Supplementary Material. Chemical ligation from O-acyl isopeptides via 8- and 11-membered cyclic transition states
|
|
|
- Hugo Patrick
- 5 years ago
- Views:
Transcription
1 Supplementary Material Chemical ligation from -acyl isopeptides via 8- and 11-membered cyclic transition states Siva S. Panda, a Mohamed Elagawany, a,b Hadi M. Marwani, c Eray Çalışkan, a Mirna El Khatib, a Alexander liferenko, a Khalid A. Alamry, c and Alan R. Katritzky* a,c a Center for Heterocyclic Compounds, Department of Chemistry, University of Florida, Gainesville, FL (USA) b Department of rganic Chemistry, College of Pharmacy, Misr University for Science and Technology, Al-Motamayez District, P.. Box: 77, Egypt c Department of Chemistry, King Abdulaziz University, Jeddah, 21589, Saudi Arabia katritzky@chem.ufl.edu Table of Contents 1. 1 H NMR and 13 C NMR spectra for compounds S2 2. HPLC chromatograms and MS Spectra for 5a and 6a S42 3. HPLC chromatograms and MS Spectra for 5f and 6f S 4. HPLC chromatograms and MS Spectra for 5c and 6c S62 5. HPLC chromatograms and MS Spectra for 9a and a S64 Page S1
2 Page S2
3 Page S3
4 Page S4
5 Page S5
6 Page S6
7 Page S7
8 Page S8
9 Page S9
10 Page S
11 Page S11
12 Page S12
13 Page S13
14 Page S14
15 Page S15
16 Page S16
17 Page S17
18 Page S18
19 Page S19
20 Page S
21 Page S21
22 Page S22
23 Page S23
24 Page S24
25 Page S25
26 Page S26
27 Page S27
28 Page S28
29 Page S29
30 Page S
31 Page S31
32 Page S32
33 Page S33
34 Page S34
35 Page S35
36 Page S36
37 Page S37
38 Page S38
39 Page S39
40 Page S
41 Page S41
42 HPLC chromatograms and MS Spectra for 5a and 6a Page S42
43 Figure S1. Lig8 NonAq, MW 9-A E:\-Spec\-Data\Seq /13/11 3:41:29 PM Lig8NNAQ, mg/ml; 5 ul injected Hydro-RP;.2;:()>55:45(15)>25:75(45)>5:95(-)/254 nm/(-)esi-ms SEQ # RT: AV: 4 SB: NL: 1.11E7 T: - c ESI sid=1. Full ms [ 1.-.] SEQ # RT: AV: 5 SB: NL: 9.1E5 F: - c ESI sid=3. Full ms [ 6.-.] Page S43
44 Figure S2. MW 9-B, Lig8 NonAq E:\-Spec\-Data\Seq /13/11 3:41:29 PM Lig8NNAQ, mg/ml; 5 ul injected Hydro-RP;.2;:()>55:45(15)>25:75(45)>5:95(-)/254 nm/(-)esi-ms SEQ #23-61 RT: AV: 5 SB: NL: 1.62E7 T: - c ESI sid=1. Full ms [ 1.-.] SEQ #28-53 RT: AV: 3 SB: NL: 8.26E5 F: - c ESI sid=3. Full ms [ 6.-.] Page S44
45 Figure S3. Lig8 NonAq: HPLC/UV/(-)ESI-MS and MSn chromatograms for the MW 9 compounds E:\-Spec\-Data\Seq /13/11 3:41:29 PM Lig8NNAQ, mg/ml; 5 ul injected Hydro-RP;.2;:()>55:45(15)>25:75(45)>5:95(-)/254 nm/(-)esi-ms RT: Relative Intensity Tim e (m in) Page S NL: 3.19E7 Base Peak F: - c ESI sid=1. Full ms [ 1.-.] MS SEQ NL: 1.68E6 Base Peak F: - c ESI sid=3. Full ms [ 6.-.] MS SEQ NL: 3.19E7 = F: - c ESI sid=1. Full ms [ 1.-.] MS SEQ NL: 1.67E7 Base Peak F: - c ESI sid=2. Full ms2 8.@37. [ 1.-4.] MS SEQ NL: 1.99E6 Base Peak F: - c sid=2. d Full ms3 8. MS SEQ NL: 1.17E-1 UV Analog SEQ
46 Figure S4. MW 9 compounds: (-)ESI-MS/MS (top and 3rd) and MS/MS/MS (2nd and bottom) for the MW 9-A (RT min) and MW 9-B (RT 23.7 min) E:\-Spec\-Data\Seq /13/11 3:41:29 PM Lig8NNAQ, mg/ml; 5 ul injected Hydro-RP;.2;:()>55:45(15)>25:75(45)>5:95(-)/254 nm/(-)esi-ms SEQ # RT: AV: 3 NL: 2.86E6 F: - c ESI sid=2. Full ms2 8.@37. [ 1.-4.] SEQ # RT: AV: 3 NL: 6.14E5 T: - c sid=2. d Full ms3 8.@ @37. [ ] SEQ # RT: AV: 4 NL: 1.2 1E 7 F: - c ESI sid=2. Full ms2 8.@37. [ 1.-4.] SEQ # RT: AV: 3 NL: 1.57E6 T: - c sid=2. d Full ms3 8.@ @37. [.-675.] Page S46
47 Figure S5. Lig8 NonAq: MW 9 isomers. The two MW 9 isomers were well separated and were characterized relatively uniquely by their (-)ESI-MS/MS product ions as shown below. The MW 9-A was the starting material while MW 9-B was SEQ /13/11 3:41:29 PM Lig8NNAQ, mg/ml; 5 ul injected Hydro-RP;.2;:()>55:45(15)>25:75(45)>5:95(-)/254 nm/(-)esi-ms RT: NL: 3.19E7 = F: - c ESI sid=1. Full ms [ 1.-.] MS SEQ (-)ESI-MS, (-)ESI-MS/MS 8 => 174 RT: BP: MW 9-A NL: 3.34E6 = F: - c ESI sid=2. Full ms2 8.@37. [ 1.-4.] MS SEQ (-)ESI-MS/MS 8 => 334 RT: BP: MW 9-B NL: 1.69E7 = F: - c ESI sid=2. Full ms2 8.@37. [ 1.-4.] MS SEQ Time (min) product. Scheme 1 shows the likely formation of the two product ions below Page S47
48 Scheme S1. The two MW 9 compounds are characterized by the formation of different major products ions from the (-)ESI- MS/MS collision-induced dissociation (CID) of their 8 [M-H]- ions. 1. Molecular Formula = C 19 H 27 N 3 7 Monoisotopic Mass = Da H 2 N H HN NH H 3 C CH 3 CH 3 (-)ESI 8 [M-H]- CID - H 3 C NH 174 CH 3 CH 3 solution phase chemistry H 3 C H 3 C CH 3 NH N HN (-)ESI 8 [M-H]- CID HN HN HN H H - H Molecular Formula = C 19 H 27 N 3 7 Monoisotopic Mass = Da 334 Page S48
49 Figure S6. Lig8 SM, MW 9, RT 19. min: With (-)ESI-MS, the MW 9 produced an 8 [M-H]- ion (top) which was dissociated to 174 (middle) which was further dissociated to (bottom). The RT and MSn spectra match those of the MW 9-A compound of Lig8 NonAq SEQ /13/11 6:41:18 PM Lig8 SM; mg/ml; 5 ul injected Hydro-RP;.2;:()>55:45(15)>25:75(45)>5:95(-)/254 nm/(-)esi-ms SEQ #812-6 RT: AV: 13 NL: 2.1E7 T: - c ESI sid=1. Full ms [ 1.-.] SEQ # RT: AV: NL: 5.95E6 F: - c ESI sid=2. Full ms2 8.@37. [ 1.-4.] SEQ # RT: AV: NL: 1.13E6 T: - c sid=2. d Full ms3 8.@ @37. [ ] Page S49
50 HPLC chromatograms and MS Spectra for 5f and 6f. Figure S7. MW 423, Rt 38.4 min: This was the most abundant MS and UV peaks. SEQ /22/11 7:31:19 AM ME36 in ul MeH; 5 ul injected XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi RT: NL: 1.22E Base Peak F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 2.25E RT: MA: BP: RT: MA: BP: 446. RT: MA: BP: MW Base Peak F: + c ESI sid=3. Full ms [ 7.-.] MS SEQ NL: 1.22E8 = F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 2.65E7 = F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 7.15E7 = F: + c ESI sid=3. Full ms [ 7.-.] MS SEQ NL: 8.93E-2 Intensity UV Analog SEQ Time (min) Page S
51 Figure S8.Sample ME36, MW 423. Due to the high concentration, self-adduct ions ( 8-958; 13-14) were also formed in addition to the 424 [M+H]+ and 445 [M+Na]+. Note the presence of other compounds also, e.g SEQ /22/11 7:31:19 AM ME36 in ul MeH; 5 ul injected XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi SEQ # RT: AV: 6 NL: 5.63E7 T: + c ESI sid=1. Full ms [ ] SEQ # RT: AV: 5 NL: 1.45E7 F: + c ESI sid=3. Full ms [ 7.-.] Page S51
52 Figure S9.Sample ME36, MW 423. (+)ESI-MSn. SEQ /22/11 7:31:19 AM ME36 in ul MeH; 5 ul injected XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi SEQ # RT: AV: 6 NL: 5.63E7 T: + c ESI sid=1. Full ms [ ] SEQ # RT: AV: 3 NL: 5.2E7 F: + c ESI sid=2. Full ms2 424.@37. [ ] SEQ # RT: AV: 2 NL: 2.E7 T: + c sid=2. d Full ms3 424.@ @37. [.-745.] Page S52
53 Figure S. ME37, MW 423 compounds: (+)ESI mass spectra of RT 38.6 min (top) and RT 39.5 min (bottom) The 324 and 368 are fragment ions. SEQ /22/11 5:54:32 AM ME37; 5 ul XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi SEQ # RT: AV: 2 SB: , NL: 2.12E8 T: + c ESI sid=1. Full ms [ ] SEQ #1551 RT: 39. AV: 1 SB: , NL: 2.49E7 T: + c ESI sid=1. Full ms [ ] Page S53
