Chemistry: classifying chemical and physical changes in various materials/substances
|
|
|
- Priscilla Arnold
- 5 years ago
- Views:
Transcription
1 Chemistry: classifying chemical and physical changes in various materials/substances Nikki Schilling, Ames, St. Paul Mn Based on original activity from Cool Chemistry Concoctions by Joe Rhatigan and Veronika Alice Gunter, p.24 Author Profile Summary In this classroom lab, students will investigate the characteristics of a chemical change. Students will also provide a definition of what constitutes a physical change through observation of several examples. Students will use/create the criteria for identifying a chemical change. Several examples of a chemical change will also be observed for students to compare to physical changes. The students will use the five characteristics that have been created to determine/judge whether or not baking soda, citric acid and water creates a chemical change. Students will write down observations of the reaction and use the five characteristics to explain why it is in fact a chemical reaction (The five characteristics will be listed for use). Learning Goals *Students will apply the five characteristics of a chemical change to real world reactions to determine the type of change that has occurred. *Students will be able to express why a given substance/mixture has had a chemical change. Concepts: -Physical change -Chemical change Key Vocabulary: -Carbon Dioxide Acid -Element Base -Chemical Citric Acid
2 Context for Use This activity would work well in 6th grade but could also be used in lower grades if needed. This would take place in the classroom, which would span 1-2 weeks depending on the number of examples and experiments you are willing to do with your students. This would follow a unit on structure of matter. This will provide students with knowledge in the physical properties of elements. This could even following unit on testing ph samples. It would provide students with an understanding of why acids and bases react to one another. The materials needed for this lesson would range from household materials to specialty items that can be found at health food stores. The desired skills for this lesson would require general knowledge of physical changes, using measuring instruments and writing observations. I would do each experiment in half an hour increments. Subject : Chemistry: General Chemistry Resource Type : Activities: Classroom Activity, Lab Activity Grade Level : Intermediate (3-5), Middle (6-8) Description and Teaching Materials Materials: -Paper -Matches -Vinegar -Milk -Citric acid -Baking soda -Water (ice) -Towel (can be paper) -Large & small plastic bags -Goggles -Lemon juice
3 -Red cabbage juice -Measuring cups or spoons In this activity I would start by comparing the difference between crumpling a piece of paper and burning a piece of paper. In this introduction students will make observations of what they have seen. First they would observe the paper being crumpled. Then their observations will be written on an observation chart. Then I would burn a small piece of paper and write observations down on the chart. I would ask this question, "Which of one was a physical change and why?" Students should be able to identify which one was a physical change by using what they already know about what qualifies as a physical change. We would have discussed why it was. From this point I would introduce the concept of a chemical change. The class would think, pair, share on why the burning paper was a chemical change. On a chart paper the students would list the characteristics of a physical change on one side. The other side would be the characteristics of a chemical change. I would help guide the students in clear characteristics. Ideally I would have the students come to five characteristics: chemical change has color change, temperature change, production of a solid, forms a gas, and change in odor. The students would use their definitions to provided them with evidence of physical and chemical changes while experimenting with the following items: Mixing vinegar and baking soda: 1-tablespoon baking soda and 20 ml of vinegar in a plastic bag (Have students put the vinegar in first then the baking soda, seal bag before mixing) Ice: On a tray observe a piece of ice melt
4 Water being soaked up into a towel: 50 ml of water and a paper towel Students will observe the water can be taken out of the towel Lemon juice and cabbage juice: 10 ml of cabbage juice and 10 ml of lemon juice It will change the colors from purple to pink Citric Acid, baking soda, and water: 1-tablespoon citric acid and 1-tablespoon baking soda in a large plastic zip lock bag Water filled halfway in a small plastic bag Place the unzipped bag with water into your large bag. Have students get as much air out of the bag before closing. Turn the bag so the water spills out. (The water will become cold and CO2 gas will fill the bag) For each experiment the students will make a hypothesis of whether it will be a physical or chemical change. Students will write and draw their observations down. They will use their observations and definitions of physical and chemical changes to decide if their hypothesis was true. After each experiment as a closer, we would have a quick share. What did we find out? Which characteristics were present in the experiment? Does anything surprise you?
5 Source Ref. Rhatigan, Joe & Gunter, Veronika Alice. Cool Chemistry Concoctions. New York: Lark, Teaching Notes and Tips I go over safety when dealing with citric acid. I make sure students understand it may look like sugar, but it is not. I would have the student wear goggles to prevent exposure to their eyes. Citric acid stings just like lemon juice. I remind students with cuts to be extra careful and if it does get into a cut to go wash it out. I also go over the information multiple times and have students repeat back the procedures to insure students have an understanding of expectations and concepts. Ideally I would have groups of two but with limited supplies I have groups of four. In their groups, jobs would be given so no one person would be doing all the work for the group. This activity is different from my past one because of the use of citric acid. The experiment with citric acid would be my last one done because my students could then use their prior knowledge to the vinegar and baking soda experiment to make a better prediction of what will happen. If you don't have time to do all the experiments, I have used the citric acid, baking soda and water to make dragon eyes. If you mix the baking soda and citric acid together in a cup and add about a half-teaspoon of baby oil to the mixture, it sticks together without changing. You can roll them into small balls and students can take them home. It will have the same effect as the experiment. You can even add food color if you like. Assessment I would give a pre- and post test to the students using the experiments. The quiz would describe the reaction and the students would identify what type of change using the characteristics of a physical and chemical change. During the week I would have students turn in their science notebook, which would have the entire student's hypothesizes, observations and conclusions from the experiments. The students will also have the option to write me questions in their notebooks and the class parking lot. I will go over topics that are still unclear using as many examples as possible.
