CP Chapter 17 Thermochemistry
|
|
|
- Shon Wilkerson
- 6 years ago
- Views:
Transcription
1 CP Chapter 17 Thermochemistry Thermochemistry Thermochemistry is the study of energy that occur during chemical reactions and phase changes (changes of state) The Nature of Energy Energy is the ability to do work or produce Heat is energy transferred from a sample of temperature to temperature. Represented by the letter Q or q. Kinetic vs. Potential Energy Two main types of energy kinetic and potential o Kinetic energy of o Potential energy due to position or energy in chemical bonds Chemical potential energy - the energy stored in a substance because of its composition Example: gasoline Law of Conservation of Energy Law of conservation of energy states that in any chemical reaction or physical process, energy can be from one form to another, but it is neither created nor destroyed. Temperature vs. Heat Temperature is a measure of the average kinetic energy of the molecular in a sample. The Kelvin temperature and kinetic energy are proportional. Temperature is a of energy. Heat is the total energy of molecular motion, dependent upon amount, size, and type of particles. Heat is. System and Surroundings System The part of the universe on which contains the or process you wish to study. Surroundings Everything in the universe than the system. When heat is transferred it can flow in or out of the system Endothermic vs. Exothermic An Exothermic process is one that (evolved) heat to its surroundings (feels warm) o Energy products < Energy reactants An Endothermic process is one that heat from the surroundings (feels cold) o Energy products > Energy reactants Exothermic Process Endothermic Process 1
2 Measuring Heat calorie - the amount of heat required to raise the temperature of one gram of pure by one degree Celsius Calorie nutritional calorie; o 1 Calorie = 1000 calories = 1 kilocalories (kcal) Joule SI unit of heat o 1 calorie = J Specific Heat = C p Specific heat of a substance is the amount of heat required to raise the temperature of one gram of that by one degree Celsius. Unit for specific heat is J/g o C Specific Heat cont... Each substance has a specific heat Water = J/g o C Gold = J/g o C Copper = J/g o C The lower the specific heat the lower the amount energy is required to raise its temperature. Calculating Heat Released and Absorbed Q = m( T)C p o Q = Heat o m is mass o T is temperature change Tfinal -Tinitial o C p is specific heat at a constant pressure The unit for heat (Q) is Joules (J) or kilojoules (kj) If reaction is heat (Q) is positive If reaction is exothermic heat (Q) is Converting Energy Units Calorie/calorie/ kilocalorie calorie/joule A cereal has 155 nutritional Calories per serving. How many calories, kilocalories and Joules is this? A person on a diet consumed 1350 Calories in one day. How many calories, kilocalories and Joules is this? 2
3 Calculating Heat If the temperature of 56.6 g of ethanol increases from 45.0 o C to 80.0 o C, how much heat has been absorbed by the ethanol? Specific heat of ethanol = 2.44 J/g o C A 4.00 g sample of a substance was heated from 274 K to 314 K and absorbed 32 J of heat, what is the specific heat of the substance? If 98,000 J of energy are added to 6200 g of water at o C, what will the change in temperature of the water be? Specific heat of water = J/g o C Calorimetry and Enthalpy Calorimetry Calorimetry is the of the heat flow into or out of a system for chemical or physical processes o Based on the law of conservation of Calorimetry Example Heat by the system is equal to the heat absorbed by the surroundings Heat absorbed by the system is equal to the heat released by the surroundings An ice cube is added to a warm cup of water. o The amount of heat used to melt the ice cube is the same amount of heat lost by the warm water. Measuring Heat Heat changes that occur during chemical and physical processes can be measured by a. A calorimeter is an device for measuring the amount of heat absorbed or released during chemical or physical processes Enthalpy and Enthalpy Changes Enthalpy (H) is the content of a system at constant Thermochemistry uses the in enthalpy( H) to study heat changes At constant pressure, Q = H 3
4 Endothermic and Exothermic Processes All chemical and physical changes release or absorb heat Exothermic process heat is released and goes from the system to the surroundings o Feels - E products < E reactants Endothermic process heat is absorbed and goes from the surrounding to the system o Feels - E products > E reactants Heat of Reaction Heat of reaction ( H rxn) The change of enthalpy in a reaction Exothermic reaction H rxn is negative o Products have a enthalpy than the reactants (Energy released) H products < H reactants Endothermic reaction H rxn is positive o Products have a enthalpy than the reactants (Energy absorbed) H products > H reactants Thermochemical Equation A thermochemical equation is a balanced chemical equation that includes the physical states of all reactants and products (1 atm, 25 o C) and the energy, expressed as a change in enthalpy, H NH 4NO + - 3(s) NH 4 (aq) + NO 3 (aq) H = 27 kj Endothermic CaO (s) + H 2O (l) Ca(OH) 2(s) H = kj Exothermic Thermochemical Equations for Endothermic Reactions Endothermic : H is positive; heat is on the side of the equation (Heat absorbed) NH 4NO + - 3(s) NH 4 (aq) + NO 3 (aq) H = 27 kj NH 4NO 3(s) + Heat + - NH 4 (aq) + NO 3 (aq) NH 4NO + - 3(s) + 27kJ NH 4 (aq) + NO 3 (aq) Thermochemical Equations for Exothermic Reactions Exothermic : H is negative; heat is on the side of the equation (Heat released) CaO (s) + H 2O (l) Ca(OH) 2(s) H = kj CaO (s) + H 2O (l) Ca(OH) 2(s) + Heat CaO (s) + H 2O (l) Ca(OH) 2(s) kj Heat of Combustion Heat of combustion is the enthalpy change for the complete burning of one mole of the substance CH 4(g) + 2O 2(g) CO 2(g) + 2H 2O(l) H = -891kJ CH 4(g) + 2O 2(g) CO 2(g) + 2H 2O(l) + Heat CH 4(g) + 2O 2(g) CO 2(g) + 2H 2O(l) + 891kJ A piece of metal with a mass of 4.68 g absorbs 2556 J of heat when its temperature increases by 182 o C. What is the specific heat of metal? How much heat is released by the combustion of g of octane, C 8H 18? Octane ΔH comb = kj/mol 4