54 Figure S11. ME37, MW 423 compounds: (+)ESI mass spectra ( 7-) of RT 38.6 min (top) and RT 39.5 min (bottom).. SEQ /22/11 5:54:32 AM ME37; 5 ul XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi SEQ # RT: AV: 3 SB: , NL: 7.87E6 F: + c ESI sid=3. Full ms [ 7.-.] SEQ # RT: AV: 2 SB: , NL: 9.6E6 F: + c ESI sid=3. Full ms [ 7.-.] Page S54
55 Figure S12. MW 423 RT 38.5 min: (+)ESI-MS/MS of the 424 [M+H]+ ions from ME37 (top) and ME 36 (bottom) were identical. SEQ /22/11 5:54:32 AM ME37; 5 ul XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi SEQ # RT: AV: 3 NL: 1.19E8 F: + c ESI sid=2. Full ms2 424.@37. [ ] 368. ME SEQ # RT: AV: 2 NL: 6.55E7 F: + c ESI sid=2. Full ms2 424.@37. [ ] 368. ME Page S55
56 Figure S13. MW 423 compounds in ME37. (+)ESI-MS/MS (top and 3 rd ) and MS/MS/MS (2 nd and 4 th ) for the 38.6 min (top 2) and RT 39.5 min (bottom 2) MW 423 compounds. The corresponding spectra are near identical. SEQ /22/11 5:54:32 AM ME37; 5 ul XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi SEQ # RT: AV: 2 NL: 1.4 1E8 F: + c ESI sid=2. Full ms2 424.@37. [ ] m/ z SEQ # RT: AV: 2 NL: 5.38E7 T: + c sid=2. d Full ms3 424.@ @37. [.-745.] m/ z SEQ # RT: AV: 2 NL: 1.36E7 F: + c ESI sid=2. Full ms2 424.@37. [ ] m/ z SEQ # RT: AV: 2 NL: 5.34E6 T: + c sid=2. d Full ms3 424.@ @37. [.-745.] m/ z Page S56
57 Figure S14. MW 423 compounds, integrated peak areas. The corresponding areas should be summed. SEQ /22/11 5:54:32 AM ME37; 5 ul XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi RT: RT: MA: BP: 368. RT: 39. MA: BP: 368. NL: 2.15E8 = F: + c ESI sid=1. Full ms [ ] MS SEQ RT: MA: BP: RT: 39. MA: BP: 424. NL: 2.E8 = F: + c ESI sid=1. Full ms [ ] MS SEQ RT: MA: BP: RT: MA: BP: NL: 5.47E7 = F: + c ESI sid=3. Full ms [ 7.-.] MS SEQ NL: 2.1E8 = F: + c ESI sid=2. Full ms2 424.@37. [ ] MS SEQ Time (min) Page S57
58 Figure S15. MW 176: This compound was not retained on the HPLC column and eluted at RT 2.8 min. Due to its ion-chemistry and abundance, it produced mostly self-adduct ions. Note also that it produced 221 [M-H+Na+Na]+ and the neutral for adduction was the MW 198 [M-H+Na]. There also appeared to be CH2 homolog at 235. The adduct ions were likely homo (all MW 198 or all MW 212) and hetero(mixture of the 2 ions). SEQ /22/11 5:54:32 AM ME37; 5 ul XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi SEQ #3-116 RT: AV: 2 NL: 2.39E6 T: + c ESI sid=1. Full ms [ ] SEQ # RT: AV: 2 SB: , NL: 1.32E6 F: + c ESI sid=3. Full ms [ 7.-.] RT: Time (min) Page S58 NL: 8.52E3 = F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 5.38E5 = F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 3.E6 = F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 6.85E6 = F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 2.46E7 TIC F: + c ESI sid=3. Full ms [ 7.-.] MS SEQ NL: 2.65E4 Base Peak F: + c ESI sid=4. Full ms2 177.@37. [.-.] MS SEQ
59 Figure S16. MW 176: (+)ESI-MS (top) and MS/MS f 177 (very weak; middle) and of 419 [2(M-H+Na)+Na]+ (bottom). SEQ /22/11 5:54:32 AM ME37; 5 ul XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi SEQ #3-116 RT: AV: 2 NL: 2.39E6 T: + c ESI sid=1. Full ms [ ] SEQ #1 RT: 2.76 AV: 1 SB: , NL: 2.6E3 F: + c ESI sid=4. Full ms2 177.@37. [.-.] SEQ #5 RT: 2. AV: 1 NL: 1.14E6 T: + c sid=1. d Full ms @37. [ 5.-8.] Page S59
60 Figure S17. MW 176: HPLC/(+)ESI-MS ion-peaks integrated. SEQ /22/11 5:54:32 AM ME37; 5 ul XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi RT: RT: 2.79 MA: BP: RT: 2.79 MA: BP: RT: 2.97 MA: 8674 BP: RT: 2.78 MA: BP: 815. RT: 2.78 MA: 7487 BP: 13.1 NL: 5.45E6 = F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 3.E6 = F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 3.7E6 = F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 5.42E6 = F: + c ESI sid=3. Full ms [ 7.-.] MS SEQ NL: 4.29E6 = F: + c ESI sid=3. Full ms [ 7.-.] MS SEQ Time (min) Page S
61 Figure S18. MW 6? The MW 6 should have produced an 671 [M+H]+ and 693 [M+Na]+ ions. There were no correlating ion-peaks for these two ions. There was a very small 671 ion-peak at RT but the signal was too low to be conclusive. The (+)ESI mass spectra for the relataively abundant 693 ion-peak at Rt does not indicate an 671. The presence of an 715 suggests this is a MW 692 compound. I have integrated the 693 ion-peak. SEQ /22/11 5:54:32 AM ME37; 5 ul XTerra MS C18;.15;:()>5:95(45-)/254 nm/(+)esi SEQ # RT: AV: 2 SB: , NL: 4.49E7 T: + c ESI sid=1. Full ms [ ] RT: NL: 4.E5 = F: + c ESI sid=1. Full ms [ ] MS SEQ RT: MA: BP: NL: 2.57E7 = F: + c ESI sid=1. Full ms [ ] MS SEQ Time (min) Page S61
62 HPLC chromatograms and MS Spectra for 5c and 6c Figure S19. MW 333, RT 18.1 min. The MW 333 compound formed 334 [M+H]+, 356 [M+Na]+ and 378 [M- H+2Na]+ ions (top) with the latter being indicative of an acidic proton. The 334 was readily dissociated to 278 (middle) which was dissociated further to 234, and several other secondary product ions (bottom). SEQ //11 1:27:36 PM ME33 in ul MeH; 5 ul injected Hydro-RP;.2;95:5()>5:95(45-)/254 nm/(+)esi SEQ # RT: AV: 5 SB: NL: 1.54E7 F: + c ESI sid=1. Full ms [ ] SEQ # RT: AV: 8 NL: 2.53E7 F: + c ESI Full ms2 334.@37. [.-3.] SEQ # RT: AV: 5 NL: 6.69E6 T: + c d Full ms3 334.@ @37. [ ] Page S62
63 Figure S. MW 333 compounds. A new MW 333 compound eluted at RT.32 min (shaded peak). The unshaded 334 and 334 => 279 peaks are due to SM compounds SEQ //11 11:47:23 AM ME34 in ul MeH; 5 ul injected Hydro-RP;.2;95:5()>5:95(45-)/254 nm/(+)esi RT: RT:.32 BP: RT:.27 BP: NL: 3.E7 Base Peak F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 1.72E7 = F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 4.65E7 = F: + c ESI Full ms2 334.@37. [.-3.] MS SEQ NL: 1.19E-1 UV Analog SEQ Intensity Time (min) Page S63
64 HPLC chromatograms and MS Spectra for 9a and a Scheme S2. taken from the submitted reaction NH 2 HN LIG11SM (SEQ-16928) Molecular Formula = C 21 H N 4 8 Monoisotopic Mass = u H HN NH H 3 C CH 3 CH 3 Products: SEQ LIG11NN-AQ SEQ-169 LIG11AQ7.65 NH CH 3 NH CH 3 NH 2 HN H 3 C CH 3 HN H 3 C CH 3 HN HN HN HN H HN H H HN NH H 3 C CH 3 CH 3 H HN H Molecular Formula = C 21 H N 4 8 Monoisotopic Mass = Da Molecular Formula = C 28 H 41 N 5 11 Monoisotopic Mass = Da Molecular Formula = C 16 H 22 N 4 6 Monoisotopic Mass = Da Page S64
65 Figure S21. LIG11SM, MW 466, RT min. The MW 466 produced 467 [M+H]+, 489 [M+Na]+ and 511 [(M- H+Na)+Na]+ ions (top). The latter is characteristic of compounds with acidic protons, e.g. RCH SEQ /1/11 6:54:43 AM LIG11SM in ul ACN; ul injected Hypurity C8;.2;:()>:(15)>:(45-)/254 nm/(+)esi-ms SEQ #966-1 RT: AV: 4 SB : , NL: 9.46E6 T: + c ESI sid=2. Full ms [ 2.-.] SEQ # 964 RT: 25.3 AV: 1 NL: 5.72E6 T: + c sid=2. d Full ms @37. [ 1.-9.] SEQ # 965 RT: 25.6 AV: 1 NL: 1.32E6 T: + c sid=2. d Full ms @ @37. [ ] Page S65
66 Figure S22. LIG11NNAQ, MW 466, RT min. The MW 466 product was expected to produce 467 [M+H]+ and 489 [M+Na]+ ions. The mass chromatograms show only one potential MW 466 compound at RT SEQ /1/11 :8:28 AM LIG11NNAQ; ul Hypurity C8;.2;:()>:(15)>:(45-)/254 nm/(+)esi-ms RT: Intensity RT: BP: 467. RT: BP: Time (min) NL: 4.17E7 Base Peak F: + c ESI sid=1. Full ms [ ] MS SEQ NL: 3.13E7 Base Peak F: + c ESI sid=2. Full ms [ 3.-.] MS SEQ NL: 4.33E5 = F: + c ESI sid=2. Full ms [ 3.-.] MS SEQ NL: 6.47E6 = F: + c ESI sid=2. Full ms [ 3.-.] MS SEQ NL: 4.69E-1 UV Analog SEQ Page S66
