RESEARCH HIGHLIGHTS. Phase Transition Enhanced Thermoelectrics David Brown
|
|
|
- Shauna Bryant
- 5 years ago
- Views:
Transcription
1 RESEARCH HIGHLIGHTS From the Resnick Sustainability Institute Graduate Research Fellows at the California Institute of Technology Phase Transition Enhanced Thermoelectrics
2 Global Significance The United States produces 28 Terrawatt-hours of energy every year, of which 58%, or 16 Terrawatthours, is lost. Much of this lost is in the form of waste-heat in the automotive sector. Technology that can capture part of that waste heat and convert it into electricity could significantly mitigate this problem, without interfering with the normal operation of a car s engine. Thermoelectrics are a physically compact and robust means of converting heat into electricity. They have been successfully demonstrated as devices in both proof-of-concept and niche applications such as the Mars Curiosity Rover. However, they are currently not efficient enough for grid-scale integration. Potential Impact In order to charge the battery of a car, extra fuel is used to run an alternator. Thermoelectrics could displace the car alternator by transforming waste heat into electricity to charge the battery. This would reduce fuel consumption by 5-10%. If successful, this project would give the same energy and carbon impact as removing 2.5 million cars from the road. This project assesses a new class of thermoelectric materials, mixed ion-electron conductors. It examines a new method for thermoelectric enhancement that utilizes the relationship between structural phase transition and electron transport. A typical thermoelectric device and a common application.
3 Project Summary We have developed a new thermoelectric material, Cu 2 Se, that shows enhanced efficiency near its structural phase transition temperature. At this temperature, the Cu+ ions in Cu 2 Se disorder and the ion conductivity increases. The entropy changes rapidly and repeatably during this transformation, which we believe affects the electrical transport boosting efficiency and enhancing the thermoelectric effect. Via material engineering, including electrochemical investigations, this project aims to understand and engineer this enhanced thermoelectric effect. This project crosses the disciplines of electrochemistry, thermoelectricity, solid state chemistry, and condensed matter physics. In this diagram, the blue spheres represent selenium atoms forming a crystal lattice. The orange regions in between the atoms represent the copper atoms that flow through the crystal structure like a liquid. This liquid-like behavior is what gives the selenium-copper material its unique thermoelectric properties. Continuous transformation of the XRD spectrum of Cu 2 Se on slow heating (1K/min). The continuous disappearance and transformations of peaks are indicative of a 2nd order transformation of the structure. The same is observed under slow cooling (not shown), indicating the gradual transformation is not due to kinetic limitations.
4 The Science The thermoelectric effect is characterized by a dimensionless figure-of-merit, zt, which is proportional to the square of the entropy transported by each electron charge (the thermopower), while inversely proportional to the thermal conductivity. The thermal conductivity includes a contribution from lattice vibrations which are loss terms. Upon approaching the second order phase transition of Cu 2 Se, the thermopower increases dramatically, resulting in an increase in zt thermoelectric conversion efficiency. We believe this to be the result of a coupling between the structural transition entropy and the transport of electrons, the mechanism of which we believe to be the coupling of transport of Cu+ ions and electrons. Dramatic increase in thermoelectric efficiency near the phase transition in Cu 2 Se (blue circles) and Cu 1.97 Ag.03 Se (green squares). In a continuous phase transition, peaks that break the symmetry of the higher temperature phase (top) decrease smoothly to zero at the critical temperature T c The integrated intensity corrected for the background intensity of the Cu 2 Se XRD peak located at 26 degrees is plotted here. In this transition region fluctuations lead to correlations at a critical length scale, which changes with temperature, and also to an increase in entropy. Ordering is represented by green squares and disordering by blue rectangles. The thermoelectric figure of merit, zt (bottom), doubles in the critical region below the phase transition at 410K. The doubling in zt above 390K in Cu 2 Se is primarily due to the increase in measured thermopower (α) compared to that expected from a rigid band model (α ρ) and the measured Hall carrier concentration.
5 The Science Thermopower (α) peak in Cu 2 Se (blue circles) and Cu 1.97 Ag.03 Se (green squares). The increase in thermopower, shown with a critical exponent fit, is clearly associated with the continuous phase transition. Between 390K and 410K, there is a significant increase in the measured thermopower above that predicted by the change in Hall carrier concentration (dotted line). In this region, the entropy of the phase transition also increases the thermopower. Key Results & Future Steps We have demonstrated an anomalous 100% increase in the zt of Cu 2 Se at 406K and have shown that it cannot be simply explained through its band structure. We have shown an even larger increase in the Ag.03 Cu 1.97 Se and a highly competitive peak zt of 1.0 at 401K. Our work has shown the 2nd order nature of the phase transition in Cu 2 Se. We intend to further our study of Ag-doped Cu 2 Se in the hopes of obtaining a record zt at 400K of 1.5. We are building a combination thermoelectricity/electrochemistry apparatus that will allow us to directly measure the coupling between Cu+ transport and electron transport, thereby providing experimental proof of our proposed mechanism. It will also perform Coloumb titration, thereby allowing rapid measurement of a wide range of compositions.
6 RESEARCH HIGHLIGHTS From the Resnick Sustainability Institute Graduate Research Fellows at the California Institute of Technology Phase Transition Enhanced Thermoelectrics Global Significance The United States produces 28 Terrawatt-hours of energy every year, of which 58%, or 16 Terrawatthours, is lost. Much of this lost is in the form of waste-heat in the automotive sector. Technology that can capture part of that waste heat and convert it into electricity could significantly mitigate this problem, without interfering with the normal operation of a car s engine. Thermoelectrics are a physically compact and robust means of converting heat into electricity. They have been successfully demonstrated as devices in both proof-of-concept and niche applications such as the Mars Curiosity Rover. However, they are currently not efficient enough for grid-scale integration. Project Summary We have developed a new thermoelectric material, Cu 2 Se, that shows enhanced efficiency near its structural phase transition temperature. At this temperature, the Cu+ ions in Cu 2 Se disorder and the ion conductivity increases. The entropy changes rapidly and repeatably during this transformation, which we believe affects the electrical transport boosting efficiency and enhancing the thermoelectric effect. Via material engineering, including electrochemical investigations, this project aims to understand and engineer this enhanced thermoelectric effect. The Science The thermoelectric effect is characterized by a dimensionless figure-of-merit, zt, which is proportional to the square of the entropy transported by each electron charge (the thermopower), while inversely proportional to the thermal conductivity. The thermal conductivity includes a contribution from lattice vibrations which are loss terms. Upon approaching the second order phase transition of Cu 2 Se, the thermopower increases dramatically, resulting in an increase in zt thermoelectric conversion efficiency. We believe this to be the result of a coupling between the structural transition entropy and the transport of electrons, the mechanism of which we believe to be the coupling of transport of Cu+ ions and electrons. Key Results We have demonstrated an anomalous 100% increase in the zt of Cu 2 Se at 406K and have shown that it cannot be simply explained through its band structure. We have shown an even larger increase in the Ag.03 Cu 1.97 Se and a highly competitive peak zt of 1.0 at 401K. Our work has shown the 2nd order nature of the phase transition in Cu 2 Se. We intend to further our study of Ag-doped Cu 2 Se in the hopes of obtaining a record zt at 400K of 1.5. We are building a combination thermoelectricity/ electrochemistry apparatus that will allow us to directly measure the coupling between Cu+ transport and electron transport, thereby providing experimental proof of our proposed mechanism. It will also perform Coloumb titration, thereby allowing rapid measurement of a wide range of compositions.
