Homeotic genes and the arthropod head: Expression patterns of the labial, proboscipedia, and Deformed genes in crustaceans and insects
|
|
|
- Dale Randall
- 5 years ago
- Views:
Transcription
1 Proc. Natl. Acad. Sci. USA Vol. 96, pp , August 1999 Evolution Homeotic genes and the arthropod head: Expression patterns of the labial, proboscipedia, and Deformed genes in crustaceans and insects ARHAT ABZHANOV AND THOMAS C. KAUFMAN* Howard Hughes Medical Institute, Department of Biology, Indiana University, Bloomington, IN Communicated by Edward B. Lewis, California Institute of Technology, Pasadena, CA, June 25, 1999 (received for review March 22, 1999) ABSTRACT cdna fragments of the homologues of the Drosophila head homeotic genes labial (lab), proboscipedia (pb), and Deformed (Dfd) have been isolated from the crustacean Porcellio scaber. Because the accumulation domains of the head homeotic complex (Hox) genes had not been previously reported for crustaceans, we studied the expression patterns of these genes in P. scaber embryos by using in situ hybridization. The P. scaber lab homologue is expressed in the developing second antennal segment and its appendages. This expression domain in crustaceans and in the homologous intercalary segment of insects suggests that the lab gene specified this metamere in the last common ancestor of these two groups. The expression domain of the P. scaber pb gene is in the posterior part of the second antennal segment. This domain, in contrast to that in insects, is colinear with the domains of other head genes in P. scaber, and it differs from the insect pb gene expression domain in the posterior mouthparts, suggesting that the insect and crustacean patterns evolved independently from a broader ancestral domain similar to that found in modern chelicerates. P. scaber Dfd is expressed in the mandibular segment and paragnaths (a pair of ventral mouthpart structures associated with the stomodeum) and differs from insects, where expression is in the mandibular and maxillary segments. Thus, like pb, Dfd shows a divergent Hox gene deployment. We conclude that homologous structures of the mandibulate head display striking differences in their underlying developmental programs related to Hox gene expression. Homologues of the Drosophila homeotic complex (HOM-C) genes from various arthropods have been the subject of intense research and comparative analysis (1 10). A goal of these studies has been to understand the role of these genes in the morphological evolution of this group. The function of Hox gene products as selective transcription factors is apparently highly conserved in animals (1, 2), and the expression patterns of these genes have been used as stable molecular markers for judging evolutionary relationships, such as homologies, of structures along the anteroposterior axis (5, 7, 8). Nevertheless, for the Hox genes to serve as useful tools for phylogenetic comparisons, a better understanding of their particular evolutionary history is needed. Moreover, observations on their expression patterns should ideally be coupled with tests for developmental function (6, 10, 11). Thus far, such comparative analyses have been done primarily on Hox genes in insects, on trunk Hox genes in the crustacean Artemia franciscana, and, most recently, in the chelicerates (3 7). Some models for the roles of Hox genes in the evolution of arthropods have already been proposed (2, 4, 5). However, the vast phylogenetic distances between and among the groups studied and the small The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C solely to indicate this fact. PNAS is available online at assortment of genes analyzed make it difficult to create a complete picture of the evolution of Hox genes in arthropods. In Drosophila, the Hox genes specify structures along the anteroposterior body axis; three are necessary for the correct development of head segments and appendages: the labial (lab), proboscipedia (pb), and Deformed (Dfd) genes. The expression patterns of these genes in insects show small, distinct, and usually nonoverlapping expression domains covering one to two segments. This pattern is in sharp contrast to the expression patterns of their homologues in vertebrate (lab, pb, and Dfd are homologous to the genes of vertebrate classes 1, 2, and 4 respectively) and chelicerate embryos, where expression domains are broadly overlapping and many segments long (7, 8, 12). The fact that the trunk genes Ultrabithorax, abdominal-a, and Abdominal-B are also expressed in extended overlapping domains in insects, chelicerates, and vertebrates suggests that the insect head genes have undergone evolution to limit their expression domains. Unfortunately, in the Mandibulata, which includes the Insecta, Crustacea, and Myriapoda, no data exist for Hox head gene expression patterns for any group other than Insecta. We have chosen the crustacean Porcellio scaber, order Isopoda, as a noninsect model organism to study the expression patterns of the head Hox genes. It is important to note that our model organism belongs to the subclass Malacostraca (higher crustaceans) and is as derived as insects are in its body plan and tagmatization, relative to phylogenetically more basal groups. Moreover, the interpretations of the expression patterns reported here are based on the assumptions that: (i) the Insecta is monophyletic, with the order Thysanura a basal group; (ii) the Mandibulata are monophyletic with the Crustacea, a sister group of the Insecta; and (iii) the Chelicerata is an outgroup to the Insectan Crustacean clade (based on refs ; reviewed in refs. 18 and 19). The six-segmented mandibulate head is believed to be a primitive character shared by all three extant mandibulate groups (20 22). This feature allows reliable identification of homologous segments and appendages, and it allows predictions regarding expression patterns and possible developmental functions of the Hox genes. Based on the available insect and chelicerate expression pattern data, we proposed two conflicting predictions: (i) because malacostracan crustaceans and insects are related mandibulate groups with morphologically similar head structures, their Hox expression patterns should be similar, if not identical; or (ii) the crustacean patterns could be intermediate between those seen in chelicerates and in insects (6 8). Whereas much is known about the Abbreviations: a1, first antennal (segment); a2, second antennal (segment); EN, engrailed; Hox, homeotic complex (genes); mx1, first maxillary (segment); Ps, P. scaber. Data deposition: The sequences reported in this paper have been deposited in the GenBank database [accession nos. AF (P. scaber lab), AF (P. scaber pb), and AF (P. scaber Dfd)]. *To whom reprint requests should be addressed. kaufman@ bio.indiana.edu
2 Evolution: Abzhanov and Kaufman Proc. Natl. Acad. Sci. USA 96 (1999) expression patterns for the trunk genes of the crustacean A. franciscana (4), no expression patterns of the head genes are known for any crustacean group, although partial sequences have been published (5, 23). Here, we describe the expression patterns of lab, pb, and Dfd in the crustacean P. scaber. Our observations show that neither of the two predictions presented above is borne out. Rather, a combination of these results and our previously published data on the expression pattern of the head Hox gene Sex combs reduced (Scr) (24) demonstrates that the expression patterns of these genes in this crustacean are divergent from those seen in insects. Moreover, the observed crustacean pattern does not appear to represent a chelicerate insect intermediate. We conclude that the divergent crustacean insectan expression patterns reveal that unexpectedly dissimilar developmental processes likely underlie the specification of homologous and morphologically similar mandibulate mouthparts. MATERIALS AND METHODS Cloning of the cdna Fragments and Sequence Analysis. Total mrna was isolated from two broods (about 50 embryos) of P. scaber embryos by using the TRIzol reagent (Life Technologies) and following the manufacturer s instructions. The cdna was screened by PCR with degenerate primers designed against conserved amino acid regions of the head Hox protein (see Fig. 2). Multiple independent clones produced only repeat copies of the same sequence for the lab, pb, and Dfd genes. All primer pairs and PCR protocols have been previously described (6, 10, 11). A related PCR screen for homeoboxes that used degenerate ELEKEF- and WFQNRRencoding primers produced single unique copies of the lab, pb, and Dfd homeoboxes matching those cloned with the more specific primers. A total of 39 and 27 homeobox fragments have been cloned from the cdna pool and from genomic DNA, respectively. For example, the Hom-C fragments cloned from cdna include 12 Ubx, 9pb, 3 Scr, 3 Abd-A, and 5 lab homeobox segments with nucleotide sequences identical to the previously cloned orthologous copies of these genes. A short cdna fragment of the Hox3 class homologue was also cloned and will be described elsewhere. The sequences were compared with our homeobox sequences from miscellaneous arthropods (unpublished data), cloned in the laboratory, and with those available in the National Center for Biotechnology Information database. Whole-Mount in Situ Hybridization, Microscopy, and Photography. In situ hybridization was performed as previously described by Panganiban et al. (26), but with modifications (10), and with the exception that protease K treatment was min instead of 1 hr. The size of the in situ probes ranged from 230 bp (pb) to 260 bp (lab). Probes of similar size have been used successfully to reveal specific expression domains of a number of arthropod genes (4, 6, 8, 10, 11, 24). All procedures for mounting and photographing embryos have been described (10, 27). RESULTS Development of the P. scaber Head as Revealed by Antibody to Engrailed (EN) Protein. To assign the P. scaber head appendages to specific segments (Fig. 1A), we used the monoclonal antibody Mab4D9, which recognizes EN, to indicate the posterior of the segmental borders (28, 29). The antibody revealed six segments in the embryonic head of P. scaber: ocular, first antennal, second antennal, mandibular, first maxillary, and second maxillary (Fig. 1B). The most anterior region of the head, the labrum, develops as a pair of small appendage-like structures that fuse medially at about the 65 70% stage of development. In early embryos, the EN stripe of the ocular segment is not complete and is interrupted on the FIG. 1. Head appendages and segments of the crustacean P. scaber, ventral view, with anterior at the top. (A) A scanning electron micrograph of a developing (45 50%) embryo ( 50.) The segments indicated are as follows: a1, first antennae; a2, second antennae; lb, labrum; mn, mandibles; pg, paragnaths; mx1, first maxillae; mx2, second maxillae; T1 mxp, first thoracic appendages maxillipeds (B) The head of the embryo stained with Mab4D9 anti-en antibody. The segments indicated are as follows: oc lb, ocular labral; a1, first antennal; a2, second antennal; mn, mandibular; mx1, first maxillary; mx2, second maxillary; T1, first thoracic segments. ventral side by the labrum. The labrum itself does not express EN and, in this respect, appears to be continuous with the stomodeum. The first antennal segment bears a pair of small uniramose antennae, which are reduced in the adult. The second antennae are the largest pair of the appendages on the head. The stomodeal opening protrudes at the level of the posterior first antennal (a1) segment and extends to the posterior of the second antennal (a2) segment, which results in a ventral interruption of the EN stripes of the a1 and a2 segments. The broad mandibular EN band is seen in the developing