Signal transduction pathways that regulate cell survival and cell death
|
|
|
- Cynthia Small
- 6 years ago
- Views:
Transcription
1 Oncogene (1998) 17, 3207 ± 3213 ã 1998 Stockton Press All rights reserved 0950 ± 9232/98 $ Signal transduction pathways that regulate cell survival and cell death Tomislav Dragovich 3, Charles M Rudin 3 and Craig B Thompson*,1,2,3 1 Gwen Knapp Center for Lupus and Immunology Research, The University of Chicago, Chicago, Illinois 60637, USA; 2 Howard Hughes Medical Institute, The University of Chicago, Chicago, Illinois 60637, USA and 3 Department of Medicine, The University of Chicago, Chicago, Illinois 60637, USA Apoptosis or programmed cell death (PCD) is a physiological process critical for organ development, tissue homeostasis and elimination of defective or potentially dangerous cells in complex organisms. Apoptosis permits cell death without a concomitant in ammatory response in the surrounding tissues. The process of apoptosis depends on the reception of multiple extracellular and intracellular signals, integration and ampli cation of these signals by second messengers and nally, activation of the death e ector proteases. Defects in control of apoptotic pathways may contribute to a variety of diseases including cancer, autoimmune and neurodegenerative conditions and AIDS. While many components of the regulatory network controlling apoptosis have been de ned, the mechanisms of action and patterns of interaction of these factors remain controversial. This article summarizes some of the known aspects of signaling pathways involved in apoptosis. Keywords: signal transduction; bcl-2 proteins; caspases; death receptors Events that activate the central pathway of cell death Programmed cell death or apoptosis can be initiated by a wide variety of stimuli, including developmental signals, cellular stress and disruption of cell cycle. In contrast, execution of apoptosis is remarkably uniform, involving characteristic morphologic and biochemical changes. The morphologic hallmarks of apoptosis include membrane blebbing, volume loss, nuclear condensation and DNA fragmentation (Kerr et al., 1972). A number of the key factors involved in the regulation, coordination and execution of these events have been identi ed. Genetic studies of the nematode Caenorhabditis elegans led to identi cation of three genes essential for control and execution of apoptosis in developing worms (Hengartner and Horvitz, 1994). Mammalian homologs of all three of these genes have been identi ed. The regulation and execution of apoptosis in C. elegans seems deceptively simple. Apoptotic cell death in the worm requires CED-3, a cousin of mammalian caspases and its activator CED-4 that is homologous to mammalian Apaf-1. Additional control of cell death/survival is provided by the anti-apoptotic factor CED-9 which is *Correspondence: CB Thompson a counterpart of mammalian Bcl-2 protein (Yuan et al., 1993). Recently, a novel C. elegans protein, Egl-1, has been found to bind to and inhibit the antiapoptotic function of CED-9 (Conradt and Horvitz, 1998). The molecular basis of apoptotic signaling appears to be more complex in mammalian cells. In mammalian cells, Bcl-2 is one of a large family of CED-9 related proteins that function as either positive or negative regulators of apoptosis. Mammalian counterparts of the nematode protein CED-3 comprise a large family of cysteine proteases, called caspases, which are critical in generation of apoptotic morphology. The multiple members of these gene families may allow for the independent regulation of apoptotic signaling pathways. Based on the established interactions among the known mediators of apoptosis three classic pathways of apoptotic signaling in mammalian cell have emerged (Figure 1). The rst one is initiated by the withdrawal of growth factors and is regulated by Bcl- 2 family of proteins. This pathway results in cytochrome c release from mitochondria, activation of Apaf-1 and triggering of a caspase cascade. The other well-established apoptosis pathway involves signaling by cell surface death receptors such as TNF or Fas, which through adapter molecules can recruit and activate caspases. The third and least well characterized pathway is initiated by DNA damage. Although in part regulated by proteins such as p53 and ATM, how this pathway results in caspase activation is not known. In all three of these pathways of cell death, caspases and Bcl-2 protein family play a key role in regulation and execution of apoptosis. Caspases Multiple mammalian homologs of CED-3 have been identi ed (1 ± 13). They all cleave peptide substrates after aspartic acid residues (Alnemri et al., 1996). Caspase activation initiates a proteolytic degradation that results in apoptotic morphology (Casiano et al., 1996). Speci c inhibitors of caspases prevent cell death, or at least apoptotic morphology, in response to many of the known signals that trigger apoptosis (Miura et al., 1993; Rabizadeh et al., 1993). Overexpression of most of the known caspases triggers apoptosis in cell lines, although only a subset of caspases appears to be involved in physiological apoptosis. Caspase-9 has been most clearly linked to central pathway of apoptotic induction. Apaf-1 in response to cytochrome c binding can form a complex with caspase-9 that initiates apoptotic events in cell free system (Li et al., 1997).
2 3208 Figure 1 Multiple apoptotic signals converge on caspase activation. Pathways discussed in this review include (1) those derived from cell surface receptor engagement, represented here by Fas receptor signaling, (2) those derived from growth factor withdrawal, represented here by the IL-3 receptor, and (3) those derived from nuclear DNA damage Caspase-9 is a critical regulator of at least some forms of apoptosis. Homozygous inactivation of the caspase- 9 gene resulted in embryonic lethality manifested by severe perturbations of the cerebral cortex and forebrain (Hakem et al., 1998; Kuida et al., 1998). Caspase-9 de cient cells were resistant to cell death induced by gamma-irradiation or cytotoxic drugs although the death response to other apoptotic stimuli, including Fas receptor engagement, was preserved. Caspase activation requires proteolytic processing from inactive proenzymes. The processing sites for procaspases are themselves caspase consensus sequences. Thus caspases may cleave and activate their own as well as other caspase pro-enzymes. This may serve as an important mechanism for amplifying caspasedependent apoptotic pathways. For example, active caspase-9 cleaves procaspase-3; active caspase-3 in turn appears to be required for many of the characteristic apoptotic nuclear changes. Downstream caspases cleave and inactivate proteins crucial for the maintenance of cellular cytoskeleton, DNA repair, signal transduction and cell cycle control (Fraser and Evan, 1996; Nagata 1997). Recent studies have uncovered a possible link between cytosolic caspase activation and apoptotic nuclear changes (Enari et al., 1998; Sakahira et al., 1998). A caspase-dependent deoxyribonuclease, CAD, is inactive and localized in the cytosol when bound to the inhibitor ICAD. Caspase-3 can cleave and inactivate ICAD thus releasing CAD that then translocates to the nucleus and initiates chromosomal degradation. While caspase activation typically results in apoptotic morphology, not all programmed cell death pathways are caspase-dependent. Cells which sustain traumatic damage or are deprived of growth-factors can die even in the presence of caspase inhibitors (Rathmell and Thompson, 1998). However, in the presence of such inhibitors the morphology of dying cell lacks many morphologic features of apoptosis, including typical nuclear changes (Hirsch et al., 1997). Outside of such experimental systems, it appears that caspase activation and cellular autodigestion may invariably accompany apoptosis in vivo. The Bcl-2 family A mammalian homolog of the C. elegans ced-9 gene, bcl-2, was identi ed as one of the genes at chromosomal breakpoint of t(14 : 18) in B-cell lymphomas. Overexpression of Bcl-2 protects cells from a variety of apoptotic stimuli, such as growth-factor withdrawal, exposure to chemotherapy agents or
3 toxins, viral infection and inappropriate oncogene expression. A number of Bcl-2 related proteins have been subsequently identi ed (Reed, 1997). Although they all share domains of structural homology, some Bcl-2 family proteins promote apoptosis while the others have anti-apoptotic function. The relative levels of pro- and anti-apoptotic Bcl-2 family proteins have been suggested to function as a `rheostat' regulating the apoptotic threshold of the cell (Yang and Korsmeyer, 1996). The antagonistic function of some of the members of the family is at least in part explained by their ability to form heterodimers. The anti-apoptotic proteins Bcl-x L and Bcl-2 form heterodimers with pro-apoptotic Bax (Oltvai and Korsmeyer, 1994). An excess of Bax promotes cell death, but coexpression of Bcl-x L or Bcl-2 can neutralize this e ect. An alpha-helical domain known as BH3 and shared by multiple members of the Bcl-2 family is critical for the formation of homo- and heterodimers (Kelekar and Thompson, 1998). Some of pro-apoptotic Bcl-2 family members, such as Bad and Bid, only share homology within the BH3 domains (so-called BH3 proteins). Most Bcl-2 family proteins have a conserved C- terminal hydrophobic domain, which is necessary for insertion into the outer mitochondrial, endoplasmic reticular and outer nuclear membranes. Based on this unusual subcellular localization and homology to bacterial pore forming molecules, such as the poreforming subunit of diphteria toxin, it is suspected that some members of Bcl-2 protein family function as membrane pores or channels (Muchmore et al., 1996). Indeed, both Bcl-x L and Bcl-2 as well as the proapoptotic Bax were found to have pore-forming capacity in vitro (Minn et al., 1997; Schendel et al., 1997; Antonsson et al., 1997). Recent studies support the central role of Bcl-2 family proteins in the regulation of mitochondrial membrane integrity and cytochrome c release (Yang et al., 1995; Vander Heiden et al., 1997). Overexpression of either Bcl-2 or Bcl-x L inhibits cytochrome c release from mitochondria. In contrast, when transfected in mammalian or yeast cells, Bax overexpression results in dissipation of mitochondrial membrane potential and release of cytochrome c (Rosse et al., 1998; Manon et al., 1997). The molecular mechanism of this regulation has not been determined. Some investigators suggest that Bcl-2 