HARVESTING HEAT TO CREATE ELECTRICITY: A NEW WORLD RECORD
 HARVESTING HEAT TO CREATE ELECTRICITY: A NEW WORLD RECORD Approximately 90% of world s electricity is generated in turbines moved by hot steam, which, unfortunately, operate only at 30 to 40 percent efficiency.
HARVESTING HEAT TO CREATE ELECTRICITY: A NEW WORLD RECORD Approximately 90% of world s electricity is generated in turbines moved by hot steam, which, unfortunately, operate only at 30 to 40 percent efficiency.
Thermoelectric effect
 Thermoelectric effect See Mizutani the temperature gradient can also induce an electrical current. linearized Boltzmann transport equation in combination with the relaxation time approximation. Relaxation
Thermoelectric effect See Mizutani the temperature gradient can also induce an electrical current. linearized Boltzmann transport equation in combination with the relaxation time approximation. Relaxation
SUPPLEMENTARY INFORMATION
 Supplementary Methods Materials Synthesis The In 4 Se 3-δ crystal ingots were grown by the Bridgeman method. The In and Se elements were placed in an evacuated quartz ampoule with an excess of In (5-10
Supplementary Methods Materials Synthesis The In 4 Se 3-δ crystal ingots were grown by the Bridgeman method. The In and Se elements were placed in an evacuated quartz ampoule with an excess of In (5-10
Phases of Na x CoO 2
 Phases of Na x CoO 2 by Aakash Pushp (pushp@uiuc.edu) Abstract This paper deals with the various phases of Na x CoO 2 ranging from charge ordered insulator to Curie-Weiss metal to superconductor as the
Phases of Na x CoO 2 by Aakash Pushp (pushp@uiuc.edu) Abstract This paper deals with the various phases of Na x CoO 2 ranging from charge ordered insulator to Curie-Weiss metal to superconductor as the
Transport properties of composition tuned - and -Eu 8 Ga 16 x Ge 30+x
 Transport properties of composition tuned - and -Eu 8 Ga 16 x Ge 30+x A. Bentien, V. Pacheco,* S. Paschen, Yu. Grin, and F. Steglich Max Planck Institute for Chemical Physics of Solids, Nöthnitzer Str.
Transport properties of composition tuned - and -Eu 8 Ga 16 x Ge 30+x A. Bentien, V. Pacheco,* S. Paschen, Yu. Grin, and F. Steglich Max Planck Institute for Chemical Physics of Solids, Nöthnitzer Str.
Chapter 6. Transport in Ag 2 Se
 86 Chapter 6 Transport in Ag 2 Se This chapter is principally about the thermoelectric performance of Ag 2 Se near its phase transition. In the sample studied here the Hall carrier concentration does not
86 Chapter 6 Transport in Ag 2 Se This chapter is principally about the thermoelectric performance of Ag 2 Se near its phase transition. In the sample studied here the Hall carrier concentration does not
Chapter 3 Electrochemical methods of Analysis-Potentiometry
 Chapter 3 Electrochemical methods of Analysis-Potentiometry Electroanalytical chemistry Contents Introduction Galvanic and electrolytic cells Salt bridge Electrode potential and cell potential Indicator
Chapter 3 Electrochemical methods of Analysis-Potentiometry Electroanalytical chemistry Contents Introduction Galvanic and electrolytic cells Salt bridge Electrode potential and cell potential Indicator
RESEARCH HIGHLIGHTS. Computationally-guided Design of Polymer Electrolytes
 RESEARCH HIGHLIGHTS From the Resnick Sustainability Institute Graduate Research Fellows at the California Institute of Technology Computationally-guided Design of Polymer Electrolytes Global Significance
RESEARCH HIGHLIGHTS From the Resnick Sustainability Institute Graduate Research Fellows at the California Institute of Technology Computationally-guided Design of Polymer Electrolytes Global Significance
Work by Friction. A box slides 10 m across a surface. A frictional force of 20 N is acting on the box.
 Work by Friction A box slides 10 m across a surface. A frictional force of 20 N is acting on the box. What is the work done by friction? What happened to this energy? Work by Friction A box slides 10 m
Work by Friction A box slides 10 m across a surface. A frictional force of 20 N is acting on the box. What is the work done by friction? What happened to this energy? Work by Friction A box slides 10 m
Thermoelectric Energy Harvesting with Carbon Nanotube Systems
 Thermoelectric Energy Harvesting with Carbon Nanotube Systems Presented by Thomas C. Van Vechten, Ph.D. At the New England Nanomanufacturing Summit at UMass Lowell, June 2010 1 Outline Carbon Nanotubes
Thermoelectric Energy Harvesting with Carbon Nanotube Systems Presented by Thomas C. Van Vechten, Ph.D. At the New England Nanomanufacturing Summit at UMass Lowell, June 2010 1 Outline Carbon Nanotubes
CENTER FOR NONLINEAR AND COMPLEX SYSTEMS. Como - Italy
 CENTER FOR NONLINEAR AND COMPLEX SYSTEMS Como - Italy Providing a sustainable supply of energy to the world s population will become a major societal problem for the 21 st century as fossil fuel supplies
CENTER FOR NONLINEAR AND COMPLEX SYSTEMS Como - Italy Providing a sustainable supply of energy to the world s population will become a major societal problem for the 21 st century as fossil fuel supplies
Chapter 5. Energy Flow in the Life of a Cell
 Chapter 5 Energy Flow in the Life of a Cell Including some materials from lectures by Gregory Ahearn University of North Florida Ammended by John Crocker Copyright 2009 Pearson Education, Inc.. Review
Chapter 5 Energy Flow in the Life of a Cell Including some materials from lectures by Gregory Ahearn University of North Florida Ammended by John Crocker Copyright 2009 Pearson Education, Inc.. Review
Chapter 1. Introduction. 1.1 Summary of Introduction. 1.2 Motivation of Thermoelectric Research
 1 Chapter 1 Introduction 1.1 Summary of Introduction In this chapter I will introduce the motivation and basic concepts that underpin this thesis. In section 1.2 I will discuss the motivations for thermoelectric
1 Chapter 1 Introduction 1.1 Summary of Introduction In this chapter I will introduce the motivation and basic concepts that underpin this thesis. In section 1.2 I will discuss the motivations for thermoelectric
Instrumental Chemical Analysis. Dr. Abdul Muttaleb Jaber Professor Faculty of Pharmacy Philadelphia University Fall 2012/2013
 0510212 Instrumental Chemical Analysis Dr. Abdul Muttaleb Jaber Professor Faculty of Pharmacy Philadelphia University Fall 2012/2013 Chapter 1 Electroanalytical Methods Electroanalytical Chemistry Electroanalytical
0510212 Instrumental Chemical Analysis Dr. Abdul Muttaleb Jaber Professor Faculty of Pharmacy Philadelphia University Fall 2012/2013 Chapter 1 Electroanalytical Methods Electroanalytical Chemistry Electroanalytical
RESEARCH HIGHLIGHTS. Polymer Photonic Crystals by Self-Assembly Raymond Weitekamp
 RESEARCH HIGHLIGHTS From the Resnick Sustainability Institute Graduate Research Fellows at the California Institute of Technology Polymer Photonic Crystals by Self-Assembly Global Significance Urbanization
RESEARCH HIGHLIGHTS From the Resnick Sustainability Institute Graduate Research Fellows at the California Institute of Technology Polymer Photonic Crystals by Self-Assembly Global Significance Urbanization
SUPPLEMENTARY INFORMATION
 SI - N. Poccia et al. Evolution and Control of Oxygen Order in a Cuprate Superconductor SUPPLEMENTARY INFORMATION 1. X-ray diffraction experiments The diffraction data were collected on the x-ray diffraction
SI - N. Poccia et al. Evolution and Control of Oxygen Order in a Cuprate Superconductor SUPPLEMENTARY INFORMATION 1. X-ray diffraction experiments The diffraction data were collected on the x-ray diffraction
Sensing, Computing, Actuating
 Sensing, Computing, ctuating Sander Stuijk (s.stuijk@tue.nl) Department of Electrical Engineering Electronic Systems 2 THERMOELECTRIC EFFECT (Chapter 5.11) 3 Thermocouple cylinder head temperature (thermocouple)
Sensing, Computing, ctuating Sander Stuijk (s.stuijk@tue.nl) Department of Electrical Engineering Electronic Systems 2 THERMOELECTRIC EFFECT (Chapter 5.11) 3 Thermocouple cylinder head temperature (thermocouple)
Matter is made of atoms and molecules
 Name Per Talking to the Text Atoms and Molecules pt.2 Author Says (important ideas, vocabulary) Matter is made of atoms and molecules We have already used the term atom and molecule a couple of times.
