The E3 SUMO ligase AtSIZ1 functions in seed germination in Arabidopsis
|
|
|
- Melissa Hamilton
- 5 years ago
- Views:
Transcription
1 Physiologia Plantarum 158: ISSN The E3 SUMO ligase AtSIZ1 functions in seed germination in Arabidopsis Sung-Il Kim a,junsookwak a, Jong Tae Song b and Hak Soo Seo a,c,* a Department of Plant Science, Research Institute for Agriculture and Life Sciences, and Plant Genomics and Breeding Institute, Seoul National University, Seoul , Korea b School of Applied Biosciences, Kyungpook National University, Daegu , Korea c Bio-MAX Institute, Seoul National University, Seoul , Korea Correspondence *Corresponding author, seohs@snu.ac.kr Received 17 September 2015; revised 16 February 2016 doi: /ppl Seed germination is an important stage in the lifecycle of a plant because it determines subsequent vegetative growth and reproduction. Here, we show that the E3 SUMO ligase AtSIZ1 regulates seed dormancy and germination. The germination rates of the siz1 mutants were less than 50%, even after a short period of ripening. However, their germination rates increased to wild-type levels after cold stratification or long periods of ripening. In addition, exogenous gibberellin (GA) application improved the germination rates of the siz1 mutants to the wild-type level. In transgenic plants, suppression of AtSIZ1 caused rapid post-translational decay of SLEEPY1 (SLY1), a positive regulator of GA signaling, during germination, and inducible AtSIZ1 overexpression led to increased SLY1 levels. In addition, overexpressing wild-type SLY1 in transgenic sly1 mutants increased their germination ratios to wild-type levels, whereas the germination ratio of transgenic sly1 mutants overexpressing msly1 was similar to that of sly1. The germination ratios of siz1 mutant seeds in immature developing siliques were much lower than those of the wild-type. Moreover, SLY1 and DELAY OF GERMINATION 1 (DOG1) transcript levels were reduced in the siz1 mutants, whereas the transcript levels of DELLAand ABSCISIC ACID INSENSITIVE 3 (ABI3) were higher than those of the wild-type. Taken together, these results indicate that the reduced germination of the siz1 mutants results from impaired GA signaling due to low SLY1 levels and activity, as well as hyperdormancy due to high levels of expression of dormancy-related genes including DOG1. Abbreviations ABA, abscisic acid; ABA2, ABSISIC ACID 2; ABI3, ABA INSENSITIVE 3; ABI5, ABA INSENSITIVE 5; ACC, 1-aminocyclopropane-1-carboxylic acid; ACO, ACC oxidase; CMT3, CHROMOMETHYLASE 3; CYP707A1 and CYP707A2, abscisic acid 8 -hydroxylases; DOG1, DELAY OF GERMINATION 1; FLC, FLOWERING LOCUS C; GA, gibberellic acid; GA2ox2, GIBBERELLIN 2-OXIDASE 2; GA3ox1, GIBBERELLIN 3-OXIDASE 1; GA3ox2, GIBBERELLIN 3-OXIDASE 2; GAI, GA INSENSITIVE; GID1A, GA INSENSITIVE DWARF 1A; ICE1, INDUCER OF CBF EXPRESSION 1; LDL1, LYSINESPECIFIC DEMETHYLASE LIKE 1; LDL2, LYSINESPECIFIC DEMETHYLASE LIKE 2; NCED6/9, 9-cis-epoxycarotenoid dioxygenase 6/9; NIA1 and NIA2, nitrate reductase 1 and 2; OPDA, 12-oxophytodienoic acid; qrt-pcr, quantitative reverse transcription polymerase chain reaction; RGA, REPRESSOR OF ga1-3; RGL1, RGA-LIKE 1; RGL2, RGA-LIKE 2; RGL3, RGA-LIKE 3; SCF, Skp, Cullin, F-box; SLY1, SLEEPY1; SLY2, SLEEPY2; SPY, SPINDLY; SUMO, small ubiquitin-related modifier; VP1, Viviparous The Authors. Physiologia Plantarum published by John Wiley & Sons Ltd on behalf of Scandinavian Plant Physiology Society. This is an open access article under the terms of the Creative Commons Attribution-NonCommercial-NoDerivs License, which permits use and distribution in any medium, provided the original work is properly cited, the use is non-commercial and no modifications or adaptations are made.
2 Introduction Plants have evolved the ability to undergo seed dormancy, a major factor preventing germination of viable seeds when conditions are temporarily favorable for germination during an unfavorable season. The induction of seed dormancy during the maturation stage and its release in seeds in a dry state after a certain period of time ( after-ripening ) or after a short period of imbibition at low temperatures ( stratification ) are widespread phenomena observed in diverse species of seed plants (Ali-Rachedi et al. 2004, Yamauchi et al. 2004, Bewley et al. 2013). Seed dormancy controls germination and plays a critical role in regulating the beginning of the lifecycle of a plant. Seed germination is the first stage of plant development. The cycle of seed germination begins with imbibition of the dry seed and ends with protrusion of the radicle (Bewley 1997, Bewley et al. 2003); this process greatly influences subsequent plant growth and yield. Major internal factors in the seed affecting germination include hormones such as gibberellin (GA), abscisic acid (ABA), ethylene and auxin. Among these, ABA and GA are key regulators of this process, although they have opposite functions during germination (Holdsworth et al. 2008, Nambara et al. 2010). External factors such as light, temperature, soil water content, and nutrient availability also affect germination through their effects on metabolic pathways (Holdsworth et al. 2008). Hormonal regulation of seed dormancy, including the induction and maintenance of seed dormancy by ABA and dormancy release by GA, has been observed in many species. Thus, dormancy and germination have been intensively studied using genes involved in GA and ABA biosynthesis and other processes, revealing the antagonistic functions of these two hormones in seed dormancy and germination. For example, loss-of-function mutants of ABA biosynthesis genes including ABSCISIC ACID 2 (ABA2), ABI3 and NINE-CIS-EPOXYCAROTENOID DIOXYGENASE 6/9 (NCED6/9) show reduced seed dormancy (Koornneef et al. 1982, Giraudat et al. 1992, Leon-Kloosterziel et al. 1996, Lefebvre et al. 2006, Okamoto et al. 2006), whereas loss-of-function mutants of ABA catabolism genes such as CYP707A1 and CYP707A2 display enhanced seed dormancy (Kushiro et al. 2004, Okamoto et al. 2006, Finkelstein et al. 2008, Holdsworth et al. 2008). Quantitative trait loci analysis showed that DOG1 is a master regulator of seed dormancy (Bentsink et al. 2006). In addition, DOG1 determines the time required for dormancy release in freshly harvested seeds (Nakabayashi et al. 2012). Epigenetic studies have revealed that the histone demethylases LDL1 and LDL2, two Arabidopsis homologs of human LYSINE-SPECIFIC DEMETHYLASE 1 (LSD1), repress the expression of dormancy-related genes including DOG1, ABSISIC ACID 2 (ABA2) and ABI3 (Zhao et al. 2015). FERTILIZATION INDEPENDENT ENDOSPERM (FIE), a component of polycomb repressive complex PRC2, reduces seed dormancy by repressing ABI3 and DOG1 (Bouyer et al. 2011). SLY1, a member of the GA signaling pathway, acts as a positive regulator of seed germination (Steber et al. 1998), whereas DELLA proteins GA INSENSITIVE (GAI), REPRESSOR OF GA1-3 (RGA), RGA-LIKE 1 (RGL1), RGL2 and RGL3, as well as SPINDLY (SPY), another negative regulator of GA signaling, inhibit seed germination by maintaining the dormancy state (Lee et al. 2002, Peng and Harberd 2002, Cao et al. 2005, Penfield et al. 2006, Piskurewicz and Lopez-Molina 2009). Repression of GIBBERELLIN 2-oxidase (GA2ox, GA catabolism) inhibits seed dormancy (Yamauchi et al. 2007), whereas the induction of GIBBERELLIN 3-oxidase 1 (GA3ox1, GA biosynthesis) induces dormancy break (Footitt et al. 2013). GA receptor proteins GIBBERELLIN-INSENSITIVE DWARF1s (GID1s), which ubiquitinate and degrade DELLA proteins along with SLY1 in the presence of GA, stimulate GA signal transduction (McGinnis et al. 2003, Sun and Gubler 2004, Ueguchi-Tanaka et al. 2007, Schwechheimer 2008). Consistent with this observation, GID1- or SLY1-deficient mutants show reduced germination rates (Griffiths et al. 2006, Iuchi et al. 2007). In addition, various Omics technologies have recently been employed to investigate the molecular mechanism underlying germination in Arabidopsis and various crops. However, more studies are needed to elucidate the biochemical and molecular mechanisms underlying dormancy and germination. Sumoylation is a post-translational modification in which SUMO is covalently attached to target proteins by E3 SUMO ligase. AtSIZ1 is an Arabidopsis E3 SUMO ligase that stimulates covalent modification of target proteins by SUMO. For example, AtSIZ1 directly interacts with nitrate reductases NIA1 and NIA2, INDUCER OF CBF EXPRESSION 1 (ICE1), ABSCISIC ACID-INSENSITIVE 5 (ABI5), FLOWERING LOCUS C (FLC), SLEEPY1 (SLY1), and CHROMOMETHYLASE 3 (CMT3) and sumoylates these proteins, which regulates their stability and activity (Miura et al. 2009, Elrouby and Coupland 2010, Miller et al. 2010, Park et al. 2011, Zheng et al. 2012, Conti et al. 2014, Son et al. 2014, Kim et al. 2015b, 2015a). Thus, sumoylation regulates various cellular processes including stress, defense, and nutrient deficiency responses, nutrient assimilation, growth, flowering and hormone signaling (Hotson et al. 2003, Kurepa et al. 2003, Lois et al. 2003, Murtas et al. 2003, Catala et al. 2007, Lee et al. 2007, Miura et al. Physiol. Plant. 158,
3 2007, Conti et al. 2008, 2014, Ishida et al. 2009, Zhang et al. 2013). In addition, numerous proteins have been shown to be sumoylated, although the particular E3 SUMO ligases involved have not been identified (Conti et al. 2014). A recent study demonstrated that the dwarf phenotype of the siz1-2 mutant recovered to the wild-type phenotype after the addition of exogenous ammonium, but not nitrate, phosphate or potassium (Park et al. 2011). In addition, the SUMO ligase activity of AtSIZ1 positively regulates GA signaling by sumoylating SLY1 during vegetative growth (Kim et al. 2015a), suggesting that AtSIZ1 also participates in seed germination. Moreover, ABI5 sumoylation by AtSIZ1 inactivates ABA signaling during seed germination (Miura et al. 2009). Nevertheless, how sumoylation controls dormancy and germination remains unknown. Here, we report the effect of the SUMO ligase activity of AtSIZ1 on seed dormancy and germination. The low germination rate of partially after-ripened siz1 mutant seeds recovered to wild-type levels after cold stratification or long periods of ripening, and exogenous GA supply improved the low germination rate of siz1 seeds to wild-type levels. AtSIZ1 stabilized SLY1 during germination, whereas msly1 did not exert its function on germination. During imbibition, SLY1 expression was downregulated in the siz1 mutants, whereas the expression of DOG1, ABI3 and DELLA genes was upregulated. These results represent the first evidence that GA-regulated seed dormancy break and germination are modulated by sumoylation through E3 SUMO ligase activity. To investigate the effects of sumoylation on SLY1 function during germination, after-ripened sly1-13, sly1-13/35s SLY1 FLAG 3 and sly1-13/35s msly1 FLAG 3 seeds (Kim et al. 2015a) were plated on MS medium and incubated at 22 Cfor7days.Forstratification, the plates were incubated for 4 days at 4 C and further incubated at 22 C for 7 days. The experiment was repeated three times. After-ripened seeds were stored at room temperature (22 C) after harvest. Effects of ammonium and GA on seed germination To investigate the effects of ammonium on germination, wild-type, siz1-2 andsiz1-3 seeds that were after-ripened for 1 week were plated on MS agar medium with or without 5 mm (NH 4 ) 2 SO 4. Half of the seeds were directly incubated for 7 days in a growth chamber at 22 C under a 16 h light/8 h dark cycle, whereas the remaining seeds were stratified for 4 days at 4 C, transferred to a growth chamber at 22 C, and further incubated for 7 days under a 16 h light/8 h dark cycle. The experiment was repeated three times. To investigate the effects of GA on germination, wild-type, siz1-2 andsiz1-3 seeds that were after-ripened for 1 week were plated on MS agar medium with or without 10 μm GA 3, followed by stratification for 4 days at 4 C. The seeds were then transferred to a growth chamber at 22 C and further incubated for 7 days under a 16 h light/8 h dark cycle. The experiment was repeated three times. Materials and methods Plant materials and growth conditions Arabidopsis thaliana Columbia-0 ecotype (wild-type) and the T-DNA insertion knock-out mutants siz1-2 and siz1-3 were used in this study. For plant growth in medium, seeds were surface sterilized in commercial bleach solution (containing 5% sodium hypochlorite and 0.1% Triton X-100) for 10 min, rinsed five times in sterilized water, and stratified at 4 C for 4 days in the dark. The seeds were planted on agar plates containing MS medium, 2% sucrose and 0.8% agar (ph 5.7). All plants (including seedlings) were grown in a growth chamber at 22 C under a 16 h light/8 h dark cycle. Estimation of seed germination rates Fresh or after-ripened wild-type, siz1-2,andsiz1-3 seeds were plated on MS medium and incubated at 22 Cfor 7 days. For stratification, the plates were incubated for 4 days at 4 C and further incubated at 22 Cfor7days. Production of transgenic Arabidopsis plants To produce SLY1- or msly1 (K122R)-overexpressing plants, the corresponding full-length cdnas were amplified by polymerase chain reaction (PCR) using a forward primer and a reverse primer tagged with FLAG 3 and inserted into plant expression vector pba002. Recombinant plasmids 35S-SLY1-FLAG 3 and 35S-mSLY1-FLAG 3 were introduced into wild-type and siz1-2 Arabidopsis. To produce double transgenic plants, the full-length cdna encoding AtSIZ1 was amplified by PCR using a forward primer tagged with HA 3 and a reverse primer, and inserted into plant expression vector per8. The resulting recombinant plasmids, XVE-HA 3 -AtSIZ1, 35S-SLY1-FLAG 3 and 35S-mSLY1-FLAG 3, were introduced into Arabidopsis. Effects of AtSIZ1 on SLY1 levels in vivo Post-translational degradation of SLY1 was examined during germination using transgenic wild-type 258 Physiol. Plant. 158, 2016