5 Changes of State (Phase Changes) Phase Change A phase change is going from one of matter to another (Physical change) o Gas to liquid o Liquid to solid Changes of state either or (evolve/liberate) energy Phase Changes cont.. Endothermic phase changes (Absorb energy) o Melting (solid to liquid) o Evaporation/ (liquid to gas) Exothermic phase changes (Release energy) o (gas to liquid) o Freezing (liquid to solid) Temperature and Phase Change During a phase change, as energy is added or removed, there is temperature change. Latent Heat Since there is no change, we cannot use Q = m( T)C p to calculate enthalpy change. Instead we use latent (hidden) heat, which is the quantity of heat released or absorbed during a phase change. Units are usually Joule/gram or kilojoule/mole Changes of State Latent Heat of Vaporization is the heat required to one mole of a liquid o Going from liquid to gas o For water, H vap = 40.7 kj/mol Latent Heat of Condensation is the heat required to one mole of a gas o Going from gas to liquid o For water, H cond = kj/mol Thermochemical Equations for Vaporization and Condensation The reverse process of vaporization is condensation o The process has an opposite sign o H vap = - H cond o H 2O (l) H 2O (g) H vap = 40.7 kj/mol o H 2O (g) H 2O (l) H cond = kj/mol Changes of State Latent Heat of Fusion is the heat required to one mole of a solid substance. o Going from solid to liquid o For water, H fus = 6.01 kj/mol (Endothermic) Latent Heat of Solidification is the heat required to one mole of a liquid substance. o Going from liquid to solid o For water, H solid = kj/mol (Exothermic) Thermochemical Equations for Fusion and Solidification The reverse process of fusion is solidification. o The reverse process has an sign o Hfus = - Hsolid o H 2O (s) H 2O (l) H fus = 6.01 kj/mol o H 2O (l) H 2O (s) H solid = kj/mol 5
6 Heating Curve Explanation of Heating Curve When there is a temperature change use Q=m( T)C p to calculate the amount of energy used. When the heating curve is flat (no temperature change), there is a phase change. Use the latent heat to calculate the amount of energy used. Calculate the heat required to melt 45.6 g of water at its melting point. Latent heat of fusion ( Hfus) = 6.01 kj/mol Calculate the heat evolved to condense 75.4 g of water at its boiling point Latent heat of vaporization ( Hvap) = 40.7 kj/mol What mass of ammonia (NH 3) must be vaporizes that absorbs 345 kj of heat? Latent heat of vaporization ( Hvap) = 23.3 kj/mol 6
7 Hess s Law - Calculating Enthalpy Change (Heat of Reaction) Hess s Law states that if you can two or more equations to produce a equation for a reaction, then the sum of the enthalpy changes for the individual reactions is the change for the reaction. General principles for combining thermochemical equations. 1. If a reaction is reversed, the sign of H is reversed 2. If the coefficients are multiplied, then the H is multiplied by the same factor. Calculate the enthalpy change for: 2S (s) + 3O 2(g) 2SO 3(g) H =? S (s) + O 2(g) SO 2(g) H = -297 kj 2SO 3(g) 2SO 2(g) + O 2(g) H = 198 kj 2NO (g) + O 2(g) 2NO 2(g) H =? N 2(g) + O 2(g) - 2NO (g) H = kj N 2(g) + 2O 2(g) 2NO 2(g) H = 66.4 kj 2C (s) + O 2(g) 2CO (g) H =? C (s) + O 2(g) CO 2(g) H = kj 2CO + O 2 2CO 2 H = -566kJ 7
8 C (s) + 2H 2(g) CH 4(g) H =? C (s) + O 2(g) CO 2(g) H = kj 2H 2(g) + O 2(g) 2H 2O (l) H = kj CH 4(g) + 2O 2(g) CO 2(g) + 2H 2O (l) H = kj Standard Heat of Formation Standard heat of formation is defined as the in that accompanies the of one mole of the compound at its state from its elements in their standard state. Standard state is Every free element in its standard state is assigned a H f of. Free elements include elements and any molecule (H 2O 2N 2Cl 2Br 2I 2F 2) Hrxn = Use the standard enthalpies of formation to calculate Hrxn CH 4(g) +2O 2(g) CO 2(g) + 2H 2O (l) H f(co 2(g)) = -394kJ H f(h 2O (l)) = -286 kj H f(ch 4(g)) = -75kJ H f(o 2(g)) = 0 kj 8
9 Use Table C-13 to calculate Hrxn for the following reactions 2HF (g) H 2(g) + F 2(g) 2H 2S (g) + 3O 2(g) 2H 2O (l) + 2SO 2(g) 4Fe(s) + 3O 2(g) 2Fe 2O 3 (s) 9
10 2NO (g) + O 2(g) 2NO 2(g) 2CO (g) + O 2(g) 2CO 2(g) 10
The Nature of Energy Energy is the ability to do work or produce Heat, q or Q, is ; flows due to temperature differences (always to )
 CP Chapter 17 Thermochemistry 2014-2015 Thermochemistry Thermochemistry is the study of energy that occur during chemical and physical changes (changes of state) The Nature of Energy Energy is the ability
CP Chapter 17 Thermochemistry 2014-2015 Thermochemistry Thermochemistry is the study of energy that occur during chemical and physical changes (changes of state) The Nature of Energy Energy is the ability
Ch. 17 Thermochemistry
 Ch. 17 Thermochemistry 17.1 The Flow of Energy Energy Transformations Thermochemistry: study of energy changes in chemical reactions and changes in state Chemical potential energy: energy stored in bonds
Ch. 17 Thermochemistry 17.1 The Flow of Energy Energy Transformations Thermochemistry: study of energy changes in chemical reactions and changes in state Chemical potential energy: energy stored in bonds
Name Date Class THE FLOW OF ENERGY HEAT AND WORK
 17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
CHAPTER 17 Thermochemistry
 CHAPTER 17 Thermochemistry Thermochemistry The study of the heat changes that occur during chemical reactions and physical changes of state. Chemical Change: new substances created during chemical reaction
CHAPTER 17 Thermochemistry Thermochemistry The study of the heat changes that occur during chemical reactions and physical changes of state. Chemical Change: new substances created during chemical reaction
Energy and Chemical Change
 Energy and Chemical Change Section 15.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Energy and Chemical Change Section 15.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Chapter 15 Energy and Chemical Change
 Chapter 15 Energy and Chemical Change Chemical reactions usually absorb or release energy. Section 1: Energy Section 2: Heat Section 3: Thermochemical Equations Section 4: Calculating Enthalpy Change Section
Chapter 15 Energy and Chemical Change Chemical reactions usually absorb or release energy. Section 1: Energy Section 2: Heat Section 3: Thermochemical Equations Section 4: Calculating Enthalpy Change Section
Energy and Chemical Change
 Energy and Chemical Change Section 16.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Energy and Chemical Change Section 16.1 Energy In your textbook, read about the nature of energy. In the space at the left, write true if the statement is true; if the statement is false, change the italicized
Thermochemistry. Energy and Chemical Change
 Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Chapter 11. Thermochemistry: Heat & Chemical Change