like proteins regulate the opening of what has been referred to as mitochondrial permeability transition pore (PTP) or megachannel. The hypothetical PTP would consist of the adenine nucleotide translocator in the inner mitochondrial membrane, voltage-dependent anion channel in the outer membrane as well as other components (Marzo et al., 1998). Physical association between Bcl-2 family proteins and components of PTP remains speculative although both Bcl-x L and Bcl-2 localize to contact points between the inner and outer mitochondrial membrane and Bax was recently co-puri ed with other components of the pore (Marzo et al., 1998). In addition, some of the antiapoptotic Bcl-2 proteins may function by complexing with and inactivating factors that trigger caspase activation. Both cytochrome c and Apaf-1 have been de ned as co-factors that are critical for the activation of caspase-9 and the subsequent apoptotic cascade. As mentioned before, in C. elegans CED-9 directly interacts with CED-4 preventing it from activating caspase CED-3. In mammalian cells, Bcl-x L has been reported to interact with the mammalian CED-4 homolog Apaf-1 and by that mechanism might prevent Apaf-1 from activating downstream caspases (Hu et al., 1998). These several lines of evidence suggest that Bcl-2 family members function at a critical decision point immediate upstream of an irreversible commitment to cell death. The Bcl-2 family proteins, Apaf-1, and caspases together comprise mammalian equivalent of the C. elegans `apoptosome' consisting of Ced-9, Ced-4 and Ced-3. Loss of mitochondrial transmembrane potential and activation of caspases represent molecular hallmarks of mammalian central apoptotic pathway. Bcl-2 family proteins appear to be critical in regulation of both events. Nevertheless, signi cant gaps in this model remain. For example, although the model integrates the three critical apoptotic regulators de ned in C. elegans, it fails to explain how extramitochondrial Bcl-2 inhibits apoptosis and why it can do so even after cytochrome c is released (Rosse et al., 1998; Li et al., 1997). It does not explain the nding that caspase inhibitors do not inhibit Bax toxicity in mammalian cells suggesting that Bax may induce apoptotic changes independently of the mammalian `apoptosome' (Xiang et al., 1996). Further analysis of the molecular basis of programmed cell death execution will be required to answer these and other important questions. Receptors and second messengers that regulate cell death and survival Homeostasis in mammalian cells is dependent on the continuous integration of survival and death signals from the extracellular environment (Ra, 1992). Extracellular signals that activate apoptotic or survival pathways include peptide growth factors, cytokines and interleukins. Signaling pathways can be triggered by both soluble factors and by direct cell to cell contact. One of the most intensively studied families of receptors involved in apoptotic signaling is that of tumor necrosis factor receptor (TNFR) family (Bazzoni and Beutler, 1996). This is a family of multimeric receptors that upon activation by their respective ligands may initiate either cell death or survival signals. Extracellular domains of the TNFR family members are related, while intracellular domains di er widely both in sequence and in their a nity for various adapter molecules and second messengers. The subfamily of receptors most closely linked to induction of cell death (e.g. Fas, TNFR1 and DR3) do contain a conserved intracellular sequence known as a `death domain'. Through these domains, receptors associate with a number of intracellular signal transduction molecules that themselves contain the `death domain'. For example, the intracellular domain of activated Fas receptor interacts with that of a linker molecule called FADD (Fas-associated death domain-containing protein) (Chinnaiyan et al., 1995). Subsequent transduction of the apoptotic signal is dependent on another domain of FADD, the `death e ector domain', which interacts with additional downstream molecules. One of them is caspase-8, which after activation by FADD can initiate a cascade of caspase activation leading to 3209
4 3210 degradation of intracellular substrates and cell death (Muzio et al., 1996). TNFR1 similarly induces cell death through interaction with a related but di erent adapter molecule called TRADD (TNFR-associated death domain protein) (Hsu et al., 1996). While activation of Fas, TNFR1 and DR3 receptors induces apoptosis by associating with cytoplasmic factors such as FADD, TRADD and RIP, other receptors such as CD40, TNFR2 and CD30 promote cell survival by interacting with a distinct group of second messengers, including the TNF receptorassociated factors or TRAFs (Arch et al., 1998). To date six mammalian TRAF proteins have been identi ed. Some members of the TRAF family negatively regulate apoptosis through induction of survival pathways. For example, recruitment of TRAF2 protein to TNF receptors has been reported to promote survival and activates JNK (c-jun N- terminal kinase) and NF-kB (nuclear factor-kb) (Yeh et al., 1997). A number of recent studies have linked NF-kB activation to the induction of cell survival pathway (Liu et al., 1996; Beg and Baltimore, 1996). Additional regulatory proteins further modify signaling pathways downstream from TRAF recruitment. These include adapter proteins like A20, cellular inhibitor of apoptosis (c-iap), TRAF-interacting protein (TRIP) and TRAF family member associated NF-kB activator (I-TRAF/TANK) (Cheng and Baltimore, 1996; Lee and Choi, 1997; Rothe et al., 1996; Roy et al., 1997). TRIP inhibits NF-kB activation by TRAFs while I-TRAF/TANK augments it depending on the speci c cellular context. c-iap1 and c-iap2 interact with TRAFs and have been reported to interfere with the activation of caspases even downstream of cytochrome c release (Roy et al., 1997). FLICE inhibitory protein or FLIP, which contains two death e ector domains and an inactive caspase domain, binds to FADD and caspase-8 and inhibits death receptor-induced apoptosis (Hu et al., 1997; Thome et al., 1997). This web of interacting proteins leads to very di erent responses in di erent cells and at di erent times. Interaction of CD30 and CD40 receptors with TRAF proteins, depending on cellular context can promote either cell death or cell survival (Arch et al., 1998). It is interesting that signaling through CD30 and CD40 can promote survival of naive lymphocytes, but subsequently increases their sensitivity to death inducing signals from Fas or TNFR1 (Rathmell et al., 1996; Duckett and Thompson, 1997). This may serve as an internal control mechanism to prevent unlimited proliferation of cells that might otherwise result in development of autoimmune disease or neoplasia. Another extracellular signal for induction of apoptosis is deprivation of cells of their anchorage to extracellular matrix or from cell to cell contacts. The induction of apoptotic pathway through disruption of cell anchorage to the substratum is termed anoikis (Aplin et al., 1998). The exact signaling pathway responsible for the induction of apoptosis after cell anchorage deprivation is unknown. One of the two pathways proposed includes tyrosine kinase FAK, PI-3 kinase and Akt. The other, parallel pathway may involve the stress induced kinases including MEKK-1 and JNK (Frisch and Rouslahti, 1997). Overexpression of Bcl-2 in epithelial cells can protect the cells from anoikis. The physiological role of anoikis is unclear although disregulation of this process may be important for the ability of malignant cells to invade and metastasize elsewhere. Nuclear factors ivolved in apoptosis In addition to extracellular signals, cells can undergo apoptosis in response to internal signals including genetic abnormality. Defective or inappropriate cell cycle progression caused by a variety of genotoxic injuries can result in cell cycle arrest, and subsequent induction of apoptosis. The tumor suppressor gene p53 has been clearly linked to these pathways leading to its designation as the `guardian of the genome'. DNA strand breaks induce rapid p53 upregulation but the exact mechanism of this triggering event remains unknown. The upregulation of p53 is mostly posttranscriptional, involving both, an increase in translation and prolonged half-life. p53 is a sequence-speci c DNA-binding protein, which activates transcription of genes including Gadd45, mdm2, p21 and bax (Cox and Lane, 1995; Wyllie, 1997). Increase in bax transcription may be in part responsible for p53-induced apoptosis following DNA damage (Miyashita and Reed, 1995). However, p53 can also activate apoptosis by transcription-independent mechanisms (Caelles et al., 1994). What is more, there are p53-independent pathways that result in cellular apoptosis following DNA damage. T cell lymphoma lines derived from p53 knockout mice died rapidly after exposure to irradiation or to drugs that promote DNA strand breaks (Strasser et al., 1994). Death occurred by apoptosis and was inhibitable by Bcl-2 suggesting that Bcl-2 acts downstream of p53 signal and pointing out that there are additional, parallel and p53-independent pathways regulating DNA damage induced apoptosis in mammalian cells. Another gene implicated in DNA damage induced apoptosis is ATM (Morgan and Kastan, 1997). This gene is defective in patients with ataxia-telangiectasia disorder, who are prone to lymphoid malignancies, cerebellar degeneration and are extremely sensitive to ionizing radiation. Cells de cient in ATM do not upregulate p53 following irradiation damage, suggesting that ATM plays a role in the induction of p53. The downstream p53-dependent apoptotic pathway, however appears to be intact in ATM-de cient cells. Interactions between the molecules that sense DNA damage and molecules involved in cytosolic activation of caspases have not been established. Such connections must exist as cytosol-nuclear translocation of the nuclease CAD requires cytosolic, caspase-dependent inactivation of ICAD. How `inside-out' or nuclearcytoplasmic signal transduction is achieved remains to be elucidated. Cross-talk between the pathways Di erent pathways involved in regulation of cell proliferation and cell death may have di erent signi cance in various cells or within the same cell at di erent stages of development. We now have some evidence of the cross-talk between the components of