67 Figure S23. LIG11 NNAQ via HPLC/UV/(-)ESI-MSn, MW 466, RT min. The tentative MW 466 detected via (+)ESI-MS above produced predominantly an 465 [M-H]- ion under (-)ESI-MS conditions and thus confirmed it as being a MW 466 compound SEQ /1/11 11:43:24 AM LIG11NNAQ; ul Hypurity C8;.2;:()>:(15)>:(45-)/254 nm/(-)esi-ms SEQ # RT: AV: 7 SB: , NL: 1.13E7 T: - c ESI sid=2. Full ms [ 3.-.] RT: RT: BP: NL: 1.12E7 Base Peak F: - c ESI sid=1. Full ms [ ] MS SEQ NL: 1.94E7 Base Peak F: - c ESI sid=2. Full ms [ 3.-.] MS SEQ NL: 1.94E7 = F: - c ESI sid=2. Full ms [ 3.-.] MS SEQ NL: 5.29E-1 UV Analog SEQ Intensity Time (min) Page S67
68 Figure S24. HPLC/(-)ESI-MS mass chromatograms of 465. MW 466 compounds in (top to bottom): LIG11SM, LIG11NNAQ and LIG11 AQ nly a very minor amount of MW 466 was detected in the LIG11 AQ7.65 sample. Note the products eluted prior to the starting material. SEQ /1/11 4:14:2 PM LIG11SM in ul ACN; ul injected Hypurity C8;.2;:()>:(15)>:(45-)/254 nm/(-)esi-ms RT: RT: BP: LIG11SM, MW RT: BP: LIG11NNAQ, MW 466 NL: 3.E6 = F: - c ESI sid=2. Full ms [ 3.-.] MS SEQ NL: 1.94E7 = F: - c ESI sid=2. Full ms [ 3.-.] MS SEQ RT: BP: LIG11 AQ 7.65, MW Time (min) NL: 1.63E5 = F: - c ESI sid=2. Full ms [ 3.-.] 38.5 MS SEQ Page S68
69 Figure S25. (-)ESI-MS/MS of the 465 [M-H]- ions of the MW 466 compounds in (top to bottom): LIG11SM, LIG11NNAQ and LIG11 AQ While it was expected that the starting material and products were different, the two MW 466 products also produced different dissociation ions. SEQ /1/11 4:14:2 PM LIG11SM in ul ACN; ul injected Hypurity 2. C8;.2;:()>:(15)>:(45-)/254 nm/(-)esi-ms SEQ # RT: AV: 8 NL: 6.88E4 F: - c ESI sid=2. Full ms2 465.@37. [ ] SEQ # RT: AV: 5 NL: 1.54E7 F: - c ESI sid=2. Full ms2 465.@37. [ ] SEQ # RT: AV: 3 SB : NL: 1.29E5 F: - c ESI sid=2. Full ms2 465.@37. [ ] Page S69
70 Figure S26. LIG11SM, MW 466, (-)ESI-MSn of the 465 [M-H]- ion. 3. SEQ /1/11 4:14:2 PM LIG11SM in ul ACN; ul injected Hypurity C8;.2;:()>:(15)>:(45-)/254 nm/(-)esi-ms SEQ # RT: AV: 8 NL: 6.88E4 F: - c ESI sid=2. Full ms2 465.@37. [ ] SEQ # 959 RT: AV: 1 NL: 3.58E4 T: - c sid=2. d Full ms3 465.@ @37. [ 5.-8.] SEQ # RT: AV: 3 NL: 1.14E5 T: - c sid=2. d Full ms3 465.@ @37. [ ] Page S
71 Figure S27. LIG11 NN-AQ, MW 466, RT min: (-)ESI-MS/MS (top) and MS/MS/MS (bottom) of the 465 [M-H]- ion. 4. SEQ /1/11 11:43:24 AM LIG11NNAQ; ul Hypurity C8;.2;:()>:(15)>:(45-)/254 nm/(-)esi-ms SEQ # RT: AV: 5 NL: 1.54E7 F: - c ESI sid=2. Full ms2 465.@37. [ ] SEQ # RT: AV: 5 NL: 3.24E6 T: - c sid=2. d Full ms3 465.@ @37. [ ] Page S71
72 Figure S28. LIG11 AQ7.65, MW 466, RT min. The 465 [M-H]- was very minor in its (-)ESI-MS (top). The dominant ion was 5. It is not uncommon to see [M+59]- ions where 59 is the acetate anion (from acetic acid often used in the mobile phase). Might the 465 be an [M+59]- ion of a MW 6 compound? The 465 was dissociated to 433, 397 and 365 ( 397 may be background). The 433 was dissociated further to 365 and 246 (bottom). SEQ /1/11 2:41:12 PM LIG11AQ7.65, ul Hypurity C8;.2;:()>:(15)>:(45-)/254 nm/(-)esi-ms SEQ # RT: AV: 3 SB: , NL: 2.32E6 T: - c ESI sid=2. Full ms [ 3.-.] SEQ # RT: AV: 2 SB: , NL: 1.4 3E5 F: - c ESI sid=2. Full ms2 465.@37. [ ] SEQ # 869 RT: AV: 1 NL: 5.83E4 T: - c sid=2. d Full ms3 465.@ @37. [ ] Page S72
Analytical Technologies and Compound Identification. Daniel L. Norwood, MSPH, PhD SCĪO Analytical Consulting, LLC.
 Analytical Technologies and Compound Identification Daniel L. Norwood, MSPH, PhD SCĪ Analytical Consulting, LLC. Extractables/Leachables Characterization (USP) Characterization is the discovery,
Analytical Technologies and Compound Identification Daniel L. Norwood, MSPH, PhD SCĪ Analytical Consulting, LLC. Extractables/Leachables Characterization (USP) Characterization is the discovery,
Welcome! Course 7: Concepts for LC-MS
 Welcome! Mass Spectrometry meets Cheminformatics Tobias Kind and Julie Leary UC Davis Course 7: Concepts for LC-MS Class website: CHE 241 - Spring 28 - CRN 16583 Slides: http://fiehnlab.ucdavis.edu/staff/kind/teaching/
Welcome! Mass Spectrometry meets Cheminformatics Tobias Kind and Julie Leary UC Davis Course 7: Concepts for LC-MS Class website: CHE 241 - Spring 28 - CRN 16583 Slides: http://fiehnlab.ucdavis.edu/staff/kind/teaching/
Hypericin Analysis by LC/MS
 Hypericin Analysis by LC/MS Jeanne B. Li and Michael P. Balogh LC/MS Waters Corporation LC GC, Vol 17, No. 6, June 1999 Introduction to hypericin analysis by LC/MS Hypericin and Related Compounds O Protohypericin
Hypericin Analysis by LC/MS Jeanne B. Li and Michael P. Balogh LC/MS Waters Corporation LC GC, Vol 17, No. 6, June 1999 Introduction to hypericin analysis by LC/MS Hypericin and Related Compounds O Protohypericin
Technical Note. Introduction
 Technical Note Analysis and Characterization of Psilocybin and Psilocin Using Liquid Chromatography - Electrospray Ionization Mass Spectrometry (LC-ESI-MS) with Collision-Induced-Dissociation (CID) and
Technical Note Analysis and Characterization of Psilocybin and Psilocin Using Liquid Chromatography - Electrospray Ionization Mass Spectrometry (LC-ESI-MS) with Collision-Induced-Dissociation (CID) and
Construction and Use of API Single- Quadrupole MS Spectral Libraries.
 Paul Zavitsanos Product Marketing Manager September 11, 2003 Construction and Use of API Single- Quadrupole MS Spectral Libraries. Time: 11:00 a.m. EDT Telephone Number: 888-840-7687 International Number:
Paul Zavitsanos Product Marketing Manager September 11, 2003 Construction and Use of API Single- Quadrupole MS Spectral Libraries. Time: 11:00 a.m. EDT Telephone Number: 888-840-7687 International Number:
An Effective Workflow for Impurity Analysis Incorporating High Quality HRAM LCMS & MSMS with Intelligent Automated Data Mining
 An Effective Workflow for Impurity Analysis Incorporating High Quality HRAM LCMS & MSMS with Intelligent Automated Data Mining Dave Weil, Ph.D. and Jim Lau, Ph.D. Typical Method Conditions: 1260 UHPLC
An Effective Workflow for Impurity Analysis Incorporating High Quality HRAM LCMS & MSMS with Intelligent Automated Data Mining Dave Weil, Ph.D. and Jim Lau, Ph.D. Typical Method Conditions: 1260 UHPLC
Choosing the metabolomics platform
 GBS 748 Choosing the metabolomics platform Stephen Barnes, PhD 4 7117; sbarnes@uab.edu So, I have my samples what s next? You ve collected your samples and you may have extracted them Protein precipitation
GBS 748 Choosing the metabolomics platform Stephen Barnes, PhD 4 7117; sbarnes@uab.edu So, I have my samples what s next? You ve collected your samples and you may have extracted them Protein precipitation
De Novo Metabolite Chemical Structure Determination. Paul R. West Ph.D. Stemina Biomarker Discovery, Inc.
