Triangular Diagrams. 1. On the diagram above, plot the following (mostly mineral) compositions: silicon quartz corundum kyanite
|
|
|
- Mitchell Holmes
- 6 years ago
- Views:
Transcription
1 Triangular Diagrams Note: For this exercise, everything is plotted using mole%. Sometimes it is done with wt% but that just adds unnecessary complications. 1. On the diagram above, plot the following (mostly mineral) compositions: silicon quartz corundum kyanite
2 2. On the diagram above, plot the following (mostly mineral) compositions: silicon quartz corundum kyanite grossular, Ca3Al2Si3O12 CaTs, CaAl2SiO6 (Beware of a possible trick question.)
3 The Lever Rule Sometimes you won t have all those % lines on your diagram. You can draw them in, or use the lever rule. 3. Where on the diagram do compositions plot that contain 0% A? 0%B? 0%C? 4. Where on the diagram do compositions plot that contain 50% A? 50%B? 50%C? 5. What %A, %B, and %C are represented by the black dot? (Figure out real number values.)
4 4. What mineral (name it!) is plotted on this diagram? (You may wish to use a ruler.) 5. What is a eutectic? On this binary diagram (below), where is the binary Di-An eutectic?
5 Standard petrologic triangular diagrams are ternary compositional diagrams with a different component at each of the three corners. Liquidus temperatures (the temperatures at which crystallization begins upon cooling of a magma) are contoured much like a topographic map. The diagrams often contain eutectics and cotectics, and may contain peritectics. Numbers in the diagram below are temperatures in centigrade. 6. On the ternary diagram above where is the binary Di-An eutectic? 7. On the ternary diagram above, where is the ternary Di-An-Fo eutectic?
6 8. Using the ternary diagram on the previous page as a reference, sketch the Di-An binary phase diagram as best you can to scale with temperatures of end members and any eutectic (if present) labeled). 9. Consider the diagram shown. If a magma (of any composition) cools, what is the lowest temperature it can reach before it is all solidified? If a rock composed of a mix of Di, An, and Fo is heated, at what temperature will it begin to melt? If a rock contains some amounts of Di, An, and Fo, what is the highest temperature it can reach without being completely melted?
7 10. Triangular diagrams describe melting/crystallization in model chemical systems that approximate real rocks. What kind of rocks (name some; e.g., diorite) does the diagram on the previous page best describe? You may wish to look at the IUGS classification scheme be sure to look at the correct diagram. 11. Consider a rock containing Di, An, and Fo that has overall composition A, shown below. What %s Di, An, and Fo does this rock contain? 12. Consider a plutonic rock with composition A. What name would an igneous petrologist give to such a rock? (Consult the IUGS classification scheme.) 13. If a magma of composition A cools to 1640, it will begin to crystallize forsterite. What happens after that? Describe what happens and at what temperatures. 14. If magma A crystallizes completely, what will be present after it is all solid?
8 15. A rock of composition A is heated. At what temperature will melting begin? Which mineral will melt first? What will be the composition of the first drop of melt? If melting continues, at what temperature will the rock be entirely melted? Will all minerals in this rock melt at the same time? Or will they melt in sequence? Explain. 16. Describe in general terms how melting proceeds until the rock is entirely molten. Use arrows on the diagram to show how the magma composition changes during melting.
9 Use the lever rule again! 17. Suppose melt A cools and crystallizes. At what temperature will it be approximately 50% solidified? 18. The melt will change composition until it reaches the eutectic. What % melt will be present just when the composition reaches the eutectic? What minerals will be present just before melt composition reaches the eutectic? Just after the melt composition reaches the eutectic, what minerals will be present? 19. Suppose the melt reaches the eutectic and then the remaining melt squirts off (is separated from the crystals). What rock (name it) will that melt produce when IT crystallizes? And, what kind of rock is left behind?
Introductory Statement:
 The use of visualization and sketches of thin sections to encourage a better understanding of phase diagrams: Binary and ternary phase diagram exercises Jennifer M. Wenner Drew S. Coleman Introductory
The use of visualization and sketches of thin sections to encourage a better understanding of phase diagrams: Binary and ternary phase diagram exercises Jennifer M. Wenner Drew S. Coleman Introductory
Hirsch - Binary phase diagrams problems KEY
 Hirsch - Binary phase diagrams problems KEY Name Question 1. 1A. Using the phase rule, determine the degrees of freedom: a. at point e F = 2-3 + 1 = 0 b. within the field A + B F = 2-2 + 1 = 1 c. within
Hirsch - Binary phase diagrams problems KEY Name Question 1. 1A. Using the phase rule, determine the degrees of freedom: a. at point e F = 2-3 + 1 = 0 b. within the field A + B F = 2-2 + 1 = 1 c. within
Common non-silicate planetary minerals
 Common non-silicate planetary minerals Many of the non-silicate minerals are simple oxides. Corundum Al2O3 Al2+3 O3-2 Rutile Ti2O3 Ti2+3 O3-2 Ilmenite FeTiO3 Fe+3Ti+3O3-2 Hematite Fe2O3 Fe2+3 O3-2 Families
Common non-silicate planetary minerals Many of the non-silicate minerals are simple oxides. Corundum Al2O3 Al2+3 O3-2 Rutile Ti2O3 Ti2+3 O3-2 Ilmenite FeTiO3 Fe+3Ti+3O3-2 Hematite Fe2O3 Fe2+3 O3-2 Families
TWO COMPONENT (BINARY) PHASE DIAGRAMS. Experimental Determination of 2-Component Phase Diagrams
 Page 1 of 12 EENS 211 Earth Materials Tulane University Prof. Stephen A. Nelson TWO COMPONENT (BINARY) PHASE DIAGRAMS This document last updated on 08-Oct-2003 Experimental Determination of 2-Component
Page 1 of 12 EENS 211 Earth Materials Tulane University Prof. Stephen A. Nelson TWO COMPONENT (BINARY) PHASE DIAGRAMS This document last updated on 08-Oct-2003 Experimental Determination of 2-Component
C = 3: Ternary Systems: Example 1: Ternary Eutectic
 Phase Equilibrium C = 3: Ternary Systems: Example 1: Ternary Eutectic Adding components, becomes increasingly difficult to depict 1-C: P - T diagrams easy 2-C: isobaric T-X, isothermal P-X 3-C:?? Still
Phase Equilibrium C = 3: Ternary Systems: Example 1: Ternary Eutectic Adding components, becomes increasingly difficult to depict 1-C: P - T diagrams easy 2-C: isobaric T-X, isothermal P-X 3-C:?? Still
DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER
 Geol 2312 Igneous and Metamorphic Petrology Spring 2009 Name DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER Objective: This exercise is intended to improve understanding
