6-3 Particle model of matter Physics
|
|
|
- Anthony Miller
- 6 years ago
- Views:
Transcription
1 6-3 Particle model of matter Physics.0 A teacher uses a tray filled with table tennis balls to model how particles are arranged in materials, as shown in Figure Figure. Initially the balls are arranged in regular pattern as shown in Figure. Which state of matter is best represented by the balls in Figure? Tick one box. [ mark] solid liquid gas.2 The teacher then moves the tray from side to side so that the table tennis balls are no longer in a regular pattern. Which state of matter is now best represented by the balls? Tick one box. solid [ mark] liquid gas Page
2 .3 The teacher next performs another demonstration by moving the tray more vigorously so that some of the balls jump out of the tray. The teacher tells the students that the balls that have left the tray represent gas particles. Which two processes could this demonstration represent? Tick two boxes. boiling condensing evaporation freezing melting.4 Gases can be at different temperatures. Tick one box. The temperature of a gas is related to [ mark] the average size of the particles the average mass of the particles the average kinetic energy of the particles Page 2
3 2. Figure 2 shows some of the gas particles in a balloon. Figure 2 Describe the movement of the gas particles inside the balloon. 2.2 The gas in the balloon has a mass of kg. The balloon has a volume of m 3. Calculate the density of the gas in the balloon. Density of gas = kg/m 3 Page 3
4 2.3 The balloon is held by a diver. Above the water, the pressure is Pa. Calculate the value of the p V constant for the balloon. Give your answer in standard form to 2 significant figures. Constant = Pa m The diver dives underwater with the balloon. At a depth of 25 m, the pressure was Pa. Calculate the volume of the ball at this depth. Give your answer to 2 significant figures. Volume = m Explain what would happen to the temperature of the gas in the balloon if it was moved very quickly to the greater depth. [2 mark] Page 4
5 3.0 Figure 3 shows a fridge with a freezer compartment. Figure 3 3. Energy is transferred to cool food when it is placed in the fridge. Complete the sentence to describe how energy is transferred to cool the food. food fridge surroundings Energy is transferred from the to the, this energy is then transferred to the. Page 5
6 3.2 The fridge and freezer compartment contain water in three different states. Use your knowledge of the particle model to explain the differences in how the particles are arranged in solids liquids and gases Include in your answer any differences in how the particles move in each state of matter. [6 marks] 3.3 The temperature of the air inside the freezer compartment is 5 C. The temperature of the air inside the fridge is 5 C Explain why the particles in the freezer compartment move at a different speed than those in the main part of the fridge. Page 6
7 4.0 A student used the apparatus in Figure 4 to compare the specific heat capacities of different metals. Figure 4 The student measured the time taken to increase the temperature of each material by 0 C. Figure 5 shows the student s results. Figure 5 Material 4. The student makes the following conclusion; The specific heat capacity of concrete is five times greater than tin. Use data from the bar chart to decide if the student s conclusion is correct. Page 7
8 4.2 The iron block had a mass of.023 kg. The specific heat capacity of iron is 450 J / kg C. Calculate the energy transferred by the heater to increase the temperature of the iron block by 0 C. Use the correct equation from the physics equation sheet. Give your answer to two significant figures. [3 marks] Energy transferred = J The student used the same apparatus to heat a kg block of aluminium. He recorded the temperature of the block as it was heated from room temperature. The results are shown in Figure 6. Figure 6 Time the immersion heater is switched on for in minutes Page 8
9 4.3 After how many minutes did the student record the incorrect temperature? [ mark] Time = minutes 4.4 Draw the line of best fit for the points plotted in Figure 6. [ mark] 4.5 What was the temperature of the room? [ mark] Temperature C 4.6 Another student suggested repeating the experiment using a heater with a greater power. Explain what effect this would have on the gradient of the graph the student drew. [3 marks] Page 9
10 5. You have been given a metal cube and a small statue with an irregular shape. Describe how to measure the density of each of these objects. [6 marks] Page 0
11 MARK SCHEME Qu No. Extra Information Marks. solid.2 liquid.3 boiling evaporation.4 the average kinetic energy of the particles Qu No. Extra Information Marks 2. motion is random range of speeds or range of directions 2.2 ρ = / ρ =.3 (kg/m 3 ) allow.28 (kg/m 3 ) 2.3 pv= = (Pa m 3 ) 2.4 V = constant / p = / = (m 3 ) 2.5 work done on the gas so internal energy increases (so temperature increases) allow.3 (kg/m 3 ) with no working for 2 marks allow ecf allow (m 3 ) ignore reference to other sources of temperature change eg cold water, insulation etc Page
12 Qu No. Extra Information Marks 3. food 3.2 Level 3: Level 2: Level : fridge surroundings Indicative content 2 marks for all three in the correct place mark for 2 or in the correct place A detailed and coherent description of both the arrangement and motion of the particles in the different states of matter A coherent description of both the arrangement and motion of the particles in the different states of matter. Simple description of the arrangement and/or motion of the particles in the different states of matter No relevant content 0 Solid Particles closely packed in a regular pattern Particles vibrate about a fixed position Liquid Particles closely packed in an irregular pattern Particles are able to move relative to each other Liquid Particles are widely spread in no pattern Particles move randomly and rapidly. 3.3 Air molecules in fridge will have a greater speed. because the air is at a greater temperature so greater kinetic energy allow 2 marks for the converse. Page 2
13 Qu No. Extra Information Marks 4. conclusion is not correct because 45 / 50 = is less than 5 allow for TIn 4.2 E =.023 x 450 x 0 E = (J) Answer to 2 sig. figs. allow 2 marks for a correct answer to an incorrect number of sig figs eg (J) allow mark for an incorrect answer to an incorrect number of sig figs eg (J) if substitution is correct (minutes) 4.4 Correct line of best fit drawn ( C) 4.6 gradient would be greater because energy supplied per second would be greater. so rate of increase of temperature would be greater. or more energy supplied (in 2 minutes) so greater final temperature (so greater temperature difference) Qu No. Extra Information Marks 5. Level 3: Level 2: Clear and coherent description of both methods including equation needed to calculate density. Steps are logically ordered and could be followed by someone else to obtain valid results. Clear description of one method to measure density or partial description of both methods. Steps may not be logically ordered. Level : Basic description of measurements needed with no indication of how to use them. -2 Indicative content No relevant content 0 For both measure mass using a balance calculate density using ρ = m / V Metal cube measure length of cube s sides using a ruler calculate volume Small statue immerse in water measure volume / mass of water displaced volume of water displaced = volume of small statue Page 3
6-3 Particle model of matter Trilogy
 6-3 Particle model of matter Trilogy.0 A teacher uses a tray filled with table tennis balls to model how particles are arranged in materials, as shown in Figure Figure. Initially the balls are arranged
6-3 Particle model of matter Trilogy.0 A teacher uses a tray filled with table tennis balls to model how particles are arranged in materials, as shown in Figure Figure. Initially the balls are arranged
Name: New Document 1. Class: Date: 83 minutes. Time: 82 marks. Marks: Comments:
 New Document Name: Class: Date: Time: 83 minutes Marks: 82 marks Comments: Q. Solid, liquid and gas are three different states of matter. (a) Describe the difference between the solid and gas states, in
New Document Name: Class: Date: Time: 83 minutes Marks: 82 marks Comments: Q. Solid, liquid and gas are three different states of matter. (a) Describe the difference between the solid and gas states, in
Two students investigated the change of state of stearic acid from liquid to solid.
