Xentrivalpeptides A-Q: Depsipeptide diversification in Xenorhabdus
|
|
|
- Egbert Clement Armstrong
- 5 years ago
- Views:
Transcription
1 Supplementary Material for Xentrivalpeptides A-Q: Depsipeptide diversification in Xenorhabdus Qiuqin Zhou, Andrea Dowling, Heinrich Heide, Jens Wöhnert, Ulrich Brandt, James Baum, Richard ffrench-constant, and Helge B. Bode, * Institut für Molekulare Biowissenschaften, Goethe Universität Frankfurt, Frankfurt am Main, Germany Biosciences, University of Exeter in Cornwall, Tremough Campus, Penryn, Cornwall TR10 9EZ, UK Zentrum für Biologische Chemie, Molekulare Bioenergetik, Klinikum der Goethe Universität Frankfurt, Frankfurt am Main, Germany Monsanto Company, Chesterfield, M 63017, USA 1
2 Table S1. Results from HRESIMS (exp.) and the resulting molecular formula with the tolerances in ppm. a) protonated xentrivalpeptides and the two main MS 2 fragments. b) MS 3 fragments using fragment B as precursor. Fragment A was fragmented to fragment B by losing a water molecule. Fragmentation of 17 was shown in Figure S8. a [M + H] + fragment A fragment B Nr. molecular formula exp. theo. ppm molecular formula m/z exp. ppm molecular formula m/z exp. ppm 1 C 46 H 66 9 N C 33 H 51 7 N C 33 H 49 6 N C 40 H 62 9 N C 33 H 51 7 N C 33 H 49 6 N C 41 H 64 9 N C 33 H 51 7 N C 33 H 49 6 N C 42 H 66 9 N C 33 H 51 7 N C 33 H 49 6 N C 43 H 68 9 N C 33 H 51 7 N C 33 H 49 6 N C 45 H 72 9 N C 33 H 51 7 N C 33 H 49 6 N C 44 H 70 9 N C 33 H 51 7 N C 33 H 49 6 N C 45 H 64 9 N C 33 H 51 7 N C 33 H 49 6 N C 47 H 68 9 N C 33 H 51 7 N C 33 H 49 6 N C 39 H 68 9 N C 30 H 53 7 N C 30 H 51 6 N C 40 H 70 9 N C 30 H 53 7 N C 30 H 51 6 N C 43 H 68 9 N C 30 H 53 7 N C 30 H 51 6 N C 42 H 74 9 N C 30 H 53 7 N C 30 H 51 6 N C 47 H 68 9 N C 34 H 53 7 N C 34 H 51 6 N C 45 H 64 9 N C 32 H 49 7 N C 32 H 47 6 N C 41 H 64 9 N C 33 H 51 7 N C 32 H 47 6 N C 41 H 57 8 N fragment A in figure S7 fragment B in figure S7 2
3 b fragment C fragment D fragment E fragment F Nr. precursor chemical chemical chemical chemical m/z exp. ppm m/z exp. ppm m/z exp. ppm formula formula formula formula m/z exp. ppm C 22 H 33 3 N C 23 H 31 4 N C 23 H 33 4 N C 20 H 35 4 N C 22 H 33 3 N C 23 H 31 4 N C 23 H 33 4 N C 20 H 35 4 N C 22 H 33 3 N C 23 H 31 4 N C 23 H 33 4 N C 20 H 35 4 N C 22 H 33 3 N C 23 H 31 4 N C 23 H 33 4 N C 20 H 35 4 N C 22 H 33 3 N C 23 H 31 4 N C 23 H 33 4 N C 20 H 35 4 N C 22 H 33 3 N C 23 H 31 4 N C 23 H 33 4 N C 20 H 35 4 N C 22 H 33 3 N C 23 H 31 4 N C 23 H 33 4 N C 20 H 35 4 N C 22 H 33 3 N C 23 H 31 4 N C 23 H 33 4 N C 20 H 35 4 N C 22 H 33 3 N C 23 H 31 4 N C 23 H 33 4 N C 20 H 35 4 N * * * C 20 H 33 4 N C 20 H 35 4 N C 20 H 35 4 N * * * C 20 H 33 4 N C 20 H 35 4 N C 20 H 35 4 N * * * C 20 H 33 4 N C 20 H 35 4 N C 20 H 35 4 N C 23 H 35 3 N C 23 H 31 4 N C 24 H 35 4 N C 21 H 37 4 N C 21 H 31 3 N C 23 H 31 4 N C 22 H 31 4 N C 19 H 33 4 N * not detected; MS 3 fragments of 13 and 16 were too weak. 3
4 Table S2. Results from the advanced Marfey s method: retention times (t R, minute) of the derivatized amino acids from the hydrolyzed 1 detected at negative ion mode by HPLC-MS. Commercial L-amino acids were used as standard. nly phenylalanine in 1 has D-configuration. m/z [M-H] - t R, L-FDLA a t R, LD-FDLA a Pro(4) Thr(2) Val(1)/(5)/(6)/(7) Phe(3) a : Marfey s reagents (N α -(5-fluoro-2,4-dinitrophenyl)-L-leucinamide, abbr. L-FDLA; N α -(5-fluoro-2,4- dinitrophenyl)-d-leucinamide, abbr. D-FDLA ); mixture of L-FDLA and D-FDLA, abbr. LD-FDLA. 4
5 Table S3. NMR Spectroscopic Data (500 MHz ( 1 H), 125 MHz ( 13 C) in CDCl 3 ) of 4, δ in ppm. Amino acid numbering from N- to C-terminus. subunit position δ C δ H, mult (J in Hz) Butyryl a , m 2b , m , m a 1.03, t (7.4) L-Val(1) C= α , m β , m γ 19.6 a 0.97, d (6.6) δ , d (6.4) NH 6.39, d (5.8) L-Thr(2) C= α , d (9.9) β , m γ , d (6.5) NH d (10.0) D-Phe(3) C= α , m β , m γ δ , m ε , m ζ , m NH 7.36, m L-Pro (4) C= α , m β , m β , m γ , m γ , m δ , m δ , m L-Val(5) C= α , m β , m γ 19.6 a 0.87, d, (7.0) δ , d, (6.9) NH 6.19, d, (10.1) L-Val(6) C= α , m β , m γ , d (6.8) δ 19.6 a 0.98, d (6.8) NH 8.04, d (6.9) L-Val(7) C= α , t (7.5) β , m γ , d (6.9) δ , d (6.7) NH 8.80, bs d: doublet; m: multiplet; a : same chemical shift 5
6 Figure S1. a) HRESIMS of 1. b) MS 2 -spectra of 1 after collision induced dissociation (CID) of [M+H] +. c) MS 3 -spectra of fragment B in selected rang. d) Schema for fragmentation path of 1 and the proposed fragment structures. The fragments A-F (Table S1) were selected for the structure elucidation. 6