Exploring Acids & Bases
 Food Explorations Lab: Exploring Acids & Bases STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will use cabbage juice indicator to determine if two unknown samples are acids or
Food Explorations Lab: Exploring Acids & Bases STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will use cabbage juice indicator to determine if two unknown samples are acids or
MiSP CHEMICAL REACTIONS, L3 Teacher Guide. Introduction
 MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
The Chemistry of Color Tie Die
 Ages: 6 to 12 The Chemistry of Color Tie Die Contributor: Dr. Aaron Judy Couture, Laboratory of Atomic and Solid State Physics, Cornell University Main idea: What is an acid, and what is a base? How is
Ages: 6 to 12 The Chemistry of Color Tie Die Contributor: Dr. Aaron Judy Couture, Laboratory of Atomic and Solid State Physics, Cornell University Main idea: What is an acid, and what is a base? How is
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED)
 CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
Lesson 2. Color change
 Lesson 2 Color change T E A C H E R G U I D E Lesson summary Students meet marine chemist Sera Tuikabe, who is studying ocean acidification in the water surrounding the Republic of the Fiji Islands. Students
Lesson 2 Color change T E A C H E R G U I D E Lesson summary Students meet marine chemist Sera Tuikabe, who is studying ocean acidification in the water surrounding the Republic of the Fiji Islands. Students
Physical & Chemical PROPERTIES
 Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Post-Show. Chemistry. Periodic Table of the Elements. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Ocean Acidification in a Cup Materials
 Ocean Acidification in a Cup Ocean acidification is a problem that humans will have to deal with as we release more and more carbon dioxide into the atmosphere. This activity demonstrates how water can
Ocean Acidification in a Cup Ocean acidification is a problem that humans will have to deal with as we release more and more carbon dioxide into the atmosphere. This activity demonstrates how water can
MONDAY (12/12) TUESDAY (12/13) WEDNESDAY (12/14) THURSDAY (12/15) FRIDAY (12/16) Making Acid Rain (a lab) Quiz
 Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions
 Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Photosynthesis-Cellular Respiration Cycle
 Photosynthesis-Cellular Respiration Cycle Lesson Concept Link Photosynthesis and cellular respiration are reverse processes. Plants use photosynthesis to make food and release oxygen and plants and animals
Photosynthesis-Cellular Respiration Cycle Lesson Concept Link Photosynthesis and cellular respiration are reverse processes. Plants use photosynthesis to make food and release oxygen and plants and animals
Chapter 6, Lesson 9: Neutralizing Acids and Bases
 Chapter 6, Lesson 9: Neutralizing Acids and Bases Key Concepts ph is a measure of the concentration of H 3 O + ions in a solution. Adding an acid increases the concentration of H 3 O + ions in the solution.
Chapter 6, Lesson 9: Neutralizing Acids and Bases Key Concepts ph is a measure of the concentration of H 3 O + ions in a solution. Adding an acid increases the concentration of H 3 O + ions in the solution.
Author: Alan Moghaddam and Rachael Cunningham Title: phun with ph
 Author: Alan Moghaddam and Rachael Cunningham Title: phun with ph Synopsis The audience will be introduced to the main concepts of the ph scale. They will learn the definition of ph and the logarithmic
Author: Alan Moghaddam and Rachael Cunningham Title: phun with ph Synopsis The audience will be introduced to the main concepts of the ph scale. They will learn the definition of ph and the logarithmic
6TH GRADE UNIT 2 STUDY GUIDE
 6TH GRADE UNIT 2 STUDY GUIDE 6thgradeUnit2StudyGuide:StatesofMatter I. Vocabulary Indicate how students are going to demonstrate accountability for completing the correctives. Example Signature. Matter:
6TH GRADE UNIT 2 STUDY GUIDE 6thgradeUnit2StudyGuide:StatesofMatter I. Vocabulary Indicate how students are going to demonstrate accountability for completing the correctives. Example Signature. Matter:
Investigation 4: Fizz Quiz
 5 th Science Notebook Mixtures and Solutions Investigation 4 Investigation 4: Fizz Quiz Name: Big Question: How can matter be changed? Explain. 1 Alignment with New York State Science Standards & Performance
5 th Science Notebook Mixtures and Solutions Investigation 4 Investigation 4: Fizz Quiz Name: Big Question: How can matter be changed? Explain. 1 Alignment with New York State Science Standards & Performance
Pure Substances and Mixtures
 Pure Substances and Mixtures Unit 1 Lesson 4 Mix It Up! Make two mixtures in transparent containers with lids. 1. Find two transparent containers with tight lids. In one container, mix either salt or sugar
Pure Substances and Mixtures Unit 1 Lesson 4 Mix It Up! Make two mixtures in transparent containers with lids. 1. Find two transparent containers with tight lids. In one container, mix either salt or sugar
What Do You Think? Investigate GOALS
 Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Matter [6th grade] Digital Trinity. Trinity University. Anne Cowell Trinity University
![Matter [6th grade] Digital Trinity. Trinity University. Anne Cowell Trinity University Matter [6th grade] Digital Trinity. Trinity University. Anne Cowell Trinity University](/thumbs/78/76800104.jpg) Trinity University Digital Commons @ Trinity Understanding by Design: Complete Collection Understanding by Design 6-14-2006 Matter [6th grade] Anne Cowell Trinity University Follow this and additional
Trinity University Digital Commons @ Trinity Understanding by Design: Complete Collection Understanding by Design 6-14-2006 Matter [6th grade] Anne Cowell Trinity University Follow this and additional
CO 2. Lesson 1. Production of a gas
 Lesson 1 Production of a gas T E A C H E R G U I D E CO 2 Lesson summary Students meet volcanologist Victor Helguson, who is studying the gases released by volcanoes in Iceland. Students conduct a chemical
Lesson 1 Production of a gas T E A C H E R G U I D E CO 2 Lesson summary Students meet volcanologist Victor Helguson, who is studying the gases released by volcanoes in Iceland. Students conduct a chemical
Properties of Matter - Mass Unit
 3-5 Physical Science 3-5 Nature of Science Southern Nevada Regional Professional Development Program Properties of Matter - Mass Unit INTRODUCTION Children are often confused about matter and mass. This
3-5 Physical Science 3-5 Nature of Science Southern Nevada Regional Professional Development Program Properties of Matter - Mass Unit INTRODUCTION Children are often confused about matter and mass. This
Characteristics of Chemical Change
 Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
The grade 5 English science unit, Acids and Bases, meets the academic content standards set in the Korean curriculum, which state students should:
 This area addresses ph among different characteristics of solutions. It will be interesting for students to classify a variety of solutions into acids and bases by using the characteristics of the solutions.