Name Per Talking to the Text Atoms and Molecules pt.2 Author Says (important ideas, vocabulary) Matter is made of atoms and molecules We have already used the term atom and molecule a couple of times.
Thermoelectric materials for energy harvesting new modelling tools with predictive power
 Thermoelectric materials for energy harvesting new modelling tools with predictive power Ole Martin Løvvik 1,2 1 SINTEF Materials Physics, Norway 2 University of Oslo, Norway Thermoelectric generators
Thermoelectric materials for energy harvesting new modelling tools with predictive power Ole Martin Løvvik 1,2 1 SINTEF Materials Physics, Norway 2 University of Oslo, Norway Thermoelectric generators
DETECTORS. I. Charged Particle Detectors
 DETECTORS I. Charged Particle Detectors A. Scintillators B. Gas Detectors 1. Ionization Chambers 2. Proportional Counters 3. Avalanche detectors 4. Geiger-Muller counters 5. Spark detectors C. Solid State
DETECTORS I. Charged Particle Detectors A. Scintillators B. Gas Detectors 1. Ionization Chambers 2. Proportional Counters 3. Avalanche detectors 4. Geiger-Muller counters 5. Spark detectors C. Solid State
A Primer On Phonon Glass Electrical Crystal Material
 A Primer On Phonon Glass Electrical Crystal Material Stefan Bringuier Materials Science and Engineering, University of Arizona stefanb@email.arizona.edu http://u.arizona.edu/~stefanb May 7, 2012 Abstract
A Primer On Phonon Glass Electrical Crystal Material Stefan Bringuier Materials Science and Engineering, University of Arizona stefanb@email.arizona.edu http://u.arizona.edu/~stefanb May 7, 2012 Abstract
EE 42/100 Lecture 3: Circuit Elements, Resistive Circuits. Rev D 1/22/2012 (4:19PM) Prof. Ali M. Niknejad
 A. M. Niknejad University of California, Berkeley EE 100 / 42 Lecture 3 p. 1/22 EE 42/100 Lecture 3: Circuit Elements, Resistive Circuits ELECTRONICS Rev D 1/22/2012 (4:19PM) Prof. Ali M. Niknejad University
A. M. Niknejad University of California, Berkeley EE 100 / 42 Lecture 3 p. 1/22 EE 42/100 Lecture 3: Circuit Elements, Resistive Circuits ELECTRONICS Rev D 1/22/2012 (4:19PM) Prof. Ali M. Niknejad University
Solid State electrochemistry
 Solid State electrochemistry edited by Peter G. Bruce Department of Chemistry, University of St Andrews, Scotland IH CAMBRIDGE ^pf UNIVERSITY PRESS 1 1.1 1.2 1.3 1.4 1.5 1.6 Preface Introduction P. G.
Solid State electrochemistry edited by Peter G. Bruce Department of Chemistry, University of St Andrews, Scotland IH CAMBRIDGE ^pf UNIVERSITY PRESS 1 1.1 1.2 1.3 1.4 1.5 1.6 Preface Introduction P. G.
Thermoelectric transport of ultracold fermions : theory
 Thermoelectric transport of ultracold fermions : theory Collège de France, December 2013 Theory : Ch. Grenier C. Kollath A. Georges Experiments : J.-P. Brantut J. Meineke D. Stadler S. Krinner T. Esslinger
Thermoelectric transport of ultracold fermions : theory Collège de France, December 2013 Theory : Ch. Grenier C. Kollath A. Georges Experiments : J.-P. Brantut J. Meineke D. Stadler S. Krinner T. Esslinger
efficiency can be to Carnot primarily through the thermoelectric figure of merit, z, defined by
 USING THE COMPATIBILITY FACTOR TO DESIGN HIGH EFFICIENCY SEGMENTED THERMOELECTRIC GENERATORS G. Jeffrey Snyder*, and T. Caillat Jet Propulsion Laboratory/California Institute of Technology 4800, Oak Grove
USING THE COMPATIBILITY FACTOR TO DESIGN HIGH EFFICIENCY SEGMENTED THERMOELECTRIC GENERATORS G. Jeffrey Snyder*, and T. Caillat Jet Propulsion Laboratory/California Institute of Technology 4800, Oak Grove
Chapter 7. Transport in Cu 2 Se and Cu 1.97 Ag 0.03 Se. 7.1 Entropy and Charge Transport
 101 Chapter 7 Transport in Cu 2 Se and Cu 1.97 Ag 0.03 Se In the previous chapter I introduced super-ionic materials and band structure modeling of thermoelectrics. In that context I introduced the thermoelectric
101 Chapter 7 Transport in Cu 2 Se and Cu 1.97 Ag 0.03 Se In the previous chapter I introduced super-ionic materials and band structure modeling of thermoelectrics. In that context I introduced the thermoelectric
DC Circuits I. Physics 2415 Lecture 12. Michael Fowler, UVa
 DC Circuits I Physics 2415 Lecture 12 Michael Fowler, UVa Today s Topics Mention of AC Semiconductors and superconductors Battery emf, internal resistance Series and parallel resistances Kirchhoff s rules
DC Circuits I Physics 2415 Lecture 12 Michael Fowler, UVa Today s Topics Mention of AC Semiconductors and superconductors Battery emf, internal resistance Series and parallel resistances Kirchhoff s rules
Organic Electronic Devices
 Organic Electronic Devices Week 5: Organic Light-Emitting Devices and Emerging Technologies Lecture 5.5: Course Review and Summary Bryan W. Boudouris Chemical Engineering Purdue University 1 Understanding
Organic Electronic Devices Week 5: Organic Light-Emitting Devices and Emerging Technologies Lecture 5.5: Course Review and Summary Bryan W. Boudouris Chemical Engineering Purdue University 1 Understanding
In an electric field R and magnetic field B, the force on an electron (charge e) is given by:
 Lecture 17 Electric conduction Electrons motion in magnetic field Electrons thermal conductivity Brief review In solid state physics, we do not think about electrons zipping around randomly in real space.
