Differential Regulation of Cellulose Orientation at the Inner and Outer Face of Epidermal Cells in the Arabidopsis Hypocotyl W
|
|
|
- Dortha Thornton
- 6 years ago
- Views:
Transcription
1 The Plant Cell, Vol. 23: , July 2011, ã 2011 American Society of Plant Biologists. All rights reserved. Differential Regulation of Cellulose Orientation at the Inner and Outer Face of Epidermal Cells in the Arabidopsis Hypocotyl W Elizabeth Faris Crowell, a,b Hélène Timpano, a Thierry Desprez, a Tiny Franssen-Verheijen, c Anne-Mie Emons, c Herman Höfte, a and Samantha Vernhettes a,1 a Institut Jean-Pierre Bourgin, Unité Mixte de Recherche 1318, INRA-AgroParisTech, Institut National de la Recherche Agronomique Centre de Versailles-Grignon, Versailles, France b Membrane Traffic and Cell Division Laboratory, Institut Pasteur, Paris, France c Laboratory of Plant Cell Biology, Wageningen University, 6708 PB Wageningen, The Netherlands It is generally believed that cell elongation is regulated by cortical microtubules, which guide the movement of cellulose synthase complexes as they secrete cellulose microfibrils into the periplasmic space. Transversely oriented microtubules are predicted to direct the deposition of a parallel array of microfibrils, thus generating a mechanically anisotropic cell wall that will favor elongation and prevent radial swelling. Thus far, support for this model has been most convincingly demonstrated in filamentous algae. We found that in etiolated Arabidopsis thaliana hypocotyls, microtubules and cellulose synthase trajectories are transversely oriented on the outer surface of the epidermis for only a short period during growth and that anisotropic growth continues after this transverse organization is lost. Our data support previous findings that the outer epidermal wall is polylamellate in structure, with little or no anisotropy. By contrast, we observed perfectly transverse microtubules and microfibrils at the inner face of the epidermis during all stages of cell expansion. Experimental perturbation of cortical microtubule organization preferentially at the inner face led to increased radial swelling. Our study highlights the previously underestimated complexity of cortical microtubule organization in the shoot epidermis and underscores a role for the inner tissues in the regulation of growth anisotropy. INTRODUCTION Plant growth and development depend on the coordinated expansion of individual cells. In order for cell expansion to occur, the plant cell wall must yield to the force exerted by hydrostatic pressure (reviewed in Cosgrove, 2005). Since hydrostatic pressure is thought to be constant and uniform in normal growth conditions, it follows that the rate and direction of cell expansion is primarily determined by the properties of the cell wall. The plant cell wall is composed of long cellulose microfibrils embedded in a viscoelastic matrix. Cellulose microfibrils are deposited in parallel arrays and confer anisotropic properties to the cell wall (Probine and Preston, 1961, 1962). A widely accepted hypothesis states that cell expansion occurs along the axis perpendicular to the orientation of the cellulose microfibrils (Green, 1962). Thus, cell elongation coincides with the deposition of cellulose microfibrils in transversely oriented arrays, as has been observed, for instance, in the filamentous internodes of the alga Nitella (Roelofsen and Houwink, 1953) and Arabidopsis thaliana root epidermal cells (Sugimoto et al., 2000). The microtubule-microfibril alignment hypothesis (Ledbetter and Porter, 1963; Green, 1965; Heath, 1974) posits that cortical 1 Address correspondence to samantha.vernhettes@versailles.inra.fr. The author responsible for distribution of materials integral to the findings presented in this article in accordance with the policy described in the Instructions for Authors ( is: Samantha Vernhettes (samantha.vernhettes@versailles.inra.fr). W Online version contains Web-only data. microtubules guide motile cellulose synthase complexes (CSCs) in the plasma membrane as they secrete cellulose microfibrils into the wall. A large body of evidence shows that the orientation of the cortical microtubule array reflects the orientation of the cellulose microfibrils deposited in the cell wall (reviewed in Baskin, 2001), and CSCs have been shown to move along cortical microtubules in living cells (Paredez et al., 2006). Consequently, the orientation of cortical microtubules determines the orientation of newly deposited cellulose microfibrils in the cell wall, ultimately determining the preferential direction of cell expansion. The initial studies on the regulation of cell elongation focused on the filamentous internodes of algae like Nitella (Roelofsen and Houwink, 1953; Green, 1960), which lack the complexity and mechanical constraints of organs of higher plants, in which cell layers with extensible walls are connected to layers with less extensible walls. This generates compression and tension forces (referred to as tissue tension), in addition to the local forces of turgor pressure. External epidermal walls are generally the thickest and probably the least extensible and hence are thought to bear the forces generated in internal tissue layers (Kutschera, 1992). Recent genetic studies indeed support this idea (Savaldi- Goldstein et al., 2007), but thus far it remains unclear how the architecture of the external epidermal cell wall relates to the growth anisotropy of the organ. If the epidermis controls growth anisotropy, one would expect to find transversely oriented cellulose microfibrils in epidermal cell walls, as predicted by the experiments in Nitella. This seems to be the case for instance in Arabidopsis roots (Sugimoto et al., 2000). Instead, in hypocotyls
2 Growth Anisotropy in Higher Plants 2593 or coleoptyles, it has been repeatedly shown that cell walls of growing epidermal cells have a crossed polylamellate or helicoidal architecture, containing arrays of cellulose microfibrils deposited in all orientations (Roland et al., 1975; Neville and Levy, 1984; Refrégier et al., 2004; reviewed in Neville et al., 1976). This polylamellate architecture is, at least in the Arabidopsis lightgrown hypocotyl, generated by continuously rotating microtubule arrays that correspondingly cause rotation of cellulose synthase trajectories in the plasma membrane (Chan et al., 2010). The resulting wall architecture is predicted to be isotropic, suggesting that the cell walls of inner tissues rather than the outer epidermal walls may play a dominant role in regulating the growth anisotropy of the organ (Baskin, 2005). Although it has been noted that cortical microtubules are not always organized in the same manner on the outer tangential face and the inner tangential face of epidermal cells (Busby and Gunning, 1983; Flanders et al., 1989; Yuan et al., 1995; Hejnowicz et al., 2000; Barton and Overall, 2010), all studies of cellulose synthase proteins thus far have focused on the outer face due to difficulty in accessing the deeper regions of the tissue (for example, Paredez et al., 2006; DeBolt et al., 2007; Desprez et al., 2007; Harris and DeBolt, 2008; Paredez et al., 2008; Crowell et al., 2009; Gutierrez et al., 2009). To study how cellulose deposition is related to hypocotyl elongation, we found it pertinent to characterize both the inner and outer tangential walls of the epidermal cells (see Supplemental Figure 1 online) and used spinning disk microscopy and field emission scanning electron microscopy (FESEM) to do so. Here, we present a detailed quantitative study of the orientation of microtubules, CSC trajectories, and newly deposited microfibrils throughout the whole volume of the epidermis in etiolated Arabidopsis hypocotyls. Our results show that microtubules and cellulose synthase trajectories at the inner face of the epidermis consistently show a transverse orientation in the growing zone, whereas on the outer face, the transverse orientation occurs only transiently, after which elongation growth continues despite the absence of a preferential microtubule orientation. In addition, a reduced degree of growth anisotropy was observed in plants in which microtubules had been perturbed primarily on the inner epidermal face. The results show that epidermal cells are polarized for the orientation of the cortical microtubules, cellulose synthase trajectories, and newly deposited microfibrils. Furthermore, our data support a model in which internal cell walls and not the outer epidermal cell walls regulate the directionality of the expansion of the organ. at the maximum rate of 370 mm/h (Figure 1A). We additionally analyzed hypocotyls at 76 HAI, just as the growth rate began to decline (Figure 1A). At both time points, hypocotyls displayed distinct zones with differing growth rates (Figure 1B). The upper quarter of the hypocotyl, termed zone 1, had the highest relative elemental growth rate (REGR) at either time point (Figure 1C). The second quarter, termed zone 2, displayed a 50% decrease in the REGR compared with zone 1 (Figure 1C), and in the lower half of the hypocotyl (zones 3 and 4) the growth rate declined to near zero. Henceforth, we will refer to zone 1 as the rapid elongation zone, since cells in this part of the hypocotyl showed the highest elongation rate. To explore how cortical microtubule organization might relate to these differing growth rates, we quantified the orientations of cortical microtubules (hereafter simply referred to as microtubules) in living cells expressing green fluorescent protein tagged a-tubulin 6 (GFP-TUA6) or Arabidopsis end binding protein 1a (EB1-GFP). Global microtubule orientations per cell were calculated from measurements of all visible microtubules (see Methods and Supplemental Figure 2 online). As predicted by Green s hypothesis (Green, 1962) and based on previous reports (Le et al., 2005; Gu et al., 2008), we expected that the rapidly elongating cells in zone 1 would have primarily transverse microtubules. RESULTS Orientations of Cortical Microtubules and CSC Trajectories on the Outer Face of the Hypocotyl Epidermis We focused on the epidermal cells of etiolated hypocotyls of Arabidopsis, which follow an acropetal growth gradient (Gendreau et al., 1997; Refrégier et al., 2004). We chose to characterize seedlings starting at 71 h after induction of germination (hours after induction [HAI]; see Methods), at which time kinematic analysis under infrared light showed that hypocotyls elongated Figure 1. Characterization of Hypocotyl Elongation at 71 and 76 HAI. (A) Mean elongation rates for n $ 10 plants are represented; error bars indicate the SD. hpi, hours post induction. (B) The hypocotyl was divided into four equivalent zones as shown. In the upper quarter (zone 1), the cells elongate most rapidly. In the second quarter (zone 2), growth declines, and it reaches near zero in the bottom half of the hypocotyl (zones 3 and 4). (C) Mean REGRs for n $ 10 plants are represented for zones 1, 2, and at each time point. Error bars indicate the SD. A gradient in REGRs is found for both time points, with zone 1 having the highest REGR.