This area addresses ph among different characteristics of solutions. It will be interesting for students to classify a variety of solutions into acids and bases by using the characteristics of the solutions.
LESSON 9: Ageless Apples ESTIMATED TIME Setup: 5 10 minutes Procedure: The effects can be observed over a 24-hour time period.
 LESSON 9: Ageless Apples ESTIMATED TIME Setup: 5 10 minutes Procedure: The effects can be observed over a 24-hour time period. DESCRIPTION Place apple slices into solutions with different levels of acidity
LESSON 9: Ageless Apples ESTIMATED TIME Setup: 5 10 minutes Procedure: The effects can be observed over a 24-hour time period. DESCRIPTION Place apple slices into solutions with different levels of acidity
Matter Lesson 2. Learning Goal 3: I can describe the differences between physical and chemical changes of matter.
 Matter Lesson 2 Learning Goal 2: I can describe the differences between intensive physical properties, extensive physical properties, and chemical properties of matter. Learning Goal 3: I can describe
Matter Lesson 2 Learning Goal 2: I can describe the differences between intensive physical properties, extensive physical properties, and chemical properties of matter. Learning Goal 3: I can describe
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
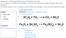 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Sc1 Collecting & using evidence, making observations, evaluating & presenting results, making fair tests and comparisons, identifying patterns
 A collection of experiments in which pupils carefully study and analyse evidence to solve a crime. Could be used as an off-timetable activity for a Science Week etc. Curriculum Links: Sc1 Collecting &
A collection of experiments in which pupils carefully study and analyse evidence to solve a crime. Could be used as an off-timetable activity for a Science Week etc. Curriculum Links: Sc1 Collecting &
Energy Transformations Activities Students engage in different activities to observe energy changing form!
 Energy Transformations Activities Students engage in different activities to observe energy changing form! Name: Energy Labs and Activities Activity 1: Static Electricity Glue this side down Procedure:
Energy Transformations Activities Students engage in different activities to observe energy changing form! Name: Energy Labs and Activities Activity 1: Static Electricity Glue this side down Procedure:
Chemical Matrix of Acids and Bases
 Chemical Matrix of Acids and Bases Overview:If you love the idea of mixing up chemicals and dream of having your own mad science lab, this one is for you. You are going to mix up solids and liquids in
Chemical Matrix of Acids and Bases Overview:If you love the idea of mixing up chemicals and dream of having your own mad science lab, this one is for you. You are going to mix up solids and liquids in
What s the Matter? An experiment demonstrating the 3 stages of matter. Michelle Garner Laura Williams
 What s the Matter? An experiment demonstrating the 3 stages of matter. Michelle Garner Laura Williams Introduction to Physical Science Ms. C. Dianne Phillips, Instructor Introduction There are three states
What s the Matter? An experiment demonstrating the 3 stages of matter. Michelle Garner Laura Williams Introduction to Physical Science Ms. C. Dianne Phillips, Instructor Introduction There are three states
By All INdICATIONS (2 Hours)
 By All INdICATIONS (2 Hours) Addresses NGSS Level of Difficulty: 5 Grade Range: 6-8 OVERVIEW In this activity, students create an acid-base indicator using red cabbage extract. Students then use this indicator
By All INdICATIONS (2 Hours) Addresses NGSS Level of Difficulty: 5 Grade Range: 6-8 OVERVIEW In this activity, students create an acid-base indicator using red cabbage extract. Students then use this indicator
TEACHER S NOTES FOR UNIT 1: KAI/OCEAN (SCIENTIFIC METHOD PLUS)
 TEACHER S NOTES FOR UNIT 1: KAI/OCEAN (SCIENTIFIC METHOD PLUS) After introducing/reviewing the scientific method (see 2 handouts), try this sequence of lessons to allow students to experiment themselves,
TEACHER S NOTES FOR UNIT 1: KAI/OCEAN (SCIENTIFIC METHOD PLUS) After introducing/reviewing the scientific method (see 2 handouts), try this sequence of lessons to allow students to experiment themselves,
Conservation of Mass in Chemical Reactions Lab. Experiment Question: What happens to the total mass of substances when a chemical reaction occurs?
 Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
UC Irvine FOCUS! 5 E Lesson Plan Title: Acid/Base-pH Lab Grade Level and Course: 8 th grade Physical Science, Grades 9-12 Chemistry Materials:
 UC Irvine FOCUS! 5 E Lesson Plan Title: Acid/Base-pH Lab Grade Level and Course: 8 th grade Physical Science, Grades 9-12 Chemistry Materials: Detergent-quart, shampoo- quart Lemon-juice-quart Vinegar
UC Irvine FOCUS! 5 E Lesson Plan Title: Acid/Base-pH Lab Grade Level and Course: 8 th grade Physical Science, Grades 9-12 Chemistry Materials: Detergent-quart, shampoo- quart Lemon-juice-quart Vinegar
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic
 Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Saturday Science Lesson Plan Fall 2008
 Saturday Science Lesson Plan Fall 2008 LEARNING OBJECTIVES STANDARDS 1.1.1 Observe, describe, draw, and sort objects carefully to learn about them. 1.2.6 Describe and compare objects in terms of number,
Saturday Science Lesson Plan Fall 2008 LEARNING OBJECTIVES STANDARDS 1.1.1 Observe, describe, draw, and sort objects carefully to learn about them. 1.2.6 Describe and compare objects in terms of number,
Follow the instructions to determine if your sample is metamorphic, sedimentary or igneous rock.
 To gather some appreciation of our world, especially our rock world, we are going to gather an assortment of rocks from our campus. You will use a rock key to classify some of the samples we collect. Procedures:
To gather some appreciation of our world, especially our rock world, we are going to gather an assortment of rocks from our campus. You will use a rock key to classify some of the samples we collect. Procedures:
2/22/2019 NEW UNIT! Chemical Interactions. Atomic Basics #19
 NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
Lab: Detecting ph of Commonly Used Acids and Bases
 Lab: Detecting ph of Commonly Used Acids and Bases FOR THE TEACHER Summary In this lab, students will use their knowledge of acids and bases to determine the acidity and basicity of every day items by
Lab: Detecting ph of Commonly Used Acids and Bases FOR THE TEACHER Summary In this lab, students will use their knowledge of acids and bases to determine the acidity and basicity of every day items by
4 th Grade Science Unit: Matter Matters Conservation of Matter Unit Snapshot
 4 th Grade Science Unit: Matter Matters Conservation of Matter Unit Snapshot Grade Level: 4 Topic: Electricity, Heat and Matter Duration: 14 days Summary This series of activities is for students to develop
4 th Grade Science Unit: Matter Matters Conservation of Matter Unit Snapshot Grade Level: 4 Topic: Electricity, Heat and Matter Duration: 14 days Summary This series of activities is for students to develop
Standards 8.5.c. I know chemical reactions usually liberate or absorbs heat.
 6 March 2013 Give an example of a physical change and a chemical change, and then describe how they are different from the other. Explain your answer in 2-3 sentences. Standards 8.5.c. I know chemical
6 March 2013 Give an example of a physical change and a chemical change, and then describe how they are different from the other. Explain your answer in 2-3 sentences. Standards 8.5.c. I know chemical
Lesson 4. Temperature change
 Lesson 4 Temperature change T E A C H E R G U I D E Lesson summary Students meet scientist Jason Williams, an industrial chemist who designs the materials and processes for making solar cells. He explains
Lesson 4 Temperature change T E A C H E R G U I D E Lesson summary Students meet scientist Jason Williams, an industrial chemist who designs the materials and processes for making solar cells. He explains
Activity 6.5 From gas to liquid to solid
 Activity 6.5 This activity is an extension of Activity 6.4a in which ice is used to make a container cold. As in Activity 6.4a, this activity will work only with sufficient water vapor in the air. Here,
Activity 6.5 This activity is an extension of Activity 6.4a in which ice is used to make a container cold. As in Activity 6.4a, this activity will work only with sufficient water vapor in the air. Here,
Mixtures, Solutions, and Suspensions
 Purpose To explore how mixtures, solutions, and suspensions form by combining and then attempting to separate various materials. Process Skills Observe, measure, predict, collect data, interpret data,
Purpose To explore how mixtures, solutions, and suspensions form by combining and then attempting to separate various materials. Process Skills Observe, measure, predict, collect data, interpret data,
Chemical Change. Day 1: 70 minutes; Day 2: minutes
 Chemical Change Lesson Concept When one substance interacts with another substance, a chemical change may occur. There are five indicators that a chemical change has occurred: gas production (bubbles),
Chemical Change Lesson Concept When one substance interacts with another substance, a chemical change may occur. There are five indicators that a chemical change has occurred: gas production (bubbles),
5th Grade. Slide 1 / 67. Slide 2 / 67. Slide 3 / 67. Matter and Its Interactions. Table of Contents: Matter and Its Interactions
 Slide 1 / 67 Slide 2 / 67 5th Grade Matter and Its Interactions 2015-11-02 www.njctl.org Table of Contents: Matter and Its Interactions Slide 3 / 67 Click on the topic to go to that section What Is Matter?
Slide 1 / 67 Slide 2 / 67 5th Grade Matter and Its Interactions 2015-11-02 www.njctl.org Table of Contents: Matter and Its Interactions Slide 3 / 67 Click on the topic to go to that section What Is Matter?
Name. Pre-Test. Directions: Circle the T if a statement is true or F if it is false (F). 1. Matter always stays the same.
 1 Pre-est Directions: Circle the if a statement is true or if it is false (). 1. Matter always stays the same. 2. A physical change is a change in size, shape or phase of matter, without the matter changing
1 Pre-est Directions: Circle the if a statement is true or if it is false (). 1. Matter always stays the same. 2. A physical change is a change in size, shape or phase of matter, without the matter changing
LESSON 1. Chemical Reactions. Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions.