Lecture 17 Electric conduction Electrons motion in magnetic field Electrons thermal conductivity Brief review In solid state physics, we do not think about electrons zipping around randomly in real space.
Physics of disordered materials. Gunnar A. Niklasson Solid State Physics Department of Engineering Sciences Uppsala University
 Physics of disordered materials Gunnar A. Niklasson Solid State Physics Department of Engineering Sciences Uppsala University Course plan Familiarity with the basic description of disordered structures
Physics of disordered materials Gunnar A. Niklasson Solid State Physics Department of Engineering Sciences Uppsala University Course plan Familiarity with the basic description of disordered structures
HALL EFFECT IN SEMICONDUCTORS
 Warsaw University of Technology Faculty of Physics Physics Laboratory I P Andrzej Kubiaczyk 30 HALL EFFECT IN SEMICONDUCTORS 1. ackground 1.1. Electron motion in electric and magnetic fields A particle
Warsaw University of Technology Faculty of Physics Physics Laboratory I P Andrzej Kubiaczyk 30 HALL EFFECT IN SEMICONDUCTORS 1. ackground 1.1. Electron motion in electric and magnetic fields A particle
Coimisiún na Scrúduithe Stáit State Examinations Commission
 2014. M35 Coimisiún na Scrúduithe Stáit State Examinations Commission LEAVING CERTIFICATE EXAMINATION, 2014 PHYSICS ORDINARY LEVEL MONDAY, 16 JUNE MORNING, 9:30 TO 12:30 Answer three questions from Section
2014. M35 Coimisiún na Scrúduithe Stáit State Examinations Commission LEAVING CERTIFICATE EXAMINATION, 2014 PHYSICS ORDINARY LEVEL MONDAY, 16 JUNE MORNING, 9:30 TO 12:30 Answer three questions from Section
Self-Check. change in pressure. 6. I can explain how the volume of a gas is changed by a change in temperature S. Coates
 Self-Check YES NO 1. I can describe how atoms move in a solid, liquid, and gas 2. I can describe the speed/energy of the atoms in a solid, liquid, and gas. 3. I can explain how the distance between atoms
Self-Check YES NO 1. I can describe how atoms move in a solid, liquid, and gas 2. I can describe the speed/energy of the atoms in a solid, liquid, and gas. 3. I can explain how the distance between atoms
Heat: the transfer of thermal energy from one substance to another. Electrons: the small particles inside an atom that have negative charge.
 GRADE 5 0 Energy: the ability to cause change OR to do work Kinetic energy: energy of motion Potential energy: energy of position (stored energy) Thermal energy: the energy that moves particles of matter
GRADE 5 0 Energy: the ability to cause change OR to do work Kinetic energy: energy of motion Potential energy: energy of position (stored energy) Thermal energy: the energy that moves particles of matter
Chapter 26 Current and Resistance
 Chapter 26 Current and Resistance Electric Current Although an electric current is a stream of moving charges, not all moving charges constitute an electric current. If there is to be an electric current
Chapter 26 Current and Resistance Electric Current Although an electric current is a stream of moving charges, not all moving charges constitute an electric current. If there is to be an electric current
Name Date Class THE NATURE OF GASES
 13.1 THE NATURE OF GASES Section Review Objectives Describe the assumptions of the kinetic theory as it applies to gases Interpret gas pressure in terms of kinetic theory Define the relationship between
13.1 THE NATURE OF GASES Section Review Objectives Describe the assumptions of the kinetic theory as it applies to gases Interpret gas pressure in terms of kinetic theory Define the relationship between
Chapter 10 States of Matter
 Chapter 10 States of Matter 1 Section 10.1 The Nature of Gases Objectives: Describe the assumptions of the kinetic theory as it applies to gases. Interpret gas pressure in terms of kinetic theory. Define
Chapter 10 States of Matter 1 Section 10.1 The Nature of Gases Objectives: Describe the assumptions of the kinetic theory as it applies to gases. Interpret gas pressure in terms of kinetic theory. Define
FREQUENTLY ASKED QUESTIONS February 21, 2017
 FREQUENTLY ASKED QUESTIONS February 21, 2017 Content Questions How do you place a single arsenic atom with the ratio 1 in 100 million? Sounds difficult to get evenly spread throughout. Yes, techniques
FREQUENTLY ASKED QUESTIONS February 21, 2017 Content Questions How do you place a single arsenic atom with the ratio 1 in 100 million? Sounds difficult to get evenly spread throughout. Yes, techniques
Class 29: Reciprocal Space 3: Ewald sphere, Simple Cubic, FCC and BCC in Reciprocal Space
 Class 29: Reciprocal Space 3: Ewald sphere, Simple Cubic, FCC and BCC in Reciprocal Space We have seen that diffraction occurs when, in reciprocal space, Let us now plot this information. Let us designate
Class 29: Reciprocal Space 3: Ewald sphere, Simple Cubic, FCC and BCC in Reciprocal Space We have seen that diffraction occurs when, in reciprocal space, Let us now plot this information. Let us designate
Indiana University, January T. Witten, University of Chicago
 Indiana University, January 2007 T. Witten, University of Chicago Force propagation in a simple solid: two pictures Add circular beads to a container one by one How does an added force reach the ground?
Indiana University, January 2007 T. Witten, University of Chicago Force propagation in a simple solid: two pictures Add circular beads to a container one by one How does an added force reach the ground?
Exercises Evaporation (page 451) 23.2 Condensation (pages )
 Exercises 23.1 Evaporation (page 451) 1. The four forms in which matter exists solid, liquid, gas, and plasma are called. 2. Water that is left out in an open container will eventually. 3. Is the following
Exercises 23.1 Evaporation (page 451) 1. The four forms in which matter exists solid, liquid, gas, and plasma are called. 2. Water that is left out in an open container will eventually. 3. Is the following
From Last Time Important new Quantum Mechanical Concepts. Atoms and Molecules. Today. Symmetry. Simple molecules.
