C-Fiber Structure Varies with Location in Peripheral Nerve
|
|
|
- April Warner
- 6 years ago
- Views:
Transcription
1 Journal of Neuropathology and Experimental Neurology Vol. 63, No. 3 Copyright 2004 by the American Association of Neuropathologists March, 2004 pp C-Fiber Structure Varies with Location in Peripheral Nerve BETH BRIANNA MURINSON, MD, PHD, AND JOHN WESLEY GRIFFIN, MD Abstract. Recent advances in regeneration and pain research have revealed gaps in the understanding of normal C-fiber anatomy. In the rat PNS, C-fiber axons assemble into Remak bundles, but beyond this, features of C-fiber organization are not defined. Systematic sampling and quantitation reveals that Remak bundles exiting from the L5 dorsal root ganglion (DRG) contain large numbers of axons, for example, 56% of unmyelinated axons were in bundles of 20 axons. This is different from distal nerve segments such as the hindpaw plantar nerve where the median number of axons per bundle is 3. The crosssectional area of unmyelinated axons in dorsal root was homogeneous near the DRG but variability in axonal area increased near the spinal cord (p ) and the mean axonal area was unchanged. Unmyelinated axons in peripheral nerve were almost always isolated from one another by Schwann cell processes; however, in dorsal root 7% to 9% of unmyelinated axons were immediately adjacent within pockets containing 2 or more axons. Remak bundles in the distal peripheral nerve clustered with other Remak bundles. We observe that multiple unmyelinated axons are juxtaposed within the C-fiber/Remak bundle and that the close association of afferent axons may have important functional implications. Key Words: Dorsal root; Peripheral nerve; Primary afferent neuron; Remak bundle; Sensory function; Sympathetic; Unmyelinated axon. INTRODUCTION Unmyelinated nerve fibers in the peripheral nervous system are often envisioned as simple but reliable conduits for signals from sensory terminations to the spinal cord. However, a new complexity is emerging. C-fiber axons are now recognized to be diverse in biophysical properties as well as modality and to exhibit complex responses to partial nerve injury (1 3). Recent studies indicate that C-fiber axons of the L4 mixed spinal nerve exhibit novel spontaneous activity after L5 mixed spinal nerve ligation and transection or L5 ventral rhizotomy, procedures that spare L4 unmyelinated axons from direct injury (4, 5). Although the mechanisms by which this spontaneous activity develops are not known, experimental evidence supports a role for peripheral nerve events. Signaling between C-fiber component cells has been demonstrated in that calcium transients arise in Remak Schwann cells in response to action potential propagation in unmyelinated axons and it is likely that multiple signaling pathways exist within the Remak bundle (6 8). These observations highlight a relative paucity of information about normal C-fiber/Remak bundle organization. Essential unresolved questions include whether multiple neurons contribute axons to a single unmyelinated nerve fiber bundle, whether these bundles typically contain 1 axon or multiple axons, and whether the relationship of axons to Remak Schwann cells changes with location. To address these questions we systematically From Departments of Neurology (BBM, JWG), Neuroscience (JWG), and Pathology (JWG), Johns Hopkins School of Medicine, Baltimore, Maryland. Correspondence to: Dr. Beth Murinson, 509 Pathology, 600 N. Wolfe Street, Baltimore, MD BethMurinson@hotmail.com Supported in part by the Pearl M. Stetler Fund (B.B.M.) and NIH characterized Remak bundle organization at defined locations in the rat PNS. The findings show important regional differences. This study examines the L5 dorsal root, the sciatic nerve, and specific branches because of direct relevance for models of neuropathic pain. The potential pathophysiological implications of the structural complexity of Remak bundles could include understanding how Remak bundle structure influences the electrochemical micro-environment of C-fiber axons and their discriminative capacity, determinants of the response of intact unmyelinated axons to partial nerve injury, and the requirements placed on Remak Schwann cells for successful regeneration. MATERIALS AND METHODS Eight- to 9-week-old male Sprague-Dawley rats weighing approximately 250 grams were used in these experiments. A total of 17 animals were studied. The animals were handled using an approved protocol. Briefly, under deep anesthesia (chloral hydrate 400 mg/kg i.p.), cardiac puncture was followed by perfusion with phosphate buffered saline and fixative. Fixation was overnight with 4% paraformaldehyde and 3% glutaraldehyde or with 5% glutaraldehyde in Sorenson s buffered saline (ph 7.4). The following tissues were collected after dissection (followed by the approximate distance from spinal cord in mm): rostral dorsal root, 0; caudal dorsal root, 30; mid-dorsal root ganglion (DRG), 32.5; mixed spinal nerve, 35; proximal sciatic nerve, 50; plantar nerve, 150. Each anatomical location was evaluated in a minimum of 3 animals. The fixed samples were dehydrated in preparation for plastic embedding. After 3 rinses with buffer, the tissues underwent a 1-hour incubation with 2% OsO 4, 3 buffer rinses, a graded alcohol series of twice for 10 minutes each in 50%, 70%, 80%, 95%, and 100% ethanol; propylene oxide for 5 minutes twice followed by propylene oxide:plastic (1:1) overnight at room temperature (9). Propylene oxide was exhausted from uncapped tubes for 3 hours, followed by immersion in fresh plastic overnight in a vacuum oven at room temperature and embedding in 246
2 C-FIBER STRUCTURE 247 fresh plastic on day 3. The plastic was polymerized over 48 hours at 60 C. Plastic components included dodenyl succinic anhydride (DDSA), nadic methyl anhydride (methyl-5-norborene-2,3-dicarboxylic anhydride, NMA), EMbed 812 (a mixture of bisphenol A/Epichchlorohydrin epoxy resin and resin modifier) and 2,4,6-(tri(dimethylaminoethyl)phenol) (DMP-30) (all from EMS, Fort Washington, PA) (10). One- m-thick sections were stained with toluidine blue (1%). Thin silver sections were mounted on Formvar-coated (EMS, Fort Washington, PA) grids of 50 or 100 mesh and stained with uranyl acetate (2.5% in 50% ethanol) and lead citrate (3%) (11). Tissues were quantitatively evaluated and photographed using a Hitachi 600A electron microscope. Photos were printed by hand and selected images were scanned for digital archiving and montage construction. Contrast and brightness were adjusted for optimal print quality but images are otherwise unaltered. Unmyelinated axons were distinguished from Schwann cells by several criteria. Compared with Schwann cells, Remak bundle axons have axoplasm that is less electron-dense and an axonal membrane that is more dense than the membrane of Schwann cells. Axons contain longitudinally oriented microtubules and do not form desmosomes. Individual Remak bundles were defined as contained within a continuous distinct basal lamina. Remak bundles were identified at 5,000 times magnification and axons were counted at 50,000 times magnification to ensure consistency. Plantar nerves were assessed in entire cross section; other tissues were systematically sampled and assessed over multiple 50 by 75 m regions selected for proximity to grid bar intersections. Overall, between 790 and 2,660 axons were counted for each sample evaluated. Clusters of Remak bundles were defined as 2 or more bundles immediately juxtaposed such that a line drawn around them would be a compact space excluding large myelinated axons. Clusters were often near 1 or more small myelinated axons. In cases where 2 small myelinated axons were in close proximity to one another, individual clusters were distinguished by including only those Remak bundles in closest juxtaposition. Axons were defined as being in a poly-axonal pocket when 2 or more axons were immediately adjacent and no extension of Schwann cell cytoplasm separated the 2 axons. Axonal areas were measured using Bioquant software (R&M Biometrics, Nashville, TN). Electron micrographs were photographed at grid bar intersections using 3,500 times magnification and digitized at 200 dpi. Approximately 100 unmyelinated axons were outlined by hand for each of the nerves studied; this number was sufficient to detect a 2-fold increase in variance at the p level. The distributions of axonal areas were compared using the Student t-test of the mean and an F- statistic for comparison of variance (12). Histograms and basic statistical analyses were carried out using a standard spreadsheet utility. RESULTS Characteristics of Unmyelinated Axons and Remak Bundles in the L5 Dorsal Root At the caudal end of the dorsal root, 2 to 3 mm from the center of the DRG, most Remak bundles contained large numbers of axons ( 20 axons). In some instances, several Remak bundles were juxtaposed so that the number of unmyelinated axons in close association could exceed 100 (Fig. 1A). In order to identify differences between the 2 ends of the dorsal root we compared the caudal dorsal root site to the rostral dorsal root at a site within 3 mm of entry into the spinal cord. In the caudal dorsal root, 56% of the unmyelinated axons were supported in bundles of 20 or more axons; axons in 1-axon bundles were infrequent (1.5%). At this site, the median number of axons/remak bundle was 9. A typical Remak bundle from the caudal dorsal root is shown in Figure 1B. By comparison, the Remak bundles in the rostral dorsal root contained a smaller number of axons/bundle (median number 5). This was significantly lower compared with the caudal DR (rank sum test, p 0.01). Examples of Remak bundles in the rostral dorsal root are shown in Figure 1C and D. The distribution of axons into Remak bundles is shown in Figure 2 for several locations along the nerve axis. The range of axons/bundle was greatest in the caudal dorsal root (Fig. 2B), where the axons/bundle varied from 1 to 50. Near the spinal cord the distribution shifted to the left and Remak bundles containing more than 20 axons were much less common (Fig. 2A). The shapes of these distributions were unusual and could not be characterized as normal, Poisson, or