 LESSON 1 Chemical Reactions Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions. By the end of this lesson... you will be able to explain ways
LESSON 1 Chemical Reactions Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions. By the end of this lesson... you will be able to explain ways
Partnerships Implementing Engineering Education Worcester Polytechnic Institute Worcester Public Schools Supported by: National Science Foundation
 Partnerships Implementing Engineering Education Chemical Engineering: K.L.1 Volcanoes Grade Level Sessions Seasonality Instructional Mode(s) Team Size WPS Benchmarks MA Frameworks Key Words K Part 1: 30
Partnerships Implementing Engineering Education Chemical Engineering: K.L.1 Volcanoes Grade Level Sessions Seasonality Instructional Mode(s) Team Size WPS Benchmarks MA Frameworks Key Words K Part 1: 30
States of Matter in Food
 Science Unit: Lesson 13: Matter States of Matter in Food Summary: Science skills: In this lesson, students explore the concept of state change, using various food and drink, in three activities: (1) Students
Science Unit: Lesson 13: Matter States of Matter in Food Summary: Science skills: In this lesson, students explore the concept of state change, using various food and drink, in three activities: (1) Students
Bay Area Scientists in Schools Presentation Plan
 Bay Area Scientists in Schools Presentation Plan Lesson Name Presenter(s) The Water Cycle UC Berkeley PhD students Grade Level 1 Standards Connection(s) Earth Sciences, physics sciences CA Science Content
Bay Area Scientists in Schools Presentation Plan Lesson Name Presenter(s) The Water Cycle UC Berkeley PhD students Grade Level 1 Standards Connection(s) Earth Sciences, physics sciences CA Science Content
Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic
 Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic Name Date DEMONSTRATION 1. Your teacher blew through a straw into a universal indicator solution until it changed color. Did
Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic Name Date DEMONSTRATION 1. Your teacher blew through a straw into a universal indicator solution until it changed color. Did
Community in the Classroom Presentation Plan
 Community in the Classroom Presentation Plan Lesson Name How Can You Tell One Clear Gas From Another? Presenter(s) Michael Grass Grade Level 5th Standards Connection(s) Abstract: Many gases are clear and
Community in the Classroom Presentation Plan Lesson Name How Can You Tell One Clear Gas From Another? Presenter(s) Michael Grass Grade Level 5th Standards Connection(s) Abstract: Many gases are clear and
OBJECTIVES: By the end of class, students will be able to
 7 th Grade Science Unit: Matter and Periodic Table Lesson: Matter 11 Law of Conservation of Mass Review Name: Date: Monday, December 12, 2016 Homeroom: OBJECTIVES: By the end of class, students will be
7 th Grade Science Unit: Matter and Periodic Table Lesson: Matter 11 Law of Conservation of Mass Review Name: Date: Monday, December 12, 2016 Homeroom: OBJECTIVES: By the end of class, students will be
Virtual Library Lesson: Oobleck, Gloop, and Glurch
 Oobleck, Gloop, and Glurch Lesson Overview Throughout this lesson, students will use inquiry skills to identify states of matter, describe physical properties, and modify the recipe to change physical
Oobleck, Gloop, and Glurch Lesson Overview Throughout this lesson, students will use inquiry skills to identify states of matter, describe physical properties, and modify the recipe to change physical
Separating the Mixture
 Separating the Mixture 40- to 1 50-minute session ACTIVITY OVERVIEW I N V E S T 5 I O N I G AT Students perform their procedures written in Activity 3, A Plan to Separate the Mixture, to physically separate
Separating the Mixture 40- to 1 50-minute session ACTIVITY OVERVIEW I N V E S T 5 I O N I G AT Students perform their procedures written in Activity 3, A Plan to Separate the Mixture, to physically separate
Chemical Reaction Lab Bagged Chemical Reactions
 Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
5 th Grade Lesson Plan: Matter and Chemical Reactions
 5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
Most people think of volcanoes as destructive. The high temperature
 37 Volcanic Landforms M O D E L I N G Most people think of volcanoes as destructive. The high temperature of volcanic lava can burn almost everything in its path. Volcanoes also release large amounts of
37 Volcanic Landforms M O D E L I N G Most people think of volcanoes as destructive. The high temperature of volcanic lava can burn almost everything in its path. Volcanoes also release large amounts of
More Chemical Changes
 Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
SPOOKY SCIENCE GUIDE
 GUIDE TABLE OF CONTENTS PACING GUIDE 2 SCIENCE SAFETY 3 SELF CARVING PUMPKIN 4 GHOST BUBBLES 7 SPOOKY CLOUD 9 TEA BAG GHOST 11 BLOODY HAND PRINT 13 QUIZ 15 SPOOKY SCIENCE GUIDE l 1 PACING GUIDE TEACHER
GUIDE TABLE OF CONTENTS PACING GUIDE 2 SCIENCE SAFETY 3 SELF CARVING PUMPKIN 4 GHOST BUBBLES 7 SPOOKY CLOUD 9 TEA BAG GHOST 11 BLOODY HAND PRINT 13 QUIZ 15 SPOOKY SCIENCE GUIDE l 1 PACING GUIDE TEACHER
Predict the effect of increased competition for abiotic and biotic resources on a food web. colored pencils graph paper ruler
 Edit File QUICK LAB Effect of Abiotic and Biotic Factors No organism exists in isolation. Organisms depend on and compete for the abiotic, or non-living, factors in its environment. For example, organisms
Edit File QUICK LAB Effect of Abiotic and Biotic Factors No organism exists in isolation. Organisms depend on and compete for the abiotic, or non-living, factors in its environment. For example, organisms
5 th GRADE PHYSICAL AND CHEMICAL TESTS
 5 th GRADE PHYSICAL AND CHEMICAL TESTS Summary: Students investigate 5 unknown white powders. They gather clues by observing the physical and chemical changes of the powders. At the end of the activity,
5 th GRADE PHYSICAL AND CHEMICAL TESTS Summary: Students investigate 5 unknown white powders. They gather clues by observing the physical and chemical changes of the powders. At the end of the activity,
Moles Lab Activity 1: PCU (Popcorn Counting Units)
 Moles Lab Activity 1: PCU (Popcorn Counting Units) Materials: A container of each of the following: Popcorn kernels Another type of beans A large unopened bag of popcorn Kernels Balance Safety goggles
Moles Lab Activity 1: PCU (Popcorn Counting Units) Materials: A container of each of the following: Popcorn kernels Another type of beans A large unopened bag of popcorn Kernels Balance Safety goggles
Unit 6M.3: Changing materials
 Unit 6M.3: Adding materials to water. Chemical reactions Skill you will use: Classifying Observing Predicting By the end of this unit you should: Know that when substances are added to water, some will
Unit 6M.3: Adding materials to water. Chemical reactions Skill you will use: Classifying Observing Predicting By the end of this unit you should: Know that when substances are added to water, some will
Copyright 2011, 2013 Gravitas Publications, Inc.
