JØRGEN OLESEN*, STEFAN RICHTER and GERHARD SCHOLTZ. Humboldt-Universität zu Berlin, Institut für Biologie, Vergleichende Zoologie, Berlin, Germany
|
|
|
- Scarlett Richards
- 6 years ago
- Views:
Transcription
1 Int. J. Dev. Biol. 45: (2001) Original Article The evolutionary transformation of phyllopodous to stenopodous limbs in the Branchiopoda (Crustacea) - Is there a common mechanism for early limb development in arthropods? JØRGEN OLESEN*, STEFAN RICHTER and GERHARD SCHOLTZ Humboldt-Universität zu Berlin, Institut für Biologie, Vergleichende Zoologie, Berlin, Germany ABSTRACT Arthropods and in particular crustaceans show a great diversity concerning their limb morphology. This makes the homologization of limbs and their parts and our understanding of evolutionary transformations of these limb types problematical. To address these problems we undertook a comparative study of the limb development of two representatives of branchiopod crustaceans, one with phyllopodous the other with stenopodous trunk limbs. The trunk limb ontogeny of a larger branchiopod, Cyclestheria hislopi ( Conchostraca ) and the raptorial cladoceran Leptodora kindtii (Haplopoda) has been examined by various methods such as SEM, Hoechst fluorescent stain and expression of the Distal-less gene. The early ontogeny of the trunk limbs in C. hislopi and L. kindtii is similar. In both species the limbs are formed as ventrally placed, elongate, subdivided limb buds. However, in C. hislopi, the portions of the early limb bud end up constituting the endites and the endopod of the phyllopodous filtratory limb in the adult, whereas in L. kindtii, similar limb bud portions end up constituting the actual segments in the segmented, stenopodous, and raptorial trunk limbs of the adults. Hence, the portions of the limbs corresponding to the endites of the phyllopodous trunk limbs in C. hislopi (and other larger branchiopods ) are homologous to the segments of the stenopodous trunk limbs in L. kindtii. It is most parsimonious to assume that the segmented trunk limbs in L. kindtii have developed from phyllopodous limbs with endites and not vice versa. This study has demonstrated at least one way in which segmented limbs have been derived from phyllopodous, multi-lobate limbs during evolution. Similar pathways can be assumed for the evolution of stenopodous, segmented and uniramous limbs in other crustaceans. Irrespective of the differences in the adult limb morphology, the early patterning of arthropod limbs seems to follow a similar principle. KEY WORDS: Distal-less, Leptodora, Cladocera, phylogeny, embryology, ontogeny Introduction Arthropods exhibit a great diversity in the morphology of their limbs. In addition to highly modified appendages used as sensory organs or as mouthparts, the trunk limbs which are involved in locomotion and in several instances in food transport vary to a high degree. One finds uniramous, biramous or polyramous legs, which are either of tube-like (stenopodous) or of flattened (phyllopodous) shape. In some cases there is only weak articulation within the limbs, in others there are distinct joints. Moreover, the number of limb segments varies between the arthropod taxa. This high degree of variety in limb shape can already be seen in Cambrian arthropod representatives (e.g., Gould, 1989; Walossek, 1993; Conway Morris, 1998; Budd, 1996). These morphological differences of arthropod limbs have caused numerous investigations and speculations dealing with comparative aspects of limb development and evolution. In particular, the following questions are controversially discussed: are limb segments homologous between higher arthropod taxa (Hessler and Newman, 1975; Abzhanov and Kaufman, 2000), do polyramous phyllopodous limbs share similar developmental gene expression patterns and principles of axis formation with uniramous stenopodous limbs (Panganiban et al., 1995; González-Crespo and Morata, 1996; Abbreviations used in this paper: a1, antenna 1; a2, antenna 2; ca, carapace; Dll, Distal-less; e1-e5, endite 1-5; en, endopod; ep, epipod; ex, exopod; la, labrum; mx1, maxilla 1; mx2, maxilla 2; SEM, Scanning Electron Microscopy; tl1, trunk limb 1; tl6, trunk limb 6; tl7, trunk limb 7; tl10, trunk limb 10. *Address correspondence to: Dr. JØrgen Olesen. Zoological Museum, University of Copenhagen, Universitetsparken 15, DK-2100 Copenhagen OE, Denmark. Fax: j1olesen@zmuc.ku.dk /2001/$25.00 UBC Press Printed in Spain
2 870 J. Olesen et al. Fig. 1. Light microscopy of adult trunk limbs of two large branchiopods and one predatory cladoceran to show diversity of trunk limbs within the Branchiopoda. (A) Triops sp. (preparation from the zoological collection of the Humboldt University). (B) Cyclestheria hislopi. (C) Leptodora kindtii (preparation from the zoological collection of the Humboldt University), lateral view of left side trunk limbs, the distal-most segment of trunk limbs 1 (left) is not shown. Abbreviations: e1-e5-endite 1-5; en, endopod; ep, epipod; ex, exopod; tl1-4, trunk limbs 1-4; arabic numbers 1-5 refer to embryonic limb portions in L. kindtii. Williams, 1998, 1999; Nulsen and Nagy, 1999; Nagy and Williams, 2001), are phyllopodous limbs plesiomorphic within arthropods or subgroups like Crustacea or did phyllopodous limbs evolve several times independently from stenopodous appendages (Lauterbach, 1978; Martin and Christiansen, 1995; Spears and Abele, 1999; Ax, 1999), can insect wings be derived from epipodites (Kukalová-Peck, 1992; Averof and Cohen, 1997)? Among the extant arthropod groups, the Crustacea shows the greatest diversity in trunk limb morphology and this high degree of diversity is also seen in each of the major crustacean taxa such as malacostracans or branchiopods. Examples of the latter are treated in the present paper. The trunk limbs of the larger branchiopods (Anostraca, Notostraca and Conchostraca ) are relatively similar. All have a high number of serially similar, more or less flattened trunk limbs (phyllopods), composed of a large corm (the basis, after Walossek, 1993), normally with 6 setose lobes (at least in adults) of variable shape along the inner margin, the proximal one normally with a differing morphology and directed into the food groove. Five of these lobes are usually considered as endites. However, the sixth, distalmost lobe, which in some cases looks like an endite, has by comparison to certain Orsten fossils been suggested to be homologous to the endopod of other Crustacea (Walossek, 1993). At the outer margin of the trunk limbs they normally have a setose exopod of variable shape and a non-setose, sac-like epipod (gill). In contrast to this, a much greater variation of trunk limb morphology is seen within the Cladocera where only 4 to 6 trunk limbs are present (Fryer, 1987; Dumont and Silva-Briano, 1998). In this paper we focus on a raptorial cladoceran - Leptodora kindtii representing the monotypic Haplopoda - which is, due to its lifestyle as a predator, among the most deviating branchiopods. This cladoceran has uniramous, stenopodous, segmented trunk limbs, and any comparison between these and the flattened, multi-lobate limb type described above for the larger branchiopods is difficult. We examined embryonic stages of Leptodora kindtii (Haplopoda) and one larger branchiopod, Cyclestheria hislopi, the presumed sister group to the Cladocera, by different morphological methods. In particular, we used a polyclonal antibody against the Distal-less (Dll) gene product (Panganiban et al., 1995). The Dll gene has been shown to play an important role for arthropod limb development and its expression patterns have been used to infer on homology of limbs and their parts respectively (e.g., Panganiban et al., 1995; Niwa et al., 1997; Popadic et al., 1998; Scholtz et al., 1998; Williams, 1998; Thomas and Telford, 1999; Abzhanov and Kaufmann, 2000; Browne and Patel, 2000; Scholtz, 2001). Examination of embryos of Cyclestheria hislopi and various cladocerans has earlier proved useful when trying to establish homologies between taxa where the adults are difficult to compare (Olesen, 1998, 1999). We address the following questions: What are the homologies between the segmented trunk limbs of the raptorial cladocerans and the typical multi-lobate, phyllopodous trunk limb of the larger branchiopods? Based on this homologisation - what evolutionary scenario for the development of the segmented limbs can be outlined? Can the evolution of the branchiopod limbs serve as a model for arthropod limb transformation during development and evolution? Results The early development of the trunk limbs in Leptodora kindtii and Cyclestheria hislopi is quite similar. In both taxa they are formed as two ventral rows of elongate, subdivided limb buds, six pairs of limb buds in L. kindtii and in C. hislopi. In C. hislopi the long limb buds first form a lateral tip, then they divide into six ventral portions (starting from proximal), attain a bifurcate tip (which appears before all ventral portions are formed), develop a primordial epipod which originates dorsal to the bifurcate tip, and then shift the orientation of the entire limb to an upright, vertical position (Figs. 2,3). L. kindtii has a similar tip formation and subdivision of the early, elongate limb bud as C. hislopi, but the number of separate portions of the limb bud is reduced to five, as revealed by SEM (Fig. 4), by Hoechst nuclear stain (Fig. 5) and by expression of the Distal-less (Dll) gene (Fig. 6). Different from the limb development in C. hislopi is that L. kindtii develops no bifurcate tip and no epipod. The early pattern of Dll expression is quite similar between C. hislopi and L. kindtii. The Dll expression in each limb is first seen in a lateral area, which later forms the tip of the appendage (Figs. 3,6). The second spot of Dll expression is found in the most proximal area of the forming limb (Figs. 3,6). In more advanced stages of both species a series of Dll positive
3 Evolution of Crustacean Limbs 871 Fig. 2. Embryonic stages of Cyclestheria hislopi ( Conchostraca ) (SEM). (A) Lateral view of stage IV. (B) Ventral view of stage VI. (C) Left side trunk limbs, ventral, stage VI. Fifth endite becomes separated from endopod between trunk limb 6 and 7 (white arrow). (D) Ventral view of trunk limbs of stage VII. (E) Lateral view of posterior trunk limbs of stage VII. Abbreviations: a1, antenna 1; a2, antenna 2; ca, carapace; la, labrum; mx1, maxilla 1; mx2, maxilla 2; tl1, trunk limb 1; e1-e5, endite 1-5; en, endopod; ex, exopod; tl6, trunk limb 6; tl7, trunk limb 7; tl10, trunk limb 10; ep, epipod. second segments in the intermediate to late parts of their ontogeny (Figs. 4,5). The limb portions of the early limb bud of C. hislopi are simple to account for in terms of their further development. The five proximal ventral portions end up constituting endites 1-5 in the adult, while the distalmost sixth portion develops into the small unsegmented endopod (Figs. 2,3). The dorsal part of the bifurcated tip of the limb buds will constitute the exopod in the adult, while the dorso-laterally placed portion will constitute the epipod (see Figs. 1,2). In L. kindtii the situation is more complicated and difficult to follow during the development, despite there are fewer limb portions to account for. Trunk limb 1-5 in the adults of L. kindtii have four segments each, one very long proximal segment followed by three smaller segments (see Sars, 1993). The elongate limb buds of early embryos consist of five portions as revealed by the expression of Dll (mentioned above, see Figs. 4-6). It is clear that the three distal (or lateral) portions of the early limb bud will end up constituting the three distal short segments present in trunk limb 1-5 in the adult, since the fate of these limb portions can be followed unambiguously during ontogeny (see Fig. 7). The fate of the two proximal portions seen in the early embryos, revealed by Dll expression, is more difficult to follow and, furthermore, the development of trunk limb 1 is slightly different from that of trunk limb 2-5. Common to trunk limbs 1-5 is that the large proximal segment in the adult arise from a fusion of two rudimentary embryonic portions indicated by the Dll expression (marked by Arabic numbers 1 and 2 on Figs. 4-6). regions appears at the upper margin of the limb anlage. In C. hislopi these expression areas correspond with the forming inner lobes. In L. kindtii the Dll positive regions are associated with the prospective limb segments (Fig. 6 C,D). It appears that Dll expression fades in the proximal limb segments of more ad- vanced embryos of L. kindtii. In contrast Dll expression is maintained in the lobes of the C. hislopi limbs throughout the observed development (Fig. 3 C,D). No Dll expression has been found in the epipod. The way in which the limbs attain their vertical orientation is also different in L. kindtii and appears more complicated than in C. hislopi. Whereas C. hislopi effectively bends the whole limb (Fig. 2), the trunk limbs of L. kindtii have a characteristic bend in the region of the first and However, even more proximal, slightly bend under the more distal part of the limb, is a limb portion which has not been Dll positive, but which nevertheless becomes included in the long proximal part of the adult trunk limb (Figs. 4 B,C, 5B, marked by upper arrow). This limb part is best visible in the anterior limb pairs. We interpret this limb portion as a part of embryonic limb Fig. 3. Embryonic stages of Cyclestheria hislopi ( Conchostraca ). (A) Ventral view of Cyclestheria hislopi, intermediate stage (stage VI) (Hoechst nuclear stain). (B) Right side trunk limbs of late stage of Cyclestheria hislopi (stage VII) (Hoechst nuclear stain). (C) Cyclestheria hislopi, ventral view, stage VII (Distal-less immunostaining). (D) Cyclestheria hislopi, ventral view, posterior part (Distal-less immunostaining). Staging after Olesen (1999). Abbreviations: a1, antenna 1; a2, antennae 2; en, endopod; ex, exopod; fu, furcae; la, labrum; md, mandible; mx1, maxilla 1; mx2, maxilla 2; tl1, trunk limb 1; arabic numbers 1-5 indicate endites 1-5.