4 and siz1-2 seeds transformed with 35S SLY1 FLAG 3. Seeds of the transgenic plants were after-ripened for 1 week and incubated in MS medium with 100 μm cycloheximide (CHX) for 4 h. Proteins were extracted from seeds collected at the indicated time points and separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). SLY1 FLAG 3 levels were examined using western blotting with anti-flag antibody. To investigate the effect of AtSIZ1 overexpression on SLY1 levels, 2-week-old light-grown (16 h light/8 h dark) plants carrying 35S SLY1 FLAG 3 and XVE HA 3 AtSIZ1 or 35S msly1 FLAG 3 and XVE HA 3 AtSIZ1 were incubated in the light on MS medium with or without β-estradiol for 15 h. The samples were ground in liquid nitrogen and the lysates were separated by SDS-PAGE. SLY1 FLAG 3 and msly1 FLAG 3 levels were examined by western blotting with anti-flag antibody. HA 3 AtSIZ1 induction was analyzed by western blotting with anti-ha antibody. Measuring the transcript levels of GAand ABA-related genes Wild-type and siz1-2 seeds that were after-ripened for 1 week were imbibed in distilled water for 36 h, and samples were collected at the indicated time points. For GA treatment, wild-type and siz1-2 seeds that were after-ripened for 1 week were imbibed in distilled water containing 10 μm GA 3 for 24 h, and samples were collected at the indicated time points. Total RNA was extracted from each sample, quantified and diluted to equal concentrations. First-strand cdna was synthesized with 5 μg of total RNA using an iscript cdna Synthesis Kit (Bio-Rad, Hercules, CA, USA). An equal volume of cdna was amplified by real-time quantitative reverse-transcription PCR (qrt-pcr) (MyiQ, Bio-Rad) according to the manufacturer s protocol (Bio-Rad). The specific primers and template cdna were combined with 25 μl of iq SYBR Green Super Mix (Bio-Rad), and the reactions were performed under the following thermal cycling conditions: 50 Cfor2min,95 C for 10 min, 40 cycles of 95 C for 15 s, and 60 C for 1 min. The C T values of target genes were normalized to the C T value of the tubulin gene and analyzed with icycler IQ software (Bio-Rad). RT-PCR primers were designed using Primer3 ( r3plus.cgi), and their specificity was verified by cloning into the pgem T-Easy vector (Promega, Madison, WI) and sequencing with an ABI 3730 l DNA Analyzer (Applied Biosystems, Foster, CA). The primer sets used for these experiments are listed in Table S1, Supporting Information. Determining seed water content Wild-type and siz1-2 seeds that were after-ripened for 1 week were weighed (on a Precisa XT 220A balance) immediately after harvest and divided into 100 mg samples. The seed samples were then imbibed in 2 ml of distilled water for 48 h at 22 C and weighed again after drying for 1 min in an oven set at 65 C to remove the remaining water. The seed moisture content was calculated as the difference between the weights of seeds before and after imbibition. The experiment was repeated three times. Examination of precocious germination Developing siliques at the long green stage were collected from wild-type, siz1-2, and siz1-3 plants, sterilized with 70% (v/v) ethanol containing 0.1% Triton X-100 for 10 min, and rinsed five times in sterilized water. Sterilized siliques were gently opened at the valve-replum margin using a sharp surgical blade, and seven siliques were plated on a 0.8% (w/v) plant agar plate. The plates were incubated in a growth chamber at 22 C for 7 days under a 16 h light/8 h dark cycle and photographed. Results The siz1 mutants display uneven, low germination ratios The Arabidopsis siz1-2 and siz1-3 plants displayed dwarfism, early flowering and abnormal seed development, suggesting that the E3 SUMO ligase AtSIZ1 controls various signal transduction pathways during growth and development. Therefore, we further investigated whether AtSIZ1 plays a regulatory role in seed germination. First, we examined the germination ratios of fresh and after-ripened siz1-2 and siz1-3 seeds. The germination ratios of fresh wild-type, siz1-2, and siz1-3 seeds were quite low under both non-treated and cold-stratified conditions (Fig. 1A D). After 1 week of ripening at 22 C, the germination rates of seeds of the siz1-2 and siz1-3 mutants recovered to approximately 40%, whereas wild-type seeds germinated at a rate of approximately 100% (Fig. 1A, C). However, the germination ratios of siz1-2 and siz1-3 seeds that were after-ripened for 1 week reached approximately 100% after cold stratification (Fig. 1B, D). After-ripening for 4 weeks or more completely recovered the germination of wild-type, siz1-2, and siz1-3 seeds under both non-treated and cold-stratified conditions (Fig. 1A D). These results suggest that AtSIZ1 is involved in seed dormancy. Physiol. Plant. 158,
5 Fig. 1. Seed germination of the siz1 mutants. The seeds of wild-type, siz1-2, and siz1-3 plants were harvested and stored for the indicated time periods at 22 C. (A) Seeds plated on MS medium and incubated for 7 days at 22 C. (B) Seeds plated on MS medium and stratified for 4 days at 4 C, followed by incubation for an additional 7 days at 22 C. Germination ratios of non-treated (C) and cold-stratified (D) samples. Error bars indicate standard deviations (n = 3). A recent study showed that the siz1-2 mutant exhibits wild-type phenotypes in response to exogenous ammonium treatment (Park et al. 2011). We therefore examined whether exogenous ammonium would recover the low germination rates of siz1-2 and siz1-3 to wild-type levels. However, the germination rates of siz1-2 and siz1-3 seeds that were after-ripened for 1 week were not recovered to wild-type levels by ammonium supply (Fig. 2A, B), suggesting that ammonium has no effect on germination. DOG1 expression is upregulated in the siz1 mutants The recovery of germination of siz1 seeds by afterripening or cold stratification (Fig. 1) suggests that the loss of AtSIZ1 causes hyperdormancy. To investigate the effect of AtSIZ1 on the expression of dormancy-related genes, we examined the expression levels of three dormancy-stimulating genes, DOG1, LDL1 and LDL2. DOG1 transcript levels are high in dormant seeds, whereas the transcript levels of histone demethylase genes LDL1 and LDL2 are downregulated in dormant seeds (Bentsink et al. 2006, Zhao et al. 2015). Wild-type and siz1-2 seeds that were after-ripened for 1 week were used for this experiment. As expected, during imbibition, DOG1 transcript levels were higher in siz1-2 than in wild-type, whereas LDL1 and LDL2 transcript levels were lower in this mutant than in wild type (Fig. 3). These results indicate that hyperdormancy induced by the loss of AtSIZ1 is caused by the altered expression of dormancy-related genes. 260 Physiol. Plant. 158, 2016
6 Fig. 2. The effect of ammonium on the germination of siz1 seeds. (A) Wild-type, siz1-2 and siz1-3 seeds after-ripened for 1 week at 22 C after harvest were plated on MS medium with or without 5 mm (NH 4 ) 2 SO 4 and incubated for 7 days at 22 C. (B) Germination ratios of the samples. Error bars indicate standard deviations (n = 3). Germination of the siz1 mutants improves in response to exogenous GA supply Because GA positively regulates seed germination and can break seed dormancy, we next investigated whether exogenous GA would recover germination in the siz1 mutants. For this experiment, we treated wild-type, siz1-2, andsiz1-3 seeds that were after-ripened for 1 week with 10 μm GA 3 and estimated their germination rates. Some of the siz1-2 and siz1-3 seeds did not germinate (Fig. 4A, B left panel and upper panel, respectively), resulting in a germination rate of approximately 40% (Fig. 3C). However, the impaired germination of siz1-2 and siz1-3 seeds was completely recovered by the addition of 10 μm GA 3 (Fig. 3A, B, right panel and lower panel, respectively; Fig. 3C), suggesting that the GA-mediated signaling pathway is defective in the siz1 mutants during germination. SLY1-mediated GA signaling is disrupted in the siz1 mutants The above results suggest that GA signaling is abnormal in the siz1 mutants and that AtSIZ1 is involved in the role of GA signaling in seed germination. We therefore examined the role of AtSIZ1 in germination. For this experiment, we chose to focus on SLY1 because it is a central regulator of seed germination in response to GA and because SLY1-mediated vegetative growth is positively controlled by AtSIZ1 in Arabidopsis (Kim et al. 2015a). To examine the effect of AtSIZ1 on SLY1 stability, we infiltrated wild-type and siz1-2 plants with Agrobacteria transformed with 35S-SLY1-FLAG 3 to produce transgenic wild-type and siz1-2 plants overexpressing SLY1-FLAG 3. Next, we evaluated the decay of SLY1-FLAG 3 during germination in the presence of CHX to compare the degradation rates of this protein between transgenic wild-type and siz1-2 plants. The degradation of SLY1-FLAG 3 occurred much more rapidly in siz1-2 plants than in wild-type plants (Fig. 5A, B), indicating that SLY1 is stabilized by AtSIZ1 during germination. To further confirm this result, we produced double transgenic plants carrying 35S SLY1 FLAG 3 and XVE HA 3 AtSIZ1 or 35S msly1 FLAG 3 and XVE HA 3 AtSIZ1; msly1isa mutant SLY1 protein in which the 122nd lysine, a sumoylation site, was mutated to arginine (Kim et al. 2015a). We then measured SLY1-FLAG 3 levels in transgenic seedlings carrying the 35S SLY1 FLAG 3 transgene and the estradiol-inducible XVE HA 3 AtSIZ1 transgene. Induction of AtSIZ1 expression increased SLY1-FLAG 3 levels up to and 2.47-fold in seedlings of two independent transgenic lines, respectively (Fig. 6A). However, seedlings of the two independent transgenic lines carrying the 35S msly1 FLAG 3 transgene and the estradiol-inducible XVE HA 3 AtSIZ1 transgene showed no changes in msly1-flag 3 levels in response to AtSIZ1 induction (Fig. 6B). These results are consistent with the results of an in vivo transient expression experiment showing that SLY1-mediated GA signaling is positively regulated by AtSIZ1 (Kim et al. 2015a). These findings suggest that SLY1 is stabilized by sumoylation through the SUMO ligase activity of AtSIZ1 during germination. SLY1 transcription is downregulated in the siz1 mutants Our transgenic analysis results strongly suggests that the defective germination of siz1 mutant seeds is also caused by abnormal GA signaling due to low levels and activity of SLY1, as this protein is unstable in the mutants. However, it is also possible that the defective germination of siz1 mutant seeds results from changes in the expression of GA- and ABA-responsive genes involved in germination. Therefore, we monitored the transcript levels of four GA-responsive genes (SLY1, SLY2, RGA, and GAI), three GA-biosynthetic genes (GA2ox2, Physiol. Plant. 158,
