KONV (Electrostatics in Biology)!X Alajos Bérczi Institute of Biophysics, BRC HAS, Szeged, Hungary 14th September, 2016! X
2 ... Most biophysics courses and textbooks (at least in Hungary) deal with electric potential differences (e.g. diffusion potential, Donnan potential, action potential, Goldman-Hodgkin-Katz potential, etc.) but neglect the electrostatics of point charges. This lecture is intended to make up for this lack by giving a selected introduction into the parts of electrostatics of point charges that will then be employed in explaining some experimental observations.... Introduction - Why electrostatics? - Science-historical background (Maxwell equations) Electrical charges in aqueous media - The Poisson-Boltzmann equation - The Debye-Hückel approximation - Electric dipoles in aqueous media - Counterion condensation Surface charges and surface potentials at interface - The Gouy-Chapman theory - Surface charge layers Employing electrostatics in biology - Determination of surface charge density and surface potential of membrane vesicles (electrophoresis, fluorescence method) - Monovalent salt dependence of enzyme reactions - Like-charge attraction in polyelectrolytes - Variety of redox potential of the very same redox center - Zn 2+ -transporting ATPase Outlook! X
3 Introduction Why electrostatics? Martin Rees (1999): Just six numbers -/- Csak hat szám =0.007; 2p+2n He -- but m=-0.7% actual / critical (?) ~0.04 (1) l the cosmological constant (?) very small Q ratio of two energies (gravitational/ rest-mass ) ~10 5 D spatial dimension 3 N the ratio of F el to F gr ~ X
4 Introduction Gravitational field: Electrostatic field: F = f m m r F = f q q r f = N m kg f = 1 = N m 4πε C always attractive attractive or repulsive For two protons or two electrons at l = 1 nm (=10 Å) distance: 2 protons F = N F = N F F = electrons F = N F = N F F =
5 Introduction Fundamental interactions in nature Gravitational Electromagnetic Weak Strong??? dark??? Relative strength ???
6 Why electrostatics in biology? Biological objects: mass (m), electrical charge(s) (q, Q), typical distances and sizes are in 0.5 nm to 1 μm range (R, r), colloid system, dielectric media ( m, p, a ), Na + Example: Na + and Cl, r =2 nm (20 Å), in water ( r =80), gravitation (Newton s law): F g f g m m r F g = N r Cl - electrostatic (Coulomb s law): F e r q q r F e = N F e >>> F g (F e = F g ) Why electrostatics? motion by diffusion: m s 1 propagation of the electromagnetic effect: m s 1
7 Why charge-charge interactions firstly? Let T=298 K (25 C), r =80, q = C, r =1 nm (5 nm), l =0.3 nm (0.5 nm), =90, thermal energy (for each degree of freedom): potential energy in charge-charge interaction: potential energy in dipole-charge interaction: potential energy in dipole-dipole interaction: E T 2 kt E E E qq d q d d r r r q q 1 2 r p1q 2 r 2 p p r where p ql + 1 3cos E T = J E q-q = J (E q-q = J) E d-q = J (E d-q = J) E d-d = J (E d-d = J) Charge induced dipole and dipole induced dipole interactions are weaker (depend on polarizability a of molecules) and their distance dependence is r 4 and r 6, respectively.
8 Electromagnetism: Maxwell equations Formulation in terms of total charge and current; The Maxwell s microscopic equations in vacuum Gauss' law: Name Differential form Integral form James Clark Maxwell Gauss' law for magnetism: Maxwell-Faraday equation (Faraday's law of induction): Ampère's circuital law (with Maxwell's correction): The four Maxwell s microscopic equations describe the electric and magnetic fields arising from varying distributions of electric charges and currents, and how those fields change in the vacuum in time. In the presence of matter, however, the following equations will modify the Maxwell s microscopic equations:
9 The Poisson-Boltzmann equation If (1) there are no time-dependent changes of the field parameters, (2) there is no magnetic field (B = 0) and (3) no electric current (J = 0), then the following 3 equations will describe the electrostatic field: (a) No curl (b) No divergence The combination of the above 4 equations will result the most general form of the Poisson equation that can be used as strating point in the discussions of almost all biophysical problems: If, furthermore, (4) there is no polarization (P = 0), (5) the aqueous medium is homogeneous and isotropic ( = 0 r = const.), (6) the distribution of ions are according to the Boltzmann statistics, and (7) under the influence of electrostatic interactions only, then the Poisson equation gets its most frequently used form the Piosson-Boltzmann equation: It should be noted that general solution of this equation does not exists.
10 3D-Coordinate systems
11 The Debye-Hückel approximation The Debye-Hückel (DH) limiting law for electrolytes. For small potentials, i.e. where ey<<kt, the exponential factor in the Boltzmann equation can be linearized (i.e. application of Taylor series expansion for all exponential terms in the Boltzmann equation), e x ~1+x+ and the Poisson-Boltzmann equation can be solved. If (i) only Coulomb interaction exists between ions (the other types of interaction are neglected), (ii) spherical symmetry can be applied for all ions, (iii) which are considered as point charges and ions around a point charge is approximated by ionic clouds with continuous charge distribution, and (iv) the medium is considered as a homogenous and isotropic continuum, we obtain the following equation for the potential distribution around a charge Ze where The Debye length is a useful measure of the spatial extent of the diffuse charge layer. The Debye lengths are in the nanoscale regime and significantly reduced by increasing ionic strength. and is the reciprocal Debye length, (1/ D ). A Debye sphere is a volume whose radius is the Debye length ( D ), outside of which charges are electrically screened. When the size of the central ion Ze is also taken into account, the potential distribution at r > R is:
12 Ion association temporary ion pair formation Ion association (temporary ion pair formation): it occurs when - the electrostatic energy of attraction between unlike charges is larger than the thermic energy; or with other words, - cations (z (+) ) and anions (z (-) ) in an aqueous medium are mutually present in each other s electrostatic field and the centre of an anion is closer to the centre of a cation than a critical distance (l crit ): l B + where is the Bjerrum length. 2 2l B 4l B l B Since l B 7 Å in an aqueous phase and at room temperature, ion pairs play important role in the properties of polyelectrolytes.
13 Dipoles and set of point charges in a well-defined space The electrostatic field of two point charges with opposite sign is the superposition of the two electrostatic fields of the point charges. Far from the dipole (i.e. when L<<r) the potential distribution will be Since r i q i R d i If +q i then The last equation would describe the superposition of the electrostatic fields of an electric point charge Q and of a permanent electric dipole p. In such case the p is called the dipole moment of a space charge.
14 Counterion condensation theory Polyelectrolytes (or polyions) are macromolecules in which a substantial portion of the constitutional units have ionizable and/or ionic groups (proteins, DNA, RNA). Polyelectrolytes which bear both cationic and anionic groups are called polyampholytes (proteins). Ion association theory predicts that such molecules exist always together with associated counterions. According to the original counterion condensation theory (G.S. Manning, 1969), if (i) the contour length of a linear polyelectrolyte is L, and (ii) this polymer chain bears N charged groups of valence z N, (the linear charge density of a polyelectrolyte molecule is z N en/l), and (iii) the valence of counterions is z ci, then counterions should condense around the polyelectrolyte molecules as long as the reduced charge spacing, l fulfils the following inequality: Remarks: (1) While the condensation of counterions is an assumption in the Manning s theory it automatically shows up in the solution of the Poisson- Boltzmann equation in cylindrical coordinates employed for rod-like polyelectrolytes. (2) While the original counterion condensation theory deals only with the polyelectrolytes (without any additional solutes in the medium), the second counterion condensation theory (Manning et al , Manning 2011) study the problem in the presence of a bias electrolyte solution. Charge renormalization: Counterion concentration profile vs. radial distance around an infinitely long rod with radius of 5 Å and linear charge density equal to the B-DNA in 0.01 M electrolyte of 1:1 type. The Poisson-Boltzmann model (solid line) and the counterion condensation model (broken line) is compared. where is the fraction of condensed z ci -valent counterions per polymer charged site. Thus, for instance, on B-DNA (L/N)1.7 Å and since l B 7 Å, z ci 0.8 in case of z ci =1 (Na + ), and z ci 0.9 in case of z ci =2 (Mg 2+ ).
15 The Gouy-Chapman theory The Gouy-Chapman theory for charged surfaces preceded the Debye-Hückel theory for electrolytes by about 10 years. The basic assumptions are identical in the two theories. Let us assume that (i) there is an infinite surface with uniform surface charge density of at the YZ plane and (ii) a homogeneous and isotropic aquous medium in the X>0 half space with (iii) point charges and the distribution of charges follows the Boltzmann statistics. In such case the Poisson-Boltzmann equation can be integrated once and we obtain the following one-dimensional form: Under the following boundary conditions the relationship between the surface charge density (), surface potential ( 0 ) and the ion concentrations (c i ) in the medium will be given as: Red curve refers to ionic medium of 20 mm 1:1 electrolyte. Blue curve refers to a medium of 20 mm (M + ), 30 mm (A ) and 5 mm (D 2+ ). is about 10 times that of the thylakoid membranes.