 Today From Last Time Important new Quantum Mechanical Concepts Indistinguishability: Symmetries of the wavefunction: Symmetric and Antisymmetric Pauli exclusion principle: only one fermion per state Spin
Today From Last Time Important new Quantum Mechanical Concepts Indistinguishability: Symmetries of the wavefunction: Symmetric and Antisymmetric Pauli exclusion principle: only one fermion per state Spin
Physics 1302W.400 Lecture 21 Introductory Physics for Scientists and Engineering II
 Physics 1302W.400 Lecture 21 Introductory Physics for Scientists and Engineering II In today s lecture, we will learn to: Calculate the resistance of a conductor depending on the material and shape Apply
Physics 1302W.400 Lecture 21 Introductory Physics for Scientists and Engineering II In today s lecture, we will learn to: Calculate the resistance of a conductor depending on the material and shape Apply
CHAPTER GENERAL CONCEPTS READING from text HOMEWORK from text OTHER ASSIGNMENTS LABORATORIES. R1: Sections 1,2 Set 1: CR 15; PP 1
 MONTPELIER HIGH SCHOOL teacher: Toni Ceckler / Room 220, phone: 225-8182, email: toni@mpsvt.org CHEMISTRY 2018-2019 GENERAL SYLLABUS Text: Chemistry: Connections to Our Changing World (Prentice Hall, 2000)
MONTPELIER HIGH SCHOOL teacher: Toni Ceckler / Room 220, phone: 225-8182, email: toni@mpsvt.org CHEMISTRY 2018-2019 GENERAL SYLLABUS Text: Chemistry: Connections to Our Changing World (Prentice Hall, 2000)
AQA (Trilogy) Combined Science GCSE Unit 6.3 Particle Model of Matter
 AQA (Trilogy) Combined Science GCSE Unit 6.3 Particle Model of Matter Test (Levels 4 9) Time allowed: 50 minutes Question Links to Student Progress Sheet Score Total Marks Available Score Estimated Grade
AQA (Trilogy) Combined Science GCSE Unit 6.3 Particle Model of Matter Test (Levels 4 9) Time allowed: 50 minutes Question Links to Student Progress Sheet Score Total Marks Available Score Estimated Grade
AS COMPETITION PAPER 2007
 AS COMPETITION PAPER 2007 Total Mark/50 Name School Town & County Time Allowed: One hour Attempt as many questions as you can. Write your answers on this question paper. Marks allocated for each question
AS COMPETITION PAPER 2007 Total Mark/50 Name School Town & County Time Allowed: One hour Attempt as many questions as you can. Write your answers on this question paper. Marks allocated for each question
Scaling analysis of snapshot spectra in the world-line quantum Monte Carlo for the transverse-field Ising chain
 TNSAA 2018-2019 Dec. 3-6, 2018, Kobe, Japan Scaling analysis of snapshot spectra in the world-line quantum Monte Carlo for the transverse-field Ising chain Kouichi Seki, Kouichi Okunishi Niigata University,
TNSAA 2018-2019 Dec. 3-6, 2018, Kobe, Japan Scaling analysis of snapshot spectra in the world-line quantum Monte Carlo for the transverse-field Ising chain Kouichi Seki, Kouichi Okunishi Niigata University,
Acid/Base Reactions & Electrochemistry
 Adult Basic Education Science Acid/Base Reactions & Electrochemistry Prerequisite: Chemistry 3102B Credit Value: 1 Chemistry Concentration Chemistry 1102 Chemistry 2102A Chemistry 2102B Chemistry 2102C
Adult Basic Education Science Acid/Base Reactions & Electrochemistry Prerequisite: Chemistry 3102B Credit Value: 1 Chemistry Concentration Chemistry 1102 Chemistry 2102A Chemistry 2102B Chemistry 2102C
Thermoelectric materials. Hyo-Jeong Moon
 Thermoelectric materials Hyo-Jeong Moon Electrical conductivity Thermoelectric materials Ratio of current density to electric field, when no temperature gradient is present. Thermal conductivity Ratio
Thermoelectric materials Hyo-Jeong Moon Electrical conductivity Thermoelectric materials Ratio of current density to electric field, when no temperature gradient is present. Thermal conductivity Ratio
PN Junction
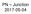 P Junction 2017-05-04 Definition Power Electronics = semiconductor switches are used Analogue amplifier = high power loss 250 200 u x 150 100 u Udc i 50 0 0 50 100 150 200 250 300 350 400 i,u dc i,u u
P Junction 2017-05-04 Definition Power Electronics = semiconductor switches are used Analogue amplifier = high power loss 250 200 u x 150 100 u Udc i 50 0 0 50 100 150 200 250 300 350 400 i,u dc i,u u
RESEARCH HIGHLIGHTS. Model Complexes for Studying Heterometallic Effects Relevant to Fuel Cell Chemistry
 RESEARCH HIGHLIGHTS From the Resnick Sustainability Institute Graduate Research Fellows at the California Institute of Technology Model Complexes for Studying Heterometallic Effects Relevant to Fuel Cell
RESEARCH HIGHLIGHTS From the Resnick Sustainability Institute Graduate Research Fellows at the California Institute of Technology Model Complexes for Studying Heterometallic Effects Relevant to Fuel Cell
Classification of Solids
 Classification of Solids Classification by conductivity, which is related to the band structure: (Filled bands are shown dark; D(E) = Density of states) Class Electron Density Density of States D(E) Examples
Classification of Solids Classification by conductivity, which is related to the band structure: (Filled bands are shown dark; D(E) = Density of states) Class Electron Density Density of States D(E) Examples
4 Energy and Rates of Chemical Reactions
 CHAPTER 14 4 and Rates of Chemical Reactions SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: How is energy involved in a chemical reaction?
CHAPTER 14 4 and Rates of Chemical Reactions SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: How is energy involved in a chemical reaction?
Gauged Cosmic Strings and Superconductor Vortices
 Gauged Cosmic Strings and Superconductor Vortices Hindmarsh&Rajantie, PRL2000 Rajantie, IJMPA2001 Kibble&Rajantie, PRB2003 Donaire,Kibble&Rajantie, cond-mat/0409172 Donaire&Rajantie, hep-ph/0508272 3 September
Gauged Cosmic Strings and Superconductor Vortices Hindmarsh&Rajantie, PRL2000 Rajantie, IJMPA2001 Kibble&Rajantie, PRB2003 Donaire,Kibble&Rajantie, cond-mat/0409172 Donaire&Rajantie, hep-ph/0508272 3 September
Module 5: Rise and Fall of the Clockwork Universe. You should be able to demonstrate and show your understanding of:
 OCR B Physics H557 Module 5: Rise and Fall of the Clockwork Universe You should be able to demonstrate and show your understanding of: 5.2: Matter Particle model: A gas consists of many very small, rapidly
OCR B Physics H557 Module 5: Rise and Fall of the Clockwork Universe You should be able to demonstrate and show your understanding of: 5.2: Matter Particle model: A gas consists of many very small, rapidly
Temperature Measurement
 MECE 3320 Measurements & Instrumentation Temperature Measurement Dr. Isaac Choutapalli Department of Mechanical Engineering University of Texas Pan American Introduction Temperature is one of the most
MECE 3320 Measurements & Instrumentation Temperature Measurement Dr. Isaac Choutapalli Department of Mechanical Engineering University of Texas Pan American Introduction Temperature is one of the most
Unit 7 Kinetics and Thermodynamics
 17.1 The Flow of Energy Heat and Work Unit 7 Kinetics and Thermodynamics I. Energy Transformations A. Temperature 1. A measure of the average kinetic energy of the particles in a sample of matter B. Heat
17.1 The Flow of Energy Heat and Work Unit 7 Kinetics and Thermodynamics I. Energy Transformations A. Temperature 1. A measure of the average kinetic energy of the particles in a sample of matter B. Heat
NEUTRALIZATION TITRATION-2 TITRATION OF AN ANTACID (Exp. 4)
 Objective NEUTRALIZATION TITRATION-2 TITRATION OF AN ANTACID (Exp. 4) The aim of this experiment is to carry out titration of antacid tablets and to determine acetic acid content of vinegar. a) Titration
Objective NEUTRALIZATION TITRATION-2 TITRATION OF AN ANTACID (Exp. 4) The aim of this experiment is to carry out titration of antacid tablets and to determine acetic acid content of vinegar. a) Titration
Chapter 10. Lesson Starter. Why did you not smell the odor of the vapor immediately? Explain this event in terms of the motion of molecules.