4 872 J. Olesen et al. Fig. 4. Embryonic stages of Leptodora kindtii (SEM). (A) Ventral view of an intermediate stage 2. (B) Left side trunk limbs of an intermediate stage 2, upper arrow points to the folded part of trunk limb 1, lower arrow points to the so-called maxillary process (see text for details). (C) Left side trunk limbs of stage 3, arrow points to basal, folded part of first trunk limb. (D) Left side of stage 4 (juvenile/sub-adult). Abbreviations: a1, antenna 1; a2, antenna 2; md, mandible; tl1, trunk limb 1; la, labrum; ca, carapace. Arabic numbers 1-5 mark limb portions. portion 1. In the adults, trunk limb 1 differs from the more posterior trunk limbs in the presence of a small, setose (two setae) socalled maxillary process situated at the broad mid region between the limb, close to bases of the limbs, but not connected to these (e.g. Sebestyén, 1931). In early embryos, the expression of Dll in the proximal segment is located more closely to the midline in the first trunk limbs than in the more posterior ones (Fig. 6D). This median expression exactly corresponds to the anlagen of the maxillary process (Fig. 5 B,C, lower arrow). We interpret this as indicating that the endite of the proximal portion of the limb migrates towards the midline of the animal, and ends up constituting the maxillary process. Hence, the maxillary process has nothing to do with the maxillae. The endites of the proximal parts of the subsequent trunk limbs remain associated with the corresponding limb portion. Discussion Limb homologies in Cyclestheria hislopi and Leptodora kindtii Since Cyclestheria hislopi is most likely the sister group to a monophyletic Cladocera (see Martin and Cash-Clark, 1995; Olesen et al., 1997; Olesen, 1999; Crease and Taylor, 1998; Ax, 1999; Taylor et al., 1999; Spears and Abele, 2000; Braband et al., 2000), there are good reasons for comparing the limb development and morphology of L. kindtii and C. hislopi. As mentioned above, the pattern of subdivision of early limb buds in C. hislopi and L. kindtii is very similar. In contrast, the limbs continue their development in two completely different directions (summarised in Fig. 7). In C. hislopi the five proximal portions end up constituting the five endites present in the adult. The sixth portion ends up constituting the distal-most lobe on the inner corm of the limb, the endopod. Most other larger branchiopods show a similar pattern of limb development as has been shown for the notostracan Triops longicaudatus (Williams and Müller, 1996) and the anostracans Branchipus stagnalis and Branchipus schaefferi (Claus, 1873; Schlögl, 1996). Since this type of development is not found outside the Branchiopoda, it has been suggested as being a synapomorphy for the Branchiopoda (Olesen, 1999). In L. kindtii, by contrast, the various portions of the early limb bud ends up constituting the actual segments in the stenopodous trunk limbs in the adult (see above for more details). Based on these ontogenetic findings, we propose the following homologies between L. kindtii and C. hislopi (and thereby other larger branchiopods). The long basal segment in L. kindtii is homologous to endites 1 and 2 and the corresponding parts of the limb corm in C. hislopi. This picture is most clear for trunk limbs 2-5. Associated to the first pair of trunk limbs in L. kindtii is a pair of so-called maxillary processes, placed on the ventral trunk area, disconnected from the actual limb. These are probably homologous to the proximal endites of the first trunk limbs in C. hislopi. The three smaller distal segments of the trunk limbs in L. kindtii correspond to endites 3-5 (including the corresponding parts of the limb corms) and the small, unsegmented endopod in C. hislopi. In C. hislopi, the ventral portions (future 5 endites, endopod and exopod) of the elongate limb bud are formed gradually from proximal to distal with the exopod and epipod formation sometime in between (Fig. 2C, see Olesen, 1999 for more details). This is reflected by the Dll expression, which is first seen in the distal tip of the limb anlagen, followed by the proximal endite, and subsequently more distal endites. The exopod and endopod originate from a bifurcation of the distal portion of the limb anlage. Except for some differences in the timing of the subdivision and the Dll expression a similar pattern of limb development has been described in the notostracan Triops longicaudatus (Williams, 1998). In L. kindtii only five limb portions are present and the two proximal parts fuse to a single segment. Therefore, the distal most limb segment corresponds not only to the fifth endite but also to the endopod of
5 Evolution of Crustacean Limbs 873 C. hislopi. It is furthermore reasonable to assume that the absence of the exopod in the limbs of L.kindtii is the result of the suppressed bifurcation of the early distal limb portion. Evolution of stenopodous trunk limbs in Leptodora kindtii The homologies between the trunk limbs in Cyclestheria hislopi and Leptodora kindtii are established in some detail above and, broadly speaking, the endites including the corresponding part of the basis of C. hislopi and other larger branchiopods are homologous to the true segments in Leptodora. But which is derived from which? The phylogenetic position of L. kindtii, as a member of the Cladocera within the Branchiopoda irrespective of its exact sister group relationship (Martin and Cash-Clark, 1995; Olesen et al., 1997; Olesen, 1998; Crease and Taylor, 1998; Schwenk et al., 1998; Taylor et al., 1999; Negrea et al., 1999; Spears and Abele, 2000; Richter et al., 2001), indicates that the segmented limbs of this taxon have been derived from the typical larger branchiopod limb and not vice versa. If the segmented limbs of L. kindtii were viewed as homologous to the segmented limbs so commonly seen in other crustaceans, and therefore retained through the whole evolution of the Branchiopoda, it would imply convergent evolution of the typical flattened trunk limb a number of times within the larger Branchiopoda and the Cladocera, which is therefore unrealistic. Hence, the similarity between the segmented limbs of L. kindtii and the similarly segmented limbs of so many other Crustacea is considered convergent. This allows for a plausible evolutionary scenario where it is possible to account convincingly for functional intermediate stages between a filter feeding branchiopod, as represented by most larger branchiopods, with their flattened, endite bearing limbs and the raptorial feeding L. kindtii, with its uniramous, stenopodous, segmented limbs. During the evolution of the segmented limb of L. kindtii from flattened, endite bearing limb, as those of the larger Branchiopoda, one must imagine the following morphological changes. 1) Transformation of the clefts between the endites gradually developing into articulations. 2) Development of tubular articles of the limb and a more rigid cuticle. 3) Loss of exopod (possibly by suppression of the bifurcation of the tip of the limb during development) and epipod (not necessarily related to loss of exopod). 4) Fusion of regions corresponding to endite 1 and 2 to one long tubular segment. 5) Alteration of endite formation (possibly by ontogenetic suppression of subdivision) so that only five limb portions are formed. The exact order in which these evolutionary changes took place, or to which extent the various steps could have been linked to each other, is not known for certain, but some suggestions can be made. One obvious possible correlation is that the appearance Fig. 5. Embryonic stages of Leptodora kindtii (Hoechst nuclear stain). (A) Ventral view of stage 2, arrow points to folded part of trunk limb 1. (B) Close-up of A, upper arrow points to folded part of trunk limb 1, lower arrow points to the so-called maxillary process. (C) Late stage 2, arrow points to so-called maxillary process. Abbreviations: a1, antenna 1; a2, antenna 2; md, mandible; tl1, trunk limb 1; tl6, trunk limb 6. Arabic numbers 1-5 mark limb portions. of articulations (step 1) can be seen as linked to the appearance of a more rigid cuticle (step 2). Two features are characteristic for the limbs of L. kindtii as compared to those of C. hislopi and other larger branchiopods, the fact that they are stenopodous and uniramous. While we cannot exclude that the appearance of these two characteristics have been evolutionary linked, we have, on the other hand, no reason to believe so. Implications for limb evolution in the Crustacea Already Borradaile (1926) speculated, based on comparative morphology, that the primitive crustacean limb was unsegmented but had endites from which true segments had developed. Also Fryer (1992) put forward the view that arthropod appendages had originated as unsegmented structures. Now it is clearly shown in the present paper that truly segmented uniramous limbs have evolved from phyllopodous, multi-lobate limbs at least once during crustacean evolution (within the Recent branchiopods). It is tempting to suggest that a similar scenario has occurred in the evolution of segmented limbs in other crustaceans and arthropods. Trunk limbs with a row of identical lobate endites - very similar and most likely homologous to those of Recent branchiopods - are a very old phenomenon, present already in the Upper Cambrian in certain crustacean Orsten -fossils, like Rehbachiella kinnekullensis Müller, 1983, Walossekia quinquespinosa Müller, 1983 and Dala peilertae Müller, 1983 (see Walossek, 1993, 1999). Accordingly, there is room for interpreting phyllopodous trunk limbs with such morphology as