posterior mandibular appendages. EN is expressed similarly in the first and second maxillary segments. The Orthologues of the lab, pb, and Dfd Genes. Fig. 2 shows an alignment of the predicted partial amino acid sequences for the recovered P. scaber genes lab, pb, and Dfd and their homologues from several insect orders and from A. franciscana. Because of high levels of Porcellio Hox sequence conservation, each cdna can be unambiguously assigned to its specific HOM-C gene class. Analysis of the alignment in Fig. 2 reveals that the lab homologues from Drosophila melanogaster, Oncopeltus fasciatus, Thermobia domestica, and P. scaber are highly similar immediately downstream of the YKWM motif and in most of the homeobox; however, the N-terminal arm of the homeodomain and the variable region just upstream are not conserved among the orthologues. All pb homologues share essentially identical homeobox and N- terminal arm regions (see Fig. 2). Porcellio has the largest variable region of all the sequences shown. Porcellio Dfd is very similar to its homologues in insects and A. franciscana. Its variable region appears to be more similar to that of insects than to that of Artemia. Hox Gene Expression Patterns. The expression patterns of lab, pb, and Dfd were obtained by using whole mount in situ hybridization to reveal transcript distribution. Expression can be detected from the 30% to the 80% stage but is best seen at the 50 60% stage. All embryos shown in Fig. 3 are at the 50 60% developmental stage. At this stage the embryo begins dorsal closure and its appendages develop unique morphologies. Additionally, the first pair of the thoracic appendages begins to transform into the mouthpart maxillipeds (24). Expression of P. scaber lab is clear in the developing a2 segment and its appendages (Fig. 3A). The embryo in Fig. 3A shows no expression in the first antennae, mandibles, or any
3 10226 Evolution: Abzhanov and Kaufman Proc. Natl. Acad. Sci. USA 96 (1999) FIG. 2. Alignments of the deduced head Hox protein sequences. Hexapeptide variable region and homeodomain sequences are aligned for four insect species and the crustaceans A. franciscana and P. scaber (some sequences were not available). All sequences are compared with their Drosophila counterparts. Dashes indicate sequence identity, breaches in the sequences indicate introduced gaps. Species are indicated in the first column: Dm, Drosophila melanogaster; Of, Oncopeltus fasciatus; Ad, Acheta domestica; Td, Thermobia domestica; Ps, P. scaber; Af, A. franciscana. Gene names are indicated in the second column: LAB, labial; PB, proboscipedia; DFD, Deformed. other mouthparts. Closer examination reveals that P. scaber lab expression is continuous within the a2 segment and circles the stomodeum (Fig. 3A). Very strong expression just anterior to the stomodeum and in the posterior part of the labrum either suggests that the anterior boundary is ventrally parasegmental or that parts of the aforementioned organs originate from the a2 segment. Fig. 4A shows the P. scaber lab expression domain as a colored overlay on a scanning electron micrograph. The expression domain of P. scaber pb appears as a narrow stripe in the posterior a2 segment; it is reminiscent of the ventral portion of the EN stripe in that segment (Fig. 1 and Fig. 3C). Like the EN stripe, the P. scaber pb stripe is interrupted medially by the stomodeum. Careful examination of the embryonic head reveals additional strong expression in a small group of cells in the posterior-lateral portion of the a2 appendages (Fig. 3D). These antennal cells form a small bud-like structure, possibly representing a rudimentary antennal exopod (Fig. 3D). The P. scaber pb stripe does not extend into the remainder of the a2 appendage and is restricted to the ventrolateral portion of the a2 segment. Based on the segment morphology and direct comparison to P. scaber lab stained embryos, we conclude that P. scaber pb expression in the a2 segment overlaps that of P. scaber lab. The exact extent of the overlap between the domains of the two genes is difficult to ascertain because of the lack of good morphological markers in the posterior a2 segment. Embryos that were stained for several more hours showed additional weak expression in the paragnaths, ventral mouthpart structures associated with the stomodeum, but this expression appears to be mesodermal (Fig. 3C). No other expression domains can be seen, even in overstained embryos. A summary of the pb expression domains is shown in Fig. 4B. In embryos of all stages that were examined, the P. scaber Dfd homologue is expressed in the developing paragnaths (Fig. 1; and Fig. 3 E and F; see Discussion). Weak expression in the mandibles is mesodermal. There is also P. scaber Dfd expression in the ventral portion of the mandibular segment, which may be parasegmental; it extends from the mid-mandibular segment into the anterior half of the first maxillary (mx1) (Fig. 3E). A much weaker expression domain in the mesoderm can be recognized in the mandibular appendages (Fig. 3E). There is not the detectable accumulation in the mx1 appendages or posterior half of the mx1 segment that one might have predicted based on the expression of Dfd in the homologous maxillary segment of insects (6). These data are summarized in Fig. 4C. The pattern of P. scaber Scr expression has been described (24). We note here only that colinearity of expression is exhibited by this gene. The anterior border of Scr accumulation is in the posterior of the mx1 segment, whereas the posterior border of Dfd resides in the anterior of this same segment. It is possible that there is some overlap of expression in the middle of mx1 but a demonstration of this point awaits further experimentation. DISCUSSION Two comparative analyses of insect and chelicerate Hox expression patterns have been published recently (7, 8). These analyses are intriguing, but they point to the necessity for studies on crustacean and myriapod Hox head genes to gain a better understanding of their evolution in arthropods (7, 8). To partially fill this need, we have determined the embryonic expression patterns for the genes lab, pb, and Dfd in P. scaber, a malacostracan crustacean, and compared our results with data from insects and chelicerates. In general, we found the P. scaber expression patterns to be well-defined and discrete, but not identical to those of insects or chelicerates (Fig. 5). Comparison of Insect and Crustacean Hox Expression Domains. lab expression in the Drosophila head is in the intercalary segment, a small metamere that is devoid of appendages, located posterior to the antennal segment and anterior to the mandibular segment. The exact role of lab in this segment is unclear, because mutants do not show an obvious homeotic transformation. Nevertheless, the gene is important for the formation of the embryonic and adult head in Drosophila, because mutants do show defects in the development of cephalic structures (for review, see ref. 30). lab expression in the intercalary segment is conserved in all of the insect species examined thus far (for review, see ref. 6). Comparison of the P. scaber lab expression domain to that of insects reveals both conservation and change. P. scaber expression is restricted to the first postantennular metamere, i.e., the a2 segment, which, based on morphological and molecular (EN expression) data, is thought to be homologous to the intercalary (22, 29 32). Moreover, innervation of this first postantennular segment is associated with the tritocerebral ganglion of the brain in all mandibulates (31). The fact that lab is expressed in a homologous segment in insects and crustaceans suggests that it was recruited for and might be involved in conserved and homologous developmental processes. However, in Porcellio, lab is expressed in the a2 appendages, whereas the adult insect intercalary segment is limbless. Interestingly, the embryos of some insects develop small transitory appendages on this segment, which might be atavisms to a more primitive state (34). It is not clear, however,
4 Evolution: Abzhanov and Kaufman Proc. Natl. Acad. Sci. USA 96 (1999) FIG. 4. Summary of Hox expression domains in P. scaber. The main domains of expression in the ectoderm are shown in red: (A) lab is expressed in the second antennal segment and its appendages. (B) pb is expressed in the posterior second antennal segment. (C) Expression of Dfd is limited to the ventral mandibular segment and paragnaths. (D) Scr mrna distribution in the first maxillary appendages, second maxillary segment, and appendages and distal half of T1 leg (24). FIG. 3. Embryonic expression patterns of the P. scaber homologues of the Drosophila genes lab, pb, and Dfd, as revealed by in situ hybridization. Abbreviations as in Fig. 1. Ventral view of embryos with anterior at the top in all panels. (A and B) 50 60% Stage embryos showing the lab expression pattern; whole embryo (A) and close-up of embryonic head (B). (C and D) 50 60% Stage embryos showing pb expression in close-up views of the embryonic head revealing details of the expression domain. Arrow points to the pb expression in the posterior-lateral second antennae (D). (E and F) 50 60% Stage embryos showing Dfd mrna distribution with dissected embryonic head (F) showing strong Dfd expression in the paragnaths. (A and E, 50; B, C, D, and F, 100.) whether lab was directly involved in appendage loss in the insects; mutations in this gene do not cause limb growth from the intercalary (30). Nevertheless, it is possible that in crustaceans, lab contributes to the unique morphology of the second as compared with the first pair of antennae. For example, in P. scaber, the first antennae are greatly reduced in size, whereas the second antennae are large and more leg-like. The pb gene is located upstream of lab in the HOM-C and is expressed in insects in the appendages of the maxillary and labial segments, where it has been shown to specify the posterior mouthparts in Drosophila and Tribolium castaneum (3, 35). Oddly, the main expression domain of pb in Drosophila is not colinear with that of lab and Dfd. In all of the insects surveyed, the anterior boundary of Dfd, the next upstream HOM-C gene relative to pb, is in the mandibular segment (10), and, based on the position of pb in the complex, one might expect its anterior border of expression to be at or anterior to this point. To rationalize this fact, it has been suggested that an ancestral insect pb gene lost its colinear expression pattern and gained a new, appendage-specific role in the maxillae and labium (35). Studies of the expression patterns of pb in several insect orders revealed another, albeit weaker, expression domain in the ventral portion of the intercalary (6). The significance of this domain is not clear, and in some groups, including Drosophila, it was found to be mesodermal. In the apterygote insect T. domestica, there is epidermal pb expression in the intercalary, but it is weak, appears late, and is transient (6, 35). These latter, more anterior patches of expression may be remnants of the posited ancestral, more extensive expression domain. The pb expression domain of P. scaber was found to be quite dissimilar from the insect pattern. In this crustacean, pb accumulation is restricted to the posterior part of the a2 segment and includes neither the mx1 nor the mx2 segments or appendages (Figs. 3A and 4B). The first and second maxillae of crustaceans are homologous to the maxillae and labium of insects, respectively (30). As pb is required for maxillary and labial appendage development in insects, it clearly cannot be performing a similar function in the P. scaber embryo. At this point, it is difficult to discern a possible developmental function for pb in P. scaber embryos.