7 Figure S2. Selected results of feeding experiments from 1-6, 8, and 9 which have the same ring structure. Proline and threonine were incorporated in all xentrivalpeptides (data not shown). Structures of R 1 and R 2 were shown in figure 4. Feeding experiments in nonlabled medium (LB medium, 12 C): incorporation of one L[2,3,3,5,5,6,6,7-2 H 8 ]phenylalanine ( 12 C H 8 -Phe), incorporation of two L[2,3,3,5,5,6,6,7-2 H]phenylalanine ( 12 C H 8 -Phe), incorporation of one DL[2,3,4,4,4,5,5,5-2 H 8 ]valine ( 12 C H 8 -Val), incorporation of two DL[2,3,4,4,4,5,5,5-2 H 8 ]valine ( 12 C H 8 -Val), incorporation of one L[2,3,3,4,5,5,5,5,5,5-2 H 10 ]leucine ( 12 C + 1 Leu), incorporation of one p-phenylacetic acid ( 12 C + 1 p-f- PAA), incorporation of one [U- 2 H 6 ]propionic acid ( 12 C H 6 -propionic acid). Feeding experiments in labled medium (ISGR - 13 C growth medium, 13 C): incorporation of one L-α-amino acid ( 13 C + 1 amino 7
8 acid), incorporation of two L-α-amino acids ( 13 C + 2 amino acids), incorporation of one butyric acid ( 13 C + 1 butyric acid). CID: collision induced dissociation. All xentrivalpeptides were fragmented to fragment A and B (Figure S1). Figure S3. Selected results of feeding experiments for 10-13, which have the same ring structure fragment B. Abbreviations see Figure S2. 8
9 Figure S4. Selected results of feeding experiments for 14. Abbreviations see Figure S2. 9
10 Figure S5. Selected results of feeding experiments for 15. Abbreviations see Figure S2. Fragment B of 15 and 16 indicated the same ring structure (Table S1). But, labeling of 16 was too weak for its characterization. 10
11 a N HN HN Pro(4) Thr(2) Val(1)/(5)/(6)/(7) NH Phe(3) Butyryl b H N N H H N H N N H N NH Figure S6. a) Subunits and selected CSY correlations (bond lines) for 4. b) Selected HMBC correlations (arrows from 1 H to 13 C) between subunits of 4. Amino acid numbering from N- to C-terminus. 11
12 a fragment A [M + H] + - Val fragment B fragment H fragment I fragment G fragment K fragment J m/z b c fragment formula m/z exp. ppm [M + H] + + -Val C 26 H 48 7 N fragment A C 28 H 42 6 N fragment B + C 28 H 40 5 N fragment G + C 26 H 32 5 N fragment H + C 24 H 37 5 N fragment I + C 26 H 30 4 N fragment J C 15 H 28 N fragment K C 17 H 21 N Figure S7. Structure elucidation of 17. a) HR-ESI MS 2 fragments. b) Proposed fragment structures. c) Molecular formula of the fragments. 12
13 Figure S8. Galleria mellonella hemocyte monolayers were incubated for 4h with (A) 2% DMS in Graces Insect Medium as a control, and (B) 100 µg/ml of 1. White arrows indicate regions of actin cytoskeletal ruffling caused by the compound. Green = FITC-phalloidin labeled f-actin, Red = polarized mitochondria, Blue = nuclei. 13
14 14
15 15
16 16
17 17
18 18
19 19
20 20
21 21
22 22
23 23
Supporting Information
 Supporting Information Figure S1.1 Positive HR-ESI-MS spectrum of compound 1 Figure S1.2 IR spectrum of compound 1 Figure S1.3 1 H NMR (600 MHz, D 2 O) spectrum of compound 1 Figure S1.4 1 H NMR (600 MHz,
Supporting Information Figure S1.1 Positive HR-ESI-MS spectrum of compound 1 Figure S1.2 IR spectrum of compound 1 Figure S1.3 1 H NMR (600 MHz, D 2 O) spectrum of compound 1 Figure S1.4 1 H NMR (600 MHz,
A Plausible Model Correlates Prebiotic Peptide Synthesis with. Primordial Genetic Code
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
Studies Leading to the Development of a Highly Selective. Colorimetric and Fluorescent Chemosensor for Lysine
 Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
A New Model for Asymmetric Amplification in Amino Acid Catalysis - Supporting information
 A New Model for Asymmetric Amplification in Amino Acid Catalysis - Supporting information Martin Klussmann, Hiroshi Iwamura, Suju P. Mathew, David H. Wells, Urvish Pandya, Alan Armstrong and Donna G. Blackmond
A New Model for Asymmetric Amplification in Amino Acid Catalysis - Supporting information Martin Klussmann, Hiroshi Iwamura, Suju P. Mathew, David H. Wells, Urvish Pandya, Alan Armstrong and Donna G. Blackmond
Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4.
 Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4. Field Desorption 5. MS MS techniques Matrix assisted
Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4. Field Desorption 5. MS MS techniques Matrix assisted
Proteins: Characteristics and Properties of Amino Acids
 SBI4U:Biochemistry Macromolecules Eachaminoacidhasatleastoneamineandoneacidfunctionalgroupasthe nameimplies.thedifferentpropertiesresultfromvariationsinthestructuresof differentrgroups.thergroupisoftenreferredtoastheaminoacidsidechain.
SBI4U:Biochemistry Macromolecules Eachaminoacidhasatleastoneamineandoneacidfunctionalgroupasthe nameimplies.thedifferentpropertiesresultfromvariationsinthestructuresof differentrgroups.thergroupisoftenreferredtoastheaminoacidsidechain.