exponential. Remak bundles of the caudal dorsal root typically contained axons of homogenous size densely packed into Schwann cell profiles. The striking homogeneity of the axons suggested a sense of order, Figure 1B. In contrast, the axons of Remak bundles in the rostral dorsal root appeared less well organized, reflecting greater diversity in axon cross-sectional area (Fig. 1C, D). Quantitative analysis demonstrated that average cross-sectional area of unmyelinated axons was not different in the rostral and caudal dorsal roots (Student t-test, p 0.28). However, the variability in cross-sectional area was strikingly increased in axons of the rostral dorsal root (F-statistic, p ) when compared with the caudal dorsal root. Associated with the increased variability in axon crosssectional area in the rostral dorsal root, there were 3 times as many very large (defined as axons with area more than 2 standard deviations above the mean) unmyelinated axons. We used this variability to test the hypothesis that axonal caliber may influence the number of axons in a Remak bundle. The Remak bundles of the rostral dorsal root were evaluated with regard to the number of axons and the cross-sectional area of the largest axon in each bundle. There was a positive correlation between the number of axons in a bundle and the cross-sectional area of the largest axon in that bundle (correlation coefficient 0.62, p 0.005). A probabilistic model confirmed that this positive correlation was entirely consistent with the random assortment of axons into Remak bundles without regard to axon caliber. There was a slight excess of large
3 248 MURINSON AND GRIFFIN
4 C-FIBER STRUCTURE 249 caliber axons in single-axon Remak bundles relative to the expectations of the random assortment model. Remak Bundle Organization in the DRG Viewed in transverse section at the mid-point of the long axis of the DRG, the DRG consists of both axonal and cellular portions. In the cellular portions, a typical Remak bundle consisted of a single axon ensheathed by Schwann cell cytoplasm with a continuous basal lamina. In the axonal portions there were Remak bundles with multiple axons but in no case more than 15 axons per Remak bundle. Overall, 57% of Remak bundles in the central DRG contained 1 axon, the median number of axons per Remak bundle was 1 and the mean was 2.1. Features of Remak Bundle Organization in the Sciatic Nerve and Branches As a population, the Remak bundles of the sciatic nerve contained more axons than did those of the distal branches in the hindpaw (plantar nerves). The median number of axons per Remak bundle was 6 in the sciatic nerve and 3 in the plantar nerves. Representative Remak bundles from the sciatic nerve and plantar nerve are shown in Figure 3A and B. The distribution of axons/ Remak bundle in the sciatic and mid-plantar nerve is shown in Figure 2C and D. The Remak bundles of the sciatic nerve contained from 1 to 26 axons while those of the plantar nerve ranged from 1 to 14 axons. Comparison of these distributions showed that the number of axons per Remak bundle in the plantar nerve was lower than in the sciatic nerve (rank sum test, p 0.01). The shape of the distribution for the proximal sciatic nerve was not normal, exponential or Poisson. Interestingly, the distribution of axons into Remak bundles in the plantar nerve was heavily weighted to a small number of axons per bundle and fit well by a negative exponential distribution (p ). The influence of location on Remak bundle structure is summarized in Figure 4. The number of axons per Remak bundle increased sharply as unmyelinated axons extended outward from the DRG. In both the peripheral nerve and dorsal root, further increases in distance from the DRG were associated with a decrease in the number of axons per Remak bundle. The variability between animals was small; indicating that although the number of axons making up an individual Remak bundle was random, it was highly constrained. Within the central DRG, a large percentage of axons were supported in Remak bundles as single axons but upon exit from the DRG, single-axon Remak bundles were much less frequent, 5% in the mixed spinal nerve and 1.5% in the caudal dorsal root. Further from the DRG, the number of single-axon Remak bundles increased and was 7.2% in the plantar nerve. Even though the plantar nerves harvested in the hindpaw were no more than 1 cm from the afferent nerve terminals, the incidence of single-axon bundles there was less than 10%. The organization of Remak bundles relative to one another (tertiary structure) varied with location along the nerve axis. In the dorsal root and sciatic nerve, little tertiary structure was apparent except that near the DRG; Remak bundles with many axons were frequently found in juxtaposition (Fig. 1A). In the plantar nerve, a pattern of tertiary organization emerged; Remak bundles often clustered with other Remak bundles. Using quantitative methods we found that the typical Remak bundle cluster consisted of 2 to 6 Remak bundles in association with a small myelinated axon. Not all clusters had an associated small myelinated axon. The distribution of Remak bundles into clusters was evaluated by pooling data from the plantar nerves of several animals (Fig. 5). The number of Remak bundles per cluster was centered about a mean of 4.3 and the size distribution was truncated on the left. This distribution was well modeled by a Poisson distribution with 3.3, Poly-Axonal Pockets are Found in the Dorsal Root Most of the unmyelinated axons in the rat PNS were individually ensheathed within Schwann cell cytoplasmic extensions, physically separated from other unmyelinated axons. An exception to this was observed in the dorsal root where about 7% to 9% of axons were immediately adjacent to other axons without an intervening Schwann cell process (Fig. 1A, arrows). We refer to these structures as poly-axonal pockets. Poly-axonal pockets in the dorsal root rarely contained more than 3 axons ( 1% of Fig. 1. A: EM Photomicrograph of the L5 dorsal root near the L5 DRG, stained with uranyl acetate/lead citrate as described in methods. Unmyelinated, small myelinated, and large myelinated axons are seen. Most of the unmyelinated axons are in Remak bundles that contain large numbers of axons. Although most unmyelinated axons are isolated from one another by intervening Schwann cell cytoplasm, there are examples of axons that are immediately adjacent (described in the text as poly-axonal pockets) shown by white arrowheads. B: Remak bundle in the caudal L5 dorsal root. These axons are striking in homogeneity with respect to size and are densely assembled in the bundle. C: The Remak bundles of the rostral dorsal root (near the spinal cord) typically contain smaller numbers of axons. D: This Remak bundle from the rostral dorsal root demonstrates axons with a wide range of cross-sectional areas. Axons in the bundles of this region are more diverse in size compared with the caudal dorsal root, F-test, p Magnifications: A, B, D, 3,500; C, 6,000. Scale bars: A, 5 m; B D, 1 m.
5 250 MURINSON AND GRIFFIN all pockets); poly-axonal pockets with 2 axons were relatively common and occurred throughout the dorsal root (Table). In part because Remak bundles in the dorsal root contained many axons, we found that 28% of the Remak bundles near the DRG contained at least 1 poly-axonal pocket. Poly-axonal pockets were rare in the sciatic nerve. An additional feature of axons in the dorsal roots was a high prevalence of type 2 axon-schwann cell interactions (13). In type 2 axon-schwann cell interactions, the Schwann cell cytoplasmic projection does not fully encircle the axon so that basal lamina directly overlies part of the axonal membrane. This was in contrast to the peripheral nerve where virtually all axons were completely encircled by Schwann cells (type 1 axon-schwann cell interaction), consistent with previous studies (14). DISCUSSION This work demonstrates that most C-fiber (Remak) bundles in rat support multiple axons and that the organization of unmyelinated nerves in the rat PNS varies with location along the nerve axis. The observation that dozens of axons can be found in a single dorsal root Remak bundle indicates that multiple neurons must contribute axons to that bundle. The currently accepted ratio of centrally directed axons per sensory neuron is 2.3 (15), suggesting that a Remak bundle containing 2 dozen axons could receive axons from as many as 10 neurons. It remains to be established whether the number of axons per neuron is different for small and large DRG neurons, but it is unlikely that small neurons commonly give rise to 20 centrally directed axons. Our study is the first to definitively show that position along the nerve axis is an important factor influencing the number of axons in each Remak bundle. The amount of variation between animals in our study was small, suggesting that location along the neuraxis establishes fundamental constraints on Remak bundle structure. The factors determining the assembly of axons into Remak bundles are still undefined. Interspecies differences have been observed and Remak bundles in man, as determined distally in the sural nerve, have few axons relative to other vertebrates (16). Fig. 2. The distributions of axons into Remak bundles are displayed as histograms for selected anatomical locations. The histograms are ordered from top to bottom with respect to distance from the spinal cord: 0, 3, 5 and 15 cm. The vertical axis is number of Remak bundles and the horizontal axis is number of axons per Remak bundle. These distributions show the great variability in the number of axons per bundle and do not match any familiar probability distributions, as described in the text. A: The distribution of axons into Remak bundles for a representative rostral dorsal root near the spinal cord. The distribution appears to follow a monotonically decreasing pattern. B: In the caudal dorsal root, close to the DRG, a substantial portion of unmyelinated axons are bundled in groups of 20 or more. C: Sciatic nerve Remak bundles contained numbers of axons similar to those of the dorsal root near the spinal cord. D: Plantar nerve Remak bundles larger than 15 axons in size were not seen.
