1 - C Systems. The system H 2 O. Heat an ice at 1 atm from-5 to 120 o C. Heat vs. Temperature
|
|
|
- Darren Harris
- 5 years ago
- Views:
Transcription
1 1 - C Systems The system H 2 O Heat an ice at 1 atm from-5 to 120 o C Heat vs. Temperature Fig After Bridgman (1911) Proc. Amer. Acad. Arts and Sci., 5, ; (1936) J. Chem. Phys., 3, ; (1937) J. Chem. Phys., 5,
2 Crystallization Behavior of Melts 1. Cooling melts crystallize from a liquid to a solid over a range of temperatures (and pressures)
3 Crystallization Behavior of Melts 1. Cooling melts crystallize from a liquid to a solid over a range of temperatures (and pressures) 2. Several minerals crystallize over this T range, and the number of minerals generally increases as T decreases
4 Crystallization Behavior of Melts 1. Cooling melts crystallize from a liquid to a solid over a range of temperatures (and pressures) 2. Several minerals crystallize over this T range, and the number of minerals increases as T decreases 3. Minerals that form do so sequentially, with considerable overlap
5 Crystallization Behavior of Melts 1. Cooling melts crystallize from a liquid to a solid over a range of temperatures (and pressures) 2. Several minerals crystallize over this T range, and the number of minerals increases as T decreases 3. Minerals that form do so sequentially, with considerable overlap 4. Minerals that involve solid solution change composition as cooling progresses
6 Crystallization Behavior of Melts 1. Cooling melts crystallize from a liquid to a solid over a range of temperatures (and pressures) 2. Several minerals crystallize over this T range, and the number of minerals increases as T decreases 3. The minerals that form do so sequentially, with consideral overlap 4. Minerals that involve solid solution change composition as cooling progresses 5. The melt composition also changes during crystallization
7 Crystallization Behavior of Melts 1. Cooling melts crystallize from a liquid to a solid over a range of temperatures (and pressures) 2. Several minerals crystallize over this T range, and the number of minerals increases as T decreases 3. The minerals that form do so sequentially, with consideral overlap 4. Minerals that involve solid solution change composition as cooling progresses 5. The melt composition also changes during crystallization 6. The minerals that crystallize (as well as the sequence) depend on T and X of the melt
8 Crystallization Behavior of Melts 1. Cooling melts crystallize from a liquid to a solid over a range of temperatures (and pressures) 2. Several minerals crystallize over this T range, and the number of minerals increases as T decreases 3. The minerals that form do so sequentially, with consideral overlap 4. Minerals that involve solid solution change composition as cooling progresses 5. The melt composition also changes during crystallization 6. The minerals that crystallize (as well as the sequence) depend on T and X of the melt 7. Pressure can affect the types of minerals that form and the sequence
9 Crystallization Behavior of Melts 1. Cooling melts crystallize from a liquid to a solid over a range of temperatures (and pressures) 2. Several minerals crystallize over this T range, and the number of minerals increases as T decreases 3. The minerals that form do so sequentially, with consideral overlap 4. Minerals that involve solid solution change composition as cooling progresses 5. The melt composition also changes during crystallization 6. The minerals that crystallize (as well as the sequence) depend on T and X of the melt 7. Pressure can affect the types of minerals that form and the sequence 8. The nature and pressure of the volatiles can also affect the minerals and their sequence
10 2 - C Systems A. Systems with Complete Solid Solution 1. Plagioclase (Ab-An, NaAlSi 3 O 8 - CaAl 2 Si 2 O 8 ) Coupled substitution Fig Isobaric T-X phase diagram at atmospheric pressure. After Bowen (1913) Amer. J. Sci., 35,
11 Bulk composition a = An 60 = 60 g An + 40 g Ab X An = 60/(60+40) = 0.60
12 At 1450 o C, liquid d and plagioclase f coexist at equilibrium A continuous reaction of the type: liquid B + solid C = liquid D + solid F
13 The lever principle: Amount of liquid Amount of solid = ef de where d = the liquid composition, f = the solid composition and e = the bulk composition liquidus d de e ef f solidus
14 Note the difference between the two types of fields The blue fields are one phase fields Any point in these fields represents a true phase composition Liquid The blank field is a two phase field Any point in this field represents a bulk composition composed of two phases at the edge of the blue fields and connected by a horizontal tie-line Liquid Plagioclase plus Plagioclase
15 When X plag h, then X plag = X bulk and, according to the lever principle, the amount of liquid 0 Thus g is the composition of the last liquid to crystallize at 1340 o C for bulk X = 0.60
16 Final plagioclase to form is i when = 0.60 Now f = 1 so F = = 2 plag X An
17 * The actual temperatures and the range depend on the bulk composition Note the following: 1. The melt crystallized over a T range of 135 o C * 2. The composition of the liquid changed from b to g 3. The composition of the solid changed from c to h 4. The ratio of solid to liquid increases with cooling
18 Fractional crystallization: Remove crystals as they form so they can t undergo a continuous reaction with the melt At any T X bulk = X liq due to the removal of the crystals
19 Plagioclase Differentiation Mechanisms zoned plag Crystal Settling Perthitic pyroxene Gabbro - Plane Polarized Light Plagioclase zoning
20 2. The Olivine System Fo - Fa (Mg 2 SiO 4 - Fe 2 SiO 4 ) also a solid-solution series Fig Isobaric T-X phase diagram at atmospheric pressure After Bowen and Shairer (1932), Amer. J. Sci. 5th Ser., 24,
21 2-C Eutectic Systems Example: Diopside Anorthite No solid solution [basalt composition] Fig Isobaric T-X phase diagram at atmospheric pressure. After Bowen (1915), Amer. J. Sci. 40,
22 Cool composition a: bulk composition = An 70
23 Cool to 1455 o C (point b)
24 Continue cooling as X liq varies along the liquidus Continuous reaction: liq A anorthite + liq B
25 at 1274 o C f = 3 so F = = 0 invariant (P) T and the composition of all phases is fixed Must remain at 1274 o C as a discontinuous reaction proceeds until a phase is lost
26 Discontinuous Reaction: all at a single T Dipside+liquid=diopside +Anorthite
27 Left of the eutectic get a similar situation
28 #s are listed points in text Note the following: 1. The melt crystallizes over a T range up to ~280 o C 2. A sequence of minerals forms over this interval - And the number of minerals increases as T drops 6. The minerals that crystallize depend upon T - The sequence changes with the bulk composition
29 Augite forms before plagioclase Gabbro of the Stillwater Complex, Montana This forms on the left side of the eutectic
30 Plagioclase forms before augite Ophitic texture Diabase dike This forms on the right side of the eutectic
31 Also note: The last melt to crystallize in any binary eutectic mixture is the eutectic composition Equilibrium melting is the opposite of equilibrium crystallization Thus the first melt of any mixture of Di and An must be the eutectic composition as well
32 Fractional crystallization: No effect on the liquid path The composition of the final rock is affected Fig Isobaric T-X phase diagram at atmospheric pressure. After Bowen (1915), Amer. J. Sci. 40,
33 C. Binary Peritectic Systems Three phases enstatite = forsterite + SiO 2 Figure Isobaric T-X phase diagram of the system Fo-Silica at 0.1 MPa. After Bowen and Anderson (1914) and Grieg (1927). Amer. J. Sci.