 Two students investigated the change of state of stearic acid from liquid to solid. They measured how the temperature of stearic acid changed over 5 minutes as it changed from liquid to solid. Figure shows
Two students investigated the change of state of stearic acid from liquid to solid. They measured how the temperature of stearic acid changed over 5 minutes as it changed from liquid to solid. Figure shows
AQA (Trilogy) Combined Science GCSE Unit 6.3 Particle Model of Matter
 AQA (Trilogy) Combined Science GCSE Unit 6.3 Particle Model of Matter Test (Levels 4 9) Time allowed: 50 minutes Question Links to Student Progress Sheet Score Total Marks Available Score Estimated Grade
AQA (Trilogy) Combined Science GCSE Unit 6.3 Particle Model of Matter Test (Levels 4 9) Time allowed: 50 minutes Question Links to Student Progress Sheet Score Total Marks Available Score Estimated Grade
TEACHER NOTES: ICE CUBE POSTER
 TEACHER NOTES: NATIONAL CURRICULUM LINKS THE PARTICULATE NATURE OF MATTER the properties of the different states of matter (solid, liquid and gas) in terms of the particle model, including gas pressure
TEACHER NOTES: NATIONAL CURRICULUM LINKS THE PARTICULATE NATURE OF MATTER the properties of the different states of matter (solid, liquid and gas) in terms of the particle model, including gas pressure
4.3.1 Changes of state and the particle model Density of materials. ρ = m. Content. Key opportunities for skills development
 4.3 Particle model of matter The particle model is widely used to predict the behaviour of solids, liquids and gases and this has many applications in everyday life. It helps us to explain a wide range
4.3 Particle model of matter The particle model is widely used to predict the behaviour of solids, liquids and gases and this has many applications in everyday life. It helps us to explain a wide range
Physics PH1FP. (Jun14PH1FP01) General Certificate of Secondary Education Foundation Tier June Unit Physics P1. Unit Physics P1 TOTAL
 Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials Question Mark Science A Unit Physics P1 Physics Unit Physics P1 Thursday 12 June 2014 General
Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials Question Mark Science A Unit Physics P1 Physics Unit Physics P1 Thursday 12 June 2014 General
PAPER 2 THEORY QUESTIONS
 PAPER 2 THEORY QUESTIONS 1 Fig. 1.1 shows the arrangement of atoms in a solid block. Fig. 1.1 (a) End X of the block is heated. Energy is conducted to end Y, which becomes warm. (i) Explain how heat is
PAPER 2 THEORY QUESTIONS 1 Fig. 1.1 shows the arrangement of atoms in a solid block. Fig. 1.1 (a) End X of the block is heated. Energy is conducted to end Y, which becomes warm. (i) Explain how heat is
PURE PHYSICS THERMAL PHYSICS (PART I)
 PURE PHYSICS THERMAL PHYSICS (PART I) 1 The kinetic theory of matter states that all matters are made up of or, which are in and motion. forces hold the atoms or molecules together. The nature of these
PURE PHYSICS THERMAL PHYSICS (PART I) 1 The kinetic theory of matter states that all matters are made up of or, which are in and motion. forces hold the atoms or molecules together. The nature of these
Electricity and Energy 1 Content Statements
 Keep this in good condition, it will help you pass your final exams. The school will only issue one paper copy per pupil. An e-copy will be placed on the school s web-site. Electricity and Energy 1 Content
Keep this in good condition, it will help you pass your final exams. The school will only issue one paper copy per pupil. An e-copy will be placed on the school s web-site. Electricity and Energy 1 Content
Solids (cont.) Describe the movement of particles in a solid and the forces between them.
 Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
1. Thermal energy is transferred through the glass windows of a house mainly by. D. radiation and convection. (1)
 1. Thermal energy is transferred through the glass windows of a house mainly by A. conduction. B. radiation. C. conduction and convection. D. radiation and convection. 2. The specific latent heat of vaporization
1. Thermal energy is transferred through the glass windows of a house mainly by A. conduction. B. radiation. C. conduction and convection. D. radiation and convection. 2. The specific latent heat of vaporization
Rashid School for Boys. Year 7 Science. Particles. Name: Form:
 Rashid School for Boys Year Science Particles Name: Form: 1 By the end of this topic.. Unit Particles Level 3 I know that ice melts when it gets too warm and that liquid water turns into solid water (ice)
Rashid School for Boys Year Science Particles Name: Form: 1 By the end of this topic.. Unit Particles Level 3 I know that ice melts when it gets too warm and that liquid water turns into solid water (ice)
SAM Teachers Guide Phase Change Overview Learning Objectives Possible Student Pre/Misconceptions
 SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion and observing and manipulating
SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion and observing and manipulating
NAME: ACTIVITY SHEETS PHYSICS AND CHEMISTRY (SECONDARY 3 rd YEAR)
 NAME: ACTIVITY SHEETS PHYSICS AND CHEMISTRY (SECONDARY 3 rd YEAR) ACTIVITY 1: Matter Lesson 2 THE PARTICULATE NATURE OF MATTER 1-What is matter? 2-What is a particle (corpuscle)? Set some examples 3-What
NAME: ACTIVITY SHEETS PHYSICS AND CHEMISTRY (SECONDARY 3 rd YEAR) ACTIVITY 1: Matter Lesson 2 THE PARTICULATE NATURE OF MATTER 1-What is matter? 2-What is a particle (corpuscle)? Set some examples 3-What
Kinetic Theory of Matter. Matter & Energy
 Kinetic Theory of Matter Matter & Energy 1 Kinetic Theory of Matter All matter is made up of atoms and molecules that act as tiny particles. 2 Kinetic Theory of Matter These tiny particles are always in
Kinetic Theory of Matter Matter & Energy 1 Kinetic Theory of Matter All matter is made up of atoms and molecules that act as tiny particles. 2 Kinetic Theory of Matter These tiny particles are always in
Kinetic Theory of Matter
 1 Temperature and Thermal Energy Kinetic Theory of Matter The motion of the particles in matter is described by kinetic theory of matter. Matter is composed of particles that are atoms, molecules, or ions
1 Temperature and Thermal Energy Kinetic Theory of Matter The motion of the particles in matter is described by kinetic theory of matter. Matter is composed of particles that are atoms, molecules, or ions
Name... Class... Date... Specific heat capacity and specific latent heat
 Specific heat capacity and specific latent heat Specification references: P3.2.2 Temperature changes in a system and specific heat capacity P3.2.3 Changes of heat and specific latent heat Aims This is
Specific heat capacity and specific latent heat Specification references: P3.2.2 Temperature changes in a system and specific heat capacity P3.2.3 Changes of heat and specific latent heat Aims This is
SAM Teachers Guide Phase Change Overview Learning Objectives Possible Student Pre/Misconceptions
 SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion. Students observe and manipulate
SAM Teachers Guide Phase Change Overview Students review the atomic arrangements for each state of matter, following trajectories of individual atoms to observe their motion. Students observe and manipulate
GraspIT AQA Particle Model Questions
 A. Particle model of matter Density of materials and changes of state 1. A 45 g piece of plasticine is placed in water and 30 cm 3 of water was displaced. Calculate the density of the plasticine in kg/m
A. Particle model of matter Density of materials and changes of state 1. A 45 g piece of plasticine is placed in water and 30 cm 3 of water was displaced. Calculate the density of the plasticine in kg/m
Chemistry A: States of Matter Packet Name: Hour: Page!1. Chemistry A States of Matter Packet
 Chemistry A: States of Matter Packet Name: Hour: Page!1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page!2 Worksheet #1: States of Matter In this packet we will
Chemistry A: States of Matter Packet Name: Hour: Page!1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page!2 Worksheet #1: States of Matter In this packet we will
Solids, Liquids & Gases
 Solids, Liquids & Gases Density & Pressure...1 Density...1 Formula...1 Common densities...2 Pressure...3 Pressure in Liquids...3 Nice to know: External Pressure exerted on a closed container...4 Change
Solids, Liquids & Gases Density & Pressure...1 Density...1 Formula...1 Common densities...2 Pressure...3 Pressure in Liquids...3 Nice to know: External Pressure exerted on a closed container...4 Change
Name... Class... Date...
 Density and states of matter Specification reference: P3.1.1 Density of materials Aims In this exercise, you will consider the term density and its relationship to the and volume of a variety of different
Density and states of matter Specification reference: P3.1.1 Density of materials Aims In this exercise, you will consider the term density and its relationship to the and volume of a variety of different
40P (2 x 60 x 60) = 2.5 x 10 6 (4200)(5) P = 1.82 x 10 5 W
 NAME : F.3C ( ) Marks: /50 Form 3 Physics Assessment on Heat Time allowed: 45 minutes Section A (34 marks) 1. An indoor swimming pool containing 2.5 x 10 6 kg of water uses 40 identical heaters to maintain
NAME : F.3C ( ) Marks: /50 Form 3 Physics Assessment on Heat Time allowed: 45 minutes Section A (34 marks) 1. An indoor swimming pool containing 2.5 x 10 6 kg of water uses 40 identical heaters to maintain
Pg , Syllabus
 Pg. 169 171, 173-175 Syllabus 5.7 5.14 www.cgrahamphysics.com What do you remember? End www.cgrahamphysics.com How do particles move? 3 of 30 Boardworks Ltd 2012 4 of 30 Boardworks Ltd 2012 States of matter
Pg. 169 171, 173-175 Syllabus 5.7 5.14 www.cgrahamphysics.com What do you remember? End www.cgrahamphysics.com How do particles move? 3 of 30 Boardworks Ltd 2012 4 of 30 Boardworks Ltd 2012 States of matter
Chemistry #3 Notebook States of Matter
 Name Hour Test Date Group # Chemistry #3 Notebook States of Matter LEARNING TARGETS I CAN model the motion and arrangement of particles in typical solids, liquids and gasses. I CAN describe how the motion
Name Hour Test Date Group # Chemistry #3 Notebook States of Matter LEARNING TARGETS I CAN model the motion and arrangement of particles in typical solids, liquids and gasses. I CAN describe how the motion
M1. (a) range of speeds 1. moving in different directions accept random motion 1. internal energy 1. density = mass / volume 1. (d) / 0.