Geol 2312 Igneous and Metamorphic Petrology Spring 2009 Name DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER Objective: This exercise is intended to improve understanding
Interpreting Phase Diagrams
 Interpreting Phase Diagrams Understanding chemical reactions requires that we know something about how materials behave as the temperature and pressure change. For a single component (like quartz or ice)
Interpreting Phase Diagrams Understanding chemical reactions requires that we know something about how materials behave as the temperature and pressure change. For a single component (like quartz or ice)
To get you thinking What natural process is responsible for the appearance of these rocks? Rocks and the Rock Cycle
 To get you thinking What natural process is responsible for the appearance of these rocks? Rocks and the Rock Cycle Bell Ringer Name the 3 types of rock. Is one type of rock able to change into a different
To get you thinking What natural process is responsible for the appearance of these rocks? Rocks and the Rock Cycle Bell Ringer Name the 3 types of rock. Is one type of rock able to change into a different
1 - C Systems. The system H 2 O. Heat an ice at 1 atm from-5 to 120 o C. Heat vs. Temperature
 1 - C Systems The system H 2 O Heat an ice at 1 atm from-5 to 120 o C Heat vs. Temperature Fig. 6.7. After Bridgman (1911) Proc. Amer. Acad. Arts and Sci., 5, 441-513; (1936) J. Chem. Phys., 3, 597-605;
1 - C Systems The system H 2 O Heat an ice at 1 atm from-5 to 120 o C Heat vs. Temperature Fig. 6.7. After Bridgman (1911) Proc. Amer. Acad. Arts and Sci., 5, 441-513; (1936) J. Chem. Phys., 3, 597-605;
CERAMIC GLAZING as an IGNEOUS PROCESS
 GEOL 640: Geology through Global Arts and Artifacts CERAMIC GLAZING as an IGNEOUS PROCESS GLAZE COMPONENTS A glaze is a waterproof silica glass on the surface of a ceramic pot, and was first produced by
GEOL 640: Geology through Global Arts and Artifacts CERAMIC GLAZING as an IGNEOUS PROCESS GLAZE COMPONENTS A glaze is a waterproof silica glass on the surface of a ceramic pot, and was first produced by
A. One component system (c = 1)
 A. One component system (c = 1) Example: SiO 2 system. Since all phases in this system have the same composition, there are no compositional variables to consider. Phase equilibria can be shown completely
A. One component system (c = 1) Example: SiO 2 system. Since all phases in this system have the same composition, there are no compositional variables to consider. Phase equilibria can be shown completely
Lab 4 - Identification of Igneous Rocks
 Lab 4 - Identification of Igneous Rocks Page - Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly recognize
Lab 4 - Identification of Igneous Rocks Page - Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly recognize
Phase Diagrams and Chemographic Diagrams C:\Courses\320\fall2005\inclass, etc\57-projections.wpd; October 9, 2003 (6:09pm)
 1 Phase Diagrams and Chemographic Diagrams C:\Courses\320\fall2005\inclass, etc\57-projections.wpd; October 9, 2003 (6:09pm) Recall the phase rule: C + 2 = P + F. At a point on a phase diagram where two
1 Phase Diagrams and Chemographic Diagrams C:\Courses\320\fall2005\inclass, etc\57-projections.wpd; October 9, 2003 (6:09pm) Recall the phase rule: C + 2 = P + F. At a point on a phase diagram where two
Differentiation of Magmas By Fractional Crystallization
 Wirth Magmatic Differentiation Using M&M s 1 HANDOUT Differentiation of Magmas By Fractional Crystallization Objective The objective of this exercise is to gain first-hand knowledge of the process of magmatic
Wirth Magmatic Differentiation Using M&M s 1 HANDOUT Differentiation of Magmas By Fractional Crystallization Objective The objective of this exercise is to gain first-hand knowledge of the process of magmatic
RR#7 - Free Response
 Base your answers to questions 1 through 3 on the table below and on your knowledge of Earth science. The table shows the elements and their percent compositions by mass in the five minerals present in
Base your answers to questions 1 through 3 on the table below and on your knowledge of Earth science. The table shows the elements and their percent compositions by mass in the five minerals present in
Exam I. 1. (10 points) Give the following optical properties for the minerals listed below.
 GLY306 Petrology Exam I 1. (10 points) Give the following optical properties for the minerals listed below. Color/pleochroism Extinction angle Cleavage angle Twining Refractive index Hornblende Plagioclase
GLY306 Petrology Exam I 1. (10 points) Give the following optical properties for the minerals listed below. Color/pleochroism Extinction angle Cleavage angle Twining Refractive index Hornblende Plagioclase
2-C Eutectic Systems
 Phase Equilibrium 2-C Eutectic Systems Example: Diopside - Anorthite No solid solution * For system at this T, X, phase(s) plot where? Fig. 6.11. Isobaric T-X phase diagram at atmospheric pressure. After
Phase Equilibrium 2-C Eutectic Systems Example: Diopside - Anorthite No solid solution * For system at this T, X, phase(s) plot where? Fig. 6.11. Isobaric T-X phase diagram at atmospheric pressure. After
T-X Diagrams Answers C:\Courses\320\fall2007\in class\5000-t-x ExerciseAnswers.wpd; September 25, 2003 (11:45am) Problems
 1 T-X Diagrams Answers C:\Courses\320\fall2007\in class\5000-t-x ExerciseAnswers.wpd; September 25, 2003 (11:45am) Problems Problem 1. Look at Figure 10. One reaction (that plots as a horizontal line)
1 T-X Diagrams Answers C:\Courses\320\fall2007\in class\5000-t-x ExerciseAnswers.wpd; September 25, 2003 (11:45am) Problems Problem 1. Look at Figure 10. One reaction (that plots as a horizontal line)
EESC 4701: Igneous and Metamorphic Petrology IGNEOUS ROCK CLASSIFICATION LAB 2 HANDOUT
 EESC 4701: Igneous and Metamorphic Petrology IGNEOUS ROCK CLASSIFICATION LAB 2 HANDOUT Sources: University of Washington, Texas A&M University, University of Southern Alabama What is an igneous rock (a
EESC 4701: Igneous and Metamorphic Petrology IGNEOUS ROCK CLASSIFICATION LAB 2 HANDOUT Sources: University of Washington, Texas A&M University, University of Southern Alabama What is an igneous rock (a
UNIVERSITI SAINS MALAYSIA
 UNIVERSITI SAINS MALAYSIA First Semester Examination Academic Session 2011/2012 January 2012 EBB 512/3 Phase Diagram and Phase Equilibra Duration : 3 hours Please ensure that this examination paper contains
UNIVERSITI SAINS MALAYSIA First Semester Examination Academic Session 2011/2012 January 2012 EBB 512/3 Phase Diagram and Phase Equilibra Duration : 3 hours Please ensure that this examination paper contains
Lecture 37. Igneous geochemistry. Crystallization Let's look at how we use differences in element distribution to understand crystallization process.