 Chapter 11 Thermochemistry: Heat & Chemical Change The Flow of Energy Thermochemistry: Study of heat changes that occur during physical processes and chemical reactions Energy Energy is the capacity to
Chapter 11 Thermochemistry: Heat & Chemical Change The Flow of Energy Thermochemistry: Study of heat changes that occur during physical processes and chemical reactions Energy Energy is the capacity to
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Chapter 5 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy
 Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Thermochemistry. Energy and Chemical Change
 Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Thermochemistry Energy and Chemical Change Energy Energy can change for and flow, but it is always conserved. The Nature of Energy Energy the ability to do work or produce heat Potential energy Kinetic
Thermochemistry. Section The flow of energy
 Thermochemistry Section 17.1 - The flow of energy What is Energy? Energy is the capacity for doing work or supplying heat Energy does not have mass or volume, and it can only be detected because of its
Thermochemistry Section 17.1 - The flow of energy What is Energy? Energy is the capacity for doing work or supplying heat Energy does not have mass or volume, and it can only be detected because of its
I. The Nature of Energy A. Energy
 I. The Nature of Energy A. Energy is the ability to do work or produce heat. It exists in 2 forms: 1. Potential energy is energy due to the composition or position of an object. 2. Kinetic energy is energy
I. The Nature of Energy A. Energy is the ability to do work or produce heat. It exists in 2 forms: 1. Potential energy is energy due to the composition or position of an object. 2. Kinetic energy is energy
Thermochemistry. Energy. 1st Law of Thermodynamics. Enthalpy / Calorimetry. Enthalpy of Formation
 THERMOCHEMISTRY Thermochemistry Energy 1st Law of Thermodynamics Enthalpy / Calorimetry Hess' Law Enthalpy of Formation The Nature of Energy Kinetic Energy and Potential Energy Kinetic energy is the energy
THERMOCHEMISTRY Thermochemistry Energy 1st Law of Thermodynamics Enthalpy / Calorimetry Hess' Law Enthalpy of Formation The Nature of Energy Kinetic Energy and Potential Energy Kinetic energy is the energy
Ch. 14 Notes ENERGY AND CHEMICAL CHANGE NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics.
 Ch. 14 Notes ENERGY AND CHEMICAL CHANGE NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Energy the capacity to do work or produce heat A. two basic types of
Ch. 14 Notes ENERGY AND CHEMICAL CHANGE NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Energy the capacity to do work or produce heat A. two basic types of
Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes
 Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes Students will be expected to: Compare the molar enthalpies of several combustion reactions involving organic compounds.
Mr Chiasson Advanced Chemistry 12 / Chemistry 12 1 Unit B: Thermochemical Changes Students will be expected to: Compare the molar enthalpies of several combustion reactions involving organic compounds.
Chapter 17 Thermochemistry
 Chapter 17 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Chapter 17 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Chemical Thermodynamics
 Chemical Thermodynamics 1 Thermodynamics Thermodynamics is a Greek term which means, heat power. Thermodynamics is the study of energy and its transformations. 2 Thermodynamics Thermochemistry how we observe,
Chemical Thermodynamics 1 Thermodynamics Thermodynamics is a Greek term which means, heat power. Thermodynamics is the study of energy and its transformations. 2 Thermodynamics Thermochemistry how we observe,
Thermochemistry. Energy (and Thermochemistry) World of Chemistry Chapter 10. Energy. Energy
 Thermochemistry Thermodynamics is the science of the relationship between heat and other forms of energy. (and Thermochemistry) World of Chemistry Chapter 10 is defined as the ability to do work or produce
Thermochemistry Thermodynamics is the science of the relationship between heat and other forms of energy. (and Thermochemistry) World of Chemistry Chapter 10 is defined as the ability to do work or produce
Energy and Chemical Change
 Energy and Chemical Change Section 15.1 Energy Section 15.2 Heat Section 15.3 Thermochemical Equations Section 15.4 Calculating Enthalpy Change Section 15.5 Reaction Spontaneity Click a hyperlink or folder
Energy and Chemical Change Section 15.1 Energy Section 15.2 Heat Section 15.3 Thermochemical Equations Section 15.4 Calculating Enthalpy Change Section 15.5 Reaction Spontaneity Click a hyperlink or folder
All chemical reactions involve changes in energy. Typically this energy comes in the form of heat.
 Topic: Thermochemistry Essential Question: How does energy flow in chemical reactions? Name: Class: Date: / / Period: All chemical reactions involve changes in energy. Typically this energy comes in the
Topic: Thermochemistry Essential Question: How does energy flow in chemical reactions? Name: Class: Date: / / Period: All chemical reactions involve changes in energy. Typically this energy comes in the
Thermochemistry-Part 1
 Brad Collins Thermochemistry-Part 1 Chapter 7 Thermochemistry Thermodynamics: The study of energy Thermochemistry: The study of energy in chemical reactions Energy: The capacity to do work Work = force
Brad Collins Thermochemistry-Part 1 Chapter 7 Thermochemistry Thermodynamics: The study of energy Thermochemistry: The study of energy in chemical reactions Energy: The capacity to do work Work = force
_ + Units of Energy. Energy in Thermochemistry. Thermochemistry. Energy flow between system and surroundings. 100º C heat 50º C
 Units of Energy Like we saw with pressure, many different units are used throughout the world for energy. SI unit for energy 1kg m 1J = 2 s 2 Joule (J) calorie (cal) erg (erg) electron volts (ev) British
Units of Energy Like we saw with pressure, many different units are used throughout the world for energy. SI unit for energy 1kg m 1J = 2 s 2 Joule (J) calorie (cal) erg (erg) electron volts (ev) British
Thermochemistry: Energy Flow and Chemical Reactions
 Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
Name: Class: Date: ID: A
 Name: Class: _ Date: _ ID: A Chpter 17 review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of these phase changes is an endothermic process? a.
Name: Class: _ Date: _ ID: A Chpter 17 review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of these phase changes is an endothermic process? a.