5 di erent signaling pathways within the cell (Figure 2). Bid, a pro-apoptotic Bcl-2 family protein, is cleaved by caspase-8 in response to Fas signaling (Li et al., 1998; Luo et al., 1998). Following cleavage, the C-terminal fragment of Bid binds to mitochondria inducing the release of cytochrome c, which in turn activates Apaf-1 and initiates procaspase-9 processing. Thus, this crosstalk connects two of the classic apoptotic pathways depicted in Figure 1; that of Fas receptor mediated activation of caspase-8 with that of IL-3 withdrawalinduced mitochondrial release of cytochrome c and activation of caspase-9. Another example of the cross-talk between di erent signaling pathways that regulate cell proliferation and cell death involves Bad, a pro-apoptotic Bcl-2 family protein. Bad executes its pro-apoptotic function by binding to anti-apoptotic proteins Bcl-2 and Bcl-x L. Following growth factor receptor engagement, the growth factor dependent serine/threonine kinase Akt phosphorylates Bad (Figure 2). The phosphorylated Bad is sequestered by cytosolic protein thus increasing the levels of free Bcl-2 and Bcl-x L (Zha et al., 1996). In this example, a growth factor initiated, survival-promoting signal interferes with a Bcl-2/Bclx L -dependent regulation of the cellular apoptotic threshold (Gajewski and Thompson, 1996). It is expected that more such examples of cross-talk will be uncovered as we continue to untangle the web of apoptotic signaling pathways. Conclusion Our understanding of the complex processes that determine cell fate has advanced over the past decade yet many unanswered questions remain. The complexity and redundancy involved in regulation of apoptosis re ects its central importance for the growth and development of complex organisms. The signal transduction of apoptotic stimuli resembles a complex information network which includes systems for reception, processing of information, ampli cation, feedback, and response integration. The apoptotic threshold varies widely with the cell type and its stage of development and di erentiation. As the cells exist in a dynamic equilibrium, their threshold for apoptosis may be constantly changing. Alterations in external milieu are sensed through the cell surface receptors as well as multiple cytoplasmic and nuclear factors. These multiple signals are integrated through a complex 3211 Figure 2 Cross-talk between positive and negative apoptotic signaling pathways. Positive and negative apoptotic signals can be integrated at the level of the mitochondrial outer membrane by interactions involving the Bcl-2 family of apoptotic regulators
6 3212 network of positive and negative regulators of apoptosis that ultimately determine cell fate. Bcl-2 family members, both by their ability to interact with other mediators of apoptosis and their subcellular localization, appear to function at the point of convergence of multiple apoptotic signals. Disturbances in cellular metabolism, whether imposed by starvation, infection, toxins or by DNA damage ultimately result in mitochondrial membrane release of cytochrome c and activation of downstream caspases. Based on recent studies, mitochondrial membrane disruption appears to be a `point of no return' for the cell, an ultimate commitment to cell death. Bcl-2 family members and caspases appear to be critical factors involved in regulating the outcome of these events. Recent advances in apoptosis research have identi ed new molecular players that regulate this fundamental and complex biological process. However, there are large gaps in our ability to understand the intricate network of apoptotic signal transduction. While there is considerable information concerning apoptotic signaling pathways that involve cell surface receptors that can induce caspase activation, much less is known about Bcl-2 family proteins and their promiscuous interactions among themselves, with caspases and with components of the mitochondria. A deeper understanding of the physiological importance of numerous molecules involved in apoptotic signaling pathways requires further investigation. In addition, we need to learn more about how DNA damage initiates and regulates the execution of programmed cell death. There is every reason to believe that new and exciting advances in apoptosis research will continue to come at a rapid pace, constantly changing the eld and making reviews like this outdated by the time they reach the press. References Alnemri ES, Livingston DJ, Nicholson DW, Salvesen G, Thornberry NA, Wong WW and Juan J. (1996). Cell, 87, 171. Antonsson B, Conti F, Ciavatta Am, Montessuit S, Lewis S, Martinou I, Bernasconi L, Bernard A, Mermod JJ, Mazzei G, Maundrell K, Gambale F, Sadoul R and Martinou JC. (1997). Science, 277, 370 ± 372. Aplin AE, Howe A, Alahari SK and Juliano RL. (1998). Pharmacol. Rev., 50, 199 ± 251. Arch RH, Gedrich RW and Thompson CB. (1998). Genes & Dev., 12, 2821 ± Bazzoni F and Beutler B. (1996). N. Engl. J. Med., 334, 1717 ± Beg AA and Baltimore D. (1996). Science, 274, 782 ± 784. Caelles C, Helmberg A and Karin M. (1994). Nature, 370, 220 ± 223. Casiano CA, Martin SJ, Green DR and Tan EM. (1996). J. Exp. Med., 184, 765 ± 770. Cheng G and Baltimore D. (1996). Genes Dev., 10, 963 ± 973. Chinnaiyan AM, O'Rourke K, Tewari M and Dixit VM. (1995). Cell, 81, 505 ± 512. Conradt B and Horvitz HR. (1998). Cell, 93, 519 ± 529. Cox LS and Lane DP. (1995). Bioessays, 17, 501 ± 508. Duckett CS and Thompson CB. (1998). Genes Dev., 11, 2810 ± Enari M, Sakahira H, Yokoyama H, Okawa K, Iwamatsu A and Nagata S. (1998). Nature, 391, 43 ± 50. Fraser A and Evan G. (1996). Cell, 85, 781 ± 784. Frisch SM and Ruoslahti E. (1997). Curr. Opin. Cell Biol., 9, 701 ± 706. Gajewski TF and Thompson CB. (1996). Cell, 87, 589 ± 592. Hakem R, Hakem A, Duncan GS, Henderson JT, Woo M, Soengas MS, Elia A, de-la-pompa JL, Kagi D, Khoo W, Potter J, Yoshida R, Kaufman SA, Lowe SW, Penninger JM and Mak TW. (1998). Cell, 94, 339 ± 352. Hengartner MO and Horvitz HR. (1994). Cell, 76, 665 ± 676. Hirsch T, Marchetti P, Susin SA, Dallaporta B, Zamzami N, Marzo I, Geuskens M and Kroemer G. (1997). Oncogene, 15, 1573 ± Hsu H, Shu H-B, Pan MB and Goeddel DV. (1996). Cell, 84, 299 ± 308. Hu S, Vincenz C, Buller M and Dixit VM. (1997). J. Biol. Chem., 272, Hu Y, Benedict MA, Wu D, Inohara N and Nunez G. (1998). Proc. Natl. Acad. Sci. USA, 95, 4386 ± Kastan MB. (1997). N. Engl. J. Med., 333, 662 ± 663. Kelekar A and Thompson CB. (1998). Trends. Cell Biol., 8, 324 ± 330. Kerr JFR, Wyllie AH and Currie AR. (1972). J. Cancer, 26, 239 ± 257. Kuida K, Haydar TF, Kuan CY, Gu Y, Taya C, Karasuyama H, Su MS, Rakic P and Flavell RA. (1998). Cell, 94, 325 ± 337. Lee SY and Choi Y. (1997). J. Exp. Med., 185, 1275 ± Li H, Zhu H, Xu Cj and Yuan J. (1998). Cell, 94, 491 ± 501. Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES and Wang X. (1997). Cell, 91, 479 ± 489. Liu Z, Hsu H, Goeddel DV and Karin M. (1996). Cell, 87, 555 ± 576. Luo X, Budihardjo I, Zou H, Claughter C and Wang X. (1998). Cell, 94, 481 ± 490. Manon S, Chaudhuri B and Guerin M. (1997). FEBS Lett, 415, 29 ± 32. Marzo I, Brenner C, Zamzami N, Susin SA, Beutner G, Brdiczka D, Remy R, Xie ZH, Reed JC and Kroemer G. (1998). J. Exp. Med., 187, 1261 ± Minn AJ, Velez P, Schendel SL, Liang H, Muchmore SW, Fesik SW, Fill M and Thompson CB. (1997). Nature, 385, 353 ± 357. Miura M, Zhu H, Rotello R, Hartweig EA and Yuan J. (1993). Cell, 75, 653 ± 660. MiyashitaT and Reed JC. (1997). Blood, 81, 151 ± 157. Morgan SE and Kastan MB. (1997). Adv. Cancer Res., 71, 1±25. Muchmore SW, Sattler M, Liang H, Meadows RP, Harlan JE, Yoon HS, Nettesheim D, Chang BS, Thompson CB, Wong SL, Ng SL and Fesik SW. (1996). Nature, 381, 335 ± 341. Muzio M, Chinnaiyan AM, Kischkel FC, O'Rourke K, Shevchenko A, Ni J, Sca di C, Bretz JD, Zhang M, Gentz R, Mann M, Krammer PH, Peter ME and Dixit VM. (1996). Cell, 85, 817 ± 827. Nagata S. (1997). Cell, 88, 355 ± 365. Oltvai ZN and Korsmeyer SJ. (1994). Cell, 79, 189 ± 192. Ra MC. (1992). Nature, 356, 397 ± 400. Rabizedeh S, LaCount DJ, Friesen PD and Bredsen DE. (1993). J. Neurochem., 61, 2318 ± Rathmel JC and Thompson CB. (1998). Ann. Rev. Immunol., in press.. Rathmell JC, Townsed SE, Xu JC, Flavell RA and Godnow CC. (1996). Cell, 87, 319 ± 329. Reed JC. (1997). Nature, 387, 773 ± 776. Rosse T, Olivier R, Monney L, Rager M, Conus S, Fellay I, Jansen B and Borner C. (1998). Nature, 391, 496 ± 499.
7 Rothe M, Xiong J, Shu B, Williamson K, Goddard A and Goeddel DV. (1996). Proc. Natl. Acad. Sci. USA, 93, 8241 ± Roy N, Deveraux QL, Takanishi R, Salvesen GS and Reed JC. (1997). EMBO J, 16, 6914 ± Sakahira H, Enari M and Nagata S. (1998). Nature, 391, 96 ± 99. Schendel SL, Xie Z, Montal MO, Matsuyama S, Montal M and Reed JC. (1997). Proc. Natl. Acad. Sci. USA, 94, 5113 ± Strasser A, Harris AW, Jacks T and Cory S. (1994). Cell, 79, 329 ± 339. Thome M, Schneider P, Ho man K, Fickensher H, Meinl E, Neipel F, Mattmann C, Burns K, Bodmer JL, Schroter M, Sca di C, Krammer PH, Peter ME and Tschopp J. (1997). Nature, 386, 517. Vander Heiden MG, Chandel NS, Williamson EK, Schumaker PT and Thompson CB. (1997). Cell, 91, 627 ± 637. Wyllie A. (1997). Nature, 389, 237 ± 238. Xiang J, Chao D and Korsmeyer SJ. (1996). Proc. Natl. Acad.Sci.USA,93, ± Yang E and Korsmeyer SJ. (1996). Blood, 88, 386 ± 401. Yang E, Zha J, Jockel J, Boise LH, Thompson CB and Korsmeyer SJ. (1995). 80, 285 ± 291. Yeh WC, Shahinian A, Speiser D, Kraunus J, Billia F, Wakeham A, de-la-pompa JL, Ferrick D, Hum B, Iscove N, Ohashi P, Rothe M, Goeddel DV and Mak TW. (1997). Immunity, 7, 715 ± 725. Yuan J, Shaham S, Ledoux S, Ellis HM and Horvitz HR. (1993). Cell, 75, 641 ± 652. Zha JP, Harada H, Yang E, Jockel J and Korsmeyer SJ. (1996). Cell, 87, 619 ±
Cell Death & Trophic Factors II. Steven McLoon Department of Neuroscience University of Minnesota
 Cell Death & Trophic Factors II Steven McLoon Department of Neuroscience University of Minnesota 1 Remember? Neurotrophins are cell survival factors that neurons get from their target cells! There is a
Cell Death & Trophic Factors II Steven McLoon Department of Neuroscience University of Minnesota 1 Remember? Neurotrophins are cell survival factors that neurons get from their target cells! There is a
Apoptosis EXTRA REQUIREMETS
 Apoptosis Introduction (SLIDE 1) Organisms develop from a single cell, and in doing so an anatomy has to be created. This process not only involves the creation of new cells, but also the removal of cells
Apoptosis Introduction (SLIDE 1) Organisms develop from a single cell, and in doing so an anatomy has to be created. This process not only involves the creation of new cells, but also the removal of cells
Massive loss of neurons in embryos occurs during normal development (!)