7 Fig. 3. Transcript levels of dormancy-related genes in siz1 mutant seeds during imbibition. Seeds of wild-type and siz1-2 plants stored for the indicated time period at 22 C after harvesting were imbibed in distilled water for 12 h. Total RNA was extracted from each of the seed samples, and the expression levels of DOG1, LDL1 and LDL2 were examined by RT-PCR with gene-specific primers. DOG1, DELAY OF GERMINATION 1; LDL1, LYSINE-SPECIFIC DEMETHYLASE LIKE 1; LDL2, LYSINE-SPECIFIC DEMETHYLASE LIKE 2. GA3ox1, and GA3ox2), and an ABA-responsive gene (ABI3) insiz1-2. We imbibed wild-type and siz1-2 seeds that were after-ripened for 1 week in distilled water and measured the transcript levels of the selected genes by qrt-pcr at different time points (Fig. 7). SLY1 transcript levels were low in siz1-2 compared with wild-type. SLY1 expression was not induced in siz1-2 after imbibition, whereas its expression increased in wild-type. RGA transcript levels in wild-type and siz1-2 seeds were similar before imbibition. However, RGA expression dramatically increased in siz1-2 after 9 h of imbibition, whereas its expression was unchanged in the wild-type. The expression of GAI gradually increased in siz1-2, reaching the highest levels at 9 h, followed by a decrease to wild-type levels. Interestingly, in wild-type, GAI expression also gradually increased, peaking at 24 h, followed by a decrease to wild-type levels, although its expression level was much lower in wild-type than in siz1-2. There was little difference in the expression patterns of GA2ox2, GA3ox1 and GA3ox2 between wild-type and siz1-2 plants, although the expression of these genes in both wild-type and siz1-2 changed during imbibition. Transcript levels of the ABA-responsive gene ABI3 were higher in siz1-2 than in the wild-type, although its expression level fluctuated, and ABI3 expression did not change in wild-type seeds during imbibition. Arabidopsis contains two SLY genes, SLY1 and SLY2. We therefore also examined the expression patterns of SLY2, a homolog of SLY1. Its expression pattern was also almost the same in the wild-type and siz1-2, although its expression level increased until the 3 h time point and decreased during imbibition. We also examined AtSIZ1 expression in the wild-type, finding that AtSIZ1 expression increased dramatically until 6 h and gradually decreased thereafter. We also examined the effect of GA on the expression of SLY1 and DELLA genes RGA and GAI during germination. We imbibed wild-type and siz1-2 seeds that were after-ripened for 1 week in distilled water containing 10 μm GA 3 and measured the transcript levels of the genes by qrt-pcr at different time points (Fig. 8). SLY1 expression in both wild-type and siz1-2 was not altered by exogenous GA supply, although SLY1 transcript levels were low in siz1-2 compared with the wild-type. GA treatment had no effect on RGA or GAI transcription, although their expression levels were higher in siz1-2 than in the wild-type. These results suggest that the recovery of germination in the siz1 mutants by GA supply does not result from the change in SLY1 expression but rather from the change in its function. SLY1 modification by SUMO promotes germination One of the notable phenotypes of sly1 mutants is severe dwarfism (McGinnis et al. 2003). We previously showed that the growth of sly1-13 plants overexpressing wild-type SLY1 protein is restored to wild-type levels (Kim et al. 2015a). By contrast, the growth of sly1-13 plants overexpressing the mutated msly1 protein (K122R, a mutation at the sumoylation site) resembles that of sly1-13. Thus, we examined whether sumoylation affects the function of SLY1 during seed germination. For this experiment, we used sly1-13 and transgenic sly1-13 plants overexpressing SLY1-FLAG 3 or msly1-flag 3. First, we examined the germination ratios of sly1-13 seeds. The result show that the germination ratio of sly1-13 seeds that were after-ripened for 1 and 2 weeks was approximately 34 and 43%, respectively (Fig. 9A). However, the germination of mutant seeds recovered to wild-type levels in response to cold stratification or long periods of after-ripening 262 Physiol. Plant. 158, 2016
8 and SLY1-FLAG 3 -ormsly1-flag 3 -expressing transgenic sly1-13 seeds that were after-ripened for 2 weeks were slightly higher than those of sly1-13 and SLY1-FLAG 3 -or msly1-flag 3 -expressing transgenic sly1-13 seeds that were after-ripened for 1 week (Fig. 9A). After-ripening for long periods resulted in the recovery of germination ratios to wild-type levels (Fig. 9A). However, the germination ratios recovered to approximately 90% or more in response to cold stratification regardless of ripening period (Fig. 9B). These results suggest that the modification of SLY1 by SUMO is critical for its functioning during germination. GID1A is upregulated in the siz1 mutants Analyses of seed dormancy and transgenic plants and investigation of the expression levels of GA- and ABA-responsive genes suggested that the lower germination rate of partially after-ripened siz1 seeds is caused by high levels of DOG1 expression, low levels of SLY1 expression, and high levels of DELLA (RGA and GAI) gene and ABI3 expression, as well as low levels and activity of SLY1 (Figs 3 and 5 9). However, it is still possible that the low germination rate of siz1 mutant seeds after-ripened for 1 week may result from downregulation of GA receptor gene expression. We therefore examined the transcript levels of the GA receptor gene GA-INSENSITIVE DWARF1A (GID1A), finding that the expression of this gene increased rather strongly in the siz1-2 mutant until the 9 h time point, followed by a decrease, while its levels only slightly changed in wild-type seeds (Fig. 10). This result suggests that the lower germination rate of partially after-ripened siz1 mutant seeds is not due to GID1A expression. Fig. 4. AtSIZ1 positively regulates seed germination. (A) Germination of wild-type, siz1-2 and siz1-3 seeds after-ripened for 1 week at 22 C after harvest was examined after incubation on MS medium with or without 10 μm GA 3 for a short time (as indicated). (B) Germination and vegetative growth of wild type and siz1-2 on MS medium with or without 10 μm GA 3 for 7 days. (C) Germination ratios of wild-type, siz1-2 and siz1-3 seeds. Error bars indicate standard deviations (n = 3). (Fig. 9B). We then examined the germination ratios of sly1-13 transgenic mutant seeds. The germination ratio of after-ripened SLY1-FLAG 3 -expressing transgenic sly1-13 seeds that were after-ripened for 1 week reached approximately 90% (Fig. 9A), whereas the germination ratio msly1-flag 3 -expressing transgenic sly1-13 seeds that were after-ripened for 1 week were approximately 30% (Fig. 9A). The germination ratios of sly1-13 Precocious germination is suppressed in developing seeds of siz1 mutants Whereas quiescent seeds germinate when provided with the appropriate conditions necessary for resumption of growth of the enclosed embryo, dormant seeds germinate only when some additional hormonal, environmental, metabolic or physical conditions are met. Because the germination of partially after-ripened siz1 mutant seeds increased in response to cold stratification or GA supply, we also examined the germination rate of siz1 mutant seeds in developing immature siliques still attached to the mother plant. Wild-type, siz1-2, and siz1-3 seeds in developing siliques were directly plated onto MS medium and incubated for 7 days. Under these conditions, the siz1-2 and siz1-3 seeds germinated at a ratio of approximately 20% or less (Fig. 11A, B), whereas the germination ratio of wild-type seeds was Physiol. Plant. 158,
9 Fig. 5. Effect of AtSIZ1 on SLY1 levels. (A) Wild-type and siz1-2 plants were transformed with 35S-SLY1-FLAG 3, and transgenic wild-type and siz1-2 plants overexpressing SLY1-FLAG 3 were selected. Total proteins were extracted from transgenic wild-type and siz1-2 plants treated with 100 μm cycloheximide for the indicated length of time, and SLY1-FLAG 3 was detected by western blotting with anti-flag antibody; tubulin was used as a loading control. Numbers under lanes indicate relative intensities. Protein levels were normalized to a value of 1.00 for SLY1-FLAG 3 at time 0. (B) SLY1-FLAG 3 levels during degradation. Relative levels of SLY1-FLAG 3 shown in (A) were normalized to numerical values based on a value of 1.0 for protein levels at time 0. Closed and open circles indicate SLY1-FLAG 3 in wild-type and siz1-2 plants, respectively. Fig. 6. SLY1 is stabilized by AtSIZ1 in vivo. Double transgenic plants harboring 35S SLY1 FLAG 3 and XVE HA 3 AtSIZ1 (A) or 35S msly1 (K122R) FLAG 3 and XVE HA 3 AtSIZ1 (B) were incubated in liquid medium with β-estradiol to induce AtSIZ1 expression. After incubation for 15 h, HA 3 -AtSIZ1, SLY1-FLAG 3 and msly1-flag 3 levels were assessed using western blotting with anti-ha or anti-flag antibodies. Tubulin was used as a loading control. Numbers under lanes indicate relative intensities. Protein levels were normalized to a value of 1.00 for SLY1 or msly1 in the inducer lane in both panels. over 50% (Fig. 11A, B), which is much higher than that of non-ripened fresh wild-type, siz1-2, andsiz1-3 seeds (Fig. 1C, D). These results indicate that the loss of AtSIZ1 significantly alters the dormancy of developing seeds and increases seed dormancy during maturation. Water content is low in siz1 mutant seeds The water content of seeds is an important factor for germination. Accumulating data demonstrate that a certain water content in seeds is required for GA biosynthesis, ABA degradation and the expression of various genes required to induce germination (Weitbrecht et al. 2011). We found that the water content of siz1-2 seeds was approximately 10% lower than that of wild-type seeds (Fig. 12), suggesting that the low germination rate of siz1-2 seeds may be partially due to abnormal metabolism due to low water content during germination. Discussion Among GA signaling pathway components, seed germination is controlled, at least in part, by the degradation of SLY1-mediated DELLA proteins (which negatively regulate GA signaling) via the E3 ligase activity of SCF SLY1 264 Physiol. Plant. 158, 2016
10 Fig. 7. Transcript levels of GA-biosynthetic genes and ABA- or GA-responsive genes in siz1-2 seeds during imbibition. Seeds of wild-type and siz1-2 plants were imbibed in distilled water and sampled at the indicated time points. Total RNA was extracted from the seeds, and the expression levels of SLY1, RGA, GAI, ABI3, GA2ox2, GA3ox1, GA3ox2 and SLY2 were examined by RT-PCR with gene-specific primers. SLY1, SLEEPY1; RGA, Repressor of ga1-3; GAI, GA-insensitive; ABI3, ABSCISIC ACID-INSENSITIVE3; GA2ox2, GIBBERELLIN 2-oxidase 2; GA3ox1, GIBBERELLIN 3-oxidase 1; GA3ox2, GIBBERELLIN 2-oxidase 2; SLY2, SLEEPY2. Filled circle, wild-type plants; open circle, siz1-2 mutant. (Dill et al. 2004, Fu et al. 2004, Itoh et al. 2005). Recent reports suggest that the degradation of DELLA proteins is stimulated by SLY1 sumoylation through the SUMO ligase activity of AtSIZ1 during vegetative growth (Kim et al. 2015a). Among ABA signaling pathway components, seed germination is mainly controlled by transcription factors ABI3 (Ooms et al. 1993), ABI5 (Miura et al. 2009) and Viviparous1 (VP1) (McKibbin et al. 2002, Gerjets et al. 2010). DOG1 is another major factor that controls seed germination via ABA signaling (Teng et al. 2008, Graeber et al. 2010). The ethylene and jasmonate signaling pathways are also involved in seed germination. For instance, the 1-aminocyclopropane-1-carboxylic acid (ACC) oxidase ACO mediates the final step in ethylene biosynthesis, which regulates ethylene production during seed germination (Chiwocha et al. 2005, Kucera et al. 2005, Linkies et al. 2009), and ACO levels and ethylene production are tightly linked to seed germination in various plants (Leubner-Metzger et al. 1998, Petruzzelli et al. 2000, Chiwocha et al. 2005, Hermann et al. 2007, Linkies et al. 2009). Jasmonic acid and methyl jasmonate inhibit seed germination in various plants (Wilen et al. 1991, Norastehnia et al. 2007, Miersch et al. 2008, Oh et al. 2009, Zalewski et al. 2010), and 12-oxophytodienoic acid (OPDA), a precursor of methyl jasmonate, also has a strong inhibitory effect on seed germination (Dave et al. 2011). All of these findings indicate that germination is controlled by various hormone-signaling pathways and that protein modification is also an important post-translational mechanism for the regulation of seed germination. The siz1-2 mutant exhibits dwarfism (Catala et al. 2007), and the sly1 mutant displays severe dwarfism and dark green coloration (McGinnis et al. 2003). The dwarf phenotype of the siz1-2 mutant is caused by low levels and activity of SLY1 (Kim et al. 2015a), leading Physiol. Plant. 158,