 Reading White Chapter 7 Lecture 37 Igneous geochemistry Today: Using trace elements to study 1. crystallization 2. melting Crystallization Let's look at how we use differences in element distribution to
Reading White Chapter 7 Lecture 37 Igneous geochemistry Today: Using trace elements to study 1. crystallization 2. melting Crystallization Let's look at how we use differences in element distribution to
Feldspars. Structure. The feldspars are by far the most abundant group of minerals and are found in igneous, metamorphic and many sedimentary rocks.
 Feldspars The feldspars are by far the most abundant group of minerals and are found in igneous, metamorphic and many sedimentary rocks. Structure Felsdpars are framework silicates where each silica tetrahedra
Feldspars The feldspars are by far the most abundant group of minerals and are found in igneous, metamorphic and many sedimentary rocks. Structure Felsdpars are framework silicates where each silica tetrahedra
LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES
 Geology 316 (Petrology) (03/26/2012) Name LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES INTRODUCTION Ultramafic rocks are igneous rocks containing less than 10% felsic minerals (quartz + feldspars
Geology 316 (Petrology) (03/26/2012) Name LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES INTRODUCTION Ultramafic rocks are igneous rocks containing less than 10% felsic minerals (quartz + feldspars
Internet Interactive Rock Cycle
 Internet Interactive Rock Cycle Directions: Go to the website http://www.uky.edu/as/geology/howell/goodies/elearning/module05swf.swf and answer the questions below. Part I: Igneous Rocks (click on Igneous
Internet Interactive Rock Cycle Directions: Go to the website http://www.uky.edu/as/geology/howell/goodies/elearning/module05swf.swf and answer the questions below. Part I: Igneous Rocks (click on Igneous
Phase Equilibrium. Phase Rule. Phase Diagram
 Phase Equilibrium Phase Rule Phase Diagram Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS Prof. Paper, 1004. Makaopuhi Lava Lake
Phase Equilibrium Phase Rule Phase Diagram Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS Prof. Paper, 1004. Makaopuhi Lava Lake
Unit 3 Lesson 2 The Rock Cycle. Copyright Houghton Mifflin Harcourt Publishing Company
 Let s Rock! What is rock? Rock is a naturally occurring solid mixture of one or more minerals that may also include organic matter. Most rock is made of minerals, but some rock is made of nonmineral material
Let s Rock! What is rock? Rock is a naturally occurring solid mixture of one or more minerals that may also include organic matter. Most rock is made of minerals, but some rock is made of nonmineral material
Mineral Stability and Phase Diagrams Introduction
 1 of 10 10/10/2002 2:50 PM Prof. Stephen A. Nelson Geology 211 Tulane University Mineralogy and Phase Diagrams Introduction This document last updated on 10-Oct-2002 As we discussed previously, there are
1 of 10 10/10/2002 2:50 PM Prof. Stephen A. Nelson Geology 211 Tulane University Mineralogy and Phase Diagrams Introduction This document last updated on 10-Oct-2002 As we discussed previously, there are
Magma Formation and Behavior
 Magma Formation and Behavior Introduction: The study of body waves as they pass through Earth's interior provides strong evidence that the Earth's mantle is composed almost entirely of solid ultramafic
Magma Formation and Behavior Introduction: The study of body waves as they pass through Earth's interior provides strong evidence that the Earth's mantle is composed almost entirely of solid ultramafic
Igneous Rocks. How Do Igneous Rocks Form? Liquid to Solid
 Igneous Rocks Answering the Big Question The activities in this lesson will help students answer the Big Question by modeling the result of different cooling rates of magma and lava and by learning how
Igneous Rocks Answering the Big Question The activities in this lesson will help students answer the Big Question by modeling the result of different cooling rates of magma and lava and by learning how
Lab 3 - Identification of Igneous Rocks
 Lab 3 - Identification of Igneous Rocks Page - 1 Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly
Lab 3 - Identification of Igneous Rocks Page - 1 Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly
OUTCOMES BASED LEARNING MATRIX. Course: Physical Geology Department: _Physical Science. Study the text and lecture material
 OUTCOMES BASED LEARNING MATRIX Course: Physical Geology Department: _Physical Science Physical Geology This course is intended to acquaint students with the physical structure of the Earth, the nature
OUTCOMES BASED LEARNING MATRIX Course: Physical Geology Department: _Physical Science Physical Geology This course is intended to acquaint students with the physical structure of the Earth, the nature
PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS
 PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS PLATE TECTONICS TO IGNEOUS ROCKS Internal Heat Seafloor Spreading/Plate Tectonics Volcanism Plate Boundary Intra-plate (hot spot) Divergent Convergent Igneous
PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS PLATE TECTONICS TO IGNEOUS ROCKS Internal Heat Seafloor Spreading/Plate Tectonics Volcanism Plate Boundary Intra-plate (hot spot) Divergent Convergent Igneous
Phase Equilibria C:\a-StudioClassroom\minex20.doc; July 7, 2005
 1 Phase Equilibria C:\a-StudioClassroom\minex20.doc; July 7, 2005 S/mole V/mole E/mole J/mol-K cc/mol J/mol grossular 255.5 125.3-6656700 quartz 41.46 22.688-910700 anorthite 199.3 100.79-4243040 wollastonite
1 Phase Equilibria C:\a-StudioClassroom\minex20.doc; July 7, 2005 S/mole V/mole E/mole J/mol-K cc/mol J/mol grossular 255.5 125.3-6656700 quartz 41.46 22.688-910700 anorthite 199.3 100.79-4243040 wollastonite
Giant Crystals from Maine
 Maine Geologic Facts and Localities May, 2006 Text by Woodrow B. Thompson, Department of Agriculture, Conservation & Forestry 1 Photo by Pegmatites The 2006 Maine Mineral Symposium had a talk and exhibits
Maine Geologic Facts and Localities May, 2006 Text by Woodrow B. Thompson, Department of Agriculture, Conservation & Forestry 1 Photo by Pegmatites The 2006 Maine Mineral Symposium had a talk and exhibits
Differentiation of Magmas By Fractional Crystallization Modified from Karl Wirth, rev. July 2011
 M&M s Magma Chamber 1 Differentiation of Magmas By Fractional Crystallization Modified from Karl Wirth, rev. July 2011 Objective The objective of this exercise is to gain first-hand knowledge of the process