5 10228 Evolution: Abzhanov and Kaufman Proc. Natl. Acad. Sci. USA 96 (1999) FIG. 5. (A) Diagram of the Hox gene expression pattern boundaries in the Crustacea (P. scaber), Insecta, and Chelicerata (refs. 6 8; unpublished data). Detailed expression pattern of the Scr homologue of P. scaber has been described elsewhere (24). Columns represent homologous segments. Parasegments and mandibulate segments (as in Crustacea) are labeled at the top of the diagram and chelicerate segments are labeled at the bottom. Bars indicate Hox gene expression patterns. Black bars indicate primary domains (strong expression) and gray bars indicate secondary domains (weak, transient, late, or variable expression). (B) Model for evolution of the homeotic gene pb expression pattern. Schematic head segments and structures are shown for insects, P. scaber, and a hypothetical mandibulate ancestor. Segmental expression of pb homologues is shown in dark gray. The segments are labeled according to the accepted conventions in the respective taxa. Abbreviations as in Figs. 1 4, except that in the schematic Insecta head, ic marks the intercalary segment and lab indicates the labial segment. In D. melanogaster, the expression domain of Dfd includes the mandibular and maxillary segments and appendages. It has been shown genetically to be required for the normal development of both mandibles and maxillae in Drosophila (30). As noted above, Dfd expression has been found to be very similar in all insect groups studied, including the basal insect T. domestica (10, 11), and the mandibles and maxillae of insects are homologous to the mandibles and first maxillae (maxillulae) of crustaceans (31). Comparison of the Dfd expression domains in P. scaber and insects reveals that the crustacean domain is smaller (Figs. 3E and 4C). P. scaber Dfd is expressed strongly in the paragnaths and the mandibular segment, but not the mandibular appendages. The paragnaths are associated with the stomodeum, but their exact embryonic origin is obscure. Some authors have concluded that these structures are sternal protrusions of the mandibular segment associated with the mouth, reduced appendages associated with the mandibles, or even structures homologous to the insect hypopharynx (31, 37, 38). Whatever their allegiance, paragnaths are found in a diversity of crustaceans (37). Thus, P. scaber Dfd is not expressed in the mandibles or maxillae (only mesodermal expression is detected in mandibles), where Dfd function is required in insects, suggesting that Dfd has a different developmental function in the crustacean head. It is not known which, if any, selector gene is expressed in the ectoderm of the crustacean mandibles proper. Comparison with Chelicerate Hox Gene Expression Domains. A comparison of the crustacean insect and chelicerate patterns of Hox gene expression is made difficult by the uncertainty of segmental homologies between the two groups. The more traditional view, based on anatomy and patterns of innervation, concludes that the segment associated with the deuterocerebral ganglion of the central nervous system is greatly reduced or absent in modern chelicerates (31). If this conclusion is correct, the more anterior ocular protocerebral segment would be homologous to the same segment in insects and crustaceans, whereas the next posterior segment in the spider would correspond to the intercalary tritocerebral segment of insects and to the second antennal segment of crustaceans. Thus, the homologue of the insect antennal and the crustacean first antennal segment would be absent in chelicerates (31). More recently, this question of head segment homology has been revisited by using the patterns of Hox gene expression as a basis for determining the presence or absence of the deuterocerebral segment in chelicerates (7, 8). These authors, using the spider Cupiennius salei, the mite Archegozetes longisetosus, and several head Hox genes as probes, have concluded that this segment is present (7, 8). For the purposes of this discussion we will take this point of view. However, we must stress that the use of only the expression patterns of the Hox genes as a primary reference for anatomical homology is inappropriate. Indeed, we show in this work that boundaries of expression patterns can and do change during evolution, and only some general tendencies, such as colinearity, persist. Nonetheless, in chelicerates, lab is expressed anteriorly in the developing pedipalps (homologous to the appendages of the intercalary segment). This expression extends posteriorly to the fourth pair of walking limbs, which would correspond to the first pair of the thoracic appendages in insects and crustaceans (maxillipeds in P. scaber) (7, 8). Therefore, the anterior boundary of lab expression appears to be conserved among chelicerates, crustaceans, and insects. It is notable, however, that the posterior limit of expression is not conserved. The integumentary expression of lab is limited to the intercalary or second antennal segment in insects and crustaceans, respectively, whereas in chelicerates, expression extends posteriorly a further four full segments (Fig. 5A). In A. longisetosus, pb is expressed in a broad domain from the pedipalps to the third pair of walking legs, where it is accumulated in the appendages (7). This boundary is colinear with both lab and Dfd, and is thus similar to the relative expression domains of these genes in vertebrates, annelids, and Porcellio (39 41); but it is dissimilar to that seen in insects (6). Thus, the anterior boundary of pb expression in the a2 segment of P. scaber appears to resemble that seen in chelicerates rather than that in insects. However, it should also be noted that, as for lab, the expression domain of chelicerate pb extends further posteriorly than that in insects and crustaceans (Fig. 5A). Dfd expression has been examined in both C. salei and A. longisetosus. In C. salei, Dfd is expressed in all walking legs (7). The mite shows a similar expression domain in the L1 L4 legs, with additional accumulation covering all opistosomal (abdominal) segments except the most terminal ones (8). The anterior boundary of Dfd accumulation appears to be conserved and located in the mandibular (insects) and homologous L1 (chelicerates) segments (Fig. 5A). If one assigns the paragnaths to the mandibular segment, the anterior boundary of Dfd expression then appears to be similar, albeit not identical, in P. scaber vis-a-vis insects and chelicerates. How-
6 Evolution: Abzhanov and Kaufman Proc. Natl. Acad. Sci. USA 96 (1999) ever, both the chelicerate and insect Dfd domains are clearly broader than those seen in the crustacean. Hox Genes and the Evolution of Mandibulate Head Structures. As noted above, the extended and broadly overlapping expression domains in chelicerates are reminiscent of those in vertebrates and are probably closer to an ancestral state. In contrast, the expression domains in insects and crustaceans are more resolved and segment-specific (Fig. 4). Based on morphological and recent molecular evidence, the Crustacea belongs to the monophyletic group Mandibulata, which is a close sister group to the Insecta (13 19). Thus, crustaceans represent an ideal case for study of the evolution of the homologous head Hox gene expression patterns and possible functions in the homologous structures of insects. Comparison of the expression patterns of the crustacean and insect Hox genes demonstrates that there is conservation of segment affinity (e.g., lab) and spatial colinearity (e.g., lab, pb, Dfd, and Scr) of expression (Figs. 4 and 5A; and ref. 24). In addition, the anterior boundaries of the lab and Dfd genes appear to be conserved in insects, crustaceans, and chelicerates. However, there is also divergence of the observed expression domains (e.g., pb and Dfd) (Figs. 4 and 5A). Consequently, substantial variation in the deployment of the Hox genes, and presumably in the developmental processes regulated by them, can be seen in homologous and morphologically similar crustacean and insect head structures. Genes involved in the development of mandibles and posterior mouthparts in insects are expressed in novel, though still colinear domains. For example, in insects, the maxillary and labial mouthparts express pb, whereas in P. scaber, the homologous appendages both express and probably depend on Scr, a different head homeotic gene (refs. 6, 11, 24; also see Fig. 4). We hypothesize that the mandibulate head evolved prior to the establishment of the defined head Hox gene expression domains, which have been recruited to their current regions and developmental functions independently in crustaceans and insects (Fig. 5B). This model involves an intermediate hypothetical mandibulate ancestor that did not have segmentspecific expression domains and probably resembled the pattern of expression seen in modern chelicerates. The specification of individual segments and mouthparts in such an animal would depend on the redundant and or fractional functions of multiple Hox genes, and would be facilitated by the subsequent evolution of more distinct expression domains (Figs. 4 and 5). That is, the head Hox genes functioned in a manner analogous to the genes of the D. melanogaster Bithorax complex. To test this model and to better understand the evolution of the Hox genes and head structures, further studies across different crustacean and myriapod groups will be required. We thank R. Turner for the scanning electron microscopy; M. Peterson and A. Popadic for providing primers and protocols; Thierry Rigaud for identifying the woodlouse species and culture suggestions; N. Patel for the EN INV antibody; Dee Verostko for administrative assistance; and G. Scholtz, Kevin Cook, S. Glueck, and T. Powers for valuable comments on the manuscript. This work was supported by the Howard Hughes Medical Institute. T.C.K. is an Investigator of the Howard Hughes Medical Institute. 1. Carroll, S. B. (1995) Nature (London) 376, Warren, R. & Carroll, S. B. (1995) Curr. Opin. Genet. Dev. 5, Beeman, R. W., Stuart, J. J., Brown, S. J. & Denell, R. E. (1993) BioEssays 15, Averof, M. & Akam, M. (1995) Nature (London) 376, Akam, M. (1995) Philos. Trans. R. Soc. Lond. B 349, Rogers, B. T. & Kaufman, T. C. (1997) Int. Rev. Cytol. 174, Damen, W. G. M., Hausdorf, M., Seyfarth, E-A. & Tautz, D. (1998) Proc. Natl. Acad. Sci. USA 95, Telford, M. J. & Thomas, R. H. (1998) Proc. Natl. Acad. Sci. USA 95, Kelsh, R., Weinzierl, R. O. J., White, R. A. H. & Akam, M. (1994) Dev. Genet. 15, Rogers, B. T., Peterson, M. D. & Kaufman, T. C. (1997) Development (Cambridge, U.K.) 124, Peterson, M. D., Rogers, B. T., Popadic, A. & Kaufman, T. C. (1999) Dev. Genes. Evol. 209, Manak, J. R. & Scott, M. P. (1994) Development (Cambridge, U.K.) Suppl. 1994, Friedrich, M. & Tautz, D. (1995) Nature (London) 376, Osorio, D., Averof, M. & Bacon, J. P. (1995) TREE 10, Ballard, J. W., Olsen, G. J., Faith, D. P., Odgers, W. A., Rowell, D. M. & Atkinson, P. W. (1992) Science 258, Boore, J. L., Collins, T. M., Stanton, D., Daehler, L. L. & Brown, W. M. (1995) Nature (London) 376, Boore, J. L., Lavrov, D. V. & Brown, W. M. (1998) Nature (London) 392, Gilbert, S. F. & Raunio, A. M. (1997) Embryology: Constructing the Organism (Sinauer, Sunderland, MA). 19. Zrzavy, J., Hypsa, V. & Vlaskova, M. (1997) in Arthropod Relationships, eds. Fortey, R. A. & Thomas, R. H. (Chapman & Hall, London), pp ). 20. Nielsen, C. (1989) Animal Evolution: Interrelationships of the Living Phyla (Oxford Univ. Press, Oxford). 21. Telford, M. J. & Thomas, R. H. (1995) Nature (London) 376, Scholtz, G. (1997) in Arthropod Relationships, eds. Fortey, R. A. & Thomas, R. H. (Chapman & Hall, London), pp Mouchel-Vielh, E., Rigolot, C., Gibert, J-M. & Deutsch, J. S. (1998) Mol. Phylogenet. Evol. 9, Abzhanov, A. & Kaufman, T. C. (1999) Development (Cambridge, U.K.) 126, Whitington, P. M., Leach, D. & Sandeman, R. (1993) Development (Cambridge, U.K.) 118, Panganiban, G., Nagy, L. & Carroll, S. (1994) Curr. Biol. 4, Gorman, M. J. & Kaufman, T. C. (1995) Genetics 140, Patel, N. H., Martin-Blanco, E., Coleman, K. G., Pole, S. J., Ellis, M. C., Kornberg, T. B. & Goodman, C. S. (1989) Cell 58, Scholtz, G. (1995) Zoology 98, Kaufman, T. C., Seeger, M. A. & Olsen, G. (1990) Adv. Genet. 27, Beklemishev, W. N. (1964) Principles of the Comparative Anatomy of Invertebrates (Univ. of Chicago Press, Chicago), pp , Schmidt-Ott, U. & Technau, G. M. (1992) Development (Cambridge, U.K.) 116, Rogers, B. T. & Kaufman, T. C. (1996) Development (Cambridge, U.K.) 122, Tamarelle, M. (1984) Int. J. Insect Morphol. Embryol. 13, Denell, R. E., Brown, S. J. & Beeman, R. W. (1996) Semin. Cell. Dev. Biol. 7, Peterson, M. D. (1998) Ph.D. thesis (Indiana Univ., Bloomington). 37. Kaestner, A. (1970) Invertebrate Zoology (Interscience, New York), Vol Schram, F. R. (1986) Crustacea (Oxford Univ. Press, Oxford). 39. Hanken, J. (1993) Am. Zool. 33, Keynes, R. (1994) Annu. Rev. Neurosci. 17, Matthew, J. K., Master, V. A., Lokhorst, D. K., Nardelli- Haefliger, D., Wedeen, C. J., Martindale, M. Q. & Shankland, M. (1997) Dev. Biology 190,
