CHAPTER 16: REACTION ENERGY AND CHAPTER 17: REACTION KINETICS. Honors Chemistry Ms. Agostine
|
|
|
- Brittney Lang
- 5 years ago
- Views:
Transcription
1 CHAPTER 16: REACTION ENERGY AND CHAPTER 17: REACTION KINETICS Honors Chemistry Ms. Agostine
2 16.1 Thermochemistry Definition: study of the transfers of energy as heat that accompany chemical reactions and physical changes Measured by a calorimeter
3 Measuring Heat (Review) Units that measure heat/energy: CALORIES! Calorie (cal): the quantity of heat that raises the temperature of 1 g of pure water by 1 o C 1 Calorie = 1000 calories = 1 kilocalorie SI Units: joule (J) Conversion: 1 J = cal or 1 cal = J
4 Specific Heat (c) (Review) How the quantity of heat is actually measured Specific Heat (c) amount of energy required to raise the heat 1 gram of a substance by 1 degree Celsius q = m c T (heat quantity) = (mass)(spec. heat)(temp change) or rearranged to be: c = q m T
5 Specific Heat Water has a high specific heat and metals have low specific heat Therefore it takes less energy to heat up a metal then a nonmetal like water! That is why the beach is cooler in the summer then the city!
6 Heat of Reaction Definition: Quantity of heat released or absorbed during a chemical reaction Enthalpy (H): heat content of a system Enthalpy Change ( H): amount of heat absorbed or lost by a system during a process Can have Exothermic and Endothermic reactions
7 Enthalpy of Reaction Thermochemical Equation : equation that includes the quantity of heat released or absorbed during the reaction as written Depends on the amounts of reactants and products: H 2 (g) + ½ O 2 (g) H 2 O (g) kJ
8 Exothermic Reactions Energy is released to surroundings Products have LESS energy then reactants H is negative 2 H 2 (g) + O 2 (g) 2 H 2 O (g) H = kj
9 Endothermic Reactions Energy is absorbed from surroundings Products have MORE energy then reactants H is positive 2 H 2 O (g) 2 H 2 (g) + O 2 (g) H = kj
10 Standard Molar Heat of Formation ( H fo ) Definition: heat released or absorbed when one mole of a compound is formed by combination of its elements at standard room temperature of 25 o C Appendix Table A-14 p. 862 H 2 (g) + ½ O 2 (g) H 2 O (l) H o f = kj Na (s) + ½ Cl 2 (g) NaCl (s) H o f = kj S (s) + O 2 (g) SO 2 (g) H o f = kj Elements in their standard states have H o f = 0 kj A negative H o f indicates a substance is more stable than the free elements
11 Standard Heats of Formation
12 Molar Heat of Combustion ( H comb ) Definition: heat released when one mole of a compound combusts Appendix Table A-5, p. 856 C 3 H 8 (g) + 5 O 2 (g) 3 CO 2 (g) + 4 H 2 O (l) H comb = kj C 8 H 18 (g) O 2 (g) 8 CO 2 (g) + 9 H 2 O (l) H comb = kj
13 Calculating Heats of Reaction Hess s Law: the overall enthalpy change in a reaction is equal to the sum of enthalpy changes for the individual steps in the process In order for a reaction to occur: all reactants must have their bonds broken all products must have their bonds formed
14 Calculating Heats of Reactions Hess s Law of Heat Summation: If you add two or more thermochemical equations to give a final equation, then you can also add the heats of reaction to give the final heat of reaction H o = H f o products - H f o reactants ( = sum) This will also show if the net reaction is exothermic (-) or endothermic (+).
15 Sample Problem What is the H f o for the reaction between gaseous carbon monoxide with oxygen to form gaseous carbon dioxide? Balanced equation: 2 CO (g) + O 2 (g) 2 CO 2 (g) LOOK UP H f o CO (g) = kj/mol H f o O 2 (g) = 0 kj/mol (free element) H f o CO 2 (g) = kj/mol
16 2 CO (g) + O 2 (g) 2 CO 2 (g) H o = H f o products - H f o reactants H o = [2 mol x kj/mol] [2 mol x kj/mol] H o = [-791 kj] [-221 kj] H o = kj Therefore, this reaction is an exothermic process!
17 Calculating H Reaction from the Heats of Formation Reactions PbCl 2 + Cl 2 PbCl 4, H o =? Look up: H f o PbCl 2 = kj/mol Rxn of the formation: Pb + Cl 2 PbCl 2 H f o Cl 2 = 0 kj/mol b/c it s an element H f o PbCl 4 = kj/mol Rxn of the formation: Pb + 2 Cl 2 PbCl 4 In the reaction: PbCl 2 has to be broken, so reverse the reaction PbCl 4 has to be made, keep the reaction as written
18 Calculating H Reaction from the Heats of Formation Reactions H f o PbCl 2 = kj/mol FLIP IT! Rxn: PbCl 2 Pb + Cl 2 H f o Cl 2 = 0 kj/mol b/c it s an element H f o PbCl 4 = kj/mol Rxn: Pb + 2 Cl 2 PbCl 4 Complete: PbCl 2 + Pb + 2 Cl 2 Pb + Cl 2 + PbCl 4 Simplify: PbCl 2 + Pb + 2 Cl 2 Pb + Cl 2 + PbCl 4 Net: PbCl 2 + Cl 2 PbCl 4 Add H f o = kj kj = 30.2 kj
19 Calculating H Reaction from the Heats of Formation Reactions Check Answer with: H o = H f o products - H f o reactants H o = [1 mol x kj/mol] [1 mol x kj/mol] H o = [ kj] [ kj] H o = kj Therefore, this reaction is an endothermic process!
20 Calculating H Reaction from the Heats of Formation Reactions Calculate the heat of the reaction for the decomposition of calcium carbonate when heated CaCO 3 (s) CaO (s) + CO(g) H o =?
21 Solution Look up: H f o CaCO 3 = kj/mol Rxn : Ca + C + 3/2 O 2 CaCO 3 H f o CaO = kj/mol Rxn: Ca + ½ O 2 CaO H f o CO 2 = kj/mol Rxn: C + O 2 CO 2 In the reaction: CaCO 3 has to be broken, so reverse the rxn CaO & CO 2 has to be made, keep the rxns as written
22 Answer: Complete equation: CaCO 3 + Ca + C + 3/2 O 2 Ca + C + CaO + 3/2 O 2 + CO 2 x x x x x x Net Eqn: CaCO 3 (s) CaO (s) + CO(g) Add up heats of formation: CaCO 3 is reversed: kj CaO is the same: CO 2 is the same: TOTAL: kj kj kj, endothermic
23 Answer: Check your answer: H = products - reactants = [ kj kj] [-1207 kj] = kj
24 Section 16.2 Driving Force of Reactions What makes a reaction spontaneous? Ex: Ball rolls down a hill obvious, but why? Driving force: Potential energy is too high, so nature wants to lower it. SPONTANEOUS! Nature has a tendency of reactions to occur that lead to a lower energy state Therefore: ball will NOT roll up a hill!
