Study of adhesion between microspheres and rubber surfaces accompanied by meniscus formation and sedimentation
|
|
|
- Amie Fleming
- 5 years ago
- Views:
Transcription
1 DOI /s x RESEARCH Open Access Study of adhesion between microspheres and rubber surfaces accompanied by meniscus formation and sedimentation Shoko Mishima, Hiroaki Iikura and Toshiaki Ougizawa * *Correspondence: tougizawa@op.titech.ac.jp Graduate School of Science and Engineering, Tokyo Institute of Technology, Ookayama, Meguro ku, Tokyo , Japan Abstract This paper reports on the adhesion characteristics between microspheres and rubber surfaces. Silica, polystyrene, and poly(methyl methacrylate) microspheres were deposited on cis-1,4-polybutadiene (BR) films. A BR meniscus formed on the sphere surfaces when the film thickness was less than the diameters of the spheres. Additionally, the attractive forces acting on the spheres in the direction of the BR films were examined via atomic force microscopy. Sedimentation of the spheres occurred for films with thicknesses much greater than the diameters of the microspheres in all systems. Interestingly, this wetting process occurred even in the silica/br system, despite the incompatibility of these materials. The driving force for meniscus formation is the difference between the surface free energy of BR (γ BR ) and that of the spheres (γ sphere ). For all systems, γ BR is lower than γ sphere, i.e., the BR surface is more stable than those of the spheres, and thus a meniscus forms to stabilize the system. Once a meniscus formed, a downward force acted on the spheres to embed them into the BR film. Sedimentation eventually ceased when the angle between the tangential line of the sphere and the rubber surface became equal to the equilibrium contact angle determined by Young s equation. Interestingly, the sedimentation behavior was nearly identical for spheres with various surface free energy values except in terms of their final positions. The same sedimentation phenomena were studied with crosslinked BR films. In contrast to the experiments performed using various types of spheres, the sedimentation behavior varied with different rubber characteristics. The results of these studies indicate that the sedimentation behavior mainly depends on the physical properties of the rubbers used, although the physical properties of the spheres are in determining their final depth. Keywords: Adhesion, Interface, Surface free energy, Meniscus, Sedimentation Background Pressure-sensitive adhesive (PSA) is a common means of adhesion used in various fields. Examples of daily use include adhesive tape or sticky paper. PSAs are also used in industrial fields such as building materials and cars. However, the phenomenon of adhesion is complex because of the many factors involved, such as surface free energy, viscoelasticity, and cohesion. Many studies have been conducted to investigate the nature The Author(s) 217. This article is distributed under the terms of the Creative Commons Attribution 4. International License ( which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
2 Page 2 of 13 of adhesion, most of which have focused on the various adhesion properties between spheres and substrates. One of the most important aspects involved is the adhesive force. In particular, many theories have been proposed to address deformation induced by the adhesion of spheres on substrates. For example, Johnson et al. [1] investigated the deformation of macroscopic gelatin spheres and presented a theory known as the Johnson Kendall Roberts (JKR) theory, which takes a thermodynamic approach to adhesion. An adhesion energy between a rubber ball and a flat smooth glass surface was investigated by Roberts et al. [2]. They observed the time-dependence of contact radius and surface energy with the equation based on JKR theory. These values eventually reached the certain values. The detail of time-dependence of fracture and adhesion energy was investigated by Presson et al. [3]. Furthermore, Derjaguin et al. [4] proposed the Derjaguin Muller Toporov (DMT) theory, which includes long-range forces. The JKR theory and the DMT theory each have a certain range of validity [5]. In addition to these theories, the Maugis Pollock (MP) theory was presented by Maugis et al. [6]. In comparison with the JKR and DMT theories, which assume purely elastic deformation, the MP theory is unique in addressing plastic deformation. High degrees of deformation in substrates have also been studied, for example in a paper by Rimai et al. using a plasticized polystyrene substrate and soda-lime glass spheres [7 9]. Under these conditions, Rimai et al. observed sedimentation of the spheres into the substrate and extended the JKR, DMT, and MP theories to include an understanding of the substrate deformation. Aside from deformation studies, systems of spheres and substrates have also been investigated to determine the fundamental physical properties of polymers. For instance, Sharp et al. [1] directly assessed the near-surface properties of polystyrene films by monitoring how gold nanoparticles became embedded. The adhesive force between spheres and substrates coated with low viscosity liquids, such as water or silicon oil, have been investigated using a capillary bridge with atomic force microscopy (AFM) [11 14]. The measured force has been fitted to a hydrodynamic force, van der Waals attraction, electrostatic repulsion, hydrodynamic drag, and the restoring force of a cantilever [11, 13]. In addition to these forces, Ally et al. [14] has taken capillary forces into account, which has been a topic of interest among researchers for many years. Cross [15] and Pietsch [16] and their coworkers showed that the capillary force component results from the direct action of the surface free energy, with further details recently reported by Butt et al. [17]. In contrast to studies of deformation and adhesive force performed on low viscosity materials, few reports on the initiation of the adhesion process and wetting progress in viscoelastic materials are available. Therefore, the relationship between the initial stage of stickiness and the physical properties of polymers is not well understood. In this study, we have investigated the adhesion characteristics between micro-scale spheres and rubber films with large thicknesses relative to the sphere diameters. Viscoelastic materials such as rubber generally require a long time to become wetted. To address this problem, we used spheres with sizes on the order of microns. For such small spheres, wetting at the surface proceeds in a sufficiently short time that we were able to observe the process. Additionally, we were able to observe deformation of the viscoelastic material caused by wetting. We focused on four main factors in order to investigate
3 Page 3 of 13 the process of adhesion, including two physical properties of the spheres, the surface free energy and the sphere size, in addition to the surface free energy, and viscoelasticity of rubber. Experimental Materials cis-1,4-polybutadiene (BR) (BR1) was supplied by JSR Corp. The glass transition temperature T g of this material is 18 C, while the number average of the molecular weight M n is 121,193 and M w /M n is Dicumyl peroxide (DCP), which is a crosslinking agent for BR, was purchased from Sigma-Aldrich, Inc. Three types of silica spheres (Hyprecia) were obtained from Ube-Exsymo Co., Ltd with diameters of 5, 1, and 5 μm. Polystyrene (PS) spheres (SBX-12) and poly(methyl methacrylate) (PMMA) spheres (MB-8) were supplied by Sekisui Plastics Co., Ltd with diameters of 12 and 8 μm, respectively. Rubber film preparation Rubber films were prepared with two thicknesses. To prepare thin BR films with thicknesses around 5 nm, a BR solution in toluene (3. wt%) was spin-coated at 2 rpm for 3 s onto a single-crystal silicon wafer with a native oxide layer (Mitsubishi Materials Trading Corp.). The film was dried for at least 24 h at room temperature and the film thickness was evaluated with an ellipsometer (SA-11; Photonic Lattice, Inc.). Rubber films with large thicknesses compared to the diameter of the spheres were prepared by a solvent cast method. A BR solution in toluene (1 wt%) was cast onto a silicon wafer and the resulting film was dried for 24 h at room temperature in ambient conditions and for 24 h under vacuum. The film thickness was determined to be around 16 μm by scanning electron microscopy (SEM) (SM-2; Topcon Corp.). For thicker films, a crosslinked BR film was also made using a BR solution in toluene (1 wt%; DCP 1.5 phr for rubber). After drying in ambient conditions followed by vacuum drying, the film was annealed at 13 C for, 15, 2, 25, and 3 min to induce crosslinking. Hereafter, the crosslinked samples are denote by the crosslinking time appended to BR (e.g., BR15 describes a BR film crosslinked for 15 min). The surface free energies of the rubber films were evaluated with the Owens Wendt equation [18]. Because two types of contact angles are required for this evaluation, we used ultrapure water and diiodomethane, and determined the contact angles with a Dropmaster 3 from Kyowa Interface Science Co., Ltd. The storage modulus G and the loss modulus G of the rubber were measured with a rheometer (parallel plate, φ = 25 mm, 2 C, Rheometrics dynamic mechanical spectrometer RDS-77; Rheometrics, Inc.). The shear moduli of the rubber films were evaluated from the force-distance curve measured by atomic force microscopy (AFM) (MFP-3D-BIO-J; Asylum Research) and the Johnson Kenall Roberts (JKR) theory [1] was applied to the evaluation. The JKR theory is known to be suitable for highly adhesive systems with low stiffness and large tip radii, and can therefore be applied to the system in this study. In several reports on this theory [19, 2], the 2-point method has been proposed for determining the elastic modulus, E. The shear modulus, G, can then be calculated using the following equation:
4 Page 4 of 13 G = E 2(1 + ν) = E 3 (1) where ν is the Poisson s ratio, which was assumed to be.5 in this study. The value of.5 is a typical Poisson s ratio for a non-compressive elastomer. Test for adhesion of spheres to rubber surfaces The spheres were deposited onto the rubber films, then nitrogen gas was blown across the rubber surfaces to remove any unattached spheres. The interfacial area between the spheres and the rubber was imaged using SEM after resting the samples for a designated length of time. Force distance curves from particle probe measurements The adhesion between the silica spheres and thin rubber films was investigated by AFM. These measurements used a silicon cantilever with a silica sphere at the tip (23.9 N/m spring constant, 1 μm silica sphere diameter, 4 nm/s scanning rate, Novascan Technologies, Inc.). The attractive force was also measured by AFM by first moving the sample stage to approach the cantilever and pausing the approach after contact occurred between the substrate and the cantilever following a jump in the force curve. Then, the measured force returned to N, the sample stage was fixed, and the changes in detected force with time were recorded. Equilibrium contact angle measurements A 5-nm-thick BR film was produced using the above method. The film was scratched with tweezers, and submicron fragments of BR that remained attached to the tip of the tweezers were placed on a flat silica glass surface. Changes in the contact angle with time were observed by SEM. Results and discussion Figure 1 shows an SEM image of the interface between a silica sphere with a 1 μm diameter and a BR film with a 5 nm thickness. The image clearly illustrates the formation of a BR meniscus on the silica sphere. Adhesion of the silica occurred quickly, in <1 s, so the rubber meniscus must have formed immediately after the spheres were deposited. Generally, contact between hydrophilic and hydrophobic materials tends to result in smaller contact areas; yet, the hydrophobic BR formed a meniscus on the hydrophilic silica surface. This phenomenon can be understood in terms of the differences in surface free energy. The surface free energy of BR is 28.8 mj/m 2 and that of silica is >3 mj/m 2. In other words, a BR/air system is more stable than a silica/air system, so the BR tends to cover the silica sphere surface. As a result, a BR meniscus formed on the silica sphere surface even though they are not compatible. In addition to silica, the surface free energies of PS and PMMA are 43 and 39 mj/m 2, respectively, which are both larger than that of BR, also resulting in the formation of a BR meniscus on PS and PMMA sphere surfaces.