 M. (a) range of speeds moving in different directions accept random motion (b) internal energy (c) density = mass / volume (d) 0.00254 / 0.04 0.8 accept 0.8 with no working shown for the 2 calculation
M. (a) range of speeds moving in different directions accept random motion (b) internal energy (c) density = mass / volume (d) 0.00254 / 0.04 0.8 accept 0.8 with no working shown for the 2 calculation
The Can Demonstration
 The Can Demonstration With your table, make a prediction as to what will happen to the can. Discuss why you think that the can imploded. What are some reasons? https://www.youtube.com/watch?v=xg5niowf_zw&t=28s
The Can Demonstration With your table, make a prediction as to what will happen to the can. Discuss why you think that the can imploded. What are some reasons? https://www.youtube.com/watch?v=xg5niowf_zw&t=28s
Chapter 1 Heating Processes
 Chapter 1 Heating Processes Section 1.1 Heat and temperature Worked example: Try yourself 1.1.1 CALCULATING THE CHANGE IN INTERNAL ENERGY A student places a heating element and a paddle wheel apparatus
Chapter 1 Heating Processes Section 1.1 Heat and temperature Worked example: Try yourself 1.1.1 CALCULATING THE CHANGE IN INTERNAL ENERGY A student places a heating element and a paddle wheel apparatus
CLASSIFIED 2 PRESSURE THERMAL PHYSICS MR. HUSSAM SAMIR
 CLASSIFIED 2 PRESSURE THERMAL PHYSICS MR. HUSSAM SAMIR 1. The diagram shows a simple mercury barometer. If atmospheric pressure increases, what happens to level X and to level Y? 2. Four flower vases have
CLASSIFIED 2 PRESSURE THERMAL PHYSICS MR. HUSSAM SAMIR 1. The diagram shows a simple mercury barometer. If atmospheric pressure increases, what happens to level X and to level Y? 2. Four flower vases have
Thermal Effects. IGCSE Physics
 Thermal Effects IGCSE Physics Starter What is the difference between heat and temperature? What unit is thermal energy measured in? And what does it depend on? In which direction does heat flow? Heat (Thermal
Thermal Effects IGCSE Physics Starter What is the difference between heat and temperature? What unit is thermal energy measured in? And what does it depend on? In which direction does heat flow? Heat (Thermal
Put sufficient ice cubes into water (1 M) and wait for equilibrium (both exist) (1 M)
 NAME : F.5 ( ) Marks: /70 FORM FOUR PHYSICS REVISION TEST on HEAT Allowed: 70 minutes This paper consists of two sections. Section A (50 marks) consists of the structure-type questions, and Section B (20
NAME : F.5 ( ) Marks: /70 FORM FOUR PHYSICS REVISION TEST on HEAT Allowed: 70 minutes This paper consists of two sections. Section A (50 marks) consists of the structure-type questions, and Section B (20
ENERGY IN CHEMISTRY. R. Ashby Duplication by permission only.
 CH 11 TOPIC 28 CHANGING STATES OF MATTER 1 You have mastered this topic when you can: 1) define or describe: ENERGY, POTENTIAL ENERGY, KINETIC ENERGY & KINETIC MOLECULAR THEORY 2) define or describe HEAT
CH 11 TOPIC 28 CHANGING STATES OF MATTER 1 You have mastered this topic when you can: 1) define or describe: ENERGY, POTENTIAL ENERGY, KINETIC ENERGY & KINETIC MOLECULAR THEORY 2) define or describe HEAT
GraspIT AQA Particle Model Questions
 A. Particle model of matter Density of materials and changes of state 1. A 45 g piece of plasticine is placed in water and displaces 30 cm 3 of water. Calculate the density of the plasticine in kg/m 3
A. Particle model of matter Density of materials and changes of state 1. A 45 g piece of plasticine is placed in water and displaces 30 cm 3 of water. Calculate the density of the plasticine in kg/m 3
KS3 Science. Heat and Energy
 KS3 Science Heat and Energy Heat and Energy Key Words Write a definition for each of the key words listed below Key words States of matter Melt Freeze Evaporate Condense Heat Conduction Convention Radiation
KS3 Science Heat and Energy Heat and Energy Key Words Write a definition for each of the key words listed below Key words States of matter Melt Freeze Evaporate Condense Heat Conduction Convention Radiation
What is a change of state? What happens during a change of state? What can happen when a substance loses or gains energy?
 CHAPTER 3 3 Changes of State SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a change of state? What happens during a change
CHAPTER 3 3 Changes of State SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a change of state? What happens during a change
Matter. Energy- which is a property of matter!! Matter: anything that takes up space and has mass
 Matter Matter: anything that takes up space and has mass Can you think of anything that is not made of matter? Energy- which is a property of matter!! Matter is made up of moving particles! Instead of
Matter Matter: anything that takes up space and has mass Can you think of anything that is not made of matter? Energy- which is a property of matter!! Matter is made up of moving particles! Instead of
1. This question is about modelling the thermal processes involved when a person is running.
 1. This question is about modelling the thermal processes involved when a person is running. When running, a person generates thermal energy but maintains approximately constant temperature. (a) Explain
1. This question is about modelling the thermal processes involved when a person is running. When running, a person generates thermal energy but maintains approximately constant temperature. (a) Explain
Chapter 7.1. States of Matter
 Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Orchard School. New Document 1 Name: Class: Date: 129 minutes. Time: 126 marks. Marks: Comments: Page 1
 New Document Name: Class: Date: Time: Marks: 29 minutes 26 marks Comments: Page Q. The ph scale is a measure of the acidity or alkalinity of a solution. (a) Solution Draw one line from each solution to
New Document Name: Class: Date: Time: Marks: 29 minutes 26 marks Comments: Page Q. The ph scale is a measure of the acidity or alkalinity of a solution. (a) Solution Draw one line from each solution to
Estimate, for this water, the specific heat capacity, specific heat capacity =... J kg 1 K 1. the specific latent heat of vaporisation.