16 The Gouy-Chapman theory If there is only one biner z:z electrolyte present in the aqueous medium (e.g. z = 1 in case of NaCl and z = 2 in case of MgSO 4 ), and the bulk concentration of the electrolyte is c( ), the one dimensional potential distribution, ψ(x), is given by the following equations: where 2 mm MgATP -2 - anion r 0 =5 Å T=300 K 0.15 M NaCl r>r (r ion ) (1/)=0.256 nm 0.1 As m -2 Red arrows and curve 1 Gouy-Chapman theory Blue arrows and curve 2 Debye-Hückel theory Distribution of the potential in the electric double layer at 4 different surface charge densities in a 100 mm 1:1 electrolyte solution (left panel) and in 4 different 1:1 electrolyte solutions at fixed surface charge density (right panel)
17 Surface charge layers The Gouy-Chapman theory has been improved by taking into account the spatial distribution of surface charges at the interface and the penetration of electrolytes into the membrane phase. The interface region between aqueous and membrane phases is a region of continuous changes from point of view of both size and composition. Charged groups (e.g. phosphate), dipoles (e.g. =C=O and water), and induced dipoles determine both the relative permittivity ( r ) and the Debye length ( 1 ) in this region. Analytical form for the potential function thus can hardly be derived, however, numerical approximations do exist for some special cases.
18 Surface charge layers z:z electrolite With the following boundary conditions of and ( ) = 0 the problem can be solved completely. After introducing the dimensionless potential (y) and the reciprocal Debye length ( and ) in both compartments the potential distribution will be given as Potential distribution at membrane surface with space charge layer of thickness d = 10, 5, 0 Å (curves 1, 2, and 3, respectively). z = 1, c() = 0.1 M, e d d = = Cm 2 and m = a (solid lines) or m = 0.5 a (broken lines).
19
20 Surface charge density and surface potential determination (1) According to the Helmholtz-Smoluchowski equation, a particle s electrophoretic velocity (v el ) is proportianal to the applied electric field strength (E), the zeta potential (the potential at the hydrodynamic plane of shear SE on the picture - z), and inversely proportianal to the dynamic viscosity of the medium () Charge distribution around an electrophoretic particle (liposome) in electrolyte. The equation has been derived with the following assumptions: the radius of the vesicle is much higher than the Debye length (generally higher than 100 nm; e.g. colloid particles), the charges are at the particle/solution interface, the surface is smooth, and there is no specific adsorption influencing the hydrodynamic plane of shear (this is hardly fulfilled). A more general formulation is the Henry s equation which takes into account the ionic strength of the medium (via the Debye length 1/) and can be used for charged particles with radius R Uncertainity of surface potential with identical zeta potential; the real surface charge density cannot be inferred from zeta potential. where f H (R) is the Henry s function (which approaches 1 for small R and 3/2 for large R). Problem unsolved here: the relation between and 0?
21 Surface charge density and surface potential determination (2) The simplest spectroscopic method for determining the surface charge density of membrane vesicles uses the relative fluorescence intensity values of a water soluble dye which are obtained when the quenched fluorescence in the electric double layer is released by replacing the dye molecules with monovalent or divalent cations. We have seen that With a proper choice of fluorescent dye (e.g. 9-aminoacridine: 9AA), and ionic solutions [e.g. KCl and (HM)Br 2 or (DM)Br 2 ], and assuming that fluorescence quenching of fluorescent molecules in the electric double layer is independent of the valence of cations used for the titration, both s and y 0 can be determined as follows... PM vesicles (bar 1 nm) Flourescence titration
22 Surface charge density and surface potential determination (3) Let us introduce K = 2000FkTε ε e and y = exp F RT then the equation for surface charge density given before will be as follows: σ = K c y 1 where c i and z i is the concentration and the valency of the i-th ion in the solution. Titration of the surface potential with 1:1 and 2:1 electrolytes in the presence of some buffer solution we may write for the surface charge density and surface potential σ = K c + c y + y 2 σ = K c y + y 2 + c y + 2y 3 for the 1:1 electrolyte, and for the 2:1 electrolyte. The surface potential ( o ) and the surface charge density () can be calculated as ψ = 2kT e arc sinh 2 H c and σ = K c + c H c where H c = c c 3 c c 2 and c m and c d are the monovalent and divalent cation concentrations, respectively. Bérczi A and Møller IM (1993) Eur. Biophys. J. 22:
23 Monovalent salt dependence of enzyme reactions According to the Michaelis-Menten kinetics We have seen that The surface potential depends both on the surface charge density and on the concentration of all ions in the medium. Thus any change in the concentration of any ion will affect the apparent value of the Michaelis-Menten constants. where and z S is the valence of the substrate. (1) Shift of the ph optimum by monovalent salts. Let 0, 01 0, 1:1 electrolyte, z S 0, and let change the monovalent salt concentration c m2 =10c m1. As consequence, but 02 0, and thus the surface ph value increases (the surface concentration of H + decreases) and the optimum ph value will shift to a lower ph value. (2) Activation, inhibition by monovalent salts. Let 0, 01 0, 1:1 electrolyte, z S 0, and let change the monovalent salt concentration c m2 =10c m1. As consequence, but 02 0, and thus [S] 0 increases and K M, app decreases.
24 Like charge attraction of polyelectrolytes (1) At the beginning it was known that multivalent ions generate attractions between like-charged polyelectrolytes in a wide range of systems, while monovalent ions do not. It was also known that different ion valences are required to condense different polyelectrolytes. How multivalent does an ion have to be before it can condense a given polyelectrolyte? Using the charge-tunable M13 filamentous virus system and a family of artificial homologous divalent ions of different effective sizes, a multivalent ion-polyelectrolyte phase diagram was constructed, and an experimentally motivated general criterion for like-charged attraction based on the ion size and the Debye length was established. M13 virus with ~2700 major coat proteins Dimethonium cations (n=1,2,3,4) M13 virus 5 mm Tris-HCl, 1 mm NaN 3 Small Angle X-ray Scattering (SAXS) measurements When >d (the Gouy-Chapman length around the polyelectrolyte charges is larger than the size of divalent cations), local charge inversion is possible. This may result in condensation ( association ) of polyelectrolytes.
25 Like charge attraction of polyelectrolytes (2) The basic elements of the second counterion condensation theory (Ray and Manning, ) are as follows. For two parallel polyions (separated by distance r and in z:z electrolyte), there are three components of the free energy: - the ionic interaction within each polyion, W p (r); summation over all Debye-Hückel pairwise interactions among the renormalized charges within each polyion, - the ionic interaction between the two polyions, W pp (r) K 0 (r); summation over all Debye-Hückel pairwise interactions among the renormalized charges, one member of the pair on one polyion, the other on the other polyion, - the work of transfer of a subpopulation of counterions from the bulk solution to the condensed layer on the two polyions, W transfer (r); According to the second counterion condensation theory, W(r) comes in three separated pieces: - the near distances: a < r < (e) 1 where a is the cutoff distance of the closest approach, 1 is the Debye length, - the intermediate distance: (e) 1 < r < 1 where 1 is the Debye length, - the far region: r > 1 where 1 is the Debye length. In the three different regions different approximations are taken for the expansion of the zeroth-order modified Bessel function of the second kind, K 0 (r).
26 Born model for solvation... and the redox potential of proteins The simples model for charcterizing a charged particle in a medium is based on the Born model. According to this model the electrostatic self-energy of an ion with charge Q and effective radius R (Born radius of ion) in free space (vacuum) is given as and in a medium as Thus the Born free energy of hydration or solvation (i.e. the energy needed for moving the charge from the free space into the medium) can be defined as the difference of W m and W v Similarly to this, the energy needed for moving the charge Q from the aqueous medium into a hydrophobic medium can be defined as the difference of W h and W a Problems: - infinite media, - the dipole character of the medium is neglected,
27 Zn 2+ -transporting ATPase (ZntA); a putative mechanism (Nature 2014, doi: /nature13618) Putative zinc transport mechanism of ZntA from Shigella sonnei. Phosphorylation events in the intracellular domains drive large conformational changes that permit alternating access to transport sites in the membrane about 50 Å from the ATP-targeted catalytic aspartate. According to the model, (1) a high-affinity state (E1), which is open to the intracellular space, binds to Zn 2+ and enters an occluded state. (2) This state then undergoes phosphorylation. (3) Completion of this event (E1P) triggers the release of the Zn 2+, establishing an outward-facing, low-affinity state (E2P). (4) Release of the inorganic phosphate (Pi) yields the fully dephosphorylated conformation (E2), which is followed by... (5)... restoration of the inward-facing conformation (E1), which initiates a new reaction cycle.