 De Novo Metabolite Chemical Structure Determination Paul R. West Ph.D. Stemina Biomarker Discovery, Inc. As a part of non targeted metabolomic analysis, when peaks in the mass spectra are thought to be
De Novo Metabolite Chemical Structure Determination Paul R. West Ph.D. Stemina Biomarker Discovery, Inc. As a part of non targeted metabolomic analysis, when peaks in the mass spectra are thought to be
BUFOTENINE Latest Revision: August 16, 2005
 BUFOTENINE Latest Revision: August 16, 25 H N HO N CH 3 CH 3 1. SYNONYMS CFR: Bufotenine CAS #: Base: 487-93-4 Other Names: N,N-dimethylserotonin, 5-hydroxy-N,N-dimethyltryptamine, mappine 2. CHEMICAL
BUFOTENINE Latest Revision: August 16, 25 H N HO N CH 3 CH 3 1. SYNONYMS CFR: Bufotenine CAS #: Base: 487-93-4 Other Names: N,N-dimethylserotonin, 5-hydroxy-N,N-dimethyltryptamine, mappine 2. CHEMICAL
Supplementary Materials. Synthesis of Reusable Silica Nanosphere-Supported Pt(IV) Complex for. Formation of Disulfide Bonds in Peptides
 Supplementary Materials Synthesis of Reusable Silica Nanosphere-Supported Pt(IV) Complex for Formation of Disulfide Bonds in Peptides Xiaonan Hou, Xiaowei Zhao, Yamei Zhang, Aiying Han, Shuying Huo*, and
Supplementary Materials Synthesis of Reusable Silica Nanosphere-Supported Pt(IV) Complex for Formation of Disulfide Bonds in Peptides Xiaonan Hou, Xiaowei Zhao, Yamei Zhang, Aiying Han, Shuying Huo*, and
MS Interpretation I. Identification of the Molecular Ion
 MS Interpretation I Identification of the Molecular Ion Molecular Ion: EI Requirements for the Molecular Ion Must be the highest m/z peak in the spectrum Highest Isotope Cluster Must be an odd-electron
MS Interpretation I Identification of the Molecular Ion Molecular Ion: EI Requirements for the Molecular Ion Must be the highest m/z peak in the spectrum Highest Isotope Cluster Must be an odd-electron
The new Water Screening PCDL
 The new Water Screening PCDL Content and integration in suspect and non-target screening Dr. Thomas Glauner Senior LC/MS Applications Scientist EMEA Market Development Team 1 Accurate mass screening and
The new Water Screening PCDL Content and integration in suspect and non-target screening Dr. Thomas Glauner Senior LC/MS Applications Scientist EMEA Market Development Team 1 Accurate mass screening and
Collision Cross Section: Ideal elastic hard sphere collision:
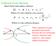 Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Mass Spectrometry. General Principles
 General Principles Mass Spectrometer: Converts molecules to ions Separates ions (usually positively charged) on the basis of their mass/charge (m/z) ratio Quantifies how many units of each ion are formed
General Principles Mass Spectrometer: Converts molecules to ions Separates ions (usually positively charged) on the basis of their mass/charge (m/z) ratio Quantifies how many units of each ion are formed
Degradation of Rapamycin and its Ring-Opened Isomer: Role of Base Catalysis Yuri V. Il ichev*, Lori Alquier, and Cynthia A.
 Degradation of Rapamycin and its Ring-Opened Isomer: Role of Base Catalysis Yuri V. Il ichev*, Lori Alquier, and Cynthia A. Maryanoff Supplementary Information Table S1. Results of fitting of the kinetic
Degradation of Rapamycin and its Ring-Opened Isomer: Role of Base Catalysis Yuri V. Il ichev*, Lori Alquier, and Cynthia A. Maryanoff Supplementary Information Table S1. Results of fitting of the kinetic
LC-MS compatible Separation of the Fungicide Spiroxamine
 Spiroxamine is a systemic fungicide, which was brought to the market by Bayer CropScience. The substance is a mixture of diastereomers A and B again consisting of 4 enantiomers A1, A2, B1 and B2 (fig.
Spiroxamine is a systemic fungicide, which was brought to the market by Bayer CropScience. The substance is a mixture of diastereomers A and B again consisting of 4 enantiomers A1, A2, B1 and B2 (fig.
MS Interpretation II. Fragmentation
 MS Interpretation II Fragmentation Ionization E Electron Ionization (EI): Even-electron neutrals yield odd-electron radical cations. M(EE) EI - 1e - M (E) Electron can come from anywhere. EI EI even electron
MS Interpretation II Fragmentation Ionization E Electron Ionization (EI): Even-electron neutrals yield odd-electron radical cations. M(EE) EI - 1e - M (E) Electron can come from anywhere. EI EI even electron
Multiple Fragmentation Methods for Small Molecule Characterization on a Dual Pressure Linear Ion Trap Orbitrap Hybrid Mass Spectrometer
 Application ote: 54 Multiple Fragmentation Methods for Small Molecule Characterization on a Dual Pressure Linear Ion Trap rbitrap Hybrid Mass Spectrometer Kate Comstock, Yingying Huang; Thermo Fisher Scientific,
Application ote: 54 Multiple Fragmentation Methods for Small Molecule Characterization on a Dual Pressure Linear Ion Trap rbitrap Hybrid Mass Spectrometer Kate Comstock, Yingying Huang; Thermo Fisher Scientific,
Chapter 5. Complexation of Tholins by 18-crown-6:
 5-1 Chapter 5. Complexation of Tholins by 18-crown-6: Identification of Primary Amines 5.1. Introduction Electrospray ionization (ESI) is an excellent technique for the ionization of complex mixtures,
5-1 Chapter 5. Complexation of Tholins by 18-crown-6: Identification of Primary Amines 5.1. Introduction Electrospray ionization (ESI) is an excellent technique for the ionization of complex mixtures,
MS-based proteomics to investigate proteins and their modifications
 MS-based proteomics to investigate proteins and their modifications Francis Impens VIB Proteomics Core October th 217 Overview Mass spectrometry-based proteomics: general workflow Identification of protein
MS-based proteomics to investigate proteins and their modifications Francis Impens VIB Proteomics Core October th 217 Overview Mass spectrometry-based proteomics: general workflow Identification of protein
MassHunter TOF/QTOF Users Meeting
 MassHunter TOF/QTOF Users Meeting 1 Qualitative Analysis Workflows Workflows in Qualitative Analysis allow the user to only see and work with the areas and dialog boxes they need for their specific tasks
MassHunter TOF/QTOF Users Meeting 1 Qualitative Analysis Workflows Workflows in Qualitative Analysis allow the user to only see and work with the areas and dialog boxes they need for their specific tasks
MASS SPECTRA measure a compound s Mol. Wt. This ionization type is called: electron impact MS
 MASS SPECTRA measure a compound s Mol. Wt. p. 213 M + Molecule e - Molecule + 2 e - + + Mole cule + + Mol ecule IONIZATION CHAMBER repellor plate accelerating plates variable field magnet + Mo + lecule
MASS SPECTRA measure a compound s Mol. Wt. p. 213 M + Molecule e - Molecule + 2 e - + + Mole cule + + Mol ecule IONIZATION CHAMBER repellor plate accelerating plates variable field magnet + Mo + lecule
Integrated approach to HPLC method development: Using all the tools in the chromatographer s toolbox
 Integrated approach to HPLC method development: Using all the tools in the chromatographer s toolbox Rosario LoBrutto Pharmaceutical and Analytical Development Novartis Pharmaceuticals Overall Strategy
Integrated approach to HPLC method development: Using all the tools in the chromatographer s toolbox Rosario LoBrutto Pharmaceutical and Analytical Development Novartis Pharmaceuticals Overall Strategy
Chromatographic Analysis
 Chromatographic Analysis Distribution of Analytes between Phases An analyte is in equilibrium between the two phases [S 1 ] [S 2 ] (in phase 1) (in phase 2) AS [S2 ] K 2 A S [S1 ] 1 AS, A 1 S Activity
Chromatographic Analysis Distribution of Analytes between Phases An analyte is in equilibrium between the two phases [S 1 ] [S 2 ] (in phase 1) (in phase 2) AS [S2 ] K 2 A S [S1 ] 1 AS, A 1 S Activity
Dissociation of Even-Electron Ions
 Dissociation of Even-Electron Ions Andrea Raffaelli CNR Istituto di Fisiologia Clinica Via Moruzzi, 1, 56124 Pisa. E-Mail: andrea.raffaelli@cnr.it Web: http://raffaelli.ifc.cnr.it A Simple? ESI Spectrum
Dissociation of Even-Electron Ions Andrea Raffaelli CNR Istituto di Fisiologia Clinica Via Moruzzi, 1, 56124 Pisa. E-Mail: andrea.raffaelli@cnr.it Web: http://raffaelli.ifc.cnr.it A Simple? ESI Spectrum