3 2594 The Plant Cell However, only a subpopulation of the epidermal cells in zone 1 exhibited transverse alignment of microtubules on their outer surface (Figure 2A). A collar of cells with transversely aligned microtubules was observed to encompass the entire circumference of the hypocotyl; however, this collar was surprisingly short, never exceeding two cells in a file along the length of the hypocotyl (see below; Figure 3). The cells with synchronized transverse microtubules were found ;1.2 mm below the apical hook (for a total hypocotyl length of mm). Immediately above or below this region, microtubule orientations were variable and unsynchronized between cells (Figures 2B and 3). The pooled measurements of microtubule orientations from all cells in zone 1 were plotted to visualize the distribution of angles (Figure 2C). The peak in the distribution corresponded to the transverse angles, indicating that transversely oriented microtubules were slightly more abundant in this population of cells. Cells just a few millimeters farther from the apical hook, in zone 2, were still elongating with an REGR of 0.10 h 21 (Figure 1C). However, the microtubule orientations on the outer surface were unsynchronized among these cells and variable. The plot of the distribution of all measured microtubules illustrated the absence of a dominant peak at the transverse angles (Figure 2D). We next examined how the situation evolved over the course of the next 5 h. Although the hypocotyl continued to elongate at a high rate of 345 mm/h at 76 HAI (Figure 1A) and zone 1 exhibited a high REGR of 0.16 h 21 (Figure 1C), the ring of cells with synchronized transverse microtubules was no longer present. At this time point, only 23% of the cells in zone 1 had transverse microtubules (Table 1). Relatively equal proportions of cells with transverse, oblique, and longitudinal microtubule arrays were found in the rapid elongation zone (Table 1). Indeed, the pooled microtubule measurements from all cells in zone 1 yielded an isotropic distribution of orientations, where no statistically significant peaks could be detected (Figures 4A and 4B). Total microtubule orientations on the outer face were isotropic both in the small cells in the apical hook (Figure 4A) and in the cells of the rapid elongation zone (Figure 4B). Considered together, these results indicate that transversely oriented microtubules do not always predominate on the outer face of rapidly elongating cells. We also analyzed microtubule orientations in other regions of the hypocotyl. Cells in zone 2 likewise showed no bias toward transversely oriented microtubules (Figure 4C, Table 1). Finally, microtubules in cells nearer the base of the hypocotyl (zones 3 and 4), which at this stage had ceased elongation (Gendreau et al., 1997), showed a strong, statistically significant bias toward longitudinal orientations (Figure 4D, Table 1). Together, our data show that at 76 HAI, no zone with a preferential transverse microtubule orientation could be observed on the outer face of the epidermis, contrary to expectations based on the relatively high elongation rate of these hypocotyls (Figure 1A). It has been convincingly demonstrated that microtubules guide the movement of CSCs in etiolated hypocotyl epidermal cells (Paredez et al., 2006). Consequently, we expect that the trajectories of CSCs on the outer epidermal face should also be widely distributed over many angles. To test this hypothesis, we acquired time series of the movement of GFP-CESA3 labeled complexes (Desprez et al., 2007) at 76 HAI (see Supplemental Figure 3 online). In data from 15 plants, only 20 cells out of 113 (18%) showed transverse CSC trajectories (between and 22.58) on the outer surface of zone 1. These results further support the close relationship between CSC movement and microtubule orientation. In conclusion, cellulose microfibrils are not deposited strictly in a transverse orientation on the outer wall of elongating cells. Orientations of Cortical Microtubules, CSC Trajectories, and Cellulose Microfibrils on the Inner Face of the Hypocotyl Epidermis We next asked if microtubule organization on the inner epidermal face is correlated with the growth anisotropy of the hypocotyl. Remarkably, microtubules on the inner face in zone 1 at 76 HAI were consistently found in a transverse orientation (22.58 to 22.58) (Table 2, Figure 4F) and were particularly well aligned and parallel to each other (average variance per cell = 427, compared with 2508 for the outer face). Cells in the apical hook likewise had well-aligned, transverse microtubules (Table 2, Figure 4E). Every cell examined throughout zone 1 had a statistically significant bias toward the transverse orientation (Table 2). Even the more slowly elongating cells in zone 2 (Figure 1C) exhibited a strong bias toward transverse microtubules on the inner face (Figure 4G). This result was further confirmed for elongating cells at 71 HAI (Figures 2E, 2F, and 3). The only cells in which transverse microtubules did not predominate on the inner face were the cells in zones 3 and 4 at the base of the hypocotyl (Figure 4H), which grew slowly or had stopped growing (Figure 1C). In sum, only a small fraction of elongating cells had transversely oriented microtubules on the outer face, while all elongating cells had transversely oriented microtubules on the inner face (Figure 4). This indicates that microtubule orientation is consistently biased toward the transverse on the inner epidermal face in elongating cells, while the orientation varies widely on the outer face and is not correlated with growth. The transgenic lines and imaging methods used did not allow the visualization of the microtubule orientation in cortical tissue layers. We therefore performed antitubulin immunolabeling of longitudinal sections through the hypocotyl. In all sections, a transverse microtubule alignment was observed in the cortical cells (see Supplemental Figure 4 online), thus confirming previous observations for microtubules (Furutani et al., 2000) and cellulose microfibrils (Refrégier et al., 2004) in this cell type. In addition to drawing inferences from populations of cells, we also acquired confocal z-stacks of entire cells to determine if outer and inner surfaces shared the same alignments (175 cells). No correlation was found between the preferred orientation on either face of the epidermis in zone 1 (Figures 5A and 5B), although some microtubules were longitudinal or oblique on both faces of the cell in the mature cells of zones 3 and 4 (Figures 5C and 5D). By generating orthogonal projections of the z-stacks, we could observe continuous, presumably bundled microtubules that extended from the outer face to the inner face (Figure 6A). Microinjection of fluorescent tubulin and immunofluorescence staining of tubulin in other plant species led to similar observations (Flanders et al., 1989; Yuan et al., 1995). While the microtubules facing the inner tangential and anticlinal walls exhibited similar orientations, the curvature of the continuous
4 Growth Anisotropy in Higher Plants 2595 Figure 2. Microtubules at the Inner Face Align Independently of Those on the Outer Wall of Etiolated Hypocotyls. Quantifications were performed in zones 1 and 2 of etiolated hypocotyls at 71 HAI in GFP-TUA6 expressing plants. Bars = 10 mm. (A) A collar of synchronized cells with transversely oriented microtubules is found in zone 1. (B) Cells just below those pictured in (A) lack synchronization and have variably oriented microtubules. (C) and (D) Distributions of microtubule orientations at 71 HAI, in zone 1 on the outer epidermal face ([C], 34 cells from nine plants) and in zone 2 on the outer epidermal face ([D], 53 cells from 14 plants). The total number of microtubules measured (N) is indicated in the top right corner of each graph. Angles corresponding to the transverse orientation (22.58 $u> 22.58) are highlighted in gray at the center of each distribution (see Supplemental Figure 2 online for the definition of orientations). In zone 2, no dominant peak is found at the transverse angles, and the distribution is relatively isotropic. (E) Transversely oriented microtubules on the inner face in zone 1 at 71 HAI. (F) The distributions of orientations of microtubules on the inner epidermal face in zones 1 and 2 (pooled measurements from 94 cells in 11 plants). The total number of microtubules measured (N) is indicated in the top right of each graph. Angles corresponding to the transverse orientation (22.58 $u> 22.58) are highlighted in gray at the center of the graph.
5 2596 The Plant Cell Figure 3. Only a Small Fraction of Cells Have Transversely Oriented Microtubules on the Outer Face, While All Elongating Cells Have Transversely Oriented Microtubules on the Inner Face. Images of a single representative GFP-TUA6 expressing plant showing microtubule orientations on the outer face (left panel) and inner face (right panel) throughout zone 1 at 71 HAI. The insets show enlarged views of the boxed regions. Bars = 10 mm. bundles changed abruptly at the limit between the outer face and anticlinal walls (Figure 6A). It seems therefore unlikely that microtubule orientation is regulated by a property of the microtubules themselves and suggests that their orientation may be regulated by a property of the local environment. Differences in microtubule orientation on the outer and inner faces were consistent regardless of whether we visualized the GFP-TUA6 marker or the EB1-GFP marker (Figure 6B). Interestingly, our observations of EB1-GFP served to demonstrate that microtubules are also highly dynamic on the inner face. We quantified the velocity of EB1 plus-end comets on both faces of the epidermis (Figure 6C). The mean velocity on the outer face was mm/min (n = 1119 comets), similar to previous reports (Chan et al., 2003; Crowell et al., 2009). Similarly, the mean velocity on the inner face was mm/min (n = 1825 comets), which was not significantly different from the velocity measured on the outer face at a 99% confidence interval. Together, this indicates that a mechanism must continually operate to orient nascent microtubules in the transverse orientation on the inner face, but the dynamics of microtubule polymerization are unlikely to be at the origin of these differences in orientation. We next sought to determine if CSC trajectories on the inner epidermal face reflect the observed orientations of microtubules. In 10 independent experiments (15 cells from 12 different plants), and regardless of the orientation of the trajectories on the outer face (Figure 7A), we consistently observed CSCs migrating along perfectly transverse trajectories on the inner face of cells in the rapid elongation zone (Figure 7B). This result suggests that cellulose microfibrils are deposited in the transverse orientation on the inner face of these cells. We further investigated the organization of the cellulose microfibrils themselves using scanning electron microscopy and FESEM, techniques that permit visualization of cellulose microfibril orientation in the most recently deposited cell wall layer. Transverse arrays of cellulose microfibrils were evident on the inner walls of epidermal cells in zones 1 and 2 (see Supplemental Figure 5 online), in accordance with previously published results (Sugimoto et al., 2000; Refrégier et al., 2004). We were able to occasionally obtain epidermal cells in which the wall had torn open and was retained after fixation (Figure 7C). In these cases, it was possible to visualize the cellulose microfibrils on all faces of the same cell, from the outer wall to the inner wall (Figure 7D). Interestingly, cellulose microfibrils could be found in oblique or longitudinal orientations on the outer wall, quickly transitioned through mixed orientations, and finally were found in perfectly transverse orientations on the inner wall and portions of the radial side wall (Figure 7D). These findings confirm the predictions based on our measurements of microtubule orientation and illustrate that cellulose microfibrils can be deposited in strikingly different orientations on the outer and inner walls of the same cell. Relationship between Cortical Microtubules in the Inner Tissues and Cell Expansion Our data indicate that deposition of transversely oriented cellulose microfibrils in the outer epidermal wall is spatially and Table 1. Results of the Classification of Individual Cells According to the Major Orientation of Their Microtubules at 76 HAI in the Outer Face Outer Face Longitudinal Oblique Transverse Oblique + Mixed Apical hook 12 (17%) 17 (24%) 10 (14%) 20 (28%) 12 (17%) Zone 1 14 (20%) 14 (20%) 16 (23%) 11 (16%) 14 (20%) 2 7 (16%) 10 (23%) 3 (7%) 12 (26%) 10 (23%) 3 and 4 49 (83%) 5 (8%) 1 (2%) 3 (5%) 1 (2%) See Supplemental Figure 2 online for a definition of the orientations denoted in the tables. Each cell was classified by examination of its microtubule distribution and by testing if any major peaks were significant (see Methods). The number of cells in each class is noted, with the percentage of the total number of cells shown in parentheses. Cells with more than one peak in their distribution were counted according to which peak was significant or counted as mixed if two peaks were significant.
6 Growth Anisotropy in Higher Plants 2597 temporally restricted. By contrast, deposition of transverse cellulose microfibrils is maintained in the inner epidermal wall over a much larger region of the hypocotyl for longer periods of growth. The microtubule orientation on the inner face is perpendicular to the cell elongation axis, as would be predicted for elongating cells. Furthermore, the degree of anisotropy on the inner face is closely correlated with the growth status of the cells. Deposition of transversely oriented cortical microtubules on the inner epidermal wall precedes rapid cell elongation (in the apical hook, Figure 4E) and is maintained during cell elongation (Figures 4F and 4G), but gradually dissipates as the cells cease to grow (Figure 4H). These findings support the hypothesis that the walls inside the organ primarily control the growth direction, whereas the extensibility of the thick external epidermal walls limits growth independently of the growth direction. To test this hypothesis, one has to envisage a treatment that reduces the anisotropy of inner walls, including the internal tangential epidermal wall, without changing the outer epidermal wall. Such a treatment should lead to increased radial organ expansion or not depending on whether the growth anisotropy is regulated by the inner or outer walls of the organ, respectively. Previous observations of changes in microtubule organization in plants expressing a GFP-fused microtubule binding domain of the mammalian microtubule-associated protein 4 (GFP-MBD) prompted us to examine in detail transgenic lines stably overexpressing this protein (Marc et al., 1998). On the outer epidermal face, microtubules in GFP-MBD expressing plants showed, like the GFP-TUA6 or EB1-GFP control lines at the same growth stage, parallel arrays that were widely distributed over all angles, but with a slight bias toward negative oblique angles (Figure 8A). By contrast, on the inner epidermal face of the GFP-MBD line, microtubule orientations showed a highly reduced anisotropy: they were less transverse and less parallel than in the GFP-TUA6 line (average variance per cell = 911 for GFP-MBD and 427 for GFP-TUA6), with a bias toward oblique angles (Figure 8B, compare with Figure 5B). In conclusion, overexpression of GFP-MBD reduced the anisotropy Figure 4. Microtubules Are Transverse on the Inner Face of Elongating Cells. The distribution of orientations of microtubules at 76 HAI on the outer epidermal face ([A] to [D]) and inner epidermal face ([E] to [H]; see Supplemental Figure 1 online). The distributions present pooled measurements of microtubules from multiple cells in multiple plants (outer face: n = 243 cells, n = 33 plants; inner face: n = 205 cells, n = 28 plants). The total number of microtubules measured (N) is indicated in the top right corner of each graph. The growth zones correspond to those outlined in Figure 1B. Microtubule orientations are shown for the apical hook region of zone 1 ([A] and [E]), the rapid elongation zone ([B] and [F]), zone 2 ([C] and [G]), and zones 3 and 4 ([D] and [H]). Angles corresponding to the transverse orientation (22.58 $u> 22.58) are highlighted in gray at the center of each distribution (see Supplemental Figure 2 online for the definition of orientations). Table 2. Results of the Classification of Individual Cells According to the Major Orientation of Their Microtubules at 76 HAI in the Inner Face Inner Face Longitudinal Oblique Transverse Oblique + Mixed Apical hook 0 (0%) 0 (0%) 17 (100%) 0 (0%) 0 (0%) Zone 1 0 (0%) 0 (0%) 45 (100%) 0 (0%) 0 (0%) 2 0 (0%) 1 (2%) 43 (91%) 3 (6%) 0 (0%) 3 and 4 22 (47%) 7 (15%) 8 (17%) 6 (13%) 4 (9%) See Supplemental Figure 2 online for a definition of the orientations denoted in the tables. Each cell was classified by examination of its microtubule distribution and by testing if any major peaks were significant (see Methods). The number of cells in each class is noted, with the percentage of the total number of cells shown in parentheses. Cells with more than one peak in their distribution were counted according to which peak was significant or counted as mixed if two peaks were significant.