Certificate Of Analysis
 Certificate f Analysis Compound: -Hydroxy Propranolol Molecular Formula: C H N. (mw base =.) Structure: MW =. base H 8 0 H C H N -Hydroxy Propranolol "FR CHEMICAL USE NLY" Synonyms: (CA index name) -[(-Methylethyl)amino]--(-{-hydroxy}-naphthalenyloxy)--Propanol
Certificate f Analysis Compound: -Hydroxy Propranolol Molecular Formula: C H N. (mw base =.) Structure: MW =. base H 8 0 H C H N -Hydroxy Propranolol "FR CHEMICAL USE NLY" Synonyms: (CA index name) -[(-Methylethyl)amino]--(-{-hydroxy}-naphthalenyloxy)--Propanol
Collision Cross Section: Ideal elastic hard sphere collision:
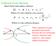 Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
(b) How many hydrogen atoms are in the molecular formula of compound A? [Consider the 1 H NMR]
![(b) How many hydrogen atoms are in the molecular formula of compound A? [Consider the 1 H NMR] (b) How many hydrogen atoms are in the molecular formula of compound A? [Consider the 1 H NMR]](/thumbs/89/97638524.jpg) CHEM 6371/4511 Name: The exam consists of interpretation of spectral data for compounds A-C. The analysis of each structure is worth 33.33 points. Compound A (a) How many carbon atoms are in the molecular
CHEM 6371/4511 Name: The exam consists of interpretation of spectral data for compounds A-C. The analysis of each structure is worth 33.33 points. Compound A (a) How many carbon atoms are in the molecular
Organic Chemistry II (CHE ) Examination I February 11, Name (Print legibly): Key. Student ID#:
 rganic hemistry II (HE 232-001) Examination I February 11, 2009 Name (Print legibly): Key (last) (first) Student ID#: PLEASE observe the following: You are allowed to have scratch paper (provided by me),
rganic hemistry II (HE 232-001) Examination I February 11, 2009 Name (Print legibly): Key (last) (first) Student ID#: PLEASE observe the following: You are allowed to have scratch paper (provided by me),
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature14137 1. Supplementary Methods 2. Supplementary Text 2.1 Physico-Chemical Properties and Structural Elucidation of Compound 1. 2.2 Physico-Chemical Properties and Structural Elucidation
doi:10.1038/nature14137 1. Supplementary Methods 2. Supplementary Text 2.1 Physico-Chemical Properties and Structural Elucidation of Compound 1. 2.2 Physico-Chemical Properties and Structural Elucidation
THE JOURNAL OF ANTIBIOTICS MAY Total Structures of Colistin Minor Components YOSHITOMO IKAI, HISAO OKA, JUNKO HAYAKAWA, NORIHISA KAWAMURA,
 THE JOURNAL OF ANTIBIOTICS MAY 1998 Total Structures of Colistin Minor Components YOSHITOMO IKAI, HISAO OKA, JUNKO HAYAKAWA, NORIHISA KAWAMURA, Aichi Prefectural Institute of Public Health, Tsuji-machi,
THE JOURNAL OF ANTIBIOTICS MAY 1998 Total Structures of Colistin Minor Components YOSHITOMO IKAI, HISAO OKA, JUNKO HAYAKAWA, NORIHISA KAWAMURA, Aichi Prefectural Institute of Public Health, Tsuji-machi,
HSQC spectra for three proteins
 HSQC spectra for three proteins SH3 domain from Abp1p Kinase domain from EphB2 apo Calmodulin What do the spectra tell you about the three proteins? HSQC spectra for three proteins Small protein Big protein
HSQC spectra for three proteins SH3 domain from Abp1p Kinase domain from EphB2 apo Calmodulin What do the spectra tell you about the three proteins? HSQC spectra for three proteins Small protein Big protein
BENG 183 Trey Ideker. Protein Sequencing
 BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
Supplementary Information. Synthesis and biological activity of a CXCR4-targeting bis(cyclam) lipid
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supplementary Information Synthesis and biological activity of a CXCR4-targeting
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supplementary Information Synthesis and biological activity of a CXCR4-targeting
Diastereomeric resolution directed towards chirality. determination focussing on gas-phase energetics of coordinated. sodium dissociation
 Supplementary Information Diastereomeric resolution directed towards chirality determination focussing on gas-phase energetics of coordinated sodium dissociation Authors: samu Kanie*, Yuki Shioiri, Koji
Supplementary Information Diastereomeric resolution directed towards chirality determination focussing on gas-phase energetics of coordinated sodium dissociation Authors: samu Kanie*, Yuki Shioiri, Koji
The resonance frequency of the H b protons is dependent upon the orientation of the H a protons with respect to the external magnetic field:
 Spin-Spin Splitting in Alkanes The signal arising from a proton or set of protons is split into (N+1) lines by the presence of N adjacent nuclei Example 1: Bromoethane The resonance frequency of the H
Spin-Spin Splitting in Alkanes The signal arising from a proton or set of protons is split into (N+1) lines by the presence of N adjacent nuclei Example 1: Bromoethane The resonance frequency of the H
Synthesis and Evaluation of Antibacterial Activity of Bottromycins
 Supporting Information Synthesis and Evaluation of Antibacterial Activity of Bottromycins Takeshi Yamada,,, Miu Yagita, Yutaka Kobayashi, Goh Sennari, Hiroyuki Shimamura, Hidehito Matsui, Yuki Horimatsu,
Supporting Information Synthesis and Evaluation of Antibacterial Activity of Bottromycins Takeshi Yamada,,, Miu Yagita, Yutaka Kobayashi, Goh Sennari, Hiroyuki Shimamura, Hidehito Matsui, Yuki Horimatsu,
Supplementary Material
 10.1071/CH17506_AC CSIRO 2018 Australian Journal of Chemistry 2018, 71(5), 341-347 Supplementary Material Magnesium alkoxide complexes of (benzimidazolylmethyl)amino ligands: Synthesis and applications
10.1071/CH17506_AC CSIRO 2018 Australian Journal of Chemistry 2018, 71(5), 341-347 Supplementary Material Magnesium alkoxide complexes of (benzimidazolylmethyl)amino ligands: Synthesis and applications
Lecture 15: Realities of Genome Assembly Protein Sequencing
 Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
CM Chemical Spectroscopy and Applications. Final Examination Solution Manual AY2013/2014
 NANYANG TECHNOLOGICAL UNIVERSITY DIVISION OF CHEMISTRY AND BIOLOGICAL CHEMISTRY SCHOOL OF PHYSICAL & MATHEMATICAL SCIENCES CM 3011 - Chemical Spectroscopy and Applications Final Examination Solution Manual
NANYANG TECHNOLOGICAL UNIVERSITY DIVISION OF CHEMISTRY AND BIOLOGICAL CHEMISTRY SCHOOL OF PHYSICAL & MATHEMATICAL SCIENCES CM 3011 - Chemical Spectroscopy and Applications Final Examination Solution Manual
SUPPLEMENTARY INFORMATION
 SUPPLEMETARY IFRMATI DI: 10.1038/CHEM.1108 A Route to Enantiopure RA Precursors from early Racemic Starting Materials Jason E. Hein, Eric Tse, and Donna G. Blackmond Department of Chemistry The Scripps
SUPPLEMETARY IFRMATI DI: 10.1038/CHEM.1108 A Route to Enantiopure RA Precursors from early Racemic Starting Materials Jason E. Hein, Eric Tse, and Donna G. Blackmond Department of Chemistry The Scripps
CHM 292 Final Exam Answer Key
 CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
Room Temperature N-Arylation of Amino Acids and Peptides Using Copper(I) and β-diketone
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2015 Room Temperature -Arylation of Amino Acids and Peptides Using Copper(I)
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2015 Room Temperature -Arylation of Amino Acids and Peptides Using Copper(I)
*Assignments could be reversed. *
 Name Key 5 W-Exam No. Page I. (6 points) Identify the indicated pairs of hydrogens in each of the following compounds as (i) homotopic, (ii) enantiotopic, or (iii) diastereotopic s. Write the answers as
Name Key 5 W-Exam No. Page I. (6 points) Identify the indicated pairs of hydrogens in each of the following compounds as (i) homotopic, (ii) enantiotopic, or (iii) diastereotopic s. Write the answers as
Chemistry Chapter 22
 hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
Chem 2320 Exam 1. January 30, (Please print)
 Chem 2320 Exam 1 January 30, 2006 Name: (first) (last) (Please print) Last 4 digits of I.D. I. Multiple Choice ( /20) Score /60 II /15 III /25 Total score /100 I. Multiple choice questions. (3 points each).
Chem 2320 Exam 1 January 30, 2006 Name: (first) (last) (Please print) Last 4 digits of I.D. I. Multiple Choice ( /20) Score /60 II /15 III /25 Total score /100 I. Multiple choice questions. (3 points each).
Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry
 Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry Application Note Biologics and Biosimilars Author Edgar Naegele Agilent Technologies, Inc.
Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry Application Note Biologics and Biosimilars Author Edgar Naegele Agilent Technologies, Inc.
Corygaline A, Hexahydrobenzophenanthridine Alkaloid with. Unusual Carbon Skeleton from Corydalis bungeana Turcz.
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information for Corygaline A, Hexahydrobenzophenanthridine Alkaloid
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Supporting Information for Corygaline A, Hexahydrobenzophenanthridine Alkaloid
Rubiaceae-type cyclopeptides from Galianthe thalictroides. Patrícia de Oliveira Figueiredo, * Renata Trentin Perdomo, Wilson Hino Kato Junior, Marcos
 Rubiaceae-type cyclopeptides from Galianthe thalictroides Patrícia de liveira Figueiredo, * Renata Trentin Perdomo, Wilson Hino Kato Junior, Marcos Vinícius Gonçalves de liveira Barros, Maria de Fatima
Rubiaceae-type cyclopeptides from Galianthe thalictroides Patrícia de liveira Figueiredo, * Renata Trentin Perdomo, Wilson Hino Kato Junior, Marcos Vinícius Gonçalves de liveira Barros, Maria de Fatima
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
SUPPLEMENTARY MATERIAL
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 SUPPLEMENTARY MATERIAL Two rare antioxidative prenylated terpenoids from loop-root Asiatic mangrove
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 SUPPLEMENTARY MATERIAL Two rare antioxidative prenylated terpenoids from loop-root Asiatic mangrove
The Depsipeptide Methodology for Solid Phase Peptide Synthesis: Circumventing Side Reactions and Development of an Automated Technique via
 Supporting Information The Depsipeptide Methodology for Solid Phase Peptide Synthesis: Circumventing Side Reactions and Development of an Automated Technique via Depsidipeptide Units. Irene Coin, Rudolf
Supporting Information The Depsipeptide Methodology for Solid Phase Peptide Synthesis: Circumventing Side Reactions and Development of an Automated Technique via Depsidipeptide Units. Irene Coin, Rudolf
Molecular Selective Binding of Basic Amino Acids by a Water-soluble Pillar[5]arene
![Molecular Selective Binding of Basic Amino Acids by a Water-soluble Pillar[5]arene Molecular Selective Binding of Basic Amino Acids by a Water-soluble Pillar[5]arene](/thumbs/77/75727507.jpg) Electronic supplementary information Molecular Selective Binding of Basic Amino Acids y a Water-solule Pillar[5]arene Chunju Li,* a, Junwei Ma, a Liu Zhao, a Yanyan Zhang, c Yihua Yu, c Xiaoyan Shu, a
Electronic supplementary information Molecular Selective Binding of Basic Amino Acids y a Water-solule Pillar[5]arene Chunju Li,* a, Junwei Ma, a Liu Zhao, a Yanyan Zhang, c Yihua Yu, c Xiaoyan Shu, a
Your Name: Question 1. 2D-NMR: C 6 H 10 O 2. (20 points)
 Question 1. 2D-NMR: C 6 H 10 O 2. (20 points) Integrations show signals 3H 1 & 5, 2H for signal 4, and 1H each for signals 2 and 3. - Draw the structure. - Assign the hydrogens to signals 1 5 (that is,
Question 1. 2D-NMR: C 6 H 10 O 2. (20 points) Integrations show signals 3H 1 & 5, 2H for signal 4, and 1H each for signals 2 and 3. - Draw the structure. - Assign the hydrogens to signals 1 5 (that is,
Ibuprofen. Example of benchtop NMR on small organic molecules
 R Ibuprofen Example of benchtop NMR on small organic molecules Ibuprofen (C 13 H 18 O 2 ) is a non-steroidal antiinflammatory drug (NSAID) and is commonly used for pain relief, fever reduction and against
R Ibuprofen Example of benchtop NMR on small organic molecules Ibuprofen (C 13 H 18 O 2 ) is a non-steroidal antiinflammatory drug (NSAID) and is commonly used for pain relief, fever reduction and against
Supporting Information
 Supporting Information Wiley-VCH 2006 69451 Weinheim, Germany A Highly Enantioselective Brønsted Acid Catalyst for the Strecker Reaction Magnus Rueping, * Erli Sugiono and Cengiz Azap General: Unless otherwise
Supporting Information Wiley-VCH 2006 69451 Weinheim, Germany A Highly Enantioselective Brønsted Acid Catalyst for the Strecker Reaction Magnus Rueping, * Erli Sugiono and Cengiz Azap General: Unless otherwise
NMRis the most valuable spectroscopic technique for organic chemists because it maps the carbon-hydrogen framework of a molecule.
 Chapter 13: Nuclear magnetic resonance spectroscopy NMRis the most valuable spectroscopic technique for organic chemists because it maps the carbon-hydrogen framework of a molecule. 13.2 The nature of
Chapter 13: Nuclear magnetic resonance spectroscopy NMRis the most valuable spectroscopic technique for organic chemists because it maps the carbon-hydrogen framework of a molecule. 13.2 The nature of
Amino acids: Optimization in underivatized formula and fragment patterns in LC/ESI-MS analysis. Yoshinori Takano (JAMSTEC)
 (a) Positive ion mode (ph < 7) Amino acids: ptimization in underivatized formula and fragment patterns in L/ESI-MS analysis R 1 R 2 N + HA R 3 Base Acid R 1 R N H + 2 R 3 + A - Sample [M + H] + (b) Negative
(a) Positive ion mode (ph < 7) Amino acids: ptimization in underivatized formula and fragment patterns in L/ESI-MS analysis R 1 R 2 N + HA R 3 Base Acid R 1 R N H + 2 R 3 + A - Sample [M + H] + (b) Negative
Hour Examination # 4
 CHEM 346 Organic Chemistry I Fall 2014 Exam # 4 Solutions Key Page 1 of 12 CHEM 346 Organic Chemistry I Fall 2014 Instructor: Paul Bracher Hour Examination # 4 Wednesday, December 3 rd, 2014 6:00 8:00
CHEM 346 Organic Chemistry I Fall 2014 Exam # 4 Solutions Key Page 1 of 12 CHEM 346 Organic Chemistry I Fall 2014 Instructor: Paul Bracher Hour Examination # 4 Wednesday, December 3 rd, 2014 6:00 8:00
Spectroscopy in Organic Chemistry. Types of Spectroscopy in Organic
 Spectroscopy in Organic Chemistry Spectroscopy Spectrum dealing with light, or more specifically, radiation Scope to see Organic Spectroscopy therefore deals with examining how organic molecules interact
Spectroscopy in Organic Chemistry Spectroscopy Spectrum dealing with light, or more specifically, radiation Scope to see Organic Spectroscopy therefore deals with examining how organic molecules interact
SUPPORTING INFORMATION Antimicrobial activity of chlorinated amino acids and peptides