 Illustrations: Rebecca W. Keller, PhD Copyright 2011, 2013 Gravitas Publications, Inc. All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted,
Illustrations: Rebecca W. Keller, PhD Copyright 2011, 2013 Gravitas Publications, Inc. All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted,
Chapter 6, Lesson 1: What is a Chemical Reaction?
 Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Chapter 5, Lesson 5 Using Dissolving to Identify an Unknown
 Chapter 5, Lesson 5 Using Dissolving to Identify an Unknown Key Concepts Different substances are made from different atoms, ions, or molecules, which interact with water in different ways. Since dissolving
Chapter 5, Lesson 5 Using Dissolving to Identify an Unknown Key Concepts Different substances are made from different atoms, ions, or molecules, which interact with water in different ways. Since dissolving
Name Date Class. Chapter 19 Acids, Bases, and Salts EXPERIMENT. ESTIMATION OF ph PURPOSE BACKGROUND MATERIALS (PER PAIR)
 Chapter 19 Acids, Bases, and Salts EXPERIMENT ESTIMATION OF Text Reference Sections 19.2 PURPOSE To estimate the of solutions by using acid base indicators. BACKGROUND If you are interested in gardening,
Chapter 19 Acids, Bases, and Salts EXPERIMENT ESTIMATION OF Text Reference Sections 19.2 PURPOSE To estimate the of solutions by using acid base indicators. BACKGROUND If you are interested in gardening,
Describing Matter Laboratory
 Describing Matter Laboratory Name: 5 th Grade PSI Science Score: / 5 Experiment Question: How is matter identified? What are the observable properties of matter? Hypothesis Starters: 1. Your eyes help
Describing Matter Laboratory Name: 5 th Grade PSI Science Score: / 5 Experiment Question: How is matter identified? What are the observable properties of matter? Hypothesis Starters: 1. Your eyes help
Science Grade 01 Unit 07 Exemplar Lesson 02: Investigating the Moon, the Stars, and the Sky
 Grade 1 Unit: 07 Lesson: 02 Suggested Duration: 5 days Grade 01 Unit 07 Exemplar Lesson 02: Investigating the Moon, the Stars, and the Sky This lesson is one approach to teaching the State Standards associated
Grade 1 Unit: 07 Lesson: 02 Suggested Duration: 5 days Grade 01 Unit 07 Exemplar Lesson 02: Investigating the Moon, the Stars, and the Sky This lesson is one approach to teaching the State Standards associated
Cool Chemistry. Middle School TEKS. Vocabulary
 Cool Chemistry Middle School TEKS Sixth Grade: 6.5A, 6.5B, 6.5C, 6.5D Seventh Grade: 7.6A, 7.6B, 7.6C Eighth Grade: 8.5A, 8.5B, 8.5C, 8.5E Vocabulary chemical change, chemical reaction, cirrhosis of the
Cool Chemistry Middle School TEKS Sixth Grade: 6.5A, 6.5B, 6.5C, 6.5D Seventh Grade: 7.6A, 7.6B, 7.6C Eighth Grade: 8.5A, 8.5B, 8.5C, 8.5E Vocabulary chemical change, chemical reaction, cirrhosis of the
Problem How can I determine if different propellants will make a rocket travel faster?
 National Aeronautics and Space Administration SPEED IT UP Student Section Student Name Lesson Objective In this lesson, you will simulate how different propellant systems affect the velocity, or speed
National Aeronautics and Space Administration SPEED IT UP Student Section Student Name Lesson Objective In this lesson, you will simulate how different propellant systems affect the velocity, or speed
Introduction. Objectives
 Experiment: Acids, Bases, and Buffers * Introduction Many common household solutions contain acids and bases. Acid-base indicators, such as litmus and red cabbage juice, turn different colors in acidic
Experiment: Acids, Bases, and Buffers * Introduction Many common household solutions contain acids and bases. Acid-base indicators, such as litmus and red cabbage juice, turn different colors in acidic
Lesson 1 Matter and Its Properties
 Lesson 1 Student Labs and Activities Page Launch Lab 8 Content Vocabulary 9 Lesson Outline 10 MiniLab 12 Content Practice A 13 Content Practice B 14 Math Skills 15 School to Home 16 Key Concept Builders
Lesson 1 Student Labs and Activities Page Launch Lab 8 Content Vocabulary 9 Lesson Outline 10 MiniLab 12 Content Practice A 13 Content Practice B 14 Math Skills 15 School to Home 16 Key Concept Builders
Lab: Cabbages in Chemistry 3pts ec printing in COLOR / 2pts B&W
 Lab: Cabbages in Chemistry 3pts ec printing in COLOR / 2pts B&W Telltale Colors 1. In the mixing tray, place 5 drops of the chemical in 13 compartments 2. DIP test the ph paper & record the ph measurement
Lab: Cabbages in Chemistry 3pts ec printing in COLOR / 2pts B&W Telltale Colors 1. In the mixing tray, place 5 drops of the chemical in 13 compartments 2. DIP test the ph paper & record the ph measurement
Name Period Date. Lab 1: Mass of Ice Materials: beaker, ice and balance.