7 2598 The Plant Cell expansion in GFP-MBD hypocotyls compared with wild-type and GFP-TUA6 lines (Figures 8D and 8E; see Supplemental Figure 6 online), suggesting that the anisotropy of internal cell walls is indeed required to maintain the growth direction of the organ. Cortical cells showed the largest surface increase (75%) on sections, compared with epidermal cells, which showed a 50 or 20% surface increase, respectively (Figure 8E), depending on whether or not they faced anticlinal cortical walls (Figure 8F). These increases in radial expansion were accompanied by a reduced cell elongation (in zone 1, mean cell length GFP- MBD = 233 mm; GFP-TUA6 = 312 mm; P value = )anda reduced hypocotyl length (mean length GFP-MBD = 8.62 mm; GFP-TUA6 = 9.35 mm; P value = 0.01). DISCUSSION Figure 5. Microtubules Have Different Orientations on Opposite Faces of the Same Cells. Maximum projected z-stacks showing GFP-TUA6 labeled microtubules on the outer epidermal face (left panel) and inner epidermal face (right panel) of the same cells at 76 HAI. Images acquired in the apical hook (A), zone 1 (B), zone 3 (C), and zone 4 (D) are shown. The transverse microtubule alignment on the inner face is progressively lost as the cells cease elongating ([C] and [D], right panels). Images are representative of n > 50 hypocotyls sampled. Bar = 10 mm. in the orientation of microtubules on the inner epidermal walls and possibly walls of other internal cell layers, while changing to a lesser extent the microtubule orientation on the outer epidermal wall (Figure 8C). This suggests that GFP-MBD did not induce major chances in the architecture of the outer epidermal walls, although we cannot entirely rule out that the extensibility of the walls has changed through an unknown but microtubule-dependent mechanism as observed for instance by Lucas et al. (2011). Interestingly, scanning electron microscopy and transverse sections midway through the hypocotyl showed increased radial In this report, we provide a full characterization of microtubule orientations, CSC trajectories, and newly deposited cellulose microfibril orientations in the epidermis of Arabidopsis etiolated hypocotyls. This led to a number of interesting observations: First, we observed a perfect parallel between the orientation of microtubule orientations, CSC trajectories, and newly deposited cellulose microfibrils on the external and internal faces of the epidermal cells. Although technical limitations prevented us from being able to visualize all three components simultaneously, our quantitative statistical approach allowed us to draw firm conclusions. These new data confirm and extend previous findings that microtubules guide the trajectories of GFP-labeled CSCs, which in turn orient the deposition of the microfibrils. By imaging CSCs and microtubules on the inner epidermal surface, we were able to visualize cellulose deposition on the same wall surface that has been characterized by FESEM (Sugimoto et al., 2000; Refrégier et al., 2004). It should be kept in mind that after deposition, the orientation of microfibrils could change during growth according to the multinet growth theory (Roelofsen and Houwink, 1953). This is also true for the Arabidopsis dark-grown hypocotyl, in which, during a rapid elongation phase, the outer cell wall thins and cellulose microfibrils in older wall layers reorient toward a longitudinal orientation (Refrégier et al., 2004; Derbyshire et al., 2007; Anderson et al., 2010). Second, microtubules, CSC trajectories, and newly deposited cellulose microfibrils on the outer wall of epidermis cells did not show a preferential orientation during most of the elongation period. This fits with observations on pea (Pisum sativum) internodes, dandelion (Taraxacum officinale) peduncles, oat (Avena sativa) coleoptiles, and soybean (Glycine max) hypocotyls, which showed that these organs were able to elongate despite a net longitudinal orientation of cellulose microfibrils in the outer epidermal walls (Iwata and Hogetsu, 1989; Paolillo, 2000). This is also in agreement with the recent study on the mor1 mutant showing that there are different orientations of cellulose microfibrils in outer walls (Fujita et al., 2011). In addition, the outer walls of the hypocotyl epidermis in multiple species have been shown to consist of layers of cellulose in a complete range of orientations (Roland et al., 1975; Neville and Levy, 1984; Refrégier et al., 2004; reviewed in Neville et al., 1976). A recent study on lightgrown Arabidopsis seedlings shows that microtubules on the
8 Growth Anisotropy in Higher Plants 2599 Figure 6. The Same Microtubules Can Extend from the Outer to the Inner Face and Have Similar Dynamics. (A) Montage of maximum and orthogonal projections of a confocal z-stack showing GFP-TUA6 labeled microtubules on the inner face, side region, and outer face of one epidermal cell in zone 3. The arrowheads indicate example microtubule bundles that extend from the inner face to the outer face of the cell, with a sudden change in orientation on the outer face (right side of image). Other unmarked examples are also visible. (B) Maximum projected 5-min time series acquired at a 20-s interval showing the trajectories of EB1-GFP comets following the polymerizing ends of microtubules. EB1-GFP trajectories on the outer face (left panel) and inner face (right panel) of the same epidermal cell. Bars = 10 mm. (C) Velocity distribution of EB1-GFP comets on the outer and inner faces (outer: n = 1119; inner: n = 1825). outer face of the epidermis and cellulose synthase trajectories in the plasma membrane cycle through a number of different orientations, thus producing a polylamellate cell wall architecture (Chan et al., 2010). The rotary movements of the microtubule array have also been proposed for the hypocotyl epidermis of etiolated sunflower (Helianthus annuus; Hejnowicz, 2005). Although observation of long-term microtubule behavior is not feasible on dark-grown seedlings, our observations of the wide distribution in angles on the outer face of elongating epidermal cells suggest that microtubules may also undergo rotary movements in etiolated conditions and thus generate the polylamellate external cell wall found in immature hypocotyl epidermal cells. Third, and this is the most surprising finding, growing epidermal cells are polarized in the orientation of microtubules, with no preferential orientation on the outer face and a strictly transverse orientation on the inner face in elongating cells. Differences in microtubule orientation on opposing faces of the epidermis have been noted previously (Busby and Gunning, 1983; Flanders et al. 1989; Yuan et al., 1995; Hejnowicz et al., 2000; Barton and Overall, 2010), but their relationship to organ expansion was not established. As discussed below, the transverse alignment of microtubules on the inner epidermal face has implications for the regulation of the growth anisotropy of the organ and raises new questions on the establishment and maintenance of such a polarity in the epidermis. Regulation of Growth Anisotropy by Inner Tissues and Not by Outer Epidermal Walls in the Hypocotyl The finding that microtubules and microfibrils are transversely oriented on the inner epidermal wall (Figures 4 and 7) extends previous observations that microtubules (Furutani et al., 2000) and newly deposited microfibrils (Refrégier et al., 2004) are transverse in growing cortical cells of the Arabidopsis hypocotyl. Transversely oriented microfibrils have also been observed in the parenchyma of other species (Hogetsu, 1986; Iwata and Hogetsu, 1989; Abe et al., 1995; Baskin et al., 1999; Crow and Murphy, 2000). Likewise, in Arabidopsis roots, transversely
9 2600 The Plant Cell Figure 7. Cellulose Synthase Trajectories and Cellulose Microfibrils Are Also Oriented Transversely on the Inner Face. (A) and (B) Representative average projected 10-min time series acquired at a 30-s interval showing the trajectories of GFP-CESA3 labeled CSCs in the same cell in zone 1 on the outer (A) and inner (B) face. On the inner face, the CSCs migrate along strictly transverse trajectories (B). Bar = 5 mm. (C) Low-magnification scanning electron microscopy image of an etiolated hypocotyl, showing an epidermal cell wall that has torn open. The numbers indicate positions subsequently analyzed at higher magnification. (D) Montage of several high magnification images in the boxed region in (C), showing the gradual change in cellulose microfibril orientation and texture from the outer wall to the inner wall. Bar = 500 nm. aligned cellulose microfibrils are found on the inner epidermal walls in elongating cells (Sugimoto et al., 2000). However, cortical microtubule organization seems to be distinct in roots and shoots, since microtubules were shown to have a transverse orientation on both inner and outer faces of the root epidermis throughout the cell elongation zone (Sugimoto et al., 2000). When the inner tissue layers are separated from the epidermal layer, the inner tissues have a tendency to elongate, while the epidermis bends toward its outer surface (reviewed in Kutschera and Niklas, 2007). This can be illustrated both in isolated tissues and in epidermal cells of Arabidopsis mutants with cell adhesion defects (Bouton et al., 2002). These observations form the basis of the tissue-tension model of growth (Peters and Tomos, 1996). This model posits that the highly anisotropic cell walls inside the organ transmit a large axial force to the epidermis, which drives elongation of the organ despite an outer cell wall that lacks anisotropy (Baskin, 2005). Interestingly, consistent with a critical role for internal tissues in the regulation of growth directionality, hypocotyl cortex and endodermis show increased radial expansion in mutants for the microtubule-associated protein SPIRAL1, in which the orientation of microtubules is selectively perturbed in ground tissues (Furutani et al., 2000). Furthermore, we showed
10 Growth Anisotropy in Higher Plants 2601 Figure 8. Microtubule Orientation Is Perturbed on the Inner Epidermal Face of GFP-MBD Expressing Plants. (A) and (B) Extended depth of field images of GFP-MBD labeled microtubules on the outer (A) and inner (B) epidermal face of the same cells in zone 1. The microtubules are abnormally oriented in oblique angles on the inner face ([B]; compare with Figure 5B, right panel). Bar = 10 mm. (C) The distribution of orientations of microtubules on the outer and inner epidermal face in zone 1 of GFP-MBD expressing plants at 76 HAI (pooled measurements of 78 cells from three plants). The total number of microtubules measured (N) is indicated in the top right of each graph. Angles corresponding to the transverse orientation (22.58 $u> 22.58) are highlighted in gray at the center of each distribution. The microtubules on the inner face of the GFP-MBD line are less parallel and more obliquely oriented than in the GFP-TUA6 line (cf. with Figure 4F). (D) Photographs of hypocotyl cross sections from a Wassilewskija wild-type (top) and GFP-MBD (bottom) line. The asterisk shows an epidermal cell facing an anticlinal cortical wall. Bar = 100 mm. (E) Distributions of epidermal and cortical cell surface areas measured in the Wassilewskija wild-type (wt)and GFP-MBD line (n = 75 to 160 cells, n =4 plants). Note the significant overall increase in cortical cell surface area in the GFP-MBD line. (F) Distributions of epidermal cell surface areas for the subpopulation facing anticlinal inner walls (n = 67 cells, n = 4 plants). This subpopulation of epidermal cells undergoes radial swelling in the GFP-MBD line.