5 Page 5 of μm Fig. 1 SEM image of the interface between a thin BR film and a silica sphere. 1 μm sphere diameter; 5 nm BR film thickness Figure 2a displays the force-distance curve for a thin BR film (5 nm thickness) measured by a cantilever with a silica sphere tip. As shown in Fig. 2a, the cantilever first adhered to the BR film by van der Waals forces (point A in Fig. 2a); then, the cantilever returned to a straight position by lifting the sample stage (from point B to C) until the force decreased to zero. Afterward, the sample stage was fixed at point C. The force curves measured after fixing the sample stage are shown in Fig. 2b. The data plot indicates that downward forces were detected and the cantilever gradually bent, reflecting an attractive force on the silica sphere as a result of the meniscus (Fig. 3), known as the meniscus force, F meniscus. The meniscus force consists of two components [17]: F meniscus = F Lap + F cap (2) the Laplace force, F Lap, and the capillary force, F cap, which is induced by the Laplace pressure, Δp. This pressure is generated in the vicinity of the interface between two fluids (a).5 (b) C A -2 Force [μn] Force [nn] B Approach Sample stage displacement [μm] Time [sec] Fig. 2 AFM measurement of a thin BR film using a cantilever attached to a silica sphere tip. a Force-distance curve of a thin BR film. b Variation in the attractive force with time. Conditions: 5 nm BR film thickness; 1 μm silica sphere diameter; 23.9 N/m cantilever spring constant; 4 nm/s scanning rate
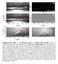
6 Page 6 of 13 Silica sphere R 2 r R BR R 1 Silicon wafer Fig. 3 Schematic of a meniscus between a sphere and a flat substrate L that have different surface free energies. F Lap is described by the following equations [17, 21, 22]: F Lap = S p = πr 2 p (3) p = γ L ( 1 R R 2 )(R 1 >, R 2 < ) (4) where S is the contact area, (1/R 1 + 1/R 2 ) is the surface curvature of an interface, and γ L is the surface free energy of the liquid. The F cap portion of F meniscus, is induced by γ L at the contact line of a meniscus [14, 17, 23, 24]: F cap = lγ L sin(φ + θ) = 2πrγ L sin(φ + θ) (5) where l is the length of the contact line, and the angles φ and θ are shown in Fig. 3. The direction of γ L is tangential to the line of liquid around the contact line. These forces produced an attractive force that acted on the sphere at the tip of the AFM cantilever. The meniscus of rubber formed immediately on the sphere surface after point B, then the meniscus force acted on the sphere and the sphere became embedded in the BR film. As a result, the cantilever was bent and a force was detected (Fig. 2b). Because the attractive force acts on the spheres in the direction of the rubber film, we used thicker rubber films in place of the thin film to investigate the wetting process. These conditions resulted in sedimentation of the silica spheres into the rubber film. Figure 4 shows SEM images of this sedimentation, as well as similar sedimentation features for PS spheres and PMMA spheres. The sedimentation ratio, δ/d, was calculated from SEM images to compare the sedimentation behavior of each sphere type, where the term δ is the sedimentation depth and D is the diameter of the spheres. Figure 5 shows the time-dependence of sedimentation for all three types of spheres. According to Fig. 5, the sedimentation behavior indicated by the shape of the curves was nearly identical for all spheres. Additionally, the sedimentation velocities are also similar for all spheres, even for spheres with very different surface free energies. In comparison to the silica/air
7 Page 7 of 13 (a) (b) (c) 2.μm (d) 2. μm 2. μm 2. μm Fig. 4 SEM images of silica spheres deposited on a thick BR film. 16 μm BR film thickness; 5 μm silica sphere diameter; resting times: a 5 min, b 3 min, c 1 h, and d 792 h 1 D Time [day] δ/d [%] Silica PS PMMA Time [sec] Fig. 5 Resting-time-dependence of sedimentation ratios for silica, PS, and PMMA spheres in a thick BR film. Sphere diameters: silica 1 μm, PS 12 μm, PMMA 8 μm; BR film thickness: 16 μm system, the polymer/air systems are more stable as a result of their smaller surface free energy. Therefore, the silica spheres were expected to be more rapidly embedded into the rubber film than the polymeric spheres, however a different result was obtained. Figure 5 indicates that the sedimentation behavior does not depend strongly on the physical properties of the spheres. These results are also in agreement with Eqs. (4) and (5), which indicate that the attractive force depends only on the physical properties of the rubber. Additionally, we assume a drag force occurs by the viscoelastic deformation of rubber. We discuss the dependence of viscoelasticity of rubber later. Figure 5 also shows that sedimentation eventually ceased at a certain depth, depending on the type of sphere. The final depths were 85% for silica spheres, 95% for PS spheres, and 96% for PMMA spheres. The angle between the rubber surface and the tangential
8 Page 8 of 13 line to the sphere at its final position, θ end, was calculated using the final depth, δ/d. These values were 45, 26, and 23, respectively. According to Eqs. (4) and (5), the sedimentation should cease when the meniscus has vanished. Disappearance of the meniscus requires that Δp = and θ = π φ, then F Lap = and F cap =. Next, we considered the factors that determine the final sedimentation position of the spheres. The terminal contact angle is assumed to be equal to the equilibrium contact angle, θ eq, in Young s equation [25]: γ S = γ SL + γ L cos θ eq (6) Here, γ S and γ L are the surface free energy of the solid and liquid, respectively, and γ SL is the interfacial free energy between the liquid and the solid. For a sphere at the liquid gas interface, the sphere is stable at the position where the forces on the tangential line around the contact line are balanced [1, 26]. Namely, in equilibrium, Young s equation is valid for the tangential line to the sphere. To verify whether this theory also holds in our system, in which spheres are present at a viscoelastic material gas interface, we determined θ eq by the following experiment and compared the values of θ eq with θ end. Pieces of BR with an equilibrium contact area diameter of around 3 μm were placed on silica glass substrates. The time-dependence of the contact angle is shown in Fig. 6. The BR pieces gradually spread along the surface of the silica glass. The final contact angle reached a value of 43, which is broadly consistent with the θ end value of 45. This result indicates that a sphere at a viscoelastic material gas interface, as well as the liquid gas system, is in equilibrium when θ end becomes equal to θ eq. We also confirmed whether the value of θ eq was appropriate comparing the apparent surface free energy reported by Roberts et al. [2]. We calculated the interfacial free energy using Young s equation [Eq. (6)], and it was about 278 mj/m 2. Then, we also calculated the work of adhesion, w, using Dupré equation [21]: γ SL = γ S + γ L w (7) w between the BR and the glass substrate was about 51 mj/m 2. Robert and his coworker reported w of natural rubber (NR) glass system was 6 mj/m 2. BR and NR are 8 Contact angle [ ] Silica glass Time [day] Fig. 6 Resting-time-dependence of contact angle between BR pieces and silica glass
9 Page 9 of 13 considered to have similar adhesion properties, therefore our w value is considered to be an appropriate value. Additionally, our experiments show new method to obtain an interfacial free energy between a solid and a viscoelastic material directly using Young s equation by just placing the micron-order viscoelastic piece on the solid. Dependence on sphere size To confirm the influence of sphere size on the sedimentation behavior, various sizes of silica spheres were deposited onto a thick BR film without a crosslinking agent. Figure 7a shows the time-dependence of sedimentation. The velocities clearly varied among the three spheres sizes, however, the final sedimentation ratios had very similar values. The plots in Fig. 7a, with a reference sphere size of D = 5 μm, can be superposed on one another by shifting the data along the time axis (Fig. 7b). Figure 8 displays a log log plot of the relationship between the shift factors, a t, and the sphere diameter, which Time [day] 1 8 (a) (b) δ/d [%] μm 1μm 5μm δ/d [%] μm 1μm 5μm Time [sec] Time forreference sphere [sec] Fig. 7 Resting-time-dependence of sedimentation ratios of silica spheres with various diameters in a thick BR film. a Raw data, b data shifted along the time axis. The reference sphere size is 5 μm y = x R² =.995 Log (a t ) Log (D) Fig. 8 Shift factors for plotted data in Fig. 7b versus sphere diameter. The dotted line represents a linear relationship
10 Page 1 of 13 approximates a straight line. The slope of 2.5 for this line indicates that the total time for sedimentation is proportional to D to the power of 2.5. This result clearly shows the relaxation process is dominated by time-dependent fracture energy, therefore we have to clarify the relationship between D and a t in the near future. In contrast to the velocity, the sedimentation behavior does not depend on sphere size (i.e., the curve shapes were nearly identical), as the shape of all plots was consistent. As described above, the final depth depends only on the physical properties of the spheres and the rubber: γ S, γ L, and γ SL. The sphere size affects the velocity but not the overall behavior. Dependence on rubber type As discussed above, the type of sphere does not affect the intermediate stage of sedimentation behavior, however the final sphere depth depends significantly on the material. Therefore, we considered the effect of different types of rubber. For this investigation, we observed the sedimentation behavior of silica spheres into crosslinked BR films. Figure 9 shows the cross-linking time-dependence of the storage modulus G and the loss modulus G. This result indicates that the fluidity of the BR film decreases with crosslinking time. Figure 1 shows the cross-linking time-dependence of the surface free energy (Fig. 1a) and the shear modulus (Fig. 1b). Both values increased with 1 5 Crosslinking time 45 min G, G [MPa] 15 min min min min 45 min [rad/s] Fig. 9 Cross-linking time-dependence of the storage modulus G (solid lines) and the loss modulus G (dashed lines) for a thick BR film 4 (a) 16 (b) Surface free energy [mj/m 2 ] G [MPa] Crosslinking time [min] Crosslinking time [min] Fig. 1 Cross-linking time-dependence of a surface free energy and b shear modulus G for a thick BR film