6 C-FIBER STRUCTURE 251 TABLE Distribution of Axons into Schwann Cell Pockets in the Dorsal Root Rostral dorsal root (n) Caudal dorsal root (n) 1 axon/pocket 2 axons/pocket 3 axons/pocket Percentage poly-axonal* 1069 (93%) 72 (6%) 12 (1%) (91%) 16 (7%) 3 (2%) 9.3 n Number of axons counted in each category. * Percentage of poly-axonal axons in 2-axon and 3-axon pockets. Fig. 3. EM Photomicrographs of Remak bundles in the sciatic and plantar nerves stained with uranyl acetate/lead citrate. A: Sciatic nerve Remak bundles may incorporate dozens of axons. All the axons in this figure are enclosed within a single basal lamina, although some division is suggested by the band of collagen fibers running through the bundle. Collagen was much more prominent in the peripheral nerve than in dorsal root. Although some axons are separated by only a very slender extension of Schwann cell cytoplasm, there are no poly-axonal Schwann cell pockets, as seen in the dorsal root in Figure 1A. B: Remak bundles of the plantar nerve contain fewer axons and single axon-remak bundles are much more common. Remak bundles are often found clustered in close association with other Remak bundles and with small myelinated axons, described in the text and Figure 5. Magnifications: 10,000. Scale bars: 1 m. As in human sural nerve, Remak bundles of the rat hindpaw form clusters that are identical to the groupings of Remak bundles described by Gasser as typical of skin nerves (17). These clusters are recognized by electrophysiologists who observe that C fibers often have a close physical association with 1 or more A fibers. We found the number of Remak bundles per cluster was distributed as a Poisson function, compatible with the hypothesis that the formation of clusters of Remak bundles is based largely on physical proximity. This is consistent with developmental studies of peripheral nerve which indicate that unmyelinated nerve fibers arise from a persistent core of axons that do not myelinate (18, 19). An additional factor that may play a role in determining the number of axons per Remak bundle is axon caliber. We explored this possibility in the dorsal root by measuring axon cross-sectional area (axonal area). It is known that average axonal area and diameter do not vary along the length of the S1 and S2 spinal nerve roots in the rat (20). We confirmed that mean axonal area did not vary along the length of the L5 dorsal root but noted a marked increase in the variation of axonal area in the rostral dorsal root. The reasons for this increased variation in axonal area are not known. Because there was an increased number of very large unmyelinated axons in the dorsal root near the spinal cord, we wanted to re-visit the idea that large axons drive the sorting of axons into smaller bundles, resulting in one-to-one axon-to-schwann cell association and ultimately myelination (21). In examining the relationship between the cross-sectional area of axons in a Remak bundle and the number of axons in that bundle we found that all the Remak bundles with 2 or more axons contained large caliber axons at a frequency consistent with random assortment. There were Remak bundles with single axons in which the axons were larger than expected, which may represent a premyelinating state for the axon-schwann cell unit. Important questions arise from the observation that several axons are supported in a single Remak bundle. These include questions about the electrochemical environment of C fibers in the Remak bundle, the response of nociceptors to partial nerve injury, and implications for regeneration. The role of the non-myelinating Schwann cell in defining the electrical environment of the unmyelinated axon is unresolved (17, 22) and recent studies provide direct evidence that Remak Schwann cells are electrochemically responsive to C fiber action potentials (6, 7). The electron microscopic appearance of Remak bundles prompted early concerns that unmyelinated axons were likely to interfere the function of one another (23). Detailed morphological analysis lead to the conclusion that the exchange of axons between Remak bundles was frequent enough to prevent adjacent axons
7 252 MURINSON AND GRIFFIN Fig. 5. Remak bundle clustering in the plantar nerves. This graph shows the number of Remak bundles in clusters of bundles in the plantar nerves of the hindpaw. The vertical axis is the number of clusters and the horizontal axis is number of Remak bundles per cluster. Typically 1 or 2 small myelinated nerves were found in the midst of a group of Remak bundles (Fig. 4A), but clusters could occur without small myelinated nerves. These data are pooled from several animals. An average of 4.3 Remak bundles were clustered together. The distribution was not different from normal but was well represented by a Poisson distribution ( 3.3, 2 test). Fig. 4. A: The L5 spinal root system extending from spinal cord to hindpaw, indicating location of systematic sampling. Arrows are labeled with distance from the spinal cord in cm as determined by averaging measurements from 3 animals. The sampling locations (from left to right) are referred to in the text as the rostral dorsal root, caudal dorsal root, mid-drg, mixed spinal nerve, proximal sciatic nerve, and plantar nerve. Figure is not to scale. B: Mean and median number of axons per Remak bundle. The vertical axis is the number of axons per bundle and the horizontal axis is distance from the spinal cord in cm. The mean number of axons per Remak bundle (filled circles with standard deviation bars) varied with distance from the primary sensory neuron. The median number of axons (open triangles) paralleled the mean. C: The percent of axons in bundles with a single axon is shown as open triangles and is lowest from undergoing supra-threshold voltage changes in response to nearby action potentials. This does not mean that interaction does not occur, but rather that the interaction is not sufficient to induce de novo action potentials along the length of the nerve under normal circumstances. Experimental evidence is actually in favor of limited interaction between C fiber axons. This includes coupling of C fiber action potentials in the periphery, conduction velocity speeding and slowing, as well as the detection of C fiber action potentials with complex morphology, suggesting entrainment of adjacent axons (24) (R. Meyer and S. McMahon, personal communication). It has been shown that the cross-sectional area of a Remak bundle is generally proportional to the number of axons supported by the Schwann cell and that the cross-sectional area of Schwann cell cytoplasm in a Remak bundle is approximately equal to axoplasm area (25, 26). This means that on average each unmyelinated axon is wrapped by a thickness of Schwann cell approximately 40% the radius of the axon (the solution to the equation: r 2 a (r a r c ) 2 r 2 a where r a is the axon radius and r c is the thickness of Schwann cell cytoplasm wrapping around the axon). Measurements show that the thickness of Schwann cell cytoplasm between unmyelinated axons is often much less, as little as 50 nanometers, resulting in a distance of 100 nanometers between axons. Knowing that the conductivity of this Schwann cell extension is much higher in the dorsal root near the DRG (1.5%) and highest in the center of the DRG (57%, data not shown). Distance from the spinal cord is marked on the x-axis and corresponds to the specific anatomic locations indicated in panel (A).
8 C-FIBER STRUCTURE 253 than that of myelin and that the Schwann cell is not electrically inert suggests that the multiple axons sharing Remak bundles may not be fully isolated from one another electrically. Beyond this, we find that nearly 10% of axons in the dorsal root are supported in poly-axonal pockets where electrical isolation is impossible. The possibility remains that axonal proximity may influence spatial and modality-specific discrimination of sensory inputs from the C fiber system and that certain pathological states may enhance the interaction of adjacent axons. Close proximity of unmyelinated axons in a shared Remak bundle may have implications for the response of unmyelinated axons to partial nerve injury. Electrophysiological studies following both ventral rhizotomy and mixed spinal nerve transection have shown increased spontaneous activity in C fibers (4, 5, 27, 28). The spontaneous activity of L4 C-fibers following L5 ventral rhizotomy is particularly interesting because in rat this lesion essentially spares all C-fibers from direct injury. We have demonstrated myelinated axons undergoing Wallerian degeneration closely juxtaposed to C-fibers in peripheral nerves and have proposed that this proximity implicates diffusible factors in the signaling of post-injury spontaneous activity of peripheral origin (4). Following mixed spinal nerve transection, there is an even larger percentage of L4 C-fibers exhibiting spontaneous activity. Possible explanations include the larger number of myelinated axons injured by this lesion or an interaction of unmyelinated axons arising from adjacent sensory ganglia; this is an area of ongoing research (5). In summary, degenerating nerve fibers may release factors that alter the function of neighboring uninjured axons. Because mixtures of intact and degenerating axons are almost universal to neuropathic pain models, it seems likely that post-injury events in the peripheral nerve play a contributory role. Finally, optimizing the response of Schwann cells after injury and improving C fiber regeneration are important motivations for the study of nerve fiber anatomy. Recovery of distal C fiber function takes place only over a period of months and may be incomplete (29), resulting in abnormal sensory functioning or neuropathic pain (30). There is an emerging appreciation that C fibers are diverse and have a variety of trophic factor requirements (2, 31). Our observation that Remak bundles near the DRG contain large numbers of axons shows that Remak Schwann cells support axons arising from multiple neuronal cell bodies. The idea that multiple small DRG neurons associate with single Remak Schwann cells suggests that following nerve injury, Remak Schwann cells may need to produce a variety of trophic factors to promote successful regeneration. ACKNOWLEDGMENTS The authors gratefully acknowledge the technical contributions of Mrs. Carol Rubright and Mrs. Nancy Crouse. The contributions of Dr. Yongbo Li to this work are appreciated. Thanks to Richard Meyer and Stephen McMahon for helpful discussions of this work. REFERENCES 1. Weidner C, Schmidt R, Schmelz M, Hilliges M, Handwerker HO, Torebjork HE. Time course of post-excitatory effects separates afferent human C fibre classes. J Physiol 2000;527: Schmidt R, Schmelz M, Forster C, Ringkamp M, Torebjork E, Handwerker H. Novel classes of responsive and unresponsive C nociceptors in human skin. J Neurosci 1995;15: Orstavik K, Weidner C, Schmidt R, et al. Pathological C-fibres in patients with a chronic painful condition. Brain 2003;126: Wu G, Ringkamp M, Murinson BB, et al. Degeneration of myelinated efferent fibers induces spontaneous activity in uninjured C- fiber afferents. J Neurosci 2002;22: Wu G, Ringkamp M, Hartke TV, et al. Early onset of spontaneous activity in uninjured C-fiber nociceptors after injury to neighboring nerve fibers. J Neurosci 2001;21:RC Mayer C, Quasthoff S, Grafe P. Confocal imaging reveals activitydependent intracellular Ca2 transients in nociceptive human C fibres. Pain 1999;81: Wachtler J, Mayer C, Grafe P. Activity-dependent intracellular Ca2 transients in unmyelinated nerve fibres of the isolated adult rat vagus nerve. Pflugers Arch 1998;435: Hilliges M, Weidner C, Schmelz M, et al. ATP responses in human C nociceptors. Pain 2002;98: Pease DC. Histological techniques for electron microscopy. New York: Academic Press, Millonig G. Manual of biological electron microscopy. Vercelli, Italy: Mario Saviolo, Hayat MA. Basic electron microscopic technique. New York: Van Nostrand-Reinhold, Dixon WJ, Massey FJ. Inference: Two populations. In: Dixon WJ, Massey FJ, eds. Introduction to statistical analysis. New York: Mc- Graw-Hill Book Company, 1983: Brown MJ, Martin JR, Asbury AK. Painful diabetic neuropathy. Arch Neurol 1976;33: Sima AF, Robertson DM. Peripheral neuropathy in the diabetic mutant mouse. Laboratory Investigation 1979;40: Langford LA, Coggeshall RE. Branching of sensory axons in the peripheral nerve of the rat. J Comp Neurol 1981;203: Kanda T, Tsukagoshi H, Oda M, Miyamoto K, Tanabe H. Morphological changes in unmyelinated nerve fibres in the sural nerve with age. Brain 1991;114: Gasser HS. Properties of dorsal root unmedullated fibers on the two sides of the ganglion. J Gen Physiol 1955;38: Mirsky R, Jessen KR, Brennan A, et al. Schwann cells as regulators of nerve development. J Physiol Paris 2002;96: Friede RL, Samorajski T. Myelin formation in the sciatic nerve of the rat. A quantitative electron microscopic, histochemical and radioautographic study. J Neuropathol Exp Neurol 1968;27: Suh YS, Chung K, Coggeshall RE. A study of axonal diameters and areas in lumbosacral roots and nerves in the rat. J Comp Neurol 1984;222: Friede RL, Samorajski T. Relation between the number of myelin lamellae and axon circumference in fibers of vagus and sciatic nerves of mice. J Comp Neurol 1967;130: Scriven DR. Modeling repetitive firing and bursting in a small unmyelinated nerve fiber. Biophys J 1981;35: Ochoa J. Structure and function of the peripheral nervous system. In: Dyck PJ, ed. Peripheral neuropathy. Williams and Wilkins, 1980: Meyer RA, Campbell JN. Myelinated nociceptive afferents account for the hyperalgesia that follows a burn to the hand. Science 1981; 213:
9 254 MURINSON AND GRIFFIN 25. Pannese E, Arcidiacono G, Frattola D, Rigamonti L, Procacci P, Ledda M. Quantitative relationships between axoplasm and Schwann cell sheath in unmyelinated nerve fibres. An electron microscope study. J Anat 1988;159: Samorajski T, Friede RL. Size-dependent distribution of axoplasm, Schwann cell cytoplasm, and mitochondri in the peripheral nerve fibers of mouse. Anat Rec 1968;161: Li Y, Dorsi MJ, Meyer RA, Belzberg AJ. Mechanical hyperalgesia after an L5 spinal nerve lesion in the rat is not dependent on input from injured nerve fibers. Pain 2000;85: Sheth RN, Dorsi MJ, Li Y, et al. Mechanical hyperalgesia after an L5 ventral rhizotomy or an L5 ganglionectomy in the rat. Pain 2002;96: Bester H, Allchorne AJ, Woolf CJ. Recovery of C-fiber-induced extravasation following peripheral nerve injury in the rat. Exp Neurol 1998;154: Ramer MS, Bisby MA. Adrenergic innervation of rat sensory ganglia following proximal or distal painful sciatic neuropathy: Distinct mechanisms revealed by anti- NGF treatment. Eur J Neurosci 1999; 11: Molliver DC, Snider WD. Nerve growth factor receptor TrkA is down-regulated during postnatal development by a subset of dorsal root ganglion neurons. J Comp Neurol 1997;381: Received July 1, 2003 Revision received November 25, 2003 Accepted December 2, 2003
Nervous Tissue. Neurons Neural communication Nervous Systems
 Nervous Tissue Neurons Neural communication Nervous Systems What is the function of nervous tissue? Maintain homeostasis & respond to stimuli Sense & transmit information rapidly, to specific cells and
Nervous Tissue Neurons Neural communication Nervous Systems What is the function of nervous tissue? Maintain homeostasis & respond to stimuli Sense & transmit information rapidly, to specific cells and
Nervous System Organization
 The Nervous System Nervous System Organization Receptors respond to stimuli Sensory receptors detect the stimulus Motor effectors respond to stimulus Nervous system divisions Central nervous system Command
The Nervous System Nervous System Organization Receptors respond to stimuli Sensory receptors detect the stimulus Motor effectors respond to stimulus Nervous system divisions Central nervous system Command
Nervous Tissue. Neurons Electrochemical Gradient Propagation & Transduction Neurotransmitters Temporal & Spatial Summation
 Nervous Tissue Neurons Electrochemical Gradient Propagation & Transduction Neurotransmitters Temporal & Spatial Summation What is the function of nervous tissue? Maintain homeostasis & respond to stimuli
Nervous Tissue Neurons Electrochemical Gradient Propagation & Transduction Neurotransmitters Temporal & Spatial Summation What is the function of nervous tissue? Maintain homeostasis & respond to stimuli
Xenopus laevis (Daudin)
 The Structure of Myelin Sheaths in the Central Nervous System of Xenopus laevis (Daudin) By A. PETERS, Ph.D. (From the Department of Anatomy, University of Edinburgh, Scotland) PLATES 43 To 47 (Received
The Structure of Myelin Sheaths in the Central Nervous System of Xenopus laevis (Daudin) By A. PETERS, Ph.D. (From the Department of Anatomy, University of Edinburgh, Scotland) PLATES 43 To 47 (Received
Neurons and Nervous Systems
 34 Neurons and Nervous Systems Concept 34.1 Nervous Systems Consist of Neurons and Glia Nervous systems have two categories of cells: Neurons, or nerve cells, are excitable they generate and transmit electrical
34 Neurons and Nervous Systems Concept 34.1 Nervous Systems Consist of Neurons and Glia Nervous systems have two categories of cells: Neurons, or nerve cells, are excitable they generate and transmit electrical
The Nervous System. Nervous System Organization. Nerve Tissue. Two parts to the nervous system 11/27/2016
 The Nervous System Nervous System Organization Animals must be able to respond to environmental stimuli. Three functions of the nervous system: Sensory input conduction of signals from sensory receptors.
The Nervous System Nervous System Organization Animals must be able to respond to environmental stimuli. Three functions of the nervous system: Sensory input conduction of signals from sensory receptors.
Nervous System Organization
 The Nervous System Chapter 44 Nervous System Organization All animals must be able to respond to environmental stimuli -Sensory receptors = Detect stimulus -Motor effectors = Respond to it -The nervous
The Nervous System Chapter 44 Nervous System Organization All animals must be able to respond to environmental stimuli -Sensory receptors = Detect stimulus -Motor effectors = Respond to it -The nervous
NEURONS, SENSE ORGANS, AND NERVOUS SYSTEMS CHAPTER 34
 NEURONS, SENSE ORGANS, AND NERVOUS SYSTEMS CHAPTER 34 KEY CONCEPTS 34.1 Nervous Systems Are Composed of Neurons and Glial Cells 34.2 Neurons Generate Electric Signals by Controlling Ion Distributions 34.3
NEURONS, SENSE ORGANS, AND NERVOUS SYSTEMS CHAPTER 34 KEY CONCEPTS 34.1 Nervous Systems Are Composed of Neurons and Glial Cells 34.2 Neurons Generate Electric Signals by Controlling Ion Distributions 34.3
Cellular Neuroanatomy II The Prototypical Neuron: Neurites. Reading: BCP Chapter 2
 Cellular Neuroanatomy II The Prototypical Neuron: Neurites Reading: BCP Chapter 2 Major Internal Features of a Neuron The neuron is the functional unit of the nervous system. A typical neuron has a soma
Cellular Neuroanatomy II The Prototypical Neuron: Neurites Reading: BCP Chapter 2 Major Internal Features of a Neuron The neuron is the functional unit of the nervous system. A typical neuron has a soma
Aberrant Mitochondria with Longitudinal Cristae Observed in the Normal Rat Hepatic Parenchymal Cell. Takuma Saito and Kazuo Ozawa
 Okajimas Fol. anat. jap., 44 : 357-363, 1968 Aberrant Mitochondria with Longitudinal Cristae Observed in the Normal Rat Hepatic Parenchymal Cell By Takuma Saito and Kazuo Ozawa Department of Anatomy, Kansai
Okajimas Fol. anat. jap., 44 : 357-363, 1968 Aberrant Mitochondria with Longitudinal Cristae Observed in the Normal Rat Hepatic Parenchymal Cell By Takuma Saito and Kazuo Ozawa Department of Anatomy, Kansai
Neurochemistry 1. Nervous system is made of neurons & glia, as well as other cells. Santiago Ramon y Cajal Nobel Prize 1906
 Neurochemistry 1 Nervous system is made of neurons & glia, as well as other cells. Santiago Ramon y Cajal Nobel Prize 1906 How Many Neurons Do We Have? The human brain contains ~86 billion neurons and
Neurochemistry 1 Nervous system is made of neurons & glia, as well as other cells. Santiago Ramon y Cajal Nobel Prize 1906 How Many Neurons Do We Have? The human brain contains ~86 billion neurons and
Overview Organization: Central Nervous System (CNS) Peripheral Nervous System (PNS) innervate Divisions: a. Afferent
 Overview Organization: Central Nervous System (CNS) Brain and spinal cord receives and processes information. Peripheral Nervous System (PNS) Nerve cells that link CNS with organs throughout the body.
Overview Organization: Central Nervous System (CNS) Brain and spinal cord receives and processes information. Peripheral Nervous System (PNS) Nerve cells that link CNS with organs throughout the body.
Ch 7. The Nervous System 7.1 & 7.2
 Ch 7 The Nervous System 7.1 & 7.2 SLOs Describe the different types of neurons and supporting cells, and identify their functions. Identify the myelin sheath and describe how it is formed in the CNS and
Ch 7 The Nervous System 7.1 & 7.2 SLOs Describe the different types of neurons and supporting cells, and identify their functions. Identify the myelin sheath and describe how it is formed in the CNS and
CONTRACTION BANDS AT SHORT SARCOMERE LENGTH IN CHICK MUSCLE
 CONTRACTION BANDS AT SHORT SARCOMERE LENGTH IN CHICK MUSCLE MARTIN HAGOPIAN. From the Department of Pathology, New York Medical College, New York 10029 INTRODUCTION The sliding filament model for contraction
CONTRACTION BANDS AT SHORT SARCOMERE LENGTH IN CHICK MUSCLE MARTIN HAGOPIAN. From the Department of Pathology, New York Medical College, New York 10029 INTRODUCTION The sliding filament model for contraction
Information processing. Divisions of nervous system. Neuron structure and function Synapse. Neurons, synapses, and signaling 11/3/2017
 Neurons, synapses, and signaling Chapter 48 Information processing Divisions of nervous system Central nervous system (CNS) Brain and a nerve cord Integration center Peripheral nervous system (PNS) Nerves
Neurons, synapses, and signaling Chapter 48 Information processing Divisions of nervous system Central nervous system (CNS) Brain and a nerve cord Integration center Peripheral nervous system (PNS) Nerves
Physiology 2 nd year. Neuroscience Optional Lecture
 Academic year 2018/2019 Physiology 2 nd year Semester 1 Curricula Nervous system physiology Blood physiology Acid-base equilibrium Bibliography: Boron & Boulpaep Medical Physiology, 3 rd edition Physiology
Academic year 2018/2019 Physiology 2 nd year Semester 1 Curricula Nervous system physiology Blood physiology Acid-base equilibrium Bibliography: Boron & Boulpaep Medical Physiology, 3 rd edition Physiology
Nervous Systems: Neuron Structure and Function
 Nervous Systems: Neuron Structure and Function Integration An animal needs to function like a coherent organism, not like a loose collection of cells. Integration = refers to processes such as summation
Nervous Systems: Neuron Structure and Function Integration An animal needs to function like a coherent organism, not like a loose collection of cells. Integration = refers to processes such as summation
Atypical Neural Sheaths Formed by Muller Cells in Chicken Retina
 Okajimas Folia Anat. Jpn., 57(2-3) : 79-88, August 1980 Atypical Neural Sheaths Formed by Muller Cells in Chicken Retina By YOSHIRO INOUE, YOSHIHIRO SUGIHARA,Yozo NISHIMURA and KAZUYO SHIMAI Department
Okajimas Folia Anat. Jpn., 57(2-3) : 79-88, August 1980 Atypical Neural Sheaths Formed by Muller Cells in Chicken Retina By YOSHIRO INOUE, YOSHIHIRO SUGIHARA,Yozo NISHIMURA and KAZUYO SHIMAI Department
Neurons, Synapses, and Signaling
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 48 Neurons, Synapses, and Signaling
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 48 Neurons, Synapses, and Signaling
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo
 Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
The growth rate of sensory nerve fibres in the mammalian embryo
 Development 00, 307-3 (987) Printed in Great Britain The Company of Biologists Limited 987 307 The growth rate of sensory nerve fibres in the mammalian embryo ALUN M. DAVIES Department of Anatomy, Si George's
Development 00, 307-3 (987) Printed in Great Britain The Company of Biologists Limited 987 307 The growth rate of sensory nerve fibres in the mammalian embryo ALUN M. DAVIES Department of Anatomy, Si George's
Chapter 48 Neurons, Synapses, and Signaling
 Chapter 48 Neurons, Synapses, and Signaling Concept 48.1 Neuron organization and structure reflect function in information transfer Neurons are nerve cells that transfer information within the body Neurons
Chapter 48 Neurons, Synapses, and Signaling Concept 48.1 Neuron organization and structure reflect function in information transfer Neurons are nerve cells that transfer information within the body Neurons
Introduction Principles of Signaling and Organization p. 3 Signaling in Simple Neuronal Circuits p. 4 Organization of the Retina p.