11 2602 The Plant Cell in this study that expression of GFP-MBD preferentially perturbed microtubule anisotropy at the inner epidermal face and caused increased radial expansion of the organ. It is interesting to note that epidermal cells in this line also showed increased radial expansion. The extent of the radial expansion (50 or 20% depending on whether or not they faced anticlinal cortical walls) was less pronounced than that of the cortical cells (75%) and was proportional to the fraction of the cell surface that is exposed to the highly anisotropic inner periclinal walls ( % [SE] and % of their perimeter for the two classes of epidermal cells). These observations are consistent with the hypothesis that the reduced anisotropy of the inner periclinal walls of the epidermis and cortex in the GFP-MBD line is responsible for the increased radial expansion of the organ. In conclusion, the results support the hypothesis that the directionality of organ expansion is regulated by the highly anisotropic walls inside the organ rather than the outer epidermal walls. In this view, the controlled yielding of the thick external wall, in part through the secretion of wall remodeling agents (Pelletier et al., 2010), will regulate the growth rate of the organ independently of the growth direction. While this manuscript was under review, Chan et al. (2011) published a similar study on light-grown hypocotyls with essentially the same conclusions as our study on dark-grown hypocotyls. They invariably observed a transient transverse orientation of the cortical microtubules facing the outer tangential wall, concomitant with growth acceleration. In our study, however, we showed that rapid elongation growth could occur even in the absence of such a transient fixation of the transverse orientation on the outer face. In addition, we further corroborate the proposed model for the regulation of growth anisotropy of the hypocotyl by the experimental perturbation of the cortical microtubules facing the inner tangential epidermal wall. The Epidermis Is Polarized for Cortical Microtubule Orientation One intriguing question raised by this study is how the coordinated transverse alignment of microtubules on the inner face is maintained, while microtubules cycle between different orientations on the outer face. This is not related to differences in the dynamics of microtubule polymerization, since no difference in the motility of the plus end marker EB1-GFP could be observed on the two faces of the epidermal cells. It is also unlikely to be related to intrinsic properties of the microtubules themselves, since the same microtubules can be visualized extending from the outer to inner face. It is possible that mechanical force fields may underlie the synchronous alignment of microtubules transverse to the elongation axis in cortical cells and on the inner epidermal face. For example, in the epidermis of the shoot apical meristem, microtubules align parallel to the axis of maximal stress, resulting in the coordinated alignment of microtubules (Hamant et al., 2008). Microtubules in peeled epidermis were also found to reorient depending on the stress anisotropy applied to the cells (Hejnowicz et al., 2000). Such a mechanical feedback mechanism would explain the maintenance of the cell wall anisotropy of the inner cell walls, but it does not explain the establishment of the transverse orientation of microtubules in inner layers while excluding the outer face. In etiolated hypocotyls, the establishment of the transverse orientation appears to take place well before elongation, since we consistently found that cells in the apical hook also showed transverse microtubules on the inner face before elongating (Figure 4E). Interestingly, the situation in light-grown hypocotyls appears to be different, since microtubules initially rotate on the inner and outer faces preceding elongation (Chan et al., 2011). In conclusion, our quantitative analysis of microtubules, CSC trajectories, and cellulose microfibril orientations on both the outer and inner faces of the shoot epidermis presents a coherent model for the regulation of growth anisotropy in this tissue. It will now be interesting to see how and when exactly during hypocotyl development the epidermis becomes polarized in its cortical microtubule orientation. METHODS Plant Material and in Vitro Growth Conditions Transgenic Arabidopsis thaliana plants expressing GFP-TUA6 (Ueda et al., 1999), end binding protein 1a fused to GFP (EB1-GFP; Chan et al., 2003), GFP fused to the cellulose synthase catalytic subunit 3 (GFP- CESA3; Desprez et al., 2007), or GFP fused to the microtubule binding domain of microtubule-associated protein 4 (GFP-MBD; Marc et al., 1998) were sown in chambers, as previously described (Chan et al., 2007), or on plates containing Estelle and Somerville medium (Estelle and Somerville, 1987) without Suc. Seeds were subjected to cold treatment at 48C for 48 h, exposed to fluorescent white light for 5 h, and cultured for an additional 66 or 73 h in darkness at 208C. The number of hours after induction was counted from the start of the light exposure. The GFP- TUA6, EB1-GFP, and GFP-CESA3 transgenic lines have been extensively studied previously and exhibit growth patterns indistinguishable from those of the wild type (Ueda et al., 1999; Chan et al., 2003; Desprez et al., 2007). Cell and Hypocotyl Length Measurements Hypocotyl length measurements were performed on seedlings grown for 71 or 76 h in darkness on Estelle and Somerville medium (Estelle and Somerville, 1987) without Suc. Hypocotyl length was measured on photos in ImageJ. To determine the REGR, the hypocotyls were photographed under infrared light using a Nikon D70 camera with a 50-mm macro-objective, from 71 to 73 HAI or from 76 to 78 HAI. Each hypocotyl was divided into four equivalent zones: the uppermost quarter including the apical hook (zone 1), the second quarter (zone 2), and the lower half (zones 3 and 4). In each zone, the strain was determined by measuring the change in distance between two arbitrarily chosen natural marks at the hypocotyl surface (i.e., anticlinal cell walls) at the two time points, divided by the distance at the first time point. GFP-MBD and GFP-TUA6 seedlings were visualized using a Leica DMRB microscope. Cell lengths in zone 1 were measured using ImageJ. Cell Wall Preparation and FESEM FESEM was performed as described for Figure 6B (Refrégier et al. 2004). For Figure 7D, seedlings were treated as described by Sugimoto et al. (2000), except for the fixation (seedlings were incubated in 3% glutaraldehyde in 0.1 M phosphate buffer, ph 7.2, at room temperature for at least 3 h and then seedlings were incubated in 20% Suc for at least 1 h). For scanning electron microscopy, 3-d-old etiolated seedlings were imaged with a Hirox SH-1500 (Hirox Europe-Jyfel) microscope at 2308C.
12 Growth Anisotropy in Higher Plants 2603 Image contrast was improved using Photoshop CS (Adobe Systems). Immunolocalization Using Indirect Immunofluorescence Analysis Seedlings were fixed in 1.5% formaldehyde, 0.5% glutaraldehyde, and 0.05% triton in microtubule-stabilizing buffer (MTSB) and put under vacuum for 2 h. Samples were dehydrated, waxed, and dewaxed as described (Masclaux-Daubresse et al., 2006). Longitudinal sections (12 mm) were prepared using a microtome (Jung RM2055; Leica) and air-dried on polylysine-coated glass slides. Samples were blocked with 5% (w/v) BSA in PBS and incubated with mouse antibody against tubulin (Monoclonal B5 1-2; Sigma-Aldrich) diluted at 1:1000. After three washes of 5 min each in PBS, slides were incubated with goat anti-mouse IgG conjugated with Alexa 488 (Molecular Probes) diluted at 1:200. Cross Sections and Surface Area Measurements Seedlings were fixed in 2% (w/v) formaldehyde, 1% (w/v) glutaraldehyde, and 1% caffeine and put under vacuum for 15 min. Seedlings were embedded in historesin (Technovit 7100; Kulzer) following the manufacturer s instructions. Sections 5 mm thick were cut using a microtome (Jung RM2055; Leica). The cross sections were visualized using a Leica DMRB microscope. Surface areas were measured using ImageJ. Spinning Disk Microscopy Imaging of 3-d-old, live seedlings was performed on an Axiovert 200M microscope (Zeiss) equipped with a Yokogawa CSU22 spinning disk, Zeiss 3100/1.4 numerical aperture oil objective, and Andor EMCCD ixon DU 895 camera (Plateforme d Imagerie Dynamique, Institut Pasteur) or on an Axio Observer Z1 Zeiss microscope (Zeiss) equipped with a Yokogawa CSU-X1 spinning disk, Zeiss 3100/1.4 numerical aperture oil objective, and Roper EMCCD Quantum 512C (Plateforme d Imagerie Institut Jean- Pierre Bourgin-Institut National de la Recherche Agronomique, Versailles). A 488-nm diode-pumped solid-state laser was used for excitation of GFP, and emission was collected using a band-pass 488/25 filter (Semrock). Z-stacks were acquired with an interval of 0.3 to 0.5 mm and binning. Time series were acquired with a time interval of 10 to 15 s and a z-slice interval of 0.3 mm. For visualization of CESA trajectories on the inner epidermal face, laser intensity was increased and the time interval optimized to reduce photobleaching. To avoid changes in microtubule orientation due to exposure to laser light, all images were acquired <5 min after mounting the slide. Cell viability was verified by monitoring cytoplasmic streaming. Image Analysis The orientation of all visible microtubules was quantified using ImageJ software (W. Rasband, National Institutes of Health) on maximum or average projections of z-stacks. Measurements were adjusted to represent the orientation relative to the cell long axis (see Supplemental Figure 3 online for the definition of orientations). Orthogonal z-projections were created using the reslice tool in ImageJ. Extended depth of field images were prepared using an ImageJ plugin (Biomedical Imaging Group; Forster et al., 2004). Figures and drawings were prepared with Photoshop CS (Adobe Systems). Statistical analysis of microtubule orientations was performed using the Physics Analysis Workstation (PAW) software (CERN). Distributions of all microtubule orientations for each cell were generated using PAW. Each cell was classified according to the major orientation of its microtubules using a statistical test based on the position and height of the peaks in the distribution (see Supplemental Figure 3 online for the definition of orientations). Statistically significant peaks or troughs in the distributions were detected using a binning of 18. A peak or trough was considered significant if the difference of the absolute value of the count in that bin and the mean count was >2 times the SD (s). The statistical test was calibrated visually by examining the distributions of microtubules for each individual cell and the raw images. Only cells for which 30 or more measurements could be obtained were analyzed. Supplemental Data The following materials are available in the online version of this article. Supplemental Figure 1. Cross Section through an Arabidopsis Hypocotyl, Showing the Two Distinct Regions of the Epidermis Studied in This Report. Supplemental Figure 2. Definition of Angles of Orientation Relative to the Cell Long Axis. Supplemental Figure 3. Representative Average Projected 10-min Time Series Acquired at a 20-s Interval, Showing the Trajectories of GFP-CESA3 Labeled Cellulose Synthase Complexes on the Outer Face of Zone 1 Epidermal Cells at 76 HAI. Supplemental Figure 4. Representative Maximum Projected z-stack Showing Antitubulin Immunostaining of Cortical Cells in Zone 1 of the Hypocotyl at 76 HAI. Supplemental Figure 5. Representative FESEM Images of Transverse Cellulose Microfibrils on the Inner Wall of an Epidermal Cell in Zone 1 (Left) and Zone 2 (Right) at 76 HAI (Courtesy of G. Refrégier). Supplemental Figure 6. Scanning Electron Microscopy Images of 3-d-Old Etiolated Hypocotyls of Arabidopsis, Expressing Either GFP- TUA6 or GFP-MBD. ACKNOWLEDGMENTS We thank the three anonymous reviewers for offering constructive comments. We thank Clive Lloyd and Jordi Chan (John Innes Centre, Norwich, UK) for stimulating discussions and for their critical reading of the manuscript. We thank G. Refrégier for contributing FESEM images of the inner epidermal face of Arabidopsis hypocotyls. We also thank J.-L. Lecouey (Laboratoire de Physique Corpusculaire, Caen, France) for assisting us in creating graphs with PAW. We thank O. Grandjean (PlateformedeCytologieetd ImagerieVégétale, Institut Jean-Pierre Bourgin, Institut National de la Recherche Agronomique) for technical assistance. E.F.C. was supported by the National Agency for Research Project IMACEL (ANR-06-BLAN-0262). Part of this work was supported by the European Union Framework Program 6 (FP6) CASPIC NEST-CT and FP6 program AGRON-OMICS. The Plateforme de Cytologie et d Imagerie Végétale is supported in part by the Région Ile-de-France. Received May 12, 2011; revised June 10, 2011; accepted June 20, 2011; published July 8, REFERENCES Abe, H., Funada, R., Ohtani, J., and Fukazawa, K. (1995). Changes in the arrangement of microtubules and microfibrils in differentiating conifer tracheids during the expansion of cells. Ann. Bot. (Lond.) 75: Anderson, C.T., Carroll, A., Akhmetova, L., and Somerville, C. (2010). Real-time imaging of cellulose reorientation during cell wall expansion in Arabidopsis roots. Plant Physiol. 152:
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
Alteration of Oriented Deposition of Cellulose Microfibrils by Mutation of a Katanin-Like Microtubule-Severing Protein
 The Plant Cell, Vol. 14, 2145 2160, September 2002, www.plantcell.org 2002 American Society of Plant Biologists Alteration of Oriented Deposition of Cellulose Microfibrils by Mutation of a Katanin-Like
The Plant Cell, Vol. 14, 2145 2160, September 2002, www.plantcell.org 2002 American Society of Plant Biologists Alteration of Oriented Deposition of Cellulose Microfibrils by Mutation of a Katanin-Like
The rotation of cellulose synthase trajectories is microtubule dependent and influences the texture of epidermal cell walls in Arabidopsis hypocotyls
 3490 Research Article The rotation of cellulose synthase trajectories is microtubule dependent and influences the texture of epidermal cell walls in Arabidopsis hypocotyls Jordi Chan 1, Elizabeth Crowell
3490 Research Article The rotation of cellulose synthase trajectories is microtubule dependent and influences the texture of epidermal cell walls in Arabidopsis hypocotyls Jordi Chan 1, Elizabeth Crowell
THE PENNSYLVANIA STATE UNIVERSITY THE SCHREYER HONORS COLLEGE DEPARTMENT OF BIOCHEMISTRY AND MOLECULAR BIOLOGY
 THE PENNSYLVANIA STATE UNIVERSITY THE SCHREYER HONORS COLLEGE DEPARTMENT OF BIOCHEMISTRY AND MOLECULAR BIOLOGY The Effects of 1-Butanol on Cellulose Synthase Dynamics and Cellulose Production in Arabidopsis
THE PENNSYLVANIA STATE UNIVERSITY THE SCHREYER HONORS COLLEGE DEPARTMENT OF BIOCHEMISTRY AND MOLECULAR BIOLOGY The Effects of 1-Butanol on Cellulose Synthase Dynamics and Cellulose Production in Arabidopsis
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
Regulation of Growth Anisotropy in Well-Watered and Water-Stressed Maize Roots. II. Role of Cortical Microtubules and Cellulose Microfibrils 1
 Plant Physiology, February 1999, Vol. 119, pp. 681 692, www.plantphysiol.org 1999 American Society of Plant Physiologists Regulation of Growth Anisotropy in Well-Watered and Water-Stressed Maize Roots.