 SUPPORTING INFORMATION Antimicrobial activity of chlorinated amino acids and peptides Melanie S. A. Coker*, Wan-Ping Hu, Senti T Senthilmohan and Anthony J. Kettle Free Radical Research Group, University
SUPPORTING INFORMATION Antimicrobial activity of chlorinated amino acids and peptides Melanie S. A. Coker*, Wan-Ping Hu, Senti T Senthilmohan and Anthony J. Kettle Free Radical Research Group, University
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 205 Supporting Information ortho-alkenylation of anilines with aromatic terminal alkynes over nanosized
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 205 Supporting Information ortho-alkenylation of anilines with aromatic terminal alkynes over nanosized
Amino Acid Side Chain Induced Selectivity in the Hydrolysis of Peptides Catalyzed by a Zr(IV)-Substituted Wells-Dawson Type Polyoxometalate
 Amino Acid Side Chain Induced Selectivity in the Hydrolysis of Peptides Catalyzed by a Zr(IV)-Substituted Wells-Dawson Type Polyoxometalate Stef Vanhaecht, Gregory Absillis, Tatjana N. Parac-Vogt* Department
Amino Acid Side Chain Induced Selectivity in the Hydrolysis of Peptides Catalyzed by a Zr(IV)-Substituted Wells-Dawson Type Polyoxometalate Stef Vanhaecht, Gregory Absillis, Tatjana N. Parac-Vogt* Department
, by reacting CH 3 with ethanoic anhydride, (CH 3
 1 A chemist prepares and analyses some esters. (a) The chemist prepares an ester of propan-2-ol, H 3 H(H)H 3, by reacting H 3 H(H)H 3 with ethanoic anhydride, (H 3 ) 2. Using structural formulae, write
1 A chemist prepares and analyses some esters. (a) The chemist prepares an ester of propan-2-ol, H 3 H(H)H 3, by reacting H 3 H(H)H 3 with ethanoic anhydride, (H 3 ) 2. Using structural formulae, write
Supplementary Information
 Supplementary Information C aryl -C alkyl bond formation from Cu(ClO 4 ) 2 -mediated oxidative cross coupling reaction between arenes and alkyllithium reagents through structurally well-defined Ar-Cu(III)
Supplementary Information C aryl -C alkyl bond formation from Cu(ClO 4 ) 2 -mediated oxidative cross coupling reaction between arenes and alkyllithium reagents through structurally well-defined Ar-Cu(III)
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2017 1 Electronic Supplementary Information Depolymerization of poly-(bisphenol A carbonate)
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2017 1 Electronic Supplementary Information Depolymerization of poly-(bisphenol A carbonate)
Proton NMR. Four Questions
 Proton NMR Four Questions How many signals? Equivalence Where on spectrum? Chemical Shift How big? Integration Shape? Splitting (coupling) 1 Proton NMR Shifts Basic Correlation Chart How many 1 H signals?
Proton NMR Four Questions How many signals? Equivalence Where on spectrum? Chemical Shift How big? Integration Shape? Splitting (coupling) 1 Proton NMR Shifts Basic Correlation Chart How many 1 H signals?
CHEM4. (JAN12CHEM401) WMP/Jan12/CHEM4. General Certificate of Education Advanced Level Examination January 2012
 Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials General Certificate of Education Advanced Level Examination January 2012 Question 1 2 Mark
Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials General Certificate of Education Advanced Level Examination January 2012 Question 1 2 Mark
Structure Determination: Nuclear Magnetic Resonance Spectroscopy
 Structure Determination: Nuclear Magnetic Resonance Spectroscopy Why This Chapter? NMR is the most valuable spectroscopic technique used for structure determination More advanced NMR techniques are used
Structure Determination: Nuclear Magnetic Resonance Spectroscopy Why This Chapter? NMR is the most valuable spectroscopic technique used for structure determination More advanced NMR techniques are used
Supporting Information
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2012 Subcellular Localization and Activity of Gambogic Acid Gianni Guizzunti,* [b] Ayse Batova, [a] Oraphin Chantarasriwong,
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2012 Subcellular Localization and Activity of Gambogic Acid Gianni Guizzunti,* [b] Ayse Batova, [a] Oraphin Chantarasriwong,
Supplementary Material
 10.1071/CH13324_AC CSIRO 2013 Australian Journal of Chemistry 2013, 66(12), 1570-1575 Supplementary Material A Mild and Convenient Synthesis of 1,2,3-Triiodoarenes via Consecutive Iodination/Diazotization/Iodination
10.1071/CH13324_AC CSIRO 2013 Australian Journal of Chemistry 2013, 66(12), 1570-1575 Supplementary Material A Mild and Convenient Synthesis of 1,2,3-Triiodoarenes via Consecutive Iodination/Diazotization/Iodination
Supporting Information. Table S General information General procedure for synthesis of compounds 4 and
 Yang Li, Lipeng Wu, Helfried Neumann and Matthias Beller* Leibniz-Institut für Katalyse e.v. an der Universität Rostock, Albert Einstein Str. 29a, 18059 Rostock, Germany Supporting Information Table S1...
Yang Li, Lipeng Wu, Helfried Neumann and Matthias Beller* Leibniz-Institut für Katalyse e.v. an der Universität Rostock, Albert Einstein Str. 29a, 18059 Rostock, Germany Supporting Information Table S1...
A General and Efficient Method for the Palladium Catalysed Conversion of Allylic Alcohols into their Corresponding Dienes
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2016 A General and Efficient Method for the Palladium Catalysed Conversion of
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2016 A General and Efficient Method for the Palladium Catalysed Conversion of
CSIRO Land & Water, Black Mountain Science and Innovation Park, Acton, ACT 2601, Australia
 Isolation of the (+)-pinoresinol-mineralizing Pseudomonas sp. SG-MS2 and elucidation of its catabolic pathway Madhura Shettigar a,b, Sahil Balotra a, David Cahill b, Andrew C. Warden a, Michael J. Lacey
Isolation of the (+)-pinoresinol-mineralizing Pseudomonas sp. SG-MS2 and elucidation of its catabolic pathway Madhura Shettigar a,b, Sahil Balotra a, David Cahill b, Andrew C. Warden a, Michael J. Lacey
Chemistry 224 Bioorganic Chemistry Friday, Sept. 29, This Exam is closed book and closed notes. Please show all your work!