 1 A kettle is rated as 2.3 kw. A mass of 750 g of water at 20 C is poured into the kettle. When the kettle is switched on, it takes 2.0 minutes for the water to start boiling. In a further 7.0 minutes,
1 A kettle is rated as 2.3 kw. A mass of 750 g of water at 20 C is poured into the kettle. When the kettle is switched on, it takes 2.0 minutes for the water to start boiling. In a further 7.0 minutes,
Science Notebook Layout DONʼT COPY UNDERLINED TEXT Mr. Needhamʼs web page:
 20 Rating cocoas 10/ See teacher for data or copy from someone in your group Analysis of cocoa 10/4 21 What made test unfair? Bias: influence in an unfair way. How could bian have influenced that outcome
20 Rating cocoas 10/ See teacher for data or copy from someone in your group Analysis of cocoa 10/4 21 What made test unfair? Bias: influence in an unfair way. How could bian have influenced that outcome
Chapter 22 States of matter. Section 1 matter Section 2 Changes of State
 Chapter 22 States of matter Section 1 matter Section 2 Changes of State States of Matter is a physical property ***Matter is made of atoms Atoms form chemical bonds to make matter **** Atoms vibrate constantly
Chapter 22 States of matter Section 1 matter Section 2 Changes of State States of Matter is a physical property ***Matter is made of atoms Atoms form chemical bonds to make matter **** Atoms vibrate constantly
Chapter: Heat and States
 Table of Contents Chapter: Heat and States of Matter Section 1: Temperature and Thermal Energy Section 2: States of Matter Section 3: Transferring Thermal Energy Section 4: Using Thermal Energy 1 Temperature
Table of Contents Chapter: Heat and States of Matter Section 1: Temperature and Thermal Energy Section 2: States of Matter Section 3: Transferring Thermal Energy Section 4: Using Thermal Energy 1 Temperature
Physical Science Chapter 5 Cont3. Temperature & Heat
 Physical Science Chapter 5 Cont3 Temperature & Heat What are we going to study? Heat Transfer Phases of Matter The Kinetic Theory of Gases Thermodynamics Specific Heat (Capacity) Specific Heat Latent Heat
Physical Science Chapter 5 Cont3 Temperature & Heat What are we going to study? Heat Transfer Phases of Matter The Kinetic Theory of Gases Thermodynamics Specific Heat (Capacity) Specific Heat Latent Heat
IGCSE Double Award Extended Coordinated Science
 IGCSE Double Award Extended Coordinated Science Physics 5 - Thermal Properties of Matter Thermal Expansion You need to know thermal expansions for solids, liquids, and gases, and their applications. Thermal
IGCSE Double Award Extended Coordinated Science Physics 5 - Thermal Properties of Matter Thermal Expansion You need to know thermal expansions for solids, liquids, and gases, and their applications. Thermal
4.1. Physics Module Form 4 Chapter 4 - Heat GCKL UNDERSTANDING THERMAL EQUILIBRIUM. What is thermal equilibrium?
 Physics Module Form 4 Chapter 4 - Heat GCKL 2010 4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. (, Temperature ) is a form of energy that flows from a hot body to a cold body.
Physics Module Form 4 Chapter 4 - Heat GCKL 2010 4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. (, Temperature ) is a form of energy that flows from a hot body to a cold body.
(ii) the total kinetic energy of the gas molecules (1 mark) (iii) the total potential energy of the gas molecules (1 mark)
 NAME : F.5 ( ) Marks: /70 FORM FOUR PHYSICS REVISION TEST on HEAT Allowed: 70 minutes This paper consists of two sections. Section A (50 marks) consists of the structure-type questions, and Section B (20
NAME : F.5 ( ) Marks: /70 FORM FOUR PHYSICS REVISION TEST on HEAT Allowed: 70 minutes This paper consists of two sections. Section A (50 marks) consists of the structure-type questions, and Section B (20
Topic review. hi.com.au Science Resource Centre. Using scientific language 1. You should be familiar with the words in the following list.
 Topic review Using scientific language 1. You should be familiar with the words in the following list. condense solid boiling matter gas diffusion melting solidify particles substance liquid property compression
Topic review Using scientific language 1. You should be familiar with the words in the following list. condense solid boiling matter gas diffusion melting solidify particles substance liquid property compression
Year 7 Science. 7C1: The Particle Model. PPA Challenge
 Year 7 Science 7C1: The Particle Model PPA Challenge Name: Form: Task Sheet 1 (Bronze Challenge): The Particle Model Use the words in the box to label the diagram below. This particle diagram shows the
Year 7 Science 7C1: The Particle Model PPA Challenge Name: Form: Task Sheet 1 (Bronze Challenge): The Particle Model Use the words in the box to label the diagram below. This particle diagram shows the
Atoms and molecules are in motion and have energy
 Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
NAME DATE CLASS TEST DATE:
 1 TEST DATE: 2 Vocabulary Chapter 8 Solids, liquids, and gases Condensation Crystals Evaporation Heat of fusion Heat of vaporization Kinetic theory of matter Plasma States of matter Thermal expansion Chapter
1 TEST DATE: 2 Vocabulary Chapter 8 Solids, liquids, and gases Condensation Crystals Evaporation Heat of fusion Heat of vaporization Kinetic theory of matter Plasma States of matter Thermal expansion Chapter
PHYSICS (SPECIFICATION A) Unit 2 Mechanics and Molecular Kinetic Theory
 Surname Centre Number Other Names Candidate Number Leave blank Candidate Signature General Certificate of Education January 2003 Advanced Subsidiary Examination PHYSICS (SPECIFICATION A) Unit 2 Mechanics
Surname Centre Number Other Names Candidate Number Leave blank Candidate Signature General Certificate of Education January 2003 Advanced Subsidiary Examination PHYSICS (SPECIFICATION A) Unit 2 Mechanics
Thermal Properties Of Matter
 Thermal Properties Of Matter 3.2.2 Explain why different substances have different specific heat capacities. Heat two same size objects of different materials for the same amount of time they will not
Thermal Properties Of Matter 3.2.2 Explain why different substances have different specific heat capacities. Heat two same size objects of different materials for the same amount of time they will not
Matter & Energy. Objectives: properties and structures of the different states of matter.
 Matter & Energy Objectives: 1. Use the kinetic theory to describe the properties and structures of the different states of matter. 2. Describe energy transfers involved in changes of state. 3. Describe
Matter & Energy Objectives: 1. Use the kinetic theory to describe the properties and structures of the different states of matter. 2. Describe energy transfers involved in changes of state. 3. Describe
1. (a) Methane can be a gas, a liquid or a solid. In the diagram below, arrows P, Q, R and S represent changes of state.
 1. (a) Methane can be a gas, a liquid or a solid. In the diagram below, arrows P, Q, R and S represent changes of state. The boxes on the right show the arrangement of particles of methane in the three
1. (a) Methane can be a gas, a liquid or a solid. In the diagram below, arrows P, Q, R and S represent changes of state. The boxes on the right show the arrangement of particles of methane in the three
CHAPTER 3 TEST REVIEW
 IB PHYSICS Name: Period: Date: # Marks: 52 Raw Score: IB Curve: DEVIL PHYSICS BADDEST CLASS ON CAMPUS CHAPTER 3 TEST REVIEW 1. Water at a temperature of 0 C is kept in a thermally insulated container.
IB PHYSICS Name: Period: Date: # Marks: 52 Raw Score: IB Curve: DEVIL PHYSICS BADDEST CLASS ON CAMPUS CHAPTER 3 TEST REVIEW 1. Water at a temperature of 0 C is kept in a thermally insulated container.