Plant Physiology, February 1999, Vol. 119, pp. 681 692, www.plantphysiol.org 1999 American Society of Plant Physiologists Regulation of Growth Anisotropy in Well-Watered and Water-Stressed Maize Roots.
Dynamic Microtubules and the Texture of Plant Cell Walls
 CHAPTER SEVEN Dynamic Microtubules and the Texture of Plant Cell Walls Clive Lloyd Contents 1. Introduction 288 1.1. The multinet-growth hypothesis and hoop reinforcement 289 1.2. Microtubule hoops 290
CHAPTER SEVEN Dynamic Microtubules and the Texture of Plant Cell Walls Clive Lloyd Contents 1. Introduction 288 1.1. The multinet-growth hypothesis and hoop reinforcement 289 1.2. Microtubule hoops 290
BOTANY LAB #1 MITOSIS AND PLANT TISSUES
 Mitosis and cytokinesis in plants BOTANY LAB #1 MITOSIS AND PLANT TISSUES In plants the formation of new cells takes place in specialized regions of meristematic tissue. Meristematic tissues contain immature,
Mitosis and cytokinesis in plants BOTANY LAB #1 MITOSIS AND PLANT TISSUES In plants the formation of new cells takes place in specialized regions of meristematic tissue. Meristematic tissues contain immature,
Plant Structure. Lab Exercise 24. Objectives. Introduction
 Lab Exercise Plant Structure Objectives - Be able to identify plant organs and give their functions. - Learn distinguishing characteristics between monocot and dicot plants. - Understand the anatomy of
Lab Exercise Plant Structure Objectives - Be able to identify plant organs and give their functions. - Learn distinguishing characteristics between monocot and dicot plants. - Understand the anatomy of
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
Quantitative Changes in Microtubule Distribution Correlate with Guard Cell Function in Arabidopsis
 RESEARCH ARTICLE Quantitative Changes in Microtubule Distribution Correlate with Guard Cell Function in Arabidopsis William R. Eisinger a,b, Viktor Kirik c, Charlotte Lewis a, David W. Ehrhardt b and Winslow
RESEARCH ARTICLE Quantitative Changes in Microtubule Distribution Correlate with Guard Cell Function in Arabidopsis William R. Eisinger a,b, Viktor Kirik c, Charlotte Lewis a, David W. Ehrhardt b and Winslow
Bring Your Text to Lab!!!
 Bring Your Text to Lab!!! Vascular Plant Anatomy: Flowering Plants Objectives: 1. To observe what the basic structure of vascular plants is, and how and where this form originates. 2. To begin to understand
Bring Your Text to Lab!!! Vascular Plant Anatomy: Flowering Plants Objectives: 1. To observe what the basic structure of vascular plants is, and how and where this form originates. 2. To begin to understand
The mode of development in animals and plants is different
 The mode of development in animals and plants is different Outcome of animal embryogenesis is a mini edition of the adult Outcome of plant embryogenesis is a simple structure with -root apical meristem
The mode of development in animals and plants is different Outcome of animal embryogenesis is a mini edition of the adult Outcome of plant embryogenesis is a simple structure with -root apical meristem
Bald cypress Taxodium distichum in a swamp in North Carolina
 Bald cypress Taxodium distichum in a swamp in North Carolina Bald cypress is another deciduous gymnosperm. It is native to the SE United States. It can tolerate a wide range of soil conditions. It is not
Bald cypress Taxodium distichum in a swamp in North Carolina Bald cypress is another deciduous gymnosperm. It is native to the SE United States. It can tolerate a wide range of soil conditions. It is not
ROOT STRUCTURE: EXTERNAL ROOT
 ROOT STRUCTURE: EXTERNAL ROOT Material: Botany I cards #9 - #12, #13 - #18, #19 - # 23 Presentation: 1. The external parts of the root have been introduced to the children: the root hairs, the root tips,
ROOT STRUCTURE: EXTERNAL ROOT Material: Botany I cards #9 - #12, #13 - #18, #19 - # 23 Presentation: 1. The external parts of the root have been introduced to the children: the root hairs, the root tips,
UNIT 6 - STRUCTURES OF FLOWERING PLANTS & THEIR FUNCTIONS
 6.1 Plant Tissues A tissue is a group of cells with common function, structures or both. In plants we can find 2 types of tissues: Meristem Permanent tissues Meristem is found in regions with continuous
6.1 Plant Tissues A tissue is a group of cells with common function, structures or both. In plants we can find 2 types of tissues: Meristem Permanent tissues Meristem is found in regions with continuous
Plant Structure and Function Extension
 Plant Structure and Function Extension NGSSS: SC.912.L.14.7 Relate the structure of each of the major plant organs and tissues to physiological processes. (AA) Part 1A: Leaves The leaf of a plant serves
Plant Structure and Function Extension NGSSS: SC.912.L.14.7 Relate the structure of each of the major plant organs and tissues to physiological processes. (AA) Part 1A: Leaves The leaf of a plant serves
Neurite formation & neuronal polarization. The cytoskeletal components of neurons have characteristic distributions and associations
 Mechanisms of neuronal migration & Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 The cytoskeletal
Mechanisms of neuronal migration & Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 The cytoskeletal
On the role of stress anisotropy in the growth of stems
 Journal of Experimental Botany, Vol. 64, No. 15, pp. 4697 4707, 2013 doi:10.1093/jxb/ert176 Advance Access publication 3 August, 2013 Review paper On the role of stress anisotropy in the growth of stems
Journal of Experimental Botany, Vol. 64, No. 15, pp. 4697 4707, 2013 doi:10.1093/jxb/ert176 Advance Access publication 3 August, 2013 Review paper On the role of stress anisotropy in the growth of stems
23-. Shoot and root development depend on ratio of IAA/CK
 Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
EFFECTS OF GIBBERELLIC ACID ON INTERNODE GROWTH AND STARCH CONTENTS OF EUCALYPTUS CAMALDULENSIS SEEDLINGS
 New Phytol. {ig()) S, ioiyio22. EFFECTS OF GIBBERELLIC ACID ON INTERNODE GROWTH AND STARCH CONTENTS OF EUCALYPTUS CAMALDULENSIS SEEDLINGS BY E. P. BACHELARD Department of Forestry, Australian National
New Phytol. {ig()) S, ioiyio22. EFFECTS OF GIBBERELLIC ACID ON INTERNODE GROWTH AND STARCH CONTENTS OF EUCALYPTUS CAMALDULENSIS SEEDLINGS BY E. P. BACHELARD Department of Forestry, Australian National
Primary Plant Body: Embryogenesis and the Seedling
 BIOL 221 Concepts of Botany Primary Plant Body: Embryogenesis and the Seedling (Photo Atlas: Figures 1.29, 9.147, 9.148, 9.149, 9.150, 9.1, 9.2) A. Introduction Plants are composed of fewer cell types,
BIOL 221 Concepts of Botany Primary Plant Body: Embryogenesis and the Seedling (Photo Atlas: Figures 1.29, 9.147, 9.148, 9.149, 9.150, 9.1, 9.2) A. Introduction Plants are composed of fewer cell types,
Plant Cell Biology. Ram Dixit Washington University in St. Louis
 Plant Cell Biology Ram Dixit Washington University in St. Louis July 10, 2017 Lecture Goals 1. Introduction to the major components of plant cells and how they differ from animal cells 2. Understand the
Plant Cell Biology Ram Dixit Washington University in St. Louis July 10, 2017 Lecture Goals 1. Introduction to the major components of plant cells and how they differ from animal cells 2. Understand the
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated
 Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Actions of auxin. Hormones: communicating with chemicals History: Discovery of a growth substance (hormone- auxin)
 Hormones: communicating with chemicals History- discovery of plant hormone. Auxin Concepts of hormones Auxin levels are regulated by synthesis/degradation, transport, compartmentation, conjugation. Polar
Hormones: communicating with chemicals History- discovery of plant hormone. Auxin Concepts of hormones Auxin levels are regulated by synthesis/degradation, transport, compartmentation, conjugation. Polar
Chapter 6. Biology of Flowering Plants. Anatomy Seedlings, Meristems, Stems, and Roots
 BOT 3015L (Outlaw/Sherdan/Aghoram); Page 1 of 6 Chapter 6 Biology of Flowering Plants Anatomy Seedlings, Meristems, Stems, and Roots Objectives Seedling germination and anatomy. Understand meristem structure
BOT 3015L (Outlaw/Sherdan/Aghoram); Page 1 of 6 Chapter 6 Biology of Flowering Plants Anatomy Seedlings, Meristems, Stems, and Roots Objectives Seedling germination and anatomy. Understand meristem structure
Neurite formation & neuronal polarization
 Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 An immature neuron in cell culture first sprouts
Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 An immature neuron in cell culture first sprouts
 Question 1: What are the factors affecting the rate of diffusion? Diffusion is the passive movement of substances from a region of higher concentration to a region of lower concentration. Diffusion of
Question 1: What are the factors affecting the rate of diffusion? Diffusion is the passive movement of substances from a region of higher concentration to a region of lower concentration. Diffusion of
Supplementary Methods
 Supplementary Methods Modeling of magnetic field In this study, the magnetic field was generated with N52 grade nickel-plated neodymium block magnets (K&J Magnetics). The residual flux density of the magnets
Supplementary Methods Modeling of magnetic field In this study, the magnetic field was generated with N52 grade nickel-plated neodymium block magnets (K&J Magnetics). The residual flux density of the magnets
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. HSP21 expression in 35S:HSP21 and hsp21 knockdown plants. (a) Since no T- DNA insertion line for HSP21 is available in the publicly available T-DNA collections,
Supplementary Figure 1 Supplementary Figure 1. HSP21 expression in 35S:HSP21 and hsp21 knockdown plants. (a) Since no T- DNA insertion line for HSP21 is available in the publicly available T-DNA collections,
Plants. Plant Form and Function. Tissue Systems 6/4/2012. Chapter 17. Herbaceous (nonwoody) Woody. Flowering plants can be divided into two groups:
 Monocots Dicots 6/4/2012 Plants Plant Form and Function Chapter 17 Herbaceous (nonwoody) In temperate climates, aerial parts die back Woody In temperate climates, aerial parts persist The Plant Body Functions
Monocots Dicots 6/4/2012 Plants Plant Form and Function Chapter 17 Herbaceous (nonwoody) In temperate climates, aerial parts die back Woody In temperate climates, aerial parts persist The Plant Body Functions
ABSORPTION OF WATER MODE OF WATER ABSORPTION ACTIVE AND PASSIVE ABSORPTION AND FACTORS AFFECTING ABSORPTION.