 Introduction Principles of Signaling and Organization p. 3 Signaling in Simple Neuronal Circuits p. 4 Organization of the Retina p. 5 Signaling in Nerve Cells p. 9 Cellular and Molecular Biology of Neurons
Introduction Principles of Signaling and Organization p. 3 Signaling in Simple Neuronal Circuits p. 4 Organization of the Retina p. 5 Signaling in Nerve Cells p. 9 Cellular and Molecular Biology of Neurons
2401 : Anatomy/Physiology
 Dr. Chris Doumen Week 6 2401 : Anatomy/Physiology Action Potentials NeuroPhysiology TextBook Readings Pages 400 through 408 Make use of the figures in your textbook ; a picture is worth a thousand words!
Dr. Chris Doumen Week 6 2401 : Anatomy/Physiology Action Potentials NeuroPhysiology TextBook Readings Pages 400 through 408 Make use of the figures in your textbook ; a picture is worth a thousand words!
BIOLOGY. 1. Overview of Neurons 11/3/2014. Neurons, Synapses, and Signaling. Communication in Neurons
 CAMPBELL BIOLOGY TENTH EDITION 48 Reece Urry Cain Wasserman Minorsky Jackson Neurons, Synapses, and Signaling Lecture Presentation by Nicole Tunbridge and Kathleen Fitzpatrick 1. Overview of Neurons Communication
CAMPBELL BIOLOGY TENTH EDITION 48 Reece Urry Cain Wasserman Minorsky Jackson Neurons, Synapses, and Signaling Lecture Presentation by Nicole Tunbridge and Kathleen Fitzpatrick 1. Overview of Neurons Communication
The Use of Teased Preparations and Frozen Sections in Quantitative Studies of Mammalian Peripheral Nerve. By C. P. WENDELL-SMITH AND P. L.
 499 The Use of Teased Preparations and Frozen Sections in Quantitative Studies of Mammalian Peripheral Nerve By C. P. WENDELL-SMITH AND P. L. WILLIAMS (From the Department of Anatomy, Guy's Hospital Medical
499 The Use of Teased Preparations and Frozen Sections in Quantitative Studies of Mammalian Peripheral Nerve By C. P. WENDELL-SMITH AND P. L. WILLIAMS (From the Department of Anatomy, Guy's Hospital Medical
PROPERTY OF ELSEVIER SAMPLE CONTENT - NOT FINAL. The Nervous System and Muscle
 The Nervous System and Muscle SECTION 2 2-1 Nernst Potential 2-2 Resting Membrane Potential 2-3 Axonal Action Potential 2-4 Neurons 2-5 Axonal Conduction 2-6 Morphology of Synapses 2-7 Chemical Synaptic
The Nervous System and Muscle SECTION 2 2-1 Nernst Potential 2-2 Resting Membrane Potential 2-3 Axonal Action Potential 2-4 Neurons 2-5 Axonal Conduction 2-6 Morphology of Synapses 2-7 Chemical Synaptic
Cells. Steven McLoon Department of Neuroscience University of Minnesota
 Cells Steven McLoon Department of Neuroscience University of Minnesota 1 Microscopy Methods of histology: Treat the tissue with a preservative (e.g. formaldehyde). Dissect the region of interest. Embed
Cells Steven McLoon Department of Neuroscience University of Minnesota 1 Microscopy Methods of histology: Treat the tissue with a preservative (e.g. formaldehyde). Dissect the region of interest. Embed
Tokuhiro JSHIHARA, Chotatsu TSUKAYAMA, Fumiya UCHINO
 (39) JOURNAL OF ELECTRON MICROSCOPY 39 Vol. 22, No. I, 39-44, 1973 Intramitochondrial Filamentous Structures in Human Reticulum Cells in the Bone Marrow Tokuhiro JSHIHARA, Chotatsu TSUKAYAMA, Fumiya UCHINO
(39) JOURNAL OF ELECTRON MICROSCOPY 39 Vol. 22, No. I, 39-44, 1973 Intramitochondrial Filamentous Structures in Human Reticulum Cells in the Bone Marrow Tokuhiro JSHIHARA, Chotatsu TSUKAYAMA, Fumiya UCHINO
Nervous system. 3 Basic functions of the nervous system !!!! !!! 1-Sensory. 2-Integration. 3-Motor
 Nervous system 3 Basic functions of the nervous system 1-Sensory 2-Integration 3-Motor I. Central Nervous System (CNS) Brain Spinal Cord I. Peripheral Nervous System (PNS) 2) Afferent towards afferent
Nervous system 3 Basic functions of the nervous system 1-Sensory 2-Integration 3-Motor I. Central Nervous System (CNS) Brain Spinal Cord I. Peripheral Nervous System (PNS) 2) Afferent towards afferent
Curtis et al. Il nuovo Invito alla biologia.blu BIOLOGY HIGHLIGHTS KEYS
 BIOLOGY HIGHLIGHTS KEYS Watch the videos and download the transcripts of this section at: online.scuola.zanichelli.it/curtisnuovoinvitoblu/clil > THE HUMAN NERVOUS SYSTEM 2. WARM UP a) The structures that
BIOLOGY HIGHLIGHTS KEYS Watch the videos and download the transcripts of this section at: online.scuola.zanichelli.it/curtisnuovoinvitoblu/clil > THE HUMAN NERVOUS SYSTEM 2. WARM UP a) The structures that
Morphology and Ultrastructure of Staphylococcal L Colonies: Light, Scanning,
 JOURNAL OF BACTERIOLOGY, Feb. 1973, p. 1049-1053 Copyright ( 1973 American Society for Microbiology Vol. 113, No. 2 Printed in U.S.A. Morphology and Ultrastructure of Staphylococcal L Colonies: Light,
JOURNAL OF BACTERIOLOGY, Feb. 1973, p. 1049-1053 Copyright ( 1973 American Society for Microbiology Vol. 113, No. 2 Printed in U.S.A. Morphology and Ultrastructure of Staphylococcal L Colonies: Light,
Neural Tissue. PowerPoint Lecture Presentations prepared by Jason LaPres. Lone Star College North Harris Pearson Education, Inc.
 12 Neural Tissue PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris An Introduction to the Nervous System The Nervous System Includes all neural tissue in the body
12 Neural Tissue PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris An Introduction to the Nervous System The Nervous System Includes all neural tissue in the body
Dendrites - receives information from other neuron cells - input receivers.
 The Nerve Tissue Neuron - the nerve cell Dendrites - receives information from other neuron cells - input receivers. Cell body - includes usual parts of the organelles of a cell (nucleus, mitochondria)
The Nerve Tissue Neuron - the nerve cell Dendrites - receives information from other neuron cells - input receivers. Cell body - includes usual parts of the organelles of a cell (nucleus, mitochondria)
University of Jordan School of Medicine MD Program Curriculum
 University of Jordan School of Medicine MD Program Curriculum Course Title: General Histology Course number: 0502111 Credit Hours: 2 credits Academic Year Level: 1st year, 2nd semester 2016/2017 Course
University of Jordan School of Medicine MD Program Curriculum Course Title: General Histology Course number: 0502111 Credit Hours: 2 credits Academic Year Level: 1st year, 2nd semester 2016/2017 Course
Chapter 37 Active Reading Guide Neurons, Synapses, and Signaling
 Name: AP Biology Mr. Croft Section 1 1. What is a neuron? Chapter 37 Active Reading Guide Neurons, Synapses, and Signaling 2. Neurons can be placed into three groups, based on their location and function.
Name: AP Biology Mr. Croft Section 1 1. What is a neuron? Chapter 37 Active Reading Guide Neurons, Synapses, and Signaling 2. Neurons can be placed into three groups, based on their location and function.