CHAPTER 17: THERMOCHEMISTRY. Mrs. Brayfield
 CHAPTER 17: THERMOCHEMISTRY Mrs. Brayfield REVIEW What is the law of conservation of energy? It states that energy cannot be created or destroyed So the energy of any process is the same THERMOCHEMISTRY
CHAPTER 17: THERMOCHEMISTRY Mrs. Brayfield REVIEW What is the law of conservation of energy? It states that energy cannot be created or destroyed So the energy of any process is the same THERMOCHEMISTRY
Name Class Date. As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings.
 Name Class Date Thermochemistry 17.1 The Flow of Energy As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings. Process Cause Effect endothermic
Name Class Date Thermochemistry 17.1 The Flow of Energy As you read Lesson 17.1, use the cause and effect chart below. Complete the chart with the terms system and surroundings. Process Cause Effect endothermic
Introduction to Thermochemistry. Thermochemistry Unit. Definition. Terminology. Terminology. Terminology 07/04/2016. Chemistry 30
 Thermochemistry Unit Introduction to Thermochemistry Chemistry 30 Definition Thermochemistry is the branch of chemistry concerned with the heat produced and used in chemical reactions. Most of thermochemistry
Thermochemistry Unit Introduction to Thermochemistry Chemistry 30 Definition Thermochemistry is the branch of chemistry concerned with the heat produced and used in chemical reactions. Most of thermochemistry
Energy Transformations
 Thermochemistry Energy Transformations Thermochemistry - concerned with heat changes that occur during chemical reactions Energy - capacity for doing work or supplying heat weightless, odorless, tasteless
Thermochemistry Energy Transformations Thermochemistry - concerned with heat changes that occur during chemical reactions Energy - capacity for doing work or supplying heat weightless, odorless, tasteless
Chemistry Chapter 16. Reaction Energy
 Chemistry Reaction Energy Section 16.1.I Thermochemistry Objectives Define temperature and state the units in which it is measured. Define heat and state its units. Perform specific-heat calculations.
Chemistry Reaction Energy Section 16.1.I Thermochemistry Objectives Define temperature and state the units in which it is measured. Define heat and state its units. Perform specific-heat calculations.
Reaction Energy. Thermochemistry
 Reaction Energy Thermochemistry Thermochemistry The study of the transfers of energy as heat that accompany chemical reactions & physical changes Thermochemistry -In studying heat changes, think of defining
Reaction Energy Thermochemistry Thermochemistry The study of the transfers of energy as heat that accompany chemical reactions & physical changes Thermochemistry -In studying heat changes, think of defining
Thermochemistry. The study of energy changes that occur during chemical reactions and changes in state.
 Energy Thermochemistry The study of energy changes that occur during chemical reactions and changes in state. The Nature of Energy Energy - the ability to do work or produce heat Energy is stored in the
Energy Thermochemistry The study of energy changes that occur during chemical reactions and changes in state. The Nature of Energy Energy - the ability to do work or produce heat Energy is stored in the
Chapter 8. Thermochemistry 강의개요. 8.1 Principles of Heat Flow. 2) Magnitude of Heat Flow. 1) State Properties. Basic concepts : study of heat flow
 강의개요 Basic concepts : study of heat flow Chapter 8 Thermochemistry Calorimetry : experimental measurement of the magnitude and direction of heat flow Thermochemical Equations Copyright 2005 연세대학교이학계열일반화학및실험
강의개요 Basic concepts : study of heat flow Chapter 8 Thermochemistry Calorimetry : experimental measurement of the magnitude and direction of heat flow Thermochemical Equations Copyright 2005 연세대학교이학계열일반화학및실험
Name Date Class THERMOCHEMISTRY
 Name Date Class 17 THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY HEAT AND WORK (pages 505 510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Name Date Class 17 THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY HEAT AND WORK (pages 505 510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Chapter 8 Thermochemistry
 William L Masterton Cecile N. Hurley http://academic.cengage.com/chemistry/masterton Chapter 8 Thermochemistry Edward J. Neth University of Connecticut Outline 1. Principles of heat flow 2. Measurement
William L Masterton Cecile N. Hurley http://academic.cengage.com/chemistry/masterton Chapter 8 Thermochemistry Edward J. Neth University of Connecticut Outline 1. Principles of heat flow 2. Measurement
Section 9: Thermodynamics and Energy
 Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Energy. Different types of energy exist (heat, potential, kinetic, chemical, nuclear etc.)
 Change in Energy Energy Different types of energy exist (heat, potential, kinetic, chemical, nuclear etc.) Heat - the energy transferred between objects that are at different temperatures. Unit of heat
Change in Energy Energy Different types of energy exist (heat, potential, kinetic, chemical, nuclear etc.) Heat - the energy transferred between objects that are at different temperatures. Unit of heat
CHEM 1105 S10 March 11 & 14, 2014
 CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
THERMOCHEMISTRY & DEFINITIONS
 THERMOCHEMISTRY & DEFINITIONS Thermochemistry is the study of the study of relationships between chemistry and energy. All chemical changes and many physical changes involve exchange of energy with the
THERMOCHEMISTRY & DEFINITIONS Thermochemistry is the study of the study of relationships between chemistry and energy. All chemical changes and many physical changes involve exchange of energy with the
Thermochemistry: Heat and Chemical Change
 Thermochemistry: Heat and Chemical Change 1 Heat or Thermal Energy (q) Heat is a form of energy Is heat the same as temperature? Heat flows between two objects at different temperatures. Hot Cold 2 Chemical
Thermochemistry: Heat and Chemical Change 1 Heat or Thermal Energy (q) Heat is a form of energy Is heat the same as temperature? Heat flows between two objects at different temperatures. Hot Cold 2 Chemical
Chemistry Heat Review. Heat: Temperature: Enthalpy: Calorimetry: Activation energy:
 Chemistry Heat Review Name Date Vocabulary Heat: Temperature: Enthalpy: Calorimetry: Activation energy: Formulas Heat of phase change Heat for temperature increase Heat of reaction Endothermic/Exothermic
Chemistry Heat Review Name Date Vocabulary Heat: Temperature: Enthalpy: Calorimetry: Activation energy: Formulas Heat of phase change Heat for temperature increase Heat of reaction Endothermic/Exothermic
Unit 15 Energy and Thermochemistry Notes
 Name Period CRHS Academic Chemistry Unit 15 Energy and Thermochemistry Notes Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Name Period CRHS Academic Chemistry Unit 15 Energy and Thermochemistry Notes Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Unit 15 Energy and Thermochemistry Notes
 Name KEY Period CRHS Academic Chemistry Unit 15 Energy and Thermochemistry Notes Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Name KEY Period CRHS Academic Chemistry Unit 15 Energy and Thermochemistry Notes Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Chapter 6 Problems: 9, 19, 24, 25, 26, 27, 31-33, 37, 39, 43, 45, 47, 48, 53, 55, 57, 59, 65, 67, 73, 78-82, 85, 89, 93
 Chapter 6 Problems: 9, 19, 24, 25, 26, 27, 31-33, 37, 39, 43, 45, 47, 48, 53, 55, 57, 59, 65, 67, 73, 78-82, 85, 89, 93 Chapter 6 Thermochemistry The study of chemical reactions and the energy changes
Chapter 6 Problems: 9, 19, 24, 25, 26, 27, 31-33, 37, 39, 43, 45, 47, 48, 53, 55, 57, 59, 65, 67, 73, 78-82, 85, 89, 93 Chapter 6 Thermochemistry The study of chemical reactions and the energy changes
THERMOCHEMISTRY. This section explains the relationship between energy and heat, and distinguishes between heat capacity and specific heat.