25 Entropy (S) Definition: property that describes the order of a system Nature tends toward disorder Hint: easier to mess up or to clean up a room? We measure S in J/K mol Temperature can effect S
26 Free Energy Depends on two things: Enthalpy: H Entropy: S and the Temperature of the system Gibb s Free Energy: combined enthalpy-entropy of a system G = H - T S If G = (-) = rxn will be favorable/spontaneous If If G = (+) = rxn will be unfavorable/nonspontaneous Note: G o, H o, & S o = at standard temperature
27 Enthalpy, Entropy & Free Energy H S G - Value (exothermic) - Value (exothermic) + Value (endothermic) + Value (endothermic) + Value (more random) - Value (less random) + Value (more random) - Value (less random) Always negative lower temperatures higher temps Never negative
28 Free Energy Sample Problem For the reaction NH 4 Cl (s) NH 3 (g) + HCl (g), at K, H o = 176 kj/mol and S o = kj/(mol K). Calculate G o, and tell whether this reaction is spontaneous in the forward direction at this temperature.
29 Free Energy Sample Problem G = H - T S G = 176 kj/mol [298 K x kj/(mol K)] G = 176 kj/mol 84.9 kj/mol G = 91 kj/mol Non spontaneous because G = positive!
30 Chapter 17: Reaction Kinetics
31 Collision Theory
32 Section 17.1 The Reaction Process Reaction Mechanism: step-by-step sequence of reactions by which the overall chemical change occurs Why does the rate of chemical reactions vary so widely? Collision Theory: Reactant molecules must collide in the proper orientation and with sufficient energy Only a fraction of collisions meet these requirements
33 Collision Theory Consider: Ozone + smog nitrogen dioxide + oxygen
34 Collisions of NO and O3
35 Energy Diagrams Reactants must overcome an energy barrier before they can change to products Activation Energy (E a ): Minimum amount of energy that reactants must have before they can be converted to products
36 Energy Diagrams
37 Energy Diagrams Activated Complex: An unstable, high-energy chemical species that must be formed as reactants convert to products The in-between stage. Designated with a double superscript dagger ( ) such as {O 3 NO}
38 Energy Diagram for NO and O3
39 Exothermic vs. Endothermic
40 Energy Diagrams with Activated Complexes Example: The following is exothermic: 2 HI (g) H 2 (g) + I 2 (g) Draw an energy diagram that shows the relative energies of the reactants, products and the activated complex. Label the diagram with molecular representations of the reactants, products, and a possible structure for the activated complex.
41 Section Reaction Rates What makes milk and other foods spoil more quickly? What happens if you accidentally add a drop of bleach to a dark wash? A cup? What dissolves faster granulated sugar or a sugar cube?
42 Reaction Rates Factors that influence reaction rates 1. Temperature 2. Reaction concentration 3. Surface area 4. Presence of a catalyst Looking at each in the terms of Collision Theory!
43 Concentration Rxns go faster when the concentration of one or more of the reactants increases Increases in concentration increases the number of reactants per unit volume Because molecules are closer together, the number of collisions per unit time increases
44 Surface Area Similar to increase concentration More surface area means more molecules or atoms exposed to the reaction collisions Ex) granulated sugar vs. sugar cubes
45 Temperature The average kinetic energy of a substance increases when the temperature rises Increases the fraction of collisions that are effective Molecules move faster at higher temperatures, so they collide more frequently More molecules attain the required activation energy
46 Temperature Temperature effects on reaction rates are significant For a typical reaction, the rate approximately doubles for every 10 o C increase in temperature
47 Fraction of Collisions vs. Collision Different Temperatures
48 Catalysts Catalyst: A substance that alters the pathway in which a reaction occurs without itself being consumed in the reaction Lower-energy pathway with lower activation energy This way, increases the rate of a reaction Great fraction of reactants can achieve the new minimum energy requirement
49 Catalyst
50 Rate Laws for Reactions Rate Law: an equation that relates reaction rate and concentrations of reactants Specific Rate Laws for the directions of reactions Forward rate law is different then the reverse reaction rate law
51 Rate Laws for Reactions 2 H 2 (g) + 2 NO (g) N 2 (g) + 2 H 2 O (g) Rate Law of the forward reaction: Depends on concentrations of H 2 and NO Depends on temperature R = k [reactants] coeffient R = reaction rate K = rate constant (takes into account temperature) R = k [H 2 ] 2 [NO] 2
Chemistry Chapter 16. Reaction Energy
 Chemistry Reaction Energy Section 16.1.I Thermochemistry Objectives Define temperature and state the units in which it is measured. Define heat and state its units. Perform specific-heat calculations.
Chemistry Reaction Energy Section 16.1.I Thermochemistry Objectives Define temperature and state the units in which it is measured. Define heat and state its units. Perform specific-heat calculations.