Novel regulation of the homeotic gene Scr associated with a crustacean leg-to-maxilliped appendage transformation.
 Novel regulation of the homeotic gene Scr associated with a crustacean leg-to-maxilliped appendage transformation. The Harvard community has made this article openly available. Please share how this access
Novel regulation of the homeotic gene Scr associated with a crustacean leg-to-maxilliped appendage transformation. The Harvard community has made this article openly available. Please share how this access
Homeotic genes in flies. Sem 9.3.B.6 Animal Science
 Homeotic genes in flies Sem 9.3.B.6 Animal Science So far We have seen that identities of each segment is determined by various regulators of segment polarity genes In arthopods, and in flies, each segment
Homeotic genes in flies Sem 9.3.B.6 Animal Science So far We have seen that identities of each segment is determined by various regulators of segment polarity genes In arthopods, and in flies, each segment
Cap n collar differentiates the mandible from the maxilla in the beetle Tribolium castaneum
 Coulcher and Telford EvoDevo 2012, 3:25 RESEARCH Open Access Cap n collar differentiates the mandible from the maxilla in the beetle Tribolium castaneum Joshua F Coulcher and Maximilian J Telford * Abstract
Coulcher and Telford EvoDevo 2012, 3:25 RESEARCH Open Access Cap n collar differentiates the mandible from the maxilla in the beetle Tribolium castaneum Joshua F Coulcher and Maximilian J Telford * Abstract
Understanding the genetic basis of morphological evolution: the role of homeotic genes in the diversification of the arthropod bauplan
 Int. J. Dev. Biol. 42: 453-461 (1998) Understanding the genetic basis of morphological evolution: the role of homeotic genes in the diversification of the arthropod bauplan ALEKSANDAR POPADIC, ARHAT ABZHANOV,
Int. J. Dev. Biol. 42: 453-461 (1998) Understanding the genetic basis of morphological evolution: the role of homeotic genes in the diversification of the arthropod bauplan ALEKSANDAR POPADIC, ARHAT ABZHANOV,
Analysis of nubbin expression patterns in insects
 EVOLUTION & DEVELOPMENT 6:5, 310 324 (2004) Analysis of nubbin expression patterns in insects Hua Li and Aleksandar Popadić Department of Biological Sciences, Wayne State University, Detroit, MI 48202,
EVOLUTION & DEVELOPMENT 6:5, 310 324 (2004) Analysis of nubbin expression patterns in insects Hua Li and Aleksandar Popadić Department of Biological Sciences, Wayne State University, Detroit, MI 48202,
Exploring the myriapod body plan: expression patterns of the ten Hox genes in a centipede
 Development 129, 1225-1238 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV5988 1225 Exploring the myriapod body plan: expression patterns of the ten Hox genes in a centipede
Development 129, 1225-1238 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV5988 1225 Exploring the myriapod body plan: expression patterns of the ten Hox genes in a centipede
MOLECULAR CONTROL OF EMBRYONIC PATTERN FORMATION
 MOLECULAR CONTROL OF EMBRYONIC PATTERN FORMATION Drosophila is the best understood of all developmental systems, especially at the genetic level, and although it is an invertebrate it has had an enormous
MOLECULAR CONTROL OF EMBRYONIC PATTERN FORMATION Drosophila is the best understood of all developmental systems, especially at the genetic level, and although it is an invertebrate it has had an enormous
Homeotic Genes and Body Patterns
 Homeotic Genes and Body Patterns Every organism has a unique body pattern. Although specialized body structures, such as arms and legs, may be similar in makeup (both are made of muscle and bone), their
Homeotic Genes and Body Patterns Every organism has a unique body pattern. Although specialized body structures, such as arms and legs, may be similar in makeup (both are made of muscle and bone), their
Axis determination in flies. Sem 9.3.B.5 Animal Science
 Axis determination in flies Sem 9.3.B.5 Animal Science All embryos are in lateral view (anterior to the left). Endoderm, midgut; mesoderm; central nervous system; foregut, hindgut and pole cells in yellow.
Axis determination in flies Sem 9.3.B.5 Animal Science All embryos are in lateral view (anterior to the left). Endoderm, midgut; mesoderm; central nervous system; foregut, hindgut and pole cells in yellow.
From DNA to Diversity
 From DNA to Diversity Molecular Genetics and the Evolution of Animal Design Sean B. Carroll Jennifer K. Grenier Scott D. Weatherbee Howard Hughes Medical Institute and University of Wisconsin Madison,
From DNA to Diversity Molecular Genetics and the Evolution of Animal Design Sean B. Carroll Jennifer K. Grenier Scott D. Weatherbee Howard Hughes Medical Institute and University of Wisconsin Madison,
18.4 Embryonic development involves cell division, cell differentiation, and morphogenesis
 18.4 Embryonic development involves cell division, cell differentiation, and morphogenesis An organism arises from a fertilized egg cell as the result of three interrelated processes: cell division, cell
18.4 Embryonic development involves cell division, cell differentiation, and morphogenesis An organism arises from a fertilized egg cell as the result of three interrelated processes: cell division, cell
Evolution of Transcription factor function: Homeotic (Hox) proteins
 Evolution of Transcription factor function: Homeotic (Hox) proteins Hox proteins regulate morphology in cellular zones on the anterior-posterior axis of embryos via the activation/repression of unknown
Evolution of Transcription factor function: Homeotic (Hox) proteins Hox proteins regulate morphology in cellular zones on the anterior-posterior axis of embryos via the activation/repression of unknown
A pair-rule gene circuit defines segments sequentially in the short-germ insect Tribolium castaneum
 A pair-rule gene circuit defines segments sequentially in the short-germ insect Tribolium castaneum Chong Pyo Choe, Sherry C. Miller, and Susan J. Brown* Division of Biology, Kansas State University, Manhattan,
A pair-rule gene circuit defines segments sequentially in the short-germ insect Tribolium castaneum Chong Pyo Choe, Sherry C. Miller, and Susan J. Brown* Division of Biology, Kansas State University, Manhattan,
Chapter 18 Lecture. Concepts of Genetics. Tenth Edition. Developmental Genetics
 Chapter 18 Lecture Concepts of Genetics Tenth Edition Developmental Genetics Chapter Contents 18.1 Differentiated States Develop from Coordinated Programs of Gene Expression 18.2 Evolutionary Conservation
Chapter 18 Lecture Concepts of Genetics Tenth Edition Developmental Genetics Chapter Contents 18.1 Differentiated States Develop from Coordinated Programs of Gene Expression 18.2 Evolutionary Conservation
As a result of advancements in the past two decades, the
 Differential expression patterns of the hox gene are associated with differential growth of insect hind legs Najmus S. Mahfooz*, Hua Li*, and Aleksandar Popadić Department of Biological Sciences, Wayne
Differential expression patterns of the hox gene are associated with differential growth of insect hind legs Najmus S. Mahfooz*, Hua Li*, and Aleksandar Popadić Department of Biological Sciences, Wayne
Drosophila Life Cycle
 Drosophila Life Cycle 1 Early Drosophila Cleavage Nuclei migrate to periphery after 10 nuclear divisions. Cellularization occurs when plasma membrane folds in to divide nuclei into cells. Drosophila Superficial
Drosophila Life Cycle 1 Early Drosophila Cleavage Nuclei migrate to periphery after 10 nuclear divisions. Cellularization occurs when plasma membrane folds in to divide nuclei into cells. Drosophila Superficial
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila November 6, 2007 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Developmental Biology Biology 4361 Axis Specification in Drosophila November 6, 2007 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Midterm 1. Average score: 74.4 Median score: 77
 Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila November 2, 2006 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Developmental Biology Biology 4361 Axis Specification in Drosophila November 2, 2006 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
5/4/05 Biol 473 lecture
 5/4/05 Biol 473 lecture animals shown: anomalocaris and hallucigenia 1 The Cambrian Explosion - 550 MYA THE BIG BANG OF ANIMAL EVOLUTION Cambrian explosion was characterized by the sudden and roughly simultaneous
5/4/05 Biol 473 lecture animals shown: anomalocaris and hallucigenia 1 The Cambrian Explosion - 550 MYA THE BIG BANG OF ANIMAL EVOLUTION Cambrian explosion was characterized by the sudden and roughly simultaneous
Lecture 7. Development of the Fruit Fly Drosophila
 BIOLOGY 205/SECTION 7 DEVELOPMENT- LILJEGREN Lecture 7 Development of the Fruit Fly Drosophila 1. The fruit fly- a highly successful, specialized organism a. Quick life cycle includes three larval stages
BIOLOGY 205/SECTION 7 DEVELOPMENT- LILJEGREN Lecture 7 Development of the Fruit Fly Drosophila 1. The fruit fly- a highly successful, specialized organism a. Quick life cycle includes three larval stages
Analysis of the Wnt gene repertoire in an onychophoran provides new insights into the evolution of segmentation
 Hogvall et al. EvoDevo 2014, 5:14 RESEARCH Analysis of the Wnt gene repertoire in an onychophoran provides new insights into the evolution of segmentation Open Access Mattias Hogvall 1, Anna Schönauer
Hogvall et al. EvoDevo 2014, 5:14 RESEARCH Analysis of the Wnt gene repertoire in an onychophoran provides new insights into the evolution of segmentation Open Access Mattias Hogvall 1, Anna Schönauer
The analysis of arthropods and their relatives may present the best opportunity for an integrated approach to the
 Research Paper 547 Evolution of the entire arthropod Hox gene set predated the origin and radiation of the onychophoran/arthropod clade Jennifer K. Grenier*, Theodore L. Garber*, Robert Warren*, Paul M.