 ABSORPTION OF WATER MODE OF WATER ABSORPTION ACTIVE AND PASSIVE ABSORPTION AND FACTORS AFFECTING ABSORPTION. PRELUDE OF WATER POTENTIAL Most organisms are comprised of at least 70% or more water. Some
ABSORPTION OF WATER MODE OF WATER ABSORPTION ACTIVE AND PASSIVE ABSORPTION AND FACTORS AFFECTING ABSORPTION. PRELUDE OF WATER POTENTIAL Most organisms are comprised of at least 70% or more water. Some
GS Analysis of Microarray Data
 GS01 0163 Analysis of Microarray Data Keith Baggerly and Kevin Coombes Section of Bioinformatics Department of Biostatistics and Applied Mathematics UT M. D. Anderson Cancer Center kabagg@mdanderson.org
GS01 0163 Analysis of Microarray Data Keith Baggerly and Kevin Coombes Section of Bioinformatics Department of Biostatistics and Applied Mathematics UT M. D. Anderson Cancer Center kabagg@mdanderson.org
TORSIONS INDUCED BY AUXIN
 JUNE 1947 VOL. 46, No. i TORSIONS INDUCED BY AUXIN BY R. SNOW Fellow of Magdalen College, Oxford (With I figure in the text) It was reported in a previous paper of this series (1945, p. 77) that petioles
JUNE 1947 VOL. 46, No. i TORSIONS INDUCED BY AUXIN BY R. SNOW Fellow of Magdalen College, Oxford (With I figure in the text) It was reported in a previous paper of this series (1945, p. 77) that petioles
Supplemental Figures S1 S5
 Beyond reduction of atherosclerosis: PON2 provides apoptosis resistance and stabilizes tumor cells Ines Witte (1), Sebastian Altenhöfer (1), Petra Wilgenbus (1), Julianna Amort (1), Albrecht M. Clement
Beyond reduction of atherosclerosis: PON2 provides apoptosis resistance and stabilizes tumor cells Ines Witte (1), Sebastian Altenhöfer (1), Petra Wilgenbus (1), Julianna Amort (1), Albrecht M. Clement
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2215 Figure S1 Number of egfp-vps4a bursts versus cellular expression levels. The total number of egfp-vps4a bursts, counted at the end of each movie (frame 2000, after 1h 28 min) are plotted
DOI: 10.1038/ncb2215 Figure S1 Number of egfp-vps4a bursts versus cellular expression levels. The total number of egfp-vps4a bursts, counted at the end of each movie (frame 2000, after 1h 28 min) are plotted
QUANTIFICATION OF EMBOLI BY VISUALIZATION OF AIR FILLED XYLEM VESSELS
 QUANTIFICATION OF EMBOLI BY VISUALIZATION OF AIR FILLED XYLEM VESSELS J. Nijsse and U. van Meeteren Wageningen University Plant Sciences Horticultural Production Chains Marijkeweg 22 6709 PG Wageningen
QUANTIFICATION OF EMBOLI BY VISUALIZATION OF AIR FILLED XYLEM VESSELS J. Nijsse and U. van Meeteren Wageningen University Plant Sciences Horticultural Production Chains Marijkeweg 22 6709 PG Wageningen
Stems and Transport in Vascular Plants. Herbaceous Stems. Herbaceous Dicot Stem 3/12/2012. Chapter 34. Basic Tissues in Herbaceous Stems.
 Bud scale Terminal bud Stems and Transport in Plants One year's growth Terminal bud scale scars Axillary bud Leaf scar Node Internode Node Chapter 34 Lenticels Terminal bud scale scars Bundle scars A Woody
Bud scale Terminal bud Stems and Transport in Plants One year's growth Terminal bud scale scars Axillary bud Leaf scar Node Internode Node Chapter 34 Lenticels Terminal bud scale scars Bundle scars A Woody
Embryo Development. Embryo Development. Embryo Development. Embryo Development (Cont.) Vegetative Plant Development
 Vegetative Plant Development Chapter 37 Embryo Development Begins once the egg cell is fertilized -The growing pollen tube enters angiosperm embryo sac and releases two sperm cells -One sperm fertilizes
Vegetative Plant Development Chapter 37 Embryo Development Begins once the egg cell is fertilized -The growing pollen tube enters angiosperm embryo sac and releases two sperm cells -One sperm fertilizes
Introduction to Biology
 Introduction to Biology Biology The Study of Life Life arose more than 3.5 billion years ago First organisms (living things) were single celled Only life on Earth for millions of years Organisms changed
Introduction to Biology Biology The Study of Life Life arose more than 3.5 billion years ago First organisms (living things) were single celled Only life on Earth for millions of years Organisms changed
THE BEHAVIOUR OF CHLOROPLASTS DURING CELL DIVISION OF ISOETES LACUSTRIS L.
 New Phytol (1974) 73, 139-142. THE BEHAVIOUR OF CHLOROPLASTS DURING CELL DIVISION OF ISOETES LACUSTRIS L. BY JEAN M. WHATLEY Botany School, University of Oxford (Received 2 July 1973) SUMMARY Cells in
New Phytol (1974) 73, 139-142. THE BEHAVIOUR OF CHLOROPLASTS DURING CELL DIVISION OF ISOETES LACUSTRIS L. BY JEAN M. WHATLEY Botany School, University of Oxford (Received 2 July 1973) SUMMARY Cells in
Introduction to Biology
 1 Introduction to Biology 2 Biology The Study of Life Life arose more than 3.5 billion years ago First organisms (living things) were single celled Only life on Earth for millions of years Organisms changed
1 Introduction to Biology 2 Biology The Study of Life Life arose more than 3.5 billion years ago First organisms (living things) were single celled Only life on Earth for millions of years Organisms changed
Plant Anatomy and Tissue Structures
 Plant Anatomy and Tissue Structures The Two Major Plant Systems Reproductive shoot (flower) Terminal bud Node Internode Angiosperm plants have threse major organs: Roots Stems Leaves & Flowers Terminal
Plant Anatomy and Tissue Structures The Two Major Plant Systems Reproductive shoot (flower) Terminal bud Node Internode Angiosperm plants have threse major organs: Roots Stems Leaves & Flowers Terminal
Non Permanent Tissues - Meristematic Tissue
 PLANT TISSUES Non Permanent Tissues - Meristematic Tissue Undifferentiated plant cells that are continually dividing by mitosis Large thin walled cells No vacuole Dense cytoplasm Large nucleus Found at
PLANT TISSUES Non Permanent Tissues - Meristematic Tissue Undifferentiated plant cells that are continually dividing by mitosis Large thin walled cells No vacuole Dense cytoplasm Large nucleus Found at
Lecture 19. A Sieve Plate with large Sieve Pores. Secondary Phloem. Secondary phloem (cont d)
 Lecture 19 Secondary phloem (cont d) Secondary Phloem in Tilia americana (American Basswood) Secondary Phloem of Tilia Stained with Toluidine Blue & viewed with Crossed Polarizers. Secondary Phloem A Sieve
Lecture 19 Secondary phloem (cont d) Secondary Phloem in Tilia americana (American Basswood) Secondary Phloem of Tilia Stained with Toluidine Blue & viewed with Crossed Polarizers. Secondary Phloem A Sieve
 Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark the regions where active cell division and rapid division
Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark the regions where active cell division and rapid division
SESSION 6: SUPPORT AND TRANSPORT SYSTEMS IN PLANTS PART 1
 SESSION 6: SUPPORT AND TRANSPORT SYSTEMS IN PLANTS PART 1 KEY CONCEPTS In this session we will focus on summarising what you need to know about: - Anatomy of dicotyledonous plants Root and stem: distribution
SESSION 6: SUPPORT AND TRANSPORT SYSTEMS IN PLANTS PART 1 KEY CONCEPTS In this session we will focus on summarising what you need to know about: - Anatomy of dicotyledonous plants Root and stem: distribution
Bio Factsheet. Transport in Plants. Number 342
 Number 342 Transport in Plants This Factsheet: Explains why plants need a transport system Describes what plants transport Describes the tissues which carry out transport Outlines the position of the xylem
Number 342 Transport in Plants This Factsheet: Explains why plants need a transport system Describes what plants transport Describes the tissues which carry out transport Outlines the position of the xylem
Identification of factors involved in Xylem Cell Differentiation Aarush Mohit Mittal 1, 2
 Identification of factors involved in Xylem Cell Differentiation Aarush Mohit Mittal 1, 2 1 Department of Biological Sciences and Bio-Engineering, Indian Institute of Technology, Kanpur, India 2 Department
Identification of factors involved in Xylem Cell Differentiation Aarush Mohit Mittal 1, 2 1 Department of Biological Sciences and Bio-Engineering, Indian Institute of Technology, Kanpur, India 2 Department
Anatomy of Flowering Plants. K C Meena PGT Biology
 Anatomy of Flowering Plants K C Meena PGT Biology Tissues A group of similar cells performing same function. Types of plant tissues - Meristematic tissues and permanent tissues. Meristematic tissues Have
Anatomy of Flowering Plants K C Meena PGT Biology Tissues A group of similar cells performing same function. Types of plant tissues - Meristematic tissues and permanent tissues. Meristematic tissues Have
PARTICLE MEASUREMENT IN CLEAN ROOM TECHNOLOGY
 WHITEPAPER ENGLISH PARTICLE MEASUREMENT IN CLEAN ROOM TECHNOLOGY PARTICLE MEASUREMENT Particle measurement in cleanrooms. WP1508006-0100-EN, V1R0, 2015-08 PARTICLE MEASUREMENT IN CLEAN ROOM TECHNOLOGY
WHITEPAPER ENGLISH PARTICLE MEASUREMENT IN CLEAN ROOM TECHNOLOGY PARTICLE MEASUREMENT Particle measurement in cleanrooms. WP1508006-0100-EN, V1R0, 2015-08 PARTICLE MEASUREMENT IN CLEAN ROOM TECHNOLOGY
Class XI Chapter 6 Anatomy of Flowering Plants Biology
 Class XI Chapter 6 Anatomy of Flowering Plants Biology Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark
Class XI Chapter 6 Anatomy of Flowering Plants Biology Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark
DNA or RNA metabolism (1%) Signal transduction (2%) Development (2%) Other cellular processes (17%)
 Fig. 35-24 Other metabolism (18%) DNA or RNA metabolism (1%) Signal transduction (2%) Development (2%) Unknown (24%) Energy pathways (3%) Cell division and organization (3%) Transport (4%) Transcription
Fig. 35-24 Other metabolism (18%) DNA or RNA metabolism (1%) Signal transduction (2%) Development (2%) Unknown (24%) Energy pathways (3%) Cell division and organization (3%) Transport (4%) Transcription
BIOLOGY. Chapter 10 CELL REPRODUCTION PowerPoint Image Slideshow
 BIOLOGY Chapter 10 CELL REPRODUCTION PowerPoint Image Slideshow FIGURE 10.1 A sea urchin begins life as a single cell that (a) divides to form two cells, visible by scanning electron microscopy. After
BIOLOGY Chapter 10 CELL REPRODUCTION PowerPoint Image Slideshow FIGURE 10.1 A sea urchin begins life as a single cell that (a) divides to form two cells, visible by scanning electron microscopy. After
Last time: Obtaining information from a cloned gene
 Last time: Obtaining information from a cloned gene Objectives: 1. What is the biochemical role of the gene? 2. Where and when is the gene expressed (transcribed)? 3. Where and when is the protein made?