28 Zn 2+ -transporting ATPase (ZntA); a putative mechanism (Nature 2014, doi: /nature13618) Putative zinc transport mechanism of ZntA from Shigella sonnei. A transport cycle based on schematic models of the E1 and E1P states and the E2P and E2-P i structures. (1) In the presence of intracellular zinc, Zn 2+ enters the ATPase through the electronegative funnel (red). (2) Upon Zn 2+ binding to the intramembranous ion-binding site (grey circle),... (3)... F210 and M187 occlude the ion entry funnel, preventing backflow of Zn 2+. (4) Substantial domain rearrangements in transition to the E2P state open the extracellular pathway, lowering the affinity for Zn 2+ and... (5)... mediating Zn 2+ release,... (6)... possibly stimulated by K693. (7) Dephosphorylation triggers closure of the transmembrane domain, in which K693 (as a built-in counter ion) forms a salt bridge with D714. Upon dephosphorylation, the side chains move to their initial positions before an E2 to E1 transition is stimulated by the presence of intracellular Zn 2+.
29 Outlook Deficiencies in and/or problems with the standard Poisson-Boltzmann approaches (the linearized and the non-linearized forms) are well known and originate from (or go back to) the two basic assumptions of the theory. The Poisson- Boltzmann theory, for instance, underestimates the actual potential at highly charged surfaces by 15 25% for monovalent and divalent electrolytes. Neglecting of (a) ion correlation, (b) dielectric saturation and (c) finite size of ions and solvent molecules largely contribute to the inaccuracy of the theory. The way to overcome the problem is possible only after 1) abandoning the assumption (constraint) for the dielectric permittivity of the medium; as soon as the length scale of the order of a few Ångstroms becomes important, approximation of aqueous solution as a uniform dielectric medium is no longer sufficient and the Poisson-Boltzmann equation has the following more general, nonhomogeneous form: ε ψ r + P = ρ r ε r ψ r = ρ r F z ε 1000ε c exp z eψ r kt where f (r) is the charge density function of fixed charges (on molecules), r (r) refers to the electrostatic inhomogeneity of the medium (space-dependent relative dielectric constant), otherwise as in earlier, 2) taking into account the finite size of ions and the charge distribution inside the macromolecules (see the term f (r) in the above equation), and 3) introducing non-electrostatic component in the model. All of these three modifications have been introduced by nowadays and modeling of any biological processes on the basis of electrostatic interactions between the participants has been the most promising and fruitful in theoretical biophysics and provides the background for molecular modelling. The past 100+ years have proven that electrostatic interaction is the only interaction among the 4 fundamental interactions in nature that governs the biological processes and provide perfect starting point for quantitative discussion of any biological problems. We must, however, also be aware of that as soon as we want to go as deep as atomic level, the only proper theoretical background is quantum physics (quantum electro-dynamics).
30 Thank you for your attention! This work is supported by the European Union, co-financed by the European Social Fund, within the framework of " Practiceoriented, student-friendly modernization of the biomedical education for strengthening the international competitiveness of the rural Hungarian universities " TÁMOP C-13/1/KONV project.
ELŐADÁS KIVONAT CLASSROOM LECTURE HANDOUT. financed by the program
 TÁMOP4.1.1.C13/1/KONV0140001 projekt Az élettudományiklinikai felsőoktatás gyakorlatorientált és hallgatóbarát korszerűsítése a vidéki képzőhelyek nemzetközi versenyképességének erősítésére program keretében
TÁMOP4.1.1.C13/1/KONV0140001 projekt Az élettudományiklinikai felsőoktatás gyakorlatorientált és hallgatóbarát korszerűsítése a vidéki képzőhelyek nemzetközi versenyképességének erősítésére program keretében
Lecture 3 Charged interfaces
 Lecture 3 Charged interfaces rigin of Surface Charge Immersion of some materials in an electrolyte solution. Two mechanisms can operate. (1) Dissociation of surface sites. H H H H H M M M +H () Adsorption
Lecture 3 Charged interfaces rigin of Surface Charge Immersion of some materials in an electrolyte solution. Two mechanisms can operate. (1) Dissociation of surface sites. H H H H H M M M +H () Adsorption
The change in free energy on transferring an ion from a medium of low dielectric constantε1 to one of high dielectric constant ε2:
 The Born Energy of an Ion The free energy density of an electric field E arising from a charge is ½(ε 0 ε E 2 ) per unit volume Integrating the energy density of an ion over all of space = Born energy:
The Born Energy of an Ion The free energy density of an electric field E arising from a charge is ½(ε 0 ε E 2 ) per unit volume Integrating the energy density of an ion over all of space = Born energy:
2 Structure. 2.1 Coulomb interactions
 2 Structure 2.1 Coulomb interactions While the information needed for reproduction of living systems is chiefly maintained in the sequence of macromolecules, any practical use of this information must
2 Structure 2.1 Coulomb interactions While the information needed for reproduction of living systems is chiefly maintained in the sequence of macromolecules, any practical use of this information must
Stability of colloidal systems
 Stability of colloidal systems Colloidal stability DLVO theory Electric double layer in colloidal systems Processes to induce charges at surfaces Key parameters for electric forces (ζ-potential, Debye
Stability of colloidal systems Colloidal stability DLVO theory Electric double layer in colloidal systems Processes to induce charges at surfaces Key parameters for electric forces (ζ-potential, Debye
Electrostatic Double Layer Force: Part III
 NPTEL Chemical Engineering Interfacial Engineering Module 3: Lecture 4 Electrostatic Double Layer Force: Part III Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati
NPTEL Chemical Engineering Interfacial Engineering Module 3: Lecture 4 Electrostatic Double Layer Force: Part III Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati
Bchem 675 Lecture 9 Electrostatics-Lecture 2 Debye-Hückel: Continued Counter ion condensation
 Bchem 675 Lecture 9 Electrostatics-Lecture 2 Debye-Hückel: Continued Counter ion condensation Ion:ion interactions What is the free energy of ion:ion interactions ΔG i-i? Consider an ion in a solution
Bchem 675 Lecture 9 Electrostatics-Lecture 2 Debye-Hückel: Continued Counter ion condensation Ion:ion interactions What is the free energy of ion:ion interactions ΔG i-i? Consider an ion in a solution
8.592J HST.452J: Statistical Physics in Biology
 Assignment # 4 8.592J HST.452J: Statistical Physics in Biology Coulomb Interactions 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions scales as Q 2 E c.
Assignment # 4 8.592J HST.452J: Statistical Physics in Biology Coulomb Interactions 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions scales as Q 2 E c.