M&M s Magma Chamber 1 Differentiation of Magmas By Fractional Crystallization Modified from Karl Wirth, rev. July 2011 Objective The objective of this exercise is to gain first-hand knowledge of the process
Chapter 6: The Phase Rule and One and Two-Component Systems aka Phase Equilibria
 Chapter 6: The Phase Rule and One and Two-Component Systems aka Phase Equilibria Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS
Chapter 6: The Phase Rule and One and Two-Component Systems aka Phase Equilibria Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS
3.012 PS 6 THERMODYANMICS SOLUTIONS Issued: Fall 2005 Due:
 3.012 PS 6 THERMODYANMICS SOLUTIONS 3.012 Issued: 11.28.05 Fall 2005 Due: THERMODYNAMICS 1. Building a binary phase diagram. Given below are data for a binary system of two materials A and B. The two components
3.012 PS 6 THERMODYANMICS SOLUTIONS 3.012 Issued: 11.28.05 Fall 2005 Due: THERMODYNAMICS 1. Building a binary phase diagram. Given below are data for a binary system of two materials A and B. The two components
e. another name for 0 elevation! f. imaginary lines that connect points of equal elevation! depression! steep slope!
 Name: Date: Period: PART 1 - Topography Basics A. Match the definition to the vocabulary word. contour interval a. shows the shape, surface features & elevation of the land topographic maps b. evenly spaced
Name: Date: Period: PART 1 - Topography Basics A. Match the definition to the vocabulary word. contour interval a. shows the shape, surface features & elevation of the land topographic maps b. evenly spaced
Edible Changing Rock (Starbursts)
 Name: Date: Hour: Lab Conduct Grade: 10 9 8 7 6 5 4 3 2 1 0 Edible Changing Rock (Starbursts) Most rocks are made of minerals. All rock begin as igneous rock which is formed when magma or lava cools and
Name: Date: Hour: Lab Conduct Grade: 10 9 8 7 6 5 4 3 2 1 0 Edible Changing Rock (Starbursts) Most rocks are made of minerals. All rock begin as igneous rock which is formed when magma or lava cools and
T-X Diagrams C:\Courses\320\fall2007\in class\5000-t-x Exercise.wpd; September 25, 2003 (11:45am)
 1 T-X Diagrams C:\Courses\320\fall2007\in class\5000-t-x Exercise.wpd; September 25, 2003 (11:45am) T-X diagrams are most often used for describing metamorphism of carbonate-rich rocks (marbles or marls)
1 T-X Diagrams C:\Courses\320\fall2007\in class\5000-t-x Exercise.wpd; September 25, 2003 (11:45am) T-X diagrams are most often used for describing metamorphism of carbonate-rich rocks (marbles or marls)
Plate tectonics, rock cycle
 Dikes, Antarctica Rock Cycle Plate tectonics, rock cycle The Rock Cycle A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one
Dikes, Antarctica Rock Cycle Plate tectonics, rock cycle The Rock Cycle A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one
UNIVERSITY OF EDINBURGH. College of Science and Engineering School of GeoSciences. Earth Materials UO4824 DEGREE EXAMINATION (MOCK) xxxxxxxxxxxxxxxxx
 UNIVERSITY OF EDINBURGH College of Science and Engineering School of GeoSciences Earth Materials UO4824 DEGREE EXAMINATION (MOCK) xxxxxxxxxxxxxxxxxxxx xxxxxxxxxxxxxxxxxxxxxx Chairman: External Examiners:
UNIVERSITY OF EDINBURGH College of Science and Engineering School of GeoSciences Earth Materials UO4824 DEGREE EXAMINATION (MOCK) xxxxxxxxxxxxxxxxxxxx xxxxxxxxxxxxxxxxxxxxxx Chairman: External Examiners:
Structure of the Earth
 And the ROCK CYCLE Structure of the Earth Compositional (Chemical) Layers Crust: Low density High in silicon (Si) and oxygen (O) Moho: Density boundary between crust and mantle Mantle: Higher density High
And the ROCK CYCLE Structure of the Earth Compositional (Chemical) Layers Crust: Low density High in silicon (Si) and oxygen (O) Moho: Density boundary between crust and mantle Mantle: Higher density High
Unit 3 Lesson 2 The Rock Cycle. Copyright Houghton Mifflin Harcourt Publishing Company
 Unit 3 Lesson 2 The Rock Cycle What are minerals? A mineral is a naturally occurring, usually inorganic solid that has a defininite crystalline structure and chemical composition. Naturally occuring =
Unit 3 Lesson 2 The Rock Cycle What are minerals? A mineral is a naturally occurring, usually inorganic solid that has a defininite crystalline structure and chemical composition. Naturally occuring =
Earth Science 11 Learning Guide Unit 5
 Earth Science 11 Learning Guide Unit 5 Name: 5-1 Minerals and mineral identification 1. For each of the following statements put a R in the space if the statement refers to a rock M in the space if the
Earth Science 11 Learning Guide Unit 5 Name: 5-1 Minerals and mineral identification 1. For each of the following statements put a R in the space if the statement refers to a rock M in the space if the
Modeling the Rock Cycle
 P a g e 1 Name Modeling the Rock Cycle Background Information: Like the Earth itself, rocks are in a constant state of change. Rocks are solids made up of different minerals. Scientists usually classify
P a g e 1 Name Modeling the Rock Cycle Background Information: Like the Earth itself, rocks are in a constant state of change. Rocks are solids made up of different minerals. Scientists usually classify
Engineering Geology ECIV 2204
 Engineering Geology ECIV 2204 Instructor : Dr. Jehad Hamad 2017-2016 Chapter (3) Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth
Engineering Geology ECIV 2204 Instructor : Dr. Jehad Hamad 2017-2016 Chapter (3) Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth
LAB 5: COMMON MINERALS IN IGNEOUS ROCKS
 EESC 2100: Mineralogy LAB 5: COMMON MINERALS IN IGNEOUS ROCKS Part 1: Minerals in Granitic Rocks Learning Objectives: Students will be able to identify the most common minerals in granitoids Students will
EESC 2100: Mineralogy LAB 5: COMMON MINERALS IN IGNEOUS ROCKS Part 1: Minerals in Granitic Rocks Learning Objectives: Students will be able to identify the most common minerals in granitoids Students will
Minerals RMS. Mineral ID Rocks Soil
 Minerals Moh Minerals RMS Mineral ID Rocks Soil 1000 1000 1000 1000 1000 2000 2000 2000 2000 2000 3000 3000 3000 3000 3000 4000 4000 4000 4000 4000 5000 5000 5000 5000 5000 What is a naturally occurring,
Minerals Moh Minerals RMS Mineral ID Rocks Soil 1000 1000 1000 1000 1000 2000 2000 2000 2000 2000 3000 3000 3000 3000 3000 4000 4000 4000 4000 4000 5000 5000 5000 5000 5000 What is a naturally occurring,
Block: Igneous Rocks. From this list, select the terms which answer the following questions.