11 Fig. 8. Examination of the effect of GA on the expression of GA-responsive genes in siz1 mutant seeds. Seeds of wild-type and siz1-2 plants were imbibed in MS medium with or without 10 μm GA 3 and sampled at the indicated time points. Total RNA was extracted from the seeds, and the expression levels of SLY1, RGA and GAI were examined by RT-PCR with gene-specific primers. SLY1, SLEEPY1; RGA, Repressor of ga1-3; GAI, GA-insensitive. Fig. 9. Seed germination of sly1 mutants and transgenic sly1 plants. The seeds of sly1-13, sly1-13/35s-sly1-flag 3 and sly1-13/35s-msly1-flag 3 plants were harvested and stored for the indicated time at 22 C. (A) Seeds plated on MS medium and incubated for 7 days at 22 C. (B) Seeds plated on MS medium and stratified for 4 days at 4 C. After further incubation at 22 C for 7 days, the germination ratios of the samples were estimated and graphed. Error bars indicate standard deviations (n = 3). to the conclusion that GA signaling during vegetative growth is positively regulated by AtSIZ1-mediated SLY1 sumoylation. Therefore, we further investigated the cause of the low seed germination rates of non-ripened or non-stratified siz1 mutantseeds, and foundthat their germination recovered to wild-type levels in response to long periods of ripening or cold stratification after harvest (Fig. 1). In addition, interestingly, exogenous supply of GA recovered the low germination rates of the siz1 mutants to wild-type levels (Fig. 4). These results suggest that the loss of AtSIZ1 causes enhanced dormancy and impaired GA signaling during germination. The dog1 mutants are completely non-dormant, which indicates that DOG1 is a key regulator of seed dormancy (Bentsink et al. 2006, Graeber et al. 2010, 2013, 2014, Nakabayashi et al. 2012). We found that DOG1 expression was upregulated in the siz1 mutants during imbibition (Fig. 3), indicating that AtSIZ1 suppresses the expression of seed dormancy-associated genes during imbibition, thereby reducing primary seed dormancy. In addition, the transcript levels of LDL1 and LDL2, encoding histone demethylases that negatively regulate DOG1 expression, were reduced in the siz1 mutants during imbibition (Fig. 3). Moreover, the DNA methylation activity of chromometylase CMT3 is stimulated by sumoylation via the E3 SUMO ligase activity of AtSIZ1 (Kim et al. 2015b). These results suggest that AtSIZ1 directly or indirectly participates in seed dormancy and germination as an epigenetic regulator. 266 Physiol. Plant. 158, 2016
12 Fig. 10. Transcript levels of GA receptor gene GID1A in the siz1 mutants. Wild-type and siz1-2 seeds were imbibed in distilled water for the indicated time period. Total RNA was isolated from wild-type and siz1-2 seeds, and GID1A expression levels were examined by quantitative RT-PCR with gene-specific primers. GID1A, GA-INSENSITIVE DWARF1A. Filled circle, wild type; open circle, siz1-2. Fig. 11. Suppression of precocious germination in siz1 siliques. (A) Immature green siliques of WT, siz1-2 andsiz1-3 plants incubated for 7 daysonagar medium. Seeds in siliquesofsiz1-2andsiz1-3 exhibited little germination. (B) Results of precocious germination tests of WT, siz1-2 and siz1-3 siliques. Error bars indicate standard deviations (SD, n = 3). A recent study demonstrated that SLY1 is directly sumoylated by AtSIZ1 and that sumoylation increases the stability and activity of SLY1 during vegetative growth (Kim et al. 2015a). Thus, we also examined the stability of SLY1 during germination. As expected, this protein was unstable in siz1-2 seeds during germination (Fig. 5) but became stabilized in the presence of sufficient levels of AtSIZ1 (Fig. 6), suggesting that SLY1 is stabilized and activated by sumoylation through the SUMO ligase activity of AtSIZ1 during germination and that SLY1 sumoylation is required for proper germination. ABA and GA are major phytohormones that control seed germination through the regulation of gene Fig. 12. Water content of siz1 seeds. The weights of after-ripened wild-type and siz1-2 seeds were measured before and after imbibition. Error bars indicate standard deviations (n = 3). expression. In the current study, we found that the poor germination of partially after-ripened siz1 mutant seeds was improved by exogenous GA (Fig. 4). This finding also suggests that germination-related gene expression may be altered in the siz1 mutants, which may explain its poor germination rate. Analysis of the transcript levels of germination-related genes revealed that during imbibition SLY1 was downregulated in the siz1-2 mutant, whereas ABI3 and DELLA genes were upregulated (Fig. 7), suggesting that the changes in germination-related gene expression due to the mutation of AtSIZ1 also contribute to the low germination rate of partially after-ripened siz1 seeds. In addition, AtSIZ1 expression also increased during the initial stage of imbibition and then decreased, as did SLY1 expression, although the expression patterns of AtSIZ1 and SLY1 were quite different, suggesting that AtSIZ1 affect SLY1-mediated germination through regulating SLY1 expression. The germination of transgenic sly1 mutants overexpressing wild-type SLY1 was recovered by after-ripening in all lines except transgenic sly1 mutants overexpressing msly1 (Fig. 9), indicating that sumoylation is a critical modification required for the functioning of SLY1 during germination. A recent study demonstrated that AtSIZ1 positively regulates GA signaling at the post-translational level during vegetative growth (Kim et al. 2015a). All these findings strongly suggest that sumoylation controls SLY1-mediated seed germination, seedling growth and vegetative development at both the transcriptional and post-translational levels. Uptake and cell-to-cell transport of GA are also highly important for seed germination and development. Therefore, the levels and stability of GA receptors may also Physiol. Plant. 158,
13 affect germination. However, our examination of the transcript levels of the GA receptor gene GID1A revealed that this gene was upregulated in partially after-ripened siz1-2 seeds during imbibition (Fig. 10), suggesting that the low germination rate of the mutant did not result from the low expression levels of this GA receptor gene. An increased primary dormancy level usually results in inhibition of preharvest sprouting or vivipary (Bewley et al. 2013, Nonogaki 2014). Examination of the effect of AtSIZ1 on vivipary showed that the germination ratio of siz1 seeds in developing siliques was lower than that of the wild-type (Fig. 11). This result suggests that the loss of AtSIZ1 enhances primary dormancy and, therefore, the difference in germination rate between seeds in wild-type and siz1 siliques results from their different levels of seed dormancy. GA and ABA play opposite roles in germination. Interestingly, sumoylation can stimulate both ABA and GA signaling during germination. ABI5 sumoylation inhibits ABA signaling during the germination stage (Miura et al. 2009). The current results show that the GA signaling pathway is positively regulated by sumoylation during the germination stage, suggesting that sumoylation is an important post-translational modification required for the control of germination. In conclusion, our results provide evidence that AtSIZ1 controls seed germination through modulating the stability and activity of SLY1 and the expression of dormancy-associated or germination-related GA and ABA signaling genes. Future studies examining the interplay between the sumoylation pathway and other signaling pathways, including GA, ABA and light signaling, will help elucidate the specific mechanisms that control seed dormancy and germination. Author contributions S. I. K., J. S. K., J. T. S. and H. S. S. designed the studies. S. I. K., and J. S. K. performed experiments. S. I. K., J. S. K., J. T. S. and H. S. S. interpreted data and wrote the manuscript. All authors commented on the results and the manuscript. Acknowledgements This work was supported by a grant from the Next-Generation BioGreen 21 Program (Plant Molecular Breeding Center) (Grant number PJ ), Rural Development Administration, Republic of Korea. References Ali-Rachedi S, Bouinot D, Wagner MH, Bonnet M, Sotta B, Grappin P, Jullien M (2004) Changes in endogenous abscisic acid levels during dormancy release and maintenance of mature seeds: studies with the Cape Verde Islands ecotype, the dormant model of Arabidopsis thaliana. Planta 219: Bentsink L, Jowett J, Hanhart CJ, Koornneef M (2006) Cloning of DOG1, a quantitative trait locus controlling seed dormancy in Arabidopsis. Proc Natl Acad Sci USA 103: Bewley JD (1997) Seed germination and dormancy. Plant Cell 9: Bewley JD, Bradford KJ, Hilhorst HWM, Nonogaki H (2003) Physiology of development, germination and dormancy, 3rd Edn. Springer, New York Bewley JD, Bradford KJ, Hilhorst HWM, Nonogaki H (2013) Seeds: Physiology of Development, Germination and Dormancy. Springer, New York Bouyer D, Roudier F, Heese M, Andersen ED, Gey D, Nowack MK, Goodrich J, Renou JP, Grini PE, Colot V, Schnittger A (2011) Polycomb repressive complex 2 controls the embryo-to-seedling phase transition. PLoS Genet 7: e Cao DN, Hussain A, Cheng H, Peng JR (2005) Loss of function of four DELLA genes leads to light- and gibberellin-independent seed germination in Arabidopsis. Planta 223: Catala R, Ouyang J, Abreu IA, Hu Y, Seo H, Zhang X, Chua NH (2007) The Arabidopsis E3 SUMO ligase SIZ1 regulates plant growth and drought responses. Plant Cell 19: Chiwocha SD, Cutler AJ, Abrams SR, Ambrose SJ, Yang J, Ross AR, Kermode AR (2005) The etr1-2 mutation in Arabidopsis thaliana affects the abscisic acid, auxin, cytokinin and gibberellin metabolic pathways during maintenance of seed dormancy, moist-chilling and germination. Plant J 42: Conti L, Price G, O Donnell E, Schwessinger B, Dominy P, Sadanandom A (2008) Small ubiquitin-like modifier proteases OVERLY TOLERANT TO SALT1 and -2 regulate salt stress responses in Arabidopsis. Plant Cell 20: Conti L, Nelis S, Zhang C, Woodcock A, Swarup R, Galbiati M, Tonelli C, Napier R, Hedden P, Bennett M, Sadanandom A (2014) Small ubiquitin-like modifier protein SUMO enables plants to control growth independently of the phytohormone gibberellin. Dev Cell 28: Dave A, Hernández ML, He Z, Andriotis VM, Vaistij E, Larson TR, Graham IA (2011) 12-oxo-phytodienoic acid accumulation during seed development represses seed germination in Arabidopsis. Plant Cell 23: Dill A, Thomas SG, Hu J, Steber CM, Sun TP (2004) The Arabidopsis F-box protein SLEEPY1 targets gibberellin signaling repressors for gibberellin-induced degradation. Plant Cell 16: Elrouby N, Coupland G (2010) Proteome-wide screens for small ubiquitin-like modifier (SUMO) substrates identify 268 Physiol. Plant. 158, 2016
14 Arabidopsis proteins implicated in diverse biological processes. Proc Natl Acad Sci USA 107: Finkelstein R, Reeves W, Ariizumi T, Steber C (2008) Molecular aspects of seed dormancy. Annu Rev Plant Biol 59: Footitt S, Huang Z, Clay HA, Mead A, Finch-Savage WE (2013) Temperature, light and nitrate sensing coordinate Arabidopsis seed dormancy cycling, resulting in winter and summer annual phenotypes. Plant J 74: Fu X, Richards DE, Fleck B, Xie D, Burton N, Harberd NP (2004) The Arabidopsis mutant sleepy1gar2-1 protein promotes plant growth by increasing the affinity of the SCFSLY1 E3 ubiquitin ligase for DELLA protein substrates. Plant Cell 16: Gerjets T, Scholefield D, Foulkes MJ, Lenton JR, Holdsworth MJ (2010) An analysis of dormancy, ABA responsiveness, after-ripening and pre-harvest sprouting in hexaploid wheat (Triticum aestivum L.) caryopses. J Exp Bot 61: Giraudat J, Hauge BM, Valon C, Smalle J, Parcy F, Goodman HM (1992) Isolation of the Arabidopsis-ABI3 gene by positional cloning. Plant Cell 4: Graeber K, Linkies A, Müller K, Wunchova A, Rott A, Leubner-Metzger G (2010) Cross-species approaches to seed dormancy and germination: conservation and biodiversity of ABA-regulated mechanisms and the Brassicaceae DOG1 genes. Plant Mol Biol 73: Graeber K, Voegele A, Büttner-Mainik A, Sperber K, Mummenhoff K, Leubner-Metzger G (2013) Spatiotemporal seed development analysis provides insight into primary dormancy induction and evolution of the Lepidium DELAY OF GERMINATION1 Genes. Plant Physiol 161: Graeber K, Linkies A, Steinbrecher T, Mummenhoff K, Tarkowská D, Turečková V, Ignatz M, Sperber K, Voegele A, de Jong H, Urbanová T, Strnad M, Leubner-Metzger G (2014) DELAY OF GERMINATION 1 mediates a conserved coat-dormancy mechanism for the temperature- and gibberellin-dependent control of seed germination. Proc Natl Acad Sci USA 111: E3571 E3580 Griffiths J, Murase K, Rieu I, Zentella R, Zhang ZL, Powers SJ, Gong F, Phillips AL, Hedden P, Sun TP, Thomas SG (2006) Genetic characterization and functional analysis of the GID1 gibberellin receptors in Arabidopsis. Plant Cell 18: Hermann K, Meinhard J, Dobrev P, Linkies A, Pesek B, Hess B, Machácková I, Fischer U, Leubner-Metzger G (2007) 1-Aminocyclopropane-1-carboxylic acid and abscisic acid during the germination of sugar beet (Beta vulgaris L.): a comparative study of fruits and seeds. J Exp Bot 58: Holdsworth MJ, Bentsink L, Soppe WJJ (2008) Molecular regulating Arabidopsis seed germination, after ripening, dormancy and germination. New Phytol 179: Hotson A, Chosed R, Shu H, Orth K, Mudgett