Unit 7 Kinetics and Thermodynamics
 17.1 The Flow of Energy Heat and Work Unit 7 Kinetics and Thermodynamics I. Energy Transformations A. Temperature 1. A measure of the average kinetic energy of the particles in a sample of matter B. Heat
17.1 The Flow of Energy Heat and Work Unit 7 Kinetics and Thermodynamics I. Energy Transformations A. Temperature 1. A measure of the average kinetic energy of the particles in a sample of matter B. Heat
Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy
 Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Thermodynamics. Thermodynamics of Chemical Reactions. Enthalpy change
 Thermodynamics 1 st law (Cons of Energy) Deals with changes in energy Energy in chemical systems Total energy of an isolated system is constant Total energy = Potential energy + kinetic energy E p mgh
Thermodynamics 1 st law (Cons of Energy) Deals with changes in energy Energy in chemical systems Total energy of an isolated system is constant Total energy = Potential energy + kinetic energy E p mgh
Energy Changes, Reaction Rates and Equilibrium. Thermodynamics: study of energy, work and heat. Kinetic energy: energy of motion
 Energy Changes, Reaction Rates and Equilibrium Thermodynamics: study of energy, work and heat Kinetic energy: energy of motion Potential energy: energy of position, stored energy Chemical reactions involve
Energy Changes, Reaction Rates and Equilibrium Thermodynamics: study of energy, work and heat Kinetic energy: energy of motion Potential energy: energy of position, stored energy Chemical reactions involve
Rates, Temperature and Potential Energy Diagrams Worksheet
 SCH4U1 ER10 Name: Date: Rates, Temperature and Potential Energy Diagrams Worksheet Part 1: 1. Use the potential energy diagram shown to the right to answer the following: a. Label the axis. y axis is potential
SCH4U1 ER10 Name: Date: Rates, Temperature and Potential Energy Diagrams Worksheet Part 1: 1. Use the potential energy diagram shown to the right to answer the following: a. Label the axis. y axis is potential
First Law of Thermodynamics
 Energy Energy: ability to do work or produce heat. Types of energy 1) Potential energy - energy possessed by objects due to position or arrangement of particles. Forms of potential energy - electrical,
Energy Energy: ability to do work or produce heat. Types of energy 1) Potential energy - energy possessed by objects due to position or arrangement of particles. Forms of potential energy - electrical,
The Nature of Energy. Chapter Six: Kinetic vs. Potential Energy. Energy and Work. Temperature vs. Heat
 The Nature of Energy Chapter Six: THERMOCHEMISTRY Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationship between chemical reactions and energy changes
The Nature of Energy Chapter Six: THERMOCHEMISTRY Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationship between chemical reactions and energy changes
Gummy Bear Demonstration:
 Name: Unit 8: Chemical Kinetics Date: Regents Chemistry Aim: _ Do Now: a) Using your glossary, define chemical kinetics: b) Sort the phrases on the SmartBoard into the two columns below. Endothermic Rxns
Name: Unit 8: Chemical Kinetics Date: Regents Chemistry Aim: _ Do Now: a) Using your glossary, define chemical kinetics: b) Sort the phrases on the SmartBoard into the two columns below. Endothermic Rxns
Chapter 15 Energy and Chemical Change
 Chapter 15 Energy and Chemical Change Chemical reactions usually absorb or release energy. Section 1: Energy Section 2: Heat Section 3: Thermochemical Equations Section 4: Calculating Enthalpy Change Section
Chapter 15 Energy and Chemical Change Chemical reactions usually absorb or release energy. Section 1: Energy Section 2: Heat Section 3: Thermochemical Equations Section 4: Calculating Enthalpy Change Section
Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes
 Thermochemistry Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes heat flows from high to low (hot cool) endothermic reactions: absorb energy
Thermochemistry Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes heat flows from high to low (hot cool) endothermic reactions: absorb energy
Thermochemistry. Energy and Chemical Change
 Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Warm up. 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]?
![Warm up. 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]? Warm up. 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]?](/thumbs/78/77877522.jpg) Warm up 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]? 4) What is the concentration of H 2 SO 4 if 30.1 ml
Warm up 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]? 4) What is the concentration of H 2 SO 4 if 30.1 ml
UNIT 15 - Reaction Energy & Reaction Kinetics. I. Thermochemistry: study of heat in chemical reactions and phase changes
 I. Thermochemistry: study of heat in chemical reactions and phase changes II. A. Heat equation (change in temperature): Q = m. C. p T 1. Q = heat (unit is Joules) 2. m = mass (unit is grams) 3. C p = specific
I. Thermochemistry: study of heat in chemical reactions and phase changes II. A. Heat equation (change in temperature): Q = m. C. p T 1. Q = heat (unit is Joules) 2. m = mass (unit is grams) 3. C p = specific
Energy Ability to produce change or do work. First Law of Thermodynamics. Heat (q) Quantity of thermal energy
 THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
Chapter 5 - Thermochemistry
 Chapter 5 - Thermochemistry Study of energy changes that accompany chemical rx s. I) Nature of Energy Energy / Capacity to do work Mechanical Work w = F x d Heat energy - energy used to cause the temperature
Chapter 5 - Thermochemistry Study of energy changes that accompany chemical rx s. I) Nature of Energy Energy / Capacity to do work Mechanical Work w = F x d Heat energy - energy used to cause the temperature
Energy and Chemical Change
 Energy and Chemical Change Section 16.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Energy and Chemical Change Section 16.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Reaction Rates & Equilibrium. What determines how fast a reaction takes place? What determines the extent of a reaction?
 Reaction Rates & Equilibrium What determines how fast a reaction takes place? What determines the extent of a reaction? Reactants Products 1 Reaction Rates Vary TNT exploding. A car rusting. Dead plants
Reaction Rates & Equilibrium What determines how fast a reaction takes place? What determines the extent of a reaction? Reactants Products 1 Reaction Rates Vary TNT exploding. A car rusting. Dead plants
UNIT 9 IB MATERIAL KINETICS & THERMODYNAMICS
 UNIT 9 IB MATERIAL KINETICS & THERMODYNAMICS Name: ESSENTIALS: Know, Understand, and Be Able To State that combustion and neutralization are exothermic processes. Calculate the heat energy change when
UNIT 9 IB MATERIAL KINETICS & THERMODYNAMICS Name: ESSENTIALS: Know, Understand, and Be Able To State that combustion and neutralization are exothermic processes. Calculate the heat energy change when
Thermochemistry. Energy and Chemical Change
 Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Energy Ability to produce change or do work. First Law of Thermodynamics. Heat (q) Quantity of thermal energy
 THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
Name Date Class THE FLOW OF ENERGY HEAT AND WORK
 17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
Energy and Chemical Change
 Energy and Chemical Change Section 15.1 Energy Section 15.2 Heat Section 15.3 Thermochemical Equations Section 15.4 Calculating Enthalpy Change Section 15.5 Reaction Spontaneity Click a hyperlink or folder
Energy and Chemical Change Section 15.1 Energy Section 15.2 Heat Section 15.3 Thermochemical Equations Section 15.4 Calculating Enthalpy Change Section 15.5 Reaction Spontaneity Click a hyperlink or folder