 Geology 12 Name: Mix and Match: Igneous Rocks Refer to the following list. Block: porphyritic volatiles mafic glassy magma mixing concordant discontinuous reaction series igneous vesicular partial melting
Geology 12 Name: Mix and Match: Igneous Rocks Refer to the following list. Block: porphyritic volatiles mafic glassy magma mixing concordant discontinuous reaction series igneous vesicular partial melting
10/20/2015. How is magma different from lava? Magma is molten rock below the Earth s surface. Lava is magma that flows out onto Earth s surface.
 Chapter 5 What are igneous rocks? How do they form? Igneous rocks are rocks that form when molten material cools and crystallizes. Molten material can be either magma or lava. How is magma different from
Chapter 5 What are igneous rocks? How do they form? Igneous rocks are rocks that form when molten material cools and crystallizes. Molten material can be either magma or lava. How is magma different from
EPSC 233. Compositional variation in minerals. Recommended reading: PERKINS, p. 286, 41 (Box 2-4).
 EPSC 233 Compositional variation in minerals Recommended reading: PERKINS, p. 286, 41 (Box 2-4). Some minerals are nearly pure elements. These are grouped under the category of native elements. This includes
EPSC 233 Compositional variation in minerals Recommended reading: PERKINS, p. 286, 41 (Box 2-4). Some minerals are nearly pure elements. These are grouped under the category of native elements. This includes
Igneous Rocks (Right Side Question)
 Igneous Rocks (Right Side Question) Record the question in your notebook! Using your understanding of rock texture (grain size, grain shape, grain pattern), explain the process that you would use to determine
Igneous Rocks (Right Side Question) Record the question in your notebook! Using your understanding of rock texture (grain size, grain shape, grain pattern), explain the process that you would use to determine
Compositional (Chemical) Layers
 Compositional (Chemical) Layers Crust: Low density High in and : Density boundary between crust and mantle Mantle: Higher density High in and Core: High in Heat Flow in the Earth Heat sources: heat from
Compositional (Chemical) Layers Crust: Low density High in and : Density boundary between crust and mantle Mantle: Higher density High in and Core: High in Heat Flow in the Earth Heat sources: heat from
The Rock Cycle The Rock Cycle illustrates the origin of igneous, sedimentary and metamorphic rocks
 The Rock Cycle The Rock Cycle illustrates the origin of igneous, sedimentary and metamorphic rocks Igneous rocks form as molten magma or lava cools and solidifies. Magma is completely or partly molten
The Rock Cycle The Rock Cycle illustrates the origin of igneous, sedimentary and metamorphic rocks Igneous rocks form as molten magma or lava cools and solidifies. Magma is completely or partly molten
GEOL 2312 Igneous and Metamorphic Petrology Spring 2016 Score / 58. Midterm 1 Chapters 1-10
 GEOL 2312 Igneous and Metamorphic Petrology Name KEY Spring 2016 Score / 58 Midterm 1 Chapters 1-10 1) Name two things that petrologists want to know about magmas (1 pt) Formation, source, composition,
GEOL 2312 Igneous and Metamorphic Petrology Name KEY Spring 2016 Score / 58 Midterm 1 Chapters 1-10 1) Name two things that petrologists want to know about magmas (1 pt) Formation, source, composition,
Plate Tectonics Scavenger Hunt
 Plate Tectonics Scavenger Hunt Directions: GO TO TEACHER WEBPAGE FOR LINKS TO WEBSITES! Read each page from the websites very carefully and use the information given to complete the scavenger hunt. SITE:
Plate Tectonics Scavenger Hunt Directions: GO TO TEACHER WEBPAGE FOR LINKS TO WEBSITES! Read each page from the websites very carefully and use the information given to complete the scavenger hunt. SITE:
Metamorphic Petrology GLY 262 Metamorphic reactions and isograds
 Metamorphic Petrology GLY 262 Metamorphic reactions and isograds What do we mean by reaction? Reaction: change in the nature or types of phases in a system=> formation of new mineral(s) ) which are stable
Metamorphic Petrology GLY 262 Metamorphic reactions and isograds What do we mean by reaction? Reaction: change in the nature or types of phases in a system=> formation of new mineral(s) ) which are stable
Rocks Rock- A group of minerals, glass, mineroid bound together in some way.