INTERMOLECULAR AND SURFACE FORCES
 INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
Module 8: "Stability of Colloids" Lecture 38: "" The Lecture Contains: Calculation for CCC (n c )
 The Lecture Contains: Calculation for CCC (n c ) Relation between surface charge and electrostatic potential Extensions to DLVO theory file:///e /courses/colloid_interface_science/lecture38/38_1.htm[6/16/2012
The Lecture Contains: Calculation for CCC (n c ) Relation between surface charge and electrostatic potential Extensions to DLVO theory file:///e /courses/colloid_interface_science/lecture38/38_1.htm[6/16/2012
The effect of surface dipoles and of the field generated by a polarization gradient on the repulsive force
 Journal of Colloid and Interface Science 263 (2003) 156 161 www.elsevier.com/locate/jcis The effect of surface dipoles and of the field generated by a polarization gradient on the repulsive force Haohao
Journal of Colloid and Interface Science 263 (2003) 156 161 www.elsevier.com/locate/jcis The effect of surface dipoles and of the field generated by a polarization gradient on the repulsive force Haohao
Electrical double layer
 Electrical double layer Márta Berka és István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry http://dragon.unideb.hu/~kolloid/ 7. lecture Adsorption of strong electrolytes from
Electrical double layer Márta Berka és István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry http://dragon.unideb.hu/~kolloid/ 7. lecture Adsorption of strong electrolytes from
Electrophoretic Light Scattering Overview
 Electrophoretic Light Scattering Overview When an electric field is applied across an electrolytic solution, charged particles suspended in the electrolyte are attracted towards the electrode of opposite
Electrophoretic Light Scattering Overview When an electric field is applied across an electrolytic solution, charged particles suspended in the electrolyte are attracted towards the electrode of opposite
Soft Matter - Theoretical and Industrial Challenges Celebrating the Pioneering Work of Sir Sam Edwards
 Soft Matter - Theoretical and Industrial Challenges Celebrating the Pioneering Work of Sir Sam Edwards One Hundred Years of Electrified Interfaces: The Poisson-Boltzmann theory and some recent developments
Soft Matter - Theoretical and Industrial Challenges Celebrating the Pioneering Work of Sir Sam Edwards One Hundred Years of Electrified Interfaces: The Poisson-Boltzmann theory and some recent developments
Aqueous solutions. Solubility of different compounds in water
 Aqueous solutions Solubility of different compounds in water The dissolution of molecules into water (in any solvent actually) causes a volume change of the solution; the size of this volume change is
Aqueous solutions Solubility of different compounds in water The dissolution of molecules into water (in any solvent actually) causes a volume change of the solution; the size of this volume change is
Electrochemical Properties of Materials for Electrical Energy Storage Applications
 Electrochemical Properties of Materials for Electrical Energy Storage Applications Lecture Note 3 October 11, 2013 Kwang Kim Yonsei Univ., KOREA kbkim@yonsei.ac.kr 39 Y 88.91 8 O 16.00 7 N 14.01 34 Se
Electrochemical Properties of Materials for Electrical Energy Storage Applications Lecture Note 3 October 11, 2013 Kwang Kim Yonsei Univ., KOREA kbkim@yonsei.ac.kr 39 Y 88.91 8 O 16.00 7 N 14.01 34 Se
Electrolytes. Chapter Basics = = 131 2[ ]. (c) From both of the above = = 120 8[
![Electrolytes. Chapter Basics = = 131 2[ ]. (c) From both of the above = = 120 8[ Electrolytes. Chapter Basics = = 131 2[ ]. (c) From both of the above = = 120 8[](/thumbs/83/88791608.jpg) Chapter 1 Electrolytes 1.1 Basics Here we consider species that dissociate into positively and negatively charged species in solution. 1. Consider: 1 H (g) + 1 Cl (g) + ()+ () = { } = (+ )+ ( ) = 167[
Chapter 1 Electrolytes 1.1 Basics Here we consider species that dissociate into positively and negatively charged species in solution. 1. Consider: 1 H (g) + 1 Cl (g) + ()+ () = { } = (+ )+ ( ) = 167[
Classical Models of the Interface between an Electrode and Electrolyte. M.Sc. Ekaterina Gongadze
 Presented at the COMSOL Conference 009 Milan Classical Models of the Interface between an Electrode and Electrolyte M.Sc. Ekaterina Gongadze Faculty of Informatics and Electrical Engineering Comsol Conference
Presented at the COMSOL Conference 009 Milan Classical Models of the Interface between an Electrode and Electrolyte M.Sc. Ekaterina Gongadze Faculty of Informatics and Electrical Engineering Comsol Conference
1. Poisson-Boltzmann 1.1. Poisson equation. We consider the Laplacian. which is given in spherical coordinates by (2)
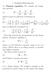 1. Poisson-Boltzmann 1.1. Poisson equation. We consider the Laplacian operator (1) 2 = 2 x + 2 2 y + 2 2 z 2 which is given in spherical coordinates by (2) 2 = 1 ( r 2 ) + 1 r 2 r r r 2 sin θ θ and in
1. Poisson-Boltzmann 1.1. Poisson equation. We consider the Laplacian operator (1) 2 = 2 x + 2 2 y + 2 2 z 2 which is given in spherical coordinates by (2) 2 = 1 ( r 2 ) + 1 r 2 r r r 2 sin θ θ and in
Foundations of. Colloid Science SECOND EDITION. Robert J. Hunter. School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS
 Foundations of Colloid Science SECOND EDITION Robert J. Hunter School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS CONTENTS 1 NATURE OF COLLOIDAL DISPERSIONS 1.1 Introduction 1 1.2 Technological
Foundations of Colloid Science SECOND EDITION Robert J. Hunter School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS CONTENTS 1 NATURE OF COLLOIDAL DISPERSIONS 1.1 Introduction 1 1.2 Technological
Multimedia : Boundary Lubrication Podcast, Briscoe, et al. Nature , ( )
 3.05 Nanomechanics of Materials and Biomaterials Thursday 04/05/07 Prof. C. Ortiz, MITDMSE I LECTURE 14: TE ELECTRICAL DOUBLE LAYER (EDL) Outline : REVIEW LECTURE #11 : INTRODUCTION TO TE ELECTRICAL DOUBLE
3.05 Nanomechanics of Materials and Biomaterials Thursday 04/05/07 Prof. C. Ortiz, MITDMSE I LECTURE 14: TE ELECTRICAL DOUBLE LAYER (EDL) Outline : REVIEW LECTURE #11 : INTRODUCTION TO TE ELECTRICAL DOUBLE
*blood and bones contain colloids. *milk is a good example of a colloidal dispersion.
 Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
Electrostatic Forces & The Electrical Double Layer
 Electrostatic Forces & The Electrical Double Layer Dry Clay Swollen Clay Repulsive electrostatics control swelling of clays in water LiquidSolid Interface; Colloids Separation techniques such as : column
Electrostatic Forces & The Electrical Double Layer Dry Clay Swollen Clay Repulsive electrostatics control swelling of clays in water LiquidSolid Interface; Colloids Separation techniques such as : column
Colloid Chemistry. La chimica moderna e la sua comunicazione Silvia Gross.
 Colloid Chemistry La chimica moderna e la sua comunicazione Silvia Gross Istituto Dipartimento di Scienze di e Scienze Tecnologie Chimiche Molecolari ISTM-CNR, Università Università degli Studi degli Studi
Colloid Chemistry La chimica moderna e la sua comunicazione Silvia Gross Istituto Dipartimento di Scienze di e Scienze Tecnologie Chimiche Molecolari ISTM-CNR, Università Università degli Studi degli Studi
SECOND PUBLIC EXAMINATION. Honour School of Physics Part C: 4 Year Course. Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS
 2757 SECOND PUBLIC EXAMINATION Honour School of Physics Part C: 4 Year Course Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS TRINITY TERM 2013 Monday, 17 June, 2.30 pm 5.45 pm 15
2757 SECOND PUBLIC EXAMINATION Honour School of Physics Part C: 4 Year Course Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS TRINITY TERM 2013 Monday, 17 June, 2.30 pm 5.45 pm 15
We have considered how Coulombic attractions and repulsions help to organize electrons in atoms and ions.
 CHEM 2060 Lecture 10: Electrostatics L10-1 Electrostatics of Atoms & Molecules We have considered how Coulombic attractions and repulsions help to organize electrons in atoms and ions. We now look at Coulombic
CHEM 2060 Lecture 10: Electrostatics L10-1 Electrostatics of Atoms & Molecules We have considered how Coulombic attractions and repulsions help to organize electrons in atoms and ions. We now look at Coulombic
István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry
 Colloid stability István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry www.kolloid.unideb.hu (Stability of lyophilic colloids see: macromolecular solutions) Stabilities 1.
Colloid stability István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry www.kolloid.unideb.hu (Stability of lyophilic colloids see: macromolecular solutions) Stabilities 1.
Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015,
 Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015, Course,Informa5on, BIOC%530% GraduateAlevel,discussion,of,the,structure,,func5on,,and,chemistry,of,proteins,and, nucleic,acids,,control,of,enzyma5c,reac5ons.,please,see,the,course,syllabus,and,
Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015, Course,Informa5on, BIOC%530% GraduateAlevel,discussion,of,the,structure,,func5on,,and,chemistry,of,proteins,and, nucleic,acids,,control,of,enzyma5c,reac5ons.,please,see,the,course,syllabus,and,
Theoretische Physik 2: Elektrodynamik (Prof. A-S. Smith) Home assignment 11
 WiSe 22..23 Prof. Dr. A-S. Smith Dipl.-Phys. Matthias Saba am Lehrstuhl für Theoretische Physik I Department für Physik Friedrich-Alexander-Universität Erlangen-Nürnberg Problem. Theoretische Physik 2:
WiSe 22..23 Prof. Dr. A-S. Smith Dipl.-Phys. Matthias Saba am Lehrstuhl für Theoretische Physik I Department für Physik Friedrich-Alexander-Universität Erlangen-Nürnberg Problem. Theoretische Physik 2:
Electrostatic Interactions in Mixtures of Cationic and Anionic Biomolecules: Bulk Structures and Induced Surface Pattern Formation
 Electrostatic Interactions in Mixtures of Cationic and Anionic Biomolecules: Bulk Structures and Induced Surface Pattern Formation Monica Olvera de la Cruz F. J. Solis, P. Gonzalez- Mozuleos (theory) E.
Electrostatic Interactions in Mixtures of Cationic and Anionic Biomolecules: Bulk Structures and Induced Surface Pattern Formation Monica Olvera de la Cruz F. J. Solis, P. Gonzalez- Mozuleos (theory) E.