 Name Period Date Testing the Law of Conservation of MASS! Introduction: Does mass change in a chemical or physical reaction? In this series of experiments you will find the answer to this question. Lab
Name Period Date Testing the Law of Conservation of MASS! Introduction: Does mass change in a chemical or physical reaction? In this series of experiments you will find the answer to this question. Lab
Physical and Chemical Changes Or How Do You Know When You ve Made Something New?
 Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Science Supply List. General Science II Glynlyon, Inc.
 Science Supply List General Science II 2016 Glynlyon, Inc. Table of Contents UNIT 1: SCIENCE AND SOCIETY... 1 UNIT 2: STRUCTURE OF MATTER (PART 1)... 1 UNIT 3: STRUCTURE OF MATTER (PART 2)... 2 UNIT 4:
Science Supply List General Science II 2016 Glynlyon, Inc. Table of Contents UNIT 1: SCIENCE AND SOCIETY... 1 UNIT 2: STRUCTURE OF MATTER (PART 1)... 1 UNIT 3: STRUCTURE OF MATTER (PART 2)... 2 UNIT 4:
This tossed salad is an example of a. Matter Unit TEST. Name: Class: Date: ID: A
 Name: Class: Date: ID: A Matter Unit TEST Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Why is lemonade a solution? a. It is one substance whose kind
Name: Class: Date: ID: A Matter Unit TEST Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Why is lemonade a solution? a. It is one substance whose kind
Section 1: Four States of Matter (pages 60-67)
 Name: Sci Number: Period: Parent Signature: My little book of: Word: Pg found States of matter Solid Four States of Matter: Section 1 definitions: (pg60) My definition Chp 3 Draw/paste examples of all
Name: Sci Number: Period: Parent Signature: My little book of: Word: Pg found States of matter Solid Four States of Matter: Section 1 definitions: (pg60) My definition Chp 3 Draw/paste examples of all
1. Read the following article and fill in the vocabulary crossword. An Atom Apart: by Leslie Cargile
 Grade 6 Science Summer Work International School of Arts and Sciences ISAS 2012 2013 Name: 1. Read the following article and fill in the vocabulary crossword. An Atom Apart: by Leslie Cargile Have you
Grade 6 Science Summer Work International School of Arts and Sciences ISAS 2012 2013 Name: 1. Read the following article and fill in the vocabulary crossword. An Atom Apart: by Leslie Cargile Have you
General Science II General Science II
 2017-2018 General Science II General Science II 2017 Glynlyon, Inc. Table of Contents UNIT 1: SCIENCE AND SOCIETY... 1 UNIT 2: STRUCTURE OF MATTER (PART 1)... 1 UNIT 3: STRUCTURE OF MATTER (PART 2)...
2017-2018 General Science II General Science II 2017 Glynlyon, Inc. Table of Contents UNIT 1: SCIENCE AND SOCIETY... 1 UNIT 2: STRUCTURE OF MATTER (PART 1)... 1 UNIT 3: STRUCTURE OF MATTER (PART 2)...
Chemical Reactions. Teachers Guide.
 Chemical Reactions. Teachers Guide. Introduction: Almost everything you will ever see in this universe is made of atoms. Each different type of atom is known as a chemical element there are currently 118
Chemical Reactions. Teachers Guide. Introduction: Almost everything you will ever see in this universe is made of atoms. Each different type of atom is known as a chemical element there are currently 118
LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EXPERIMENTS
 LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EPERIMENTS PURPOSE: To practice the scientific method of observation, hypothesis, experimentation, & conclusion. SAFETY CONCERNS: Always wear
LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EPERIMENTS PURPOSE: To practice the scientific method of observation, hypothesis, experimentation, & conclusion. SAFETY CONCERNS: Always wear
Solids, Liquids, and Gases: A First Look Teacher s Guide
 Teacher s Guide Grade Level: K-2 Curriculum Focus: Science Lesson Duration: Four class periods Program Description Solids, Liquids, and Gases: A First Look In this program students are encouraged to take
Teacher s Guide Grade Level: K-2 Curriculum Focus: Science Lesson Duration: Four class periods Program Description Solids, Liquids, and Gases: A First Look In this program students are encouraged to take
Liquid X Lab. Number of Drops Before Spilling Trial 1 Trial 2 Trial 3. Write a conclusion: How do your results for Liquid X compare to water?
 Names Block Date BIG QUESTION: Liquid X Lab Station 1 Surface Tension, Cohesion, and Adhesion Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette
Names Block Date BIG QUESTION: Liquid X Lab Station 1 Surface Tension, Cohesion, and Adhesion Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette
Matter and Its Changes
 Lesson 2 Matter and Its Changes 6.NS.3, 6.NS.5, 6.NS.6, 6.NS.7, 6.NS.10, 6.1.1, 6.1.2, 6.1.3 Skim or scan the heading, boldfaced words, and pictures in the lesson. Identify or predict three facts you will
Lesson 2 Matter and Its Changes 6.NS.3, 6.NS.5, 6.NS.6, 6.NS.7, 6.NS.10, 6.1.1, 6.1.2, 6.1.3 Skim or scan the heading, boldfaced words, and pictures in the lesson. Identify or predict three facts you will
States of Matter: A Solid Lesson where Liquids Can be a Gas!