 Rocks Rock- A group of minerals, glass, mineroid bound together in some way. All rocks fit into one of three categories: Igneous- formed by the cooling and hardening of hot molten rock Sedimentary- formed
Rocks Rock- A group of minerals, glass, mineroid bound together in some way. All rocks fit into one of three categories: Igneous- formed by the cooling and hardening of hot molten rock Sedimentary- formed
CITY OF LONDON CEMETERY TEACHERS GUIDE TO ROCK TYPE EXERCISES: LOCATION ONE
 CITY OF LONDON CEMETERY TEACHERS GUIDE TO ROCK TYPE EXERCISES: LOCATION ONE The City of London Cemetery is a haven for the geologist, with representatives from the sedimentary, igneous and metamorphic
CITY OF LONDON CEMETERY TEACHERS GUIDE TO ROCK TYPE EXERCISES: LOCATION ONE The City of London Cemetery is a haven for the geologist, with representatives from the sedimentary, igneous and metamorphic
Page 1. Name:
 Name: 1) What is the approximate density of a mineral with a mass of 262.2 grams that displaces 46 cubic centimeters of water? A) 6.1 g/cm 3 C) 1.8 g/cm 3 B) 5.7 g/cm 3 D) 12.2 g/cm 3 2) In which two Earth
Name: 1) What is the approximate density of a mineral with a mass of 262.2 grams that displaces 46 cubic centimeters of water? A) 6.1 g/cm 3 C) 1.8 g/cm 3 B) 5.7 g/cm 3 D) 12.2 g/cm 3 2) In which two Earth
Page 1. Name: 1) Which diagram best shows the grain size of some common sedimentary rocks?
 Name: 1) Which diagram best shows the grain size of some common sedimentary rocks? 1663-1 - Page 1 5) The flowchart below illustrates the change from melted rock to basalt. 2) Which processes most likely
Name: 1) Which diagram best shows the grain size of some common sedimentary rocks? 1663-1 - Page 1 5) The flowchart below illustrates the change from melted rock to basalt. 2) Which processes most likely
L.O: SLOWING STREAMS DEPOSIT (SORT) SEDIMENT HORIZONTALLY BY SIZE.
 L.O: SLOWING STREAMS DEPOSIT (SORT) SEDIMENT HORIZONTALLY BY SIZE. 1. Base your answer to the following question on the profile shown below, which shows the pattern of horizontal sorting produced at a
L.O: SLOWING STREAMS DEPOSIT (SORT) SEDIMENT HORIZONTALLY BY SIZE. 1. Base your answer to the following question on the profile shown below, which shows the pattern of horizontal sorting produced at a
Lab Activity on Igneous Processes
 Lab Activity on Igneous Processes 2002 Ann Bykerk-Kauffman, Dept. of Geological and Environmental Sciences, California State University, Chico * Objectives When you have completed this lab you should be
Lab Activity on Igneous Processes 2002 Ann Bykerk-Kauffman, Dept. of Geological and Environmental Sciences, California State University, Chico * Objectives When you have completed this lab you should be
Rock Cycle. Presented by Kesler Science
 Presented by Kesler Science Essential Questions: What processes are involved in the formation and classification of metamorphic, sedimentary, and igneous rocks? Sediments A model that describes the formation,
Presented by Kesler Science Essential Questions: What processes are involved in the formation and classification of metamorphic, sedimentary, and igneous rocks? Sediments A model that describes the formation,
Solid Earth materials:
 Solid Earth materials: Elements minerals rocks Nonuniform distribution of matter Molten core Contains most heavy elements Iron, nickel Thin surface crust Mostly lighter elements 8 elements make up 98.6%
Solid Earth materials: Elements minerals rocks Nonuniform distribution of matter Molten core Contains most heavy elements Iron, nickel Thin surface crust Mostly lighter elements 8 elements make up 98.6%
Geology Lab: The Properties of Minerals & Igneous Rocks. Part 1: Minerals
 Geology 101, Lab Section (20 Points) Your First & Last Name: Geology Lab: The Properties of Minerals & Igneous Rocks Part 1: Minerals This section designed to introduce you to the most common minerals
Geology 101, Lab Section (20 Points) Your First & Last Name: Geology Lab: The Properties of Minerals & Igneous Rocks Part 1: Minerals This section designed to introduce you to the most common minerals
Unsaved Test, Version: 1 1
 Name: Key Concepts Choose the letter of the best answer. 1. Which of the following is true? A. Some minerals have no crystal structure. B. Some minerals are liquids. C. Minerals are always solids. D. Minerals
Name: Key Concepts Choose the letter of the best answer. 1. Which of the following is true? A. Some minerals have no crystal structure. B. Some minerals are liquids. C. Minerals are always solids. D. Minerals
NOTE OUTLINE : Chap 5 & 6: Rocks
 Name Period NOTE OUTLINE : Chap 5 & 6: Rocks Objectives 1. Identify and explain characteristics of igneous rocks. This means that if I am given an igneous rock I: a. Can use grain size to identify a rock
Name Period NOTE OUTLINE : Chap 5 & 6: Rocks Objectives 1. Identify and explain characteristics of igneous rocks. This means that if I am given an igneous rock I: a. Can use grain size to identify a rock
Rocks: Materials of the Solid Earth
 1 Rocks: Materials of the Solid Earth Presentation modified from: Instructor Resource Center on CD-ROM, Foundations of Earth Science,, 4 th Edition, Lutgens/Tarbuck, Rock Cycle Igneous Rocks Today 2 Rock
1 Rocks: Materials of the Solid Earth Presentation modified from: Instructor Resource Center on CD-ROM, Foundations of Earth Science,, 4 th Edition, Lutgens/Tarbuck, Rock Cycle Igneous Rocks Today 2 Rock
EPS 50 Lab 2: Igneous Rocks Grotzinger and Jordan, Chapter 4
 Name: EPS 50 Lab 2: Igneous Rocks Grotzinger and Jordan, Chapter 4 Introduction In the previous lab, we learned about mineral characteristics, properties and identities as well as the three basic rock
Name: EPS 50 Lab 2: Igneous Rocks Grotzinger and Jordan, Chapter 4 Introduction In the previous lab, we learned about mineral characteristics, properties and identities as well as the three basic rock
IGNEOUS ROCKS. SECTION 5.1 What are igneous rocks?
 Date Period Name IGNEOUS ROCKS SECTION.1 What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. basaltic
Date Period Name IGNEOUS ROCKS SECTION.1 What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. basaltic
A Rock is A group of minerals that have been put together in several different ways.