Lecture 5: Electrostatic Interactions & Screening
 Lecture 5: Electrostatic Interactions & Screening Lecturer: Prof. Brigita Urbanc (brigita@drexel.edu) PHYS 461 & 561, Fall 2009-2010 1 A charged particle (q=+1) in water, at the interface between water
Lecture 5: Electrostatic Interactions & Screening Lecturer: Prof. Brigita Urbanc (brigita@drexel.edu) PHYS 461 & 561, Fall 2009-2010 1 A charged particle (q=+1) in water, at the interface between water
(Na++ K +)-ATPase in artificial lipid vesicles: influence of the concentration of mono- and divalent cations on the pumping rate
 254 Biochimica et Biophysica Acta 862 (1986) 254-264 Elsevier BBA 72961 (Na++ K +)-ATPase in artificial lipid vesicles: influence of the concentration of mono- and divalent cations on the pumping rate
254 Biochimica et Biophysica Acta 862 (1986) 254-264 Elsevier BBA 72961 (Na++ K +)-ATPase in artificial lipid vesicles: influence of the concentration of mono- and divalent cations on the pumping rate
V. Electrostatics. MIT Student
 V. Electrostatics Lecture 26: Compact Part of the Double Layer MIT Student 1 Double-layer Capacitance 1.1 Stern Layer As was discussed in the previous lecture, the Gouy-Chapman model predicts unphysically
V. Electrostatics Lecture 26: Compact Part of the Double Layer MIT Student 1 Double-layer Capacitance 1.1 Stern Layer As was discussed in the previous lecture, the Gouy-Chapman model predicts unphysically
SECOND PUBLIC EXAMINATION. Honour School of Physics Part C: 4 Year Course. Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS
 2757 SECOND PUBLIC EXAMINATION Honour School of Physics Part C: 4 Year Course Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS TRINITY TERM 2011 Monday, 27 June, 9.30 am 12.30 pm Answer
2757 SECOND PUBLIC EXAMINATION Honour School of Physics Part C: 4 Year Course Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS TRINITY TERM 2011 Monday, 27 June, 9.30 am 12.30 pm Answer
Surface interactions part 1: Van der Waals Forces
 CHEM-E150 Interfacial Phenomena in Biobased Systems Surface interactions part 1: Van der Waals Forces Monika Österberg Spring 018 Content Colloidal stability van der Waals Forces Surface Forces and their
CHEM-E150 Interfacial Phenomena in Biobased Systems Surface interactions part 1: Van der Waals Forces Monika Österberg Spring 018 Content Colloidal stability van der Waals Forces Surface Forces and their
V = 2ze 2 n. . a. i=1
 IITS: Statistical Physics in Biology Assignment # 3 KU Leuven 5/29/2013 Coulomb Interactions & Polymers 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions
IITS: Statistical Physics in Biology Assignment # 3 KU Leuven 5/29/2013 Coulomb Interactions & Polymers 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions
INTRODUCTION TO FINITE ELEMENT METHODS ON ELLIPTIC EQUATIONS LONG CHEN
 INTROUCTION TO FINITE ELEMENT METHOS ON ELLIPTIC EQUATIONS LONG CHEN CONTENTS 1. Poisson Equation 1 2. Outline of Topics 3 2.1. Finite ifference Method 3 2.2. Finite Element Method 3 2.3. Finite Volume
INTROUCTION TO FINITE ELEMENT METHOS ON ELLIPTIC EQUATIONS LONG CHEN CONTENTS 1. Poisson Equation 1 2. Outline of Topics 3 2.1. Finite ifference Method 3 2.2. Finite Element Method 3 2.3. Finite Volume
UvA-DARE (Digital Academic Repository) Electrokinetics in porous media Luong, D.T. Link to publication
 UvA-DARE (Digital Academic Repository) Electrokinetics in porous media Luong, D.T. Link to publication Citation for published version (APA): Luong, D. T. (2014). Electrokinetics in porous media General
UvA-DARE (Digital Academic Repository) Electrokinetics in porous media Luong, D.T. Link to publication Citation for published version (APA): Luong, D. T. (2014). Electrokinetics in porous media General
Chapter 3. Crystal Binding
 Chapter 3. Crystal Binding Energy of a crystal and crystal binding Cohesive energy of Molecular crystals Ionic crystals Metallic crystals Elasticity What causes matter to exist in three different forms?
Chapter 3. Crystal Binding Energy of a crystal and crystal binding Cohesive energy of Molecular crystals Ionic crystals Metallic crystals Elasticity What causes matter to exist in three different forms?
Fundamental Interactions: 6 Forces
 Fundamental Interactions: 6 Forces In nuclear and high-energy physics 6 fundamental forces are recognized, which describe the structure of matter. - the strong interaction - the weak interaction act inside
Fundamental Interactions: 6 Forces In nuclear and high-energy physics 6 fundamental forces are recognized, which describe the structure of matter. - the strong interaction - the weak interaction act inside
Theory and Applications of Dielectric Materials Introduction
 SERG Summer Seminar Series #11 Theory and Applications of Dielectric Materials Introduction Tzuyang Yu Associate Professor, Ph.D. Structural Engineering Research Group (SERG) Department of Civil and Environmental
SERG Summer Seminar Series #11 Theory and Applications of Dielectric Materials Introduction Tzuyang Yu Associate Professor, Ph.D. Structural Engineering Research Group (SERG) Department of Civil and Environmental
Charged Interfaces & electrokinetic
 Lecture Note #7 Charged Interfaces & electrokinetic phenomena Reading: Shaw, ch. 7 Origin of the charge at colloidal surfaces 1. Ionization Proteins acquire their charge by ionization of COOH and NH 2
Lecture Note #7 Charged Interfaces & electrokinetic phenomena Reading: Shaw, ch. 7 Origin of the charge at colloidal surfaces 1. Ionization Proteins acquire their charge by ionization of COOH and NH 2
Origin of the Electrophoretic Force on DNA in Nanopores. Biological and Soft Systems - Cavendish Laboratory
 Origin of the Electrophoretic Force on DNA in Nanopores Ulrich F. Keyser Biological and Soft Systems - Cavendish Laboratory Acknowledgements Delft Cees Dekker, Nynke H. Dekker, Serge G. Lemay R. Smeets,
Origin of the Electrophoretic Force on DNA in Nanopores Ulrich F. Keyser Biological and Soft Systems - Cavendish Laboratory Acknowledgements Delft Cees Dekker, Nynke H. Dekker, Serge G. Lemay R. Smeets,
Electrolyte Solutions
 Chapter 8 Electrolyte Solutions In the last few chapters of this book, we will deal with several specific types of chemical systems. The first one is solutions and equilibria involving electrolytes, which
Chapter 8 Electrolyte Solutions In the last few chapters of this book, we will deal with several specific types of chemical systems. The first one is solutions and equilibria involving electrolytes, which
International Journal of Engineering & Technology IJET-IJENS Vol:18 No:03 1
 International Journal of Engineering & Technology IJET-IJENS Vol:18 No:03 1 Analytical Derivation of Diffusio-osmosis Electric Potential and Velocity Distribution of an Electrolyte in a Fine Capillary
International Journal of Engineering & Technology IJET-IJENS Vol:18 No:03 1 Analytical Derivation of Diffusio-osmosis Electric Potential and Velocity Distribution of an Electrolyte in a Fine Capillary
Molecular Driving Forces
 Molecular Driving Forces Statistical Thermodynamics in Chemistry and Biology SUBGfittingen 7 At 216 513 073 / / Ken A. Dill Sarina Bromberg With the assistance of Dirk Stigter on the Electrostatics chapters
Molecular Driving Forces Statistical Thermodynamics in Chemistry and Biology SUBGfittingen 7 At 216 513 073 / / Ken A. Dill Sarina Bromberg With the assistance of Dirk Stigter on the Electrostatics chapters
957 Lecture #13 of 18
 Lecture #13 of 18 957 958 Q: What was in this set of lectures? A: B&F Chapter 2 main concepts: Section 2.1 : Section 2.3: Salt; Activity; Underpotential deposition Transference numbers; Liquid junction
Lecture #13 of 18 957 958 Q: What was in this set of lectures? A: B&F Chapter 2 main concepts: Section 2.1 : Section 2.3: Salt; Activity; Underpotential deposition Transference numbers; Liquid junction
Colloidal Suspension Rheology Chapter 1 Study Questions
 Colloidal Suspension Rheology Chapter 1 Study Questions 1. What forces act on a single colloidal particle suspended in a flowing fluid? Discuss the dependence of these forces on particle radius. 2. What
Colloidal Suspension Rheology Chapter 1 Study Questions 1. What forces act on a single colloidal particle suspended in a flowing fluid? Discuss the dependence of these forces on particle radius. 2. What
Solvation and Macromolecular Structure. The structure and dynamics of biological macromolecules are strongly influenced by water:
 Overview Solvation and Macromolecular Structure The structure and dynamics of biological macromolecules are strongly influenced by water: Electrostatic effects: charges are screened by water molecules
Overview Solvation and Macromolecular Structure The structure and dynamics of biological macromolecules are strongly influenced by water: Electrostatic effects: charges are screened by water molecules
Contents. 2. Fluids. 1. Introduction