Introduction and summary of the chapters
 Introduction and summary of the chapters 1. Electroreception Electroreception is the ability of animal species to detect weak electric fields. It is mediated by a sensory system that occurs in some aquatic
Introduction and summary of the chapters 1. Electroreception Electroreception is the ability of animal species to detect weak electric fields. It is mediated by a sensory system that occurs in some aquatic
Universality of sensory-response systems
 excite.org(anism): Electrical Signaling Universality of sensory-response systems Three step process: sensation-integration-response Bacterial chemotaxis Madigan et al. Fig. 8.24 Rick Stewart (CBMG) Human
excite.org(anism): Electrical Signaling Universality of sensory-response systems Three step process: sensation-integration-response Bacterial chemotaxis Madigan et al. Fig. 8.24 Rick Stewart (CBMG) Human
Electron and Light Microscope Studies of Endamoeba terrapinae
 Proceedings of the Iowa Academy of Science Volume 68 Annual Issue Article 81 1961 Electron and Light Microscope Studies of Endamoeba terrapinae Marilyn Driml Cornell College Copyright Copyright 1961 by
Proceedings of the Iowa Academy of Science Volume 68 Annual Issue Article 81 1961 Electron and Light Microscope Studies of Endamoeba terrapinae Marilyn Driml Cornell College Copyright Copyright 1961 by
Neurons, Synapses, and Signaling
 Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Intravital Imaging Reveals Ghost Fibers as Architectural Units Guiding Myogenic Progenitors during Regeneration
 Cell Stem Cell Supplemental Information Intravital Imaging Reveals Ghost Fibers as Architectural Units Guiding Myogenic Progenitors during Regeneration Micah T. Webster, Uri Manor, Jennifer Lippincott-Schwartz,
Cell Stem Cell Supplemental Information Intravital Imaging Reveals Ghost Fibers as Architectural Units Guiding Myogenic Progenitors during Regeneration Micah T. Webster, Uri Manor, Jennifer Lippincott-Schwartz,
Chapter 9. Nerve Signals and Homeostasis
 Chapter 9 Nerve Signals and Homeostasis A neuron is a specialized nerve cell that is the functional unit of the nervous system. Neural signaling communication by neurons is the process by which an animal
Chapter 9 Nerve Signals and Homeostasis A neuron is a specialized nerve cell that is the functional unit of the nervous system. Neural signaling communication by neurons is the process by which an animal
Supplementary Figure 1-10 with Figure legends
 332 coordinates mechanotransduction and growth cone bifurcation in sensory neurons Li Yang Chiang 1, Kate Poole, Beatriz E. Oliveira, Neuza Duarte,Yinth Andrea Bernal Sierra, Leena Bruckner Tuderman, Manuel
332 coordinates mechanotransduction and growth cone bifurcation in sensory neurons Li Yang Chiang 1, Kate Poole, Beatriz E. Oliveira, Neuza Duarte,Yinth Andrea Bernal Sierra, Leena Bruckner Tuderman, Manuel
BIOLOGY 11/10/2016. Neurons, Synapses, and Signaling. Concept 48.1: Neuron organization and structure reflect function in information transfer
 48 Neurons, Synapses, and Signaling CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Lecture Presentation by Nicole Tunbridge and Kathleen Fitzpatrick Concept 48.1: Neuron organization
48 Neurons, Synapses, and Signaling CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Lecture Presentation by Nicole Tunbridge and Kathleen Fitzpatrick Concept 48.1: Neuron organization
Effects of Betaxolol on Hodgkin-Huxley Model of Tiger Salamander Retinal Ganglion Cell
 Effects of Betaxolol on Hodgkin-Huxley Model of Tiger Salamander Retinal Ganglion Cell 1. Abstract Matthew Dunlevie Clement Lee Indrani Mikkilineni mdunlevi@ucsd.edu cll008@ucsd.edu imikkili@ucsd.edu Isolated
Effects of Betaxolol on Hodgkin-Huxley Model of Tiger Salamander Retinal Ganglion Cell 1. Abstract Matthew Dunlevie Clement Lee Indrani Mikkilineni mdunlevi@ucsd.edu cll008@ucsd.edu imikkili@ucsd.edu Isolated
Control and Integration. Nervous System Organization: Bilateral Symmetric Animals. Nervous System Organization: Radial Symmetric Animals
 Control and Integration Neurophysiology Chapters 10-12 Nervous system composed of nervous tissue cells designed to conduct electrical impulses rapid communication to specific cells or groups of cells Endocrine
Control and Integration Neurophysiology Chapters 10-12 Nervous system composed of nervous tissue cells designed to conduct electrical impulses rapid communication to specific cells or groups of cells Endocrine
BIOL Anatomy and Physiology I ( version L )
 BIOL 2113 - Anatomy and Physiology I ( version 213L ) Course Title Course Development Learning Support Anatomy and Physiology I Standard No Course Description Introduces the anatomy and physiology of the
BIOL 2113 - Anatomy and Physiology I ( version 213L ) Course Title Course Development Learning Support Anatomy and Physiology I Standard No Course Description Introduces the anatomy and physiology of the
Neurons. General neuron anatomy. BIOL 164 Human Biology Ch 7 Neurons. Nervous system:
 BIOL 164 Human Biology Ch 7 Neurons Nervous system: Neurons Integrates and coordinates the body s ac3vi3es Provides rapid and brief responses to s3muli Major divisions: Central nervous system (CNS) brain
BIOL 164 Human Biology Ch 7 Neurons Nervous system: Neurons Integrates and coordinates the body s ac3vi3es Provides rapid and brief responses to s3muli Major divisions: Central nervous system (CNS) brain
MIT 9.14 Class 2 Neuroanatomical techniques
 9.14 - Brain Structure and its Origins Spring 2005 Massachusetts Institute of Technology Instructor: Professor Gerald Schneider A sketch of the central nervous system and its origins G. Schneider 2005
9.14 - Brain Structure and its Origins Spring 2005 Massachusetts Institute of Technology Instructor: Professor Gerald Schneider A sketch of the central nervous system and its origins G. Schneider 2005
The Nervous System. What did you learn at school today? Neurophysiology!
 The Nervous System What did you learn at school today? Neurophysiology! The Nervous System Controls heart rate, emotions, memories, consciousness, and much more. The most intricate and beautifully complex
The Nervous System What did you learn at school today? Neurophysiology! The Nervous System Controls heart rate, emotions, memories, consciousness, and much more. The most intricate and beautifully complex
Action Potentials & Nervous System. Bio 219 Napa Valley College Dr. Adam Ross
 Action Potentials & Nervous System Bio 219 Napa Valley College Dr. Adam Ross Review: Membrane potentials exist due to unequal distribution of charge across the membrane Concentration gradients drive ion
Action Potentials & Nervous System Bio 219 Napa Valley College Dr. Adam Ross Review: Membrane potentials exist due to unequal distribution of charge across the membrane Concentration gradients drive ion
Neurophysiology. Danil Hammoudi.MD
 Neurophysiology Danil Hammoudi.MD ACTION POTENTIAL An action potential is a wave of electrical discharge that travels along the membrane of a cell. Action potentials are an essential feature of animal
Neurophysiology Danil Hammoudi.MD ACTION POTENTIAL An action potential is a wave of electrical discharge that travels along the membrane of a cell. Action potentials are an essential feature of animal
Neurons, Synapses, and Signaling
 Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
MEMBRANE POTENTIALS AND ACTION POTENTIALS:
 University of Jordan Faculty of Medicine Department of Physiology & Biochemistry Medical students, 2017/2018 +++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ Review: Membrane physiology
University of Jordan Faculty of Medicine Department of Physiology & Biochemistry Medical students, 2017/2018 +++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ Review: Membrane physiology
Ch 33. The nervous system
 Ch 33 The nervous system AP bio schedule Tuesday Wed Thursday Friday Plant test Animal behavior lab Nervous system 25 Review Day (bring computer) 27 Review Day (bring computer) 28 Practice AP bio test
Ch 33 The nervous system AP bio schedule Tuesday Wed Thursday Friday Plant test Animal behavior lab Nervous system 25 Review Day (bring computer) 27 Review Day (bring computer) 28 Practice AP bio test
Supplementary Information
 Supplementary Information Tuning Ranvier node and internode properties in myelinated axons to adjust action potential timing Marc C. Ford, Olga Alexandrova, Lee Cossell, Annette Stange Marten, James Sinclair,
Supplementary Information Tuning Ranvier node and internode properties in myelinated axons to adjust action potential timing Marc C. Ford, Olga Alexandrova, Lee Cossell, Annette Stange Marten, James Sinclair,
Electrical Signaling. Lecture Outline. Using Ions as Messengers. Potentials in Electrical Signaling
 Lecture Outline Electrical Signaling Using ions as messengers Potentials in electrical signaling Action Graded Other electrical signaling Gap junctions The neuron Using Ions as Messengers Important things
Lecture Outline Electrical Signaling Using ions as messengers Potentials in electrical signaling Action Graded Other electrical signaling Gap junctions The neuron Using Ions as Messengers Important things
Nervous System Part II
 Nervous System Part II 175 Types of Neurons 1. Motor Neurons 2. Sensory Neurons 3. Interneurons 176 Motor (Efferent) Neurons take information from the CNS to effectors (muscles or glands). Characterized
Nervous System Part II 175 Types of Neurons 1. Motor Neurons 2. Sensory Neurons 3. Interneurons 176 Motor (Efferent) Neurons take information from the CNS to effectors (muscles or glands). Characterized
NOTE: LOOK ON MY WEBSITE FOR THE MUSCLE LABELING POWER POINT/PDF Part I. Identify the parts of the neuron that are labeled below.