Chapter 3. States of Matter
 Chapter 3 States of Matter 1. Solid 2. Liquid 3. Gas States of Matter Two More (discuss later) Plasma Bose-Einstein condensate States of Matter Solid (definite shape and volume) Particles are tightly packed
Chapter 3 States of Matter 1. Solid 2. Liquid 3. Gas States of Matter Two More (discuss later) Plasma Bose-Einstein condensate States of Matter Solid (definite shape and volume) Particles are tightly packed
Chapter 8. Chapter 8. Preview. Bellringer. Chapter 8. Particles of Matter. Objectives. Chapter 8. Particles of Matter, continued
 States of Matter Preview Bellringer Section 2 Behavior of Gases In the kitchen, you might find three different forms of water. What are these three forms of water, and where exactly in the kitchen would
States of Matter Preview Bellringer Section 2 Behavior of Gases In the kitchen, you might find three different forms of water. What are these three forms of water, and where exactly in the kitchen would
4-1 / 5-1 Atomic structure and the periodic table Chemistry and Trilogy
 4- / 5- Atomic structure and the periodic table Chemistry and Trilogy.0 A student separated a mixture of two alcohols, ethanol (boiling point 78 C) and butanol (boiling point 8 C). The apparatus is shown
4- / 5- Atomic structure and the periodic table Chemistry and Trilogy.0 A student separated a mixture of two alcohols, ethanol (boiling point 78 C) and butanol (boiling point 8 C). The apparatus is shown
Compiled and rearranged by Sajit Chandra Shakya
 1 (a) (i) The kinetic theory of gases leads to the equation m = kt. (b) Explain the significance of the quantity m... the equation to suggest what is meant by the absolute zero of temperature...
1 (a) (i) The kinetic theory of gases leads to the equation m = kt. (b) Explain the significance of the quantity m... the equation to suggest what is meant by the absolute zero of temperature...
P6 Molecules and matter. Student Book answers. P6.1 Density. Question Answer Marks Guidance. 1 a m 3 (= 0.80 m 0.60 m 0.
 P6. Density a 0.024 m 3 (= 0.80 m 0.60 m 0.05 m) b = 2500 kg/m 3 2 a 36 g 48 g = 88 g 2 b =. g/cm 3 3 a i 0.000 40 m 3 (= 0.0 m 0.080 m 0.05 m) 3 a ii = 9 000 kg/m 3 3 b v = = 7.9 0 8 m 3 thickness t =
P6. Density a 0.024 m 3 (= 0.80 m 0.60 m 0.05 m) b = 2500 kg/m 3 2 a 36 g 48 g = 88 g 2 b =. g/cm 3 3 a i 0.000 40 m 3 (= 0.0 m 0.080 m 0.05 m) 3 a ii = 9 000 kg/m 3 3 b v = = 7.9 0 8 m 3 thickness t =
SOLIDS, LIQUIDS, AND GASES
 CHAPTER 2 SOLIDS, LIQUIDS, AND GASES SECTION 2 1 States of Matter (pages 56-60) This section explains how shape, volume, and the motion of particles are useful in describing solids, liquids, and gases.
CHAPTER 2 SOLIDS, LIQUIDS, AND GASES SECTION 2 1 States of Matter (pages 56-60) This section explains how shape, volume, and the motion of particles are useful in describing solids, liquids, and gases.
Table of Contents. Diagnostic Pre-test... 5 Lesson 1: What Is an Atom? Lesson 5: Gases. Lesson 6: Melting and Freezing. Lesson 2: What Are Molecules?
 Table of Contents Diagnostic Pre-test................. 5 Lesson 1: What Is an Atom? What Can You See?................ 11 What Is Matter Made Of?........... 12 Describing the Atom............... 13 What
Table of Contents Diagnostic Pre-test................. 5 Lesson 1: What Is an Atom? What Can You See?................ 11 What Is Matter Made Of?........... 12 Describing the Atom............... 13 What
4.1. Physics Module Form 4 Chapter 4 - Heat GCKL UNDERSTANDING THERMAL EQUILIBRIUM. What is thermal equilibrium?
 4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. ( Heat, Temperature ) is a form of energy that flows from a hot body to a cold body. 2. The SI unit for ( heat, temperature) is Joule,
4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. ( Heat, Temperature ) is a form of energy that flows from a hot body to a cold body. 2. The SI unit for ( heat, temperature) is Joule,
SOLIDS, LIQUIDS AND GASES
 CHEMIS TRY CONTENTS 17 SOLIDS, LIQUIDS AND GASES 17 Solids, Liquids and Gases 147 18 Solutions and Crystallisation 155 19 Separating Mixtures 162 20 Elements, Compounds and Mixtures 171 All materials exist
CHEMIS TRY CONTENTS 17 SOLIDS, LIQUIDS AND GASES 17 Solids, Liquids and Gases 147 18 Solutions and Crystallisation 155 19 Separating Mixtures 162 20 Elements, Compounds and Mixtures 171 All materials exist
CIE Physics IGCSE. Topic 2: Thermal Physics
 CIE Physics IGCSE Topic 2: Thermal Physics Summary Notes Simple kinetic molecular model of matter Molecular model Solids Molecules close together in regular pattern Strong intermolecular forces of attraction
CIE Physics IGCSE Topic 2: Thermal Physics Summary Notes Simple kinetic molecular model of matter Molecular model Solids Molecules close together in regular pattern Strong intermolecular forces of attraction
Matter. Gas. Solid Liquid. Both shape and volume are not fixed. It has a fixed shape and a fixed volume.
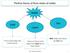 Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Chemistry A: States of Matter Packet Name: Hour: Page 1. Chemistry A States of Matter Packet
 Chemistry A: States of Matter Packet Name: Hour: Page 1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page 2 Worksheet #1: States of Matter In this packet we will
Chemistry A: States of Matter Packet Name: Hour: Page 1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page 2 Worksheet #1: States of Matter In this packet we will
Chapter 3. Preview. Section 1 Three States of Matter. Section 2 Behavior of Gases. Section 3 Changes of State. States of Matter.