Last time: Obtaining information from a cloned gene Objectives: 1. What is the biochemical role of the gene? 2. Where and when is the gene expressed (transcribed)? 3. Where and when is the protein made?
The three principal organs of seed plants are roots, stems, and leaves.
 23 1 Specialized Tissues in Plants Seed Plant Structure The three principal organs of seed plants are roots, stems, and leaves. 1 of 34 23 1 Specialized Tissues in Plants Seed Plant Structure Roots: absorb
23 1 Specialized Tissues in Plants Seed Plant Structure The three principal organs of seed plants are roots, stems, and leaves. 1 of 34 23 1 Specialized Tissues in Plants Seed Plant Structure Roots: absorb
Contains ribosomes attached to the endoplasmic reticulum. Genetic material consists of linear chromosomes. Diameter of the cell is 1 m
 1. (a) Complete each box in the table, which compares a prokaryotic and a eukaryotic cell, with a tick if the statement is correct or a cross if it is incorrect. Prokaryotic cell Eukaryotic cell Contains
1. (a) Complete each box in the table, which compares a prokaryotic and a eukaryotic cell, with a tick if the statement is correct or a cross if it is incorrect. Prokaryotic cell Eukaryotic cell Contains
Identifying cells using Immunofluorscence Rachel Scalzo Bio-300, 001 Professor Bentley 9/24/2014
 Identifying cells using Immunofluorscence Rachel Scalzo Bio-300, 001 Professor Bentley 9/24/2014 2 Abstract In this experiment, we used immunofluorescence to isolate certain organelles and see where they
Identifying cells using Immunofluorscence Rachel Scalzo Bio-300, 001 Professor Bentley 9/24/2014 2 Abstract In this experiment, we used immunofluorescence to isolate certain organelles and see where they
Lab Exercise 4: Primary Growth and Tissues in Stems
 Lab Exercise 4: Primary Growth and Tissues in Stems Tissues of the plant body can be classified in a variety of ways: functionally (based on the tissue function, e.g. vascular tissue ), morphologically
Lab Exercise 4: Primary Growth and Tissues in Stems Tissues of the plant body can be classified in a variety of ways: functionally (based on the tissue function, e.g. vascular tissue ), morphologically
BIO1PS 2012 Plant Science Lecture 4 Hormones Pt. I
 BIO1PS 2012 Plant Science Lecture 4 Hormones Pt. I Dr. Michael Emmerling Department of Botany Room 410 m.emmerling@latrobe.edu.au Hormones and Ghost gum Eucalyptus papuana Coordination ~3 Lectures Leaves
BIO1PS 2012 Plant Science Lecture 4 Hormones Pt. I Dr. Michael Emmerling Department of Botany Room 410 m.emmerling@latrobe.edu.au Hormones and Ghost gum Eucalyptus papuana Coordination ~3 Lectures Leaves
Supplementary Figure 1. Phenotype of the HI strain.
 Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Supplementary Figure 1. Phenotype of the HI strain. (A) Phenotype of the HI and wild type plant after flowering (~1month). Wild type plant is tall with well elongated inflorescence. All four HI plants
Hybrid Gold Superstructures: Synthesis and. Specific Cell Surface Protein Imaging Applications
 Supporting Information Hybrid Gold Nanocube@Silica@Graphene-Quantum-Dot Superstructures: Synthesis and Specific Cell Surface Protein Imaging Applications Liu Deng, Ling Liu, Chengzhou Zhu, Dan Li and Shaojun
Supporting Information Hybrid Gold Nanocube@Silica@Graphene-Quantum-Dot Superstructures: Synthesis and Specific Cell Surface Protein Imaging Applications Liu Deng, Ling Liu, Chengzhou Zhu, Dan Li and Shaojun
Amanda Rawson Independent Research Project Report Bio219 Cell Biology. December 3, 2008
 Evidence That Treatment with Cytochalasin B Increases the Average Velocity of Cytoplasmic Streaming in Amoebae Proteus Amanda Rawson Independent Research Project Report Bio219 Cell Biology December 3,
Evidence That Treatment with Cytochalasin B Increases the Average Velocity of Cytoplasmic Streaming in Amoebae Proteus Amanda Rawson Independent Research Project Report Bio219 Cell Biology December 3,
Photoelastic Experiments with a Transmitted-Light Polariscope
 Photoelastic Experiments with a Transmitted-Light Polariscope Objectives: To become familiar with a Transmitted-Light Polariscope To physically see the effects of loading various specimens and their unique
Photoelastic Experiments with a Transmitted-Light Polariscope Objectives: To become familiar with a Transmitted-Light Polariscope To physically see the effects of loading various specimens and their unique
Supplemental Data. Perrella et al. (2013). Plant Cell /tpc
 Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Tuesday, February 11, Chapter 3. Load and Stress Analysis. Dr. Mohammad Suliman Abuhaiba, PE
 1 Chapter 3 Load and Stress Analysis 2 Chapter Outline Equilibrium & Free-Body Diagrams Shear Force and Bending Moments in Beams Singularity Functions Stress Cartesian Stress Components Mohr s Circle for
1 Chapter 3 Load and Stress Analysis 2 Chapter Outline Equilibrium & Free-Body Diagrams Shear Force and Bending Moments in Beams Singularity Functions Stress Cartesian Stress Components Mohr s Circle for
Plant Tissues and Organs. Topic 13 Plant Science Subtopics , ,
 Plant Tissues and Organs Topic 13 Plant Science Subtopics 13.1.2, 13.1.3, 13.1.4 Objectives: List and describe the major plant organs their structure and function List and describe the major types of plant
Plant Tissues and Organs Topic 13 Plant Science Subtopics 13.1.2, 13.1.3, 13.1.4 Objectives: List and describe the major plant organs their structure and function List and describe the major types of plant
SUPPLEMENTARY INFORMATION
 PRC2 represses dedifferentiation of mature somatic cells in Arabidopsis Momoko Ikeuchi 1 *, Akira Iwase 1 *, Bart Rymen 1, Hirofumi Harashima 1, Michitaro Shibata 1, Mariko Ohnuma 1, Christian Breuer 1,
PRC2 represses dedifferentiation of mature somatic cells in Arabidopsis Momoko Ikeuchi 1 *, Akira Iwase 1 *, Bart Rymen 1, Hirofumi Harashima 1, Michitaro Shibata 1, Mariko Ohnuma 1, Christian Breuer 1,
GFP GAL bp 3964 bp
 Supplemental Data. Møller et al. (2009) Shoot Na + exclusion and increased salinity tolerance engineered by cell type-specific alteration of Na + transport in Arabidopsis Supplemental Figure 1. Salt-sensitive
Supplemental Data. Møller et al. (2009) Shoot Na + exclusion and increased salinity tolerance engineered by cell type-specific alteration of Na + transport in Arabidopsis Supplemental Figure 1. Salt-sensitive
Chapter 3. Load and Stress Analysis
 Chapter 3 Load and Stress Analysis 2 Shear Force and Bending Moments in Beams Internal shear force V & bending moment M must ensure equilibrium Fig. 3 2 Sign Conventions for Bending and Shear Fig. 3 3
Chapter 3 Load and Stress Analysis 2 Shear Force and Bending Moments in Beams Internal shear force V & bending moment M must ensure equilibrium Fig. 3 2 Sign Conventions for Bending and Shear Fig. 3 3
II. SIMPLE TISSUES Bot 404--Fall A. Introduction to Tissues (DIAGRAM allow a full page)
 II. SIMPLE TISSUES Bot 404--Fall 2004 A. Introduction to Tissues (DIAGRAM allow a full page) B. Definitions Adaxial = facing the axil; upper surface of leaf Abaxial = facing away from the axil; lower surface
II. SIMPLE TISSUES Bot 404--Fall 2004 A. Introduction to Tissues (DIAGRAM allow a full page) B. Definitions Adaxial = facing the axil; upper surface of leaf Abaxial = facing away from the axil; lower surface
purpose of this Chapter is to highlight some problems that will likely provide new
 119 Chapter 6 Future Directions Besides our contributions discussed in previous chapters to the problem of developmental pattern formation, this work has also brought new questions that remain unanswered.
119 Chapter 6 Future Directions Besides our contributions discussed in previous chapters to the problem of developmental pattern formation, this work has also brought new questions that remain unanswered.
Understanding Cambial Behaviour. The key to wood quality
 Understanding Cambial Behaviour The key to wood quality A brief history Terminology Dormancy and reactivation Growth of derivatives and wall formation Pitting and plasmodesmata A brief history Nehemiah
Understanding Cambial Behaviour The key to wood quality A brief history Terminology Dormancy and reactivation Growth of derivatives and wall formation Pitting and plasmodesmata A brief history Nehemiah
Supplemental material
 Supplemental material THE JOURNAL OF CELL BIOLOGY Civelekoglu-Scholey et al., http://www.jcb.org/cgi/content/full/jcb.200908150/dc1 Figure S1. Spindle dynamics in Drosophila embryos and in vitro gliding
Supplemental material THE JOURNAL OF CELL BIOLOGY Civelekoglu-Scholey et al., http://www.jcb.org/cgi/content/full/jcb.200908150/dc1 Figure S1. Spindle dynamics in Drosophila embryos and in vitro gliding
Name: Plant stems and leaves (p. 1 of )
 Name: Plant stems and leaves (p. 1 of ) Introduction: Plants have a variety of configurations but the same basic structures. The three main parts of a plant are the roots, stems, and leaves. The tracheids
Name: Plant stems and leaves (p. 1 of ) Introduction: Plants have a variety of configurations but the same basic structures. The three main parts of a plant are the roots, stems, and leaves. The tracheids
GS Analysis of Microarray Data
 GS01 0163 Analysis of Microarray Data Keith Baggerly and Kevin Coombes Section of Bioinformatics Department of Biostatistics and Applied Mathematics UT M. D. Anderson Cancer Center kabagg@mdanderson.org
GS01 0163 Analysis of Microarray Data Keith Baggerly and Kevin Coombes Section of Bioinformatics Department of Biostatistics and Applied Mathematics UT M. D. Anderson Cancer Center kabagg@mdanderson.org
Supplemental Data. Gao et al. (2012). Plant Cell /tpc
 Supplemental Figure 1. Plant EMP Proteins. (A) The Accession numbers of the 12 EMP members from Arabidopsis. (B) Phylogenetic analysis of EMP proteins from Arabidopsis, human and yeast using the Mac Vector
Supplemental Figure 1. Plant EMP Proteins. (A) The Accession numbers of the 12 EMP members from Arabidopsis. (B) Phylogenetic analysis of EMP proteins from Arabidopsis, human and yeast using the Mac Vector
Transport in Vascular Plants
 Chapter 36 Transport in Vascular Plants PowerPoint Lectures for Biology, Seventh Edition Neil Campbell and Jane Reece Lectures by Chris Romero Vascular tissue Transports nutrients throughout a plant; such
Chapter 36 Transport in Vascular Plants PowerPoint Lectures for Biology, Seventh Edition Neil Campbell and Jane Reece Lectures by Chris Romero Vascular tissue Transports nutrients throughout a plant; such
Ginkgo leaf. Ginkgo is dioecious, separate sexes: male and female plants are separate. Monoecious plants have both male and female parts.