 Preview Lesson Starter Objectives The Kinetic-Molecular Theory of Gases The Kinetic-Molecular Theory and the Nature of Gases Deviations of Real Gases from Ideal Behavior Section 1 The Kinetic-Molecular
Preview Lesson Starter Objectives The Kinetic-Molecular Theory of Gases The Kinetic-Molecular Theory and the Nature of Gases Deviations of Real Gases from Ideal Behavior Section 1 The Kinetic-Molecular
OPTIMIZATION OF AN INDUSTRIAL HEAT EXCHANGER BY THE MINIMALIZATION OF ENTROPY SVOČ FST 2017
 OPTIMIZATION OF AN INDUSTRIAL HEAT EXCHANGER BY THE MINIMALIZATION OF ENTROPY SVOČ FST 217 Bc. Richard Pisinger, University of West Bohemia, Univerzitní 8, 36 14 Pilsen Czech Republic ABSTRACT A second
OPTIMIZATION OF AN INDUSTRIAL HEAT EXCHANGER BY THE MINIMALIZATION OF ENTROPY SVOČ FST 217 Bc. Richard Pisinger, University of West Bohemia, Univerzitní 8, 36 14 Pilsen Czech Republic ABSTRACT A second
2 Changes of State KEY IDEAS READING TOOLBOX ADDING AND REMOVING ENERGY. States of Matter. As you read this section, keep these questions in mind:
 CHAPTER 3 States of Matter 2 Changes of State SECTION KEY IDEAS As you read this section, keep these questions in mind: What happens when a substance changes from one state of matter to another? What happens
CHAPTER 3 States of Matter 2 Changes of State SECTION KEY IDEAS As you read this section, keep these questions in mind: What happens when a substance changes from one state of matter to another? What happens
Molecules and Condensed Matter
 Chapter 42 Molecules and Condensed Matter PowerPoint Lectures for University Physics, Thirteenth Edition Hugh D. Young and Roger A. Freedman Lectures by Wayne Anderson Goals for Chapter 42 To understand
Chapter 42 Molecules and Condensed Matter PowerPoint Lectures for University Physics, Thirteenth Edition Hugh D. Young and Roger A. Freedman Lectures by Wayne Anderson Goals for Chapter 42 To understand
6-3 Particle model of matter Trilogy
 6-3 Particle model of matter Trilogy.0 A teacher uses a tray filled with table tennis balls to model how particles are arranged in materials, as shown in Figure Figure. Initially the balls are arranged
6-3 Particle model of matter Trilogy.0 A teacher uses a tray filled with table tennis balls to model how particles are arranged in materials, as shown in Figure Figure. Initially the balls are arranged
A MODEL BASED APPROACH TO EXHAUST THERMOELECTRICS. Quazi Hussain, David Brigham, and Clay Maranville Research & Advanced Engineering
 A MODEL BASED APPROACH TO EXHAUST HEAT RECOVERY USING THERMOELECTRICS Quazi Hussain, David Brigham, and Clay Maranville Research & Advanced Engineering Ford Motor Company Objective Investigate potential
A MODEL BASED APPROACH TO EXHAUST HEAT RECOVERY USING THERMOELECTRICS Quazi Hussain, David Brigham, and Clay Maranville Research & Advanced Engineering Ford Motor Company Objective Investigate potential
Unit 13 Review: Types of Bonding and Phase Changes (Yes, I know that the outline levels are doing stupid things partway through this document.
 Unit 13 Review: Types of Bonding and Phase Changes (Yes, I know that the outline levels are doing stupid things partway through this document. I m sorry about that, but I m not going to spend any more
Unit 13 Review: Types of Bonding and Phase Changes (Yes, I know that the outline levels are doing stupid things partway through this document. I m sorry about that, but I m not going to spend any more
Lecture 20: Semiconductor Structures Kittel Ch 17, p , extra material in the class notes
 Lecture 20: Semiconductor Structures Kittel Ch 17, p 494-503, 507-511 + extra material in the class notes MOS Structure Layer Structure metal Oxide insulator Semiconductor Semiconductor Large-gap Semiconductor
Lecture 20: Semiconductor Structures Kittel Ch 17, p 494-503, 507-511 + extra material in the class notes MOS Structure Layer Structure metal Oxide insulator Semiconductor Semiconductor Large-gap Semiconductor
1. a Draw a labelled diagram of an atom that has 9 protons and electrons and 9 neutrons.