Research Paper 547 Evolution of the entire arthropod Hox gene set predated the origin and radiation of the onychophoran/arthropod clade Jennifer K. Grenier*, Theodore L. Garber*, Robert Warren*, Paul M.
SCIENTIFIC EVIDENCE TO SUPPORT THE THEORY OF EVOLUTION. Using Anatomy, Embryology, Biochemistry, and Paleontology
 SCIENTIFIC EVIDENCE TO SUPPORT THE THEORY OF EVOLUTION Using Anatomy, Embryology, Biochemistry, and Paleontology Scientific Fields Different fields of science have contributed evidence for the theory of
SCIENTIFIC EVIDENCE TO SUPPORT THE THEORY OF EVOLUTION Using Anatomy, Embryology, Biochemistry, and Paleontology Scientific Fields Different fields of science have contributed evidence for the theory of
Evolution and Development Evo-Devo
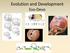 Evolution and Development Evo-Devo Darwin wrote a book on barnacles. Plate 1 (Lepas), from A monograph on the sub-class Cirripedia, by Charles Darwin. Comparative embryology There is an obvious similarity
Evolution and Development Evo-Devo Darwin wrote a book on barnacles. Plate 1 (Lepas), from A monograph on the sub-class Cirripedia, by Charles Darwin. Comparative embryology There is an obvious similarity
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila July 9, 2008 Drosophila Development Overview Fertilization Cleavage Gastrulation Drosophila body plan Oocyte formation Genetic control
Developmental Biology Biology 4361 Axis Specification in Drosophila July 9, 2008 Drosophila Development Overview Fertilization Cleavage Gastrulation Drosophila body plan Oocyte formation Genetic control
Metameric body plans are found in diverse metazoan phyla,
 Expression patterns of hairy, even-skipped, and runt in the spider Cupiennius salei imply that these genes were segmentation genes in a basal arthropod Wim G. M. Damen*, Mathias Weller, and Diethard Tautz
Expression patterns of hairy, even-skipped, and runt in the spider Cupiennius salei imply that these genes were segmentation genes in a basal arthropod Wim G. M. Damen*, Mathias Weller, and Diethard Tautz
Tissue- and stage-specific control of homeotic and segmentation gene expression in Drosophila embryos by the polyhomeotic gene
 Development 103, 733-741 (1988) Printed in Great Britain The Company of Biologists Limited 1988 733 Tissue- and stage-specific control of homeotic and segmentation gene expression in Drosophila embryos
Development 103, 733-741 (1988) Printed in Great Britain The Company of Biologists Limited 1988 733 Tissue- and stage-specific control of homeotic and segmentation gene expression in Drosophila embryos
RALF JANSSEN* Uppsala University, Department of Earth Sciences, Palaeobiology, Uppsala, Sweden
 Int. J. Dev. Biol. 58: 343-347 (2014) doi: 10.1387/ijdb.140058rj www.intjdevbiol.com Gene expression suggests double-segmental and single-segmental patterning mechanisms during posterior segment addition
Int. J. Dev. Biol. 58: 343-347 (2014) doi: 10.1387/ijdb.140058rj www.intjdevbiol.com Gene expression suggests double-segmental and single-segmental patterning mechanisms during posterior segment addition
8/23/2014. Introduction to Animal Diversity
 Introduction to Animal Diversity Chapter 32 Objectives List the characteristics that combine to define animals Summarize key events of the Paleozoic, Mesozoic, and Cenozoic eras Distinguish between the
Introduction to Animal Diversity Chapter 32 Objectives List the characteristics that combine to define animals Summarize key events of the Paleozoic, Mesozoic, and Cenozoic eras Distinguish between the
Arthropod Hox genes: insights on the evolutionary forces that shape gene functions Michalis Averof
 386 Arthropod Hox genes: insights on the evolutionary forces that shape gene functions Michalis Averof Comparative studies suggest that gene duplication, changes in cis-regulatory elements and changes
386 Arthropod Hox genes: insights on the evolutionary forces that shape gene functions Michalis Averof Comparative studies suggest that gene duplication, changes in cis-regulatory elements and changes
Evolving role of Antennapedia protein in arthropod limb patterning
 Development 129, 3555-3561 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV7965 3555 Evolving role of Antennapedia protein in arthropod limb patterning Yasuhiro Shiga 1, *, Ryusuke
Development 129, 3555-3561 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV7965 3555 Evolving role of Antennapedia protein in arthropod limb patterning Yasuhiro Shiga 1, *, Ryusuke
Why Flies? stages of embryogenesis. The Fly in History
 The Fly in History 1859 Darwin 1866 Mendel c. 1890 Driesch, Roux (experimental embryology) 1900 rediscovery of Mendel (birth of genetics) 1910 first mutant (white) (Morgan) 1913 first genetic map (Sturtevant
The Fly in History 1859 Darwin 1866 Mendel c. 1890 Driesch, Roux (experimental embryology) 1900 rediscovery of Mendel (birth of genetics) 1910 first mutant (white) (Morgan) 1913 first genetic map (Sturtevant
Developmental genetics: finding the genes that regulate development
 Developmental Biology BY1101 P. Murphy Lecture 9 Developmental genetics: finding the genes that regulate development Introduction The application of genetic analysis and DNA technology to the study of
Developmental Biology BY1101 P. Murphy Lecture 9 Developmental genetics: finding the genes that regulate development Introduction The application of genetic analysis and DNA technology to the study of
Piecing It Together. 1) The envelope contains puzzle pieces for 5 vertebrate embryos in 3 different stages of
 Piecing It Together 1) The envelope contains puzzle pieces for 5 vertebrate embryos in 3 different stages of development. Lay out the pieces so that you have matched up each animal name card with its 3
Piecing It Together 1) The envelope contains puzzle pieces for 5 vertebrate embryos in 3 different stages of development. Lay out the pieces so that you have matched up each animal name card with its 3
Genomes and Their Evolution
 Chapter 21 Genomes and Their Evolution PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from
Chapter 21 Genomes and Their Evolution PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from
Development of Drosophila
 Development of Drosophila Hand-out CBT Chapter 2 Wolpert, 5 th edition March 2018 Introduction 6. Introduction Drosophila melanogaster, the fruit fly, is found in all warm countries. In cooler regions,
Development of Drosophila Hand-out CBT Chapter 2 Wolpert, 5 th edition March 2018 Introduction 6. Introduction Drosophila melanogaster, the fruit fly, is found in all warm countries. In cooler regions,
Diverse Adaptations of an Ancestral Gill: A Common Evolutionary Origin for Wings, Breathing Organs, and Spinnerets
 Current Biology, Vol. 12, 1711 1716, October 1, 2002, 2002 Elsevier Science Ltd. All rights reserved. PII S0960-9822(02)01126-0 Diverse Adaptations of an Ancestral Gill: A Common Evolutionary Origin for
Current Biology, Vol. 12, 1711 1716, October 1, 2002, 2002 Elsevier Science Ltd. All rights reserved. PII S0960-9822(02)01126-0 Diverse Adaptations of an Ancestral Gill: A Common Evolutionary Origin for
Commentary A review of the correlation of tergites, sternites, and leg pairs in diplopods Ralf Janssen, Nikola-Michael Prpic and WimGMDamen*
 Frontiers in Zoology BioMed Central Commentary A review of the correlation of tergites, sternites, and leg pairs in diplopods Ralf Janssen, Nikola-Michael Prpic and WimGMDamen* Open Access Address: Department
Frontiers in Zoology BioMed Central Commentary A review of the correlation of tergites, sternites, and leg pairs in diplopods Ralf Janssen, Nikola-Michael Prpic and WimGMDamen* Open Access Address: Department
2. Der Dissertation zugrunde liegende Publikationen und Manuskripte. 2.1 Fine scale mapping in the sex locus region of the honey bee (Apis mellifera)
 2. Der Dissertation zugrunde liegende Publikationen und Manuskripte 2.1 Fine scale mapping in the sex locus region of the honey bee (Apis mellifera) M. Hasselmann 1, M. K. Fondrk², R. E. Page Jr.² und
2. Der Dissertation zugrunde liegende Publikationen und Manuskripte 2.1 Fine scale mapping in the sex locus region of the honey bee (Apis mellifera) M. Hasselmann 1, M. K. Fondrk², R. E. Page Jr.² und
Expression of pair rule gene orthologs in the blastoderm of a myriapod: evidence for pair rule-like mechanisms?