6 874 J. Olesen et al. Fig. 6. Embryonic stages of Leptodora kindtii (Distal-less immunostaining). (A) Early stage 1. (B) Stage slightly later than A. (C) Late stage 1/early stage 2. (D) Close-up of C. Abbreviations: a1, antenna 1; a2, antenna 2; fu, furcae; md, mandible; tl1, trunk limb 1; tl6, trunk limb 6. Arabic numbers 1-5 mark limb portions. ancestral also for other Recent Crustacean groups. We therefore suggest the transformation of limb endites into true segments, as a working hypothesis for the evolution of other Crustacea with segmented limbs. However, studies focussing on small but well-identified aspects of limb evolution, with high attention to segmentation homologies, much in the spirit of Boxshall and Huys (1992), are pivotal if we wish to understand such questions and the evolution of crustacean and arthropod appendages in general. The best candidate for a similar scenario of limb transformation from phyllopodous to stenopodous would be the Malacostraca. Foliaceous trunk limbs are found in the malacostracan subgroup Leptostracana which is most likely the sister group to the remaining malacostracans with stenopodous limbs, the Eumalacostraca (Richter and Scholtz, 2001). Accordingly, it is reasonable to assume that the stenopodous segmented limbs of the Eumalacostraca have evolved from foliaceous limbs similar to those of Recent leptostracans. However, whether foliaceous limbs are really plesiomorphic for the Malacostraca depends on the homology between phyllopodous limbs of branchiopods and leptostracans which has been suggested, for instance, by Lauterbach (1975), Schram (1986), and Ax (1999). On the other hand, this homology has been rejected by other authors (Dahl, 1987; Walossek, 1993; Martin and Christiansen, 1995; Spears and Abele, 1999). As a last remark, we find it interesting to note that Walossek s (1993) hypothesis about the origin of the crustacean coxa from an (proximal) endite receives some support from our results. The described transformation of endites into true limb segments within branchiopods are, at the general level, essentially the same thing as the transformation events (from the proximal endite to a coxa (limb segment)) hypothesised by Walossek (1993) The P/D axis is the same in uniramous and polyramous arthropod limbs Our data clearly show that similar limb anlagen in terms of morphology and expression of Dll can result in highly different multi-lobed, phyllopodous and stenopodous, uniramous limbs. This provides a solution to the problem of the comparability of the axis formation in uniramous and multilobed limbs raised and discussed by Williams and Nagy (1996), Williams (1999), Nulsen and Nagy (1999), and Nagy and Williams (2001). Williams (1999) discussed whether the phyllopodous limbs possess one P/D axis as the uniramous Drosophila melanogaster legs or whether each lobe of the branchiopod leg is equivalent to a Drosophila limb in terms of axis formation. Our results now directly support the conclusions by Williams and Nagy (1996), that the latter hypothesis is unlikely and polyramous limbs are evolutionarily transformed into uniramous limbs (and probably vice versa) without changes of the early axis determination as shown by the Dll expression. This is indicated by the fact that in all cases the earliest Dll expression is found in the prospective distal tip of the limb anlagen irrespective of their shape (Panganiban et al., 1995; Niwa et al., 1997; Williams, 1998; Scholtz et al., 1998; Popadic et al., 1998; Mittmann and Scholtz, 2001). This is in good agreement with the function of the Dll gene in the insect Drosophila melanogaster and in the spider Cupiennius salei where Dll has been found to be responsible for differentiation of the P/D axis and the formation of the distal limb elements (Cohen and Jürgens, 1989; Schoppmeier and Damen, 2001). Interestingly enough, the second (in time) area of Dll expression is found in the most proximal part of the limb anlage - if an endite is at least transitorily formed. This is again true for the anlagen of phyllopodous and stenopodous limbs and can be found even in hexapod mouthparts (Niwa et al., 1997; Scholtz et al., 1998). This sequence of limb differentiation starting with the distalmost elements followed by the most proximal ones is also found in the morphological differentiation sequence (Fryer, 1983; Dorn and Hoffmann, 1983; Olesen, 1999) and in the sequence of the expression of genes involved in the subdivision of the limbs such as annulin (Bastiani et al., 1992) and DSS-8 (Norbeck and Denburg, 1991). All these data suggest an intercalary limb differentiation as has been proposed for Drosophila (Goto and Hayashi, 1999) as a general feature for arthropod limb patterning during development. The early establishment of a main P/D axis together with an intercalary differentiation and a similar set of genes (Abzhanov and Kaufman, 2000) allows or even supports the manifold evolution of arthropod limb morphology and function which makes this group so successful.
7 Evolution of Crustacean Limbs 875 Fig. 7. Trunk limb homologies between Cyclestheria hislopi (left side) and Leptodora kindtii (right side). Upper part of figure shows embryos, lower part shows adults. The expression domains of Dll are indicated by brown dots. The six ventral portions of the embryonic limb bud in Cyclestheria develop into the median endites and the endopod in the limb of the adult. In Leptodora, the five ventral portions of the early limb bud develop into true segments in the adult. This suggests homology between the median trunk limb endites of Cyclestheria (and thereby other larger branchiopods ) and the segments of the raptorial trunk limbs of Leptodora. The coloured lines indicate presumed homologous limb parts between Cyclestheria and Leptodora. The dotted line with a? indicates the inclusion of the reduced exopod in the tip of the limb of Leptodora caused by the suggested suppression of the bifurcation of the early distal tip of the limb anlage. See text for details. Materials and Methods The material of Cyclestheria hislopi (Baird, 1859) was collected in Colombia in 1994 (see Olesen et al., 1997) and in Northern Territory, Australia in Leptodora kindtii (Focke, 1844) was collected in 1998 and 1999 in the Tegeler See, Berlin. The material for scanning electron microscopy (SEM) was fixed in glutaraldehyde, formaldehyde or Bouins s Fluid, dehydrated and critical point dried following standard procedures. The other material was fixed in PEM-FA (0.1 PIPES (ph 6.95), 2.0 mm EGTA, 0.1 mm MgSO 4, 3.7% formaldehyde) and stored in methanol. The immunostaining and fluorescent staining followed Scholtz et al. (1998). The staging of the embryos of C. hislopi followed the guidelines provided by Olesen (1999). The embryos of L. kindtii were grouped into 4 appropriate stage categories based on their degree of development. Acknowledgements We thank Jean Deutsch for helpful comments on the manuscript. The Distal-less antibody was generously provided by Grace Panganiban. The work was supported by a postdoctoral grant to JO from the Danish Natural Science Research Council. Hendrik Müller helped with preparing and photographing embryos of C. hislopi. We are especially grateful to Werner Richter who kindly assisted us in the collection of Leptodora kindtii in the Tegeler See. Collecting of C. hislopi was supported by a Visiting Fellowship of the Australian Museum to SR. SR also acknowledges the generous support of the Environmental Research Institute of Supervising Scientists and of Dr. Chris Humphrey during collecting of C. hislopi. References ABZHANOV, A. and KAUFMAN, T.C. (2000). Homologs of Drosophila appendage genes in the patterning of arthropod limbs. Dev. Biol. 227: AVEROF, M. and COHEN, S.M. (1997). Evolutionary origin of insect wings from ancestral gills. Nature 385: AX, P. (1999). Das System der Metazoa II. Gustav Fischer Verlag, Stuttgart. BASTIANI, M.J., DE COUET, H.G., QUINN, J.M.A., KARLSTROM, R.O., KORTLA, K., GOODMAN, C.S. and BALL, E.E. (1992). Position specific expression of the annulin protein during grasshopper embryogenesis. Dev. Biol. 154: BORRADAILE, L.A. (1926). Notes upon Crustacean limbs. Ann. Mag. Nat. Hist. Ser (98), BOXSHALL, G.A. and HUYS, R. (1992). A homage to homology: Patterns of copepod evolution. Acta Zool. 73(5): BRABAND, A., RICHTER, S., HIESEL, R. and SCHOLTZ, G. (2000). Phylogeny of the Branchiopoda (Crustacea) inferred by DNA data. Zoology 103 Suppl.III: 99. BROWNE, W.E. and PATEL, N.H. (2000). Molecular genetics of crustacean feeding appendage development and diversification. Sem. Cell. Dev. Biol. 11: BUDD, G. (1996). The morphology of Opabinia regalis and the reconstruction of the arthropod stem-group. Lethaia 29: CLAUS, C. (1873). Zur Kenntnis des Baues und der Entwicklung von Branchipus stagnalis und Apus cancriformis. Abhandlungen der Königlichen Gesellschaft der Wissenschaften in Göttingen 18: , pls COHEN, S.M. and JÜRGENS, G. (1989). Proximal-distal pattern formation in Drosophila: cell autonomous requirement for Distal-less gene activity in limb development. EMBO J. 8: CONWAY MORRIS, S. (1998). The Crucible of Creation. Oxford University Press, Oxford. CREASE, T.J. and. TAYLOR, D.J. (1998). The origin and evolution of variableregion helices in V4 and V7 of the small-subunit ribosomal RNA of branchiopod crustaceans. Mol. Biol. Evol. 15: DAHL, E. (1987). Malacostraca maltreated the case of the Phyllocarida. J. Crust. Biol. 7: DORN, A. and HOFFMANN, P. (1983). Segmentation and differentiation of appendages during embryogenesis of the milkweed bug, Oncopeltus fasciatus: a scanning electron microscopical study. Zool. Jb. Anat. 109: DUMONT, H.J. and SILVA-BRIANO, M. (1998). A reclassification of the anomopod families Macrothricidae and Chydoridae, with a creation of a new suborder, the Rapopoda (Crustacea: Branchiopoda). Hydrobiologia 384: FRYER, G. (1983). Functional ontogenetic changes in Branchinecta ferox (Milne- Edwards) (Crustacea: Anostraca). Phil. Trans. R. Soc. Lond. B 303: FRYER, G. (1987). A new classification of the branchiopod Crustacea. Zool. J. Linn. Soc. 91: FRYER, G. (1992). The origin of the Crustacea. Acta Zool. 73(5): GONZÁLEZ-CRESPO, S. and MORATA, G. (1996). Genetic evidence for the subdivision of the arthropod limb into coxopodite and telopodite. Development 122: GOTO, S. and HAYASHI, S. (1999). Proximal to distal cell communication in the Drosophila leg provides a basis for an intercalary mechanism of limb patterning. Development 126: GOULD, S.J. (1989). Wonderful Life. The Burgess shale and the nature of history. Norton, New York. HESSLER, R.R. and NEWMAN, W.A. (1975). A trilobitomorph origin for the Crustacea. Fossils and Strata 4:
8 876 J. Olesen et al. KUKALOVÁ-PECK, J. (1992). The Uniramia do not exist: the ground plan of the Pterygota as revealed by Permian Diaphanopterodea from Russia (Insecta: Paleodictyopteridea). Can. J. Zool. 70: LAUTERBACH, K.-E. (1975). Über die Herkunft der Malacostraca (Crustacea). Zool. Anz. 194: LAUTERBACH, K.-E. (1978). Gedanken zur Evolution der Euarthropoden-Extremität. Zool. Jb. Anat. 99: MARTIN, J. W and CASH-CLARK, C. (1995). The external morphology of the onychopod «cladoceran» genus Bythotrephes (Crustacea, Branchiopoda, Onychopoda, Cercopagididae), with notes on the morphology and phylogeny of the order Onychopoda. Zoologica Scripta 24: MARTIN, J.W. and CHRISTIANSEN, J.C. (1995). A morphological comparison of the phyllopodous thoracic limbs of a leptostracan (Nebalia sp.) and a spinicaudate conchostracan (Leptestheria sp.), with comments on the use of Phyllopoda as a taxonomic category. Can. J. Zool. 73: MITTMANN, B. and SCHOLTZ, G. (2001). Distal-less expression in embryos of Limulus polyphemus (Chelicerata, Xiphosura) and Lepisma saccharina (Insecta, Zygentoma) suggests a role for the development of mechanoreceptors, chemoreceptors, and the CNS. Dev. Genes Evol. 211: NAGY, L.M. and WILLIAMS, T.A. (2001). Comparative limb development as a tool for understanding the evolutionary diversification of limbs in arthropods: challenging the modularity paradigm. In The Character Concept in Evolutionary Biology (Ed. G.P. Wagner). Academic Press, New York, pp NEGREA, S., BOTNARIUC, N. and DUMONT, H. (1999). Phylogeny, evolution and classification of the Branchiopoda (Crustacea). Hydrobiologia 412: NIWA, N., SAITOH, M., OHUCHI, H., YOSHIOKA, H., NOJI, S. (1997). Correlation between Distal-less expression patterns and structures of appendages in development of the two-spotted cricket, Gryllus bimaculatus. Zool. Sci. 14: NORBECK, B.A. and DENBURG, J.L. (1991). Pattern formation during insect leg segmentation: studies with a prepattern of a cell surface antigen. Roux s Arch. Dev. Biol. 199: NULSEN, C. and NAGY, L.M. (1999). The role of wingless in the development of multibranched crustacean limbs. Dev. Genes Evol. 209: OLESEN, J. (1998). A phylogenetic analysis of the Conchostraca and Cladocera (Crustacea, Branchiopoda, Diplostraca). Zool. J. Linnean Soc. 122: OLESEN, J. (1999). Larval and post-larval development of the branchiopod clam shrimp Cyclestheria hislopi (Baird, 1859) Crustacea, Branchiopoda, Conchostraca, Spinicaudata). Acta Zoologica 80: OLESEN, J, MARTIN, J.W. and ROESSLER, E.W. (1997). External morphology of the male of Cyclestheria hislopi (Baird, 1859) Crustacea, Branchiopoda, Spinicaudata), with comparison of male claspers among the Conchostraca and Cladocera and its bearing on phylogeny of the bivalved Branchiopoda. Zoologica Scripta 25: PANGANIBAN, G., SEBRING, A., NAGY, L. and CARROLL, S. (1995). The development of crustacean limbs and the evolution of arthropods. Science 270: POPADIC, A., PANGANIBAN, G., RUSCH, D., SHEAR, W.A. and KAUFMAN, T.C. (1998). Molecular evidence for the gnathobasic derivation of arthropod mandibles and for the appendicular origin of the labrum and other structures. Dev. Genes Evol. 208: RICHTER, S., BRABAND, A., ALADIN, N. and SCHOLTZ, G. (2001). The phylogenetic relationships of predatory water fleas (Cladocera: Onychopoda, Haplopoda) inferred from 12S rdna. Mol. Phylogenet. Evol. 19: RICHTER, S. and SCHOLTZ, G. (2001). Phylogenetic analysis of the Malacostraca (Crustacea). J. Zool. Syst. Evol. Res. 39: SARS G. O. (1993). On the freshwater crustaceans occurring in the vicinity of Christania. University Bergen Press (translation of an 1861 handwritten manuscript, University of Oslo Library). SCHLÖGL, T. (1996). Die postembryonale Entwicklung der Männchen des Feenkrebses Branchipus schaefferi. Stapfia 42: SCHOLTZ, G. (2001). Evolution of developmental patterns in arthropods the analysis of gene expression and its bearing on morphology and phylogenetics. Zoology 103: SCHOLTZ, G., MITTMANN, B. and GERBERDING, M. (1998). The pattern of Distal-less expression in the mouthparts of crustaceans, myriapods and insects: new evidence for a gnathobasic mandible and the common origin of the Mandibulata. Int. J. Dev. Biol. 42: SCHOPPMEIER, M. and DAMEN, W.G.M. (2001). Double-stranded RNA interference in the spider Cupiennius salei: the role of Distal-less is evolutionarily conserved in arthropod appendage formation. Dev. Genes Evol. 211: SCHRAM F.R. (1986). Crustacea. Oxford University Press, Oxford. SCHWENK, K., SAND, A., BOERSMA, M., BREHM, M., MADER, E., OFFERHAUS, D. and SPAAK, P. (1998). Genetic markers, genealogies and biogeographic patterns in the Cladocera. Aquatic Ecology 32: SEBESTYÉN, O. (1931). Contribution to the biology and morphology of Leptodora kindtii (Focke) (Crustacea, Cladocera). Magy. Biol. Kutatointez. Munkai 4: SPEARS, T. and ABELE, L.G. (1999). Phylogenetic relationships of crustaceans with foliaceous limbs: an 18S rdna study of Branchiopoda, Cephalocarida, and Phyllocarida. J. Crust. Biol. 19: SPEARS, T. and ABELE, L.G. (2000). Branchiopod monophyly and interordinal phylogeny inferred from 18S ribosomal DNA. J. Crust. Biol. 20: TAYLOR, D. J., CREASE, T.J. and BROWN, W.M. (1999). Phylogenetic evidence for a single long-lived clade of crustacean cyclic parthenogens and its implications for the evolution of sex. Proc. R. Soc. London. B 266: THOMAS, R.H. and TELFORD, M.J. (1999). Appendage development in embryos of the oribatid mite Archegozetes longisetus (Acari, Oribatei, Trhypochthoniidae) Acta Zool. 80: WALOSSEK, D. (1993). The Upper Cambrian Rehbachiella and the phylogeny of Branchiopoda and Crustacea. Fossils and Strata 32: WALOSSEK, D. (1999). On the Cambrian diversity of Crustacea. In Crustaceans and the Biodiversity Crisis. Proceedings of the Fourth International Crustacean Congress, Amsterdam, the Netherlands, July 20-24, 1998, vol. 1. (Eds. Schram, F.R. and von Vaupel Klein, J.C.). Brill, Leiden, pp WILLIAMS, T.A. (1998). Distalless expression in crustaceans and the patterning of branched limbs. Dev. Genes Evol. 207: WILLIAMS, T.A. (1999). Morphogenesis and homology in arthropod limbs. Amer. Zool. 39: WILLIAMS, T.A. and MÜLLER, G.B. (1996). Limb development in a primitive crustacean, Triops longicaudatus: subdivision of the early limb bud gives rise to multibranched limbs. Dev. Genes Evol. 206: WILLIAMS, T.A. and NAGY, L.M. (1996). Comparative limb development in insects and crustaceans. Sem. Cell Dev. Biol. 7: Received: June 2001 Reviewed by Referees: August 2001 Modified by Authors and Accepted for Publication: October 2001
Jùrgen Olesen. Acta Zoologica (Stockholm) 80: 163±184 (April 1999)
 Acta Zoologica (Stockholm) 80: 163±184 (April 1999) Larval and post-larval development of the branchiopod clam shrimp Cyclestheria hislopi (Baird, 1859) (Crustacea, Branchiopoda, Conchostraca, Spinicaudata)
Acta Zoologica (Stockholm) 80: 163±184 (April 1999) Larval and post-larval development of the branchiopod clam shrimp Cyclestheria hislopi (Baird, 1859) (Crustacea, Branchiopoda, Conchostraca, Spinicaudata)
On the Ontogeny of Leptodora kindtii (Crustacea, Branchiopoda, Cladocera), with Notes on the Phylogeny of the Cladocera
 JOURNAL OF MORPHOLOGY 256:235 259 (2003) On the Ontogeny of Leptodora kindtii (Crustacea, Branchiopoda, Cladocera), with Notes on the Phylogeny of the Cladocera Jørgen Olesen, 1,2 * Stefan Richter, 1 and
JOURNAL OF MORPHOLOGY 256:235 259 (2003) On the Ontogeny of Leptodora kindtii (Crustacea, Branchiopoda, Cladocera), with Notes on the Phylogeny of the Cladocera Jørgen Olesen, 1,2 * Stefan Richter, 1 and
Phylogeny of Branchiopoda (Crustacea) Character Evolution and Contribution of Uniquely Preserved Fossils
 Arthropod Systematics & Phylogeny 3 67 (1) 3 39 Museum für Tierkunde Dresden, eissn 1864-8312, 17.6.2009 Phylogeny of Branchiopoda (Crustacea) Character Evolution and Contribution of Uniquely Preserved
Arthropod Systematics & Phylogeny 3 67 (1) 3 39 Museum für Tierkunde Dresden, eissn 1864-8312, 17.6.2009 Phylogeny of Branchiopoda (Crustacea) Character Evolution and Contribution of Uniquely Preserved
Diverse Adaptations of an Ancestral Gill: A Common Evolutionary Origin for Wings, Breathing Organs, and Spinnerets
 Current Biology, Vol. 12, 1711 1716, October 1, 2002, 2002 Elsevier Science Ltd. All rights reserved. PII S0960-9822(02)01126-0 Diverse Adaptations of an Ancestral Gill: A Common Evolutionary Origin for
Current Biology, Vol. 12, 1711 1716, October 1, 2002, 2002 Elsevier Science Ltd. All rights reserved. PII S0960-9822(02)01126-0 Diverse Adaptations of an Ancestral Gill: A Common Evolutionary Origin for
AN UPDATED PHYLOGENY OF THE CONCHOSTRACA CLADOCERA CLADE (BRANCHIOPODA, DIPLOSTRACA)
 AN UPDATED PHYLOGENY OF THE CONCHOSTRACA CLADOCERA CLADE (BRANCHIOPODA, DIPLOSTRACA) BY JØRGEN OLESEN 1 ) Humboldt Universität zu Berlin, Institut für Biologie, Vergleichende Zoologie, Philippstrasse 13,
AN UPDATED PHYLOGENY OF THE CONCHOSTRACA CLADOCERA CLADE (BRANCHIOPODA, DIPLOSTRACA) BY JØRGEN OLESEN 1 ) Humboldt Universität zu Berlin, Institut für Biologie, Vergleichende Zoologie, Philippstrasse 13,
Phylogeny of Branchiopoda (Crustacea) based on a combined analysis of morphological data and six molecular loci
 Cladistics Cladistics 23 (2007) 301 336 10.1111/j.1096-0031.2007.00148.x Phylogeny of Branchiopoda (Crustacea) based on a combined analysis of morphological data and six molecular loci Stefan Richter 1,3
Cladistics Cladistics 23 (2007) 301 336 10.1111/j.1096-0031.2007.00148.x Phylogeny of Branchiopoda (Crustacea) based on a combined analysis of morphological data and six molecular loci Stefan Richter 1,3
Københavns Universitet
 university of copenhagen Københavns Universitet The unique dorsal brood pouch of Thermosbaenacea (Crustacea, Malacostraca) and description of an advanced developmental stage of Tulumella unidens from the
university of copenhagen Københavns Universitet The unique dorsal brood pouch of Thermosbaenacea (Crustacea, Malacostraca) and description of an advanced developmental stage of Tulumella unidens from the
Hypothesis Testing in Evolutionary Developmental Biology: A Case Study from Insect Wings
 Journal of Heredity 2004:95(5):382 396 doi:10.1093/jhered/esh064 ª 2004 The American Genetic Association Hypothesis Testing in Evolutionary Developmental Biology: A Case Study from Insect Wings E. L. JOCKUSCH
Journal of Heredity 2004:95(5):382 396 doi:10.1093/jhered/esh064 ª 2004 The American Genetic Association Hypothesis Testing in Evolutionary Developmental Biology: A Case Study from Insect Wings E. L. JOCKUSCH
Cap n collar differentiates the mandible from the maxilla in the beetle Tribolium castaneum