 page 1 of 6 hemistry 224 ame Bioorganic hemistry Friday, ept. 29, 2000 Exam 1 100 points This Exam is closed book and closed notes Please show all your work! tereochemistry counts as indicated! eatness
page 1 of 6 hemistry 224 ame Bioorganic hemistry Friday, ept. 29, 2000 Exam 1 100 points This Exam is closed book and closed notes Please show all your work! tereochemistry counts as indicated! eatness
Supporting Information. Labeled Ligand Displacement: Extending NMR-based Screening of Protein Targets
 Supporting Information Labeled Ligand Displacement: Extending NMR-based Screening of Protein Targets Steven L. Swann, Danying Song, Chaohong Sun, Philip J. Hajduk, and Andrew M. Petros Global Pharmaceutical
Supporting Information Labeled Ligand Displacement: Extending NMR-based Screening of Protein Targets Steven L. Swann, Danying Song, Chaohong Sun, Philip J. Hajduk, and Andrew M. Petros Global Pharmaceutical
Supporting Information. for. Inhibition of peptide aggregation by means of
 Supporting Information for Inhibition of peptide aggregation by means of enzymatic phosphorylation Kristin Folmert 1, Malgorzata Broncel 2, Hans v. Berlepsch 1, Christopher H. Ullrich 3, Mary-Ann Siegert
Supporting Information for Inhibition of peptide aggregation by means of enzymatic phosphorylation Kristin Folmert 1, Malgorzata Broncel 2, Hans v. Berlepsch 1, Christopher H. Ullrich 3, Mary-Ann Siegert
Electronic Supplementary Information
 Electronic Supplementary Information A new chemo-enzymatic route to chiral 2-hydroxy-4-phenylbutyrates by combining lactonase-mediated resolution with hydrogenation over Pd/C Bing Chen, a Hai-Feng Yin,
Electronic Supplementary Information A new chemo-enzymatic route to chiral 2-hydroxy-4-phenylbutyrates by combining lactonase-mediated resolution with hydrogenation over Pd/C Bing Chen, a Hai-Feng Yin,
CHAPTER 8 ISOLATION AND CHARACTERIZATION OF PHYTOCONSTITUENTS BY COLUMN CHROMATOGRAPHY
 146 CHAPTER 8 ISLATIN AND CHARACTERIZATIN F PHYTCNSTITUENTS BY CLUMN CHRMATGRAPHY 8.1 INTRDUCTIN Column chromatography is an isolation technique in which the phytoconstituents are being eluted by adsorption.
146 CHAPTER 8 ISLATIN AND CHARACTERIZATIN F PHYTCNSTITUENTS BY CLUMN CHRMATGRAPHY 8.1 INTRDUCTIN Column chromatography is an isolation technique in which the phytoconstituents are being eluted by adsorption.
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Self-Assembled Arrays of Peptide Nanotubes by Vapor Deposition Method Lihi Adler-Abramovich 1, Daniel Aronov 2, Peter Beker 2, Maya Yevnin 2, Shiri Stempler 1, Ludmila Buzhansky
SUPPLEMENTARY INFORMATION Self-Assembled Arrays of Peptide Nanotubes by Vapor Deposition Method Lihi Adler-Abramovich 1, Daniel Aronov 2, Peter Beker 2, Maya Yevnin 2, Shiri Stempler 1, Ludmila Buzhansky
Supplementary Material
 ARKIVC 0 (iii) S-S9 Supplementary Material Lewis acid-catalyzed Diels-Alder reaction of -cyclopentenones with Danishefsky s diene: double bond isomerization of tetrahydro-hindene-,(7ah)-diones, and attempts
ARKIVC 0 (iii) S-S9 Supplementary Material Lewis acid-catalyzed Diels-Alder reaction of -cyclopentenones with Danishefsky s diene: double bond isomerization of tetrahydro-hindene-,(7ah)-diones, and attempts
Electronic Supplementary Information
 Electronic Supplementary Information 1. Reagents All the L-amino acids and L-glutathione (reduced form) were purchased from Sangon Biotch. o. (Shanghai, hina); Methyl orange, potassium tetrachloropalladate(ii),
Electronic Supplementary Information 1. Reagents All the L-amino acids and L-glutathione (reduced form) were purchased from Sangon Biotch. o. (Shanghai, hina); Methyl orange, potassium tetrachloropalladate(ii),
MASS SPECTROMETRY. Topics
 MASS SPECTROMETRY MALDI-TOF AND ESI-MS Topics Principle of Mass Spectrometry MALDI-TOF Determination of Mw of Proteins Structural Information by MS: Primary Sequence of a Protein 1 A. Principles Ionization:
MASS SPECTROMETRY MALDI-TOF AND ESI-MS Topics Principle of Mass Spectrometry MALDI-TOF Determination of Mw of Proteins Structural Information by MS: Primary Sequence of a Protein 1 A. Principles Ionization:
SUPPLEMENTARY INFORMATION
 5 N 4 8 20 22 24 2 28 4 8 20 22 24 2 28 a b 0 9 8 7 H c (kda) 95 0 57 4 28 2 5.5 Precipitate before NMR expt. Supernatant before NMR expt. Precipitate after hrs NMR expt. Supernatant after hrs NMR expt.
5 N 4 8 20 22 24 2 28 4 8 20 22 24 2 28 a b 0 9 8 7 H c (kda) 95 0 57 4 28 2 5.5 Precipitate before NMR expt. Supernatant before NMR expt. Precipitate after hrs NMR expt. Supernatant after hrs NMR expt.
Electronic Supplementary Material (ESI) for Chemical Communications This journal is The Royal Society of Chemistry 2012
 Ring Expansion of Alkynyl Cyclopropanes to Highly substituted Cyclobutenes via a N-Sulfonyl-1,2,3-Triazole Intermediate Renhe Liu, Min Zhang, Gabrielle Winston-Mcerson, and Weiping Tang* School of armacy,
Ring Expansion of Alkynyl Cyclopropanes to Highly substituted Cyclobutenes via a N-Sulfonyl-1,2,3-Triazole Intermediate Renhe Liu, Min Zhang, Gabrielle Winston-Mcerson, and Weiping Tang* School of armacy,
De Novo Metabolite Chemical Structure Determination. Paul R. West Ph.D. Stemina Biomarker Discovery, Inc.
 De Novo Metabolite Chemical Structure Determination Paul R. West Ph.D. Stemina Biomarker Discovery, Inc. As a part of non targeted metabolomic analysis, when peaks in the mass spectra are thought to be
De Novo Metabolite Chemical Structure Determination Paul R. West Ph.D. Stemina Biomarker Discovery, Inc. As a part of non targeted metabolomic analysis, when peaks in the mass spectra are thought to be
12/6/12. Dr. Sanjeeva Srivastava IIT Bombay. Primary Structure. Secondary Structure. Tertiary Structure. Quaternary Structure.
 Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
Stereoselectivity of Proline / Cyclobutane Amino Acid-Containing Peptide. Organocatalysts for Asymmetric Aldol Additions: a Rationale
 Stereoselectivity of Proline / Cyclobutane Amino Acid-Containing Peptide Organocatalysts for Asymmetric Aldol Additions: a Rationale Ona Illa, *, Oriol Porcar-Tost, Carme Robledillo, Carlos Elvira, Pau
Stereoselectivity of Proline / Cyclobutane Amino Acid-Containing Peptide Organocatalysts for Asymmetric Aldol Additions: a Rationale Ona Illa, *, Oriol Porcar-Tost, Carme Robledillo, Carlos Elvira, Pau
Light-Controlled Switching of a Non- Photoresponsive Molecular Shuttle
 Supporting Information Light-Controlled Switching of a Non- Photoresponsive Molecular Shuttle Liu-Pan Yang, a,b Fei Jia, a Jie-Shun Cui, a Song-Bo Lu, a and Wei Jiang* a a Department of Chemistry, South
Supporting Information Light-Controlled Switching of a Non- Photoresponsive Molecular Shuttle Liu-Pan Yang, a,b Fei Jia, a Jie-Shun Cui, a Song-Bo Lu, a and Wei Jiang* a a Department of Chemistry, South
Institut für Pharmazeutische Biologie und Biotechnologie, Philipps-Universität. Marburg, Deutschhausstrasse 17A, Marburg, Germany
 Electronic Supplementary Material for: Substrate promiscuity of secondary metabolite enzymes: Prenylation of hydroxynaphthalenes by fungal indole prenyltransferases Xia Yu, Xiulan Xie, and Shu-Ming Li
Electronic Supplementary Material for: Substrate promiscuity of secondary metabolite enzymes: Prenylation of hydroxynaphthalenes by fungal indole prenyltransferases Xia Yu, Xiulan Xie, and Shu-Ming Li
Supporting Information (SI) Isolation and Confirmation of the Proposed Cleistanthol Biogenic Link from Croton Insularis
 SI1 Supporting Information (SI) Isolation and Confirmation of the Proposed Cleistanthol Biogenic Link from Croton Insularis Lidia A. Maslovskaya, Andrei I. Savchenko, Victoria A. Gordon, Paul W. Reddell,
SI1 Supporting Information (SI) Isolation and Confirmation of the Proposed Cleistanthol Biogenic Link from Croton Insularis Lidia A. Maslovskaya, Andrei I. Savchenko, Victoria A. Gordon, Paul W. Reddell,
Organic Chemistry II (CHE ) Examination I March 1, Name (Print legibly): _KEY _. Student ID#: _
 Organic Chemistry II (CHE 232-002) Examination I March 1, 2007 Name (Print legibly): _KEY _ (last) (first) Student ID#: _ PLEASE observe the following: You are allowed to have scratch paper (provided by
Organic Chemistry II (CHE 232-002) Examination I March 1, 2007 Name (Print legibly): _KEY _ (last) (first) Student ID#: _ PLEASE observe the following: You are allowed to have scratch paper (provided by
Organic Chemistry II* (CHE ) Examination I March 1, Name (Print legibly): KEY. Student ID#:
 Organic Chemistry II* (CHE 232-002) Examination I March 1, 2007 Name (Print legibly): KEY (last) (first) Student ID#: PLEASE observe the following: You are allowed to have scratch paper (provided by me),
Organic Chemistry II* (CHE 232-002) Examination I March 1, 2007 Name (Print legibly): KEY (last) (first) Student ID#: PLEASE observe the following: You are allowed to have scratch paper (provided by me),
Tuning Porosity and Activity of Microporous Polymer Network Organocatalysts by Co-Polymerisation
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Tuning Porosity and Activity of Microporous Polymer Network Organocatalysts
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Tuning Porosity and Activity of Microporous Polymer Network Organocatalysts
A prevalent intraresidue hydrogen bond stabilizes proteins
 Supplementary Information A prevalent intraresidue hydrogen bond stabilizes proteins Robert W. Newberry 1 & Ronald T. Raines 1,2 * 1 Department of Chemistry and 2 Department of Biochemistry, University
Supplementary Information A prevalent intraresidue hydrogen bond stabilizes proteins Robert W. Newberry 1 & Ronald T. Raines 1,2 * 1 Department of Chemistry and 2 Department of Biochemistry, University
Synthesis of Levulinic Acid based Poly(amine-co-ester)s
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2018 Synthesis of Levulinic Acid based Poly(amine-co-ester)s Yann Bernhard, Lucas Pagies, Sylvain
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2018 Synthesis of Levulinic Acid based Poly(amine-co-ester)s Yann Bernhard, Lucas Pagies, Sylvain
Supporting Information
 Supporting Information Synthesis of Nitrosobenzene Derivatives via Nitrosodesilylation Reaction Corinna Kohlmeyer, Maike Klüppel and Gerhard Hilt a) Institut für Chemie, Universität Oldenburg, Carl-von-Ossietzky-Straße
Supporting Information Synthesis of Nitrosobenzene Derivatives via Nitrosodesilylation Reaction Corinna Kohlmeyer, Maike Klüppel and Gerhard Hilt a) Institut für Chemie, Universität Oldenburg, Carl-von-Ossietzky-Straße
CHEM Chapter 13. Nuclear Magnetic Spectroscopy (Homework) W
 CHEM 2423. Chapter 13. Nuclear Magnetic Spectroscopy (Homework) W Short Answer 1. For a nucleus to exhibit the nuclear magnetic resonance phenomenon, it must be magnetic. Magnetic nuclei include: a. all
CHEM 2423. Chapter 13. Nuclear Magnetic Spectroscopy (Homework) W Short Answer 1. For a nucleus to exhibit the nuclear magnetic resonance phenomenon, it must be magnetic. Magnetic nuclei include: a. all
Protein Structure Bioinformatics Introduction
 1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
CHEM311 FALL 2005 Practice Exam #3
 EM311 FALL 2005 Practice Exam #3 Instructions: This is a multiple choice / short answer practice exam. For the multiple-choice questions, there may be more than one correct answer. If so, then circle as
EM311 FALL 2005 Practice Exam #3 Instructions: This is a multiple choice / short answer practice exam. For the multiple-choice questions, there may be more than one correct answer. If so, then circle as
Degradation of Rapamycin and its Ring-Opened Isomer: Role of Base Catalysis Yuri V. Il ichev*, Lori Alquier, and Cynthia A.