34 C. Binary Peritectic Systems Figure Isobaric T-X phase diagram of the system Fo-Silica at 0.1 MPa. After Bowen and Anderson (1914) and Grieg (1927). Amer. J. Sci.
35 Figure Isobaric T-X phase diagram of the system Fo-Silica at 0.1 MPa. After Bowen and Anderson (1914) and Grieg (1927). Amer. J. Sci.
36 i = peritectic point 1557 o C have colinear Fo-En-liq geometry indicates a reaction: Fo + liq = En consumes olivine (and liquid) resorbed textures When is the reaction finished? 1557 k i m d c Fo Bulk X En
37
38
39 y k x i m 1557 d c 1543 Cr Fo En bulk X
40 Cool X = n At 1960 o C hit solvus exsolution 2 liquids o and p f = 2 F = 1 both liquids follow solvus At 1695 o C get Crst also Reaction? Immiscible Liquids Mafic-rich liquid 1695 Silica-rich liquid Crst
Phase Equilibrium. Phase Rule. Phase Diagram
 Phase Equilibrium Phase Rule Phase Diagram Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS Prof. Paper, 1004. Makaopuhi Lava Lake
Phase Equilibrium Phase Rule Phase Diagram Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS Prof. Paper, 1004. Makaopuhi Lava Lake
Chapter 6: The Phase Rule and One and Two-Component Systems aka Phase Equilibria
 Chapter 6: The Phase Rule and One and Two-Component Systems aka Phase Equilibria Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS
Chapter 6: The Phase Rule and One and Two-Component Systems aka Phase Equilibria Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS
2-C Eutectic Systems
 Phase Equilibrium 2-C Eutectic Systems Example: Diopside - Anorthite No solid solution * For system at this T, X, phase(s) plot where? Fig. 6.11. Isobaric T-X phase diagram at atmospheric pressure. After
Phase Equilibrium 2-C Eutectic Systems Example: Diopside - Anorthite No solid solution * For system at this T, X, phase(s) plot where? Fig. 6.11. Isobaric T-X phase diagram at atmospheric pressure. After
TWO COMPONENT (BINARY) PHASE DIAGRAMS. Experimental Determination of 2-Component Phase Diagrams
 Page 1 of 12 EENS 211 Earth Materials Tulane University Prof. Stephen A. Nelson TWO COMPONENT (BINARY) PHASE DIAGRAMS This document last updated on 08-Oct-2003 Experimental Determination of 2-Component
Page 1 of 12 EENS 211 Earth Materials Tulane University Prof. Stephen A. Nelson TWO COMPONENT (BINARY) PHASE DIAGRAMS This document last updated on 08-Oct-2003 Experimental Determination of 2-Component
A. One component system (c = 1)
 A. One component system (c = 1) Example: SiO 2 system. Since all phases in this system have the same composition, there are no compositional variables to consider. Phase equilibria can be shown completely
A. One component system (c = 1) Example: SiO 2 system. Since all phases in this system have the same composition, there are no compositional variables to consider. Phase equilibria can be shown completely
C = 3: Ternary Systems: Example 1: Ternary Eutectic
 Phase Equilibrium C = 3: Ternary Systems: Example 1: Ternary Eutectic Adding components, becomes increasingly difficult to depict 1-C: P - T diagrams easy 2-C: isobaric T-X, isothermal P-X 3-C:?? Still
Phase Equilibrium C = 3: Ternary Systems: Example 1: Ternary Eutectic Adding components, becomes increasingly difficult to depict 1-C: P - T diagrams easy 2-C: isobaric T-X, isothermal P-X 3-C:?? Still
Interpreting Phase Diagrams
 Interpreting Phase Diagrams Understanding chemical reactions requires that we know something about how materials behave as the temperature and pressure change. For a single component (like quartz or ice)
Interpreting Phase Diagrams Understanding chemical reactions requires that we know something about how materials behave as the temperature and pressure change. For a single component (like quartz or ice)
Common non-silicate planetary minerals
 Common non-silicate planetary minerals Many of the non-silicate minerals are simple oxides. Corundum Al2O3 Al2+3 O3-2 Rutile Ti2O3 Ti2+3 O3-2 Ilmenite FeTiO3 Fe+3Ti+3O3-2 Hematite Fe2O3 Fe2+3 O3-2 Families
Common non-silicate planetary minerals Many of the non-silicate minerals are simple oxides. Corundum Al2O3 Al2+3 O3-2 Rutile Ti2O3 Ti2+3 O3-2 Ilmenite FeTiO3 Fe+3Ti+3O3-2 Hematite Fe2O3 Fe2+3 O3-2 Families
Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and. MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout)
 Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and 20 MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout) Chain silicate eg Diopside Mg and Fe ions link SiO 3 chains The chain runs up and down
Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and 20 MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout) Chain silicate eg Diopside Mg and Fe ions link SiO 3 chains The chain runs up and down
Hirsch - Binary phase diagrams problems KEY
 Hirsch - Binary phase diagrams problems KEY Name Question 1. 1A. Using the phase rule, determine the degrees of freedom: a. at point e F = 2-3 + 1 = 0 b. within the field A + B F = 2-2 + 1 = 1 c. within
Hirsch - Binary phase diagrams problems KEY Name Question 1. 1A. Using the phase rule, determine the degrees of freedom: a. at point e F = 2-3 + 1 = 0 b. within the field A + B F = 2-2 + 1 = 1 c. within
Feldspars. Structure. The feldspars are by far the most abundant group of minerals and are found in igneous, metamorphic and many sedimentary rocks.