 Coulcher and Telford EvoDevo 2012, 3:25 RESEARCH Open Access Cap n collar differentiates the mandible from the maxilla in the beetle Tribolium castaneum Joshua F Coulcher and Maximilian J Telford * Abstract
Coulcher and Telford EvoDevo 2012, 3:25 RESEARCH Open Access Cap n collar differentiates the mandible from the maxilla in the beetle Tribolium castaneum Joshua F Coulcher and Maximilian J Telford * Abstract
Patterns of Evolution
 Patterns of Evolution A tree that represents an estimate (hypothesis) of evolutionary relatedness is a phylogeny Classifications can be based on groupings within a phylogeny Groupings can be categorized
Patterns of Evolution A tree that represents an estimate (hypothesis) of evolutionary relatedness is a phylogeny Classifications can be based on groupings within a phylogeny Groupings can be categorized
Classifications can be based on groupings g within a phylogeny
 Patterns of Evolution A tree that represents an estimate (hypothesis) of evolutionary relatedness is a phylogeny Classifications can be based on groupings g within a phylogeny y Groupings can be categorized
Patterns of Evolution A tree that represents an estimate (hypothesis) of evolutionary relatedness is a phylogeny Classifications can be based on groupings g within a phylogeny y Groupings can be categorized
Arthropoda ARTHRO JOINTED PODA FEET
 Arthropoda ARTHRO JOINTED PODA FEET The arthropods are a group of animals which has attained the greatest biological success largest number of species and individuals and occupy the greatest number of
Arthropoda ARTHRO JOINTED PODA FEET The arthropods are a group of animals which has attained the greatest biological success largest number of species and individuals and occupy the greatest number of
Classification and Phylogeny
 Classification and Phylogeny The diversity it of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize without a scheme
Classification and Phylogeny The diversity it of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize without a scheme
Homeotic genes and the arthropod head: Expression patterns of the labial, proboscipedia, and Deformed genes in crustaceans and insects
 Proc. Natl. Acad. Sci. USA Vol. 96, pp. 10224 10229, August 1999 Evolution Homeotic genes and the arthropod head: Expression patterns of the labial, proboscipedia, and Deformed genes in crustaceans and
Proc. Natl. Acad. Sci. USA Vol. 96, pp. 10224 10229, August 1999 Evolution Homeotic genes and the arthropod head: Expression patterns of the labial, proboscipedia, and Deformed genes in crustaceans and
Non-independence in Statistical Tests for Discrete Cross-species Data
 J. theor. Biol. (1997) 188, 507514 Non-independence in Statistical Tests for Discrete Cross-species Data ALAN GRAFEN* AND MARK RIDLEY * St. John s College, Oxford OX1 3JP, and the Department of Zoology,
J. theor. Biol. (1997) 188, 507514 Non-independence in Statistical Tests for Discrete Cross-species Data ALAN GRAFEN* AND MARK RIDLEY * St. John s College, Oxford OX1 3JP, and the Department of Zoology,
Classification and Phylogeny
 Classification and Phylogeny The diversity of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize without a scheme
Classification and Phylogeny The diversity of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize without a scheme
SCIENTIFIC EVIDENCE TO SUPPORT THE THEORY OF EVOLUTION. Using Anatomy, Embryology, Biochemistry, and Paleontology
 SCIENTIFIC EVIDENCE TO SUPPORT THE THEORY OF EVOLUTION Using Anatomy, Embryology, Biochemistry, and Paleontology Scientific Fields Different fields of science have contributed evidence for the theory of
SCIENTIFIC EVIDENCE TO SUPPORT THE THEORY OF EVOLUTION Using Anatomy, Embryology, Biochemistry, and Paleontology Scientific Fields Different fields of science have contributed evidence for the theory of
/. W. Martin and C. E. Cash-Clark
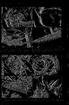 78 /. W. Martin and C. E. Cash-Clark 7 ig. 16. A. Outer view of spine-like exopods of thoracopods 1-3. Exopod of t4 also visible (although not spine-like). B. Exopod of thoracopod wo. Note small seta at
78 /. W. Martin and C. E. Cash-Clark 7 ig. 16. A. Outer view of spine-like exopods of thoracopods 1-3. Exopod of t4 also visible (although not spine-like). B. Exopod of thoracopod wo. Note small seta at
ESS 345 Ichthyology. Systematic Ichthyology Part II Not in Book
 ESS 345 Ichthyology Systematic Ichthyology Part II Not in Book Thought for today: Now, here, you see, it takes all the running you can do, to keep in the same place. If you want to get somewhere else,
ESS 345 Ichthyology Systematic Ichthyology Part II Not in Book Thought for today: Now, here, you see, it takes all the running you can do, to keep in the same place. If you want to get somewhere else,
Chapter 16: Reconstructing and Using Phylogenies
 Chapter Review 1. Use the phylogenetic tree shown at the right to complete the following. a. Explain how many clades are indicated: Three: (1) chimpanzee/human, (2) chimpanzee/ human/gorilla, and (3)chimpanzee/human/
Chapter Review 1. Use the phylogenetic tree shown at the right to complete the following. a. Explain how many clades are indicated: Three: (1) chimpanzee/human, (2) chimpanzee/ human/gorilla, and (3)chimpanzee/human/
Novel regulation of the homeotic gene Scr associated with a crustacean leg-to-maxilliped appendage transformation.
 Novel regulation of the homeotic gene Scr associated with a crustacean leg-to-maxilliped appendage transformation. The Harvard community has made this article openly available. Please share how this access
Novel regulation of the homeotic gene Scr associated with a crustacean leg-to-maxilliped appendage transformation. The Harvard community has made this article openly available. Please share how this access
Chapter 12: Aquatic Mandibulates
 Chapter 12: Aquatic Mandibulates Phylum Arthropoda Subphylum: Crustacea (Latin crusta = shell) Class: Malacostraca Order: Decapoda Order: Euphausiacea Order: Amphipoda Order: Isopoda Class: Maxillopoda
Chapter 12: Aquatic Mandibulates Phylum Arthropoda Subphylum: Crustacea (Latin crusta = shell) Class: Malacostraca Order: Decapoda Order: Euphausiacea Order: Amphipoda Order: Isopoda Class: Maxillopoda
Post-embryonic development of remipede crustaceans
 EVOLUTION & DEVELOPMENT 9:2, 117 121 (2007) Post-embryonic development of remipede crustaceans Stefan Koenemann, a, Frederick R. Schram, b Armin Bloechl, a Thomas M. Iliffe, c Mario Hoenemann, a and Christoph
EVOLUTION & DEVELOPMENT 9:2, 117 121 (2007) Post-embryonic development of remipede crustaceans Stefan Koenemann, a, Frederick R. Schram, b Armin Bloechl, a Thomas M. Iliffe, c Mario Hoenemann, a and Christoph
Molecular phylogeny of the Branchiopoda (Crustacea) Multiple approaches suggest a diplostracan ancestry of the Notostraca
 Molecular Phylogenetics and Evolution 41 (2006) 182 194 www.elsevier.com/locate/ympev Molecular phylogeny of the Branchiopoda (Crustacea) Multiple approaches suggest a diplostracan ancestry of the Notostraca
Molecular Phylogenetics and Evolution 41 (2006) 182 194 www.elsevier.com/locate/ympev Molecular phylogeny of the Branchiopoda (Crustacea) Multiple approaches suggest a diplostracan ancestry of the Notostraca
Analysis of nubbin expression patterns in insects
 EVOLUTION & DEVELOPMENT 6:5, 310 324 (2004) Analysis of nubbin expression patterns in insects Hua Li and Aleksandar Popadić Department of Biological Sciences, Wayne State University, Detroit, MI 48202,
EVOLUTION & DEVELOPMENT 6:5, 310 324 (2004) Analysis of nubbin expression patterns in insects Hua Li and Aleksandar Popadić Department of Biological Sciences, Wayne State University, Detroit, MI 48202,
Chapter 26 Phylogeny and the Tree of Life
 Chapter 26 Phylogeny and the Tree of Life Chapter focus Shifting from the process of how evolution works to the pattern evolution produces over time. Phylogeny Phylon = tribe, geny = genesis or origin
Chapter 26 Phylogeny and the Tree of Life Chapter focus Shifting from the process of how evolution works to the pattern evolution produces over time. Phylogeny Phylon = tribe, geny = genesis or origin
How should we organize the diversity of animal life?
 How should we organize the diversity of animal life? The difference between Taxonomy Linneaus, and Cladistics Darwin What are phylogenies? How do we read them? How do we estimate them? Classification (Taxonomy)
How should we organize the diversity of animal life? The difference between Taxonomy Linneaus, and Cladistics Darwin What are phylogenies? How do we read them? How do we estimate them? Classification (Taxonomy)
PHYLOGENY & THE TREE OF LIFE
 PHYLOGENY & THE TREE OF LIFE PREFACE In this powerpoint we learn how biologists distinguish and categorize the millions of species on earth. Early we looked at the process of evolution here we look at
PHYLOGENY & THE TREE OF LIFE PREFACE In this powerpoint we learn how biologists distinguish and categorize the millions of species on earth. Early we looked at the process of evolution here we look at
Classification, Phylogeny yand Evolutionary History
 Classification, Phylogeny yand Evolutionary History The diversity of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize
Classification, Phylogeny yand Evolutionary History The diversity of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize
Resolving arthropod relationships: Present and future insights from evo-devo studies
 Resolving arthropod relationships: Present and future insights from evo-devo studies STEVEN HRYCAJ & ALEKSANDAR POPADIC Department Of Biological Sciences, 5047 Gullen Mall, Wayne State University, Detroit,
Resolving arthropod relationships: Present and future insights from evo-devo studies STEVEN HRYCAJ & ALEKSANDAR POPADIC Department Of Biological Sciences, 5047 Gullen Mall, Wayne State University, Detroit,
Comparative Mouthpart Morphology and Evolution of the Carnivorous Heptageniidae (Ephemeroptera) 1
 l Aquatic Insects, Vol. 8 (1986). No. 2, pp. 83-89 0165-0424/86/0802-0083 $3.00 Swets & Zeitlinger Comparative Mouthpart Morphology and Evolution of the Carnivorous Heptageniidae (Ephemeroptera) 1 by W.
l Aquatic Insects, Vol. 8 (1986). No. 2, pp. 83-89 0165-0424/86/0802-0083 $3.00 Swets & Zeitlinger Comparative Mouthpart Morphology and Evolution of the Carnivorous Heptageniidae (Ephemeroptera) 1 by W.