Application Note. Edgar Naegele. Abstract
 Agilent MassHunter Fast computer aided analysis of LC/ESI-TF data from complex natural product extracts Part 1: Analysis of Agilent 6210 TF data with the Molecular Feature Extractor in MassHunter Workstation
Agilent MassHunter Fast computer aided analysis of LC/ESI-TF data from complex natural product extracts Part 1: Analysis of Agilent 6210 TF data with the Molecular Feature Extractor in MassHunter Workstation
Varying nucleotide synthesis reaction conditions
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2016 Varying nucleotide synthesis reaction conditions In order to obtain maximum yields
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2016 Varying nucleotide synthesis reaction conditions In order to obtain maximum yields
Application Note. Edgar Naegele. Abstract
 Fast identification of main drug metabolites by quadrupole time-of-flight LC/MS Measuring accurate MS and MS/MS data with the Agilent 651 Q-TOF LC/MS and identification of main meta-bolites by comparison
Fast identification of main drug metabolites by quadrupole time-of-flight LC/MS Measuring accurate MS and MS/MS data with the Agilent 651 Q-TOF LC/MS and identification of main meta-bolites by comparison
Highly Specific near-infrared Fluorescent probe for the Real-Time Detection of β-glucuronidase in Various Living Cells and Animals
 Supplementary information Highly Specific near-infrared Fluorescent probe for the Real-Time Detection of β-glucuronidase in Various Living Cells and Animals Yinzhu Jin,, Xiangge Tian,, Lingling Jin, Yonglei
Supplementary information Highly Specific near-infrared Fluorescent probe for the Real-Time Detection of β-glucuronidase in Various Living Cells and Animals Yinzhu Jin,, Xiangge Tian,, Lingling Jin, Yonglei
Electrospray Ion Trap Mass Spectrometry. Introduction
 Electrospray Ion Source Electrospray Ion Trap Mass Spectrometry Introduction The key to using MS for solutions is the ability to transfer your analytes into the vacuum of the mass spectrometer as ionic
Electrospray Ion Source Electrospray Ion Trap Mass Spectrometry Introduction The key to using MS for solutions is the ability to transfer your analytes into the vacuum of the mass spectrometer as ionic
Halogenated Compounds from Directed Fermentation of Penicillium concentricum, an Endophytic Fungus of the Liverwort Trichocolea tomentella
 Supporting Information for Halogenated Compounds from Directed Fermentation of Penicillium concentricum, an Endophytic Fungus of the Liverwort Trichocolea tomentella Tehane Ali, Masanori Inagaki, Hee-byung
Supporting Information for Halogenated Compounds from Directed Fermentation of Penicillium concentricum, an Endophytic Fungus of the Liverwort Trichocolea tomentella Tehane Ali, Masanori Inagaki, Hee-byung
Mass Spectrometry. Hyphenated Techniques GC-MS LC-MS and MS-MS
 Mass Spectrometry Hyphenated Techniques GC-MS LC-MS and MS-MS Reasons for Using Chromatography with MS Mixture analysis by MS alone is difficult Fragmentation from ionization (EI or CI) Fragments from
Mass Spectrometry Hyphenated Techniques GC-MS LC-MS and MS-MS Reasons for Using Chromatography with MS Mixture analysis by MS alone is difficult Fragmentation from ionization (EI or CI) Fragments from
Strategies for the Analysis of Therapeutic Peptides in Biofluids by LC-MS/MS. Lee Goodwin
 Strategies for the Analysis of Therapeutic Peptides in Biofluids by LC-MS/MS Lee Goodwin Sample Preparation Chromatography Detection General Strategies Examples New Approaches Summary Outline ABUNDANCE
Strategies for the Analysis of Therapeutic Peptides in Biofluids by LC-MS/MS Lee Goodwin Sample Preparation Chromatography Detection General Strategies Examples New Approaches Summary Outline ABUNDANCE
Chemistry Instrumental Analysis Lecture 37. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 37 Most analytes separated by HPLC are thermally stable and non-volatile (liquids) (unlike in GC) so not ionized easily by EI or CI techniques. MS must be at
Chemistry 4631 Instrumental Analysis Lecture 37 Most analytes separated by HPLC are thermally stable and non-volatile (liquids) (unlike in GC) so not ionized easily by EI or CI techniques. MS must be at
Novel LC/MS-Compatible Stationary Phase with Polar Selectivity
 Novel LC/MS-Compatible Stationary Phase with Polar Selectivity Carmen T. Santasania and David S. Bell Supelco 595 N. Harrison Rd., Bellefonte, PA 16823 T403168 GJU Introduction Stationary phases based
Novel LC/MS-Compatible Stationary Phase with Polar Selectivity Carmen T. Santasania and David S. Bell Supelco 595 N. Harrison Rd., Bellefonte, PA 16823 T403168 GJU Introduction Stationary phases based
Tridip Sheet, Raja Banerjee*
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Supplementary information The C NN motif: an intrinsic lover of sulfate and phosphate ions
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Supplementary information The C NN motif: an intrinsic lover of sulfate and phosphate ions
Reversed Phase Solvents
 Part 1. General Chromatographic Theory Part 2. verview of HPLC Media Part 3. The Role of the Mobile Phase in Selectivity Part 4. Column Care and Use Reversed Phase Solvents 2 Solvents for RP Chromatography
Part 1. General Chromatographic Theory Part 2. verview of HPLC Media Part 3. The Role of the Mobile Phase in Selectivity Part 4. Column Care and Use Reversed Phase Solvents 2 Solvents for RP Chromatography
Unit 12: Acids & Bases. Aim: What are the definitions and properties of an acid and a base? Properties of an Acid. Taste Sour.
 Unit 12: Acids & Bases Aim: What are the definitions and properties of an acid and a base? Mar 23 12:08 PM Properties of an Acid 3. Are electrolytes. (Dissociate and conduct electricity when aq) 2. Turns
Unit 12: Acids & Bases Aim: What are the definitions and properties of an acid and a base? Mar 23 12:08 PM Properties of an Acid 3. Are electrolytes. (Dissociate and conduct electricity when aq) 2. Turns
DISSOCIATION OF GAS IONS IN AIR BEFORE MASS SPECTROMETERS USING ELECTRIC FIELDS FROM FIELD DEPENDENT MOBILITY SPECTROMETERS
 DISSOCIATION OF GAS IONS IN AIR BEFORE MASS SPECTROMETERS USING ELECTRIC FIELDS FROM FIELD DEPENDENT MOBILITY SPECTROMETERS X. An, J.A. Stone, and G.A. Eiceman * Department of Chemistry and Biochemistry
DISSOCIATION OF GAS IONS IN AIR BEFORE MASS SPECTROMETERS USING ELECTRIC FIELDS FROM FIELD DEPENDENT MOBILITY SPECTROMETERS X. An, J.A. Stone, and G.A. Eiceman * Department of Chemistry and Biochemistry
Structural Analysis by In-Depth Impurity Search Using MetID Solution and High Accuracy MS/MS
 C146-E118 Structural Analysis by In-Depth Impurity Search Using MetID Solution and High Accuracy MS/MS Technical Report vol.16 1. Introduction MetID Solution is a software application that was developed
C146-E118 Structural Analysis by In-Depth Impurity Search Using MetID Solution and High Accuracy MS/MS Technical Report vol.16 1. Introduction MetID Solution is a software application that was developed
Profiling of Diferulates (Plant Cell Wall Cross- Linkers) Using Ultrahigh-performance Liquid. Chromatography-Tandem Mass Spectrometry
 Supporting Information for: Profiling of Diferulates (Plant Cell Wall Cross- Linkers) Using Ultrahigh-performance Liquid Chromatography-Tandem Mass Spectrometry Ramin Vismeh a,b, Fachuang Lu c,d, Shishir
Supporting Information for: Profiling of Diferulates (Plant Cell Wall Cross- Linkers) Using Ultrahigh-performance Liquid Chromatography-Tandem Mass Spectrometry Ramin Vismeh a,b, Fachuang Lu c,d, Shishir
A New Model for Asymmetric Amplification in Amino Acid Catalysis - Supporting information
 A New Model for Asymmetric Amplification in Amino Acid Catalysis - Supporting information Martin Klussmann, Hiroshi Iwamura, Suju P. Mathew, David H. Wells, Urvish Pandya, Alan Armstrong and Donna G. Blackmond
A New Model for Asymmetric Amplification in Amino Acid Catalysis - Supporting information Martin Klussmann, Hiroshi Iwamura, Suju P. Mathew, David H. Wells, Urvish Pandya, Alan Armstrong and Donna G. Blackmond
Caspase-1 Specific Light-up Probe with Aggregation-Induced Emission. Characteristics for Inhibitor Screening of Coumarin-Originated Natural.