 Anatomy & Physiology Nervous System Part I 2/26/16 NOTE: LOOK ON MY WEBSITE FOR THE MUSCLE LABELING POWER POINT/PDF Part I. Identify the parts of the neuron that are labeled below. 1. 2. 3. 5. 4. 6. Part
Anatomy & Physiology Nervous System Part I 2/26/16 NOTE: LOOK ON MY WEBSITE FOR THE MUSCLE LABELING POWER POINT/PDF Part I. Identify the parts of the neuron that are labeled below. 1. 2. 3. 5. 4. 6. Part
Brunswick School Department: Grades Essential Understandings
 Essential Understandings Essential Questions Essential Knowledge Vocabulary Essential Skills The Nervous system controls all voluntary and involuntary actions of the body. Neurons are the main functional
Essential Understandings Essential Questions Essential Knowledge Vocabulary Essential Skills The Nervous system controls all voluntary and involuntary actions of the body. Neurons are the main functional
A Dynamical Model of Fast Intrinsic Optical Signal of Neural Burstings
 A Dymical Model of Fast Intrinsic Optical Sigl of Neural Burstings Jianzhong Su Hanli Liu Yuanbo Peng Technical Report 2007-20 http://www.uta.edu/math/preprint/ A Dymical Model of Fast Intrinsic Optical
A Dymical Model of Fast Intrinsic Optical Sigl of Neural Burstings Jianzhong Su Hanli Liu Yuanbo Peng Technical Report 2007-20 http://www.uta.edu/math/preprint/ A Dymical Model of Fast Intrinsic Optical
12-The Nervous System. Taft College Human Physiology
 12-The Nervous System Taft College Human Physiology Introduction To The Nervous System The nervous system is a wired system with discrete pathways (nerves) and local actions. The effects of nervous stimulation
12-The Nervous System Taft College Human Physiology Introduction To The Nervous System The nervous system is a wired system with discrete pathways (nerves) and local actions. The effects of nervous stimulation
Experiment 3: Nerve Conduction
 Experiment 3: Nerve Conduction Overview The interior of a cell is negatively charged with respect to the outside, and the magnitude of the potential difference is usually between 50 and 80 mv. Some cells,
Experiment 3: Nerve Conduction Overview The interior of a cell is negatively charged with respect to the outside, and the magnitude of the potential difference is usually between 50 and 80 mv. Some cells,
Neurons, Synapses, and Signaling
 Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lectures for Biology, Eighth Edition Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp and Janette Lewis Copyright
Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lectures for Biology, Eighth Edition Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp and Janette Lewis Copyright
LOW-POWER ELECTRON MICROSCOPY OF THE ROOT CAP REGION OF EUCALYPT MYCORRHIZAS
 New Phytol. (1968) 67, 663-665. LOW-POWER ELECTRON MICROSCOPY OF THE ROOT CAP REGION OF EUCALYPT MYCORRHIZAS BY G. A. CHILVERS Botany Department, School of General Studies, Australian National University,
New Phytol. (1968) 67, 663-665. LOW-POWER ELECTRON MICROSCOPY OF THE ROOT CAP REGION OF EUCALYPT MYCORRHIZAS BY G. A. CHILVERS Botany Department, School of General Studies, Australian National University,
Geert Geeven. April 14, 2010
 iction of Gene Regulatory Interactions NDNS+ Workshop April 14, 2010 Today s talk - Outline Outline Biological Background Construction of Predictors The main aim of my project is to better understand the
iction of Gene Regulatory Interactions NDNS+ Workshop April 14, 2010 Today s talk - Outline Outline Biological Background Construction of Predictors The main aim of my project is to better understand the
Cellular Neurobiology BIPN 140 Fall 2016 Problem Set #8
 Cellular Neurobiology BIPN 140 Fall 2016 Problem Set #8 1. Inductive signaling is a hallmark of vertebrate and mammalian development. In early neural development, there are multiple signaling pathways
Cellular Neurobiology BIPN 140 Fall 2016 Problem Set #8 1. Inductive signaling is a hallmark of vertebrate and mammalian development. In early neural development, there are multiple signaling pathways
Single-Compartment Neural Models
 Single-Compartment Neural Models BENG/BGGN 260 Neurodynamics University of California, San Diego Week 2 BENG/BGGN 260 Neurodynamics (UCSD) Single-Compartment Neural Models Week 2 1 / 18 Reading Materials
Single-Compartment Neural Models BENG/BGGN 260 Neurodynamics University of California, San Diego Week 2 BENG/BGGN 260 Neurodynamics (UCSD) Single-Compartment Neural Models Week 2 1 / 18 Reading Materials
Introduction to Neural Networks U. Minn. Psy 5038 Spring, 1999 Daniel Kersten. Lecture 2a. The Neuron - overview of structure. From Anderson (1995)
 Introduction to Neural Networks U. Minn. Psy 5038 Spring, 1999 Daniel Kersten Lecture 2a The Neuron - overview of structure From Anderson (1995) 2 Lect_2a_Mathematica.nb Basic Structure Information flow:
Introduction to Neural Networks U. Minn. Psy 5038 Spring, 1999 Daniel Kersten Lecture 2a The Neuron - overview of structure From Anderson (1995) 2 Lect_2a_Mathematica.nb Basic Structure Information flow:
THE FREQUENCY OF HETEROCYSTS IN THE NOSTOC PHYCOBIONT OF THE LICHEN PELTIGERA CANINA WILLD.
 New Phytol. (1972) 71, 11-13. THE FREQUENCY OF HETEROCYSTS IN THE NOSTOC PHYCOBIONT OF THE LICHEN PELTIGERA CANINA WILLD. BY H. BRONWEN GRIFFITHS, A. D. GREENWOOD AND J. W. MILLBANK Department of Botany,
New Phytol. (1972) 71, 11-13. THE FREQUENCY OF HETEROCYSTS IN THE NOSTOC PHYCOBIONT OF THE LICHEN PELTIGERA CANINA WILLD. BY H. BRONWEN GRIFFITHS, A. D. GREENWOOD AND J. W. MILLBANK Department of Botany,
Nervous System: Part II How A Neuron Works
 Nervous System: Part II How A Neuron Works Essential Knowledge Statement 3.E.2 Continued Animals have nervous systems that detect external and internal signals, transmit and integrate information, and
Nervous System: Part II How A Neuron Works Essential Knowledge Statement 3.E.2 Continued Animals have nervous systems that detect external and internal signals, transmit and integrate information, and
Association of Tobacco Rattle Virus with Mitochondria
 J. gen. ViroL (I968), 3, I2I-I24 With 3 plates Printed in Great Britain I2I Association of Tobacco Rattle Virus with Mitochondria (Accepted 8 February I968) As part of a study of the way in which tobacco
J. gen. ViroL (I968), 3, I2I-I24 With 3 plates Printed in Great Britain I2I Association of Tobacco Rattle Virus with Mitochondria (Accepted 8 February I968) As part of a study of the way in which tobacco
Purpose: Perception, Movement, Learning, Memory, Thinking, Communication Functions:
 Nervous System Purpose: Perception, Movement, Learning, Memory, Thinking, Communication Functions: Sensory Input: Obtaining stimulation from the environment (light, heat, pressure, vibration, chemical,
Nervous System Purpose: Perception, Movement, Learning, Memory, Thinking, Communication Functions: Sensory Input: Obtaining stimulation from the environment (light, heat, pressure, vibration, chemical,
Transmission Electron Microscope Technique for Risk Assessment of Manufactured Nanomaterials
 Transmission Electron Microscope Technique for Risk Assessment of Manufactured Nanomaterials Kazuhiro Yamamoto and Miyabi Makino National Institute of Advanced Industrial Science and Technology (AIST),
Transmission Electron Microscope Technique for Risk Assessment of Manufactured Nanomaterials Kazuhiro Yamamoto and Miyabi Makino National Institute of Advanced Industrial Science and Technology (AIST),
HUMAN ANATOMY & PHYSIOLOGY STRUCTURE 30
 HUMAN ANATOMY & PHYSIOLOGY STRUCTURE 30 Description This second-year course in biology emphasizes the workings of the human body. The course is offered in the fall semester and meets six (6) periods per
HUMAN ANATOMY & PHYSIOLOGY STRUCTURE 30 Description This second-year course in biology emphasizes the workings of the human body. The course is offered in the fall semester and meets six (6) periods per
THE LOCALIZATION OF FUNCTION IN THE ROOT OF AN INSECT SEGMENTAL NERVE
 Exp. Biol. (1963), 40, SS3-s6i 553 A 2 plates and 2 text-figures Printed in Great Britain THE LOCALIZATION OF FUNCTION IN THE ROOT OF AN INSECT SEGMENTAL NERVE BY ANN FIELDEN* Department of Zoology, University
Exp. Biol. (1963), 40, SS3-s6i 553 A 2 plates and 2 text-figures Printed in Great Britain THE LOCALIZATION OF FUNCTION IN THE ROOT OF AN INSECT SEGMENTAL NERVE BY ANN FIELDEN* Department of Zoology, University
* ra. VOL. 53, 1965 ANATOMY: D. BODIAN 419. FIG. 1.-Synaptic contact in spinal motoneuron of a chimpanzee. Large dendrite (D) shows
 A SUGGESTIVE RELATIONSHIP OF NERVE CELL RNA WITh1 SPECIFIC SYNAPTIC SITES* BY DAVID BODIAN DEPARTMENT OF ANATOMY, JOHNS HOPKINS UNIVERSITY SCHOOL OF MEDICINE Communicated December 29, 1964 In a previous
A SUGGESTIVE RELATIONSHIP OF NERVE CELL RNA WITh1 SPECIFIC SYNAPTIC SITES* BY DAVID BODIAN DEPARTMENT OF ANATOMY, JOHNS HOPKINS UNIVERSITY SCHOOL OF MEDICINE Communicated December 29, 1964 In a previous
Ch 8: Neurons: Cellular and Network Properties, Part 1
 Developed by John Gallagher, MS, DVM Ch 8: Neurons: Cellular and Network Properties, Part 1 Objectives: Describe the Cells of the NS Explain the creation and propagation of an electrical signal in a nerve
Developed by John Gallagher, MS, DVM Ch 8: Neurons: Cellular and Network Properties, Part 1 Objectives: Describe the Cells of the NS Explain the creation and propagation of an electrical signal in a nerve
Detailed Learning Outcomes
 Detailed Learning Outcomes Following lectures, workshops and directed study activities, students should: 1. Understand the principles behind the study of bioscience in relation to the profession of SLT
Detailed Learning Outcomes Following lectures, workshops and directed study activities, students should: 1. Understand the principles behind the study of bioscience in relation to the profession of SLT
Biomedical Instrumentation
 Biomedical Instrumentation Winter 1393 Bonab University The Origin of BioPotentials Bioelectric Signals Bioelectrical potential is a result of electrochemical activity across the membrane of the cell.
Biomedical Instrumentation Winter 1393 Bonab University The Origin of BioPotentials Bioelectric Signals Bioelectrical potential is a result of electrochemical activity across the membrane of the cell.