Introduction to Thermochemistry. Thermochemistry Unit. Definition. Terminology. Terminology. Terminology 07/04/2016. Chemistry 30
 Thermochemistry Unit Introduction to Thermochemistry Chemistry 30 Definition Thermochemistry is the branch of chemistry concerned with the heat produced and used in chemical reactions. Most of thermochemistry
Thermochemistry Unit Introduction to Thermochemistry Chemistry 30 Definition Thermochemistry is the branch of chemistry concerned with the heat produced and used in chemical reactions. Most of thermochemistry
Section 1 - Thermochemistry
 Reaction Energy Section 1 - Thermochemistry Virtually every chemical reaction is accompanied by a change in energy. Chemical reactions usually absorb or release energy as heat. You learned in Chapter 12
Reaction Energy Section 1 - Thermochemistry Virtually every chemical reaction is accompanied by a change in energy. Chemical reactions usually absorb or release energy as heat. You learned in Chapter 12
Chapter 6. Thermochemistry
 Chapter 6. Thermochemistry 1 1. Terms to Know: thermodynamics thermochemistry energy kinetic energy potential energy heat heat vs. temperature work work of expanding gases work of expanding gases under
Chapter 6. Thermochemistry 1 1. Terms to Know: thermodynamics thermochemistry energy kinetic energy potential energy heat heat vs. temperature work work of expanding gases work of expanding gases under
The following gas laws describes an ideal gas, where
 Alief ISD Chemistry STAAR Review Reporting Category 4: Gases and Thermochemistry C.9.A Describe and calculate the relations between volume, pressure, number of moles, and temperature for an ideal gas as
Alief ISD Chemistry STAAR Review Reporting Category 4: Gases and Thermochemistry C.9.A Describe and calculate the relations between volume, pressure, number of moles, and temperature for an ideal gas as
_ + Units of Energy. Energy in Thermochemistry. Thermochemistry. Energy flow between system and surroundings. 100º C heat 50º C
 Units of Energy Like we saw with pressure, many different units are used throughout the world for energy. SI unit for energy 1kg m 1J = 2 s 2 Joule (J) calorie (cal) erg (erg) electron volts (ev) British
Units of Energy Like we saw with pressure, many different units are used throughout the world for energy. SI unit for energy 1kg m 1J = 2 s 2 Joule (J) calorie (cal) erg (erg) electron volts (ev) British
The Nature of Energy Energy is the ability to do work or produce Heat, q or Q, is ; flows due to temperature differences (always to )
 CP Chapter 17 Thermochemistry 2014-2015 Thermochemistry Thermochemistry is the study of energy that occur during chemical and physical changes (changes of state) The Nature of Energy Energy is the ability
CP Chapter 17 Thermochemistry 2014-2015 Thermochemistry Thermochemistry is the study of energy that occur during chemical and physical changes (changes of state) The Nature of Energy Energy is the ability
I. The Nature of Energy A. Energy
 I. The Nature of Energy A. Energy is the ability to do work or produce heat. It exists in 2 forms: 1. Potential energy is energy due to the composition or position of an object. 2. Kinetic energy is energy
I. The Nature of Energy A. Energy is the ability to do work or produce heat. It exists in 2 forms: 1. Potential energy is energy due to the composition or position of an object. 2. Kinetic energy is energy
Calculate the mass of L of oxygen gas at 25.0 C and 1.18 atm pressure.
 148 Calculate the mass of 22650 L of oxygen gas at 25.0 C and 1.18 atm pressure. 1 - Convert the volume of oxygen gas to moles using IDEAL GAS EQUATION 2 - Convert moles oxygen gas to mass using formula
148 Calculate the mass of 22650 L of oxygen gas at 25.0 C and 1.18 atm pressure. 1 - Convert the volume of oxygen gas to moles using IDEAL GAS EQUATION 2 - Convert moles oxygen gas to mass using formula
Thermochemistry. Energy (and Thermochemistry) World of Chemistry Chapter 10. Energy. Energy
 Thermochemistry Thermodynamics is the science of the relationship between heat and other forms of energy. (and Thermochemistry) World of Chemistry Chapter 10 is defined as the ability to do work or produce
Thermochemistry Thermodynamics is the science of the relationship between heat and other forms of energy. (and Thermochemistry) World of Chemistry Chapter 10 is defined as the ability to do work or produce
Reaction Rates & Equilibrium. What determines how fast a reaction takes place? What determines the extent of a reaction?
 Reaction Rates & Equilibrium What determines how fast a reaction takes place? What determines the extent of a reaction? Reactants Products 1 Reaction Rates Vary TNT exploding. A car rusting. Dead plants
Reaction Rates & Equilibrium What determines how fast a reaction takes place? What determines the extent of a reaction? Reactants Products 1 Reaction Rates Vary TNT exploding. A car rusting. Dead plants
Selected Questions on Chapter 5 Thermochemistry
 Selected Questions on Chapter 5 Thermochemistry Circle the correct answer: 1) At what velocity (m/s) must a 20.0 g object be moving in order to possess a kinetic energy of 1.00 J? A) 1.00 B) 100 10 2 C)
Selected Questions on Chapter 5 Thermochemistry Circle the correct answer: 1) At what velocity (m/s) must a 20.0 g object be moving in order to possess a kinetic energy of 1.00 J? A) 1.00 B) 100 10 2 C)
Thermochemistry. Section The flow of energy
 Thermochemistry Section 17.1 - The flow of energy What is Energy? Energy is the capacity for doing work or supplying heat Energy does not have mass or volume, and it can only be detected because of its
Thermochemistry Section 17.1 - The flow of energy What is Energy? Energy is the capacity for doing work or supplying heat Energy does not have mass or volume, and it can only be detected because of its
Chapter 8 Thermochemistry: Chemical Energy. Chemical Thermodynamics
 Chapter 8 Thermochemistry: Chemical Energy Chapter 8 1 Chemical Thermodynamics Chemical Thermodynamics is the study of the energetics of a chemical reaction. Thermodynamics deals with the absorption or
Chapter 8 Thermochemistry: Chemical Energy Chapter 8 1 Chemical Thermodynamics Chemical Thermodynamics is the study of the energetics of a chemical reaction. Thermodynamics deals with the absorption or
Spontaneity, Entropy, and Free Energy
 Spontaneity, Entropy, and Free Energy A ball rolls spontaneously down a hill but not up. Spontaneous Processes A reaction that will occur without outside intervention; product favored Most reactants are
Spontaneity, Entropy, and Free Energy A ball rolls spontaneously down a hill but not up. Spontaneous Processes A reaction that will occur without outside intervention; product favored Most reactants are
Collision Theory. Unit 12: Chapter 18. Reaction Rates. Activation Energy. Reversible Reactions. Reversible Reactions. Reaction Rates and Equilibrium
 Collision Theory For reactions to occur collisions between particles must have Unit 12: Chapter 18 Reaction Rates and Equilibrium the proper orientation enough kinetic energy See Both In Action 1 2 Activation
Collision Theory For reactions to occur collisions between particles must have Unit 12: Chapter 18 Reaction Rates and Equilibrium the proper orientation enough kinetic energy See Both In Action 1 2 Activation
Chapter 11 Thermochemistry Heat and Chemical Change
 Chemistry/ PEP Name: Date: Chapter 11 Thermochemistry Heat and Chemical Change Chapter 11:1 35, 57, 60, 61, 71 Section 11.1 The Flow of Energy - Heat 1. When 435 of heat is added to 3.4 g of olive oil
Chemistry/ PEP Name: Date: Chapter 11 Thermochemistry Heat and Chemical Change Chapter 11:1 35, 57, 60, 61, 71 Section 11.1 The Flow of Energy - Heat 1. When 435 of heat is added to 3.4 g of olive oil
Name: Class: Date: ID: A
 Name: Class: _ Date: _ ID: A Chpter 17 review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of these phase changes is an endothermic process? a.