 I Name _ Date _ Class _ THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY-HEAT AND WORK (pages 505-510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
I Name _ Date _ Class _ THERMOCHEMISTRY SECTION 17.1 THE FLOW OF ENERGY-HEAT AND WORK (pages 505-510) This section explains the relationship between energy and heat, and distinguishes between heat capacity
Heat. Heat Terminology 04/12/2017. System Definitions. System Definitions
 System Definitions Heat Physical Science 20 Ms. Hayduk Heat Terminology System: the part of the universe being studied (big Earth, or small one atom) Surroundings: the part of the universe outside the
System Definitions Heat Physical Science 20 Ms. Hayduk Heat Terminology System: the part of the universe being studied (big Earth, or small one atom) Surroundings: the part of the universe outside the
Name Date Class SECTION 16.1 PROPERTIES OF SOLUTIONS
 SOLUTIONS Practice Problems In your notebook, solve the following problems. SECTION 16.1 PROPERTIES OF SOLUTIONS 1. The solubility of CO 2 in water at 1.22 atm is 0.54 g/l. What is the solubility of carbon
SOLUTIONS Practice Problems In your notebook, solve the following problems. SECTION 16.1 PROPERTIES OF SOLUTIONS 1. The solubility of CO 2 in water at 1.22 atm is 0.54 g/l. What is the solubility of carbon
Gilbert Kirss Foster. Chapter 9. Thermochemistry. Energy Changes in Chemical Reactions
 Gilbert Kirss Foster Chapter 9 Thermochemistry Energy Changes in Chemical Reactions Chapter Outline 9.1 Energy as a Reactant or Product 9.2 Transferring Heat and Doing Work 9.3 Enthalpy and Enthalpy Changes
Gilbert Kirss Foster Chapter 9 Thermochemistry Energy Changes in Chemical Reactions Chapter Outline 9.1 Energy as a Reactant or Product 9.2 Transferring Heat and Doing Work 9.3 Enthalpy and Enthalpy Changes
Chapter 5. Thermochemistry
 Chapter 5 Thermochemistry Dr. A. Al-Saadi 1 Preview Introduction to thermochemistry: Potential energy and kinetic energy. Chemical energy. Internal energy, work and heat. Exothermic vs. endothermic reactions.
Chapter 5 Thermochemistry Dr. A. Al-Saadi 1 Preview Introduction to thermochemistry: Potential energy and kinetic energy. Chemical energy. Internal energy, work and heat. Exothermic vs. endothermic reactions.
Chapter 6. Thermochemistry
 Chapter 6 Thermochemistry Section 5.6 The Kinetic Molecular Theory of Gases http://www.scuc.txed.net/webpages/dmackey/files /chap06notes.pdf ..\..\..\..\..\..\Videos\AP Videos\Thermochemistry\AP
Chapter 6 Thermochemistry Section 5.6 The Kinetic Molecular Theory of Gases http://www.scuc.txed.net/webpages/dmackey/files /chap06notes.pdf ..\..\..\..\..\..\Videos\AP Videos\Thermochemistry\AP
Chapter 5 Principles of Chemical Reactivity: Energy and Chemical Reactions
 Chapter 5 Principles of Chemical Reactivity: Energy and Chemical Reactions Jeffrey Mack California State University, Sacramento Energy & Chemistry Questions that need to be addressed: How do we measure
Chapter 5 Principles of Chemical Reactivity: Energy and Chemical Reactions Jeffrey Mack California State University, Sacramento Energy & Chemistry Questions that need to be addressed: How do we measure
2 Copyright Pearson Education, Inc., or its affiliates. All Rights Reserved.
 CHEMISTRY & YOU Chapter 17 Thermochemistry 17.1 The Flow of Energy 17. Measuring and Expressing Enthalpy Changes 17.3 Heat in Changes of State 17.4 Calculating Heats of Reaction Why does sweating help
CHEMISTRY & YOU Chapter 17 Thermochemistry 17.1 The Flow of Energy 17. Measuring and Expressing Enthalpy Changes 17.3 Heat in Changes of State 17.4 Calculating Heats of Reaction Why does sweating help
Thermochemistry HW. PSI Chemistry
 Thermochemistry HW PSI Chemistry Name Energy 1) Objects can possess energy as: (a) endothermic energy (b) potential energy A) a only B) b only C) c only D) a and c E) b and c (c) kinetic energy 2) The
Thermochemistry HW PSI Chemistry Name Energy 1) Objects can possess energy as: (a) endothermic energy (b) potential energy A) a only B) b only C) c only D) a and c E) b and c (c) kinetic energy 2) The
This reaction is ENDOTHERMIC. Energy is being transferred from the room/flask/etc. (the SURROUNDINGS) to the reaction itself (the SYSTEM).