 A Rock is A group of minerals that have been put together in several different ways. Depending on how they are put together, rocks are classified as: 1. Sedimentary 2. Igneous 3. Metamorphic Sedimentary
A Rock is A group of minerals that have been put together in several different ways. Depending on how they are put together, rocks are classified as: 1. Sedimentary 2. Igneous 3. Metamorphic Sedimentary
Welcome to Class 5: Earth Geology. Remember: sit only in the first 10 rows of the room
 Welcome to Class 5: Earth Geology Remember: sit only in the first 10 rows of the room What s going to happen today? 1. You will learn: Sublime is more than a dead heroin addict s band. 2. You will better
Welcome to Class 5: Earth Geology Remember: sit only in the first 10 rows of the room What s going to happen today? 1. You will learn: Sublime is more than a dead heroin addict s band. 2. You will better
Thursday, October 4 th
 Thursday, October 4 th Objective: We will use and define the different ways to classify igneous rocks. Warm-up: 1. Which type of lava is most viscous? 2. Which type of lava has the least amount of silicate?
Thursday, October 4 th Objective: We will use and define the different ways to classify igneous rocks. Warm-up: 1. Which type of lava is most viscous? 2. Which type of lava has the least amount of silicate?
8 th Earth Science Chapter 4 Rocks Name Section 1 The Rock Cycle:
 8 th Earth Science Chapter 4 Rocks Name Section 1 The Rock Cycle: Most rock used for stone contains one or more common minerals, called rock-forming minerals, such as, feldspar,, or. When you look closely,
8 th Earth Science Chapter 4 Rocks Name Section 1 The Rock Cycle: Most rock used for stone contains one or more common minerals, called rock-forming minerals, such as, feldspar,, or. When you look closely,
Which rock is shown? A) slate B) dunite C) gneiss D) quartzite
 1. Which metamorphic rock will have visible mica crystals and a foliated texture? A) marble B) quartzite C) schist D) slate 2. The recrystallization of unmelted material under high temperature and pressure
1. Which metamorphic rock will have visible mica crystals and a foliated texture? A) marble B) quartzite C) schist D) slate 2. The recrystallization of unmelted material under high temperature and pressure
GY303 Igneous & Metamorphic Petrology. Lecture 7: Magma Sources and Tectonic Environments
 GY303 Igneous & Metamorphic Petrology Lecture 7: Magma Sources and Tectonic Environments Factors controlling Magma production Source rock composition Amount of fluids, especially H 2 O Pressure (Depth)
GY303 Igneous & Metamorphic Petrology Lecture 7: Magma Sources and Tectonic Environments Factors controlling Magma production Source rock composition Amount of fluids, especially H 2 O Pressure (Depth)
The Gibbs Phase Rule F = 2 + C - P
 The Gibbs Phase Rule The phase rule allows one to determine the number of degrees of freedom (F) or variance of a chemical system. This is useful for interpreting phase diagrams. F = 2 + C - P Where F
The Gibbs Phase Rule The phase rule allows one to determine the number of degrees of freedom (F) or variance of a chemical system. This is useful for interpreting phase diagrams. F = 2 + C - P Where F
Student Name: College: Grade:
 Student Name: College: Grade: Physical Geology Laboratory IGNEOUS MINERALS AND ROCKS IDENTIFICATION - INTRODUCTION & PURPOSE: In this lab you will learn to identify igneous rocks in hand samples from their
Student Name: College: Grade: Physical Geology Laboratory IGNEOUS MINERALS AND ROCKS IDENTIFICATION - INTRODUCTION & PURPOSE: In this lab you will learn to identify igneous rocks in hand samples from their
Igneous Rocks: Formed by Fire (p )
 READING QUESTIONS: Chapter 2 (Rocks) PSCI 131 18/WI 70 pts NAME DUE: Monday, January 29 Igneous Rocks: Formed by Fire (p. 34-43) 1. What is magma? 2. Extrusive igneous rocks form at the Earth s, and intrusive
READING QUESTIONS: Chapter 2 (Rocks) PSCI 131 18/WI 70 pts NAME DUE: Monday, January 29 Igneous Rocks: Formed by Fire (p. 34-43) 1. What is magma? 2. Extrusive igneous rocks form at the Earth s, and intrusive
12 Chemistry (Mg,Fe) 2 SiO 4 Olivine is forms what is called an isomorphous solid solution series that ranges between two end members: Forsterite Mg
 11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
Minerals and Rocks. Test Review Activity. Begin
 Minerals and Rocks Test Review Activity Begin What type of rock is this? How can you tell? Question #1 If the crystals Sample in this felsic Digneous rock are 3-6 mm in size, what rock is it? Last Question
Minerals and Rocks Test Review Activity Begin What type of rock is this? How can you tell? Question #1 If the crystals Sample in this felsic Digneous rock are 3-6 mm in size, what rock is it? Last Question
Rock Identification. invisible rhyolite andesite basalt komatiite. visible granite diorite gabbro peridotite
 Rock Identification The samples in this lab are arranged into four groups: igneous, sedimentary, metamorphic, and unknown. Study the igneous, sedimentary, and metamorphic collections to get an idea of
Rock Identification The samples in this lab are arranged into four groups: igneous, sedimentary, metamorphic, and unknown. Study the igneous, sedimentary, and metamorphic collections to get an idea of
Teaching With Projections in the Geosciences David W. Mogk Dept. of Earth Sciences Montana State University
 Teaching With Projections in the Geosciences David W. Mogk Dept. of Earth Sciences Montana State University Geoscientists are trained to represent multi-component datasets by projecting onto relatively
Teaching With Projections in the Geosciences David W. Mogk Dept. of Earth Sciences Montana State University Geoscientists are trained to represent multi-component datasets by projecting onto relatively
Earth Science 11: Earth Materials: Rock Cycle
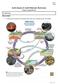 Name: Date: Earth Science 11: Earth Materials: Rock Cycle Chapter 2, pages 44 to 46 2.1: Rock Cycle What is a Rock? A solid mass of mineral or mineral-like matter that occurs naturally as part of our planet
Name: Date: Earth Science 11: Earth Materials: Rock Cycle Chapter 2, pages 44 to 46 2.1: Rock Cycle What is a Rock? A solid mass of mineral or mineral-like matter that occurs naturally as part of our planet
lava magma pyroclastic materials lava flow igneous rock volcanic (extrusive igneous) rock plutonic (intrusive igneous) rock felsic magma mafic magma