 Feldspars The feldspars are by far the most abundant group of minerals and are found in igneous, metamorphic and many sedimentary rocks. Structure Felsdpars are framework silicates where each silica tetrahedra
Feldspars The feldspars are by far the most abundant group of minerals and are found in igneous, metamorphic and many sedimentary rocks. Structure Felsdpars are framework silicates where each silica tetrahedra
DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER
 Geol 2312 Igneous and Metamorphic Petrology Spring 2009 Name DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER Objective: This exercise is intended to improve understanding
Geol 2312 Igneous and Metamorphic Petrology Spring 2009 Name DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER Objective: This exercise is intended to improve understanding
12 Chemistry (Mg,Fe) 2 SiO 4 Olivine is forms what is called an isomorphous solid solution series that ranges between two end members: Forsterite Mg
 11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
Environments of Mineral Formation. Stability Diagrams
 Environments of Mineral Formation Unary, Binary, and Ternary Mineral Stability Diagrams Minerals of differing composition (or polymorphs of the same mineral) that coexist at a set of pressure (P) temperature
Environments of Mineral Formation Unary, Binary, and Ternary Mineral Stability Diagrams Minerals of differing composition (or polymorphs of the same mineral) that coexist at a set of pressure (P) temperature
Introductory Statement:
 The use of visualization and sketches of thin sections to encourage a better understanding of phase diagrams: Binary and ternary phase diagram exercises Jennifer M. Wenner Drew S. Coleman Introductory
The use of visualization and sketches of thin sections to encourage a better understanding of phase diagrams: Binary and ternary phase diagram exercises Jennifer M. Wenner Drew S. Coleman Introductory
Exam I. 1. (10 points) Give the following optical properties for the minerals listed below.
 GLY306 Petrology Exam I 1. (10 points) Give the following optical properties for the minerals listed below. Color/pleochroism Extinction angle Cleavage angle Twining Refractive index Hornblende Plagioclase
GLY306 Petrology Exam I 1. (10 points) Give the following optical properties for the minerals listed below. Color/pleochroism Extinction angle Cleavage angle Twining Refractive index Hornblende Plagioclase
Chapter 9: Trace Elements
 Lecture 13 Introduction to Trace Elements Wednesday, March 9, 2005 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8-2. Harker variation diagram for 310 analyzed volcanic rocks
Lecture 13 Introduction to Trace Elements Wednesday, March 9, 2005 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8-2. Harker variation diagram for 310 analyzed volcanic rocks
Differentiation of Magmas By Fractional Crystallization
 Wirth Magmatic Differentiation Using M&M s 1 HANDOUT Differentiation of Magmas By Fractional Crystallization Objective The objective of this exercise is to gain first-hand knowledge of the process of magmatic
Wirth Magmatic Differentiation Using M&M s 1 HANDOUT Differentiation of Magmas By Fractional Crystallization Objective The objective of this exercise is to gain first-hand knowledge of the process of magmatic
The Gibbs Phase Rule F = 2 + C - P
 The Gibbs Phase Rule The phase rule allows one to determine the number of degrees of freedom (F) or variance of a chemical system. This is useful for interpreting phase diagrams. F = 2 + C - P Where F
The Gibbs Phase Rule The phase rule allows one to determine the number of degrees of freedom (F) or variance of a chemical system. This is useful for interpreting phase diagrams. F = 2 + C - P Where F
GEOL 2312 Igneous and Metamorphic Petrology Spring 2016 Score / 58. Midterm 1 Chapters 1-10
 GEOL 2312 Igneous and Metamorphic Petrology Name KEY Spring 2016 Score / 58 Midterm 1 Chapters 1-10 1) Name two things that petrologists want to know about magmas (1 pt) Formation, source, composition,
GEOL 2312 Igneous and Metamorphic Petrology Name KEY Spring 2016 Score / 58 Midterm 1 Chapters 1-10 1) Name two things that petrologists want to know about magmas (1 pt) Formation, source, composition,
Phase transitions and exsolution phenomena in pyroxenes
 Phase transitions and exsolution phenomena in pyroxenes Cleavage in the pyroxenes 001 100 010 110 110 Optical micrograph showing two cleavages at 90 o Exsolution lamellae in pyroxenes Because exsolution
Phase transitions and exsolution phenomena in pyroxenes Cleavage in the pyroxenes 001 100 010 110 110 Optical micrograph showing two cleavages at 90 o Exsolution lamellae in pyroxenes Because exsolution
Metamorphic Petrology GLY 262 Metamorphic reactions and isograds
 Metamorphic Petrology GLY 262 Metamorphic reactions and isograds What do we mean by reaction? Reaction: change in the nature or types of phases in a system=> formation of new mineral(s) ) which are stable
Metamorphic Petrology GLY 262 Metamorphic reactions and isograds What do we mean by reaction? Reaction: change in the nature or types of phases in a system=> formation of new mineral(s) ) which are stable
Differentiation of Magmas By Fractional Crystallization Modified from Karl Wirth, rev. July 2011
 M&M s Magma Chamber 1 Differentiation of Magmas By Fractional Crystallization Modified from Karl Wirth, rev. July 2011 Objective The objective of this exercise is to gain first-hand knowledge of the process
M&M s Magma Chamber 1 Differentiation of Magmas By Fractional Crystallization Modified from Karl Wirth, rev. July 2011 Objective The objective of this exercise is to gain first-hand knowledge of the process
LAB 6: COMMON MINERALS IN IGNEOUS ROCKS
 GEOLOGY 17.01: Mineralogy LAB 6: COMMON MINERALS IN IGNEOUS ROCKS Part 2: Minerals in Gabbroic Rocks Learning Objectives: Students will be able to identify the most common silicate minerals in gabbroic
GEOLOGY 17.01: Mineralogy LAB 6: COMMON MINERALS IN IGNEOUS ROCKS Part 2: Minerals in Gabbroic Rocks Learning Objectives: Students will be able to identify the most common silicate minerals in gabbroic
Chapter 9: Trace Elements
 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8.2. Harker variation diagram for 310 analyzed volcanic rocks from Crater Lake (Mt. Mazama), Oregon Cascades. Data compiled by Rick
Chapter 9: Trace Elements Note magnitude of major element changes Figure 8.2. Harker variation diagram for 310 analyzed volcanic rocks from Crater Lake (Mt. Mazama), Oregon Cascades. Data compiled by Rick
Mon. Oct. 09, Reading: For Friday. Andrews-Hanna et al (GRAIL Procellarium region)
 Mon. Oct. 09, 2017 Reading: For Friday Zuber et al. 2013 Grail Lunar Gravity Andrews-Hanna et al. 2013 (GRAIL Procellarium region) Makeup lecture days -- This Friday?? No Class Oct. 16, 18, 20 Today: Lunar
Mon. Oct. 09, 2017 Reading: For Friday Zuber et al. 2013 Grail Lunar Gravity Andrews-Hanna et al. 2013 (GRAIL Procellarium region) Makeup lecture days -- This Friday?? No Class Oct. 16, 18, 20 Today: Lunar
Chapter 4 Rocks & Igneous Rocks
 Chapter 4 Rocks & Igneous Rocks Rock Definition A naturally occurring consolidated mixture of one or more minerals e.g, marble, granite, sandstone, limestone Rock Definition Must naturally occur in nature,
Chapter 4 Rocks & Igneous Rocks Rock Definition A naturally occurring consolidated mixture of one or more minerals e.g, marble, granite, sandstone, limestone Rock Definition Must naturally occur in nature,
Block: Igneous Rocks. From this list, select the terms which answer the following questions.