Name: Class: _ Date: _ ID: A Chpter 17 review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of these phase changes is an endothermic process? a.
CP Chapter 17 Thermochemistry
 CP Chapter 17 Thermochemistry Thermochemistry Thermochemistry is the study of energy that occur during chemical reactions and phase changes (changes of state) The Nature of Energy Energy is the ability
CP Chapter 17 Thermochemistry Thermochemistry Thermochemistry is the study of energy that occur during chemical reactions and phase changes (changes of state) The Nature of Energy Energy is the ability
Thermochemistry Chapter 4
 Thermochemistry Chapter 4 Thermochemistry is the study of energy changes that occur during chemical reactions Focus is on heat and matter transfer between the system and the surroundings Energy The ability
Thermochemistry Chapter 4 Thermochemistry is the study of energy changes that occur during chemical reactions Focus is on heat and matter transfer between the system and the surroundings Energy The ability
Practice Test: Energy and Rates of Reactions
 Practice Test: Energy and Rates of Reactions NAME: /65 Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. (20 marks) 1. What is the symbol for
Practice Test: Energy and Rates of Reactions NAME: /65 Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. (20 marks) 1. What is the symbol for
33. a. Heat is absorbed from the water (it gets colder) as KBr dissolves, so this is an endothermic process.
 31. This is an endothermic reaction so heat must be absorbed in order to convert reactants into products. The high temperature environment of internal combustion engines provides the heat. 33. a. Heat
31. This is an endothermic reaction so heat must be absorbed in order to convert reactants into products. The high temperature environment of internal combustion engines provides the heat. 33. a. Heat
HC- Kinetics and Thermodynamics Test Review Stations
 HC- Kinetics and Thermodynamics Test Review Stations Station 1- Collision Theory and Factors Affecting Reaction Rate 1. Explain the collision theory of reactions. 2. Fill out the following table to review
HC- Kinetics and Thermodynamics Test Review Stations Station 1- Collision Theory and Factors Affecting Reaction Rate 1. Explain the collision theory of reactions. 2. Fill out the following table to review
CHAPTER 16 REVIEW. Reaction Energy. SHORT ANSWER Answer the following questions in the space provided.
 CHAPTER 16 REVIEW Reaction Energy SECTION 1 SHORT ANSWER Answer the following questions in the space provided. 1. For elements in their standard state, the value of H 0 f is 0. 2. The formation and decomposition
CHAPTER 16 REVIEW Reaction Energy SECTION 1 SHORT ANSWER Answer the following questions in the space provided. 1. For elements in their standard state, the value of H 0 f is 0. 2. The formation and decomposition
Note: 1 calorie = 4.2 Joules
 Enthalpy Changes All substances contain chemical energy, called enthalpy. Like any kind of energy it is measured in Joules (previously energy was measured in Calories). When reactions happen, energy is
Enthalpy Changes All substances contain chemical energy, called enthalpy. Like any kind of energy it is measured in Joules (previously energy was measured in Calories). When reactions happen, energy is
Reaction Energy. Thermochemistry
 Reaction Energy Thermochemistry Thermochemistry The study of the transfers of energy as heat that accompany chemical reactions & physical changes Thermochemistry -In studying heat changes, think of defining
Reaction Energy Thermochemistry Thermochemistry The study of the transfers of energy as heat that accompany chemical reactions & physical changes Thermochemistry -In studying heat changes, think of defining
11B, 11E Temperature and heat are related but not identical.
 Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
CHAPTER 17 Thermochemistry
 CHAPTER 17 Thermochemistry Thermochemistry The study of the heat changes that occur during chemical reactions and physical changes of state. Chemical Change: new substances created during chemical reaction
CHAPTER 17 Thermochemistry Thermochemistry The study of the heat changes that occur during chemical reactions and physical changes of state. Chemical Change: new substances created during chemical reaction
Unit 13 Kinetics & Equilibrium Page 1 of 14 Chemistry Kinetics, Entropy, Equilibrium, LeChatelier s Principle, K, Unit 13 Quiz: Unit 13 Test:
 Unit 13 Kinetics & Equilibrium Page 1 of 14 Chemistry Kinetics, Entropy, Equilibrium, LeChatelier s Principle, K, Unit 13 Quiz: Unit 13 Test: Final Project: VOCABULARY: 1 Chemical equilibrium 2 equilibrium
Unit 13 Kinetics & Equilibrium Page 1 of 14 Chemistry Kinetics, Entropy, Equilibrium, LeChatelier s Principle, K, Unit 13 Quiz: Unit 13 Test: Final Project: VOCABULARY: 1 Chemical equilibrium 2 equilibrium
Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes
 Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes Students will be expected to: Compare the molar enthalpies of several combustion reactions involving organic compounds.
Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes Students will be expected to: Compare the molar enthalpies of several combustion reactions involving organic compounds.
Thermochemistry Lecture
 Thermochemistry Lecture Jennifer Fang 1. Enthalpy 2. Entropy 3. Gibbs Free Energy 4. q 5. Hess Law 6. Laws of Thermodynamics ENTHALPY total energy in all its forms; made up of the kinetic energy of the
Thermochemistry Lecture Jennifer Fang 1. Enthalpy 2. Entropy 3. Gibbs Free Energy 4. q 5. Hess Law 6. Laws of Thermodynamics ENTHALPY total energy in all its forms; made up of the kinetic energy of the
This reaction is ENDOTHERMIC. Energy is being transferred from the room/flask/etc. (the SURROUNDINGS) to the reaction itself (the SYSTEM).