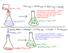 151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
The Nature of Energy. Chapter Six: Kinetic vs. Potential Energy. Energy and Work. Temperature vs. Heat
 The Nature of Energy Chapter Six: THERMOCHEMISTRY Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationship between chemical reactions and energy changes
The Nature of Energy Chapter Six: THERMOCHEMISTRY Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationship between chemical reactions and energy changes
Chapter 11. Thermochemistry. 1. Let s begin by previewing the chapter (Page 292). 2. We will partner read Pages
 Chapter 11 Thermochemistry 1. Let s begin by previewing the chapter (Page 292). 2. We will partner read Pages 293-94 The Flow of energy - heat Thermochemistry concerned with the heat changes that occur
Chapter 11 Thermochemistry 1. Let s begin by previewing the chapter (Page 292). 2. We will partner read Pages 293-94 The Flow of energy - heat Thermochemistry concerned with the heat changes that occur
First Law of Thermodynamics
 Energy Energy: ability to do work or produce heat. Types of energy 1) Potential energy - energy possessed by objects due to position or arrangement of particles. Forms of potential energy - electrical,
Energy Energy: ability to do work or produce heat. Types of energy 1) Potential energy - energy possessed by objects due to position or arrangement of particles. Forms of potential energy - electrical,
Chapter 6: Thermochemistry
 Chapter 6: Thermochemistry 1. Light the Furnace: The Nature of Energy and Its Transformations a. Thermochemistry is the study of the relationships between chemistry and energy i. This means that we will
Chapter 6: Thermochemistry 1. Light the Furnace: The Nature of Energy and Its Transformations a. Thermochemistry is the study of the relationships between chemistry and energy i. This means that we will
Energy Ability to produce change or do work. First Law of Thermodynamics. Heat (q) Quantity of thermal energy
 THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
Chapter 5 - Thermochemistry
 Chapter 5 - Thermochemistry Study of energy changes that accompany chemical rx s. I) Nature of Energy Energy / Capacity to do work Mechanical Work w = F x d Heat energy - energy used to cause the temperature
Chapter 5 - Thermochemistry Study of energy changes that accompany chemical rx s. I) Nature of Energy Energy / Capacity to do work Mechanical Work w = F x d Heat energy - energy used to cause the temperature
Energy: Heat, Nutrients, and Enthalpy Page 13
 V. Thermochemistry A. Thermochemistry is concerned with the B. Such reactions and changes are. VI. Chemical Reactions A. Chemical compounds contain B. Chemical potential energy. C. In chemical reactions,
V. Thermochemistry A. Thermochemistry is concerned with the B. Such reactions and changes are. VI. Chemical Reactions A. Chemical compounds contain B. Chemical potential energy. C. In chemical reactions,
THERMOCHEMISTRY CHAPTER 11
 THERMOCHEMISTRY CHAPTER 11 ENERGY AND HEAT nthermochemistry: The study of the energy changes that accompany chemical reactions and changes in the physical states of matter. ENERGY AND HEAT nwork: Energy
THERMOCHEMISTRY CHAPTER 11 ENERGY AND HEAT nthermochemistry: The study of the energy changes that accompany chemical reactions and changes in the physical states of matter. ENERGY AND HEAT nwork: Energy
Ch. 6 Enthalpy Changes
 Ch. 6 Enthalpy Changes Energy: The capacity to do work. In Physics, there are 2 main types of energy Kinetic (energy of motion) = ½ mv 2 Potential (energy of position due to gravity)= mgh In Chemistry,
Ch. 6 Enthalpy Changes Energy: The capacity to do work. In Physics, there are 2 main types of energy Kinetic (energy of motion) = ½ mv 2 Potential (energy of position due to gravity)= mgh In Chemistry,
Slide 1 / Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy. a only b only c only a and c b and c
 Slide 1 / 84 1 Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy A B C D E a only b only c only a and c b and c Slide 2 / 84 2 The internal energy of a system
Slide 1 / 84 1 Objects can possess energy as: (a) endothermic energy (b) potential energy (c) kinetic energy A B C D E a only b only c only a and c b and c Slide 2 / 84 2 The internal energy of a system
Warm up. 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]?
![Warm up. 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]? Warm up. 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]?](/thumbs/78/77877522.jpg) Warm up 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]? 4) What is the concentration of H 2 SO 4 if 30.1 ml
Warm up 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]? 4) What is the concentration of H 2 SO 4 if 30.1 ml
Energy and Chemical Change
 Energy and Chemical Change Reviewing Vocabulary Match the definition in Column A with the term in Column B. h e d p c f a r m t j i s l u k n q g o Column A 1. The ability to do work or produce heat 2.
Energy and Chemical Change Reviewing Vocabulary Match the definition in Column A with the term in Column B. h e d p c f a r m t j i s l u k n q g o Column A 1. The ability to do work or produce heat 2.
Brown, LeMay Ch 5 AP Chemistry Monta Vista High School
 Brown, LeMay Ch 5 AP Chemistry Monta Vista High School 1 From Greek therme (heat); study of energy changes in chemical reactions Energy: capacity do work or transfer heat Joules (J), kilo joules (kj) or
Brown, LeMay Ch 5 AP Chemistry Monta Vista High School 1 From Greek therme (heat); study of energy changes in chemical reactions Energy: capacity do work or transfer heat Joules (J), kilo joules (kj) or
Chapter 16 Theories of Energy Changes
 {Read p. 624 and 626 to understand concepts} Class discussion for chapter 17.3 Chapter 16 Theories of Energy Changes Section 16.1A Temperature change and Heat THERMODYNAMICS - the study of energy and energy
{Read p. 624 and 626 to understand concepts} Class discussion for chapter 17.3 Chapter 16 Theories of Energy Changes Section 16.1A Temperature change and Heat THERMODYNAMICS - the study of energy and energy
ENTHALPY CHANGE CHAPTER 4
 ENTHALPY CHANGE CHAPTER 4 ENTHALPY Is the total energy of a system. E k = Kinetic energy. Vibrational Rotational Translational E due to motion H = E k + E p E P = Potential energy Attractive force b/w
ENTHALPY CHANGE CHAPTER 4 ENTHALPY Is the total energy of a system. E k = Kinetic energy. Vibrational Rotational Translational E due to motion H = E k + E p E P = Potential energy Attractive force b/w