 Geology 12 Name: Mix and Match: Igneous Rocks Refer to the following list. Block: porphyritic volatiles mafic glassy magma mixing concordant discontinuous reaction series igneous vesicular partial melting
Geology 12 Name: Mix and Match: Igneous Rocks Refer to the following list. Block: porphyritic volatiles mafic glassy magma mixing concordant discontinuous reaction series igneous vesicular partial melting
Earth Science 232 Petrography
 Earth Science 232 Petrography Course notes by Shaun Frape and Alec Blyth Winter 2002 1 Petrology - Introduction Some Definitions Petra Greek for rock Logos Greek for disclosure or explanation Petrology
Earth Science 232 Petrography Course notes by Shaun Frape and Alec Blyth Winter 2002 1 Petrology - Introduction Some Definitions Petra Greek for rock Logos Greek for disclosure or explanation Petrology
Triangular Diagrams. 1. On the diagram above, plot the following (mostly mineral) compositions: silicon quartz corundum kyanite
 Triangular Diagrams Note: For this exercise, everything is plotted using mole%. Sometimes it is done with wt% but that just adds unnecessary complications. 1. On the diagram above, plot the following (mostly
Triangular Diagrams Note: For this exercise, everything is plotted using mole%. Sometimes it is done with wt% but that just adds unnecessary complications. 1. On the diagram above, plot the following (mostly
Partial melting of mantle peridotite
 Partial melting of mantle peridotite 1100 1200 1300 1400 1500 (TºC) Depth (km) 50 100 150 Plag lherzolite (ol-opx-cpx-pl) Spinel lherzolite (Ol-opx-cpx-sp) Garnet lherzolite (Ol-opx-cpx-gar) Graphite Diamond
Partial melting of mantle peridotite 1100 1200 1300 1400 1500 (TºC) Depth (km) 50 100 150 Plag lherzolite (ol-opx-cpx-pl) Spinel lherzolite (Ol-opx-cpx-sp) Garnet lherzolite (Ol-opx-cpx-gar) Graphite Diamond
Chapter 4 8/27/2013. Igneous Rocks. and Intrusive Igneous Activity. Introduction. The Properties and Behavior of Magma and Lava
 Introduction Chapter 4 Igneous rocks form by the cooling of magma (or lava). Large parts of the continents and all the oceanic crust are composed of. and Intrusive Igneous Activity The Properties and Behavior
Introduction Chapter 4 Igneous rocks form by the cooling of magma (or lava). Large parts of the continents and all the oceanic crust are composed of. and Intrusive Igneous Activity The Properties and Behavior
Melting Behavior of Simplified Lherzolite in the System CaO-MgO-Al 2 O 3 -SiO 2 -Na 2 O from 7 to 35 kbar
 Melting Behavior of Simplified Lherzolite in the System --Al 2 O 3 - -Na 2 O from 7 to 35 kbar by MICHAEL J. WALTER* AND DEAN C. PRESNALL Center for Lithospheric Studies, University of Texas at Dallas,
Melting Behavior of Simplified Lherzolite in the System --Al 2 O 3 - -Na 2 O from 7 to 35 kbar by MICHAEL J. WALTER* AND DEAN C. PRESNALL Center for Lithospheric Studies, University of Texas at Dallas,
amphibole PART 3 Pyroxene: augite CHAIN SILICATES
 amphibole PART 3 Pyroxene: augite CHAIN SILICATES CHAIN SILICATES = INOSILICATES inos = chains Basic structural group: Si 2 O 6 (each tetrahedra shared two corners) Simple or double chains linked by cations
amphibole PART 3 Pyroxene: augite CHAIN SILICATES CHAIN SILICATES = INOSILICATES inos = chains Basic structural group: Si 2 O 6 (each tetrahedra shared two corners) Simple or double chains linked by cations
GY303 Igneous & Metamorphic Petrology. Lecture 7: Magma Sources and Tectonic Environments
 GY303 Igneous & Metamorphic Petrology Lecture 7: Magma Sources and Tectonic Environments Factors controlling Magma production Source rock composition Amount of fluids, especially H 2 O Pressure (Depth)
GY303 Igneous & Metamorphic Petrology Lecture 7: Magma Sources and Tectonic Environments Factors controlling Magma production Source rock composition Amount of fluids, especially H 2 O Pressure (Depth)
A Rock is a solid aggregate of minerals.
 Quartz A Rock is a solid aggregate of minerals. Orthoclase Feldspar Plagioclase Feldspar Biotite Four different minerals are obvious in this piece of Granite. The average automobile contains: Minerals
Quartz A Rock is a solid aggregate of minerals. Orthoclase Feldspar Plagioclase Feldspar Biotite Four different minerals are obvious in this piece of Granite. The average automobile contains: Minerals
Imagine the first rock and the cycles that it has been through.
 A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one type of rocky material gets transformed into another The Rock Cycle Representation
A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one type of rocky material gets transformed into another The Rock Cycle Representation
Name Class Date STUDY GUIDE FOR CONTENT MASTERY
 Igneous Rocks What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. extrusive igneous rock intrusive
Igneous Rocks What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. extrusive igneous rock intrusive
Plate tectonics, rock cycle
 Dikes, Antarctica Rock Cycle Plate tectonics, rock cycle The Rock Cycle A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one
Dikes, Antarctica Rock Cycle Plate tectonics, rock cycle The Rock Cycle A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one
GLY 155 Introduction to Physical Geology, W. Altermann
 Earth Materials Systematic subdivision of magmatic rocks Subdivision of magmatic rocks according to their mineral components: Content of quartz SiO 2 ( free quartz presence) Quartz with conchoidal breakage
Earth Materials Systematic subdivision of magmatic rocks Subdivision of magmatic rocks according to their mineral components: Content of quartz SiO 2 ( free quartz presence) Quartz with conchoidal breakage
IGNEOUS ROCKS. SECTION 5.1 What are igneous rocks?
 Date Period Name IGNEOUS ROCKS SECTION.1 What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. basaltic
Date Period Name IGNEOUS ROCKS SECTION.1 What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. basaltic
UNIVERSITY OF EDINBURGH. College of Science and Engineering School of GeoSciences. Earth Materials UO4824 DEGREE EXAMINATION (MOCK) xxxxxxxxxxxxxxxxx
 UNIVERSITY OF EDINBURGH College of Science and Engineering School of GeoSciences Earth Materials UO4824 DEGREE EXAMINATION (MOCK) xxxxxxxxxxxxxxxxxxxx xxxxxxxxxxxxxxxxxxxxxx Chairman: External Examiners:
UNIVERSITY OF EDINBURGH College of Science and Engineering School of GeoSciences Earth Materials UO4824 DEGREE EXAMINATION (MOCK) xxxxxxxxxxxxxxxxxxxx xxxxxxxxxxxxxxxxxxxxxx Chairman: External Examiners:
Name Class Date. In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements.