11 Page 11 of 13 greater crosslinking times. The reason for increasing of the surface free energy as the crosslinking proceeds is increases of a cohesive force and a density of the BR. To study the sedimentation behavior, silica spheres with a diameter of 5 μm were deposited on BR, BR15, and BR3 films. The results are shown in Fig. 11a. The plots for BR15 and BR3 were shifted to compare the sedimentation behavior of these samples with that of BR (Fig. 11b). Figure 11b demonstrates that the shapes of the curves are very similar, whereas the final sphere depth for BR3 is different from that of the other samples. Details are provided below. First, the surface free energy of BR is nearly the same as that of BR15 according to Fig. 1a; however, the shear modulus increase with crosslinking time. Therefore, the flow of BR15 from the bottom to the side of a sphere and the sedimentation of BR15 were slower than that of BR. This result suggests that the sedimentation velocity relies on the viscoelasticity of the rubber film. Secondly, BR3 clearly exhibited a different terminal sedimentation position than the other two samples. Because the surface free energy of BR3 is larger than those of BR and BR15, the positions predicted by Young s equation and thus the final depths were different. These results indicate that the sedimentation mechanism is strongly influenced by the physical properties of the rubber film, such as surface free energy and viscoelasticity. These results are also in agreement with Eqs. (4) and (5), which indicate that the attractive force depends only on the physical properties of the rubber. Conclusions We have reported the adhesion characteristics between microspheres and rubber surfaces. For thin rubber films with thicknesses less than the sphere diameters, the formation of a rubber meniscus was observed on the sphere surfaces. In the case of thick rubber films, with large thicknesses compared to the sphere diameters, sedimentation of the microspheres occurred. These studies demonstrate certain important features about the sedimentation mechanisms: A meniscus formed immediately on the sphere surfaces after they were deposited. This result indicates that an attractive force develops in a short time span (<1 s). Time [day] 1 8 (a) (b) δ/d [%] 6 4 Crosslinking time min 2 15 min 3 min Time [sec] Time forreference rubber[sec] Fig. 11 Resting-time-dependence of sedimentation ratios of silica spheres in a crosslinked BR film. a Raw data, b data shifted along the time axis. The reference crosslinking time is min. 5 μm silica sphere diameter; 16 μm BR film thickness δ/d [%] Crosslinking time min 15 min 3 min
12 Page 12 of 13 The system was stabilized as a result of meniscus formation and microspheres becoming embedded in the rubber film. Young s equation is valid to describe the final sphere depth. In other words, the angle between the rubber surface and the tangential line of the sphere in its final position, θ end, is equal to the equilibrium contact angle, θ eq, in Young s equation. The following relationships between the sedimentation phenomena and material factors were also determined: Sedimentation behavior mainly depends on the physical properties of the rubber. Sedimentation behavior is nearly independent of the physical properties of the spheres. The physical properties of the spheres are particularly important in determining their final depth. Abbreviations BR: cis-1,4-polybutadiene; NR: natural rubber; PSA: pressure-sensitive adhesive; JKR: Johnson Kendall Roberts; DMT: Derjaguin Muller Toporov; MP: Maugis Pollock; AFM: atomic force microscopy; DCP: dicumyl peroxide; PS: polystyrene; PMMA: poly(methyl methacrylate); SEM: scanning electron microscopy; T g : glass transition temperature; M n : number average of the molecular weight; M w : weight average of the molecular weight; M w /M n : molecular weight distribution; G : storage modulus; G : loss modulus; E: elastic modulus; G: shear modulus; ν: Poisson s ratio; F meniscus : meniscus force; F Lap : Laplace force; F cap : capillary force; Δp: Laplace pressure; S: contact area; R 1, R 2 : curvature radii of an interface; (1/R 1 + 1/R 2 ): surface curvature of an interface; γ S : surface free energy of the solid; γ L : surface free energy of the liquid; γ SL : interfacial free energy between the liquid and the solid; l: length of the contact line; φ: filling angle; θ: contact angle; D: diameter of the sphere; δ: sedimentation depth; δ/d: sedimentation ratio; θ end : angle between the rubber surface and the tangential line to the sphere at its final position; θ eq : equilibrium contact angle in Young s equation; w: work of adhesion; a t : shift factor. Authors contributions SM prepared the manuscript, observed sedimentation of PS sphere and PMMA sphere. HI conceived of the study, did observations with system of silica sphere and BR film, and carried out AFM measurement. TO conceived of the study, participated in its design and coordination, and revised the draft manuscript. All authors read and approved the final manuscript. Acknowledgements Not applicable. Competing interests The authors declare that they have no competing interests. Availability of data and materials All materials we used were supplied by the companies which we wrote in the material section of main manuscript. Received: 1 November 216 Accepted: 26 January 217 References 1. Johnson KL, Kendall K, Roberts AD. Surface energy and the contact of elastic solids. Proc R Soc Lond Ser A. 1971;324(1558): Roberts AD, Thomas AG. The adhesion and friction of smooth rubber surfaces. Wear. 1975;33: Persson BNJ, Albohr O, Heinrich G, Ueba H. Crack propagation in rubber-like materials. J Phys: Condens Matter. 25;17:R Derjaguin BV, Muller VM, Toporov YuP. Effect of contact deformations on the adhesion of particles. J Colloid Interface Sci. 1975;53(2): Muller VM, Yushchenko VS, Derjaguin BV. On the influence of molecular forces on the deformation of an elastic sphere and its sticking to a rigid plane. J Colloid Interface Sci. 198;77(1): Maugis D, Pollock HM. Surface forces, deformation and adherence at metal microcontacts. Acta Metall. 1984;32(9): Rimai DS, Quesnel DJ, Busnaina AA. The adhesion of dry particles in the nanometer to micrometer-size range. Colloids Surf. 2;165(1 3):3 1.
13 Page 13 of Rimai DS, Schaefer DM, Bowen RC, Quesnel DJ. The time dependence of particle engulfment. Langmuir. 22;18(12): Rimai DS, Ezenyilimba MC, Quesnel DJ. Effects of electrostatic and van der Waals interactions on the adhesion of spherical 7 μm particles. J Adhes. 25;81(3 4): Sharp JS, Teichroeb JH, Forrest JA. The properties of free polymer surfaces and their influence on the glass transition temperature of thin polystyrene films. Eur Phys J E. 24;15(4): Guriyanova S, Bonaccurso E. Influence of wettability and surface charge on the interaction between an aqueous electrolyte solution and a solid surface. Phys Chem Chem Phys. 28;1: Butt H-J, Cappella B, Kappl M. Force measurements with the atomic force microscope: technique, interpretation and applications. Surf Sci Rep. 25;59(1 6): Guriyanova S, Semin B, Rodrigues TS, Butt H-J, Bonaccurso E. Hydrodynamic drainage force in a highly confined geometry: role of surface roughness on different length scales. Microfluid Nanofluid. 21;8(5): Ally J, Vittorias E, Amirfazli A, Kappl M, Bonaccurso E, McNamee CE, Butt H-J. Interaction of a microsphere with a solid-supported liquid film. Langmuir. 21;26(14): Cross NL, Picknett RG. Particle adhesion in the presence of a liquid film. In: Johnson HR, Littler DJ, editors. International conference on mechanism of corrosion by fuel impurities, Marchwood Marchwood: Butterworths; p Pietsch W, Rumpf H. Haftkraft, kaplliardruck, flüssigkeitsvolumen und grenzwinkel einer flüssigkeitsbrücke zwischen zwei kugeln. Chem Ing Technol. 1967;39(15): Butt H-J, Kappl M. Normal capillary forces. Adv Colloid Interface Sci. 29;146: Owens DK, Wendt RC. Estimation of the surface free energy of polymers. J Appl Polym Sci. 1969;13(8): Sun Y, Akhremitchev B, Walker GC. Using the adhesive interaction between atomic force microscopy tips and polymer surfaces to measure the elastic modulus of compliant samples. Langmuir. 24;2(14): Nagai S, Fujinami S, Nakajima K, Nishi T. Nanorheological investigation of polymeric surfaces by atomic force microscopy. Compos Interfaces. 29;16(1): Israelachvili JN. Intermolecular and surface forces. 2nd ed. London: Academic press; Matthewson MJ. Adhesion of spheres by thin liquid films. Philos Mag A. 1988;57(2): Orr FM, Scriven LE. Pendular rings between solids: meniscus properties and capillary force. J Fluid Mech. 1975;67(4): de Gennes PG, Brochard-Wyart F, Quéré D. Gouttes, bulles, perles et ondes. Paris: Belin; Young T. An essay on the cohesion of fluids. Philos Trans R Soc. 185;95: Butt H-J, Graf K, Kappl M. Physics and chemistry of interfaces. Weinheim: Wiley-VCH Verlag & Co. KGaA; 23.