The texts, tables and images contained in this lecture notes are not my own, they can be found on: References supplied
 The texts, tables and images contained in this lecture notes are not my own, they can be found on: References supplied Atlases or The web sites Source: http://www.science.smith.edu/departments/biochem/biochem_353/buffer_pre
The texts, tables and images contained in this lecture notes are not my own, they can be found on: References supplied Atlases or The web sites Source: http://www.science.smith.edu/departments/biochem/biochem_353/buffer_pre
Animal structure and function
 Animal structure and function The nervous system Parts of the nervous system 43C, 44B, 45D Brain structure and function Eyes Retina Neurons: How neurons communicate: Resting potential: The resting
Animal structure and function The nervous system Parts of the nervous system 43C, 44B, 45D Brain structure and function Eyes Retina Neurons: How neurons communicate: Resting potential: The resting
Peripheral Nerve II. Amelyn Ramos Rafael, MD. Anatomical considerations
 Peripheral Nerve II Amelyn Ramos Rafael, MD Anatomical considerations 1 Physiologic properties of the nerve Irritability of the nerve A stimulus applied on the nerve causes the production of a nerve impulse,
Peripheral Nerve II Amelyn Ramos Rafael, MD Anatomical considerations 1 Physiologic properties of the nerve Irritability of the nerve A stimulus applied on the nerve causes the production of a nerve impulse,
Vertebrate Physiology 437 EXAM I NAME, Section (circle): am pm 23 September Exam is worth 100 points. You have 75 minutes.
 1 Vertebrate Physiology 437 EXAM I NAME, Section (circle): am pm 23 September 2004. Exam is worth 100 points. You have 75 minutes. True or False (write true or false ; 10 points total; 1 point each) 1.
1 Vertebrate Physiology 437 EXAM I NAME, Section (circle): am pm 23 September 2004. Exam is worth 100 points. You have 75 minutes. True or False (write true or false ; 10 points total; 1 point each) 1.
CYTOLOGY & HISTOLOGY THE STUDY OF CELLS AND TISSUES
 NAME: DATE: PARTNER: CYTOLOGY & HISTOLOGY THE STUDY OF CELLS AND TISSUES For ease of study, multicellular animals are often examined at various levels of structural organization. Starting from the most
NAME: DATE: PARTNER: CYTOLOGY & HISTOLOGY THE STUDY OF CELLS AND TISSUES For ease of study, multicellular animals are often examined at various levels of structural organization. Starting from the most
6.3.4 Action potential
 I ion C m C m dφ dt Figure 6.8: Electrical circuit model of the cell membrane. Normally, cells are net negative inside the cell which results in a non-zero resting membrane potential. The membrane potential
I ion C m C m dφ dt Figure 6.8: Electrical circuit model of the cell membrane. Normally, cells are net negative inside the cell which results in a non-zero resting membrane potential. The membrane potential
T H E J O U R N A L O F C E L L B I O L O G Y
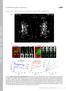 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Eisner et al., http://www.jcb.org/cgi/content/full/jcb.201312066/dc1 Figure S1. Mitochondrial continuity in adult skeletal muscle. (A)
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Eisner et al., http://www.jcb.org/cgi/content/full/jcb.201312066/dc1 Figure S1. Mitochondrial continuity in adult skeletal muscle. (A)
Introduction to Embryology. He who sees things grow from the beginning will have the finest view of them.
 He who sees things grow from the beginning will have the finest view of them. Aristotle 384 322 B.C. Introduction to Embryology This lecture will introduce you to the science of developmental biology or
He who sees things grow from the beginning will have the finest view of them. Aristotle 384 322 B.C. Introduction to Embryology This lecture will introduce you to the science of developmental biology or
A Neurosurgeon s Perspectives of Diffusion Tensor Imaging(DTI) Diffusion Tensor MRI (DTI) Background and Relevant Physics.
 A Neurosurgeon s Perspectives of Diffusion Tensor Imaging(DTI) Kalai Arasu Muthusamy, D.Phil(Oxon) Senior Lecturer & Consultant Neurosurgeon. Division of Neurosurgery. University Malaya Medical Centre.
A Neurosurgeon s Perspectives of Diffusion Tensor Imaging(DTI) Kalai Arasu Muthusamy, D.Phil(Oxon) Senior Lecturer & Consultant Neurosurgeon. Division of Neurosurgery. University Malaya Medical Centre.
Fundamentals of the Nervous System and Nervous Tissue
 Chapter 11 Part B Fundamentals of the Nervous System and Nervous Tissue Annie Leibovitz/Contact Press Images PowerPoint Lecture Slides prepared by Karen Dunbar Kareiva Ivy Tech Community College 11.4 Membrane
Chapter 11 Part B Fundamentals of the Nervous System and Nervous Tissue Annie Leibovitz/Contact Press Images PowerPoint Lecture Slides prepared by Karen Dunbar Kareiva Ivy Tech Community College 11.4 Membrane
Membrane Potentials, Action Potentials, and Synaptic Transmission. Membrane Potential
 Cl Cl - - + K + K+ K + K Cl - 2/2/15 Membrane Potentials, Action Potentials, and Synaptic Transmission Core Curriculum II Spring 2015 Membrane Potential Example 1: K +, Cl - equally permeant no charge
Cl Cl - - + K + K+ K + K Cl - 2/2/15 Membrane Potentials, Action Potentials, and Synaptic Transmission Core Curriculum II Spring 2015 Membrane Potential Example 1: K +, Cl - equally permeant no charge
BIO 210: Anatomy and Physiology Text: Fundamentals of Anatomy and Physiology 9ed. Chapter 12 NEURAL TISSUE
 NAME COURSE BIO 210: Anatomy and Physiology Text: Fundamentals of Anatomy and Physiology 9ed. Chapter 12 NEURAL TISSUE Like a telephone switchboard, the nervous system directs a countless number of incoming
NAME COURSE BIO 210: Anatomy and Physiology Text: Fundamentals of Anatomy and Physiology 9ed. Chapter 12 NEURAL TISSUE Like a telephone switchboard, the nervous system directs a countless number of incoming
Cell Death & Trophic Factors II. Steven McLoon Department of Neuroscience University of Minnesota
 Cell Death & Trophic Factors II Steven McLoon Department of Neuroscience University of Minnesota 1 Remember? Neurotrophins are cell survival factors that neurons get from their target cells! There is a
Cell Death & Trophic Factors II Steven McLoon Department of Neuroscience University of Minnesota 1 Remember? Neurotrophins are cell survival factors that neurons get from their target cells! There is a
UNUSUAL MITOCHONDRIAL CRISTAE IN THE VINEGAR EELWORM
 UNUSUAL MITOCHONDRIAL CRISTAE IN THE VINEGAR EELWORM BERT M. ZUCKERMAN, MARIAN KISIEL, and STANLEY HIMMELHOCH. From the Laboratory of Experimental Biology, University of Massachusetts, East Wareham, Massachusetts
UNUSUAL MITOCHONDRIAL CRISTAE IN THE VINEGAR EELWORM BERT M. ZUCKERMAN, MARIAN KISIEL, and STANLEY HIMMELHOCH. From the Laboratory of Experimental Biology, University of Massachusetts, East Wareham, Massachusetts
(From the Department of Anatomy and the Beaumont-May Institute of Neurology, Washington University School of Medicine, St. Louis)
 ELECTRON MICROSCOPIC OBSERVATIONS OF THE OLFACTORY MUCOSA AND OLFACTORY NERVE* BY A. J. DE LORENZO, PH.D. (From the Department of Anatomy and the Beaumont-May Institute of Neurology, Washington University
ELECTRON MICROSCOPIC OBSERVATIONS OF THE OLFACTORY MUCOSA AND OLFACTORY NERVE* BY A. J. DE LORENZO, PH.D. (From the Department of Anatomy and the Beaumont-May Institute of Neurology, Washington University
The Nervous System. Nerve Impulses. Resting Membrane Potential. Overview. Nerve Impulses. Resting Membrane Potential
 The Nervous System Overview Nerve Impulses (completed12/03/04) (completed12/03/04) How do nerve impulses start? (completed 19/03/04) (completed 19/03/04) How Fast are Nerve Impulses? Nerve Impulses Nerve
The Nervous System Overview Nerve Impulses (completed12/03/04) (completed12/03/04) How do nerve impulses start? (completed 19/03/04) (completed 19/03/04) How Fast are Nerve Impulses? Nerve Impulses Nerve
GRE Quantitative Reasoning Practice Questions
 GRE Quantitative Reasoning Practice Questions y O x 7. The figure above shows the graph of the function f in the xy-plane. What is the value of f (f( ))? A B C 0 D E Explanation Note that to find f (f(
GRE Quantitative Reasoning Practice Questions y O x 7. The figure above shows the graph of the function f in the xy-plane. What is the value of f (f( ))? A B C 0 D E Explanation Note that to find f (f(
Anatomy and Physiology 4601
 Anatomy and Physiology 4601 Description Basic concepts of human anatomy and physiology will be explored in this health (life) sciencefocused course. Using a systems approach, students will learn about
Anatomy and Physiology 4601 Description Basic concepts of human anatomy and physiology will be explored in this health (life) sciencefocused course. Using a systems approach, students will learn about
Integrated CME Project Mathematics I-III 2013
 A Correlation of -III To the North Carolina High School Mathematics Math I A Correlation of, -III, Introduction This document demonstrates how, -III meets the standards of the Math I. Correlation references
A Correlation of -III To the North Carolina High School Mathematics Math I A Correlation of, -III, Introduction This document demonstrates how, -III meets the standards of the Math I. Correlation references
Essential Question Content Skills Assessment Standards. Anatomical terms. Homeostatic mechanisms. Characteristics of Life. Requirements for life
 Honors Anatomy & Physiology 2012 Michael Harshaw, Melissa Castone Essential Question Content Skills Assessment Standards Month Introduction September- 1.5 weeks How do Anatomy and Physiology relate to
Honors Anatomy & Physiology 2012 Michael Harshaw, Melissa Castone Essential Question Content Skills Assessment Standards Month Introduction September- 1.5 weeks How do Anatomy and Physiology relate to