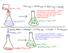 151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
Ch. 17 Thermochemistry
 Ch. 17 Thermochemistry 17.1 The Flow of Energy Energy Transformations Thermochemistry: study of energy changes in chemical reactions and changes in state Chemical potential energy: energy stored in bonds
Ch. 17 Thermochemistry 17.1 The Flow of Energy Energy Transformations Thermochemistry: study of energy changes in chemical reactions and changes in state Chemical potential energy: energy stored in bonds
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Learning Outcomes: Interconvert energy units Distinguish between the system and the surroundings in thermodynamics Calculate internal energy from heat and work and state sign
Chapter 5 Thermochemistry Learning Outcomes: Interconvert energy units Distinguish between the system and the surroundings in thermodynamics Calculate internal energy from heat and work and state sign
Chemistry 101 Chapter 10 Energy
 Chemistry 101 Chapter 10 Energy Energy: the ability to do work or produce heat. Kinetic energy (KE): is the energy of motion. Any object that is moving has kinetic energy. Several forms of kinetic energy
Chemistry 101 Chapter 10 Energy Energy: the ability to do work or produce heat. Kinetic energy (KE): is the energy of motion. Any object that is moving has kinetic energy. Several forms of kinetic energy
Name: Thermochemistry. Practice Test C. General Chemistry Honors Chemistry
 Name: Thermochemistry C Practice Test C General Chemistry Honors Chemistry 1 Objective 1: Use the relationship between mass, specific heat, and temperature change to calculate the heat flow during a chemical
Name: Thermochemistry C Practice Test C General Chemistry Honors Chemistry 1 Objective 1: Use the relationship between mass, specific heat, and temperature change to calculate the heat flow during a chemical
Chemistry. Friday, March 30 th Monday, April 9 th, 2018
 Chemistry Friday, March 30 th Monday, April 9 th, 2018 Do-Now: BrainPOP: Heat 1. Write down today s FLT 2. Distinguish between exothermic and endothermic processes. 3. What is the specific heat of water?
Chemistry Friday, March 30 th Monday, April 9 th, 2018 Do-Now: BrainPOP: Heat 1. Write down today s FLT 2. Distinguish between exothermic and endothermic processes. 3. What is the specific heat of water?
OCR Chemistry A H432
 All the energy changes we have considered so far have been in terms of enthalpy, and we have been able to predict whether a reaction is likely to occur on the basis of the enthalpy change associated with
All the energy changes we have considered so far have been in terms of enthalpy, and we have been able to predict whether a reaction is likely to occur on the basis of the enthalpy change associated with
CHEM 1105 S10 March 11 & 14, 2014
 CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
AP CHEMISTRY NOTES 4-1 THERMOCHEMISTRY: ENTHALPY AND ENTROPY
 AP CHEMISTRY NOTES 4-1 THERMOCHEMISTRY: ENTHALPY AND ENTROPY Reaction Rate how fast a chemical reaction occurs Collision Theory In order for a chemical reaction to occur, the following conditions must
AP CHEMISTRY NOTES 4-1 THERMOCHEMISTRY: ENTHALPY AND ENTROPY Reaction Rate how fast a chemical reaction occurs Collision Theory In order for a chemical reaction to occur, the following conditions must
Chapter 17: Energy and Kinetics
 Pages 510-547 S K K Chapter 17: Energy and Kinetics Thermochemistry: Causes of change in systems Kinetics: Rate of reaction progress (speed) Heat, Energy, and Temperature changes S J J Heat vs Temperature
Pages 510-547 S K K Chapter 17: Energy and Kinetics Thermochemistry: Causes of change in systems Kinetics: Rate of reaction progress (speed) Heat, Energy, and Temperature changes S J J Heat vs Temperature
CHEMISTRY 12 UNIT II EQUILIBRIUM D Learning Goals
 CHEMISTRY 12 UNIT II EQUILIBRIUM D Learning Goals 1. Chemical equilibrium is said to by dynamic because a. The reaction proceeds quickly b. The mass of the reactants is decreasing c. The macroscopic properties
CHEMISTRY 12 UNIT II EQUILIBRIUM D Learning Goals 1. Chemical equilibrium is said to by dynamic because a. The reaction proceeds quickly b. The mass of the reactants is decreasing c. The macroscopic properties
UNIT #10: Reaction Rates Heat/Energy in Chemical Reactions Le Chatlier s Principle Potential Energy Diagrams
 UNIT #10: Reaction Rates Heat/Energy in Chemical Reactions Le Chatlier s Principle Potential Energy Diagrams NAME: 1. REACTION RATES a) The speed of a chemical reaction determined by the change in concentration
UNIT #10: Reaction Rates Heat/Energy in Chemical Reactions Le Chatlier s Principle Potential Energy Diagrams NAME: 1. REACTION RATES a) The speed of a chemical reaction determined by the change in concentration
Unit 6 Kinetics and Equilibrium.docx
 6-1 Unit 6 Kinetics and Equilibrium At the end of this unit, you ll be familiar with the following: Kinetics: Reaction Rate Collision Theory Reaction Mechanism Factors Affecting Rate of Reaction: o Nature
6-1 Unit 6 Kinetics and Equilibrium At the end of this unit, you ll be familiar with the following: Kinetics: Reaction Rate Collision Theory Reaction Mechanism Factors Affecting Rate of Reaction: o Nature
Advanced Chemistry Practice Problems
 Thermodynamics: Review of Thermochemistry 1. Question: What is the sign of DH for an exothermic reaction? An endothermic reaction? Answer: ΔH is negative for an exothermic reaction and positive for an
Thermodynamics: Review of Thermochemistry 1. Question: What is the sign of DH for an exothermic reaction? An endothermic reaction? Answer: ΔH is negative for an exothermic reaction and positive for an
Quantities in Chemical Reactions
 Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Bond Enthalpy and Activation Energy
 Bond Enthalpy and Activation Energy Energy of a Chemical Reaction ΔH = ΔH (bonds broken) - ΔH (bonds formed) Add up all the energies of the broken bonds Add up all the energies of the bonds that are reformed
Bond Enthalpy and Activation Energy Energy of a Chemical Reaction ΔH = ΔH (bonds broken) - ΔH (bonds formed) Add up all the energies of the broken bonds Add up all the energies of the bonds that are reformed
Thermochemistry: Energy Flow and Chemical Reactions
 Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
CRHS Academic Chemistry Unit 15 Thermochemistry HOMEWORK. Due Date Assignment On-Time (100) Late (70)
 Name KEY Period CRHS Academic Chemistry Unit 15 Thermochemistry HOMEWORK Due Date Assignment On-Time (100) Late (70) 15.1 15.2 15.3 15.4 Warm Ups Extra Credit Notes, Homework, Exam Reviews and Their KEYS
Name KEY Period CRHS Academic Chemistry Unit 15 Thermochemistry HOMEWORK Due Date Assignment On-Time (100) Late (70) 15.1 15.2 15.3 15.4 Warm Ups Extra Credit Notes, Homework, Exam Reviews and Their KEYS
Chapter 7 Chemical Reactions: Energy, Rates, and Equilibrium
 Chapter 7 Chemical Reactions: Energy, Rates, and Equilibrium Introduction This chapter considers three factors: a) Thermodynamics (Energies of Reactions) a reaction will occur b) Kinetics (Rates of Reactions)
Chapter 7 Chemical Reactions: Energy, Rates, and Equilibrium Introduction This chapter considers three factors: a) Thermodynamics (Energies of Reactions) a reaction will occur b) Kinetics (Rates of Reactions)
Chapter 5: Thermochemistry. Molecular Kinetic Energy -Translational energy E k, translational = 1/2mv 2 -Rotational energy 5.