 States of Matter Preview Section 1 Three States of Matter Section 2 Behavior of Gases Section 3 Changes of State Concept Mapping Section 1 Three States of Matter Bellringer In the kitchen, you might find
States of Matter Preview Section 1 Three States of Matter Section 2 Behavior of Gases Section 3 Changes of State Concept Mapping Section 1 Three States of Matter Bellringer In the kitchen, you might find
Part A (Level 1) A Matching (3 marks, 1 mark each) B True or false questions (7 marks, 1 mark each) Name: ( ) Time and Marks Class: Date:
 S1 Science Test Unit Name: ( ) Time and Marks Class: Date: Part A: 35 min / 100 marks Parts A & B: 45 min / 120 marks Note: 1 Attempt ALL questions. 2 Write your answers in the spaces provided on the Answer
S1 Science Test Unit Name: ( ) Time and Marks Class: Date: Part A: 35 min / 100 marks Parts A & B: 45 min / 120 marks Note: 1 Attempt ALL questions. 2 Write your answers in the spaces provided on the Answer
Section 16.3 Phase Changes
 Section 16.3 Phase Changes Solid Liquid Gas 3 Phases of Matter Density of Matter How packed matter is (The amount of matter in a given space) Solid: Liquid: Gas: High Density Medium Density Low Density
Section 16.3 Phase Changes Solid Liquid Gas 3 Phases of Matter Density of Matter How packed matter is (The amount of matter in a given space) Solid: Liquid: Gas: High Density Medium Density Low Density
Thermal Physics. Temperature (Definition #1): a measure of the average random kinetic energy of all the particles of a system Units: o C, K
 Thermal Physics Internal Energy: total potential energy and random kinetic energy of the molecules of a substance Symbol: U Units: J Internal Kinetic Energy: arises from random translational, vibrational,
Thermal Physics Internal Energy: total potential energy and random kinetic energy of the molecules of a substance Symbol: U Units: J Internal Kinetic Energy: arises from random translational, vibrational,
Solids, Liquids, and Gases
 Date Class _ Solids, Liquids, and Gases Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. _ 1. The surface of water can act like a sort of skin due to a property
Date Class _ Solids, Liquids, and Gases Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. _ 1. The surface of water can act like a sort of skin due to a property
Name... Class... Date...
 Latent heat Specification references: P3.2.3 Changes of heat and specific latent heat M1a, M3b, M3c, M3d, M4a Aims In this worksheet, you will work through two worked examples designed to allow you to
Latent heat Specification references: P3.2.3 Changes of heat and specific latent heat M1a, M3b, M3c, M3d, M4a Aims In this worksheet, you will work through two worked examples designed to allow you to
International General Certificate of Secondary Education UNIVERSITY OF CAMBRIDGE LOCAL EXAMINATIONS SYNDICATE
 Centre Number Candidate Number Candidate Name International General Certificate of Secondary Education UNIVERSITY OF CAMBRIDGE LOCAL EXAMINATIONS SYNDICATE PHYSICS 0625/2 PAPER 2 Friday 10 NOVEMBER 2000
Centre Number Candidate Number Candidate Name International General Certificate of Secondary Education UNIVERSITY OF CAMBRIDGE LOCAL EXAMINATIONS SYNDICATE PHYSICS 0625/2 PAPER 2 Friday 10 NOVEMBER 2000
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 2, LESSON 1 HEAT, TEMPERATURE, AND CONDUCTION MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state
The Next Generation Science Standards (NGSS) CHAPTER 2, LESSON 1 HEAT, TEMPERATURE, AND CONDUCTION MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state
Name Class Date. What is a change of state? What happens during a change of state? What can happen when a substance loses or gains energy?
 CHAPTER 2 3 Changes of State SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a change of state? What happens during a change
CHAPTER 2 3 Changes of State SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a change of state? What happens during a change
HEAT HISTORY. D. Whitehall
 1 HEAT HISTORY 18 th Century In the 18 th century it was assumed that there was an invisible substance called caloric. When objects got it was assumed that they gained caloric, therefore hot objects should
1 HEAT HISTORY 18 th Century In the 18 th century it was assumed that there was an invisible substance called caloric. When objects got it was assumed that they gained caloric, therefore hot objects should
NATIONAL 5 PHYSICS THERMODYNAMICS
 NATIONAL 5 PHYSICS THERMODYNAMICS HEAT AND TEMPERATURE Heat and temperature are not the same thing! Heat Heat is a type of energy. Like all types of energy it is measured in joules (J). The heat energy
NATIONAL 5 PHYSICS THERMODYNAMICS HEAT AND TEMPERATURE Heat and temperature are not the same thing! Heat Heat is a type of energy. Like all types of energy it is measured in joules (J). The heat energy
3.3 Phase Changes Charactaristics of Phase Changes phase change
 A large iceberg contains enough fresh water to supply millions of people with water for a year. As it moves into warmer areas, the ice changes to liquid water and eventually disappears. What happens when
A large iceberg contains enough fresh water to supply millions of people with water for a year. As it moves into warmer areas, the ice changes to liquid water and eventually disappears. What happens when
LEVEL ZERO VOICE CATALYST (10 minutes, individual work):
 LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Predict the properties of a metal. 2. Predict the properties of a nonmetal. 3. Which is more reactive? Ca or Cs? 4. How many electrons does N
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Predict the properties of a metal. 2. Predict the properties of a nonmetal. 3. Which is more reactive? Ca or Cs? 4. How many electrons does N
PhysicsAndMathsTutor.com 1
 PhysicsAndMathsTutor.com 1 Q1. In an experiment to measure the temperature of the flame of a Bunsen burner, a lump of copper of mass 0.12 kg is heated in the flame for several minutes. The copper is then
PhysicsAndMathsTutor.com 1 Q1. In an experiment to measure the temperature of the flame of a Bunsen burner, a lump of copper of mass 0.12 kg is heated in the flame for several minutes. The copper is then
Liquids & Solids: Section 12.3
 Liquids & Solids: Section 12.3 MAIN IDEA: The particles in and have a range of motion and are not easily. Why is it more difficult to pour syrup that is stored in the refrigerator than in the cabinet?
Liquids & Solids: Section 12.3 MAIN IDEA: The particles in and have a range of motion and are not easily. Why is it more difficult to pour syrup that is stored in the refrigerator than in the cabinet?