 Types of Cell Death Apoptosis (Programmed Cell Death) : Cell-Autonomous Stereotypic Rapid Clean (dead cells eaten) Necrosis : Not Self-Initiated Not Stereotypic Can Be Slow Messy (injury can spread) Apoptosis
Types of Cell Death Apoptosis (Programmed Cell Death) : Cell-Autonomous Stereotypic Rapid Clean (dead cells eaten) Necrosis : Not Self-Initiated Not Stereotypic Can Be Slow Messy (injury can spread) Apoptosis
Apoptosis: Death Comes for the Cell
 Apoptosis: Death Comes for the Cell Joe W. Ramos joeramos@hawaii.edu From Ingmar Bergman s The Seventh Seal 1 2 Mutations in proteins that regulate cell proliferation, survival and death can contribute
Apoptosis: Death Comes for the Cell Joe W. Ramos joeramos@hawaii.edu From Ingmar Bergman s The Seventh Seal 1 2 Mutations in proteins that regulate cell proliferation, survival and death can contribute
Bio 3411, Fall 2006, Lecture 19-Cell Death.
 Types of Cell Death Questions : Apoptosis (Programmed Cell Death) : Cell-Autonomous Stereotypic Rapid Clean (dead cells eaten) Necrosis : Not Self-Initiated Not Stereotypic Can Be Slow Messy (injury can
Types of Cell Death Questions : Apoptosis (Programmed Cell Death) : Cell-Autonomous Stereotypic Rapid Clean (dead cells eaten) Necrosis : Not Self-Initiated Not Stereotypic Can Be Slow Messy (injury can
APOPTOSIS REGULATOR BCL-2
 Papers on Anthropology Apoptosis regulator XXII, 2013, BCL-2 pp. 63 67 P. Hussar, M. Žuravskaja, M. Kärner APOPTOSIS REGULATOR BCL-2 Piret Hussar 1, Maria Žuravskaja 2, Martin Kärner 2 1 Institute of Anatomy,
Papers on Anthropology Apoptosis regulator XXII, 2013, BCL-2 pp. 63 67 P. Hussar, M. Žuravskaja, M. Kärner APOPTOSIS REGULATOR BCL-2 Piret Hussar 1, Maria Žuravskaja 2, Martin Kärner 2 1 Institute of Anatomy,
Cell Survival Curves (Chap. 3)
 Cell Survival Curves (Chap. 3) Example of pedigress of EMT6 mouse cells Clone The in vitro cell survival curve Cell Survival Assay Dye exclusion assay: membrane integrity MTT assay: respiratory activity
Cell Survival Curves (Chap. 3) Example of pedigress of EMT6 mouse cells Clone The in vitro cell survival curve Cell Survival Assay Dye exclusion assay: membrane integrity MTT assay: respiratory activity
Death signaling. Color PDF file of handouts can be found at Wu lab web-page:
 Death signaling Hao Wu References 1. Wang, X. The expanding role of mitochondria in apoptosis. Genes Dev 15, 2922-2933. (2001). 2. Fesik, S. W. Insights into programmed cell death through structural biology.
Death signaling Hao Wu References 1. Wang, X. The expanding role of mitochondria in apoptosis. Genes Dev 15, 2922-2933. (2001). 2. Fesik, S. W. Insights into programmed cell death through structural biology.
Programmed Cell Death
 Programmed Cell Death Dewajani Purnomosari Department of Histology and Cell Biology Faculty of Medicine Universitas Gadjah Mada d.purnomosari@ugm.ac.id What is apoptosis? a normal component of the development
Programmed Cell Death Dewajani Purnomosari Department of Histology and Cell Biology Faculty of Medicine Universitas Gadjah Mada d.purnomosari@ugm.ac.id What is apoptosis? a normal component of the development
IAPs. Structure of BIR2 Zn finger domain from XIAP
 IAPs The only known endogenous caspase inhibitors are members of a second family of proteins identified by apoptosis research, the IAP (inhibitor of apoptosis proteins) family. IAPs were originally described
IAPs The only known endogenous caspase inhibitors are members of a second family of proteins identified by apoptosis research, the IAP (inhibitor of apoptosis proteins) family. IAPs were originally described
Signal Transduction. Dr. Chaidir, Apt
 Signal Transduction Dr. Chaidir, Apt Background Complex unicellular organisms existed on Earth for approximately 2.5 billion years before the first multicellular organisms appeared.this long period for
Signal Transduction Dr. Chaidir, Apt Background Complex unicellular organisms existed on Earth for approximately 2.5 billion years before the first multicellular organisms appeared.this long period for
Mechanisms. Cell Death. Carol M. Troy, MD PhD August 24, 2009 PHENOMENOLOGY OF CELL DEATH I. DEVELOPMENT 8/20/2009
 Mechanisms of Cell Death Carol M. Troy, MD PhD August 24, 2009 PHENOMENOLOGY OF CELL DEATH I. DEVELOPMENT A. MORPHOGENESIS: SCULPTING/SHAPING STRUCTURES CREATION OF CAVITIES AND TUBES FUSION OF TISSUE
Mechanisms of Cell Death Carol M. Troy, MD PhD August 24, 2009 PHENOMENOLOGY OF CELL DEATH I. DEVELOPMENT A. MORPHOGENESIS: SCULPTING/SHAPING STRUCTURES CREATION OF CAVITIES AND TUBES FUSION OF TISSUE
Mechanisms of Cell Death
 Mechanisms of Cell Death CELL DEATH AND FORMATION OF THE SEMICIRCULAR CANALS Carol M. Troy, MD PhD August 24, 2009 FROM: Fekete et al., Development 124: 2451 (1997) PHENOMENOLOGY OF CELL DEATH I. DEVELOPMENT
Mechanisms of Cell Death CELL DEATH AND FORMATION OF THE SEMICIRCULAR CANALS Carol M. Troy, MD PhD August 24, 2009 FROM: Fekete et al., Development 124: 2451 (1997) PHENOMENOLOGY OF CELL DEATH I. DEVELOPMENT
Chang 1. Characterization of the Drosophila Ortholog of the Mammalian Anti-apoptotic Protein Aven
 Chang 1 Characterization of the Drosophila Ortholog of the Mammalian Anti-apoptotic Protein Aven Joy Chang Georgetown University Senior Digital Thesis May 1, 2006 Chang 2 Table of Contents BACKGROUND 4
Chang 1 Characterization of the Drosophila Ortholog of the Mammalian Anti-apoptotic Protein Aven Joy Chang Georgetown University Senior Digital Thesis May 1, 2006 Chang 2 Table of Contents BACKGROUND 4
Reception The target cell s detection of a signal coming from outside the cell May Occur by: Direct connect Through signal molecules
 Why Do Cells Communicate? Regulation Cells need to control cellular processes In multicellular organism, cells signaling pathways coordinate the activities within individual cells that support the function
Why Do Cells Communicate? Regulation Cells need to control cellular processes In multicellular organism, cells signaling pathways coordinate the activities within individual cells that support the function
ADAM FAMILY. ephrin A INTERAZIONE. Eph ADESIONE? PROTEOLISI ENDOCITOSI B A RISULTATO REPULSIONE. reverse. forward
 ADAM FAMILY - a family of membrane-anchored metalloproteases that are known as A Disintegrin And Metalloprotease proteins and are key components in protein ectodomain shedding Eph A INTERAZIONE B ephrin
ADAM FAMILY - a family of membrane-anchored metalloproteases that are known as A Disintegrin And Metalloprotease proteins and are key components in protein ectodomain shedding Eph A INTERAZIONE B ephrin
Stopping death cold Milton H Werner
 Minireview 879 Stopping death cold Milton H Werner The three-dimensional structure of Bcl-x L, an inhibitor of apoptosis, suggests how different combinations of proteins in the same family might be utilized
Minireview 879 Stopping death cold Milton H Werner The three-dimensional structure of Bcl-x L, an inhibitor of apoptosis, suggests how different combinations of proteins in the same family might be utilized
Apoptosis & Autophagy
 SPETSAI SUMMER SCHOOL 2010 Host Microbe Interactions Cellular Response to Infection: Apoptosis & Autophagy Christoph Dehio There are many ways to die Apoptosis: Historical perspective Process of programmed
SPETSAI SUMMER SCHOOL 2010 Host Microbe Interactions Cellular Response to Infection: Apoptosis & Autophagy Christoph Dehio There are many ways to die Apoptosis: Historical perspective Process of programmed