 TEACHER GUIDE STATES OF MATTER 60 Minute Physical Science Lesson Science- to- Go! Program Grades: 1-3 States of Matter: A Solid Lesson where Liquids Can be a Gas! Description Your classroom will be converted
TEACHER GUIDE STATES OF MATTER 60 Minute Physical Science Lesson Science- to- Go! Program Grades: 1-3 States of Matter: A Solid Lesson where Liquids Can be a Gas! Description Your classroom will be converted
Chapter 1, Lesson 3: The Ups and Downs of Thermometers
 Chapter 1, Lesson 3: The Ups and Downs of Thermometers Key Concepts The way a thermometer works is an example of heating and cooling a liquid. When heated, the molecules of the liquid in the thermometer
Chapter 1, Lesson 3: The Ups and Downs of Thermometers Key Concepts The way a thermometer works is an example of heating and cooling a liquid. When heated, the molecules of the liquid in the thermometer
6.2 The ph Scale and Indicators
 6.2 The ph Scale and Indicators In May 2001, 49 people who attended a dance festival in Dauphin, Manitoba, became sick within a week. The evidence suggests that the source of the illness was the hotel
6.2 The ph Scale and Indicators In May 2001, 49 people who attended a dance festival in Dauphin, Manitoba, became sick within a week. The evidence suggests that the source of the illness was the hotel
Mixing Acids and Bases
 Mixing Acids and Bases Acids and bases are two different kinds of substances. They each produce different kinds of charged particles, called ions, when they dissolve. An acid produces positive hydrogen
Mixing Acids and Bases Acids and bases are two different kinds of substances. They each produce different kinds of charged particles, called ions, when they dissolve. An acid produces positive hydrogen
KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator
 KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator By Darby Sloss and Marianne Smith Edited by Anne Starace Abstract Chemistry is an important part of our lives. Kitchen Chemistry
KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator By Darby Sloss and Marianne Smith Edited by Anne Starace Abstract Chemistry is an important part of our lives. Kitchen Chemistry
Bust-A-Myth Particles of Matter and Heat Transfer. Students will use the online Research Gadget and experimentation to bust or confirm the myth:
 ACTIVITY Bust-A-Myth Particles of Matter and Heat Transfer Approximate Classroom Time: 100-150 minutes Students will use the online Research Gadget and experimentation to bust or confirm the myth: A winter
ACTIVITY Bust-A-Myth Particles of Matter and Heat Transfer Approximate Classroom Time: 100-150 minutes Students will use the online Research Gadget and experimentation to bust or confirm the myth: A winter
Bay Area Scientists in Schools Presentation Plan
 Bay Area Scientists in Schools Presentation Plan Lesson Name: Chemistry Magic! How do we know if there is a chemical change or only a state change? Presenter: Melissa Juedemann Grade Level 5 th _Standards
Bay Area Scientists in Schools Presentation Plan Lesson Name: Chemistry Magic! How do we know if there is a chemical change or only a state change? Presenter: Melissa Juedemann Grade Level 5 th _Standards
INTRODUCTION TO LESSON CLUSTER 5
 INTRODUCTION TO LESSON CLUSTER 5 EXPLAINING DISSOLVING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain dissolving of solids in liquids in terms of molecules. Lesson
INTRODUCTION TO LESSON CLUSTER 5 EXPLAINING DISSOLVING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain dissolving of solids in liquids in terms of molecules. Lesson
Liquid X Lab. Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties?
 Names Block Date Liquid X Lab Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette to carefully place drops of each liquid
Names Block Date Liquid X Lab Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette to carefully place drops of each liquid
Recommended Resources: The following resources may be useful in teaching this
 Unit C: Plant Physiology Lesson 1: Understanding Plant Physiology Student Learning Objectives: Instruction in this lesson should result in students achieving the following objectives: 1. Describe the process
Unit C: Plant Physiology Lesson 1: Understanding Plant Physiology Student Learning Objectives: Instruction in this lesson should result in students achieving the following objectives: 1. Describe the process
Bubbles and Colors and Smells Oh My! A lesson about physical and chemical changes By Glenda Kitts, Arlene Cuellar, and Maritza Aquino
 Glenda Kitts, Arlene Cuellar, Maritza Aquino Last Updated on 9/17/2017 Bubbles and Colors and Smells Oh My! A lesson about physical and chemical changes By Glenda Kitts, Arlene Cuellar, and Maritza Aquino
Glenda Kitts, Arlene Cuellar, Maritza Aquino Last Updated on 9/17/2017 Bubbles and Colors and Smells Oh My! A lesson about physical and chemical changes By Glenda Kitts, Arlene Cuellar, and Maritza Aquino
OBJECTIVES: By the end of class, students will be able to. SWBAT identify and describe physical and chemical changes of matter.
 7 th Grade Science Name: Unit: Matter and Periodic Table Lesson: Matter 4 Chemical and Physical Changes Part 2 Date: Thursday, December 1, 2016 Homeroom: OBJECTIVES: By the end of class, students will
7 th Grade Science Name: Unit: Matter and Periodic Table Lesson: Matter 4 Chemical and Physical Changes Part 2 Date: Thursday, December 1, 2016 Homeroom: OBJECTIVES: By the end of class, students will
Geology, Part 2: What Is the Mystery Rock?
 Geology, Part 2: What Is the Mystery Rock? You and your partner will be given 14 rocks to identify. Using the chart provided, follow the steps by observing your rocks closely and using the tools available.
Geology, Part 2: What Is the Mystery Rock? You and your partner will be given 14 rocks to identify. Using the chart provided, follow the steps by observing your rocks closely and using the tools available.