 Chapter 5: Thermochemistry 1. Thermodynamics 2. Energy 3. Specific Heat 4. Enthalpy 5. Enthalpies of Reactions 6. Hess s Law 7. State Functions 8. Standard Enthalpies of Formation 9. Determining Enthalpies
Chapter 5: Thermochemistry 1. Thermodynamics 2. Energy 3. Specific Heat 4. Enthalpy 5. Enthalpies of Reactions 6. Hess s Law 7. State Functions 8. Standard Enthalpies of Formation 9. Determining Enthalpies
Chemistry 30 Review Sessions THERMOCHEMISTRY
 Chemistry 30 Review Sessions THERMOCHEMISTRY Curriculum bullets Use Q = mcδt to analyze heat transfer. Use calorimetry data to determine the enthalpy changes in chemical reactions. Calorimeters rely on
Chemistry 30 Review Sessions THERMOCHEMISTRY Curriculum bullets Use Q = mcδt to analyze heat transfer. Use calorimetry data to determine the enthalpy changes in chemical reactions. Calorimeters rely on
Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions
 Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions Terms, definitions, topics Joule, calorie (Re-read p 57-58) Thermochemistry Exothermic reaction Endothermic reaction
Accelerated Chemistry Study Guide Chapter 12, sections 1 and 2: Heat in Chemical Reactions Terms, definitions, topics Joule, calorie (Re-read p 57-58) Thermochemistry Exothermic reaction Endothermic reaction
Chapter 7 Chemical Reactions
 Chapter 7 Chemical Reactions Chemical Equation --> is a representation of a chemical reaction in which the reactants and products are expressed as formulas Reactants --> substances that undergo change
Chapter 7 Chemical Reactions Chemical Equation --> is a representation of a chemical reaction in which the reactants and products are expressed as formulas Reactants --> substances that undergo change
Name AP CHEM / / Collected AP Exam Essay Answers for Chapter 16
 Name AP CHEM / / Collected AP Exam Essay Answers for Chapter 16 1980 - #7 (a) State the physical significance of entropy. Entropy (S) is a measure of randomness or disorder in a system. (b) From each of
Name AP CHEM / / Collected AP Exam Essay Answers for Chapter 16 1980 - #7 (a) State the physical significance of entropy. Entropy (S) is a measure of randomness or disorder in a system. (b) From each of
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Chapter 5 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Thermochemistry Notes
 Name: Thermochemistry Notes I. Thermochemistry deals with the changes in energy that accompany a chemical reaction. Energy is measured in a quantity called enthalpy, represented as H. The change in energy
Name: Thermochemistry Notes I. Thermochemistry deals with the changes in energy that accompany a chemical reaction. Energy is measured in a quantity called enthalpy, represented as H. The change in energy
Energy in Chemical Reaction Reaction Rates Chemical Equilibrium. Chapter Outline. Energy 6/29/2013
 Energy in Chemical Reaction Reaction Rates Chemical Equilibrium Chapter Outline Energy change in chemical reactions Bond dissociation energy Reaction rate Chemical equilibrium, Le Châtelier s principle
Energy in Chemical Reaction Reaction Rates Chemical Equilibrium Chapter Outline Energy change in chemical reactions Bond dissociation energy Reaction rate Chemical equilibrium, Le Châtelier s principle
Chapter 6 Problems: 9, 19, 24, 25, 26, 27, 31-33, 37, 39, 43, 45, 47, 48, 53, 55, 57, 59, 65, 67, 73, 78-82, 85, 89, 93
 Chapter 6 Problems: 9, 19, 24, 25, 26, 27, 31-33, 37, 39, 43, 45, 47, 48, 53, 55, 57, 59, 65, 67, 73, 78-82, 85, 89, 93 Chapter 6 Thermochemistry The study of chemical reactions and the energy changes
Chapter 6 Problems: 9, 19, 24, 25, 26, 27, 31-33, 37, 39, 43, 45, 47, 48, 53, 55, 57, 59, 65, 67, 73, 78-82, 85, 89, 93 Chapter 6 Thermochemistry The study of chemical reactions and the energy changes
AP CHEMISTRY SCORING GUIDELINES
 Mean 5.64 out of 9 pts AP CHEMISTRY Question 1 CO(g) + 1 2 O 2 (g) CO 2 (g) 1. The combustion of carbon monoxide is represented by the equation above. (a) Determine the value of the standard enthalpy change,
Mean 5.64 out of 9 pts AP CHEMISTRY Question 1 CO(g) + 1 2 O 2 (g) CO 2 (g) 1. The combustion of carbon monoxide is represented by the equation above. (a) Determine the value of the standard enthalpy change,
Unit 15 Energy and Thermochemistry Notes
 Name KEY Period CRHS Academic Chemistry Unit 15 Energy and Thermochemistry Notes Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Name KEY Period CRHS Academic Chemistry Unit 15 Energy and Thermochemistry Notes Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Energy and Chemical Change
 Energy and Chemical Change Section 15.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Energy and Chemical Change Section 15.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Chapter 3. Thermochemistry: Energy Flow and Chemical Change. 5.1 Forms of Energy and Their Interconversion
 Chapter 3 Thermochemistry: Energy Flow and Chemical Change 5.1 Forms of Energy and Their Interconversion 5.2 Enthalpy: Chemical Change at Constant Pressure 5.3 Calorimetry: Measuring the Heat of a Chemical
Chapter 3 Thermochemistry: Energy Flow and Chemical Change 5.1 Forms of Energy and Their Interconversion 5.2 Enthalpy: Chemical Change at Constant Pressure 5.3 Calorimetry: Measuring the Heat of a Chemical