MB (2003) Xanthomonas type III effector XopD targets SUMO-conjugated proteins in planta. Mol Microbiol 50: Ishida T, Fujiwara S, Miura K, Stacey N, Yoshimura M, Schneider K, Adachi S, Minamisawa K, Umeda M, Sugimoto K (2009) SUMO E3 ligase HIGH PLOIDY2 regulates endocycle onset and meristem maintenance in Arabidopsis. Plant Cell 21: Itoh H, Sasaki A, Ueguchi-Tanaka M, Ishiyama K, Kobayashi M, Hasegawa Y, Minami E, Ashikari M, Matsuoka M (2005) Dissection of the phosphorylation of rice DELLA protein, SLENDER RICE1. Plant Cell Physiol 46: Iuchi S, Suzuki H, Kim YC, Iuchi A, Kuromori T, Ueguchi-Tanaka M, Asami T, Yamaguchi I, Matsuoka M, Kobayashi M, Nakajima M (2007) Multiple loss-of-function of Arabidopsis gibberellin receptor AtGID1s completely shuts down a gibberellin signal. Plant J 50: Kim SI, Park BS, Kim DY, Yeu SY, Song SI, Song JT, Seo HS (2015a) E3 SUMO ligase AtSIZ1 positively regulates SLY1-mediated GA signaling and plant development. Biochem J 469: Kim DY, Han YJ, Kim SI, Song JT, Seo HS (2015b) Arabidopsis CMT3 activity is positively regulated by AtSIZ1-mediated sumoylation. Plant Sci 239: Koornneef M, Jorna ML, Derswan D, Karssen CM (1982) The isolation of abscisic-acid (ABA) deficient mutants by selection of induced revertants in non-germinating gibberellin sensitive lines of Arabidopsis thaliana (L) heynh. Theor Appl Genet 61: Kucera B, Cohn MA, Leubner-Metzger G (2005) Plant hormone interactions during seed dormancy release and germination. Seed Sci Res 15: Kurepa J, Walker JM, Smalle J, Gosink MM, Davis SJ, Durham TL, Sung DY, Vierstra RD (2003) The small ubiquitin-like modifier (SUMO) protein modification system in Arabidopsis. Accumulation of SUMO1 and -2 conjugates is increased by stress. J Biol Chem 278: Kushiro T, Okamoto M, Nakabayashi K, Yamagishi K, Kitamura S, Asami T, Hirai N, Koshiba T, Kamiya Y, Nambara E (2004) The Arabidopsis cytochrome P450 CYP707A encodes ABA 8 -hydroxylases: key enzymes in ABA catabolism. EMBO J 23: Lee S, Cheng H, King KE, Wang W, He Y, Hussain A, Lo J, Harberd NP, Peng J (2002) Gibberellin regulates Arabidopsis seed germination via RGL2, a GAI/RGA-like gene whose expression is upregulated following imbibition. Genes Dev 16: Lee J, Nam J, Park HC, Na G, Miura K, Jin JB, Yoo CY, Baek D, Kim DH, Jeong JC, Kim D, Lee SY, Salt DE, Mengiste T, Gong Q, Ma S, Bohnert HJ, Kwak SS, Bressan RA, Hasegawa PM, Yun DJ (2007) Salicylic acid-mediated Physiol. Plant. 158,
Life Science Journal 2014;11(9) Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis
 Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis Sung-Il Kim 1, Sang Ik Song 3, Hak Soo Seo 1, 2, 4 * 1 Department of Plant Science and Research Institute of Agriculture
Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis Sung-Il Kim 1, Sang Ik Song 3, Hak Soo Seo 1, 2, 4 * 1 Department of Plant Science and Research Institute of Agriculture
Chapter 2 Regulation of Seed Dormancy Cycling in Seasonal Field Environments
 Chapter 2 Regulation of Seed Dormancy Cycling in Seasonal Field Environments W.E. Finch-Savage and S. Footitt Seeds Select the Habitat, Climate Space and Season in Which to Germinate by Sensing and Responding
Chapter 2 Regulation of Seed Dormancy Cycling in Seasonal Field Environments W.E. Finch-Savage and S. Footitt Seeds Select the Habitat, Climate Space and Season in Which to Germinate by Sensing and Responding
SOMNUS, a CCCH-Type Zinc Finger Protein in Arabidopsis, Negatively Regulates Light-Dependent Seed Germination Downstream of PIL5 W
 The Plant Cell, Vol. 20: 1260 1277, May 2008, www.plantcell.org ª 2008 American Society of Plant Biologists SOMNUS, a CCCH-Type Zinc Finger Protein in Arabidopsis, Negatively Regulates Light-Dependent
The Plant Cell, Vol. 20: 1260 1277, May 2008, www.plantcell.org ª 2008 American Society of Plant Biologists SOMNUS, a CCCH-Type Zinc Finger Protein in Arabidopsis, Negatively Regulates Light-Dependent
The combined use of Arabidopsis thaliana and Lepidium sativum to find conserved mechanisms of seed germination within the Brassicaceae family
 www.seedbiology.de The combined use of Arabidopsis thaliana and Lepidium sativum to find conserved mechanisms of seed germination within the Brassicaceae family Linkies, A., Müller, K., Morris, K., Gräber,
www.seedbiology.de The combined use of Arabidopsis thaliana and Lepidium sativum to find conserved mechanisms of seed germination within the Brassicaceae family Linkies, A., Müller, K., Morris, K., Gräber,
Genetic Characterization and Functional Analysis of the GID1 Gibberellin Receptors in Arabidopsis W
 The Plant Cell, Vol. 18, 3399 3414, December 2006, www.plantcell.org ª 2006 American Society of Plant Biologists Genetic Characterization and Functional Analysis of the GID1 Gibberellin Receptors in Arabidopsis
The Plant Cell, Vol. 18, 3399 3414, December 2006, www.plantcell.org ª 2006 American Society of Plant Biologists Genetic Characterization and Functional Analysis of the GID1 Gibberellin Receptors in Arabidopsis
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated
 Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Plant Growth and Development
 Plant Growth and Development Concept 26.1 Plants Develop in Response to the Environment Factors involved in regulating plant growth and development: 1. Environmental cues (e.g., day length) 2. Receptors
Plant Growth and Development Concept 26.1 Plants Develop in Response to the Environment Factors involved in regulating plant growth and development: 1. Environmental cues (e.g., day length) 2. Receptors
23-. Shoot and root development depend on ratio of IAA/CK
 Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Cytokinin. Fig Cytokinin needed for growth of shoot apical meristem. F Cytokinin stimulates chloroplast development in the dark
 Cytokinin Abundant in young, dividing cells Shoot apical meristem Root apical meristem Synthesized in root tip, developing embryos, young leaves, fruits Transported passively via xylem into shoots from
Cytokinin Abundant in young, dividing cells Shoot apical meristem Root apical meristem Synthesized in root tip, developing embryos, young leaves, fruits Transported passively via xylem into shoots from
Major Plant Hormones 1.Auxins 2.Cytokinins 3.Gibberelins 4.Ethylene 5.Abscisic acid
 Plant Hormones Lecture 9: Control Systems in Plants What is a Plant Hormone? Compound produced by one part of an organism that is translocated to other parts where it triggers a response in target cells
Plant Hormones Lecture 9: Control Systems in Plants What is a Plant Hormone? Compound produced by one part of an organism that is translocated to other parts where it triggers a response in target cells
Dormancy in Plants. TheRoleofDormancyinPlants under Natural and Agricultural Conditions. Introduction. Advanced article
 Wim JJ Soppe, Max Planck Institute for Plant Breeding Research, Cologne, Germany Leónie Bentsink, Wageningen University, Wageningen, The Netherlands Introduction Advanced article Article Contents The Role
Wim JJ Soppe, Max Planck Institute for Plant Breeding Research, Cologne, Germany Leónie Bentsink, Wageningen University, Wageningen, The Netherlands Introduction Advanced article Article Contents The Role
10 DE-repression of seed germination by GA signaling
 10 DE-repression of seed germination by GA signaling Camille M. Steber 10.1 Introduction This chapter explores evidence that the ubiquitin proteasome pathway plays a role in gibberellin (GA) stimulation
10 DE-repression of seed germination by GA signaling Camille M. Steber 10.1 Introduction This chapter explores evidence that the ubiquitin proteasome pathway plays a role in gibberellin (GA) stimulation
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL
 GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. HSP21 expression in 35S:HSP21 and hsp21 knockdown plants. (a) Since no T- DNA insertion line for HSP21 is available in the publicly available T-DNA collections,
Supplementary Figure 1 Supplementary Figure 1. HSP21 expression in 35S:HSP21 and hsp21 knockdown plants. (a) Since no T- DNA insertion line for HSP21 is available in the publicly available T-DNA collections,
can affect division, elongation, & differentiation of cells to another region of plant where they have an effect
 Note that the following is a rudimentary outline of the class lecture; it does not contain everything discussed in class. Plant Hormones Plant Hormones compounds regulators growth or can affect division,
Note that the following is a rudimentary outline of the class lecture; it does not contain everything discussed in class. Plant Hormones Plant Hormones compounds regulators growth or can affect division,
Useful Propagation Terms. Propagation The application of specific biological principles and concepts in the multiplication of plants.
 Useful Propagation Terms Propagation The application of specific biological principles and concepts in the multiplication of plants. Adventitious Typically describes new organs such as roots that develop
Useful Propagation Terms Propagation The application of specific biological principles and concepts in the multiplication of plants. Adventitious Typically describes new organs such as roots that develop
Releasing the brakes of plant growth: how GAs shutdown DELLA proteins
 Journal of Experimental Botany, Vol. 60, No. 4, pp. 1085 1092, 2009 doi:10.1093/jxb/ern301 Advance Access publication 28 November, 2009 REVIEW PAPER Releasing the brakes of plant growth: how GAs shutdown
Journal of Experimental Botany, Vol. 60, No. 4, pp. 1085 1092, 2009 doi:10.1093/jxb/ern301 Advance Access publication 28 November, 2009 REVIEW PAPER Releasing the brakes of plant growth: how GAs shutdown
Supplemental Data. Perrella et al. (2013). Plant Cell /tpc
 Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Ph.D. thesis. Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana
 Ph.D. thesis Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana Written by: Edit Ábrahám Temesváriné Supervisors: Dr. László Szabados
Ph.D. thesis Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana Written by: Edit Ábrahám Temesváriné Supervisors: Dr. László Szabados
HRS1 Acts as a Negative Regulator of Abscisic Acid Signaling to Promote Timely Germination of Arabidopsis Seeds
 HRS1 Acts as a Negative Regulator of Abscisic Acid Signaling to Promote Timely Germination of Arabidopsis Seeds Chongming Wu 1,2., Juanjuan Feng 1,2., Ran Wang 1,2, Hong Liu 1,2, Huixia Yang 1,2, Pedro
HRS1 Acts as a Negative Regulator of Abscisic Acid Signaling to Promote Timely Germination of Arabidopsis Seeds Chongming Wu 1,2., Juanjuan Feng 1,2., Ran Wang 1,2, Hong Liu 1,2, Huixia Yang 1,2, Pedro
Epigenetics and Flowering Any potentially stable and heritable change in gene expression that occurs without a change in DNA sequence
 Epigenetics and Flowering Any potentially stable and heritable change in gene expression that occurs without a change in DNA sequence www.plantcell.org/cgi/doi/10.1105/tpc.110.tt0110 Epigenetics Usually
Epigenetics and Flowering Any potentially stable and heritable change in gene expression that occurs without a change in DNA sequence www.plantcell.org/cgi/doi/10.1105/tpc.110.tt0110 Epigenetics Usually
Reproduction, Seeds and Propagation
 Reproduction, Seeds and Propagation Diploid (2n) somatic cell Two diploid (2n) somatic cells Telophase Anaphase Metaphase Prophase I One pair of homologous chromosomes (homologues) II Homologues condense
Reproduction, Seeds and Propagation Diploid (2n) somatic cell Two diploid (2n) somatic cells Telophase Anaphase Metaphase Prophase I One pair of homologous chromosomes (homologues) II Homologues condense
Temperature, light and nitrate sensing coordinate Arabidopsis seed dormancy cycling, resulting in winter and summer annual phenotypes
 The Plant Journal (2013) 74, 1003 1015 doi: 10.1111/tpj.12186 Temperature, light and nitrate sensing coordinate Arabidopsis seed dormancy cycling, resulting in winter and summer annual phenotypes Steven
The Plant Journal (2013) 74, 1003 1015 doi: 10.1111/tpj.12186 Temperature, light and nitrate sensing coordinate Arabidopsis seed dormancy cycling, resulting in winter and summer annual phenotypes Steven
NATURAL VARIATION IN THE CYTOKININ METABOLIC NETWORK IN ARABIDOPSIS THALIANA
 NATURAL VARIATION IN THE CYTOKININ METABOLIC NETWORK IN ARABIDOPSIS THALIANA PŘÍRODNÍ VARIACE METABOLISMU CYTOKININŮ U ARABIDOPSIS THALIANA Samsonová Z. 1, 2, 3, Kuklová A. 1, 2, Mazura P. 1, 2, Rotková
NATURAL VARIATION IN THE CYTOKININ METABOLIC NETWORK IN ARABIDOPSIS THALIANA PŘÍRODNÍ VARIACE METABOLISMU CYTOKININŮ U ARABIDOPSIS THALIANA Samsonová Z. 1, 2, 3, Kuklová A. 1, 2, Mazura P. 1, 2, Rotková
THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING. AnitaHajdu. Thesis of the Ph.D.
 THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING AnitaHajdu Thesis of the Ph.D. dissertation Supervisor: Dr. LászlóKozma-Bognár - senior research associate Doctoral
THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING AnitaHajdu Thesis of the Ph.D. dissertation Supervisor: Dr. LászlóKozma-Bognár - senior research associate Doctoral
Is that artificial turf or real grass? Its thicker than Bermuda!
 Is that artificial turf or real grass? Its thicker than Bermuda! 1 Using Plant Growth Regulators Growth regulators DO NOT interfere with plant respiration, photosynthesis, or other internal plant functions
Is that artificial turf or real grass? Its thicker than Bermuda! 1 Using Plant Growth Regulators Growth regulators DO NOT interfere with plant respiration, photosynthesis, or other internal plant functions
Plant Development. Chapter 31 Part 1
 Plant Development Chapter 31 Part 1 Impacts, Issues Foolish Seedlings, Gorgeous Grapes Gibberellin and other plant hormones control the growth and development of plants environmental cues influence hormone
Plant Development Chapter 31 Part 1 Impacts, Issues Foolish Seedlings, Gorgeous Grapes Gibberellin and other plant hormones control the growth and development of plants environmental cues influence hormone
Voegele et al. Journal of Experimental Botany Embryo growth, testa permeability and endosperm weakening are major
 Voegele et al. Journal of Experimental Botany 2012 1 Embryo growth, testa permeability and endosperm weakening are major targets for the environmentally regulated inhibition of Lepidium sativum seed germination
Voegele et al. Journal of Experimental Botany 2012 1 Embryo growth, testa permeability and endosperm weakening are major targets for the environmentally regulated inhibition of Lepidium sativum seed germination
Arabidopsis thaliana. Lucia Strader. Assistant Professor, Biology
 Arabidopsis thaliana Lucia Strader Assistant Professor, Biology Arabidopsis as a genetic model Easy to grow Small genome Short life cycle Self fertile Produces many progeny Easily transformed HIV E. coli
Arabidopsis thaliana Lucia Strader Assistant Professor, Biology Arabidopsis as a genetic model Easy to grow Small genome Short life cycle Self fertile Produces many progeny Easily transformed HIV E. coli
Plant Molecular and Cellular Biology Lecture 10: Plant Cell Cycle Gary Peter
 Plant Molecular and Cellular Biology Lecture 10: Plant Cell Cycle Gary Peter 9/10/2008 1 Learning Objectives Explain similarities and differences between fungal, mammalian and plant cell cycles Explain
Plant Molecular and Cellular Biology Lecture 10: Plant Cell Cycle Gary Peter 9/10/2008 1 Learning Objectives Explain similarities and differences between fungal, mammalian and plant cell cycles Explain
EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA
 EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA Item Type text; Electronic Thesis Authors Bergstrand, Lauren Janel Publisher The University of Arizona. Rights Copyright is held by the author. Digital
EXPRESSION OF THE FIS2 PROMOTER IN ARABIDOPSIS THALIANA Item Type text; Electronic Thesis Authors Bergstrand, Lauren Janel Publisher The University of Arizona. Rights Copyright is held by the author. Digital
Introduction. Gene expression is the combined process of :
 1 To know and explain: Regulation of Bacterial Gene Expression Constitutive ( house keeping) vs. Controllable genes OPERON structure and its role in gene regulation Regulation of Eukaryotic Gene Expression
1 To know and explain: Regulation of Bacterial Gene Expression Constitutive ( house keeping) vs. Controllable genes OPERON structure and its role in gene regulation Regulation of Eukaryotic Gene Expression
CONTROL OF PLANT GROWTH AND DEVELOPMENT BI-2232 RIZKITA R E
 CONTROL OF PLANT GROWTH AND DEVELOPMENT BI-2232 RIZKITA R E The development of a plant the series of progressive changes that take place throughout its life is regulated in complex ways. Factors take part
CONTROL OF PLANT GROWTH AND DEVELOPMENT BI-2232 RIZKITA R E The development of a plant the series of progressive changes that take place throughout its life is regulated in complex ways. Factors take part
Selection and characterisation of the awake mutants with altered seed dormancy in response to temperature in Arabidopsis thaliana (L.) Heyn.
 Selection and characterisation of the awake mutants with altered seed dormancy in response to temperature in Arabidopsis thaliana (L.) Heyn. Submitted by FABIO FEDI to the University of Exeter as a thesis
Selection and characterisation of the awake mutants with altered seed dormancy in response to temperature in Arabidopsis thaliana (L.) Heyn. Submitted by FABIO FEDI to the University of Exeter as a thesis
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach
 Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids
 Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids Plant growth conditions The soil was a 1:1 v/v mixture of loamy soil and organic compost. Initial soil water content was determined
Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids Plant growth conditions The soil was a 1:1 v/v mixture of loamy soil and organic compost. Initial soil water content was determined
Genetic and Molecular Regulation by DELLA Proteins of Trichome Development in Arabidopsis 1[W][OA]
![Genetic and Molecular Regulation by DELLA Proteins of Trichome Development in Arabidopsis 1[W][OA] Genetic and Molecular Regulation by DELLA Proteins of Trichome Development in Arabidopsis 1[W][OA]](/thumbs/94/120779971.jpg) Genetic and Molecular Regulation by DELLA Proteins of Trichome Development in Arabidopsis 1[W][OA] Yinbo Gan, Hao Yu, Jinrong Peng, and Pierre Broun* Centre for Novel Agricultural Products, Department
Genetic and Molecular Regulation by DELLA Proteins of Trichome Development in Arabidopsis 1[W][OA] Yinbo Gan, Hao Yu, Jinrong Peng, and Pierre Broun* Centre for Novel Agricultural Products, Department
Chapter 1 Introduction
 Chapter 1 Introduction 1. INTRODUCTION Plants being sessile are exposed to environmental stresses mainly abiotic, caused by non-living effects of environment (temperature extremes, drought, and salinity)
Chapter 1 Introduction 1. INTRODUCTION Plants being sessile are exposed to environmental stresses mainly abiotic, caused by non-living effects of environment (temperature extremes, drought, and salinity)
Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering
 Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering by Valverde et. Al Published in Science 2004 Presented by Boyana Grigorova CBMG 688R Feb. 12, 2007 Circadian Rhythms: The Clock Within
Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering by Valverde et. Al Published in Science 2004 Presented by Boyana Grigorova CBMG 688R Feb. 12, 2007 Circadian Rhythms: The Clock Within
A Role for Multiple Circadian Clock Genes in the Response to Signals That Break Seed Dormancy in Arabidopsis W
 The Plant Cell, Vol. 21: 1722 1732, June 2009, www.plantcell.org ã 2009 American Society of Plant Biologists A Role for Multiple Circadian Clock Genes in the Response to Signals That Break Seed Dormancy
The Plant Cell, Vol. 21: 1722 1732, June 2009, www.plantcell.org ã 2009 American Society of Plant Biologists A Role for Multiple Circadian Clock Genes in the Response to Signals That Break Seed Dormancy
Electromagenetic spectrum
 Light Controls of Plant Development 1 Electromagenetic spectrum 2 Light It is vital for photosynthesis and is also necessary to direct plant growth and development. It acts as a signal to initiate and
Light Controls of Plant Development 1 Electromagenetic spectrum 2 Light It is vital for photosynthesis and is also necessary to direct plant growth and development. It acts as a signal to initiate and
Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport
 Ph.D. thesis Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport Zsigmond Laura Supervisor: Dr. Szabados László Arabidopsis Molecular Genetic Group Institute of Plant
Ph.D. thesis Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport Zsigmond Laura Supervisor: Dr. Szabados László Arabidopsis Molecular Genetic Group Institute of Plant
Abscisic acid content of a nondormant sunflower (Helianthus annuus L.) mutant
 Abscisic acid content of a nondormant sunflower (Helianthus annuus L.) mutant Brady A. Vick 1, C.C. Jan 2 1 USDA Agric. Res. Service, P.O. Box 677, Fargo, ND 81, USA, E-mail: brady.vick@ars.usda.gov 2
Abscisic acid content of a nondormant sunflower (Helianthus annuus L.) mutant Brady A. Vick 1, C.C. Jan 2 1 USDA Agric. Res. Service, P.O. Box 677, Fargo, ND 81, USA, E-mail: brady.vick@ars.usda.gov 2
Questions for Biology IIB (SS 2006) Wilhelm Gruissem
 Questions for Biology IIB (SS 2006) Plant biology Wilhelm Gruissem The questions for my part of Biology IIB, Plant Biology, are provided for self-study and as material for the exam. Please note that the
Questions for Biology IIB (SS 2006) Plant biology Wilhelm Gruissem The questions for my part of Biology IIB, Plant Biology, are provided for self-study and as material for the exam. Please note that the
Summary. Keywords: hormone metabolites, ethylene response mutant, auxins, cytokinins, abscisic acid, gibberellins, dormancy, germination.