 Degradation of Rapamycin and its Ring-Opened Isomer: Role of Base Catalysis Yuri V. Il ichev*, Lori Alquier, and Cynthia A. Maryanoff Supplementary Information Table S1. Results of fitting of the kinetic
Degradation of Rapamycin and its Ring-Opened Isomer: Role of Base Catalysis Yuri V. Il ichev*, Lori Alquier, and Cynthia A. Maryanoff Supplementary Information Table S1. Results of fitting of the kinetic
Chapter 5. Complexation of Tholins by 18-crown-6:
 5-1 Chapter 5. Complexation of Tholins by 18-crown-6: Identification of Primary Amines 5.1. Introduction Electrospray ionization (ESI) is an excellent technique for the ionization of complex mixtures,
5-1 Chapter 5. Complexation of Tholins by 18-crown-6: Identification of Primary Amines 5.1. Introduction Electrospray ionization (ESI) is an excellent technique for the ionization of complex mixtures,
Chapter 20: Identification of Compounds
 Chemists are frequently faced with the problem of identifying unknown compounds. Environmental scientists may have to identify pollutants in soils and water, synthetic chemists may want to confirm that
Chemists are frequently faced with the problem of identifying unknown compounds. Environmental scientists may have to identify pollutants in soils and water, synthetic chemists may want to confirm that
Supporting Information for
 Page of 0 0 0 0 Submitted to The Journal of Organic Chemistry S Supporting Information for Syntheses and Spectral Properties of Functionalized, Water-soluble BODIPY Derivatives Lingling Li, Junyan Han,
Page of 0 0 0 0 Submitted to The Journal of Organic Chemistry S Supporting Information for Syntheses and Spectral Properties of Functionalized, Water-soluble BODIPY Derivatives Lingling Li, Junyan Han,
Supporting Information
 Electronic upplementary Material (EI) for rganic Chemistry rontiers. This journal is the Partner rganisations 0 upporting Information Convenient ynthesis of Pentafluoroethyl Thioethers via Catalytic andmeyer
Electronic upplementary Material (EI) for rganic Chemistry rontiers. This journal is the Partner rganisations 0 upporting Information Convenient ynthesis of Pentafluoroethyl Thioethers via Catalytic andmeyer
Supporting Information
 Supporting Information Wiley-VCH 25 69451 Weinheim, Germany Direct Asymmetric α-fluorination of Aldehydes [**] Derek D. Steiner, Nobuyuki Mase, Carlos F. Barbas III* [*] Prof. Dr. C. F. Barbas III, Derek
Supporting Information Wiley-VCH 25 69451 Weinheim, Germany Direct Asymmetric α-fluorination of Aldehydes [**] Derek D. Steiner, Nobuyuki Mase, Carlos F. Barbas III* [*] Prof. Dr. C. F. Barbas III, Derek
NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY
 NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY A STUDENT SHOULD BE ABLE TO: 1. Identify and explain the processes involved in proton ( 1 H) and carbon-13 ( 13 C) nuclear magnetic resonance
NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY A STUDENT SHOULD BE ABLE TO: 1. Identify and explain the processes involved in proton ( 1 H) and carbon-13 ( 13 C) nuclear magnetic resonance
HWeb27 ( ; )
 HWeb27 (9.1-9.2; 9.12-9.18) 28.1. Which of the following cannot be determined about a compound by mass spectrometry? [a]. boiling point [b]. molecular formula [c]. presence of heavy isotopes (e.g., 2 H,
HWeb27 (9.1-9.2; 9.12-9.18) 28.1. Which of the following cannot be determined about a compound by mass spectrometry? [a]. boiling point [b]. molecular formula [c]. presence of heavy isotopes (e.g., 2 H,
Electronic Supplementary Information. Jiani Wang, Lei Zhang, Qiong Qi, Shunhua Li* and Yunbao Jiang
 Electronic Supplementary Information Specific ratiometric fluorescent sensing of Hg 2+ via the formation of mercury(ii) barbiturate coordination polymers Jiani Wang, Lei Zhang, Qiong Qi, Shunhua Li* and
Electronic Supplementary Information Specific ratiometric fluorescent sensing of Hg 2+ via the formation of mercury(ii) barbiturate coordination polymers Jiani Wang, Lei Zhang, Qiong Qi, Shunhua Li* and
Supplementary Table S1: Response evaluation of FDA- approved drugs
 SUPPLEMENTARY DATA, FIGURES AND TABLE BIOLOGICAL DATA Spheroids MARY-X size distribution, morphology and drug screening data Supplementary Figure S1: Spheroids MARY-X size distribution. Spheroid size was
SUPPLEMENTARY DATA, FIGURES AND TABLE BIOLOGICAL DATA Spheroids MARY-X size distribution, morphology and drug screening data Supplementary Figure S1: Spheroids MARY-X size distribution. Spheroid size was
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2015 Supporting Information Photo-Induced Chemistry for the Design of ligonucleotide
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2015 Supporting Information Photo-Induced Chemistry for the Design of ligonucleotide
Brønsted Base-Catalyzed Reductive Cyclization of Alkynyl. α-iminoesters through Auto-Tandem Catalysis
 Supporting Information Brønsted Base-Catalyzed Reductive Cyclization of Alkynyl α-iminoesters through Auto-Tandem Catalysis Azusa Kondoh, b and Masahiro Terada* a a Department of Chemistry, Graduate School
Supporting Information Brønsted Base-Catalyzed Reductive Cyclization of Alkynyl α-iminoesters through Auto-Tandem Catalysis Azusa Kondoh, b and Masahiro Terada* a a Department of Chemistry, Graduate School
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I Dr. Glen B Legge
 EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
Structure solving based on IR, UV-Vis, MS, 1 H and 13 C NMR spectroscopic data. Problem solving session
 Structure solving based on IR, UV-Vis, MS, 1 H and 13 C NMR spectroscopic data Problem solving session S. SANKARARAMAN DEPARTMENT OF CHEMISTRY INDIAN INSTITUTE OF TECHNOLOGY MADRAS CHENNAI 600036 sanka@iitm.ac.in
Structure solving based on IR, UV-Vis, MS, 1 H and 13 C NMR spectroscopic data Problem solving session S. SANKARARAMAN DEPARTMENT OF CHEMISTRY INDIAN INSTITUTE OF TECHNOLOGY MADRAS CHENNAI 600036 sanka@iitm.ac.in
On the Structure of Palau amine: Evidence for the Revised Relative Configuration from Chemical Synthesis
 n the Structure of Palau amine: Evidence for the Revised Relative Configuration from Chemical Synthesis Brian A. Lanman, Larry E. verman,* Ralph Paulini, and Nicole S. White Contribution from the Department
n the Structure of Palau amine: Evidence for the Revised Relative Configuration from Chemical Synthesis Brian A. Lanman, Larry E. verman,* Ralph Paulini, and Nicole S. White Contribution from the Department
(07) 2 (c) 2 (c) (i) Calculate the ph of this buffer solution at 25 oc (3 marks) (Extra space)
 7 The value of Ka for methanoic acid is 1.78 10 4 mol dm 3 at 25 oc. A buffer solution is prepared containing 2.35 10 2 mol of methanoic acid and Question 1: N/A 1.84 10 2 mol of sodium methanoate in 1.00
7 The value of Ka for methanoic acid is 1.78 10 4 mol dm 3 at 25 oc. A buffer solution is prepared containing 2.35 10 2 mol of methanoic acid and Question 1: N/A 1.84 10 2 mol of sodium methanoate in 1.00
Supporting Information
 Supporting Information Wiley-VCH 2007 69451 Weinheim, Germany SUPPRTIG MATERIAL Base Dependence in Copper-catalyzed Huisgen Reactions: Efficient Formation of Bistriazoles Yu Angell and Kevin Burgess *
Supporting Information Wiley-VCH 2007 69451 Weinheim, Germany SUPPRTIG MATERIAL Base Dependence in Copper-catalyzed Huisgen Reactions: Efficient Formation of Bistriazoles Yu Angell and Kevin Burgess *
* * Cambridge International Examinations Cambridge International Advanced Level
 *3272601861* Cambridge International Examinations Cambridge International Advanced Level CHEMISTRY 9701/42 Paper 4 Structured Questions ctober/november 2014 2 hours Candidates answer on the Question Paper.
*3272601861* Cambridge International Examinations Cambridge International Advanced Level CHEMISTRY 9701/42 Paper 4 Structured Questions ctober/november 2014 2 hours Candidates answer on the Question Paper.
Pd(II) Catalyzed C3-selective arylation of pyridine with (hetero)arenes SUPPORTING INFORMATION
 Pd(II) Catalyzed C3-selective arylation of pyridine with (hetero)arenes Guo-Lin Gao,, Wujiong Xia, Pankaj Jain and Jin-Quan Yu *, Department of Chemistry, The Scripps Research Institute, 10550 N. Torrey
Pd(II) Catalyzed C3-selective arylation of pyridine with (hetero)arenes Guo-Lin Gao,, Wujiong Xia, Pankaj Jain and Jin-Quan Yu *, Department of Chemistry, The Scripps Research Institute, 10550 N. Torrey