 Supporting Information Caspase-1 Specific Light-up Probe with Aggregation-Induced Emission Characteristics for Inhibitor Screening of Coumarin-Originated Natural Products Hao Lin, ^ Haitao Yang, ^ Shuai
Supporting Information Caspase-1 Specific Light-up Probe with Aggregation-Induced Emission Characteristics for Inhibitor Screening of Coumarin-Originated Natural Products Hao Lin, ^ Haitao Yang, ^ Shuai
First Prebiotic Generation of a Ribonucleotide from Adenine, D Ribose and Trimetaphosphate
 Supplementary information First Prebiotic Generation of a Ribonucleotide from Adenine, D Ribose and Trimetaphosphate Graziano Baccolini, a * Carla Boga, a and Gabriele Micheletti a a* Department of Organic
Supplementary information First Prebiotic Generation of a Ribonucleotide from Adenine, D Ribose and Trimetaphosphate Graziano Baccolini, a * Carla Boga, a and Gabriele Micheletti a a* Department of Organic
Bioanalytical Chem: 4590: LC-MSMS of analgesics LC-MS Experiment Liquid Chromatography Mass Spectrometry (LC/MS)
 Liquid Chromatography Mass Spectrometry (LC/MS) Prelab Questions: Questions to be answered before doing the experiment. The answers are due at the beginning of each experiment without exception (the questions
Liquid Chromatography Mass Spectrometry (LC/MS) Prelab Questions: Questions to be answered before doing the experiment. The answers are due at the beginning of each experiment without exception (the questions
TOMAHAQ Method Construction
 TOMAHAQ Method Construction Triggered by offset mass accurate-mass high-resolution accurate quantitation (TOMAHAQ) can be performed in the standard method editor of the instrument, without modifications
TOMAHAQ Method Construction Triggered by offset mass accurate-mass high-resolution accurate quantitation (TOMAHAQ) can be performed in the standard method editor of the instrument, without modifications
ACD/AutoChrom Assisted Method Development for Challenging Separations. Vera Leshchinskaya February 7, 2018
 ACD/AutoChrom Assisted Method Development for Challenging Separations Vera Leshchinskaya February 7, 2018 Introduction Resolution of isomeric species is one of the most challenging areas of separation
ACD/AutoChrom Assisted Method Development for Challenging Separations Vera Leshchinskaya February 7, 2018 Introduction Resolution of isomeric species is one of the most challenging areas of separation
ANALYTICAL REPORT 1 CUMYL-4CN-BINACA (C22H24N4O) 1-(4-cyanobutyl)-N-(1-methyl-1-phenylethyl)-1H-indazole-3-carboxamide
 Vodovodna 95 1000 Ljubljana SLOVENIJA T: +386 (0)1 428 44 93 E: nfl@policija.si www.policija.si Remark other NPS detected: none ANALYTICAL REPORT 1 CUMYL-4CN-BINACA (C22H24N4O) 1-(4-cyanobutyl)-N-(1-methyl-1-phenylethyl)-1H-indazole-3-carboxamide
Vodovodna 95 1000 Ljubljana SLOVENIJA T: +386 (0)1 428 44 93 E: nfl@policija.si www.policija.si Remark other NPS detected: none ANALYTICAL REPORT 1 CUMYL-4CN-BINACA (C22H24N4O) 1-(4-cyanobutyl)-N-(1-methyl-1-phenylethyl)-1H-indazole-3-carboxamide
Synthesis of High-Mannose 1-Thio Glycans and Their Conjugation to Protein
 Electronic Supplementary Material (ESI) for Organic. This journal is The Royal Society of Chemistry 2014 Supplementary Data for Synthesis of High-Mannose 1-Thio Glycans and Their Conjugation to Protein
Electronic Supplementary Material (ESI) for Organic. This journal is The Royal Society of Chemistry 2014 Supplementary Data for Synthesis of High-Mannose 1-Thio Glycans and Their Conjugation to Protein
Method Development for Chiral LC/MS/MS Analysis of Acidic Stereoisomeric Pharmaceutical Compounds with Polysaccharide-based Stationary Phases
 Method Development for Chiral LC/MS/MS Analysis of Acidic Stereoisomeric Pharmaceutical Compounds with Polysaccharide-based Stationary Phases Liming Peng, Swapna Jayapalan, and Tivadar Farkas Phenomenex,
Method Development for Chiral LC/MS/MS Analysis of Acidic Stereoisomeric Pharmaceutical Compounds with Polysaccharide-based Stationary Phases Liming Peng, Swapna Jayapalan, and Tivadar Farkas Phenomenex,
Overview. Introduction. NMR hyphenation modes HPLC-SPE-NMR. Advantages and limitations. Examples
 NMR hyphenation verview Introduction NMR hyphenation modes HPLC-SPE-NMR Advantages and limitations Examples Hyphenated techniques Two or more analytical techniques combined into one integrated and automated
NMR hyphenation verview Introduction NMR hyphenation modes HPLC-SPE-NMR Advantages and limitations Examples Hyphenated techniques Two or more analytical techniques combined into one integrated and automated
Making Sense of Differences in LCMS Data: Integrated Tools
 Making Sense of Differences in LCMS Data: Integrated Tools David A. Weil Agilent Technologies MassHunter Overview Page 1 March 2008 How Clean is our Water?... Page 2 Chemical Residue Analysis.... From
Making Sense of Differences in LCMS Data: Integrated Tools David A. Weil Agilent Technologies MassHunter Overview Page 1 March 2008 How Clean is our Water?... Page 2 Chemical Residue Analysis.... From
COMPARISON GUIDE. to C18 Reversed Phase HPLC Columns. Comparison Data on 60 Commonly Used C18 Phases. Stationary Phase Specifications
 COMPARISON GUIDE to C18 Reversed Phase HPLC Columns Comparison Data on 60 Commonly Used C18 Phases Stationary Phase Specifications Phases Compared According to Relative Hydrophobicity Phases Compared According
COMPARISON GUIDE to C18 Reversed Phase HPLC Columns Comparison Data on 60 Commonly Used C18 Phases Stationary Phase Specifications Phases Compared According to Relative Hydrophobicity Phases Compared According
Choosing Columns and Conditions for the Best Peak Shape
 Choosing Columns and Conditions for the Best Peak Shape What is Good Peak Shape and Why is it Important? Good peak shape can be defined as a symmetrical or gaussian peak and poor peak shape can include
Choosing Columns and Conditions for the Best Peak Shape What is Good Peak Shape and Why is it Important? Good peak shape can be defined as a symmetrical or gaussian peak and poor peak shape can include
What is Tandem Mass Spectrometry? (MS/MS)
 What is Tandem Mass Spectrometry? (MS/MS) Activate MS1 MS2 Gas Surface Photons Electrons http://www.iupac.org/goldbook/t06250.pdf Major difference between MS and MS/MS: two analysis steps Ionization Analysis
What is Tandem Mass Spectrometry? (MS/MS) Activate MS1 MS2 Gas Surface Photons Electrons http://www.iupac.org/goldbook/t06250.pdf Major difference between MS and MS/MS: two analysis steps Ionization Analysis
Tandem MS = MS / MS. ESI-MS give information on the mass of a molecule but none on the structure
 Tandem MS = MS / MS ESI-MS give information on the mass of a molecule but none on the structure In tandem MS (MSMS) (pseudo-)molecular ions are selected in MS1 and fragmented by collision with gas. collision
Tandem MS = MS / MS ESI-MS give information on the mass of a molecule but none on the structure In tandem MS (MSMS) (pseudo-)molecular ions are selected in MS1 and fragmented by collision with gas. collision
Supporting Information. Detection and Occurrence of Chlorinated By-products of Bisphenol A, Nonylphenol and
 1 2 3 Supporting Information Detection and Occurrence of Chlorinated By-products of Bisphenol A, Nonylphenol and Estrogens in Drinking Water of China: Comparison to the Parent Compounds 4 5 6 7 8 1 Laboratory
1 2 3 Supporting Information Detection and Occurrence of Chlorinated By-products of Bisphenol A, Nonylphenol and Estrogens in Drinking Water of China: Comparison to the Parent Compounds 4 5 6 7 8 1 Laboratory
Supporting Information
 Supporting Information Online Hydrophobic Interaction Chromatography-Mass Spectrometry for the Analysis of Intact Monoclonal Antibodies Bifan Chen a, Ziqing Lin b,c, Andrew J. Alpert d, Cexiong Fu e, Qunying
Supporting Information Online Hydrophobic Interaction Chromatography-Mass Spectrometry for the Analysis of Intact Monoclonal Antibodies Bifan Chen a, Ziqing Lin b,c, Andrew J. Alpert d, Cexiong Fu e, Qunying
Isotopic-Labeling and Mass Spectrometry-Based Quantitative Proteomics
 Isotopic-Labeling and Mass Spectrometry-Based Quantitative Proteomics Xiao-jun Li, Ph.D. Current address: Homestead Clinical Day 4 October 19, 2006 Protein Quantification LC-MS/MS Data XLink mzxml file
Isotopic-Labeling and Mass Spectrometry-Based Quantitative Proteomics Xiao-jun Li, Ph.D. Current address: Homestead Clinical Day 4 October 19, 2006 Protein Quantification LC-MS/MS Data XLink mzxml file
PesticideScreener. Innovation with Integrity. Comprehensive Pesticide Screening and Quantitation UHR-TOF MS
 PesticideScreener Comprehensive Pesticide Screening and Quantitation Innovation with Integrity UHR-TOF MS The Challenge of Comprehensive Pesticide Residue Analysis The use of pesticides to reduce crop
PesticideScreener Comprehensive Pesticide Screening and Quantitation Innovation with Integrity UHR-TOF MS The Challenge of Comprehensive Pesticide Residue Analysis The use of pesticides to reduce crop
Quantitative Screening of 46 Illicit Drugs in Urine using Exactive Ultrahigh Resolution and Accurate Mass system
 Quantitative Screening of 46 Illicit Drugs in Urine using Exactive Ultrahigh Resolution and Accurate Mass system Kevin Mchale Thermo Fisher Scientific, San Jose CA Presentation Overview Anabolic androgenic
Quantitative Screening of 46 Illicit Drugs in Urine using Exactive Ultrahigh Resolution and Accurate Mass system Kevin Mchale Thermo Fisher Scientific, San Jose CA Presentation Overview Anabolic androgenic
Method Development Kits
 Method Development Kits sales representative for ordering information or contact...77 Method Development Kits First column choice for Pharma Development USP L C Method Development Kits Maximize retention
Method Development Kits sales representative for ordering information or contact...77 Method Development Kits First column choice for Pharma Development USP L C Method Development Kits Maximize retention
Analysis of Stachydrine in Leonurus japonicus Using an Agilent ZORBAX RRHD HILIC Plus Column with LC/ELSD and LC/MS/MS
 Analysis of Stachydrine in Leonurus japonicus Using an Agilent ZORBAX RRHD HILIC Plus Column with LC/ELSD and LC/MS/MS Application Note Traditional Chinese Medicine Author Rongjie Fu Agilent Technologies
Analysis of Stachydrine in Leonurus japonicus Using an Agilent ZORBAX RRHD HILIC Plus Column with LC/ELSD and LC/MS/MS Application Note Traditional Chinese Medicine Author Rongjie Fu Agilent Technologies
SUPPORTING INFORMATION Antimicrobial activity of chlorinated amino acids and peptides