 Janssen et al. BMC Developmental Biology 2012, 12:15 RESEARCH ARTICLE Expression of pair rule gene orthologs in the blastoderm of a myriapod: evidence for pair rule-like mechanisms? Ralf Janssen 1*, Wim
Janssen et al. BMC Developmental Biology 2012, 12:15 RESEARCH ARTICLE Expression of pair rule gene orthologs in the blastoderm of a myriapod: evidence for pair rule-like mechanisms? Ralf Janssen 1*, Wim
Warm-Up- Review Natural Selection and Reproduction for quiz today!!!! Notes on Evidence of Evolution Work on Vocabulary and Lab
 Date: Agenda Warm-Up- Review Natural Selection and Reproduction for quiz today!!!! Notes on Evidence of Evolution Work on Vocabulary and Lab Ask questions based on 5.1 and 5.2 Quiz on 5.1 and 5.2 How
Date: Agenda Warm-Up- Review Natural Selection and Reproduction for quiz today!!!! Notes on Evidence of Evolution Work on Vocabulary and Lab Ask questions based on 5.1 and 5.2 Quiz on 5.1 and 5.2 How
Comparative analysis of neurogenesis in the myriapod Glomeris marginata (Diplopoda) suggests more similarities to chelicerates than to insects
 Development 130, 2161-2171 2003 The Company of Biologists Ltd doi:10.1242/dev.00442 2161 Comparative analysis of neurogenesis in the myriapod Glomeris marginata (Diplopoda) suggests more similarities to
Development 130, 2161-2171 2003 The Company of Biologists Ltd doi:10.1242/dev.00442 2161 Comparative analysis of neurogenesis in the myriapod Glomeris marginata (Diplopoda) suggests more similarities to
Supporting Information
 Supporting Information Pueyo et al. 10.1073/pnas.0804093105 SI Text Periplaneta americana (Delta GenBank Accession Number FJ222590). MR- WTQQTRVQGAVVVVILAALQQVCCSGVFELRLKSF- INDYGKDSVGQCCSGTPSPGTKACSGPCRTRFRVCL-
Supporting Information Pueyo et al. 10.1073/pnas.0804093105 SI Text Periplaneta americana (Delta GenBank Accession Number FJ222590). MR- WTQQTRVQGAVVVVILAALQQVCCSGVFELRLKSF- INDYGKDSVGQCCSGTPSPGTKACSGPCRTRFRVCL-
Evolutionary Developmental Biology
 Evolutionary Developmental Biology a.k.a. EVO-DEVO Paedomorphosis is common among salamanders. Note how this hellbender (top) and mudpuppy (bottom) both have gills, paddle tails, and weaker limbs... Top:
Evolutionary Developmental Biology a.k.a. EVO-DEVO Paedomorphosis is common among salamanders. Note how this hellbender (top) and mudpuppy (bottom) both have gills, paddle tails, and weaker limbs... Top:
Workshop: The Evolution of Animalia body symmetry embryonic germ layers ontogenetic origins I. What is an Animal? II. Germ Layers
 Workshop: The Evolution of Animalia by Dana Krempels Perhaps even more than the other Eukarya, Animalia is characterized by a distinct progression of complexity in form and function as one moves from the
Workshop: The Evolution of Animalia by Dana Krempels Perhaps even more than the other Eukarya, Animalia is characterized by a distinct progression of complexity in form and function as one moves from the
Building the brain (1): Evolutionary insights
 Building the brain (1): Evolutionary insights Historical considerations! Initial insight into the general role of the brain in human behaviour was already attained in antiquity and formulated by Hippocrates
Building the brain (1): Evolutionary insights Historical considerations! Initial insight into the general role of the brain in human behaviour was already attained in antiquity and formulated by Hippocrates
v Scientists have identified 1.3 million living species of animals v The definition of an animal
 Biosc 41 9/10 Announcements BIOSC 041 v Genetics review: group problem sets Groups of 3-4 Correct answer presented to class = 2 pts extra credit Incorrect attempt = 1 pt extra credit v Lecture: Animal
Biosc 41 9/10 Announcements BIOSC 041 v Genetics review: group problem sets Groups of 3-4 Correct answer presented to class = 2 pts extra credit Incorrect attempt = 1 pt extra credit v Lecture: Animal
Posterior patterning genes and the identification of a unique body region in the brine shrimp Artemia franciscana
 Development First posted epress online online 15 October publication 2003 as date 10.1242/dev.00835 15 October 2003 Access the most recent version at http://dev.biologists.org/lookup/doi/10.1242/dev.00835
Development First posted epress online online 15 October publication 2003 as date 10.1242/dev.00835 15 October 2003 Access the most recent version at http://dev.biologists.org/lookup/doi/10.1242/dev.00835
Developmental Biology
 Developmental Biology 361 (2012) 427 438 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Evolution of Developmental Control
Developmental Biology 361 (2012) 427 438 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Evolution of Developmental Control
Expression of myriapod pair rule gene orthologs
 RESEARCH Open Access Expression of myriapod pair rule gene orthologs Ralf Janssen 1*, Graham E Budd 1, Nikola-Michael Prpic 2, Wim GM Damen 3 Abstract Background: Segmentation is a hallmark of the arthropods;
RESEARCH Open Access Expression of myriapod pair rule gene orthologs Ralf Janssen 1*, Graham E Budd 1, Nikola-Michael Prpic 2, Wim GM Damen 3 Abstract Background: Segmentation is a hallmark of the arthropods;
Gene expression analysis of potential morphogen signalling modifying factors in Panarthropoda
 https://doi.org/10.1186/s13227-018-0109-y EvoDevo RESEARCH Open Access Gene expression analysis of potential morphogen signalling modifying factors in Panarthropoda Mattias Hogvall, Graham E. Budd and
https://doi.org/10.1186/s13227-018-0109-y EvoDevo RESEARCH Open Access Gene expression analysis of potential morphogen signalling modifying factors in Panarthropoda Mattias Hogvall, Graham E. Budd and
Hox gene expression in the harvestman Phalangium opilio reveals divergent patterning of the chelicerate opisthosoma
 EVOLUTION & DEVELOPMENT 14:5, 450 463 (2012) DOI: 10.1111/j.1525-142X.2012.00565.x Hox gene expression in the harvestman Phalangium opilio reveals divergent patterning of the chelicerate opisthosoma Prashant
EVOLUTION & DEVELOPMENT 14:5, 450 463 (2012) DOI: 10.1111/j.1525-142X.2012.00565.x Hox gene expression in the harvestman Phalangium opilio reveals divergent patterning of the chelicerate opisthosoma Prashant
Expression of engrailed during segmentation in grasshopper and crayfish
 Development 107, 201-212 (1989) Printed in Great Britain The Company of Biologists Limited 201 Expression of engrailed during segmentation in grasshopper and crayfish NIPAM H. PATEL 1, THOMAS B. KORNBERG
Development 107, 201-212 (1989) Printed in Great Britain The Company of Biologists Limited 201 Expression of engrailed during segmentation in grasshopper and crayfish NIPAM H. PATEL 1, THOMAS B. KORNBERG
Developmental Biology Lecture Outlines
 Developmental Biology Lecture Outlines Lecture 01: Introduction Course content Developmental Biology Obsolete hypotheses Current theory Lecture 02: Gametogenesis Spermatozoa Spermatozoon function Spermatozoon
Developmental Biology Lecture Outlines Lecture 01: Introduction Course content Developmental Biology Obsolete hypotheses Current theory Lecture 02: Gametogenesis Spermatozoa Spermatozoon function Spermatozoon
Outline. v Definition and major characteristics of animals v Dividing animals into groups based on: v Animal Phylogeny
 BIOSC 041 Overview of Animal Diversity: Animal Body Plans Reference: Chapter 32 Outline v Definition and major characteristics of animals v Dividing animals into groups based on: Body symmetry Tissues
BIOSC 041 Overview of Animal Diversity: Animal Body Plans Reference: Chapter 32 Outline v Definition and major characteristics of animals v Dividing animals into groups based on: Body symmetry Tissues
2/23/09. Regional differentiation of mesoderm. Morphological changes at early postgastrulation. Segments organize the body plan during embryogenesis
 Regional differentiation of mesoderm Axial Paraxial Intermediate Somatic Splanchnic Chick embryo Morphological changes at early postgastrulation stages Segments organize the body plan during embryogenesis
Regional differentiation of mesoderm Axial Paraxial Intermediate Somatic Splanchnic Chick embryo Morphological changes at early postgastrulation stages Segments organize the body plan during embryogenesis
Development Team. Developmental Biology Axis Specification in Drosophila. Head, Department of Zoology, University of Delhi
 Paper No. : 11 Module : 6 Development Team Principal Investigator: Prof. Neeta Sehgal Head, Department of Zoology, University of Delhi Paper Coordinator: Prof. Namita Agrawal Department of Zoology, University
Paper No. : 11 Module : 6 Development Team Principal Investigator: Prof. Neeta Sehgal Head, Department of Zoology, University of Delhi Paper Coordinator: Prof. Namita Agrawal Department of Zoology, University
Drosophila Somatic Anterior-Posterior Axis (A-P Axis) Formation
 Home Biol 4241 Luria-Delbruck 1943 Hershey-Chase 1952 Meselson-Stahl 1958 Garapin et al. 1978 McClintock 1953 King-Wilson 1975 Sanger et al. 1977 Rothberg et al. 2011 Jeffreys et al. 1985 Bacterial Genetics
Home Biol 4241 Luria-Delbruck 1943 Hershey-Chase 1952 Meselson-Stahl 1958 Garapin et al. 1978 McClintock 1953 King-Wilson 1975 Sanger et al. 1977 Rothberg et al. 2011 Jeffreys et al. 1985 Bacterial Genetics
Developmental Biology 3230 Midterm Exam 1 March 2006
 Name Developmental Biology 3230 Midterm Exam 1 March 2006 1. (20pts) Regeneration occurs to some degree to most metazoans. When you remove the head of a hydra a new one regenerates. Graph the inhibitor
Name Developmental Biology 3230 Midterm Exam 1 March 2006 1. (20pts) Regeneration occurs to some degree to most metazoans. When you remove the head of a hydra a new one regenerates. Graph the inhibitor