1.4 Enthalpy. What is chemical energy?
 1.4 Enthalpy What is chemical energy? Chemical energy is a form of potential energy which is stored in chemical bonds. Chemical bonds are the attractive forces that bind atoms together. As a reaction takes
1.4 Enthalpy What is chemical energy? Chemical energy is a form of potential energy which is stored in chemical bonds. Chemical bonds are the attractive forces that bind atoms together. As a reaction takes
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Energy -Very much a chemistry topic Every chemical change has an accompanying change of. Combustion of fossil fuels The discharging a battery Metabolism of foods If we are to
Chapter 5 Thermochemistry Energy -Very much a chemistry topic Every chemical change has an accompanying change of. Combustion of fossil fuels The discharging a battery Metabolism of foods If we are to
Thermochemistry. Chapter 6. Dec 19 8:52 AM. Thermochemistry. Energy: The capacity to do work or to produce heat
 Chapter 6 Dec 19 8:52 AM Intro vocabulary Energy: The capacity to do work or to produce heat Potential Energy: Energy due to position or composition (distance and strength of bonds) Kinetic Energy: Energy
Chapter 6 Dec 19 8:52 AM Intro vocabulary Energy: The capacity to do work or to produce heat Potential Energy: Energy due to position or composition (distance and strength of bonds) Kinetic Energy: Energy
Thermochemistry Ch. 8
 Definitions I. Energy (E): capacity to do work. II. Heat (q): transfer of energy from a body at a high temp. to a body at a low temp. III. Reaction perspectives: A. System: the focus. B. Surroundings:
Definitions I. Energy (E): capacity to do work. II. Heat (q): transfer of energy from a body at a high temp. to a body at a low temp. III. Reaction perspectives: A. System: the focus. B. Surroundings:
Name Date Class THERMOCHEMISTRY
 Name Date Class 17 THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY HEAT AND WORK (pages 505 510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Name Date Class 17 THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY HEAT AND WORK (pages 505 510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Slide 1 / Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy. a only b only c only a and c b and c
 Slide 1 / 84 1 Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy A B C D E a only b only c only a and c b and c Slide 2 / 84 2 The internal energy of a system
Slide 1 / 84 1 Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy A B C D E a only b only c only a and c b and c Slide 2 / 84 2 The internal energy of a system
Thermochemistry. Mr.V
 Thermochemistry Mr.V Introduction to Energy changes System Surroundings Exothermic Endothermic Internal energy Enthalpy Definitions System A specified part of the universe which is under investigation
Thermochemistry Mr.V Introduction to Energy changes System Surroundings Exothermic Endothermic Internal energy Enthalpy Definitions System A specified part of the universe which is under investigation
Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat.
 CHEM134- F18 Dr. Al- Qaisi Chapter 06: Thermodynamics Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat. Energy is anything that has the capacity
CHEM134- F18 Dr. Al- Qaisi Chapter 06: Thermodynamics Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat. Energy is anything that has the capacity
Quantities in Chemical Reactions
 Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Chapter 8. Thermochemistry 강의개요. 8.1 Principles of Heat Flow. 2) Magnitude of Heat Flow. 1) State Properties. Basic concepts : study of heat flow
 강의개요 Basic concepts : study of heat flow Chapter 8 Thermochemistry Calorimetry : experimental measurement of the magnitude and direction of heat flow Thermochemical Equations Copyright 2005 연세대학교이학계열일반화학및실험
강의개요 Basic concepts : study of heat flow Chapter 8 Thermochemistry Calorimetry : experimental measurement of the magnitude and direction of heat flow Thermochemical Equations Copyright 2005 연세대학교이학계열일반화학및실험
Energy & Chemistry. Internal Energy (E) Energy and Chemistry. Potential Energy. Kinetic Energy. Energy and Chemical Reactions: Thermochemistry or
 Page III-5-1 / Chapter Five Lecture Notes Energy & Chemistry Energy and Chemical Reactions: Thermochemistry or Thermodynamics Chapter Five Burning peanuts supplies sufficient energy to boil a cup of water
Page III-5-1 / Chapter Five Lecture Notes Energy & Chemistry Energy and Chemical Reactions: Thermochemistry or Thermodynamics Chapter Five Burning peanuts supplies sufficient energy to boil a cup of water
Section 9: Thermodynamics and Energy
 Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Date: SCH 4U Name: ENTHALPY CHANGES
 Date: SCH 4U Name: ENTHALPY CHANGES Enthalpy (H) = heat content of system (heat, latent heat) Enthalpy = total energy of system + pressure volume H = E + PV H = E + (PV) = final conditions initial conditions
Date: SCH 4U Name: ENTHALPY CHANGES Enthalpy (H) = heat content of system (heat, latent heat) Enthalpy = total energy of system + pressure volume H = E + PV H = E + (PV) = final conditions initial conditions
How many ml of 0.250M potassium permangenate are needed to react with 3.36 g of iron(ii) sulfate?
 115 How many ml of 0.250M potassium permangenate are needed to react with 3.36 g of iron(ii) sulfate? 1) Convert 3.36 grams iron(ii) sulfate to moles. Use FORMULA WEIGHT. 2) Convert moles iron(ii) sulfate
115 How many ml of 0.250M potassium permangenate are needed to react with 3.36 g of iron(ii) sulfate? 1) Convert 3.36 grams iron(ii) sulfate to moles. Use FORMULA WEIGHT. 2) Convert moles iron(ii) sulfate
CHEMISTRY. Chapter 5 Thermochemistry
 CHEMISTRY The Central Science 8 th Edition Chapter 5 Thermochemistry Dr. Kozet YAPSAKLI The Nature of Energy Kinetic and Potential Energy Potential energy can be converted into kinetic energy. E p = mgh
CHEMISTRY The Central Science 8 th Edition Chapter 5 Thermochemistry Dr. Kozet YAPSAKLI The Nature of Energy Kinetic and Potential Energy Potential energy can be converted into kinetic energy. E p = mgh