 SUPPORTING INFORMATION Antimicrobial activity of chlorinated amino acids and peptides Melanie S. A. Coker*, Wan-Ping Hu, Senti T Senthilmohan and Anthony J. Kettle Free Radical Research Group, University
SUPPORTING INFORMATION Antimicrobial activity of chlorinated amino acids and peptides Melanie S. A. Coker*, Wan-Ping Hu, Senti T Senthilmohan and Anthony J. Kettle Free Radical Research Group, University
Probing Hydrogen Bond Energies by Mass Spectrometry
 Probing Hydrogen Bond Energies by Mass Spectrometry Hai-Feng Su, Lan Xue,* Yun-Hua Li, Shui-Chao Lin, Yi-Mei Wen, Rong-Bin Huang, Su-Yuan Xie,* and Lan-Sun Zheng State Key Laboratory for Physical Chemistry
Probing Hydrogen Bond Energies by Mass Spectrometry Hai-Feng Su, Lan Xue,* Yun-Hua Li, Shui-Chao Lin, Yi-Mei Wen, Rong-Bin Huang, Su-Yuan Xie,* and Lan-Sun Zheng State Key Laboratory for Physical Chemistry
Shodex TM ODP2 HP series columns
 HPLC Columns Shodex TM ODP2 HP series columns Better retention of highly polar substances Technical notebook No. 6 Contents 1. Introduction 1-1. Specifications 1-2. Eluent Compatibility of ODP2 HP Series
HPLC Columns Shodex TM ODP2 HP series columns Better retention of highly polar substances Technical notebook No. 6 Contents 1. Introduction 1-1. Specifications 1-2. Eluent Compatibility of ODP2 HP Series
Mass Spectrometry. Electron Ionization and Chemical Ionization
 Mass Spectrometry Electron Ionization and Chemical Ionization Mass Spectrometer All Instruments Have: 1. Sample Inlet 2. Ion Source 3. Mass Analyzer 4. Detector 5. Data System http://www.asms.org Ionization
Mass Spectrometry Electron Ionization and Chemical Ionization Mass Spectrometer All Instruments Have: 1. Sample Inlet 2. Ion Source 3. Mass Analyzer 4. Detector 5. Data System http://www.asms.org Ionization
Inertsil ODS-EP Technical Information
 Technical Information Advantages of The selectivity is completely different from those of conventional columns such as ODS column due to its specific polar group in the stationary phase. Durability can
Technical Information Advantages of The selectivity is completely different from those of conventional columns such as ODS column due to its specific polar group in the stationary phase. Durability can
A fluorescent ph probe for acidic organelle in living cells
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Supporting Information for A fluorescent ph probe for acidic organelle in
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Supporting Information for A fluorescent ph probe for acidic organelle in
Investigation of retention mechanisms in HILIC chromatography: Important considerations for robust method development
 Investigation of retention mechanisms in HILIC chromatography: Important considerations for robust method development Anders Fridström Sigma- Aldrich GMBH Abstract Investigation of Retention Mechanisms
Investigation of retention mechanisms in HILIC chromatography: Important considerations for robust method development Anders Fridström Sigma- Aldrich GMBH Abstract Investigation of Retention Mechanisms
Accelerate Unknown Detection in Emerging Drug Testing Using Thermo Scientific Compound Discoverer and mzcloud
 Accelerate Unknown Detection in Emerging Drug Testing Using Thermo Scientific Compound Discoverer and mzcloud Ed Goucher ClinTox Marketing Manager Thermo Fisher Scientific v1 The world leader in serving
Accelerate Unknown Detection in Emerging Drug Testing Using Thermo Scientific Compound Discoverer and mzcloud Ed Goucher ClinTox Marketing Manager Thermo Fisher Scientific v1 The world leader in serving
Analysis - HPLC A.136. Primesep 5 µm columns B.136
 Primesep 5 µm columns Primesep columns feature double functionality of the bonding i.e : alkyl chain with anionic or cationic group, chelating group. This feature creates unique selectivities when using
Primesep 5 µm columns Primesep columns feature double functionality of the bonding i.e : alkyl chain with anionic or cationic group, chelating group. This feature creates unique selectivities when using
Dilution(*) Chromatography
 WA20264 Poster # 184, HPLC 2002, Montreal, 4-5 June 2002 At-Column Column-Dilution for Preparative Dilution(*) Chromatography Cecilia Mazza, Jie Cavanaugh, Ziling Lu,Tom Sirard,Tom Wheat and Uwe Neue Waters
WA20264 Poster # 184, HPLC 2002, Montreal, 4-5 June 2002 At-Column Column-Dilution for Preparative Dilution(*) Chromatography Cecilia Mazza, Jie Cavanaugh, Ziling Lu,Tom Sirard,Tom Wheat and Uwe Neue Waters
Breaking the speed limit: Fast, high-resolution peptide and tryptic digest separations using fused-core particles
 Breaking the speed limit: Fast, high-resolution peptide and tryptic digest separations using fused-core particles Stephanie Schuster 1, Barry Boyes 1,2, and Darryl Johnson 2 1 Advanced Materials Technology,
Breaking the speed limit: Fast, high-resolution peptide and tryptic digest separations using fused-core particles Stephanie Schuster 1, Barry Boyes 1,2, and Darryl Johnson 2 1 Advanced Materials Technology,
Xentrivalpeptides A-Q: Depsipeptide diversification in Xenorhabdus
 Supplementary Material for Xentrivalpeptides A-Q: Depsipeptide diversification in Xenorhabdus Qiuqin Zhou, Andrea Dowling, Heinrich Heide, Jens Wöhnert, Ulrich Brandt, James Baum, Richard ffrench-constant,
Supplementary Material for Xentrivalpeptides A-Q: Depsipeptide diversification in Xenorhabdus Qiuqin Zhou, Andrea Dowling, Heinrich Heide, Jens Wöhnert, Ulrich Brandt, James Baum, Richard ffrench-constant,
The Theory of HPLC. Ion Pair Chromatography
 The Theory of HPLC Ion Pair Chromatography i Wherever you see this symbol, it is important to access the on-line course as there is interactive material that cannot be fully shown in this reference manual.
The Theory of HPLC Ion Pair Chromatography i Wherever you see this symbol, it is important to access the on-line course as there is interactive material that cannot be fully shown in this reference manual.
Enantioselective copper catalysed C H insertion reactions of 2- sulfonyl-2-diazoacetamides to form γ-lactams. Supporting Information
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Enantioselective copper catalysed C H insertion reactions of 2- sulfonyl-2-diazoacetamides
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Enantioselective copper catalysed C H insertion reactions of 2- sulfonyl-2-diazoacetamides
sample was a solution that was evaporated in the spectrometer (such as with ESI-MS) ions such as H +, Na +, K +, or NH 4
 Introduction to Spectroscopy V: Mass Spectrometry Basic Theory: Unlike other forms of spectroscopy used in structure elucidation of organic molecules mass spectrometry does not involve absorption/emission
Introduction to Spectroscopy V: Mass Spectrometry Basic Theory: Unlike other forms of spectroscopy used in structure elucidation of organic molecules mass spectrometry does not involve absorption/emission
Analysis of Illegal Dyes in Food Matrices using Automated Online Sample Preparation with LC/MS
 Application Note: 56 Analysis of Illegal Dyes in Food Matrices using Automated Online Sample Preparation with LC/MS Yang Shi, Catherine Lafontaine, Matthew Berube, John Fink, François Espourteille Thermo
Application Note: 56 Analysis of Illegal Dyes in Food Matrices using Automated Online Sample Preparation with LC/MS Yang Shi, Catherine Lafontaine, Matthew Berube, John Fink, François Espourteille Thermo
Supplemental Information
 Supplemental Information Neutralizing positive charges at the surface of a protein lowers its rate of amide hydrogen exchange without altering its structure or increasing its thermostability. Bryan F.
Supplemental Information Neutralizing positive charges at the surface of a protein lowers its rate of amide hydrogen exchange without altering its structure or increasing its thermostability. Bryan F.
WADA Technical Document TD2015IDCR
 MINIMUM CRITERIA FOR CHROMATOGRAPHIC-MASS SPECTROMETRIC CONFIRMATION OF THE IDENTITY OF ANALYTES FOR DOPING CONTROL PURPOSES. The ability of a method to identify an analyte is a function of the entire
MINIMUM CRITERIA FOR CHROMATOGRAPHIC-MASS SPECTROMETRIC CONFIRMATION OF THE IDENTITY OF ANALYTES FOR DOPING CONTROL PURPOSES. The ability of a method to identify an analyte is a function of the entire
Acknowledgement: 4F-PBP (C14H18FNO) T: +386 (0) E: Vodovodna Ljubljana SLOVENIJA
 Vodovodna 95 1000 Ljubljana SLOVENIJA T: +386 (0)1 428 44 93 E: nfl@policija.si www.policija.si 4F-PBP (C14H18FNO) Acknowledgement: Sample was kindly provided by the Laboratório de Polícia Cientifica da
Vodovodna 95 1000 Ljubljana SLOVENIJA T: +386 (0)1 428 44 93 E: nfl@policija.si www.policija.si 4F-PBP (C14H18FNO) Acknowledgement: Sample was kindly provided by the Laboratório de Polícia Cientifica da
Supplementary information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supplementary information 1. Composition of the nanoparticles 2. Dynamic light scattering 3. Scanning
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supplementary information 1. Composition of the nanoparticles 2. Dynamic light scattering 3. Scanning
Supporting Information. A fluorogenic assay for screening Sirt6 modulators
 This journal is The Royal Society of Chemistry 213 Supporting Information A fluorogenic assay for screening Sirt6 modulators Jing Hu, Bin He, Shiva Bhargava, Hening Lin* Department of Chemistry and Chemical
This journal is The Royal Society of Chemistry 213 Supporting Information A fluorogenic assay for screening Sirt6 modulators Jing Hu, Bin He, Shiva Bhargava, Hening Lin* Department of Chemistry and Chemical
Ch. 22 Mass Spectrometry (MS)
 Ch. Mass Spectroetry (MS).1 MS easures ass of atos, olecules, or fragents of olecules -1. What is MS? Gaseous ato fro condensed phase ionized Accelerated & separated By ass to charge ratio /z M=00: 1 ion
Ch. Mass Spectroetry (MS).1 MS easures ass of atos, olecules, or fragents of olecules -1. What is MS? Gaseous ato fro condensed phase ionized Accelerated & separated By ass to charge ratio /z M=00: 1 ion
Hai-Bin Yang, Xing Fan, Yin Wei,* Min Shi*
 Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2015 Solvent-controlled Nucleophilic Trifloromethylthiolation of Morita- Baylis-Hillman
Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2015 Solvent-controlled Nucleophilic Trifloromethylthiolation of Morita- Baylis-Hillman
New Approaches in the Analysis of Amadori Compounds
 7 TH Wartburg Symposium on Flavor Chemistry and Biology Eisenach, April 21 st - April 23 rd, 2004 New Approaches in the Analysis of Amadori Compounds T. Davidek, I. Blank, K. Kraehenhuehl, J. Hau and S.