^ ^ LIBRARY Division of Crustace;
 /re/// ^ ^ LIBRARY Division of Crustace; Larval Development of Helice tridens wuana Rathbun ^Y RTEBRAT and H. tridens tridens de Haan (Crustacea, Brachyura) ZOOLOGY Reared in the Laboratory Crustacea By
/re/// ^ ^ LIBRARY Division of Crustace; Larval Development of Helice tridens wuana Rathbun ^Y RTEBRAT and H. tridens tridens de Haan (Crustacea, Brachyura) ZOOLOGY Reared in the Laboratory Crustacea By
Biology 211 (2) Week 1 KEY!
 Biology 211 (2) Week 1 KEY Chapter 1 KEY FIGURES: 1.2, 1.3, 1.4, 1.5, 1.6, 1.7 VOCABULARY: Adaptation: a trait that increases the fitness Cells: a developed, system bound with a thin outer layer made of
Biology 211 (2) Week 1 KEY Chapter 1 KEY FIGURES: 1.2, 1.3, 1.4, 1.5, 1.6, 1.7 VOCABULARY: Adaptation: a trait that increases the fitness Cells: a developed, system bound with a thin outer layer made of
Systematics Lecture 3 Characters: Homology, Morphology
 Systematics Lecture 3 Characters: Homology, Morphology I. Introduction Nearly all methods of phylogenetic analysis rely on characters as the source of data. A. Character variation is coded into a character-by-taxon
Systematics Lecture 3 Characters: Homology, Morphology I. Introduction Nearly all methods of phylogenetic analysis rely on characters as the source of data. A. Character variation is coded into a character-by-taxon
Evolution of the Arthropod Mandible:
 Evolution of the Arthropod Mandible: a molecular developmental perspective Joshua Frederick Coulcher UCL Submitted for the Degree of Doctor of Philosophy September 2011 Declaration I, Joshua Frederick
Evolution of the Arthropod Mandible: a molecular developmental perspective Joshua Frederick Coulcher UCL Submitted for the Degree of Doctor of Philosophy September 2011 Declaration I, Joshua Frederick
Phytogeny, evolution and classification of the Branchiopoda (Crustacea)
 l - I y d w b U M w 412: 101-2 12, i m (f) 1999 K hm er Academie Publisher.';. Printed in the Netherlands. 191 Phytogeny, evolution and classification of the Branchiopoda (Crustacea) Stefan Negrea1, Nicolae
l - I y d w b U M w 412: 101-2 12, i m (f) 1999 K hm er Academie Publisher.';. Printed in the Netherlands. 191 Phytogeny, evolution and classification of the Branchiopoda (Crustacea) Stefan Negrea1, Nicolae
Importance of Taxonomic Collections
 Importance of Taxonomic Collections Document earth s biodiversity Facilitate the process of researching relationships among and within different groups of organisms Study ecological processes using special
Importance of Taxonomic Collections Document earth s biodiversity Facilitate the process of researching relationships among and within different groups of organisms Study ecological processes using special
Conservation and diversification of gene function during mouthpart development in Onthophagus beetles
 EVOLUTION & DEVELOPMENT 13:3, 280 289 (2011) DOI: 10.1111/j.1525-142X.2011.00479.x Conservation and diversification of gene function during mouthpart development in Onthophagus beetles Franck Simonnet
EVOLUTION & DEVELOPMENT 13:3, 280 289 (2011) DOI: 10.1111/j.1525-142X.2011.00479.x Conservation and diversification of gene function during mouthpart development in Onthophagus beetles Franck Simonnet
Arthropods (Arthropoda)
 Arthropods (Arthropoda) Davide Pisani Laboratory of Evolutionary Biology, Department of Biology, The National University of Ireland, Maynooth, Co. Kildare, Ireland (davide. pisani@nuim.ie) Abstract Living
Arthropods (Arthropoda) Davide Pisani Laboratory of Evolutionary Biology, Department of Biology, The National University of Ireland, Maynooth, Co. Kildare, Ireland (davide. pisani@nuim.ie) Abstract Living
18.4 Embryonic development involves cell division, cell differentiation, and morphogenesis
 18.4 Embryonic development involves cell division, cell differentiation, and morphogenesis An organism arises from a fertilized egg cell as the result of three interrelated processes: cell division, cell
18.4 Embryonic development involves cell division, cell differentiation, and morphogenesis An organism arises from a fertilized egg cell as the result of three interrelated processes: cell division, cell
Biologists have used many approaches to estimating the evolutionary history of organisms and using that history to construct classifications.
 Phylogenetic Inference Biologists have used many approaches to estimating the evolutionary history of organisms and using that history to construct classifications. Willi Hennig developed d the techniques
Phylogenetic Inference Biologists have used many approaches to estimating the evolutionary history of organisms and using that history to construct classifications. Willi Hennig developed d the techniques
Understanding the genetic basis of morphological evolution: the role of homeotic genes in the diversification of the arthropod bauplan
 Int. J. Dev. Biol. 42: 453-461 (1998) Understanding the genetic basis of morphological evolution: the role of homeotic genes in the diversification of the arthropod bauplan ALEKSANDAR POPADIC, ARHAT ABZHANOV,
Int. J. Dev. Biol. 42: 453-461 (1998) Understanding the genetic basis of morphological evolution: the role of homeotic genes in the diversification of the arthropod bauplan ALEKSANDAR POPADIC, ARHAT ABZHANOV,
The post-embryonic development of Remipedia (Crustacea) additional results and new insights
 Dev Genes Evol (2009) 219:131 145 DOI 10.1007/s00427-009-0273-0 ORIGINAL ARTICLE The post-embryonic development of Remipedia (Crustacea) additional results and new insights Stefan Koenemann & Jørgen Olesen
Dev Genes Evol (2009) 219:131 145 DOI 10.1007/s00427-009-0273-0 ORIGINAL ARTICLE The post-embryonic development of Remipedia (Crustacea) additional results and new insights Stefan Koenemann & Jørgen Olesen
Chapter 26: Phylogeny and the Tree of Life Phylogenies Show Evolutionary Relationships
 Chapter 26: Phylogeny and the Tree of Life You Must Know The taxonomic categories and how they indicate relatedness. How systematics is used to develop phylogenetic trees. How to construct a phylogenetic
Chapter 26: Phylogeny and the Tree of Life You Must Know The taxonomic categories and how they indicate relatedness. How systematics is used to develop phylogenetic trees. How to construct a phylogenetic
Structural rrna characters support monophyly of raptorial limbs and paraphyly of limb specialization in water fleas
 Received 6 November 2002 ccepted 3 December 2002 Published online 12 March 2003 Structural rrn characters support monophyly of raptorial and paraphyly of limb specialization in water fleas Timothy D. Swain
Received 6 November 2002 ccepted 3 December 2002 Published online 12 March 2003 Structural rrn characters support monophyly of raptorial and paraphyly of limb specialization in water fleas Timothy D. Swain
Macroevolution Part I: Phylogenies
 Macroevolution Part I: Phylogenies Taxonomy Classification originated with Carolus Linnaeus in the 18 th century. Based on structural (outward and inward) similarities Hierarchal scheme, the largest most
Macroevolution Part I: Phylogenies Taxonomy Classification originated with Carolus Linnaeus in the 18 th century. Based on structural (outward and inward) similarities Hierarchal scheme, the largest most
Homeotic genes in flies. Sem 9.3.B.6 Animal Science
 Homeotic genes in flies Sem 9.3.B.6 Animal Science So far We have seen that identities of each segment is determined by various regulators of segment polarity genes In arthopods, and in flies, each segment
Homeotic genes in flies Sem 9.3.B.6 Animal Science So far We have seen that identities of each segment is determined by various regulators of segment polarity genes In arthopods, and in flies, each segment
Phylogeny is the evolutionary history of a group of organisms. Based on the idea that organisms are related by evolution
 Bio 1M: Phylogeny and the history of life 1 Phylogeny S25.1; Bioskill 11 (2ndEd S27.1; Bioskills 3) Bioskills are in the back of your book Phylogeny is the evolutionary history of a group of organisms
Bio 1M: Phylogeny and the history of life 1 Phylogeny S25.1; Bioskill 11 (2ndEd S27.1; Bioskills 3) Bioskills are in the back of your book Phylogeny is the evolutionary history of a group of organisms
Evolving role of Antennapedia protein in arthropod limb patterning
 Development 129, 3555-3561 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV7965 3555 Evolving role of Antennapedia protein in arthropod limb patterning Yasuhiro Shiga 1, *, Ryusuke
Development 129, 3555-3561 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV7965 3555 Evolving role of Antennapedia protein in arthropod limb patterning Yasuhiro Shiga 1, *, Ryusuke
Evolutionary Developmental Biology
 Evolutionary Developmental Biology a.k.a. EVO-DEVO Paedomorphosis is common among salamanders. Note how this hellbender (top) and mudpuppy (bottom) both have gills, paddle tails, and weaker limbs... Top:
Evolutionary Developmental Biology a.k.a. EVO-DEVO Paedomorphosis is common among salamanders. Note how this hellbender (top) and mudpuppy (bottom) both have gills, paddle tails, and weaker limbs... Top:
8/23/2014. Introduction to Animal Diversity
 Introduction to Animal Diversity Chapter 32 Objectives List the characteristics that combine to define animals Summarize key events of the Paleozoic, Mesozoic, and Cenozoic eras Distinguish between the
Introduction to Animal Diversity Chapter 32 Objectives List the characteristics that combine to define animals Summarize key events of the Paleozoic, Mesozoic, and Cenozoic eras Distinguish between the
Outline. Classification of Living Things
 Outline Classification of Living Things Chapter 20 Mader: Biology 8th Ed. Taxonomy Binomial System Species Identification Classification Categories Phylogenetic Trees Tracing Phylogeny Cladistic Systematics
Outline Classification of Living Things Chapter 20 Mader: Biology 8th Ed. Taxonomy Binomial System Species Identification Classification Categories Phylogenetic Trees Tracing Phylogeny Cladistic Systematics
Forty. Annelids. The. group of in humid. elongate, worm-like. bodies with
 WEEK 2: INSECT MACROEVOLUTION Forty million years ago some insects were trapped in tree resin and preserved in what became amber. These trapped insects look almost exactly the same as insects around us
WEEK 2: INSECT MACROEVOLUTION Forty million years ago some insects were trapped in tree resin and preserved in what became amber. These trapped insects look almost exactly the same as insects around us
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila November 2, 2006 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Developmental Biology Biology 4361 Axis Specification in Drosophila November 2, 2006 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Introduction to Lepidoptera
 Introduction to Lepidoptera Taxonomic Workshop for Early Detection of Important Tortricidae and Other Lepidopteran Agricultural and Silvicultural Pests UMass Amherst 15-17 July 2013 Todd M. Gilligan, Ph.D.