 CHAPTER 5 Igneous Rocks SECTION 5.1 What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. basaltic
CHAPTER 5 Igneous Rocks SECTION 5.1 What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. basaltic
Lab 3 - Identification of Igneous Rocks
 Lab 3 - Identification of Igneous Rocks Page - 1 Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly
Lab 3 - Identification of Igneous Rocks Page - 1 Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly
EAS 4550: Geochemistry Problem Set 4 Solutions Due Sept. 27, 2017
 1. Interaction parameters for the enstatite diopside solid solution have been determined as follows: WH-En = 34.0 kj/mol, WH-Di = 24.74 kj/mol (assume WV and WS are 0). (a) Use the asymmetric solution
1. Interaction parameters for the enstatite diopside solid solution have been determined as follows: WH-En = 34.0 kj/mol, WH-Di = 24.74 kj/mol (assume WV and WS are 0). (a) Use the asymmetric solution
Classification of Igneous Rocks
 Classification of Igneous Rocks Textures: Glassy- no crystals formed Aphanitic- crystals too small to see by eye Phaneritic- can see the constituent minerals Fine grained- < 1 mm diameter Medium grained-
Classification of Igneous Rocks Textures: Glassy- no crystals formed Aphanitic- crystals too small to see by eye Phaneritic- can see the constituent minerals Fine grained- < 1 mm diameter Medium grained-
Igneous and Metamorphic Rock Forming Minerals. Department of Geology Mr. Victor Tibane SGM 210_2013
 Igneous and Metamorphic Rock Forming Minerals Department of Geology Mr. Victor Tibane 1 SGM 210_2013 Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011
Igneous and Metamorphic Rock Forming Minerals Department of Geology Mr. Victor Tibane 1 SGM 210_2013 Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011
Engineering Geology ECIV 2204
 Engineering Geology ECIV 2204 Instructor : Dr. Jehad Hamad 2017-2016 Chapter (3) Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth
Engineering Geology ECIV 2204 Instructor : Dr. Jehad Hamad 2017-2016 Chapter (3) Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth
9/4/2015. Feldspars White, pink, variable Clays White perfect Quartz Colourless, white, red, None
 ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks Chapter 3.0: Weathering & soils Chapter 4.0: Geological
ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks Chapter 3.0: Weathering & soils Chapter 4.0: Geological
Name Class Date STUDY GUIDE FOR CONTENT MASTERY
 Igneous Rocks What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. extrusive igneous rock intrusive
Igneous Rocks What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. extrusive igneous rock intrusive
Phase Diagram. Remains virtually constant in most applications Most phase diagrams at 1 atm
 Phase Diagram Reading: West 11-1 Also called equilibrium or constitutional diagram Represent the relationships between temperature and composition and quantities of phase at equilibrium Pressure also influences
Phase Diagram Reading: West 11-1 Also called equilibrium or constitutional diagram Represent the relationships between temperature and composition and quantities of phase at equilibrium Pressure also influences
Lab 4 - Identification of Igneous Rocks
 Lab 4 - Identification of Igneous Rocks Page - Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly recognize
Lab 4 - Identification of Igneous Rocks Page - Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly recognize
Lecture 36. Igneous geochemistry
 Lecture 36 Igneous geochemistry Reading - White Chapter 7 Today 1. Overview 2. solid-melt distribution coefficients Igneous geochemistry The chemistry of igneous systems provides clues to a number of important
Lecture 36 Igneous geochemistry Reading - White Chapter 7 Today 1. Overview 2. solid-melt distribution coefficients Igneous geochemistry The chemistry of igneous systems provides clues to a number of important
Chapter 4 Up from the Inferno: Magma and Igneous Rocks
 Chapter 4 Up from the Inferno: Magma and Igneous Rocks Up from the Inferno: Magma and Igneous Rocks Updated by: Rick Oches, Professor of Geology & Environmental Sciences Bentley University Waltham, Massachusetts
Chapter 4 Up from the Inferno: Magma and Igneous Rocks Up from the Inferno: Magma and Igneous Rocks Updated by: Rick Oches, Professor of Geology & Environmental Sciences Bentley University Waltham, Massachusetts
Lecture 6 - Igneous Rocks and Volcanoes
 Lecture 6 - Igneous Rocks and Volcanoes Learning objectives Understand and be able to predict where and why magma will be forming at different tectonic settings Understand the factors controlling magma
Lecture 6 - Igneous Rocks and Volcanoes Learning objectives Understand and be able to predict where and why magma will be forming at different tectonic settings Understand the factors controlling magma
LAB 2: SILICATE MINERALS
 GEOLOGY 640: Geology through Global Arts and Artifacts LAB 2: SILICATE MINERALS FRAMEWORK SILICATES The framework silicates quartz and feldspar are the most common minerals in Earth s crust. Quartz (SiO
GEOLOGY 640: Geology through Global Arts and Artifacts LAB 2: SILICATE MINERALS FRAMEWORK SILICATES The framework silicates quartz and feldspar are the most common minerals in Earth s crust. Quartz (SiO
Constitution of Magmas. Magmas. Gas Law. Composition. Atomic Structure of Magma. Structural Model. PV = nrt H 2 O + O -2 = 2(OH) -
 Constitution of Magmas Magmas Best, Ch. 8 Hot molten rock T = 700-1200 degrees C Composed of ions or complexes Phase Homogeneous Separable part of the system With an interface Composition Most components
Constitution of Magmas Magmas Best, Ch. 8 Hot molten rock T = 700-1200 degrees C Composed of ions or complexes Phase Homogeneous Separable part of the system With an interface Composition Most components
Rocks: Materials of the Solid Earth
 1 Rocks: Materials of the Solid Earth Presentation modified from: Instructor Resource Center on CD-ROM, Foundations of Earth Science,, 4 th Edition, Lutgens/Tarbuck, Rock Cycle Igneous Rocks Today 2 Rock
1 Rocks: Materials of the Solid Earth Presentation modified from: Instructor Resource Center on CD-ROM, Foundations of Earth Science,, 4 th Edition, Lutgens/Tarbuck, Rock Cycle Igneous Rocks Today 2 Rock
LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES
 Geology 316 (Petrology) (03/26/2012) Name LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES INTRODUCTION Ultramafic rocks are igneous rocks containing less than 10% felsic minerals (quartz + feldspars
Geology 316 (Petrology) (03/26/2012) Name LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES INTRODUCTION Ultramafic rocks are igneous rocks containing less than 10% felsic minerals (quartz + feldspars
Wed. Oct. 04, Makeup lecture time? Will Friday noon work for everyone? No class Oct. 16, 18, 20?