Contents. Preface XI Symbols and Abbreviations XIII. 1 Introduction 1
 V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
! Importance of Particle Adhesion! History of Particle Adhesion! Method of measurement of Adhesion! Adhesion Induced Deformation
 ! Importance of Particle Adhesion! History of Particle Adhesion! Method of measurement of Adhesion! Adhesion Induced Deformation! JKR and non-jkr Theory! Role of Electrostatic Forces! Conclusions Books:
! Importance of Particle Adhesion! History of Particle Adhesion! Method of measurement of Adhesion! Adhesion Induced Deformation! JKR and non-jkr Theory! Role of Electrostatic Forces! Conclusions Books:
Forces Acting on Particle
 Particle-Substrate Interactions: Microscopic Aspects of Adhesion Don Rimai NexPress Solutions LLC. Rochester, NY 14653-64 Email: donald_rimai@nexpress.com (Edited for course presentation by ) Part Role
Particle-Substrate Interactions: Microscopic Aspects of Adhesion Don Rimai NexPress Solutions LLC. Rochester, NY 14653-64 Email: donald_rimai@nexpress.com (Edited for course presentation by ) Part Role
A General Equation for Fitting Contact Area and Friction vs Load Measurements
 Journal of Colloid and Interface Science 211, 395 400 (1999) Article ID jcis.1998.6027, available online at http://www.idealibrary.com on A General Equation for Fitting Contact Area and Friction vs Load
Journal of Colloid and Interface Science 211, 395 400 (1999) Article ID jcis.1998.6027, available online at http://www.idealibrary.com on A General Equation for Fitting Contact Area and Friction vs Load
Supporting Information. Interfacial Shear Strength of Multilayer Graphene Oxide Films
 Supporting Information Interfacial Shear Strength of Multilayer Graphene Oxide Films Matthew Daly a,1, Changhong Cao b,1, Hao Sun b, Yu Sun b, *, Tobin Filleter b, *, and Chandra Veer Singh a, * a Department
Supporting Information Interfacial Shear Strength of Multilayer Graphene Oxide Films Matthew Daly a,1, Changhong Cao b,1, Hao Sun b, Yu Sun b, *, Tobin Filleter b, *, and Chandra Veer Singh a, * a Department
Physics and Chemistry of Interfaces
 Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
ADHESION OF AN AXISYMMETRIC ELASTIC BODY: RANGES OF VALIDITY OF MONOMIAL APPROXIMATIONS AND A TRANSITION MODEL
 ADHESION OF AN AXISYMMETRIC ELASTIC BODY: RANGES OF VALIDITY OF MONOMIAL APPROXIMATIONS AND A TRANSITION MODEL A Thesis Presented By Fouad Oweiss to The Department of Mechanical and Industrial Engineering
ADHESION OF AN AXISYMMETRIC ELASTIC BODY: RANGES OF VALIDITY OF MONOMIAL APPROXIMATIONS AND A TRANSITION MODEL A Thesis Presented By Fouad Oweiss to The Department of Mechanical and Industrial Engineering
Fabrication of ordered array at a nanoscopic level: context
 Fabrication of ordered array at a nanoscopic level: context Top-down method Bottom-up method Classical lithography techniques Fast processes Size limitations it ti E-beam techniques Small sizes Slow processes
Fabrication of ordered array at a nanoscopic level: context Top-down method Bottom-up method Classical lithography techniques Fast processes Size limitations it ti E-beam techniques Small sizes Slow processes
Atomic Force Microscopy imaging and beyond
 Atomic Force Microscopy imaging and beyond Arif Mumtaz Magnetism and Magnetic Materials Group Department of Physics, QAU Coworkers: Prof. Dr. S.K.Hasanain M. Tariq Khan Alam Imaging and beyond Scanning
Atomic Force Microscopy imaging and beyond Arif Mumtaz Magnetism and Magnetic Materials Group Department of Physics, QAU Coworkers: Prof. Dr. S.K.Hasanain M. Tariq Khan Alam Imaging and beyond Scanning
Use of chemically modified AFM tips as a powerful tool for the determination of surface energy of functionalised surfaces
 J. Phys. IV France 124 (2005) 129 134 C EDP Sciences, Les Ulis DOI: 10.1051/jp4:2005124020 Use of chemically modified AFM tips as a powerful tool for the determination of surface energy of functionalised
J. Phys. IV France 124 (2005) 129 134 C EDP Sciences, Les Ulis DOI: 10.1051/jp4:2005124020 Use of chemically modified AFM tips as a powerful tool for the determination of surface energy of functionalised
SUPPLEMENTARY INFORMATION
 Supplementary Information for Manuscript: Nanoscale wear as a stress-assisted chemical reaction Supplementary Methods For each wear increment, the diamond indenter was slid laterally relative to the silicon
Supplementary Information for Manuscript: Nanoscale wear as a stress-assisted chemical reaction Supplementary Methods For each wear increment, the diamond indenter was slid laterally relative to the silicon
Specific ion effects on the interaction of. hydrophobic and hydrophilic self assembled
 Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Basic Laboratory. Materials Science and Engineering. Atomic Force Microscopy (AFM)
 Basic Laboratory Materials Science and Engineering Atomic Force Microscopy (AFM) M108 Stand: 20.10.2015 Aim: Presentation of an application of the AFM for studying surface morphology. Inhalt 1.Introduction...
Basic Laboratory Materials Science and Engineering Atomic Force Microscopy (AFM) M108 Stand: 20.10.2015 Aim: Presentation of an application of the AFM for studying surface morphology. Inhalt 1.Introduction...
Princeton University. Adhsion and Interfacial Failure in Drug Eluting Stents
 Princeton University Adhsion and Interfacial Failure in Drug Eluting Stents Princeton University Background and Introduction Cardiovascular disease is the leading cause of death across the world It represents
Princeton University Adhsion and Interfacial Failure in Drug Eluting Stents Princeton University Background and Introduction Cardiovascular disease is the leading cause of death across the world It represents
Interfacial forces and friction on the nanometer scale: A tutorial
 Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Instabilities in Thin Polymer Films: From Pattern Formation to Rupture
 Instabilities in Thin Polymer Films: From Pattern Formation to Rupture John R. Dutcher*, Kari Dalnoki-Veress Η, Bernie G. Nickel and Connie B. Roth Department of Physics, University of Guelph, Guelph,
Instabilities in Thin Polymer Films: From Pattern Formation to Rupture John R. Dutcher*, Kari Dalnoki-Veress Η, Bernie G. Nickel and Connie B. Roth Department of Physics, University of Guelph, Guelph,
Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Features of static and dynamic friction profiles in one and two dimensions on polymer and atomically flat surfaces using atomic force microscopy
 Features of static and dynamic friction profiles in one and two dimensions on polymer and atomically flat surfaces using atomic force microscopy Author Watson, Gregory, Watson, Jolanta Published 008 Journal
Features of static and dynamic friction profiles in one and two dimensions on polymer and atomically flat surfaces using atomic force microscopy Author Watson, Gregory, Watson, Jolanta Published 008 Journal
PARTICLE ADHESION AND REMOVAL IN POST-CMP APPLICATIONS
 PARTICLE ADHESION AND REMOVAL IN POST-CMP APPLICATIONS George Adams, Ahmed A. Busnaina and Sinan Muftu the oratory Mechanical, Industrial, and Manufacturing Eng. Department Northeastern University, Boston,
PARTICLE ADHESION AND REMOVAL IN POST-CMP APPLICATIONS George Adams, Ahmed A. Busnaina and Sinan Muftu the oratory Mechanical, Industrial, and Manufacturing Eng. Department Northeastern University, Boston,
Atomic force microscopy study of polypropylene surfaces treated by UV and ozone exposure: modification of morphology and adhesion force
 Ž. Applied Surface Science 144 145 1999 627 632 Atomic force microscopy study of polypropylene surfaces treated by UV and ozone exposure: modification of morphology and adhesion force H.-Y. Nie ), M.J.
Ž. Applied Surface Science 144 145 1999 627 632 Atomic force microscopy study of polypropylene surfaces treated by UV and ozone exposure: modification of morphology and adhesion force H.-Y. Nie ), M.J.