1. Fill in the blanks with the following: kinetic, potential, chemical, thermal. One word will be used twice.
 Thermo Worksheets Name Class Period Types of Energy and the Law of Conservation of Energy 1. Fill in the blanks with the following: kinetic, potential, chemical, thermal. One word will be used twice. Solar
Thermo Worksheets Name Class Period Types of Energy and the Law of Conservation of Energy 1. Fill in the blanks with the following: kinetic, potential, chemical, thermal. One word will be used twice. Solar
Measuring and Expressing Enthalpy Changes. Copyright Pearson Prentice Hall. Measuring and Expressing Enthalpy Changes. Calorimetry
 Measuring and Expressing Enthalpy Changes A burning match releases heat to its surroundings in all directions. How much heat does this exothermic reaction release? You will learn to measure heat flow in
Measuring and Expressing Enthalpy Changes A burning match releases heat to its surroundings in all directions. How much heat does this exothermic reaction release? You will learn to measure heat flow in
Chapter Objectives. Chapter 9 Energy and Chemistry. Chapter Objectives. Energy Use and the World Economy. Energy Use and the World Economy
 Chapter Objectives Larry Brown Tom Holme www.cengage.com/chemistry/brown Chapter 9 Energy and Chemistry Explain the economic importance of conversions between different forms of energy and the inevitability
Chapter Objectives Larry Brown Tom Holme www.cengage.com/chemistry/brown Chapter 9 Energy and Chemistry Explain the economic importance of conversions between different forms of energy and the inevitability
Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes
 Thermochemistry Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes heat flows from high to low (hot cool) endothermic reactions: absorb energy
Thermochemistry Thermochemistry: the study of energy (in the from of heat) changes that accompany physical & chemical changes heat flows from high to low (hot cool) endothermic reactions: absorb energy
Energy Ability to produce change or do work. First Law of Thermodynamics. Heat (q) Quantity of thermal energy
 THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
THERMOCHEMISTRY Thermodynamics Study of energy and its interconversions Energy is TRANSFORMED in a chemical reaction (POTENTIAL to KINETIC) HEAT (energy transfer) is also usually produced or absorbed -SYSTEM:
Thermochemistry Chapter 4
 Thermochemistry Chapter 4 Thermochemistry is the study of energy changes that occur during chemical reactions Focus is on heat and matter transfer between the system and the surroundings Energy The ability
Thermochemistry Chapter 4 Thermochemistry is the study of energy changes that occur during chemical reactions Focus is on heat and matter transfer between the system and the surroundings Energy The ability
To calculate heat (q) for a given temperature change: heat (q) = (specific heat) (mass) ( T) where T = T f T i
 Use your textbook or other resources available to answer the following questions General Information: Thermochemistry Phase Change A change in the physical form/state but not a change in the chemical identity
Use your textbook or other resources available to answer the following questions General Information: Thermochemistry Phase Change A change in the physical form/state but not a change in the chemical identity
Name: General Chemistry Chapter 11 Thermochemistry- Heat and Chemical Change
 Name: General Chemistry Chapter 11 Thermochemistry- Heat and Chemical Change Notepack 1 Section 11.1: The Flow of Energy Heat (Pages 293 299) 1. Define the following terms: a. Thermochemistry b. Energy
Name: General Chemistry Chapter 11 Thermochemistry- Heat and Chemical Change Notepack 1 Section 11.1: The Flow of Energy Heat (Pages 293 299) 1. Define the following terms: a. Thermochemistry b. Energy
- Joule (J): SI unit for energy. It's defined based on the equation for kinetic energy. from. mass. velocity
 153 ENERGY UNITS - calorie (cal): the amount of energy required to change the temperature of one gram of water by one degree Celsius (or Kelvin) 1g 1g add one calorie of energy - Calories in food? The
153 ENERGY UNITS - calorie (cal): the amount of energy required to change the temperature of one gram of water by one degree Celsius (or Kelvin) 1g 1g add one calorie of energy - Calories in food? The
11B, 11E Temperature and heat are related but not identical.
 Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Chapter 5. Thermochemistry
 Chapter 5 Thermochemistry Energy Thermodynamics Study of the relationship between heat, work, and other forms of energy Thermochemistry A branch of thermodynamics Focuses on the study of heat given off
Chapter 5 Thermochemistry Energy Thermodynamics Study of the relationship between heat, work, and other forms of energy Thermochemistry A branch of thermodynamics Focuses on the study of heat given off
Name Chemistry / / Understanding Phase Changes
 Name Chemistry / / Understanding Phase Changes As a piece of ice is exposed to a warmer environment, it begins to absorb heat. The heat causes the solid molecules to vibrate faster. Eventually, the ice
Name Chemistry / / Understanding Phase Changes As a piece of ice is exposed to a warmer environment, it begins to absorb heat. The heat causes the solid molecules to vibrate faster. Eventually, the ice
Topic 5: Energetics. Heat & Calorimetry. Thursday, March 22, 2012
 Topic 5: Energetics Heat & Calorimetry 1 Heat is energy that is transferred from one object to another due to a difference in temperature Temperature is a measure of the average kinetic energy of a body
Topic 5: Energetics Heat & Calorimetry 1 Heat is energy that is transferred from one object to another due to a difference in temperature Temperature is a measure of the average kinetic energy of a body
Energy, Heat and Temperature. Introduction
 Energy, Heat and Temperature Introduction 3 basic types of energy: Potential (possibility of doing work because of composition or position) Kinetic (moving objects doing work) Radiant (energy transferred
Energy, Heat and Temperature Introduction 3 basic types of energy: Potential (possibility of doing work because of composition or position) Kinetic (moving objects doing work) Radiant (energy transferred
Chapter 6: Thermochemistry
 Chapter 6: Thermochemistry Section 6.1: Introduction to Thermochemistry Thermochemistry refers to the study of heat flow or heat energy in a chemical reaction. In a study of Thermochemistry the chemical
Chapter 6: Thermochemistry Section 6.1: Introduction to Thermochemistry Thermochemistry refers to the study of heat flow or heat energy in a chemical reaction. In a study of Thermochemistry the chemical
Thermochemistry (chapter 5)
 Thermochemistry (chapter 5) Basic Definitions: Thermochemistry = the study of the energy changes that accompany physical and chemical changes of matter. Energy is defined as the ability to do work or the
Thermochemistry (chapter 5) Basic Definitions: Thermochemistry = the study of the energy changes that accompany physical and chemical changes of matter. Energy is defined as the ability to do work or the
THE ENERGY OF THE UNIVERSE IS CONSTANT.