 The Plant Journal (5) 42, 35 48 doi: 1.1111/j.1365-313X.5.2359.x The etr1-2 mutation in Arabidopsis thaliana affects the abscisic acid, auxin, cytokinin and gibberellin metabolic pathways during maintenance
The Plant Journal (5) 42, 35 48 doi: 1.1111/j.1365-313X.5.2359.x The etr1-2 mutation in Arabidopsis thaliana affects the abscisic acid, auxin, cytokinin and gibberellin metabolic pathways during maintenance
Identifying key genes involved in accumulation of ABA during drought in rice
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 6 Number 11 (2017) pp. 5482-5487 Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2017.611.527
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 6 Number 11 (2017) pp. 5482-5487 Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2017.611.527
Model plants and their Role in genetic manipulation. Mitesh Shrestha
 Model plants and their Role in genetic manipulation Mitesh Shrestha Definition of Model Organism Specific species or organism Extensively studied in research laboratories Advance our understanding of Cellular
Model plants and their Role in genetic manipulation Mitesh Shrestha Definition of Model Organism Specific species or organism Extensively studied in research laboratories Advance our understanding of Cellular
Beyond gibberellins and abscisic acid: how ethylene and jasmonates control seed germination
 Plant Cell Rep (2012) 31:253 270 DOI 10.1007/s00299-011-1180-1 REVIEW Beyond gibberellins and abscisic acid: how ethylene and jasmonates control seed germination Ada Linkies Gerhard Leubner-Metzger Received:
Plant Cell Rep (2012) 31:253 270 DOI 10.1007/s00299-011-1180-1 REVIEW Beyond gibberellins and abscisic acid: how ethylene and jasmonates control seed germination Ada Linkies Gerhard Leubner-Metzger Received:
Plant Growth Hormones - The Key Players In The Process Of Seed Germination
 Plant Growth Hormones - The Key Players In The Process Of Seed Germination Ameeta Sharma 1, Nikita Jain 2 1,2 Department of Biotechnology, The IIS University, Jaipur, Rajasthan, India. Abstract: Plant
Plant Growth Hormones - The Key Players In The Process Of Seed Germination Ameeta Sharma 1, Nikita Jain 2 1,2 Department of Biotechnology, The IIS University, Jaipur, Rajasthan, India. Abstract: Plant
Increasing Processing Tomato Fruit Soluble Solids
 Increasing Processing Tomato Fruit Soluble Solids Diane M Beckles Department of Plant Sciences dmbeckles@ucdavis.edu Processing Tomato Conference @ UC Davis December 13 th 2018 Soil Micronutrients Cultivar
Increasing Processing Tomato Fruit Soluble Solids Diane M Beckles Department of Plant Sciences dmbeckles@ucdavis.edu Processing Tomato Conference @ UC Davis December 13 th 2018 Soil Micronutrients Cultivar
SEED DORMANCY. Seed dormancy definitions. Seed dormancy. Seed dormancy 10/14/2013
 Seed dormancy definitions SEED DORMANCY Seed dormancy is a survival mechanism by which seeds can delay germination until the right environmental conditions for seedling growth and development. Seed dormancy
Seed dormancy definitions SEED DORMANCY Seed dormancy is a survival mechanism by which seeds can delay germination until the right environmental conditions for seedling growth and development. Seed dormancy
Plant hormones. Characteristics
 Plant hormones Plant hormones (also known as phytohormones) are chemicals that regulate plant growth, which, in the UK, are termed 'plant growth substances'. Plant hormones are signal molecules produced
Plant hormones Plant hormones (also known as phytohormones) are chemicals that regulate plant growth, which, in the UK, are termed 'plant growth substances'. Plant hormones are signal molecules produced
Growth Regulator Effects on Flowering in Maize
 Growth Regulator Effects on Flowering in Maize Eric Bumann July 14, 2008 My Background Research Associate at Pioneer Hi-Bred in Johnston, IA Production research 5 years in greenhouse research B.S. in Horticulture
Growth Regulator Effects on Flowering in Maize Eric Bumann July 14, 2008 My Background Research Associate at Pioneer Hi-Bred in Johnston, IA Production research 5 years in greenhouse research B.S. in Horticulture
Abscisic Acid Represses Growth of the Arabidopsis Embryonic Axis after Germination by Enhancing Auxin Signaling W
 The Plant Cell, Vol. 21: 2253 2268, August 2009, www.plantcell.org ã 2009 American Society of Plant Biologists Abscisic Acid Represses Growth of the Arabidopsis Embryonic Axis after Germination by Enhancing
The Plant Cell, Vol. 21: 2253 2268, August 2009, www.plantcell.org ã 2009 American Society of Plant Biologists Abscisic Acid Represses Growth of the Arabidopsis Embryonic Axis after Germination by Enhancing
Genetic interaction between rice PLASTOCHRON genes and the gibberellin pathway in leaf development
 Mimura and Itoh Rice 2014, 7:25 SHORT REPORT Open Access Genetic interaction between rice PLASTOCHRON genes and the gibberellin pathway in leaf development Manaki Mimura and Jun-Ichi Itoh * Abstract Background:
Mimura and Itoh Rice 2014, 7:25 SHORT REPORT Open Access Genetic interaction between rice PLASTOCHRON genes and the gibberellin pathway in leaf development Manaki Mimura and Jun-Ichi Itoh * Abstract Background:
Regulation of Phosphate Homeostasis by microrna in Plants
 Regulation of Phosphate Homeostasis by microrna in Plants Tzyy-Jen Chiou 1 *, Kyaw Aung 1,2, Shu-I Lin 1,3, Chia-Chune Wu 1, Su-Fen Chiang 1, and Chun-Lin Su 1 Abstract Upon phosphate (Pi) starvation,
Regulation of Phosphate Homeostasis by microrna in Plants Tzyy-Jen Chiou 1 *, Kyaw Aung 1,2, Shu-I Lin 1,3, Chia-Chune Wu 1, Su-Fen Chiang 1, and Chun-Lin Su 1 Abstract Upon phosphate (Pi) starvation,
Flowering Time Control in Plants -How plants know the time to flower?
 Advanced Molecular and Cell Biology II, 2015/12/04 Flowering Time Control in Plants -How plants know the time to flower? Masaki NIWA Grad. Sch. Biostudies, Kyoto Univ. Why can plants bloom every year in
Advanced Molecular and Cell Biology II, 2015/12/04 Flowering Time Control in Plants -How plants know the time to flower? Masaki NIWA Grad. Sch. Biostudies, Kyoto Univ. Why can plants bloom every year in
Examples of Epigenetics
 Examples of Computational EvoDevo, University of Leipzig WS 2016/17 How do plants know that winter is over? external input: light, photoperiodic external input: temperature receptive tissue: meristem,
Examples of Computational EvoDevo, University of Leipzig WS 2016/17 How do plants know that winter is over? external input: light, photoperiodic external input: temperature receptive tissue: meristem,
The Time Required for Dormancy Release in Arabidopsis Is Determined by DELAY OF GERMINATION1 Protein Levels in Freshly Harvested Seeds OA
 The Plant Cell, Vol. 24: 2826 2838, July 2012, www.plantcell.org ã 2012 American Society of Plant Biologists. All rights reserved. The Time Required for Dormancy Release in Arabidopsis Is Determined by
The Plant Cell, Vol. 24: 2826 2838, July 2012, www.plantcell.org ã 2012 American Society of Plant Biologists. All rights reserved. The Time Required for Dormancy Release in Arabidopsis Is Determined by
Gibberellin Signaling: A Theme and Variations on DELLA Repression 1
 Update on Gibberellin Signaling Gibberellin Signaling: A Theme and Variations on DELLA Repression 1 Amber L. Hauvermale, Tohru Ariizumi, and Camille M. Steber* Molecular Plant Sciences Program (A.L.H.,
Update on Gibberellin Signaling Gibberellin Signaling: A Theme and Variations on DELLA Repression 1 Amber L. Hauvermale, Tohru Ariizumi, and Camille M. Steber* Molecular Plant Sciences Program (A.L.H.,
Prospecting for Green Revolution Genes
 Learning Objectives: Prospecting for Green Revolution Genes 1) Discover how changes in individual genes produce phenotypic change 2) Learn to apply bioinformatics tools to identify groups of related genes
Learning Objectives: Prospecting for Green Revolution Genes 1) Discover how changes in individual genes produce phenotypic change 2) Learn to apply bioinformatics tools to identify groups of related genes
Role of Abscisic Acid in Seed Dormancy
 J Plant Growth Regul (2005) 24:319 344 DOI: 10.1007/s00344-005-0110-2 Role of Abscisic Acid in Seed Dormancy Allison R. Kermode* Department of Biological Sciences, Simon Fraser University, 8888 University
J Plant Growth Regul (2005) 24:319 344 DOI: 10.1007/s00344-005-0110-2 Role of Abscisic Acid in Seed Dormancy Allison R. Kermode* Department of Biological Sciences, Simon Fraser University, 8888 University
Dormancy break in seeds of Impatiens glandulifera Royle
 Dormancy break in seeds of Impatiens glandulifera Royle BY PAULINE M. MUMFORD* School of Biological Sciences, University of Birmingham, P.O. Box 363, Birmingham B15 2TT, U.K. (Received 27 April 1989; accepted
Dormancy break in seeds of Impatiens glandulifera Royle BY PAULINE M. MUMFORD* School of Biological Sciences, University of Birmingham, P.O. Box 363, Birmingham B15 2TT, U.K. (Received 27 April 1989; accepted
** * * * Col-0 cau1 CAU1. Actin2 CAS. Actin2. Supplemental Figure 1. CAU1 affects calcium accumulation.
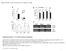 Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Complete all warm up questions Focus on operon functioning we will be creating operon models on Monday
 Complete all warm up questions Focus on operon functioning we will be creating operon models on Monday 1. What is the Central Dogma? 2. How does prokaryotic DNA compare to eukaryotic DNA? 3. How is DNA
Complete all warm up questions Focus on operon functioning we will be creating operon models on Monday 1. What is the Central Dogma? 2. How does prokaryotic DNA compare to eukaryotic DNA? 3. How is DNA
REVERSAL BY PHENOLIC COMPOUNDS OF ABSCISIC ACID-INDUCED INHIBITION OF IN VITRO ACTIVITY OF AMYLASE FROM SEEDS OF TRITICUM AESTIVUM 1..
 New Phytol. (1986) 103, 293-297 REVERSAL BY PHENOLIC COMPOUNDS OF ABSCISIC ACID-INDUCED INHIBITION OF IN VITRO ACTIVITY OF AMYLASE FROM SEEDS OF TRITICUM AESTIVUM 1.. BY SHASHI SHARMA, S. S. SHARMA AND
New Phytol. (1986) 103, 293-297 REVERSAL BY PHENOLIC COMPOUNDS OF ABSCISIC ACID-INDUCED INHIBITION OF IN VITRO ACTIVITY OF AMYLASE FROM SEEDS OF TRITICUM AESTIVUM 1.. BY SHASHI SHARMA, S. S. SHARMA AND
Regulation of Abscisic Acid Signaling by the Ethylene Response Pathway in Arabidopsis
 The Plant Cell, Vol. 12, 1117 1126, July 2000, www.plantcell.org 2000 American Society of Plant Physiologists Regulation of Abscisic Acid Signaling by the Ethylene Response Pathway in Arabidopsis Majid
The Plant Cell, Vol. 12, 1117 1126, July 2000, www.plantcell.org 2000 American Society of Plant Physiologists Regulation of Abscisic Acid Signaling by the Ethylene Response Pathway in Arabidopsis Majid
Aihua Liu 1., Feng Gao 1., Yuri Kanno 3, Mark C. Jordan 2, Yuji Kamiya 3, Mitsunori Seo 3, Belay T. Ayele 1 * Abstract.
 Regulation of Wheat Seed Dormancy by After-Ripening Is Mediated by Specific Transcriptional Switches That Induce Changes in Seed Hormone Metabolism and Signaling Aihua Liu 1., Feng Gao 1., Yuri Kanno 3,
Regulation of Wheat Seed Dormancy by After-Ripening Is Mediated by Specific Transcriptional Switches That Induce Changes in Seed Hormone Metabolism and Signaling Aihua Liu 1., Feng Gao 1., Yuri Kanno 3,
Cold and Light Control Seed Germination through the bhlh Transcription Factor SPATULA
 Current Biology, Vol. 15, 1998 2006, November 22, 2005, ª2005 Elsevier Ltd All rights reserved. DOI 10.1016/j.cub.2005.11.010 Cold and Light Control Seed Germination through the bhlh Transcription Factor
Current Biology, Vol. 15, 1998 2006, November 22, 2005, ª2005 Elsevier Ltd All rights reserved. DOI 10.1016/j.cub.2005.11.010 Cold and Light Control Seed Germination through the bhlh Transcription Factor
Curriculum vitae Xigang Liu
 Curriculum vitae Xigang Liu 1, EDUCATION: 09/1993-07/1997 B.S. Major: Biology. College of Life Sciences, Hebei Normal University Academic Degree Paper: RAPD analysis of Taigu genic male-sterile wheat and
Curriculum vitae Xigang Liu 1, EDUCATION: 09/1993-07/1997 B.S. Major: Biology. College of Life Sciences, Hebei Normal University Academic Degree Paper: RAPD analysis of Taigu genic male-sterile wheat and
Plant Growth and Development
 1. Define plasticity. Give an example? A: Plant Growth and Development The ability of the plants to follow different pathways in response to the environment or phases of life to form different kinds of
1. Define plasticity. Give an example? A: Plant Growth and Development The ability of the plants to follow different pathways in response to the environment or phases of life to form different kinds of
Ethylene: The Gaseous Hormone
 Ethylene: The Gaseous Hormone History: 1. 19 th century: coal gas was used for street illumination, it was observed that trees in the vicinity of streetlamps defoliated more extensively than other trees.
Ethylene: The Gaseous Hormone History: 1. 19 th century: coal gas was used for street illumination, it was observed that trees in the vicinity of streetlamps defoliated more extensively than other trees.
Common Effects of Abiotic Stress Factors on Plants
 Common Effects of Abiotic Stress Factors on Plants Plants are living organisms which lack ability of locomotion. Animals can move easily from one location to other. Immovable property of plants makes it
Common Effects of Abiotic Stress Factors on Plants Plants are living organisms which lack ability of locomotion. Animals can move easily from one location to other. Immovable property of plants makes it
Seed Dormancy and Germination
 Seed Dormancy and Germination Author(s): Leónie Bentsink and Maarten Koornneef Source: The Arabidopsis Book, Published By: The American Society of Plant Biologists https://doi.org/10.1199/tab.0119 URL:
Seed Dormancy and Germination Author(s): Leónie Bentsink and Maarten Koornneef Source: The Arabidopsis Book, Published By: The American Society of Plant Biologists https://doi.org/10.1199/tab.0119 URL:
POTASSIUM IN PLANT GROWTH AND YIELD. by Ismail Cakmak Sabanci University Istanbul, Turkey
 POTASSIUM IN PLANT GROWTH AND YIELD by Ismail Cakmak Sabanci University Istanbul, Turkey Low K High K High K Low K Low K High K Low K High K Control K Deficiency Cakmak et al., 1994, J. Experimental Bot.