A. Incorrect! The Cell Cycle contains 4 distinct phases: (1) G 1, (2) S Phase, (3) G 2 and (4) M Phase.
 Molecular Cell Biology - Problem Drill 21: Cell Cycle and Cell Death Question No. 1 of 10 1. Which of the following statements about the cell cycle is correct? Question #1 (A) The Cell Cycle contains 3
Molecular Cell Biology - Problem Drill 21: Cell Cycle and Cell Death Question No. 1 of 10 1. Which of the following statements about the cell cycle is correct? Question #1 (A) The Cell Cycle contains 3
Apoptosis in Mammalian Cells
 Apoptosis in Mammalian Cells 7.16 2-10-05 Apoptosis is an important factor in many human diseases Cancer malignant cells evade death by suppressing apoptosis (too little apoptosis) Stroke damaged neurons
Apoptosis in Mammalian Cells 7.16 2-10-05 Apoptosis is an important factor in many human diseases Cancer malignant cells evade death by suppressing apoptosis (too little apoptosis) Stroke damaged neurons
RANK. Alternative names. Discovery. Structure. William J. Boyle* SUMMARY BACKGROUND
 RANK William J. Boyle* Department of Cell Biology, Amgen, Inc., One Amgen Center Drive, Thousand Oaks, CA 91320-1799, USA * corresponding author tel: 805-447-4304, fax: 805-447-1982, e-mail: bboyle@amgen.com
RANK William J. Boyle* Department of Cell Biology, Amgen, Inc., One Amgen Center Drive, Thousand Oaks, CA 91320-1799, USA * corresponding author tel: 805-447-4304, fax: 805-447-1982, e-mail: bboyle@amgen.com
 http://cwp.embo.org/w09-13/index.html Roads to Ruin is s o t p o ap healthy cell autophagic cell death ne cr os is Thanks to Seamus Martin! Evolution of apoptosis signalling cascades Inhibitor Adopted
http://cwp.embo.org/w09-13/index.html Roads to Ruin is s o t p o ap healthy cell autophagic cell death ne cr os is Thanks to Seamus Martin! Evolution of apoptosis signalling cascades Inhibitor Adopted
12/5/2014. The cell cycle and cell death. The cell cycle: cells duplicate their contents and divide
 The cell cycle and cell death The cell cycle: cells duplicate their contents and divide 1 The cell cycle may be divided into 4 phases Eucaryotic cell division: Mitosis (nuclear division) Cytokinesis (cell
The cell cycle and cell death The cell cycle: cells duplicate their contents and divide 1 The cell cycle may be divided into 4 phases Eucaryotic cell division: Mitosis (nuclear division) Cytokinesis (cell
S1 Gene ontology (GO) analysis of the network alignment results
 1 Supplementary Material for Effective comparative analysis of protein-protein interaction networks by measuring the steady-state network flow using a Markov model Hyundoo Jeong 1, Xiaoning Qian 1 and
1 Supplementary Material for Effective comparative analysis of protein-protein interaction networks by measuring the steady-state network flow using a Markov model Hyundoo Jeong 1, Xiaoning Qian 1 and
Drosophila Apoptosis and the Regulation of the Caspase Cascade
 Drosophila Apoptosis and the Regulation of the Caspase Cascade Kate Stafford March 18, 2005 Abstract The caspase cascade in Drosophila is controlled primarily by DIAP1 (Drosophila inhibitor of apoptosis),
Drosophila Apoptosis and the Regulation of the Caspase Cascade Kate Stafford March 18, 2005 Abstract The caspase cascade in Drosophila is controlled primarily by DIAP1 (Drosophila inhibitor of apoptosis),
Gene Control Mechanisms at Transcription and Translation Levels
 Gene Control Mechanisms at Transcription and Translation Levels Dr. M. Vijayalakshmi School of Chemical and Biotechnology SASTRA University Joint Initiative of IITs and IISc Funded by MHRD Page 1 of 9
Gene Control Mechanisms at Transcription and Translation Levels Dr. M. Vijayalakshmi School of Chemical and Biotechnology SASTRA University Joint Initiative of IITs and IISc Funded by MHRD Page 1 of 9
The Caspase System: a potential role in muscle proteolysis and meat quality? Tim Parr
 The Caspase System: a potential role in muscle proteolysis and meat quality? Tim Parr Caroline Kemp, Ron Bardsley,, Peter Buttery Division of Nutritional Sciences, School of Biosciences, University of
The Caspase System: a potential role in muscle proteolysis and meat quality? Tim Parr Caroline Kemp, Ron Bardsley,, Peter Buttery Division of Nutritional Sciences, School of Biosciences, University of
Lecture 10: Cyclins, cyclin kinases and cell division
 Chem*3560 Lecture 10: Cyclins, cyclin kinases and cell division The eukaryotic cell cycle Actively growing mammalian cells divide roughly every 24 hours, and follow a precise sequence of events know as
Chem*3560 Lecture 10: Cyclins, cyclin kinases and cell division The eukaryotic cell cycle Actively growing mammalian cells divide roughly every 24 hours, and follow a precise sequence of events know as
Chemical aspects of the cell. Compounds that induce cell proliferation, differentiation and death
 Chemical aspects of the cell Compounds that induce cell proliferation, differentiation and death Cell proliferation The cell cycle Chapter 17 Molecular Biology of the Cell 2 Cell cycle Chapter 17 Molecular
Chemical aspects of the cell Compounds that induce cell proliferation, differentiation and death Cell proliferation The cell cycle Chapter 17 Molecular Biology of the Cell 2 Cell cycle Chapter 17 Molecular
Sig2GRN: A Software Tool Linking Signaling Pathway with Gene Regulatory Network for Dynamic Simulation
 Sig2GRN: A Software Tool Linking Signaling Pathway with Gene Regulatory Network for Dynamic Simulation Authors: Fan Zhang, Runsheng Liu and Jie Zheng Presented by: Fan Wu School of Computer Science and
Sig2GRN: A Software Tool Linking Signaling Pathway with Gene Regulatory Network for Dynamic Simulation Authors: Fan Zhang, Runsheng Liu and Jie Zheng Presented by: Fan Wu School of Computer Science and
VDAC regulation by the Bcl-2 family of proteins
 Review (2000) 7, 1174 ± 1181 ã 2000 Macmillan Publishers Ltd All rights reserved 1350-9047/00 $15.00 www.nature.com/cdd VDAC regulation by the Bcl-2 family of proteins Y Tsujimoto*,1 and S Shimizu 1 1
Review (2000) 7, 1174 ± 1181 ã 2000 Macmillan Publishers Ltd All rights reserved 1350-9047/00 $15.00 www.nature.com/cdd VDAC regulation by the Bcl-2 family of proteins Y Tsujimoto*,1 and S Shimizu 1 1
Apoptosis: Death comes for the Cell. Apoptosis: Programmed Cell Death. Apoptosis. Necrosis
 poptosis: eath comes for the Cell Mutations in proteins that regulate cell proliferation, survival and death can contribute to oncogenesis Joe W. Ramos jramos@crch.hawaii.edu From Ingmar Bergman s The
poptosis: eath comes for the Cell Mutations in proteins that regulate cell proliferation, survival and death can contribute to oncogenesis Joe W. Ramos jramos@crch.hawaii.edu From Ingmar Bergman s The
Apoptosis and Carcinogenesis
 Death Becomes Us: Apoptosis and Carcinogenesis by Michele M. Cox Department of Biology, University of the Virgin Islands Part I What Is Cancer? Hi, Derek. Studying for your Cell and Molec final? asked
Death Becomes Us: Apoptosis and Carcinogenesis by Michele M. Cox Department of Biology, University of the Virgin Islands Part I What Is Cancer? Hi, Derek. Studying for your Cell and Molec final? asked
Regulation and signaling. Overview. Control of gene expression. Cells need to regulate the amounts of different proteins they express, depending on
 Regulation and signaling Overview Cells need to regulate the amounts of different proteins they express, depending on cell development (skin vs liver cell) cell stage environmental conditions (food, temperature,
Regulation and signaling Overview Cells need to regulate the amounts of different proteins they express, depending on cell development (skin vs liver cell) cell stage environmental conditions (food, temperature,
Mechanisms of Cell Death
 Mechanisms of Cell Death Etiology of cell death Major Factors Accidental Genetic Necrosis Apoptosis Necrosis: The sum of the morphologic changes that follow cell death in a living tissue or organ Apoptosis:
Mechanisms of Cell Death Etiology of cell death Major Factors Accidental Genetic Necrosis Apoptosis Necrosis: The sum of the morphologic changes that follow cell death in a living tissue or organ Apoptosis:
Lipniacki 2004 Ground Truth
 Abstract Lipniacki 2004 Ground Truth The two-feedback-loop regulatory module of nuclear factor kb (NF-kB) signaling pathway is modeled by means of ordinary differential equations. signaling pathway: https://en.wikipedia.org/wiki/signaling_pathway
Abstract Lipniacki 2004 Ground Truth The two-feedback-loop regulatory module of nuclear factor kb (NF-kB) signaling pathway is modeled by means of ordinary differential equations. signaling pathway: https://en.wikipedia.org/wiki/signaling_pathway
Monte Carlo study elucidates the type 1/type 2 choice in apoptotic death signaling in normal and cancer cells
 Cells 2013, 2, 1-x manuscripts; doi:10.3390/cells20x000x OPEN ACCESS cells ISSN 2073-4409 www.mdpi.com/journal/cells Article Monte Carlo study elucidates the type 1/type 2 choice in apoptotic death signaling
Cells 2013, 2, 1-x manuscripts; doi:10.3390/cells20x000x OPEN ACCESS cells ISSN 2073-4409 www.mdpi.com/journal/cells Article Monte Carlo study elucidates the type 1/type 2 choice in apoptotic death signaling
Proteases for Cell Suicide: Functions and Regulation of Caspases
 MICROBIOLOGY AND MOLECULAR BIOLOGY REVIEWS, Dec. 2000, p. 821 846 Vol. 64, No. 4 1092-2172/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Proteases for Cell Suicide:
MICROBIOLOGY AND MOLECULAR BIOLOGY REVIEWS, Dec. 2000, p. 821 846 Vol. 64, No. 4 1092-2172/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Proteases for Cell Suicide:
Delivery. Delivery Processes. Delivery Processes: Distribution. Ultimate Toxicant
 Delivery Ultimate Toxicant The chemical species that reacts with the endogenous target. Toxicity depends on the concentration (dose) of the ultimate toxicant at the target site Delivery Processes Absorption