 Wed. Oct. 04, 2017 Reading: For Friday: Bugiolacchi et al. 2008 Laurence et al. 1998" Makeup lecture time? Will Friday noon work for everyone? No class Oct. 16, 18, 20? Today: Finish Lunar overview (from
Wed. Oct. 04, 2017 Reading: For Friday: Bugiolacchi et al. 2008 Laurence et al. 1998" Makeup lecture time? Will Friday noon work for everyone? No class Oct. 16, 18, 20? Today: Finish Lunar overview (from
Introduction. Volcano a vent where molten rock comes out of Earth
 Introduction Volcano a vent where molten rock comes out of Earth Example: Kilauea Volcano, Hawaii Hot (~1,200 o C) lava pools around the volcanic vent. Hot, syrupy lava runs downhill as a lava flow. The
Introduction Volcano a vent where molten rock comes out of Earth Example: Kilauea Volcano, Hawaii Hot (~1,200 o C) lava pools around the volcanic vent. Hot, syrupy lava runs downhill as a lava flow. The
Directed Reading. Section: Rocks and the Rock Cycle. made of a. inorganic matter. b. solid organic matter. c. liquid organic matter. d. chemicals.
 Skills Worksheet Directed Reading Section: Rocks and the Rock Cycle 1. The solid part of Earth is made up of material called a. glacial ice. b. lava. c. rock. d. wood. 2. Rock can be a collection of one
Skills Worksheet Directed Reading Section: Rocks and the Rock Cycle 1. The solid part of Earth is made up of material called a. glacial ice. b. lava. c. rock. d. wood. 2. Rock can be a collection of one
23/9/2013 ENGINEERING GEOLOGY. Chapter 2: Rock classification:
 ENGINEERING GEOLOGY Chapter 2: Rock classification: ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks
ENGINEERING GEOLOGY Chapter 2: Rock classification: ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks
Textures of Igneous Rocks
 Page 1 of 6 EENS 212 Prof. Stephen A. Nelson Petrology Tulane University This document last updated on 12-Feb-2004 Introduction to Igneous Rocks An igneous rock is any crystalline or glassy rock that forms
Page 1 of 6 EENS 212 Prof. Stephen A. Nelson Petrology Tulane University This document last updated on 12-Feb-2004 Introduction to Igneous Rocks An igneous rock is any crystalline or glassy rock that forms
Lecture 24 Hawaii. Hawaii
 Lecture 24 Hawaii Friday, April 22 nd 2005 Hawaii The Hawaiian Islands, in the middle of the Pacific Ocean, are volcanic islands at the end of a long chain of submerged volcanoes. These volcanoes get progressively
Lecture 24 Hawaii Friday, April 22 nd 2005 Hawaii The Hawaiian Islands, in the middle of the Pacific Ocean, are volcanic islands at the end of a long chain of submerged volcanoes. These volcanoes get progressively
Metamorphic Petrology GLY 262 Lecture 3: An introduction to metamorphism (II)
 Metamorphic Petrology GLY 262 Lecture 3: An introduction to metamorphism (II) Metamorphic processes Metamorphism is very complex and involves a large number of chemical and physical processes occurring
Metamorphic Petrology GLY 262 Lecture 3: An introduction to metamorphism (II) Metamorphic processes Metamorphism is very complex and involves a large number of chemical and physical processes occurring
AMHERST COLLEGE Department of Geology Geology 41: Environmental and Solid Earth Geophysics
 AMHERST COLLEGE Department of Geology Geology 41: Environmental and Solid Earth Geophysics Lab 1: Meteorites EQUIPMENT: notebook and pen only In this lab, we will examine thin sections and hand samples
AMHERST COLLEGE Department of Geology Geology 41: Environmental and Solid Earth Geophysics Lab 1: Meteorites EQUIPMENT: notebook and pen only In this lab, we will examine thin sections and hand samples
Ultramafic rocks. Types of Ultramafic Rocks. Spinel lherzolite xenolith
 Ultramafic rocks Definition: Color Index > 90, i.e., less than 10% felsic minerals. Not to be confused with Ultrabasic Rocks which are rocks with
Ultramafic rocks Definition: Color Index > 90, i.e., less than 10% felsic minerals. Not to be confused with Ultrabasic Rocks which are rocks with
GLY 155 Introduction to Physical Geology, W. Altermann. Grotzinger Jordan. Understanding Earth. Sixth Edition
 Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011 by W. H. Freeman and Company Chapter 4: Igneous Rocks: Solids from Melts 1 About Igneous Rocks Igneous
Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011 by W. H. Freeman and Company Chapter 4: Igneous Rocks: Solids from Melts 1 About Igneous Rocks Igneous
Principles of Igneous and Metamorphic Petrology John D. Winter Second Edition
 Principles of Igneous and Metamorphic Petrology John D. Winter Second Edition Pearson Education Limited Edinburgh Gate Harlow Essex CM20 2JE England and Associated Companies throughout the world Visit
Principles of Igneous and Metamorphic Petrology John D. Winter Second Edition Pearson Education Limited Edinburgh Gate Harlow Essex CM20 2JE England and Associated Companies throughout the world Visit
Occurrence of mafic-ultramafic rocks
 Occurrence of mafic-ultramafic rocks Mantle-derived magmas Oceanic Lithospheric mantle Continental lithospheric mantle Ultramafic xenoliths Oman ophiolite harzburgite upper mantle both from: http://www.bris.ac.uk/depts/geol/vft/oman.html
Occurrence of mafic-ultramafic rocks Mantle-derived magmas Oceanic Lithospheric mantle Continental lithospheric mantle Ultramafic xenoliths Oman ophiolite harzburgite upper mantle both from: http://www.bris.ac.uk/depts/geol/vft/oman.html
Mineral Stability and Phase Diagrams Introduction
 1 of 10 10/10/2002 2:50 PM Prof. Stephen A. Nelson Geology 211 Tulane University Mineralogy and Phase Diagrams Introduction This document last updated on 10-Oct-2002 As we discussed previously, there are
1 of 10 10/10/2002 2:50 PM Prof. Stephen A. Nelson Geology 211 Tulane University Mineralogy and Phase Diagrams Introduction This document last updated on 10-Oct-2002 As we discussed previously, there are
N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0
 N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0 is initial number of parents, D* is number of radiogenic daughter atoms, and λ is the decay
N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0 is initial number of parents, D* is number of radiogenic daughter atoms, and λ is the decay
GEOLOGY. Subject : GEOLOGY (For under graduate student.) Paper No. : Paper 02 Introduction to Geology 02
 GEOLOGY Subject : GEOLOGY (For under graduate student.) Paper No. : Paper 02 Introduction to Geology 02 Topic No. & Title : 37 Magma Bowen Series (Part 01) Academic Script What is Igneous Petrology? Igneous
GEOLOGY Subject : GEOLOGY (For under graduate student.) Paper No. : Paper 02 Introduction to Geology 02 Topic No. & Title : 37 Magma Bowen Series (Part 01) Academic Script What is Igneous Petrology? Igneous
EPSC 233. Compositional variation in minerals. Recommended reading: PERKINS, p. 286, 41 (Box 2-4).