Notes on Rubber Friction
 Notes on Rubber Friction 2011 A G Plint Laws of Friction: In dry sliding between a given pair of materials under steady conditions, the coefficient of friction may be almost constant. This is the basis
Notes on Rubber Friction 2011 A G Plint Laws of Friction: In dry sliding between a given pair of materials under steady conditions, the coefficient of friction may be almost constant. This is the basis
Nano-Scale Effect in Adhesive Friction of Sliding Rough Surfaces
 Journal of Nanoscience and Nanoengineering Vol. 1, No. 4, 015, pp. 06-13 http://www.aiscience.org/journal/jnn Nano-Scale Effect in Adhesive Friction of Sliding Rough Surfaces Prasanta Sahoo * Department
Journal of Nanoscience and Nanoengineering Vol. 1, No. 4, 015, pp. 06-13 http://www.aiscience.org/journal/jnn Nano-Scale Effect in Adhesive Friction of Sliding Rough Surfaces Prasanta Sahoo * Department
SUPPLEMENTARY NOTES Supplementary Note 1: Fabrication of Scanning Thermal Microscopy Probes
 SUPPLEMENTARY NOTES Supplementary Note 1: Fabrication of Scanning Thermal Microscopy Probes Fabrication of the scanning thermal microscopy (SThM) probes is summarized in Supplementary Fig. 1 and proceeds
SUPPLEMENTARY NOTES Supplementary Note 1: Fabrication of Scanning Thermal Microscopy Probes Fabrication of the scanning thermal microscopy (SThM) probes is summarized in Supplementary Fig. 1 and proceeds
Chapter 3 Contact Resistance Model with Adhesion between Contact
 Chapter 3 Contact Resistance Model with Adhesion between Contact Surfaces In this chapter, I develop a contact resistance model that includes adhesion between contact surfaces. This chapter is organized
Chapter 3 Contact Resistance Model with Adhesion between Contact Surfaces In this chapter, I develop a contact resistance model that includes adhesion between contact surfaces. This chapter is organized
Lecture 12: Biomaterials Characterization in Aqueous Environments
 3.051J/20.340J 1 Lecture 12: Biomaterials Characterization in Aqueous Environments High vacuum techniques are important tools for characterizing surface composition, but do not yield information on surface
3.051J/20.340J 1 Lecture 12: Biomaterials Characterization in Aqueous Environments High vacuum techniques are important tools for characterizing surface composition, but do not yield information on surface
Analysis of contact deformation between a coated flat plate and a sphere and its practical application
 Computer Methods and Experimental Measurements for Surface Effects and Contact Mechanics VII 307 Analysis of contact deformation between a coated flat plate and a sphere and its practical application T.
Computer Methods and Experimental Measurements for Surface Effects and Contact Mechanics VII 307 Analysis of contact deformation between a coated flat plate and a sphere and its practical application T.
Outline Scanning Probe Microscope (SPM)
 AFM Outline Scanning Probe Microscope (SPM) A family of microscopy forms where a sharp probe is scanned across a surface and some tip/sample interactions are monitored Scanning Tunneling Microscopy (STM)
AFM Outline Scanning Probe Microscope (SPM) A family of microscopy forms where a sharp probe is scanned across a surface and some tip/sample interactions are monitored Scanning Tunneling Microscopy (STM)
Effect of Tabor parameter on hysteresis losses during adhesive contact
 Effect of Tabor parameter on hysteresis losses during adhesive contact M. Ciavarella a, J. A. Greenwood b, J. R. Barber c, a CEMEC-Politecnico di Bari, 7015 Bari - Italy. b Department of Engineering, University
Effect of Tabor parameter on hysteresis losses during adhesive contact M. Ciavarella a, J. A. Greenwood b, J. R. Barber c, a CEMEC-Politecnico di Bari, 7015 Bari - Italy. b Department of Engineering, University
Rubber Friction and Tire Dynamics:
 Mitglied der Helmholtz-Gemeinschaft Rubber Friction and Tire Dynamics: A Comparison of Theory with Experimental Data 21. April 2015 Boris Lorenz Bo Persson Motivation Tire models are often used to simulate
Mitglied der Helmholtz-Gemeinschaft Rubber Friction and Tire Dynamics: A Comparison of Theory with Experimental Data 21. April 2015 Boris Lorenz Bo Persson Motivation Tire models are often used to simulate
STUDIES ON NANO-INDENTATION OF POLYMERIC THIN FILMS USING FINITE ELEMENT METHODS
 STUDIES ON NANO-INDENTATION OF POLYMERIC THIN FILMS USING FINITE ELEMENT METHODS Shen Xiaojun, Yi Sung, Lallit Anand Singapore-MIT Alliance E4-04-0, 4 Engineering Drive 3, Singapore 7576 Zeng Kaiyang Institute
STUDIES ON NANO-INDENTATION OF POLYMERIC THIN FILMS USING FINITE ELEMENT METHODS Shen Xiaojun, Yi Sung, Lallit Anand Singapore-MIT Alliance E4-04-0, 4 Engineering Drive 3, Singapore 7576 Zeng Kaiyang Institute
Mesoscale Science and Technology
 Mesoscale Science and Technology Classical Sciences Atomistic & Molecular Sciences Applications: - Lubrication - Photonics - Fuel Cells - Data Storage The realm of the Mesoscale fosters new perceptions
Mesoscale Science and Technology Classical Sciences Atomistic & Molecular Sciences Applications: - Lubrication - Photonics - Fuel Cells - Data Storage The realm of the Mesoscale fosters new perceptions
RHK Technology Brief
 The Atomic Force Microscope as a Critical Tool for Research in Nanotribology Rachel Cannara and Robert W. Carpick Nanomechanics Laboratory, University of Wisconsin Madison Department of Engineering Physics,
The Atomic Force Microscope as a Critical Tool for Research in Nanotribology Rachel Cannara and Robert W. Carpick Nanomechanics Laboratory, University of Wisconsin Madison Department of Engineering Physics,
Evaluation of energy dissipation involving adhesion hysteresis in spherical contact between a glass lens and a PDMS block
 DOI 10.1186/s40563-017-0082-z RESEARCH Open Access Evaluation of energy dissipation involving adhesion hysteresis in spherical contact between a glass lens and a PDMS block Dooyoung Baek 1*, Pasomphone
DOI 10.1186/s40563-017-0082-z RESEARCH Open Access Evaluation of energy dissipation involving adhesion hysteresis in spherical contact between a glass lens and a PDMS block Dooyoung Baek 1*, Pasomphone
Mohamed Daoud Claudine E.Williams Editors. Soft Matter Physics. With 177 Figures, 16 of them in colour
 Mohamed Daoud Claudine E.Williams Editors Soft Matter Physics With 177 Figures, 16 of them in colour Contents 1. Droplets: CapiUarity and Wetting 1 By F. Brochard-Wyart (With 35 figures) 1.1 Introduction
Mohamed Daoud Claudine E.Williams Editors Soft Matter Physics With 177 Figures, 16 of them in colour Contents 1. Droplets: CapiUarity and Wetting 1 By F. Brochard-Wyart (With 35 figures) 1.1 Introduction
Supporting Information
 Supporting Information Atomic Force Microscopy Nanomechanical Mapping Visualizes Interfacial Broadening between Networks Due to Chemical Exchange Reactions Changfei He, Shaowei Shi, *, Xuefei Wu, Thomas
Supporting Information Atomic Force Microscopy Nanomechanical Mapping Visualizes Interfacial Broadening between Networks Due to Chemical Exchange Reactions Changfei He, Shaowei Shi, *, Xuefei Wu, Thomas
Microfluidics 2 Surface tension, contact angle, capillary flow
 MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
Effects of Nanoparticle Coating on the Adhesion of Emulsion Aggregation Toner Particles
 Journal of Adhesion Science and Technology 24 (2010) 371 387 brill.nl/jast Effects of Nanoparticle Coating on the Adhesion of Emulsion Aggregation Toner Particles Xiarong Tong, Weiqiang Ding and Cetin
Journal of Adhesion Science and Technology 24 (2010) 371 387 brill.nl/jast Effects of Nanoparticle Coating on the Adhesion of Emulsion Aggregation Toner Particles Xiarong Tong, Weiqiang Ding and Cetin
Supplementary Material
 Mangili et al. Supplementary Material 2 A. Evaluation of substrate Young modulus from AFM measurements 3 4 5 6 7 8 Using the experimental correlations between force and deformation from AFM measurements,
Mangili et al. Supplementary Material 2 A. Evaluation of substrate Young modulus from AFM measurements 3 4 5 6 7 8 Using the experimental correlations between force and deformation from AFM measurements,
Adhesion at Poly(Butylacrylate) Poly(Dimethylsiloxane) Interfaces
 The Journal of Adhesion, 83:741 760, 2007 Copyright # Taylor & Francis Group, LLC ISSN: 0021-8464 print=1545-5823 online DOI: 10.1080/00218460701585840 Adhesion at Poly(Butylacrylate) Poly(Dimethylsiloxane)
The Journal of Adhesion, 83:741 760, 2007 Copyright # Taylor & Francis Group, LLC ISSN: 0021-8464 print=1545-5823 online DOI: 10.1080/00218460701585840 Adhesion at Poly(Butylacrylate) Poly(Dimethylsiloxane)
Praktikum zur. Materialanalytik
 Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Introduction. Sample kit content
 Introduction Polymer blend sample preparation TN01062 Preparation of polymer samples available from the Lateral force, Phase imaging, and Force modulation mode kits The SBS-PMMA, SBS-PS, and SBR-PMMA samples
Introduction Polymer blend sample preparation TN01062 Preparation of polymer samples available from the Lateral force, Phase imaging, and Force modulation mode kits The SBS-PMMA, SBS-PS, and SBR-PMMA samples
Contents. Preface XIII. 1 General Introduction 1 References 6
 VII Contents Preface XIII 1 General Introduction 1 References 6 2 Interparticle Interactions and Their Combination 7 2.1 Hard-Sphere Interaction 7 2.2 Soft or Electrostatic Interaction 7 2.3 Steric Interaction
VII Contents Preface XIII 1 General Introduction 1 References 6 2 Interparticle Interactions and Their Combination 7 2.1 Hard-Sphere Interaction 7 2.2 Soft or Electrostatic Interaction 7 2.3 Steric Interaction
INTERMOLECULAR AND SURFACE FORCES
 INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
Quantitative Nanomechanical Measurements in HybriD TM Mode Atomic Force Microscopy
 Application Note 090 Quantitative Nanomechanical Measurements in HybriD TM Mode Atomic Force Microscopy Sergei Magonov 1, Marko Surtchev 1, Sergey Belikov 1, Ivan Malovichko 2 and Stanislav Leesment 2
Application Note 090 Quantitative Nanomechanical Measurements in HybriD TM Mode Atomic Force Microscopy Sergei Magonov 1, Marko Surtchev 1, Sergey Belikov 1, Ivan Malovichko 2 and Stanislav Leesment 2
Imaging Methods: Scanning Force Microscopy (SFM / AFM)
 Imaging Methods: Scanning Force Microscopy (SFM / AFM) The atomic force microscope (AFM) probes the surface of a sample with a sharp tip, a couple of microns long and often less than 100 Å in diameter.