Delivery Ultimate Toxicant The chemical species that reacts with the endogenous target. Toxicity depends on the concentration (dose) of the ultimate toxicant at the target site Delivery Processes Absorption
Study Guide 11 & 12 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Study Guide 11 & 12 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) The receptors for a group of signaling molecules known as growth factors are
Study Guide 11 & 12 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) The receptors for a group of signaling molecules known as growth factors are
A MATHEMATICAL MODEL FOR DYNAMIC SIMULATION OF APOPTOSIS PATHWAYS
 A MATHEMATICAL MODEL FOR DYNAMIC SIMULATION OF APOPTOSIS PATHWAYS Paolo Massari Department of Information Engineering Bioengineering University of Padova April 2014 Advisor: Gianna Maria Toolo Coadvisor:
A MATHEMATICAL MODEL FOR DYNAMIC SIMULATION OF APOPTOSIS PATHWAYS Paolo Massari Department of Information Engineering Bioengineering University of Padova April 2014 Advisor: Gianna Maria Toolo Coadvisor:
Apoptosis and Cancer. Carol M. Troy, MD, PhD October 26, 2016
 Apoptosis and Cancer Carol M. Troy, MD, PhD October 26, 2016 PHENOMENOLOGY OF CELL DEATH -1 I. DEVELOPMENT A. MORPHOGENESIS: SCULPTING/SHAPING STRUCTURES FUSION OF TISSUE MASSES (PALATE/NEURAL TUBE) CREATION
Apoptosis and Cancer Carol M. Troy, MD, PhD October 26, 2016 PHENOMENOLOGY OF CELL DEATH -1 I. DEVELOPMENT A. MORPHOGENESIS: SCULPTING/SHAPING STRUCTURES FUSION OF TISSUE MASSES (PALATE/NEURAL TUBE) CREATION
Mitochondria: execution central
 FEBS Letters 482 (2000) 6^12 FEBS 24121 Minireview Mitochondria: execution central Roberta A. Gottlieb* Molecular and Experimental Medicine, MEM 220, The Scripps Research Institute, 10550 North Torrey
FEBS Letters 482 (2000) 6^12 FEBS 24121 Minireview Mitochondria: execution central Roberta A. Gottlieb* Molecular and Experimental Medicine, MEM 220, The Scripps Research Institute, 10550 North Torrey
BCL-2 family members and the mitochondria in apoptosis
 REVIEW BCL-2 family members and the mitochondria in apoptosis Atan Gross, 1 James M. McDonnell, 2 and Stanley J. Korsmeyer 1,3 1 Departments of Pathology and Medicine, Dana-Farber Cancer Institute, Harvard
REVIEW BCL-2 family members and the mitochondria in apoptosis Atan Gross, 1 James M. McDonnell, 2 and Stanley J. Korsmeyer 1,3 1 Departments of Pathology and Medicine, Dana-Farber Cancer Institute, Harvard
Biol403 - Receptor Serine/Threonine Kinases
 Biol403 - Receptor Serine/Threonine Kinases The TGFβ (transforming growth factorβ) family of growth factors TGFβ1 was first identified as a transforming factor; however, it is a member of a family of structurally
Biol403 - Receptor Serine/Threonine Kinases The TGFβ (transforming growth factorβ) family of growth factors TGFβ1 was first identified as a transforming factor; however, it is a member of a family of structurally
COMPUTER SIMULATION OF DIFFERENTIAL KINETICS OF MAPK ACTIVATION UPON EGF RECEPTOR OVEREXPRESSION
 COMPUTER SIMULATION OF DIFFERENTIAL KINETICS OF MAPK ACTIVATION UPON EGF RECEPTOR OVEREXPRESSION I. Aksan 1, M. Sen 2, M. K. Araz 3, and M. L. Kurnaz 3 1 School of Biological Sciences, University of Manchester,
COMPUTER SIMULATION OF DIFFERENTIAL KINETICS OF MAPK ACTIVATION UPON EGF RECEPTOR OVEREXPRESSION I. Aksan 1, M. Sen 2, M. K. Araz 3, and M. L. Kurnaz 3 1 School of Biological Sciences, University of Manchester,
Pro-apoptotic cascade activates BID, which oligomerizes BAK or BAX into pores that result in the release of cytochrome c
 Review (2000) 7, 1166 ± 1173 ã 2000 Macmillan Publishers Ltd All rights reserved 1350-9047/00 $15.00 www.nature.com/cdd Pro-apoptotic cascade activates BID, which oligomerizes BAK or BAX into pores that
Review (2000) 7, 1166 ± 1173 ã 2000 Macmillan Publishers Ltd All rights reserved 1350-9047/00 $15.00 www.nature.com/cdd Pro-apoptotic cascade activates BID, which oligomerizes BAK or BAX into pores that
Cell Cell Communication in Development
 Biology 4361 Developmental Biology Cell Cell Communication in Development June 25, 2008 Cell Cell Communication Concepts Cells in developing organisms develop in the context of their environment, including
Biology 4361 Developmental Biology Cell Cell Communication in Development June 25, 2008 Cell Cell Communication Concepts Cells in developing organisms develop in the context of their environment, including
Apoptosis regulators and their role in tumorigenesis
 Biochimica et Biophysica Acta 1551 (2001) F1^F37 www.bba-direct.com Review Apoptosis regulators and their role in tumorigenesis Martin Zo«rnig a; *, Anne-Odile Hueber b, Wiebke Baum a, Gerard Evan c b
Biochimica et Biophysica Acta 1551 (2001) F1^F37 www.bba-direct.com Review Apoptosis regulators and their role in tumorigenesis Martin Zo«rnig a; *, Anne-Odile Hueber b, Wiebke Baum a, Gerard Evan c b
Validation of Petri Net Apoptosis Models Using P-Invariant Analysis
 2009 IEEE International Conference on Control and Automation Christchurch, New Zealand, December 9-11, 2009 WeMT5.1 Validation of Petri Net Apoptosis Models Using P-Invariant Analysis Ian Wee Jin Low,
2009 IEEE International Conference on Control and Automation Christchurch, New Zealand, December 9-11, 2009 WeMT5.1 Validation of Petri Net Apoptosis Models Using P-Invariant Analysis Ian Wee Jin Low,
16 CONTROL OF GENE EXPRESSION
 16 CONTROL OF GENE EXPRESSION Chapter Outline 16.1 REGULATION OF GENE EXPRESSION IN PROKARYOTES The operon is the unit of transcription in prokaryotes The lac operon for lactose metabolism is transcribed
16 CONTROL OF GENE EXPRESSION Chapter Outline 16.1 REGULATION OF GENE EXPRESSION IN PROKARYOTES The operon is the unit of transcription in prokaryotes The lac operon for lactose metabolism is transcribed
Life Sciences 1a: Section 3B. The cell division cycle Objectives Understand the challenges to producing genetically identical daughter cells
 Life Sciences 1a: Section 3B. The cell division cycle Objectives Understand the challenges to producing genetically identical daughter cells Understand how a simple biochemical oscillator can drive the
Life Sciences 1a: Section 3B. The cell division cycle Objectives Understand the challenges to producing genetically identical daughter cells Understand how a simple biochemical oscillator can drive the
Analysis of retinoblastoma protein function in the regulation of apoptosis
 Analysis of retinoblastoma protein function in the regulation of apoptosis Analyse der Funktion des Retinoblastoma-Proteins in der Apoptose-Regulation Dissertation zur Erlangung des akademischen Grades
Analysis of retinoblastoma protein function in the regulation of apoptosis Analyse der Funktion des Retinoblastoma-Proteins in der Apoptose-Regulation Dissertation zur Erlangung des akademischen Grades
 Cell-cell communication in development Review of cell signaling, paracrine and endocrine factors, cell death pathways, juxtacrine signaling, differentiated state, the extracellular matrix, integrins, epithelial-mesenchymal
Cell-cell communication in development Review of cell signaling, paracrine and endocrine factors, cell death pathways, juxtacrine signaling, differentiated state, the extracellular matrix, integrins, epithelial-mesenchymal
Examination paper for Bi3016 Molecular Cell Biology
 Department of Biology Examination paper for Bi3016 Molecular Cell Biology Academic contact during examination: Per Winge Phone: 99369359 Examination date: 20 th December 2017 Examination time (from-to):
Department of Biology Examination paper for Bi3016 Molecular Cell Biology Academic contact during examination: Per Winge Phone: 99369359 Examination date: 20 th December 2017 Examination time (from-to):
Cells to Tissues. Peter Takizawa Department of Cell Biology
 Cells to Tissues Peter Takizawa Department of Cell Biology From one cell to ensembles of cells. Multicellular organisms require individual cells to work together in functional groups. This means cells
Cells to Tissues Peter Takizawa Department of Cell Biology From one cell to ensembles of cells. Multicellular organisms require individual cells to work together in functional groups. This means cells
BIOCHEMICAL PATHWAYS OF CASPASE ACTIVATION DURING APOPTOSIS
 ?Annu. Rev. Cell Dev. Biol. 1999. 15:269 90 Copyright c 1999 by Annual Reviews. All rights reserved BIOCHEMICAL PATHWAYS OF CASPASE ACTIVATION DURING APOPTOSIS Imawati Budihardjo, Holt Oliver, Michael
?Annu. Rev. Cell Dev. Biol. 1999. 15:269 90 Copyright c 1999 by Annual Reviews. All rights reserved BIOCHEMICAL PATHWAYS OF CASPASE ACTIVATION DURING APOPTOSIS Imawati Budihardjo, Holt Oliver, Michael
Cytochrome c, cytochrome b 5 and electron transfer
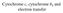 Cytochrome c, cytochrome b 5 and electron transfer Covalent bonds Cytochrome c is a major player in membrane associated electron transport systems in bacteria and mitochondria. Cytochrome c has two axial
Cytochrome c, cytochrome b 5 and electron transfer Covalent bonds Cytochrome c is a major player in membrane associated electron transport systems in bacteria and mitochondria. Cytochrome c has two axial
cellular division cell division cell cycle cell cycle kinases chapter 18-19
 cellular division chapter 18-19 cell division when? growth replacement of older cells production of specialized cells asexual reproduction sexual reproduction production of gametes prokaryotic binary fission
cellular division chapter 18-19 cell division when? growth replacement of older cells production of specialized cells asexual reproduction sexual reproduction production of gametes prokaryotic binary fission
Transport between cytosol and nucleus
 of 60 3 Gated trans Lectures 9-15 MBLG 2071 The n GATED TRANSPORT transport between cytoplasm and nucleus (bidirectional) controlled by the nuclear pore complex active transport for macro molecules e.g.
of 60 3 Gated trans Lectures 9-15 MBLG 2071 The n GATED TRANSPORT transport between cytoplasm and nucleus (bidirectional) controlled by the nuclear pore complex active transport for macro molecules e.g.