 Contents 1. Introduction 2. Fluids 3. Physics of Microfluidic Systems 4. Microfabrication Technologies 5. Flow Control 6. Micropumps 7. Sensors 8. Ink-Jet Technology 9. Liquid Handling 10.Microarrays 11.Microreactors
Contents 1. Introduction 2. Fluids 3. Physics of Microfluidic Systems 4. Microfabrication Technologies 5. Flow Control 6. Micropumps 7. Sensors 8. Ink-Jet Technology 9. Liquid Handling 10.Microarrays 11.Microreactors
PH 202-1E Fall Electric Forces and Electric Fields. Lectures 1-4. Chapter 18 (Cutnell & Johnson, Physics 6 th edition)
 PH 202-1E Fall 2006 Electric Forces and Electric Fields Lectures 1-4 Chapter 18 (Cutnell & Johnson, Physics 6 th edition) 1 Electric Force and Electric Charge Qualitatively, a force is any push or pull
PH 202-1E Fall 2006 Electric Forces and Electric Fields Lectures 1-4 Chapter 18 (Cutnell & Johnson, Physics 6 th edition) 1 Electric Force and Electric Charge Qualitatively, a force is any push or pull
Physical Chemistry - Problem Drill 01: Chemistry and Physics Review
 Physical Chemistry - Problem Drill 01: Chemistry and Physics Review No. 1 of 10 1. Chemical bonds are considered to be the interaction of their electronic structures of bonding atoms involved, with the
Physical Chemistry - Problem Drill 01: Chemistry and Physics Review No. 1 of 10 1. Chemical bonds are considered to be the interaction of their electronic structures of bonding atoms involved, with the
Density Functional Theory for the Nonspecific Binding of Salt to Polyelectrolytes: Thermodynamic Properties
 Biophysical Journal Volume 78 February 2000 699 706 699 Density Functional Theory for the Nonspecific Binding of Salt to Polyelectrolytes: Thermodynamic Properties Chandra N. Patra and Arun Yethiraj Department
Biophysical Journal Volume 78 February 2000 699 706 699 Density Functional Theory for the Nonspecific Binding of Salt to Polyelectrolytes: Thermodynamic Properties Chandra N. Patra and Arun Yethiraj Department
Supporting Information for. Concentration dependent effects of bovine serum albumin on graphene
 Supporting Information for Concentration dependent effects of bovine serum albumin on graphene oxide colloidal stability in aquatic environment Binbin Sun, Yinqing Zhang, Wei Chen, Kunkun Wang, Lingyan
Supporting Information for Concentration dependent effects of bovine serum albumin on graphene oxide colloidal stability in aquatic environment Binbin Sun, Yinqing Zhang, Wei Chen, Kunkun Wang, Lingyan
An Overview of the Concept, Measurement, Use and Application of Zeta Potential. David Fairhurst, Ph.D. Colloid Consultants, Ltd
 An Overview of the Concept, Measurement, Use and Application of Zeta Potential David Fairhurst, Ph.D. Colloid Consultants, Ltd Fundamental Parameters that control the Nature and Behavior of all Particulate
An Overview of the Concept, Measurement, Use and Application of Zeta Potential David Fairhurst, Ph.D. Colloid Consultants, Ltd Fundamental Parameters that control the Nature and Behavior of all Particulate
Colloid stability. Lyophobic sols. Stabilization of colloids.
 Colloid stability. Lyophobic sols. Stabilization of colloids. Lyophilic and lyophobic sols Sols (lyosols) are dispersed colloidal size particles in a liquid medium (=solid/liquid dispersions) These sols
Colloid stability. Lyophobic sols. Stabilization of colloids. Lyophilic and lyophobic sols Sols (lyosols) are dispersed colloidal size particles in a liquid medium (=solid/liquid dispersions) These sols
Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers
 Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers Hong-ming Ding 1 & Yu-qiang Ma 1,2, 1 National Laboratory of Solid State Microstructures and Department
Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers Hong-ming Ding 1 & Yu-qiang Ma 1,2, 1 National Laboratory of Solid State Microstructures and Department
Single action pressing (from top)
 www.komage.de Single action pressing (from top) Double action pressing with fixed die Typical course of the pressure during pressing and ejection (Single action) Upper punch Pressure Lower punch Time Green
www.komage.de Single action pressing (from top) Double action pressing with fixed die Typical course of the pressure during pressing and ejection (Single action) Upper punch Pressure Lower punch Time Green
ELECTRO MAGNETIC FIELDS
 SET - 1 1. a) State and explain Gauss law in differential form and also list the limitations of Guess law. b) A square sheet defined by -2 x 2m, -2 y 2m lies in the = -2m plane. The charge density on the
SET - 1 1. a) State and explain Gauss law in differential form and also list the limitations of Guess law. b) A square sheet defined by -2 x 2m, -2 y 2m lies in the = -2m plane. The charge density on the
1044 Lecture #14 of 18
 Lecture #14 of 18 1044 1045 Q: What s in this set of lectures? A: B&F Chapter 13 main concepts: Section 1.2.3: Diffuse double layer structure Sections 13.1 & 13.2: Gibbs adsorption isotherm; Electrocapillary
Lecture #14 of 18 1044 1045 Q: What s in this set of lectures? A: B&F Chapter 13 main concepts: Section 1.2.3: Diffuse double layer structure Sections 13.1 & 13.2: Gibbs adsorption isotherm; Electrocapillary
Solutions and Non-Covalent Binding Forces
 Chapter 3 Solutions and Non-Covalent Binding Forces 3.1 Solvent and solution properties Molecules stick together using the following forces: dipole-dipole, dipole-induced dipole, hydrogen bond, van der
Chapter 3 Solutions and Non-Covalent Binding Forces 3.1 Solvent and solution properties Molecules stick together using the following forces: dipole-dipole, dipole-induced dipole, hydrogen bond, van der
Graduate Written Examination Fall 2014 Part I
 Graduate Written Examination Fall 2014 Part I University of Minnesota School of Physics and Astronomy Aug. 19, 2014 Examination Instructions Part 1 of this exam consists of 10 problems of equal weight.
Graduate Written Examination Fall 2014 Part I University of Minnesota School of Physics and Astronomy Aug. 19, 2014 Examination Instructions Part 1 of this exam consists of 10 problems of equal weight.
Biochemistry 675, Lecture 8 Electrostatic Interactions
 Biochemistry 675, Lecture 8 Electrostatic Interactions Previous Classes Hydrophobic Effect Hydrogen Bonds Today: Electrostatic Interactions Reading: Handout:P.R. Bergethon & E.R. Simons, Biophysical Chemistry,
Biochemistry 675, Lecture 8 Electrostatic Interactions Previous Classes Hydrophobic Effect Hydrogen Bonds Today: Electrostatic Interactions Reading: Handout:P.R. Bergethon & E.R. Simons, Biophysical Chemistry,
Protein separation and characterization
 Address:800 S Wineville Avenue, Ontario, CA 91761,USA Website:www.aladdin-e.com Email USA: tech@aladdin-e.com Email EU: eutech@aladdin-e.com Email Asia Pacific: cntech@aladdin-e.com Protein separation
Address:800 S Wineville Avenue, Ontario, CA 91761,USA Website:www.aladdin-e.com Email USA: tech@aladdin-e.com Email EU: eutech@aladdin-e.com Email Asia Pacific: cntech@aladdin-e.com Protein separation
Electrostatic correlations and fluctuations for ion binding to a finite length polyelectrolyte
 THE JOURNAL OF CHEMICAL PHYSICS 122, 044903 2005 Electrostatic correlations and fluctuations for ion binding to a finite length polyelectrolyte Zhi-Jie Tan and Shi-Jie Chen a) Department of Physics and
THE JOURNAL OF CHEMICAL PHYSICS 122, 044903 2005 Electrostatic correlations and fluctuations for ion binding to a finite length polyelectrolyte Zhi-Jie Tan and Shi-Jie Chen a) Department of Physics and
Unit-1 Electrostatics-1
 1. Describe about Co-ordinate Systems. Co-ordinate Systems Unit-1 Electrostatics-1 In order to describe the spatial variations of the quantities, we require using appropriate coordinate system. A point
1. Describe about Co-ordinate Systems. Co-ordinate Systems Unit-1 Electrostatics-1 In order to describe the spatial variations of the quantities, we require using appropriate coordinate system. A point
Soft Matter and Biological Physics
 Dr. Ulrich F. Keyser - ufk20 (at) cam.ac.uk Soft Matter and Biological Physics Question Sheet Michaelmas 2011 Version: November 2, 2011 Question 0: Sedimentation Initially consider identical small particles
Dr. Ulrich F. Keyser - ufk20 (at) cam.ac.uk Soft Matter and Biological Physics Question Sheet Michaelmas 2011 Version: November 2, 2011 Question 0: Sedimentation Initially consider identical small particles
CHEMISTRY XL-14A CHEMICAL BONDS
 CHEMISTRY XL-14A CHEMICAL BONDS July 16, 2011 Robert Iafe Office Hours 2 July 18-July 22 Monday: 2:00pm in Room MS-B 3114 Tuesday-Thursday: 3:00pm in Room MS-B 3114 Chapter 2 Overview 3 Ionic Bonds Covalent
CHEMISTRY XL-14A CHEMICAL BONDS July 16, 2011 Robert Iafe Office Hours 2 July 18-July 22 Monday: 2:00pm in Room MS-B 3114 Tuesday-Thursday: 3:00pm in Room MS-B 3114 Chapter 2 Overview 3 Ionic Bonds Covalent
Colloid Chemistry. La chimica moderna e la sua comunicazione Silvia Gross.