 EPSC 233 Compositional variation in minerals Recommended reading: PERKINS, p. 286, 41 (Box 2-4). Some minerals are nearly pure elements. These are grouped under the category of native elements. This includes
EPSC 233 Compositional variation in minerals Recommended reading: PERKINS, p. 286, 41 (Box 2-4). Some minerals are nearly pure elements. These are grouped under the category of native elements. This includes
Geology 314 Accretion of the Bushveld complex
 Geology 314 Accretion of the Bushveld complex Foreword: This document is not a stand-alone document; it s written as an example showing how the documents of the prac 9 (Geol 314, Stellenbosch, 2007) can
Geology 314 Accretion of the Bushveld complex Foreword: This document is not a stand-alone document; it s written as an example showing how the documents of the prac 9 (Geol 314, Stellenbosch, 2007) can
WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY
 WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY Brandon E. Schwab Department of Geology Humboldt State University
WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY Brandon E. Schwab Department of Geology Humboldt State University
CERAMIC GLAZING as an IGNEOUS PROCESS
 GEOL 640: Geology through Global Arts and Artifacts CERAMIC GLAZING as an IGNEOUS PROCESS GLAZE COMPONENTS A glaze is a waterproof silica glass on the surface of a ceramic pot, and was first produced by
GEOL 640: Geology through Global Arts and Artifacts CERAMIC GLAZING as an IGNEOUS PROCESS GLAZE COMPONENTS A glaze is a waterproof silica glass on the surface of a ceramic pot, and was first produced by
EARTH SCIENCE. Geology, the Environment and the Universe. Chapter 5: Igneous Rocks
 EARTH SCIENCE Geology, the Environment and the Universe Chapter 5: Igneous Rocks CHAPTER 5 Igneous Rocks Section 5.1 What are igneous rocks? Section 5.2 Classification of Igneous Rocks Click a hyperlink
EARTH SCIENCE Geology, the Environment and the Universe Chapter 5: Igneous Rocks CHAPTER 5 Igneous Rocks Section 5.1 What are igneous rocks? Section 5.2 Classification of Igneous Rocks Click a hyperlink
Igneous Rock. Magma Chamber Large pool of magma in the lithosphere
 Igneous Rock Magma Molten rock under the surface Temperature = 600 o 1400 o C Magma Chamber Large pool of magma in the lithosphere Magma chamber - most all magma consists of silicon and oxygen (silicate)
Igneous Rock Magma Molten rock under the surface Temperature = 600 o 1400 o C Magma Chamber Large pool of magma in the lithosphere Magma chamber - most all magma consists of silicon and oxygen (silicate)
Oxide Structures & Networks
 Oxide Structures & Networks Unit Cell: Primitive Tetragonal (a = b c) 2TiO 2 per unit cell Motif: 2Ti at (0, 0, 0); ( 1 / 2, 1 / 2, 1 / 2 ) & 4O at ±(0.3, 0.3, 0); ±(0.8, 0.2, 1 / 2 ) Ti: 6 (octahedral
Oxide Structures & Networks Unit Cell: Primitive Tetragonal (a = b c) 2TiO 2 per unit cell Motif: 2Ti at (0, 0, 0); ( 1 / 2, 1 / 2, 1 / 2 ) & 4O at ±(0.3, 0.3, 0); ±(0.8, 0.2, 1 / 2 ) Ti: 6 (octahedral
Sphene (Titanite) Plane polarized light. Honey brown/orange Wedge-shaped crystals
 Sphene (Titanite) Plane polarized light Honey brown/orange Wedge-shaped crystals Sphene (Titanite) Crossed nicols High-order, washedout interference colors (light orange) #1 Rule for (Heavy) Radiogenic
Sphene (Titanite) Plane polarized light Honey brown/orange Wedge-shaped crystals Sphene (Titanite) Crossed nicols High-order, washedout interference colors (light orange) #1 Rule for (Heavy) Radiogenic
Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100
 Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100 Ray Rector - Instructor Major Concepts 1) Igneous rocks form directly from the crystallization of a magma or lava 2)
Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100 Ray Rector - Instructor Major Concepts 1) Igneous rocks form directly from the crystallization of a magma or lava 2)
Phase Transformations
 Phase Transformations Chapter 8 of Atkins: Sections 8.4-8.6 Temperature- Composition Diagrams Distillation of Mixtures Azeotropes Immiscible Liquids Liquid- Liquid Phase Diagrams Phase Separations Critical
Phase Transformations Chapter 8 of Atkins: Sections 8.4-8.6 Temperature- Composition Diagrams Distillation of Mixtures Azeotropes Immiscible Liquids Liquid- Liquid Phase Diagrams Phase Separations Critical
Topic 5 : Crystal chemistry
 GEOL360 LECTURE NOTES: T5 : CRYSTAL CHEMISTRY 1/15 GEOL360 Topic 5 : Crystal chemistry 5.1 Introduction what is a crystal? A crystal is a homogeneous, solid body of a chemical element, compound, or isomorphous
GEOL360 LECTURE NOTES: T5 : CRYSTAL CHEMISTRY 1/15 GEOL360 Topic 5 : Crystal chemistry 5.1 Introduction what is a crystal? A crystal is a homogeneous, solid body of a chemical element, compound, or isomorphous
The Nature of Igneous Rocks
 The Nature of Igneous Rocks Form from Magma Hot, partially molten mixture of solid liquid and gas Mineral crystals form in the magma making a crystal slush Gases - H 2 O, CO 2, etc. - are dissolved in
The Nature of Igneous Rocks Form from Magma Hot, partially molten mixture of solid liquid and gas Mineral crystals form in the magma making a crystal slush Gases - H 2 O, CO 2, etc. - are dissolved in
Igneous Rocks. Igneous Rocks. Genetic Classification of
 Igneous Rocks Fig. 5.1 Genetic Classification of Igneous Rocks Intrusive: crystallized from slowly cooling magma intruded within the Earth s crust; e.g. granite, gabbro 1 Fig. 5.2 Genetic Classification
Igneous Rocks Fig. 5.1 Genetic Classification of Igneous Rocks Intrusive: crystallized from slowly cooling magma intruded within the Earth s crust; e.g. granite, gabbro 1 Fig. 5.2 Genetic Classification
Melts of garnet lherzolite: experiments, models and comparison to melts of pyroxenite and carbonated lherzolite
 Melts of garnet lherzolite: experiments, models and comparison to melts of pyroxenite and carbonated lherzolite The MIT Faculty has made this article openly available. Please share how this access benefits
Melts of garnet lherzolite: experiments, models and comparison to melts of pyroxenite and carbonated lherzolite The MIT Faculty has made this article openly available. Please share how this access benefits
Cloud Condensation Chemistry. in Low-Mass Objects. Katharina Lodders Washington University, St. Louis
 Cloud Condensation Chemistry in Low-Mass Objects Katharina Lodders Washington University, St. Louis For a recent review on this topic, see Lodders & Fegley 2006, Astrophysics Update 2, Springer, p. 1 ff.