 magma lava lava flow pyroclastic materials igneous rock volcanic (extrusive igneous) rock plutonic (intrusive igneous) rock felsic magma mafic magma intermediate magma viscosity magma chamber Bowen s reaction
magma lava lava flow pyroclastic materials igneous rock volcanic (extrusive igneous) rock plutonic (intrusive igneous) rock felsic magma mafic magma intermediate magma viscosity magma chamber Bowen s reaction
Lecture 3 Rocks and the Rock Cycle Dr. Shwan Omar
 Rocks A naturally occurring aggregate of one or more minerals (e.g., granite), or a body of non-crystalline material (e.g., obsidian glass), or of solid organic material (e.g., coal). Rock Cycle A sequence
Rocks A naturally occurring aggregate of one or more minerals (e.g., granite), or a body of non-crystalline material (e.g., obsidian glass), or of solid organic material (e.g., coal). Rock Cycle A sequence
Name Class Date STUDY GUIDE FOR CONTENT MASTERY
 Igneous Rocks What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. extrusive igneous rock intrusive
Igneous Rocks What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. extrusive igneous rock intrusive
Chapter 11 Review Book Earth Materials Minerals and Rocks
 Chapter 11 Review Book Earth Materials Minerals and Rocks Define the Vocabulary 1. bioclastic sedimentary rocks 2. chemical sedimentary rocks 3. Clastic sedimentary rocks 4. cleavage 5. contact metamorphism
Chapter 11 Review Book Earth Materials Minerals and Rocks Define the Vocabulary 1. bioclastic sedimentary rocks 2. chemical sedimentary rocks 3. Clastic sedimentary rocks 4. cleavage 5. contact metamorphism
Igneous petrology EOSC 321 Laboratory 8: Intermediate and Felsic Volcanic Rocks. Pyroclastic Rocks
 321 Lab 8 Instructor: L. Porritt - 1 - Igneous petrology EOSC 321 Laboratory 8: Intermediate and Felsic Volcanic Rocks. Pyroclastic Rocks Learning Goals. After this Lab, you should be able: Identify fine-grained
321 Lab 8 Instructor: L. Porritt - 1 - Igneous petrology EOSC 321 Laboratory 8: Intermediate and Felsic Volcanic Rocks. Pyroclastic Rocks Learning Goals. After this Lab, you should be able: Identify fine-grained
Geology 314 Accretion of the Bushveld complex
 Geology 314 Accretion of the Bushveld complex Foreword: This document is not a stand-alone document; it s written as an example showing how the documents of the prac 9 (Geol 314, Stellenbosch, 2007) can
Geology 314 Accretion of the Bushveld complex Foreword: This document is not a stand-alone document; it s written as an example showing how the documents of the prac 9 (Geol 314, Stellenbosch, 2007) can
Environments of Mineral Formation. Stability Diagrams
 Environments of Mineral Formation Unary, Binary, and Ternary Mineral Stability Diagrams Minerals of differing composition (or polymorphs of the same mineral) that coexist at a set of pressure (P) temperature
Environments of Mineral Formation Unary, Binary, and Ternary Mineral Stability Diagrams Minerals of differing composition (or polymorphs of the same mineral) that coexist at a set of pressure (P) temperature
Lab 2: The rock cycle, minerals and igneous rocks. Rocks are divided into three major categories on the basis of their origin:
 Geology 101 Name(s): Lab 2: The rock cycle, minerals and igneous rocks Rocks are divided into three major categories on the basis of their origin: Igneous rocks (from the Latin word, ignis = fire) are
Geology 101 Name(s): Lab 2: The rock cycle, minerals and igneous rocks Rocks are divided into three major categories on the basis of their origin: Igneous rocks (from the Latin word, ignis = fire) are
Chapter 4 Rocks & Igneous Rocks
 Chapter 4 Rocks & Igneous Rocks Rock Definition A naturally occurring consolidated mixture of one or more minerals e.g, marble, granite, sandstone, limestone Rock Definition Must naturally occur in nature,
Chapter 4 Rocks & Igneous Rocks Rock Definition A naturally occurring consolidated mixture of one or more minerals e.g, marble, granite, sandstone, limestone Rock Definition Must naturally occur in nature,
Primary Structures in Igneous Rocks. Engr. Sultan Ahmed Khoso
 Primary Structures in Igneous Rocks Engr. Sultan Ahmed Khoso Primary Structures in Igneous Rocks Primary Structures developed in the igneous rock during the time of cooling, crystallization and solidification
Primary Structures in Igneous Rocks Engr. Sultan Ahmed Khoso Primary Structures in Igneous Rocks Primary Structures developed in the igneous rock during the time of cooling, crystallization and solidification
1. A solid material made up of one or more minerals or grains or other substances, including the remains of once-living things is a.
 1. A solid material made up of one or more minerals or grains or other substances, including the remains of once-living things is a. 2. A solid, inorganic material that forms naturally on or beneath Earth
1. A solid material made up of one or more minerals or grains or other substances, including the remains of once-living things is a. 2. A solid, inorganic material that forms naturally on or beneath Earth
Faults, Fossils, Rocks and Minerals Review:
 Faults, Fossils, Rocks and Minerals Review: 1. The preserved remains or traces of organisms that lived in the past are. - Fossils 2. How do Fossils form? - A dead organism becomes buried in sediment 3.
Faults, Fossils, Rocks and Minerals Review: 1. The preserved remains or traces of organisms that lived in the past are. - Fossils 2. How do Fossils form? - A dead organism becomes buried in sediment 3.
This slide show is intended to help you understand important types of rocks.
 Rocks Mrs. Comer This slide show is intended to help you understand important types of rocks. The diagram in the next slide represents the ROCK CYCLE a scheme that represents the processes of continuous
Rocks Mrs. Comer This slide show is intended to help you understand important types of rocks. The diagram in the next slide represents the ROCK CYCLE a scheme that represents the processes of continuous
Table 7.1 Mineralogy of metamorphic rocks related to protolith and grade
 Geology 101 Name(s): Lab 7: Metamorphic rocks Metamorphic rocks have been subjected to sufficient heat and/or pressure to melt some of their constituent minerals, but not all of them. As a result of this
Geology 101 Name(s): Lab 7: Metamorphic rocks Metamorphic rocks have been subjected to sufficient heat and/or pressure to melt some of their constituent minerals, but not all of them. As a result of this