 Colloid Chemistry La chimica moderna e la sua comunicazione Silvia Gross Istituto Dipartimento di Scienze di e Scienze Tecnologie Chimiche Molecolari ISTM-CNR, Università Università degli Studi degli Studi
Colloid Chemistry La chimica moderna e la sua comunicazione Silvia Gross Istituto Dipartimento di Scienze di e Scienze Tecnologie Chimiche Molecolari ISTM-CNR, Università Università degli Studi degli Studi
INTRODUCTION. Introduction. Discrete charges: Electric dipole. Continuous charge distributions. Flux of a vector field
 Chapter 2 THE ELECTRC FELD ntroduction Discrete charges: Electric dipole Continuous charge distributions Flux of a vector field Flux of an electric field for a spherical Gaussian surface concentric to
Chapter 2 THE ELECTRC FELD ntroduction Discrete charges: Electric dipole Continuous charge distributions Flux of a vector field Flux of an electric field for a spherical Gaussian surface concentric to
free space (vacuum) permittivity [ F/m]
![free space (vacuum) permittivity [ F/m] free space (vacuum) permittivity [ F/m]](/thumbs/79/79624275.jpg) Electrostatic Fields Electrostatic fields are static (time-invariant) electric fields produced by static (stationary) charge distributions. The mathematical definition of the electrostatic field is derived
Electrostatic Fields Electrostatic fields are static (time-invariant) electric fields produced by static (stationary) charge distributions. The mathematical definition of the electrostatic field is derived
Final exam. Please write your name on the exam and keep an ID card ready. You may use a calculator (but no computer or smart phone) and a dictionary.
 Biophysics of Macromolecules Prof. D. Braun and Prof. J. Lipfert SS 2015 Final exam Final exam Name: Student number ( Matrikelnummer ): Please write your name on the exam and keep an ID card ready. You
Biophysics of Macromolecules Prof. D. Braun and Prof. J. Lipfert SS 2015 Final exam Final exam Name: Student number ( Matrikelnummer ): Please write your name on the exam and keep an ID card ready. You
Electrostatics of membrane adhesion
 Electrostatics of membrane adhesion S. Marcelja Department of Applied Mathematics, Research School of Physical Sciences and Engineering, The Australian National University, Canberra ACT 6, Australia ABSTRACT
Electrostatics of membrane adhesion S. Marcelja Department of Applied Mathematics, Research School of Physical Sciences and Engineering, The Australian National University, Canberra ACT 6, Australia ABSTRACT
Indiana University Physics P331: Theory of Electromagnetism Review Problems #3
 Indiana University Physics P331: Theory of Electromagnetism Review Problems #3 Note: The final exam (Friday 1/14 8:00-10:00 AM will be comprehensive, covering lecture and homework material pertaining to
Indiana University Physics P331: Theory of Electromagnetism Review Problems #3 Note: The final exam (Friday 1/14 8:00-10:00 AM will be comprehensive, covering lecture and homework material pertaining to
Peter Debye and Electrochemistry
 Peter Debye and Electrochemistry A K Shukla and T Prem Kumar A K Shukla is Professor and Chairman, Solid State and Structural Chemistry Unit, Indian Institute of Science, Bangalore. His research interests
Peter Debye and Electrochemistry A K Shukla and T Prem Kumar A K Shukla is Professor and Chairman, Solid State and Structural Chemistry Unit, Indian Institute of Science, Bangalore. His research interests
On the Chemical Free Energy of the Electrical Double Layer
 1114 Langmuir 23, 19, 1114-112 On the Chemical Free Energy of the Electrical Double Layer Marian Manciu and Eli Ruckenstein* Department of Chemical Engineering, State University of New York at Buffalo,
1114 Langmuir 23, 19, 1114-112 On the Chemical Free Energy of the Electrical Double Layer Marian Manciu and Eli Ruckenstein* Department of Chemical Engineering, State University of New York at Buffalo,
Peptide folding in non-aqueous environments investigated with molecular dynamics simulations Soto Becerra, Patricia
 University of Groningen Peptide folding in non-aqueous environments investigated with molecular dynamics simulations Soto Becerra, Patricia IMPORTANT NOTE: You are advised to consult the publisher's version
University of Groningen Peptide folding in non-aqueous environments investigated with molecular dynamics simulations Soto Becerra, Patricia IMPORTANT NOTE: You are advised to consult the publisher's version
Computational Neuroscience. Session 2-1
 Computational Neuroscience. Session 2-1 Dr. Marco A Roque Sol 06/04/2018 All living cells exhibit an electrical potential difference between the inner and outer surface of the cytoplasmic membrane. This
Computational Neuroscience. Session 2-1 Dr. Marco A Roque Sol 06/04/2018 All living cells exhibit an electrical potential difference between the inner and outer surface of the cytoplasmic membrane. This
16 years ago TODAY (9/11) at 8:46, the first tower was hit at 9:03, the second tower was hit. Lecture 2 (9/11/17)
 16 years ago TODAY (9/11) at 8:46, the first tower was hit at 9:03, the second tower was hit By Anthony Quintano - https://www.flickr.com/photos/quintanomedia/15071865580, CC BY 2.0, https://commons.wikimedia.org/w/index.php?curid=38538291
16 years ago TODAY (9/11) at 8:46, the first tower was hit at 9:03, the second tower was hit By Anthony Quintano - https://www.flickr.com/photos/quintanomedia/15071865580, CC BY 2.0, https://commons.wikimedia.org/w/index.php?curid=38538291
Inorganic Pharmaceutical Chemistry
 Inorganic Pharmaceutical Chemistry Lecture No. 4 Date :25/10 /2012 Dr. Mohammed Hamed --------------------------------------------------------------------------------------------------------------------------------------
Inorganic Pharmaceutical Chemistry Lecture No. 4 Date :25/10 /2012 Dr. Mohammed Hamed --------------------------------------------------------------------------------------------------------------------------------------
Research Statement. Shenggao Zhou. November 3, 2014
 Shenggao Zhou November 3, My research focuses on: () Scientific computing and numerical analysis (numerical PDEs, numerical optimization, computational fluid dynamics, and level-set method for interface
Shenggao Zhou November 3, My research focuses on: () Scientific computing and numerical analysis (numerical PDEs, numerical optimization, computational fluid dynamics, and level-set method for interface
An Introduction to namic Light Scattering by Macromole cules
 An Introduction to namic Light Scattering by Macromole cules Kenneth S. Schmitz Department of Chemistry University of Missouri-Kansas Kansas City, Missouri City ACADEMIC PRESS, INC. Harcourt Brace Jovanovich,
An Introduction to namic Light Scattering by Macromole cules Kenneth S. Schmitz Department of Chemistry University of Missouri-Kansas Kansas City, Missouri City ACADEMIC PRESS, INC. Harcourt Brace Jovanovich,
Chapter 11. Intermolecular Forces and Liquids & Solids
 Chapter 11 Intermolecular Forces and Liquids & Solids The Kinetic Molecular Theory of Liquids & Solids Gases vs. Liquids & Solids difference is distance between molecules Liquids Molecules close together;
Chapter 11 Intermolecular Forces and Liquids & Solids The Kinetic Molecular Theory of Liquids & Solids Gases vs. Liquids & Solids difference is distance between molecules Liquids Molecules close together;
V. Electrostatics Lecture 24: Diffuse Charge in Electrolytes
 V. Electrostatics Lecture 24: Diffuse Charge in Electrolytes MIT Student 1. Poisson-Nernst-Planck Equations The Nernst-Planck Equation is a conservation of mass equation that describes the influence of