Cloud Condensation Chemistry in Low-Mass Objects Katharina Lodders Washington University, St. Louis For a recent review on this topic, see Lodders & Fegley 2006, Astrophysics Update 2, Springer, p. 1 ff.
Quartz. ! Naturally occurring - formed by nature. ! Solid - not liquid or gas. Liquid water is not a mineral
 GEOL 110 - Minerals, Igneous Rocks Minerals Diamond Azurite Quartz Why Study Minerals?! Rocks = aggregates of minerals! Importance to Society?! Importance to Geology? 5 part definition, must satisfy all
GEOL 110 - Minerals, Igneous Rocks Minerals Diamond Azurite Quartz Why Study Minerals?! Rocks = aggregates of minerals! Importance to Society?! Importance to Geology? 5 part definition, must satisfy all
The 3 types of rocks:
 Igneous Rocks and Intrusive Igneous Activity The 3 types of rocks:! Sedimentary! Igneous! Metamorphic Marble 1 10/7/15 SEDIMENTARY ROCKS Come from rocks sediments (rock fragments, sand, silt, etc.) Fossils
Igneous Rocks and Intrusive Igneous Activity The 3 types of rocks:! Sedimentary! Igneous! Metamorphic Marble 1 10/7/15 SEDIMENTARY ROCKS Come from rocks sediments (rock fragments, sand, silt, etc.) Fossils
Supplementary information. Jadeite in Chelyabinsk meteorite and the nature of an impact event on its parent. body
 Supplementary information Jadeite in Chelyabinsk meteorite and the nature of an impact event on its parent body Shin Ozawa 1*, Masaaki Miyahara 1, 2, Eiji Ohtani 1, 3, Olga N. Koroleva 4, Yoshinori Ito
Supplementary information Jadeite in Chelyabinsk meteorite and the nature of an impact event on its parent body Shin Ozawa 1*, Masaaki Miyahara 1, 2, Eiji Ohtani 1, 3, Olga N. Koroleva 4, Yoshinori Ito
Framework Silicates & Building the Crust. JD Price
 Framework Silicates & Building the Crust JD Price Silicate Structure Silicate Structure (SiO2) Zeolites Zeolites have cage-like structures that store water (or other large molecules). Typically found in
Framework Silicates & Building the Crust JD Price Silicate Structure Silicate Structure (SiO2) Zeolites Zeolites have cage-like structures that store water (or other large molecules). Typically found in
Effect of tectonic setting on chemistry of mantle-derived melts
 Effect of tectonic setting on chemistry of mantle-derived melts Lherzolite Basalt Factors controlling magma composition Composition of the source Partial melting process Fractional crystallization Crustal
Effect of tectonic setting on chemistry of mantle-derived melts Lherzolite Basalt Factors controlling magma composition Composition of the source Partial melting process Fractional crystallization Crustal
Earth and Planetary Materials
 Earth and Planetary Materials Spring 2013 Lecture 4 2013.01.16 Example Beryl Be 3 Al 2 (SiO 3 ) 6 Goshenite Aquamarine Emerald Heliodor Red beryl Morganite pure Fe 2+ & Fe 3+ Cr 3+ Fe 3+ Mn 3+ Mn 2+ Rules
Earth and Planetary Materials Spring 2013 Lecture 4 2013.01.16 Example Beryl Be 3 Al 2 (SiO 3 ) 6 Goshenite Aquamarine Emerald Heliodor Red beryl Morganite pure Fe 2+ & Fe 3+ Cr 3+ Fe 3+ Mn 3+ Mn 2+ Rules
Pyroxene, amphibole, and feldspar
 Pyroxene, amphibole, and feldspar Sometimes, common minerals in igneous and metamorphic rocks are hard to distinguish, unless you know the tricks. Colored mineral such as pyroxene and amphibole can be
Pyroxene, amphibole, and feldspar Sometimes, common minerals in igneous and metamorphic rocks are hard to distinguish, unless you know the tricks. Colored mineral such as pyroxene and amphibole can be
Igneous Rocks. Igneous Rocks - 1. Environment of Formation - Magma - Plutonic - rock that formed within the Earth. Intrusive - Earth s crust.
 Name: Date: Period: Minerals and Rocks The Physical Setting: Earth Science CLASS NOTES - Methods to classify igneous rocks: 1. Environment of Formation - Magma - Plutonic - rock that formed within the
Name: Date: Period: Minerals and Rocks The Physical Setting: Earth Science CLASS NOTES - Methods to classify igneous rocks: 1. Environment of Formation - Magma - Plutonic - rock that formed within the
Metamorphism (means changed form
 Metamorphism (means changed form) is recrystallization without melting of a previously existing rock at depth in response to a change in the environment of temperature, pressure, and fluids. Common minerals
Metamorphism (means changed form) is recrystallization without melting of a previously existing rock at depth in response to a change in the environment of temperature, pressure, and fluids. Common minerals
Lecture 8: Igneous Petrogenesis. Igneous rock classification Phase relations Mantle melting Trace element geochemistry
 Lecture 8: Igneous Petrogenesis Igneous rock classification Phase relations Mantle melting Trace element geochemistry >70% of Earth s annual volcanic budget is erupted in the oceans. Igneous Rock Classification:
Lecture 8: Igneous Petrogenesis Igneous rock classification Phase relations Mantle melting Trace element geochemistry >70% of Earth s annual volcanic budget is erupted in the oceans. Igneous Rock Classification:
Earth Science 11: Earth Materials: Rock Cycle
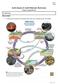 Name: Date: Earth Science 11: Earth Materials: Rock Cycle Chapter 2, pages 44 to 46 2.1: Rock Cycle What is a Rock? A solid mass of mineral or mineral-like matter that occurs naturally as part of our planet
Name: Date: Earth Science 11: Earth Materials: Rock Cycle Chapter 2, pages 44 to 46 2.1: Rock Cycle What is a Rock? A solid mass of mineral or mineral-like matter that occurs naturally as part of our planet