Regulation of apoptosis by BH3 domains in a cell-free system Sabina C. Cosulich*, Vivienne Worrall, Philip J. Hedge, Stephen Green and Paul R.
 Research Paper 913 Regulation of apoptosis by BH3 domains in a cell-free system Sabina C. Cosulich*, Vivienne Worrall, Philip J. Hedge, Stephen Green and Paul R. Clarke* Background: The family of proteins
Research Paper 913 Regulation of apoptosis by BH3 domains in a cell-free system Sabina C. Cosulich*, Vivienne Worrall, Philip J. Hedge, Stephen Green and Paul R. Clarke* Background: The family of proteins
Caenorhabditis elegans CED-4 stimulates CED-3 processing and CED-3-induced apoptosis Somasekar Seshagiri* and Lois K. Miller*
 Research Paper 455 Caenorhabditis elegans CED-4 stimulates CED-3 processing and CED-3-induced apoptosis Somasekar Seshagiri* and Lois K. Miller* Background: Programmed cell death or apoptosis is a key
Research Paper 455 Caenorhabditis elegans CED-4 stimulates CED-3 processing and CED-3-induced apoptosis Somasekar Seshagiri* and Lois K. Miller* Background: Programmed cell death or apoptosis is a key
International Journal of Pharma and Bio Sciences V1(2)2010 PETRI NET IMPLEMENTATION OF CELL SIGNALING FOR CELL DEATH
 SHRUTI JAIN *1, PRADEEP K. NAIK 2, SUNIL V. BHOOSHAN 1 1, 3 Department of Electronics and Communication Engineering, Jaypee University of Information Technology, Solan-173215, India 2 Department of Biotechnology
SHRUTI JAIN *1, PRADEEP K. NAIK 2, SUNIL V. BHOOSHAN 1 1, 3 Department of Electronics and Communication Engineering, Jaypee University of Information Technology, Solan-173215, India 2 Department of Biotechnology
CELL CYCLE AND GROWTH REGULATION
 CELL CYCLE AND GROWTH REGULATION The cell cycle is the set of stages through which a cell progresses from one division to the next. Interphase is the period between mitotic cell divisions; divided into
CELL CYCLE AND GROWTH REGULATION The cell cycle is the set of stages through which a cell progresses from one division to the next. Interphase is the period between mitotic cell divisions; divided into
Toxicological Targets. Russell L. Carr Department of Basic Sciences College of Veterinary Medicine
 Toxicological Targets Russell L. Carr Department of Basic Sciences College of Veterinary Medicine Toxicology Definitions = study of poisons Poison = any agent capable of producing a deleterious response
Toxicological Targets Russell L. Carr Department of Basic Sciences College of Veterinary Medicine Toxicology Definitions = study of poisons Poison = any agent capable of producing a deleterious response
N-Bak: a neuron-specific splice variant of Bak with anti-apoptotic properties
 N-Bak: a neuron-specific splice variant of Bak with anti-apoptotic properties Yun-Fu Sun Institute of Biotechnology and Division of Animal Physiology, Department of Biosciences, Faculty of Science, University
N-Bak: a neuron-specific splice variant of Bak with anti-apoptotic properties Yun-Fu Sun Institute of Biotechnology and Division of Animal Physiology, Department of Biosciences, Faculty of Science, University
Types of biological networks. I. Intra-cellurar networks
 Types of biological networks I. Intra-cellurar networks 1 Some intra-cellular networks: 1. Metabolic networks 2. Transcriptional regulation networks 3. Cell signalling networks 4. Protein-protein interaction
Types of biological networks I. Intra-cellurar networks 1 Some intra-cellular networks: 1. Metabolic networks 2. Transcriptional regulation networks 3. Cell signalling networks 4. Protein-protein interaction
Chem Lecture 10 Signal Transduction
 Chem 452 - Lecture 10 Signal Transduction 111202 Here we look at the movement of a signal from the outside of a cell to its inside, where it elicits changes within the cell. These changes are usually mediated
Chem 452 - Lecture 10 Signal Transduction 111202 Here we look at the movement of a signal from the outside of a cell to its inside, where it elicits changes within the cell. These changes are usually mediated
Mechanisms of Cell Death
 Mechanisms of Cell Death Etiology of cell death Major Factors Accidental Genetic Necrosis Apoptosis Necrosis: The sum of the morphologic changes that follow cell death in a living tissue or organ Apoptosis:
Mechanisms of Cell Death Etiology of cell death Major Factors Accidental Genetic Necrosis Apoptosis Necrosis: The sum of the morphologic changes that follow cell death in a living tissue or organ Apoptosis:
Richik N. Ghosh, Linnette Grove, and Oleg Lapets ASSAY and Drug Development Technologies 2004, 2:
 1 3/1/2005 A Quantitative Cell-Based High-Content Screening Assay for the Epidermal Growth Factor Receptor-Specific Activation of Mitogen-Activated Protein Kinase Richik N. Ghosh, Linnette Grove, and Oleg
1 3/1/2005 A Quantitative Cell-Based High-Content Screening Assay for the Epidermal Growth Factor Receptor-Specific Activation of Mitogen-Activated Protein Kinase Richik N. Ghosh, Linnette Grove, and Oleg
CHAPTER 3. Cell Structure and Genetic Control. Chapter 3 Outline
 CHAPTER 3 Cell Structure and Genetic Control Chapter 3 Outline Plasma Membrane Cytoplasm and Its Organelles Cell Nucleus and Gene Expression Protein Synthesis and Secretion DNA Synthesis and Cell Division
CHAPTER 3 Cell Structure and Genetic Control Chapter 3 Outline Plasma Membrane Cytoplasm and Its Organelles Cell Nucleus and Gene Expression Protein Synthesis and Secretion DNA Synthesis and Cell Division
The Inhibition of the Nuclear Transport of Caspase-7 by its Prodomain
 The Inhibition of the Nuclear Transport of Caspase-7 by its Prodomain Yoshio Yaoita Division of Embryology and Genetics, Laboratory for Amphibian Biology, Graduate School of Science, Hiroshima University,
The Inhibition of the Nuclear Transport of Caspase-7 by its Prodomain Yoshio Yaoita Division of Embryology and Genetics, Laboratory for Amphibian Biology, Graduate School of Science, Hiroshima University,
Activation of a receptor. Assembly of the complex
 Activation of a receptor ligand inactive, monomeric active, dimeric When activated by growth factor binding, the growth factor receptor tyrosine kinase phosphorylates the neighboring receptor. Assembly
Activation of a receptor ligand inactive, monomeric active, dimeric When activated by growth factor binding, the growth factor receptor tyrosine kinase phosphorylates the neighboring receptor. Assembly
6 Mechanotransduction
 6.1 Motivation The process of converting physical forces into biochemical signals and integrating these signals into the cellular response is referred to as mechnotransduction [11, 20]. To fully understand
6.1 Motivation The process of converting physical forces into biochemical signals and integrating these signals into the cellular response is referred to as mechnotransduction [11, 20]. To fully understand
Domain 6: Communication
 Domain 6: Communication 6.1: Cell communication processes share common features that reflect a shared evolutionary history. (EK3.D.1) 1. Introduction to Communication Communication requires the generation,
Domain 6: Communication 6.1: Cell communication processes share common features that reflect a shared evolutionary history. (EK3.D.1) 1. Introduction to Communication Communication requires the generation,
MAPK kinase kinase regulation of SAPK/JNK pathways
 MAPK kinase kinase regulation of SAPK/JNK pathways Lisa Stalheim and Gary L. Johnson Abstract SAPK/JNK members of the MAPK family are regulated by at least fourteen known MAPK kinase kinases (MKKKs). In
MAPK kinase kinase regulation of SAPK/JNK pathways Lisa Stalheim and Gary L. Johnson Abstract SAPK/JNK members of the MAPK family are regulated by at least fourteen known MAPK kinase kinases (MKKKs). In
Signal Transduction Phosphorylation Protein kinases. Misfolding diseases. Protein Engineering Lysozyme variants
 Signal Transduction Phosphorylation Protein kinases Misfolding diseases Protein Engineering Lysozyme variants Cells and Signals Regulation The cell must be able to respond to stimuli Cellular activities
Signal Transduction Phosphorylation Protein kinases Misfolding diseases Protein Engineering Lysozyme variants Cells and Signals Regulation The cell must be able to respond to stimuli Cellular activities
Death versus survival: functional interaction between the apoptotic and stress-inducible heat shock protein pathways
 Death versus survival: functional interaction between the apoptotic and stress-inducible heat shock protein pathways Helen M. Beere J Clin Invest. 2005;115(10):2633-2639. https://doi.org/10.1172/jci26471.
Death versus survival: functional interaction between the apoptotic and stress-inducible heat shock protein pathways Helen M. Beere J Clin Invest. 2005;115(10):2633-2639. https://doi.org/10.1172/jci26471.