V. Electrostatics Lecture 24: Diffuse Charge in Electrolytes MIT Student 1. Poisson-Nernst-Planck Equations The Nernst-Planck Equation is a conservation of mass equation that describes the influence of
Harris: Quantitative Chemical Analysis, Eight Edition CHAPTER 25: CHROMATOGRAPHIC METHODS AND CAPILLARY ELECTROPHORESIS
 Harris: Quantitative Chemical Analysis, Eight Edition CHAPTER 25: CHROMATOGRAPHIC METHODS AND CAPILLARY ELECTROPHORESIS CHAPTER 25: Opener Aa CHAPTER 25: Opener Ab CHAPTER 25: Opener B 25-1 Ion-Exchange
Harris: Quantitative Chemical Analysis, Eight Edition CHAPTER 25: CHROMATOGRAPHIC METHODS AND CAPILLARY ELECTROPHORESIS CHAPTER 25: Opener Aa CHAPTER 25: Opener Ab CHAPTER 25: Opener B 25-1 Ion-Exchange
Chapter 12 Intermolecular Forces and Liquids
 Chapter 12 Intermolecular Forces and Liquids Jeffrey Mack California State University, Sacramento Why? Why is water usually a liquid and not a gas? Why does liquid water boil at such a high temperature
Chapter 12 Intermolecular Forces and Liquids Jeffrey Mack California State University, Sacramento Why? Why is water usually a liquid and not a gas? Why does liquid water boil at such a high temperature
Specific ion effects on the interaction of. hydrophobic and hydrophilic self assembled
 Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Chapter 13: Phenomena
 Chapter 13: Phenomena Phenomena: Scientists measured the bond angles of some common molecules. In the pictures below each line represents a bond that contains 2 electrons. If multiple lines are drawn together
Chapter 13: Phenomena Phenomena: Scientists measured the bond angles of some common molecules. In the pictures below each line represents a bond that contains 2 electrons. If multiple lines are drawn together
CE 530 Molecular Simulation
 1 CE 530 Molecular Simulation Lecture 14 Molecular Models David A. Kofke Department of Chemical Engineering SUNY Buffalo kofke@eng.buffalo.edu 2 Review Monte Carlo ensemble averaging, no dynamics easy
1 CE 530 Molecular Simulation Lecture 14 Molecular Models David A. Kofke Department of Chemical Engineering SUNY Buffalo kofke@eng.buffalo.edu 2 Review Monte Carlo ensemble averaging, no dynamics easy
Lec.1 Chemistry Of Water
 Lec.1 Chemistry Of Water Biochemistry & Medicine Biochemistry can be defined as the science concerned with the chemical basis of life. Biochemistry can be described as the science concerned with the chemical
Lec.1 Chemistry Of Water Biochemistry & Medicine Biochemistry can be defined as the science concerned with the chemical basis of life. Biochemistry can be described as the science concerned with the chemical
UNIT I ELECTROSTATIC FIELDS
 UNIT I ELECTROSTATIC FIELDS 1) Define electric potential and potential difference. 2) Name few applications of gauss law in electrostatics. 3) State point form of Ohm s Law. 4) State Divergence Theorem.
UNIT I ELECTROSTATIC FIELDS 1) Define electric potential and potential difference. 2) Name few applications of gauss law in electrostatics. 3) State point form of Ohm s Law. 4) State Divergence Theorem.
Surface Forces & Liquid Films (Answers to Exercise Problems)
 //5 Surface Forces & Liquid Films (nswers to Exercise Problems) Wuge H. Briscoe wuge.briscoe@bris.ac.uk URL: wugebrisco7.wix.com/briscoegroup Exercise : van der Waals forces & liquid films When octane
//5 Surface Forces & Liquid Films (nswers to Exercise Problems) Wuge H. Briscoe wuge.briscoe@bris.ac.uk URL: wugebrisco7.wix.com/briscoegroup Exercise : van der Waals forces & liquid films When octane
Dipole - Electronegativity - Nonpolar covalent bond - Partial charges - Polar covalent bond - Polarity -
 Chemistry Electronegativity & Polarity Guided Inquiry (Textbook 10.3) Use your knowledge of chemical bonding, the textbook, your classmates, the PhET simulation and the internet to answer the questions
Chemistry Electronegativity & Polarity Guided Inquiry (Textbook 10.3) Use your knowledge of chemical bonding, the textbook, your classmates, the PhET simulation and the internet to answer the questions
CHAPTER 2. COULOMB S LAW AND ELECTRONIC FIELD INTENSITY. 2.3 Field Due to a Continuous Volume Charge Distribution
 CONTENTS CHAPTER 1. VECTOR ANALYSIS 1. Scalars and Vectors 2. Vector Algebra 3. The Cartesian Coordinate System 4. Vector Cartesian Coordinate System 5. The Vector Field 6. The Dot Product 7. The Cross
CONTENTS CHAPTER 1. VECTOR ANALYSIS 1. Scalars and Vectors 2. Vector Algebra 3. The Cartesian Coordinate System 4. Vector Cartesian Coordinate System 5. The Vector Field 6. The Dot Product 7. The Cross
Chemistry 452/ August 2012
 Chemistry 45/456 7 August 0 End- of-term Examination Professor G. Drobny Enter your answers into a blue or green Composition Book. Perform only the number of problems required. Answers must be given in
Chemistry 45/456 7 August 0 End- of-term Examination Professor G. Drobny Enter your answers into a blue or green Composition Book. Perform only the number of problems required. Answers must be given in
Complicated, short range. þq 1 Q 2 /4p3 0 r (Coulomb energy) Q 2 u 2 /6(4p3 0 ) 2 ktr 4. u 2 1 u2 2 =3ð4p3 0Þ 2 ktr 6 ðkeesom energyþ
 Bonding ¼ Type of interaction Interaction energy w(r) Covalent, metallic Complicated, short range Charge charge þq 1 Q 2 /4p3 0 r (Coulomb energy) Charge dipole Qu cos q/4p3 0 r 2 Q 2 u 2 /6(4p3 0 ) 2
Bonding ¼ Type of interaction Interaction energy w(r) Covalent, metallic Complicated, short range Charge charge þq 1 Q 2 /4p3 0 r (Coulomb energy) Charge dipole Qu cos q/4p3 0 r 2 Q 2 u 2 /6(4p3 0 ) 2
Chapter 4. Electrostatic Fields in Matter
 Chapter 4. Electrostatic Fields in Matter 4.1. Polarization 4.2. The Field of a Polarized Object 4.3. The Electric Displacement 4.4. Linear Dielectrics 4.5. Energy in dielectric systems 4.6. Forces on
Chapter 4. Electrostatic Fields in Matter 4.1. Polarization 4.2. The Field of a Polarized Object 4.3. The Electric Displacement 4.4. Linear Dielectrics 4.5. Energy in dielectric systems 4.6. Forces on
Supporting Text Z = 2Γ 2+ + Γ + Γ [1]
![Supporting Text Z = 2Γ 2+ + Γ + Γ [1] Supporting Text Z = 2Γ 2+ + Γ + Γ [1]](/thumbs/91/105230015.jpg) Supporting Text RNA folding experiments are typically carried out in a solution containing a mixture of monovalent and divalent ions, usually MgCl 2 and NaCl or KCl. All three species of ions, Mg, M +
Supporting Text RNA folding experiments are typically carried out in a solution containing a mixture of monovalent and divalent ions, usually MgCl 2 and NaCl or KCl. All three species of ions, Mg, M +
MUDRA PHYSICAL SCIENCES
 MUDRA PHYSICAL SCIENCES VOLUME- PART B & C MODEL QUESTION BANK FOR THE TOPICS:. Electromagnetic Theory UNIT-I UNIT-II 7 4. Quantum Physics & Application UNIT-I 8 UNIT-II 97 (MCQs) Part B & C Vol- . Electromagnetic
MUDRA PHYSICAL SCIENCES VOLUME- PART B & C MODEL QUESTION BANK FOR THE TOPICS:. Electromagnetic Theory UNIT-I UNIT-II 7 4. Quantum Physics & Application UNIT-I 8 UNIT-II 97 (MCQs) Part B & C Vol- . Electromagnetic
Molecular Forces in Biological Systems - Electrostatic Interactions; - Shielding of charged objects in solution
 Molecular Forces in Biological Systems - Electrostatic Interactions; - Shielding of charged objects in solution Electrostatic self-energy, effects of size and dielectric constant q q r r ε ε 1 ε 2? δq
Molecular Forces in Biological Systems - Electrostatic Interactions; - Shielding of charged objects in solution Electrostatic self-energy, effects of size and dielectric constant q q r r ε ε 1 ε 2? δq