Biosc 41 9/10 Announcements
 Biosc 41 9/10 Announcements v Genetics review: group problem sets Groups of 3-4 Correct answer presented to class = 2 pts extra credit Incorrect attempt = 1 pt extra credit v Lecture: Animal Body Plans
Biosc 41 9/10 Announcements v Genetics review: group problem sets Groups of 3-4 Correct answer presented to class = 2 pts extra credit Incorrect attempt = 1 pt extra credit v Lecture: Animal Body Plans
Arthropods (Arthropoda)
 Arthropods (Arthropoda) Davide Pisani Laboratory of Evolutionary Biology, Department of Biology, The National University of Ireland, Maynooth, Co. Kildare, Ireland (davide. pisani@nuim.ie) Abstract Living
Arthropods (Arthropoda) Davide Pisani Laboratory of Evolutionary Biology, Department of Biology, The National University of Ireland, Maynooth, Co. Kildare, Ireland (davide. pisani@nuim.ie) Abstract Living
Resolving arthropod relationships: Present and future insights from evo-devo studies
 Resolving arthropod relationships: Present and future insights from evo-devo studies STEVEN HRYCAJ & ALEKSANDAR POPADIC Department Of Biological Sciences, 5047 Gullen Mall, Wayne State University, Detroit,
Resolving arthropod relationships: Present and future insights from evo-devo studies STEVEN HRYCAJ & ALEKSANDAR POPADIC Department Of Biological Sciences, 5047 Gullen Mall, Wayne State University, Detroit,
Delimiting the conserved features of hunchback function for the trunk organization of insects
 RESEARCH ARTICLE 881 Development 135, 881-888 (2008) doi:10.1242/dev.018317 Delimiting the conserved features of hunchback function for the trunk organization of insects Henrique Marques-Souza 1, *, Manuel
RESEARCH ARTICLE 881 Development 135, 881-888 (2008) doi:10.1242/dev.018317 Delimiting the conserved features of hunchback function for the trunk organization of insects Henrique Marques-Souza 1, *, Manuel
Conservation and diversification of gene function during mouthpart development in Onthophagus beetles
 EVOLUTION & DEVELOPMENT 13:3, 280 289 (2011) DOI: 10.1111/j.1525-142X.2011.00479.x Conservation and diversification of gene function during mouthpart development in Onthophagus beetles Franck Simonnet
EVOLUTION & DEVELOPMENT 13:3, 280 289 (2011) DOI: 10.1111/j.1525-142X.2011.00479.x Conservation and diversification of gene function during mouthpart development in Onthophagus beetles Franck Simonnet
Chapter 16: Reconstructing and Using Phylogenies
 Chapter Review 1. Use the phylogenetic tree shown at the right to complete the following. a. Explain how many clades are indicated: Three: (1) chimpanzee/human, (2) chimpanzee/ human/gorilla, and (3)chimpanzee/human/
Chapter Review 1. Use the phylogenetic tree shown at the right to complete the following. a. Explain how many clades are indicated: Three: (1) chimpanzee/human, (2) chimpanzee/ human/gorilla, and (3)chimpanzee/human/
Morphogens in biological development: Drosophila example
 LSM5194 Morphogens in biological development: Drosophila example Lecture 29 The concept of morphogen gradients The concept of morphogens was proposed by L. Wolpert as a part of the positional information
LSM5194 Morphogens in biological development: Drosophila example Lecture 29 The concept of morphogen gradients The concept of morphogens was proposed by L. Wolpert as a part of the positional information
!!!!!!!! DB3230 Midterm 2 12/13/2013 Name:
 1. (10 pts) Draw or describe the fate map of a late blastula stage sea urchin embryo. Draw or describe the corresponding fate map of the pluteus stage larva. Describe the sequence of gastrulation events
1. (10 pts) Draw or describe the fate map of a late blastula stage sea urchin embryo. Draw or describe the corresponding fate map of the pluteus stage larva. Describe the sequence of gastrulation events
Genes controlling segmental specification in the Drosophila thorax (homeosis/insect segments/determination/clonal analysis)
 Proc. NatL Acad. Sci. USA Vol. 79, pp. 7380-7384, December 1982 Developmental Biology Genes controlling segmental specification in the Drosophila thorax (homeosis/insect segments/determination/clonal analysis)
Proc. NatL Acad. Sci. USA Vol. 79, pp. 7380-7384, December 1982 Developmental Biology Genes controlling segmental specification in the Drosophila thorax (homeosis/insect segments/determination/clonal analysis)
Hox gene duplications correlate with posterior heteronomy in scorpions
 Downloaded from rspb.royalsocietypublishing.org on August 13, 2014 Hox gene duplications correlate with posterior heteronomy in scorpions Prashant P. Sharma, Evelyn E. Schwager, Cassandra G. Extavour and
Downloaded from rspb.royalsocietypublishing.org on August 13, 2014 Hox gene duplications correlate with posterior heteronomy in scorpions Prashant P. Sharma, Evelyn E. Schwager, Cassandra G. Extavour and
Trans-regulatory functions in the Abdominal-B gene of the bithorax complex
 Development 101, 117-122 (1987) Printed in Great Britain The Company of Biologists Limited 1987 117 Trans-regulatory functions in the Abdominal-B gene of the bithorax complex JORDI CASANOVA 1 and ROBERT
Development 101, 117-122 (1987) Printed in Great Britain The Company of Biologists Limited 1987 117 Trans-regulatory functions in the Abdominal-B gene of the bithorax complex JORDI CASANOVA 1 and ROBERT
3/8/ Complex adaptations. 2. often a novel trait
 Chapter 10 Adaptation: from genes to traits p. 302 10.1 Cascades of Genes (p. 304) 1. Complex adaptations A. Coexpressed traits selected for a common function, 2. often a novel trait A. not inherited from
Chapter 10 Adaptation: from genes to traits p. 302 10.1 Cascades of Genes (p. 304) 1. Complex adaptations A. Coexpressed traits selected for a common function, 2. often a novel trait A. not inherited from
Hypothesis Testing in Evolutionary Developmental Biology: A Case Study from Insect Wings
 Journal of Heredity 2004:95(5):382 396 doi:10.1093/jhered/esh064 ª 2004 The American Genetic Association Hypothesis Testing in Evolutionary Developmental Biology: A Case Study from Insect Wings E. L. JOCKUSCH
Journal of Heredity 2004:95(5):382 396 doi:10.1093/jhered/esh064 ª 2004 The American Genetic Association Hypothesis Testing in Evolutionary Developmental Biology: A Case Study from Insect Wings E. L. JOCKUSCH
Unicellular: Cells change function in response to a temporal plan, such as the cell cycle.
 Spatial organization is a key difference between unicellular organisms and metazoans Unicellular: Cells change function in response to a temporal plan, such as the cell cycle. Cells differentiate as a
Spatial organization is a key difference between unicellular organisms and metazoans Unicellular: Cells change function in response to a temporal plan, such as the cell cycle. Cells differentiate as a
UNIVERSITY OF YORK BIOLOGY. Developmental Biology
 Examination Candidate Number: UNIVERSITY OF YORK BSc Stage 2 Degree Examinations 2017-18 Department: BIOLOGY Title of Exam: Developmental Biology Desk Number: Time allowed: 1 hour and 30 minutes Total
Examination Candidate Number: UNIVERSITY OF YORK BSc Stage 2 Degree Examinations 2017-18 Department: BIOLOGY Title of Exam: Developmental Biology Desk Number: Time allowed: 1 hour and 30 minutes Total
Drosophila melanogaster- Morphogen Gradient
 NPTEL Biotechnology - Systems Biology Drosophila melanogaster- Morphogen Gradient Dr. M. Vijayalakshmi School of Chemical and Biotechnology SASTRA University Joint Initiative of IITs and IISc Funded by
NPTEL Biotechnology - Systems Biology Drosophila melanogaster- Morphogen Gradient Dr. M. Vijayalakshmi School of Chemical and Biotechnology SASTRA University Joint Initiative of IITs and IISc Funded by
The Amphioxus Model System
 EGF, epithelium and The Amphioxus Model System Guest Editor Zbynek Kozmik Institute of Molecular Genetics of the Czech Academy of Sciences, Prague, Czech Republic Volume 61 Nos. 10/11/12 Special Issue
EGF, epithelium and The Amphioxus Model System Guest Editor Zbynek Kozmik Institute of Molecular Genetics of the Czech Academy of Sciences, Prague, Czech Republic Volume 61 Nos. 10/11/12 Special Issue
Neuronal cell death in grasshopper embryos: variable patterns in different species, clutches, and clones
 J. Embryol. exp. Morph. 78, 169-182 (1983) Printed in Great Britain The Company of Biologists Limited 1983 Neuronal cell death in grasshopper embryos: variable patterns in different species, clutches,
J. Embryol. exp. Morph. 78, 169-182 (1983) Printed in Great Britain The Company of Biologists Limited 1983 Neuronal cell death in grasshopper embryos: variable patterns in different species, clutches,
Hox genes and evolution of body plan
 Hox genes and evolution of body plan Prof. LS Shashidhara Indian Institute of Science Education and Research (IISER), Pune ls.shashidhara@iiserpune.ac.in 2009 marks 150 years since Darwin and Wallace proposed
Hox genes and evolution of body plan Prof. LS Shashidhara Indian Institute of Science Education and Research (IISER), Pune ls.shashidhara@iiserpune.ac.in 2009 marks 150 years since Darwin and Wallace proposed
Comparative Mouthpart Morphology and Evolution of the Carnivorous Heptageniidae (Ephemeroptera) 1
 l Aquatic Insects, Vol. 8 (1986). No. 2, pp. 83-89 0165-0424/86/0802-0083 $3.00 Swets & Zeitlinger Comparative Mouthpart Morphology and Evolution of the Carnivorous Heptageniidae (Ephemeroptera) 1 by W.
l Aquatic Insects, Vol. 8 (1986). No. 2, pp. 83-89 0165-0424/86/0802-0083 $3.00 Swets & Zeitlinger Comparative Mouthpart Morphology and Evolution of the Carnivorous Heptageniidae (Ephemeroptera) 1 by W.