 Ginkgo leaf Figure 22-30 Ginkgo tree. Ginkgo is dioecious, separate sexes: male and female plants are separate. Monoecious plants have both male and female parts. The vein pattern is dichotomous: Divided
Ginkgo leaf Figure 22-30 Ginkgo tree. Ginkgo is dioecious, separate sexes: male and female plants are separate. Monoecious plants have both male and female parts. The vein pattern is dichotomous: Divided
Plant Structure. Objectives At the end of this sub section students should be able to:
 Name: 3.2 Organisation and the Vascular Structures 3.2.1 Flowering plant structure and root structure Objectives At the end of this sub section students should be able to: 1. Label a diagram of the external
Name: 3.2 Organisation and the Vascular Structures 3.2.1 Flowering plant structure and root structure Objectives At the end of this sub section students should be able to: 1. Label a diagram of the external
Report. Impaired Cellulose Synthase Guidance Leads to Stem Torsion and Twists Phyllotactic Patterns in Arabidopsis
 Current Biology 23, 895 900, May 20, 2013 ª2013 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2013.04.013 Impaired Cellulose Synthase Guidance Leads to Stem Torsion and Twists Phyllotactic
Current Biology 23, 895 900, May 20, 2013 ª2013 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2013.04.013 Impaired Cellulose Synthase Guidance Leads to Stem Torsion and Twists Phyllotactic
DEVELOPMENTAL GENETICS OF ARABIDOPSIS THALIANA
 DEVELOPMENTAL GENETICS OF ARABIDOPSIS THALIANA CHASE BALLARD LINDA EAN HECTOR LOPEZ DR. JOANNA WERNER-FRACZEK IN COLLABORATION WITH DR. PATRICIA SPRINGER S LAB AT UCR AND ROBERT KOBLE PURPOSE OF RESEARCH
DEVELOPMENTAL GENETICS OF ARABIDOPSIS THALIANA CHASE BALLARD LINDA EAN HECTOR LOPEZ DR. JOANNA WERNER-FRACZEK IN COLLABORATION WITH DR. PATRICIA SPRINGER S LAB AT UCR AND ROBERT KOBLE PURPOSE OF RESEARCH
with others and thus regenerate a functioning conductive system. Regeneration
 388 BOTANY: SINNOTT AND BLOCH PROC. N. A. S. VISIBLE EXPRESSION OF CYTOPLASMIC PA TTERN IN THE DIFFERENTIATION OF XYLEM STRANDS BY EDMUND W. SINOTT AND ROBERT BLOCH OsBORN BOTANCAL LABORATORY, YALE UNIVERSITY
388 BOTANY: SINNOTT AND BLOCH PROC. N. A. S. VISIBLE EXPRESSION OF CYTOPLASMIC PA TTERN IN THE DIFFERENTIATION OF XYLEM STRANDS BY EDMUND W. SINOTT AND ROBERT BLOCH OsBORN BOTANCAL LABORATORY, YALE UNIVERSITY
Heather Currinn, Benjamin Guscott, Zita Balklava, Alice Rothnie and Thomas Wassmer*
 Online Resources APP controls the formation of PI(3,5)P 2 vesicles through its binding of the PIKfyve complex. Cellular and Molecular Life Sciences Heather Currinn, Benjamin Guscott, Zita Balklava, Alice
Online Resources APP controls the formation of PI(3,5)P 2 vesicles through its binding of the PIKfyve complex. Cellular and Molecular Life Sciences Heather Currinn, Benjamin Guscott, Zita Balklava, Alice
LOW-POWER ELECTRON MICROSCOPY OF THE ROOT CAP REGION OF EUCALYPT MYCORRHIZAS
 New Phytol. (1968) 67, 663-665. LOW-POWER ELECTRON MICROSCOPY OF THE ROOT CAP REGION OF EUCALYPT MYCORRHIZAS BY G. A. CHILVERS Botany Department, School of General Studies, Australian National University,
New Phytol. (1968) 67, 663-665. LOW-POWER ELECTRON MICROSCOPY OF THE ROOT CAP REGION OF EUCALYPT MYCORRHIZAS BY G. A. CHILVERS Botany Department, School of General Studies, Australian National University,
Transport of substances in plants
 Transport of substances in plants We have already looked at why many organisms need transport systems with special reference to surface area and volume. The larger the volume : surface area ratio, the
Transport of substances in plants We have already looked at why many organisms need transport systems with special reference to surface area and volume. The larger the volume : surface area ratio, the
Water Relations in Viticulture BRIANNA HOGE AND JIM KAMAS
 Water Relations in Viticulture BRIANNA HOGE AND JIM KAMAS Overview Introduction Important Concepts for Understanding water Movement through Vines Osmosis Water Potential Cell Expansion and the Acid Growth
Water Relations in Viticulture BRIANNA HOGE AND JIM KAMAS Overview Introduction Important Concepts for Understanding water Movement through Vines Osmosis Water Potential Cell Expansion and the Acid Growth
SUPPLEMENTARY INFORMATION
 GP2 Type I-piliated bacteria FAE M cell M cell pocket idc T cell mdc Generation of antigenspecific T cells Induction of antigen-specific mucosal immune response Supplementary Figure 1 Schematic diagram
GP2 Type I-piliated bacteria FAE M cell M cell pocket idc T cell mdc Generation of antigenspecific T cells Induction of antigen-specific mucosal immune response Supplementary Figure 1 Schematic diagram
Chapter 21: Plant Structure & Function
 Chapter 21: Plant Structure & Function Chapter 21: Plant Structure & Function All organisms must: Take in certain materials, e.g. O 2, food, drink Eliminate other materials, e.g. CO 2, waste products Chapter
Chapter 21: Plant Structure & Function Chapter 21: Plant Structure & Function All organisms must: Take in certain materials, e.g. O 2, food, drink Eliminate other materials, e.g. CO 2, waste products Chapter
Overview of Plant Tissues
 Plant Tissue Growth Key Concepts Overview of Plant Tissues Seed-bearing vascular plants have a shoot system with stems, leaves, and reproductive parts Most also have a root system These systems consist
Plant Tissue Growth Key Concepts Overview of Plant Tissues Seed-bearing vascular plants have a shoot system with stems, leaves, and reproductive parts Most also have a root system These systems consist
High photostability and enhanced fluorescence of gold nanoclusters by silver doping-supporting information
 High photostability and enhanced fluorescence of gold nanoclusters by silver doping-supporting information Size measurements Figure S1 P2 FTIR measurements Figure S2 P2 XPS measurements Figure S3 P3 Photo-physical
High photostability and enhanced fluorescence of gold nanoclusters by silver doping-supporting information Size measurements Figure S1 P2 FTIR measurements Figure S2 P2 XPS measurements Figure S3 P3 Photo-physical
Regulation of Growth Anisotropy in Well-Watered and Water-Stressed Maize Roots
 Plant Physiol. (1997) 115: 101-1 11 Regulation of Growth Anisotropy in Well-Watered and Water-Stressed Maize Roots 1. Spatial Distribution of Longitudinal, Radial, and Tangential Expansion Rates Benjamin
Plant Physiol. (1997) 115: 101-1 11 Regulation of Growth Anisotropy in Well-Watered and Water-Stressed Maize Roots 1. Spatial Distribution of Longitudinal, Radial, and Tangential Expansion Rates Benjamin
STRAIN ANALYSIS OF HDPE BUTT FUSION JOINT IN SIDE BEND TEST
 STRAIN ANALYSIS OF HDPE BUTT FUSION JOINT IN SIDE BEND TEST Meng Xiangli (Shawn), McElroy Manufacturing, Inc., Tulsa, OK Abstract The standard practice ASTM F3183-16 (F3183) has been developed for conducting
STRAIN ANALYSIS OF HDPE BUTT FUSION JOINT IN SIDE BEND TEST Meng Xiangli (Shawn), McElroy Manufacturing, Inc., Tulsa, OK Abstract The standard practice ASTM F3183-16 (F3183) has been developed for conducting
T H E J O U R N A L O F C E L L B I O L O G Y
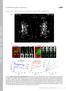 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Eisner et al., http://www.jcb.org/cgi/content/full/jcb.201312066/dc1 Figure S1. Mitochondrial continuity in adult skeletal muscle. (A)
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Eisner et al., http://www.jcb.org/cgi/content/full/jcb.201312066/dc1 Figure S1. Mitochondrial continuity in adult skeletal muscle. (A)
STRESS STRAIN AND DEFORMATION OF SOLIDS, STATES OF STRESS
 1 UNIT I STRESS STRAIN AND DEFORMATION OF SOLIDS, STATES OF STRESS 1. Define: Stress When an external force acts on a body, it undergoes deformation. At the same time the body resists deformation. The
1 UNIT I STRESS STRAIN AND DEFORMATION OF SOLIDS, STATES OF STRESS 1. Define: Stress When an external force acts on a body, it undergoes deformation. At the same time the body resists deformation. The
Fig (1):Layers of seconday cell wall
 Dr. Alaa J. Taha, Dr. Rana Alroomi and Dr. Hadeel Al-Newani :Secondary cell wall Although the secondary wall commonly is thought of as being deposited after the increase in surface area of the primary
Dr. Alaa J. Taha, Dr. Rana Alroomi and Dr. Hadeel Al-Newani :Secondary cell wall Although the secondary wall commonly is thought of as being deposited after the increase in surface area of the primary
4) Please cite Dagda et al J Biol Chem 284: , for any publications or presentations resulting from use or modification of the macro.
 Supplement Figure S1. Algorithmic quantification of mitochondrial morphology in SH- SY5Y cells treated with known fission/fusion mediators. Parental SH-SY5Y cells were transiently transfected with an empty
Supplement Figure S1. Algorithmic quantification of mitochondrial morphology in SH- SY5Y cells treated with known fission/fusion mediators. Parental SH-SY5Y cells were transiently transfected with an empty
Visit For All NCERT solutions, CBSE sample papers, Question papers, Notes for Class 6 to 12. Chapter-6 ANATOMY OF FLOWERING PLANTS
 Chapter-6 ANATOMY OF FLOWERING PLANTS POINTS TO REMEMBER Anatomy : Anatomy is the study of internal structure of organisms. Plant anatomy includes organisation and structure of tissues. Tissue : A group
Chapter-6 ANATOMY OF FLOWERING PLANTS POINTS TO REMEMBER Anatomy : Anatomy is the study of internal structure of organisms. Plant anatomy includes organisation and structure of tissues. Tissue : A group
THE ROOTS OF WILD RICE. ZIZANIA AQUATICA L.
 THE ROOTS OF WILD RICE. ZIZANIA AQUATICA L. E. L. STOVER, Eastern Illinois State Teachers College. This grass grows from Maine to Minnesota in aquatic habitats (2 and 5). It is common in marsh lands all
THE ROOTS OF WILD RICE. ZIZANIA AQUATICA L. E. L. STOVER, Eastern Illinois State Teachers College. This grass grows from Maine to Minnesota in aquatic habitats (2 and 5). It is common in marsh lands all
Variation in Microstructure and Mechanical Properties of Tre Gai bamboo (Bambusa stenostachya) with Position in the Culm
 Variation in Microstructure and Mechanical Properties of Tre Gai bamboo (Bambusa stenostachya) with Position in the Culm 3.054 Report May 14, 2014 Abstract Bamboo, with its high strength-to-weight ratio
Variation in Microstructure and Mechanical Properties of Tre Gai bamboo (Bambusa stenostachya) with Position in the Culm 3.054 Report May 14, 2014 Abstract Bamboo, with its high strength-to-weight ratio