2. State the direction of heat transfer between the surroundings and the water in the bottle from 7 a.m. to 3 p.m.
 Base your answers to questions 1 through 3 on the information below. A student investigated heat transfer using a bottle of water. The student placed the bottle in a room at 20.5 C. The student measured
Base your answers to questions 1 through 3 on the information below. A student investigated heat transfer using a bottle of water. The student placed the bottle in a room at 20.5 C. The student measured
Specific heat capacity
 Specific heat capacity Investigating the specific heat capacity of different metals. In this practical you will: heat up blocks of different metals using an electric heater measure the mass and temperature
Specific heat capacity Investigating the specific heat capacity of different metals. In this practical you will: heat up blocks of different metals using an electric heater measure the mass and temperature
Find the rate of rise of water temperature at the beginning of the heating process. Rate of rise of temperature = K s -1 (1)
 HEAT ENERGY ANSWERS 1. A student pours 500 g of water into an aluminium saucepan of mass 1.20 kg, heats it over a steady flame and records the temperature as it heats up. the temperatures are plotted as
HEAT ENERGY ANSWERS 1. A student pours 500 g of water into an aluminium saucepan of mass 1.20 kg, heats it over a steady flame and records the temperature as it heats up. the temperatures are plotted as
1.0 The distance taken for a car to stop after an emergency depends on two things:
 4-5 Forces Physics.0 The distance taken for a car to stop after an emergency depends on two things: The thinking distance: how far the car travels while the driver processes the information. The braking
4-5 Forces Physics.0 The distance taken for a car to stop after an emergency depends on two things: The thinking distance: how far the car travels while the driver processes the information. The braking
PHYSICS 220. Lecture 24. Textbook Sections Lecture 25 Purdue University, Physics 220 1
 PHYSICS 220 Lecture 24 Heat Textbook Sections 14.4 14.5 Lecture 25 Purdue University, Physics 220 1 Exam 2 Average: 96.7 out of 150 Std Dev: 30.5 Lecture 25 Purdue University, Physics 220 2 Overview Last
PHYSICS 220 Lecture 24 Heat Textbook Sections 14.4 14.5 Lecture 25 Purdue University, Physics 220 1 Exam 2 Average: 96.7 out of 150 Std Dev: 30.5 Lecture 25 Purdue University, Physics 220 2 Overview Last
Term Info Picture. Anything that has mass and takes up space; everything is made of matter.
 Characteristics, Changes, and States of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. B. Develop and use models to describe the movement of particles
Characteristics, Changes, and States of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. B. Develop and use models to describe the movement of particles
THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES
 THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES The particle model of a gas A gas has no fixed shape or volume, but always spreads out to fill any container. There are
THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES The particle model of a gas A gas has no fixed shape or volume, but always spreads out to fill any container. There are
O-LEVELS REQUIREMENT. Name: Class: Date: THERMAL PROPETIES OF MATTER
 Name: Class: Date: Unit 11 THERMAL PROPETIES OF MATTER 82465685 calvinkongphysics@yahoo.com NOTES O-LEVELS REQUIREMENT Candidates should be able to: 1. describe a rise in temperature of a body in terms
Name: Class: Date: Unit 11 THERMAL PROPETIES OF MATTER 82465685 calvinkongphysics@yahoo.com NOTES O-LEVELS REQUIREMENT Candidates should be able to: 1. describe a rise in temperature of a body in terms
KINETIC PARTICLE THEORY
 KINETIC PARTICLE THEORY IMPORTANT DEFINITIONS: The mixing process in gases or solutions due to the random motion of particles is called Diffusion. The process by which a liquid changes into a vapour at
KINETIC PARTICLE THEORY IMPORTANT DEFINITIONS: The mixing process in gases or solutions due to the random motion of particles is called Diffusion. The process by which a liquid changes into a vapour at
Name Date Class THE NATURE OF GASES
 13.1 THE NATURE OF GASES Section Review Objectives Describe the assumptions of the kinetic theory as it applies to gases Interpret gas pressure in terms of kinetic theory Define the relationship between
13.1 THE NATURE OF GASES Section Review Objectives Describe the assumptions of the kinetic theory as it applies to gases Interpret gas pressure in terms of kinetic theory Define the relationship between
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 3, LESSON 1: WHAT IS DENSITY? MS-PS1-1. Develop models to describe the atomic composition of simple molecules and extended structures. DISCIPLINARY
The Next Generation Science Standards (NGSS) CHAPTER 3, LESSON 1: WHAT IS DENSITY? MS-PS1-1. Develop models to describe the atomic composition of simple molecules and extended structures. DISCIPLINARY
INTRODUCTION TO LESSON CLUSTER 7
 INTRODUCTION TO LESSON CLUSTER 7 EXPLAINING MELTING AND SOLIDIFYING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain melting and solidifying, by reference to the molecular
INTRODUCTION TO LESSON CLUSTER 7 EXPLAINING MELTING AND SOLIDIFYING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain melting and solidifying, by reference to the molecular
Post-Show HOT AND COLD. Gases. Liquids. Solids. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show HOT AND COLD After the Show We recently presented a Hot and Cold show at your school, and thought you and your students might like to continue investigating this topic.
Traveling Science Shows Post-Show HOT AND COLD After the Show We recently presented a Hot and Cold show at your school, and thought you and your students might like to continue investigating this topic.
States of Matter 1 of 21 Boardworks Ltd 2016
 States of Matter 1 of 21 Boardworks Ltd 2016 States of Matter 2 of 21 Boardworks Ltd 2016 What are the three states of matter? 3 of 21 Boardworks Ltd 2016 At any given temperature, all substances exist
States of Matter 1 of 21 Boardworks Ltd 2016 States of Matter 2 of 21 Boardworks Ltd 2016 What are the three states of matter? 3 of 21 Boardworks Ltd 2016 At any given temperature, all substances exist
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education
 UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *6024898353* PHYSICS 0625/31 Paper 3 Extended October/November 2009 1 hour 15 minutes Candidates
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *6024898353* PHYSICS 0625/31 Paper 3 Extended October/November 2009 1 hour 15 minutes Candidates
Lockerbie Academy. S1 Science. Elements, Compounds and Mixtures Homework Booklet
 Lockerbie Academy S Science Elements, Compounds and Mixtures Homework Booklet Homework From Lesson - Research and Design Researching Use the internet or books from a library to research the element below
Lockerbie Academy S Science Elements, Compounds and Mixtures Homework Booklet Homework From Lesson - Research and Design Researching Use the internet or books from a library to research the element below