Section 4 Professor Donald McFarlane
 Characteristics Section 4 Professor Donald McFarlane Lecture 11 Animals: Origins and Bauplans Multicellular heterotroph Cells lack cell walls Most have nerves, muscles, capacity to move at some point in
Characteristics Section 4 Professor Donald McFarlane Lecture 11 Animals: Origins and Bauplans Multicellular heterotroph Cells lack cell walls Most have nerves, muscles, capacity to move at some point in
Classification and Phylogeny
 Classification and Phylogeny The diversity of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize without a scheme
Classification and Phylogeny The diversity of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize without a scheme
178 Part 3.2 SUMMARY INTRODUCTION
 178 Part 3.2 Chapter # DYNAMIC FILTRATION OF VARIABILITY WITHIN EXPRESSION PATTERNS OF ZYGOTIC SEGMENTATION GENES IN DROSOPHILA Surkova S.Yu. *, Samsonova M.G. St. Petersburg State Polytechnical University,
178 Part 3.2 Chapter # DYNAMIC FILTRATION OF VARIABILITY WITHIN EXPRESSION PATTERNS OF ZYGOTIC SEGMENTATION GENES IN DROSOPHILA Surkova S.Yu. *, Samsonova M.G. St. Petersburg State Polytechnical University,
Embryogenesis of an insect nervous system II: A second class of neuron precursor cells and the origin of the intersegmental connectives
 J. Embryol. exp. Morph. Vol. 61,pp. 317-330, 1981 3J7 Printed in Great Britain @ Company of Biologists Limited 1981 Embryogenesis of an insect nervous system II: A second class of neuron precursor cells
J. Embryol. exp. Morph. Vol. 61,pp. 317-330, 1981 3J7 Printed in Great Britain @ Company of Biologists Limited 1981 Embryogenesis of an insect nervous system II: A second class of neuron precursor cells
This is the submitted version of a paper published in Development, Genes and Evolution.
 http://www.diva-portal.org Preprint This is the submitted version of a paper published in Development, Genes and Evolution. Citation for the original published paper (version of record): Eriksson, B.,
http://www.diva-portal.org Preprint This is the submitted version of a paper published in Development, Genes and Evolution. Citation for the original published paper (version of record): Eriksson, B.,
Classification and Phylogeny
 Classification and Phylogeny The diversity it of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize without a scheme
Classification and Phylogeny The diversity it of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize without a scheme
Conservation of arthropod midline netrin accumulation revealed with a cross-reactive antibody provides evidence for midline cell homology
 EVOLUTION & DEVELOPMENT 11:3, 260 268 (2009) DOI: 10.1111/j.1525-142X.2009.00328.x Conservation of arthropod midline netrin accumulation revealed with a cross-reactive antibody provides evidence for midline
EVOLUTION & DEVELOPMENT 11:3, 260 268 (2009) DOI: 10.1111/j.1525-142X.2009.00328.x Conservation of arthropod midline netrin accumulation revealed with a cross-reactive antibody provides evidence for midline
Evolution of the Skeleton
 Evolution of the Skeleton Reading Benton Chapter 5 The Vertebrate Archetype (Richard Owen) Next week Tuesday (17 Sept): Paper Discussion Purnell, M. A. 2001. Scenarios, selection and the ecology of early
Evolution of the Skeleton Reading Benton Chapter 5 The Vertebrate Archetype (Richard Owen) Next week Tuesday (17 Sept): Paper Discussion Purnell, M. A. 2001. Scenarios, selection and the ecology of early
Non-independence in Statistical Tests for Discrete Cross-species Data
 J. theor. Biol. (1997) 188, 507514 Non-independence in Statistical Tests for Discrete Cross-species Data ALAN GRAFEN* AND MARK RIDLEY * St. John s College, Oxford OX1 3JP, and the Department of Zoology,
J. theor. Biol. (1997) 188, 507514 Non-independence in Statistical Tests for Discrete Cross-species Data ALAN GRAFEN* AND MARK RIDLEY * St. John s College, Oxford OX1 3JP, and the Department of Zoology,
Sonic hedgehog (Shh) signalling in the rabbit embryo
 Sonic hedgehog (Shh) signalling in the rabbit embryo In the first part of this thesis work the physical properties of cilia-driven leftward flow were characterised in the rabbit embryo. Since its discovery
Sonic hedgehog (Shh) signalling in the rabbit embryo In the first part of this thesis work the physical properties of cilia-driven leftward flow were characterised in the rabbit embryo. Since its discovery
"PRINCIPLES OF PHYLOGENETICS: ECOLOGY AND EVOLUTION" Integrative Biology 200B Spring 2011
 "PRINCIPLES OF PHYLOGENETICS: ECOLOGY AND EVOLUTION" Integrative Biology 200B Spring 2011 Evolution and development ("evo-devo") The last frontier in our understanding of biological forms is an understanding
"PRINCIPLES OF PHYLOGENETICS: ECOLOGY AND EVOLUTION" Integrative Biology 200B Spring 2011 Evolution and development ("evo-devo") The last frontier in our understanding of biological forms is an understanding
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11589 Supplementary Figure 1 Ciona intestinalis and Petromyzon marinus neural crest expression domain comparison. Cartoon shows dorsal views of Ciona mid gastrula (left) and Petromyzon
doi:10.1038/nature11589 Supplementary Figure 1 Ciona intestinalis and Petromyzon marinus neural crest expression domain comparison. Cartoon shows dorsal views of Ciona mid gastrula (left) and Petromyzon
(http://www.cliffsnotes.com/study_guide/evidence-for-evolution.topicarticleid-8741,articleid-8636.html)
 Teacher Wrap-up Background Information Darwin noted the striking similarity among embryos of complex animals such as humans, chickens, frogs, reptiles, and fish. He wrote that the uniformity is evidence
Teacher Wrap-up Background Information Darwin noted the striking similarity among embryos of complex animals such as humans, chickens, frogs, reptiles, and fish. He wrote that the uniformity is evidence
The evolution of selector gene function: Expression dynamics and regulatory interactions of tiptop/teashirt across Arthropoda
 DOI: 10.1111/ede.12270 RESEARCH The evolution of selector gene function: Expression dynamics and regulatory interactions of tiptop/teashirt across Arthropoda Logan E. March Rachel M. Smaby Emily V. W.
DOI: 10.1111/ede.12270 RESEARCH The evolution of selector gene function: Expression dynamics and regulatory interactions of tiptop/teashirt across Arthropoda Logan E. March Rachel M. Smaby Emily V. W.
Big Idea #1: The process of evolution drives the diversity and unity of life
 BIG IDEA! Big Idea #1: The process of evolution drives the diversity and unity of life Key Terms for this section: emigration phenotype adaptation evolution phylogenetic tree adaptive radiation fertility
BIG IDEA! Big Idea #1: The process of evolution drives the diversity and unity of life Key Terms for this section: emigration phenotype adaptation evolution phylogenetic tree adaptive radiation fertility
Phylogeny and systematics. Why are these disciplines important in evolutionary biology and how are they related to each other?
 Phylogeny and systematics Why are these disciplines important in evolutionary biology and how are they related to each other? Phylogeny and systematics Phylogeny: the evolutionary history of a species
Phylogeny and systematics Why are these disciplines important in evolutionary biology and how are they related to each other? Phylogeny and systematics Phylogeny: the evolutionary history of a species
Classification, Phylogeny yand Evolutionary History
 Classification, Phylogeny yand Evolutionary History The diversity of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize
Classification, Phylogeny yand Evolutionary History The diversity of life is great. To communicate about it, there must be a scheme for organization. There are many species that would be difficult to organize
Cellular origins of bilateral symmetry in glossiphoniid leech embryos
 Hydrobiologia 402: 285 290, 1999. A.W.C. Dorresteijn & W. Westheide (eds), Reproductive Strategies and Developmental Patterns in Annelids. 1999 Kluwer Academic Publishers. Printed in the Netherlands. 285
Hydrobiologia 402: 285 290, 1999. A.W.C. Dorresteijn & W. Westheide (eds), Reproductive Strategies and Developmental Patterns in Annelids. 1999 Kluwer Academic Publishers. Printed in the Netherlands. 285
The effect of lethal mutations and deletions within the bithorax complex upon the identity of caudal metameres in the Drosophila embryo
 /. Embryol. exp. Morph. 93, 153-166 (1986) 153 Printed in Great Britain The Company of Biologists Limited 1986 The effect of lethal mutations and deletions within the bithorax complex upon the identity
/. Embryol. exp. Morph. 93, 153-166 (1986) 153 Printed in Great Britain The Company of Biologists Limited 1986 The effect of lethal mutations and deletions within the bithorax complex upon the identity
Dr. Amira A. AL-Hosary
 Phylogenetic analysis Amira A. AL-Hosary PhD of infectious diseases Department of Animal Medicine (Infectious Diseases) Faculty of Veterinary Medicine Assiut University-Egypt Phylogenetic Basics: Biological
Phylogenetic analysis Amira A. AL-Hosary PhD of infectious diseases Department of Animal Medicine (Infectious Diseases) Faculty of Veterinary Medicine Assiut University-Egypt Phylogenetic Basics: Biological