Imaging Methods: Scanning Force Microscopy (SFM / AFM) The atomic force microscope (AFM) probes the surface of a sample with a sharp tip, a couple of microns long and often less than 100 Å in diameter.
Lecture 7 Contact angle phenomena and wetting
 Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Generalized Wenzel equation for contact angle of droplets on spherical rough solid substrates
 Science Front Publishers Journal for Foundations and Applications of Physics, 3 (2), (2016) (sciencefront.org) ISSN 2394-3688 Generalized Wenzel equation for contact angle of droplets on spherical rough
Science Front Publishers Journal for Foundations and Applications of Physics, 3 (2), (2016) (sciencefront.org) ISSN 2394-3688 Generalized Wenzel equation for contact angle of droplets on spherical rough
Van der Waals Interaction between Polymer Aggregates and Substrate Surface Analyzed by Atomic Force Microscope (AFM)
 Journal of Photopolymer Science and Technology Volume 15,Number 1(2002)127-132 2002TAPJ L Van der Waals Interaction between Polymer Aggregates and Substrate Surface Analyzed by Atomic Force Microscope
Journal of Photopolymer Science and Technology Volume 15,Number 1(2002)127-132 2002TAPJ L Van der Waals Interaction between Polymer Aggregates and Substrate Surface Analyzed by Atomic Force Microscope
Dispersion of rgo in Polymeric Matrices by Thermodynamically Favorable Self-Assembly of GO at Oil-Water Interfaces
 Supporting information for Dispersion of rgo in Polymeric Matrices by Thermodynamically Favorable Self-Assembly of GO at Oil-Water Interfaces Saeed Zajforoushan Moghaddam, Sina Sabury and Farhad Sharif*
Supporting information for Dispersion of rgo in Polymeric Matrices by Thermodynamically Favorable Self-Assembly of GO at Oil-Water Interfaces Saeed Zajforoushan Moghaddam, Sina Sabury and Farhad Sharif*
Abstract. The principles and applicability of surface structure and hydrophobicity of polymers (PS, PDMS),
 Contact Angle Goniometer: Hydrophobicity of Biomaterial Surfaces and Protein Coatings Eman Mousa Alhajji North Carolina State University Department of Materials Science and Engineering MSE 255 Lab Report
Contact Angle Goniometer: Hydrophobicity of Biomaterial Surfaces and Protein Coatings Eman Mousa Alhajji North Carolina State University Department of Materials Science and Engineering MSE 255 Lab Report
CHAPTER 1 Fluids and their Properties
 FLUID MECHANICS Gaza CHAPTER 1 Fluids and their Properties Dr. Khalil Mahmoud ALASTAL Objectives of this Chapter: Define the nature of a fluid. Show where fluid mechanics concepts are common with those
FLUID MECHANICS Gaza CHAPTER 1 Fluids and their Properties Dr. Khalil Mahmoud ALASTAL Objectives of this Chapter: Define the nature of a fluid. Show where fluid mechanics concepts are common with those
Mechanics of wafer bonding: Effect of clamping
 JOURNAL OF APPLIED PHYSICS VOLUME 95, NUMBER 1 1 JANUARY 2004 Mechanics of wafer bonding: Effect of clamping K. T. Turner a) Massachusetts Institute of Technology, Cambridge, Massachusetts 0219 M. D. Thouless
JOURNAL OF APPLIED PHYSICS VOLUME 95, NUMBER 1 1 JANUARY 2004 Mechanics of wafer bonding: Effect of clamping K. T. Turner a) Massachusetts Institute of Technology, Cambridge, Massachusetts 0219 M. D. Thouless
Supporting Information
 Supporting Information Wiley-VCH 2013 69451 Weinheim, Germany Colloidal Clusters by Using Emulsions and Dumbbell-Shaped Particles: Experiments and Simulations** Bo Peng,* Frank Smallenburg,* Arnout Imhof,
Supporting Information Wiley-VCH 2013 69451 Weinheim, Germany Colloidal Clusters by Using Emulsions and Dumbbell-Shaped Particles: Experiments and Simulations** Bo Peng,* Frank Smallenburg,* Arnout Imhof,
A van der Waals Force-Based Adhesion Model for Micromanipulation
 Journal of Adhesion Science and Technology 24 (2010) 2415 2428 brill.nl/jast A van der Waals Force-Based Adhesion Model for Micromanipulation S. Alvo a,b,, P. Lambert b,c, M. Gauthier b and S. Régnier
Journal of Adhesion Science and Technology 24 (2010) 2415 2428 brill.nl/jast A van der Waals Force-Based Adhesion Model for Micromanipulation S. Alvo a,b,, P. Lambert b,c, M. Gauthier b and S. Régnier
Supplementary Information. Synthesis of soft colloids with well controlled softness
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supplementary Information Synthesis of soft colloids with well controlled softness Fuhua Luo, Zhifeng
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supplementary Information Synthesis of soft colloids with well controlled softness Fuhua Luo, Zhifeng
Module 3: "Thin Film Hydrodynamics" Lecture 11: "" The Lecture Contains: Micro and Nano Scale Hydrodynamics with and without Free Surfaces
 The Lecture Contains: Micro and Nano Scale Hydrodynamics with and without Free Surfaces Order of Magnitude Analysis file:///e /courses/colloid_interface_science/lecture11/11_1.htm[6/16/2012 1:39:56 PM]
The Lecture Contains: Micro and Nano Scale Hydrodynamics with and without Free Surfaces Order of Magnitude Analysis file:///e /courses/colloid_interface_science/lecture11/11_1.htm[6/16/2012 1:39:56 PM]
Effects of Size, Humidity, and Aging on Particle Removal
 LEVITRONIX Ultrapure Fluid Handling and Wafer Cleaning Conference 2009 February 10, 2009 Effects of Size, Humidity, and Aging on Particle Removal Jin-Goo Park Feb. 10, 2009 Department t of Materials Engineering,
LEVITRONIX Ultrapure Fluid Handling and Wafer Cleaning Conference 2009 February 10, 2009 Effects of Size, Humidity, and Aging on Particle Removal Jin-Goo Park Feb. 10, 2009 Department t of Materials Engineering,
Reaction at the Interfaces
 Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Stress Overshoot of Polymer Solutions at High Rates of Shear
 Stress Overshoot of Polymer Solutions at High Rates of Shear K. OSAKI, T. INOUE, T. ISOMURA Institute for Chemical Research, Kyoto University, Uji, Kyoto 611-0011, Japan Received 3 April 2000; revised
Stress Overshoot of Polymer Solutions at High Rates of Shear K. OSAKI, T. INOUE, T. ISOMURA Institute for Chemical Research, Kyoto University, Uji, Kyoto 611-0011, Japan Received 3 April 2000; revised
Lecture Note October 1, 2009 Nanostructure characterization techniques
 Lecture Note October 1, 29 Nanostructure characterization techniques UT-Austin PHYS 392 T, unique # 5977 ME 397 unique # 1979 CHE 384, unique # 151 Instructor: Professor C.K. Shih Subjects: Applications
Lecture Note October 1, 29 Nanostructure characterization techniques UT-Austin PHYS 392 T, unique # 5977 ME 397 unique # 1979 CHE 384, unique # 151 Instructor: Professor C.K. Shih Subjects: Applications
Contact, Adhesion and Rupture of Elastic Solids
 D. Maugis Contact, Adhesion and Rupture of Elastic Solids With 186 Figures Springer Contents г 1. Elements of Surface Physics 1 1.1 Introduction 1 1.2 Van der Waals Forces Between Molecules 3 1.2.1 The
D. Maugis Contact, Adhesion and Rupture of Elastic Solids With 186 Figures Springer Contents г 1. Elements of Surface Physics 1 1.1 Introduction 1 1.2 Van der Waals Forces Between Molecules 3 1.2.1 The
Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Supplementary Information Durable Gels with Ultra-low Adhesion to Ice D.
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Supplementary Information Durable Gels with Ultra-low Adhesion to Ice D.