7 TH Wartburg Symposium on Flavor Chemistry and Biology Eisenach, April 21 st - April 23 rd, 2004 New Approaches in the Analysis of Amadori Compounds T. Davidek, I. Blank, K. Kraehenhuehl, J. Hau and S.
Interazioni di ioni con elettroni (ECD, ETD) e fotoni (Ion spectroscopy) Gianluca Giorgi. via Aldo Moro Siena
 Interazioni di ioni con elettroni (ECD, ETD) e fotoni (Ion spectroscopy) Gianluca Giorgi Università degli Studi di Siena Dipartimento di Biotecnologie, Chimica e Farmacia via Aldo Moro 53100 Siena e-mail:
Interazioni di ioni con elettroni (ECD, ETD) e fotoni (Ion spectroscopy) Gianluca Giorgi Università degli Studi di Siena Dipartimento di Biotecnologie, Chimica e Farmacia via Aldo Moro 53100 Siena e-mail:
Simplified Approaches to Impurity Identification using Accurate Mass UPLC/MS
 Simplified Approaches to Impurity Identification using Accurate Mass UPLC/MS Marian Twohig, Michael D. Jones, Dominic Moore, Peter Lee, and Robert Plumb Waters Corporation, Milford, MA, USA APPLICATION
Simplified Approaches to Impurity Identification using Accurate Mass UPLC/MS Marian Twohig, Michael D. Jones, Dominic Moore, Peter Lee, and Robert Plumb Waters Corporation, Milford, MA, USA APPLICATION
Carbon Carbon Bond Activation in Saturated Hydrocarbons by Field-Assisted Nitrogen Fixation Guangtao Li, Xin Li, Zheng Ouyang, and R.
 Carbon Carbon Bond Activation in Saturated Hydrocarbons by Field-Assisted Nitrogen Fixation Guangtao Li, Xin Li, Zheng Ouyang, and R. Graham Cooks* Department of Chemistry, Purdue University560 Oval Drive,
Carbon Carbon Bond Activation in Saturated Hydrocarbons by Field-Assisted Nitrogen Fixation Guangtao Li, Xin Li, Zheng Ouyang, and R. Graham Cooks* Department of Chemistry, Purdue University560 Oval Drive,
Multi-stage Mass Spectrometry up to MS 4 on a QTof System
 Valerie Koppen, 1 Russell Mortishire-Smith, 2 Filip Cuyckens 1 1 Janssen R&D, Beerse, Belgium 2 Waters Corporation, Wilmslow, United Kingdom APPLICATION BENEFITS This application note illustrates how multi-stage
Valerie Koppen, 1 Russell Mortishire-Smith, 2 Filip Cuyckens 1 1 Janssen R&D, Beerse, Belgium 2 Waters Corporation, Wilmslow, United Kingdom APPLICATION BENEFITS This application note illustrates how multi-stage
Quantitative analysis of mitragynine in human urine by high performance liquid chromatography-tandem mass spectrometry
 Quantitative analysis of mitragynine in human urine by high performance liquid chromatography-tandem mass spectrometry Shijun Lua, Buu N. Trana, Jamie L. Nelsenb, Kenneth M. Aldousa. Journal of Chromatography
Quantitative analysis of mitragynine in human urine by high performance liquid chromatography-tandem mass spectrometry Shijun Lua, Buu N. Trana, Jamie L. Nelsenb, Kenneth M. Aldousa. Journal of Chromatography
Certificate Of Analysis
 Certificate f Analysis Compound: -Hydroxy Propranolol Molecular Formula: C H N. (mw base =.) Structure: MW =. base H 8 0 H C H N -Hydroxy Propranolol "FR CHEMICAL USE NLY" Synonyms: (CA index name) -[(-Methylethyl)amino]--(-{-hydroxy}-naphthalenyloxy)--Propanol
Certificate f Analysis Compound: -Hydroxy Propranolol Molecular Formula: C H N. (mw base =.) Structure: MW =. base H 8 0 H C H N -Hydroxy Propranolol "FR CHEMICAL USE NLY" Synonyms: (CA index name) -[(-Methylethyl)amino]--(-{-hydroxy}-naphthalenyloxy)--Propanol
Agilent TOF Screening & Impurity Profiling Julie Cichelli, PhD LC/MS Small Molecule Workshop Dec 6, 2012
 1 Agilent TOF Screening & Impurity Profiling Julie Cichelli, PhD LC/MS Small Molecule Workshop Dec 6, 2012 Review: Technology for Accurate Mass Analysis: TOF LC/MS Mass measurements accurate to several
1 Agilent TOF Screening & Impurity Profiling Julie Cichelli, PhD LC/MS Small Molecule Workshop Dec 6, 2012 Review: Technology for Accurate Mass Analysis: TOF LC/MS Mass measurements accurate to several
Using TOF for Screening and Quantitation of Sudan Red Colorants in Food Application
 Using TOF for Screening and Quantitation of Sudan Red Colorants in Food Application Food Authors Yanyan Fang Agilent Technologies, Inc. 412 Ying Lun Road Pu Dong, Shanghai 200131 China Michael Zumwalt
Using TOF for Screening and Quantitation of Sudan Red Colorants in Food Application Food Authors Yanyan Fang Agilent Technologies, Inc. 412 Ying Lun Road Pu Dong, Shanghai 200131 China Michael Zumwalt
A Platform to Identify Endogenous Metabolites Using a Novel High Performance Orbitrap MS and the mzcloud Library
 A Platform to Identify Endogenous Metabolites Using a Novel High Performance Orbitrap MS and the mzcloud Library Junhua Wang, 1 David A. Peake, 1 Robert Mistrik, 2 Yingying Huang 1 1 Thermo Fisher Scientific
A Platform to Identify Endogenous Metabolites Using a Novel High Performance Orbitrap MS and the mzcloud Library Junhua Wang, 1 David A. Peake, 1 Robert Mistrik, 2 Yingying Huang 1 1 Thermo Fisher Scientific
William E. Barber, Ph.D. Applications Chemist October
 William E. Barber, Ph.D. Applications Chemist ctober 0 00 Secrets of Good Peak Shape in HPLC Time: :00 p.m. CET Telephone Number: + 44 0 76 088 Chairperson: John Vis The Secrets of Good Peak Shape in HPLC
William E. Barber, Ph.D. Applications Chemist ctober 0 00 Secrets of Good Peak Shape in HPLC Time: :00 p.m. CET Telephone Number: + 44 0 76 088 Chairperson: John Vis The Secrets of Good Peak Shape in HPLC
CHAPTER Identification of side products in the synthesis of MMBC. As shown in the previous chapter, MMBC can be produced with high
 113 CHAPTER 6 IDENTIFICATION OF SIDE PRODUCTS IN THE PRODUCTION OF METHYL 4- (METHOXYMETHYL) BENZENE CARBOXYLATE (MMBC) FROM METHYL 5- (METHOXYMETHYL)-FURAN-2-CARBOXYLATE (MMFC) AND ETHYLENE 6.1 Identification
113 CHAPTER 6 IDENTIFICATION OF SIDE PRODUCTS IN THE PRODUCTION OF METHYL 4- (METHOXYMETHYL) BENZENE CARBOXYLATE (MMBC) FROM METHYL 5- (METHOXYMETHYL)-FURAN-2-CARBOXYLATE (MMFC) AND ETHYLENE 6.1 Identification
Ligations of O-acyl threonine units to give native peptides via 5-, 8-, 9- and 10-membered cyclic transition states
 Ligations of O-acyl threonine units to give native peptides via 5-, 8-, 9- and 10-membered cyclic transition states Siva S. Panda,* a Sumaira Liaqat, a,b Anand D. Tiwari, a Hadi M. Marwani, c Hassan M.
Ligations of O-acyl threonine units to give native peptides via 5-, 8-, 9- and 10-membered cyclic transition states Siva S. Panda,* a Sumaira Liaqat, a,b Anand D. Tiwari, a Hadi M. Marwani, c Hassan M.
- Supporting Information -
 Protomers of Benzocaine: Solvent and Permittivity Dependence Stephan Warnke, Jongcheol Seo, Jasper Boschmans, Frank Sobott, James H. Scrivens, Christian Bleiholder,,? Michael T. Bowers, Sandy Gewinner,
Protomers of Benzocaine: Solvent and Permittivity Dependence Stephan Warnke, Jongcheol Seo, Jasper Boschmans, Frank Sobott, James H. Scrivens, Christian Bleiholder,,? Michael T. Bowers, Sandy Gewinner,