Introduction to Lepidoptera Taxonomic Workshop for Early Detection of Important Tortricidae and Other Lepidopteran Agricultural and Silvicultural Pests UMass Amherst 15-17 July 2013 Todd M. Gilligan, Ph.D.
8/23/2014. Phylogeny and the Tree of Life
 Phylogeny and the Tree of Life Chapter 26 Objectives Explain the following characteristics of the Linnaean system of classification: a. binomial nomenclature b. hierarchical classification List the major
Phylogeny and the Tree of Life Chapter 26 Objectives Explain the following characteristics of the Linnaean system of classification: a. binomial nomenclature b. hierarchical classification List the major
Bulletin Zoölogisch Museum
 Bulletin Zoölogisch Museum UNIVERSITEIT VAN AMSTERDAM Vol.11 No. 15 1988 Redescription of johanna Monod, 1926 Virgin Isls (Isopoda) from St. John, Hans Georg Müller Summary Based on the type material,
Bulletin Zoölogisch Museum UNIVERSITEIT VAN AMSTERDAM Vol.11 No. 15 1988 Redescription of johanna Monod, 1926 Virgin Isls (Isopoda) from St. John, Hans Georg Müller Summary Based on the type material,
As a result of advancements in the past two decades, the
 Differential expression patterns of the hox gene are associated with differential growth of insect hind legs Najmus S. Mahfooz*, Hua Li*, and Aleksandar Popadić Department of Biological Sciences, Wayne
Differential expression patterns of the hox gene are associated with differential growth of insect hind legs Najmus S. Mahfooz*, Hua Li*, and Aleksandar Popadić Department of Biological Sciences, Wayne
Biology 1B Evolution Lecture 2 (February 26, 2010) Natural Selection, Phylogenies
 1 Natural Selection (Darwin-Wallace): There are three conditions for natural selection: 1. Variation: Individuals within a population have different characteristics/traits (or phenotypes). 2. Inheritance:
1 Natural Selection (Darwin-Wallace): There are three conditions for natural selection: 1. Variation: Individuals within a population have different characteristics/traits (or phenotypes). 2. Inheritance:
Lecture V Phylogeny and Systematics Dr. Kopeny
 Delivered 1/30 and 2/1 Lecture V Phylogeny and Systematics Dr. Kopeny Lecture V How to Determine Evolutionary Relationships: Concepts in Phylogeny and Systematics Textbook Reading: pp 425-433, 435-437
Delivered 1/30 and 2/1 Lecture V Phylogeny and Systematics Dr. Kopeny Lecture V How to Determine Evolutionary Relationships: Concepts in Phylogeny and Systematics Textbook Reading: pp 425-433, 435-437
Hexapoda Origins: Monophyletic, Paraphyletic or Polyphyletic? Rob King and Matt Kretz
 Hexapoda Origins: Monophyletic, Paraphyletic or Polyphyletic? Rob King and Matt Kretz Outline Review Hexapod Origins Response to Hexapod Origins How the same data = different trees Arthropod Origins The
Hexapoda Origins: Monophyletic, Paraphyletic or Polyphyletic? Rob King and Matt Kretz Outline Review Hexapod Origins Response to Hexapod Origins How the same data = different trees Arthropod Origins The
Microbial Taxonomy. Microbes usually have few distinguishing properties that relate them, so a hierarchical taxonomy mainly has not been possible.
 Microbial Taxonomy Traditional taxonomy or the classification through identification and nomenclature of microbes, both "prokaryote" and eukaryote, has been in a mess we were stuck with it for traditional
Microbial Taxonomy Traditional taxonomy or the classification through identification and nomenclature of microbes, both "prokaryote" and eukaryote, has been in a mess we were stuck with it for traditional
Antennal Regeneration in Daphnia Magna
 The Ohio State University Knowledge Bank kb.osu.edu Ohio Journal of Science (Ohio Academy of Science) Ohio Journal of Science: Volume 35, Issue 2 (March, 1935) 1935-03 Antennal Regeneration in Daphnia
The Ohio State University Knowledge Bank kb.osu.edu Ohio Journal of Science (Ohio Academy of Science) Ohio Journal of Science: Volume 35, Issue 2 (March, 1935) 1935-03 Antennal Regeneration in Daphnia
Chapter 26 Phylogeny and the Tree of Life
 Chapter 26 Phylogeny and the Tree of Life Biologists estimate that there are about 5 to 100 million species of organisms living on Earth today. Evidence from morphological, biochemical, and gene sequence
Chapter 26 Phylogeny and the Tree of Life Biologists estimate that there are about 5 to 100 million species of organisms living on Earth today. Evidence from morphological, biochemical, and gene sequence
PR1VATE LIBRARY OE WILLIAM L P.EIER_.S
 PR1VATE LIBRARY OE WILLIAM L P.EIER_.S PENTAGENllDAE: A NEW FAMILY OF EPHEMEROIDEA (EPHEMEROPTERA) 1 2 W. P. McCAFFERTY 3 Department of Entomology University of Georgia Athens, Georgia 30601 ABSTRACT On
PR1VATE LIBRARY OE WILLIAM L P.EIER_.S PENTAGENllDAE: A NEW FAMILY OF EPHEMEROIDEA (EPHEMEROPTERA) 1 2 W. P. McCAFFERTY 3 Department of Entomology University of Georgia Athens, Georgia 30601 ABSTRACT On
Chapter Chemical Uniqueness 1/23/2009. The Uses of Principles. Zoology: the Study of Animal Life. Fig. 1.1
 Fig. 1.1 Chapter 1 Life: Biological Principles and the Science of Zoology BIO 2402 General Zoology Copyright The McGraw Hill Companies, Inc. Permission required for reproduction or display. The Uses of
Fig. 1.1 Chapter 1 Life: Biological Principles and the Science of Zoology BIO 2402 General Zoology Copyright The McGraw Hill Companies, Inc. Permission required for reproduction or display. The Uses of
Phylogeny 9/8/2014. Evolutionary Relationships. Data Supporting Phylogeny. Chapter 26
 Phylogeny Chapter 26 Taxonomy Taxonomy: ordered division of organisms into categories based on a set of characteristics used to assess similarities and differences Carolus Linnaeus developed binomial nomenclature,
Phylogeny Chapter 26 Taxonomy Taxonomy: ordered division of organisms into categories based on a set of characteristics used to assess similarities and differences Carolus Linnaeus developed binomial nomenclature,
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila November 6, 2007 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Developmental Biology Biology 4361 Axis Specification in Drosophila November 6, 2007 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Midterm 1. Average score: 74.4 Median score: 77
 Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
Principles of Experimental Embryology
 Biology 4361 Developmental Biology Principles of Experimental Embryology September 19, 2006 Major Research Questions How do forces outside the embryo affect its development? (Environmental Developmental
Biology 4361 Developmental Biology Principles of Experimental Embryology September 19, 2006 Major Research Questions How do forces outside the embryo affect its development? (Environmental Developmental
Assessing an Unknown Evolutionary Process: Effect of Increasing Site- Specific Knowledge Through Taxon Addition
 Assessing an Unknown Evolutionary Process: Effect of Increasing Site- Specific Knowledge Through Taxon Addition David D. Pollock* and William J. Bruno* *Theoretical Biology and Biophysics, Los Alamos National
Assessing an Unknown Evolutionary Process: Effect of Increasing Site- Specific Knowledge Through Taxon Addition David D. Pollock* and William J. Bruno* *Theoretical Biology and Biophysics, Los Alamos National
CHAPTERS 24-25: Evidence for Evolution and Phylogeny
 CHAPTERS 24-25: Evidence for Evolution and Phylogeny 1. For each of the following, indicate how it is used as evidence of evolution by natural selection or shown as an evolutionary trend: a. Paleontology
CHAPTERS 24-25: Evidence for Evolution and Phylogeny 1. For each of the following, indicate how it is used as evidence of evolution by natural selection or shown as an evolutionary trend: a. Paleontology
Reconstructing the history of lineages
 Reconstructing the history of lineages Class outline Systematics Phylogenetic systematics Phylogenetic trees and maps Class outline Definitions Systematics Phylogenetic systematics/cladistics Systematics
Reconstructing the history of lineages Class outline Systematics Phylogenetic systematics Phylogenetic trees and maps Class outline Definitions Systematics Phylogenetic systematics/cladistics Systematics
From DNA to Diversity
 From DNA to Diversity Molecular Genetics and the Evolution of Animal Design Sean B. Carroll Jennifer K. Grenier Scott D. Weatherbee Howard Hughes Medical Institute and University of Wisconsin Madison,
From DNA to Diversity Molecular Genetics and the Evolution of Animal Design Sean B. Carroll Jennifer K. Grenier Scott D. Weatherbee Howard Hughes Medical Institute and University of Wisconsin Madison,
"PRINCIPLES OF PHYLOGENETICS: ECOLOGY AND EVOLUTION" Integrative Biology 200 Spring 2018 University of California, Berkeley
 "PRINCIPLES OF PHYLOGENETICS: ECOLOGY AND EVOLUTION" Integrative Biology 200 Spring 2018 University of California, Berkeley D.D. Ackerly Feb. 26, 2018 Maximum Likelihood Principles, and Applications to
"PRINCIPLES OF PHYLOGENETICS: ECOLOGY AND EVOLUTION" Integrative Biology 200 Spring 2018 University of California, Berkeley D.D. Ackerly Feb. 26, 2018 Maximum Likelihood Principles, and Applications to
Integrative Biology 200 "PRINCIPLES OF PHYLOGENETICS" Spring 2018 University of California, Berkeley
 Integrative Biology 200 "PRINCIPLES OF PHYLOGENETICS" Spring 2018 University of California, Berkeley B.D. Mishler Feb. 14, 2018. Phylogenetic trees VI: Dating in the 21st century: clocks, & calibrations;
Integrative Biology 200 "PRINCIPLES OF PHYLOGENETICS" Spring 2018 University of California, Berkeley B.D. Mishler Feb. 14, 2018. Phylogenetic trees VI: Dating in the 21st century: clocks, & calibrations;
Amira A. AL-Hosary PhD of infectious diseases Department of Animal Medicine (Infectious Diseases) Faculty of Veterinary Medicine Assiut
 Amira A. AL-Hosary PhD of infectious diseases Department of Animal Medicine (Infectious Diseases) Faculty of Veterinary Medicine Assiut University-Egypt Phylogenetic analysis Phylogenetic Basics: Biological
Amira A. AL-Hosary PhD of infectious diseases Department of Animal Medicine (Infectious Diseases) Faculty of Veterinary Medicine Assiut University-Egypt Phylogenetic analysis Phylogenetic Basics: Biological
Chapter 19: Taxonomy, Systematics, and Phylogeny
 Chapter 19: Taxonomy, Systematics, and Phylogeny AP Curriculum Alignment Chapter 19 expands on the topics of phylogenies and cladograms, which are important to Big Idea 1. In order for students to understand
Chapter 19: Taxonomy, Systematics, and Phylogeny AP Curriculum Alignment Chapter 19 expands on the topics of phylogenies and cladograms, which are important to Big Idea 1. In order for students to understand
Chapter 10. Objectives. Contrast the pre-darwin world view w/ the post-darwin world view Examine early ideas about evolution.