Particle removal in linear shear flow: model prediction and experimental validation
 Particle removal in linear shear flow: model prediction and experimental validation M.L. Zoeteweij, J.C.J. van der Donck and R. Versluis TNO Science and Industry, P.O. Box 155, 600 AD Delft, The Netherlands
Particle removal in linear shear flow: model prediction and experimental validation M.L. Zoeteweij, J.C.J. van der Donck and R. Versluis TNO Science and Industry, P.O. Box 155, 600 AD Delft, The Netherlands
A CONTACT-MECHANICS BASED MODEL FOR DISHING AND EROSION IN
 Mat. Res. Soc. Symp. Proc. Vol. 671 001 Materials Research Society A CONTACT-MECHANICS BASED MODEL FOR DISHING AND EROSION IN CHEMICAL-MECHANICAL POLISHING Joost J. Vlassak Division of Engineering and
Mat. Res. Soc. Symp. Proc. Vol. 671 001 Materials Research Society A CONTACT-MECHANICS BASED MODEL FOR DISHING AND EROSION IN CHEMICAL-MECHANICAL POLISHING Joost J. Vlassak Division of Engineering and
spreading of drops on soft surfaces
 Supplementary Material on Electrically modulated dynamic spreading of drops on soft surfaces Ranabir Dey 1, Ashish Daga 1, Sunando DasGupta 2,3, Suman Chakraborty 1,3 1 Department of Mechanical Engineering,
Supplementary Material on Electrically modulated dynamic spreading of drops on soft surfaces Ranabir Dey 1, Ashish Daga 1, Sunando DasGupta 2,3, Suman Chakraborty 1,3 1 Department of Mechanical Engineering,
3.032 Problem Set 2 Solutions Fall 2007 Due: Start of Lecture,
 3.032 Problem Set 2 Solutions Fall 2007 Due: Start of Lecture, 09.21.07 1. In the beam considered in PS1, steel beams carried the distributed weight of the rooms above. To reduce stress on the beam, it
3.032 Problem Set 2 Solutions Fall 2007 Due: Start of Lecture, 09.21.07 1. In the beam considered in PS1, steel beams carried the distributed weight of the rooms above. To reduce stress on the beam, it
Thermodynamically Stable Emulsions Using Janus Dumbbells as Colloid Surfactants
 Thermodynamically Stable Emulsions Using Janus Dumbbells as Colloid Surfactants Fuquan Tu, Bum Jun Park and Daeyeon Lee*. Description of the term notionally swollen droplets When particles are adsorbed
Thermodynamically Stable Emulsions Using Janus Dumbbells as Colloid Surfactants Fuquan Tu, Bum Jun Park and Daeyeon Lee*. Description of the term notionally swollen droplets When particles are adsorbed
Analytical Prediction of Particle Detachment from a Flat Surface by Turbulent Air Flows
 Chiang Mai J. Sci. 2011; 38(3) 503 Chiang Mai J. Sci. 2011; 38(3) : 503-507 http://it.science.cmu.ac.th/ejournal/ Short Communication Analytical Prediction of Particle Detachment from a Flat Surface by
Chiang Mai J. Sci. 2011; 38(3) 503 Chiang Mai J. Sci. 2011; 38(3) : 503-507 http://it.science.cmu.ac.th/ejournal/ Short Communication Analytical Prediction of Particle Detachment from a Flat Surface by
Probing the Hydrophobic Interaction between Air Bubbles and Partially. Hydrophobic Surfaces Using Atomic Force Microscopy
 Supporting Information for Probing the Hydrophobic Interaction between Air Bubbles and Partially Hydrophobic Surfaces Using Atomic Force Microscopy Chen Shi, 1 Derek Y.C. Chan, 2.3 Qingxia Liu, 1 Hongbo
Supporting Information for Probing the Hydrophobic Interaction between Air Bubbles and Partially Hydrophobic Surfaces Using Atomic Force Microscopy Chen Shi, 1 Derek Y.C. Chan, 2.3 Qingxia Liu, 1 Hongbo
AFM Imaging In Liquids. W. Travis Johnson PhD Agilent Technologies Nanomeasurements Division
 AFM Imaging In Liquids W. Travis Johnson PhD Agilent Technologies Nanomeasurements Division Imaging Techniques: Scales Proteins 10 nm Bacteria 1μm Red Blood Cell 5μm Human Hair 75μm Si Atom Spacing 0.4nm
AFM Imaging In Liquids W. Travis Johnson PhD Agilent Technologies Nanomeasurements Division Imaging Techniques: Scales Proteins 10 nm Bacteria 1μm Red Blood Cell 5μm Human Hair 75μm Si Atom Spacing 0.4nm
Frictional characteristics of exfoliated and epitaxial graphene
 Frictional characteristics of exfoliated and epitaxial graphene Young Jun Shin a,b, Ryan Stromberg c, Rick Nay c, Han Huang d, Andrew T. S. Wee d, Hyunsoo Yang a,b,*, Charanjit S. Bhatia a a Department
Frictional characteristics of exfoliated and epitaxial graphene Young Jun Shin a,b, Ryan Stromberg c, Rick Nay c, Han Huang d, Andrew T. S. Wee d, Hyunsoo Yang a,b,*, Charanjit S. Bhatia a a Department
Influence of Surface Energy of Polymer Films on Spreading and Adhesion of UV-Flexo Inks
 acta graphica 219 short communication received: 08-04-2013. accepted: 17-07-2013. Influence of Surface Energy of Polymer Films on Spreading and Adhesion of UV-Flexo Inks Authors Vyacheslav Repeta Ukrainian
acta graphica 219 short communication received: 08-04-2013. accepted: 17-07-2013. Influence of Surface Energy of Polymer Films on Spreading and Adhesion of UV-Flexo Inks Authors Vyacheslav Repeta Ukrainian
Effect of Non-Ionic Surfactants on Dispersion and. Polar Interactions in the Adsorption of Cellulases. onto Lignin
 Supporting Information Effect of Non-Ionic Surfactants on Dispersion and Polar Interactions in the Adsorption of Cellulases onto Lignin Feng Jiang, Chen Qian, Alan R. Esker and Maren Roman, * Macromolecules
Supporting Information Effect of Non-Ionic Surfactants on Dispersion and Polar Interactions in the Adsorption of Cellulases onto Lignin Feng Jiang, Chen Qian, Alan R. Esker and Maren Roman, * Macromolecules
A Short Course on Nanoscale Mechanical Characterization: The Theory and Practice of Contact Probe Techniques
 A Short Course on Nanoscale Mechanical Characterization: The Theory and Practice of Contact Probe Techniques Tuesday, Part 1: Contact Mechanics -Hertz & Boussinesq Problem -Adhesive elastic contact mechanics
A Short Course on Nanoscale Mechanical Characterization: The Theory and Practice of Contact Probe Techniques Tuesday, Part 1: Contact Mechanics -Hertz & Boussinesq Problem -Adhesive elastic contact mechanics
Wetting & Adhesion on Soft Surfaces Young s Law is dead long live Young s Law. Eric Dufresne
 Wetting & Adhesion on Soft Surfaces Young s Law is dead long live Young s Law Eric Dufresne KITP 2014 Wetting Adhesion 3mm 30 um Young s Law relates contact line geometry and material properties in equilibrium
Wetting & Adhesion on Soft Surfaces Young s Law is dead long live Young s Law Eric Dufresne KITP 2014 Wetting Adhesion 3mm 30 um Young s Law relates contact line geometry and material properties in equilibrium
Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
W. DING, A. J. HOWARD, M.D. MURTHY PERI and C. CETINKAYA*
 Philosophical Magazine, Vol. 87, No. 36, 21 December 2007, 5685 5696 Rolling resistance moment of microspheres on surfaces: contact measurements W. DING, A. J. HOWARD, M.D. MURTHY PERI and C. CETINKAYA*
Philosophical Magazine, Vol. 87, No. 36, 21 December 2007, 5685 5696 Rolling resistance moment of microspheres on surfaces: contact measurements W. DING, A. J. HOWARD, M.D. MURTHY PERI and C. CETINKAYA*
MPIKG Public Access Author Manuscript
 MPIKG Public Access Author Manuscript Max Planck Institute of of Colloids and Interfaces Author Manuscript Published in final edited form as: Stocco, A., & Möhwald, H. (2015). The influence of long-range
MPIKG Public Access Author Manuscript Max Planck Institute of of Colloids and Interfaces Author Manuscript Published in final edited form as: Stocco, A., & Möhwald, H. (2015). The influence of long-range
Colloidal Suspension Rheology Chapter 1 Study Questions
 Colloidal Suspension Rheology Chapter 1 Study Questions 1. What forces act on a single colloidal particle suspended in a flowing fluid? Discuss the dependence of these forces on particle radius. 2. What
Colloidal Suspension Rheology Chapter 1 Study Questions 1. What forces act on a single colloidal particle suspended in a flowing fluid? Discuss the dependence of these forces on particle radius. 2. What
Contents. Preface XIII
 V Contents Preface XIII 1 General Introduction 1 1.1 Fundamental Knowledge Required for Successful Dispersion of Powders into Liquids 1 1.1.1 Wetting of Powder into Liquid 1 1.1.2 Breaking of Aggregates
V Contents Preface XIII 1 General Introduction 1 1.1 Fundamental Knowledge Required for Successful Dispersion of Powders into Liquids 1 1.1.1 Wetting of Powder into Liquid 1 1.1.2 Breaking of Aggregates
ISCST shall not be responsible for statements or opinions contained in papers or printed in its publications.