 Topic review Using scientific language Design and construct a crossword using the following words: atom; proton; molecule; ion; lattice; shell; element; compound; bond; conductor; insulator; electrolysis;
Topic review Using scientific language Design and construct a crossword using the following words: atom; proton; molecule; ion; lattice; shell; element; compound; bond; conductor; insulator; electrolysis;
Structure of Surfaces
 Structure of Surfaces C Stepped surface Interference of two waves Bragg s law Path difference = AB+BC =2dsin ( =glancing angle) If, n =2dsin, constructive interference Ex) in a cubic lattice of unit cell
Structure of Surfaces C Stepped surface Interference of two waves Bragg s law Path difference = AB+BC =2dsin ( =glancing angle) If, n =2dsin, constructive interference Ex) in a cubic lattice of unit cell
Relaxation in Glass: Review of Thermodynamics. Lecture 11: Thermodynamics in the Glass Transition Region
 Relaxation in Glass: Review of hermodynamics Lecture 11: hermodynamics in the Glass ransition Region hermodynamic Functions 1 st Derivatives emperature Dependence of the Entropy swmartin@iastate.edu MI:
Relaxation in Glass: Review of hermodynamics Lecture 11: hermodynamics in the Glass ransition Region hermodynamic Functions 1 st Derivatives emperature Dependence of the Entropy swmartin@iastate.edu MI:
MOLECULAR DYNAMICS SIMULATION OF THERMAL CONDUCTIVITY OF NANOCRYSTALLINE COMPOSITE FILMS
 Proceedings of HT 2007 2007 ASME-JSME Thermal Engineering Summer Heat Transfer Conference July 8 12, 2007, Vancouver, British Columbia, Canada HT2007-1520 MOLECULAR DYNAMICS SIMULATION OF THERMAL CONDUCTIVITY
Proceedings of HT 2007 2007 ASME-JSME Thermal Engineering Summer Heat Transfer Conference July 8 12, 2007, Vancouver, British Columbia, Canada HT2007-1520 MOLECULAR DYNAMICS SIMULATION OF THERMAL CONDUCTIVITY
First-Hand Investigation: Modeling of Semiconductors
 perform an investigation to model the behaviour of semiconductors, including the creation of a hole or positive charge on the atom that has lost the electron and the movement of electrons and holes in
perform an investigation to model the behaviour of semiconductors, including the creation of a hole or positive charge on the atom that has lost the electron and the movement of electrons and holes in
When Done Test. Pg. 324 #1-9 Pg. 325 #1-5 Pg. 325 #1-4
 When Done Test Pg. 324 #1-9 Pg. 325 #1-5 Pg. 325 #1-4 The Nature of Oxidation- Reduction Reactions I will be able to explain how oxidation and reduction reactions involve the transfer of electrons. I will
When Done Test Pg. 324 #1-9 Pg. 325 #1-5 Pg. 325 #1-4 The Nature of Oxidation- Reduction Reactions I will be able to explain how oxidation and reduction reactions involve the transfer of electrons. I will
Conduction and Convection
 Conduction and Convection Convection Currents Definition Convection is the transfer of heat in liquids and gases. The hotter the liquid/gas the particles move faster and spread out. This means the gas/liquid
Conduction and Convection Convection Currents Definition Convection is the transfer of heat in liquids and gases. The hotter the liquid/gas the particles move faster and spread out. This means the gas/liquid
6-3 Particle model of matter Physics
 6-3 Particle model of matter Physics.0 A teacher uses a tray filled with table tennis balls to model how particles are arranged in materials, as shown in Figure Figure. Initially the balls are arranged
6-3 Particle model of matter Physics.0 A teacher uses a tray filled with table tennis balls to model how particles are arranged in materials, as shown in Figure Figure. Initially the balls are arranged
EXTENSION 6. Chapter 3 Encounters with Electricity: Electrical Energy in the Home Unit 3.2 Electric Circuits and Electric Charge
 EXTENSION 6 Chapter 3 Encounters with Electricity: Electrical Energy in the Home Unit 3.2 Electric Circuits and Electric Charge A view of the Atom and Electric Charge Figure 1 The size of the nucleus is
EXTENSION 6 Chapter 3 Encounters with Electricity: Electrical Energy in the Home Unit 3.2 Electric Circuits and Electric Charge A view of the Atom and Electric Charge Figure 1 The size of the nucleus is
Lab 12.1 Calorimetry. Name School Date
 Name School Date Lab 12.1 Calorimetry Purpose To investigate the flow of heat between two bodies due to a difference in their temperatures. To investigate the flow of heat involved in changes in phase.
Name School Date Lab 12.1 Calorimetry Purpose To investigate the flow of heat between two bodies due to a difference in their temperatures. To investigate the flow of heat involved in changes in phase.
Superinsulator: a new topological state of matter
 Superinsulator: a new topological state of matter M. Cristina Diamantini Nips laboratory, INFN and Department of Physics and Geology University of Perugia Coll: Igor Lukyanchuk, University of Picardie
Superinsulator: a new topological state of matter M. Cristina Diamantini Nips laboratory, INFN and Department of Physics and Geology University of Perugia Coll: Igor Lukyanchuk, University of Picardie
INTRODUCTION Strained Silicon Monochromator Magnesium Housing Windows for Monochromator Shutter and Collimator Fission Detector HOPG Monochromator
 Design for a Four-Blade Neutron Interferometer INTRODUCTION Strained Silicon Monochromator The neutron beam used for this interferometer is separated from the NIST reactor's main beam using a strained
Design for a Four-Blade Neutron Interferometer INTRODUCTION Strained Silicon Monochromator The neutron beam used for this interferometer is separated from the NIST reactor's main beam using a strained
Thermionic power generation at high temperatures using SiGe/ Si superlattices
 JOURNAL OF APPLIED PHYSICS 101, 053719 2007 Thermionic power generation at high temperatures using SiGe/ Si superlattices Daryoosh Vashaee a and Ali Shakouri Jack Baskin School of Engineering, University
JOURNAL OF APPLIED PHYSICS 101, 053719 2007 Thermionic power generation at high temperatures using SiGe/ Si superlattices Daryoosh Vashaee a and Ali Shakouri Jack Baskin School of Engineering, University
Nernst effect and Kondo scattering in (CeLa)Cu 2 Si 2
 Nernst effect and Kondo scattering in (CeLa)Cu 2 Si 2 Peijie Sun MPI Chemical Physics of Solids, Dresden (IOP-CAS, from April 212) Collaborators: C. Geibel, F. Steglich 1 Outline Introduction to magneto-thermoelectric
Nernst effect and Kondo scattering in (CeLa)Cu 2 Si 2 Peijie Sun MPI Chemical Physics of Solids, Dresden (IOP-CAS, from April 212) Collaborators: C. Geibel, F. Steglich 1 Outline Introduction to magneto-thermoelectric
SnSe: a remarkable new thermoelectric material
 SnSe: a remarkable new thermoelectric material A radioisotope thermoelectric generator (RTG) is an electrical generator that uses an array of thermocouples to convert the heat released by the decay of
SnSe: a remarkable new thermoelectric material A radioisotope thermoelectric generator (RTG) is an electrical generator that uses an array of thermocouples to convert the heat released by the decay of
Electric systems, energy and non dualistic logic
 Electric systems, energy and non dualistic logic Marcus Albert Reid October 2008 Basic energy conversion mechanism within electric systems 1 The energy flow within an electric circuit T.D. Lee, (Nobelist)
Electric systems, energy and non dualistic logic Marcus Albert Reid October 2008 Basic energy conversion mechanism within electric systems 1 The energy flow within an electric circuit T.D. Lee, (Nobelist)
Direct and Indirect Semiconductor
 Direct and Indirect Semiconductor Allowed values of energy can be plotted vs. the propagation constant, k. Since the periodicity of most lattices is different in various direction, the E-k diagram must
Direct and Indirect Semiconductor Allowed values of energy can be plotted vs. the propagation constant, k. Since the periodicity of most lattices is different in various direction, the E-k diagram must