 Objectives Section 1 Early Ideas About Evolution Contrast the pre-darwin world view w/ the post-darwin world view Examine early ideas about evolution. Section 1 Science Before Darwin God created man in
Objectives Section 1 Early Ideas About Evolution Contrast the pre-darwin world view w/ the post-darwin world view Examine early ideas about evolution. Section 1 Science Before Darwin God created man in
Commentary A review of the correlation of tergites, sternites, and leg pairs in diplopods Ralf Janssen, Nikola-Michael Prpic and WimGMDamen*
 Frontiers in Zoology BioMed Central Commentary A review of the correlation of tergites, sternites, and leg pairs in diplopods Ralf Janssen, Nikola-Michael Prpic and WimGMDamen* Open Access Address: Department
Frontiers in Zoology BioMed Central Commentary A review of the correlation of tergites, sternites, and leg pairs in diplopods Ralf Janssen, Nikola-Michael Prpic and WimGMDamen* Open Access Address: Department
Phylogeny and systematics. Why are these disciplines important in evolutionary biology and how are they related to each other?
 Phylogeny and systematics Why are these disciplines important in evolutionary biology and how are they related to each other? Phylogeny and systematics Phylogeny: the evolutionary history of a species
Phylogeny and systematics Why are these disciplines important in evolutionary biology and how are they related to each other? Phylogeny and systematics Phylogeny: the evolutionary history of a species
Biology. Slide 1 of 24. End Show. Copyright Pearson Prentice Hall
 Biology 1 of 24 18-2 Modern Evolutionary Classification 2 of 24 18-2 Modern Evolutionary Classification Evolutionary Classification Evolutionary Classification Phylogeny is the study of evolutionary relationships
Biology 1 of 24 18-2 Modern Evolutionary Classification 2 of 24 18-2 Modern Evolutionary Classification Evolutionary Classification Evolutionary Classification Phylogeny is the study of evolutionary relationships
Anatomy of a tree. clade is group of organisms with a shared ancestor. a monophyletic group shares a single common ancestor = tapirs-rhinos-horses
 Anatomy of a tree outgroup: an early branching relative of the interest groups sister taxa: taxa derived from the same recent ancestor polytomy: >2 taxa emerge from a node Anatomy of a tree clade is group
Anatomy of a tree outgroup: an early branching relative of the interest groups sister taxa: taxa derived from the same recent ancestor polytomy: >2 taxa emerge from a node Anatomy of a tree clade is group
Patterning of the Adult Mandibulate Mouthparts in the Red Flour Beetle, Tribolium castaneum
 INVESTIGATION Patterning of the Adult Mandibulate Mouthparts in the Red Flour Beetle, Tribolium castaneum David R. Angelini,*,,1 Frank W. Smith, Ariel C. Aspiras,* Moto Kikuchi, and Elizabeth L. Jockusch
INVESTIGATION Patterning of the Adult Mandibulate Mouthparts in the Red Flour Beetle, Tribolium castaneum David R. Angelini,*,,1 Frank W. Smith, Ariel C. Aspiras,* Moto Kikuchi, and Elizabeth L. Jockusch
Phylogenies & Classifying species (AKA Cladistics & Taxonomy) What are phylogenies & cladograms? How do we read them? How do we estimate them?
 Phylogenies & Classifying species (AKA Cladistics & Taxonomy) What are phylogenies & cladograms? How do we read them? How do we estimate them? Carolus Linneaus:Systema Naturae (1735) Swedish botanist &
Phylogenies & Classifying species (AKA Cladistics & Taxonomy) What are phylogenies & cladograms? How do we read them? How do we estimate them? Carolus Linneaus:Systema Naturae (1735) Swedish botanist &
RALF JANSSEN* Uppsala University, Department of Earth Sciences, Palaeobiology, Uppsala, Sweden
 Int. J. Dev. Biol. 58: 343-347 (2014) doi: 10.1387/ijdb.140058rj www.intjdevbiol.com Gene expression suggests double-segmental and single-segmental patterning mechanisms during posterior segment addition
Int. J. Dev. Biol. 58: 343-347 (2014) doi: 10.1387/ijdb.140058rj www.intjdevbiol.com Gene expression suggests double-segmental and single-segmental patterning mechanisms during posterior segment addition
Need for systematics. Applications of systematics. Linnaeus plus Darwin. Approaches in systematics. Principles of cladistics
 Topics Need for systematics Applications of systematics Linnaeus plus Darwin Approaches in systematics Principles of cladistics Systematics pp. 474-475. Systematics - Study of diversity and evolutionary
Topics Need for systematics Applications of systematics Linnaeus plus Darwin Approaches in systematics Principles of cladistics Systematics pp. 474-475. Systematics - Study of diversity and evolutionary
Classification in General
 Classification in General Domains Carl Woese-1980s Based upon modern techniques Sequence of rrna in ribosomes trna Plasma membrane lipid structure Sensitivity to antibiotics Three cell types based upon
Classification in General Domains Carl Woese-1980s Based upon modern techniques Sequence of rrna in ribosomes trna Plasma membrane lipid structure Sensitivity to antibiotics Three cell types based upon
Heredity and Evolution
 Heredity and Variation Heredity and Evolution Living organisms have certain recognisable heritable features such as height, complexion, colour of hair and eyes, shape of nose and chin etc. These are called
Heredity and Variation Heredity and Evolution Living organisms have certain recognisable heritable features such as height, complexion, colour of hair and eyes, shape of nose and chin etc. These are called
Unit 2 Biodiversity Ch. 4 Patterns of Life
 Unit 2 Biodiversity Ch. 4 Patterns of Life Name: 4.1 Characteristics of Life In order to be considered living, an organism must possess the following Six (6) characteristics: 1. Living things are organized
Unit 2 Biodiversity Ch. 4 Patterns of Life Name: 4.1 Characteristics of Life In order to be considered living, an organism must possess the following Six (6) characteristics: 1. Living things are organized
Using phylogenetics to estimate species divergence times... Basics and basic issues for Bayesian inference of divergence times (plus some digression)
 Using phylogenetics to estimate species divergence times... More accurately... Basics and basic issues for Bayesian inference of divergence times (plus some digression) "A comparison of the structures
Using phylogenetics to estimate species divergence times... More accurately... Basics and basic issues for Bayesian inference of divergence times (plus some digression) "A comparison of the structures
Lecture 11 Friday, October 21, 2011
 Lecture 11 Friday, October 21, 2011 Phylogenetic tree (phylogeny) Darwin and classification: In the Origin, Darwin said that descent from a common ancestral species could explain why the Linnaean system
Lecture 11 Friday, October 21, 2011 Phylogenetic tree (phylogeny) Darwin and classification: In the Origin, Darwin said that descent from a common ancestral species could explain why the Linnaean system
Frank D. Ferrari, Viatcheslav N. Ivanenko, and Hans-Uwe Dahms
 BODY ARCHITECTURE AND RELATIONSHIPS AMONG BASAL COPEPODS Frank D. Ferrari, Viatcheslav N. Ivanenko, and Hans-Uwe Dahms (FDF, correspondence, ferrarif@si.edu) IZ/MSC, MRC-534, National Museum of Natural
BODY ARCHITECTURE AND RELATIONSHIPS AMONG BASAL COPEPODS Frank D. Ferrari, Viatcheslav N. Ivanenko, and Hans-Uwe Dahms (FDF, correspondence, ferrarif@si.edu) IZ/MSC, MRC-534, National Museum of Natural
Carolin Haug 1*, Wafaa S Sallam 2, Andreas Maas 3, Dieter Waloszek 3, Verena Kutschera 3 and Joachim T Haug 1
 Haug et al. Frontiers in Zoology 2012, 9:31 RESEARCH Open Access Tagmatization in Stomatopoda reconsidering functional units of modern-day mantis shrimps (Verunipeltata, Hoplocarida) and implications for
Haug et al. Frontiers in Zoology 2012, 9:31 RESEARCH Open Access Tagmatization in Stomatopoda reconsidering functional units of modern-day mantis shrimps (Verunipeltata, Hoplocarida) and implications for
CLASSIFICATION OF LIVING THINGS. Chapter 18
 CLASSIFICATION OF LIVING THINGS Chapter 18 How many species are there? About 1.8 million species have been given scientific names Nearly 2/3 of which are insects 99% of all known animal species are smaller
CLASSIFICATION OF LIVING THINGS Chapter 18 How many species are there? About 1.8 million species have been given scientific names Nearly 2/3 of which are insects 99% of all known animal species are smaller
EXTERNAL ANATOMY OF INSECTS
 External Anatomy of Insects 1 The insect s exoskeleton is made up of a series of plates EXTERNAL ANATOMY OF INSECTS These plates make up the insect s exoskeleton. These plates are connected by joints or
External Anatomy of Insects 1 The insect s exoskeleton is made up of a series of plates EXTERNAL ANATOMY OF INSECTS These plates make up the insect s exoskeleton. These plates are connected by joints or
The process by which the genetic structure of populations changes over time.
 Evolution The process by which the genetic structure of populations changes over time. Divergent evolution is the accumulation of differences between groups which can lead to the formation of new species.
Evolution The process by which the genetic structure of populations changes over time. Divergent evolution is the accumulation of differences between groups which can lead to the formation of new species.
Developmental Biology Lecture Outlines
 Developmental Biology Lecture Outlines Lecture 01: Introduction Course content Developmental Biology Obsolete hypotheses Current theory Lecture 02: Gametogenesis Spermatozoa Spermatozoon function Spermatozoon
Developmental Biology Lecture Outlines Lecture 01: Introduction Course content Developmental Biology Obsolete hypotheses Current theory Lecture 02: Gametogenesis Spermatozoa Spermatozoon function Spermatozoon
Introduction to Animal Diversity Lecture 7 Winter 2014
 Introduction to Animal Diversity Lecture 7 Winter 2014 Evolution of Animals 1 Prokaryotes Eukaryotes Prokaryotes No nucleus Nucleoid region Simple No membrane bound organelles Smaller (1-5 nm) Evolutionarily
Introduction to Animal Diversity Lecture 7 Winter 2014 Evolution of Animals 1 Prokaryotes Eukaryotes Prokaryotes No nucleus Nucleoid region Simple No membrane bound organelles Smaller (1-5 nm) Evolutionarily
Outline. v Definition and major characteristics of animals v Dividing animals into groups based on: v Animal Phylogeny
 BIOSC 041 Overview of Animal Diversity: Animal Body Plans Reference: Chapter 32 Outline v Definition and major characteristics of animals v Dividing animals into groups based on: Body symmetry Tissues
BIOSC 041 Overview of Animal Diversity: Animal Body Plans Reference: Chapter 32 Outline v Definition and major characteristics of animals v Dividing animals into groups based on: Body symmetry Tissues