 Chapter 6 Thermochemistry.notebook Chapter 6: Thermochemistry Jan 29 1:37 PM 6.1 The Nature of Energy Thermodynamics: The study of energy and its interconversions Energy: the capacity to do work or to
Chapter 6 Thermochemistry.notebook Chapter 6: Thermochemistry Jan 29 1:37 PM 6.1 The Nature of Energy Thermodynamics: The study of energy and its interconversions Energy: the capacity to do work or to
Enthalpies of Reaction
 Enthalpies of Reaction Enthalpy is an extensive property Magnitude of H is directly related to the amount of reactant used up in a process. CH 4 (g) + 2O 2 (g) CO 2 (g) + 2H 2 O(l) H = 890 kj 2CH 4 (g)
Enthalpies of Reaction Enthalpy is an extensive property Magnitude of H is directly related to the amount of reactant used up in a process. CH 4 (g) + 2O 2 (g) CO 2 (g) + 2H 2 O(l) H = 890 kj 2CH 4 (g)
CHEMISTRY: Chapter 10 Prep-Test
 CHEMISTRY: Chapter 10 Prep-Test Matching Match each item with the correct statement below. a. calorimeter d. temperature b. calorie e. specific heat c. joule f. heat 1. quantity of heat needed to raise
CHEMISTRY: Chapter 10 Prep-Test Matching Match each item with the correct statement below. a. calorimeter d. temperature b. calorie e. specific heat c. joule f. heat 1. quantity of heat needed to raise
CHEMISTRY. Chapter 5 Thermochemistry
 CHEMISTRY The Central Science 8 th Edition Chapter 5 Thermochemistry Dr. Kozet YAPSAKLI The Nature of Energy Kinetic and Potential Energy Potential energy can be converted into kinetic energy. E p = mgh
CHEMISTRY The Central Science 8 th Edition Chapter 5 Thermochemistry Dr. Kozet YAPSAKLI The Nature of Energy Kinetic and Potential Energy Potential energy can be converted into kinetic energy. E p = mgh
Energy, Heat and Chemical Change
 Energy, Heat and Chemical Change Chemistry 35 Fall 2000 Thermochemistry A part of Thermodynamics dealing with energy changes associated with physical and chemical reactions Why do we care? -will a reaction
Energy, Heat and Chemical Change Chemistry 35 Fall 2000 Thermochemistry A part of Thermodynamics dealing with energy changes associated with physical and chemical reactions Why do we care? -will a reaction
Thermochemistry (chapter 5)
 Thermochemistry (chapter 5) Is the study of the energy changes that accompany physical and chemical changes. Energy is defined as the ability to do work or the capacity to produce change. The forms of
Thermochemistry (chapter 5) Is the study of the energy changes that accompany physical and chemical changes. Energy is defined as the ability to do work or the capacity to produce change. The forms of
Chapter 17: Energy and Kinetics
 Pages 510-547 S K K Chapter 17: Energy and Kinetics Thermochemistry: Causes of change in systems Kinetics: Rate of reaction progress (speed) Heat, Energy, and Temperature changes S J J Heat vs Temperature
Pages 510-547 S K K Chapter 17: Energy and Kinetics Thermochemistry: Causes of change in systems Kinetics: Rate of reaction progress (speed) Heat, Energy, and Temperature changes S J J Heat vs Temperature
Topic 05 Energetics : Heat Change. IB Chemistry T05D01
 Topic 05 Energetics 5.1-5.2: Heat Change IB Chemistry T05D01 5.1 Exothermic and endothermic reactions - 1 hour 5.1.1 Define the terms exothermic reaction, endothermic reaction and standard enthalpy change
Topic 05 Energetics 5.1-5.2: Heat Change IB Chemistry T05D01 5.1 Exothermic and endothermic reactions - 1 hour 5.1.1 Define the terms exothermic reaction, endothermic reaction and standard enthalpy change
Unit 14. States of Matter & Thermochemistry
 Unit 14 Flashback: States of Matter & Thermochemistry Characteristic Solids Liquids Gases Shape Volume Density Fluidity Compressibility Picture Phase Diagram Shows the relationship between solid, liquid,
Unit 14 Flashback: States of Matter & Thermochemistry Characteristic Solids Liquids Gases Shape Volume Density Fluidity Compressibility Picture Phase Diagram Shows the relationship between solid, liquid,
Selected Questions on Chapter 5 Thermochemistry
 Selected Questions on Chapter 5 Thermochemistry Circle the correct answer: 1) At what velocity (m/s) must a 20.0 g object be moving in order to possess a kinetic energy of 1.00 J? A) 1.00 B) 100 10 2 C)
Selected Questions on Chapter 5 Thermochemistry Circle the correct answer: 1) At what velocity (m/s) must a 20.0 g object be moving in order to possess a kinetic energy of 1.00 J? A) 1.00 B) 100 10 2 C)
Ch 6. Energy and Chemical Change. Brady & Senese, 5th Ed.
 Ch 6. Energy and Chemical Change Brady & Senese, 5th Ed. Energy Is The Ability To Do Work Energy is the ability to do work (move mass over a distance) or transfer heat Types: kinetic and potential kinetic:
Ch 6. Energy and Chemical Change Brady & Senese, 5th Ed. Energy Is The Ability To Do Work Energy is the ability to do work (move mass over a distance) or transfer heat Types: kinetic and potential kinetic:
Chapter 6 Thermochemistry 許富銀
 Chapter 6 Thermochemistry 許富銀 6.1 Chemical Hand Warmers Thermochemistry: the study of the relationships between chemistry and energy Hand warmers use the oxidation of iron as the exothermic reaction: Nature
Chapter 6 Thermochemistry 許富銀 6.1 Chemical Hand Warmers Thermochemistry: the study of the relationships between chemistry and energy Hand warmers use the oxidation of iron as the exothermic reaction: Nature
Chapter 8 Thermochemistry: Chemical Energy. Chemical Thermodynamics
 Chapter 8 Thermochemistry: Chemical Energy Chapter 8 1 Chemical Thermodynamics Chemical Thermodynamics is the study of the energetics of a chemical reaction. Thermodynamics deals with the absorption or
Chapter 8 Thermochemistry: Chemical Energy Chapter 8 1 Chemical Thermodynamics Chemical Thermodynamics is the study of the energetics of a chemical reaction. Thermodynamics deals with the absorption or
Chapter 8 Thermochemistry: Chemical Energy
 Chapter 8 Thermochemistry: Chemical Energy 國防醫學院生化學科王明芳老師 2011-11-8 & 2011-11-15 Chapter 8/1 Energy and Its Conservation Conservation of Energy Law: Energy cannot be created or destroyed; it can only be
Chapter 8 Thermochemistry: Chemical Energy 國防醫學院生化學科王明芳老師 2011-11-8 & 2011-11-15 Chapter 8/1 Energy and Its Conservation Conservation of Energy Law: Energy cannot be created or destroyed; it can only be