 Modeling of Drop Motion on Solid Surfaces with Wettability Gradients J. B. McLaughlin, Sp. S. Saravanan, N. Moumen, and R. S. Subramanian Department of Chemical Engineering Clarkson University Potsdam,
Modeling of Drop Motion on Solid Surfaces with Wettability Gradients J. B. McLaughlin, Sp. S. Saravanan, N. Moumen, and R. S. Subramanian Department of Chemical Engineering Clarkson University Potsdam,
Physical properties of porous membranes. Membranes D f S BET [m 2 /g] d peak [nm]
![Physical properties of porous membranes. Membranes D f S BET [m 2 /g] d peak [nm] Physical properties of porous membranes. Membranes D f S BET [m 2 /g] d peak [nm]](/thumbs/79/80072104.jpg) The Sol-Gel Preparation and Characterization of Nanoporous Silica Membrane with Controlled Pore Size T. Fujii, T. Izumi, Dept. of Food Sci., Niigata Univ. of Pharm. & Appl. Life Sci., Niitsu, Niigata 956-8603,
The Sol-Gel Preparation and Characterization of Nanoporous Silica Membrane with Controlled Pore Size T. Fujii, T. Izumi, Dept. of Food Sci., Niigata Univ. of Pharm. & Appl. Life Sci., Niitsu, Niigata 956-8603,
characterize antiadhesive surfaces
 Capillary bridge formation and breakage: a test to characterize antiadhesive surfaces TITLE RUNNING HEAD: Capillary bridge for antiadhesive surfaces. Laurianne Vagharchakian 1, Frédéric Restagno 2, Liliane
Capillary bridge formation and breakage: a test to characterize antiadhesive surfaces TITLE RUNNING HEAD: Capillary bridge for antiadhesive surfaces. Laurianne Vagharchakian 1, Frédéric Restagno 2, Liliane
For an imposed stress history consisting of a rapidly applied step-function jump in
 Problem 2 (20 points) MASSACHUSETTS INSTITUTE OF TECHNOLOGY DEPARTMENT OF MECHANICAL ENGINEERING CAMBRIDGE, MASSACHUSETTS 0239 2.002 MECHANICS AND MATERIALS II SOLUTION for QUIZ NO. October 5, 2003 For
Problem 2 (20 points) MASSACHUSETTS INSTITUTE OF TECHNOLOGY DEPARTMENT OF MECHANICAL ENGINEERING CAMBRIDGE, MASSACHUSETTS 0239 2.002 MECHANICS AND MATERIALS II SOLUTION for QUIZ NO. October 5, 2003 For
Foundations of. Colloid Science SECOND EDITION. Robert J. Hunter. School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS
 Foundations of Colloid Science SECOND EDITION Robert J. Hunter School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS CONTENTS 1 NATURE OF COLLOIDAL DISPERSIONS 1.1 Introduction 1 1.2 Technological
Foundations of Colloid Science SECOND EDITION Robert J. Hunter School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS CONTENTS 1 NATURE OF COLLOIDAL DISPERSIONS 1.1 Introduction 1 1.2 Technological
Hydrophilization of Fluoropolymers and Silicones
 2017 Adhesive and Sealant Council Spring Meeting Hydrophilization of Fluoropolymers and Silicones Aknowledgements: Wei Chen Mount Holyoke College NSF, NIH, Dreyfus, ACS-RF, MHC Bryony Coupe, Mamle Quarmyne,
2017 Adhesive and Sealant Council Spring Meeting Hydrophilization of Fluoropolymers and Silicones Aknowledgements: Wei Chen Mount Holyoke College NSF, NIH, Dreyfus, ACS-RF, MHC Bryony Coupe, Mamle Quarmyne,
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa.
 Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
REMOVING A PIECE OF ADHESIVE TAPE FROM A HORIZONTAL SURFACE
 REMOVING A PIECE OF ADHESIVE TAPE FROM A HORIZONTAL SURFACE Hossein Azizinaghsh a, Hamid Ghaednia b a Sharif University of Technology, School of Computer Engineering, I. R. Iran Hossein.Azizi@gmail.com
REMOVING A PIECE OF ADHESIVE TAPE FROM A HORIZONTAL SURFACE Hossein Azizinaghsh a, Hamid Ghaednia b a Sharif University of Technology, School of Computer Engineering, I. R. Iran Hossein.Azizi@gmail.com
The Origins of Surface and Interfacial Tension
 The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
Sliding Friction in the Frenkel-Kontorova Model
 arxiv:cond-mat/9510058v1 12 Oct 1995 to appear in "The Physics of Sliding Friction", Ed. B.N.J. Persson (Kluwer Academic Publishers), 1995 Sliding Friction in the Frenkel-Kontorova Model E. Granato I.N.P.E.,
arxiv:cond-mat/9510058v1 12 Oct 1995 to appear in "The Physics of Sliding Friction", Ed. B.N.J. Persson (Kluwer Academic Publishers), 1995 Sliding Friction in the Frenkel-Kontorova Model E. Granato I.N.P.E.,
DLVO interaction between the spheres
 DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
Research Article Adhesion on Nanoorganized Multilayers
 Advances in Physical Chemistry Volume, Article ID 8543, 6 pages doi:.55//8543 Research Article Adhesion on Nanoorganized Multilayers Yolla Kazzi and Houssein Awada Faculty of Sciences, Lebanese University,
Advances in Physical Chemistry Volume, Article ID 8543, 6 pages doi:.55//8543 Research Article Adhesion on Nanoorganized Multilayers Yolla Kazzi and Houssein Awada Faculty of Sciences, Lebanese University,
Scanning Tunneling Microscopy
 Scanning Tunneling Microscopy References: 1. G. Binnig, H. Rohrer, C. Gerber, and Weibel, Phys. Rev. Lett. 49, 57 (1982); and ibid 50, 120 (1983). 2. J. Chen, Introduction to Scanning Tunneling Microscopy,
Scanning Tunneling Microscopy References: 1. G. Binnig, H. Rohrer, C. Gerber, and Weibel, Phys. Rev. Lett. 49, 57 (1982); and ibid 50, 120 (1983). 2. J. Chen, Introduction to Scanning Tunneling Microscopy,
General concept and defining characteristics of AFM. Dina Kudasheva Advisor: Prof. Mary K. Cowman
 General concept and defining characteristics of AFM Dina Kudasheva Advisor: Prof. Mary K. Cowman Overview Introduction History of the SPM invention Technical Capabilities Principles of operation Examples
General concept and defining characteristics of AFM Dina Kudasheva Advisor: Prof. Mary K. Cowman Overview Introduction History of the SPM invention Technical Capabilities Principles of operation Examples
Supporting Information
 Supporting Information Analysis Method for Quantifying the Morphology of Nanotube Networks Dusan Vobornik*, Shan Zou and Gregory P. Lopinski Measurement Science and Standards, National Research Council
Supporting Information Analysis Method for Quantifying the Morphology of Nanotube Networks Dusan Vobornik*, Shan Zou and Gregory P. Lopinski Measurement Science and Standards, National Research Council
Nanosphere Lithography
 Nanosphere Lithography Derec Ciafre 1, Lingyun Miao 2, and Keita Oka 1 1 Institute of Optics / 2 ECE Dept. University of Rochester Abstract Nanosphere Lithography is quickly emerging as an efficient, low
Nanosphere Lithography Derec Ciafre 1, Lingyun Miao 2, and Keita Oka 1 1 Institute of Optics / 2 ECE Dept. University of Rochester Abstract Nanosphere Lithography is quickly emerging as an efficient, low
Micro-Rheology Measurements with the NanoTracker
 Micro-Rheology Measurements with the NanoTracker JPK s NanoTracker optical tweezers system is a versatile high resolution force measurement tool. It is based on the principle of optical trapping and uses
Micro-Rheology Measurements with the NanoTracker JPK s NanoTracker optical tweezers system is a versatile high resolution force measurement tool. It is based on the principle of optical trapping and uses
A Hydrophilic/Hydrophobic Janus Inverse-Opal
 Supporting information A Hydrophilic/Hydrophobic Janus Inverse-Opal Actuator via Gradient Infiltration Dajie Zhang #, Jie Liu //#, Bo Chen *, Yong Zhao, Jingxia Wang * //, Tomiki Ikeda, Lei Jiang //. CAS
Supporting information A Hydrophilic/Hydrophobic Janus Inverse-Opal Actuator via Gradient Infiltration Dajie Zhang #, Jie Liu //#, Bo Chen *, Yong Zhao, Jingxia Wang * //, Tomiki Ikeda, Lei Jiang //. CAS
MECHANICAL TESTS ON EPOXY RESIN NANOSCALE MODULUS MEASUREMENT AND LONG TERM CREEP BEHAVIOR
 MECHANICAL TESTS ON EPOXY RESIN NANOSCALE MODULUS MEASUREMENT AND LONG TERM CREEP BEHAVIOR By CHANGHUA LIU A DISSERTATION PRESENTED TO THE GRADUATE SCHOOL OF THE UNIVERSITY OF FLORIDA IN PARTIAL FULFILLMENT
MECHANICAL TESTS ON EPOXY RESIN NANOSCALE MODULUS MEASUREMENT AND LONG TERM CREEP BEHAVIOR By CHANGHUA LIU A DISSERTATION PRESENTED TO THE GRADUATE SCHOOL OF THE UNIVERSITY OF FLORIDA IN PARTIAL FULFILLMENT
Application of atomic force spectroscopy (AFS) to studies of adhesion phenomena: a review
 J. Adhesion Sci. Technol., Vol. 19, No. 3 5, pp. 365 405 (2005) VSP 2005. Also available online - www.vsppub.com Application of atomic force spectroscopy (AFS) to studies of adhesion phenomena: a review
J. Adhesion Sci. Technol., Vol. 19, No. 3 5, pp. 365 405 (2005) VSP 2005. Also available online - www.vsppub.com Application of atomic force spectroscopy (AFS) to studies of adhesion phenomena: a review