POTASSIUM IN PLANT GROWTH AND YIELD by Ismail Cakmak Sabanci University Istanbul, Turkey Low K High K High K Low K Low K High K Low K High K Control K Deficiency Cakmak et al., 1994, J. Experimental Bot.
GIBBERELLIN HORMONE SIGNAL PERCEPTION: DOWN-REGULATING DELLA REPRESSORS OF PLANT GROWTH AND DEVELOPMENT
 Annual Plant Reviews (2016) 49, 153 188 doi: 10.1002/9781119210436.ch6 http://onlinelibrary.wiley.com Chapter 6 GIBBERELLIN HORMONE SIGNAL PERCEPTION: DOWN-REGULATING DELLA REPRESSORS OF PLANT GROWTH AND
Annual Plant Reviews (2016) 49, 153 188 doi: 10.1002/9781119210436.ch6 http://onlinelibrary.wiley.com Chapter 6 GIBBERELLIN HORMONE SIGNAL PERCEPTION: DOWN-REGULATING DELLA REPRESSORS OF PLANT GROWTH AND
Genetic controls of apple fruit-specific auxin metabolism. PI: Yanmin Zhu Co-PI(2): James Mattheis
 FINAL PROJECT REPORT Project Title: Genetic controls of apple fruit-specific auxin metabolism PI: Yanmin Zhu Co-PI(2): James Mattheis Organization: TFRL-ARS-USDA Organization: TFRL-ARS-USDA Telephone:
FINAL PROJECT REPORT Project Title: Genetic controls of apple fruit-specific auxin metabolism PI: Yanmin Zhu Co-PI(2): James Mattheis Organization: TFRL-ARS-USDA Organization: TFRL-ARS-USDA Telephone:
Prokaryotic Regulation
 Prokaryotic Regulation Control of transcription initiation can be: Positive control increases transcription when activators bind DNA Negative control reduces transcription when repressors bind to DNA regulatory
Prokaryotic Regulation Control of transcription initiation can be: Positive control increases transcription when activators bind DNA Negative control reduces transcription when repressors bind to DNA regulatory
Nature Genetics: doi: /ng Supplementary Figure 1. The phenotypes of PI , BR121, and Harosoy under short-day conditions.
 Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Supplementary Figure 1 The phenotypes of PI 159925, BR121, and Harosoy under short-day conditions. (a) Plant height. (b) Number of branches. (c) Average internode length. (d) Number of nodes. (e) Pods
Chapter 6. General discussion
 Chapter 6 General discussion 67 Chapter 6 General discussion Plants react in various ways on environmental stress conditions. The riverplain species Rumex palustris responds to submergence with an upward
Chapter 6 General discussion 67 Chapter 6 General discussion Plants react in various ways on environmental stress conditions. The riverplain species Rumex palustris responds to submergence with an upward
BIO1PS 2012 Plant Science Lecture 4 Hormones Pt. I
 BIO1PS 2012 Plant Science Lecture 4 Hormones Pt. I Dr. Michael Emmerling Department of Botany Room 410 m.emmerling@latrobe.edu.au Hormones and Ghost gum Eucalyptus papuana Coordination ~3 Lectures Leaves
BIO1PS 2012 Plant Science Lecture 4 Hormones Pt. I Dr. Michael Emmerling Department of Botany Room 410 m.emmerling@latrobe.edu.au Hormones and Ghost gum Eucalyptus papuana Coordination ~3 Lectures Leaves
ABA plays a central role in mediating the regulatory effects of nitrate on root branching in Arabidopsis
 The Plant Journal (2001) 28(6), 655662 ABA plays a central role in mediating the regulatory effects of nitrate on root branching in Arabidopsis Laurent Signora 1,2, Ive De Smet 2,², Christine H. Foyer
The Plant Journal (2001) 28(6), 655662 ABA plays a central role in mediating the regulatory effects of nitrate on root branching in Arabidopsis Laurent Signora 1,2, Ive De Smet 2,², Christine H. Foyer
Supplemental Data. Chen and Thelen (2010). Plant Cell /tpc
 Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Seed dormancy cycling and the regulation of dormancy mechanisms to time germination in variable field environments
 Journal of Experimental Botany, Vol. 68, No. 4 pp. 843 856, 2017 doi:10.1093/jxb/erw477 Advance Access publication 24 January 2017 REVIEW PAPER Seed dormancy cycling and the regulation of dormancy mechanisms
Journal of Experimental Botany, Vol. 68, No. 4 pp. 843 856, 2017 doi:10.1093/jxb/erw477 Advance Access publication 24 January 2017 REVIEW PAPER Seed dormancy cycling and the regulation of dormancy mechanisms
Biology 213 Exam 3 Practice Key
 Biology 213 Practice Key 1. (4) Explain the difference between a macronutrient and a micronutrient and cite two examples of each category? Macronutrients are the minerals needed by the plant in greater
Biology 213 Practice Key 1. (4) Explain the difference between a macronutrient and a micronutrient and cite two examples of each category? Macronutrients are the minerals needed by the plant in greater
Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function
 Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function Brian D. Gregory Department of Biology Penn Genome Frontiers Institute University of Pennsylvania
Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function Brian D. Gregory Department of Biology Penn Genome Frontiers Institute University of Pennsylvania
REVIEW SESSION. Wednesday, September 15 5:30 PM SHANTZ 242 E
 REVIEW SESSION Wednesday, September 15 5:30 PM SHANTZ 242 E Gene Regulation Gene Regulation Gene expression can be turned on, turned off, turned up or turned down! For example, as test time approaches,
REVIEW SESSION Wednesday, September 15 5:30 PM SHANTZ 242 E Gene Regulation Gene Regulation Gene expression can be turned on, turned off, turned up or turned down! For example, as test time approaches,
A study on genetic aspects of seed dormancy
 Available online at http://www.ijabbr.com International journal of Advanced Biological and Biomedical Research Volume 2, Issue 1, 2014: 211-215 A study on genetic aspects of seed dormancy Sanaz Kamali
Available online at http://www.ijabbr.com International journal of Advanced Biological and Biomedical Research Volume 2, Issue 1, 2014: 211-215 A study on genetic aspects of seed dormancy Sanaz Kamali
10/4/2017. Chapter 39
 Chapter 39 1 Reception 1 Reception 2 Transduction CYTOPLASM CYTOPLASM Cell wall Plasma membrane Phytochrome activated by light Cell wall Plasma membrane Phytochrome activated by light cgmp Second messenger
Chapter 39 1 Reception 1 Reception 2 Transduction CYTOPLASM CYTOPLASM Cell wall Plasma membrane Phytochrome activated by light Cell wall Plasma membrane Phytochrome activated by light cgmp Second messenger
16 CONTROL OF GENE EXPRESSION
 16 CONTROL OF GENE EXPRESSION Chapter Outline 16.1 REGULATION OF GENE EXPRESSION IN PROKARYOTES The operon is the unit of transcription in prokaryotes The lac operon for lactose metabolism is transcribed
16 CONTROL OF GENE EXPRESSION Chapter Outline 16.1 REGULATION OF GENE EXPRESSION IN PROKARYOTES The operon is the unit of transcription in prokaryotes The lac operon for lactose metabolism is transcribed
3.B.1 Gene Regulation. Gene regulation results in differential gene expression, leading to cell specialization.
 3.B.1 Gene Regulation Gene regulation results in differential gene expression, leading to cell specialization. We will focus on gene regulation in prokaryotes first. Gene regulation accounts for some of
3.B.1 Gene Regulation Gene regulation results in differential gene expression, leading to cell specialization. We will focus on gene regulation in prokaryotes first. Gene regulation accounts for some of
Supplementary Figure 1 Characterization of wild type (WT) and abci8 mutant in the paddy field.
 Supplementary Figure 1 Characterization of wild type (WT) and abci8 mutant in the paddy field. A, Phenotypes of 30-day old wild-type (WT) and abci8 mutant plants grown in a paddy field under normal sunny
Supplementary Figure 1 Characterization of wild type (WT) and abci8 mutant in the paddy field. A, Phenotypes of 30-day old wild-type (WT) and abci8 mutant plants grown in a paddy field under normal sunny
CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON
 PROKARYOTE GENES: E. COLI LAC OPERON CHAPTER 13 CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON Figure 1. Electron micrograph of growing E. coli. Some show the constriction at the location where daughter
PROKARYOTE GENES: E. COLI LAC OPERON CHAPTER 13 CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON Figure 1. Electron micrograph of growing E. coli. Some show the constriction at the location where daughter
Chapter 39. Plant Reactions. Plant Hormones 2/25/2013. Plants Response. What mechanisms causes this response? Signal Transduction Pathway model
 Chapter 39 Plants Response Plant Reactions Stimuli & a Stationary life Animals respond to stimuli by changing behavior Move toward positive stimuli Move away from negative stimuli Plants respond to stimuli
Chapter 39 Plants Response Plant Reactions Stimuli & a Stationary life Animals respond to stimuli by changing behavior Move toward positive stimuli Move away from negative stimuli Plants respond to stimuli
Expression of genes associated with the biosynthetic pathways of abscisic acid, gibberellin, and ethylene during the germination of lettuce seeds
 Expression of genes associated with the biosynthetic pathways of abscisic acid, gibberellin, and ethylene during the germination of lettuce seeds A.C.S. Clemente 1, R.M. Guimarães 1, D.C. Martins 2, L.A.A.
Expression of genes associated with the biosynthetic pathways of abscisic acid, gibberellin, and ethylene during the germination of lettuce seeds A.C.S. Clemente 1, R.M. Guimarães 1, D.C. Martins 2, L.A.A.
CONTROL SYSTEMS IN PLANTS
 AP BIOLOGY PLANTS FORM & FUNCTION ACTIVITY #5 NAME DATE HOUR CONTROL SYSTEMS IN PLANTS HORMONES MECHANISM FOR HORMONE ACTION Plant Form and Function Activity #5 page 1 CONTROL OF CELL ELONGATION Plant
AP BIOLOGY PLANTS FORM & FUNCTION ACTIVITY #5 NAME DATE HOUR CONTROL SYSTEMS IN PLANTS HORMONES MECHANISM FOR HORMONE ACTION Plant Form and Function Activity #5 page 1 CONTROL OF CELL ELONGATION Plant
A novel role for histone methyltransferase KYP SUVH4 in the control of Arabidopsis primary seed dormancy
 Research A novel role for histone methyltransferase KYP SUVH4 in the control of Arabidopsis primary seed dormancy Jian Zheng 1,2 *, Fengying Chen 1 *, Zhi Wang 1,3, Hong Cao 1, Xiaoying Li 1, Xin Deng
Research A novel role for histone methyltransferase KYP SUVH4 in the control of Arabidopsis primary seed dormancy Jian Zheng 1,2 *, Fengying Chen 1 *, Zhi Wang 1,3, Hong Cao 1, Xiaoying Li 1, Xin Deng
Analysis of regulatory function of circadian clock. on photoreceptor gene expression
 Thesis of Ph.D. dissertation Analysis of regulatory function of circadian clock on photoreceptor gene expression Tóth Réka Supervisor: Dr. Ferenc Nagy Biological Research Center of the Hungarian Academy
Thesis of Ph.D. dissertation Analysis of regulatory function of circadian clock on photoreceptor gene expression Tóth Réka Supervisor: Dr. Ferenc Nagy Biological Research Center of the Hungarian Academy
UNIVERSITY OF AGRICULTURAL SCIENCES AND VETERINARY MEDICINE
 UNIVERSITY OF AGRICULTURAL SCIENCES AND VETERINARY MEDICINE FACULTY OF HORTICULTURE OANA CIUZAN ROLE OF THE GLYCINE-RICH RNA-BINDING PROTEINS IN PLANT EARLY DEVELOPMENT AND ABIOTIC STRESS RESPONSE ABSTRACT
UNIVERSITY OF AGRICULTURAL SCIENCES AND VETERINARY MEDICINE FACULTY OF HORTICULTURE OANA CIUZAN ROLE OF THE GLYCINE-RICH RNA-BINDING PROTEINS IN PLANT EARLY DEVELOPMENT AND ABIOTIC STRESS RESPONSE ABSTRACT
CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-15 PLANT GROWTH AND DEVELOPMENT
 CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-15 PLANT GROWTH AND DEVELOPMENT Root, stem leaves, flower, fruits and seeds arise in orderly manner in plants. The sequence of growth is as follows-
CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-15 PLANT GROWTH AND DEVELOPMENT Root, stem leaves, flower, fruits and seeds arise in orderly manner in plants. The sequence of growth is as follows-