Bio-Plex Pro RBM Apoptosis Assays
 Acute Phase Apoptosis Cancer Cardiovascular Disease Cytokines Chemokines, Growth Factors Diabetes Gene Expression Genotyping Immunoglobulin Isotyping MicroRNA Expression Bio-Plex Pro RBM Apoptosis Assays
Acute Phase Apoptosis Cancer Cardiovascular Disease Cytokines Chemokines, Growth Factors Diabetes Gene Expression Genotyping Immunoglobulin Isotyping MicroRNA Expression Bio-Plex Pro RBM Apoptosis Assays
The EGF Signaling Pathway! Introduction! Introduction! Chem Lecture 10 Signal Transduction & Sensory Systems Part 3. EGF promotes cell growth
 Chem 452 - Lecture 10 Signal Transduction & Sensory Systems Part 3 Question of the Day: Who is the son of Sevenless? Introduction! Signal transduction involves the changing of a cell s metabolism or gene
Chem 452 - Lecture 10 Signal Transduction & Sensory Systems Part 3 Question of the Day: Who is the son of Sevenless? Introduction! Signal transduction involves the changing of a cell s metabolism or gene
Cell-Cell Communication in Development
 Biology 4361 - Developmental Biology Cell-Cell Communication in Development June 23, 2009 Concepts Cell-Cell Communication Cells develop in the context of their environment, including: - their immediate
Biology 4361 - Developmental Biology Cell-Cell Communication in Development June 23, 2009 Concepts Cell-Cell Communication Cells develop in the context of their environment, including: - their immediate
AP Biology Gene Regulation and Development Review
 AP Biology Gene Regulation and Development Review 1. What does the regulatory gene code for? 2. Is the repressor by default active/inactive? 3. What changes the repressor activity? 4. What does repressor
AP Biology Gene Regulation and Development Review 1. What does the regulatory gene code for? 2. Is the repressor by default active/inactive? 3. What changes the repressor activity? 4. What does repressor
p53-mediated caspase activation Biochemical Society Transactions (200 I) Volume 29, part 6
 Biochemical Society Transactions (200 I) Volume 29, part 6 Mechanisms of p53-dependent apoptosis M. Schuler* and D. R. Green+' *Department of Medicine 111, Johannes Gutenbelg University, 55 I0 I Mainz,
Biochemical Society Transactions (200 I) Volume 29, part 6 Mechanisms of p53-dependent apoptosis M. Schuler* and D. R. Green+' *Department of Medicine 111, Johannes Gutenbelg University, 55 I0 I Mainz,
Graduate Institute t of fanatomy and Cell Biology
 Cell Adhesion 黃敏銓 mchuang@ntu.edu.tw Graduate Institute t of fanatomy and Cell Biology 1 Cell-Cell Adhesion and Cell-Matrix Adhesion actin filaments adhesion belt (cadherins) cadherin Ig CAMs integrin
Cell Adhesion 黃敏銓 mchuang@ntu.edu.tw Graduate Institute t of fanatomy and Cell Biology 1 Cell-Cell Adhesion and Cell-Matrix Adhesion actin filaments adhesion belt (cadherins) cadherin Ig CAMs integrin
ON/OFF and Beyond - A Boolean Model of Apoptosis
 Rebekka Schlatter 1 *, Kathrin Schmich 2, Ima Avalos Vizcarra 1,3, Peter Scheurich 4, Thomas Sauter 5, Christoph Borner 6, Michael Ederer 1,7, Irmgard Merfort 2, Oliver Sawodny 1 1 Institute for System
Rebekka Schlatter 1 *, Kathrin Schmich 2, Ima Avalos Vizcarra 1,3, Peter Scheurich 4, Thomas Sauter 5, Christoph Borner 6, Michael Ederer 1,7, Irmgard Merfort 2, Oliver Sawodny 1 1 Institute for System
Unraveling Apoptosis Signalling using Linear Control Methods: Linking the Loop Gain to Reverting the Decision to Undergo Apoptosis
 Preprints of the 9th International Symposium on Advanced Control of Chemical Processes The International Federation of Automatic Control TuPoster.3 Unraveling Apoptosis Signalling using Linear Control
Preprints of the 9th International Symposium on Advanced Control of Chemical Processes The International Federation of Automatic Control TuPoster.3 Unraveling Apoptosis Signalling using Linear Control
Host-Pathogen Interaction. PN Sharma Department of Plant Pathology CSK HPKV, Palampur
 Host-Pathogen Interaction PN Sharma Department of Plant Pathology CSK HPKV, Palampur-176062 PATHOGEN DEFENCE IN PLANTS A BIOLOGICAL AND MOLECULAR VIEW Two types of plant resistance response to potential
Host-Pathogen Interaction PN Sharma Department of Plant Pathology CSK HPKV, Palampur-176062 PATHOGEN DEFENCE IN PLANTS A BIOLOGICAL AND MOLECULAR VIEW Two types of plant resistance response to potential
16 The Cell Cycle. Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization
 The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
AP Curriculum Framework with Learning Objectives
 Big Ideas Big Idea 1: The process of evolution drives the diversity and unity of life. AP Curriculum Framework with Learning Objectives Understanding 1.A: Change in the genetic makeup of a population over
Big Ideas Big Idea 1: The process of evolution drives the diversity and unity of life. AP Curriculum Framework with Learning Objectives Understanding 1.A: Change in the genetic makeup of a population over
CLARP, a death effector domain-containing protein interacts with caspase-8 and regulates apoptosis
 Proc. Natl. Acad. Sci. USA Vol. 94, pp. 10717 10722, September 1997 Cell Biology CLARP, a death effector domain-containing protein interacts with caspase-8 and regulates apoptosis NAOHIRO INOHARA, TAKEYOSHI
Proc. Natl. Acad. Sci. USA Vol. 94, pp. 10717 10722, September 1997 Cell Biology CLARP, a death effector domain-containing protein interacts with caspase-8 and regulates apoptosis NAOHIRO INOHARA, TAKEYOSHI
Analysis and Simulation of Biological Systems
 Analysis and Simulation of Biological Systems Dr. Carlo Cosentino School of Computer and Biomedical Engineering Department of Experimental and Clinical Medicine Università degli Studi Magna Graecia Catanzaro,
Analysis and Simulation of Biological Systems Dr. Carlo Cosentino School of Computer and Biomedical Engineering Department of Experimental and Clinical Medicine Università degli Studi Magna Graecia Catanzaro,
TRAIL-induced apoptosis requires Bax-dependent mitochondrial release of Smac/DIABLO
 TRAIL-induced apoptosis requires Bax-dependent mitochondrial release of Smac/DIABLO Yibin Deng, Yahong Lin, and Xiangwei Wu 1 Huffington Center on Aging and Department of Molecular and Cellular Biology,
TRAIL-induced apoptosis requires Bax-dependent mitochondrial release of Smac/DIABLO Yibin Deng, Yahong Lin, and Xiangwei Wu 1 Huffington Center on Aging and Department of Molecular and Cellular Biology,
COMMENTARY Roles of caspases in apoptosis, development, and cytokine maturation revealed by homozygous gene deficiencies
 Journal of Cell Science 113, 753-757 (2000) Printed in Great Britain The Company of Biologists Limited 2000 JCS0884 753 COMMENTARY Roles of caspases in apoptosis, development, and cytokine maturation revealed
Journal of Cell Science 113, 753-757 (2000) Printed in Great Britain The Company of Biologists Limited 2000 JCS0884 753 COMMENTARY Roles of caspases in apoptosis, development, and cytokine maturation revealed
cellular division cell division cell cycle cell cycle kinases chapter 18-19
 cellular division chapter 18-19 cell division when? growth replacement of older cells production of specialized cells asexual reproduction sexual reproduction production of gametes prokaryotic binary fission
cellular division chapter 18-19 cell division when? growth replacement of older cells production of specialized cells asexual reproduction sexual reproduction production of gametes prokaryotic binary fission
Modelling intrinsic apoptosis
 Modelling intrinsic apoptosis In this part of the course we will model the steps that take place inside a cell when intrinsic apoptosis is triggered. The templates and instructions for preparing the models
Modelling intrinsic apoptosis In this part of the course we will model the steps that take place inside a cell when intrinsic apoptosis is triggered. The templates and instructions for preparing the models
Embedded together: The life and death consequences of interaction of the Bcl-2 family with membranes
 DOI 10.1007/s10495-007-0746-4 Embedded together: The life and death consequences of interaction of the Bcl-2 family with membranes Brian Leber Jialing Lin David W. Andrews Published online: 15 February
DOI 10.1007/s10495-007-0746-4 Embedded together: The life and death consequences of interaction of the Bcl-2 family with membranes Brian Leber Jialing Lin David W. Andrews Published online: 15 February
Role of mitochondria transmembrane potential in lymphocyte
 Original Article Fac Med Baghdad 2009; Vol. 51, No. 1 Received Mar.2008 Accepted May 2008 Role of mitochondria transmembrane potential in lymphocyte apoptosis * PhD Summary: Background: Mitochondria play
Original Article Fac Med Baghdad 2009; Vol. 51, No. 1 Received Mar.2008 Accepted May 2008 Role of mitochondria transmembrane potential in lymphocyte apoptosis * PhD Summary: Background: Mitochondria play
What are mitochondria?
 What are mitochondria? What are mitochondria? An intracellular organelle. There are 100 to 1000s of mitochondria/cell. Most mitochondria come from the mother. Mitochondria have their own DNA Mitochondria
What are mitochondria? What are mitochondria? An intracellular organelle. There are 100 to 1000s of mitochondria/cell. Most mitochondria come from the mother. Mitochondria have their own DNA Mitochondria
BCL-2, BCL-X L Sequester BH3 Domain-Only Molecules Preventing BAX- and BAK-Mediated Mitochondrial Apoptosis
 Molecular Cell, Vol. 8, 705 711, September, 2001, Copyright 2001 by Cell Press BCL-2, BCL-X L Sequester BH3 Domain-Only Molecules Preventing BAX- and BAK-Mediated Mitochondrial Apoptosis Emily H.-Y. A.
Molecular Cell, Vol. 8, 705 711, September, 2001, Copyright 2001 by Cell Press BCL-2, BCL-X L Sequester BH3 Domain-Only Molecules Preventing BAX- and BAK-Mediated Mitochondrial Apoptosis Emily H.-Y. A.
A A A A B B1
 LEARNING OBJECTIVES FOR EACH BIG IDEA WITH ASSOCIATED SCIENCE PRACTICES AND ESSENTIAL KNOWLEDGE Learning Objectives will be the target for AP Biology exam questions Learning Objectives Sci Prac Es Knowl
LEARNING OBJECTIVES FOR EACH BIG IDEA WITH ASSOCIATED SCIENCE PRACTICES AND ESSENTIAL KNOWLEDGE Learning Objectives will be the target for AP Biology exam questions Learning Objectives Sci Prac Es Knowl