Advanced Analytical Chemistry Lecture 12. Chem 4631
 Advanced Analytical Chemistry Lecture 12 Chem 4631 What is a fuel cell? An electro-chemical energy conversion device A factory that takes fuel as input and produces electricity as output. O 2 (g) H 2 (g)
Advanced Analytical Chemistry Lecture 12 Chem 4631 What is a fuel cell? An electro-chemical energy conversion device A factory that takes fuel as input and produces electricity as output. O 2 (g) H 2 (g)
The Oxford Solid State Basics
 The Oxford Solid State Basics Steven H. Simon University of Oxford OXFORD UNIVERSITY PRESS Contents 1 About Condensed Matter Physics 1 1.1 What Is Condensed Matter Physics 1 1.2 Why Do We Study Condensed
The Oxford Solid State Basics Steven H. Simon University of Oxford OXFORD UNIVERSITY PRESS Contents 1 About Condensed Matter Physics 1 1.1 What Is Condensed Matter Physics 1 1.2 Why Do We Study Condensed
Physics 127b: Statistical Mechanics. Landau Theory of Second Order Phase Transitions. Order Parameter
 Physics 127b: Statistical Mechanics Landau Theory of Second Order Phase Transitions Order Parameter Second order phase transitions occur when a new state of reduced symmetry develops continuously from
Physics 127b: Statistical Mechanics Landau Theory of Second Order Phase Transitions Order Parameter Second order phase transitions occur when a new state of reduced symmetry develops continuously from
IGCSE Double Award Extended Coordinated Science
 IGCSE Double Award Extended Coordinated Science Physics 5 - Thermal Properties of Matter Thermal Expansion You need to know thermal expansions for solids, liquids, and gases, and their applications. Thermal
IGCSE Double Award Extended Coordinated Science Physics 5 - Thermal Properties of Matter Thermal Expansion You need to know thermal expansions for solids, liquids, and gases, and their applications. Thermal
Experimental Setup for the Measurement of Low Temperature Seebeck Coefficient of Single Crystal and Bulk Materials
 Journal of Control & Instrumentation IN: 9-697 (online), IN: 347-737 (print) Volume 5, Issue www.stmjournals.com Experimental etup for the Measurement of Low Temperature eebeck Coefficient of ingle Crystal
Journal of Control & Instrumentation IN: 9-697 (online), IN: 347-737 (print) Volume 5, Issue www.stmjournals.com Experimental etup for the Measurement of Low Temperature eebeck Coefficient of ingle Crystal
Thermal Sensors and Actuators
 Thermal Sensors and Actuators Part I Fundamentals of heat transfer Heat transfer occurs where there is a temperature gradient until an equilibrium is reached. Four major mechanism Thermal conduction Natural
Thermal Sensors and Actuators Part I Fundamentals of heat transfer Heat transfer occurs where there is a temperature gradient until an equilibrium is reached. Four major mechanism Thermal conduction Natural
Semiconductor Detectors
 Semiconductor Detectors Summary of Last Lecture Band structure in Solids: Conduction band Conduction band thermal conductivity: E g > 5 ev Valence band Insulator Charge carrier in conductor: e - Charge
Semiconductor Detectors Summary of Last Lecture Band structure in Solids: Conduction band Conduction band thermal conductivity: E g > 5 ev Valence band Insulator Charge carrier in conductor: e - Charge
Clean Energy: Thermoelectrics and Photovoltaics. Akram Boukai Ph.D.
 Clean Energy: Thermoelectrics and Photovoltaics Akram Boukai Ph.D. Solar Energy Use Hydrocarbons vs. Photons Arabian Oil: 600 years Sun: 1.5 billion years The Sun can Power both Solar Cells and Thermoelectrics
Clean Energy: Thermoelectrics and Photovoltaics Akram Boukai Ph.D. Solar Energy Use Hydrocarbons vs. Photons Arabian Oil: 600 years Sun: 1.5 billion years The Sun can Power both Solar Cells and Thermoelectrics
Thermoelectric effect
 Hiroyuki KOIZUMI 1. Principle Thermoelectric effect Seebeck effect Temperature difference ΔT Voltage difference ΔV Peltier effect I Q Thomson effect I Current Q Heat transfer Thermoelectric effect Seebeck
Hiroyuki KOIZUMI 1. Principle Thermoelectric effect Seebeck effect Temperature difference ΔT Voltage difference ΔV Peltier effect I Q Thomson effect I Current Q Heat transfer Thermoelectric effect Seebeck
David J. Starling Penn State Hazleton PHYS 214
 Being virtually killed by a virtual laser in a virtual space is just as effective as the real thing, because you are as dead as you think you are. -Douglas Adams, Mostly Harmless David J. Starling Penn
Being virtually killed by a virtual laser in a virtual space is just as effective as the real thing, because you are as dead as you think you are. -Douglas Adams, Mostly Harmless David J. Starling Penn
Thermoelectric Oxide Materials For Electric Power Generation
 Thermoelectric Oxide Materials For Electric Power Generation Kunihito Koumoto Nagoya University, Graduate School of Engineering CREST, Japan Science and Technology Agency 1. Thermoelectric Energy Conversion
Thermoelectric Oxide Materials For Electric Power Generation Kunihito Koumoto Nagoya University, Graduate School of Engineering CREST, Japan Science and Technology Agency 1. Thermoelectric Energy Conversion
Doped Semiconductors *
 OpenStax-CNX module: m1002 1 Doped Semiconductors * Bill Wilson This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 1.0 To see how we can make silicon a useful
OpenStax-CNX module: m1002 1 Doped Semiconductors * Bill Wilson This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 1.0 To see how we can make silicon a useful
Figure 1. Load resistance / Ω. Use data from Figure 1 to calculate the current in the load at the peak power. (3) Page 1 of 27
 Figure shows data for the variation of the power output of a photovoltaic cell with load resistance. The data were obtained by placing the cell in sunlight. The intensity of the energy from the Sun incident
Figure shows data for the variation of the power output of a photovoltaic cell with load resistance. The data were obtained by placing the cell in sunlight. The intensity of the energy from the Sun incident
DIFFUSION IN SOLIDS. IE-114 Materials Science and General Chemistry Lecture-5
 DIFFUSION IN SOLIDS IE-114 Materials Science and General Chemistry Lecture-5 Diffusion The mechanism by which matter is transported through matter. It is related to internal atomic movement. Atomic movement;
DIFFUSION IN SOLIDS IE-114 Materials Science and General Chemistry Lecture-5 Diffusion The mechanism by which matter is transported through matter. It is related to internal atomic movement. Atomic movement;
Properties of Vapors
 Properties of Vapors Topics for Discussion The Pressure/Temperature Relationship Vaporization Condensation Enthalpy Properties of Vapors Topics for Discussion Entropy Properties of Substances Saturated
Properties of Vapors Topics for Discussion The Pressure/Temperature Relationship Vaporization Condensation Enthalpy Properties of Vapors Topics for Discussion Entropy Properties of Substances Saturated
General Chemistry for ISE students: Final examination WS 2010 (minimum point number: 50 maximum grade at 100 points maximum point number: 115)
 General Chemistry for ISE students: Final examination WS 2010 (minimum point number: 50 maximum grade at 100 points maximum point number: 115) 1 Which statements on the properties of an electron are correct,
General Chemistry for ISE students: Final examination WS 2010 (minimum point number: 50 maximum grade at 100 points maximum point number: 115) 1 Which statements on the properties of an electron are correct,
Foundations of Chemistry
 Foundations of Chemistry Physical Changes Key Concepts How can a change in energy affect the state of matter? What happens when something dissolves? What is meant by conservation of mass? What do you think?
Foundations of Chemistry Physical Changes Key Concepts How can a change in energy affect the state of matter? What happens when something dissolves? What is meant by conservation of mass? What do you think?
